Published online Sep 26, 2021. doi: 10.12998/wjcc.v9.i27.8207
Peer-review started: April 19, 2021
First decision: June 23, 2021
Revised: July 9, 2021
Accepted: August 6, 2021
Article in press: August 6, 2021
Published online: September 26, 2021
Processing time: 149 Days and 18.3 Hours
Awake craniotomy has been widely used for tumor resection, epilepsy surgery, deep brain stimulation, and carotid endarterectomy. The report on awake artery malformation clipping is rare, especially for anesthesia management.
A 62-year-old female diagnosed with malformation of anterior cerebral artery at the right side. We clipped the artery malformation with intraoperative neuromonitoring (IONM) in awake craniotomy. Spontaneous respiration was maintained throughout the procedure by nasopharyngeal airway during the surgery success
The technique of monitoring anesthesia care can be performed successfully for the patient with IONM.
Core Tip: In this case, due to the patient’s strong request for the integrity of motor function, the neurosurgeon explained the advantages and disadvantages of different surgery plans to her. This patient chose the awake craniotomy finally. Awake cranio
- Citation: Zhou HY, Chen HY, Li Y. Anesthetic technique for awake artery malformation clipping with motor evoked potential and somatosensory evoked potential: A case report. World J Clin Cases 2021; 9(27): 8207-8213
- URL: https://www.wjgnet.com/2307-8960/full/v9/i27/8207.htm
- DOI: https://dx.doi.org/10.12998/wjcc.v9.i27.8207
