Published online Oct 16, 2022. doi: 10.12998/wjcc.v10.i29.10787
Peer-review started: June 28, 2022
First decision: July 14, 2022
Revised: July 22, 2022
Accepted: September 8, 2022
Article in press: September 8, 2022
Published online: October 16, 2022
Processing time: 92 Days and 18.9 Hours
Sperm granuloma is a rare disease in clinical andrology and its incidence is still unclear worldwide. According to the existing literature, sperm granuloma often occurs unilaterally. Clinical and ultrasound features are similar to epididymal tuberculosis, chronic epididymitis and other diseases. Sperm granuloma is usually diagnosed based on postoperative histopathological and immunohistochemical examination.
A 46-year-old man was admitted to the hospital due to the presence of a left scrotal mass for 3 mo and aggravation of pain for 1 wk. The lesions at both sites were surgically resected. Postoperative pathological examination showed that the left spermatic cord mass and the right epididymal mass were consistent with sperm granuloma. The sperm granulomas then recurred 3 mo after surgery. There is little change in the local mass so far.
The case report is helpful for our understanding of this disease. In clinical diagnosis, it should be distinguished from epididymal tuberculosis, chronic epididymitis and other diseases. Color Doppler ultrasound can be used as a preferred examination method but postoperative pathological examination is still needed for diagnosis.
Core Tip: We reported on a rare case of sperm granuloma which occurred bilaterally in the left spermatic cord and on the right epididymis. Remarkably, the granuloma recurred at 3 mo after surgery and this recurrence has not been reported before. Since the patient has no history of vasectomy, tumor or trauma, it is more likely that this case of sperm granuloma is merely caused by inflammation which is rare for sperm granuloma. This case report can greatly enhance our understanding of sperm granuloma and it is helpful for differential diagnosis of various epididymal lesions in clinical practice.
- Citation: Lv DY, Xie HJ, Cui F, Zhou HY, Shuang WB. Bilateral occurrence of sperm granulomas in the left spermatic cord and on the right epididymis: A case report. World J Clin Cases 2022; 10(29): 10787-10793
- URL: https://www.wjgnet.com/2307-8960/full/v10/i29/10787.htm
- DOI: https://dx.doi.org/10.12998/wjcc.v10.i29.10787
Sperm granuloma (SG) is a rare disease and its incidence is still unclear worldwide. The first case was reported in 1949[1] and ultrasonographic findings were first described in 1982[2]. In the past 10 years, only a few cases of SGs have been reported[3-5], let alone the cases of bilateral and postoperative recurrent SGs. According to the existing literature, SG is common in the epididymis and occasionally occurs in the spermatic cord and testes. SG is usually seen in unilateral nodules. Most of the patients visited a doctor with complaint of scrotal pain and induration.
Herein, we report a rare type of SG which occurred bilaterally, in both the left spermatic cord and right epididymis. The postoperative pathological diagnosis was consistent with the manifestation of SG and it recurred 3 mo after surgery.
A 46-year-old man reported having found a bean-like mass in the left scrotum approximately 3 mo prior.
The patient described his having felt a bean-like mass in the left scrotum without obvious inducement 3 mo prior. He described the scrotal mass as firm and associated with mild pain but without the scrotal skin having any redness or itching. The patient denied having any other symptoms, such as lower urinary tract symptoms, hot flashes and night sweats, fever and shiver, or progressive emaciation. Over time, the mass had gradually increased in size, accompanied by irregular swelling and pain. Anti-infective treatment did not relieve the symptoms, and ultimately he presented himself to the urology clinic of our hospital.
The patient denied a history of tuberculosis or vasectomy.
The patient denied any family history of disease of the scrotum.
The physical examination showed a firm mass measuring 4 cm × 3 cm × 2 cm in the left scrotum with no obvious tenderness. The light transmission test was negative.
