Published online May 26, 2019. doi: 10.12998/wjcc.v7.i10.1200
Peer-review started: January 31, 2019
First decision: April 18, 2019
Revised: April 25, 2019
Accepted: May 2, 2019
Article in press: May 2, 2019
Published online: May 26, 2019
Processing time: 116 Days and 20.6 Hours
Licorice-induced severe hypokalemic rhabdomyolysis is clinically rare. Gitelman syndrome (GS) is the most common inherited renal tubular disease, while diabetes is one of the most prevalent diseases in the world. Recently, some studies have found that GS patients had higher diabetic morbidity. However, the coexistence of these three diseases has yet to be reported.
We report the case of a 62-year-old Chinese man who was admitted with weakness in the extremities, muscle pain, and dark-colored urine. He had consumed liquorice water daily for seven days prior to admission. The laboratory tests revealed a serum potassium level of 1.84 mmol/L, magnesium 0.68 mmol/L, creatinine phosphokinase (CK) 10117 IU/L, and marked hemoglobinuria. Fractional chloride excretion and fractional magnesium excretion were increased. Plasma renin activity and aldosterone concentration were within the normal ranges. Sequence analysis of the SLC12A3 gene revealed that he had compound heterozygous mutations. The diagnosis of liquorice-induced severe hypokalemic rhabdomyolysis with GS and diabetes was thus genetically confirmed. Serum potassium and CK quickly improved with potassium replacement therapy, hydration, and discontinuation of liquorice ingestion. Upon follow-up at 3 mo, the levels of CK, myoglobin, and potassium remained normal, and magnesium was above 0.6 mmol/L.
This case emphasizes that liquorice consumption and GS should be considered causes of hypokalemia and that the diabetic status of GS patients should be noted in the clinic.
Core tip: This is the first reported case of liquorice-induced severe hypokalemic rhabdomyolysis with Gitelman syndrome and diabetes.
- Citation: Yang LY, Yin JH, Yang J, Ren Y, Xiang CY, Wang CY. Liquorice-induced severe hypokalemic rhabdomyolysis with Gitelman syndrome and diabetes: A case report. World J Clin Cases 2019; 7(10): 1200-1205
- URL: https://www.wjgnet.com/2307-8960/full/v7/i10/1200.htm
- DOI: https://dx.doi.org/10.12998/wjcc.v7.i10.1200
Rhabdomyolysis is a relatively rare but potentially lethal condition. Hypokalemia is a common electrolyte disorder and an established cause of rhabdomyolysis[1]. Frequent causes of hypokalemia are intestinal and urinary potassium loss through diarrhea or the use of diuretics. Liquorice-induced pseudo-hyperaldosteronism and Gitelman syndrome (GS) are well known but rare causes of hypokalemia. GS is an autosomal recessive disorder, which was first described by Gitelman, Graham, and Welt[2] in 1966. Symptoms of liquorice-induced hypokalemia and GS are usually mild. How-ever, GS combined with liquorice-induced pseudo-hyperaldosteronism may cause weakness followed by paralysis, and even rhabdomyolysis or ventricular fibrillation, which can be deadly if left untreated. Furthermore, hypokalemia and hypo-magnesemia in GS patients may cause abnormal glucose metabolism[3]. Here, we report the first case of liquorice-induced severe hypokalemic rhabdomyolysis with GS and diabetes.
A 62-year-old Chinese man complained of intermittent weakness in the extremities (particularly the calves), muscle pain, and walking difficulty.
The patient’s weakness in the extremities started 30 years ago, but spontaneously recovered in one week after each instance. Twenty years ago, he was admitted to the hospital because of difficulty walking upstairs and a diagnosis of hypokalemia was made. The patient’s symptoms quickly improved with potassium replacement therapy. Despite any medicine treatment, his serum potassium levels ranged from 2.8 to 3.1 mmol/L in the past 10 years. The patient presented with muscle pain and walking difficulty 15 d ago. He took potassium chloride sustained-release tablets 1.5 g per day for 5 d. Furthermore, he described dark-colored urine for 3 days. Thus, the patient was sent to the hospital for further evaluation. There was no history of diarrhea or vomiting, signs of infection or alcohol intoxication. He denied ever taking any statins. Upon further questioning, a detailed history revealed that he had con-sumed liquorice water daily for 7 days preceding admission.
