Published online Nov 26, 2020. doi: 10.12998/wjcc.v8.i22.5657
Peer-review started: June 3, 2020
First decision: September 13, 2020
Revised: September 22, 2020
Accepted: October 13, 2020
Article in press: October 13, 2020
Published online: November 26, 2020
Processing time: 175 Days and 9.6 Hours
In myelodysplastic syndrome (MDS), oxidative stress is closely related to iron overload and DNA damage. A recent study suggested the possibility that increased oxidative stress causes not only iron overload but also disease progression of MDS with DNA damage. We present a case of MDS with decreased reactive oxygen species (ROS) production in peripheral white blood cells (WBCs) and decreased diacron-reactive oxygen metabolites (d-ROMs) in serum after azacitidine therapy.
A 74-year-old man presented to the hematological department with the chief complaint of anemia. His vital signs were within normal limits at admission with a heart rate of 80 bpm and blood pressure of 135/60 mmHg. Laboratory tests indicated pancytopenia, a WBC count of 2190 cells/µL, a hemoglobin level of 6.2 g/dL and a platelet count of 7.4 × 104/µL. The patient was diagnosed with MDS with fibrosis after a bone marrow examination. This case showed decreased ROS production in WBCs, d-ROMs in serum and Wilms’ tumor 1 after azacitidine therapy, after which his hematopoiesis recovered.
Azacitidine therapy can improve hematopoiesis and decrease ROS and d-ROM production.
Core Tip: Accumulation of reactive oxygen species (ROS) contributes to the development and progression of cancer. We reported a case of myelodysplastic syndrome that showed decreased ROS production by white blood cells, decreased diacron-reactive oxygen metabolites (d-ROMs) in serum and decreased Wilms’ tumor 1 after azacitidine therapy, which can improve hematopoiesis by decreasing ROS and d-ROMs production.
- Citation: Hasunuma H, Shimizu N, Yokota H, Tatsuno I. Azacitidine decreases reactive oxygen species production in peripheral white blood cells: A case report. World J Clin Cases 2020; 8(22): 5657-5662
- URL: https://www.wjgnet.com/2307-8960/full/v8/i22/5657.htm
- DOI: https://dx.doi.org/10.12998/wjcc.v8.i22.5657
DNA damage caused by oxidative stress plays an important role in the initiation and carcinogenic process of mutation. A recent study suggested that oxidative stress leads to increased mutation frequency in a murine model of myelodysplastic syndrome (MDS)[1,2] and that additional mutations lead to MDS progression. Patients with MDS often require blood transfusion and consequently develop iron overload, which causes oxidative stress. Antioxidant therapy and iron chelation are therefore considered suitable for managing patients with MDS[3,4]. A correlation has also been reported between serum ferritin levels and serum reactive oxygen species (ROS) levels in patients with MDS patients[4]. MDS is a heterogeneous group of clonal hematopoietic stem cell disorders that transform into acute myeloid leukemia. DNA hypermethylation is one of the most important pathogenic mechanisms of MDS[1]. Azacitidine is the first DNA hypomethylating agent that has demonstrated superiority over conventional therapy in treating MDS in patients unable to undergo allogeneic stem cell transplantation. A relationship between oxidative stress and hypermethylation has also been reported; oxidative stress is likely to be involved in MDS disease extension[1]. We report on a patient with MDS and fibrosis, who presented with decreased ROS in the peripheral white blood cells (WBCs), decreased diacron-reactive oxygen metabolites (d-ROMs) in serum and decreased Wilms’ tumor 1 (WT1). The patient was withdrawn from transfusion dependence after azacitidine therapy. The Ethics Committee of Toho University approved this study, and we obtained the patient’s informed consent.
A 74-year-old man presented with dyspnea on exertion that started two months earlier.
The patient presented anemia and a hemoglobin level of 6.4 g/dL during an ophthalmology consultation for cataract treatment. The patient presented dyspnea on exertion, a symptom that had aggravated over the past two months. The patient visited the hematological department.
