Published online Apr 15, 2020. doi: 10.4251/wjgo.v12.i4.424
Peer-review started: December 11, 2019
First decision: December 26, 2019
Revised: December 28, 2019
Accepted: March 22, 2020
Article in press: March 22, 2020
Published online: April 15, 2020
Processing time: 126 Days and 4.9 Hours
Reports in the field of robotic surgery for rectal cancer are increasing year by year. However, most of these studies enroll patients at a relatively early stage and have small sample sizes. In fact, studies only on patients with locally advanced rectal cancer (LARC) and with relatively large sample sizes are lacking.
To investigate whether the short-term outcomes differed between robotic-assisted proctectomy (RAP) and laparoscopic-assisted proctectomy (LAP) for LARC.
The clinicopathological data of patients with LARC who underwent robotic- or laparoscopic-assisted radical surgery between January 2015 and October 2019 were collected retrospectively. To reduce patient selection bias, we used the clinical baseline characteristics of the two groups of patients as covariates for propensity-score matching (PSM) analysis. Short-term outcomes were compared between the two groups.
The clinical features were well matched in the PSM cohort. Compared with the LAP group, the RAP group had less intraoperative blood loss, lower volume of pelvic cavity drainage, less time to remove the pelvic drainage tube and urinary catheter, longer distal resection margin and lower rates of conversion (P < 0.05). However, the time to recover bowel function, the harvested lymph nodes, the postoperative length of hospital stay, and the rate of unplanned readmission within 30 days postoperatively showed no difference between the two groups (P > 0.05). The rates of total complications and all individual complications were similar between the RAP and LAP groups (P > 0.05).
This retrospective study indicated that RAP is a safe and feasible method for LARC with better short-term outcomes than LAP, but we have to admit that the clinically significant of part of indicators are relatively small in the practical situation.
Core tip: For patients with locally advanced rectal cancer, there is no consensus regarding whether robotic-assisted proctectomy and laparoscopic-assisted proctectomy is more beneficial. We conducted this retrospective cohort study to compare the short-term outcomes of robotic and laparoscopic for the rectal surgery. Compared with the laparoscopic-assisted proctectomy group, the robotic-assisted proctectomy group had less intraoperative blood loss, lower volume of pelvic cavity drainage, less time to remove the pelvic drainage tube and urinary catheter, longer distal resection margin and lower rates of conversion. Furthermore, the time to recover bowel function, the harvested lymph nodes, the postoperative length of hospital stay, the rate of unplanned readmission within 30 d postoperatively, and the rates of total complications showed no difference between the two groups.
- Citation: Ye SP, Zhu WQ, Liu DN, Lei X, Jiang QG, Hu HM, Tang B, He PH, Gao GM, Tang HC, Shi J, Li TY. Robotic- vs laparoscopic-assisted proctectomy for locally advanced rectal cancer based on propensity score matching: Short-term outcomes at a colorectal center in China. World J Gastrointest Oncol 2020; 12(4): 424-434
- URL: https://www.wjgnet.com/1948-5204/full/v12/i4/424.htm
- DOI: https://dx.doi.org/10.4251/wjgo.v12.i4.424
Colorectal cancer (CRC) is a major health concern worldwide, ranking third in men and second in women in terms of incidence, and the mortality ranks fourth in men and third in women[1]. CRC is more common in developed countries than in developing countries, but the incidence and mortality rates of CRC are rising quickly in developing countries, whereas they are stable or declining only in highly developed countries[2,3]. The proportion of colon cancer and rectal cancer also varies geographically. In the United States, rectal cancer accounts for only 28% of colorectal cancers according to colorectal cancer statistics (2017), and in the European Union, approximately 35% of CRC cases are rectal cancer[3,4]. However, the proportion of rectal cancer has reached up to 59.4%-71% of the total CRC cases in China[5]. Currently, approximately 60%-70% of CRC patients are diagnosed at an advanced stage because CRC is a disease largely without obvious symptoms during the early stage[2,6].
Currently, surgical resection is still the most effective treatment for locally advanced rectal cancer (LARC)[7]. For resectable LARC, total mesorectal excision (TME) is a standard surgical method[8]. In the past three decades, the adoption of different approaches for TME has been increasing rapidly, especially in the field of minimally invasive surgery (MIS)[2,7,9]. Several randomized controlled trials (RCTs) indicated that laparoscopic-assisted proctectomy (LAP) showed better short-term outcomes and similar long-term survival times when compared to the open proctectomy[10-13]. However, it cannot be denied that resection of rectal cancer with straight and nonarticulating laparoscopic instruments in the narrow pelvis is difficult, especially in obese male patients[14].
As another MIS system, the robotic surgery system is thought to be able to overcome some limitations of laparoscopy[15]. Over the years, the reports of robotic-assisted proctectomy have increased rapidly[16]. However, few of them have focused only on LARC, and most of them have relatively small sample sizes. In our country, most of patients with rectal cancer are diagnosed at an advanced stage due to the lack of screening, and lack of public and professional awareness of the disease[17]. Therefore, we conducted this retrospective cohort study to compare the short-term outcomes of robotic-assisted proctectomy (RAP) and laparoscopic-assisted proctectomy (LAP) for LARC.
This retrospective cohort study was approved by the institutional review board of our hospital (The First Affiliated Hospital of Nanchang University) and complied with the requirements of the Helsinki Declaration. From December 2014, our gastrointestinal center began to use the Da Vinci robotic surgery system. Since then, patients who suffered rectal cancer and had MIS planned could select their preferred operation method and sign the written operative informed consent before the operation. The choice is based on the patient's full understanding of the potential merits and demerits of the two surgical methods (RAP and LAP). All of the patients were diagnosed, staged, and evaluated using colonoscopy, chest and abdomen enhanced computed tomography, pelvic enhanced magnetic resonance imaging (MRI), tissue biopsy, CEA, CA199. All data of interest of consecutive patients who underwent RAP and LAP from December 2014 to August 2019 were extracted from the electronic medical record system maintained in our hospital.