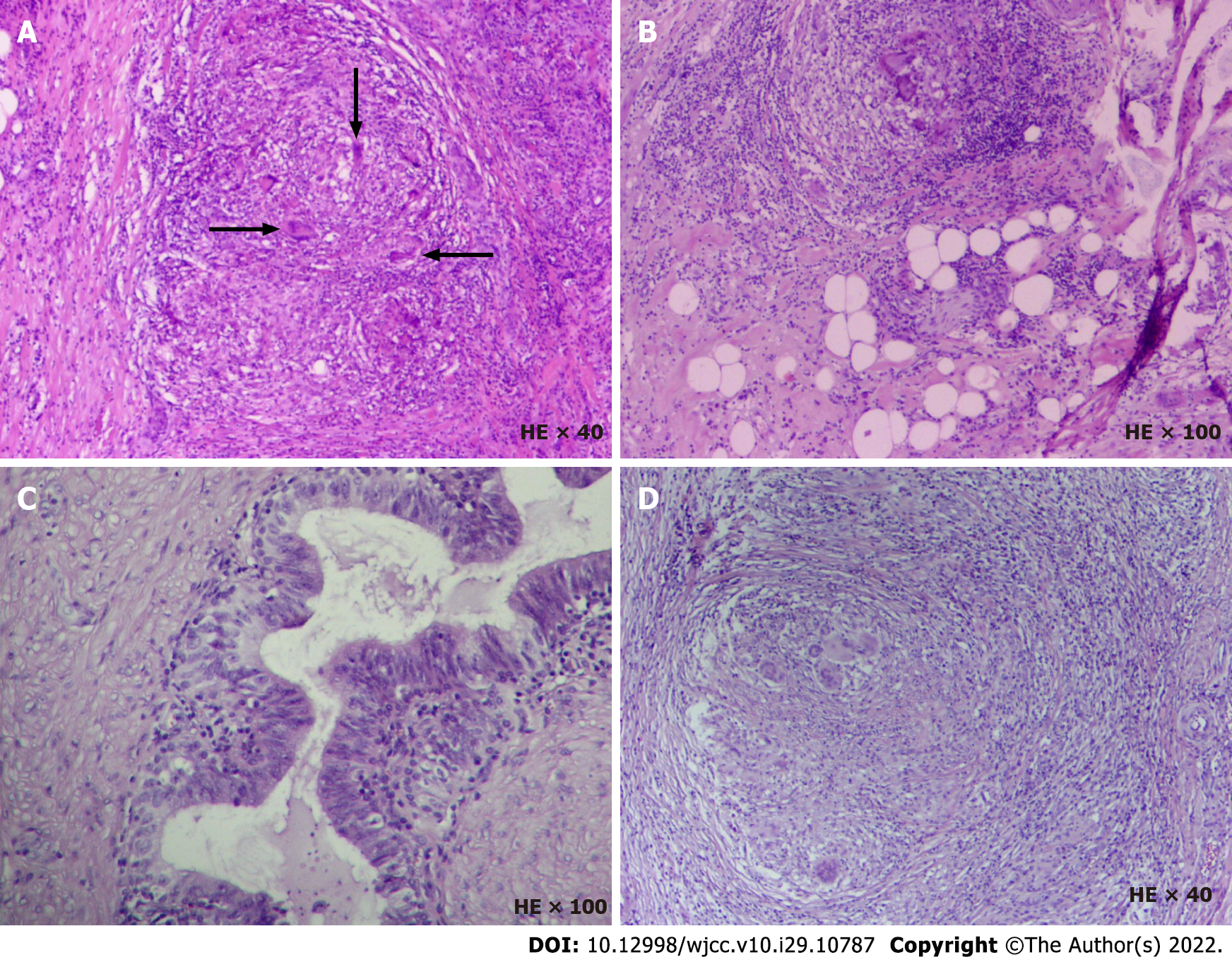
The mass was negative for three tumor markers including alpha-fetoprotein, human chorionic gonadotropin and lactate dehydrogenase. The surgical specimen was stained with hematoxylin and eosin, and histopathological observation showed that the left spermatic cord mass and the right epididymal mass were consistent with SG (Figure 1). The surgical specimen was processed using indirect immunohistochemistry for CK (Beijing Zhongshanjinqiao Biotechnology Co., Ltd, Beijing, China) and CD68 (Beijing Zhongshanjinqiao Biotechnology Co., Ltd), as well as subjected to acid-fast stain (BaSO Diagnostics Inc., Zhuhai, China); these experiments were performed according to the manufacturer’s protocols. The results for the left spermatic cord mass were as follows: acid-fast (-), CK (-) and CD68 (+).
