Published online Mar 16, 2021. doi: 10.12998/wjcc.v9.i8.1901
Peer-review started: October 15, 2020
First decision: December 13, 2020
Revised: December 24, 2020
Accepted: January 28, 2021
Article in press: January 28, 2021
Published online: March 16, 2021
Processing time: 135 Days and 4.1 Hours
Leptospira is an uncommon pathogen for adult severe community-acquired pneumonia and its nonspecific manifestations and limited diagnostic tests make it difficult to identify. Although conventional penicillin remains efficacious to treat leptospirosis, failure in early diagnosis and treatment can lead to progression into a deadly syndrome with multiple organ dysfunction. Next generation sequencing is of great value to understand cases with infection of unknown cause, which could help in the diagnosis of uncertain Leptospira infection.
We recently managed a patient with fever, cough and dyspnea on admission that progressed into persistent adult respiratory distress syndrome, hemoptysis and hematuria after admission. In this case, the rare Leptospira infection was clouded by the positive influenza tests at admission, delaying early Leptospira-targeted antibiotics administration. Next generation sequencing, a novel molecular diagnostic tool, provided a key hint to uncover the crucial pathogen, Leptospira interrogans, further supported by the possible occupational exposure history. Subsequent conventional penicillin and mechanical respiratory support were administrated to cure the patient successfully without any sequela.
Clinicians must pay attention to possible exposure history and keep uncommon Leptospira in mind when managing pneumonia with unknown causes.
Core Tip: Pneumonia caused by Leptospira interrogans is rare but life-threatening. In the early phase, the nonspecific symptoms of Leptospira pneumonia often lead clinicians to misdiagnosis as influenza or other illness. We herein report a 54-year-old man who presented with cough, fever and dyspnea and was misdiagnosed with influenza but was confirmed with Leptospira infection with the help of next generation sequencing. Possible exposure history and next generation sequencing during management of infections with unknown causes are important.
- Citation: Bao QH, Yu L, Ding JJ, Chen YJ, Wang JW, Pang JM, Jin Q. Severe community-acquired pneumonia caused by Leptospira interrogans: A case report and review of literature. World J Clin Cases 2021; 9(8): 1901-1908
- URL: https://www.wjgnet.com/2307-8960/full/v9/i8/1901.htm
- DOI: https://dx.doi.org/10.12998/wjcc.v9.i8.1901
Community-acquired pneumonia (CAP) is a common but multifactorial disease, and nearly a quarter are attributed to Streptococcus pneumonia[1]. Leptospira interrogans is a rare pathogen also responsible for adult CAP with a high fatality rate. With the increase of international travel, global warming and urban slum settlements, leptospirosis is re-emerging as an infectious disease worldwide, not only limited to tropic developing areas[2-4]. Relatively few cases have been reported in China in recent years. However, morbidity and mortality of Leptospira infection are thought to be underestimated because of lack of clinical vigilance, nonspecific clinical symptoms and limited availability of rapid and accurate diagnostic tests, which poses challenges to timely and effective clinical judgment and treatment[5-7].
This phenomenon may also result in little systematic research and understanding of the relationship between pneumonia and Leptospira. The application of next generation sequencing (NGS) has greatly promoted our knowledge of pneumonia-related pathogens ranging from their subtypes to their pathogenic characteristics[8]. Herein, we report a case of a 54-year-old man with severe pneumonia caused by Leptospira, which was misdiagnosed as viral pneumonia at admission. In this paper, we will discuss how we made the final diagnosis of Leptospira infection. This case will enhance the understanding of Leptospira infection and remind clinicians to remember it when managing pneumonia of unknown cause.
A 54-year-old man presented with a 4 d history of cough with white phlegm, chills and fever as well as dyspnea for 1 d. No hemoptysis, chest pain, edema or rash were found.
Before this visit, he had been given 1.5 g of intravenous cefuroxime but received little improvement. According to the above examinations, the initial diagnosis was considered severe pneumonia caused by influenza virus. Thus, the patient was treated with methylprednisolone (40 mg, q12 h), oseltamivir (75 mg, bid), tienam (0.5 g, q8 h) and moxifloxacin (0.4 g qd). Unexpectedly the next morning, the patient’s condition worsened into cough with bloody sputum and shortness of breath with decreased SpO2 to 60%. Hence, the patient was managed with intubation and continuous mechanical ventilation of positive end-expiratory pressure 8 cm H2O, FiO2 40% and SpO2 99%. Caspofungin was added to the initial therapeutic plan.
