Published online Sep 26, 2022. doi: 10.12998/wjcc.v10.i27.9602
Peer-review started: May 15, 2022
First decision: June 8, 2022
Revised: June 29, 2022
Accepted: August 16, 2022
Article in press: August 16, 2022
Published online: September 26, 2022
Processing time: 123 Days and 20.1 Hours
The current coronavirus disease 2019 (COVID-19) pandemic has had devastating impact on populations around the world. The high mortality rates in patients with COVID-19 has been attributed to the influence of severe acute respiratory syndrome coronavirus-2 (SARS-CoV-2), its causative viral agent, on several physiological systems in human body, including the respiratory, cardiovascular, and neurological systems. There is emerging evidence on propensity of this virus to attack cardiovascular system. However, various pathophysiological mecha
Core Tip: It is well established that severe acute respiratory syndrome coronavirus-2 has a high propensity to attack cardiovascular system with various clinical manifestations namely myocarditis and myocardial injury, cardiac arrest, acute coronary syndromes, hypercoagulability and venous thromboembolism, as well as heart failure. Currently, there is no established, specific therapy for the treatment of coronavirus disease-induced cardiovascular involvement and there is ongoing research on this topic.
- Citation: Chatzis DG, Magounaki K, Pantazopoulos I, Bhaskar SMM. COVID-19 and the cardiovascular system-current knowledge and future perspectives. World J Clin Cases 2022; 10(27): 9602-9610
- URL: https://www.wjgnet.com/2307-8960/full/v10/i27/9602.htm
- DOI: https://dx.doi.org/10.12998/wjcc.v10.i27.9602
Coronavirus disease 2019 (COVID-19) pandemic initially began in Wuhan, China in December 2019. Since 15th of February 2021, more than 109 million infections and approximately 2.4 million deaths have been reported[1]. In the beginning, COVID-19 was considered an acute respiratory distress syndrome[2]. However, it was proven to be a multiple organ disease also affecting the heart[2,3]. Studies revealed that cases of critically ill COVID-19 patients had cardiovascular complications such as myocardial damage, venous thromboembolic events, and acute coronary syndromes[4]. The presence of underlying cardiovascular comorbidities in COVID-19 patients is associated with severe disease and poor prognosis including increased risk of onset of cardiovascular complications, increased progression of underlying cardiovascular disease and risk of increased mortality both in general population as well as in diagnosed patients[5].
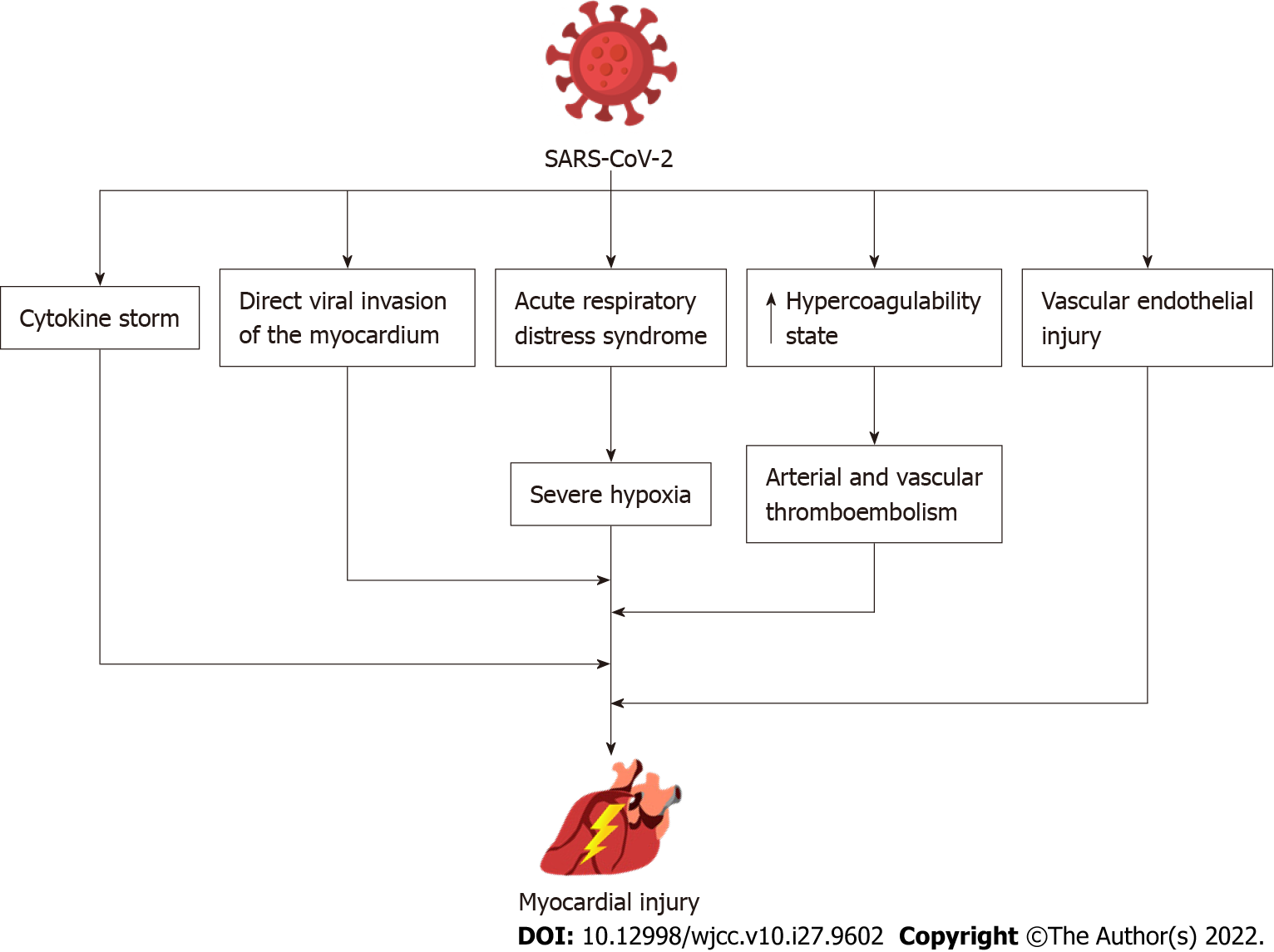
The pathophysiological mechanisms by which the severe acute respiratory syndrome coronavirus-2 (SARS-CoV-2) impacts the myocardium is still under investigation[6]. Cardiac cells express the angiotensin-converting enzyme 2 (ACE2) receptor through which the SARS-CoV-2 can bind and cause direct cardiac impairment and dysfunction of endothelial cells of coronary vessels[7]. Generally, the ACE2 has a cardioprotective role causing vasodilation, and having anti-fibrotic, anti-oxidative and anti-hypertrophic effects[8]. SARS-CoV-2 induces ACE2 downregulation which negatively affects its function, thus reduce its cardioprotective role and anti-inflammatory function[6]. Moreover, the heightened immunological response and the related cytokine storm can suppress cardiac contractility. It could also promote cardiovascular inflammation, atheromatic plaque instability, thrombocytosis, and oxygen supply-demand imbalance which may further contribute to cardiac dysfunction[7,8] (Figure 1).
