Published online Dec 26, 2022. doi: 10.12998/wjcc.v10.i36.13364
Peer-review started: August 16, 2022
First decision: September 5, 2022
Revised: September 17, 2022
Accepted: December 5, 2022
Article in press: December 5, 2022
Published online: December 26, 2022
Processing time: 132 Days and 15.9 Hours
Solitary plasmacytoma and unicentric Castleman disease (UCD) are rare lymphoproliferative disorders characterized by monoclonal plasma cells and a single set of locally enlarged lymph nodes, respectively.
A 48-year-old Han Chinese man presented to our department with a neck mass and progressive foreign body sensation in his throat. 18F-FDG positron emission tomography revealed focally increased radioactivity centered around the hyoid, and computed tomography (CT) revealed osteolytic lesions. Histopathology revealed Castleman-like features and CD138/CD38-positive mature plasma cells. Systemic work-up ruled out the possibility of POEMS syndrome, lymphoma, and multiple myeloma, leading to a final diagnosis of solitary hyoid plasmacytoma with UCD. The patient underwent partial hyoid resection and selective neck dissection, followed by intensity-modulated radiotherapy. 99mTc-MDP single-photon emission computed tomography/CT reevaluation showed neither local recurrence nor distant bone metastasis at the 40-mo follow-up.
The diagnostic process and differential diagnosis of this rare case provided valuable educational information to clinicians.
Core Tip: In this study, we described the clinical presentations, diagnosis and differential diagnosis, management, outcome, and relative long-term follow-up for a man simultaneously affected with solitary plasmacytoma (SP) of the hyoid bone and unicentric Castleman disease. To our knowledge, this is the first reported case presented with these two rare clinical entities. Besides the comprehensive histopathological examination, 18F-FDG positron emission tomography/computed tomography (CT) and 99mTc-MDP single-photon emission computed tomography (SPECT)/CT were broadly used in our clinical practice of differential diagnosis, disease staging, and follow-up monitoring, highlighting the advantages of nuclear medicine and other imaging techniques in the management of SP in the head and neck. Additionally, the diagnostic process and differential diagnosis of this patient provided valuable educational information to clinicians.
- Citation: Zhang YH, He YF, Yue H, Zhang YN, Shi L, Jin B, Dong P. Solitary hyoid plasmacytoma with unicentric Castleman disease: A case report and review of literature. World J Clin Cases 2022; 10(36): 13364-13372
- URL: https://www.wjgnet.com/2307-8960/full/v10/i36/13364.htm
- DOI: https://dx.doi.org/10.12998/wjcc.v10.i36.13364
Solitary plasmacytoma (SP) is a rare disease characterized by abnormal proliferation of monoclonal plasma cells, with a cumulative incidence of 0.15/100000[1,2]. Solitary bone plasmacytoma (SBP) is a subtype of SP that occurs primarily in the red-marrow-containing axial skeleton and long bones[3] and accounts for 70% of all SP cases[1]. SBP with minimal (< 10%) bone marrow plasma cells progresses to multiple myeloma (MM) within 3 years in approximately 60% of patients[2,4]. All SP are radiation-sensitive; therefore, radiotherapy (RT) is recommended as the first-line treatment strategy for the long-term disease control in these patients[3,5].
Unicentric Castleman disease (UCD) is a rare lymphoproliferative disorder with unknown prevalence and belongs to a heterogeneous group of conditions characterized by similar histopathological features but has distinct pathological etiologies, clinical symptoms, management, and outcomes[6,7]. For patients with resectable UCD, complete surgical excision is the optimal first-line treatment, with a 5-year overall survival of up to 90%; even after a 10-year follow-up, disease relapse is rare[6,8].
Here, we report the case of a 48-year-old man with SBP of the hyoid and UCD along with the clinical diagnosis, management process, and follow-up. Moreover, we reviewed the literature of related SBP cases to provide a comparative experience in guiding the management of this rare disease.
A 48-year-old Han Chinese man presented with a neck mass and progressive foreign body sensation in his throat.
The patient reported no sore throat, dyspnea, or dysphagia during the course of the disease.
The patient had no history of past illness.
The patient had no personal or family history.
Basic physical examination showed no obvious abnormalities.
Laboratory tests showed that the routine blood and urine results as well as blood biochemical indices, including serum calcium-albumin-creatinine, blood urea nitrogen, lactate dehydrogenase, β2-microglobulin, and 24-hour urine for total protein, moreover, the results of serum immunoglobulin and complement assay (Supplementary Table 1), were all within normal limits.
