Published online Dec 26, 2019. doi: 10.12998/wjcc.v7.i24.4299
Peer-review started: October 7, 2019
First decision: October 23, 2019
Revised: October 31, 2019
Accepted: November 15, 2019
Article in press: November 15, 2019
Published online: December 26, 2019
Processing time: 79 Days and 4.3 Hours
Post-transplant lymphoproliferative disorder (PTLD) is a fatal complication of solid organ transplantation or allogenic hematopoietic stem cell transplantation that is associated with immunosuppressive therapy. Potential manifestations are diverse, ranging from reactive lymphoid hyperplasia to high-grade lymphoma. PTLD is usually of B-cell origin and associated with Epstein-Barr virus (EBV) infection. Herein, we describe a case of PTLD involving the peritoneal omentum. There has been only case of PTLD as a diffuse large B-cell lymphoma (DLBCL) in the peritoneum.
The patient was a 62-year-old man who had been receiving immunosuppressive therapy with tacrolimus since undergoing a liver transplant 15 years prior. He reported that he had experienced abdominal discomfort and anorexia 1 month prior to the current admission. Abdominal pelvic computed tomography (CT) revealed peritoneal and omental mass-like lesions without bowel obstruction. Ultrasonography-guided biopsy was performed, and he was histologically diagnosed with EBV-negative DLBCL. Positron emission tomography (PET)-CT depicted peritoneum and omentum involvement only, without any lymphadenopathy or organ masses, including in the gastrointestinal tract. Six cycles of chemotherapy with a “R-CHOP“ regimen (rituximab-cyclophosphamide, doxorubicin, vincristine, prednisolone) were administered, and PET-CT performed thereafter indicated complete remission.
This is the first report of isolated peritoneal lymphomatosis defined as PTLD in a liver transplant recipient.
Core tip: Post-transplant lymphoproliferative disorder (PTLD) is a fatal complication of solid organ transplantation or allogenic hematopoietic stem cell transplantation that is associated with immunosuppressive therapy. Herein we present the first report of isolated peritoneal lymphomatosis defined as PTLD in a liver transplant recipient.
- Citation: Kim HB, Hong R, Na YS, Choi WY, Park SG, Lee HJ. Isolated peritoneal lymphomatosis defined as post-transplant lymphoproliferative disorder after a liver transplant: A case report. World J Clin Cases 2019; 7(24): 4299-4306
- URL: https://www.wjgnet.com/2307-8960/full/v7/i24/4299.htm
- DOI: https://dx.doi.org/10.12998/wjcc.v7.i24.4299
Post-transplant lymphoproliferative disorder (PTLD) is a fatal disease that occurs in patients who have undergone solid organ transplantation and allogenic hematopoietic stem cell transplantation. Its incidence varies depending on the organ transplanted, and after liver transplantation the incidence of PTLD is approximately 1.0%–5.5%[1,2]. Epstein-Barr virus (EBV) infection is known to be the most common etiology. It is typically classified into four histologic types; early lesion, polymorphic, monomorphic, and Hodgkin-like, and it can also be categorized as early-onset (< 2 years) or late-onset (≥ 2 years)[3-5]. PTLD in the form of extranodal lymphoma is slightly more common than other forms, and the gastrointestinal tract, transplanted organs, and central nervous system are the most frequently affected sites[2,6-9]. To date, no cases of peritoneal lymphomatosis have been reported.
Herein, we describe the case of a patient with diffuse large B-cell lymphoma (DLBCL) constituting late-onset monomorphic PTLD, which developed 15 years after a liver transplant. The case involved isolated peritoneal lymphomatosis, which has never been reported in the context of PTLD, and complete remission was achieved via chemotherapy.
A 62-year-old man was admitted due to reduced appetite and abdominal distension, which he had reportedly been experiencing for the last month.
The patient had been followed up regularly since undergoing a liver transplant 15 years prior due to fulminant hepatic failure associated with hepatitis B, and he had been taking tacrolimus 2 mg regularly since that transplant. He had been taking linagliptin and metformin for the past 10 years due to diabetes mellitus. Abdominopelvic computed tomography (CT) had been conducted 6 months prior to the current presentation as part of a periodic check-up, and it had not depicted any abnormal findings. He had developed abdominal discomfort 1 month prior to the current admission, and 2 weeks after its initial onset he developed abdominal distension that was so severe that he could not eat. He then visited the hospital for testing.
There was no tenderness upon abdominal palpation but distension was severe, with fluid wave and shifting dullness indicating ascites.
Complete blood count results were as follows, with normal ranges in parentheses: white blood cells 2.1 × 103/μL (4.0–10.0 103/μL), hemoglobin 13.8 g/dL (12–16 g/dL), platelets 318 × 103/μL (150–400 103/μL). Blood biochemistry results were total bilirubin 0.6 mg/dL (0.2–1.1 mg/dL), aspartate aminotransferase 21 U/L (5–40 U/L), alanine aminotransferase 9 U/L (5–40 U/L), albumin 3.7 g/dL (3.5–5.2 g/dL), blood urea nitrogen 14.4 mg/dL (8.0–20.0 mg/dL), creatinine 0.9 mg/dL (0.5–1.3 mg/dL), C-reactive protein 10.8 mg/dL (0.0–0.5 mg/dL). Among the tumor markers tested for, carcinoembryonic antigen was normal (1.3 ng/mL; normal range 0.0–5.0 ng/mL) but lactate dehydrogenase was elevated (746 U/L; normal range 200–450 U/L).