The past medical history revealed only type 2 diabetes mellitus since the age of 52. For that reason, he was treated with 10 mg of dapagliflozin and 25 mg of alogliptin daily.
The patient denied tobacco, alcohol, and illicit drug use. His family history was unre-markable, and there were no similar cases in his family.
On admission, the patient’s body weight was 60.0 kg, and his height was 168.2 cm. His blood pressure was 128/92 mmHg and his pulse rate was 86 beats/min. There was generalized paralysis in all four limbs (power grade 3/5). There was no sensory deficit. The examination of the thyroid and other systems was unremarkable.
Table 1 shows laboratory data of the patient. Serum potassium level was 1.84 mmol/L. Serum magnesium was 0.68 mmol/L. Creatinine phosphokinase (CK) was 10117 IU/L, and he had marked hemoglobinuria. Fractional chloride excretion and fractional magnesium excretion were calculated as 1.9% and 4.4%, respectively. Urinary calcium/creatinine ratio was calculated as 0.24. Plasma renin activity and aldosterone concentration were within the normal ranges. Serum alkaline pho-sphatase and parathormone levels were normal. According to the laboratory tests, the level of CK at presentation was 30 times more than the upper limit of the normal range. Furthermore, the level of serum myoglobin (MYO) was significantly increased. Consequently, rhabdomyolysis was diagnosed.
| Parameter | Test value before treatments | Test value after treatments | Reference range |
| Potassium (mmol/L) | 1.841 | 4.05 | 3.5-5.5 |
| Sodium (mmol/L) | 144 | 138 | 137-147 |
| Chlorine (mmol/L) | 981 | 99 | 99-110 |
| Calcium (mmol/L) | 2.31 | 2.14 | 2.11-2.52 |
| Magnesium (mmol/L) | 0.681 | 0.78 | 0.75-1.02 |
| CK (U/L) | 101171 | 275 | 50-310 |
| Myoglobin(μg/L) | >41501 | 98.4 | 17.4-105.7 |
| HbA1c (%) | 7.03%1 | - | 4.8-5.9 % |
| 8 am cortisol (nmol/L) | 423.5 | - | 171-536 |