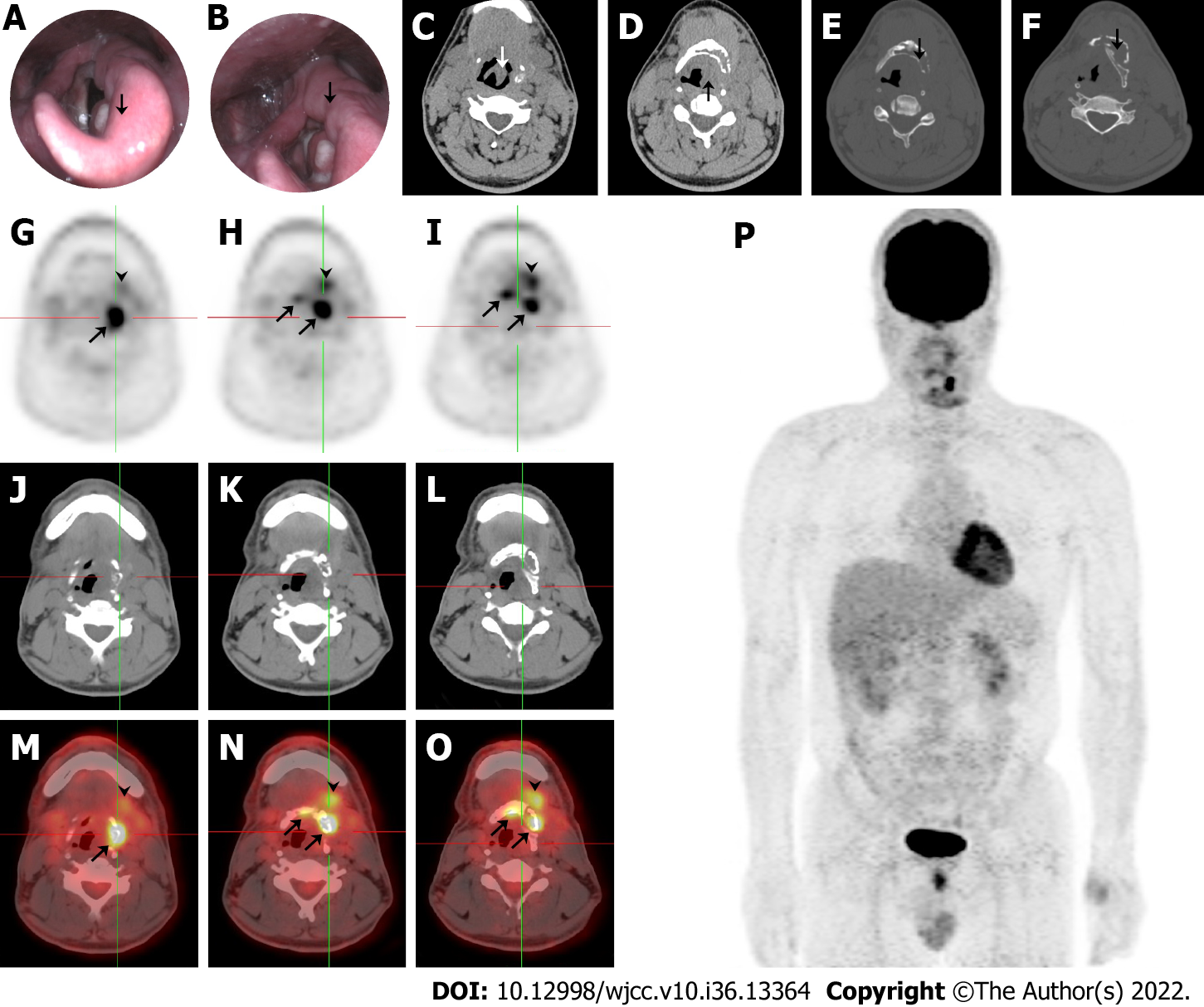
Electronic laryngoscopy and plain computed tomography (CT) showed thickening of the left epiglottis (Figure 1A and C), arytenoid and aryepiglottic folds (Figure 1B and D), osteolytic lesions of the left hyoid (Figure 1E), and thyroid cartilage plate (Figure 1F). The left piriform fossa was occluded, and enlarged cervical and submandibular lymph nodes were observed bilaterally. The patient underwent selective neck dissection and partial hyoid resection to relieve neck compression, and a final diagnosis was made. 18F-FDG positron emission tomography (PET)/CT revealed focally increased radioactivity centered around the hyoid body and its left greater horn (SUVmax = 8.8) and the left submandibular and submental soft tissue (SUVmax = 4.8) exhibited on PET (Figure 1G-I), CT (Figure 1J-L), and PET/CT fusion (Figure 1M-O). Osteolytic lesions were identified on the plain CT bone window. PET maximum-intensity projection image (Figure 1P) showed no other sites with abnormal 18F-FDG uptake, except for the perihyoid region. These findings were suggestive of a malignancy.
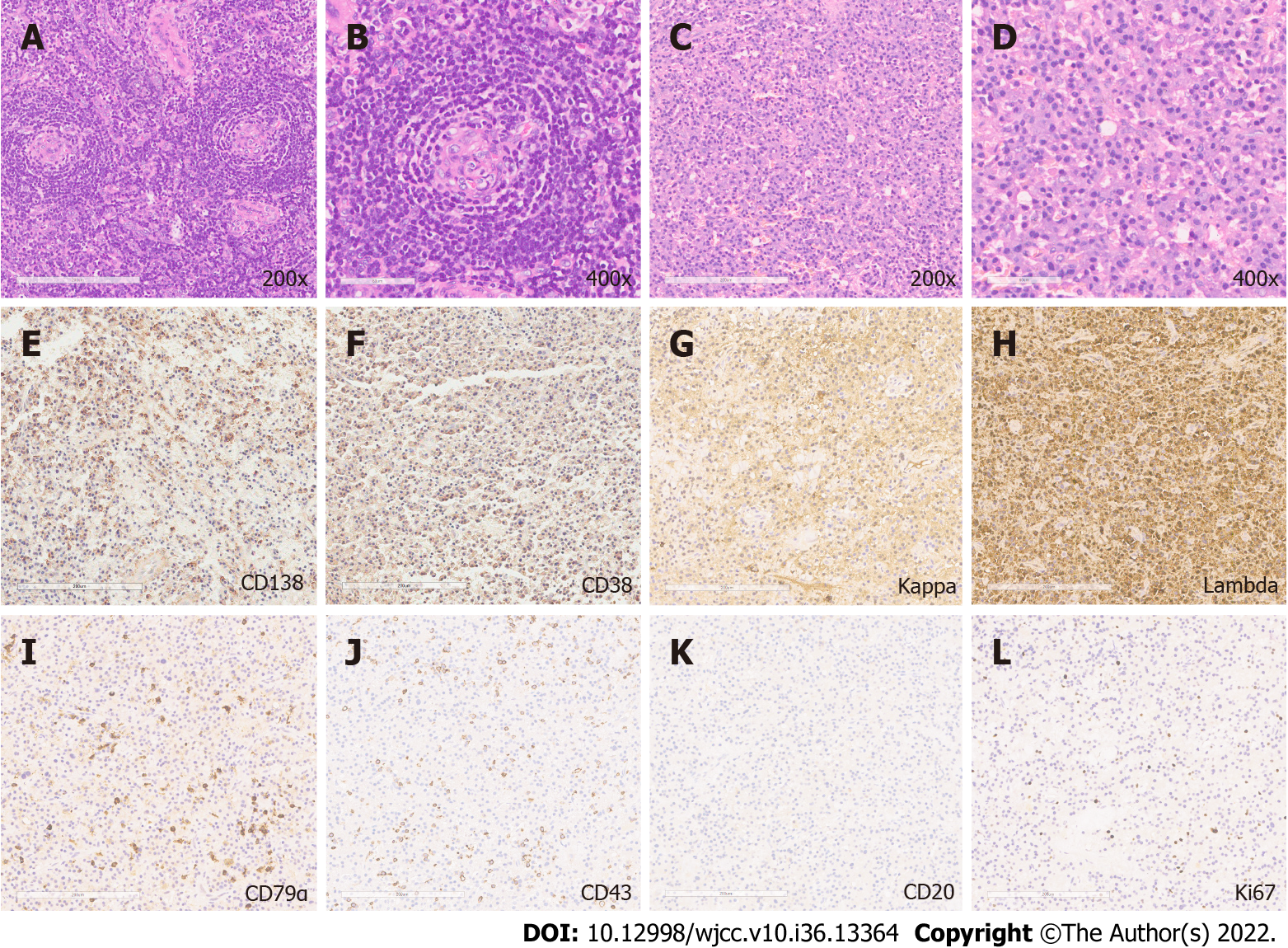
Histological results revealed angiofollicular lymph node hyperplasia of the hyaline-vascular type arranged in an onion-bulb appearance (Figure 2A and B, 200 × and 400 ×), and the presence of mature plasma cells characterized by an oval shape, abundant basophilic cytoplasm, perinuclear halo, round eccentric nuclei, and indiscernible nucleoli[6,7,9] (Figure 2C and D, 200 × and 400 ×). Histopathological results were positive for plasma cell markers [CD138 (Figure 2E), CD38 (Figure 2F), κ (Figure 2G) and λ (Figure 2H) light chains, CD79α (Figure 2I), IgG, IgM], and focal expression of T-cell markers [CD43 (Figure 