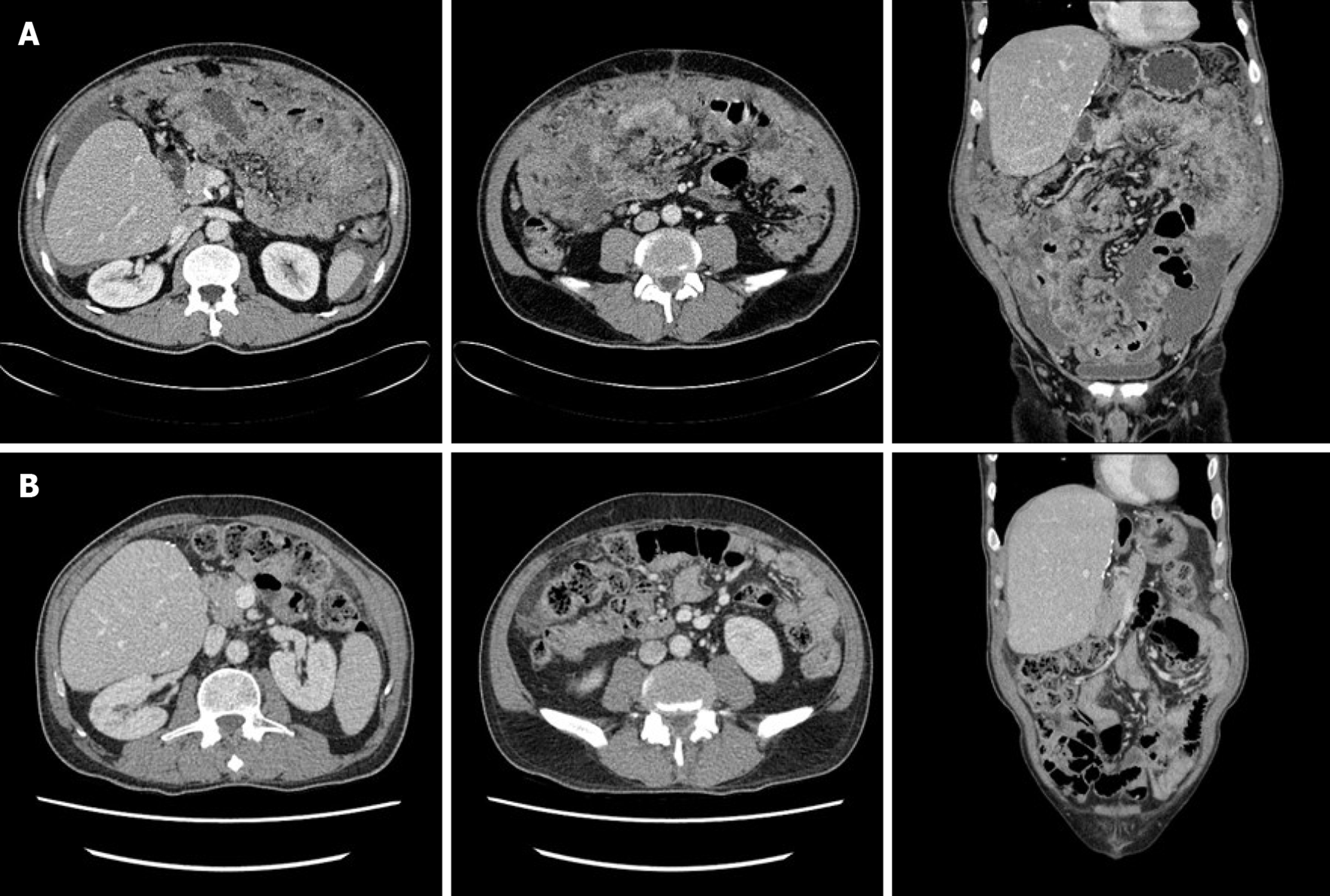
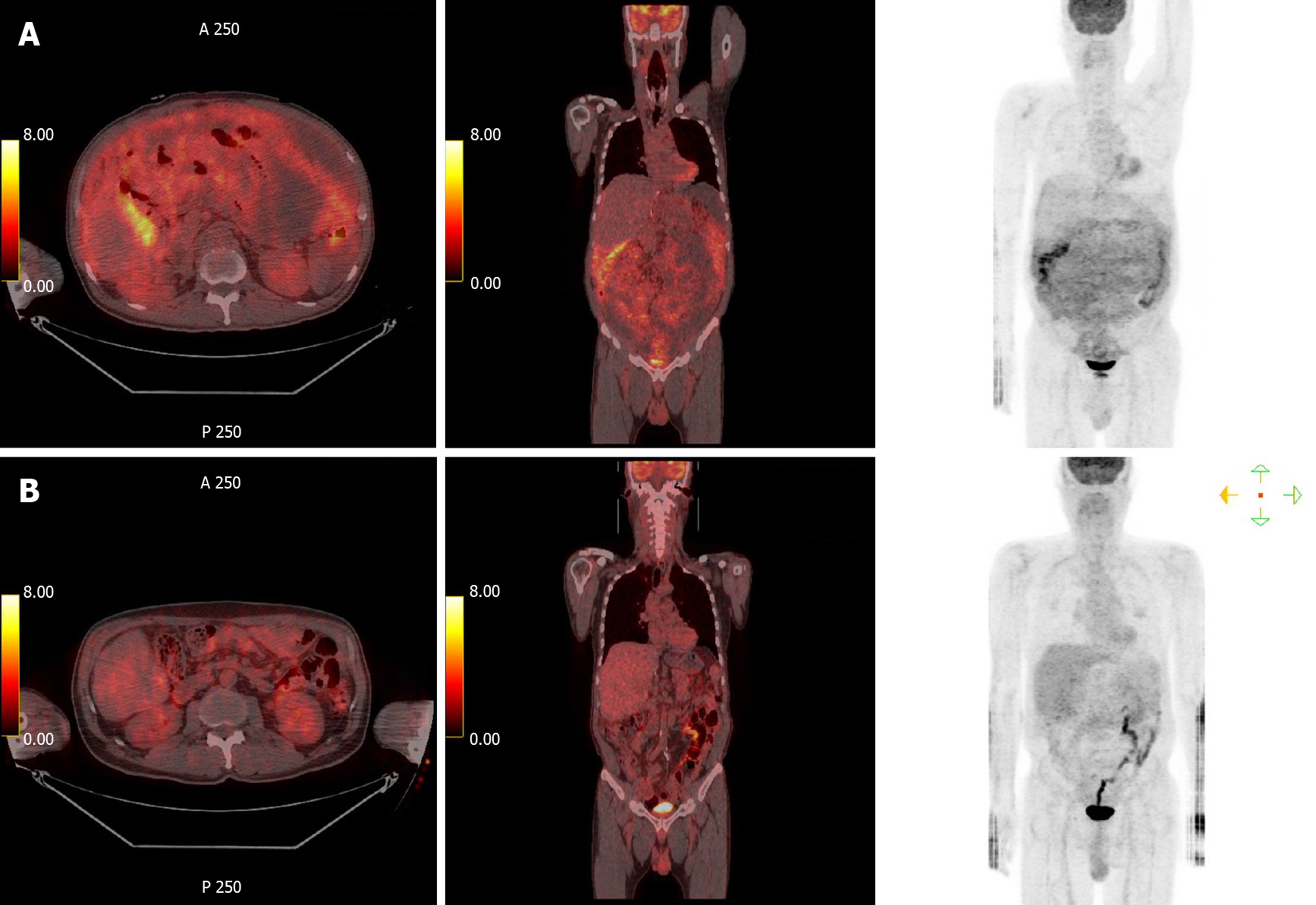
Abdominopelvic CT depicted diffuse peritoneal thickening, omental masses and nodules, and ascites with omental fat infiltration, but no mass-like lesions or bowel obstruction were evident in the gastrointestinal tract (Figure 1). A primary peritoneal disease such as tuberculous peritonitis or malignant mesothelioma was suspected. Positron emission tomography (PET)-CT was performed, and it did not depict abnormal hypermetabolism in solid organs, lymph nodes, or digestive organs but it did reveal diffuse hypermetabolism in the peritoneum and omentum (Figure 2).
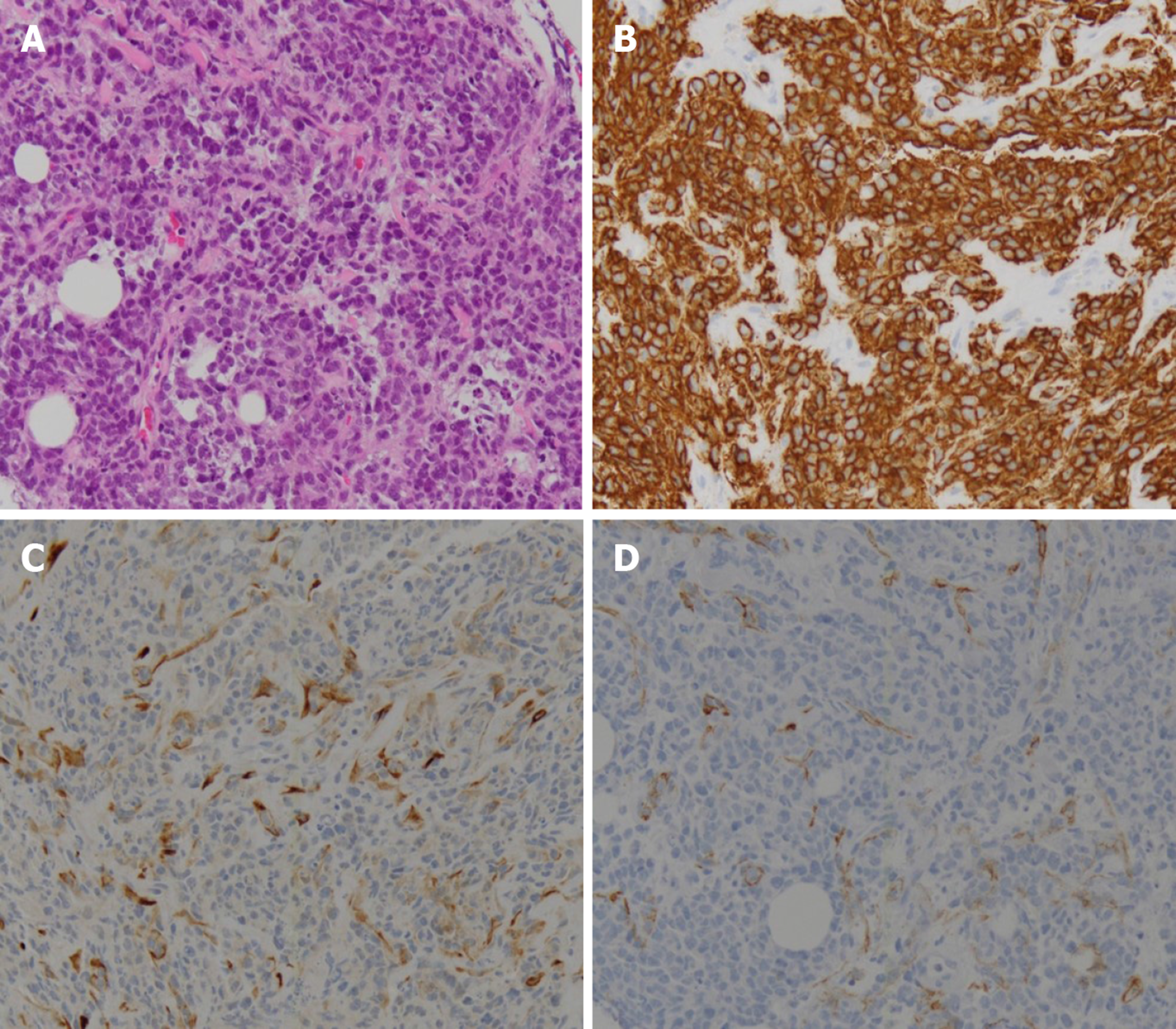
Histological examination was performed on the omental mass using a percutaneous ultrasonography guided core needle, and the mass was definitively diagnosed as DLBCL that was positive for CD20, CD45, and B-cell lymphoma 6, and negative for CD3 and cytokeratin (Figure 3). Other tissue markers were negative for EBV and human herpesvirus 8 (HHV-8). Serum EBV PCR was negative as was serum EBV viral capsid IgM testing, indicating that manifest EBV infection was not present. Serum EBV viral capsid IgG testing was positive. Hepatitis B antigen testing was negative, hepatitis B antibody testing was positive, and hepatitis C antibody testing was negative. Based on these results, the patient was diagnosed with DLBCL as an EBV-negative monomorphic