Published online Jul 6, 2021. doi: 10.12998/wjcc.v9.i19.4990
Peer-review started: January 27, 2021
First decision: February 27, 2021
Revised: March 14, 2021
Accepted: May 15, 2021
Article in press: May 15, 2021
Published online: July 6, 2021
Processing time: 148 Days and 14.9 Hours
Coronavirus disease 2019 (COVID-19), caused by the infection of a novel coronavirus [severe acute respiratory syndrome coronavirus 2 (SARS-CoV-2)], has become a pandemic. The infection has resulted in about one hundred million COVID-19 cases and millions of deaths. Although SARS-CoV-2 mainly spreads through the air and impairs the function of the respiratory system, it also attacks the gastrointestinal epithelial cells through the same receptor, angiotensin converting enzyme 2 receptor, which results in gastroenteric symptoms and potential fecal-oral transmission. Besides the infection of SARS-CoV-2, the treatments of COVID-19 also contribute to the gastroenteric manifestations due to the adverse drug reactions of anti-COVID-19 drugs. In this review, we update the clinical features, basic studies, and clinical practices of COVID-19-associated gastroenteric manifestations.
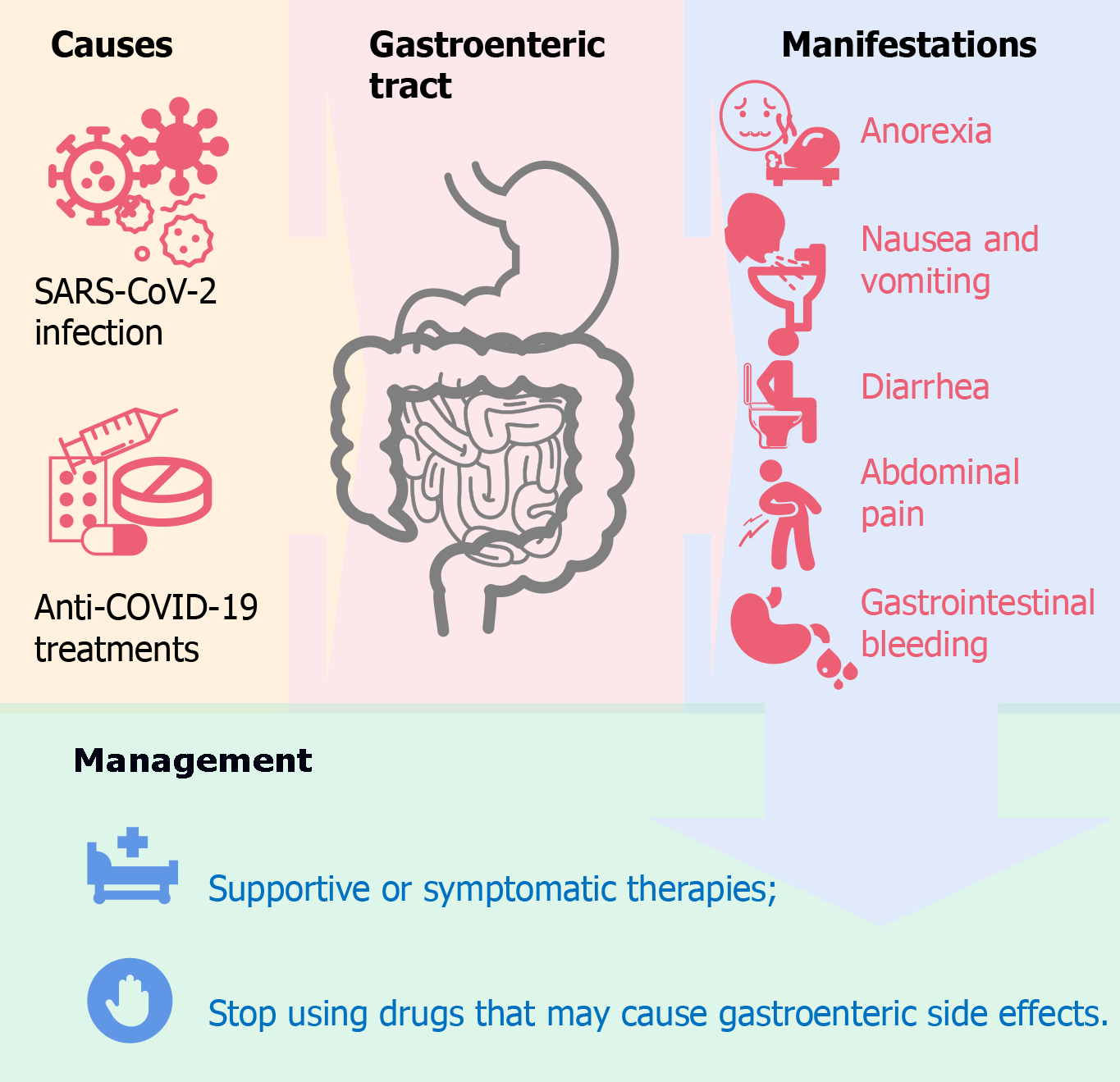
Core Tip: Gastroenteric manifestations, including anorexia, nausea, vomiting, diarrhea, abdominal pain, and gastrointestinal bleeding, can worsen the situation of patients with coronavirus disease 2019 (COVID-19). More efforts should be spared to understand its pathology and mechanism. Besides the infection of severe acute respiratory syndrome coronavirus 2 virus, the treatments of COVID-19 also contribute to the gastroenteric manifestations due to the adverse drug reactions of anti-COVID-19 drugs. Timely supportive or symptomatic treatment and withdrawal of adverse drug reaction-inducing drugs can help to manage COVID-19-associated gastroenteric manifestations.
