Published online Sep 26, 2020. doi: 10.12998/wjcc.v8.i18.4169
Peer-review started: May 29, 2020
First decision: June 13, 2020
Revised: June 23, 2020
Accepted: August 22, 2020
Article in press: August 22, 2020
Published online: September 26, 2020
Processing time: 115 Days and 12.4 Hours
Insulinomas are the most common type of functioning endocrine neoplasms of the pancreas presenting hypoglycemic symptoms. Patients characteristically develop symptoms while fasting, but some patients have reported symptoms only in the postprandial state. Repeated and prolonged hypoglycemic episodes can reduce the awareness of adrenergic symptoms, and patients may have amnesia, which delays diagnosis.
We describe a case of a 24-year-old underweight patient who showed hypoglycemic symptoms for almost 6 years. Although patients with insulinoma characteristically develop symptoms while fasting, this young man had hypoglycemic symptoms up to one hour postprandially, especially after high-sugar meals and after physical activity. The fasting tests and imaging methods performed at local hospitals were evaluated as negative for abnormal results. However, brown adipose tissue exhibited increased metabolic activity, and some muscle groups had histological changes as indicated by positron emission tomography with 2-deoxy-2-[fluorine-18]fluoro-D-glucose integrated with computed tomography. Glycogen deficiency was also histologically confirmed. The patient’s symptoms progressed over the years and occurred more frequently, i.e., several times a month, and the patient had reduced awareness of adrenergic symptoms. The follow-up fasting test was positive, and the imaging results showed a tumor in the head of the pancreas. The patient underwent laparotomy with enucleation of the insulinoma.
Weight gain and fasting hypoglycemia are not necessarily characteristics of insulinoma. In prolonged cases, adrenergic symptoms can be suppressed.
Core Tip: Postprandial hypoglycemia and hypoglycemia following physical exercise associated with unconsciousness/amnesia and a low body weight were the clinical features of insulinoma in a 24-year-old man for over 6 years. The consequences of the chronic counterregulatory adrenergic response were also indirectly indicated by the increased metabolic activity of brown adipose tissue and decrease in muscle glycogen content, although the fasting test and other imaging results were initially evaluated as normal. Therefore, if insulinoma is clinically suspected, the fasting test should be repeated. The patient was underweight with a very small amount of visceral fat, which increases the risk of perioperative complications.
- Citation: Prídavková D, Samoš M, Kyčina R, Adamicová K, Kalman M, Belicová M, Mokáň M. Insulinoma presenting with postprandial hypoglycemia and a low body mass index: A case report. World J Clin Cases 2020; 8(18): 4169-4176
- URL: https://www.wjgnet.com/2307-8960/full/v8/i18/4169.htm
- DOI: https://dx.doi.org/10.12998/wjcc.v8.i18.4169
Insulinomas are characterized clinically by hypoglycemic symptoms resulting from neuroglycopenia and the catecholamine response[1] and are the most common functioning endocrine neoplasms of the pancreas, accounting for 20.9% of all pancreatic endocrine tumors[2]. They arise from pancreatic beta cells and are the most common cause of endogenous hyperinsulinemic hypoglycemia. The incidence is 1-4 cases per million persons per year[3]. Most patients diagnosed with insulinoma are aged between 30 and 60 years and are predominantly female[4,5]. Insulinomas usually present as small, well-demarcated, solitary nodules that may arise in any part of the gland[1,6,7]. Of diagnosed insulinomas, approximately 10% involve multiple tumors, less than 10% are malignant, and 5%-10% are associated with multiple endocrine neoplasia syndrome-1[7-10].
The classical diagnosis of insulinoma depends on satisfying the following criteria of Whipple’s triad, which remain the cornerstone of the screening process: (1) Hypoglycemia; (2) Neuroglycopenic symptoms; and (3) Prompt relief of symptoms following the administration of glucose[3]. In a consensus report by the United States Endocrine Society[12], the following diagnostic criteria were proposed: Endogenous hyperinsulinism documented by the findings of symptoms, signs, or both with plasma concentrations of glucose Ë‚ 3.0 mmol/L (55 mg/dL), insulin ≥ 3.0 µIU/mL (18 pmol/L), C-peptide ≥ 0.6 ng/mL (0.2 nmol/L), and proinsulin ≥ 5.0 pmol/L.