| 4 pm cortisol (nmol/L) | 183.7 | - | 64-327 |
| 0 am cortisol (nmol/L) | 279.1 | - | - |
| ACTH (pmol/L) | 4.76 | - | 1.6-13.9 |
| pH | 7.471 | 7.40 | 7.35-7.45 |
| HCO3- (mmol/L) | 321 | 24 | 21-26 |
| BE (mmol/L) | 7.11 | 2.4 | -3-3 |
| Urinary pH | 8.01 | 6.0 | 5-6 |
| Urinary sodium (mmol/24 h) | 2401 | - | 40-220 |
| Urinary potassium (mmol/24 h) | 1091 | - | 25-125 |
| Urinary chloride (mmol/24 h) | 2601 | - | 110-250 |
| Urinary calcium (mmol/24 h) | 2.01 | - | 2.5-7.5 |
| Urinary magnesium (mmol/24 h) | 4 | - | 3.0-4.5 |
| Urinary free cortisol (mmol/24 h) | 379.681 | - | 100-379 |
| FECl | 1.9%1 | - | - |
| FEMg | 4.4%1 | - | - |
| Urinary calcium/creatinine | 0.24 | - | - |
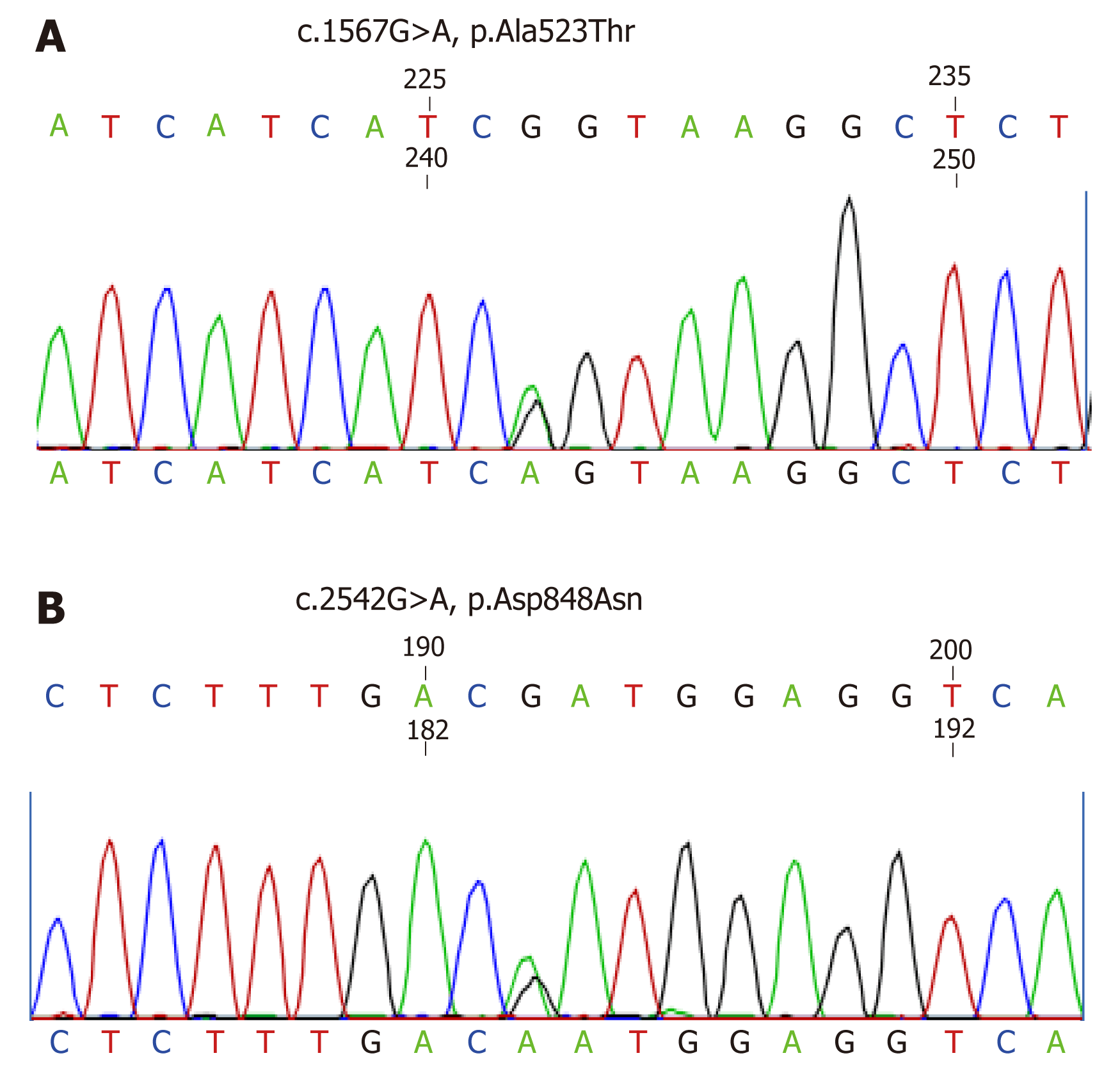
Rhabdomyolysis can have various causes, such as metabolic disease, direct trauma to muscle, muscle necrosis due to ischemia, muscle inflammation, or exposure to drugs and toxins[2]. Recent studies suggest that liquorice can induce hypokalemia and muscle weakness and can even lead to life-threatening rhabdomyolysis[4]. However, considering the medical history, the patient had a history of hypokalemia for 30 years. Moreover, considering the results of the laboratory tests including hypomagnesemia, normal plasma renin activity, and aldosterone concentration, the etiology of pre-existing hypokalemia should be considered. With informed consent, we investigated the SLC12A3 gene of the patient. Sequence analysis of the SLC12A3 gene of the patient revealed that he had compound heterozygous mutations. One missense mutation was a heterozygous G to A base pair substitution at position 1567 in exon 12, which caused an Ala to Thr substitution at position 523. A second missense mutation, a hetero-zygous G to A base pair substitution at position 2542 in exon 21, causing an Asp to Asn substitution at position 848, was also present (Figure 1).