- Citation: Chen ZR, Liu J, Liao ZG, Zhou J, Peng HW, Gong F, Hu JF, Zhou Y. COVID-19 and gastroenteric manifestations . World J Clin Cases 2021; 9(19): 4990-4997
- URL: https://www.wjgnet.com/2307-8960/full/v9/i19/4990.htm
- DOI: https://dx.doi.org/10.12998/wjcc.v9.i19.4990
The Coronaviridae family can infect both humans and animals, of which seven types of coronaviruses (CoVs) can infect humans[1]. Severe acute respiratory syndrome coronavirus (SARS-CoV) and Middle East respiratory syndrome-CoV emerged in the 21st century and were endemic locally[2,3]. Since late 2019 through January 2021, SARS-CoV-2 has accounted for over one hundred million confirmed cases and over two million deaths in all countries and all seven continents (https://www.arcgis.com/apps/opsdashboard/index.html#/bda7594740fd40299423467b48e9ecf6). SARS-CoV-2 is a beta coronavirus that originated in bats[4]. It is still poorly understood how those zoonotic coronaviruses were transmitted from bats to humans despite some clues[5,6].
The RNA of SARS-CoV-2 can be detected in the blood, kidneys, liver, heart, brain, and other organs of infected patients[7]. The infection of SARS-CoV-2 mainly occurs in the respiratory tract. The clinical manifestations of the SARS-CoV-2 infection range from asymptomatic infection, mild influenza-like symptoms (mainly fever, cough, and fatigue), severe pneumonia, multiorgan failure, and even death[8]. Some of the COVID-19 patients who suffer from the disease induced by the infection of SARS-CoV-2 have gastroenteric manifestations, mainly manifested as anorexia, nausea, vomiting, diarrhea, and abdominal pain[9,10]. The mechanism of COVID-19-associated gastroenteric manifestations has not been fully demonstrated. Also, the anti-COVID-19 treatments can induce gastroenteric side effects that may confuse with or superimpose with the COVID-19-associated gastroenteric manifestations. This review introduces the clinical features, mechanism, and management of COVID-19-associated gastroenteric manifestations.
Respiratory symptoms, including fever, fatigue, and dry cough, are the most common manifestations among COVID-19 patients. In addition, some patients suffered gastrointestinal tract disorders, including anorexia, nausea, vomiting, diarrhea, and abdominal pain[8]. The incidence of gastroenteric manifestations among COVID-19 patients varied between initial reports and subsequent studies[11]. Guan et al[12] and Wang et al[13] reported that gastroenteric manifestations can be found among 2% to 10% of COVID-19 patients. However, some meta-analyses found that the prevalence can be 9.8%, 33.4%, and 50.5%[14-16]. This divergence can be explained partly by the difference of included patients, which consist of outpatients and hospitalized patients. The RNA of SARS-CoV-2 can be detected in the stools of infected people, and the fecal viral shedding duration and positive viral rectal swabs can last for a long time after the respiratory specimens or nasopharyngeal swabs turned negative in both adults and children[17]. Those findings proposed the potential fecal-oral transmission. But the viral RNA detected in stools does not fully equate to live viruses, and further studies need to be done.
Typical gastrointestinal symptoms of COVID-19 patients consist of anorexia, nausea and vomiting, diarrhea, and abdominal pain[18]. In addition to the above-mentioned typical symptoms, some specific symptoms can be observed, such as gastrointestinal bleeding[18,19]. Noteworthily, the above-mentioned gastroenteric manifestations were observed before fever in 14 (10.1%) patients in a study[13]. Most typical gastro
Anorexia, which manifests as loss of appetite, is the most common gastroenteric manifestations, and the prevalence ranged from 1.0% to 79%[22,23]. The mechanism of COVID-19 associated anorexia is still poorly understood. Gustatory dysfunction of COVID-19 patients may explain it partly[23]. Gustatory dysfunction was markedly associated with olfactory dysfunction, which is a specific feature of early SARS-CoV-2 infection[24].
Nausea and vomiting, as a symptom of COVID-19, may result from the SARS-CoV-2 impact on the digestive system and/or central nervous system[22]. The prevalence of nausea and vomiting ranged from 7.8% to 22.7%[22]. Some anti-COVID-19 treatments can also induce nausea and vomiting, which is independent of the viral infection. In addition, treatment-induced nausea and vomiting may be merged with disease-induced nausea and vomiting and worsen the clinical status of COVID-19 patients.
Diarrhea, a common disorder of COVID-19 patients, can be induced by the intestinal SARS-CoV-2 infection itself, the disturbance of gut microbiota, and some anti-COVID-19 treatments (such as oseltamivir and abidol hydrochloride). The prevalence has been reported as 5%-10%, 2%-50%, and 22.1% in three independent reports[14,15,25]. Twenty-two percent of COVID-19-associated diarrhea were not treatment-induced[22]. Considering the potential fecal-oral transmission of SARS-CoV-2, more attention should be paid to COVID-19-associated diarrhea as a public health issue that could lead to the community transmission of COVID-19.
Abdominal pain was less common among COVID-19 patients compared with the above-mentioned gastrointestinal symptoms[22]. The mechanism of COVID-19 associated abdominal pain has also not been well elucidated yet. The SARS-CoV-2 infection-induced visceral pain and/or the involvement of the peritoneum may contribute to COVID-19 associated abdominal pain[22].
