Published online Jun 16, 2024. doi: 10.12998/wjcc.v12.i17.3259
Revised: April 21, 2024
Accepted: April 28, 2024
Published online: June 16, 2024
Processing time: 94 Days and 2 Hours
Prostate cancer is the second most common cancer among men worldwide, and prostate-specific antigen (PSA) is often used in clinical practice to screen for prostate cancer. Normal total PSA (tPSA) level initially excludes prostate cancer. Here, we report a case of prostate cancer with elevated free PSA density (fPSAD).
A patient diagnosed with benign prostatic hyperplasia underwent prostatectomy, and the postoperative pathological results showed acinar adenocarcinoma of the prostate. The patient is currently undergoing endocrine chemotherapy.
We provide a clinical reference for diagnosis and treatment of patients with normal tPSA but elevated fPSAD.
Core Tip: The patient was diagnosed with prostatic hyperplasia before surgery, and the postoperative pathological findings suggested prostate cancer. Total prostate-specific antigen (PSA) was within the normal range, while free PSA density (fPSAD) was elevated. We emphasize that PSA levels < 4 ng/mL may not always correlate with low cancer risk, highlighting the importance of fPSAD.
- Citation: Huang DH, Hu YX, Guo S, Yang WJ. Prostate cancer with elevated free prostate-specific antigen density: A case report. World J Clin Cases 2024; 12(17): 3259-3264
- URL: https://www.wjgnet.com/2307-8960/full/v12/i17/3259.htm
- DOI: https://dx.doi.org/10.12998/wjcc.v12.i17.3259