PTLD that had developed 15 years after a liver transplant. The condition was further classified as an isolated peritoneal stage IIE DLBCL with lymph node infiltration but no bone marrow or other solid organ infiltration. The International Prognostic Index score was in the intermediate-low-risk group, as the patient was 62 years of age with elevated lactate dehydrogenase.
The dosage of tacrolimus, an immunosuppressor, should be reduced in patients with PTLD, but he did not want to reduce the dose of the tacrolimus; the tacrolimus level was maintained at a slightly low level of 4. Two cycles of an “RCHOP” (rituximab-cyclophosphamide, doxorubicin, vincristine, prednisolone) regimen, the standard treatment for DLBCL, were administered without a dosage reduction. While on chemotherapy diabetes was not controlled, so response evaluation was performed via CT. CT depicted marked peritoneal mass reduction, indicating a partial response, so 6 cycles were administered without dosage reduction. Subsequent CT scanning did not depict any signs of omental mass or ascites, but there was still mild haziness in the omental fat (Figure 1). PET-CT following an aggressive diabetes management regimen indicated complete remission, with loss of diffuse hypermetabolism lesion in the peritoneum (Figure 2).
Although PET-CT suggested complete remission, CT was performed 2 mo later to monitor the previously observed mild haziness on omentum fat. The haziness had reduced, suggesting that the omentum was undergoing a healing process. The patient currently remains under observation.
PTLD is defined as a lymphoma that develops after solid organ or hematopoietic stem cell transplantation. Since some researchers reported five cases of PTLD as a severe complication of transplantation in 1966, the incidence of PTLD has risen in conjunction with an exponential increase in the number of transplants performed. Although the incidence varies depending on the organ transplanted and the use of immunosuppressors, the incidence of PTLD in cases of liver transplantation is approximately 1.0%–5.5%[1,2]. Of the four pathological types, monomorphic PTLD exhibits the typical characteristics of a malignant lymphoma. DLBCL is the most common, followed by Burkitt lymphoma and aggressive T-cell lymphoma[10].
Approximately 50%–70% of PTLD cases are early-onset, more than 80% of these early onset cases are of B-cell origin, and more than 90% are EBV-positive[4,11-13]. Conversely, EBV-negativity is more common in cases of late-onset PTLD[14]. EBVinduced pathogenesis of PTLD is relatively well known. With immunosurveillance by T lymphocytes diminished via immunosuppressors, which are used to prevent rejection responses in transplant recipients, primary infection or reactivation of EBV may thwart regulation of the proliferation of transformed B-cells, ultimately causing PTLD[15,16].