Gastrointestinal bleeding is a rare specific manifestation of COVID-19 patients and causes hematemesis or melena. Several gastrointestinal bleeding cases of COVID-19 patients have been reported, and it is more fatal than general gastrointestinal manifestations[18,19,26]. Cavaliere et al[19] reported 6 COVID-19 patients with upper gastrointestinal bleeding and concluded that COVID-19 patients with upper gastrointestinal bleeding should be treated without endoscopy. But there are still some controversies because some disorders (pulmonary embolism, myocardial infarction, and renal failure) and the intake of substances (iron supplements, bismuth subsalicylate, and foods such as blood soup) can lead to coffee-ground hematemesis episodes or black stools similar to melena[27]. Thus, despite those patients’ responses to conservative treatment in 24 h[19], Duan et al[27] states that endoscopy is still essential to find the cause of hematemesis or melena.
Pathological research confirmed that SARS-CoV-2 mainly targets the respiratory system and causes the impairment of pulmonary function. COVID-19 has similar pathological features of SARS and Middle East respiratory syndrome[20]. In addition, autopsy and endoscopy studies demonstrated that the infection of SARS-CoV-2 also causes lesions in the gastroenteric tract. Some researchers analyzed 28 autopsy studies that include 341 COVID-19 cases, and concluded that most studies focused on pulmonary alterations, and only a few pieces of information are given about the features of other organs and systemic findings[28-30]. Elsoukkary et al[29] found that the color of the stomach mucosa turned crimson, and some bleeding points can be seen on the stomach mucosa via the gross examination of a COVID-19 death autopsy, while the color of the intestine has not changed[31]. In the intestine autopsy of an 85-year-old man with COVID-19, segmental dilatation and stenosis alternating was found. However, it is still not clear whether it is induced by the infection of SARS-CoV-2[31]. Furthermore, viral particles were found in enterocytes in the large intestine after COVID-19 death autopsy through electron microscope[32]. Those findings provide further evidence of the possibility of the fecal-oral transmission of SARS-CoV-2.
Efforts to discover the mechanism of SARS-CoV-2 infecting gastroenteric tract cells led to studies involving cell lines, animals, and organoids[33,34]. Four human intestinal epithelial cell lines are permissive to SARS-CoV-2 infection, including T84, C2BBe1, Caco-2, and CL14[8]. Human organoids and bat intestinal organoids also support the infection of SARS-CoV-2[35,36]. Organoids are self-assembled, differentiated, nontransformed, and three-dimensional microphysiologically culture systems that contain multiple cell types similar to the cells in a specific tissue[37]. Organoids can model niche-mimicking and form similar architectures of original organs[37]. There are unique advantages for the utilization of organoids in medical research, especially infectious diseases, cancer, genetic diseases, and novel drug discovery. Human intestinal organoids are the most advanced accessible tool for the simulation of SARS-CoV-2 infection of gastroenteric tract cells[37]. Multiple intestinal epithelial cell types, such as enterocytes, goblet cells, tuft cells, enteroendocrine cells, and Paneth cells, can be found in human intestinal organoids.
Studies with the above-mentioned models have demonstrated the process that SARS-CoV-2 infects gastroenteric tract cells. There are four structural proteins in SARS-CoV-2: spike, envelope, membrane, and nucleocapsid. The spike protein binds to the adhesion receptor of hosts first, then binds to the entry receptor and activates a fusion of the viral and cellular membranes. Angiotensin converting enzyme 2 (ACE2) is the entry receptor for SARS-CoV-2, and transmembrane protease serine 2 is a spike priming protease[8,20]. There is a higher expression of ACE2 in the gastroenteric tract than in the respiratory tract[38]. Generally, the higher expression of ACE2 in a tissue, the more likely it was infected by SARS-CoV-2. The human colon expressed ACE2 much lower than stomach epithelial cells and enterocytes from the small intestine[39]. Consequently, human colon organoids are affected to a lesser extent than small intestine organoids, and SARS-CoV-2 infects only enterocytes, not goblet cells, enteroendocrine cells, tuft cells, or Paneth cells[36].
The infection of SARS-CoV-2 can induce alterations in gut microbiota[40]. The alterations in gut microbiota, the increase of opportunistic pathogens, and the loss of salutary bacteria contribute to the gastroenteric manifestations[41]. Bacterial species Collinsella aerofaciens, Collinsella tanakaei, Streptococcus infantis, and Morganella morganii are enriched in the stools of COVID-19 patients with higher SARS-CoV-2 infectivity[41]. In another study, the reduction of an anti-inflammatory bacterium, Faecalibacterium prausnitzii, and the increase of Coprobacillus, Clostridium ramosum, and Clostridium hathewayi were found to be correlated with the severity of COVID-19[42]. Management to alter the gut microbiota, such as the utilization of intestinal microecological preparations, may help to relieve disease symptoms[42,43].