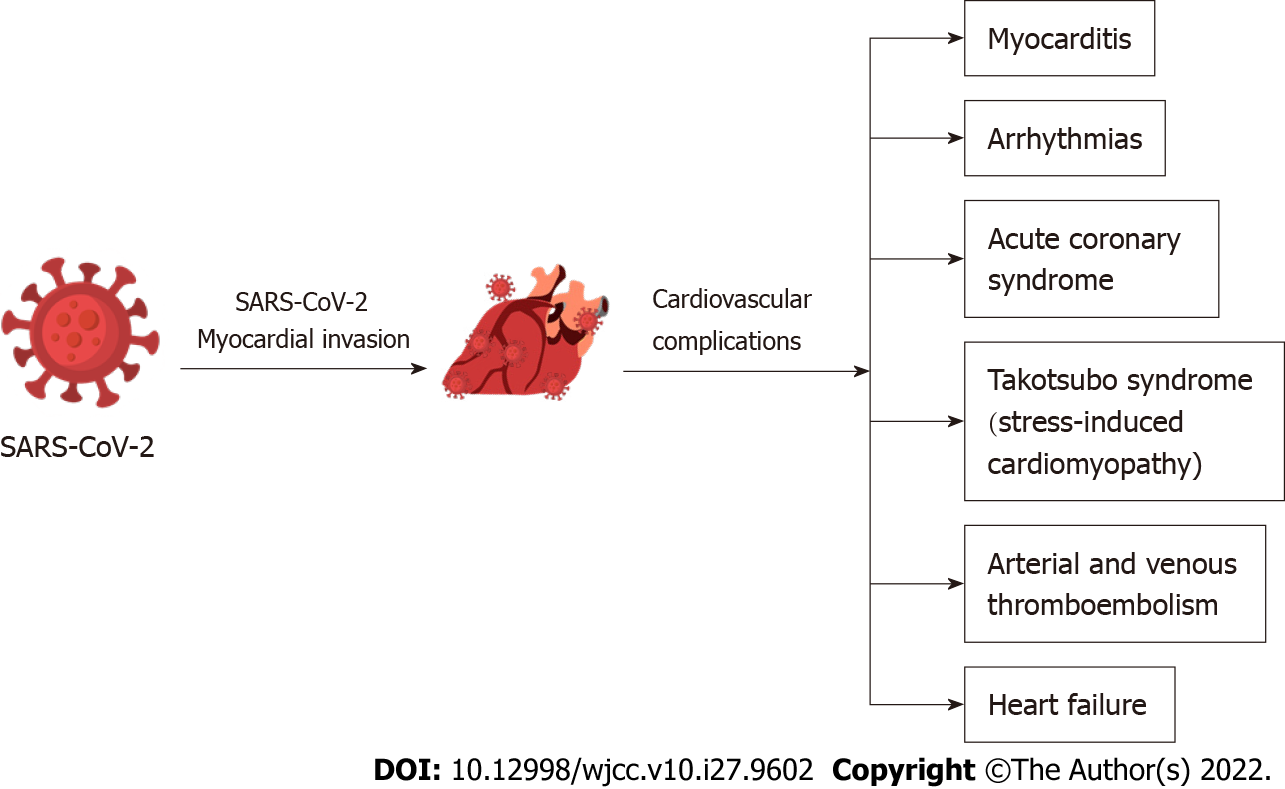
Although the major clinical manifestation of COVID-19 is viral respiratory infection, it may also be linked to cardiovascular disease[9]. The later were observed particularly in elderly individuals, those with underlying comorbidities and cardiovascular risk factors like malignancies, obesity, diabetes mellitus, and hypertension[10]. A current study showed that obesity accounted for 30% of COVID-19 hospitalized patients, hypertension for 26%, heart failure (HF) for 12% and diabetes mellitus for 21%[2]. Several clinical cardiovascular complications have been associated with SARS-CoV-2 infection including myocarditis, arrhythmias, pericardial effusion, acute coronary syndromes, myocardial injury and myocardial infarction, new onset or progression of preexisting HF, arterial and venous thromboembolism, cardiogenic shock and cardiac arrest[7,10,11] (Figure 2). Moreover, recent studies have indicated that the main laboratory findings of hospitalized COVID-19 patients were the elevated levels of high sensitivity cardiac troponin I, creatine kinase-MB, N-terminal pro-brain natriuretic peptide (NT-proBNP), and C-reactive protein. Moreover, electrocardiogram (ECG) findings may include diffuse ST-segment elevation and ST-segment depression[12].