Patients with insulinoma characteristically develop symptoms while fasting (73%-80%)[2,15], but 6% of patients report symptoms only in the postprandial state, and 21% of patients report symptoms in both the postprandial and fasting states[15]. Although fasting hypoglycemia has been considered the main trait of insulinoma, postprandial hypoglycemia has also been occasionally reported as the predominant feature[16-18]. Weight gain is found in only 25%-42% of patients[10,19,20], and monthly changes in body weight are significantly correlated with the tumor size and serum insulin concentration[20]. Weight gain in insulinoma can be attributed to overeating to treat the hypoglycemia symptoms[21]. Furthermore, voluntary weight loss induced by lowering insulin resistance accelerates the time to the clinical presentation of an asymptomatic insulinoma[22], and malnutrition, body fat and muscle depletion can contribute to hypoglycemia due to the limited substrates available for gluconeogenesis and gly-cogenolysis.
A 24-year-old man was admitted in September 2019 for a preoperative evaluation before surgery for a recently diagnosed symptomatic insulinoma.
The patient’s first episode of hypoglycemia occurred 6 years prior and presented as weakness, sweating and tremor. The symptoms occurred up to one hour postprandially, especially after eating high-sugar meals and after performing physical activity. In addition to the adrenergic hypoglycemic symptoms, the patient also reported the taste of acetone in the mouth. These difficulties progressed over the years, occurred more frequently up to several times a month and involved other neuroglycopenic symptoms, including multiple episodes of loss of consciousness associated with amnesia.
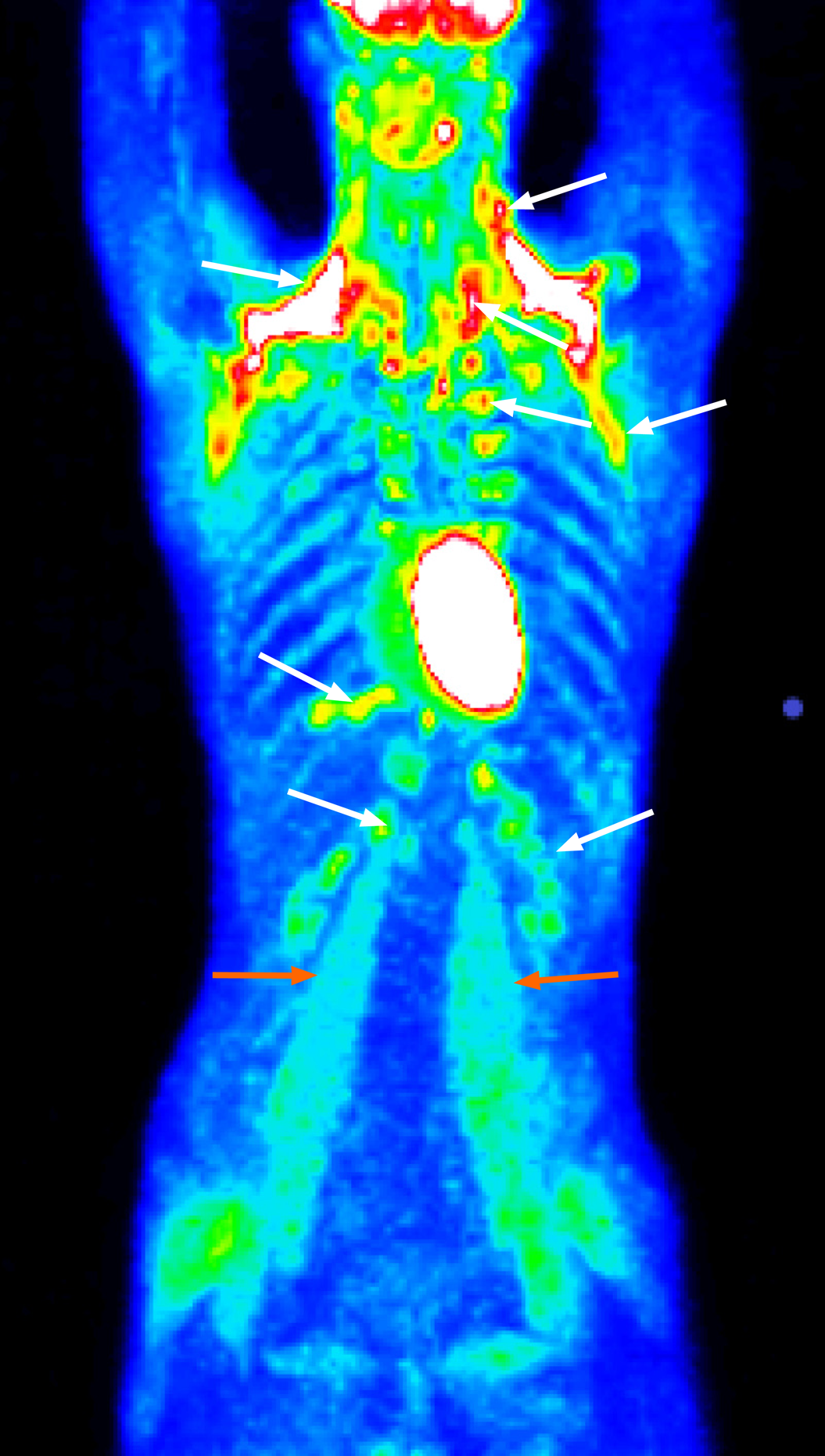
In 2015, emergency medical services confirmed a blood glucose level of 2.0 mmol/L, and the patient was referred to a local hospital to undergo a 72-h fasting test. The test was classified as negative, and further examinations were conducted. Abdominal ultrasonography and multidetector computed tomography (CT) of the chest and abdomen were negative for abnormal findings. CT examination integrated with 2-deoxy-2-[fluorine-18]fluoro-D-glucose did not confirm the presence of tumors but showed that there was enhanced metabolic activity of brown adipose tissue (BAT) bilaterally in the neck, paravertebral, supra- and infraclavicular, axillar, mediastinal, jugular, diaphragmatic and perinephric regions. Increased activity was also found