2J), CD45], and CD56; however, deficiency of B-cell markers [CD20 (Figure 2K), CD19] was observed. The Ki-67 proliferation index was 3% (Figure 2L). No paraproteins and pathological elevation of κ and λ chains were detected by the serum protein electrophoresis and free light-chain assays[4,10]. Bone marrow biopsy indicated normal trilineage hematopoiesis with only 0.2% plasma cells in all leukocytes[4]. Additionally, polyneuropathy and other minor symptoms were absent.
Systemic work-up ruled out the possibility of POEMS syndrome, lymphoma, and MM[4,11,12], thus leading to the diagnosis of SP of the hyoid, with UCD. Inflammatory factors, such as TNFα, IL-6, IL1β, TGFβ1, and VEGFα (Supplementary Figure 1), were increased, confirming the multiple cytokine-producing property of SBP[13,14].
The patient underwent intensity-modulated RT[5] for the hyoid bone and high-risk subclinical areas, with a total dose of 50.4 Gy, divided into 28 fractions[10].
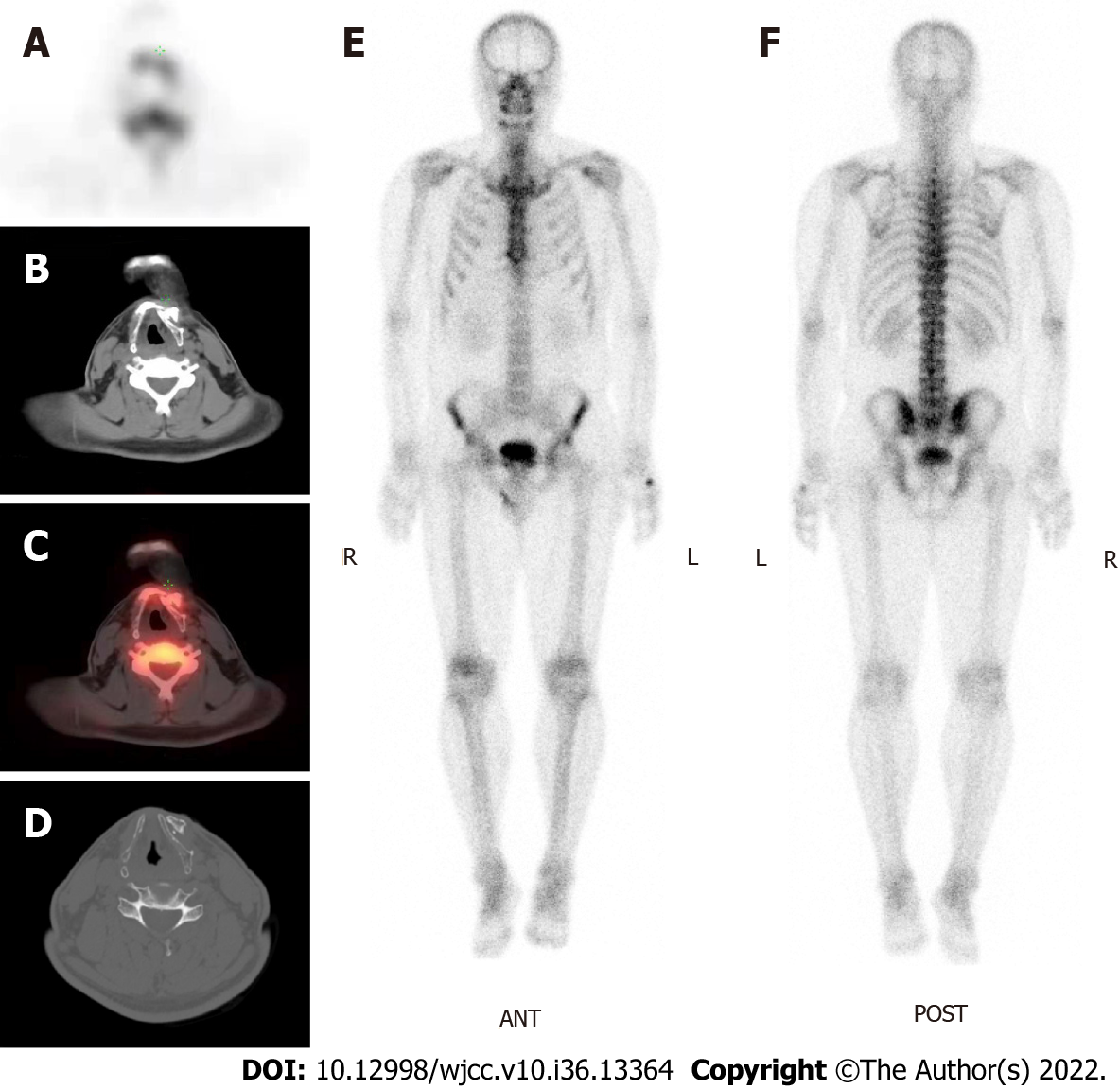
No acute toxicity was observed during RT. After 3 mo, contrast-enhanced CT revealed a slightly shrunken tumor boundary. The patient underwent CT and 18F-FDG PET/CT or 99mTc-MDP single-photon emission computed tomography (SPECT)/CT examinations at 6 and 12 mo, respectively. Recent 99mTc-MDP SPECT/CT reevaluation showed a slight increase in radioactivity around the hyoid, considering postoperative changes rather than local recurrence on SPECT (Figure 3A), CT (Figure 3B), and SPECT/CT fusion (Figure 3C). Focal bone destruction and secondary hyperosteogeny were also observed in the CT bone window (Figure 3D). Whole-body SPECT revealed no evidence of distant bone metastasis (Figure 3E and F). The patient was symptomatic and progression-free at the 40-mo follow-up visit.