Awake craniotomy aims to minimize the risk of postoperative neurologic deficits. Over the last decades, awake craniotomy has been widely used for epilepsy lesions, glioma near the functional procedure. Awake brain mapping may facilitate resection with the preservation of functions such as speech and memory, that cannot be monitored in asleep patients. Few cases of awake craniotomy for intracranial artery malformation were reported. Awake craniotomy along with intraoperative neuromonitoring improve the treatment for artery malformation clipping. We present a case of awake artery malformation clipping with motor evoked potential (MEP) and somatosensory evoked potential (SSEP).
A 62-year-old female was treated in our hospital with malformation of an anterior cerebral artery at the right side.
The malformation of the right anterior cerebral artery was found by medical exa
The patient had no past history.
The patient had no personal or family history.
The physical examination of the patient was normal.
Preoperative blood biochemistry, electrocardiogram, chest X-ray were normal.
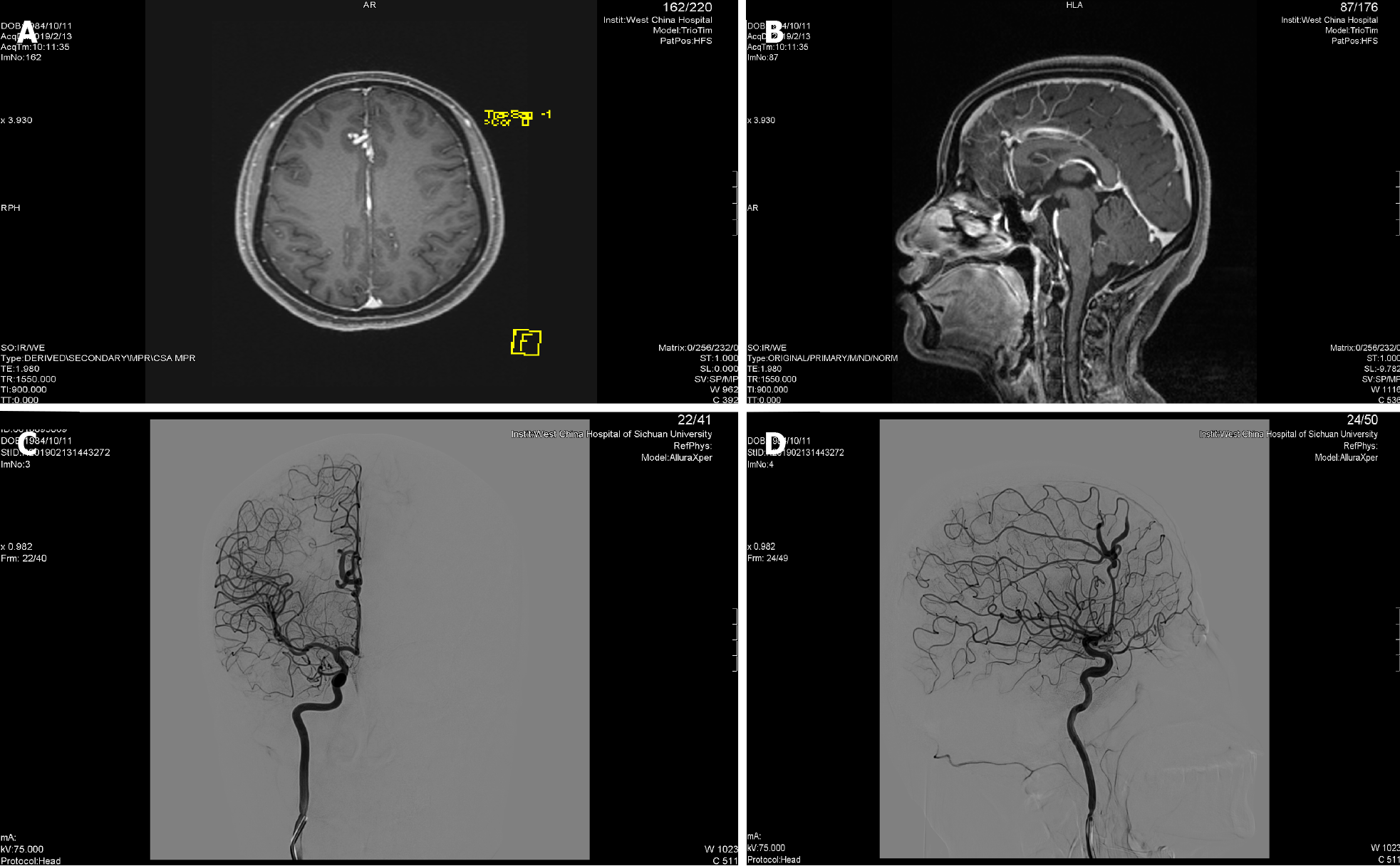
The brain computed tomography (CT) shows multiple nodular and stripy high-density shadows in the right frontal lobe and anterior cerebral falx. The magnetic resonance imaging (MRI) revealed A3 and A4 segment malformation of the right anterior cerebral artery before admission (Figure 1A and B). After the patient was admitted to the hospital, the patient was diagnosed with right anterior cerebral artery malfor