Gastroenteric disorders were the most frequent adverse drug reactions (ADRs) for the patients with COVID-19[44]. There is still a lack of specific treatment for COVID-19. Most anti-COVID-19 therapies are supportive and symptomatic treatments. Numerous drugs and chemical or biological entities have been reported as inhibitors of SARS-CoV-2 or the cures of COVID-19. Those drugs, including interferon, ribavirin, arbidol (umifenovir), lopinavir/ritonavir, hydroxychloroquine/chloroquine, azithromycin, remdesivir, etc. are controversial for their unproven and contradictory anti-COVID-19 effects[45]. Nevertheless, those drugs have been associated with many ADRs, of which gastrointestinal reaction are the most common[46]. Some researchers analyzed 1452 ADRs caused by antiviral agents named ribavirin, chloroquine phosphate, arbidol, and lopinavir/ritonavir and revealed that all four drugs can induce gastroenteric manifestations[47]. Gastroenteric ADRs are very common among lopinavir/ritonavir or arbidol treated patients (46.50% and 45.71%, respectively)[47,48] and less common among ribavirin or chloroquine phosphate treated patients (17.63% and 2.99%, respectively)[47]. Remdesivir can induce diarrhea, nausea, and vomiting in 3% to 9% and 3% to 5% of patients with COVID-19[49,50]. The pathology and mechanism of COVID-19 treatment-associated gastroenteric manifestations are still not revealed. Hydroxychloroquine can alter gut microbiota[51], but its correlation with gastroenteric symp
There is still no report or guideline on the treatment of COVID-19-associated gastroenteric manifestations[20]. Supportive and symptomatic therapies are suggested for the management of COVID-19-associated gastroenteric manifestations. Supportive care includes the intake of nutrients, calories, fluid, and salt to maintain balance. For patients with mild dehydration, oral rehydration is enough. But for patients with severe dehydration, intravenous rehydration should be considered. For symptomatic treatments, antiemetic drugs, such as metoclopramide and ondansetron, are suggested for patients with nausea and vomiting. Antidiarrheal medicines, like loperamide, are suggested for patients with diarrhea[20]. Intestinal microecological agents may help to mitigate the symptoms induced by the disorder of intestinal bacteria[52-54].
Gastroenteric manifestations, including anorexia, nausea, vomiting, diarrhea, abdominal pain, and gastrointestinal bleeding, can be found in most COVID-19 patients, which appears to be associated with worse clinical outcomes. More patients with gastrointestinal symptoms were hospitalized compared with patients without gastrointestinal symptoms[55]. Because most gastroenteric symptoms are milder than respiratory symptoms, physicians may underestimate the risk of it. In addition, anti-COVID-19 treatments can induce gastroenteric disorders independently. The merging of SARS-CoV-2 infection-induced manifestations and COVID-19 treatment-induced manifestations may worsen the situation of patients with COVID-19. Timely supportive or symptomatic treatment and withdrawal of ADR-inducing drugs should be considered (Figure 1).
The authors thank all the doctors, nurses, pharmacists, and other peers who sacrificed their lives for the discovery, treatment, control, and prevention of COVID-19. Special thanks to Mr. Guo-Nan Chen and Miss She-Lian Li.
Manuscript source: Invited Manuscript
Specialty type: Medicine, Research and Experimental
Country/Territory of origin: China
Peer-review report’s scientific quality classification
Grade A (Excellent): 0
Grade B (Very good): 0
Grade C (Good): C
Grade D (Fair): 0
Grade E (Poor): 0
P-Reviewer: Kanzaki N S-Editor: Ma YJ L-Editor: Filipodia P-Editor: Wang LL
| 1. | Galanopoulos M, Gkeros F, Doukatas A, Karianakis G, Pontas C, Tsoukalas N, Viazis N, Liatsos C, Mantzaris GJ. COVID-19 pandemic: Pathophysiology and manifestations from the gastrointestinal tract. World J Gastroenterol. 2020;26:4579-4588. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in CrossRef: 98] [Cited by in RCA: 103] [Article Influence: 20.6] [Reference Citation Analysis (5)] |
| 2. | Peng H, Chen Z, Wang Y, Ren S, Xu T, Lai X, Wen J, Zhao M, Zeng C, Du L, Zhang Y, Cao L, Hu J, Wei X, Hong T. Systematic Review and Pharmacological Considerations for Chloroquine and Its Analogs in the Treatment for COVID-19. Front Pharmacol. 2020;11:554172. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 5] [Cited by in RCA: 7] [Article Influence: 1.4] [Reference Citation Analysis (0)] |
| 3. | Dhama K, Khan S, Tiwari R, Sircar S, Bhat S, Malik YS, Singh KP, Chaicumpa W, Bonilla-Aldana DK, Rodriguez-Morales AJ. Coronavirus Disease 2019-COVID-19. Clin Microbiol Rev. 2020;33. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 689] [Cited by in RCA: 604] [Article Influence: 120.8] [Reference Citation Analysis (0)] |
