Published online Nov 16, 2021. doi: 10.12998/wjcc.v9.i32.9711
Peer-review started: June 20, 2021
First decision: July 2, 2021
Revised: July 28, 2021
Accepted: September 15, 2021
Article in press: September 15, 2021
Published online: November 16, 2021
Processing time: 142 Days and 5.9 Hours
Gastric cancer (GC) is one of the most common malignancies worldwide and surgery remains the only potentially curative treatment option for it. Although a significant proportion of GC patients are found with distant metastases already at the initial diagnosis. Peritoneal dissemination is the most common site of metastases. Positive peritoneal cytology (Cy1) is associated with poor long-term outcomes; thus, these patients are considered as stage IV even if macroscopic carcinomatosis is absent. Currently, there is no clear evidence for the most optimal treatment for this distinct subpopulation of the stage IV cohort. Available strategies vary from palliative chemotherapy to upfront gastrectomy. This comprehensive review summarized current evidence of different treatment strategies for Cy1 GC including roles of surgery, systemic and intraperitoneal chemotherapy.
Core Tip: Positive peritoneal cytology (Cy1) is associated with poor long-term outcomes; thus, these patients are considered as stage IV even if macroscopic carcinomatosis is absent. The evidence for the most efficient treatment of these patients is conflicting. We herein review current knowledge and the outcomes of different approaches for Cy1 gastric cancers.
- Citation: Bausys A, Gricius Z, Aniukstyte L, Luksta M, Bickaite K, Bausys R, Strupas K. Current treatment strategies for patients with only peritoneal cytology positive stage IV gastric cancer. World J Clin Cases 2021; 9(32): 9711-9721
- URL: https://www.wjgnet.com/2307-8960/full/v9/i32/9711.htm
- DOI: https://dx.doi.org/10.12998/wjcc.v9.i32.9711
Gastric cancer (GC) remains an important health care issue as it is the fifth most common and the fourth most deadly cancer worldwide[1]. Surgery is the only potentially curative treatment option for it[2,3]. Although up to 30%-40% of GC patients already have distant metastases at the initial diagnosis and typically they are not candidates for radical surgery[4,5]. Peritoneal dissemination is the most common site of metastases[6]. Peritoneal lavage cytology at staging laparoscopy is the modern standard to detect peritoneal spread even before visible peritoneal carcinomatosis (PC) could be detected[7-9]. Positive cytology alone (Cy1) is a negative prognostic factor for recurrence and survival[10]; thus, it is defined as metastatic (M1) factor and Cy1 patients are considered as stage IV even in absence of macroscopic carcinomatosis.
Current clinical practice guidelines by the European Society for Medical Oncology (ESMO)[11] and National Comprehensive Cancer Network (NCCN) recommend palliative chemotherapy for Cy1 patients with a possibility for re-staging through treatment. Although, Japanese GC treatment guidelines distinguish Cy1 patients as a distinct subpopulation of the stage IV cohort and suggest considering neoadjuvant chemotherapy followed by D2 gastrectomy if other non-curative factors are absent[12]. Such discrepancies and a lack of standardization arise from the gap of current knowledge for the most efficient treatment of patients with only Cy1 stage IV GC. Therefore, this review aimed to summarize the current evidence for peritoneal dissemination in GC and various available treatment options for Cy1 stage IV patients.
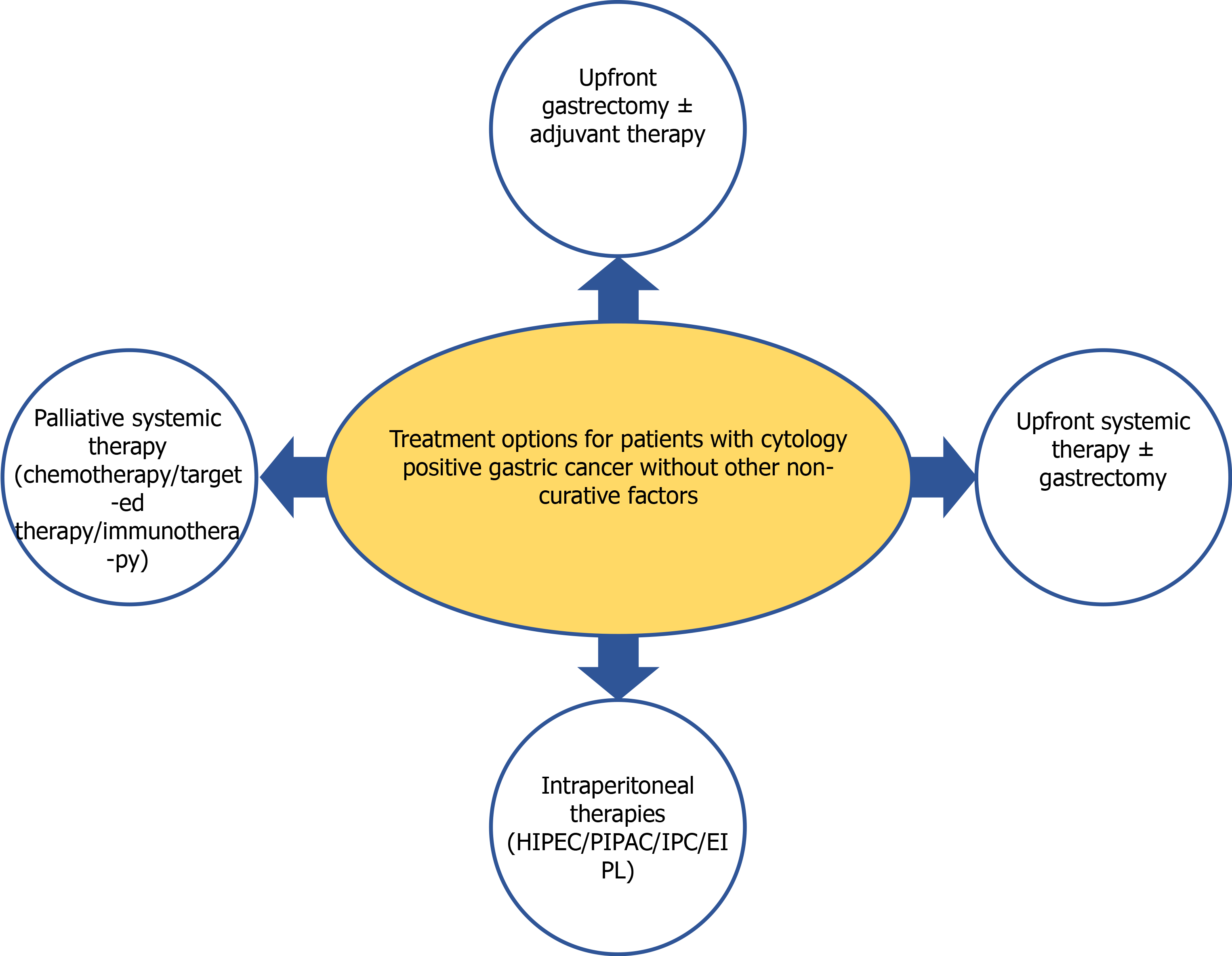
Patients with locally advanced [that penetrates subserosal connective tissue, serosa, or adjacent structures (T3 or T4) or more advanced N-stage] GC, unfavorable histological subtypes (diffuse type and/or signet ring cell component), or primary scirrhous type GC are at higher risk for peritoneal metastases[13,14]. The development of these metastases is a multistep process which includes: (1) Cancer cells detachment from the primary tumor; (2) Survival in the microenvironment of the peritoneal cavity; (3) Malignant cells attachment to peritoneal mesothelial cells and invasion through basement membrane; and (4) Tumor growth and the onset of neoangiogenesis[15]. However, not all free intraperitoneal cancer cells seed into the peritoneum and turn into PC nodes. Most of these cells die even after successful attachment to the peritoneum, because of the peritoneal-blood barrier[15]. Further, mesothelium, the innermost monolayer of the peritoneum, has some basic protective mechanism against the adhesion of exogenous cells[15]. PC develops only after some sub-population of free GC cells manage to penetrate the submesothelial space by producing specific growth factors and matrix metalloproteinases, which induce the contraction of mesothelial cells, exposing the submesothelial basement membrane[15]. The presence of free GC cells in the peritoneal cavity represents the initial stages of PC development, however, currently, there are no methods to determine at what exact stage this multistep process has been diagnosed. Thus, it remains unclear if the treatment concept for Cy1 patients should aim to treat the present peritoneal disease or should aim to prevent its further development. Because of such controversies, different strategies have been adopted for Cy1 GC worldwide (Figure 1).