The definitive diagnosis of this patient was the malformation of the right anterior cerebral artery.
In this case, due to the patient’s strong request for the integrity of motor function, the neurosurgeon explained the advantages and disadvantages of different surgery plans to her. This patient chose the awake craniotomy finally. The written informed consent was obtained from the patient preoperatively. The electrocardiogram, noninvasive blood pressure, pulse oximetry, bispectral index (BIS) were monitored continuously. A Mayfield frame was used with ropivacaine prior to pin insertion. The patient was placed in a supine position comfortably.
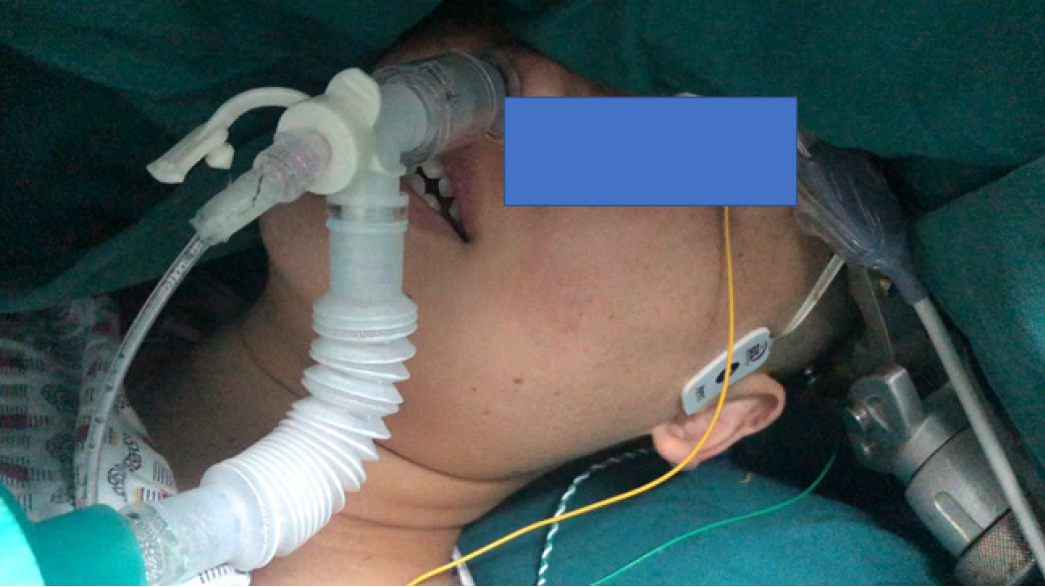
Motor function was evaluated by the electrophysiologist preoperatively. Anesthesia was induced with propofol target controlled infusion of 2 ug/mL, remifentanil 0.02-0.04 ug/kg/min and dexmedetomidine 0.5 ug/kg/h. After the patient’s consciousness disappeared, a nasopharyngeal airway was placed to supply oxygen and monitor end-tidal carbon dioxide (Figure 2). The transcranial stimulating electrodes were placed for MEP and SSEP. Latency and amplitude of MEP and SSEP were recorded. Warning criteria for MEP and SSEP was a 10% increase in latency or a 50% decrease in ampli
Scalp nerve block including supraorbital nerve, auriculotemporal nerve, greater occipital nerve and lesser occipital nerve bilaterally, was performed with 0.2% ro