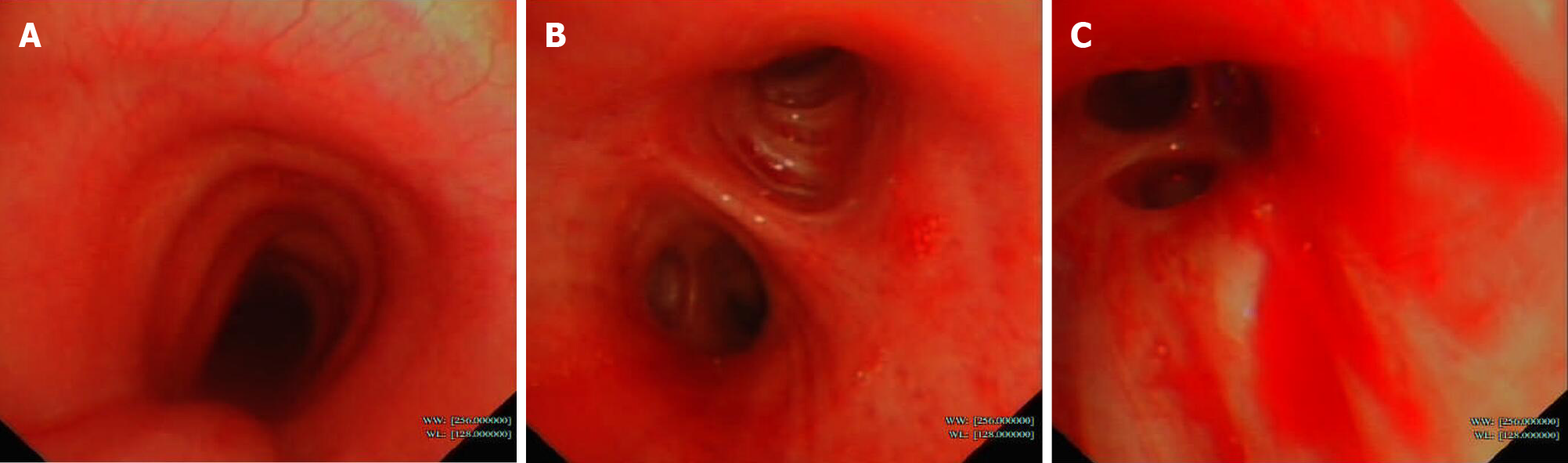
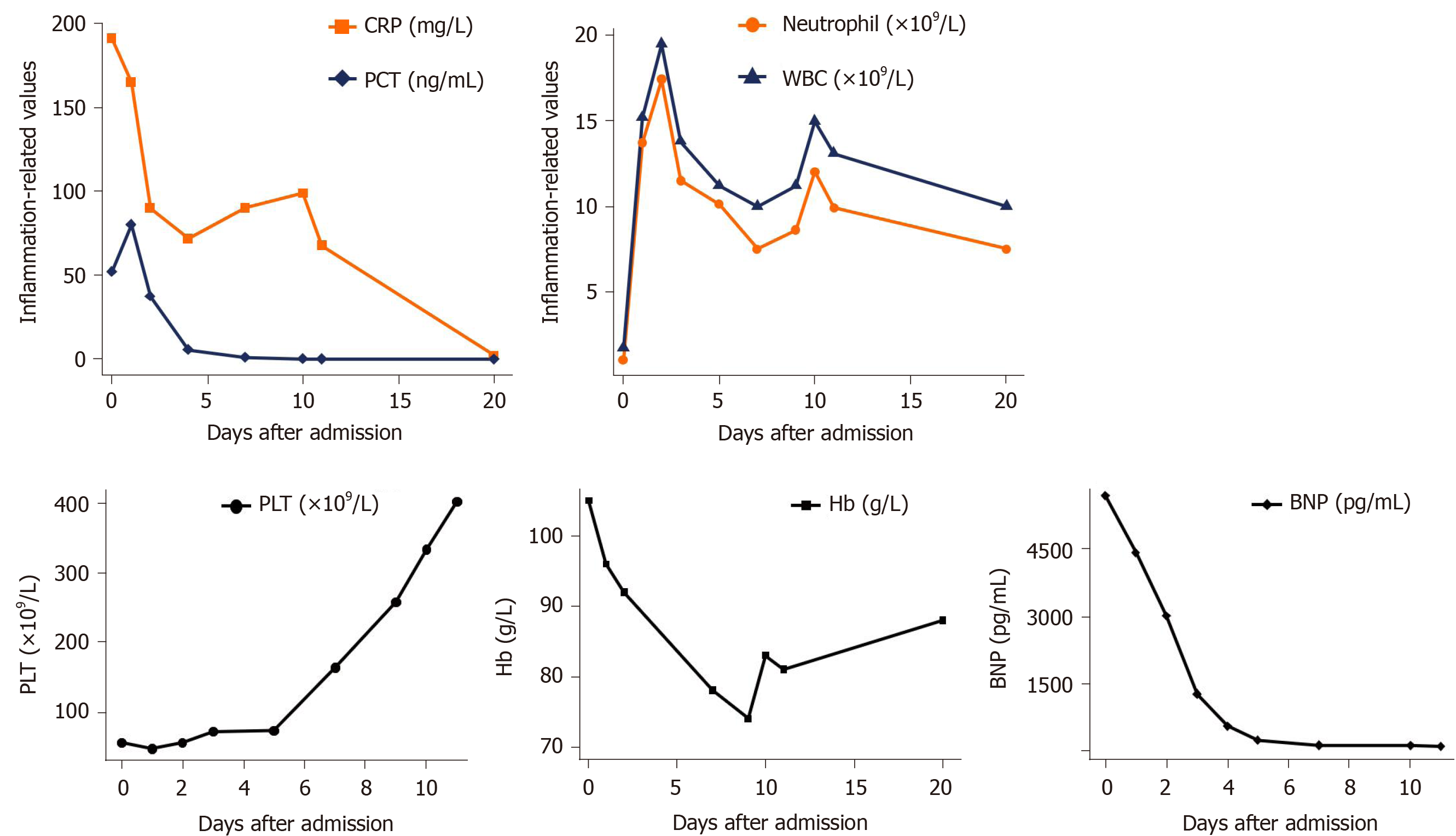
Further examinations were also performed to find the essential causes. Sputum smear test showed no positive findings. Bronchoscopy showed sporadic mucosal petechiae and bloody sputum in the bilateral bronchi (Figure 1), and bronchoalveolar lavage fluid (BALF) was collected. Echocardiography revealed left ventricular diastolic dysfunction and moderate tricuspid regurgitation with pulmonary arterial hypertension. The patient was negative for acid-fast bacilli stain, fungi test of BALF, tumor makers and a panel of respiratory viruses and autoantibodies. Under the current comprehensive treatment covering anti-infections of virus, bacteria and fungi, the patient did not improve and had persistent hemoptysis and hematuria under assisted ventilation. Inflammation and myocardial impairment-related laboratory values were elevated, and hemoglobin and platelet counts remained below normal levels (Figure 2).
The NGS report of the BALF on day 6 after admission indicated the cause. To our surprise, NGS indicated an infection of Leptospira interrogans. Subsequently, it was retrospectively interviewed that the patient worked as a rubbish cleaner of rivers with high exposure risk of Leptospira infection.
The patient reported a 30 yr history of smoking and drinking. He denied any history of hypertension, diabetes or other chronic disease.
The patient and his families denied any history of hypertension, diabetes or other chronic disease.
On examination, his temperature was 37.5 °C, pulse was 118/min, respiratory rate was 19/min, and blood pressure was 81/54 mmHg at admission. He was in a conscious state but in compulsive position with cyanotic face. His superficial lymph nodes were not swollen. Lung examination showed bilateral rales. Other physical examinations were normal.
Arterial blood gas analysis showed lower PaO2 of 64 mmHg and SpO2 of 94.3% with 2 L/min oxygen support. Laboratory investigations revealed decreased white blood cell count of 1.7 × 109/L and platelet count of 56 × 109/L, elevated C-reactive protein of 190.81 mg/L, creatinine phosphokinase of 372 U/L, creatinine of 117 μmol/L and activated partial thromboplastin ratio of 1.30 and a positive urine occult blood test. The throat swab for influenza virus antigen was positive.