Prostate-specific antigen (PSA) is produced by prostate epithelial cells in normal life activities, and is a specific marker of prostate cancer[1]. A normal PSA level is 0-4 ng/mL. Total PSA (tPSA) has some diagnostic specificity for prostate cancer, mainly in patients with serum tPSA between 4 and 10 ng/mL (gray zone). In addition, PSA levels < 4 ng/mL may not always be associated with a lower cancer risk. Therefore, the free PSA density (fPSAD) index was introduced, calculated by free PSA (fPSA)/prostate volume and the optimal cutoff value was 0.062, which has potential for clinical application[2]. Here, we report a patient with normal tPSA and elevated fPSAD who was diagnosed with prostate cancer after surgery.
An elderly man was admitted to our hospital for > 1 year due to difficulty urinating. In the 2 years prior to this, he underwent transurethral resection of the prostate (TURP), and postoperative pathology revealed benign prostatic hyperplasia.
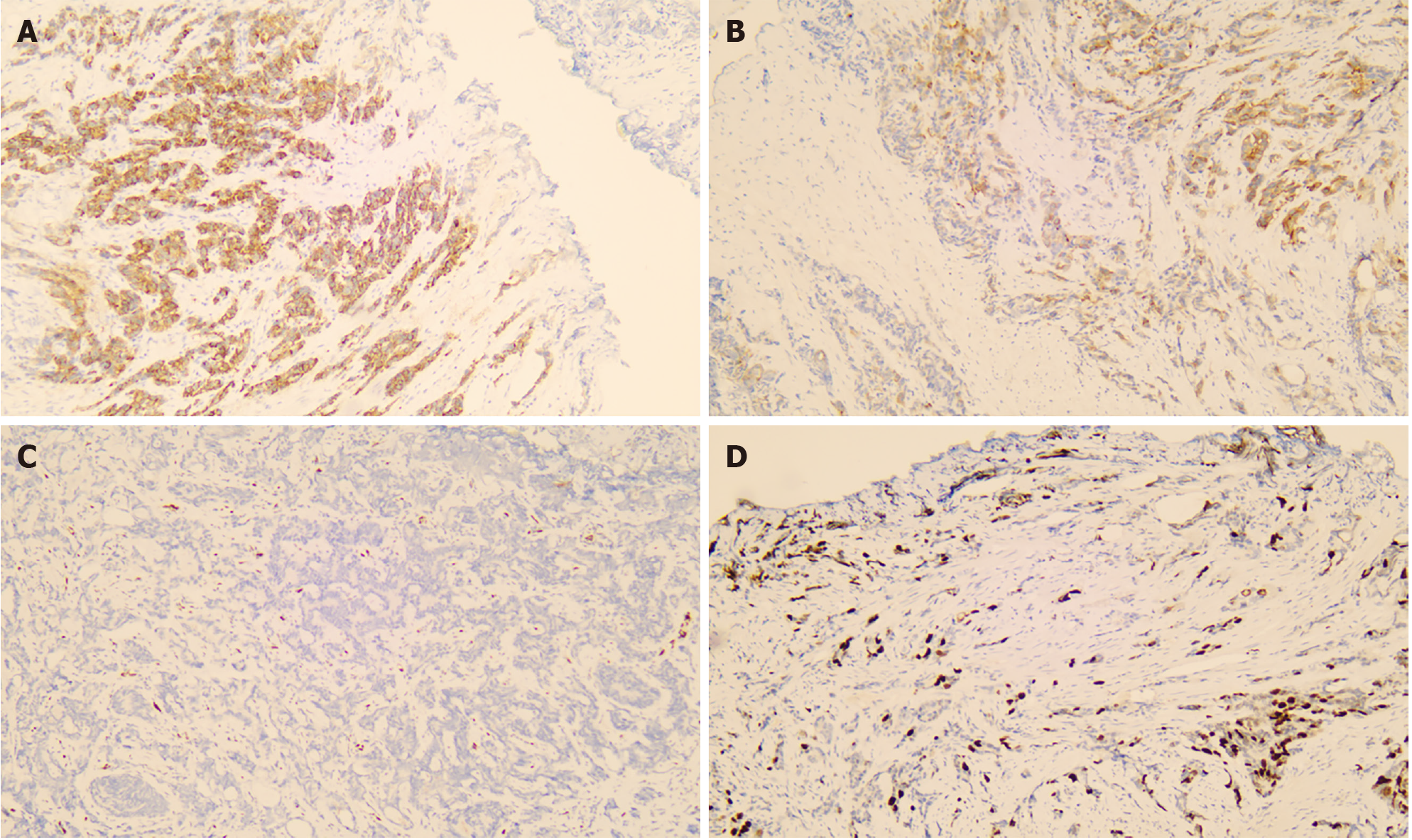
The postoperative pathological report showed prostate acinar adenocarcinoma, with a Gleason score of 4 + 3 = 7 (Figure 1). In general, a positive early growth response gene is an immunohistochemical marker of prostate cancer that may indicate the presence of prostate cancer in a patient.
The patient visited another hospital 2 years ago for benign prostatic hyperplasia and underwent TURP. The postoperative pathological result was benign prostatic hyperplasia.
The patient had no history of smoking or alcohol consumption, and no related family history.
The patient had bulging and tenderness in the bladder area, but was otherwise unremarkable.
The laboratory examination results at admission were as follows: tPSA 3.81 ng/mL, fPSA 1.82 ng/mL, and fPSA/tPSA 0.48.
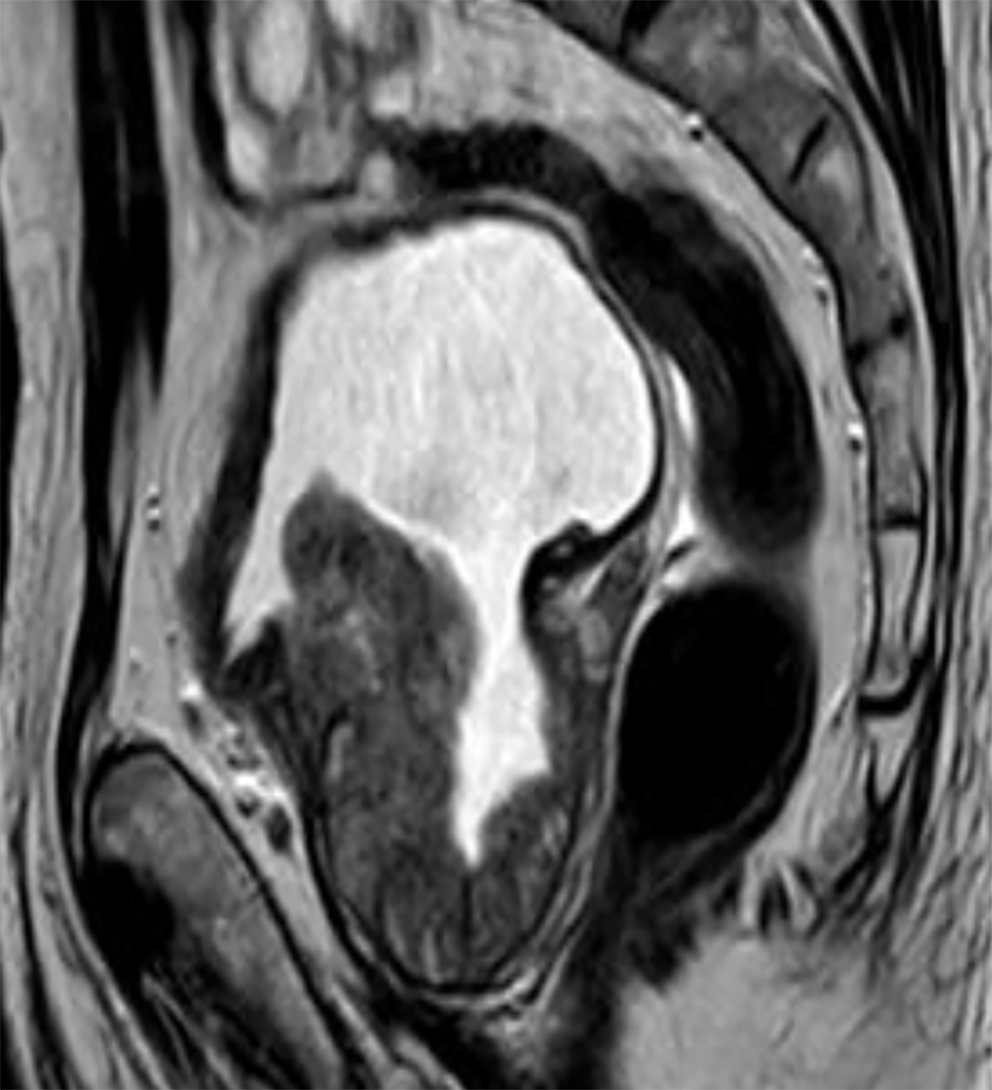
Magnetic resonance imaging of the prostate showed a consistent appearance of benign prostatic hyperplasia with a prostate volume of 4.1 cm × 2.9 cm × 1.9 cm (Figure 2).