Surgery remains the only potentially curative treatment option for GC[3]. However, Cy1 represents stage IV disease, thus, despite it may be technically resectable, the biological rationale for surgery is controversial. The results of the randomized controlled trial (RCT) by the Japan Clinical Oncology Group (JCOG 0705) and Korea GC Association (KGCA01), comparing gastrectomy + chemotherapy vs chemotherapy alone in advanced GC with a single non-curable factor, showed no advantage of surgery for patients with PC[16,17]. Nonetheless, palliative chemotherapy is associated with disappointing long-term outcomes and Cy1 patients represent the distinct subpopulation of GC patients with peritoneal dissemination. Therefore, more aggressive treatment strategies including surgical resections are utilized for these patients in some centers.
Upfront radical gastrectomy followed by adjuvant S-1 monotherapy was investigated in a phase II single-arm (CCOG0301) study which enrolled 48 Cy1 GC patients across the multiple treatment centers in Japan[18]. Long-term follow-up showed 5-year overall (OS) and relapse-free survival rates were 26% and 21%, respectively. Peritoneal recurrence occurred in 62% of enrolled patients[18]. Similar results were confirmed by other groups from the East[19-21]. Kano et al[19] presented a retrospective study with a median follow-up of almost 10 years. Radical gastrectomy followed by adjuvant S-1 chemotherapy resulted in a 17.8% 5-year OS rate and peritoneal recurrence rate of 52.9%[19]. Further, the study documented the benefit of adjuvant S-1 monotherapy, as the median survival increased to 22.3 mo compared to 11.8 mo in the surgery alone group[19]. The benefit of adjuvant therapy was confirmed in another study from Korea by Shim et al[20]. Adjuvant chemotherapy by TS-1 ± cisplatin or oxaliplatin plus capecitabine (XELOX) or oxaliplatin + 5-FU (FOLFOX) improved median disease-free survival (DFS) (11.63 vs 6.98 mo, P < 0.001) and OS (25.50 vs 12.11 mo, P < 0.001)[20]. No significant differences were observed between the regimen of postoperative chemotherapy and survival[20], thus the most optimal regimen remains unclear. Another retrospective study by Komatsu et al[21] analyzed upfront gastrectomy followed by adjuvant S-1 based chemotherapy in 51 Cy1 GC patients, with a special focus on the impact of surgical radicality. Radical gastrectomy with ≥ D2 Lymphonodectomy was superior compared to palliative gastrectomy with the 5-year OS of 48.2% vs 18.2%, respectively[21]. Further, the impact of surgery for Cy1 GC treatment was presented in another recent study from China[22]. Forty-eight Cy1 GC patients underwent upfront gastrectomy (75%; n = 36) or gastrectomy after neoadjuvant chemotherapy (25%; n = 12)[22]. The median OS and DFS were 22 and 16.5 mo, respectively[22]. However, the study did not provide a comparison of long-term outcomes between patients who received upfront surgery and neoadjuvant treatment[22]. In contrast, such a comparison was performed by Mezhir et al[23] In a Western cohort. Neoadjuvant therapy failed to improve DSS (1.7 vs 0.9, P = 0.76), although the relatively small sample size in the upfront surgery (n = 29) and neoadjuvant treatment groups (n = 23) should be taken into consideration[23].
Together, the current evidence indicates that radical upfront gastrectomy is feasible for Cy1 GC patients, and adjuvant chemotherapy is necessary to improve long-term outcomes. Although, most of the evidence for the upfront surgery arises from small-scale Eastern studies. Such treatment strategy needs further investigation in large-scale high-quality surgical trials, including the patients from Western parts of the world.
As mentioned previously, Cy1 GC represents the stage IV disease, thus ESMO and NCCN guidelines suggest considering systemic treatment (chemotherapy) as it improves survival and quality of life compared to best supportive care[11]. Doublet or triplet platinum/fluoropyrimidine combinations ± trastuzumab is recommended as a first-line palliative treatment[11]. Although there is no evidence for the most appropriate chemotherapy regimen to treat peritoneal metastases in GC[24], therefore, different schemes are adopted in clinical practice.
Several studies investigated the rates of conversion from positive to negative cytology following initial treatment by systemic chemotherapy[23-25]. The reported rates of conversion varied between 48.9% and 72.2% after treatment by various platinum/fluoropyrimidine combinations with or without docetaxel or trastuzumab[23-25]. Such conversion from positive to negative cytology results in improved oncological outcomes. Mezhir et al[23] showed increased disease-specific survival (2.5 vs 1.4 years) in those who converted to negative cytology. Similar, Yasufuku et al[25] and Aizawa et al[24] demonstrated improved 3-year (76.9% vs 10.5%) and 5-year (34.6% vs 17.6%) OS rates, respectively.
The high rate of conversions from positive to negative cytology and the clinical benefit of it proposes to consider the initial chemotherapy not as a palliative, but as neoadjuvant treatment. Further, the study by Badgwell et al[26] suggested, that palliative treatment may be inferior to neoadjuvant chemotherapy, despite only 41.6% of patients treated with it underwent surgery at some point of the treatment. Neoadjuvant therapy group showed a notably higher 3-year OS rate of 12% compared to 0% in patients who were considered as having incurable stage IV disease, therefore scheduled for palliative therapy only.
The upfront systemic therapy is the most promising when the conversion of cytological status is achieved, especially if converted patients can be allocated for further surgical treatment. The most effective chemotherapy regimens and the optimal number of cycles for conversions remain unknown, thus, future studies should elucidate these unclarities.