The mechanisms involved in EBV-negative PTLD remain unclear. The “hit and run” hypothesis suggests that EBV infection occurs but is then lost after causing PTLD[17,18]. Some reports suggest that other viruses such as HHV-8, hepatitis B virus, hepatitis C virus, and cytomegalovirus may be causative, and some reports note that PTLD can have similar characteristics to p53 mutation lymphoma[19-23]. The manifestation of PTLD varies widely, from asymptomatic to fulminant onset. Extranodal lymphoma is relatively common, and the gastrointestinal tract (20%–30%), transplanted organ (10%–15%), and central nervous system (5%–20%) are commonly affected[2,6-9]. To date however, there have been no reports of primary peritoneal onset as was observed in the present case.
PTLD has a poorer prognosis than malignant lymphoma, and the 5-year survival rate of DLBCL as a PTLD is approximately 40%–60%[2,9,24]. It is not clear whether the prognosis of malignant lymphoma that develops as PTLD is poor or whether transplant recipients’ underlying disease and reduced organ function and infection due to immunosuppressors are the main problems, but the International Prognostic Index score is evidently an important prognostic factor[2,25,26].
For PTLD treatment, lowering the dose of immunosuppressors is suggested in most cases if it has not manifested as an EBV-related aggressive lymphoma, but in practice, this is difficult. It has been reported that rituximab therapy alone can be effective in DLBCL, but the R-CHOP regimen is still considered the standard treatment; thus, further research is warranted[27,28].
It is known that malignant lymphoma can develop in any part of the body, but malignant lymphomas that involve the omentum or peritoneum are very rare[29]. Peritoneal lymphomatosis progresses in a manner similar to peritoneal cancer metastasis, where peritoneal thickening with malignant ascites and diffuse infiltration of omental fat-known as omental cake-are observed and the patient complains of severe abdominal discomfort and distension. Usually high-grade malignant lymphoma severely infiltrates the gastrointestinal tract or abdominal lymph node and the peritoneum, but isolated peritoneal lymphomatosis that is restricted to the peritoneum and omentum without involving other gastrointestinal organs is very rare, with no such cases reported as a PTLD to date[30-34]. Thus, there are no data available on the likely prognosis of the present patient.
Some potentially relevant observations pertaining to therapeutic effects and prognoses of primary effusion lymphoma, a body cavity-based lymphoma, may warrant consideration. Primary effusion lymphoma is a rare but officially acknowledged DLBCL in the WHO classification system that specifically occurs in the serosal surface of the pleura or pericardium in the body cavity[35]. HHV-8, which occurs as a result of human immunodeficiency virus infection, is known to be the major cause of primary effusion lymphoma but it commonly affects patients receiving immunosuppressant therapy after a transplant[36]. Given the growing number of reports of HHV-8-negative primary effusion lymphoma, some studies suggest that it should be differentiated from HHV-8-positive lymphoma, but relevant research data are scarce[37]. However, in light of the fact that it occurs in immunosuppressed patients and in the body cavity, the possibility of peritoneal expression of primary effusion lymphoma can be considered.
Peritoneal lymphomatosis is treated with chemotherapy, and unlike peritoneal carcinomatosis, it is curable. Although therapeutic efficacy is predicted to be low due to problems such as reduced peritoneal infiltration by anticancer agents, relevant data are lacking. The present patient is in stage II and classified as low-to-intermediate-risk based on the International Prognostic Index, and PTLD has a relatively low cure rate. Thus, despite the fact that he has currently achieved complete remission, close follow-up will be maintained, given the poor prognosis of primary pleural effusion lymphoma and the possibility of relapse.
This is a valuable report as it presents an extremely rare case of isolated peritoneal lymphomatosis after liver transplantation. Moreover, it is the first report of isolated peritoneal lymphomatosis defined as PTLD in a liver transplant recipient.