According to various monitoring parameters, the dose was titrated to maintain BIS at 60 to 80, end-tidal carbon dioxide (ETCO2) at 35 to 50mmol/L, respiratory rate (RR) at 10-15 beats/min, heart rate at 50-100 beats/min, and mean artery pressure at 60 to 80 mmHg. Continuous intravenous infusion with propofol 1.4-1.6 ug/kg, remifentanil 0.02-0.04 ug/kg/min and dexmedetomidine 0.5 ug/kg/h maintained sedation during the pre-awake period. The dura mater was infiltrated with 1% lidocaine before inci
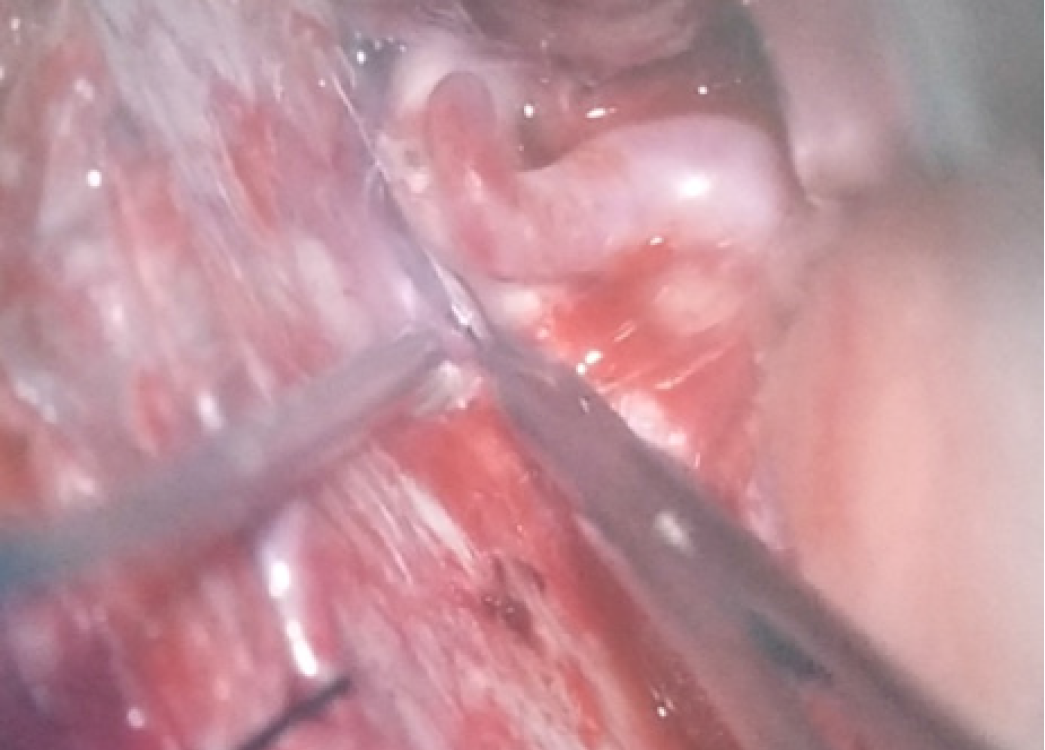
Once the artery malformation was identified (Figure 3), all anesthetics were inter
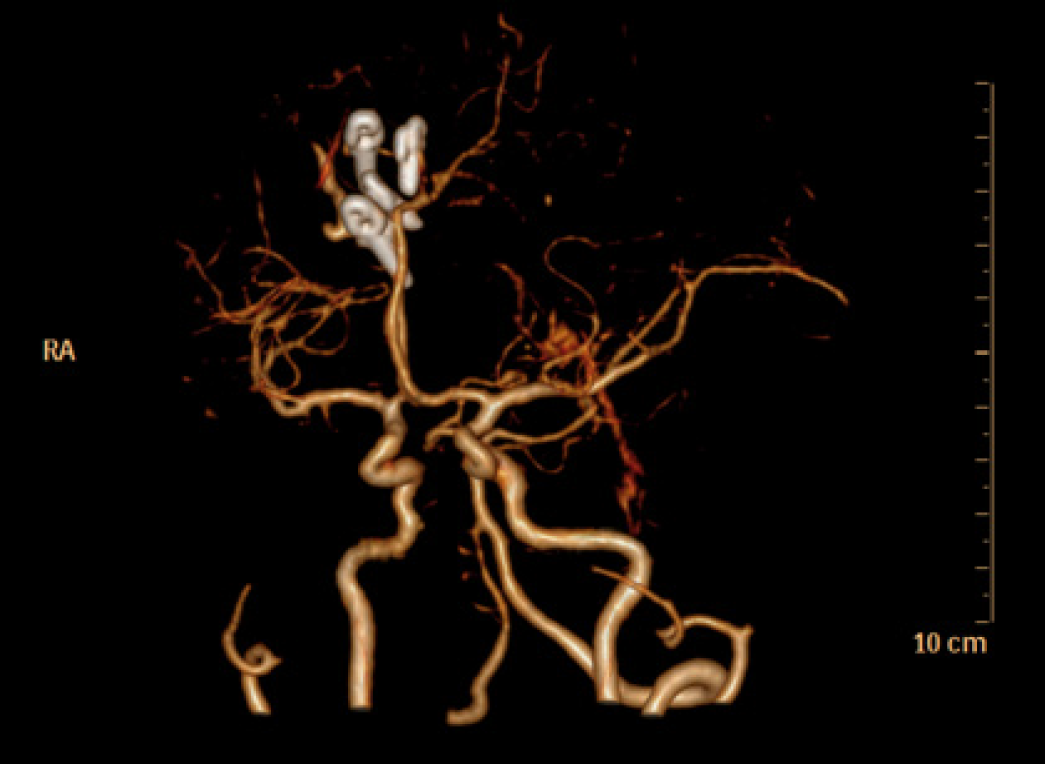
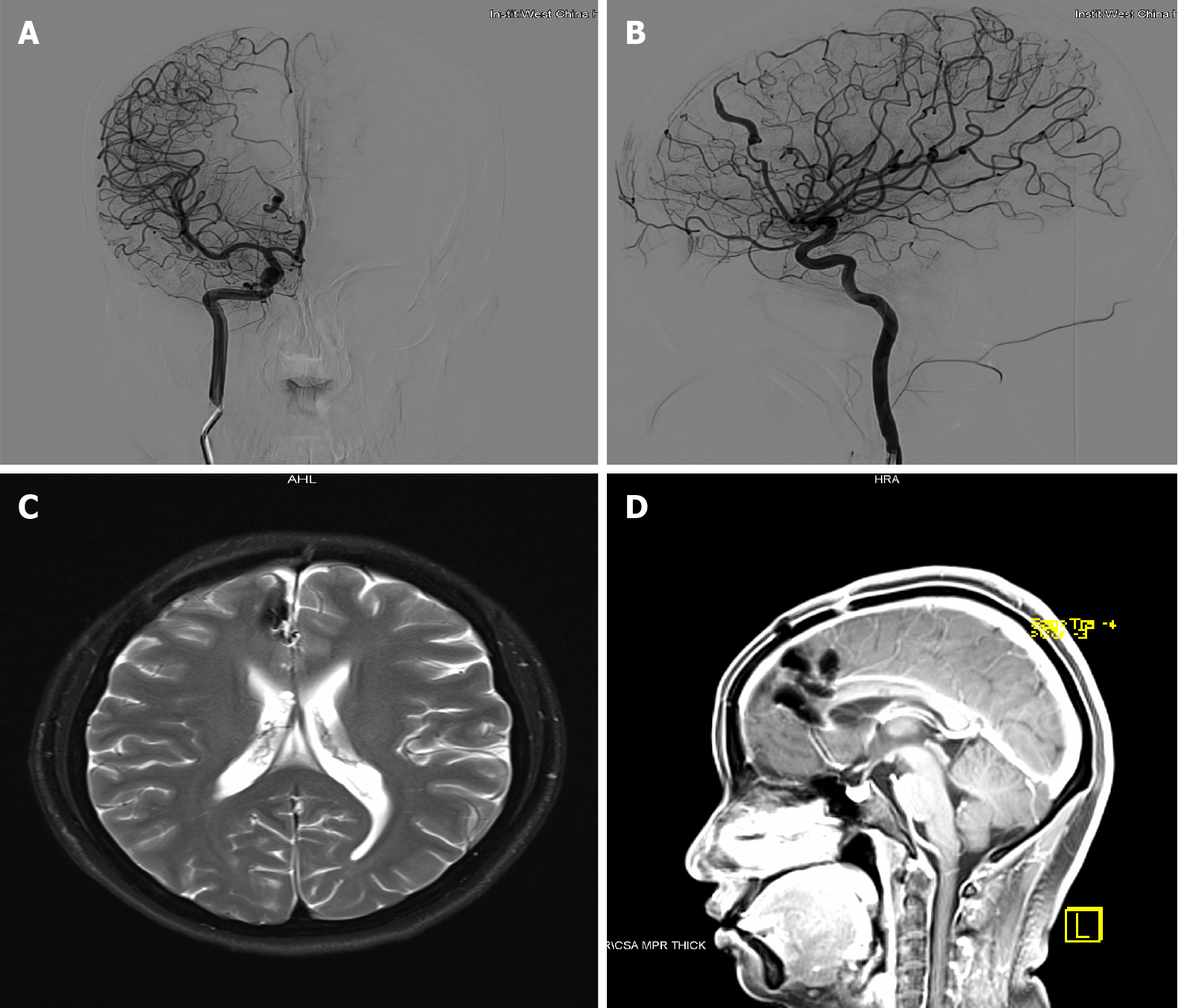
Postoperative CT angiograph image confirmed the complete clipping of the artery malformation (Figure 4). There were no tortuous segments on DSA (Figure 5A and B), and no ischemic lesions on MRI (Figure 5C and D). The patient was treated with neurotrophic drugs postoperatively.
The patient was discharged without neurological deficits or other complications ten days after the surgery. The Modified Rankin Scale was 0. There was no psychological disorder for 1-year follow-up.
The incidence of pure arterial malformations is low. Pure arterial malformations have a benign natural history, and it was usually managed conservatively. Due to the rare incidence, the optimal treatment for pure arterial malformation is controversial[1]. In this case, the arterial malformation with many branches runs into the central cortex, which may affect the motor and linguistic function. Furthermore, based on the ima