As shown previously, systemic chemotherapy in a neoadjuvant or adjuvant setting plays an important role to improve Cy1 GC patients’ outcomes. Although, systemic chemotherapy is considered to be limited efficacy for peritoneal dissemination because of the peritoneal-plasma barrier[27]. Therefore, direct intraperitoneal therapies have been suggested as a more effective alternative for these patients.
The rationale for intraperitoneal chemotherapy (IPC) application is the possibility to achieve high local concentration while keeping the low systemic concentration of cytotoxic drug[28]. These pharmacokinetic features of the method increase the therapeutic efficacy and decrease systemic toxicity. The possible limitation of IPC for the PC is the limited penetration of the drug. The maximum estimated depth of drug penetration is 3 to 5 mm, although actual penetration range from a few cell layers to a few millimeters[28]. Despite this shortcoming of the method for PC, it does not preclude the eradication of free intraperitoneal cancer cells. Thus, IPC was investigated as an attractive option for Cy1 GC patients.
Imano et al[29] conducted a pilot clinicopathological study to investigate intraperitoneal administration of 80 mg/m2 paclitaxel at the end of the radical D2 gastrectomy for 10 Cy1 GC patients. Pharmacokinetic analysis showed that the peak plasma concentration of paclitaxel did not reach the cytotoxic threshold level of 0.1 mol/L, while intraperitoneal drug concentration was about 6773 folds higher[29]. Such IPC cleared the peritoneal cytology as no viable cancer cells were found at 24 and 48 h after IPC[29]. Following radical surgery with IPC majority of patients received adjuvant S1 based chemotherapy[29]. Long-term outcome analysis showed a promising 3-years survival rate of 56% and the peritoneal recurrence rate of 30%[29]. Further, the authors compared these survival outcomes with a historical cohort who received gastrectomy alone and concluded that IPC significantly improves the survival of Cy1 GC patients[29]. Another study on IPC for Cy1 GC investigated the additional benefit of extensive peritoneal lavage (EIPL)[30]. Shimada et al[30] study included 22 Cy1 GC patients who underwent: (1) Gastrectomy; (2) Gastrectomy + IPC with 100 mg cisplatin; or (3) Gastrectomy + IPC + EIPL by peritoneal cavity washing with 10 Liters of physiologic saline solution. Postoperatively all patients received adjuvant 5-FU based chemotherapy[30]. Long-term outcomes analysis showed 2-year OS rates of 0%, 14.3%, and 57.1% in groups 1, 2 and 3, respectively. Further EIPL reduced the peritoneal recurrence rate to 42.9% compared to 85.7% and 100% in gastrectomy + IPC and gastrectomy groups, respectively. Cancer cell detection analysis in the peritoneal lavage by reverse transcriptase-polymerase chain reaction (RT-PCR) suggested 10 Liters of physiologic saline as an optimal amount to flush out the free cancer cells from the peritoneal cavity[30]. Because of the promising results in the retrospective study, the gastrectomy + EIPL + IPC strategy was tested in the subsequent multicenter RCT[31]. The study included 88 Cy1 GC patients and randomly allocated them to three previously mentioned treatment strategies[31]. This prospective study confirmed the superiority of EIPL + IPC, as the 5-year OS increased to 43.8% compared to 4.6% and 0% in IPC and gastrectomy alone groups, respectively. Further EIPL + IPC significantly reduced the peritoneal recurrence rate to 40.0% compared to 79.3% in IPC and 89.7% in gastrectomy alone groups. After the promising results of the retrospective study were confirmed in the subsequent RCT, authors recommended considering EIPL-IPC therapy as a standard prophylactic strategy for peritoneal dissemination in Cy1 GC patients[31]. However, some conflicting data on the efficacy of EIPL was presented in a recent EXPEL study. This high-quality, open-label, multicentre, phase 3 surgical RCT, conducted at 22 hospitals from South Korea, China, Japan, Malaysia, Hong Kong, and Singapore enrolled 800 patients to evaluate the potential benefit of EIPL after upfront radical gastrectomy for cT3-4 GC[32]. However, EIPL by 10 Liters of saline did not improve 3-year OS [77.0% vs 76.7%; HR: 1.09 (95%CI: 0.78-1.52); P = 0.62], DFS [64.8% vs 69.4%; HR: 1.12 (95%CI: 0.86-1.47); P = 0.40], and 3-year cumulative incidence for peritoneal recurrence [7.9% vs 6.6%; HR: 1.33 (95%CI: 0.73-2.42); P = 0.35]. Moreover, EPIL was associated with higher risk of adverse events (RR = 1.58, P = 0.019)[32,33].
Hyperthermic IPC (HIPEC) is another available method for peritoneal malignancy. It combines the benefit of IPC with the potential advantages of hyperthermia. Experimental and clinical evidence indicates that hyperthermia at a range of 41 to 43 °C destroys malignant cells by selectively increasing the number of lysosomes and lysosomal enzyme activity in malignant cells leading to increased destructive capacity[28]. Also, hyperthermia decrease blood flow in most of the malignant tumors in contrast to the opposite effect in normal tissues[28]. Such effects, together with inhibition of oxidative metabolism in malignant cells promote cell death of the more sensitive malignant cells[28]. Further, heat promotes the cytotoxic effect of the chemotherapeutical agents [28]. Thus, HIPEC was widely investigated for peritoneal disease treatment including studies in Cy1 GC patients. Meta-analysis of randomized and high-quality non-randomized trials on HIPEC for prevention and treatment of peritoneal disease in GC patients found no difference in the 3-year OS (RR = 0.99, P = 0.85) for patients with PC[34]. Although, HIPEC obtained advantages in preventing peritoneal metastases (RR = 0.63; 95%CI: 0.45-0.88; P < 0.01) in high-risk patients, including Cy1 GC patients[34]. Also, HIPEC might be applied in a neoadjuvant setting as showed by Badgwell et al[35] in a single-arm phase II study. Nineteen stage IV GC patients only by positive cytology (n = 6) or limited PC (n = 13) received up to 5 cycles of neoadjuvant laparoscopic HIPEC after initial systemic chemotherapy. In total seven (36.8%) of these converted to negative cytology and no PC and 5 of them received radical gastrectomy[35]. It is important to emphasize that the conversion rate of 66.6% (4 of 6 patients) in Cy1 patients was considerably high[35]. This aggressive treatment resulted in a 3-year OS rate of 43.5%, and the median survival of patients who received gastrectomy was 29 mo. After encouraging results of the study Badgwell et al[36] conducted another single-arm phase II study for an even more aggressive approach. Twenty patients with limited PC (n = 14) or Cy1 (n = 6) were treated with initial systemic chemotherapy followed by 1-2 Laparoscopic HIPEC procedures and then subsequent gastrectomy with a cytoreduction and intraoperative HIPEC[36]. Such an aggressive treatment resulted in a 28% 3-year OS[36]. However, it is important to note, that subgroup of Cy1 patients had a very promising result of such treatment, as 50% (n = 3) of Cy1 were alive and recurrence-free at 32-49 mo after diagnosis. Despite the encouraging initial results on HIPEC for Cy1 patients, there is a lack of data from high-quality large-scale RCTs. Currently, an ongoing phase III GASTRICHIP trial[37] is designed to evaluate the effect of HIPEC in patients with a high risk of peritoneal recurrence, including Cy1 patients after neoadjuvant chemotherapy[37]. The long-term outcomes will be available in 2023 and the results will elucidate some unclarities regarding HIPEC's role for Cy1 GC patients[34].