The inclusion criteria were as follows: (1) Patient was diagnosed with LARC [cT3-4aN0-2M0 or cT1-4aN1-2M0 according to the 8th edition of the American Joint Committee on Cancer criteria (AJCC)]; (2) The distance from the lower border of the tumor to the anal verge was less than 15 cm; and (3) The patient underwent RAP or LAP.
The exclusion criteria were as follows: (1) Rectal cancer staged at 0, I, or IV (AJCC); (2) Recurrent rectal cancer, sarcoma, melanoma, or carcinosarcoma; (3) Multivisceral resection; (4) Totally robotic surgery or totally laparoscopic surgery; (5) Sigmoidostomy only; (6) Emergency surgery; (7) Severe pelvic adhesion; and (8) Invasion to adjacent organs (T4b) or distant metastasis.
To reduce the influence of potential bias caused by the limitations of this retrospective cohort study, we conducted propensity score matching (PSM) based on a logistic regression model with a match tolerance value of 0.01. We conducted one-to-one nearest-neighbor matching with covariates as follows: age, sexual status, body mass index (BMI), tumor size, ASA classification, serum CEA level, distance between the inferior margin of the tumor and the anal margin, tumor TNM stage, Dixon’s procedure or Miles’s procedure.
The short-term outcomes were compared between the two groups. In this study, the operation time was defined as after the sterile surgical towel was laid in the operation area to completion of the skin suture. The postoperative complications were defined as any deviation from the normal postoperative procedure[18]. The criteria to remove the pelvic drainage tube were similar to previously described[15]. The discharge criteria were as follows: (1) The passing of at least 5 d since surgery; (2) Successful administration of a semifluid diet and no need for intravenous nutrition; (3) A lack of complications or the presence of complications that did not require hospitalization; (4) The presence of sound mental status; and (5) The removal of all tubes.
The RAP and LAP procedures were performed by the same mini-invasive surgery team. The Da Vinci® Si system (Intuitive Surgical, Sunnyvale, CA, United States) and the KARL STORZ® HD system (KARL STORZ Endoskope, Tuttlingen, Germany) were used for RAP and LAP procedures respectively. Endotracheal intubation and general anesthesia and urethral catheter were adopted for all patients. Patients were placed in the modified lithotomy or herringbone position for Dixon’s procedure and in the lithotomy position for Miles’s procedure. The operating table was adjusted to the Trendelenburg position with a declination of 15-30°, and the right side declined with 10-15°. The medial-to-lateral approach was used in all total mesorectal excision procedures. All patients were placed with pelvic drainage tube according to Expert consensus on robotic surgery for colorectal cancer (2015 edition)[19].
The RAP procedure adopted five trocars. A 12-mm trocar was inserted in 3-4 cm to the right of and above the umbilicus for the camera, and an 8 mm trocar was inserted 6-8 cm below the costal margin of the left midclavicular line for R1. An 8 mm trocar was placed at 6-8 cm above the pubic symphysis of the midline of the abdomen for R2. An 8 mm trocar was inserted in McBurney point for R3, and a 12 mm trocar was inserted 8 cm below the point of R1 for the assistant. The intraperitoneal exploration and operation steps of RAP refer to the Chinese expert consensus on robotic surgery for colorectal cancer (2015 edition)[19].
The LAP procedure also adopted five trocars. A 10-mm trocar was inserted in the superior border of the umbilicus for the camera, a 12-mm trocar was inserted in the McBurney point as the main operating hole, and two 5-mm trocars were inserted in the outer edge of the rectus abdominis on the left and right sides of the umbilicus for the chief surgeon and assistant. A 5-mm trocar was inserted near the left-McBurney point for assistance. Most of the operation steps in the LAP were similar to the RAP.
In Dixon’s procedure, the Endo GIA was used to separate the rectum (more than 2 cm below the inferior edge of the tumor), and then, a 4-6 cm left lower abdominal rectus incision was made for operative specimen extraction. The stapler holder was inserted into the proximal colon after the removal of the specimen, and the anastomosis was performed with a 29 mm circular stapler that was inserted into the anus. Surgeons decided whether the terminal ileostomy was necessary according to the distance between the tumor and the anal margin, the anastomotic condition and their experience. Finally, a pelvic drainage tube was placed behind the anastomosis.
In Miles’s procedure, the Endo GIA was used to separate the bowel (more than 10 cm above the superior edge of the tumor) when dissected to the levator ani muscle plane, and a perineal procedure was performed manually. A permanent sigmoid colostomy was placed in the lower left abdomen. A drainage tube was placed in the pelvis.
PSM (a logistic regression model with a match caliper value of 0.01) and all statistical analyses were conducted using SPSS 22.0 software (IBM, NY, United States). A chi-squared test (Fisher's exact test) was adopted to compare categorical variables, which are expressed as numbers with percentages. A Mann-Whitney U test (Student’s t-test) was adopted to compare continuous variables, which are shown as mean ± SD and median (range). If P < 0.05, the result was considered statistically significant.
