Published online Feb 16, 2022. doi: 10.12998/wjcc.v10.i5.1654
Peer-review started: August 13, 2021
First decision: October 20, 2021
Revised: November 5, 2021
Accepted: December 31, 2021
Article in press: December 31, 2021
Published online: February 16, 2022
Processing time: 181 Days and 22.5 Hours
Superior mesenteric artery syndrome is a disease with a complex diagnosis, and it is associated with complications that make it even harder to identify. Currently, a frequent association with psychiatric disorders has been noted. Despite numerous case reports and case series, the variability of the disease has not allowed the development of protocols regarding diagnosis and management.
A 33-year-old woman presented with abdominal pain, nausea, and bile vomiting over the last 15 mo, associated with a 15-kg weight loss over the last three months. After the onset of the symptoms, the patient was diagnosed with anxiety-depressive disorder and treated appropriately. Standard examinations excluded an organic cause, and the cause of the symptoms was considered psychogenic. The persistence of symptoms, even under treatment, prompted a computer tomography angiography examination of the abdomen and pelvis. The examination identified emergence at a sharp angle of 13.7° of the superior mesenteric artery, with a reduced distance between the artery and the anterior wall of the aorta up to a maximum of 8 mm. A diagnosis of aortomesenteric clamp was established. Surgical treatment by laparoscopic duodenojejunostomy was performed. Postoperative evolution was marked by a patent anastomosis at 1 mo, with a 10-kg weight gain and improvement of the associated anxiety.
This case report underlines two major aspects. One aspect refers to the predisposition of patients with superior mesenteric artery syndrome to develop psychiatric disorders, with an excellent outcome when proper treatment is administered. The second aspect underlines the key role of a multidisciplinary approach and follow-up.
Core Tip: Wilkie’s syndrome is a rare vasculo-intestinal obstructive pathology that is difficult to diagnose due to nonspecific symptoms and the ability to mimic or coexist with other functional disorders. Its association with psychiatric eating disorders makes its differential diagnosis even more complex, allowing it to progress toward late, chronic stages. Untreated disease is associated with significant morbidity and mortality due to complications. It is important to consider its impact on psychosocial status and the appropriate approach to make an exclusion diagnosis.
- Citation: Apostu RC, Chira L, Colcear D, Lebovici A, Nagy G, Scurtu RR, Drasovean R. Wilkie’s syndrome as a cause of anxiety-depressive disorder: A case report and review of literature. World J Clin Cases 2022; 10(5): 1654-1666
- URL: https://www.wjgnet.com/2307-8960/full/v10/i5/1654.htm
- DOI: https://dx.doi.org/10.12998/wjcc.v10.i5.1654
Superior mesenteric artery (SMA) syndrome is a rare cause of proximal small bowel obstruction, defined as compression of the third part of the duodenum between the aorta and superior mesenteric artery[1]. The syndrome has been given many names, including chronic duodenal ileus, Wilkie’s syndrome, arteriomesenteric duodenal compression syndrome, and cast syndrome[1,2].
Its incidence ranges between 0.005 and 0.3%[1,3-7]. In fluoroscopic studies, the reported incidence is 0.01%[8]. A reported incidence of 0.8% was reported by Neri et al[9] in a prospective study, while other reports mention a frequency of up to 2.4%[10]. In patients with gastric bypass surgery, the reported prevalence is 0.6%[11].
There are approximately 400 cases reported in the literature. SMA syndrome diagnosed after Roux-en-Y gastric bypass has been reported for 14 cases, while one case was reported after sleeve gastrectomy[12].
Despite numerous case reports, no large trials have been published, the diagnosis is often delayed, and patients are treated with ineffective symptomatic therapies[1,13].
The importance of this pathology is derived from its association with significant morbidity and mortality due to a delayed diagnosis. This is why it is considered an important differential diagnosis, especially in a setting of recent weight loss[1]. Additionally, the choice of treatment requires individual evaluation.
We present the case of a 33-year-old female patient diagnosed with Wilkie’s syndrome 15 mo after the onset of symptoms, initially assumed to be anxiety-depressive disorder.
A 33-year-old female patient presented to the emergency department for diffuse, progressive abdominal pain, nausea, bile vomiting, loss of appetite, and weight loss of 15 kg in the last three months. The pain was characteristically located in the epigastrium, with diffuse extension in the rest of the abdomen, after food ingestion.
The symptoms began in the last year, after the initiation of a planned weight loss diet. Over time, her food tolerance gradually decreased, accentuating the symptoms. She presented multiple times to the emergency department with the same symptoms. At every evaluation, an organic cause was excluded, fluid resuscitation was performed, and a psychogenic origin of the symptoms was suspected, given her past medical history.
She was previously diagnosed with Hashimoto thyroiditis and was under levothyroxine replacement therapy at 125 µg/d. After the onset of her symptoms, she was diagnosed with major anxiety-depressive disorder and placed under treatment with alprazolam 0.5 mg, 1 mg/24 h, and escitalopram 10 mg, 25 mg/24 h.
The patient had a history of irregular diets, with attempts to lose weight under normal weight conditions. Her history included surgical repair of bilateral inguinal hernia and an episode of acute pancreatitis of unknown cause.
Physical examination revealed a cachectic patient with an anxious face and a body mass index (BMI) of 17.8 kg/m² (normal level 18.5-24.9 kg/m²) at 45 kg and 159 cm height, with tenderness in the epigastrium.
Laboratory results showed an elevated hemoconcentration [hemoglobin 16.8 g/dL (n.v. 12-15.5 g/dL), hematocrit 48.8% (n.v. 37%-47%)], with hyposodemia [131 mmol/L (n.v. 136-146 mmol/L)], and hypocloremia [91 mmol/L (n.v. 101-109 mmol/L)], which were easily corrected with fluid resuscitation.
Upper digestive endoscopy: Given her symptoms in the superior abdomen, upper digestive endoscopy was performed, and esophagitis and gastritis secondary to bile reflux in the gastric antrum were detected. The first and second parts of the duodenum also had acute erosion and flattened folds. Biopsies were performed to exclude celiac disease and confirm chronic duodenitis. Treatment was initiated with proton pump inhibitor 40 mg, 80 mg/24 h, sucralfate 2 tb/12 h, and prokinetic domperidone 10 mg, 30 mg/24 h. Vitamins were prescribed.