The 12-lead electrocardiogram (ECG) demonstrated a sinus rhythm with inverted T-waves on leads V1-V6 and ST-depression on leads V4-V6, but no prolongation of the QTc interval and U waves. Abdominal sonography did not reveal renal stones or nephrocalcinosis. Peripheral neuropathy in both lower limbs was found by examining the electromyogram. Muscle biopsy was rejected by the patient.
Liquorice-induced severe hypokalemic rhabdomyolysis with GS syndrome and diabetes.
The patient’s signs and symptoms improved with potassium replacement therapy (10 g of potassium chloride injection daily), hydration, and discontinuation of liquorice ingestion. His serum potassium was increased to normal within 5 days. Correspondingly, CK decreased to normal within 8 days. Moreover, the ECG returned to normal on the third hospital day. Five weeks later, the patient was started on antisterone (20 mg, po, bid) and potassium magnesium aspartate tablets (2 tablets, po, tid), following the SLC12A3 gene results.
Upon follow-up at 3 mo, the levels of CK, MYO, and potassium remained normal and magnesium was above 0.6 mmol/L.
Liquorice is the root of Glycyrrhiza glabra and is mostly recognized as a flavoring agent. It has also been used in herbal medicine and even in anti-inflammatory, antiviral, antimicrobial, antioxidative, hepatoprotective, and cardioprotective properties[5]. Glycyrrhizic acid or its hydrolytic product, glycyrrhetinic acid, is found in liquorice extracts and has a well-known mineralcorticoid activity, inhibiting 11 beta-hydroxysteroid dehydrogenase type 2, the enzyme that converts cortisol to cortisone[4]. Glycyrrhizic acid can also directly bind to the mineralocorticoid receptor or suppress 5-β-reductase activity and can therefore slow down the hepatic meta-bolism of aldosterone, resulting in pseudo-hyperaldosteronism and suppression of plasma renin activity, which manifests as hypokalemia, hypertension, and metabolic alkalosis[6]. The onset and severity of the symptoms depend on the dose and duration of liquorice intake, as well as individual susceptibility[7]. GS is characterized by hypokalemia, hypomagnesemia, metabolic alkalosis, and hypocalciuria, with secondary renin-angiotensin-aldosterone system (RAAS) activation and normal blood pressure[8]. With an incidence of 1 in 40000, GS is one of the prevalent autosomal recessive diseases[9]. The SLC12A3 gene is located on chromosome 16 and comprises 26 exons. Liu et al[10] analyzed the characteristics of the genotype and phenotype in 67 patients with GS. They found that compound heterozygous mutations were detected in 42 (62.7%) patients, 10 (14.9%) patients carried homozygous mutations, and 11 patients had only one heterozygous mutation. Of note, there were four patients who had three different mutations, while three unrelated (5.7%) families were found to have triple SLC12A3 mutant alleles[10]. Mutations in the SLC12A3 gene encoding the thiazide sensitive Na+Cl– cotransporter (NCCT) on the apical membrane of distal convoluted tubule cells are usually responsible for GS. Until now, more than 180 mutations in the SLC12A3 gene have been identified in patients with GS[11]. Impaired NCCT function leads to decreased Na+ and Cl– reabsorption in the distal convoluted tubule. Secondary hyperaldosteronism results in renal potassium wasting[12]. Both mutations in this case have been previously reported[13,14]. Unfortunately, the patient’s father was not currently alive. Only his asymptomatic mother was alive; thus, his carrier status could not be ascertained. There were no other family members who underwent genetic testing. This is a deficiency of our case.