in several muscle groups (e.g., psoas major muscles) (Figure 1). This finding was attributed to a reactive stress reaction. The presence of autoimmune primary muscle disease was excluded, but a muscle biopsy showed focal glycogen deficits. The episodes of hypoglycemia occurred repeatedly, but the patient became less aware of the decreasing intensity of the adrenergic symptoms. A second fasting test (in August 2019) was performed with a resulting decrease in glycemia to 1.9 mmol/L (34 mg/dL) in the 29th h of the test and an increase in insulin to 5.45 µIU/L (normal range: 3.0-25 µIU/L). C-peptide was 0.42 nmol/L (normal range: 0.25-0.60 nmol/L).
The patient had no specific history of past illness.
The patient had no specific personal or family history.
The patient’s weight was 52 kg, and his body mass index was 17.0 kg/m2. His skin, skin adnexa and mucosa showed no pathological changes. The physical examination showed no obvious cardiovascular or respiratory abnormalities. The abdomen was soft without pathological resistance.
Upon hospitalization, the blood glucose level was 4.2 mmol/L, glycated hemoglobin-IFCC was 3.1% (normal range: 2.8%-4.8%); the values of C-peptide and insulin, lipid, mineral, protein, acid-base balance, renal and hepatic parameters were in the normal range. The diurnal rhythm of cortisol, function of the thyroid, levels of human growth hormone, adrenocorticotropic hormone, metanephrines, normetanephrines and values of oncomarkers were normal.
Endoscopic ultrasonography of the pancreas revealed a well-defined oval lesion (14.7 mm × 13.1 mm) in the head of the pancreas. The lesion was hypervascular with a higher echogenicity and a hyperechoic periphery. A CT scan of the pancreas imaged the homogenous lesion (10.5 mm × 8.0 mm) during the arterial phase of the examination.
The patient was finally diagnosed with insulinoma.
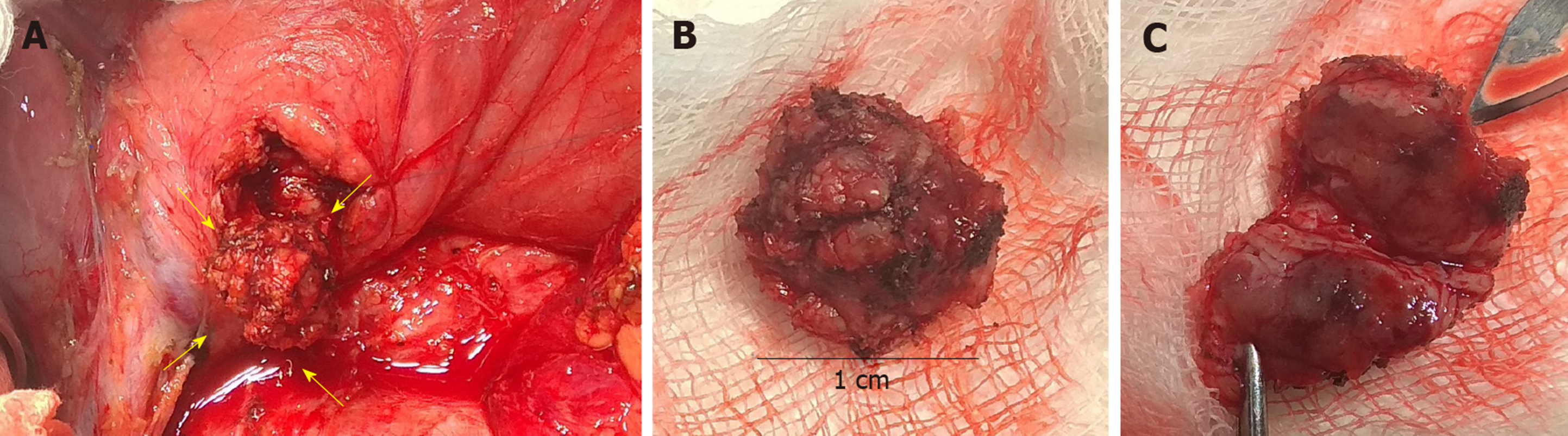
The patient underwent a laparotomy with enucleation of insulinoma (sized 1.0 cm) (Figure 2) and cholecystectomy. The perioperative findings noted a very small amount of visceral fat. The insulinoma was pressing against the common bile duct without any infiltration. Because possible lesion-involvement or sealing of the pancreatic duct by a harmonic scalpel (performed perioperatively during pancreatography, which showed a course of pancreatic duct only 2 cm from the papilla) could not be ruled out, Roux-en-Y anastomosis and nutritive lateral jejunojejunostomy was performed. Significant bleeding occurred from the base of the enucleated tumor, and the patient underwent a second surgical intervention.