| 4. | Zhou P, Yang XL, Wang XG, Hu B, Zhang L, Zhang W, Si HR, Zhu Y, Li B, Huang CL, Chen HD, Chen J, Luo Y, Guo H, Jiang RD, Liu MQ, Chen Y, Shen XR, Wang X, Zheng XS, Zhao K, Chen QJ, Deng F, Liu LL, Yan B, Zhan FX, Wang YY, Xiao GF, Shi ZL. A pneumonia outbreak associated with a new coronavirus of probable bat origin. Nature. 2020;579:270-273. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 15248] [Cited by in RCA: 14100] [Article Influence: 2820.0] [Reference Citation Analysis (1)] |
| 5. | Lam TT, Jia N, Zhang YW, Shum MH, Jiang JF, Zhu HC, Tong YG, Shi YX, Ni XB, Liao YS, Li WJ, Jiang BG, Wei W, Yuan TT, Zheng K, Cui XM, Li J, Pei GQ, Qiang X, Cheung WY, Li LF, Sun FF, Qin S, Huang JC, Leung GM, Holmes EC, Hu YL, Guan Y, Cao WC. Identifying SARS-CoV-2-related coronaviruses in Malayan pangolins. Nature. 2020;583:282-285. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 1019] [Cited by in RCA: 1199] [Article Influence: 239.8] [Reference Citation Analysis (0)] |
| 6. | Xiao K, Zhai J, Feng Y, Zhou N, Zhang X, Zou JJ, Li N, Guo Y, Li X, Shen X, Zhang Z, Shu F, Huang W, Li Y, Chen RA, Wu YJ, Peng SM, Huang M, Xie WJ, Cai QH, Hou FH, Chen W, Xiao L, Shen Y. Isolation of SARS-CoV-2-related coronavirus from Malayan pangolins. Nature. 2020;583:286-289. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 414] [Cited by in RCA: 493] [Article Influence: 98.6] [Reference Citation Analysis (0)] |
| 7. | Puelles VG, Lütgehetmann M, Lindenmeyer MT, Sperhake JP, Wong MN, Allweiss L, Chilla S, Heinemann A, Wanner N, Liu S, Braun F, Lu S, Pfefferle S, Schröder AS, Edler C, Gross O, Glatzel M, Wichmann D, Wiech T, Kluge S, Pueschel K, Aepfelbacher M, Huber TB. Multiorgan and Renal Tropism of SARS-CoV-2. N Engl J Med. 2020;383:590-592. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 1254] [Cited by in RCA: 1421] [Article Influence: 284.2] [Reference Citation Analysis (0)] |
| 8. | Synowiec A, Szczepański A, Barreto-Duran E, Lie LK, Pyrc K. Severe Acute Respiratory Syndrome Coronavirus 2 (SARS-CoV-2): a Systemic Infection. Clin Microbiol Rev. 2021;34. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 124] [Cited by in RCA: 149] [Article Influence: 37.3] [Reference Citation Analysis (0)] |
| 9. | Cha MH, Regueiro M, Sandhu DS. Gastrointestinal and hepatic manifestations of COVID-19: A comprehensive review. World J Gastroenterol. 2020;26:2323-2332. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in CrossRef: 124] [Cited by in RCA: 107] [Article Influence: 21.4] [Reference Citation Analysis (2)] |
| 10. | Ai JW, Zi H, Wang Y, Huang Q, Wang N, Li LY, Pei B, Ji J, Zeng XT. Clinical Characteristics of COVID-19 Patients With Gastrointestinal Symptoms: An Analysis of Seven Patients in China. Front Med (Lausanne). 2020;7:308. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 21] [Cited by in RCA: 17] [Article Influence: 3.4] [Reference Citation Analysis (0)] |
| 11. | El Ouali S, Achkar JP, Lashner B, Regueiro M. Gastrointestinal manifestations of COVID-19. Cleve Clin J Med. 2021;. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 18] [Cited by in RCA: 16] [Article Influence: 4.0] [Reference Citation Analysis (0)] |
| 12. | Guan WJ, Ni ZY, Hu Y, Liang WH, Ou CQ, He JX, Liu L, Shan H, Lei CL, Hui DSC, Du B, Li LJ, Zeng G, Yuen KY, Chen RC, Tang CL, Wang T, Chen PY, Xiang J, Li SY, Wang JL, Liang ZJ, Peng YX, Wei L, Liu Y, Hu YH, Peng P, Wang JM, Liu JY, Chen Z, Li G, Zheng ZJ, Qiu SQ, Luo J, Ye CJ, Zhu SY, Zhong NS; China Medical Treatment Expert Group for Covid-19. Clinical Characteristics of Coronavirus Disease 2019 in China. N Engl J Med. 2020;382:1708-1720. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 19202] [Cited by in RCA: 18852] [Article Influence: 3770.4] [Reference Citation Analysis (7)] |
| 13. | Wang D, Hu B, Hu C, Zhu F, Liu X, Zhang J, Wang B, Xiang H, Cheng Z, Xiong Y, Zhao Y, Li Y, Wang X, Peng Z. Clinical Characteristics of 138 Hospitalized Patients With 2019 Novel Coronavirus-Infected Pneumonia in Wuhan, China. JAMA. 2020;323:1061-1069. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 14113] [Cited by in RCA: 14752] [Article Influence: 2950.4] [Reference Citation Analysis (0)] |
| 14. | Cheung KS, Hung IFN, Chan PPY, Lung KC, Tso E, Liu R, Ng YY, Chu MY, Chung TWH, Tam AR, Yip CCY, Leung KH, Fung AY, Zhang RR, Lin Y, Cheng HM, Zhang AJX, To KKW, Chan KH, Yuen KY, Leung WK. Gastrointestinal Manifestations of SARS-CoV-2 Infection and Virus Load in Fecal Samples From a Hong Kong Cohort: Systematic Review and Meta-analysis. Gastroenterology. 2020;159:81-95. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 1113] [Cited by in RCA: 1130] [Article Influence: 226.0] [Reference Citation Analysis (1)] |
| 15. | Sultan S, Altayar O, Siddique SM, Davitkov P, Feuerstein JD, Lim JK, Falck-Ytter Y, El-Serag HB; AGA Institute. AGA Institute Rapid Review of the Gastrointestinal and Liver Manifestations of COVID-19, Meta-Analysis of International Data, and Recommendations for the Consultative Management of Patients with COVID-19. Gastroenterology 2020; 159: 320-334. e27. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 309] [Cited by in RCA: 292] [Article Influence: 58.4] [Reference Citation Analysis (1)] |