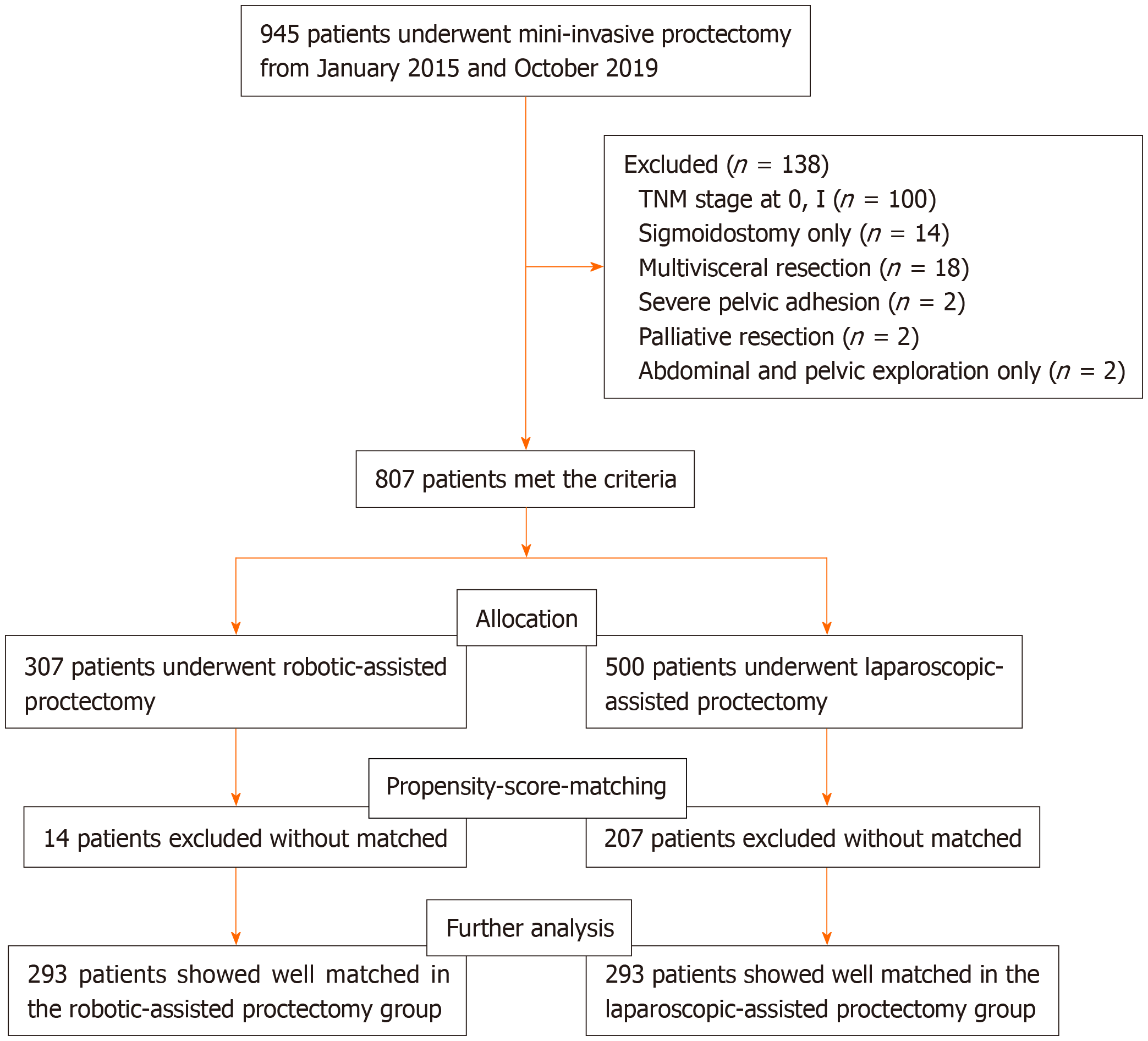
The flow chart of patient selection is displayed in Figure 1. Between January 2015 and October 2019, 945 patients underwent MIS for rectal cancer, and 807 patients met the eligibility criteria, including 500 patients in the LAP group and 307 patients in the RAP group. Finally, the PSM cohort included 586 patients for further analysis, including 338 male and 248 females, with an average age of 59.94 years (range from 26 to 89 years). After PSM, the clinicopathological features (age, sexual, BMI, tumor size, ASA classification, CEA, distance between the inferior verge of the tumor and the anal verge, tumor TNM stage, Dixon’s or Miles’s procedure) of patients between the two groups were well matched (Table 1, P > 0.05).
| Features | RAP (n = 293) | LAP (n = 293) | P value |
| Gender | 0.8671 | ||
| Male | 168 (57.3) | 170 (58.0) | |
| Female | 125 (42.7) | 123 (42.0) | |
| Age, yr | 60 ± 12, 61 (26-89) | 60 ± 11, 60 (29-86) | 0.9693 |
| Body mass index, kg/m2 | 23.16 ± 2.51 | 23.23 ± 2.48 | 0.7212 |
| Serum CEA level, ng/mL | 14.3 ± 46.7, 4.9 (0.8-760.3) | 14.5 ± 30.9, 5.8 (0.2-412.8) | 0.4293 |
| Type of resection | 0.2641 | ||
| Dixon | 240 (81.9) | 250 (85.3) | |
| Miles | 53 (18.1) | 43 (14.7) | |
| pTNM | 0.6861 | ||
| II | 64 (21.8) | 60 (20.5) | |
| III | 229 (78.2) | 233 (79.5) | |
| ASA classification | 0.5601 | ||
| 1 | 144 (49.1) | 149 (50.9) | |
| 2 | 104 (35.5) | 106 (36.9) | |
| 3 | 45 (15.4) | 36 (12.3) | |
| Diameter of neoplasm, cm | 4.2 ± 1.5, 4.0 (0.2-10.0) | 4.3 ± 1.3, 4.3 (0.3-9.0) | 0.5543 |
| Distance of tumor and anal, cm4 | 6.7 ± 2.8, 7.0 (2.0-14.0) | 6.8 ± 2.8, 7.0 (2.0-14.0) | 0.5753 |
Table 2 presents the short-term outcomes of the patients in the RAP group and LAP group. The operation time for the RAP group was longer than that for the LAP group [171 ± 42 (120-385) min vs 145 ± 42 (80-350) min, P = 0.000], but the intraoperative blood loss was less in the RAP group than in the LAP group [106 ± 114 (30–1500) mL vs 138 ± 111 (40–1200) mL, P = 0.000]. For bowel function, the time to first flatus, time on the liquid diet, and time on the semiliquid diet in the RAP group were similar to those in the LAP group (P = 0.534, 0.396, 0.194, respectively). Interestingly, the RAP group had a lower volume of pelvic drainage postoperatively [362 ± 457 (80-4020) mL vs 465 ± 564 (100-3820) mL, P = 0.000] and a shorter time to remove the pelvic drainage tube [7.1 ± 4.2 (4.0-29.0) d vs 7.8 ± 4.9 (4.0-28.0) d, P = 0.000] compared with the LAP group. The time to remove the urinary catheter was significantly shorter in the RAP group than in the LAP group [3.2 ± 1.0 (2.0-7.0) d vs 3.8 ± 1.2 (2.0-14.0) d, P = 0.000]. The distal resection margin was longer in the RAP group than the LAP group [2.7 ± 0.6 (1.8-7.1) cm vs 2.5 ± 0.5 (1.6-6.8) cm, P = 0.000]. Moreover, the RAP group was associated with lower rates of conversion to open surgery than LAP group (2.4% vs 5.8%, P = 0.037). However, the numbers of harvested lymph nodes, the rates of perineural invasion and vascular invasion, and the postoperative length of hospital stay did not differ between the RAP group and LAP group (P = 0.349, 0.557, 0.334, 0.461, respectively).