The patient had a past medical history of cataract and ossification of a posterior longitudinal ligament.
The patient had been a smoker for 50 years.
Vital signs were within normal limits at presentation, with a heart rate of 80 bpm, blood pressure of 135/60 mmHg, and a temperature of 36.1 ℃. His height and weight were 171 cm and 75 kg, respectively.
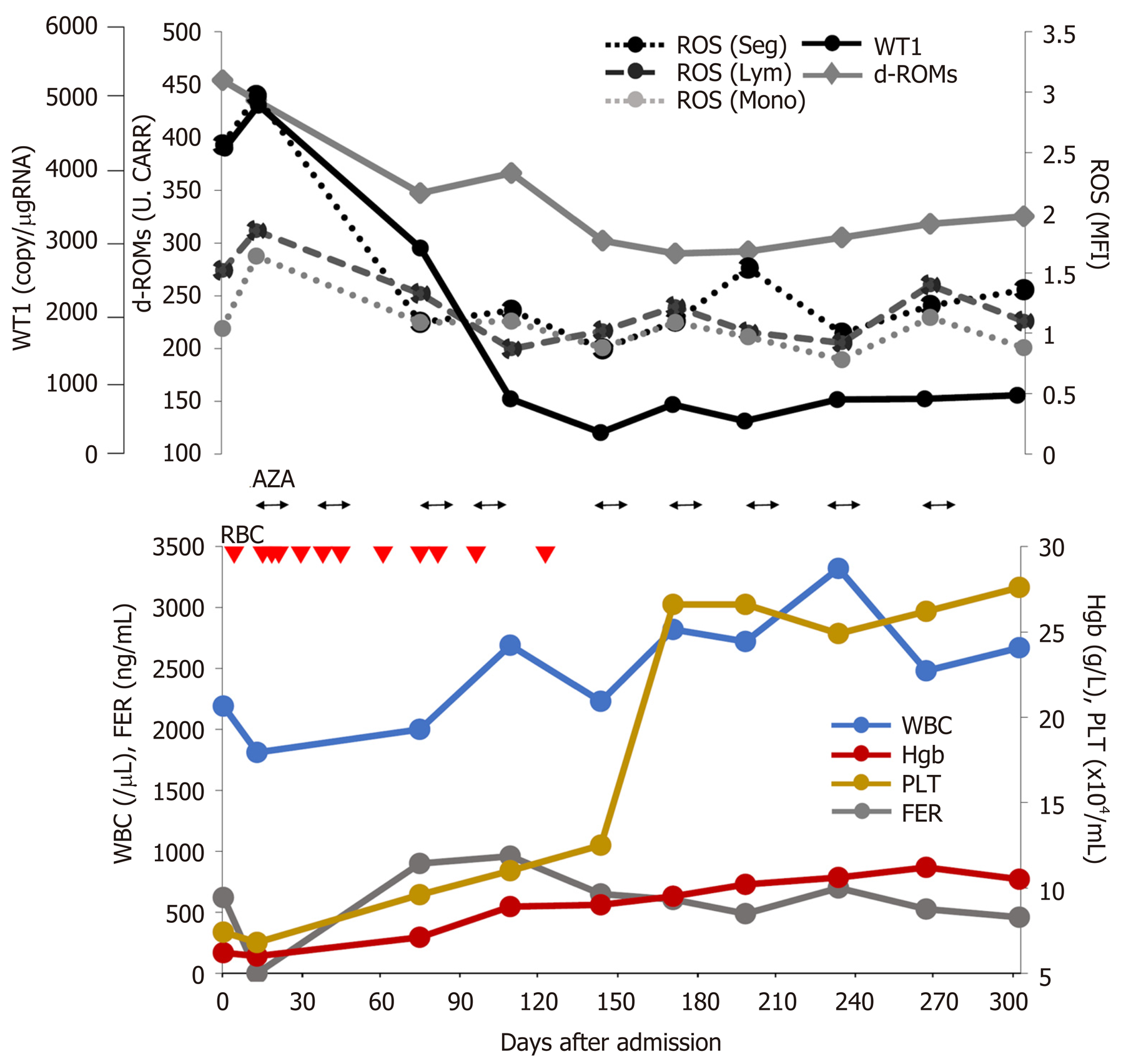
Laboratory tests indicated a WBC count of 2190 cells/µL, a hemoglobin level of 6.2 g/dL, a platelet count of 7.4 × 104/µL, a lactate dehydrogenase level of 283 IU/L, and a ferritin level of 620.41 ng/mL. The WT1 mRNA was 4800/µg RNA of peripheral WBCs, and the serum d-ROM level was 435 Carratelli units. The bone marrow aspiration was dry tap, and the bone marrow biopsy showed MDS with fibrosis.