Plasma cell disease comprises a heterogeneous group of disorders characterized by an increase in the number of monoclonal plasma cells[4]. Abnormally proliferative cells can secrete a large quantity of monoclonal immunoglobulin and invade and damage various tissues or organs, such as the bones, bone marrow, kidneys, hearts, lungs, and immune system. Precision clinical intervention is needed for the accurate diagnosis of these diseases. In the present study, the histology of the neck mass showed Castleman’s lesion, such as vitreous degeneration with an "onion skin" appearance, and monoclonal proliferation of plasma cells, resembling the typical features of CD variant of POEMS syndrome. However, the patient did not meet the other major or minor criteria required for a definite diagnosis of POEMS syndrome or MM[4,11,12]; therefore, led to a final diagnosed with SBP of the hyoid and UCD. The National Comprehensive Cancer Network recommends using primary RT of a total dose of 40–50 Gy in 1.8–2.0 Gy per fraction over 4 wk, for patients with SP in whom the surgery is depended on clinical necessity[10]; in addition, for patients with UCD, excisional surgery is the optimal strategy with the benefit of long-term disease control[6,8]. The patient received intensity-modulated RT according to the guidelines after extended dissection of the neoplastic mass adjoining the primary lesion.
The hyoid bone is an atypical location and is extremely rare for the growth of SP; to the best of our knowledge, only three cases have been reported[15-17]. Goel et al[15] presented the first case treated with local RT followed by chemotherapy, and the hyoid swelling was significantly reduced in size during the 18-mo follow-up. Danaci et al[16] described the second case of a 60-year-old man. This patient underwent total excision of the hyoid mass but was not subsequently treated with RT or chemotherapy, and was alive and progression-free during the 24-mo follow-up period[16]. The third patient was diagnosed with non-secretory MM stage IIIA and presented with plasma cell infiltration into the hyoid bone. This patient received chemotherapy and anti-hypercalcemic therapy; however, the prognosis was not referred to in the report[17].
SP can be divided into SBP and solitary extramedullary plasmacytoma based on its origin from the bone marrow or soft tissue organs[4]. Abnormal monoclonal plasma cells and bone marrow stromal cells (BMSCs) in SBP strongly express multiple cytokines and their corresponding receptors that promote the growth and survival of tumor cells or angiogenesis through autocrine and paracrine mechanisms[13,14]. Plasma cell-derived matrix metalloproteinases participate in the degradation of the extracellular matrix and basement membrane, leading to chronic inflammation and bone-destructive lesions[18]. Abnormal plasma cells also induce local angiogenesis and stimulate neovascularization depending on secreted cytokines, such as ANG-1, CXCL12, and VEGFα, which supply nutrition for tumor cell growth[19-21]. Cytokine homeostasis, particularly that secreted by BMSCs, acts as a regulatory mechanism for tumor growth[22]. Some reports indicate that aberrant activation of gp130-dependent and Notch signaling may provide strong advantages for the growth of plasmacytomas[19,23-25]. CD and POEMS syndrome are also associated with the elevation of several classical cytokines in serum, such as IL-6, VEGFα, TNFα, IL1β, and TGFβ1 secreted by proinflammatory cells, as found in SBP. CD and SP can present independently, concurrently, or sequentially with POEMS[26-28]. Patients with CD and/or POEMS syndrome usually have an upregulation of serum VEGFα and IL-6 mediated cytokine storm, which might impair cerebral vessels, resulting in vasculopathy of the central nervous system and an increased risk of cerebral infarction[26,29,30].
Recently, with the development of high-throughput sequencing technology, genetic analyses have been broadly applied to reveal the molecular mechanisms of UCD and idiopathic multicentric Castleman disease (iMCD)[31-35]. Overall, two recurrent hotspot somatic mutations, PDGFRB (NM_002609.4, c.1997A > G, p.N666S) and NCOA4 (NM_005437.4, c.781 > T, p.L261F), were identified in 17% and 18% of the tested UCD and iMCD cases, respectively[33,34]. In a patient with iMCD accompanied by severe peripheral neuropathy, somatic alterations were identified in genes related to neuronal development, such as PDLIM5, SEC24B, ZFHX3, and PACRG[36]. Another patient harbored a somatic JAK1 missense mutation, but normal serum IL-6 had a complete response rate to the IL-6 antibody siltuximab, implicating the essential roles of the IL-6/IL-6R/JAK1/STAT pathway and its mutations in the pathogenesis of iMCD[37]. Somatic mutations were prone to enrichment in the mitogen-activated protein kinase and interleukin pathways in UCD and iMCD; however, genes affecting chromatin remodeling were solely enriched in iMCD[38]. Moreover, patients with iMCD have a high prevalence of germline MEFV variants that modify their clinical phenotypes and treatment responses[39,40]. Transcriptome profiling also revealed that UCD and MCD are involved in unique genes, pathways, and cell types[35].