| Operative outcomes | RAP (n = 293) | LAP (n = 293) | P value |
| Operative time, min | 171 ± 42, 160 (120-385) | 145 ± 42, 133 (80-350) | 0.0003 |
| Operative blood loss, mL | 106 ± 114, 80 (30-1500) | 138 ± 111, 100 (40-1200) | 0.0003 |
| Median time to first flatus, h | 57 ± 11, 56 (28-98) | 56 ± 9, 56 (28-100) | 0.5343 |
| Median time to liquid diet, h | 69 ± 10, 67 (38-108) | 69 ± 9, 69 (38-110) | 0.3963 |
| Median time to semi-liquid diet, h | 83 ± 10, 82 (51-122) | 83 ± 9, 83 (53-123) | 0.1943 |
| Median volume of pelvic drainage, mL | 362 ± 457, 290 (80-4020) | 465 ± 564, 310 (100-3820) | 0.000 3 |
| Median time to remove pelvic drainage tube, d | 7.1 ± 4.2, 6.0 (4.0-29.0) | 7.8 ± 4.9, 7.0 (4.0-28.0) | 0.0003 |
| Median time to remove urinary catheter, d | 3.2 ± 1.0, 3.0 (2.0-7.0) | 3.8 ± 1.2, 4.0 (2.0-14.0) | 0.0003 |
| Numbers of retrieved lymph nodes | 16.0 ± 3.8 | 15.7 ± 3.7 | 0.3492 |
| Perineural invasion | 0.5571 | ||
| Yes | 124 (42.3) | 117 (39.9) | |
| No | 169 (57.7) | 176 (60.1) | |
| Vascular invasion | 0.3341 | ||
| Yes | 91 (31.1) | 102 (34.8) | |
| No | 202 (68.9) | 191 (65.2) | |
| Distal resection margin, cm | 2.7 ± 0.6, 2.7 (1.8-7.1) | 2.5 ± 0.5, 2.5 (1.6-6.8) | 0.0003 |
| Conversion to open laparotomy | 7 (2.4) | 17 (5.8) | 0.0371 |
| Postoperative length of stay, d | 9.1 ± 4.9, 8.0 (5.0-32.0) | 9.2 ± 5.0, 8.0 (5.0-30.0) | 0.4613 |
Table 3 shows the differences in complications between the LARC patients of both groups. In the minimally invasive cohort, there were 85 patients (14.5%) with complications, including 39 patients in the RAP group and 46 patients in the LAP group. The rates of complications were not significantly different between the two groups (13.3% vs 15.7%, P = 0.412). The incidence of single (10.6% vs 11.9%) or multiple complications (2.7% vs 3.8%) was similar between the two groups (P = 0.601, P = 0.484, respectively). The numbers of overall complications were comparable (16.0% vs 19.5%, P = 0.280) between the RAP group and the LAP group. Table 3 details the incidence of each complication in both groups, and the rates of each complication showed no significant difference (P > 0.05). The severity of complications based on the Clavien-Dindo classification was not different between the two groups (P > 0.05). Four patients in the RAP group underwent reoperation due to intra-abdominal hemorrhage, necrosis of the enterostomy, anastomotic hemorrhage, and anastomotic leakage, respectively. Three patients in the LAP group underwent reoperation owing to intra-abdominal hemorrhage, anastomotic leakage, and small intestinal fistula, respectively. The rates of reoperation were similar between the two groups (P = 1.000). Moreover, the rates of unplanned readmission within 30 days postoperatively were homologous between the two groups (P = 1.000). Unfortunately, one patient in the RAP group and two patients in the LAP group died because of complications (P = 1.000).