Incidental prostate tumor accompanied by normal tPSA and elevated fPSAD.
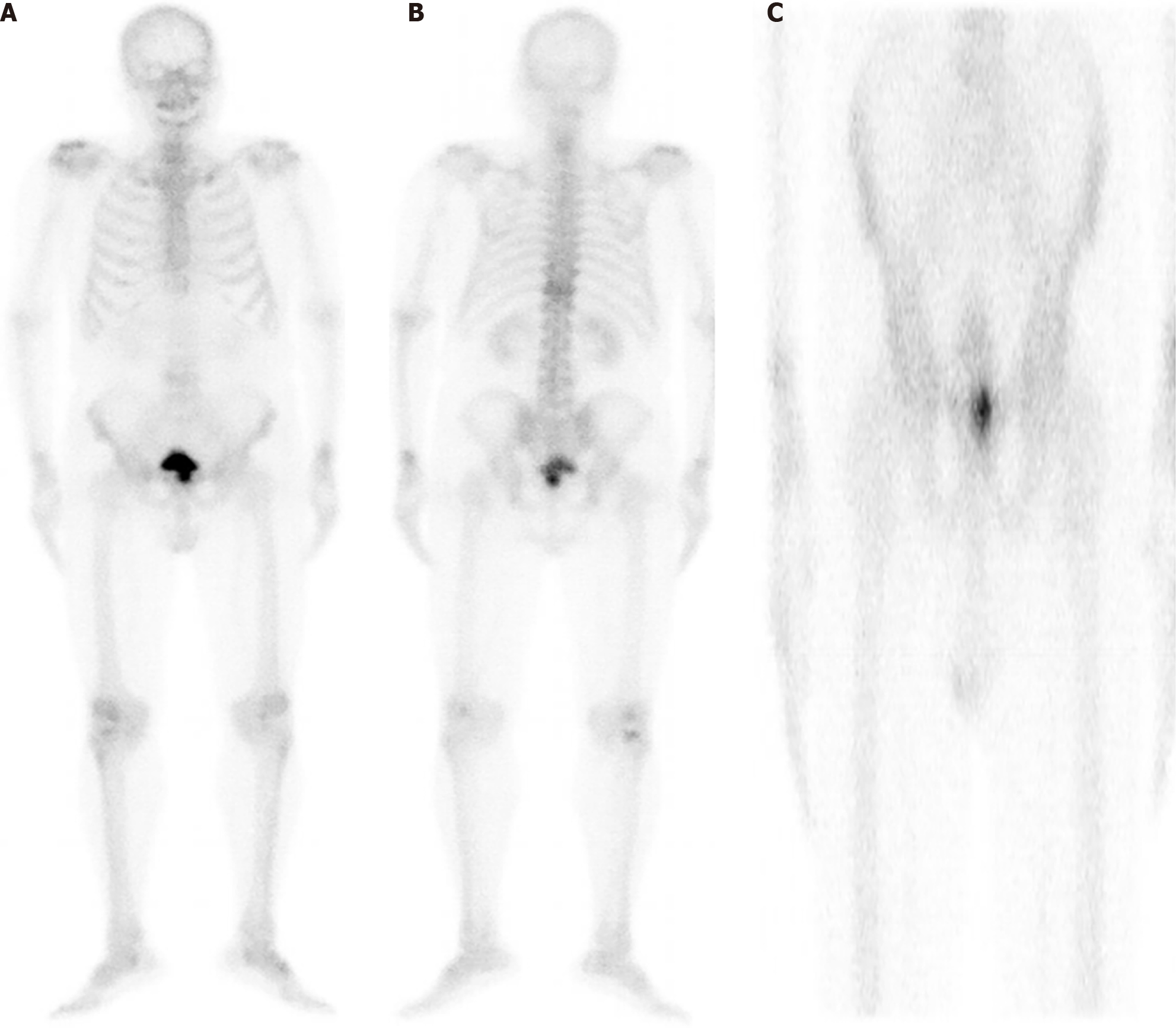
The patient was preliminarily diagnosed with benign prostatic hyperplasia through preoperative auxiliary examinations. After excluding surgical contraindications, TURP was performed, and symptomatic treatment such as fluid replacement was given postoperatively. The postoperative pathological results indicated a prostate tumor. On postoperative day 7, the patient was able to urinate smoothly after removing the catheter. Postoperative emission computed tomography showed no signs of bone metastasis. The drug was well marked, clear bone development, and visible kidney and bladder images. Bilateral thoracic symmetry, rib ribs, T11 vertebral flat, imaging agent distribution, more clear axial bone vertebral structure, no obvious abnormalities concentration and sparse area, equivalent to the pubic joint location imaging agent increased distribution, fault imaging tips for urine residue, more than imaging of limbs long tubular bone and joints no obvious abnormalities (Figure 3).
The patient received endocrine therapy with bicalutamide and goserelin, and PSA was in the normal range after follow-up.
Immunoreactive PSA comes in two forms; the majority of which is bound to serum protein and 10%-30% is fPSA[3]. fPSA is also known as kallikrein 3[4]. Most laboratories use serum fPSA levels of 0.4-1.3 as reference values. Serum fPSA is slightly elevated in patients with benign prostatic hyperplasia without inflammation and in patients with chronic inflammation. Higher-grade lesions (high-grade intraepithelial neoplasia and prostate cancer) were associated with fPSA > 5 ng/mL. Since overlapping high values have also been observed in acute prostatitis, we should be more cautious about high-grade lesions that have not yet been diagnosed after TURP and may require radical prostatectomy for evaluation[5].