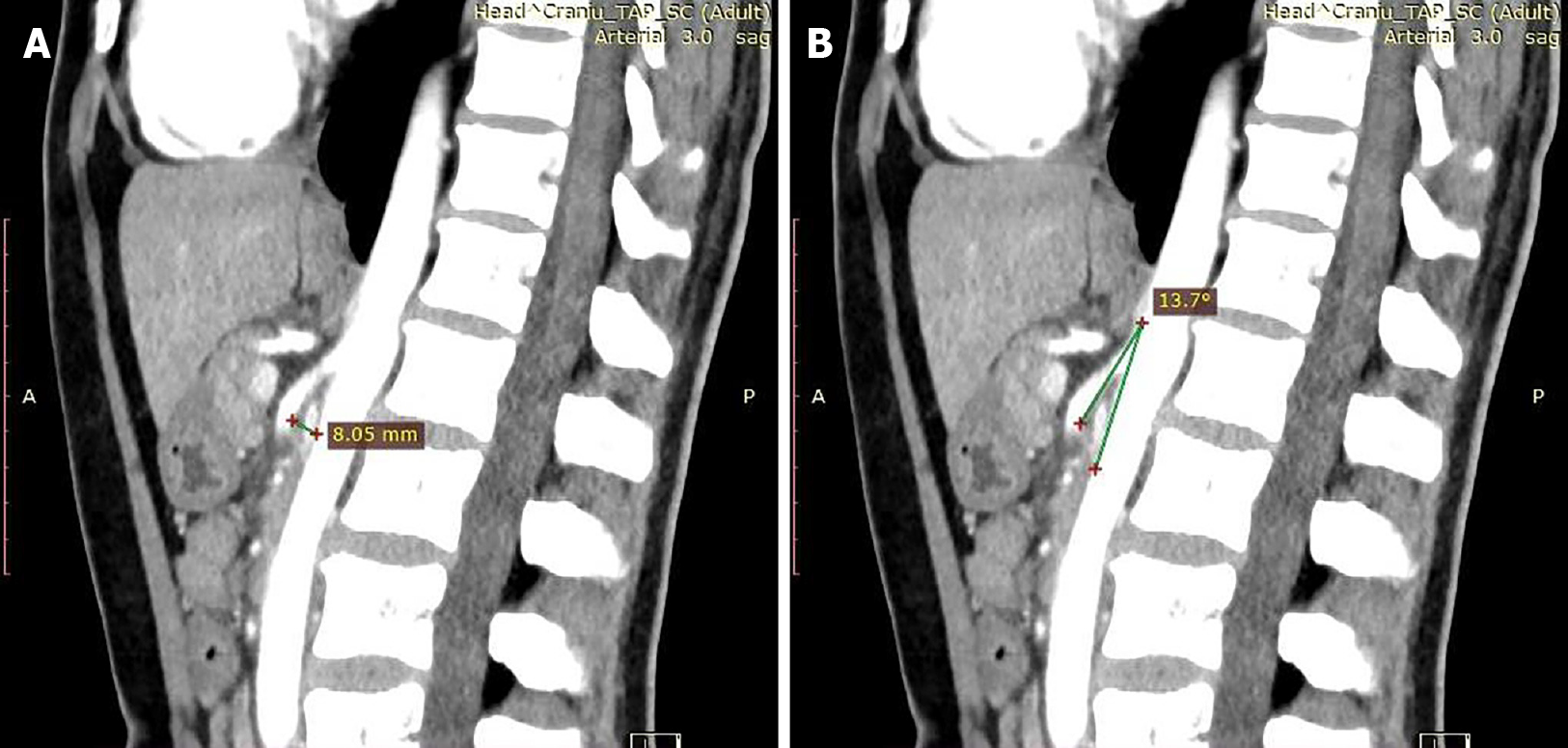
Angio-computed tomography: After one month, given the persistence of the symptoms, computer tomography angiography (Angio-CT) of the abdomen and pelvis was performed. The results revealed emergence at a sharp angle of 13.7° of the superior mesenteric artery, with a reduced distance between the SMA and the anterior wall of the aorta up to a maximum of 8 mm (Figure 1). In this clinical-biological context, a diagnosis of aortomesenteric clamp was established.
A psychiatric consultation was performed, and the diagnosis of major anxiety-depressive disorder was confirmed. Treatment was continued with alprazolam 0.5 mg, 1 mg/24 h and escitalopram 10 mg, 25 mg/24 h, and regular evaluations were scheduled.
The patient presented with a long history (over 1 year) of vomiting, bloating and abdominal pain, symptoms that worsened over the last 3 mo, leading to severe weight loss and the development of a mixed anxiety-depressive disorder. Considering the symptoms, radiologic criteria and failed conservative therapy, we concluded that a surgical approach was the only appropriate treatment.
Given the long period of persisting symptoms, the severity of the weight loss and the absence of major dehydration or electrolyte abnormalities upon admission, due to fluid resuscitation, nutritional and vitamin supplements used in the period before the diagnosis, the patient shows an indication for surgical treatment with laparoscopic duodenojejunostomy.
The angio-CT images helped narrow the differential diagnosis by showing some characteristic features of aortomesenteric clamp without any other morphological changes on CT to suggest other pathological conditions.