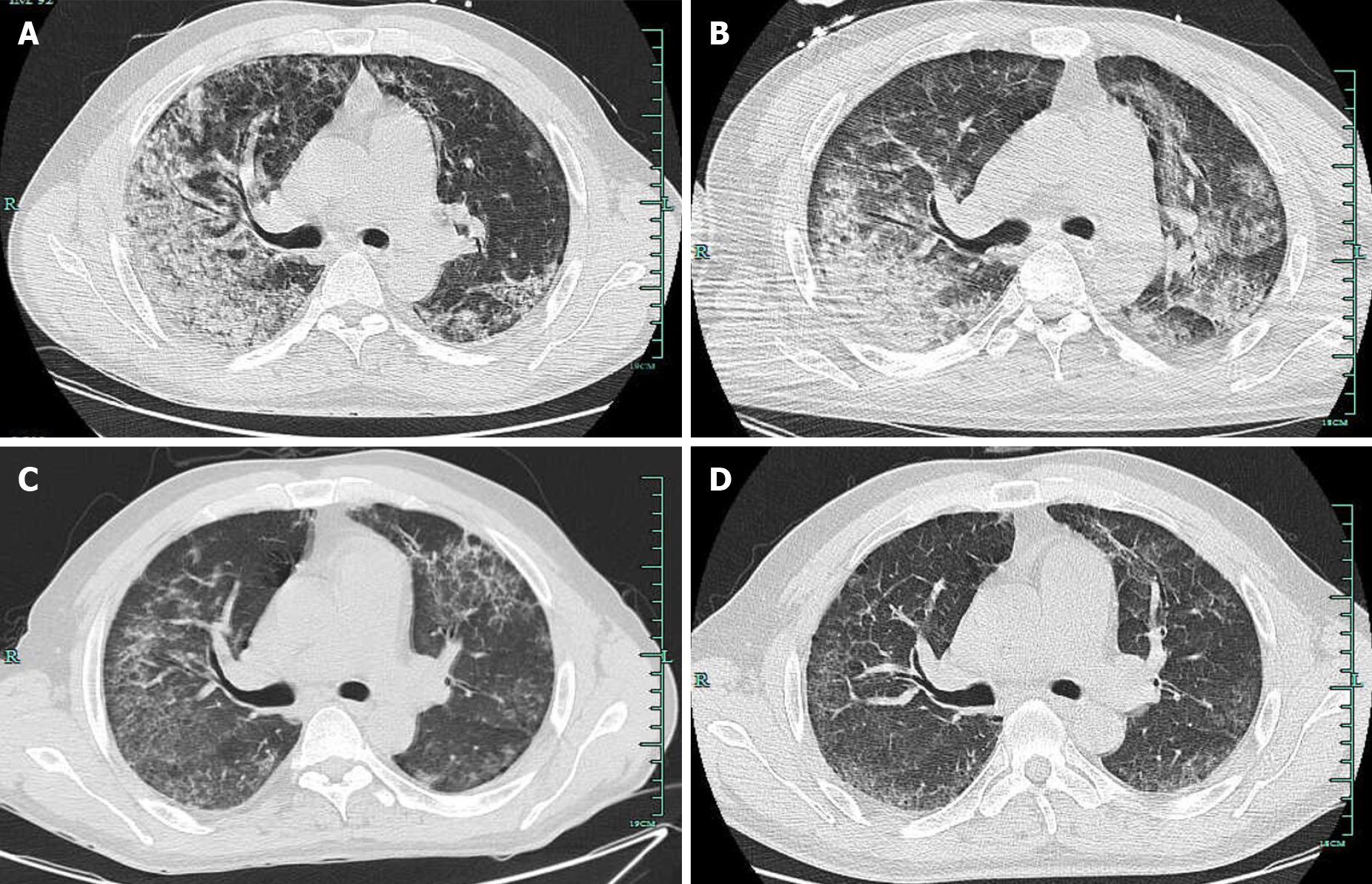
Chest computed tomography (CT) indicated infectious lesions of both lungs and emphysema with pulmonary bullae (Figure 3A).
Severe pneumonia with Leptospira interrogans infection.
After identification of the Leptospira pathogen, the patients finally received penicillin (400000 U, q12 h, im) for 1 wk as well as respiratory support, while previous administration of oseltamivir and caspofungin was stopped. After 1 wk, with advice of the infection department, the usage of penicillin was doubled for 2 d and then doubled again for another 1 wk.
After about 1 wk of penicillin treatment, the patient obviously got better with significantly relieved cough and hemoptysis as well as improved oxygenation status without mechanical ventilation (PaO2 78 mmHg, SpO2 95.3 under 5 L/min oxygen flow). Additionally, echocardiography of mild mitral and tricuspid regurgitation and decreased brain natriuretic peptide suggested the improvement of myocardial damage. Chest CT also showed the bilateral lung inflammation was greatly resolved (Figure 3C). After a total of 16 d of penicillin, the patient was discharged home without any complaint. Most laboratory values were returning to normal (Figure 2). Follow-up chest CT after discharge was almost completely cleared with no recurrence (Figure 3D).
