Published online Apr 26, 2021. doi: 10.12998/wjcc.v9.i12.2763
Peer-review started: December 8, 2020
First decision: December 30, 2020
Revised: January 1, 2021
Accepted: February 12, 2021
Article in press: February 12, 2021
Published online: April 26, 2021
Processing time: 127 Days and 19.5 Hours
The proton pump inhibitors (PPIs), used to reduce gastric acid secretion, represent one of the most widely used pharmaceutical classes in the world. Their consumption as a risk factor for the evolution of severe forms of severe acute respiratory syndrome coronavirus 2 (SARS-CoV-2) infection has been investigated as well as the mortality of these patients. These risks also appear to be linked to the duration and the dosage. On the other hand, several studies have emerged with regard to the protective or therapeutic effects of these drugs. More and more evidence underlines the immunomodulatory and anti-fibrotic role of PPIs. In addition, their ability to alkalize the contents of endosomes and lysosomes serves as an obstacle to the entry of the virus into the host cells.
To identify studies on the relationship between the intake of PPIs and coronavirus disease 2019 (COVID-19) in patients affected by SARS-CoV-2 infection, with the main objective of evaluating the outcomes related to severity and mortality.
A literature review was performed in November 2020. The MEDLINE/PubMed, Cochrane Library, EMBASE and Google Scholar databases were searched for all relevant articles published in English on this topic. The search terms were identified by means of controlled vocabularies, such as the National Library of Medicine’s MESH (Medical Subject Headings) and keywords. The MESH terms and keywords used were as follows: “COVID-19”, “proton pump inhibitors”, ”PPIs”, “SARS-CoV-2”, “outcomes”, “severity” and “mortality”. The inclusion criteria regarding the studies considered in our analysis were: meta-analysis, case-control, hospital-based case-control, population-based case-control, retrospective studies, online survey, as well as cohort-studies, while articles not published as full reports, such as conference abstracts, case reports and editorials were excluded. We tried to summarize and pool all the data if available.
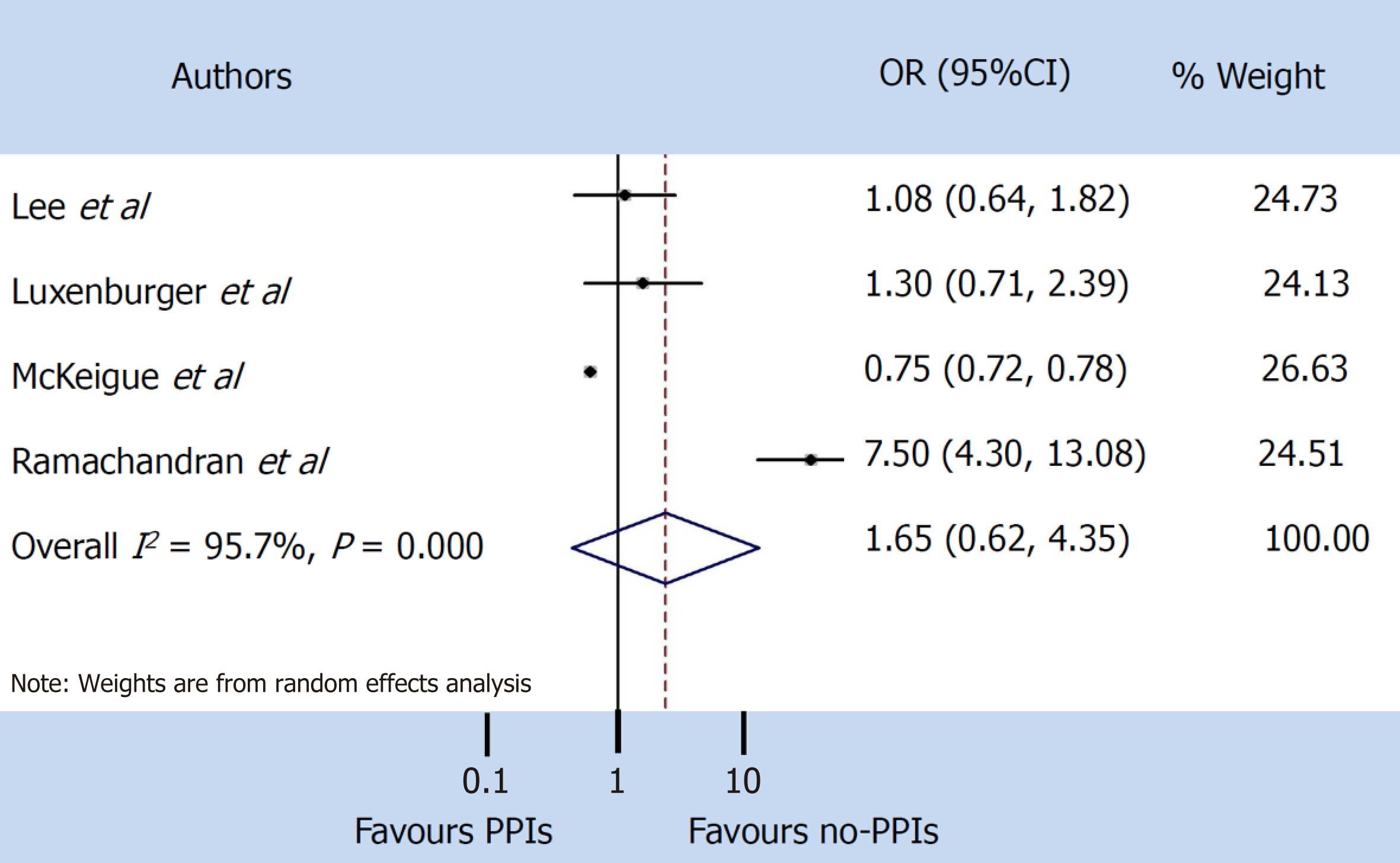
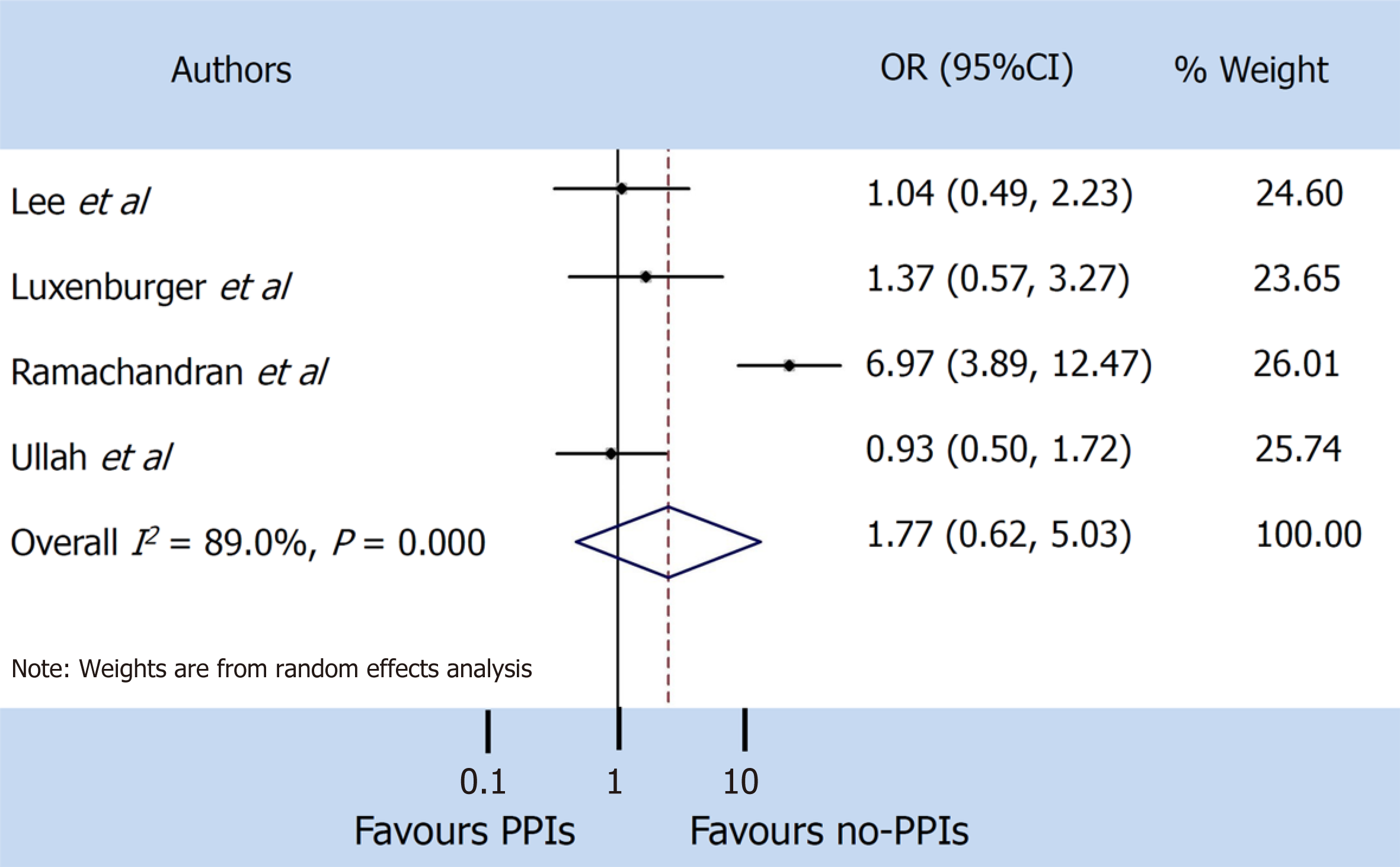
A total of 9 studies were found that described the use of PPIs, of which only 5 clearly reported the severity and mortality data in SARS-CoV-2 patients. Our pooled incidence analysis of severe events did not differ between patients with and without PPIs (odds ratio 1.65, 95% confidence interval: 0.62-4.35) (P = 0.314), or for mortality (odds ratio 1.77, 95% confidence interval: 0.62-5.03) (P = 0.286).
Detailed and larger case studies are needed to accurately understand the role of PPIs in this viral infection.
Core Tip: In the scenario of the current pandemic caused by severe acute respiratory syndrome coronavirus 2 (SARS-CoV-2), it is essential to identify possible risk factors for the occurrence of this disease, including pharmacological therapies commonly taken by the population. Among these drugs, proton pump inhibitors represent one of the classes most widely used. In this review, we have tried to explore the link between the use of proton pump inhibitors and SARS-CoV-2 infection, and to analyze the strengths and weaknesses of this relationship, using the currently available data.