| Complications | RAP (n = 293) | LAP (n = 293) | P value |
| Patients with complications | 39 (13.3) | 46 (15.7) | 0.4121 |
| Single complication | 31 (10.6) | 35 (11.9) | 0.6011 |
| Multiple complications | 8 (2.7) | 11 (3.8) | 0.4841 |
| Overall complications | 47 (16.0) | 57 (19.5) | 0.280 1 |
| Wound infection | 8 (27) | 6 (2.0) | 0.5881 |
| Delayed gastric emptying | 0 (0.0) | 1 (0.3) | 1.0003 |
| Intestinal obstruction | 2 (0.7) | 3 (1.0) | 1.0002 |
| Intra-abdominal hemorrhage | 1 (0.3) | 2 (0.7) | 1.0002 |
| Anastomotic leakage | 13 (4.4) | 14 (4.8) | 0.8441 |
| Anastomotic bleeding | 1 (0.3) | 1 (0.3) | 1.0002 |
| Pneumonia | 4 (1.4) | 5 (1.7) | 1.0002 |
| Pleural effusion | 1 (0.3) | 1 (0.3) | 1.0002 |
| Infection of presacral space | 1 (0.3) | 5 (1.7) | 0.2182 |
| Intra-abdominal infections | 1 (0.3) | 2 (0.7) | 1.000 2 |
| Infection of incision in perineum | 6 (2.0) | 5 (1.7) | 0.7611 |
| Small intestinal fistula | 1 (0.3) | 1 (0.3) | 1.0002 |
| Peristomal wound infection | 1 (0.3) | 0 (0.0) | 1.0003 |
| Necrosis of enterostomy | 1 (0.3) | 0 (0.0) | 1.0003 |
| Urinary retention | 6 (2.0) | 10 (3.4) | 0.3111 |
| Cerebral infarction | 0 (0.0) | 1 (0.3) | 1.0003 |
| Clavien-Dindo classification | |||
| I | 15 (5.1) | 16 (5.5) | 0.8541 |
| II | 13 (4.4) | 20 (6.8) | 0.2101 |
| IIIa | 14 (4.8) | 16 (5.5) | 0.7081 |
| IIIb | 4 (1.4) | 3 (1.0) | 1.0002 |
| V | 1 (0.3) | 2 (0.7) | 1.0002 |
| ≥ III | 19 (6.5) | 21 (7.2) | 0.7431 |
| Reoperation | 4 (1.4) | 3 (1.0) | 1.0002 |
| Mortality | 1 (0.3) | 2 (0.7) | 1.0002 |
| Unplanned readmission within 30 d after operation | 2 (0.7) | 5 (1.7) | 0.4472 |
TME is a standard procedure for rectal cancer, with a high degree of difficulty, especially in male patients with a narrow pelvis, a large tumor, and high BMI. Robotic TME, as another MIS method, was invented to overcome some inherent limitations of laparoscopy and is becoming increasingly used around the world[20]. However, studies only focus on patients with LARC and with relatively large sample sizes are lacking. Therefore, to evaluate the safety and feasibility of robotic surgery for TME, we conducted this large retrospective cohort study. Because this is a retrospective study, to make the two groups of patients be more comparable, we used the basic characteristics of patients to conduct a PSM[21]. Because 807 patients met the eligibility criteria, 307 patients in the RAP group and 500 patients in the LAP group met the criteria. To obtain a better match of patients between the two groups, we conducted a 1:1 PSM with a caliper value of 0.01. Finally, there were 293 patients in each group for further analysis. The LAP group included 293 patients with the average BMI of 23.21 (17.26-29.86) kg/m2, the RAP group included 293 patients with the average BMI of 23.18 (16.82-29.73) kg/m2.
In the current research, the RAP group spent more time in the TME procedure than the LAP group, which is consistent with the results of previous studies[22]. The potential reason for the long operation time of the robot group has been explained in our previous research[23]. It is not difficult to find that the operation time reported in this study is shorter than in other similar studies[22,24]. This may be closely related to the calculation method of operation time and the proficiency of the operation team. In addition, our MIS team (including operating room nurses) has extensive experience in the TME procedure. The volume of blood loss during operation is an intuitive factor reflecting the quality of operation, which is often closely related to blood transfusion[25]. A new meta-analysis including thirty-six clinical observational studies with a total of 174036 patients indicated that perioperative transfusion causes an adverse survival prognosis and increases complications after surgery[26]. Our results show that the intraoperative blood loss was less in the RAP group than in the LAP group, which is similar to the result of previous studies[27].
The present study shows that the RAP group had a lower volume of postoperative pelvic drainage and a shorter time to remove pelvic drainage tubes than the LAP group. This is the first study to focus on the volume of postoperative pelvic drainage and the time to remove pelvic drainage for rectal cancer MIS surgery. A meta-analysis enrolling 1510 procedures showed that robotic for rectal surgery is the better method to complete mesorectum[28]. This may be the potential cause of the lower volume of postoperative pelvic drainage in the RAP group. We also found that the time to remove the urinary catheter was obviously shorter in the RAP group than in the LAP group, which was similar to our previous studies[23]. This result may shows that urinary function is damaged less in robotic TME thanks to such advantages as three-dimensional stability and high-definition images, easier identification of the pelvic nerve, and flexible instruments that facilitate fine dissection[29], this needs to be confirmed by long-term follow-up of urinary and sexual function. Mary’s study indicated that urinary catheter removal before 3 days after surgery was related with urinary retention[29]. This is the underlying reason for the average catheter time of 3.5 days in the current study. The optimal time to remove urinary catheter after rectal surgery can refer to the results of an ongoing RCT study[30]. In addition, the RAP group was associated with lower rates of conversion to open surgery than LAP group, was consistent with others’ researches[31]. Some researches indicated that lower rates of conversion are associated with lower complication rates and better long-term outcomes[32,33]. Moreover, the distal resection margin in the RAP group was significantly longer than that in the LAP group, which is similar to the Patriti’s study[34]. The potential advantage of a longer distal margin is that it can reduce the residual of skip lesions. However, the radial margin and the quality of TME were not for analysis in the current study due to the fact that our center only began to analyze these two indicators last year.
The recovery of bowel function is very important for postoperative recovery. This study shows that there is no difference between the two groups in the time to first flatus, time on a liquid diet, or time on a semiliquid diet. This is mainly because the small intestine is often in the right upper abdomen during the operation, and the operation area is mainly in the pelvis, so it has little impact on the small intestine and adjacent colon. In addition, the postoperative hospital stay, the rates of perineural invasion and vascular invasion, and the number of harvested lymph nodes were not significantly different between the two groups. The median hospital stay (8 d) after operation in the current study was similar to Perez’s study[16]. In our center, one of the discharge criteria was that the passing of at least 5 days since surgery, this is due to the underdeveloped primary medical treatment in the region of Jiangxi Province and affected by the clinical pathway. This may be one of the underlying reasons for no significant difference in postoperatively hospital stay between the two groups.