Another new and emerging technique for a peritoneal disease is pressurized IPC (PIPAC). During PIPAC, laparoscopic access is obtained to create a pneumoperitoneum of 12 mmHg and nebulized chemotherapy is applied to create therapeutic capnoperitoneum for 30 min[38]. The rationale for PIPAC includes: (1) Optimization of drug distribution by applying an aerosol rather than a liquid solution; (2) Applying increased intraperitoneal hydrostatic pressure to increase drug penetration to the target; and (3) Limiting blood outflow during drug application[39,40]. Further, the minimally invasive approach of PIPAC allows multiple applications of the procedure and objective reassessment of the response through laparoscopy and biopsies[39]. Similar to laparoscopic HIPEC, PIPAC may be utilized in a neoadjuvant setting and also in combination with systemic therapy. Several retrospective and prospective phase II studies suggested that PIPAC may be a safe and promising option for GC patients with PC[41-44], although, there is a lack of data for its efficacy in a specific cohort of Cy1 patients.
All above-mentioned treatment strategies could be considered as experimental, as the standard treatment option for M1 GC remains palliative systemic therapy[11]. Doublet or triplet platinum/fluoropyrimidine combinations are recommended for fit patients with M1 GC (including Cy1 patients) as standard conventional chemotherapy options[11]. Although, such treatment remains associated with poor outcomes[45], thus novel treatment options, like targeted therapy and immunotherapy, are of interest for these patients.
One of the available options, already included in a clinical practice guideline is trastuzumab - a monoclonal antibody targeting human epidermal growth factor receptor 2 (HER2). It induces antibody-dependent cellular cytotoxicity, inhibits HER2-mediated signaling, and prevents cleavage of the extracellular domain of HER2[46]. Large scale ToGA RCT showed that trastuzumab in combination with chemotherapy increases the survival of advanced or M1 HER2-positive GC patients[47]. A recent study showed trastuzumab deruxtecan, a humanized monoclonal anti-HER2 antibody attached to a cytotoxic topoisomerase I inhibitor through a cleavable linker is available and effective as a third-line treatment for HER2 positive GC patients[48]. Some other HER-2 targeting agents such as lapatinib, trastuzumab emtansine, pertuzumab are also available, although their efficacy remains controversial[49_52]. Another available targeted therapy agent is ramucirumab, a fully humanized monoclonal antibody against vascular endothelial growth factor receptor 2[53]. This angiogenesis inhibitor was included in treatment guidelines as a second-line treatment option for patients with M1 GC after encouraging results of the REGARD and RAINBOW studies[54,55].
Another novel and promising drug class for M1 GC is immune checkpoint inhibitors. Some of these drugs improve antitumor T-cell activity by inhibiting immune checkpoints such as the programmed death-1 receptor (PD-1) and programmed death-ligand 1 (PDL1). PD1 is expressed on the surface of activated T cells that regulate their proliferation and activation and PDL1 is a major ligand for PD-1 expressed in some cancers, including GC cells[56,57]. Nivolumab is one of the available immune checkpoint inhibitors recommended in combination with fluorou
Despite some promising results of novel targeted therapy and immunotherapy drugs for M1 GC, the exact benefit for a distinct cohort of Cy1 GC patients remains unclear, as none of the current studies investigated this distinct subpopulation. Further studies are needed, to elucidate, the potential of novel systemic therapies for these patients.
The knowledge provided by the current evidence has some limitations. First, most of the available studies are relatively small in sample size. Second, many different treatment strategies including upfront gastrectomy, surgery after neoadjuvant systemic therapy, and IPC have been described for Cy1 GC, however, there is a lack of studies that would have compared them with each other. Thus, further large-scale international cohort studies comparing different treatments are needed to establish the most promising options. After, these should be tested in subsequent multi-center randomized control trials to provide robust evidence on the most efficient treatment for Cy1 patients.
Positive peritoneal cytology is associated with poor long-term outcomes in GC patients. Although, current evidence indicates, that this distinct subpopulation of the stage IV cohort may benefit from more aggressive treatment than palliative chemotherapy. Available strategies include upfront gastrectomy followed by adjuvant therapy, neoadjuvant chemotherapy option, and different methods of IPC utilization. Although, the most optimal treatment remains unclear because there is a lack of comparative studies. Thus, further clinical trials are needed to establish the best treatment option for Cy1 GC.
Provenance and peer review: Invited article; Externally peer reviewed
Specialty type: Medicine, research and experimental
Country/Territory of origin: Lithuania
Peer-review report’s scientific quality classification
Grade A (Excellent): A
Grade B (Very good): 0
Grade C (Good): 0
Grade D (Fair): 0
Grade E (Poor): 0
P-Reviewer: An T S-Editor: Wang JJ L-Editor: A P-Editor: Xing YX
| 1. | Sung H, Ferlay J, Siegel RL, Laversanne M, Soerjomataram I, Jemal A, Bray F. Global Cancer Statistics 2020: GLOBOCAN Estimates of Incidence and Mortality Worldwide for 36 Cancers in 185 Countries. CA Cancer J Clin. 2021;71:209-249. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 75126] [Cited by in RCA: 64630] [Article Influence: 16157.5] [Reference Citation Analysis (176)] |
| 2. | Stratilatovas E, Baušys A, Baušys R, Sangaila E. Mortality after gastrectomy: a 10 year single institution experience. Acta Chir Belg. 2015;115:123-130. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 14] [Cited by in RCA: 11] [Article Influence: 1.1] [Reference Citation Analysis (0)] |
| 3. | Bausys R, Bausys A, Vysniauskaite I, Maneikis K, Stratilatovas E, Strupas K. Surgical treatment outcomes of patients with T1-T2 gastric cancer: does the age matter when excellent treatment results are expected? World J Surg Oncol. 2018;16:79. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 7] [Cited by in RCA: 14] [Article Influence: 2.0] [Reference Citation Analysis (0)] |