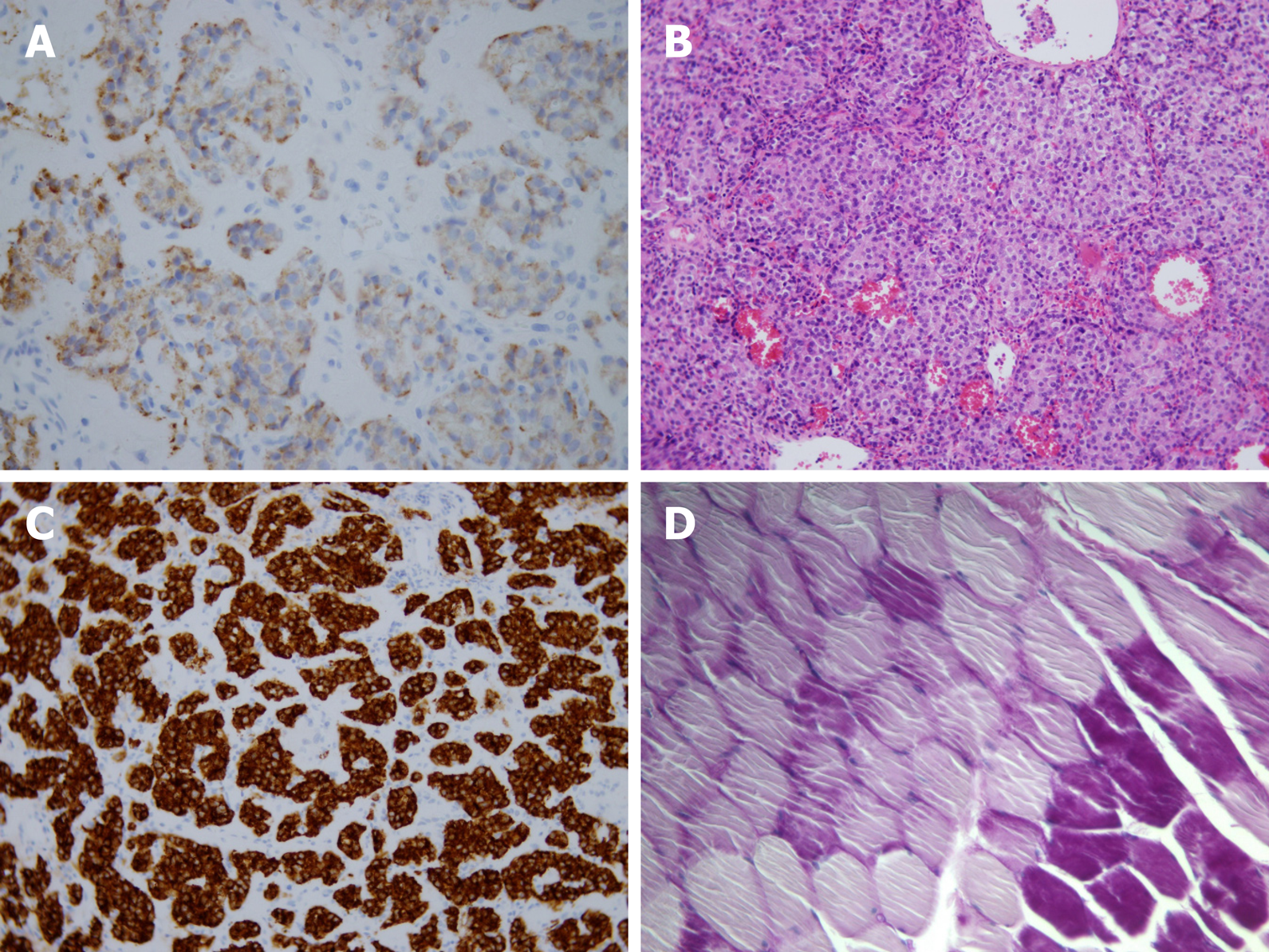
The final histological findings (Figure 3) revealed a well-differentiated insulinoma G1 (WHO 2017) (33), staging pT1pNxL0, V0, Pn0. The immunohistochemical studies of the lesion confirmed positivity for the neuroendocrine markers synaptophysin and chromogranin A, but the latter was less pronounced. The immunohistochemical studies found that the lesion sections were also positive for insulin, CK8/18 and CD56; the staining for glucagon and vimentin were negative. Lymphatic, vascular and perineural invasion was not confirmed, and the proliferative activity (Ki67) was 2.4%. The borders of the tumor were without neoplastic infiltration. The patient was released with normal levels of plasmatic glycemia. During the 6-mo follow-up period, the patient remained asymptomatic.
Delay in the diagnosis of insulinoma is common because the symptoms largely precede the occurrence of an apparent tumor, and there may be misattribution of the symptoms to psychiatric, cardiac or neurological disorders[23]. Several studies confirmed that 20%–64% of patients are misdiagnosed with a psychiatric, seizure or other neurological disorder before the final diagnosis of insulinoma is made[24,25]. The interval between the onset of symptoms and the diagnosis of insulinoma ranges from 1 mo to 30 years, with a median of 24 mo; only 28% of patients are diagnosed within 1 year of symptom onset, and the diagnosis is delayed beyond 5 years in 19% of patients[19]. The patient presented here also initially underwent psychological and psychiatric examinations with a suspicion of eating disorders with the re-commendation for psychotherapeutic guidance. The clinical manifestations of hypoglycemia, especially the adrenergic symptoms, are relatively well recognizable. However, patients with long-term illness may develop insufficient awareness of hypoglycemia with the risk of developing neuroglycopenic manifestations without awareness of adrenergic symptomatology[2], and these manifestations also occurred in our patient (episodes of short unconsciousness with amnesia). This phenomenon occurs because the set point of catecholamine secretion in response to hypoglycemia is lowered[26]. The prolonged time to the diagnosis of insulinoma in this case was likely also due to a different interpretation of the results of the fasting test performed at the local hospital. Furthermore, it appears that 9% of patients with insulinomas could be overlooked using the older recommended value of insulin ≥ 5 µIU/mL[11]. A prolonged fasting test (72 h) may not be diagnostic for 1% of insulinomas[27], which was likely the case in our patient (at the beginning of the diagnostic process).