- Citation: Zippi M, Fiorino S, Budriesi R, Micucci M, Corazza I, Pica R, de Biase D, Gallo CG, Hong W. Paradoxical relationship between proton pump inhibitors and COVID-19: A systematic review and meta-analysis. World J Clin Cases 2021; 9(12): 2763-2777
- URL: https://www.wjgnet.com/2307-8960/full/v9/i12/2763.htm
- DOI: https://dx.doi.org/10.12998/wjcc.v9.i12.2763
A global pandemic was declared in February 2020 by the World Health Organization. The causative agent for this pandemic, which initiated in the city of Wuhan in Hubei Province, China, is a novel coronavirus, named severe acute respiratory syndrome coronavirus 2 (SARS-CoV-2), which leads to coronavirus disease 2019 (COVID-19)[1]. This pathogen shows similarities to two other previously identified coronaviruses, viz., Severe Acute Respiratory Syndrome (SARS-CoV) in 2003 and Middle Eastern Respiratory Syndrome (MERS-CoV) in 2012[2]. Although the main post-infection manifestations due to this virus include respiratory symptoms and pneumonia, it has been found to attack and to affect almost all organs and organ systems, including the gastrointestinal tract[3]. Recently, Tian et al[4] highlighted the presence of gastrointestinal symptoms in patients infected with SARS-CoV-2, with an incidence ranging from 3% up to 79%. This virus binds to the host cells through the spike (S) glycoprotein present on its envelope. Various studies have shown how this protein binds to the angiotensin converting enzyme 2 (ACE-2) receptors expressed on the cell membrane in human cells[5,6]. In the digestive system, ACE-2 receptors are expressed in the intestine, liver and pancreas[7-10]. In fact, the same SARS-CoV-2 RNA has also been found in the stool[7-10].
Proton pump inhibitors (PPIs) represent a class of drugs which suppress gastric acid secretion[11]. The United States Food and Drug Administration (FDA) has approved these molecules as long-term therapy for several gastrointestinal diseases, including peptic ulcer, Barrett esophagus, gastroesophageal reflux disease (GERD), Zollinger-Ellison syndrome and as a preventive treatment for gastrointestinal bleeding due to nonsteroidal anti-inflammatory drugs[12]. The first PPI approved by the FDA in the United States in 1989 was omeprazole, followed by lansoprazole in 1995, rabeprazole in 1999, pantoprazole in 2000 and esomeprazole in 2001[13]. From the first launch on the market of the first PPI, these molecules have become one of the most commonly used drugs worldwide, being prescribed to outpatients and during hospitalizations, and self-prescriptions as over-the-counter[11]. In a study carried out by Rotman and Bishop[14] considering only outpatient visits, it was shown that the prescription rate of PPIs increased from 4% in the year 2002 to 9.2% in 2009. A Spanish study analyzed the frequency of PPIs use before, during hospitalization and at the time of discharge and found that the percentage of patients in each category was 28.65%, 82.62% and 54.75%, respectively[15]. It is noteworthy that once the use of PPI is started by hospitalized patients, more than 50% of them continue it for nearly 3-6 mo after their discharge, without an appropriate indication[16].
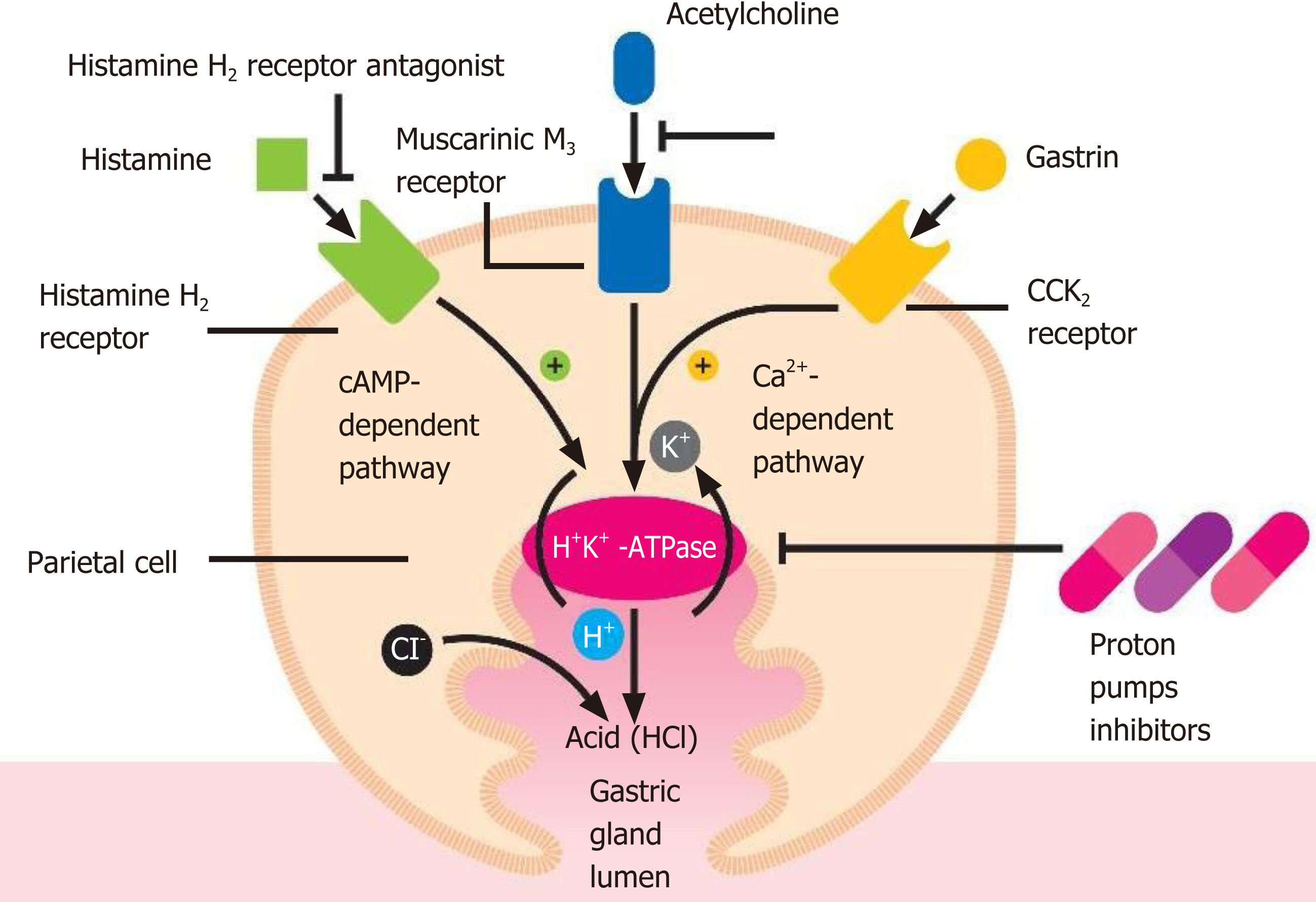
Acid secretion is regulated by the activity of the gastric parietal cells, also named oxyntic cells. These are a type of epithelial cell and are responsible for the production of hydrochloric acid (HCl), which results in an acidic pH with values between 1.5 and 3.5, and affects other intrinsic factors[17]. Their localization is predominantly at the level of the gastric glands of the fundus and in the body of the stomach[17]. This secretion of acid is regulated via various pathways, such as paracrine, endocrine and neural pathways. These cells are considered “dynamic”, as they are able to modify their morphology between the resting state and activity[17,18]. The production of HCl occurs through stimulating molecules, such as gastrin, acetylcholine and histamine when they bind to their respective receptors viz., CCK2 or Cholecystokinin2, Muscarinic M3 and histamine H2 at the level of the basolateral cellular membranes[17]. The basic pharmacophore of all the available PPIs is 2-pyridylmethylsulfinylbenzimidazole, i.e., as they have the same basic structural framework and differ only in the nature of substituents present in the rings of pyridine and benzimidazole[19,20]. These PPIs interact with the gastric H,K-ATPase or “proton pump”, an α,β-heterodimeric enzyme located internally with respect to the cytoplasmic membrane of the resting parietal cells[19]. When the parietal cell is activated, the H,K-ATPase enzyme moves from the cytoplasm to the apical portion of its membrane[21]. Of the two subunits of the gastric H,K-ATPase, the α sub-unit has catalytic function, while the β sub-unit is fundamentally responsible for the proper functioning of this enzyme. It binds to the membrane, thereby preventing this enzyme from going back into the cytoplasm[22]. The function of this protein is to exchange cytoplasmic hydronium with potassium present in the gastric lumen, which is associated with the secretion of chlorine leading to the formation of HCl[23]. It is due to this activity that the PPIs come into play, irreversibly blocking the gastric H,K-ATPase and thus preventing acid secretion (Figure 1).