| 4. | Bernards N, Creemers GJ, Nieuwenhuijzen GA, Bosscha K, Pruijt JF, Lemmens VE. No improvement in median survival for patients with metastatic gastric cancer despite increased use of chemotherapy. Ann Oncol. 2013;24:3056-3060. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 90] [Cited by in RCA: 121] [Article Influence: 10.1] [Reference Citation Analysis (0)] |
| 5. | Zhang Y, Lin Y, Duan J, Xu K, Mao M, Wang X. A Population-Based Analysis of Distant Metastasis in Stage IV Gastric Cancer. Med Sci Monit. 2020;26:e923867. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 5] [Cited by in RCA: 13] [Article Influence: 2.6] [Reference Citation Analysis (0)] |
| 6. | Shi B, Lin H, Zhang M, Lu W, Qu Y, Zhang H. Gene Regulation and Targeted Therapy in Gastric Cancer Peritoneal Metastasis: Radiological Findings from Dual Energy CT and PET/CT. J Vis Exp. 2018;. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 3] [Cited by in RCA: 4] [Article Influence: 0.6] [Reference Citation Analysis (0)] |
| 7. | Nakajima T, Harashima S, Hirata M, Kajitani T. Prognostic and therapeutic values of peritoneal cytology in gastric cancer. Acta Cytol. 1978;22:225-229. [PubMed] |
| 8. | Bando E, Yonemura Y, Takeshita Y, Taniguchi K, Yasui T, Yoshimitsu Y, Fushida S, Fujimura T, Nishimura G, Miwa K. Intraoperative lavage for cytological examination in 1,297 patients with gastric carcinoma. Am J Surg. 1999;178:256-262. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 242] [Cited by in RCA: 242] [Article Influence: 9.3] [Reference Citation Analysis (0)] |
| 9. | Yamaguchi T, Takashima A, Nagashima K, Makuuchi R, Aizawa M, Ohashi M, Tashiro K, Yamada T, Kinoshita T, Hata H, Kawachi Y, Kawabata R, Tsuji T, Hihara J, Sakamoto T, Fukagawa T, Katai H, Higuchi K, Boku N. Efficacy of Postoperative Chemotherapy After Resection that Leaves No Macroscopically Visible Disease of Gastric Cancer with Positive Peritoneal Lavage Cytology (CY1) or Localized Peritoneum Metastasis (P1a): A Multicenter Retrospective Study. Ann Surg Oncol. 2020;27:284-292. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 15] [Cited by in RCA: 34] [Article Influence: 5.7] [Reference Citation Analysis (0)] |
| 10. | Allen CJ, Newhook TE, Vreeland TJ, Das P, Minsky BD, Blum M, Song S, Ajani J, Ikoma N, Mansfield PF, Roy-Chowdhuri S, Badgwell BD. Yield of peritoneal cytology in staging patients with gastric and gastroesophageal cancer. J Surg Oncol. 2019;120:1350-1357. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 20] [Cited by in RCA: 19] [Article Influence: 3.2] [Reference Citation Analysis (0)] |
| 11. | Smyth EC, Verheij M, Allum W, Cunningham D, Cervantes A, Arnold D; ESMO Guidelines Committee. Gastric cancer: ESMO Clinical Practice Guidelines for diagnosis, treatment and follow-up. Ann Oncol. 2016;27:v38-v49. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 869] [Cited by in RCA: 1118] [Article Influence: 124.2] [Reference Citation Analysis (0)] |
| 12. | Japanese Gastric Cancer Association. Japanese gastric cancer treatment guidelines 2018 (5th edition). Gastric Cancer. 2021;24:1-21. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 735] [Cited by in RCA: 1336] [Article Influence: 334.0] [Reference Citation Analysis (2)] |
| 13. | Rau B, Brandl A, Piso P, Pelz J, Busch P, Demtröder C, Schüle S, Schlitt HJ, Roitman M, Tepel J, Sulkowski U, Uzunoglu F, Hünerbein M, Hörbelt R, Ströhlein M, Beckert S, Königsrainer I, Königsrainer A; Peritoneum Surface Oncology Group and members of the StuDoQ|Peritoneum Registry of the German Society for General and Visceral Surgery (DGAV). Peritoneal metastasis in gastric cancer: results from the German database. Gastric Cancer. 2020;23:11-22. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 54] [Cited by in RCA: 83] [Article Influence: 16.6] [Reference Citation Analysis (0)] |
| 14. | Thomassen I, van Gestel YR, van Ramshorst B, Luyer MD, Bosscha K, Nienhuijs SW, Lemmens VE, de Hingh IH. Peritoneal carcinomatosis of gastric origin: a population-based study on incidence, survival and risk factors. Int J Cancer. 2014;134:622-628. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 412] [Cited by in RCA: 414] [Article Influence: 37.6] [Reference Citation Analysis (0)] |
| 15. | Kanda M, Kodera Y. Molecular mechanisms of peritoneal dissemination in gastric cancer. World J Gastroenterol. 2016;22:6829-6840. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in CrossRef: 99] [Cited by in RCA: 120] [Article Influence: 13.3] [Reference Citation Analysis (1)] |
| 16. | Fujitani K, Yang HK, Kurokawa Y, Park DJ, Tsujinaka T, Park BJ, Fukuda H, Noh SH, Boku N, Bang YJ, Sasako M, Lee JI; Gastric Cancer Surgical Study Group of Japan Clinical Oncology Group; Korea Gastric Cancer Association. Randomized controlled trial comparing gastrectomy plus chemotherapy with chemotherapy alone in advanced gastric cancer with a single non-curable factor: Japan Clinical Oncology Group Study JCOG 0705 and Korea Gastric Cancer Association Study KGCA01. Jpn J Clin Oncol. 2008;38:504-506. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 49] [Cited by in RCA: 45] [Article Influence: 2.6] [Reference Citation Analysis (0)] |
| 17. | Jamel S, Markar SR, Malietzis G, Acharya A, Athanasiou T, Hanna GB. Prognostic significance of peritoneal lavage cytology in staging gastric cancer: systematic review and meta-analysis. Gastric Cancer. 2018;21:10-18. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 46] [Cited by in RCA: 73] [Article Influence: 10.4] [Reference Citation Analysis (0)] |
| 18. | Kodera Y, Ito S, Mochizuki Y, Ohashi N, Tanaka C, Kobayashi D, Kojima H, Matsui T, Kondo K, Fujiwara M. Long-term follow up of patients who were positive for peritoneal lavage cytology: final report from the CCOG0301 study. Gastric Cancer. 2012;15:335-337. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 54] [Cited by in RCA: 64] [Article Influence: 4.9] [Reference Citation Analysis (0)] |
| 19. | Kano K, Aoyama T, Maezawa Y, Nakajima T, Ikeda K, Yamada T, Sato T, Oshima T, Rino Y, Masuda M, Ogata T, Cho H, Yoshikawa T. The survival and prognosticators of peritoneal cytology-positive gastric cancer patients who received upfront gastrectomy and subsequent S-1 chemotherapy. Int J Clin Oncol. 2017;22:887-896. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 10] [Cited by in RCA: 14] [Article Influence: 1.8] [Reference Citation Analysis (0)] |