Repeated psychiatric evaluations prior to the diagnosis of Wilkie's syndrome revealed the presence of an anxiety-depressive disorder that was attributed to somatic symptoms. These psychiatric symptoms persisted after the somatic disease was clarified. Pre- and postoperative antidepressant and anxiolytic treatment with 10 mg/d escitalopramum and 0.5 mg/d alprazolamum helped to relieve the symptoms. Specialized monitoring of the progression of her emotional state after discharge with re-evaluation of treatment is recommended.
The final preoperative diagnosis of the presented case was Wilkie’s syndrome in a 33-year-old female patient with a secondary anxiety-depressive disorder.
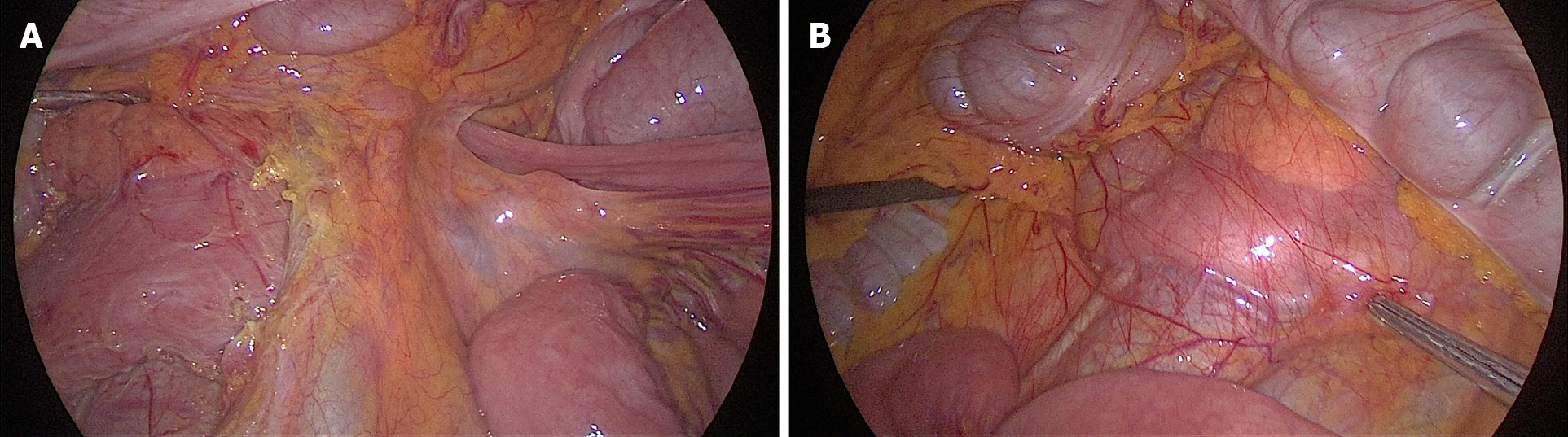
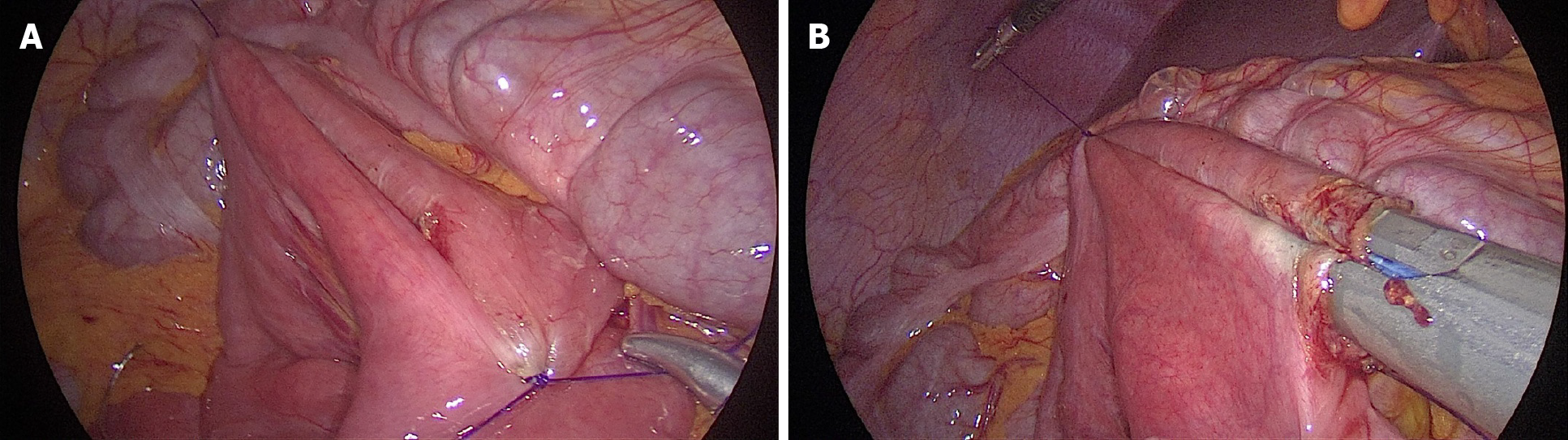
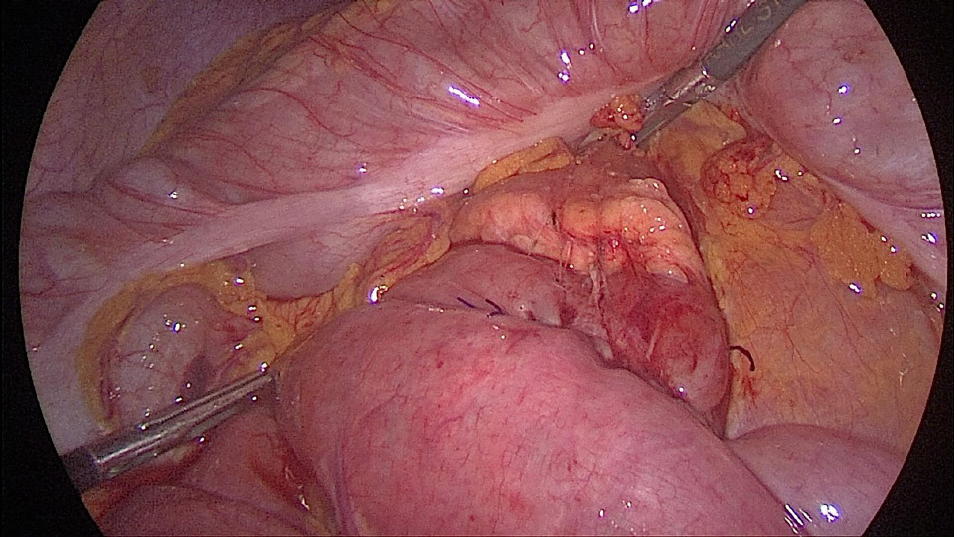
Laparoscopic duodenojejunostomy was performed. A standard surgical technique was followed. After pneumoperitoneum was induced, four ports were placed: A periombilical 10 mm optic port, 5 mm port in the right lower quadrant, 12 mm working port in the left quadrant, midclavicular line, at the level of the umbilicus, and a 5 mm port in the left upper quadrant, below the costal margin. During exploration, a dilated duodenum in the first and second parts was noticed, with a collapsed third part, without ascites or other pathological changes (Figure 2). The second portion of the duodenum was mobilized by a partial Kocher maneuver. At 25 cm distal to the ligament of Treitz, a jejunal loop was prepared and positioned near the duodenum. A side-to-side duodenojejunostomy was performed using a -60 mm endoscopic liner articulated stapler (Figure 3). The common enterotomy was closed with 3-0 polydioxanone absorbable sutures (Figure 4).
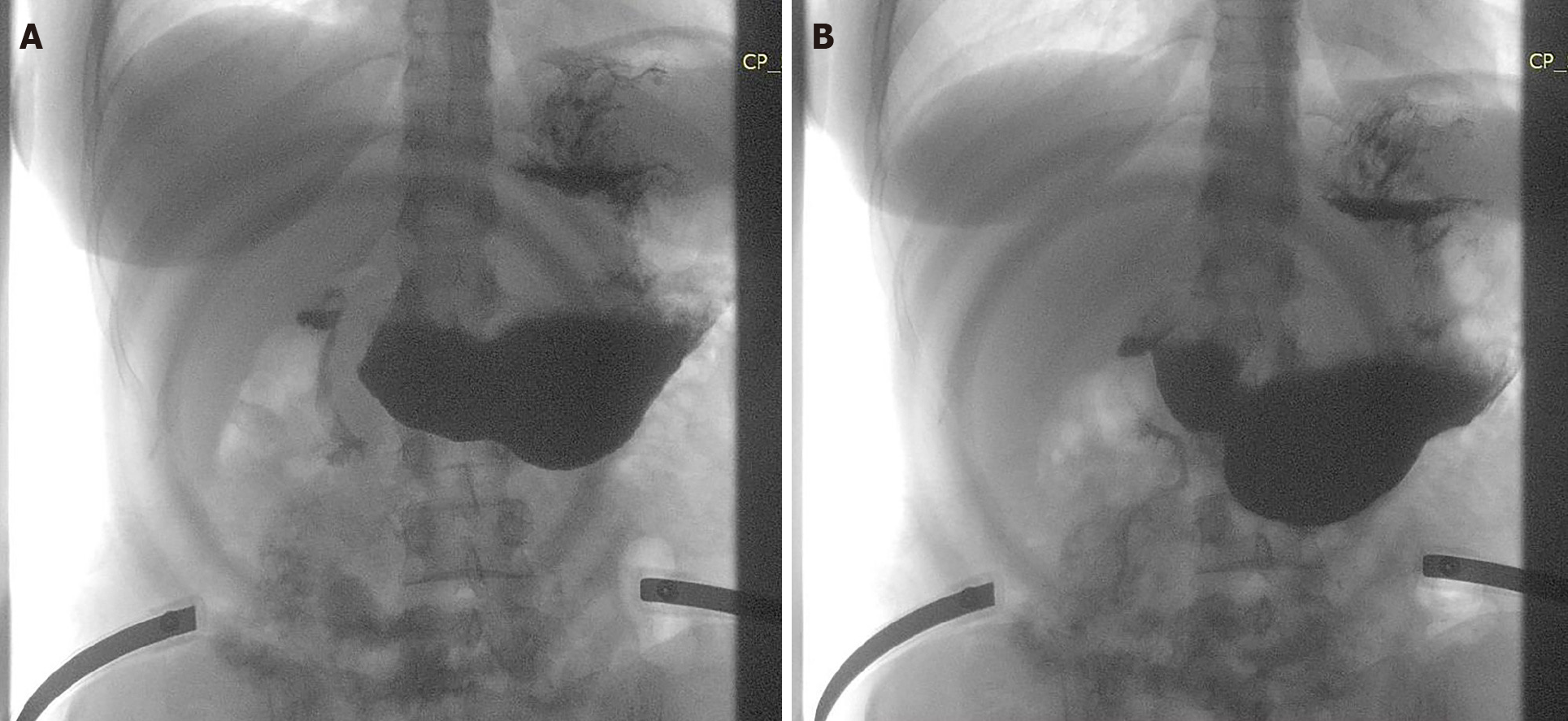
The postoperative course was uneventful, and the patient was discharged home. Nutritional and gastroenterological monitoring was initiated, while psychiatric monitoring was continued. At the 1-mo evaluation, the patient presented a 10 kg weight gain, with complete remission of symptoms (Figure 5). A barium study was conducted showing a free passage of the contrast through the anastomosis (Figure 6).