Manuscript source: Unsolicited manuscript
Specialty type: Medicine, research and experimental
Country of origin: South Korea
Peer-review report classification
Grade A (Excellent): A
Grade B (Very good): B, B
Grade C (Good): 0
Grade D (Fair): 0
Grade E (Poor): 0
P-Reviewer: Nacif L, Shrestha B, Sun WB S-Editor: Ma YJ L-Editor: A E-Editor: Liu JH
| 1. | Opelz G, Döhler B. Lymphomas after solid organ transplantation: a collaborative transplant study report. Am J Transplant. 2004;4:222-230. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 783] [Cited by in RCA: 753] [Article Influence: 35.9] [Reference Citation Analysis (0)] |
| 2. | Dierickx D, Tousseyn T, Sagaert X, Fieuws S, Wlodarska I, Morscio J, Brepoels L, Kuypers D, Vanhaecke J, Nevens F, Verleden G, Van Damme-Lombaerts R, Renard M, Pirenne J, De Wolf-Peeters C, Verhoef G. Single-center analysis of biopsy-confirmed posttransplant lymphoproliferative disorder: incidence, clinicopathological characteristics and prognostic factors. Leuk Lymphoma. 2013;54:2433-2440. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 81] [Cited by in RCA: 103] [Article Influence: 8.6] [Reference Citation Analysis (0)] |
| 3. | Campo E, Swerdlow SH, Harris NL, Pileri S, Stein H, Jaffe ES. The 2008 WHO classification of lymphoid neoplasms and beyond: evolving concepts and practical applications. Blood. 2011;117:5019-5032. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 1268] [Cited by in RCA: 1446] [Article Influence: 103.3] [Reference Citation Analysis (0)] |
| 4. | Hsiao CY, Lee PH, Ho CM, Wu YM, Ho MC, Hu RH. Post-transplant malignancy in liver transplantation: a single center experience. Medicine (Baltimore). 2014;93:e310. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 24] [Cited by in RCA: 19] [Article Influence: 1.7] [Reference Citation Analysis (0)] |
| 5. | Engels EA, Jennings LW, Everly MJ, Landgren O, Murata K, Yanik EL, Pfeiffer RM, Onaca N, Klintmalm GB. Donor-specific Antibodies, Immunoglobulin-free Light Chains, and BAFF Levels in Relation to Risk of Late-onset PTLD in Liver Recipients. Transplant Direct. 2018;4:e353. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 5] [Cited by in RCA: 7] [Article Influence: 1.0] [Reference Citation Analysis (0)] |
| 6. | Styczynski J, Gil L, Tridello G, Ljungman P, Donnelly JP, van der Velden W, Omar H, Martino R, Halkes C, Faraci M, Theunissen K, Kalwak K, Hubacek P, Sica S, Nozzoli C, Fagioli F, Matthes S, Diaz MA, Migliavacca M, Balduzzi A, Tomaszewska A, Camara Rde L, van Biezen A, Hoek J, Iacobelli S, Einsele H, Cesaro S; Infectious Diseases Working Party of the European Group for Blood and Marrow Transplantation. Response to rituximab-based therapy and risk factor analysis in Epstein Barr Virus-related lymphoproliferative disorder after hematopoietic stem cell transplant in children and adults: a study from the Infectious Diseases Working Party of the European Group for Blood and Marrow Transplantation. Clin Infect Dis. 2013;57:794-802. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 143] [Cited by in RCA: 169] [Article Influence: 14.1] [Reference Citation Analysis (0)] |
| 7. | Aull MJ, Buell JF, Trofe J, First MR, Alloway RR, Hanaway MJ, Wagoner L, Gross TG, Beebe T, Woodle ES. Experience with 274 cardiac transplant recipients with posttransplant lymphoproliferative disorder: a report from the Israel Penn International Transplant Tumor Registry. Transplantation. 2004;78:1676-1682. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 52] [Cited by in RCA: 37] [Article Influence: 1.8] [Reference Citation Analysis (0)] |
| 8. | Caillard S, Porcher R, Provot F, Dantal J, Choquet S, Durrbach A, Morelon E, Moal V, Janbon B, Alamartine E, Pouteil Noble C, Morel D, Kamar N, Buchler M, Mamzer MF, Peraldi MN, Hiesse C, Renoult E, Toupance O, Rerolle JP, Delmas S, Lang P, Lebranchu Y, Heng AE, Rebibou JM, Mousson C, Glotz D, Rivalan J, Thierry A, Etienne I, Moal MC, Albano L, Subra JF, Ouali N, Westeel PF, Delahousse M, Genin R, Hurault de Ligny B, Moulin B. Post-transplantation lymphoproliferative disorder after kidney transplantation: report of a nationwide French registry and the development of a new prognostic score. J Clin Oncol. 2013;31:1302-1309. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 100] [Cited by in RCA: 113] [Article Influence: 9.4] [Reference Citation Analysis (0)] |
| 9. | Kinch A, Baecklund E, Backlin C, Ekman T, Molin D, Tufveson G, Fernberg P, Sundström C, Pauksens K, Enblad G. A population-based study of 135 lymphomas after solid organ transplantation: The role of Epstein-Barr virus, hepatitis C and diffuse large B-cell lymphoma subtype in clinical presentation and survival. Acta Oncol. 2014;53:669-679. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 62] [Cited by in RCA: 70] [Article Influence: 6.4] [Reference Citation Analysis (0)] |
| 10. | Morovic A, Jaffe ES, Raffeld M, Schrager JA. Metachronous EBV-associated B-cell and T-cell posttransplant lymphoproliferative disorders in a heart transplant recipient. Am J Surg Pathol. 2009;33:149-154. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 24] [Cited by in RCA: 23] [Article Influence: 1.4] [Reference Citation Analysis (0)] |