18F-FDG PET/CT exhibits superior sensitivity and specificity for SBP lesions. It can detect minimal residual disease inside and outside the bone marrow at the same time in one examination, which cannot be identified by χ-ray and magnetic resonance imaging[41,42]. In the early stages of patients with SBP after RT, 18F-FDG PET/CT performs poorly in disease staging; however, a high SUVmax value is a regular prognostic indicator for progression[43]. 99mTc-MDP scintigraphy shows a relatively low sensitivity to detect bone lesions of SBP due to the suppressed differentiation of osteoblasts[44] and can be more applicable for evaluating the complications of SBP[45]. 99mTc-MIBI scintigraphy shows the same sensitivity and specificity as 18F-FDG PET/CT in the detection of SBP lesions[46] and even better performance in the detection of diffuse bone marrow involvement, but it is inefficient in the detection of focal lesions[47]. 18F-FDG PET/CT has the ability to simultaneously collect anatomical structure and metabolic information and can be used for the initial staging and response monitoring of CD[48]. It has high specificity and sensitivity for the identification of lymph node lesions in patients with CD[49] and is an important imaging marker for judging the severity and prognosis of the disease. A variety of metabolic parameters in 18F-FDG PET/CT, including SUVmax, SUVmean, SUVpeak, metabolic tumor volume, and total lesion glycolysis, can be used for the differential diagnosis of UCD and other diseases[50].
To our knowledge, this is the first reported case presented with these two rare clinical entities (SP of the hyoid bone and UCD). In addition to comprehensive histopathological analysis, 18F-FDG PET/CT and 99mTc-MDP SPECT/CT are routinely used in the clinical practice of differential diagnosis, disease staging, and follow-up monitoring, highlighting the advantages of nuclear medical and other imaging techniques in the management of SP in the head and neck. In addition, the diagnostic and management processes provided valuable educational information to clinicians.
We sincerely thank Dr. Ya-Qiong Ren and Yu-Qi Shen at the Chinese Academy of Medical Sciences & Peking Union Medical College for staining of inflammatory factors of SBP and their independent figure combination and optimization.
Provenance and peer review: Unsolicited article; Externally peer reviewed.
Peer-review model: Single blind
Specialty type: Medicine, research and experimental
Country/Territory of origin: China
Peer-review report’s scientific quality classification
Grade A (Excellent): 0
Grade B (Very good): B
Grade C (Good): C
Grade D (Fair): 0
Grade E (Poor): 0
P-Reviewer: Ankrah AO, Netherlands; Rachh S, India S-Editor: Liu XF L-Editor: A P-Editor: Liu XF
| 1. | Caers J, Paiva B, Zamagni E, Leleu X, Bladé J, Kristinsson SY, Touzeau C, Abildgaard N, Terpos E, Heusschen R, Ocio E, Delforge M, Sezer O, Beksac M, Ludwig H, Merlini G, Moreau P, Zweegman S, Engelhardt M, Rosiñol L. Diagnosis, treatment, and response assessment in solitary plasmacytoma: Updated recommendations from a European Expert Panel. J Hematol Oncol. 2018;11:10. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 148] [Cited by in RCA: 184] [Article Influence: 26.3] [Reference Citation Analysis (0)] |
| 2. | Ohana N, Rouvio O, Nalbandyan K, Sheinis D, Benharroch D. Classification of Solitary Plasmacytoma, Is it more Intricate than Presently Suggested? J Cancer. 2018;9:3894-3897. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 7] [Cited by in RCA: 5] [Article Influence: 0.7] [Reference Citation Analysis (0)] |
| 3. | Knobel D, Zouhair A, Tsang RW, Poortmans P, Belkacémi Y, Bolla M, Oner FD, Landmann C, Castelain B, Ozsahin M, Rare Cancer Network. Prognostic factors in solitary plasmacytoma of the bone: A multicenter Rare Cancer Network study. BMC Cancer. 2006;6:118. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 148] [Cited by in RCA: 119] [Article Influence: 6.3] [Reference Citation Analysis (0)] |
| 4. | Rajkumar SV, Dimopoulos MA, Palumbo A, Blade J, Merlini G, Mateos MV, Kumar S, Hillengass J, Kastritis E, Richardson P, Landgren O, Paiva B, Dispenzieri A, Weiss B, LeLeu X, Zweegman S, Lonial S, Rosinol L, Zamagni E, Jagannath S, Sezer O, Kristinsson SY, Caers J, Usmani SZ, Lahuerta JJ, Johnsen HE, Beksac M, Cavo M, Goldschmidt H, Terpos E, Kyle RA, Anderson KC, Durie BG, Miguel JF. International Myeloma Working Group updated criteria for the diagnosis of multiple myeloma. Lancet Oncol. 2014;15:e538-e548. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 2320] [Cited by in RCA: 3152] [Article Influence: 286.5] [Reference Citation Analysis (0)] |
| 5. | Tsang RW, Campbell BA, Goda JS, Kelsey CR, Kirova YM, Parikh RR, Ng AK, Ricardi U, Suh CO, Mauch PM, Specht L, Yahalom J. Radiation Therapy for Solitary Plasmacytoma and Multiple Myeloma: Guidelines From the International Lymphoma Radiation Oncology Group. Int J Radiat Oncol Biol Phys. 2018;101:794-808. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 77] [Cited by in RCA: 131] [Article Influence: 18.7] [Reference Citation Analysis (0)] |
| 6. | Carbone A, Borok M, Damania B, Gloghini A, Polizzotto MN, Jayanthan RK, Fajgenbaum DC, Bower M. Castleman disease. Nat Rev Dis Primers. 2021;7:84. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 77] [Cited by in RCA: 109] [Article Influence: 27.3] [Reference Citation Analysis (0)] |