At the 5-mo follow-up, the patient was asymptomatic, with a 15 kg weight gain. Considering the favorable postoperative outcome and the degree of weight recovery, the patient continued nutritional and psychiatric monitoring, and psychiatric treatment ceased. Long-term evaluation will establish the degree of involvement of psychiatric pathology in the clinical picture, and final conclusions will be drawn.
Superior mesenteric artery syndrome occurs preferentially in young adults and adolescents, with an age range of 10 to 39 years, but cases in 88-year-old patients have also been reported[14]. It occurs more commonly in women, with a 3:2 ratio compared to men[1,4,13].
The primary reported cause of this disease is the loss of the fat pad between the aorta and SMA, resulting in a narrower angle between the vessels and compression of the duodenum[1,12]. This syndrome can be congenital or acquired[1]. Based on this fact, two types of SMA syndrome have been described. The irreversible one (either repeated or permanent) is caused by congenital or developmental factors, such as high insertion of the ligament of Treitz, a low origin of the SMA[5], a short ligament of Treitz, abnormal rotation of the intestines, adhesions due to surgery or duodenal stenosis due to inflammation. Abdominal aortic aneurysms or mesenteric root neoplasms have also been reported to be linked[1,15]. For this type, surgery has been reported as essential for treatment. The reversible type (transient), caused by gravity, for example, can be treated with a conservative approach, including positional changes, intestinal tract decompression and improved nutrition[15]. Furthermore, there might be a genetic component, as suggested by Castro et al[16], while 40% of cases have no identifiable risk factors[10].
A rare association has been reported after inferior vena cava (IVC) filter placement with a local inflammatory response and compression of the duodenum between the IVC and the SMA[17].
The main difficulty in establishing the diagnosis of SMA syndrome resides in the fact that patients tend to be asymptomatic until significant weight loss is registered, secondary to intentional dieting or illness. The most frequently reported cause used to be the corrective treatment of scoliosis, where a relative lengthening of the spine increases the tension on the mesentery and narrows the angle between the vessels[13]. Today, things tend to be different, with the majority of published reports suggesting a very common association with a psychiatric disease[4,13]. SMA syndrome can appear secondary to the disease but can also cause psychological and social problems, including depression and anorexia, due to the severity of the disease[2].
This case report is an example of SMA syndrome as a cause of anxiety-depressive disorder. In this patient, the persistence of symptoms for a long period of time, without identifying an organic cause, led to the development of an anxiety-depressive disorder that was interpreted as the source of the symptoms. The condition’s progression, with the persistence of symptoms despite treatment of the anxiety, triggered extension of the investigations, with the establishment of a final diagnosis.
A characteristic of this disease is significant weight loss, described in different situations as cachexia (AIDS, malabsorption, cancer, paraplegia), hypermetabolism or dietary conditions (anorexia nervosa, drug use). Patients usually register a marked weight loss of > 5 kg[7]. Less frequent reports are incriminating weight loss following surgery, secondary to bariatric surgery, esophagectomy or abdominal trauma, in its etiology[1,12,13,18,19].
The diagnosis is based on symptoms and radiological testing[1]. It needs a high index of suspicion because of its vague and nonspecific symptoms[12]. Additionally, the measurements performed on imaging investigations must also be correlated with the clinical complaints[8], and even this diagnostic approach cannot exclude all motility disorder cases[4]. Thus, a realistic diagnosis can only be established with a comprehensive panel of investigations, a specific history of symptoms and characteristic imaging changes.
The most frequently reported symptoms are abdominal pain (92%), nausea, vomiting (77%), and weight loss (69.2%)[1,4,13,20,21], while in some cases, a predominance of vomiting (70%), nausea (66.3%), abdominal pain (65%), anorexia (33.8%), postprandial fullness (33.8%), and early satiety (12.5%) was reported[3,22]. Another reported characteristic is epigastric pain, which worsens in the supine position[1,4,10]. A sign that can help suggest the diagnosis is the relief of the patient’s symptoms when leaning forward[12] or in the left lateral decubitus position[13]. In our case, weight loss was followed by abdominal pain, nausea, and vomiting, a vicious cycle followed by a marked reduction in weight.
The heterogeneous presentation is aggravated by the presence of comorbidities, such as mental and behavioral disorders (21.3%), infectious disorders (12.5%), and disorders of the nervous system (11.3%)[6].
An acute course of the disease has been described with life-threatening dilatation of the stomach[1]. Despite that, a long-standing onset seems to be more characteristic, with symptom persistence between 6 and 24 mo[7,13], while a time lag of 1 to 51 mo until diagnosis is often described[4,6].
Usually, physical examination and laboratory findings are nonspecific[3]. Despite the marked weight loss, patients have serum albumin and serum proteins within normal limits, while frequent electrolyte abnormalities are encountered[13].
The most commonly used imaging modalities are contrast-enhanced computed tomography and MRA[23]. Computed tomography (CT) angiography is a more specific investigation, while endoscopy and ultrasound are adjunctive diagnostic methods[6].
CT angiography measures the aortomesenteric angle and the distance between the two vessels. A normal aortomesenteric angle is between 25° and 87° with different subintervals in different reports[1-4,8,10,13,17]. The normal reported distance between the two vessels is 10 mm-34 mm[2,3,8,21], confirmed by imaging studies[8]. The mean reported angle measurements are 12°[13] and 13.5°[8], with a mean measured distance of 4.4 mm[8]. However, there are variations regarding the angle dimensions, between 6°-22° in different reports[1,3,4,8,10,13,21], while reported variations regarding distance are between 2 mm and < 10 mm[1-4,8,10,13,21]. It has been suggested that the distance is more accurate for SMA diagnosis than the angle[7].
Cases with SMA syndrome with an angle and distance overlapping the normal range have also been reported. An aorto-mesenteric angle < 8°-46° and a distance < 8 mm have been specifically reported, compared to 25°-87° and 10-34 mm identified in control groups as normal values. In these cases, other diagnostic methods must be considered, such as gastric-emptying scintigraphy[23].
Reports suggest an association of the degree of angulation with (BMI)[10,24]. Alzervi found a significant reduction in the aortomesenteric angle and distance in patients with SMA syndrome compared to other patients with acute or chronic abdominal pain, while BMI was positively correlated with the aortomesenteric angle and distance. Thus, BMI could be used as a screening factor for SMA syndrome[21].
Characteristic CT findings are not just numerical cutoff but also distension of the stomach and proximal duodenum or narrowing of the duodenum at the SMA level. Association with compression of the left renal vein, isolated left renal vein thrombosis, enlargement of the left gonadal vein, or left-sided venous collaterals are additional elements to consider for a positive diagnosis[8].
Rotational CT from the supine to prone position can demonstrate improved outflow passing the SMA[12]. Additionally, CT has replaced magnetic resonance enterography (MRE) as a standard investigation[1,21]. The advantage of showing anatomical variants and consequences, such as delayed transit and obstruction, is highlighted in limited reports using MRE[25].
Diagnosis can also be confirmed by mesenteric artery ultrasonography[12]. Color Doppler ultrasound can also measure the aortomesenteric angle[23].
Endoscopy is useful to diagnose complications, such as esophagitis, reflux gastritis, stasis, and chronic duodenal obstruction[1,13,21]. However, a suspicion of obstruction of the third part of the duodenum is raised only in some cases during endoscopy[13]. A more specific finding for the diagnosis is pulsatile extrinsic compression of the duodenum, excluding other diseases of the superior digestive tract[23].