In the last decade various reports have suggested that PPIs can favor the more severe course and mortality of illnesses in patients suffering from conditions, such as virosis, due to their side effects[24-26]. They include (1) inhibition of the enzymatic activity of DDAH (dimethylarginine dimethylaminohydrolase), with consequent cascade inhibition of nitric oxide synthase and a relative increase in inflammatory and thrombotic phenomena, with mainly cardiovascular sequelae; and (2) damage mainly to the renal tubules with the appearance of recurrent acute interstitial nephritis[24,27]. PPIs are also able to inhibit neutrophil function, thereby compromising the immune response to infections, as neutrophils are at the initial forefront against external agents[28]. This dampened neutrophilic immune response can make the individual more prone to infections and the disease more severe, also in the context of SARS-CoV-2 infection. Supporting this fact, several studies have shown that some viral infections (influenza virus, rotavirus, norovirus and, Middle East respiratory syndrome coronavirus infections) are more prevalent in PPIs users[29]. In a meta-analysis conducted by Eom et al[30], the use of these drugs was described to increase the risk of developing pneumonia, initially due to their gastric acid suppression followed by high digestive tract bacterial overgrowth. Ultimately the bacteria are translocated and colonize the lungs by microaspirations. However, this finding has only been partially confirmed by two other studies. A study led by Luxenburger et al[31] found that GERD in 9 (14.5%) of 62 patients included in their study was an indication for PPIs treatment and another study by Almario et al[32] found similar results in 109 (3.2%) of 3386 patients. The hypothesis that treatment with PPIs may increase the risk of secondary infections in hospitalized patients has not yet been fully confirmed. Luxenburger et al[31] also demonstrated how the use of this therapy could increase the risk of contracting secondary infections and, subsequently, of developing acute respiratory distress syndrome. In a previous study carried out on SARS-CoV-1, Darnell et al[33] observed that a gastric pH ≤ 3 has the capacity to alter the infectivity of this virus, while further lower values of pH resulting from acid suppression by PPIs could not inactivate the virus. This could represent fundamental information, as SARS-CoV-2 is very similar to SARS-CoV-1 and it has been proven that SARS-CoV-2 can cause infection in the gastrointestinal tract and in the airways[34]. Another opposite view on the use of PPIs during COVID-19 disease is that their administration may result in decreased absorption of several vitamins[35]. Moreover, PPIs seem to decrease vitamin C bioavailability and as a result leads to a reduction of vitamin C and its antioxidant form, ascorbic acid concentration in gastric juices. These effects were clinically reported by several researchers[36,37]. The above-mentioned observation could be correlated to recent findings demonstrating the efficacy of this vitamin both in prophylaxis and in severe forms of COVID-19. It has been shown by Feyaerts et al[38] that low doses (0.5-2 g/d) of vitamin C could benefit patients if taken early in severe forms of COVID-19 and in severe later stages of the infection, while high dosages are able to reduce some mediators of inflammation, such as interleukin-6 and endothelin-1[38]. The benefits of using high-dose vitamin C in severe cases have also been shown by Hoang et al[39]. With regard to the role of magnesium and vitamin D in this infection and how are they related to PPIs, several studies have shown that hypomagnesemia is one of the side effects reported in patients with long-term use of PPIs[40]. Low levels of serum magnesium may be associated with a wide spectrum of symptoms, including fatigue, tetany, delirium, convulsions and arrhythmias[40]. Chronic use of PPIs has been shown to increase the risk of bone fractures, whose incidence is decreased by the use of vitamin D in low doses[40]. Magnesium (Mg2+) is a cation present in the human body, ranking second among the intracellular cations and fourth among extracellular cations, whose intestinal absorption occurs through two proteins located on the apical membrane of enterocytes, TRPM6 and TRMP7 (transient receptor potential melastatin family)[41,42]. These optimally work in an acidic intraluminal environment[43]. PPIs can decrease the activity of TRPM6, resulting in a reduction in magnesium absorption and in the onset of hypomagnesemia[44]. Several functions are attributed to this element, including the regulation of calcium and potassium ion channels[41]. The lipid-soluble vitamin D needs magnesium to assume its active form (1,25[OH]2D)[45]. More and more studies have shown a link between low levels of vitamin D and increased susceptibility to contracting the SARS-CoV-2 infection, and in the severity of the clinical course[46,47]. In a recent meta-analysis, which included six studies[31,48-52] with a total of 5884 hospitalized COVID-19 patients, Hariyanto et al[53] showed how the use of PPIs was significantly associated with an increased risk of severity and mortality in COVID-19 cases. Aby et al[54] also underlined the high rate of mortality in such patients (COVID-19 positive and PPIs users). This was a retrospective study conducted in the American population, but details of the data have not been published. The authors also highlighted how the association between mortality and PPIs intake tends to decrease by considering and by correcting some factors, such as age, cardiovascular, pulmonary and renal comorbidities. They argued that other causes associated with the use of PPIs may affect mortality[54].
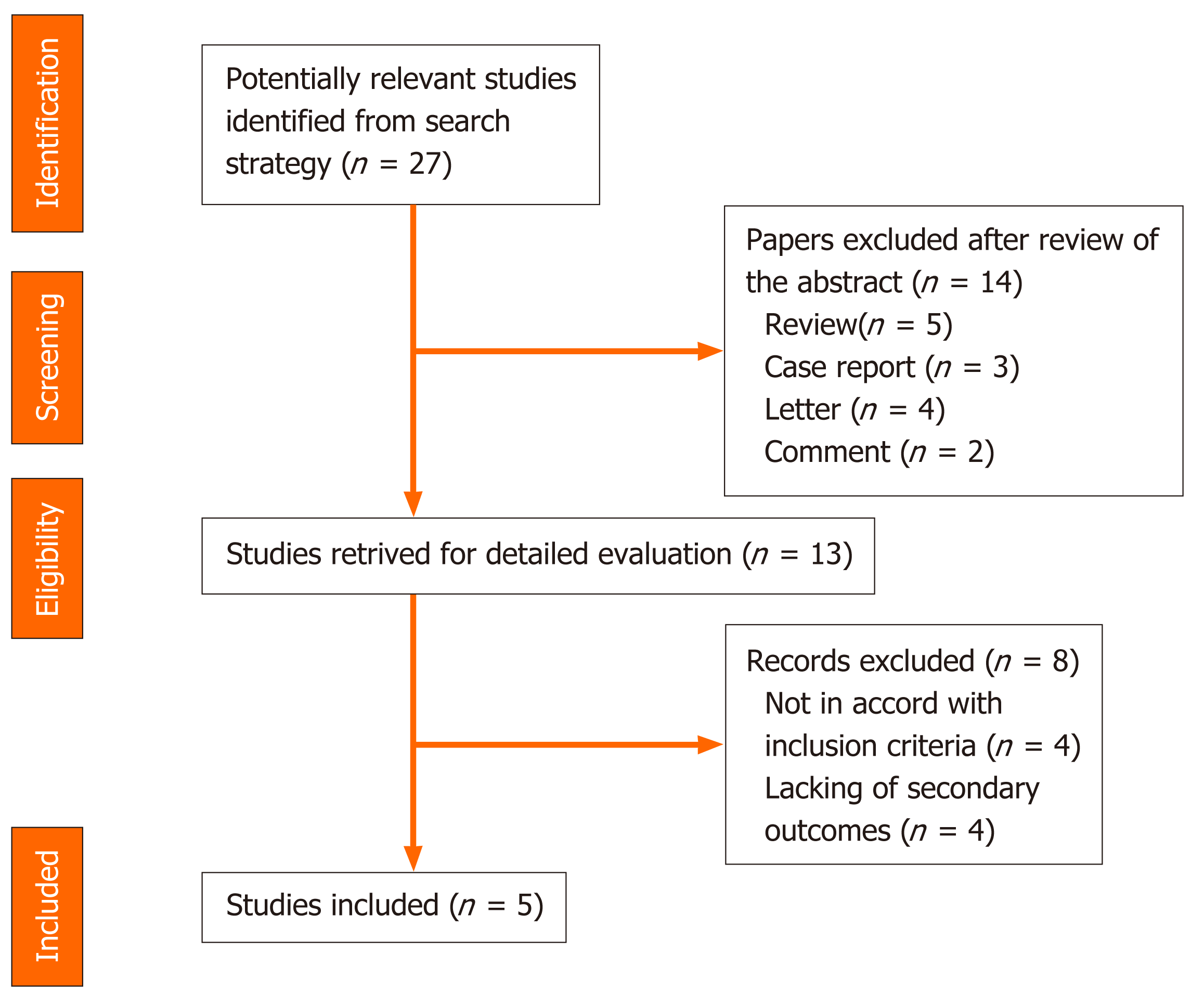
A systematic computer-based search of available published articles was carried out in order to identify relevant studies on the association between PPIs and COVID-19 (primary outcome) and the consequent related severity and mortality (secondary outcomes). Four electronic databases, including the MEDLINE/PubMed, the Cochrane Library, EMBASE, and Google Scholar were searched up to November 2020, for all relevant articles published in English on this topic. Also, relevant articles, published in other languages were taken into account, and information was taken from their English abstracts. The search terms were identified by means of controlled vocabularies, such as the National Library of Medicine’s MESH (Medical Subject Headings) and keywords. The MESH terms and keywords used were: “COVID-19”, “proton pump inhibitors”, ”PPIs”, “SARS-CoV-2”, “outcomes”, “severity” and “mortality”. All studies that contained material on this specific topic were considered. The inclusion criteria regarding the studies considered in our analysis were: meta-analyses, case-control, hospital-based case-control, population-based case-control as well as cohort-studies, while papers not published as full reports, such as conference abstracts, case reports, and editorials were excluded. Data extraction was performed by two authors (de Biase D and Hong W), in parallel and in independent mode. The other two authors (Pica R and Gallo CG), independently extracted and tabulated all relevant data from the selected studies. Lastly, the accuracy of data extrapolated was checked by Fiorino S, and when discrepancies concerning the results were found among the selected works, a consensus between all authors was mandatory. In order to avoid possible duplicates, we searched the first author’s name of the research, as well as the place and the period of the subjects’ enrolment. In the case of different versions of the same study, only the most recent was considered (Figure 2).
We summarized and collated all the available information from the final analysis of the studies, identifying five works from which the data were extracted and collected where possible the two secondary outcomes: severe events, defined as the incidence of patients needing organ support or intensive care, and mortality, namely death occurring during hospitalization. Studies were excluded from the analysis if either the outcomes of interest were not mentioned or it was impossible to calculate them from the published results. Similarly, other studies were ruled out if the sample size was less than ten and if focused on animals. We used forest plots to estimate the pooled effect sizes of each study (Table 1), with 95%CI, to provide a visual summary of the data. Heterogeneity among studies was evaluated by the Cochran Q-test and I-squared index, with I2 values above 75% as thresholds for high heterogeneity. When heterogeneity was present, we used a random-effects model (DerSimonian-Laird model). Otherwise, a fixed-effects model (Mantel-Haenszel) was used to compute the overall effects.