The main pathogens of CAP in China are Streptococcus pneumoniae and Mycoplasma pneumoniae. Other common related-pathogens include Hemophilus influenzae, Chlamydia, Klebsiella pneumoniae and Staphylococcus aureus[1]. Respiratory virus, especially influenza, rhinovirus and coronavirus, is reported to be responsible for a third of adult pneumonia cases[9]. By comparison, pneumonia caused by Leptospira is rare and seldom reported. Leptospira interrogans, detected in this case, is one type of pathogenic spirochetes and could cause zoonotic leptospirosis. Leptospirosis is estimated to affect over one million people and accounts for about 60000 deaths annually worldwide[3]. Clinical manifestations of leptospirosis vary, ranging from subclinical status, mild influenza-like illness to severe multiple organ dysfunction, including kidney, liver, heart and lung. The typical severe syndrome, named “Weil’s disease”, is characterized by jaundice and renal failure with hemorrhagic complications[10].
Pulmonary involvement of leptospirosis occurs in 20%-70% of patients[11] and is mostly secondary to renal and hepatic dysfunction as an immunity-mediated status[12,13], including mild to severe pneumonia, deadly alveolar hemorrhage, adult respiratory distress syndrome, etc.[12] and manifesting as cough, dyspnea, hemoptysis, etc. As observed in this case lung involvement of severe pneumonia was primary and main manifestations were relatively few. However, mortality from severe lung involvement like adult respiratory distress syndrome and pulmonary hemorrhage greatly exceeds that from Weil’s disease (> 50% vs > 10%), although optimal treatment is given[14]. It is the nonspecificity and diversity of clinical symptoms, particularly atypical manifestations, that lead to lack of clinical suspicion, which delays targeted detection and treatment of leptospirosis and increases mortality.
Common laboratory biochemical tests of leptospirosis usually show no specific abnormalities compared with most infectious diseases. For Leptospira-related pneumonia, chest CT may show ground-glass opacities, consolidation and small pleural effusion, which could also be observed in radiographs of influenza pneumonia[12,15]. Thus, it is possible to misdiagnose this case as influenza pneumonia, especially based on evidence from the initial throat swab test. During hospitalization, bronchoscopy with BALF collection was required because it could help to identify hemorrhage even if it remained occult and to obtain samples for etiological tests. Overall, for differential diagnosis, persistent hemorrhagic complications and related abnormal values, which are relatively few in influenza-associated pneumonia under corresponding antiviral treatment, it may be the only hint to raise doubt of the initial diagnosis and treatment plan.
For the final etiological diagnosis, serological tests and isolation of Leptospira are usually considered as the prior standard diagnostic methods for leptospirosis, including conventional microscopic agglutination test and enzyme-linked immunosorbent assay[7,16], whereas patient autoimmune status, previous medications with immunosuppressor and antibiotics and timing of specimen collection lead to an inevitable bias in etiological tests[17]. Polymerase chain reaction, as the representative of current molecular tests, is another widely-used and reliable diagnostic tool[7]. However, these methods share several common drawbacks, such as failing to discriminate among widely-diverse Leptospira serotypes and only targeting some specific pathogens based on clinical prejudgments[7,18].
NGS, applied in our case, is a rapid and high-throughput assay that determines the entire nucleotide sequences of a sample in a single run without any target primers or probes[19]. NGS could identify a variety of microorganisms at one detection and provide data about typing, virulence and antimicrobial resistance simultaneously[8]. Many previous reports confirmed that NGS offers great advantages in diagnosis and management of infectious diseases of undetermined, uncommon or emerging origins[20-22] and were superior to traditional detecting techniques. It should be noted that not all microorganisms detected by NGS, like internal colonizing bacteria, bear responsibility for the diseases. To identify the crucial pathogens, clinicians need to combine NGS results with contact history, clinical symptoms and other laboratory tests to make a comprehensive interpretation.
Penicillin remains the first choice for treatment of leptospirosis recommended by the World Health Organization because no penicillin-resistant strains have been found so far. Other antibiotics like doxycycline, tetracycline and amoxicillin are also preferred. Early and suitable administration of antibiotics could shorten disease course, decrease mortality and prevent deterioration[22]. For patients with severe pulmonary illness of leptospirosis, as observed in, hemodynamic and respiratory support are urgent. Mechanical ventilation should be a priority to deal with severe pulmonary hemorrhage[23]. Recently, extracorporeal membrane oxygenation, as a salvage intervention, is also confirmed to benefit the management of severe pulmonary leptospirosis[18]. Immunomodulation therapy is expected to promote recovery from leptospirosis. Rodrigo et al[24] reviewed that steroids may only improve the outcomes of pulmonary leptospirosis but not benefit other clinical subtypes. Several reports and trials supported utilization of glucocorticoids, and Shenoy et al[25] concluded use of corticosteroids within the first 24 h of onset of pulmonary illness could reduce the need for assisted ventilation[16,25-27]. Additionally, activated factor VII, desmopressin cyclophosphamide and hemofiltration have been used in several refractory cases[28,29].