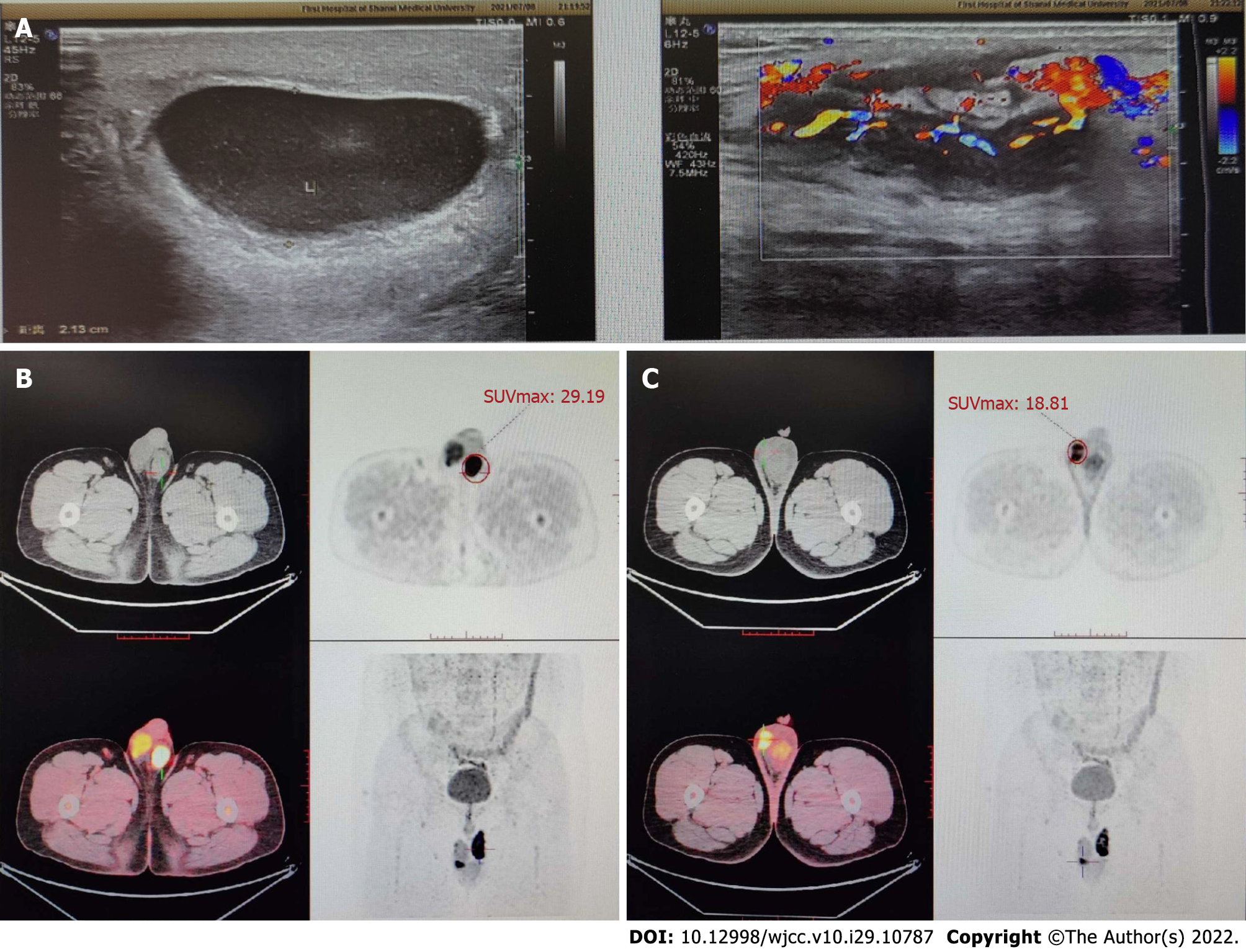
Color Doppler ultrasound (CDU) of the scrotum and positron emission tomography/computed tomography images are shown in Figure 2.
Postoperative and intraoperative pathological examination showed that the left spermatic cord mass and the right epididymal mass were consistent with SG.