An important index to evaluate the safety and feasibility of the procedure is the incidence of postoperative complications. In the PSM cohort, there were 39 and 46 patients with complications in the RAP and LAP groups, respectively, showing no significant difference (13.3% vs 15.7%, P = 0.412). The incidence of complications was within acceptable limits, which is similar to previous studies[27]. The rates of total complications were similar between the two groups (16.0% vs 19.5%, P = 0.280). For the subgroup analysis, there were fewer patients with urinary retention in the RAP group (2.0%) than in the LAP group (3.4%), but this difference was not statistically significant (P = 0.311). Moreover, one patient had a presacral space infection in the robotic group, whereas five cases had a presacral space infection in the laparoscopic group (P = 0.218). On the severity of complications, the rates of Clavien-Dindo classifications (I, II, IIIa, IIIb, V, ≥ III) were equivalent. Unfortunately, 1 patient in the robotic group and 2 patients in the laparoscopic group died. All these deaths were attributed to complications (P = 1.000).
Finally, we do not deny that there are some deficiencies in this study. First, this is a nonrandomized controlled study with possible case selection bias. Second, we did not study the long-term oncology outcomes or cost differences between the two groups. Furthermore, there was no evaluation of the long-term differences in urogenital function.
In summary, robotic rectal surgery for LARC is safe and feasible. In the present study, the results showed that RAP for LARC was associated with less intraoperative blood loss, less volume of pelvic drainage, shorter time to remove the pelvic drainage tube and urinary catheter, lower rates of conversion, and longer distal margin than LAP. When adopting this conclusion, we need to pay attention to whether many statistically significant indicators have clinical significance. More multicenter randomized controlled studies remain to be required for the true advantages of the RAP.
Rectal cancer is a major cause of cancer-related deaths, particularly in advanced stage cases. More and more studies about minimally invasive surgery for rectal cancer are reported. However, few of them have focused only on locally advanced rectal cancer (LARC), and most of them have relatively small sample sizes. Indeed, true benefits of minimally invasive surgery (robot or laparoscopy) for LARC are still controversial.
We hope to provide clinical guidance for minimally invasive (robotic-assisted or laparoscopic-assisted) surgery of LARC.
To investigate the optimal mini-invasive proctectomy methods (robotic-assisted or laparoscopic-assisted) for advanced rectal cancer.
We retrospectively collected the clinicopathological data of patients with LARC who underwent minimally invasive surgery from January 2015 to October 2019. The propensity-score matching analysis was used to reduce patient selection bias of the current retrospective cohort study. The clinical baseline data, intraoperative and postoperative outcomes and postoperative complications were compared between the two groups.
In the current study, 293 patients were enrolled in each group. The robotic-assisted proctectomy (RAP) was associated with less intraoperative blood loss (P = 0.000), lower volume of pelvic cavity drainage (P = 0.000), less time to remove the pelvic drainage tube and urinary catheter (P = 0.000 and 0.000), longer distal resection margin (P = 0.000) and lower rates of conversion (P = 0.037) as compared with the laparoscopic-assisted proctectomy. However, the time to recover bowel function, the harvested lymph nodes, the postoperative length of hospital stay, the rate of unplanned readmission within 30 days postoperatively, complications and its subgroups were similar between the two groups, all P > 0.05.
The current retrospective cohort study revealed that RAP is a safe and feasible surgery. Compared with laparoscopic-assisted proctectomy, RAP has many advantages for LARC.
The conclusions of the present retrospective cohort study may help surgeon to develop clinical guidelines with regard to mini-invasive surgery methods in the field of LARC more perfectly. We believe that research on robotic surgery is a hot topic in the field of colorectal cancer in the future. And, high quality multicenter randomized controlled clinical trial is the optimal method for future research.
Manuscript source: Invited Manuscript
Specialty type: Oncology
Country/Territory of origin: China
Peer-review report’s scientific quality classification
Grade A (Excellent): 0
Grade B (Very good): B
Grade C (Good): C
Grade D (Fair): D
Grade E (Poor): 0
P-Reviewer: Aykan NF, Rosen SA, Rutegard J S-Editor: Gong ZM L-Editor: A E-Editor: Qi LL
| 1. | Bray F, Ferlay J, Soerjomataram I, Siegel RL, Torre LA, Jemal A. Global cancer statistics 2018: GLOBOCAN estimates of incidence and mortality worldwide for 36 cancers in 185 countries. CA Cancer J Clin. 2018;68:394-424. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 53206] [Cited by in RCA: 55839] [Article Influence: 7977.0] [Reference Citation Analysis (132)] |
| 2. | Dekker E, Tanis PJ, Vleugels JLA, Kasi PM, Wallace MB. Colorectal cancer. Lancet. 2019;394:1467-1480. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 1570] [Cited by in RCA: 3029] [Article Influence: 504.8] [Reference Citation Analysis (3)] |
| 3. | Arnold M, Sierra MS, Laversanne M, Soerjomataram I, Jemal A, Bray F. Global patterns and trends in colorectal cancer incidence and mortality. Gut. 2017;66:683-691. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 3058] [Cited by in RCA: 3306] [Article Influence: 413.3] [Reference Citation Analysis (3)] |