Barium or gastrografin contrast studies are classic diagnostic procedures, and specific findings are dilatation of the stomach and the first and second parts of the duodenum and the failure of contrast passage in the third part of the duodenum, with antiperistaltic flow[13,23,26]. However, these changes are not specific to SMA syndrome[23].
Gastric-emptying scintigraphy is a known imaging modality to evaluate gastroparesis and gastric motility, and it might be of great value for a differential diagnosis between SMA syndrome and gastric motility disorders, especially in patients with diabetes. This investigation provides a qualitative and quantitative analysis of gastric motility, which might allow differential diagnosis of similar diseases and an assessment of the degree of obstruction or stenosis[23]. Other differentials include internal hernia, adhesive disease, intussusception[12], and megaduodenum[13].
Another difficult situation occurs in cases associated with systemic sclerosis, in which gastrointestinal involvement creates a clinical picture similar to SMA syndrome, with progression toward malnutrition[27]. Another rare condition that has to be differentiated is aortoduodenal syndrome, namely, obstruction of the third portion of the duodenum by a large abdominal aortic aneurysm[28].
If the disease is unrecognized and left untreated, severe complications may occur, such as malnutrition, dehydration, electrolyte abnormalities, gastric pneumatosis and portal venous gas, gastric perforation, duodenal compromise, ischemia, necrosis[1,17], or gastrointestinal hemorrhage[1]. Patients with severe hypokalemia, metabolic alkalosis, and acute kidney injury resulting in cardiorespiratory arrest have also been described[29]. Recognition of the condition, even in advanced stages with massive gastric dilatation and gastric ischemia lesions, can lead to remission of the pathological changes with decompression and endoscopic surveillance[15,30], without the need for surgery.
The management of SMA syndrome is individualized, with either conservative treatment and nutritional support or invasive treatment[3]. Multidisciplinary management is necessary[1]. Given that there is a common association with psychiatric disease or dietary conditions[1,4,13], psychotherapy is an important part of treatment[31], while patients should also be educated on lifestyle changes. Most patients require a dietary consultation for their significant weight loss[1] and a close clinical follow-up by a gastroenterologist and a nutritionist[23].
In the conservative approach, the primary focus should be on weight regain with mass restoration while monitoring the caloric needs with care to avoid refeeding syndrome and recovery of the electrolyte balance[2,5]. Treatment includes nasogastric decompression, prokinetic agents, fluid resuscitation, electrolyte correction, parenteral nutrition, nasogastric tube insertion, nutritional support through hyperalimentation[1], and the removal of precipitating factors. Nutrition may be provided by small, frequent meals or nasojejunal feeding, with the aim of providing a high-calorie diet to increase the mesenteric fat and expand the angle[26]. Positional changes and different maneuvers have also been reported as aids in tolerating enteral feeds, such as the prone position, left lateral decubitus, knee-chest maneuver, or Hayes maneuver. In cases of failure, parenteral nutrition should be considered[5].
Retrospective studies have reported successful treatment of SMA syndrome in short-term follow-up with enteral nutrition[15,17,26,30-34]. After a mean period of 10 mo with enteral therapy, 65% of patients were without symptoms, while in 15%, their symptoms had improved. Nutrition was administered via a nasal jejunal tube and percutaneous endoscopic gastrostomy with a jejunal extension tube. The reported complications were all catheter-related[35].
The success rates for medical management are reported to be between 14% and 83%[6,10], with a 71.3% success rate at a 5-mo follow-up[35]. Predictors of a poor response are a chronic course of disease, prolonged medical treatment, or associated psychosocial disorders[6].
In children, successful treatment was in most reports obtained by the medical approach, with a decline in the need for surgery from 70% to 14%. An acute presentation was most often registered, while weight loss was not a necessary condition. The expected outcome in these cases was excellent[36].
However, as the syndrome progresses, nutritional support becomes less successful, and surgical correction is necessary[17]. Recently, many studies have suggested an earlier surgical approach, since prolonged medical therapy is associated with multiple admissions, a lower success rate, and increased disease recurrence. They recommended no more than 3 mo for a trial of conservative management[6,22].
More than 75% of patients with SMA syndrome require surgical intervention[24]. Surgery usually follows a period of refeeding and the correction of electrolyte disorders. Although significant preoperative weight loss is a known risk factor for postoperative complications, there are no data about an optimal period for nutritional support, either enteral or parenteral, or indications for its use. According to some reports, surgery may be safely performed in the presence of normal serum proteins using preoperative nutritional supplementation on an individual basis. Other reports suggest nasogastric decompression and total parenteral nutrition for 7 d before surgery[2].
Several procedures have been described as a possible surgical treatment for SMA syndrome: duodenal circular drainage, derotation procedure, transposition of the SMA to the infrarenal aorta, anterior transposition of the third part of the duodenum, Billroth II gastrectomy, gastrojejunostomy, Ladd’s procedure, Strong’s operation and transabdominal or laparoscopic duodenojejunostomy[1,6,13]. All of these procedures are associated with nutritional loss, blind loop syndrome, gastric bile reflux[2,5] or a risk of duodenal entrapment between the pancreaticoduodenal arteries and a persistence of the symptoms in 25% of cases after the Strong procedure[5,10,13]. Based on their possible complications, these procedures have all been abandoned, except for duodenojejunostomy[6]. Duodenojejunostomy can significantly improve symptoms, physical activity, emotional well-being, and social functioning[12]. Laparoscopic duodenojejunostomy has the advantages of an acceptable operating time, a faster recovery, reduced postoperative pain, shorter hospitalization, reduced risk of incisional hernia, and a good cosmetic outcome[1,4,6,10,16,26], which makes it important in young patients with associated psychosocial symptoms secondary to the severity of their disease[2]. Blind loop syndrome is the most common complication after this type of procedure[13].
To close the common enterotomy, a mini laparotomy and a manual suture were performed in most of the reported cases to avoid a narrowed anastomosis. An alternative to this approach is to close the enterotomy with a laparoscopic suture or a linear stapler in a complete laparoscopic maneuver[26], as was done in our patient.
The success rate of surgical treatment is reported to be as high as 75%-100%[1,4,8,10,16]. Minimally invasive procedures, including robotic and single-port approaches[37], have reported a success rate of more than 90% with a 7% complication rate[6], while laparoscopic duodenojejunostomy alone has a success rate of 96%[1,10].
In Lee et al[22]’s case series, where half of the patients presented with associated diseases, a medical treatment success rate of 71.3%, with a 15.8% recurrence rate, was reported, while surgical management had a 92.9% success rate.
The postoperative course is marked by a major weight gain, usually registered during the first postoperative 6 mo with a mean gain between 5 and 15 kg[13].
Intermediate follow-up results were reported by Chang et al[4] for 26 patients treated with laparoscopic or robotic duodenojejunostomy, with the majority of cases secondary to psychiatric disease. Only 33.3% declared resolution or improvement of their symptoms, while 30% of patients were still severely symptomatic, with 19% of patients still requiring nutritional supplementation. Recurrence was associated with a patent anastomosis and the concomitant presence of dysmotility disorders, such as gastroparesis or global intestinal dysmotility, with an indication for intestinal transplant[4]. An important conclusion was that radiographic findings of SMA syndrome should not automatically be assumed to exclude intestinal dysmotility syndrome[4,20]. Given the overlapping symptomatology, gastrointestinal transit studies should always be included in the preoperative workup[4].