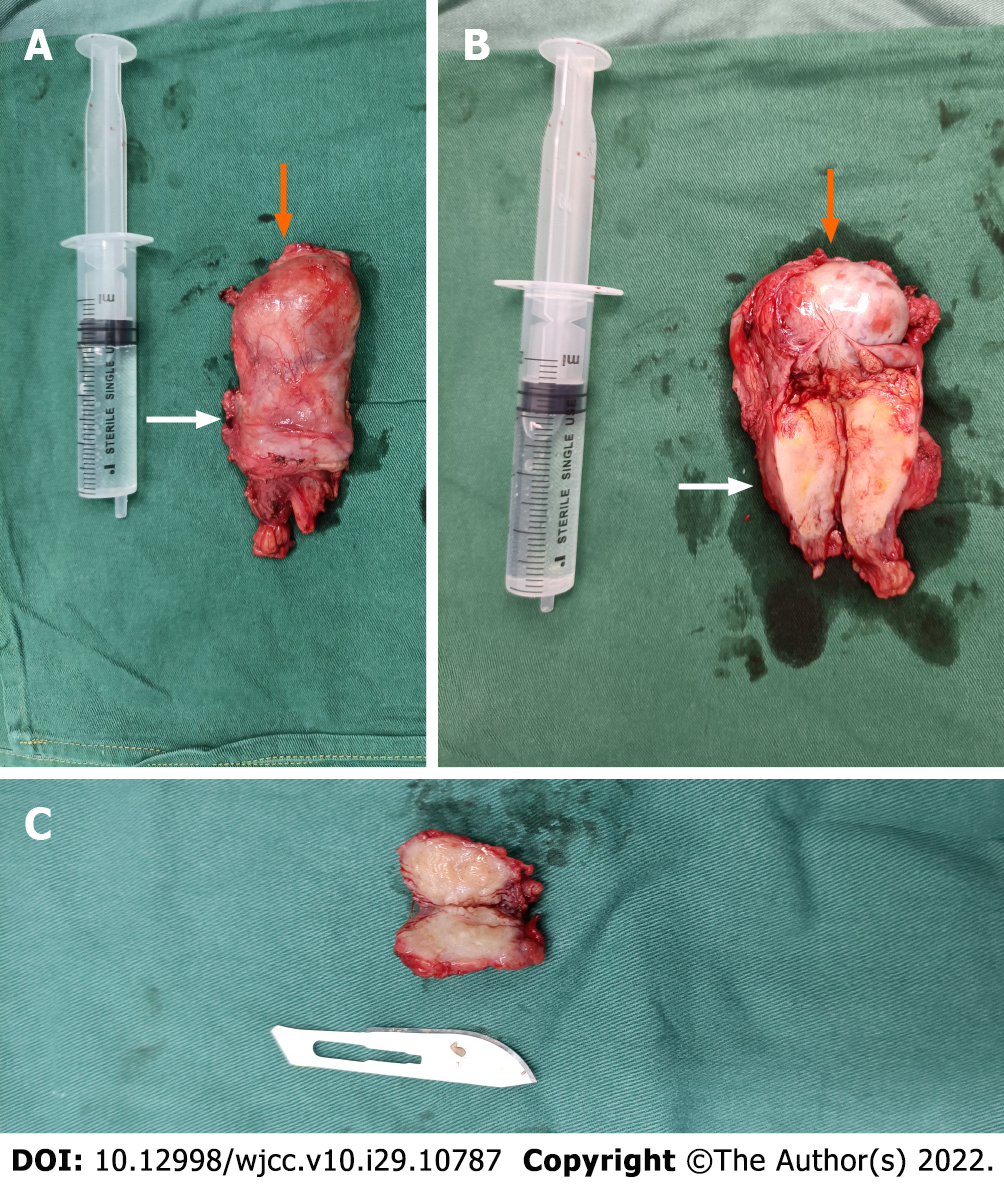
To relieve the symptoms, the patient underwent surgery with general anesthesia. During the operation, a fusiform hard mass was seen in the spermatic region of the left proximal epididymis which was about 5.0 cm × 3.0 cm × 2.8 cm in size (Figure 3A and B). Because the blood supply to the left testis could not be preserved after the resection of the left spermatic cord tumor, left orchiectomy was performed. The interoperative frozen section analysis indicated inflammatory hyperplasia in the tumor tissue. A hard mass of about 2.0 cm × 1.5 cm × 1.3 cm was found in the tail of the right epididymis and the boundary was still clear. Therefore, the resection of the right epididymis was performed (Figure 3C). After surgery, the patient was given symptomatic supportive treatment and was discharged after 1 wk.
Three months after surgery, the patient went to the outpatient clinic of our hospital once again because of a right scrotal mass. CDU of the scrotum and spermatic cord showed that after left orchiectomy, there was no obvious abnormality; the right epididymis was enlarged and the echo of parenchyma was uneven. The color Doppler flow imaging showed an increase in blood flow signals. Considering the possibility of a recurrence of the lesion in the right epididymis, the patient was asked to stay for continuous observation. There is little change in the local mass so far.