| 4. | Siegel RL, Miller KD, Fedewa SA, Ahnen DJ, Meester RGS, Barzi A, Jemal A. Colorectal cancer statistics, 2017. CA Cancer J Clin. 2017;67:177-193. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 2526] [Cited by in RCA: 2912] [Article Influence: 364.0] [Reference Citation Analysis (3)] |
| 5. | Gu J, Chen N. Current status of rectal cancer treatment in China. Colorectal Dis. 2013;15:1345-1350. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 38] [Cited by in RCA: 39] [Article Influence: 3.3] [Reference Citation Analysis (0)] |
| 6. | Maida M, Macaluso FS, Ianiro G, Mangiola F, Sinagra E, Hold G, Maida C, Cammarota G, Gasbarrini A, Scarpulla G. Screening of colorectal cancer: present and future. Expert Rev Anticancer Ther. 2017;17:1131-1146. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 143] [Cited by in RCA: 126] [Article Influence: 15.8] [Reference Citation Analysis (0)] |
| 7. | Glynne-Jones R, Wyrwicz L, Tiret E, Brown G, Rödel C, Cervantes A, Arnold D; ESMO Guidelines Committee. Rectal cancer: ESMO Clinical Practice Guidelines for diagnosis, treatment and follow-up. Ann Oncol. 2017;28:iv22-iv40. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 1112] [Cited by in RCA: 1199] [Article Influence: 149.9] [Reference Citation Analysis (0)] |
| 8. | Benson AB, Venook AP, Al-Hawary MM, Cederquist L, Chen YJ, Ciombor KK, Cohen S, Cooper HS, Deming D, Engstrom PF, Grem JL, Grothey A, Hochster HS, Hoffe S, Hunt S, Kamel A, Kirilcuk N, Krishnamurthi S, Messersmith WA, Meyerhardt J, Mulcahy MF, Murphy JD, Nurkin S, Saltz L, Sharma S, Shibata D, Skibber JM, Sofocleous CT, Stoffel EM, Stotsky-Himelfarb E, Willett CG, Wuthrick E, Gregory KM, Gurski L, Freedman-Cass DA. Rectal Cancer, Version 2.2018, NCCN Clinical Practice Guidelines in Oncology. J Natl Compr Canc Netw. 2018;16:874-901. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 447] [Cited by in RCA: 683] [Article Influence: 113.8] [Reference Citation Analysis (0)] |
| 9. | São Julião GP, Habr-Gama A, Vailati BB, Araujo SEA, Fernandez LM, Perez RO. New Strategies in Rectal Cancer. Surg Clin North Am. 2017;97:587-604. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 21] [Cited by in RCA: 31] [Article Influence: 3.9] [Reference Citation Analysis (0)] |
| 10. | Fleshman J, Branda M, Sargent DJ, Boller AM, George V, Abbas M, Peters WR, Maun D, Chang G, Herline A, Fichera A, Mutch M, Wexner S, Whiteford M, Marks J, Birnbaum E, Margolin D, Larson D, Marcello P, Posner M, Read T, Monson J, Wren SM, Pisters PW, Nelson H. Effect of Laparoscopic-Assisted Resection vs Open Resection of Stage II or III Rectal Cancer on Pathologic Outcomes: The ACOSOG Z6051 Randomized Clinical Trial. JAMA. 2015;314:1346-1355. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 755] [Cited by in RCA: 830] [Article Influence: 83.0] [Reference Citation Analysis (0)] |
| 11. | van der Pas MH, Haglind E, Cuesta MA, Fürst A, Lacy AM, Hop WC, Bonjer HJ; COlorectal cancer Laparoscopic or Open Resection II (COLOR II) Study Group. Laparoscopic versus open surgery for rectal cancer (COLOR II): short-term outcomes of a randomised, phase 3 trial. Lancet Oncol. 2013;14:210-218. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 1030] [Cited by in RCA: 1214] [Article Influence: 101.2] [Reference Citation Analysis (0)] |
| 12. | Hida K, Okamura R, Sakai Y, Konishi T, Akagi T, Yamaguchi T, Akiyoshi T, Fukuda M, Yamamoto S, Yamamoto M, Nishigori T, Kawada K, Hasegawa S, Morita S, Watanabe M; Japan Society of Laparoscopic Colorectal Surgery. Open versus Laparoscopic Surgery for Advanced Low Rectal Cancer: A Large, Multicenter, Propensity Score Matched Cohort Study in Japan. Ann Surg. 2018;268:318-324. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 56] [Cited by in RCA: 85] [Article Influence: 14.2] [Reference Citation Analysis (0)] |
| 13. | Kang SB, Park JW, Jeong SY, Nam BH, Choi HS, Kim DW, Lim SB, Lee TG, Kim DY, Kim JS, Chang HJ, Lee HS, Kim SY, Jung KH, Hong YS, Kim JH, Sohn DK, Kim DH, Oh JH. Open versus laparoscopic surgery for mid or low rectal cancer after neoadjuvant chemoradiotherapy (COREAN trial): short-term outcomes of an open-label randomised controlled trial. Lancet Oncol. 2010;11:637-645. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 687] [Cited by in RCA: 754] [Article Influence: 50.3] [Reference Citation Analysis (0)] |
| 14. | Araujo SE, Seid VE, Klajner S. Robotic surgery for rectal cancer: current immediate clinical and oncological outcomes. World J Gastroenterol. 2014;20:14359-14370. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in CrossRef: 55] [Cited by in RCA: 46] [Article Influence: 4.2] [Reference Citation Analysis (0)] |
| 15. | Ye SP, Shi J, Liu DN, Jiang QG, Lei X, Qiu H, Li TY. Robotic-assisted versus conventional laparoscopic-assisted total gastrectomy with D2 lymphadenectomy for advanced gastric cancer: short-term outcomes at a mono-institution. BMC Surg. 2019;19:86. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 16] [Cited by in RCA: 25] [Article Influence: 4.2] [Reference Citation Analysis (0)] |
| 16. | Perez D, Melling N, Biebl M, Reeh M, Baukloh JK, Miro J, Polonski A, Izbicki JR, Knoll B, Pratschke J, Aigner F. Robotic low anterior resection versus transanal total mesorectal excision in rectal cancer: A comparison of 115 cases. Eur J Surg Oncol. 2018;44:237-242. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 26] [Cited by in RCA: 29] [Article Influence: 3.6] [Reference Citation Analysis (0)] |
| 17. | Deng Y. Rectal Cancer in Asian vs. Western Countries: Why the Variation in Incidence? Curr Treat Options Oncol. 2017;18:64. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 45] [Cited by in RCA: 74] [Article Influence: 9.3] [Reference Citation Analysis (0)] |