| 20. | Shim HJ, Kim HJ, Lee SH, Bae WK, Hwang EC, Cho SH, Chung IJ, Bang HJ, Hwang JE. Observational Study of Peritoneal Washing Cytology-Positive Gastric Cancer without Gross Peritoneal Metastasis in Patients who Underwent Radical D2 Gastrectomy. Sci Rep. 2020;10:9549. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 7] [Cited by in RCA: 14] [Article Influence: 2.8] [Reference Citation Analysis (0)] |
| 21. | Komatsu S, Shioaki Y, Furuke H, Ohta A, Tsuji R, Tanaka S, Kumano T, Imura KI, Shimomura K, Ikeda J, Taniguchi F, Ueshima Y, Lee CJ, Deguchi E, Ikeda E, Otsuji E. Is curative gastrectomy justified for gastric cancer with cytology positive as the only stage IV factor? Langenbecks Arch Surg. 2019;404:599-604. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 3] [Cited by in RCA: 3] [Article Influence: 0.5] [Reference Citation Analysis (0)] |
| 22. | Kang WZ, Zhong YX, Ma FH, Xue LY, Xiong JP, Ma S, Li Y, Xie YB, Quan X, Tian YT. Survival outcomes and prognostic indicators for gastric cancer patients with positive peritoneal wash cytology but no peritoneal metastasis after radical gastrectomy. World J Gastrointest Oncol. 2021;13:24-36. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in CrossRef: 4] [Cited by in RCA: 2] [Article Influence: 0.5] [Reference Citation Analysis (0)] |
| 23. | Mezhir JJ, Shah MA, Jacks LM, Brennan MF, Coit DG, Strong VE. Positive peritoneal cytology in patients with gastric cancer: natural history and outcome of 291 patients. Ann Surg Oncol. 2010;17:3173-3180. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 124] [Cited by in RCA: 130] [Article Influence: 8.7] [Reference Citation Analysis (0)] |
| 24. | Aizawa M, Nashimoto A, Yabusaki H, Nakagawa S, Matsuki A, Homma K, Kawasaki T. The clinical significance of potentially curative resection for gastric cancer following the clearance of free cancer cells in the peritoneal cavity by induction chemotherapy. Surg Today. 2015;45:611-617. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 13] [Cited by in RCA: 19] [Article Influence: 1.7] [Reference Citation Analysis (0)] |
| 25. | Yasufuku I, Nunobe S, Ida S, Kumagai K, Ohashi M, Hiki N, Sano T. Conversion therapy for peritoneal lavage cytology-positive type 4 and large type 3 gastric cancer patients selected as candidates for R0 resection by diagnostic staging laparoscopy. Gastric Cancer. 2020;23:319-327. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 23] [Cited by in RCA: 42] [Article Influence: 8.4] [Reference Citation Analysis (0)] |
| 26. | Badgwell B, Cormier JN, Krishnan S, Yao J, Staerkel GA, Lupo PJ, Pisters PW, Feig B, Mansfield P. Does neoadjuvant treatment for gastric cancer patients with positive peritoneal cytology at staging laparoscopy improve survival? Ann Surg Oncol. 2008;15:2684-2691. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 72] [Cited by in RCA: 87] [Article Influence: 5.1] [Reference Citation Analysis (0)] |
| 27. | de Bree E, Michelakis D, Stamatiou D, Romanos J, Zoras O. Pharmacological principles of intraperitoneal and bidirectional chemotherapy. Pleura Peritoneum. 2017;2:47-62. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 74] [Cited by in RCA: 72] [Article Influence: 9.0] [Reference Citation Analysis (0)] |
| 28. | González-Moreno S, González-Bayón LA, Ortega-Pérez G. Hyperthermic intraperitoneal chemotherapy: Rationale and technique. World J Gastrointest Oncol. 2010;2:68-75. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in CrossRef: 176] [Cited by in RCA: 191] [Article Influence: 12.7] [Reference Citation Analysis (0)] |
| 29. | Imano M, Imamoto H, Itoh T, Satou T, Peng YF, Yasuda A, Kato H, Nishiki K, Shiraishi O, Shinkai M, Tsubaki M, Yasuda T, Nishida S, Takeyama Y, Okuno K, Shiozaki H. Impact of intraperitoneal chemotherapy after gastrectomy with positive cytological findings in peritoneal washings. Eur Surg Res. 2011;47:254-259. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 20] [Cited by in RCA: 20] [Article Influence: 1.4] [Reference Citation Analysis (0)] |
| 30. | Shimada S, Tanaka E, Marutsuka T, Honmyo U, Tokunaga H, Yagi Y, Aoki N, Ogawa M. Extensive intraoperative peritoneal lavage and chemotherapy for gastric cancer patients with peritoneal free cancer cells. Gastric Cancer. 2002;5:168-172. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 36] [Cited by in RCA: 44] [Article Influence: 2.0] [Reference Citation Analysis (0)] |
| 31. | Kuramoto M, Shimada S, Ikeshima S, Matsuo A, Yagi Y, Matsuda M, Yonemura Y, Baba H. Extensive intraoperative peritoneal lavage as a standard prophylactic strategy for peritoneal recurrence in patients with gastric carcinoma. Ann Surg. 2009;250:242-246. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 167] [Cited by in RCA: 187] [Article Influence: 11.7] [Reference Citation Analysis (0)] |
| 32. | Yang HK, Ji J, Han SU, Terashima M, Li G, Kim HH, Law S, Shabbir A, Song KY, Hyung WJ, Kosai NR, Kono K, Misawa K, Yabusaki H, Kinoshita T, Lau PC, Kim YW, Rao JR, Ng E, Yamada T, Yoshida K, Park DJ, Tai BC, So JBY; EXPEL study group. Extensive peritoneal lavage with saline after curative gastrectomy for gastric cancer (EXPEL): a multicentre randomised controlled trial. Lancet Gastroenterol Hepatol. 2021;6:120-127. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 15] [Cited by in RCA: 25] [Article Influence: 5.0] [Reference Citation Analysis (0)] |
| 33. | Pocard M, Reymond MA. "Peritoneal failure": A new concept to explain negative results of randomized trials evaluating intraperitoneal therapies. Pleura Peritoneum. 2020;5:20200117. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 2] [Cited by in RCA: 3] [Article Influence: 0.6] [Reference Citation Analysis (0)] |
| 34. | Desiderio J, Chao J, Melstrom L, Warner S, Tozzi F, Fong Y, Parisi A, Woo Y. The 30-year experience-A meta-analysis of randomised and high-quality non-randomised studies of hyperthermic intraperitoneal chemotherapy in the treatment of gastric cancer. Eur J Cancer. 2017;79:1-14. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 168] [Cited by in RCA: 153] [Article Influence: 19.1] [Reference Citation Analysis (0)] |
| 35. | Badgwell B, Blum M, Das P, Estrella J, Wang X, Ho L, Fournier K, Royal R, Mansfield P, Ajani J. Phase II Trial of Laparoscopic Hyperthermic Intraperitoneal Chemoperfusion for Peritoneal Carcinomatosis or Positive Peritoneal Cytology in Patients with Gastric Adenocarcinoma. Ann Surg Oncol. 2017;24:3338-3344. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 100] [Cited by in RCA: 95] [Article Influence: 11.9] [Reference Citation Analysis (0)] |