The patient was diagnosed with MDS with fibrosis.
The patient was administered 75 mg/m2 of azacitidine for 7 d once a month, underwent iron chelation therapy and took deferoxamine during the azacitidine therapy.
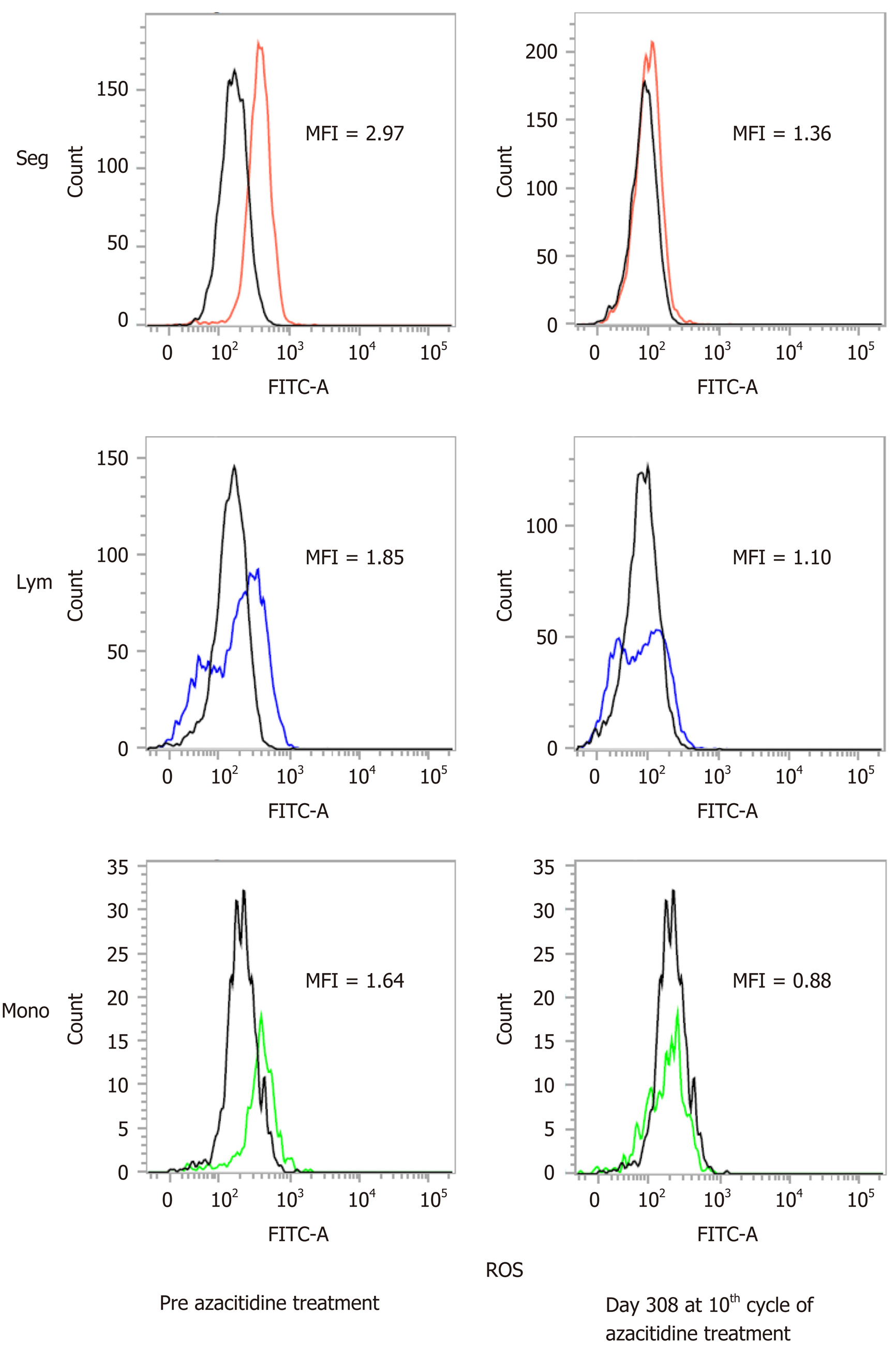
To date, the patient has undergone 11 cycles of azacitidine therapy. After the fourth cycle, he was released from transfusion. Figure 1 shows the changes in hematopoiesis during the azacitidine therapy. The patient presented with decreased ROS production in the peripheral WBCs, decreased d-ROMs in serum and decreased WT1 after azacitidine therapy. Figure 2 shows the decrease in ROS production in the WBCs after the ninth cycle of azacitidine therapy. The black line shows ROS production in a healthy control, while the colored line shows the patient’s production.
Gonçalves et al[5] reported the relationship between ROS and other mitochondrial dysfunction markers in patients with MDS. The levels of ROS stained with DCFH2-DA showed increases in each fraction of WBCs in patients with MDS, as was the case in our experiment. Moreover, patients with MDS and high ROS showed lower overall survival. The authors also analyzed the variants in genes involved in oxidative stress in patients with MDS and acute myeloid leukemia and showed that a number of superoxide dismutase 2 and glutathione peroxidase 1 genotypes increased the susceptibility for developing MDS with increased ROS production[6].