COVID-19 is associated with cardiac involvement and cardiac inflammation. A recent cohort study from Germany on patients recently recovered from COVID-19 infection demonstrated cardiac involvement and ongoing myocardial inflammation in 78% and 60% of patients on cardiovascular magnetic resonance (CMR) imaging, respectively, independent of pre-existing comorbidity, severity and overall trajectory of the acute illness and time since the COVID-19 infection[13]. In COVID-19 individuals, elevated levels of troponin along with features of myocardial damage were observed several days following the onset of fever[5]. High troponin levels were associated with poor prognosis and increased mortality rates[14]. Interestingly, in a retrospective study from Wuhan including 187 COVID-19 patients, mortality was 8% in patients without medical history of cardiovascular disease and normal troponin T levels, 13% in patients with medical history of cardiovascular disease and normal troponin T levels, 38% in patients without medical history of cardiovascular disease but elevated troponin T levels and 69% for those with both medical history of cardiovascular disease and high troponin T levels[15]. Myocardial damage was associated with significant rise in NT-proBNP, with a mean NT-proBNP value of 72 pg/mL, in those who recovered and 800 pg/mL in COVID-19 patients who died[2]. Thus, recent recommendations suggest the evaluation of cardiac enzymes such as cardiac troponin, NT-proBNP, and BNP levels in COVID-19 patients as these biomarkers are frequently increased in myocarditis[16]. The incidence rate of myocarditis may range from 4.5% to 30%[17]. The proposed pathophysiological mechanisms of COVID-19 mediated myocardial injury is still under investigation. The direct infection of myocardial cells through the adhesion of the virus in ACE2 receptors and viral replication within the myocardium is the most prominent mechanism although the excessive immunological response caused by the SARS-CoV-2 may trigger myocardial injury[14,17]. In addition, myocardial damage may be provoked by hypoxia and respiratory failure[5]. Other proposed causes of myocardial involvement or damage include cardiac inflammation including myopericarditis and myocarditis. Clinically, COVID-19 patients presented with tachycardia, third heart sound, hypotension, tachypnea, and signs of reduced cardiac output[14]. An urgent cardiac manifestation of SARS-CoV-2 infection is the development of fulminant myocarditis characterized by HF and ventricular dysfunction[16]. Usually, it appears two to three weeks following the viral infection[16]. Furthermore, non-specific abnormal electrocardiographic findings were observed in individuals with SARS-CoV-2 mediated myocarditis. The most frequent ECG findings were ST-segment changes in 40% and cardiac arrhythmias in 38% including atrial fibrillation, atrial flutter, sinus tachycardia and bradycardia, conduction blocks, premature atrial and ventricular contractions[7].
In conclusion, myocardial damage is frequently observed in individuals infected with SARS-CoV-2. However, various pathophysiologic mechanisms have been implicated in the development of COVID-19 myocardial damage[18]. Considering that troponin is a non-specific biomarker of cardiac injury, the American College of Cardiology recommends that the levels of troponin should be interpreted along with other validated clinical findings and not solely connected to acute myocardial infarction[18]. Based on the recommendations of American Heart Association, COVID-19 individuals presented with signs resembling myocarditis, they should be tested with various cardiac techniques including cardiac echocardiography, cardiac computed tomography scan, and CMR[16]. Finally, both the American Heart Association and the European Society of Cardiology highlight the diagnostic importance of endomyocardial biopsy for confirming myocarditis even though several limitations have been depicted[16].
A frequent cardiac manifestation in COVID-19 patients is arrhythmias. Emerging evidence suggests association of COVID-19 with high incidence of cardiac arrythmias. COVID-19 infection may cause injury to cardiac myocytes, thereby increasing the risk of cardiac arrythmia. Sinus tachycardia is the most prevalent arrhythmia in COVID-19 patients. However, atrial fibrillation (AF), atrial flutter, and monomorphic or polymorphic ventricular tachycardia (torsades de pointes) are the most likely pathogenic arrhythmias. A recent study found that 18% of COVID-19 individuals presented with arrhythmias and the most prevalent types were AF and atrial flutter[2]. Arévalos et al[17] showed that among the commonest arrhythmias, AF was occurred in 3.6% of COVID-19 cases, non-sustained ventricular tachycardia in 1.4%, bradyarrhythmias and cardiac arrest in 1.3%. The prevalence of AF was found to be 11% in a meta-analysis of 19 observational studies involving 21653 individuals hospitalized with COVID-19[19]. AF was more prevalent in older patients (13% in ≥ 60 years of age vs 5% in younger patients < 60 years of age) and those with severe COVID-19 (19% in severe vs 3% in mild COVID-19)[19]. Moreover, in a world-wide survey of 4526 hospitalized COVID-19 patients, 827 were identified having a new cardiac arrhythmia and cardiovascular comorbidities such as hypertension, diabetes mellitus, coronary artery disease, congestive HF were more common in the patients who developed cardiac arrhythmias[20]. Concerning the life-threatening arrhythmias (ventricular tachycardia and ventricular fibrillation), these were presented in 4%-6% of COVID-19 hospitalized patients and particularly in those with increased troponin levels[2]. Also, high NT-ProBNP was correlated to the onset of life-threatening arrhythmias[14]. Major risk factors related to arrhythmias development include hypoxemia, myocardial ischemia or injury, electrolyte imbalance (mainly hypokalaemia and hypomagnesemia), drugs that cause QT prolongation, and shock[7,17]. The presence of cardiac arrhythmias was observed mostly in COVID-19 patients hospitalized in intensive care unit (ICU) (44.4%) compared to those in non-ICU setting (6.9%)[12].