| 7. | Dispenzieri A, Fajgenbaum DC. Overview of Castleman disease. Blood. 2020;135:1353-1364. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 102] [Cited by in RCA: 282] [Article Influence: 56.4] [Reference Citation Analysis (0)] |
| 8. | van Rhee F, Oksenhendler E, Srkalovic G, Voorhees P, Lim M, Dispenzieri A, Ide M, Parente S, Schey S, Streetly M, Wong R, Wu D, Maillard I, Brandstadter J, Munshi N, Bowne W, Elenitoba-Johnson KS, Fössa A, Lechowicz MJ, Chandrakasan S, Pierson SK, Greenway A, Nasta S, Yoshizaki K, Kurzrock R, Uldrick TS, Casper C, Chadburn A, Fajgenbaum DC. International evidence-based consensus diagnostic and treatment guidelines for unicentric Castleman disease. Blood Adv. 2020;4:6039-6050. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 33] [Cited by in RCA: 126] [Article Influence: 31.5] [Reference Citation Analysis (1)] |
| 9. | Ribourtout B, Zandecki M. Plasma cell morphology in multiple myeloma and related disorders. Morphologie. 2015;99:38-62. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 23] [Cited by in RCA: 30] [Article Influence: 3.0] [Reference Citation Analysis (0)] |
| 10. | Kumar SK, Callander NS, Adekola K, Anderson L, Baljevic M, Campagnaro E, Castillo JJ, Chandler JC, Costello C, Efebera Y, Faiman M, Garfall A, Godby K, Hillengass J, Holmberg L, Htut M, Huff CA, Kang Y, Hultcrantz M, Larson S, Liedtke M, Martin T, Omel J, Shain K, Sborov D, Stockerl-Goldstein K, Weber D, Keller J, Kumar R. Multiple Myeloma, Version 3.2021, NCCN Clinical Practice Guidelines in Oncology. J Natl Compr Canc Netw. 2020;18:1685-1717. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 66] [Cited by in RCA: 139] [Article Influence: 27.8] [Reference Citation Analysis (0)] |
| 11. | Dispenzieri A. POEMS syndrome: 2021 Update on diagnosis, risk-stratification, and management. Am J Hematol. 2021;96:872-888. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 21] [Cited by in RCA: 73] [Article Influence: 18.3] [Reference Citation Analysis (0)] |
| 12. | Khouri J, Nakashima M, Wong S. Update on the Diagnosis and Treatment of POEMS (Polyneuropathy, Organomegaly, Endocrinopathy, Monoclonal Gammopathy, and Skin Changes) Syndrome: A Review. JAMA Oncol. 2021;7:1383-1391. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 3] [Cited by in RCA: 28] [Article Influence: 7.0] [Reference Citation Analysis (0)] |
| 13. | Nakayama-Ichiyama S, Yokote T, Iwaki K, Hirata Y, Akioka T, Miyoshi T, Oka S, Nishiwaki U, Masuda Y, Tsuji M, Hanafusa T. Multiple cytokine-producing solitary plasmacytoma of bone with expression of cytokine receptors. J Clin Oncol. 2011;29:e861-e863. [RCA] [PubMed] [DOI] [Full Text] [Reference Citation Analysis (0)] |
| 14. | Nakayama-Ichiyama S, Yokote T, Hirata Y, Iwaki K, Akioka T, Miyoshi T, Nishiwaki U, Masuda Y, Hiraoka N, Takayama A, Tsuji M, Hanafusa T. Multiple cytokine-producing plasmablastic solitary plasmacytoma of bone with polyneuropathy, organomegaly, endocrinology, monoclonal protein, and skin changes syndrome. J Clin Oncol. 2012;30:e91-e94. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 7] [Cited by in RCA: 7] [Article Influence: 0.5] [Reference Citation Analysis (0)] |
| 15. | Goel S, Moorjani V, Kulkarni P, Patange V, Sharma OP. Plasmacytoma of the hyoid. J Laryngol Otol. 1994;108:604-606. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 4] [Cited by in RCA: 4] [Article Influence: 0.1] [Reference Citation Analysis (0)] |
| 16. | Danaci M, Belet U, Akan H, Selçuk MB, Baştemir M, Koyuncu M. Solitary plasmacytoma of the hyoid bone. Otolaryngol Head Neck Surg. 1999;121:834-835. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 5] [Cited by in RCA: 5] [Article Influence: 0.2] [Reference Citation Analysis (0)] |
| 17. | Sychra V, Eßer D, Kosmehl H, Herold M. Unusual manifestation of a multiple myeloma in the hyoid bone. Dentomaxillofac Radiol. 2013;42:27101530. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 3] [Cited by in RCA: 3] [Article Influence: 0.2] [Reference Citation Analysis (0)] |
| 18. | Wahlgren J, Maisi P, Sorsa T, Sutinen M, Tervahartiala T, Pirilä E, Teronen O, Hietanen J, Tjäderhane L, Salo T. Expression and induction of collagenases (MMP-8 and -13) in plasma cells associated with bone-destructive lesions. J Pathol. 2001;194:217-224. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 90] [Cited by in RCA: 90] [Article Influence: 3.8] [Reference Citation Analysis (0)] |
| 19. | Hedvat CV, Comenzo RL, Teruya-Feldstein J, Olshen AB, Ely SA, Osman K, Zhang Y, Kalakonda N, Nimer SD. Insights into extramedullary tumour cell growth revealed by expression profiling of human plasmacytomas and multiple myeloma. Br J Haematol. 2003;122:728-744. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 51] [Cited by in RCA: 53] [Article Influence: 2.4] [Reference Citation Analysis (0)] |
| 20. | Nakayama T, Yao L, Tosato G. Mast cell-derived angiopoietin-1 plays a critical role in the growth of plasma cell tumors. J Clin Invest. 2004;114:1317-1325. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 95] [Cited by in RCA: 97] [Article Influence: 4.6] [Reference Citation Analysis (0)] |
| 21. | Martin SK, Diamond P, Williams SA, To LB, Peet DJ, Fujii N, Gronthos S, Harris AL, Zannettino AC. Hypoxia-inducible factor-2 is a novel regulator of aberrant CXCL12 expression in multiple myeloma plasma cells. Haematologica. 2010;95:776-784. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 67] [Cited by in RCA: 72] [Article Influence: 4.5] [Reference Citation Analysis (0)] |