| 16. | Rokkas T. Gastrointestinal involvement in COVID-19: a systematic review and meta-analysis. Ann Gastroenterol. 2020;33:355-365. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 37] [Cited by in RCA: 53] [Article Influence: 10.6] [Reference Citation Analysis (0)] |
| 17. | Wu Y, Guo C, Tang L, Hong Z, Zhou J, Dong X, Yin H, Xiao Q, Tang Y, Qu X, Kuang L, Fang X, Mishra N, Lu J, Shan H, Jiang G, Huang X. Prolonged presence of SARS-CoV-2 viral RNA in faecal samples. Lancet Gastroenterol Hepatol. 2020;5:434-435. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 1050] [Cited by in RCA: 1150] [Article Influence: 230.0] [Reference Citation Analysis (0)] |
| 18. | Li X, Huang S, Lu J, Lai R, Zhang Z, Lin X, Zheng X, Shan H. Upper Gastrointestinal Bleeding Caused by SARS-CoV-2 Infection. Am J Gastroenterol. 2020;115:1541-1542. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 18] [Cited by in RCA: 18] [Article Influence: 3.6] [Reference Citation Analysis (0)] |
| 19. | Cavaliere K, Levine C, Wander P, Sejpal DV, Trindade AJ. Management of upper GI bleeding in patients with COVID-19 pneumonia. Gastrointest Endosc. 2020;92:454-455. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 65] [Cited by in RCA: 58] [Article Influence: 11.6] [Reference Citation Analysis (0)] |
| 20. | Su S, Shen J, Zhu L, Qiu Y, He JS, Tan JY, Iacucci M, Ng SC, Ghosh S, Mao R, Liang J. Involvement of digestive system in COVID-19: manifestations, pathology, management and challenges. Therap Adv Gastroenterol. 2020;13:1756284820934626. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 43] [Cited by in RCA: 41] [Article Influence: 8.2] [Reference Citation Analysis (0)] |
| 21. | Saito H, Ozaki A, Mizuno Y, Todo K. Difficulty in diagnosing mild cases of COVID-19 without respiratory symptoms during the Novel Coronavirus Pandemic: Careful monitoring needed for patients with persistent upper gastrointestinal symptoms. Clin Case Rep. 2020;8:2787-2790. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 3] [Cited by in RCA: 1] [Article Influence: 0.2] [Reference Citation Analysis (0)] |
| 22. | Perisetti A, Goyal H, Gajendran M, Boregowda U, Mann R, Sharma N. Prevalence, Mechanisms, and Implications of Gastrointestinal Symptoms in COVID-19. Front Med (Lausanne). 2020;7:588711. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 36] [Cited by in RCA: 36] [Article Influence: 7.2] [Reference Citation Analysis (0)] |
| 23. | Yang L, Tu L. Implications of gastrointestinal manifestations of COVID-19. Lancet Gastroenterol Hepatol. 2020;5:629-630. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 63] [Cited by in RCA: 59] [Article Influence: 11.8] [Reference Citation Analysis (0)] |
| 24. | Lechien JR, Chiesa-Estomba CM, De Siati DR, Horoi M, Le Bon SD, Rodriguez A, Dequanter D, Blecic S, El Afia F, Distinguin L, Chekkoury-Idrissi Y, Hans S, Delgado IL, Calvo-Henriquez C, Lavigne P, Falanga C, Barillari MR, Cammaroto G, Khalife M, Leich P, Souchay C, Rossi C, Journe F, Hsieh J, Edjlali M, Carlier R, Ris L, Lovato A, De Filippis C, Coppee F, Fakhry N, Ayad T, Saussez S. Olfactory and gustatory dysfunctions as a clinical presentation of mild-to-moderate forms of the coronavirus disease (COVID-19): a multicenter European study. Eur Arch Otorhinolaryngol. 2020;277:2251-2261. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 1729] [Cited by in RCA: 1734] [Article Influence: 346.8] [Reference Citation Analysis (0)] |
| 25. | Hajifathalian K, Krisko T, Mehta A, Kumar S, Schwartz R, Fortune B, Sharaiha RZ; WCM-GI research group*. Gastrointestinal and Hepatic Manifestations of 2019 Novel Coronavirus Disease in a Large Cohort of Infected Patients From New York: Clinical Implications. Gastroenterology 2020; 159: 1137-1140. e2. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 99] [Cited by in RCA: 107] [Article Influence: 21.4] [Reference Citation Analysis (0)] |
| 26. | Barrett LF, Lo KB, Stanek SR, Walter JW. Self-limited gastrointestinal bleeding in COVID-19. Clin Res Hepatol Gastroenterol. 2020;44:e77-e80. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 11] [Cited by in RCA: 17] [Article Influence: 3.4] [Reference Citation Analysis (0)] |
| 27. | Duan Z, Liu K, Zhou S. The dilemma in the management of suspected upper GI bleeding in patients with COVID-19 pneumonia. Gastrointest Endosc. 2020;92:1273-1274. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 3] [Cited by in RCA: 2] [Article Influence: 0.4] [Reference Citation Analysis (0)] |
| 28. | Xiao F, Tang M, Zheng X, Liu Y, Li X, Shan H. Evidence for Gastrointestinal Infection of SARS-CoV-2. Gastroenterology 2020; 158: 1831-1833. e3. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 1985] [Cited by in RCA: 1993] [Article Influence: 398.6] [Reference Citation Analysis (1)] |