Recent observational studies from France and Italy have also reported increased risk of out of hospital cardiac arrest during the COVID-19 pandemic. Studies from Italy and France have reported 60% and 50% increase in the rate of out of hospital cardiac arrest during the peak period of COVID-19 in 2020 relative to the same time frame in 2019, respectively[21,22]. This finding on increased prevalence of cardiac arrest could be linked to COVID-19 infections, pandemic-related stress, or a delay in seeking medical help by persons experiencing cardiac symptoms. In addition, data from the Swedish registry for cardiopulmonary resuscitation showed a decrease in in-hospital cardiac arrest due to myocardial infarction from 33.9% in the pre-pandemic period to 23.9% during the pandemic. Medical history of HF or myocardial infarction were less common in COVID-19 cases whereas diabetes was more common[23].
A 20% (range 5%-38%) of COVID-19 infected patients were diagnosed with acute coronary syndrome[24,25]. Patients with chronic coronary heart disease have an increased risk of myocardial injury, compared to patients who have not previously been diagnosed with coronary heart disease (43.8% vs 14.4%)[26]. Even though the exact pathophysiologic mechanism is still under investigation, various mechanisms have been proposed like myocardial damage due to direct viral invasion and cytokine storm, oxygen supply and demand mismatch due to severe hypoxia, endothelial damage, and thrombosis of cardiac vasculature[24]. SARS-CoV-2 may lead to myocardial infarction as it induces development of thrombus and instability of atheromatic plaque[24]. Remarkably, myocardial infarction with nonobstructive coronary arteries is another common manifestation of COVID-19[24].
Takotsubo syndrome (TTS) or stress-induced cardiomyopathy has been also characterized as a manifestation of COVID-19 infection[27]. Several mechanisms including increased sympathetic response, psychological distress, cytokine storm and microvascular dysfunction in COVID-19 patients have been proposed. It is clinically manifested as a reversible and temporary left ventricular dysfunction, without considerable occlusion of the coronary arteries, caused by major physical and psychological stress[7,27]. Notably, individuals suffering from physical stress (e.g., hypoxemia, infection) have worse prognosis as opposed to those under psychological stress. It is most prevalent in postmenopausal women (> 90%) and the most common type is the apical one (> 80%)[27]. The incidence rate of TTS has been reported to be between 2% and 4%[24]. A suggested mechanism of TTS is the excessive production of catecholamines in the bloodstream which results in the excessive stimulation of sympathetic system and thus in cardiac stress[27]. Furthermore, increased levels of cortisol were found in COVID-19 infected patients which along with the high catecholamine levels may have immediate cardiotoxicity[27]. Further studies are needed to elucidate the pathophysiological mechanisms as well as the contribution of stress hormones in TTS development[27]. Though the evidence on association between stress-induced cardiomyopathy and COVID-19 is growing, further studies are required both in COVID-19 infected patients as well as general population experiencing severe psychological distress due to the COVID-19 pandemic. Understanding these interactions may inform targeted individual treatments and population-level interventions to protect individuals at high-risk of stress-induced cardiomyopathy due to COVID-19.
SARS-CoV-2 infection is characterized as a hypercoagulability state which eventually can lead to thromboembolism[9]. Both arterial and venous thromboembolism have been observed in severe cases of COVID-19 infection, presenting mainly as pulmonary embolism, disseminated intravascular coagulopathy and deep venous thrombosis[2]. Early studies revealed that critical COVID-19 individuals with thromboembolic events had poor prognosis[18]. The laboratory findings of such patients showed high D-Dimer levels, low platelets, and mild prolongation of prothrombin time[9]. Remarkably, individuals who presented with cardiac injury had higher D-Dimer levels, decreased prothrombin time, antithrombin levels and activated partial thromboplastin time as opposed to those without any sign of myocardial injury[2].
The pathophysiological mechanisms leading to hypercoagulability still remain unknown[9]. So far, several indirect and direct mechanisms of hypercoagulability have been proposed[14]. Particularly, the excessive inflammatory response, vascular endothelial injury, activation of coagulation pathways, and prolonged immobilization represent frequent manifestations of SARS-CoV-2 infection[7].
Recent meta-analyses showed that COVID-19 hospitalized patients had an incidence rate of venous thromboembolism ranging between 15% and 21% which was four times higher in ICU patients compared to their non-ICU counterparts[2]. Moreover, prior diagnosis of venous thromboembolism results in a threefold increase in mortality in patients with HF hospitalized with COVID-19[28]. Recent guidelines suggest that COVID-19 hospitalized patients should be under thromboprophylaxis with low molecular weight heparin, unfractionated heparin or fondaparinux in order to control this hypercoagulability state[2]. However, the regimen of choice, frequency, and duration of thromboprophylaxis therapy is still to be determined[2].