| 22. | Shoham T, Sternberg D, Brosh N, Krupsky M, Barda-Saad M, Zipori D. The promotion of plasmacytoma tumor growth by mesenchymal stroma is antagonized by basic fibroblast growth factor induced activin A. Leukemia. 2001;15:1102-1110. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 13] [Cited by in RCA: 13] [Article Influence: 0.5] [Reference Citation Analysis (0)] |
| 23. | Scherger AK, Al-Maarri M, Maurer HC, Schick M, Maurer S, Öllinger R, Gonzalez-Menendez I, Martella M, Thaler M, Pechloff K, Steiger K, Sander S, Ruland J, Rad R, Quintanilla-Martinez L, Wunderlich FT, Rose-John S, Keller U. Activated gp130 signaling selectively targets B cell differentiation to induce mature lymphoma and plasmacytoma. JCI Insight. 2019;4. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 9] [Cited by in RCA: 18] [Article Influence: 3.0] [Reference Citation Analysis (0)] |
| 24. | Burger R, Günther A, Klausz K, Staudinger M, Peipp M, Penas EM, Rose-John S, Wijdenes J, Gramatzki M. Due to interleukin-6 type cytokine redundancy only glycoprotein 130 receptor blockade efficiently inhibits myeloma growth. Haematologica. 2017;102:381-390. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 17] [Cited by in RCA: 20] [Article Influence: 2.2] [Reference Citation Analysis (0)] |
| 25. | Shin DM, Shaffer DJ, Wang H, Roopenian DC, Morse HC 3rd. NOTCH is part of the transcriptional network regulating cell growth and survival in mouse plasmacytomas. Cancer Res. 2008;68:9202-9211. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 20] [Cited by in RCA: 20] [Article Influence: 1.2] [Reference Citation Analysis (0)] |
| 26. | Huang J, Wang L, Zhou W, Jin J. Hyaline vascular Castleman disease associated with POEMS syndrome and cerebral infarction. Ann Hematol. 2007;86:59-61. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 7] [Cited by in RCA: 10] [Article Influence: 0.5] [Reference Citation Analysis (0)] |
| 27. | Ge Y, Da Q, Dai Y. Castleman disease of the hyaline vascular variant transforming to POEMS syndrome as endpoint: A case report. BMC Neurol. 2018;18:169. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 3] [Cited by in RCA: 3] [Article Influence: 0.4] [Reference Citation Analysis (0)] |
| 28. | Gilder H, Murphy ME, Alvi MA, Kerezoudis P, Shepherd D, Maloney PR, Yaszemski MJ, Morris JM, Dispenzieri A, Matsumoto JM, Bydon M. Skull base plasmacytoma: A unique case of POEMS syndrome with a plasmacytoma causing craniocervical instability. J Clin Neurosci. 2018;47:254-257. [RCA] [PubMed] [DOI] [Full Text] [Cited by in RCA: 2] [Reference Citation Analysis (0)] |
| 29. | Yu H, Yao F, Li Y, Li J, Cui QC. Castleman disease variant of POEMS syndrome complicated with multiple cerebral infarction: A rare case report and review of literature. Int J Clin Exp Pathol. 2015;8:13578-13583. [PubMed] |
| 30. | Garcia T, Dafer R, Hocker S, Schneck M, Barton K, Biller J. Recurrent strokes in two patients with POEMS syndrome and Castleman's disease. J Stroke Cerebrovasc Dis. 2007;16:278-284. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 15] [Cited by in RCA: 16] [Article Influence: 0.9] [Reference Citation Analysis (0)] |
| 31. | Nagy A, Bhaduri A, Shahmarvand N, Shahryari J, Zehnder JL, Warnke RA, Mughal T, Ali S, Ohgami RS. Next-generation sequencing of idiopathic multicentric and unicentric Castleman disease and follicular dendritic cell sarcomas. Blood Adv. 2018;2:481-491. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 39] [Cited by in RCA: 47] [Article Influence: 6.7] [Reference Citation Analysis (0)] |
| 32. | Baker TS, Gambino KJ, Schriefer L, Lim JY, Steinberg KM, Fajgenbaum DC, Martín García-Sancho A, Byun M. A novel FAS mutation with variable expressivity in a family with unicentric and idiopathic multicentric Castleman disease. Blood Adv. 2018;2:2959-2963. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 10] [Cited by in RCA: 17] [Article Influence: 2.8] [Reference Citation Analysis (0)] |
| 33. | Li Z, Lan X, Li C, Zhang Y, Wang Y, Xue W, Lu L, Jin M, Zhou Z, Wang X, Li L, Zhang L, Li X, Fu X, Sun Z, Wu J, Zhang X, Yu H, Nan F, Chang Y, Yan J, Wu X, Wang G, Zhang D, Young KH, Zhang M. Recurrent PDGFRB mutations in unicentric Castleman disease. Leukemia. 2019;33:1035-1038. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 57] [Cited by in RCA: 58] [Article Influence: 9.7] [Reference Citation Analysis (0)] |
| 34. | You L, Lin Q, Zhao J, Shi F, Young KH, Qian W. Whole-exome sequencing identifies novel somatic alterations associated with outcomes in idiopathic multicentric Castleman disease. Br J Haematol. 2020;188:e64-e67. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 12] [Cited by in RCA: 13] [Article Influence: 2.6] [Reference Citation Analysis (0)] |
| 35. | Wing A, Xu J, Meng W, Rosenfeld AM, Li EY, Wertheim G, Paessler M, Bagg A, Frank D, Tan K, Teachey DT, Lim MS, Prak EL, Fajgenbaum DC, Pillai V. Transcriptome and unique cytokine microenvironment of Castleman disease. Mod Pathol. 2022;35:451-461. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 5] [Cited by in RCA: 22] [Article Influence: 7.3] [Reference Citation Analysis (0)] |
| 36. | Lin Q, Wei J, Qian J, You L, Qian W. Somatic Mutations Confer Severe Peripheral Neuropathy in POEMS Syndrome-Associated Multicentric Castleman Disease. Neurosci Bull. 2020;36:664-666. [RCA] [PubMed] [DOI] [Full Text] [Reference Citation Analysis (0)] |