Although there are several reports of SMA syndrome, diagnosed as a complication of anorexia nervosa, only a few of them emphasize the evolution of psychiatric disease. Kurisu et al[38] pointed out that surgical treatment must be cautiously considered in cases of anorexia nervosa, as the long-term results are unclear. They also reported the case of a patient with anorexia nervosa and SMA syndrome treated with surgery, without postoperative improvement. In this case, a psychological approach was necessary to be continued[38]. On the other hand, Kornmehl et al[39] emphasized the overdiagnosis of anorexia nervosa, while SMA syndrome should be considered in these cases. The novelty of our case is represented by the absence of a history of psychiatric diseases, a diagnosis of the disorder after the onset of symptoms, while a restoration of her body weight was registered postoperatively, and psychiatric treatment was stopped.
In their case series, Sun et al[4] reported a mean weight gain of 3.8 kg at mid-term follow-up, with reported complications such as infection and dumping syndrome.
Cienfuegos et al[20] presented long-term results (median follow-up of 94 mo) after laparoscopic latero-lateral duodenojejunostomy, with excellent results in 61.5% of cases. Long-term results were also evaluated by Jain et al[6] with weight gain, complete symptom remission, and no recurrence.
Even though this pathology is rarely described in elderly patients, for this category and in those with significant comorbidities, endoscopic ultrasound-guided gastroenterostomy (EUS-GE) was reported as an appropriate procedure[14,40]. EUS-GE using lumen-apposing metal stents seems to be a safe and effective option[40], with a success rate of 90%, adverse events in 5% of patients and a reintervention rate of 11%[41]. A rate of 83% in preventing surgery was reported, with a mean period of 8.5 mo until symptom resolution and removal of the LAMS and a 5.6% rate of recurrent symptoms[42]. A comparison between EUS gastrojejunostomy and laparoscopic gastrojejunostomy was performed, showing a similar success rate and a significantly higher rate of complications for the surgical procedure[43-45].
In addition to the specificity of this complex pathology, some rare associations have been reported in the literature. One of them is the nutcracker syndrome[13,31]. The most commonly reported causes of this rare syndrome are anatomical variants such as posterior renal ptosis, a high course of the left renal vein and abnormal SMA branching, but the two syndromes can coexist in the context of major weight loss. Treatment for nutcracker syndrome varies from conservative management to different surgical procedures, with the best results reported after laparoscopic extravascular titanium stent placement[5].
In a small number of cases, an association with cystic fibrosis has been reported. In these cases, BMI below the 5th percentile is the best predictor of SMA syndrome. The diagnosis is difficult because of the risk of cystic fibrosis enteropathy[24]. Other rare associations are hyperthyroidism-related sympathetic hyperstimulation, vasculopathy and cerebellar infarction[46] and diabetic patients with excessive body weight loss and associated ketoacidosis after using sodium-glucose cotransporter 2 inhibitors[47]. Rare associations with rheumatoid arthritis and rheumatoid cachexia have also been reported[48].
This case report highlights the importance of SMA syndrome, especially regarding the consequences when left undiagnosed and untreated for a long period of time, as well as the numerous possibilities of its overlapping with other functional diseases or even being the cause of their onset.
The major limitation of this case report is the short follow-up, which allowed us to register the resolution of the anxiety disorder. Only a long-term follow-up will confirm that the syndrome can be a cause for the psychiatric disorder and not the other way around.
SMA syndrome is a rare occlusive pathology that needs an extensive diagnostic workup since it is hampered by a specific mimicry of psychiatric or motility disorders. The vicious cycle of its progression can be interrupted by knowing and considering the large number of differential diagnostic possibilities while individualizing the treatment. Since favorable results depend on the treatment of multiple systems, multidisciplinary management is mandatory.
Provenance and peer review: Unsolicited article; Externally peer reviewed.
Peer-review model: Single blind
Specialty type: Surgery
Country/Territory of origin: Romania
Peer-review report’s scientific quality classification
Grade A (Excellent): 0
Grade B (Very good): 0
Grade C (Good): C
Grade D (Fair): 0
Grade E (Poor): 0
P-Reviewer: Peltrini R S-Editor: Ma YJ L-Editor: A P-Editor: Ma YJ
| 1. | Van Horne N, Jackson JP. Superior mesenteric artery syndrome. In: StatPearls [Internet]. Treasure Island (FL): StatPearls Publishing; 2021 Jul 21. [PubMed] |
| 2. | Barkhatov L, Tyukina N, Fretland ÅA, Røsok BI, Kazaryan AM, Riis R, Edwin B. Superior mesenteric artery syndrome: quality of life after laparoscopic duodenojejunostomy. Clin Case Rep. 2018;6:323-329. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 11] [Cited by in RCA: 18] [Article Influence: 2.3] [Reference Citation Analysis (0)] |
| 3. | Lima Silva A, Antunes D, Cordeiro E Cunha J, Nogueira R, Fernandes D, Salazar T, Madureira Pinto C. Epigastric Pain and Weight Loss - A Case of Wilkie's Syndrome. Eur J Case Rep Intern Med. 2020;7:001557. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 1] [Cited by in RCA: 2] [Article Influence: 0.4] [Reference Citation Analysis (0)] |
| 4. | Chang J, Boules M, Rodriguez J, Walsh M, Rosenthal R, Kroh M. Laparoscopic duodenojejunostomy for superior mesenteric artery syndrome: intermediate follow-up results and a review of the literature. Surg Endosc. 2017;31:1180-1185. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 17] [Cited by in RCA: 21] [Article Influence: 2.3] [Reference Citation Analysis (0)] |
| 5. | Diab S, Hayek F. Combined Superior Mesenteric Artery Syndrome and Nutcracker Syndrome in a Young Patient: A Case Report and Review of the Literature. Am J Case Rep. 2020;21:e922619. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 6] [Cited by in RCA: 21] [Article Influence: 4.2] [Reference Citation Analysis (0)] |
| 6. | Jain N, Chopde A, Soni B, Sharma B, Saini S, Mishra S, Gupta R, Bhojwani R. SMA syndrome: management perspective with laparoscopic duodenojejunostomy and long-term results. Surg Endosc. 2021;35:2029-2038. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 9] [Cited by in RCA: 14] [Article Influence: 2.8] [Reference Citation Analysis (0)] |
| 7. | Sinagra E, Raimondo D, Albano D, Guarnotta V, Blasco M, Testai S, Marasà M, Mastrella V, Alaimo V, Bova V, Albano G, Sorrentino D, Tomasello G, Cappello F, Leone A, Rossi F, Galia M, Lagalla R, Midiri F, Morreale GC, Amvrosiadis G, Martorana G, Spampinato MG, Virgilio V, Midiri M. Superior Mesenteric Artery Syndrome: Clinical, Endoscopic, and Radiological Findings. Gastroenterol Res Pract. 2018;2018:1937416. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 30] [Cited by in RCA: 25] [Article Influence: 3.6] [Reference Citation Analysis (0)] |