Acute HF is a common manifestation of SARS-CoV-2 infection and has been related to increased mortality[29]. One third of COVID-19 patients with prior history of HF had an acute decompensation during hospitalization. However, COVID-19 can also cause “de novo” HF with an incidence ranging between 2.5% and 4.5%. It has been demonstrated that patients with new onset HF have a significantly higher mortality (46.8% vs 19.7%; P < 0.001)[30]. Numerous mechanisms of COVID-19 mediated HF have been proposed like the infiltration of myocardium by immune cells induced by the SARS-CoV-2 that may cause cardiac dysfunction, the release of cytokines may promote cardiac necrosis and eventually myocardial failure. In addition, hypoxia as a result of acute respiratory distress syndrome and respiratory failure may cause HF. Besides, endocardial dysfunction due to endothelial damage and subsequent formation of thrombi may contribute to COVID-19 associated HF[31]. Importantly, pre-existing cardiovascular comorbidities mostly arrhythmias and chronic HF were considered to be the main predictive risk factors of COVID-19 induced HF[32]. Specifically, in the initial stages of COVID-19 infection patients present with HF and preserved ejection fraction whereas in advanced stages HF with systolic dysfunction can occur. HF can occur either as a consequence of pre-existing chronic HF or recently developed HF[32].
Antiviral agents act on several stages of SARS-CoV-2 infection pathways like viral entry into human cells, as well as activation and replication of the virus[33]. Remdesivir is an antiviral drug that inhibits the replication phase of the virus leading to premature ending of viral cycle. Current studies revealed a decrease in the viral burden and pulmonary infection following therapy with remdesivir. Additionally, individuals treated with remdesivir showed limited adverse events and low mortality rates especially when it was combined with other therapies or used in early disease stages[34]. Remdesivir is given as a single loading dose of 200 mg and then as 100 mg daily infusion. However, the therapeutic dose of remdesivir does not need any adjustment in patients with renal or hepatic impairment[35]. The main side effects of remdesivir therapy included reaction on the injection site, as well as gastrointestinal and hepatic dysfunction[35].
Oseltamivir is another antiviral drug used for the management of SARS-CoV-2 infection. Even though the available data are still controversial, a beneficial effect of oseltamivir therapy was the reduction of recovery phase (1 d) of COVID-19 infected individuals[33].
Other anti-viral drugs used in COVID-19 disease include the lopinavir, ribavirin and ritonavir[34]. Lopinavir/ritonavir treatment is mostly given in moderate COVID-19 cases during the seven to ten days following symptoms onset[35]. Studies revealed that COVID-19 patients who underwent treatment with lopinavir/ritonavir had no improvement of their clinical status, viral load and 28-d mortality rate[34,35].
Chloroquine and hydroxychloroquine agents were extensively used in the beginning of the COVID-19 pandemic because they act by inhibiting the entry of virus into the host cells thus reducing the immune response[33]. Interestingly, a meta-analysis showed that the use of chloroquine and hydroxychloroquine (either alone or in combination with) azithromycin not only was ineffective, but it was also associated with a higher risk of adverse events[33]. Specifically, hydroxychloroquine can lead to cardiac arrest and death due to prolongation of QTc interval. It is recommended to assess QTc prolongation with ECG in the onset of therapy and following its termination. In general, adverse effects of these drugs occur rare but if they occur, they are serious and irreversible[18].
Importantly, several therapeutic agents used for the management of COVID-19 disease showed possible interactions with cardiovascular drugs and cardiovascular manifestations. Antiviral drugs like lopinavir and ritonavir may cause PR and QT intervals prolongation[18].
Vitamin C has been examined as a therapeutic agent in COVID-19 patients due to its anti-oxidative role[33]. However, further research is needed for determining the outcomes of vitamin C therapy in COVID-19 patients. Moreover, the use of Vitamin D as a prophylactic therapy was suggested for COVID-19 infected individuals but still the current data are limited concerning the inclusion of vitamin D in the therapeutic protocol[33].
It is critical to find low-cost, broadly available, and effective COVID-19 treatments. The idea of repurposing current drugs that are widely available and have well-understood safety profiles is of clinical and public health interest. In 2020, corticosteroids were approved by the Food and Drug Administration (FDA) as a COVID-19 therapy of critical ill patients. Dexamethasone was related to decreased mortality rates for those being on mechanical ventilation and increased need of oxygen, as well as decreased hospitalization rate[34].
Ivermectin, which is normally used as an antiparasitic medicine, is one drug that has received considerable public attention. Ivermectin is a helminthic parasite inhibitor that has been proven to be effective in treating onchocerciasis, strongyloidiasis, and ectoparasitic infection (e.g., scabies). However, in a large randomised controlled trial from Brazil, in outpatients with an early diagnosis of COVID-19, ivermectin did not lead to a lower incidence of medical admission to a hospital due to COVID-19 progression or prevented prolonged emergency department monitoring[36].
Convalescent plasma (CP) was approved by FDA as an investigational therapy for patients infected by SARS-CoV-2[34]. Patients treated with CP showed a good safety profile with decreased requirements for mechanical ventilation and improvement of inflammatory markers, oxygen levels and one-month survival rates[34]. Additionally, it may reduce the excessive immunological activation[34]. Studies found that individuals with severe COVID-19 disease showed better improvement following CP therapy. CP therapy was subsequently approved by the FDA in emergency cases[37].
In summary, although not common, cardiac arrythmia may be observed as a cardiovascular complication in COVID-19 patients. Patients with myocardial damage, myocardial ischemia, hypoxia, shock, electrolyte imbalances, or those using drugs that prolong the QT interval are all at risk of arrhythmias after COVID-19. Patients with suspected COVID-19 should have a baseline cardiac investigation (with ECG). Despite some promising therapies, further studies on new drugs and treatment of COVID-19 are warranted. Furthermore, existing therapies and future drugs need to factor in drug-to-drug and cardiovascular interactions as well as impact of COVID-19 on cardiovascular system.
Provenance and peer review: Invited article; Externally peer reviewed.