| 29. | Elsoukkary SS, Mostyka M, Dillard A, Berman DR, Ma LX, Chadburn A, Yantiss RK, Jessurun J, Seshan SV, Borczuk AC, Salvatore SP. Autopsy Findings in 32 Patients with COVID-19: A Single-Institution Experience. Pathobiology. 2021;88:56-68. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 66] [Cited by in RCA: 97] [Article Influence: 19.4] [Reference Citation Analysis (0)] |
| 30. | Maiese A, Manetti AC, La Russa R, Di Paolo M, Turillazzi E, Frati P, Fineschi V. Autopsy findings in COVID-19-related deaths: a literature review. Forensic Sci Med Pathol. 2020;. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 146] [Cited by in RCA: 141] [Article Influence: 35.3] [Reference Citation Analysis (0)] |
| 31. | Liu Q, Wang RS, Qu GQ, Wang YY, Liu P, Zhu YZ, Fei G, Ren L, Zhou YW, Liu L. Gross examination report of a COVID-19 death autopsy. Fa Yi Xue Za Zhi. 2020;36:21-23. [RCA] [PubMed] [DOI] [Full Text] [Cited by in RCA: 129] [Reference Citation Analysis (0)] |
| 32. | Bradley BT, Maioli H, Johnston R, Chaudhry I, Fink SL, Xu H, Najafian B, Deutsch G, Lacy JM, Williams T, Yarid N, Marshall DA. Histopathology and ultrastructural findings of fatal COVID-19 infections in Washington State: a case series. Lancet. 2020;396:320-332. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 549] [Cited by in RCA: 622] [Article Influence: 124.4] [Reference Citation Analysis (0)] |
| 33. | Dickson I. Organoids demonstrate gut infection by SARS-CoV-2. Nat Rev Gastroenterol Hepatol. 2020;17:383. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 14] [Cited by in RCA: 17] [Article Influence: 3.4] [Reference Citation Analysis (0)] |
| 34. | Jurado-Gomez A, Giraldez MD. Novel Coronavirus Disease-2019 and the Gastrointestinal Tract: Lessons Learned from Human Organoids. Gastroenterology. 2020;159:2245-2247. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 1] [Cited by in RCA: 1] [Article Influence: 0.2] [Reference Citation Analysis (0)] |
| 35. | Zhou J, Li C, Liu X, Chiu MC, Zhao X, Wang D, Wei Y, Lee A, Zhang AJ, Chu H, Cai JP, Yip CC, Chan IH, Wong KK, Tsang OT, Chan KH, Chan JF, To KK, Chen H, Yuen KY. Infection of bat and human intestinal organoids by SARS-CoV-2. Nat Med. 2020;26:1077-1083. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 326] [Cited by in RCA: 429] [Article Influence: 85.8] [Reference Citation Analysis (0)] |
| 36. | Lamers MM, Beumer J, van der Vaart J, Knoops K, Puschhof J, Breugem TI, Ravelli RBG, Paul van Schayck J, Mykytyn AZ, Duimel HQ, van Donselaar E, Riesebosch S, Kuijpers HJH, Schipper D, van de Wetering WJ, de Graaf M, Koopmans M, Cuppen E, Peters PJ, Haagmans BL, Clevers H. SARS-CoV-2 productively infects human gut enterocytes. Science. 2020;369:50-54. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 1245] [Cited by in RCA: 1308] [Article Influence: 261.6] [Reference Citation Analysis (0)] |
| 37. | Li M, Izpisua Belmonte JC. Organoids - Preclinical Models of Human Disease. N Engl J Med. 2019;380:569-579. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 146] [Cited by in RCA: 217] [Article Influence: 36.2] [Reference Citation Analysis (0)] |
| 38. | Huang C, Wang Y, Li X, Ren L, Zhao J, Hu Y, Zhang L, Fan G, Xu J, Gu X, Cheng Z, Yu T, Xia J, Wei Y, Wu W, Xie X, Yin W, Li H, Liu M, Xiao Y, Gao H, Guo L, Xie J, Wang G, Jiang R, Gao Z, Jin Q, Wang J, Cao B. Clinical features of patients infected with 2019 novel coronavirus in Wuhan, China. Lancet. 2020;395:497-506. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 35178] [Cited by in RCA: 30077] [Article Influence: 6015.4] [Reference Citation Analysis (3)] |
| 39. | Hamming I, Timens W, Bulthuis ML, Lely AT, Navis G, van Goor H. Tissue distribution of ACE2 protein, the functional receptor for SARS coronavirus. A first step in understanding SARS pathogenesis. J Pathol. 2004;203:631-637. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 3643] [Cited by in RCA: 4146] [Article Influence: 197.4] [Reference Citation Analysis (0)] |
| 40. | Zuo T, Zhang F, Lui GCY, Yeoh YK, Li AYL, Zhan H, Wan Y, Chung ACK, Cheung CP, Chen N, Lai CKC, Chen Z, Tso EYK, Fung KSC, Chan V, Ling L, Joynt G, Hui DSC, Chan FKL, Chan PKS, Ng SC. Alterations in Gut Microbiota of Patients With COVID-19 During Time of Hospitalization. Gastroenterology 2020; 159: 944-955. e8. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 739] [Cited by in RCA: 1057] [Article Influence: 211.4] [Reference Citation Analysis (0)] |
| 41. | Zuo T, Liu Q, Zhang F, Lui GC, Tso EY, Yeoh YK, Chen Z, Boon SS, Chan FK, Chan PK, Ng SC. Depicting SARS-CoV-2 faecal viral activity in association with gut microbiota composition in patients with COVID-19. Gut. 2021;70:276-284. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 158] [Cited by in RCA: 262] [Article Influence: 65.5] [Reference Citation Analysis (0)] |
| 42. | Akour A. Probiotics and COVID-19: is there any link? Lett Appl Microbiol. 2020;71:229-234. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 54] [Cited by in RCA: 61] [Article Influence: 12.2] [Reference Citation Analysis (0)] |