| Ref. | Type of study | Number of patients with SARS-CoV-2 | Period of intake of PPIs | Number of patients treated or not treated with PPIs (%) | Severity of patients treated or not treated with PPIs (%) | Mortality in patients treated or not treated with PPIs (%) |
| Luxenburger et al[31] | Retrospective | 152 | Home drug | 62 (40.7) | 18 + 11 = 29 (32.2) (secondary infection + ARDS) | 5 (5.6) |
| Luxenburger et al[31] | Retrospective | 152 | Home drug | 90 (59.3) | 30 + 17 = 47 (75.8) (secondary infection + ARDS) | 12 (19.4) |
| Almario et al[32] | Online survey | 3386 | Home drugs | 2661 (78.6) | NA | NA |
| Almario et al[32] | Online survey | 3386 | Home drugs | 752 (2.1) | NA | NA |
| McKeigue et al[48] | Case-control | 36391 | Home drug | 13982 (38.4) | 1177 (8.4) (severe outcomes) | NA |
| McKeigue et al[48] | Case-control | 36391 | Home drug | 22409 (61.6) | 12805 (57.1) (severe outcomes) | NA |
| Argenziano et al[49] | Retrospective | 1000 | Home drug | 163 (16.3) | 38 (23.3) (intensive care units) | NA |
| Argenziano et al[49] | Retrospective | 1000 | Home drug | 837 (83.7) | NA | NA |
| Ramachandran et al[50] | Retrospective | 295 | Home drug | 46 (15.5) | 16 + 12 = 28 (60.8) (shock + mechanical ventilation) | 16 (34.7) |
| Ramachandran et al[50] | Retrospective | 295 | Home drug | 249 (84.5) | 77 + 37 = 114 (45.7) (shock + mechanical ventilation) | 40 (16.1) |
| Lee et al[51] | Retrospective | 534 | Home drug | 267 (50) | 49 | 24 |
| Lee et al[51] | Retrospective | 534 | Home drug | 267 (50) | 32 | 14 |
| Ullah et al[52] | Retrospective | 212 (with pre-existing HPB condition) | Home drug | 109 (51.4) | NA | 27 (24.8) |
| Ullah et al[52] | Retrospective | 212 (with pre-existing HPB condition) | Home drug | 103 (48.6) | NA | 27 (26.2) |
| García-Menaya et al[55] | Retrospective | 113 (asthma and allergic patients) | Home drug | 54 (47.8) | NA | NA |
| García-Menaya et al[55] | Retrospective | 113 (asthma and allergic patients) | Home drug | 59 (52.2) | NA | NA |
| Gadiparthi et al[56] | Retrospective | 3 (all with GI bleeding) | In hospital | 3 (100) | 1 (33.3) (intensive care unit) | 2 (66.7) |
To date, according to the available data in the literature, we identified 9 papers describing the use of PPIs in SARS-CoV-2 patients. The results are summarized in Table 1.
Taking into account the study by Hariyanto et al[53], we included data on the severity of the disease, specifically for 16 patients and 77 patients with shock, respectively, in the PPI-group and in the non-PPI-group[50], 18 patients and 30 patients had secondary infection, respectively, in the PPI-group and in the non-PPI-group[31]. A study by García-Menaya et al[55] included data related to severity and mortality of the disease, but they were calculated for patients with and without allergic conditions and for overall patients. It was observed that a history of PPIs intake was related to an increased risk of admission to the intensive care unit for all patients in the cohort (P = 0.017)[55]. In some of these studies, it was possible to extrapolate the type of PPI used, the dosage and the posology. As in the study conducted by Luxenburger et al[31], the percentage of patients using PPIs was calculated to be 83.9% (52 patients) for pantoprazole, 9.7% (6 patients) for esomeprazole and 6.5% (4 patients) for omeprazole. Almario et al[32] found that people taking PPIs once daily [adjusted odds ratio (aOR) 2.15; 95% confidence interval (CI): 1.90-2.44] or twice daily (aOR 3.67; 95%CI: 2.93-4.60) showed a statistically significant risk of testing positive compared to those who did not use such drugs. On the contrary, it emerged that the use of histamine-2 receptor antagonist (H2RA) was not associated with the risk of contracting the infection. In particular, 415 individuals (5.6%) were using H2RA once daily (aOR 0.85; 95%CI: 0.74-0.99) and 143 (12.4%) twice daily (aOR 0.86; 95%CI: 0.66-1.11)[32]. Lee et al[51] noted that people who had taken PPIs for less than 30 d before contracting the virus showed an increased risk of severity compared to those who had never taken these drugs. This outcome was further confirmed by a study on a subgroup of Korean individuals who were at that time under PPIs therapy[56,57].
A random-effect model was used both for disease severity and for mortality, due to the extensive heterogeneity occurring between studies (I2 = 95.7% and I2 = 89.0% respectively). The pooled incidence of severe events did not differ between patients with and without PPIs (OR 1.65, 95%CI: 0.62-4.35) (P = 0.314), or for mortality (OR 1.77, 95%CI: 0.62-5.03) (P = 0.286) (Figure 3 and 4).
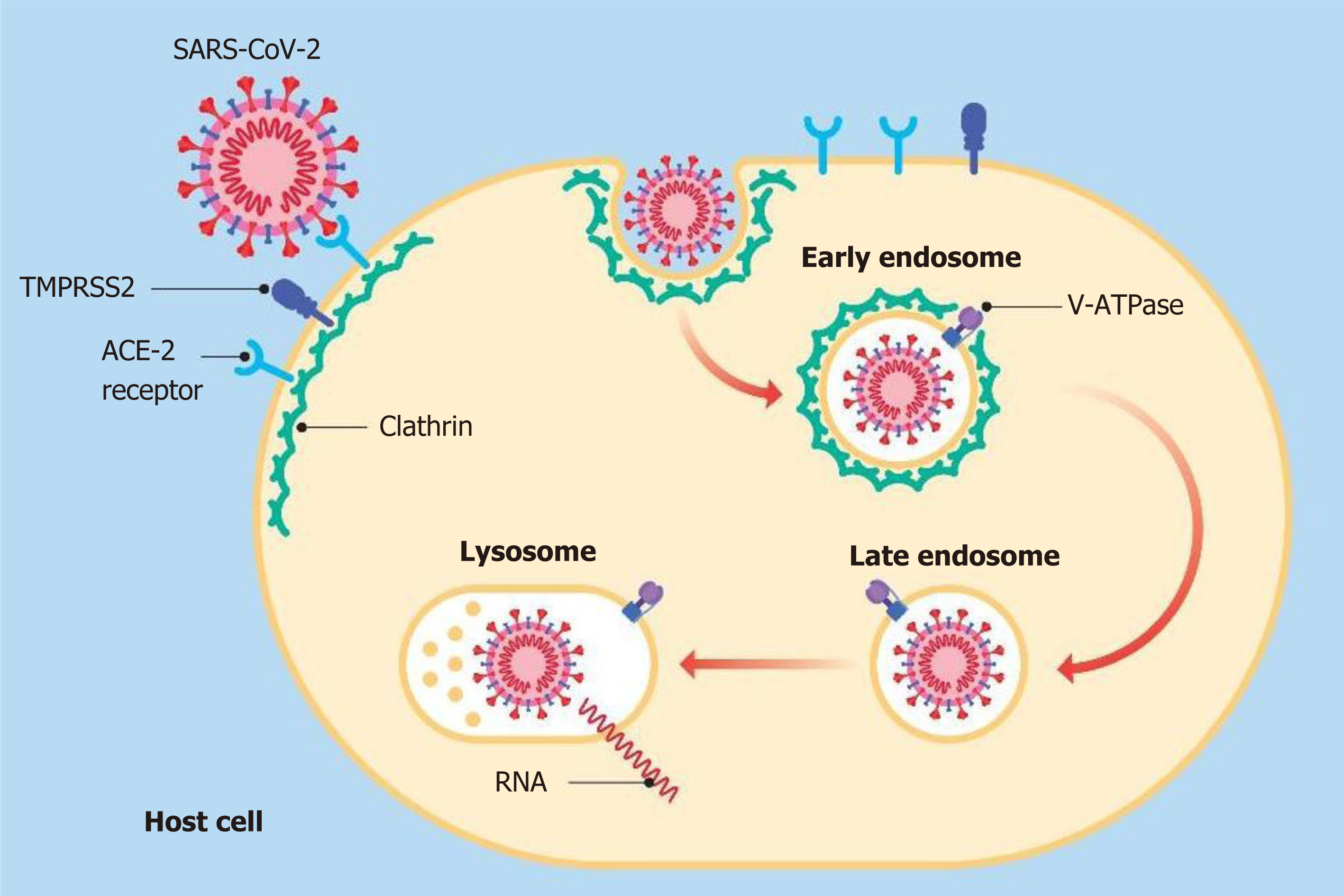
The literature, on the other hand, also provided a few studies supporting the beneficial effect of PPIs and their explicit action to counteract infections, including viral infections. It has been shown that the lowering of pH due to the gastric acid anti-secretory action of PPIs can kill ingested microorganisms and avoid gastric bacterial overgrowth[58]. These two activities are useful both in preventing infections caused by Clostridium difficile and in the development of spontaneous bacterial peritonitis[58,59]. Taştemur et al[60] hypothesized that PPIs might play a role, both in prophylaxis and in the treatment of COVID-19. Ray et al[61], based on available scientific evidence, suggested the use of PPIs for therapeutic purposes in this virosis. It has been demonstrated that both pantoprazole and omeprazole can show a modulatory and an inhibitory effect on autophagy. This physiological mechanism occurs in almost all cells and is capable of eliminating damaged or malfunctioning proteins, thereby ensuring cytoplasmic homeostasis[62-64]. Autophagy may not work properly in various diseases, such as neurodegenerative disorders, metabolic diseases, cancer and infectious diseases. Host autophagy can help to maintain homeostasis of intracellular organelles through the elimination of cellular waste, and affect the activities of some viruses to promote their proliferation and spread, and this modality is also true for SARS-CoV-2[65]. The mechanisms involved in these events are not fully understood, nevertheless it has become apparent that as the intracellular pH rises, autophagy is also stimulated[66]. For this reason, researchers are now considering particularly the use of pantoprazole as a possible molecule in the course of COVID-19 pharmacologic treatment[67]. PPIs are also able to regulate fibrogenesis, showing anti-fibrotic effects through the inhibition of molecules, such as fibronectin, collagen and matrix metalloproteinase enzymes[68]. Pulmonary fibrosis, thought to be a long-term consequence of this viral infection is still debatable, many studies have correlated the use of PPIs with clinical improvement in patients suffering from idiopathic pulmonary fibrosis[69-72]. During the previous SARS pandemic, SARS-CoV-1 virus inactivation based on intracellular pH was analyzed in alkaline conditions[73]. Pagat et al[73] in their experiments, using Vero cells and sodium hydroxide (NaOH) as an alkalizing agent, observed that the virus was active at a pH of 11, while at pH 13-13.5 it was immediately inactivated[73]. Viruses can directly enter host cells by binding their specific surface proteins to precise receptors followed by endocytosis, as in the case of SARS-CoV-2, in which the protein is a spike (S) glycoprotein and the receptor is an ACE-2[74]. This protein is organized in two functional domains: S1 subunit containing the receptor-binding domain (RBD) to bind the target cell by adhering to the ACE-2 receptor and S2 subunit, which is a second phase allowing the virus to enter into the host cell by fusing the respective membranes[75]. The fusion between S2 and the ACE-2 receptor is preceded by the action of a specific cellular protease, serine protease transmembrane 2 (TMPRSS2)[76]. In this way, the entire virus can enter into cells after formation of the SARS-CoV-2/ACE-2 complex and the relative fusion between the viral membrane and the luminal membrane of the endosome. This favors the transfer of the relative RNA to the cytosol[77-79]. The entry process also requires the intervention of other factors, such as endosomal pH acidification[80]. Bayati et al[81] found that the pathway of endocytic entry is represented by clathrin-mediated endocytosis (CME) (Figure 5). This discovery is very important, as one of the molecules used in treatment of patients affected by COVID-19 is chloroquine (CQ), which blocks CME by reducing the expression of the phosphatidylinositol binding clathrin assembly protein (PICALM)[82,83]. Aprotinin, a serine protease inhibitor, is used to treat pancreatitis and to reduce bleeding during surgery[84,85]. Its use was also assessed against influenza viruses, so much so that in Russia a formulation of this molecule in the form of an aerosol has been approved for the treatment of these viruses[86]. As previously observed by Shen et al[87], omeprazole can interfere with the acidification of lysosomes, a necessary requirement for viral replication. In a recent SARS-CoV-2-infected cell culture study, Bojkova et al[88] discovered that the administration of omeprazole, at a therapeutic plasma concentration of 8 μmol/L, has been shown to increase the efficacy of aprotinin by 2.7 times and remdesivir by 10 times. The findings showed that the combination of these two molecules (aprotinin and remdesivir) or single use, supported by the intake of omeprazole, may represent one of the possible emerging treatments for COVID-19[89]. A common feature of both endosomes and lysosomes is a low intraluminal pH[90]. Vacuolar ATPase (V-ATPase), functionally identical to H+-ATPase, expressed on the surface of lysosomes and endosomes, is fundamental for the acidification of these two organelles[91,92] (Figure 5). An increase in the pH of these organelles could inhibit viral replication[93]. Omeprazole and lansoprazole might inhibit V-ATPase in gastric mucosa and omeprazole has been shown to block this pump in rat models[94,95]. It has also been observed that both omeprazole and esomeprazole can change the location of the V-ATPase in cells[96]. The inhibition of V-ATPase by PPIs results in loss of the pH gradient across the membranes of lysosomes and endosomes, resulting in cytosolic acidification and in endolysosomal alkalinization, thus hindering viral replication and the resulting spread[97-99]. Further studies are needed to clarify these findings.