Peer-review model: Single blind
Specialty type: Cardiac and cardiovascular systems
Country/Territory of origin: Greece
Peer-review report’s scientific quality classification
Grade A (Excellent): 0
Grade B (Very good): B
Grade C (Good): C
Grade D (Fair): 0
Grade E (Poor): 0
P-Reviewer: Chavan RP, India; Saeed MAM, Egypt S-Editor: Zhang H L-Editor: A P-Editor: Zhang H
| 1. | Chung MK, Zidar DA, Bristow MR, Cameron SJ, Chan T, Harding CV 3rd, Kwon DH, Singh T, Tilton JC, Tsai EJ, Tucker NR, Barnard J, Loscalzo J. COVID-19 and Cardiovascular Disease: From Bench to Bedside. Circ Res. 2021;128:1214-1236. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 226] [Cited by in RCA: 246] [Article Influence: 61.5] [Reference Citation Analysis (2)] |
| 2. | Cenko E, Badimon L, Bugiardini R, Claeys MJ, De Luca G, de Wit C, Derumeaux G, Dorobantu M, Duncker DJ, Eringa EC, Gorog DA, Hassager C, Heinzel FR, Huber K, Manfrini O, Milicic D, Oikonomou E, Padro T, Trifunovic-Zamaklar D, Vasiljevic-Pokrajcic Z, Vavlukis M, Vilahur G, Tousoulis D. Cardiovascular disease and COVID-19: a consensus paper from the ESC Working Group on Coronary Pathophysiology & Microcirculation, ESC Working Group on Thrombosis and the Association for Acute CardioVascular Care (ACVC), in collaboration with the European Heart Rhythm Association (EHRA). Cardiovasc Res. 2021;117:2705-2729. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 117] [Cited by in RCA: 97] [Article Influence: 24.3] [Reference Citation Analysis (0)] |
| 3. | Chatzis DG, Magounaki KT, Pantazopoulos IN, Johnson EO, Tsioufis KP. COVID-19 pandemic and cardiovascular disease: where do we stand? Minerva Cardioangiol. 2020;68:347-358. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 8] [Cited by in RCA: 9] [Article Influence: 1.8] [Reference Citation Analysis (0)] |
| 4. | Ho HT, Peischard S, Strutz-Seebohm N, Klingel K, Seebohm G. Myocardial Damage by SARS-CoV-2: Emerging Mechanisms and Therapies. Viruses. 2021;13. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 6] [Cited by in RCA: 15] [Article Influence: 3.8] [Reference Citation Analysis (0)] |
| 5. | Task Force for the management of COVID-19 of the European Society of Cardiology. Corrigendum to: European Society of Cardiology guidance for the diagnosis and management of cardiovascular disease during the COVID-19 pandemic: part 1-epidemiology, pathophysiology, and diagnosis; and ESC guidance for the diagnosis and management of cardiovascular disease during the COVID-19 pandemic: part 2-care pathways, treatment, and follow-up. Eur Heart J. 2022;43:1776. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 5] [Cited by in RCA: 24] [Article Influence: 6.0] [Reference Citation Analysis (0)] |
| 6. | Abdi A, AlOtaiby S, Badarin FA, Khraibi A, Hamdan H, Nader M. Interaction of SARS-CoV-2 with cardiomyocytes: Insight into the underlying molecular mechanisms of cardiac injury and pharmacotherapy. Biomed Pharmacother. 2022;146:112518. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 16] [Cited by in RCA: 35] [Article Influence: 11.7] [Reference Citation Analysis (0)] |
| 7. | Šikić J, Planinić Z, Matišić V, Friščić T, Molnar V, Jagačić D, Vujičić L, Tudorić N, Postružin Gršić L, Ljubičić Đ, Primorac D. COVID-19: The Impact on Cardiovascular System. Biomedicines. 2021;9. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 3] [Cited by in RCA: 2] [Article Influence: 0.5] [Reference Citation Analysis (0)] |
| 8. | Magadum A, Kishore R. Cardiovascular Manifestations of COVID-19 Infection. Cells. 2020;9. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 124] [Cited by in RCA: 134] [Article Influence: 26.8] [Reference Citation Analysis (0)] |
| 9. | Nishiga M, Wang DW, Han Y, Lewis DB, Wu JC. COVID-19 and cardiovascular disease: from basic mechanisms to clinical perspectives. Nat Rev Cardiol. 2020;17:543-558. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 653] [Cited by in RCA: 886] [Article Influence: 177.2] [Reference Citation Analysis (0)] |
| 10. | Mottola FF, Verde N, Ricciolino R, Di Mauro MD, Migliaccio MG, Carfora V, Spiniello G, Coppola N; Vanvitelli Covid-Group. Cardiovascular System in COVID-19: Simply a Viewer or a Leading Actor? Life (Basel). 2020;10. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 5] [Cited by in RCA: 5] [Article Influence: 1.0] [Reference Citation Analysis (0)] |
| 11. | Kang Y, Chen T, Mui D, Ferrari V, Jagasia D, Scherrer-Crosbie M, Chen Y, Han Y. Cardiovascular manifestations and treatment considerations in COVID-19. Heart. 2020;106:1132-1141. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 252] [Cited by in RCA: 262] [Article Influence: 52.4] [Reference Citation Analysis (0)] |
| 12. | Adu-Amankwaah J, Mprah R, Adekunle AO, Ndzie Noah ML, Adzika GK, Machuki JO, Sun H. The cardiovascular aspect of COVID-19. Ann Med. 2021;53:227-236. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 36] [Cited by in RCA: 17] [Article Influence: 4.3] [Reference Citation Analysis (0)] |