| 8. | Raman SP, Neyman EG, Horton KM, Eckhauser FE, Fishman EK. Superior mesenteric artery syndrome: spectrum of CT findings with multiplanar reconstructions and 3-D imaging. Abdom Imaging. 2012;37:1079-1088. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 25] [Cited by in RCA: 30] [Article Influence: 2.3] [Reference Citation Analysis (0)] |
| 9. | Neri S, Signorelli SS, Mondati E, Pulvirenti D, Campanile E, Di Pino L, Scuderi M, Giustolisi N, Di Prima P, Mauceri B, Abate G, Cilio D, Misseri M, Scuderi R. Ultrasound imaging in diagnosis of superior mesenteric artery syndrome. J Intern Med. 2005;257:346-351. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 100] [Cited by in RCA: 114] [Article Influence: 5.7] [Reference Citation Analysis (0)] |
| 10. | Martínez H, Martínez S, Sánchez-Ussa S, Pedraza M, Cabrera LF. Laparoscopic management for Wilkie´s syndrome. Cir Cir. 2019;87:22-27. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 2] [Cited by in RCA: 2] [Article Influence: 0.3] [Reference Citation Analysis (0)] |
| 11. | Chrysikos D, Troupis T, Tsiaoussis J, Sgantzos M, Bonatsos V, Karampelias V, Piperos T, Kalles V, Theodoropoulos P, Kakaviatos D, Flessas I, Nikou E, Mariolis-Sapsakos T. Superior mesenteric artery syndrome: a rare case of upper gastrointestinal obstruction. J Surg Case Rep. 2019;2019:rjz054. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 4] [Cited by in RCA: 6] [Article Influence: 1.0] [Reference Citation Analysis (0)] |
| 12. | Andrew BD, Hamed AB, Gourash W, Ahmed BH. Laparoscopic duodenojejunostomy to manage small bowel obstruction due to superior mesenteric artery syndrome after Roux-en-Y gastric bypass. Surg Obes Relat Dis. 2021;17:242-244. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 2] [Cited by in RCA: 1] [Article Influence: 0.2] [Reference Citation Analysis (0)] |
| 13. | Merrett ND, Wilson RB, Cosman P, Biankin AV. Superior mesenteric artery syndrome: diagnosis and treatment strategies. J Gastrointest Surg. 2009;13:287-292. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 253] [Cited by in RCA: 204] [Article Influence: 12.8] [Reference Citation Analysis (0)] |
| 14. | Bronswijk M, Fransen L, Vanella G, Hiele M, van der Merwe S. Successful treatment of superior mesenteric artery syndrome by endoscopic ultrasound-guided gastrojejunostomy. Endoscopy. 2021;53:204-205. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 8] [Cited by in RCA: 15] [Article Influence: 3.8] [Reference Citation Analysis (0)] |
| 15. | Furuya Y, Wakahara T, Furuya A, Yanagie H, Yasuhara H. Rare bowel emphysema with superior mesenteric artery syndrome after surgery. Ann R Coll Surg Engl. 2020;102:e26-e28. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 1] [Cited by in RCA: 1] [Article Influence: 0.2] [Reference Citation Analysis (0)] |
| 16. | Aneiros Castro B, Cano Novillo I, García Vázquez A, Martín Alelu R, Gómez Fraile A. Wilkie's syndrome in monozygotic twins treated by 3-D laparoscopic duodenojejunostomy. Asian J Endosc Surg. 2019;12:125-127. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 6] [Cited by in RCA: 5] [Article Influence: 0.8] [Reference Citation Analysis (0)] |
| 17. | Haidar A, Davies A, Hussain A, Gregerson S, Thammineni D, Markus J. Wilkie's Weight Loss Wonder, A Case Series. Spartan Med Res J. 2020;5:13485. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 1] [Cited by in RCA: 1] [Article Influence: 0.2] [Reference Citation Analysis (0)] |
| 18. | Haider A, Sharma M, Siddiqa A. Superior Mesenteric Artery Syndrome: A Forgotten Cause of Duodenal Obstruction. Cureus. 2020;12:e10710. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 2] [Cited by in RCA: 2] [Article Influence: 0.4] [Reference Citation Analysis (0)] |
| 19. | Wong LH, Sutton TL, Spurrier RG, Zigman AF, Mayo SC. Post-Operative Superior Mesenteric Artery Syndrome Following Retroperitoneal Sarcoma Resection. Clin Pract. 2020;11:2-7. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 2] [Cited by in RCA: 4] [Article Influence: 0.8] [Reference Citation Analysis (0)] |
| 20. | Cienfuegos JA, Hurtado-Pardo L, Valentí V, Landecho MF, Vivas I, Estévez MG, Diez-Caballero A, Hernández-Lizoáin JL, Rotellar F. Minimally Invasive Surgical Approach for the Treatment of Superior Mesenteric Artery Syndrome: Long-Term Outcomes. World J Surg. 2020;44:1798-1806. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 6] [Cited by in RCA: 2] [Article Influence: 0.4] [Reference Citation Analysis (0)] |
| 21. | Alzerwi NAN. Predictors of Superior Mesenteric Artery Syndrome: Evidence from a Case-Control Study. Cureus. 2020;12:e9715. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 2] [Cited by in RCA: 1] [Article Influence: 0.2] [Reference Citation Analysis (0)] |
| 22. | Lee TH, Lee JS, Jo Y, Park KS, Cheon JH, Kim YS, Jang JY, Kang YW. Superior mesenteric artery syndrome: where do we stand today? J Gastrointest Surg. 2012;16:2203-2211. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 65] [Cited by in RCA: 81] [Article Influence: 6.2] [Reference Citation Analysis (0)] |
| 23. | Kang S, Choi BW. Clinical Usefulness of Gastric-emptying Scintigraphy in Superior Mesenteric Artery Syndrome: A Case Report. Nuklearmedizin. 2020;59:335-337. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 1] [Cited by in RCA: 1] [Article Influence: 0.2] [Reference Citation Analysis (0)] |
| 24. | Schwarz J, Sýkora J, Pomahačová R, Sýkorová A, Fremuth J, Šašek L, Vondráková R, Kreslová M. Rare cause of upper bowel obstruction arising in a 17-year-old boy with cystic fibrosis: Superior mesenteric artery (Wilkie's) syndrome. J Paediatr Child Health. 2020;56:1827-1829. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 1] [Cited by in RCA: 1] [Article Influence: 0.2] [Reference Citation Analysis (0)] |
| 25. | Cicero G, D'Angelo T, Bottari A, Costantino G, Visalli C, Racchiusa S, Marino MA, Cavallaro M, Frosina L, Blandino A, Mazziotti S. Superior Mesenteric Artery Syndrome in Patients with Crohn's Disease: A Description of 2 Cases Studied with a Novel Magnetic Resonance Enterography (MRE) Procedure. Am J Case Rep. 2018;19:431-437. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 13] [Cited by in RCA: 18] [Article Influence: 2.6] [Reference Citation Analysis (0)] |
| 26. | Yoneda A, Kanetaka K, Yamaguchi S, Koga Y, Isagawa Y, Maruya Y, Inoue Y, Torashima Y, Adachi T, Hidaka M, Kobayashi K, Ito S, Eguchi S. Complete laparoscopic duodenojejunostomy for superior mesenteric artery syndrome: Linear stapled closure of the common enterotomy. Asian J Endosc Surg. 2020;13:552-555. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 2] [Cited by in RCA: 2] [Article Influence: 0.4] [Reference Citation Analysis (0)] |
| 27. | Chua CG, Wansaicheong GK, Lim WC, Thong BY. Superior Mesenteric Artery Syndrome: A Potentially Fatal but Reversible Gastrointestinal Manifestation of Systemic Sclerosis. Case Rep Rheumatol. 2020;2020:8831417. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 1] [Cited by in RCA: 1] [Article Influence: 0.2] [Reference Citation Analysis (0)] |
| 28. | Parashar K, Gandhi D, Nepal P, Sapire J, Ahuja K, Siddiqui I. Abdominal aortic aneurysm and its association with duodenal obstruction: aortoduodenal syndrome. BJR Case Rep. 2020;6:20200040. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 1] [Cited by in RCA: 2] [Article Influence: 0.4] [Reference Citation Analysis (0)] |