| 11. | Quinlan SC, Pfeiffer RM, Morton LM, Engels EA. Risk factors for early-onset and late-onset post-transplant lymphoproliferative disorder in kidney recipients in the United States. Am J Hematol. 2011;86:206-209. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 175] [Cited by in RCA: 158] [Article Influence: 11.3] [Reference Citation Analysis (0)] |
| 12. | Morton M, Coupes B, Roberts SA, Klapper PE, Byers RJ, Vallely PJ, Ryan K, Picton ML. Epidemiology of posttransplantation lymphoproliferative disorder in adult renal transplant recipients. Transplantation. 2013;95:470-478. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 53] [Cited by in RCA: 60] [Article Influence: 5.0] [Reference Citation Analysis (0)] |
| 13. | Khedmat H, Taheri S. Ultra-early onset post-transplantation lymphoproliferative disease. Saudi J Kidney Dis Transpl. 2013;24:1144-1152. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 4] [Cited by in RCA: 4] [Article Influence: 0.3] [Reference Citation Analysis (0)] |
| 14. | Leblond V, Davi F, Charlotte F, Dorent R, Bitker MO, Sutton L, Gandjbakhch I, Binet JL, Raphael M. Posttransplant lymphoproliferative disorders not associated with Epstein-Barr virus: a distinct entity? J Clin Oncol. 1998;16:2052-2059. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 274] [Cited by in RCA: 253] [Article Influence: 9.4] [Reference Citation Analysis (0)] |
| 15. | Allen U, Preiksaitis J; AST Infectious Diseases Community of Practice. Epstein-barr virus and posttransplant lymphoproliferative disorder in solid organ transplant recipients. Am J Transplant. 2009;9 Suppl 4:S87-S96. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 101] [Cited by in RCA: 86] [Article Influence: 5.4] [Reference Citation Analysis (0)] |
| 16. | Thorley-Lawson DA, Gross A. Persistence of the Epstein-Barr virus and the origins of associated lymphomas. N Engl J Med. 2004;350:1328-1337. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 735] [Cited by in RCA: 739] [Article Influence: 35.2] [Reference Citation Analysis (0)] |
| 17. | Ambinder RF. Gammaherpesviruses and "Hit-and-Run" oncogenesis. Am J Pathol. 2000;156:1-3. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 91] [Cited by in RCA: 105] [Article Influence: 4.2] [Reference Citation Analysis (0)] |
| 18. | Shimizu N, Tanabe-Tochikura A, Kuroiwa Y, Takada K. Isolation of Epstein-Barr virus (EBV)-negative cell clones from the EBV-positive Burkitt's lymphoma (BL) line Akata: malignant phenotypes of BL cells are dependent on EBV. J Virol. 1994;68:6069-6073. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 7] [Cited by in RCA: 7] [Article Influence: 0.2] [Reference Citation Analysis (0)] |
| 19. | Mañez R, Breinig MC, Linden P, Wilson J, Torre-Cisneros J, Kusne S, Dummer S, Ho M. Posttransplant lymphoproliferative disease in primary Epstein-Barr virus infection after liver transplantation: the role of cytomegalovirus disease. J Infect Dis. 1997;176:1462-1467. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 203] [Cited by in RCA: 180] [Article Influence: 6.4] [Reference Citation Analysis (0)] |
| 20. | Buda A, Caforio A, Calabrese F, Fagiuoli S, Pevere S, Livi U, Naccarato R, Burra P. Lymphoproliferative disorders in heart transplant recipients: role of hepatitis C virus (HCV) and Epstein-Barr virus (EBV) infection. Transpl Int. 2000;13 Suppl 1:S402-S405. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 19] [Cited by in RCA: 23] [Article Influence: 1.0] [Reference Citation Analysis (0)] |
| 21. | Tsao L, Hsi ED. The clinicopathologic spectrum of posttransplantation lymphoproliferative disorders. Arch Pathol Lab Med. 2007;131:1209-1218. [RCA] [PubMed] [DOI] [Full Text] [Cited by in RCA: 3] [Reference Citation Analysis (0)] |
| 22. | Zhang A, Zhang M, Shen Y, Wang W, Zheng S. Hepatitis B virus reactivation is a risk factor for development of post-transplant lymphoproliferative disease after liver transplantation. Clin Transplant. 2009;23:756-760. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 9] [Cited by in RCA: 11] [Article Influence: 0.7] [Reference Citation Analysis (0)] |
| 23. | Courville EL, Yohe S, Chou D, Nardi V, Lazaryan A, Thakral B, Nelson AC, Ferry JA, Sohani AR. EBV-negative monomorphic B-cell post-transplant lymphoproliferative disorders are pathologically distinct from EBV-positive cases and frequently contain TP53 mutations. Mod Pathol. 2016;29:1200-1211. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 29] [Cited by in RCA: 35] [Article Influence: 3.9] [Reference Citation Analysis (0)] |
| 24. | Evens AM, David KA, Helenowski I, Nelson B, Kaufman D, Kircher SM, Gimelfarb A, Hattersley E, Mauro LA, Jovanovic B, Chadburn A, Stiff P, Winter JN, Mehta J, Van Besien K, Gregory S, Gordon LI, Shammo JM, Smith SE, Smith SM. Multicenter analysis of 80 solid organ transplantation recipients with post-transplantation lymphoproliferative disease: outcomes and prognostic factors in the modern era. J Clin Oncol. 2010;28:1038-1046. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 238] [Cited by in RCA: 248] [Article Influence: 16.5] [Reference Citation Analysis (0)] |