Prevention measures play an important role in management of leptospirosis. It is well-known that pathogenic Leptospira invades bodies via skin and mucosa abrasion. Indirect infection by contact with contaminated water or soil is more common than direct contact with infected mammals. Avoiding exposure to pathogens, such as wearing boots and goggles, is the most important protective measures for people with potential occupational risk, like the river-trash cleaner in this case. Although vaccines against Leptospira have been available since 1886, its general application still faces great challenges. Current qualified vaccines are restricted to partial serotypes and provide short-term immunity and are only available in several specific epidemic areas[30]. In addition, oral doxycycline can be used as chemoprophylaxis for high-risk people such as rescuers in floods[31].
In summary, severe CAP caused by Leptospira is uncommon, but it poses a considerable threat to public health. Although the lack of specific clinical symptoms and high-efficient diagnostic tools make it hard to discriminate from other infectious diseases, many measures could be taken to correctly diagnose the disease. First, high clinical suspicion of Leptospira infection is vital to handle severe pneumonia patients, especially those with hemorrhage complications. Second, recognition of high-risk people in contact with possible infected mammals or contaminated water or soil will give strong evidence to diagnosis. Third, NGS has great advantages in identification of undetermined or uncommon pathogens, which could provide hints for diagnosis and treatment. However, because of high costs and technical requirements, the application of NGS may be the final choice. Moreover, radiological and bronchoscopy tests are needed to provide further evidence to confirm and evaluate the lung involvement of leptospirosis. Given systematic leptospirosis, other organ functions, like kidney, liver and heart, need to be monitored. Early and overall assessment and timely comprehensive treatment composed of antibiotics, respiratory support and immunomodulation are critical to prevent the rapid progression, improve prognosis and promote rehabilitation of the severe lung leptospirosis.
We are grateful to the reviewers and editor for their constructive comments.
Manuscript source: Unsolicited manuscript
Specialty type: Medicine, research and experimental
Country/Territory of origin: China
Peer-review report’s scientific quality classification
Grade A (Excellent): 0
Grade B (Very good): 0
Grade C (Good): C
Grade D (Fair): 0
Grade E (Poor): 0
P-Reviewer: Chan SM S-Editor: Gao CC L-Editor: Filipodia P-Editor: Liu JH
| 1. | Prina E, Ranzani OT, Torres A. Community-acquired pneumonia. Lancet. 2015;386:1097-1108. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 365] [Cited by in RCA: 347] [Article Influence: 34.7] [Reference Citation Analysis (0)] |
| 2. | Bharti AR, Nally JE, Ricaldi JN, Matthias MA, Diaz MM, Lovett MA, Levett PN, Gilman RH, Willig MR, Gotuzzo E, Vinetz JM; Peru-United States Leptospirosis Consortium. Leptospirosis: a zoonotic disease of global importance. Lancet Infect Dis. 2003;3:757-771. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 1363] [Cited by in RCA: 1443] [Article Influence: 68.7] [Reference Citation Analysis (1)] |
| 3. | Costa F, Hagan JE, Calcagno J, Kane M, Torgerson P, Martinez-Silveira MS, Stein C, Abela-Ridder B, Ko AI. Global Morbidity and Mortality of Leptospirosis: A Systematic Review. PLoS Negl Trop Dis. 2015;9:e0003898. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 1095] [Cited by in RCA: 1121] [Article Influence: 112.1] [Reference Citation Analysis (2)] |
| 4. | Reis RB, Ribeiro GS, Felzemburgh RD, Santana FS, Mohr S, Melendez AX, Queiroz A, Santos AC, Ravines RR, Tassinari WS, Carvalho MS, Reis MG, Ko AI. Impact of environment and social gradient on Leptospira infection in urban slums. PLoS Negl Trop Dis. 2008;2:e228. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 225] [Cited by in RCA: 241] [Article Influence: 14.2] [Reference Citation Analysis (0)] |