Classically, insulinomas present with weight gain and fasting hypoglycemia but may also present with postprandial hypoglycemia and weight loss[22]. The patient did not have weight gain, which is noted in approximately 25%-42% of insulinoma cases[10,19,20]. Voluntary weight loss accelerates the time to clinical presentation of an asymptomatic insulinoma by lowering insulin resistance[22]. However, the patient’s diagnosis had long been stagnant, and the patient was assessed in the context of intrapsychic tension and eating disorders.
An increase in 2-deoxy-2-[fluorine-18]fluoro-D-glucose metabolic activity in BAT diffusely and muscle groups to this extent as determined using positron emission tomography/CT examinations is not common, and this phenomenon cannot be explained solely by a reactive stress response. Other factors are involved in the increased activation of BAT, such as a younger age, BAT activation in a previous scan and a lower body mass index[28]. BAT is normally present in fetuses and diminishes in adults, accounting for only 1% of the total body mass[29]. BAT is richly innervated by sympathetic nervous system efferent fibers, and sympathetic activation is the physiological activator of BAT thermogenesis. BAT is profoundly influenced by insulin sensitivity. The presence of metabolic activity of 2-deoxy-2-[fluorine-18]fluoro-D-glucose-positive BAT is associated with increased plasma catecholamines and is inversely related to central obesity. For example, BAT activation has been observed in cancer cachexia[30]. Similarly, our patient’s low body weight and small amount of visceral fat could be associated with increased BAT activity. Although the increased metabolic activity of BAT does not substantially affect glucose utilization (BAT accounts for 1% of the total body glucose utilization compared to 50% of skeletal muscles) during cold exposure in healthy subjects[30], abnormal glucose metabolism with hypoglycemia may contribute to increased metabolic activity of BAT[28] via counterregulatory elevated catecholamines. The effect of cortisol on BAT remains ambiguous; while some researchers state that cortisol inhibits BAT function, other researchers have demonstrated that cortisol has a stimulatory effect[28].