| 43. | Mak JWY, Chan FKL, Ng SC. Probiotics and COVID-19: one size does not fit all. Lancet Gastroenterol Hepatol. 2020;5:644-645. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 115] [Cited by in RCA: 172] [Article Influence: 34.4] [Reference Citation Analysis (0)] |
| 44. | Sun J, Deng X, Chen X, Huang J, Huang S, Li Y, Feng J, Liu J, He G. Incidence of Adverse Drug Reactions in COVID-19 Patients in China: An Active Monitoring Study by Hospital Pharmacovigilance System. Clin Pharmacol Ther. 2020;108:791-797. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 63] [Cited by in RCA: 74] [Article Influence: 14.8] [Reference Citation Analysis (0)] |
| 45. | Chen ZR, Zhou Y, Liu J, Peng HW, Zhou J, Zhong HL, Liu LL, Lai MF, Wei XH, Wen JH. Pharmacotherapics Advice in Guidelines for COVID-19. Front Pharmacol. 2020;11:950. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 6] [Cited by in RCA: 5] [Article Influence: 1.0] [Reference Citation Analysis (0)] |
| 46. | Javorac D, Grahovac L, Manić L, Stojilković N, Anđelković M, Bulat Z, Đukić-Ćosić D, Curcic M, Djordjevic AB. An overview of the safety assessment of medicines currently used in the COVID-19 disease treatment. Food Chem Toxicol. 2020;144:111639. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 34] [Cited by in RCA: 36] [Article Influence: 7.2] [Reference Citation Analysis (0)] |
| 47. | Ying L, Jiong T. Literature Analysis of Antiviral Agent Adverse Drug Reactions in the Treatment of COVID-19. World Notes on Antibiotics 2020; 42: 101-105. |
| 48. | Wen CY, Xie ZW, Li YP, Deng XL, Chen XT, Cao Y, Ou X, Lin WY, Li F, Cai WP, Li LH. [Real-world efficacy and safety of lopinavir/ritonavir and arbidol in treating with COVID-19 : an observational cohort study]. Zhonghua Nei Ke Za Zhi. 2020;59:E012. [RCA] [PubMed] [DOI] [Full Text] [Cited by in RCA: 19] [Reference Citation Analysis (0)] |
| 49. | Wang Y, Zhang D, Du G, Du R, Zhao J, Jin Y, Fu S, Gao L, Cheng Z, Lu Q, Hu Y, Luo G, Wang K, Lu Y, Li H, Wang S, Ruan S, Yang C, Mei C, Wang Y, Ding D, Wu F, Tang X, Ye X, Ye Y, Liu B, Yang J, Yin W, Wang A, Fan G, Zhou F, Liu Z, Gu X, Xu J, Shang L, Zhang Y, Cao L, Guo T, Wan Y, Qin H, Jiang Y, Jaki T, Hayden FG, Horby PW, Cao B, Wang C. Remdesivir in adults with severe COVID-19: a randomised, double-blind, placebo-controlled, multicentre trial. Lancet. 2020;395:1569-1578. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 2337] [Cited by in RCA: 2483] [Article Influence: 496.6] [Reference Citation Analysis (0)] |
| 50. | Grein J, Ohmagari N, Shin D, Diaz G, Asperges E, Castagna A, Feldt T, Green G, Green ML, Lescure FX, Nicastri E, Oda R, Yo K, Quiros-Roldan E, Studemeister A, Redinski J, Ahmed S, Bernett J, Chelliah D, Chen D, Chihara S, Cohen SH, Cunningham J, D'Arminio Monforte A, Ismail S, Kato H, Lapadula G, L'Her E, Maeno T, Majumder S, Massari M, Mora-Rillo M, Mutoh Y, Nguyen D, Verweij E, Zoufaly A, Osinusi AO, DeZure A, Zhao Y, Zhong L, Chokkalingam A, Elboudwarej E, Telep L, Timbs L, Henne I, Sellers S, Cao H, Tan SK, Winterbourne L, Desai P, Mera R, Gaggar A, Myers RP, Brainard DM, Childs R, Flanigan T. Compassionate Use of Remdesivir for Patients with Severe Covid-19. N Engl J Med. 2020;382:2327-2336. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 1926] [Cited by in RCA: 1884] [Article Influence: 376.8] [Reference Citation Analysis (0)] |
| 51. | Pan ZY, Chang YX, Han N, Hou FY, Lee BJY, Zhi FC, Yang RF, Bi YJ. Short-term high-dose gavage of hydroxychloroquine changes gut microbiota but not the intestinal integrity and immunological responses in mice. Life Sci. 2021;264:118450. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 11] [Cited by in RCA: 20] [Article Influence: 4.0] [Reference Citation Analysis (0)] |
| 52. | Trottein F, Sokol H. Potential Causes and Consequences of Gastrointestinal Disorders during a SARS-CoV-2 Infection. Cell Rep. 2020;32:107915. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 87] [Cited by in RCA: 92] [Article Influence: 18.4] [Reference Citation Analysis (0)] |
| 53. | Villapol S. Gastrointestinal symptoms associated with COVID-19: impact on the gut microbiome. Transl Res. 2020;226:57-69. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 194] [Cited by in RCA: 217] [Article Influence: 43.4] [Reference Citation Analysis (0)] |
| 54. | Bozkurt HS, Quigley EM. The probiotic Bifidobacterium in the management of Coronavirus: A theoretical basis. Int J Immunopathol Pharmacol. 2020;34:2058738420961304. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 40] [Cited by in RCA: 35] [Article Influence: 7.0] [Reference Citation Analysis (0)] |
| 55. | Cholankeril G, Podboy A, Aivaliotis VI, Pham EA, Spencer SP, Kim D, Ahmed A. Association of Digestive Symptoms and Hospitalization in Patients With SARS-CoV-2 Infection. Am J Gastroenterol. 2020;115:1129-1132. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 35] [Cited by in RCA: 34] [Article Influence: 6.8] [Reference Citation Analysis (0)] |