We have previously reported that the other oxidative stress markers (serum d-ROMs) transiently decreased after each azacitidine therapy. Furthermore, these oxidative stress markers strongly correlated with WT1 levels[7]. This case also suggests the mechanisms by which azacitidine prevents MDS disease progression by inhibiting oxidative stress.
Kornicka et al[8] reported that azacitidine showed reduced ROS accumulation in vitro. In the present case, we also evaluated ROS production in WBCs, in addition to serum d-ROM, ferritin and WT1 production during azacitidine therapy. Our patient was released from blood transfusion with a decrease in WT1, serum d-ROMs and ROS production in the WBCs after azacitidine therapy (Figure 1 and 2). As shown in Figure 1, there was no relationship with ferritin and oxidative stress markers. We therefore speculate that oxidative stress is related to the MDS state without iron overload. We also analyzed other patients with MDS undergoing azacitidine therapy. The patients in the responder group showed decreased ROS production in WBCs; however, the nonresponder group showed increased ROS and WT1 production (unpublished data). We therefore consider that ROS production the most suitable factor for evaluating MDS progression.
This is the first report indicating that azacitidine decreases ROS production in WBCs and decreases serum d-ROM levels in a patient with MDS. ROS, d-ROM and WT1 levels showed the same behavior during azacitidine therapy, and these parameters appear to have no relationship with ferritin levels. We speculate the novel mechanisms regarding the suppression of oxidative stress by the DNA hypomethylating agent azacitidine. Further study is therefore needed to clarify this point.