PSA is the most commonly used indicator for prostate cancer screening, with high sensitivity and low specificity[6]. Several PSA-derived parameters also appear in clinical practice, such as f/tPSA, (f/t)/PSA density and prostate health index (PHI). PHI is calculated by PSA, fPSA and p2PSA through the following formula: (p2PSA/fPSA) × (tPSA)1/2. Studies have shown that the higher the PHI, the greater the risk of prostate cancer. 4K score is also a comprehensive prostate cancer indicator, including human kallikrenase (hK2), intact PSA, fPSA and tPSA. Complete PSA refers to a free but inactive PSA. hK2 is mainly secreted by prostate epithelial cells and is present in blood, semen and urine. The 4K score provides an individualized prediction of prostate cancer without taking into account previously performed screening or biopsy, providing important information for clinical decision-making[7,8]. However, due to unsatisfactory diagnostic efficiency[9] or high cost of detection[10], This study took primary hospitals as the research object, and did not carry out p2PSA, hK2 and complete PSA related indicators, so hK2 and 4K scores are difficult to promote clinical application. Therefore, we wanted to know if there was an indicator that could more effectively diagnose prostate cancer and be clinically available. fPSAD has been shown to have potential for clinical promotion and application[2]. In evaluating the diagnostic value of fPSAD, our case verified the high consistency of fPSAD.