The role of PPIs in the progression of infection by SARS-CoV-2 is still not fully understood. Most of the published works underline how their use, both before and during hospitalization, can represent a risk factor in the course of the disease in terms of severity and mortality. Our review, based on the available studies, has some limitations: (1) most of the studies are observational or case-control studies; (2) there are no randomized controlled trials; (3) confounding aspects are present, such as statistically underpowered; and (4) statistically powered longitudinal studies in order to explore causality are lacking. These drugs possess well known anti-inflammatory and anti-fibrotic properties. If viral activity is regulated by intracellular pH, then what role can PPIs play? Thus, a paradoxical relationship between SARS-CoV-2 infection and PPIs has emerged. This review analyzed the possible effects of PPIs in COVID-19 patients with the aim of trying to understand if their administration may be deleterious or may interfere with the clinical course of the disease.
Coronavirus disease 2019 (COVID-19) is an emerging infection increasingly found in clinical practice, and is now a pandemic. Conventional pharmacotherapies, including proton pump inhibitors, are commonly used in the treatment of such patients. However, their clinical efficacy, as well as the risk of severe adverse events, remain a problem in their clinical application. Based on the fact that the use of this class of drugs seems to show both negative, particularly in terms of severity and mortality, and positive effects on the course of this disease, it is neither realistic nor definitive to consider not using these medicinal products in these patients.
Several basic and clinical studies have reported a relationship between proton pump inhibitors (PPIs) use and COVID-19. As the results obtained so far regarding these drugs are both against their use and in favor of it, these discordant conclusions may create confusion among doctors regarding their treatment choices.
The main objectives of this review were to critically analyze the available data from studies accessible from the literature. Moreover, by following the extracted outcomes, future research is needed.
We searched three electronic databases up to 30 November 2020, for studies that examined the safety and efficacy of the administration of PPIs in patients suffering from COVID-19. Two endpoints were considered: the severity and the mortality rates in this cohort. In order to calculate the estimated risks in this meta-analysis, fixed and random effects models were used.
The pooled incidence of severe events did not differ between patients with and without PPIs [odds ratio 1.65, 95% confidence interval (CI): 0.62-4.35] (P = 0.314), or for mortality (odds ratio 1.77, 95%CI: 0.62-5.03) (P = 0.286).
This review analyzed the possible effects of PPIs in COVID-19 patients with the aim of trying to understand if their administration may be deleterious or may interfere with the clinical course of the disease. The role of PPIs in the progression of severe acute respiratory syndrome coronavirus 2 infection is still not fully understood.
The paradoxical relationship between PPIs and COVID-19 should not be a confounding factor for doctors but should prompt them to proceed with further studies in this regard.
The Authors thank Dr. Varlotta A, Rome, Italy and Washington, United States, for her contribute in graphic illustration and Dr. de Vito S, Italian Medicines Agency, Rome, Italy, for the further English revision of the manuscript.
Manuscript source: Invited manuscript
Corresponding Author's Membership in Professional Societies: European Society of Gastrointestinal Endoscopy, No. 45910119.
Specialty type: Medicine, research and experimental
Country/Territory of origin: Italy
Peer-review report’s scientific quality classification
Grade A (Excellent): 0
Grade B (Very good): B
Grade C (Good): 0
Grade D (Fair): 0
Grade E (Poor): 0
P-Reviewer: Jahromi R S-Editor: Gao CC L-Editor: Webster JR P-Editor: Zhang YL
| 1. | Zheng J. SARS-CoV-2: an Emerging Coronavirus that Causes a Global Threat. Int J Biol Sci. 2020;16:1678-1685. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 650] [Cited by in RCA: 569] [Article Influence: 113.8] [Reference Citation Analysis (0)] |
| 2. | Jiang F, Deng L, Zhang L, Cai Y, Cheung CW, Xia Z. Review of the Clinical Characteristics of Coronavirus Disease 2019 (COVID-19). J Gen Intern Med. 2020;35:1545-1549. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 869] [Cited by in RCA: 703] [Article Influence: 140.6] [Reference Citation Analysis (0)] |
| 3. | Xiao F, Tang M, Zheng X, Liu Y, Li X, Shan H. Evidence for Gastrointestinal Infection of SARS-CoV-2. Gastroenterology 2020; 158: 1831-1833. e3. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 1985] [Cited by in RCA: 1993] [Article Influence: 398.6] [Reference Citation Analysis (1)] |
| 4. | Tian Y, Rong L, Nian W, He Y. Review article: gastrointestinal features in COVID-19 and the possibility of faecal transmission. Aliment Pharmacol Ther. 2020;51:843-851. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 594] [Cited by in RCA: 583] [Article Influence: 116.6] [Reference Citation Analysis (0)] |
| 5. | Ali A, Vijayan R. Dynamics of the ACE2-SARS-CoV-2/SARS-CoV spike protein interface reveal unique mechanisms. Sci Rep. 2020;10:14214. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 139] [Cited by in RCA: 165] [Article Influence: 33.0] [Reference Citation Analysis (0)] |
| 6. | Song W, Gui M, Wang X, Xiang Y. Cryo-EM structure of the SARS coronavirus spike glycoprotein in complex with its host cell receptor ACE2. PLoS Pathog. 2018;14:e1007236. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 618] [Cited by in RCA: 586] [Article Influence: 83.7] [Reference Citation Analysis (0)] |
| 7. | Ksiazek TG, Erdman D, Goldsmith CS, Zaki SR, Peret T, Emery S, Tong S, Urbani C, Comer JA, Lim W, Rollin PE, Dowell SF, Ling AE, Humphrey CD, Shieh WJ, Guarner J, Paddock CD, Rota P, Fields B, DeRisi J, Yang JY, Cox N, Hughes JM, LeDuc JW, Bellini WJ, Anderson LJ; SARS Working Group. A novel coronavirus associated with severe acute respiratory syndrome. N Engl J Med. 2003;348:1953-1966. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 3147] [Cited by in RCA: 3066] [Article Influence: 139.4] [Reference Citation Analysis (0)] |
| 8. | Zippi M, Fiorino S, Occhigrossi G, Hong W. Hypertransaminasemia in the course of infection with SARS-CoV-2: Incidence and pathogenetic hypothesis. World J Clin Cases. 2020;8:1385-1390. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in CrossRef: 23] [Cited by in RCA: 23] [Article Influence: 4.6] [Reference Citation Analysis (0)] |
| 9. | Zippi M, Hong W, Traversa G, Maccioni F, De Biase D, Gallo C, Fiorino S. Involvement of the exocrine pancreas during COVID-19 infection and possible pathogenetic hypothesis: a concise review. Infez Med. 2020;28:507-515. [PubMed] |
| 10. | Yeo C, Kaushal S, Yeo D. Enteric involvement of coronaviruses: is faecal-oral transmission of SARS-CoV-2 possible? Lancet Gastroenterol Hepatol. 2020;5:335-337. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 546] [Cited by in RCA: 554] [Article Influence: 110.8] [Reference Citation Analysis (0)] |
| 11. | Shin JM, Sachs G. Pharmacology of proton pump inhibitors. Curr Gastroenterol Rep. 2008;10:528-534. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 370] [Cited by in RCA: 320] [Article Influence: 18.8] [Reference Citation Analysis (0)] |
| 12. | Nehra AK, Alexander JA, Loftus CG, Nehra V. Proton Pump Inhibitors: Review of Emerging Concerns. Mayo Clin Proc. 2018;93:240-246. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 132] [Cited by in RCA: 157] [Article Influence: 22.4] [Reference Citation Analysis (0)] |
| 13. | Strand DS, Kim D, Peura DA. 25 Years of Proton Pump Inhibitors: A Comprehensive Review. Gut Liver. 2017;11:27-37. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 219] [Cited by in RCA: 395] [Article Influence: 49.4] [Reference Citation Analysis (0)] |
| 14. | Rotman SR, Bishop TF. Proton pump inhibitor use in the U.S. ambulatory setting, 2002-2009. PLoS One. 2013;8:e56060. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 90] [Cited by in RCA: 131] [Article Influence: 10.9] [Reference Citation Analysis (0)] |
| 15. | Ramirez E, Lei SH, Borobia AM, Piñana E, Fudio S, Muñoz R, Campos A, Carcas AJ, Frias J. Overuse of PPIs in patients at admission, during treatment, and at discharge in a tertiary Spanish hospital. Curr Clin Pharmacol. 2010;5:288-297. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 49] [Cited by in RCA: 50] [Article Influence: 3.3] [Reference Citation Analysis (0)] |
| 16. | Grant K, Al-Adhami N, Tordoff J, Livesey J, Barbezat G, Reith D. Continuation of proton pump inhibitors from hospital to community. Pharm World Sci. 2006;28:189-193. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 48] [Cited by in RCA: 57] [Article Influence: 3.0] [Reference Citation Analysis (0)] |