| 25. | Dierickx D, Tousseyn T, Morscio J, Fieuws S, Verhoef G. Validation of prognostic scores in post-transplantation lymphoproliferative disorders. J Clin Oncol. 2013;31:3443-3444. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 16] [Cited by in RCA: 16] [Article Influence: 1.3] [Reference Citation Analysis (0)] |
| 26. | Trappe RU, Choquet S, Dierickx D, Mollee P, Zaucha JM, Dreyling MH, Dührsen U, Tarella C, Shpilberg O, Sender M, Salles G, Morschhauser F, Jaccard A, Lamy T, Reinke P, Neuhaus R, Lehmkuhl H, Horst HA, Leithäuser M, Schlattmann P, Anagnostopoulos I, Raphael M, Riess H, Leblond V, Oertel S; German PTLD Study Group and the European PTLD Network. International prognostic index, type of transplant and response to rituximab are key parameters to tailor treatment in adults with CD20-positive B cell PTLD: clues from the PTLD-1 trial. Am J Transplant. 2015;15:1091-1100. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 40] [Cited by in RCA: 43] [Article Influence: 4.3] [Reference Citation Analysis (0)] |
| 27. | Trappe R, Oertel S, Leblond V, Mollee P, Sender M, Reinke P, Neuhaus R, Lehmkuhl H, Horst HA, Salles G, Morschhauser F, Jaccard A, Lamy T, Leithäuser M, Zimmermann H, Anagnostopoulos I, Raphael M, Riess H, Choquet S; German PTLD Study Group; European PTLD Network. Sequential treatment with rituximab followed by CHOP chemotherapy in adult B-cell post-transplant lymphoproliferative disorder (PTLD): the prospective international multicentre phase 2 PTLD-1 trial. Lancet Oncol. 2012;13:196-206. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 252] [Cited by in RCA: 273] [Article Influence: 19.5] [Reference Citation Analysis (0)] |
| 28. | Trappe RU, Dierickx D, Zimmermann H, Morschhauser F, Mollee P, Zaucha JM, Dreyling MH, Dührsen U, Reinke P, Verhoef G, Subklewe M, Hüttmann A, Tousseyn T, Salles G, Kliem V, Hauser IA, Tarella C, Van Den Neste E, Gheysens O, Anagnostopoulos I, Leblond V, Riess H, Choquet S. Response to Rituximab Induction Is a Predictive Marker in B-Cell Post-Transplant Lymphoproliferative Disorder and Allows Successful Stratification Into Rituximab or R-CHOP Consolidation in an International, Prospective, Multicenter Phase II Trial. J Clin Oncol. 2017;35:536-543. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 114] [Cited by in RCA: 154] [Article Influence: 17.1] [Reference Citation Analysis (0)] |
| 29. | Runyon BA, Hoefs JC. Peritoneal lymphomatosis with ascites. A characterization. Arch Intern Med. 1986;146:887-888. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 3] [Cited by in RCA: 3] [Article Influence: 0.1] [Reference Citation Analysis (0)] |
| 30. | Horger M, Müller-Schimpfle M, Yirkin I, Wehrmann M, Claussen CD. Extensive peritoneal and omental lymphomatosis with raised CA 125 mimicking carcinomatosis: CT and intraoperative findings. Br J Radiol. 2004;77:71-73. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 25] [Cited by in RCA: 28] [Article Influence: 1.3] [Reference Citation Analysis (0)] |
| 31. | Cunningham N, Ffrench-Constant S, Planche K, Gillmore R. Peritoneal lymphomatosis: a rare presentation of follicular lymphoma mimicking peritoneal carcinomatosis. BMJ Case Rep. 2015;2015:bcr2014207136. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 7] [Cited by in RCA: 10] [Article Influence: 1.0] [Reference Citation Analysis (0)] |
| 32. | Weng SC, Wu CY. Lymphoma presenting as peritoneal lymphomatosis with ascites. J Chin Med Assoc. 2008;71:646-650. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 13] [Cited by in RCA: 16] [Article Influence: 0.9] [Reference Citation Analysis (0)] |
| 33. | Kim YG, Baek JY, Kim SY, Lee DH, Park WS, Kwon Y, Kim MJ, Kang J, Lee JM. Peritoneal lymphomatosis confounded by prior history of colon cancer: a case report. BMC Cancer. 2011;11:276. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 7] [Cited by in RCA: 10] [Article Influence: 0.7] [Reference Citation Analysis (0)] |
| 34. | Choi WY, Kim JH, Choi SJ, Park J, Park YH, Lim JH, Lee MH, Kim CS, Yi HG. Peritoneal lymphomatosis confused with peritoneal carcinomatosis due to the previous history of gastric cancer: a case report. Clin Imaging. 2016;40:837-839. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 8] [Cited by in RCA: 11] [Article Influence: 1.2] [Reference Citation Analysis (0)] |
| 35. | Chen YB, Rahemtullah A, Hochberg E. Primary effusion lymphoma. Oncologist. 2007;12:569-576. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 193] [Cited by in RCA: 190] [Article Influence: 10.6] [Reference Citation Analysis (0)] |
| 36. | Carbone A, Gloghini A. PEL and HHV8-unrelated effusion lymphomas: classification and diagnosis. Cancer. 2008;114:225-227. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 53] [Cited by in RCA: 54] [Article Influence: 3.2] [Reference Citation Analysis (0)] |
| 37. | Saini N, Hochberg EP, Linden EA, Jha S, Grohs HK, Sohani AR. HHV8-Negative Primary Effusion Lymphoma of B-Cell Lineage: Two Cases and a Comprehensive Review of the Literature. Case Rep Oncol Med. 2013;2013:292301. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 5] [Cited by in RCA: 14] [Article Influence: 1.2] [Reference Citation Analysis (0)] |