Our patient had elevated fPSA and fPSAD after TURP. The pathological findings suggested prostate cancer. The fPSA of the patient was 1.82 ng/mL and the fPSAD was 0.08 ng/(mL × cm3). The important features of this case were that tPSA was within the normal range, digital rectal examination was not significantly abnormal, and magnetic resonance imaging showed signs of prostatic hyperplasia. The patient underwent TURP 2 years ago, and the postoperative pathological results indicated benign prostatic hyperplasia. For patients with normal tPSA and elevated fPSAD, adequate evaluation should be conducted before TURP to guard against the consequences of prostate cancer.
We report a case of elevated fPSAD in a patient with prostate cancer. We recommend that before TURP, even if tPSA is normal, further evaluation is needed before surgery because fPSAD is not fully understood.
| 1. | Bai X, Jiang Y, Zhang X, Wang M, Tian J, Mu L, Du Y. The Value of Prostate-Specific Antigen-Related Indexes and Imaging Screening in the Diagnosis of Prostate Cancer. Cancer Manag Res. 2020;12:6821-6826. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 5] [Cited by in RCA: 6] [Article Influence: 1.2] [Reference Citation Analysis (0)] |
| 2. | Zou BZ, Wen H, Luo HJ, Luo WC, Xie QT, Zhou MT. Value of serum free prostate-specific antigen density in the diagnosis of prostate cancer. Ir J Med Sci. 2023;192:2681-2687. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 1] [Reference Citation Analysis (0)] |
| 3. | Xu XH, Xu ZH, Yuan MZ, Guan Y, Zhao ST. Effect of prostate volume on f/tPSA value: A cross-sectional study. Andrologia. 2021;53:e13851. [RCA] [PubMed] [DOI] [Full Text] [Reference Citation Analysis (0)] |
| 4. | Fenner A. Radiotheranostic targeting of fPSA. Nat Rev Urol. 2021;18:131. [RCA] [PubMed] [DOI] [Full Text] [Reference Citation Analysis (0)] |
| 5. | Lakhey M, Ghimire R, Shrestha R, Bhatta AD. Correlation of serum free prostate-specific antigen level with histological findings in patients with prostatic disease. Kathmandu Univ Med J (KUMJ). 2010;8:158-163. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 4] [Cited by in RCA: 4] [Article Influence: 0.3] [Reference Citation Analysis (0)] |
| 6. | Mahal BA, Aizer AA, Efstathiou JA, Nguyen PL. Association of very low prostate-specific antigen levels with increased cancer-specific death in men with high-grade prostate cancer. Cancer. 2016;122:78-83. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 27] [Cited by in RCA: 44] [Article Influence: 4.4] [Reference Citation Analysis (0)] |
| 7. | Borbiev T, Kohaar I, Petrovics G. Clinical Biofluid Assays for Prostate Cancer. Cancers (Basel). 2023;16. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in RCA: 6] [Reference Citation Analysis (0)] |
| 8. | Kawada T, Shim SR, Quhal F, Rajwa P, Pradere B, Yanagisawa T, Bekku K, Laukhtina E, von Deimling M, Teoh JY, Karakiewicz PI, Araki M, Shariat SF. Diagnostic Accuracy of Liquid Biomarkers for Clinically Significant Prostate Cancer Detection: A Systematic Review and Diagnostic Meta-analysis of Multiple Thresholds. Eur Urol Oncol. 2023;. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 14] [Cited by in RCA: 10] [Article Influence: 10.0] [Reference Citation Analysis (0)] |
| 9. | Huang Y, Li ZZ, Huang YL, Song HJ, Wang YJ. Value of free/total prostate-specific antigen (f/t PSA) ratios for prostate cancer detection in patients with total serum prostate-specific antigen between 4 and 10 ng/mL: A meta-analysis. Medicine (Baltimore). 2018;97:e0249. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 33] [Cited by in RCA: 41] [Article Influence: 5.9] [Reference Citation Analysis (0)] |
| 10. | Barisiene M, Bakavicius A, Stanciute D, Jurkeviciene J, Zelvys A, Ulys A, Vitkus D, Jankevicius F. Prostate Health Index and Prostate Health Index Density as Diagnostic Tools for Improved Prostate Cancer Detection. Biomed Res Int. 2020;2020:9872146. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 10] [Cited by in RCA: 22] [Article Influence: 4.4] [Reference Citation Analysis (0)] |