Based on the above metabolic pathways, the hypokalemia, rhabdomyolysis, increased cortisol, and normal RAAS level in our case can be explained. In the past 5 years, abnormal glucose metabolism and insulin secretion have been reported in GS patients[11]. Liu’s study indicated that compared with normal Chinese adults, GS patients had a higher diabetic morbidity[10]. Hypokalemia and hypomagnesemia were thought to be the main reasons for diabetes in GS patients. However, the exact mechanism has not been well understood until recently. The pancreatic release of insulin is controlled by ATP-sensitive potassium channels and L-type calcium channels on the β cell surface. Hypokalemia may prevent the closure of these channels and consequently prevent glucose-stimulated insulin secretion[4]. It is thought that chronic hypomagnesemia may induce altered cellular glucose transport, reduce pancreatic insulin secretion, increase defective post receptor insulin signaling, and alter insulin-insulin receptor interactions[15]. Furthermore, secondary RAAS activation may lead to insulin resistance[16]. However, a study[3] in 2017 showed that glucose metabolism and insulin secretion were impaired in GS patients, but insulin sensitivity was comparable between GS patients and patients with type 2 diabetes mellitus. Therefore, further studies are required to address this question.
In conclusion, hypokalemic rhabdomyolysis is often under-recognized and can be associated with life-threatening sequelae. Vigilance and prompt treatment are crucial to improve clinical outcomes. From this case, we would like to stress the importance for physicians to keep liquorice consumption and GS in mind as a cause for hypokalemia. We suggest that genetic analysis is advisable to determine whether the suspicion of GS is warranted. At the same time, the diabetic status of GS patients should be considered.
Manuscript source: Unsolicited manuscript
Specialty type: Medicine, research and experimental
Country of origin: China
Peer-review report classification
Grade A (Excellent): 0
Grade B (Very good): B, B
Grade C (Good): 0
Grade D (Fair): 0
Grade E (Poor): 0
P-Reviewer: Chen GX, Dabla PK S-Editor: Ji FF L-Editor: Wang TQ E-Editor: Wu YXJ
| 1. | Pecnik P, Müller P, Vrabel S, Windpessl M. Two cases of hypokalaemic rhabdomyolysis: same but different. BMJ Case Rep. 2018;2018:pii: bcr-2017-223609. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 1] [Cited by in RCA: 2] [Article Influence: 0.3] [Reference Citation Analysis (0)] |
| 2. | Kumagai H, Matsumoto S, Nozu K. Hypokalemic rhabdomyolysis in a child with Gitelman's syndrome. Pediatr Nephrol. 2010;25:953-955. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 12] [Cited by in RCA: 8] [Article Influence: 0.5] [Reference Citation Analysis (0)] |
| 3. | Yuan T, Jiang L, Chen C, Peng X, Nie M, Li X, Xing X, Li X, Chen L. Glucose tolerance and insulin responsiveness in Gitelman syndrome patients. Endocr Connect. 2017;6:243-252. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 17] [Cited by in RCA: 28] [Article Influence: 3.5] [Reference Citation Analysis (0)] |
| 4. | He R, Guo WJ, She F, Miao GB, Liu F, Xue YJ, Liu YW, Wang HT, Zhang P. A rare case of hypokalemia-induced rhabdomyolysis. J Geriatr Cardiol. 2018;15:321-324. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in RCA: 3] [Reference Citation Analysis (0)] |
| 5. | de Putter R, Donck J. Low-dose liquorice ingestion resulting in severe hypokalaemic paraparesis, rhabdomyolysis and nephrogenic diabetes insipidus. Clin Kidney J. 2014;7:73-75. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 6] [Cited by in RCA: 8] [Article Influence: 0.7] [Reference Citation Analysis (0)] |