| 17. | Yao X, Forte JG. Cell biology of acid secretion by the parietal cell. Annu Rev Physiol. 2003;65:103-131. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 198] [Cited by in RCA: 184] [Article Influence: 8.4] [Reference Citation Analysis (0)] |
| 18. | Ogata T, Yamasaki Y. Ultra-high-resolution scanning electron microscopy of the continuity of cytoplasmic and luminal membranes in frog oxyntic cells. Anat Rec. 1996;245:559-567. [RCA] [PubMed] [DOI] [Full Text] [Cited by in RCA: 1] [Reference Citation Analysis (0)] |
| 19. | Roche VF. The chemically elegant proton pump inhibitors. Am J Pharm Educ. 2006;70:101. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 65] [Cited by in RCA: 63] [Article Influence: 3.3] [Reference Citation Analysis (0)] |
| 20. | Shin JM, Munson K, Vagin O, Sachs G. The gastric HK-ATPase: structure, function, and inhibition. Pflugers Arch. 2009;457:609-622. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 210] [Cited by in RCA: 180] [Article Influence: 11.3] [Reference Citation Analysis (0)] |
| 21. | Sachs G, Chang HH, Rabon E, Schackman R, Lewin M, Saccomani G. A nonelectrogenic H+ pump in plasma membranes of hog stomach. J Biol Chem. 1976;251:7690-7698. [PubMed] |
| 22. | Maeda M, Oshiman K, Tamura S, Futai M. Human gastric (H+ + K+)-ATPase gene. Similarity to (Na+ + K+)-ATPase genes in exon/intron organization but difference in control region. J Biol Chem. 1990;265:9027-9032. [PubMed] |
| 23. | Morii M, Yamauchi M, Ichikawa T, Fujii T, Takahashi Y, Asano S, Takeguchi N, Sakai H. Involvement of the H3O+-Lys-164 -Gln-161-Glu-345 charge transfer pathway in proton transport of gastric H+,K+-ATPase. J Biol Chem. 2008;283:16876-16884. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 17] [Cited by in RCA: 14] [Article Influence: 0.8] [Reference Citation Analysis (0)] |
| 24. | Savarino V, Marabotto E, Furnari M, Zingone F, Zentilin P, Savarino E. Latest insights into the hot question of proton pump inhibitor safety - a narrative review. Dig Liver Dis. 2020;52:842-852. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 18] [Cited by in RCA: 29] [Article Influence: 5.8] [Reference Citation Analysis (0)] |
| 25. | Meijvis SC, Cornips MC, Voorn GP, Souverein PC, Endeman H, Biesma DH, Leufkens HG, van de Garde EM. Microbial evaluation of proton-pump inhibitors and the risk of pneumonia. Eur Respir J. 2011;38:1165-1172. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 26] [Cited by in RCA: 28] [Article Influence: 2.0] [Reference Citation Analysis (0)] |
| 26. | Sasaki T, Nakayama K, Yasuda H, Yamaya M. A new strategy with proton pump inhibitors for the prevention of acute exacerbations in COPD. Ther Adv Respir Dis. 2011;5:91-103. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 17] [Cited by in RCA: 17] [Article Influence: 1.2] [Reference Citation Analysis (0)] |
| 27. | Savarino V, Dulbecco P, Savarino E. Are proton pump inhibitors really so dangerous? Dig Liver Dis. 2016;48:851-859. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 57] [Cited by in RCA: 60] [Article Influence: 6.7] [Reference Citation Analysis (0)] |
| 28. | Namazi MR, Jowkar F. A succinct review of the general and immunological pharmacologic effects of proton pump inhibitors. J Clin Pharm Ther. 2008;33:215-217. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 45] [Cited by in RCA: 43] [Article Influence: 2.5] [Reference Citation Analysis (0)] |
| 29. | Charpiat B, Bleyzac N, Tod M. Proton Pump Inhibitors are Risk Factors for Viral Infections: Even for COVID-19? Clin Drug Investig. 2020;40:897-899. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 22] [Cited by in RCA: 23] [Article Influence: 4.6] [Reference Citation Analysis (0)] |
| 30. | Eom CS, Jeon CY, Lim JW, Cho EG, Park SM, Lee KS. Use of acid-suppressive drugs and risk of pneumonia: a systematic review and meta-analysis. CMAJ. 2011;183:310-319. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 305] [Cited by in RCA: 277] [Article Influence: 19.8] [Reference Citation Analysis (1)] |
| 31. | Luxenburger H, Sturm L, Biever P, Rieg S, Duerschmied D, Schultheiss M, Neumann-Haefelin C, Thimme R, Bettinger D. Treatment with proton pump inhibitors increases the risk of secondary infections and ARDS in hospitalized patients with COVID-19: coincidence or underestimated risk factor? J Intern Med. 2021;289:121-124. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 44] [Cited by in RCA: 44] [Article Influence: 11.0] [Reference Citation Analysis (0)] |
| 32. | Almario CV, Chey WD, Spiegel BMR. Increased Risk of COVID-19 Among Users of Proton Pump Inhibitors. Am J Gastroenterol. 2020;115:1707-1715. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 147] [Cited by in RCA: 135] [Article Influence: 27.0] [Reference Citation Analysis (0)] |
| 33. | Darnell ME, Subbarao K, Feinstone SM, Taylor DR. Inactivation of the coronavirus that induces severe acute respiratory syndrome, SARS-CoV. J Virol Methods. 2004;121:85-91. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 476] [Cited by in RCA: 455] [Article Influence: 22.8] [Reference Citation Analysis (0)] |
| 34. | Trottein F, Sokol H. Potential Causes and Consequences of Gastrointestinal Disorders during a SARS-CoV-2 Infection. Cell Rep. 2020;32:107915. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 87] [Cited by in RCA: 92] [Article Influence: 18.4] [Reference Citation Analysis (0)] |
| 35. | Heidelbaugh JJ. Proton pump inhibitors and risk of vitamin and mineral deficiency: evidence and clinical implications. Ther Adv Drug Saf. 2013;4:125-133. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 111] [Cited by in RCA: 108] [Article Influence: 9.0] [Reference Citation Analysis (0)] |
| 36. | Mowat C, Carswell A, Wirz A, McColl KE. Omeprazole and dietary nitrate independently affect levels of vitamin C and nitrite in gastric juice. Gastroenterology. 1999;116:813-822. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 78] [Cited by in RCA: 75] [Article Influence: 2.9] [Reference Citation Analysis (0)] |
| 37. | Henry EB, Carswell A, Wirz A, Fyffe V, McColl KE. Proton pump inhibitors reduce the bioavailability of dietary vitamin C. Aliment Pharmacol Ther. 2005;22:539-545. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 37] [Cited by in RCA: 31] [Article Influence: 1.6] [Reference Citation Analysis (0)] |
| 38. | Feyaerts AF, Luyten W. Vitamin C as prophylaxis and adjunctive medical treatment for COVID-19? Nutrition. 2020;79-80:110948. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 60] [Cited by in RCA: 54] [Article Influence: 10.8] [Reference Citation Analysis (0)] |
| 39. | Hoang BX, Shaw G, Fang W, Han B. Possible application of high-dose vitamin C in the prevention and therapy of coronavirus infection. J Glob Antimicrob Resist. 2020;23:256-262. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 61] [Cited by in RCA: 62] [Article Influence: 12.4] [Reference Citation Analysis (0)] |
| 40. | Srinutta T, Chewcharat A, Takkavatakarn K, Praditpornsilpa K, Eiam-Ong S, Jaber BL, Susantitaphong P. Proton pump inhibitors and hypomagnesemia: A meta-analysis of observational studies. Medicine (Baltimore). 2019;98:e17788. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 44] [Cited by in RCA: 71] [Article Influence: 11.8] [Reference Citation Analysis (0)] |
| 41. | Katopodis P, Karteris E, Katopodis KP. Pathophysiology of Drug-Induced Hypomagnesaemia. Drug Saf. 2020;43:867-880. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 6] [Cited by in RCA: 9] [Article Influence: 2.3] [Reference Citation Analysis (0)] |
| 42. | Schmitz C, Perraud AL, Johnson CO, Inabe K, Smith MK, Penner R, Kurosaki T, Fleig A, Scharenberg AM. Regulation of vertebrate cellular Mg2+ homeostasis by TRPM7. Cell. 2003;114:191-200. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 567] [Cited by in RCA: 571] [Article Influence: 26.0] [Reference Citation Analysis (0)] |
| 43. | Thébault S, Cao G, Venselaar H, Xi Q, Bindels RJ, Hoenderop JG. Role of the alpha-kinase domain in transient receptor potential melastatin 6 channel and regulation by intracellular ATP. J Biol Chem. 2008;283:19999-20007. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 42] [Cited by in RCA: 42] [Article Influence: 2.5] [Reference Citation Analysis (0)] |
| 44. | Voets T, Nilius B, Hoefs S, van der Kemp AW, Droogmans G, Bindels RJ, Hoenderop JG. TRPM6 forms the Mg2+ influx channel involved in intestinal and renal Mg2+ absorption. J Biol Chem. 2004;279:19-25. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 457] [Cited by in RCA: 428] [Article Influence: 19.5] [Reference Citation Analysis (0)] |
| 45. | Uwitonze AM, Razzaque MS. Role of Magnesium in Vitamin D Activation and Function. J Am Osteopath Assoc. 2018;118:181-189. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 135] [Cited by in RCA: 200] [Article Influence: 28.6] [Reference Citation Analysis (1)] |
| 46. | Ali N. Role of vitamin D in preventing of COVID-19 infection, progression and severity. J Infect Public Health. 2020;13:1373-1380. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 310] [Cited by in RCA: 288] [Article Influence: 57.6] [Reference Citation Analysis (0)] |