Awake craniotomy is a reliable method of ensuring neural integrity during the excision of lesions located within or near eloquent areas. Furthermore, continuous clinical neurological monitoring can detect early deficits in the motor, sensory or language domains. Our team had rich experiences with awake craniotomy for brain tumors, but this was the first awake surgery for artery malformation clipping in our institute.
Cerebral artery malformation surgery may cause motor deficits due to brain blood flow insufficiency after clipping the artery malformation. Continuous infusion of remifentanil, dexmedetomidine and propofol has little impact on evoked potentials[2,3]. Irie et al[4] reported that the limitation of this monitoring technique is the false-negative MEP; four patients had postoperative hemiparesis without intraoperative MEP changes. Once ischemia occurs, MEP maybe not sensitive to the evaluation of movement[5]. It is reported that there is a potential advantage of awake artery mal
Anesthetic methods in awake craniotomy include monitoring anesthesia care (MAC) and asleep-awake-asleep (SAS)[7]. Both techniques can be performed success
The fixed head by headrest may be a potential risk of difficult airway, whether in the supine position or lateral position. It’s a severe challenge for the anesthesiologist to reinsert LAM or endotracheal tube intraoperatively. Maintaining airway patency is the key to improving the feasibility of artificial ventilation, especially in an emergency. Our case was accomplished with spontaneous breath by adjusting to the target concentration precisely.