| 18. | Dindo D, Demartines N, Clavien PA. Classification of surgical complications: a new proposal with evaluation in a cohort of 6336 patients and results of a survey. Ann Surg. 2004;240:205-213. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 18532] [Cited by in RCA: 24846] [Article Influence: 1183.1] [Reference Citation Analysis (0)] |
| 19. | Xu J, Qin X. Expert consensus on robotic surgery for colorectal cancer (2015 edition). Chin J Cancer. 2016;35:23. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 11] [Cited by in RCA: 16] [Article Influence: 1.8] [Reference Citation Analysis (0)] |
| 20. | Park EJ, Baik SH. Robotic Surgery for Colon and Rectal Cancer. Curr Oncol Rep. 2016;18:5. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 25] [Cited by in RCA: 32] [Article Influence: 3.6] [Reference Citation Analysis (0)] |
| 21. | Jupiter DC. Propensity Score Matching: Retrospective Randomization? J Foot Ankle Surg. 2017;56:417-420. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 50] [Cited by in RCA: 61] [Article Influence: 7.6] [Reference Citation Analysis (0)] |
| 22. | Feroci F, Vannucchi A, Bianchi PP, Cantafio S, Garzi A, Formisano G, Scatizzi M. Total mesorectal excision for mid and low rectal cancer: Laparoscopic vs robotic surgery. World J Gastroenterol. 2016;22:3602-3610. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in CrossRef: 46] [Cited by in RCA: 61] [Article Influence: 6.8] [Reference Citation Analysis (0)] |
| 23. | Ye SP, Shi J, Liu DN, Jiang QG, Lei X, Tang C, Qiu H, Li TY. [Comparative study of clinical outcomes of robot versus laparoscopic radical surgery for rectal cancer based on propensity score matching]. Zhonghua Wai Ke Za Zhi. 2019;57:447-451. [RCA] [PubMed] [DOI] [Full Text] [Cited by in RCA: 1] [Reference Citation Analysis (0)] |
| 24. | Kwak JM, Kim SH, Kim J, Son DN, Baek SJ, Cho JS. Robotic vs laparoscopic resection of rectal cancer: short-term outcomes of a case-control study. Dis Colon Rectum. 2011;54:151-156. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 142] [Cited by in RCA: 145] [Article Influence: 10.4] [Reference Citation Analysis (0)] |
| 25. | Mynster T, Nielsen HJ, Harling H, Bülow S; Danish TME-group, RANX05-group. Blood loss and transfusion after total mesorectal excision and conventional rectal cancer surgery. Colorectal Dis. 2004;6:452-457. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 17] [Cited by in RCA: 17] [Article Influence: 0.8] [Reference Citation Analysis (0)] |
| 26. | Pang QY, An R, Liu HL. Perioperative transfusion and the prognosis of colorectal cancer surgery: a systematic review and meta-analysis. World J Surg Oncol. 2019;17:7. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 38] [Cited by in RCA: 63] [Article Influence: 10.5] [Reference Citation Analysis (0)] |
| 27. | Bianchi PP, Ceriani C, Locatelli A, Spinoglio G, Zampino MG, Sonzogni A, Crosta C, Andreoni B. Robotic versus laparoscopic total mesorectal excision for rectal cancer: a comparative analysis of oncological safety and short-term outcomes. Surg Endosc. 2010;24:2888-2894. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 177] [Cited by in RCA: 190] [Article Influence: 12.7] [Reference Citation Analysis (0)] |
| 28. | Milone M, Manigrasso M, Velotti N, Torino S, Vozza A, Sarnelli G, Aprea G, Maione F, Gennarelli N, Musella M, De Palma GD. Completeness of total mesorectum excision of laparoscopic versus robotic surgery: a review with a meta-analysis. Int J Colorectal Dis. 2019;34:983-991. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 48] [Cited by in RCA: 42] [Article Influence: 7.0] [Reference Citation Analysis (0)] |
| 29. | Luca F, Valvo M, Ghezzi TL, Zuccaro M, Cenciarelli S, Trovato C, Sonzogni A, Biffi R. Impact of robotic surgery on sexual and urinary functions after fully robotic nerve-sparing total mesorectal excision for rectal cancer. Ann Surg. 2013;257:672-678. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 131] [Cited by in RCA: 128] [Article Influence: 10.7] [Reference Citation Analysis (0)] |
| 30. | Xu L, Tao ZY, Lu JY, Zhang GN, Qiu HZ, Wu B, Lin GL, Xu T, Xiao Y. A single-center, prospective, randomized clinical trial to investigate the optimal removal time of the urinary catheter after laparoscopic anterior resection of the rectum: study protocol for a randomized controlled trial. Trials. 2019;20:133. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 5] [Cited by in RCA: 6] [Article Influence: 1.0] [Reference Citation Analysis (0)] |
| 31. | Ramji KM, Cleghorn MC, Josse JM, MacNeill A, O'Brien C, Urbach D, Quereshy FA. Comparison of clinical and economic outcomes between robotic, laparoscopic, and open rectal cancer surgery: early experience at a tertiary care center. Surg Endosc. 2016;30:1337-1343. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 46] [Cited by in RCA: 61] [Article Influence: 6.1] [Reference Citation Analysis (0)] |
| 32. | Chan AC, Poon JT, Fan JK, Lo SH, Law WL. Impact of conversion on the long-term outcome in laparoscopic resection of colorectal cancer. Surg Endosc. 2008;22:2625-2630. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 102] [Cited by in RCA: 114] [Article Influence: 6.7] [Reference Citation Analysis (0)] |
| 33. | Rottoli M, Bona S, Rosati R, Elmore U, Bianchi PP, Spinelli A, Bartolucci C, Montorsi M. Laparoscopic rectal resection for cancer: effects of conversion on short-term outcome and survival. Ann Surg Oncol. 2009;16:1279-1286. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 104] [Cited by in RCA: 107] [Article Influence: 6.7] [Reference Citation Analysis (0)] |