Manuscript source: Unsolicited manuscript
Specialty type: Oncology
Country/Territory of origin: Japan
Peer-review report’s scientific quality classification
Grade A (Excellent): A
Grade B (Very good): 0
Grade C (Good): 0
Grade D (Fair): 0
Grade E (Poor): 0
P-Reviewer: Bourgoin SG S-Editor: Zhang H L-Editor: A P-Editor: Zhang YL
| 1. | Chung YJ, Robert C, Gough SM, Rassool FV, Aplan PD. Oxidative stress leads to increased mutation frequency in a murine model of myelodysplastic syndrome. Leuk Res. 2014;38:95-102. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 35] [Cited by in RCA: 41] [Article Influence: 3.4] [Reference Citation Analysis (0)] |
| 2. | Porto ML, Rodrigues BP, Menezes TN, Ceschim SL, Casarini DE, Gava AL, Pereira TM, Vasquez EC, Campagnaro BP, Meyrelles SS. Reactive oxygen species contribute to dysfunction of bone marrow hematopoietic stem cells in aged C57BL/6 J mice. J Biomed Sci. 2015;22:97. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 45] [Cited by in RCA: 61] [Article Influence: 6.1] [Reference Citation Analysis (0)] |
| 3. | Ghoti H, Amer J, Winder A, Rachmilewitz E, Fibach E. Oxidative stress in red blood cells, platelets and polymorphonuclear leukocytes from patients with myelodysplastic syndrome. Eur J Haematol. 2007;79:463-467. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 77] [Cited by in RCA: 85] [Article Influence: 4.7] [Reference Citation Analysis (0)] |
| 4. | Kikuchi S, Kobune M, Iyama S, Sato T, Murase K, Kawano Y, Takada K, Ono K, Kaneko Y, Miyanishi K, Sato Y, Hayashi T, Takimoto R, Kato J. Improvement of iron-mediated oxidative DNA damage in patients with transfusion-dependent myelodysplastic syndrome by treatment with deferasirox. Free Radic Biol Med. 2012;53:643-648. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 46] [Cited by in RCA: 49] [Article Influence: 3.8] [Reference Citation Analysis (0)] |
| 5. | Gonçalves AC, Cortesão E, Oliveiros B, Alves V, Espadana AI, Rito L, Magalhães E, Lobão MJ, Pereira A, Nascimento Costa JM, Mota-Vieira L, Sarmento-Ribeiro AB. Oxidative stress and mitochondrial dysfunction play a role in myelodysplastic syndrome development, diagnosis, and prognosis: A pilot study. Free Radic Res. 2015;49:1081-1094. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 31] [Cited by in RCA: 41] [Article Influence: 4.1] [Reference Citation Analysis (0)] |
| 6. | Gonçalves AC, Alves R, Baldeiras I, Cortesão E, Carda JP, Branco CC, Oliveiros B, Loureiro L, Pereira A, Nascimento Costa JM, Sarmento-Ribeiro AB, Mota-Vieira L. Genetic variants involved in oxidative stress, base excision repair, DNA methylation, and folate metabolism pathways influence myeloid neoplasias susceptibility and prognosis. Mol Carcinog. 2017;56:130-148. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 10] [Cited by in RCA: 17] [Article Influence: 1.9] [Reference Citation Analysis (1)] |
| 7. | Shimizu N, Hasunuma H, Watanabe Y, Matsuzawa Y, Iwashita Y, Tatsuno I, Yokota H. The Simultaneous Elevation of Oxidative Stress Markers and Wilms' Tumor 1 Gene during the Progression of Myelodysplastic Syndrome. Intern Med. 2016;55:3661-3664. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 5] [Cited by in RCA: 5] [Article Influence: 0.6] [Reference Citation Analysis (0)] |
| 8. | Kornicka K, Marycz K, Marędziak M, Tomaszewski KA, Nicpoń J. The effects of the DNA methyltranfserases inhibitor 5-Azacitidine on ageing, oxidative stress and DNA methylation of adipose derived stem cells. J Cell Mol Med. 2017;21:387-401. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 65] [Cited by in RCA: 79] [Article Influence: 8.8] [Reference Citation Analysis (0)] |