| 5. | Karpagam KB, Ganesh B. Leptospirosis: a neglected tropical zoonotic infection of public health importance-an updated review. Eur J Clin Microbiol Infect Dis. 2020;39:835-846. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 50] [Cited by in RCA: 133] [Article Influence: 26.6] [Reference Citation Analysis (0)] |
| 6. | Soo ZMP, Khan NA, Siddiqui R. Leptospirosis: Increasing importance in developing countries. Acta Trop. 2020;201:105183. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 40] [Cited by in RCA: 70] [Article Influence: 14.0] [Reference Citation Analysis (0)] |
| 7. | Marquez A, Djelouadji Z, Lattard V, Kodjo A. Overview of laboratory methods to diagnose Leptospirosis and to identify and to type leptospires. Int Microbiol. 2017;20:184-193. [RCA] [PubMed] [DOI] [Full Text] [Cited by in RCA: 9] [Reference Citation Analysis (0)] |
| 8. | Gwinn M, MacCannell D, Armstrong GL. Next-Generation Sequencing of Infectious Pathogens. JAMA. 2019;321:893-894. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 101] [Cited by in RCA: 108] [Article Influence: 18.0] [Reference Citation Analysis (0)] |
| 9. | Ruuskanen O, Lahti E, Jennings LC, Murdoch DR. Viral pneumonia. Lancet. 2011;377:1264-1275. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 786] [Cited by in RCA: 819] [Article Influence: 58.5] [Reference Citation Analysis (0)] |
| 10. | AREAN VM. The pathologic anatomy and pathogenesis of fatal human leptospirosis (Weil's disease). Am J Pathol. 1962;40:393-423. [PubMed] |
| 11. | Carvalho CR, Bethlem EP. Pulmonary complications of leptospirosis. Clin Chest Med. 2002;23:469-478. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 38] [Cited by in RCA: 44] [Article Influence: 1.9] [Reference Citation Analysis (0)] |
| 12. | Marchiori E, Lourenço S, Setúbal S, Zanetti G, Gasparetto TD, Hochhegger B. Clinical and imaging manifestations of hemorrhagic pulmonary leptospirosis: a state-of-the-art review. Lung. 2011;189:1-9. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 48] [Cited by in RCA: 53] [Article Influence: 3.5] [Reference Citation Analysis (0)] |
| 13. | De Brito T, Silva AMGD, Abreu PAE. Pathology and pathogenesis of human leptospirosis: a commented review. Rev Inst Med Trop Sao Paulo. 2018;60:e23. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 54] [Cited by in RCA: 57] [Article Influence: 8.1] [Reference Citation Analysis (0)] |
| 14. | McBride AJ, Athanazio DA, Reis MG, Ko AI. Leptospirosis. Curr Opin Infect Dis. 2005;18:376-386. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 443] [Cited by in RCA: 456] [Article Influence: 22.8] [Reference Citation Analysis (0)] |
| 15. | Koo HJ, Lim S, Choe J, Choi SH, Sung H, Do KH. Radiographic and CT Features of Viral Pneumonia. Radiographics. 2018;38:719-739. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 382] [Cited by in RCA: 413] [Article Influence: 59.0] [Reference Citation Analysis (0)] |
| 16. | Agrawal VK, Bansal A, Pujani M. A rare case of leptospirosis with isolated lung involvement. Indian J Crit Care Med. 2015;19:174-176. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 4] [Cited by in RCA: 5] [Article Influence: 0.5] [Reference Citation Analysis (0)] |
| 17. | Haake DA, Levett PN. Leptospirosis in humans. Curr Top Microbiol Immunol. 2015;387:65-97. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 195] [Cited by in RCA: 433] [Article Influence: 43.3] [Reference Citation Analysis (0)] |
| 18. | Héry G, Letheulle J, Flécher E, Quentin C, Piau C, Le Tulzo Y, Tattevin P. Massive intra-alveolar hemorrhage caused by Leptospira serovar Djasiman in a traveler returning from Laos. J Travel Med. 2015;22:212-214. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 10] [Cited by in RCA: 12] [Article Influence: 1.2] [Reference Citation Analysis (0)] |
| 19. | Deurenberg RH, Bathoorn E, Chlebowicz MA, Couto N, Ferdous M, García-Cobos S, Kooistra-Smid AM, Raangs EC, Rosema S, Veloo AC, Zhou K, Friedrich AW, Rossen JW. Application of next generation sequencing in clinical microbiology and infection prevention. J Biotechnol. 2017;243:16-24. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 286] [Cited by in RCA: 334] [Article Influence: 37.1] [Reference Citation Analysis (0)] |