| 36. | Badgwell B, Ikoma N, Murphy MB, Wang X, Estrella J, Roy-Chowdhuri S, Das P, Minsky BD, Lano E, Song S, Mansfield P, Ajani J. A Phase II Trial of Cytoreduction, Gastrectomy, and Hyperthermic Intraperitoneal Perfusion with Chemotherapy for Patients with Gastric Cancer and Carcinomatosis or Positive Cytology. Ann Surg Oncol. 2021;28:258-264. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 54] [Cited by in RCA: 48] [Article Influence: 12.0] [Reference Citation Analysis (0)] |
| 37. | Glehen O, Passot G, Villeneuve L, Vaudoyer D, Bin-Dorel S, Boschetti G, Piaton E, Garofalo A. GASTRICHIP: D2 resection and hyperthermic intraperitoneal chemotherapy in locally advanced gastric carcinoma: a randomized and multicenter phase III study. BMC Cancer. 2014;14:183. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 120] [Cited by in RCA: 146] [Article Influence: 13.3] [Reference Citation Analysis (0)] |
| 38. | Gamboa AC, Winer JH. Cytoreductive Surgery and Hyperthermic Intraperitoneal Chemotherapy for Gastric Cancer. Cancers (Basel). 2019;11. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 33] [Cited by in RCA: 43] [Article Influence: 7.2] [Reference Citation Analysis (0)] |
| 39. | Nadiradze G, Horvath P, Sautkin Y, Archid R, Weinreich FJ, Königsrainer A, Reymond MA. Overcoming Drug Resistance by Taking Advantage of Physical Principles: Pressurized Intraperitoneal Aerosol Chemotherapy (PIPAC). Cancers (Basel). 2019;12. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 27] [Cited by in RCA: 50] [Article Influence: 8.3] [Reference Citation Analysis (0)] |
| 40. | Graversen M, Detlefsen S, Bjerregaard JK, Fristrup CW, Pfeiffer P, Mortensen MB. Prospective, single-center implementation and response evaluation of pressurized intraperitoneal aerosol chemotherapy (PIPAC) for peritoneal metastasis. Ther Adv Med Oncol. 2018;10:1758835918777036. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 54] [Cited by in RCA: 59] [Article Influence: 8.4] [Reference Citation Analysis (0)] |
| 41. | Struller F, Horvath P, Solass W, Weinreich FJ, Strumberg D, Kokkalis MK, Fischer I, Meisner C, Königsrainer A, Reymond MA. Pressurized intraperitoneal aerosol chemotherapy with low-dose cisplatin and doxorubicin (PIPAC C/D) in patients with gastric cancer and peritoneal metastasis: a phase II study. Ther Adv Med Oncol. 2019;11:1758835919846402. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 40] [Cited by in RCA: 69] [Article Influence: 11.5] [Reference Citation Analysis (0)] |
| 42. | Gockel I, Jansen-Winkeln B, Haase L, Rhode P, Mehdorn M, Niebisch S, Moulla Y, Lyros O, Lordick F, Schierle K, Wittekind C, Thieme R. Pressurized Intraperitoneal Aerosol Chemotherapy (PIPAC) in Gastric Cancer Patients with Peritoneal Metastasis (PM): Results of a Single-Center Experience and Register Study. J Gastric Cancer. 2018;18:379-391. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 42] [Cited by in RCA: 58] [Article Influence: 8.3] [Reference Citation Analysis (0)] |
| 43. | Ellebæk SB, Graversen M, Detlefsen S, Lundell L, Fristrup CW, Pfeiffer P, Mortensen MB. Pressurized intraperitoneal aerosol chemotherapy (PIPAC) of peritoneal metastasis from gastric cancer: a descriptive cohort study. Clin Exp Metastasis. 2020;37:325-332. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 9] [Cited by in RCA: 27] [Article Influence: 5.4] [Reference Citation Analysis (0)] |
| 44. | Kurtz F, Struller F, Horvath P, Solass W, Bösmüller H, Königsrainer A, Reymond MA. Feasibility, Safety, and Efficacy of Pressurized Intraperitoneal Aerosol Chemotherapy (PIPAC) for Peritoneal Metastasis: A Registry Study. Gastroenterol Res Pract. 2018;2018:2743985. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 39] [Cited by in RCA: 49] [Article Influence: 7.0] [Reference Citation Analysis (0)] |
| 45. | Frattini F, Rausei S, Chiappa C, Rovera F, Boni L, Dionigi G. Prognosis and treatment of patients with positive peritoneal cytology in advanced gastric cancer. World J Gastrointest Surg. 2013;5:135-137. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in CrossRef: 7] [Cited by in RCA: 6] [Article Influence: 0.5] [Reference Citation Analysis (0)] |
| 46. | Hudis CA. Trastuzumab--mechanism of action and use in clinical practice. N Engl J Med. 2007;357:39-51. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 1728] [Cited by in RCA: 1924] [Article Influence: 106.9] [Reference Citation Analysis (0)] |
| 47. | Bang YJ, Van Cutsem E, Feyereislova A, Chung HC, Shen L, Sawaki A, Lordick F, Ohtsu A, Omuro Y, Satoh T, Aprile G, Kulikov E, Hill J, Lehle M, Rüschoff J, Kang YK; ToGA Trial Investigators. Trastuzumab in combination with chemotherapy versus chemotherapy alone for treatment of HER2-positive advanced gastric or gastro-oesophageal junction cancer (ToGA): a phase 3, open-label, randomised controlled trial. Lancet. 2010;376:687-697. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 5541] [Cited by in RCA: 5324] [Article Influence: 354.9] [Reference Citation Analysis (3)] |
| 48. | Shitara K, Bang YJ, Iwasa S, Sugimoto N, Ryu MH, Sakai D, Chung HC, Kawakami H, Yabusaki H, Lee J, Saito K, Kawaguchi Y, Kamio T, Kojima A, Sugihara M, Yamaguchi K; DESTINY-Gastric01 Investigators. Trastuzumab Deruxtecan in Previously Treated HER2-Positive Gastric Cancer. N Engl J Med. 2020;382:2419-2430. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 915] [Cited by in RCA: 828] [Article Influence: 165.6] [Reference Citation Analysis (0)] |
| 49. | Tabernero J, Hoff PM, Shen L, Ohtsu A, Shah MA, Cheng K, Song C, Wu H, Eng-Wong J, Kim K, Kang YK. Pertuzumab plus trastuzumab and chemotherapy for HER2-positive metastatic gastric or gastro-oesophageal junction cancer (JACOB): final analysis of a double-blind, randomised, placebo-controlled phase 3 study. Lancet Oncol. 2018;19:1372-1384. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 368] [Cited by in RCA: 357] [Article Influence: 51.0] [Reference Citation Analysis (0)] |