| 13. | Puntmann VO, Carerj ML, Wieters I, Fahim M, Arendt C, Hoffmann J, Shchendrygina A, Escher F, Vasa-Nicotera M, Zeiher AM, Vehreschild M, Nagel E. Outcomes of Cardiovascular Magnetic Resonance Imaging in Patients Recently Recovered From Coronavirus Disease 2019 (COVID-19). JAMA Cardiol. 2020;5:1265-1273. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 1240] [Cited by in RCA: 1468] [Article Influence: 293.6] [Reference Citation Analysis (0)] |
| 14. | Azevedo RB, Botelho BG, Hollanda JVG, Ferreira LVL, Junqueira de Andrade LZ, Oei SSML, Mello TS, Muxfeldt ES. Covid-19 and the cardiovascular system: a comprehensive review. J Hum Hypertens. 2021;35:4-11. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 240] [Cited by in RCA: 221] [Article Influence: 55.3] [Reference Citation Analysis (0)] |
| 15. | Guo T, Fan Y, Chen M, Wu X, Zhang L, He T, Wang H, Wan J, Wang X, Lu Z. Cardiovascular Implications of Fatal Outcomes of Patients With Coronavirus Disease 2019 (COVID-19). JAMA Cardiol. 2020;5:811-818. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 2516] [Cited by in RCA: 2842] [Article Influence: 568.4] [Reference Citation Analysis (0)] |
| 16. | Siripanthong B, Nazarian S, Muser D, Deo R, Santangeli P, Khanji MY, Cooper LT Jr, Chahal CAA. Recognizing COVID-19-related myocarditis: The possible pathophysiology and proposed guideline for diagnosis and management. Heart Rhythm. 2020;17:1463-1471. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 527] [Cited by in RCA: 503] [Article Influence: 100.6] [Reference Citation Analysis (0)] |
| 17. | Arévalos V, Ortega-Paz L, Rodríguez-Arias JJ, Calvo López M, Castrillo-Golvano L, Salazar-Rodríguez A, Sabaté-Tormos M, Spione F, Sabaté M, Brugaletta S. Acute and Chronic Effects of COVID-19 on the Cardiovascular System. J Cardiovasc Dev Dis. 2021;8. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 15] [Cited by in RCA: 11] [Article Influence: 2.8] [Reference Citation Analysis (0)] |
| 18. | Matsushita K, Marchandot B, Jesel L, Ohlmann P, Morel O. Impact of COVID-19 on the Cardiovascular System: A Review. J Clin Med. 2020;9. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 36] [Cited by in RCA: 32] [Article Influence: 6.4] [Reference Citation Analysis (0)] |
| 19. | Li Z, Shao W, Zhang J, Ma J, Huang S, Yu P, Zhu W, Liu X. Prevalence of Atrial Fibrillation and Associated Mortality Among Hospitalized Patients With COVID-19: A Systematic Review and Meta-Analysis. Front Cardiovasc Med. 2021;8:720129. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 42] [Cited by in RCA: 40] [Article Influence: 10.0] [Reference Citation Analysis (0)] |
| 20. | Plastiras SC, Moutsopoulos HM. Arrhythmias and Conduction Disturbances in Autoimmune Rheumatic Disorders. Arrhythm Electrophysiol Rev. 2021;10:17-25. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 4] [Cited by in RCA: 16] [Article Influence: 4.0] [Reference Citation Analysis (0)] |
| 21. | Baldi E, Sechi GM, Mare C, Canevari F, Brancaglione A, Primi R, Klersy C, Palo A, Contri E, Ronchi V, Beretta G, Reali F, Parogni P, Facchin F, Bua D, Rizzi U, Bussi D, Ruggeri S, Oltrona Visconti L, Savastano S; Lombardia CARe Researchers. Out-of-Hospital Cardiac Arrest during the Covid-19 Outbreak in Italy. N Engl J Med. 2020;383:496-498. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 436] [Cited by in RCA: 519] [Article Influence: 103.8] [Reference Citation Analysis (0)] |
| 22. | Baldi E, Sechi GM, Mare C, Canevari F, Brancaglione A, Primi R, Klersy C, Palo A, Contri E, Ronchi V, Beretta G, Reali F, Parogni P, Facchin F, Rizzi U, Bussi D, Ruggeri S, Oltrona Visconti L, Savastano S; Lombardia CARe researchers. COVID-19 kills at home: the close relationship between the epidemic and the increase of out-of-hospital cardiac arrests. Eur Heart J. 2020;41:3045-3054. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 185] [Cited by in RCA: 177] [Article Influence: 35.4] [Reference Citation Analysis (0)] |
| 23. | Sultanian P, Lundgren P, Strömsöe A, Aune S, Bergström G, Hagberg E, Hollenberg J, Lindqvist J, Djärv T, Castelheim A, Thorén A, Hessulf F, Svensson L, Claesson A, Friberg H, Nordberg P, Omerovic E, Rosengren A, Herlitz J, Rawshani A. Cardiac arrest in COVID-19: characteristics and outcomes of in- and out-of-hospital cardiac arrest. A report from the Swedish Registry for Cardiopulmonary Resuscitation. Eur Heart J. 2021;42:1094-1106. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 56] [Cited by in RCA: 86] [Article Influence: 21.5] [Reference Citation Analysis (0)] |
| 24. | Esposito L, Cancro FP, Silverio A, Di Maio M, Iannece P, Damato A, Alfano C, De Luca G, Vecchione C, Galasso G. COVID-19 and Acute Coronary Syndromes: From Pathophysiology to Clinical Perspectives. Oxid Med Cell Longev. 2021;2021:4936571. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 9] [Cited by in RCA: 28] [Article Influence: 7.0] [Reference Citation Analysis (0)] |