SG is a chronic granulomatous inflammation with low incidence which is caused by a foreign body reaction with sperm or acid-resistant lipids. Sperm overflow from damaged seminiferous tubules, epididymal ducts or vas deferens to the surrounding stroma can result in the formation of inflammatory granuloma. The breakage of these tubules is often caused by inflammation, tumor, trauma, vasectomy and surgical operation of adjacent sites. Among these various causes, vasectomy is the most common with a rate of epididymal SG after vasectomy of 41%[2]. Nevertheless, there are few reports on SG caused by inflammation, trauma or tumor alone. In this case, the patient had no history of vasectomy, tumor or trauma; therefore, inflammation was the more likely cause. Furthermore, scrotal ultrasound showed that the arterial blood flow was well filled which served as indirect evidence of inflammation.
There are three types of SGs based on their location including testicular SG, epididymal SG and spermatic cord SG[6]. The most common site is epididymis followed by spermatic cord and testis. SG is usually seen in unilateral nodules. In this case, the patient presented with bilateral SGs which occurred in the left spermatic cord and on the right epididymis. This type of case is rare and, likewise, has rarely been reported in the literature.
The diagnosis of SG mainly depends on the pathological examination of surgical specimens. CDU, CT and magnetic resonance imaging all lack specificity. However, CDU can be used as a preferable diagnostic method because CDU images can reveal histopathological characteristics of tumors at different stages[7]. At the early stage (the acute stage), there are a large number of inflammatory cells and sperm cells in the lesion site accompanied by a few fibrous cells; so, the lesions detected by CDU are hypoechoic. In addition, due to inflammatory stimulation, localized hyperemia, edema and vasodilation, there is abundant blood flow in and around the lesion. At the middle stage, inflammatory cells and sperm cells in the stroma gradually decrease but fibrous tissue gradually increases; so, the lesions detected by CDU are moderately hypoechoic. If the fibrous tissue proliferates heterogeneously or tissue begins to necrotize, the echotexture is heterogeneous. As the disease progresses, inflammatory cells and sperm cells further decrease, accompanied by intensive proliferation of fibrous tissue; so, ultrasound shows high echo. During this period, there are few inflammatory cells, and vasodilation and congestion decreases significantly; so, there is only a little stellate blood flow or no blood flow around and inside the focus[7]. In this case, based on the ultrasonographic findings and postoperative pathological examination, the lesion was considered to be in the middle pathological stage.
SGs lack typical clinical features and are often characterized by pain in the scrotum or groin, palpable hard nodules in the epididymis and seminal cord, and sometimes a slight squeezing pain. Therefore, it needs to be differentiated from epididymal tuberculosis, chronic epididymitis, epididymal tumor, epididymal cyst, semen cyst and testicular lesions. Most of the patients with epididymal tuberculosis have a history of tuberculosis, such as afternoon hot flashes, increased erythrocyte sedimentation rate, positive tuberculin test and so on. Ultrasonic images show diffuse enlargement of the epididymis, irregular shape, moderate echo or mixed echo due to calcification[8]. As for the patients with chronic epididymitis, the ultrasonographic examination shows moderate or low echo, and most have a history of acute epididymitis and are improved after anti-infective treatment[9]. With the progression of chronic epididymitis, chronic inflammatory exudates are discharged from local tissues accompanied by tissue edema with cystic changes but no hard nodules detected upon touch. In comparison, SG could be characterized by fibrous tissue hyperplasia, epididymal duct fibrosis and hard nodules near the scrotum[10]. As for epididymal tumors, they are local solid masses and are rarely reported. They can be either benign or malignant. The benign tumor has a clear boundary, with an even low internal echo or moderate-to-high internal echo. The malignant tumor grows rapidly with an uneven internal echo. As for epididymal cyst, though there is no echo in ultrasound examination, CT examination shows low-density shadow due to the presence of fluid which is easily neglected during diagnosis. As for the semen cyst, it was mostly located in the head of the epididymis and appears as round anechoic nodules while SG is mostly located in the tail of the epididymis[11].
SGs are a benign tumor with a very low risk of malignant transformation. As the disease progresses, the lesion may block the vas deferens leading to infertility. Therefore, surgical resection is recommended and should be performed as soon as possible. If the tumor is not completely removed, however, it may recur after surgery.