| 47. | Fiorino S, Zippi M, Gallo C, Sifo D, Sabbatani S, Manfredi R, Rasciti E, Rasciti L, Giampieri E, Corazza I, Leandri P, de Biase D. The rationale for a multi-step therapeutic approach based on antivirals, drugs and nutrients with immunomodulatory activity in patients with coronavirus-SARS2-induced disease of different severities. Br J Nutr. 2021;125:275-293. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 9] [Cited by in RCA: 9] [Article Influence: 2.3] [Reference Citation Analysis (0)] |
| 48. | McKeigue PM, Kennedy S, Weir A, Bishop J, McGurnaghan SJ, McAllister D, Robertson C, Wood R, Lone N, Murray J, Caparrotta TM, Smith-Palmer A, Goldberg D, McMenamin J, Ramsay C, Guthrie B, Hutchinson S, Colhoun HM. Associations of severe COVID-19 with polypharmacy in the REACT-SCOT case-control study. 2020 Preprint. Available from: medRxiv. [DOI] [Full Text] |
| 49. | Argenziano MG, Bruce SL, Slater CL, Tiao JR, Baldwin MR, Barr RG, Chang BP, Chau KH, Choi JJ, Gavin N, Goyal P, Mills AM, Patel AA, Romney MS, Safford MM, Schluger NW, Sengupta S, Sobieszczyk ME, Zucker JE, Asadourian PA, Bell FM, Boyd R, Cohen MF, Colquhoun MI, Colville LA, de Jonge JH, Dershowitz LB, Dey SA, Eiseman KA, Girvin ZP, Goni DT, Harb AA, Herzik N, Householder S, Karaaslan LE, Lee H, Lieberman E, Ling A, Lu R, Shou AY, Sisti AC, Snow ZE, Sperring CP, Xiong Y, Zhou HW, Natarajan K, Hripcsak G, Chen R. Characterization and clinical course of 1000 patients with coronavirus disease 2019 in New York: retrospective case series. BMJ. 2020;369:m1996. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 430] [Cited by in RCA: 564] [Article Influence: 112.8] [Reference Citation Analysis (0)] |
| 50. | Ramachandran P, Perisetti A, Gajendran M, Jean-Louis F, Bansal P, Dwivedi AK, Goyal H. Pre-hospitalization proton pump inhibitor use and clinical outcomes in COVID-19. Eur J Gastroenterol Hepatol. 2020;. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 26] [Cited by in RCA: 28] [Article Influence: 9.3] [Reference Citation Analysis (0)] |
| 51. | Lee SW, Ha EK, Yeniova AÖ, Moon SY, Kim SY, Koh HY, Yang JM, Jeong SJ, Moon SJ, Cho JY, Yoo IK, Yon DK. Severe clinical outcomes of COVID-19 associated with proton pump inhibitors: a nationwide cohort study with propensity score matching. Gut. 2021;70:76-84. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 163] [Cited by in RCA: 151] [Article Influence: 37.8] [Reference Citation Analysis (0)] |
| 52. | Ullah AZMD, Sivapalan L, Chelala C, Kocher HM. COVID-19 in patients with hepatobiliary and pancreatic diseases in East London: a single-centre cohort study. 2020 Preprint. Available from: medRxiv. [DOI] [Full Text] |
| 53. | Hariyanto TI, Prasetya IB, Kurniawan A. Proton pump inhibitor use is associated with increased risk of severity and mortality from coronavirus disease 2019 (COVID-19) infection. Dig Liver Dis. 2020;52:1410-1412. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 33] [Cited by in RCA: 32] [Article Influence: 6.4] [Reference Citation Analysis (0)] |
| 54. | Aby ES, Rodin H, Debes JD. Proton Pump Inhibitors and Mortality in Individuals With COVID-19. Am J Gastroenterol. 2020;115:1918. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 10] [Cited by in RCA: 9] [Article Influence: 1.8] [Reference Citation Analysis (0)] |
| 55. | García-Menaya JM, Cordobés-Durán C, Rangel-Mayoral JF, García-Martín E, Agúndez JAG. Outcomes and Laboratory and Clinical Findings of Asthma and Allergic Patients Admitted With Covid-19 in a Spanish University Hospital. Front Pharmacol. 2020;11:570721. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 5] [Cited by in RCA: 3] [Article Influence: 0.6] [Reference Citation Analysis (0)] |
| 56. | Gadiparthi C, Perisetti A, Sayana H, Tharian B, Inamdar S, Korman A. Gastrointestinal Bleeding in Patients with Severe SARS-CoV-2. Am J Gastroenterol. 2020;115:1283-1285. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 42] [Cited by in RCA: 37] [Article Influence: 7.4] [Reference Citation Analysis (0)] |
| 57. | Li GF, An XX, Yu Y, Jiao LR, Canarutto D, Yu G, Wang G, Wu DN, Xiao Y. Do proton pump inhibitors influence SARS-CoV-2 related outcomes? Gut. 2020;. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 26] [Cited by in RCA: 31] [Article Influence: 7.8] [Reference Citation Analysis (0)] |
| 58. | Kwon JH, Koh SJ, Kim W, Jung YJ, Kim JW, Kim BG, Lee KL, Im JP, Kim YJ, Kim JS, Yoon JH, Lee HS, Jung HC. Mortality associated with proton pump inhibitors in cirrhotic patients with spontaneous bacterial peritonitis. J Gastroenterol Hepatol. 2014;29:775-781. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 44] [Cited by in RCA: 48] [Article Influence: 4.4] [Reference Citation Analysis (0)] |
| 59. | Hegarty JP, Sangster W, Harris LR 3rd, Stewart DB. Proton pump inhibitors induce changes in colonocyte gene expression that may affect Clostridium difficile infection. Surgery. 2014;156:972-978. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 24] [Cited by in RCA: 25] [Article Influence: 2.3] [Reference Citation Analysis (0)] |
| 60. | Taştemur Ş, Ataseven H. Is it possible to use Proton Pump Inhibitors in COVID-19 treatment and prophylaxis? Med Hypotheses. 2020;143:110018. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 19] [Cited by in RCA: 17] [Article Influence: 3.4] [Reference Citation Analysis (0)] |
| 61. | Ray A, Sharma S, Sadasivam B. The Potential Therapeutic Role of Proton Pump Inhibitors in COVID-19: Hypotheses Based on Existing Evidences. Drug Res (Stuttg). 2020;70:484-488. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 9] [Cited by in RCA: 6] [Article Influence: 1.2] [Reference Citation Analysis (0)] |
| 62. | Cao Y, Chen M, Tang D, Yan H, Ding X, Zhou F, Zhang M, Xu G, Zhang W, Zhang S, Zhuge Y, Wang L, Zou X. The proton pump inhibitor pantoprazole disrupts protein degradation systems and sensitizes cancer cells to death under various stresses. Cell Death Dis. 2018;9:604. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 18] [Cited by in RCA: 14] [Article Influence: 2.0] [Reference Citation Analysis (0)] |
| 63. | Udelnow A, Kreyes A, Ellinger S, Landfester K, Walther P, Klapperstueck T, Wohlrab J, Henne-Bruns D, Knippschild U, Würl P. Omeprazole inhibits proliferation and modulates autophagy in pancreatic cancer cells. PLoS One. 2011;6:e20143. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 83] [Cited by in RCA: 86] [Article Influence: 6.1] [Reference Citation Analysis (0)] |
| 64. | Ravikumar B, Sarkar S, Davies JE, Futter M, Garcia-Arencibia M, Green-Thompson ZW, Jimenez-Sanchez M, Korolchuk VI, Lichtenberg M, Luo S, Massey DC, Menzies FM, Moreau K, Narayanan U, Renna M, Siddiqi FH, Underwood BR, Winslow AR, Rubinsztein DC. Regulation of mammalian autophagy in physiology and pathophysiology. Physiol Rev. 2010;90:1383-1435. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 1461] [Cited by in RCA: 1369] [Article Influence: 91.3] [Reference Citation Analysis (0)] |
| 65. | Delorme-Axford E, Klionsky DJ. Highlights in the fight against COVID-19: does autophagy play a role in SARS-CoV-2 infection? Autophagy. 2020;16:2123-2127. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 22] [Cited by in RCA: 26] [Article Influence: 5.2] [Reference Citation Analysis (0)] |
| 66. | Berezhnov AV, Soutar MP, Fedotova EI, Frolova MS, Plun-Favreau H, Zinchenko VP, Abramov AY. Intracellular pH Modulates Autophagy and Mitophagy. J Biol Chem. 2016;291:8701-8708. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 65] [Cited by in RCA: 86] [Article Influence: 9.6] [Reference Citation Analysis (0)] |
| 67. | Rubinsztein DC, Codogno P, Levine B. Autophagy modulation as a potential therapeutic target for diverse diseases. Nat Rev Drug Discov. 2012;11:709-730. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 1195] [Cited by in RCA: 1192] [Article Influence: 91.7] [Reference Citation Analysis (0)] |
| 68. | Ghebre Y, Raghu G. Proton pump inhibitors in IPF: beyond mere suppression of gastric acidity. QJM. 2016;109:577-579. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 14] [Cited by in RCA: 21] [Article Influence: 2.3] [Reference Citation Analysis (0)] |
| 69. | Raghu G, Yang ST, Spada C, Hayes J, Pellegrini CA. Sole treatment of acid gastroesophageal reflux in idiopathic pulmonary fibrosis: a case series. Chest. 2006;129:794-800. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 157] [Cited by in RCA: 138] [Article Influence: 7.3] [Reference Citation Analysis (0)] |
| 70. | Lee JS, Ryu JH, Elicker BM, Lydell CP, Jones KD, Wolters PJ, King TE Jr, Collard HR. Gastroesophageal reflux therapy is associated with longer survival in patients with idiopathic pulmonary fibrosis. Am J Respir Crit Care Med. 2011;184:1390-1394. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 307] [Cited by in RCA: 302] [Article Influence: 21.6] [Reference Citation Analysis (1)] |