The MAC technique may cause the risk of dose-dependent respiratory depression. BIS is recorded to monitor the depth of sedation. As we learned, propofol has dose-dependent depression in cardiovascular and respiratory system, while dexmede
As Abdulrauf et al[6] responded, intraoperative aneurysm rupture can be observed under both awake craniotomy and general anesthesia[14]. Intraoperative hemody
Intraoperative seizure is the most common cause for awake craniotomy failure, which is trigged by electrical cortical stimulation. The risk factors for seizure include stimulus length, current intensity, and the number of stimulations over the same cortical area. Intraoperative seizure can be easily control by ice saline and propofol, without permanent severe consequence[17]. This patient didn’t develop intraoperative seizure. Hence, awake craniotomy is safe for patient with arterial malformation.
The literature on awake artery malformation clipping is rare, especially for anesthesia management. In our case, anesthetic management with nasopharyngeal airway is safe and effective for awake artery malformation clipping with intraoperative monitoring. On the basis of mastering the basic skills, anesthesiologists use simple equipment and anesthetic drugs to perform the awake artery malformation clipping successfully. Although this scheme has some disadvantages, it can provide a reference for ane
Manuscript source: Unsolicited manuscript
Specialty type: Medicine, research and experimental
Country/Territory of origin: China
Peer-review report’s scientific quality classification
Grade A (Excellent): 0
Grade B (Very good): B
Grade C (Good): C
Grade D (Fair): 0
Grade E (Poor): 0
P-Reviewer: Chrastina J S-Editor: Fan JR L-Editor: A P-Editor: Yuan YY
| 1. | Liu TY, Xu N, Wan Z, Zhang ZM, Xu JJ, Meng H, Wang HL. Diagnosis and treatment of pure arterial malformation: Three case reports and literature review. Medicine (Baltimore). 2020;99:e20229. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 3] [Cited by in RCA: 10] [Article Influence: 2.0] [Reference Citation Analysis (0)] |
| 2. | Nunes RR, Bersot CDA, Garritano JG. Intraoperative neurophysiological monitoring in neuroanesthesia. Curr Opin Anaesthesiol. 2018;31:532-538. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 51] [Cited by in RCA: 34] [Article Influence: 4.9] [Reference Citation Analysis (0)] |
| 3. | Tobias JD, Goble TJ, Bates G, Anderson JT, Hoernschemeyer DG. Effects of dexmedetomidine on intraoperative motor and somatosensory evoked potential monitoring during spinal surgery in adolescents. Paediatr Anaesth. 2008;18:1082-1088. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 72] [Cited by in RCA: 52] [Article Influence: 3.1] [Reference Citation Analysis (0)] |
| 4. | Irie T, Yoshitani K, Ohnishi Y, Shinzawa M, Miura N, Kusaka Y, Miyazaki S, Miyamoto S. The efficacy of motor-evoked potentials on cerebral aneurysm surgery and new-onset postoperative motor deficits. J Neurosurg Anesthesiol. 2010;22:247-251. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 53] [Cited by in RCA: 52] [Article Influence: 3.5] [Reference Citation Analysis (0)] |
| 5. | Suzuki K, Mikami T, Sugino T, Wanibuchi M, Miyamoto S, Hashimoto N, Mikuni N. Discrepancy between voluntary movement and motor-evoked potentials in evaluation of motor function during clipping of anterior circulation aneurysms. World Neurosurg. 2014;82:e739-e745. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 23] [Cited by in RCA: 29] [Article Influence: 2.4] [Reference Citation Analysis (0)] |
| 6. | Abdulrauf SI, Vuong P, Patel R, Sampath R, Ashour AM, Germany LM, Lebovitz J, Brunson C, Nijjar Y, Dryden JK, Khan MQ, Stefan MG, Wiley E, Cleary RT, Reis C, Walsh J, Buchanan P. "Awake" clipping of cerebral aneurysms: report of initial series. J Neurosurg. 2017;127:311-318. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 23] [Cited by in RCA: 25] [Article Influence: 2.8] [Reference Citation Analysis (0)] |