| 37. | Patel M, Ikeda S, Pilat SR, Kurzrock R. JAK1 Genomic Alteration Associated With Exceptional Response to Siltuximab in Cutaneous Castleman Disease. JAMA Dermatol. 2017;153:449-452. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 7] [Cited by in RCA: 10] [Article Influence: 1.3] [Reference Citation Analysis (0)] |
| 38. | Butzmann A, Kumar J, Sridhar K, Gollapudi S, Ohgami RS. A Review of Genetic Abnormalities in Unicentric and Multicentric Castleman Disease. Biology (Basel). 2021;10. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 5] [Cited by in RCA: 18] [Article Influence: 4.5] [Reference Citation Analysis (0)] |
| 39. | Endo Y, Koga T, Otaki H, Sasaki D, Sumiyoshi R, Furukawa K, Tanaka Y, Katsunori Y, Kawakami A. Idiopathic multicentric Castleman disease with novel heterozygous Ile729Met mutation in exon 10 of familial Mediterranean fever gene. Rheumatology (Oxford). 2021;60:445-450. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 5] [Cited by in RCA: 5] [Article Influence: 1.0] [Reference Citation Analysis (0)] |
| 40. | Endo Y, Koga T, Ubara Y, Sumiyoshi R, Furukawa K, Kawakami A. Mediterranean fever gene variants modify clinical phenotypes of idiopathic multi-centric Castleman disease. Clin Exp Immunol. 2021;206:91-98. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 5] [Cited by in RCA: 9] [Article Influence: 2.3] [Reference Citation Analysis (0)] |
| 41. | Michaud-Robert AV, Jamet B, Bailly C, Carlier T, Moreau P, Touzeau C, Bourgeois M, Kraeber-Bodere F, Bodet-Milin C. FDG-PET/CT, a Promising Exam for Detecting High-Risk Myeloma Patients? Cancers (Basel). 2020;12. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 3] [Cited by in RCA: 3] [Article Influence: 0.6] [Reference Citation Analysis (0)] |
| 42. | Cavo M, Terpos E, Nanni C, Moreau P, Lentzsch S, Zweegman S, Hillengass J, Engelhardt M, Usmani SZ, Vesole DH, San-Miguel J, Kumar SK, Richardson PG, Mikhael JR, da Costa FL, Dimopoulos MA, Zingaretti C, Abildgaard N, Goldschmidt H, Orlowski RZ, Chng WJ, Einsele H, Lonial S, Barlogie B, Anderson KC, Rajkumar SV, Durie BGM, Zamagni E. Role of 18F-FDG PET/CT in the diagnosis and management of multiple myeloma and other plasma cell disorders: A consensus statement by the International Myeloma Working Group. Lancet Oncol. 2017;18:e206-e217. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 284] [Cited by in RCA: 355] [Article Influence: 44.4] [Reference Citation Analysis (0)] |
| 43. | Alongi P, Zanoni L, Incerti E, Fallanca F, Mapelli P, Papathanasiou N, Gianolli L, Picchio M, Bomanji J. 18F-FDG PET/CT for Early Postradiotherapy Assessment in Solitary Bone Plasmacytomas. Clin Nucl Med. 2015;40:e399-e404. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 14] [Cited by in RCA: 14] [Article Influence: 1.4] [Reference Citation Analysis (0)] |
| 44. | Vakili Sadeghi M, Sedaghat S. Is 99m Tc-methylene diphosphonate bone scintigraphy a sensitive method for detecting bone lesions in multiple myeloma? Caspian J Intern Med. 2018;9:140-143. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in RCA: 1] [Reference Citation Analysis (0)] |
| 45. | Koppula B, Kaptuch J, Hanrahan CJ. Imaging of multiple myeloma: Usefulness of MRI and PET/CT. Semin Ultrasound CT MR. 2013;34:566-577. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 11] [Cited by in RCA: 11] [Article Influence: 1.0] [Reference Citation Analysis (0)] |
| 46. | Mele A, Offidani M, Visani G, Marconi M, Cambioli F, Nonni M, Catarini M, Brianzoni E, Berbellini A, Ascoli G, Brunori M, Agostini V, Corvatta L, Isidori A, Spinelli A, Gradari M, Leoni P. Technetium-99m sestamibi scintigraphy is sensitive and specific for the staging and the follow-up of patients with multiple myeloma: A multicentre study on 397 scans. Br J Haematol. 2007;136:729-735. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 24] [Cited by in RCA: 24] [Article Influence: 1.3] [Reference Citation Analysis (0)] |
| 47. | Mosci C, Pericole FV, Oliveira GB, Delamain MT, Takahashi MES, Carvalheira JBC, Etchebehere ECSC, Santos AO, Miranda ECM, Lima MCL, Amorim BJ, de Souza CA, Lorand-Metze I, Ramos CD. 99mTc-sestamibi SPECT/CT and 18F-FDG-PET/CT have similar performance but different imaging patterns in newly diagnosed multiple myeloma. Nucl Med Commun. 2020;41:1081-1088. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 9] [Cited by in RCA: 9] [Article Influence: 1.8] [Reference Citation Analysis (0)] |
| 48. | Akosman C, Selcuk NA, Ordu C, Ercan S, Ekici ID, Oyan B. Unicentric mixed variant Castleman disease associated with Hashimoto disease: The role of PET/CT in staging and evaluating response to the treatment. Cancer Imaging. 2011;11:52-55. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 1] [Cited by in RCA: 8] [Article Influence: 0.6] [Reference Citation Analysis (0)] |
| 49. | Madan R, Chen JH, Trotman-Dickenson B, Jacobson F, Hunsaker A. The spectrum of Castleman's disease: mimics, radiologic pathologic correlation and role of imaging in patient management. Eur J Radiol. 2012;81:123-131. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 58] [Cited by in RCA: 69] [Article Influence: 4.6] [Reference Citation Analysis (0)] |
| 50. | Jiang Y, Hou G, Zhu Z, Huo L, Cheng W, Li F. The value of multiparameter 18F-FDG PET/CT imaging in differentiating retroperitoneal paragangliomas from unicentric Castleman disease. Sci Rep. 2020;10:12887. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 7] [Cited by in RCA: 9] [Article Influence: 1.8] [Reference Citation Analysis (0)] |