| 20. | Wilson MR, Naccache SN, Samayoa E, Biagtan M, Bashir H, Yu G, Salamat SM, Somasekar S, Federman S, Miller S, Sokolic R, Garabedian E, Candotti F, Buckley RH, Reed KD, Meyer TL, Seroogy CM, Galloway R, Henderson SL, Gern JE, DeRisi JL, Chiu CY. Actionable diagnosis of neuroleptospirosis by next-generation sequencing. N Engl J Med. 2014;370:2408-2417. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 609] [Cited by in RCA: 678] [Article Influence: 61.6] [Reference Citation Analysis (0)] |
| 21. | Briese T, Paweska JT, McMullan LK, Hutchison SK, Street C, Palacios G, Khristova ML, Weyer J, Swanepoel R, Egholm M, Nichol ST, Lipkin WI. Genetic detection and characterization of Lujo virus, a new hemorrhagic fever-associated arenavirus from southern Africa. PLoS Pathog. 2009;5:e1000455. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 363] [Cited by in RCA: 336] [Article Influence: 21.0] [Reference Citation Analysis (0)] |
| 22. | Palacios G, Druce J, Du L, Tran T, Birch C, Briese T, Conlan S, Quan PL, Hui J, Marshall J, Simons JF, Egholm M, Paddock CD, Shieh WJ, Goldsmith CS, Zaki SR, Catton M, Lipkin WI. A new arenavirus in a cluster of fatal transplant-associated diseases. N Engl J Med. 2008;358:991-998. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 541] [Cited by in RCA: 486] [Article Influence: 28.6] [Reference Citation Analysis (0)] |
| 23. | Goarant C. Leptospirosis: risk factors and management challenges in developing countries. Res Rep Trop Med. 2016;7:49-62. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 63] [Cited by in RCA: 72] [Article Influence: 8.0] [Reference Citation Analysis (0)] |
| 24. | Rodrigo C, Lakshitha de Silva N, Goonaratne R, Samarasekara K, Wijesinghe I, Parththipan B, Rajapakse S. High dose corticosteroids in severe leptospirosis: a systematic review. Trans R Soc Trop Med Hyg. 2014;108:743-750. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 57] [Cited by in RCA: 48] [Article Influence: 4.4] [Reference Citation Analysis (0)] |
| 25. | Shenoy VV, Nagar VS, Chowdhury AA, Bhalgat PS, Juvale NI. Pulmonary leptospirosis: an excellent response to bolus methylprednisolone. Postgrad Med J. 2006;82:602-606. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 53] [Cited by in RCA: 53] [Article Influence: 2.8] [Reference Citation Analysis (0)] |
| 26. | Niwattayakul K, Kaewtasi S, Chueasuwanchai S, Hoontrakul S, Chareonwat S, Suttinont C, Phimda K, Chierakul W, Silpasakorn S, Suputtamongkol Y. An open randomized controlled trial of desmopressin and pulse dexamethasone as adjunct therapy in patients with pulmonary involvement associated with severe leptospirosis. Clin Microbiol Infect. 2010;16:1207-1212. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 28] [Cited by in RCA: 32] [Article Influence: 2.0] [Reference Citation Analysis (0)] |
| 27. | Trivedi SV, Chavda RK, Wadia PZ, Sheth V, Bhagade PN, Trivedi SP, Clerk AM, Mevawala DM. The role of glucocorticoid pulse therapy in pulmonary involvement in leptospirosis. J Assoc Physicians India. 2001;49:901-903. [PubMed] |
| 28. | Trivedi SV, Vasava AH, Patel TC, Bhatia LC. Cyclophosphamide in pulmonary alveolar hemorrhage due to leptospirosis. Indian J Crit Care Med. 2009;13:79-84. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 18] [Cited by in RCA: 18] [Article Influence: 1.1] [Reference Citation Analysis (0)] |
| 29. | Tatopoulos A, Herbain D, Kazmierczak C, Bollaert PE, Gibot S. Parenteral use of recombinant activated factor VII during diffuse alveolar hemorrhage secondary to leptospirosis. Intensive Care Med. 2010;36:555-556. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 7] [Cited by in RCA: 8] [Article Influence: 0.5] [Reference Citation Analysis (0)] |
| 30. | Xu Y, Ye Q. Human leptospirosis vaccines in China. Hum Vaccin Immunother. 2018;14:984-993. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 18] [Cited by in RCA: 15] [Article Influence: 1.9] [Reference Citation Analysis (0)] |
| 31. | Schneider MC, Velasco-Hernandez J, Min KD, Leonel DG, Baca-Carrasco D, Gompper ME, Hartskeerl R, Munoz-Zanzi C. The Use of Chemoprophylaxis after Floods to Reduce the Occurrence and Impact of Leptospirosis Outbreaks. Int J Environ Res Public Health. 2017;14. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 29] [Cited by in RCA: 21] [Article Influence: 2.6] [Reference Citation Analysis (0)] |