| 6. | Omar HR, Komarova I, El-Ghonemi M, Fathy A, Rashad R, Abdelmalak HD, Yerramadha MR, Ali Y, Helal E, Camporesi EM. Licorice abuse: time to send a warning message. Ther Adv Endocrinol Metab. 2012;3:125-138. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 135] [Cited by in RCA: 142] [Article Influence: 10.9] [Reference Citation Analysis (0)] |
| 7. | van den Bosch AE, van der Klooster JM, Zuidgeest DM, Ouwendijk RJ, Dees A. Severe hypokalaemic paralysis and rhabdomyolysis due to ingestion of liquorice. Neth J Med. 2005;63:146-148. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 24] [Cited by in RCA: 24] [Article Influence: 1.2] [Reference Citation Analysis (0)] |
| 8. | Biagioni M, Marigliano M, Iannilli A, Cester A, Gatti S, D'Alba I, Tedeschi S, Syren ML, Cherubini V. Diabetic ketoacidosis complicated with previously unknown Gitelman syndrome in a Tunisian child. Diabetes Care. 2011;34:e107. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 10] [Cited by in RCA: 6] [Article Influence: 0.4] [Reference Citation Analysis (0)] |
| 9. | Khan J. Poster 105 Chronic Leg Weakness Complicated by Rhabdomyolysis as a Result of Gitelman Syndrome: A Case Report. PM R. 2016;8:S196. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 1] [Cited by in RCA: 1] [Article Influence: 0.1] [Reference Citation Analysis (0)] |
| 10. | Liu T, Wang C, Lu J, Zhao X, Lang Y, Shao L. Genotype/Phenotype Analysis in 67 Chinese Patients with Gitelman's Syndrome. Am J Nephrol. 2016;44:159-168. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 21] [Cited by in RCA: 37] [Article Influence: 4.1] [Reference Citation Analysis (0)] |
| 11. | Ren H, Qin L, Wang W, Ma J, Zhang W, Shen PY, Shi H, Li X, Chen N. Abnormal glucose metabolism and insulin sensitivity in Chinese patients with Gitelman syndrome. Am J Nephrol. 2013;37:152-157. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 26] [Cited by in RCA: 33] [Article Influence: 2.8] [Reference Citation Analysis (0)] |
| 12. | Akinci B, Celik A, Saygili F, Yesil S. A case of Gitelman's syndrome presenting with extreme hypokalaemia and paralysis. Exp Clin Endocrinol Diabetes. 2009;117:69-71. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 5] [Cited by in RCA: 4] [Article Influence: 0.2] [Reference Citation Analysis (0)] |
| 13. | Jang HR, Lee JW, Oh YK, Na KY, Joo KW, Jeon US, Cheong HI, Kim J, Han JS. From bench to bedside: diagnosis of Gitelman's syndrome -- defect of sodium-chloride cotransporter in renal tissue. Kidney Int. 2006;70:813-817. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 16] [Cited by in RCA: 17] [Article Influence: 0.9] [Reference Citation Analysis (0)] |
| 14. | Riveira-Munoz E, Chang Q, Godefroid N, Hoenderop JG, Bindels RJ, Dahan K, Devuyst O; Belgian Network for Study of Gitelman Syndrome. Transcriptional and functional analyses of SLC12A3 mutations: new clues for the pathogenesis of Gitelman syndrome. J Am Soc Nephrol. 2007;18:1271-1283. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 104] [Cited by in RCA: 100] [Article Influence: 5.6] [Reference Citation Analysis (0)] |
| 15. | Subasinghe CJ, Sirisena ND, Herath C, Berge KE, Leren TP, Bulugahapitiya U, Dissanayake VHW. Novel mutation in the SLC12A3 gene in a Sri Lankan family with Gitelman syndrome & coexistent diabetes: a case report. BMC Nephrol. 2017;18:140. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 9] [Cited by in RCA: 10] [Article Influence: 1.3] [Reference Citation Analysis (0)] |
| 16. | Rahimi Z, Moradi M, Nasri H. A systematic review of the role of renin angiotensin aldosterone system genes in diabetes mellitus, diabetic retinopathy and diabetic neuropathy. J Res Med Sci. 2014;19:1090-1098. [PubMed] |