We reported a rare case of SG which occurred bilaterally in the left spermatic cord and on the right epididymis. Remarkably, it recurred at 3 mo after surgery and this recurrence has not been reported before. Since the patient has no history of vasectomy, tumor or trauma, it is more likely that this case of SG is merely caused by inflammation which is rare for SG. This case report can greatly enhance our understanding of SG and it will be helpful in differential diagnosis of various epididymal lesions in clinical practice.
Provenance and peer review: Unsolicited article; Externally peer reviewed.
Peer-review model: Single blind
Specialty type: Andrology
Country/Territory of origin: China
Peer-review report’s scientific quality classification
Grade A (Excellent): 0
Grade B (Very good): 0
Grade C (Good): C, C
Grade D (Fair): 0
Grade E (Poor): 0
P-Reviewer: Arslan M, Turkey; Exbrayat JM, France S-Editor: Liu JH L-Editor: Filipodia P-Editor: Liu JH
| 1. | Friedman NB, Garske GL. Inflammatory reactions involving sperm and the seminiferous tubules; extravasation, spermatic granulomas and granulomatous orchitis. J Urol. 1949;62:363-374. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 65] [Cited by in RCA: 66] [Article Influence: 0.9] [Reference Citation Analysis (0)] |
| 2. | Dunner PS, Lipsit ER, Nochomovitz LE. Epididymal sperm granuloma simulating a testicular neoplasm. J Clin Ultrasound. 1982;10:353-355. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 11] [Cited by in RCA: 6] [Article Influence: 0.1] [Reference Citation Analysis (0)] |
| 3. | Garrido-Abad P, Díaz-Menéndez A, García-Martín L, Senra-Bravo I, Fernández-Arjona M. Tumor-like appearance of Spermatic Granuloma. Int Braz J Urol. 2019;45:634-636. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 3] [Cited by in RCA: 3] [Article Influence: 0.5] [Reference Citation Analysis (0)] |
| 4. | Theisen K, Chaudhry R, Davis A, Cannon G. Epididymal Inflammatory Pseudotumor With Downstream Sperm Granuloma in an Adolescent Patient: A Case Report and Review of the Literature. Urology. 2016;98:158-160. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 4] [Cited by in RCA: 4] [Article Influence: 0.4] [Reference Citation Analysis (0)] |
| 5. | Su JS, Farber NJ, Feldman MK, Vij SC. Sperm granuloma masquerading as a supernumerary testis. Urol Case Rep. 2020;29:101080. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 3] [Cited by in RCA: 3] [Article Influence: 0.5] [Reference Citation Analysis (0)] |
| 6. | Ye Zhanying, Liao Yanbin, Huang Weijun. Ultrasound appearance of one sperm granuloma. Zhongguo Chaosheng Yixue Za Zhi. 2020;36:384. |
| 7. | Yang Biyun, Li Huyi, Wang Liangmei. Clinical studies on ultrasonic diagnosis of inflammatory epididymal sperm granuloma. Zhongguo Xingkexue Za Zhi. 2015;24:13-14. [DOI] [Full Text] |
| 8. | Bhatt S, Rubens DJ, Dogra VS. Sonography of benign intrascrotal lesions. Ultrasound Q. 2006;22:121-136. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 45] [Cited by in RCA: 31] [Article Influence: 1.6] [Reference Citation Analysis (0)] |
| 9. | Yang DM, Kim SH, Kim HN, Kang JH, Seo TS, Hwang HY, Kim HS, Cho H. Differential diagnosis of focal epididymal lesions with gray scale sonographic, color Doppler sonographic, and clinical features. J Ultrasound Med. 2003;22:135-42; quiz 143. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 23] [Cited by in RCA: 14] [Article Influence: 0.6] [Reference Citation Analysis (0)] |
| 10. | Maher ER, Neumann HP, Richard S. von Hippel-Lindau disease: a clinical and scientific review. Eur J Hum Genet. 2011;19:617-623. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 433] [Cited by in RCA: 432] [Article Influence: 30.9] [Reference Citation Analysis (0)] |
| 11. | Kim JY, Lee YT, Kang HJ, Lee CH. Primary mucinous cystadenoma of the spermatic cord within the inguinal canal. Diagn Pathol. 2012;7:139. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 7] [Cited by in RCA: 7] [Article Influence: 0.5] [Reference Citation Analysis (0)] |