Primary muscle disease was excluded by muscle biopsy. Muscle biopsy was negative in this sense but showed reduced glycogen stores, which could be associated with prolonged counterregulatory hormonal activity. Skeletal muscles mainly express β2-adrenergic receptors and adrenaline rather than noradrenaline, which stimulates glycogen breakdown. Through the Cori cycle, skeletal muscle deposits of glycogen are broken down during adrenaline stimulation and released as lactate and converted to glucose in the liver. Indeed, the reduced glycogen content in skeletal muscles increases insulin sensitivity. The glycogen content slightly increases by the acute intake of large amounts of carbohydrates[31] and possibly leads to a deepening of hypoglycemia.
In summary, although fasting hypoglycemia has been considered a hallmark of insulinoma, postprandial hypoglycemia has also been occasionally reported as the predominant feature similar to weight loss. Repeated and prolonged hypoglycemic episodes can reduce the awareness of neurogenic and neuroglycopenic hypoglycemic symptoms in insulinoma, which delays the diagnosis of the disease. Prolonged but not continuous adrenergic stimulation in the counterregulatory response to hypoglycemia may be indicated by increased metabolic activity of BAT and reduction of muscle glycogen. A low body mass index increases the risk of postoperative complications, and a very small amount of visceral fat can be associated with a lower intraoperative core temperature. Among other things, hypothermia can lead to adverse patient outcomes, including increased blood loss with the need for blood transfer. These complications may lead to higher mortality rates and longer hospital stays, which are positively correlated with the male sex and a younger age[32].
Manuscript source: Unsolicited manuscript
Specialty type: Endocrinology and metabolism
Country/Territory of origin: Slovakia
Peer-review report’s scientific quality classification
Grade A (Excellent): 0
Grade B (Very good): 0
Grade C (Good): C
Grade D (Fair): 0
Grade E (Poor): 0
P-Reviewer: Jin W S-Editor: Ma YJ L-Editor: Filipodia P-Editor: Zhang YL
| 1. | Mathur A, Gorden P, Libutti SK. Insulinoma. Surg Clin North Am. 2009;89:1105-1121. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 82] [Cited by in RCA: 78] [Article Influence: 4.9] [Reference Citation Analysis (0)] |
| 2. | Sugawa T, Murakami T, Yabe D, Kashima R, Tatsumi M, Ooshima S, Joo E, Wada K, Yoshizawa A, Masui T, Nakamoto Y, Yamauchi Y, Kodama Y, Iemura Y, Ogura M, Yasoda A, Inagaki N. Hypoglycemia Unawareness in Insulinoma Revealed with Flash Glucose Monitoring Systems. Intern Med. 2018;57:3407-3412. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 8] [Cited by in RCA: 7] [Article Influence: 1.0] [Reference Citation Analysis (0)] |
| 3. | Okabayashi T, Shima Y, Sumiyoshi T, Kozuki A, Ito S, Ogawa Y, Kobayashi M, Hanazaki K. Diagnosis and management of insulinoma. World J Gastroenterol. 2013;19:829-837. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in CrossRef: 204] [Cited by in RCA: 270] [Article Influence: 22.5] [Reference Citation Analysis (4)] |
| 4. | Lack EE. Pancreatic endocrine neoplasms. in: Pathology of pancreas, gallbladder, extrahepatic biliary tract and ampullary region. Lack EE, New York: Oxford University Press; 2003: 323-373. |
| 5. | Grant CS. Surgical aspects of hyperinsulinemic hypoglycemia. Endocrinol Metab Clin North Am. 1999;28:533-554. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 45] [Cited by in RCA: 35] [Article Influence: 1.3] [Reference Citation Analysis (0)] |
| 6. | Tucker ON, Crotty PL, Conlon KC. The management of insulinoma. Br J Surg. 2006;93:264-275. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 124] [Cited by in RCA: 115] [Article Influence: 6.1] [Reference Citation Analysis (0)] |