| 25. | Bavishi C, Bonow RO, Trivedi V, Abbott JD, Messerli FH, Bhatt DL. Special Article - Acute myocardial injury in patients hospitalized with COVID-19 infection: A review. Prog Cardiovasc Dis. 2020;63:682-689. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 209] [Cited by in RCA: 206] [Article Influence: 41.2] [Reference Citation Analysis (0)] |
| 26. | Schiavone M, Gasperetti A, Mancone M, Kaplan AV, Gobbi C, Mascioli G, Busana M, Saguner AM, Mitacchione G, Giacomelli A, Sardella G, Viecca M, Duru F, Antinori S, Carugo S, Bartorelli AL, Tondo C, Galli M, Fedele F, Forleo GB. Redefining the Prognostic Value of High-Sensitivity Troponin in COVID-19 Patients: The Importance of Concomitant Coronary Artery Disease. J Clin Med. 2020;9. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 19] [Cited by in RCA: 29] [Article Influence: 5.8] [Reference Citation Analysis (0)] |
| 27. | Salah HM, Mehta JL. Takotsubo cardiomyopathy and COVID-19 infection. Eur Heart J Cardiovasc Imaging. 2020;21:1299-1300. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 12] [Cited by in RCA: 19] [Article Influence: 3.8] [Reference Citation Analysis (0)] |
| 28. | Mok J, Malpartida JC, O'Dell K, Davis J, Gao C, Manyam H. Vascular comorbidities worsen prognosis of patients with heart failure hospitalised with COVID-19. Open Heart. 2021;8. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 3] [Cited by in RCA: 3] [Article Influence: 0.8] [Reference Citation Analysis (0)] |
| 29. | Zuin M, Zuliani G, Roncon L, Rigatelli G. Heart failure as a complication of COVID-19 infection: systematic review and meta-analysis. Eur Heart J. 2021;42. [RCA] [DOI] [Full Text] [Cited by in Crossref: 2] [Cited by in RCA: 2] [Article Influence: 0.5] [Reference Citation Analysis (0)] |
| 30. | Tomasoni D, Inciardi RM, Lombardi CM, Tedino C, Agostoni P, Ameri P, Barbieri L, Bellasi A, Camporotondo R, Canale C, Carubelli V, Carugo S, Catagnano F, Dalla Vecchia LA, Danzi GB, Di Pasquale M, Gaudenzi M, Giovinazzo S, Gnecchi M, Iorio A, La Rovere MT, Leonardi S, Maccagni G, Mapelli M, Margonato D, Merlo M, Monzo L, Mortara A, Nuzzi V, Piepoli M, Porto I, Pozzi A, Sarullo F, Sinagra G, Volterrani M, Zaccone G, Guazzi M, Senni M, Metra M. Impact of heart failure on the clinical course and outcomes of patients hospitalized for COVID-19. Results of the Cardio-COVID-Italy multicentre study. Eur J Heart Fail. 2020;22:2238-2247. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 67] [Cited by in RCA: 100] [Article Influence: 20.0] [Reference Citation Analysis (0)] |
| 31. | Adeghate EA, Eid N, Singh J. Mechanisms of COVID-19-induced heart failure: a short review. Heart Fail Rev. 2021;26:363-369. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 55] [Cited by in RCA: 48] [Article Influence: 12.0] [Reference Citation Analysis (0)] |
| 32. | Italia L, Tomasoni D, Bisegna S, Pancaldi E, Stretti L, Adamo M, Metra M. COVID-19 and Heart Failure: From Epidemiology During the Pandemic to Myocardial Injury, Myocarditis, and Heart Failure Sequelae. Front Cardiovasc Med. 2021;8:713560. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 34] [Cited by in RCA: 76] [Article Influence: 19.0] [Reference Citation Analysis (0)] |
| 33. | Rodriguez-Guerra M, Jadhav P, Vittorio TJ. Current treatment in COVID-19 disease: a rapid review. Drugs Context. 2021;10. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 44] [Cited by in RCA: 38] [Article Influence: 9.5] [Reference Citation Analysis (0)] |
| 34. | Dong Y, Shamsuddin A, Campbell H, Theodoratou E. Current COVID-19 treatments: Rapid review of the literature. J Glob Health. 2021;11:10003. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 26] [Cited by in RCA: 22] [Article Influence: 5.5] [Reference Citation Analysis (0)] |
| 35. | Chen PL, Lee NY, Cia CT, Ko WC, Hsueh PR. A Review of Treatment of Coronavirus Disease 2019 (COVID-19): Therapeutic Repurposing and Unmet Clinical Needs. Front Pharmacol. 2020;11:584956. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 47] [Cited by in RCA: 43] [Article Influence: 8.6] [Reference Citation Analysis (0)] |
| 36. | Reis G, Silva EASM, Silva DCM, Thabane L, Milagres AC, Ferreira TS, Dos Santos CVQ, Campos VHS, Nogueira AMR, de Almeida APFG, Callegari ED, Neto ADF, Savassi LCM, Simplicio MIC, Ribeiro LB, Oliveira R, Harari O, Forrest JI, Ruton H, Sprague S, McKay P, Guo CM, Rowland-Yeo K, Guyatt GH, Boulware DR, Rayner CR, Mills EJ; Together Investigators. Effect of Early Treatment with Ivermectin among Patients with Covid-19. N Engl J Med. 2022;386:1721-1731. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 155] [Cited by in RCA: 149] [Article Influence: 49.7] [Reference Citation Analysis (0)] |
| 37. | Tsao CW, Strom JB, Chang JD, Manning WJ. COVID-19-Associated Stress (Takotsubo) Cardiomyopathy. Circ Cardiovasc Imaging. 2020;13:e011222. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 26] [Cited by in RCA: 40] [Article Influence: 8.0] [Reference Citation Analysis (0)] |