| 71. | Lee JS, Collard HR, Anstrom KJ, Martinez FJ, Noth I, Roberts RS, Yow E, Raghu G; IPFnet Investigators. Anti-acid treatment and disease progression in idiopathic pulmonary fibrosis: an analysis of data from three randomised controlled trials. Lancet Respir Med. 2013;1:369-376. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 288] [Cited by in RCA: 293] [Article Influence: 24.4] [Reference Citation Analysis (0)] |
| 72. | Ghebremariam YT, Cooke JP, Gerhart W, Griego C, Brower JB, Doyle-Eisele M, Moeller BC, Zhou Q, Ho L, de Andrade J, Raghu G, Peterson L, Rivera A, Rosen GD. Pleiotropic effect of the proton pump inhibitor esomeprazole leading to suppression of lung inflammation and fibrosis. J Transl Med. 2015;13:249. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 84] [Cited by in RCA: 100] [Article Influence: 10.0] [Reference Citation Analysis (0)] |
| 73. | Pagat AM, Seux-Goepfert R, Lutsch C, Lecouturier V, Saluzzo JF, Kusters IC. Evaluation of SARS-Coronavirus Decontamination Procedures. Appl Bio. 2007;12:100-108. [RCA] [DOI] [Full Text] [Cited by in Crossref: 25] [Cited by in RCA: 25] [Article Influence: 2.8] [Reference Citation Analysis (0)] |
| 74. | Belouzard S, Millet JK, Licitra BN, Whittaker GR. Mechanisms of coronavirus cell entry mediated by the viral spike protein. Viruses. 2012;4:1011-1033. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 824] [Cited by in RCA: 928] [Article Influence: 71.4] [Reference Citation Analysis (0)] |
| 75. | Sternberg A, Naujokat C. Structural features of coronavirus SARS-CoV-2 spike protein: Targets for vaccination. Life Sci. 2020;257:118056. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 174] [Cited by in RCA: 156] [Article Influence: 31.2] [Reference Citation Analysis (0)] |
| 76. | Hoffmann M, Kleine-Weber H, Schroeder S, Krüger N, Herrler T, Erichsen S, Schiergens TS, Herrler G, Wu NH, Nitsche A, Müller MA, Drosten C, Pöhlmann S. SARS-CoV-2 Cell Entry Depends on ACE2 and TMPRSS2 and Is Blocked by a Clinically Proven Protease Inhibitor. Cell 2020; 181: 271-280. e8. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 11946] [Cited by in RCA: 14219] [Article Influence: 2843.8] [Reference Citation Analysis (0)] |
| 77. | Glebov OO. Understanding SARS-CoV-2 endocytosis for COVID-19 drug repurposing. FEBS J. 2020;287:3664-3671. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 107] [Cited by in RCA: 129] [Article Influence: 25.8] [Reference Citation Analysis (0)] |
| 78. | Yang N, Shen HM. Targeting the Endocytic Pathway and Autophagy Process as a Novel Therapeutic Strategy in COVID-19. Int J Biol Sci. 2020;16:1724-1731. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 286] [Cited by in RCA: 318] [Article Influence: 63.6] [Reference Citation Analysis (0)] |
| 79. | Homolak J, Kodvanj I. Widely available lysosome targeting agents should be considered as potential therapy for COVID-19. Int J Antimicrob Agents. 2020;56:106044. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 34] [Cited by in RCA: 53] [Article Influence: 10.6] [Reference Citation Analysis (0)] |
| 80. | Fredericksen BL, Wei BL, Yao J, Luo T, Garcia JV. Inhibition of endosomal/lysosomal degradation increases the infectivity of human immunodeficiency virus. J Virol. 2002;76:11440-11446. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 159] [Cited by in RCA: 170] [Article Influence: 7.4] [Reference Citation Analysis (0)] |
| 81. | Bayati A, Kumar R, Francis V, McPherson PS. SARS-CoV-2 uses clathrin-mediated endocytosis to gain access into cells. 2020 Preprint. Available from: bioRxiv. [DOI] [Full Text] |
| 82. | Hu TY, Frieman M, Wolfram J. Insights from nanomedicine into chloroquine efficacy against COVID-19. Nat Nanotechnol. 2020;15:247-249. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 232] [Cited by in RCA: 206] [Article Influence: 41.2] [Reference Citation Analysis (0)] |
| 83. | Miller SE, Mathiasen S, Bright NA, Pierre F, Kelly BT, Kladt N, Schauss A, Merrifield CJ, Stamou D, Höning S, Owen DJ. CALM regulates clathrin-coated vesicle size and maturation by directly sensing and driving membrane curvature. Dev Cell. 2015;33:163-175. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 157] [Cited by in RCA: 157] [Article Influence: 15.7] [Reference Citation Analysis (0)] |
| 84. | Moggia E, Koti R, Belgaumkar AP, Fazio F, Pereira SP, Davidson BR, Gurusamy KS. Pharmacological interventions for acute pancreatitis. Cochrane Database Syst Rev. 2017;4:CD011384. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 50] [Cited by in RCA: 53] [Article Influence: 6.6] [Reference Citation Analysis (0)] |
| 85. | Kapadia BH, Torre BB, Ullman N, Yang A, Harb MA, Grieco PW, Newman JM, Harwin SF, Maheshwari AV. Reducing perioperative blood loss with antifibrinolytics and antifibrinolytic-like agents for patients undergoing total hip and total knee arthroplasty. J Orthop. 2019;16:513-516. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 12] [Cited by in RCA: 5] [Article Influence: 0.8] [Reference Citation Analysis (0)] |
| 86. | Zhirnov OP, Klenk HD, Wright PF. Aprotinin and similar protease inhibitors as drugs against influenza. Antiviral Res. 2011;92:27-36. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 88] [Cited by in RCA: 92] [Article Influence: 6.6] [Reference Citation Analysis (0)] |
| 87. | Shen LW, Mao HJ, Wu YL, Tanaka Y, Zhang W. TMPRSS2: A potential target for treatment of influenza virus and coronavirus infections. Biochimie. 2017;142:1-10. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 209] [Cited by in RCA: 209] [Article Influence: 26.1] [Reference Citation Analysis (0)] |
| 88. | Bojkova D, McGreig JE, McLaughlin KM, Masterson SG, Widera M, Krähling V, Ciesek S, Wass MN, Michaelis M, Cinatl jr J. SARS-CoV-2 and SARS-CoV differ in their cell tropism and drug sensitivity profiles. 2020 Preprint. Available from: bioRxiv. [DOI] [Full Text] |
| 89. | Aguila EJT, Cua IHY. Repurposed GI Drugs in the Treatment of COVID-19. Dig Dis Sci. 2020;65:2452-2453. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 16] [Cited by in RCA: 17] [Article Influence: 3.4] [Reference Citation Analysis (0)] |
| 90. | Hu YB, Dammer EB, Ren RJ, Wang G. The endosomal-lysosomal system: from acidification and cargo sorting to neurodegeneration. Transl Neurodegener. 2015;4:18. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 287] [Cited by in RCA: 444] [Article Influence: 44.4] [Reference Citation Analysis (0)] |
| 91. | Futai M, Sun-Wada GH, Wada Y, Matsumoto N, Nakanishi-Matsui M. Vacuolar-type ATPase: A proton pump to lysosomal trafficking. Proc Jpn Acad Ser B Phys Biol Sci. 2019;95:261-277. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 56] [Cited by in RCA: 73] [Article Influence: 12.2] [Reference Citation Analysis (0)] |
| 92. | De Milito A, Iessi E, Logozzi M, Lozupone F, Spada M, Marino ML, Federici C, Perdicchio M, Matarrese P, Lugini L, Nilsson A, Fais S. Proton pump inhibitors induce apoptosis of human B-cell tumors through a caspase-independent mechanism involving reactive oxygen species. Cancer Res. 2007;67:5408-5417. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 218] [Cited by in RCA: 232] [Article Influence: 12.9] [Reference Citation Analysis (0)] |
| 93. | Marshansky V, Futai M. The V-type H+-ATPase in vesicular trafficking: targeting, regulation and function. Curr Opin Cell Biol. 2008;20:415-426. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 368] [Cited by in RCA: 385] [Article Influence: 22.6] [Reference Citation Analysis (0)] |
| 94. | Sachs G, Prinz C, Loo D, Bamberg K, Besancon M, Shin JM. Gastric acid secretion: activation and inhibition. Yale J Biol Med. 1994;67:81-95. [PubMed] |
| 95. | Sabolić I, Brown D, Verbavatz JM, Kleinman J. H(+)-ATPases of renal cortical and medullary endosomes are differentially sensitive to Sch-28080 and omeprazole. Am J Physiol. 1994;266:F868-F877. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 19] [Cited by in RCA: 24] [Article Influence: 0.8] [Reference Citation Analysis (0)] |
| 96. | Brecher M, Schornberg KL, Delos SE, Fusco ML, Saphire EO, White JM. Cathepsin cleavage potentiates the Ebola virus glycoprotein to undergo a subsequent fusion-relevant conformational change. J Virol. 2012;86:364-372. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 117] [Cited by in RCA: 129] [Article Influence: 9.2] [Reference Citation Analysis (0)] |
| 97. | Ikemura K, Hiramatsu S, Okuda M. Drug Repositioning of Proton Pump Inhibitors for Enhanced Efficacy and Safety of Cancer Chemotherapy. Front Pharmacol. 2017;8:911. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 40] [Cited by in RCA: 51] [Article Influence: 6.4] [Reference Citation Analysis (0)] |
| 98. | Pahan P, Pahan K. Smooth or Risky Revisit of an Old Malaria Drug for COVID-19? J Neuroimmune Pharmacol. 2020;15:174-180. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 29] [Cited by in RCA: 36] [Article Influence: 7.2] [Reference Citation Analysis (0)] |
| 99. | Pamarthy S, Kulshrestha A, Katara GK, Beaman KD. The curious case of vacuolar ATPase: regulation of signaling pathways. Mol Cancer. 2018;17:41. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 115] [Cited by in RCA: 131] [Article Influence: 18.7] [Reference Citation Analysis (0)] |