| 7. | de Herder WW, Niederle B, Scoazec JY, Pauwels S, Kloppel G, Falconi M, Kwekkeboom DJ, Oberg K, Eriksson B, Wiedenmann B, Rindi G, O'Toole D, Ferone D; Frascati Consensus Conference; European Neuroendocrine Tumor Society. Well-differentiated pancreatic tumor/carcinoma: insulinoma. Neuroendocrinology. 2006;84:183-188. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 207] [Cited by in RCA: 166] [Article Influence: 9.2] [Reference Citation Analysis (0)] |
| 8. | Nikfarjam M, Warshaw AL, Axelrod L, Deshpande V, Thayer SP, Ferrone CR, Fernández-del Castillo C. Improved contemporary surgical management of insulinomas: a 25-year experience at the Massachusetts General Hospital. Ann Surg. 2008;247:165-172. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 148] [Cited by in RCA: 139] [Article Influence: 8.2] [Reference Citation Analysis (0)] |
| 9. | Lo CY, Lam KY, Fan ST. Surgical strategy for insulinomas in multiple endocrine neoplasia type I. Am J Surg. 1998;175:305-307. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 27] [Cited by in RCA: 27] [Article Influence: 1.0] [Reference Citation Analysis (0)] |
| 10. | Vezzosi D, Bennet A, Maiza JC, Buffet A, Grunenwald S, Fauvel J, Courbon F, Otal P, Carrere N, Caron Ph. Diagnosis and Treatment of Insulinomas in the Adults. In: Akin F. Basic and Clinical Endocrinology Up-to Date. IntechOpen 2011: 135-176. [DOI] [Full Text] |
| 11. | Falconi M, Eriksson B, Kaltsas G, Bartsch DK, Capdevila J, Caplin M, Kos-Kudla B, Kwekkeboom D, Rindi G, Klöppel G, Reed N, Kianmanesh R, Jensen RT; Vienna Consensus Conference participants. ENETS Consensus Guidelines Update for the Management of Patients with Functional Pancreatic Neuroendocrine Tumors and Non-Functional Pancreatic Neuroendocrine Tumors. Neuroendocrinology. 2016;103:153-171. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 1097] [Cited by in RCA: 985] [Article Influence: 109.4] [Reference Citation Analysis (1)] |
| 12. | Cryer PE, Axelrod L, Grossman AB, Heller SR, Montori VM, Seaquist ER, Service FJ; Endocrine Society. Evaluation and management of adult hypoglycemic disorders: an Endocrine Society Clinical Practice Guideline. J Clin Endocrinol Metab. 2009;94:709-728. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 712] [Cited by in RCA: 743] [Article Influence: 46.4] [Reference Citation Analysis (0)] |
| 13. | Tesfaye N, Seaquist ER. Neuroendocrine responses to hypoglycemia. Ann N Y Acad Sci. 2010;1212:12-28. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 81] [Cited by in RCA: 91] [Article Influence: 6.1] [Reference Citation Analysis (0)] |
| 14. | Thomson MJ, Mordes JP. Hypoglycemia in Pancreatic Disease. The Pancreapedia: Exocrine Pancreas Knowledge Base. 2015;. [DOI] [Full Text] |
| 15. | Placzkowski KA, Vella A, Thompson GB, Grant CS, Reading CC, Charboneau JW, Andrews JC, Lloyd RV, Service FJ. Secular trends in the presentation and management of functioning insulinoma at the Mayo Clinic, 1987-2007. J Clin Endocrinol Metab. 2009;94:1069-1073. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 207] [Cited by in RCA: 210] [Article Influence: 13.1] [Reference Citation Analysis (0)] |
| 16. | Shreenivas AV, Leung V. A rare case of insulinoma presenting with postprandial hypoglycemia. Am J Case Rep. 2014;15:488-491. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 7] [Cited by in RCA: 9] [Article Influence: 0.8] [Reference Citation Analysis (0)] |
| 17. | Kikuchi T, Chujo D, Takahashi K, Takahashi N, Tanno Y, Tonoike M, Ihana N, Tsujimoto T, Tanabe A, Kajio H. Insulinoma Presenting with Reactive Hypoglycemia: Evaluating the Effect of Tumor Resection via Continuous Glucose Monitoring. Intern Med. 2017;56:3067-3071. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 6] [Cited by in RCA: 9] [Article Influence: 1.1] [Reference Citation Analysis (0)] |
| 18. | Iida K, Ohara T, Hino Y, Nobuhara M, Ishida J, Chihara K. Glucose-responsive insulinoma in a patient with postprandial hypoglycemia in the morning. Intern Med. 2010;49:2123-2127. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 17] [Cited by in RCA: 14] [Article Influence: 0.9] [Reference Citation Analysis (0)] |