| 29. | Mohammad Kazmin NE, Kamaruzaman L, Wong Z, Fong VK, Mohd R, Mustafar R. Acute Kidney Injury Caused by Superior Mesenteric Artery Syndrome. Case Rep Nephrol. 2020;2020:8364176. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 2] [Cited by in RCA: 4] [Article Influence: 0.8] [Reference Citation Analysis (0)] |
| 30. | Sakurai Y, Hirai F, Abe M, Okaya T, Suzuki H, Sugano I. A case of gastric ischemia caused by massive gastric dilatation due to superior mesenteric artery syndrome. Clin J Gastroenterol. 2020;13:1066-1069. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 1] [Cited by in RCA: 5] [Article Influence: 1.0] [Reference Citation Analysis (0)] |
| 31. | Lin TH, Lin CC, Tsai JD. Superior mesenteric artery syndrome and nutcracker syndrome. Pediatr Neonatol. 2020;61:351-352. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 3] [Cited by in RCA: 3] [Article Influence: 0.6] [Reference Citation Analysis (0)] |
| 32. | Russell EA, Braverman RM, Vasudevan SA, Patel B. A Traumatic Quinceañera: Acute Superior Mesenteric Artery Syndrome in an Adolescent Girl. Pediatr Emerg Care. 2021;37:e203-e205. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 2] [Cited by in RCA: 2] [Article Influence: 0.5] [Reference Citation Analysis (0)] |
| 33. | Ren PLJ, Gupta A. Adolescent with Superior Mesenteric Artery Syndrome. J Radiol Case Rep. 2020;14:14-23. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 2] [Cited by in RCA: 5] [Article Influence: 1.0] [Reference Citation Analysis (0)] |
| 34. | Dimopoulou A, Zavras N, Alexopoulou E, Fessatou S, Dimopoulou D, Attilakos A. Superior mesenteric artery syndrome mimicking cyclic vomiting syndrome in a healthy 12-year-old boy. J Paediatr Child Health. 2020;56:168-170. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 3] [Cited by in RCA: 6] [Article Influence: 1.2] [Reference Citation Analysis (0)] |
| 35. | Wan S, Zhang L, Yang J, Gao X, Wang X. Superior Mesenteric Artery Syndrome Improved by Enteral Nutritional Therapy: A Retrospective Case-Series Study in a Single Institution. Ann Nutr Metab. 2020;76:37-43. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 3] [Cited by in RCA: 7] [Article Influence: 1.4] [Reference Citation Analysis (0)] |
| 36. | Biank V, Werlin S. Superior mesenteric artery syndrome in children: a 20-year experience. J Pediatr Gastroenterol Nutr. 2006;42:522-525. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 102] [Cited by in RCA: 121] [Article Influence: 6.4] [Reference Citation Analysis (0)] |
| 37. | Konstantinidis H, Charisis C, Kottos P. Robotic Strong's procedure for the treatment of superior mesenteric artery syndrome. Description of surgical technique on occasion of the first reported case in the literature. Int J Med Robot. 2018;14. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 4] [Cited by in RCA: 8] [Article Influence: 1.0] [Reference Citation Analysis (0)] |
| 38. | Kurisu K, Yamanaka Y, Yamazaki T, Yoneda R, Otani M, Takimoto Y, Yoshiuchi K. A clinical course of a patient with anorexia nervosa receiving surgery for superior mesenteric artery syndrome. J Eat Disord. 2021;9:79. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 1] [Cited by in RCA: 4] [Article Influence: 1.0] [Reference Citation Analysis (0)] |
| 39. | Kornmehl P, Weizman Z, Liss Z, Bar-Ziv J, Joseph A. Superior mesenteric artery syndrome presenting as an anorexia nervosa-like illness. J Adolesc Health Care. 1988;9:340-343. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 13] [Cited by in RCA: 13] [Article Influence: 0.4] [Reference Citation Analysis (0)] |
| 40. | Kouanda A, Watson R, Binmoeller KF, Nett A, Hamerski C. EUS-guided gastroenterostomy for duodenal obstruction secondary to superior mesenteric artery syndrome. VideoGIE. 2021;6:14-15. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 1] [Cited by in RCA: 6] [Article Influence: 1.5] [Reference Citation Analysis (0)] |
| 41. | McCarty TR, Garg R, Thompson CC, Rustagi T. Efficacy and safety of EUS-guided gastroenterostomy for benign and malignant gastric outlet obstruction: a systematic review and meta-analysis. Endosc Int Open. 2019;7:E1474-E1482. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 46] [Cited by in RCA: 72] [Article Influence: 12.0] [Reference Citation Analysis (0)] |
| 42. | James TW, Greenberg S, Grimm IS, Baron TH. EUS-guided gastroenteric anastomosis as a bridge to definitive treatment in benign gastric outlet obstruction. Gastrointest Endosc. 2020;91:537-542. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 31] [Cited by in RCA: 55] [Article Influence: 11.0] [Reference Citation Analysis (1)] |
| 43. | Tyberg A, Perez-Miranda M, Sanchez-Ocaña R, Peñas I, de la Serna C, Shah J, Binmoeller K, Gaidhane M, Grimm I, Baron T, Kahaleh M. Endoscopic ultrasound-guided gastrojejunostomy with a lumen-apposing metal stent: a multicenter, international experience. Endosc Int Open. 2016;4:E276-E281. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 174] [Cited by in RCA: 179] [Article Influence: 19.9] [Reference Citation Analysis (0)] |
| 44. | Perez-Miranda M, Tyberg A, Poletto D, Toscano E, Gaidhane M, Desai AP, Kumta NA, Fayad L, Nieto J, Barthet M, Shah R, Brauer BC, Sharaiha RZ, Kahaleh M. EUS-guided Gastrojejunostomy Versus Laparoscopic Gastrojejunostomy: An International Collaborative Study. J Clin Gastroenterol. 2017;51:896-899. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 118] [Cited by in RCA: 170] [Article Influence: 21.3] [Reference Citation Analysis (1)] |
| 45. | Xu MM, Dawod E, Gaidhane M, Tyberg A, Kahaleh M. Reverse Endoscopic Ultrasound-Guided Gastrojejunostomy for the Treatment of Superior Mesenteric Artery Syndrome: A New Concept. Clin Endosc. 2020;53:94-96. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 5] [Cited by in RCA: 11] [Article Influence: 1.8] [Reference Citation Analysis (0)] |
| 46. | Wang HK, Huang WH, Chen KT. An extensive posterior circulation infarction secondary to primary hyperthyroidism accompanied with superior mesenteric artery syndrome: A case report and description of patho-physiological association. Medicine (Baltimore). 2020;99:e22664. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 2] [Cited by in RCA: 2] [Article Influence: 0.4] [Reference Citation Analysis (0)] |
| 47. | Hirai T, Kitada M, Hayashi Y, Monno I, Takagaki Y, Shimada K, Ogura Y, Fujii M, Konishi K, Nakagawa A, Koya D. Case report of superior mesenteric artery syndrome that developed in a lean type 2 diabetes patient and was associated with rapid body weight loss after sodium-glucose cotransporter 2 inhibitor administration. J Diabetes Investig. 2020;11:1359-1362. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 3] [Cited by in RCA: 3] [Article Influence: 0.6] [Reference Citation Analysis (0)] |
| 48. | Galarza-Delgado DÁ, Flores-Alvarado DE, Compeán-Villegas JE. Superior mesenteric artery syndrome in a patient with rheumatoid arthritis and rheumatoid cachexia during the COVID-19 pandemic. Clin Rheumatol. 2021;40:2095-2096. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 1] [Cited by in RCA: 1] [Article Influence: 0.3] [Reference Citation Analysis (0)] |