| 7. | Kulikov A, Lubnin A. Anesthesia for awake craniotomy. Curr Opin Anaesthesiol. 2018;31:506-510. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 27] [Cited by in RCA: 28] [Article Influence: 4.7] [Reference Citation Analysis (0)] |
| 8. | Pouratian N, Cannestra AF, Bookheimer SY, Martin NA, Toga AW. Variability of intraoperative electrocortical stimulation mapping parameters across and within individuals. J Neurosurg. 2004;101:458-466. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 112] [Cited by in RCA: 100] [Article Influence: 4.8] [Reference Citation Analysis (0)] |
| 9. | Eseonu CI, ReFaey K, Garcia O, John A, Quiñones-Hinojosa A, Tripathi P. Awake Craniotomy Anesthesia: A Comparison of the Monitored Anesthesia Care and Asleep-Awake-Asleep Techniques. World Neurosurg. 2017;104:679-686. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 41] [Cited by in RCA: 47] [Article Influence: 5.9] [Reference Citation Analysis (0)] |
| 10. | Goodman NW, Black AM, Carter JA. Some ventilatory effects of propofol as sole anaesthetic agent. Br J Anaesth. 1987;59:1497-1503. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 118] [Cited by in RCA: 99] [Article Influence: 2.6] [Reference Citation Analysis (0)] |
| 11. | Barker N, Lim J, Amari E, Malherbe S, Ansermino JM. Relationship between age and spontaneous ventilation during intravenous anesthesia in children. Paediatr Anaesth. 2007;17:948-955. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 50] [Cited by in RCA: 34] [Article Influence: 1.9] [Reference Citation Analysis (0)] |
| 12. | Ansermino JM, Magruder W, Dosani M. Spontaneous respiration during intravenous anesthesia in children. Curr Opin Anaesthesiol. 2009;22:383-387. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 19] [Cited by in RCA: 16] [Article Influence: 1.0] [Reference Citation Analysis (0)] |
| 13. | McClellan KJ, Faulds D. Ropivacaine: an update of its use in regional anaesthesia. Drugs. 2000;60:1065-1093. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 153] [Cited by in RCA: 173] [Article Influence: 6.9] [Reference Citation Analysis (0)] |
| 14. | Agarwal N. Letter to the Editor. Intraoperative aneurysm rupture during awake clipping of cerebral aneurysms. J Neurosurg. 2018;128:1906. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 1] [Cited by in RCA: 1] [Article Influence: 0.1] [Reference Citation Analysis (0)] |
| 15. | Rajan S, Cata JP, Nada E, Weil R, Pal R, Avitsian R. Asleep-awake-asleep craniotomy: a comparison with general anesthesia for resection of supratentorial tumors. J Clin Neurosci. 2013;20:1068-1073. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 28] [Cited by in RCA: 26] [Article Influence: 2.2] [Reference Citation Analysis (0)] |
| 16. | Chowdhury T, Gray K, Sharma M, Mau C, McNutt S, Ryan C, Farou N, Bergquist P, Caldwell C, Uribe AA, Todeschini AB, Bergese SD, Bucher O, Musto G, Azazi EA, Zadeh G, Tsang DS, Mansouri SA, Kakumanu S, Venkatraghavan L. Brain Cancer Progression: A Retrospective Multicenter Comparison of Awake Craniotomy Versus General Anesthesia in High-grade Glioma Resection. J Neurosurg Anesthesiol. 2021;. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 1] [Cited by in RCA: 8] [Article Influence: 2.0] [Reference Citation Analysis (0)] |
| 17. | Boetto J, Bertram L, Moulinié G, Herbet G, Moritz-Gasser S, Duffau H. Low Rate of Intraoperative Seizures During Awake Craniotomy in a Prospective Cohort with 374 Supratentorial Brain Lesions: Electrocorticography Is Not Mandatory. World Neurosurg. 2015;84:1838-1844. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 87] [Cited by in RCA: 86] [Article Influence: 8.6] [Reference Citation Analysis (0)] |