| 19. | Dizon AM, Kowalyk S, Hoogwerf BJ. Neuroglycopenic and other symptoms in patients with insulinomas. Am J Med. 1999;106:307-310. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 126] [Cited by in RCA: 114] [Article Influence: 4.4] [Reference Citation Analysis (0)] |
| 20. | Shinden Y, Maemura K, Hashiguchi M, Kawasaki Y, Kurahara H, Mataki Y, Ino S, Sakoda M, Natsugoe S. Preoperative Changes in Body Weight in Patients with an Insulinoma JOP. J Pancreas. 2019;20:44-47. |
| 21. | Grant CS. Insulinoma. Best Pract Res Clin Gastroenterol. 2005;19:783-798. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 211] [Cited by in RCA: 209] [Article Influence: 10.5] [Reference Citation Analysis (0)] |
| 22. | Prelipcean MS, O'Neil PJ, Bell DS. Hyperinsulinemic hypoglycemia precipitated by weight loss. South Med J. 2005;98:726-728. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 2] [Cited by in RCA: 3] [Article Influence: 0.2] [Reference Citation Analysis (0)] |
| 23. | Kar P, Price P, Sawers S, Bhattacharya S, Reznek RH, Grossman AB. Insulinomas may present with normoglycemia after prolonged fasting but glucose-stimulated hypoglycemia. J Clin Endocrinol Metab. 2006;91:4733-4736. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 45] [Cited by in RCA: 39] [Article Influence: 2.1] [Reference Citation Analysis (0)] |
| 24. | Service FJ, Dale AJ, Elveback LR, Jiang NS. Insulinoma: clinical and diagnostic features of 60 consecutive cases. Mayo Clin Proc. 1976;51:417-429. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 10] [Cited by in RCA: 11] [Article Influence: 0.2] [Reference Citation Analysis (0)] |
| 25. | González-Clavijo AM, Fierro-Maya LF. Patient with neuropsychiatric symptoms and insulinoma of difficult preoperative localization. Rev Fac Med. 2014;62:637-640. [RCA] [DOI] [Full Text] [Cited by in Crossref: 2] [Cited by in RCA: 2] [Article Influence: 0.2] [Reference Citation Analysis (0)] |
| 26. | Fahmi SY, Raskin P. Case Study: An 82-Year-Old Woman Presents With Severe Hypoglycemia Induced by an Insulinoma. Clin Diabetes. 2004;22:102-104. [DOI] [Full Text] |
| 27. | Service FJ, Natt N. The prolonged fast. J Clin Endocrinol Metab. 2000;85:3973-3974. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 73] [Cited by in RCA: 71] [Article Influence: 2.8] [Reference Citation Analysis (0)] |
| 28. | Steinberg JD, Vogel W, Vegt E. Factors influencing brown fat activation in FDG PET/CT: a retrospective analysis of 15,000+ cases. Br J Radiol. 2017;90:20170093. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 38] [Cited by in RCA: 50] [Article Influence: 6.3] [Reference Citation Analysis (0)] |
| 29. | Iyer RB, Guo CC, Perrier N. Adrenal pheochromocytoma with surrounding brown fat stimulation. AJR Am J Roentgenol. 2009;192:300-301. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 28] [Cited by in RCA: 28] [Article Influence: 1.8] [Reference Citation Analysis (0)] |
| 30. | Carpentier AC, Blondin DP, Virtanen KA, Richard D, Haman F, Turcotte É E. Brown Adipose Tissue Energy Metabolism in Humans. Front Endocrinol (Lausanne). 2018;9:447. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 213] [Cited by in RCA: 221] [Article Influence: 31.6] [Reference Citation Analysis (0)] |
| 31. | Jensen J, Rustad PI, Kolnes AJ, Lai YC. The role of skeletal muscle glycogen breakdown for regulation of insulin sensitivity by exercise. Front Physiol. 2011;2:112. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 198] [Cited by in RCA: 266] [Article Influence: 19.0] [Reference Citation Analysis (0)] |
| 32. | Miyazaki R, Hoka S, Yamaura K. Visceral fat, but not subcutaneous fat, is associated with lower core temperature during laparoscopic surgery. PLoS One. 2019;14:e0218281. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 3] [Cited by in RCA: 7] [Article Influence: 1.2] [Reference Citation Analysis (0)] |