| 50. | Thuss-Patience PC, Shah MA, Ohtsu A, Van Cutsem E, Ajani JA, Castro H, Mansoor W, Chung HC, Bodoky G, Shitara K, Phillips GDL, van der Horst T, Harle-Yge ML, Althaus BL, Kang YK. Trastuzumab emtansine versus taxane use for previously treated HER2-positive locally advanced or metastatic gastric or gastro-oesophageal junction adenocarcinoma (GATSBY): an international randomised, open-label, adaptive, phase 2/3 study. Lancet Oncol. 2017;18:640-653. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 265] [Cited by in RCA: 388] [Article Influence: 48.5] [Reference Citation Analysis (0)] |
| 51. | Satoh T, Xu RH, Chung HC, Sun GP, Doi T, Xu JM, Tsuji A, Omuro Y, Li J, Wang JW, Miwa H, Qin SK, Chung IJ, Yeh KH, Feng JF, Mukaiyama A, Kobayashi M, Ohtsu A, Bang YJ. Lapatinib plus paclitaxel versus paclitaxel alone in the second-line treatment of HER2-amplified advanced gastric cancer in Asian populations: TyTAN--a randomized, phase III study. J Clin Oncol. 2014;32:2039-2049. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 507] [Cited by in RCA: 490] [Article Influence: 44.5] [Reference Citation Analysis (0)] |
| 52. | Hecht JR, Bang YJ, Qin SK, Chung HC, Xu JM, Park JO, Jeziorski K, Shparyk Y, Hoff PM, Sobrero A, Salman P, Li J, Protsenko SA, Wainberg ZA, Buyse M, Afenjar K, Houé V, Garcia A, Kaneko T, Huang Y, Khan-Wasti S, Santillana S, Press MF, Slamon D. Lapatinib in Combination With Capecitabine Plus Oxaliplatin in Human Epidermal Growth Factor Receptor 2-Positive Advanced or Metastatic Gastric, Esophageal, or Gastroesophageal Adenocarcinoma: TRIO-013/LOGiC--A Randomized Phase III Trial. J Clin Oncol. 2016;34:443-451. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 466] [Cited by in RCA: 477] [Article Influence: 53.0] [Reference Citation Analysis (0)] |
| 53. | Young K, Smyth E, Chau I. Ramucirumab for advanced gastric cancer or gastro-oesophageal junction adenocarcinoma. Therap Adv Gastroenterol. 2015;8:373-383. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 18] [Cited by in RCA: 17] [Article Influence: 1.7] [Reference Citation Analysis (0)] |
| 54. | Fuchs CS, Tomasek J, Yong CJ, Dumitru F, Passalacqua R, Goswami C, Safran H, Dos Santos LV, Aprile G, Ferry DR, Melichar B, Tehfe M, Topuzov E, Zalcberg JR, Chau I, Campbell W, Sivanandan C, Pikiel J, Koshiji M, Hsu Y, Liepa AM, Gao L, Schwartz JD, Tabernero J; REGARD Trial Investigators. Ramucirumab monotherapy for previously treated advanced gastric or gastro-oesophageal junction adenocarcinoma (REGARD): an international, randomised, multicentre, placebo-controlled, phase 3 trial. Lancet. 2014;383:31-39. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 1541] [Cited by in RCA: 1574] [Article Influence: 143.1] [Reference Citation Analysis (0)] |
| 55. | Wilke H, Muro K, Van Cutsem E, Oh SC, Bodoky G, Shimada Y, Hironaka S, Sugimoto N, Lipatov O, Kim TY, Cunningham D, Rougier P, Komatsu Y, Ajani J, Emig M, Carlesi R, Ferry D, Chandrawansa K, Schwartz JD, Ohtsu A; RAINBOW Study Group. Ramucirumab plus paclitaxel versus placebo plus paclitaxel in patients with previously treated advanced gastric or gastro-oesophageal junction adenocarcinoma (RAINBOW): a double-blind, randomised phase 3 trial. Lancet Oncol. 2014;15:1224-1235. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 1613] [Cited by in RCA: 1768] [Article Influence: 160.7] [Reference Citation Analysis (0)] |
| 56. | Sexton RE, Al Hallak MN, Diab M, Azmi AS. Gastric cancer: a comprehensive review of current and future treatment strategies. Cancer Metastasis Rev. 2020;39:1179-1203. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 459] [Cited by in RCA: 460] [Article Influence: 92.0] [Reference Citation Analysis (0)] |
| 57. | Akyala AI, Verhaar AP, Peppelenbosch MP. Immune checkpoint inhibition in gastric cancer: A systematic review. Journal of Cellular Immunotherapy. 2018;4:49-55. [RCA] [DOI] [Full Text] [Cited by in Crossref: 7] [Cited by in RCA: 7] [Article Influence: 1.0] [Reference Citation Analysis (0)] |
| 58. | Janjigian YY, Shitara K, Moehler M, Garrido M, Salman P, Shen L, Wyrwicz L, Yamaguchi K, Skoczylas T, Campos Bragagnoli A, Liu T, Schenker M, Yanez P, Tehfe M, Kowalyszyn R, Karamouzis MV, Bruges R, Zander T, Pazo-Cid R, Hitre E, Feeney K, Cleary JM, Poulart V, Cullen D, Lei M, Xiao H, Kondo K, Li M, Ajani JA. First-line nivolumab plus chemotherapy versus chemotherapy alone for advanced gastric, gastro-oesophageal junction, and oesophageal adenocarcinoma (CheckMate 649): a randomised, open-label, phase 3 trial. Lancet. 2021;398:27-40. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 1962] [Cited by in RCA: 1898] [Article Influence: 474.5] [Reference Citation Analysis (1)] |
| 59. | Kang YK, Boku N, Satoh T, Ryu MH, Chao Y, Kato K, Chung HC, Chen JS, Muro K, Kang WK, Yeh KH, Yoshikawa T, Oh SC, Bai LY, Tamura T, Lee KW, Hamamoto Y, Kim JG, Chin K, Oh DY, Minashi K, Cho JY, Tsuda M, Chen LT. Nivolumab in patients with advanced gastric or gastro-oesophageal junction cancer refractory to, or intolerant of, at least two previous chemotherapy regimens (ONO-4538-12, ATTRACTION-2): a randomised, double-blind, placebo-controlled, phase 3 trial. Lancet. 2017;390:2461-2471. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 1283] [Cited by in RCA: 1714] [Article Influence: 214.3] [Reference Citation Analysis (0)] |
| 60. | Fuchs CS, Doi T, Jang RW, Muro K, Satoh T, Machado M, Sun W, Jalal SI, Shah MA, Metges JP, Garrido M, Golan T, Mandala M, Wainberg ZA, Catenacci DV, Ohtsu A, Shitara K, Geva R, Bleeker J, Ko AH, Ku G, Philip P, Enzinger PC, Bang YJ, Levitan D, Wang J, Rosales M, Dalal RP, Yoon HH. Safety and Efficacy of Pembrolizumab Monotherapy in Patients With Previously Treated Advanced Gastric and Gastroesophageal Junction Cancer: Phase 2 Clinical KEYNOTE-059 Trial. JAMA Oncol. 2018;4:e180013. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 1413] [Cited by in RCA: 1461] [Article Influence: 208.7] [Reference Citation Analysis (0)] |
| 61. | Shitara K, Van Cutsem E, Bang YJ, Fuchs C, Wyrwicz L, Lee KW, Kudaba I, Garrido M, Chung HC, Lee J, Castro HR, Mansoor W, Braghiroli MI, Karaseva N, Caglevic C, Villanueva L, Goekkurt E, Satake H, Enzinger P, Alsina M, Benson A, Chao J, Ko AH, Wainberg ZA, Kher U, Shah S, Kang SP, Tabernero J. Efficacy and Safety of Pembrolizumab or Pembrolizumab Plus Chemotherapy vs Chemotherapy Alone for Patients With First-line, Advanced Gastric Cancer: The KEYNOTE-062 Phase 3 Randomized Clinical Trial. JAMA Oncol. 2020;6:1571-1580. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 821] [Cited by in RCA: 867] [Article Influence: 173.4] [Reference Citation Analysis (1)] |