INTRODUCTION
The systemic nature of cirrhosis is becoming increasingly evident, expanding well beyond liver function and direct Newtonian complications of portal hypertension (PHT). Disease severity ranges from advanced chronic liver disease (ACLD) with mild portal hypertension, to decompensated disease and a late decompensation stage, with significant, progressive hemodynamic impairment[1]. Systemic involvement includes individual organ dysfunctions accompanying classical events (such as the hepato-renal syndrome in ascites or hepatic encephalopathy in variceal bleeding), longstanding slowly progressive entities (sarcopenia), or the ultimate multi organic storm: acute-on-chronic liver failure.
The kidneys, brain, heart, and lungs are closely intertwined with liver function and consecutive vascular alterations. However, while kidney dysfunction and hepatic encephalopathy represent common clinical issues that are regularly recognized, treated, and studied by hepatologists across the board, pulmonary involvement is typically overlooked and examined only in expert settings and liver transplantation units, despite their relatively high prevalence. Lung involvement in liver disease consists of three major entities: Hepato-pulmonary syndrome (HPS), porto-pulmonary hypertension (PoPH), and hepatic hydrothorax (HH). All three conditions have distinct pathophysiology, which is in close correlation to the adapted homeostasis of ACLD. The current minireview aims to shed light on the three complications in a structured manner, from definitions to summarizing the main research themes, and, ultimately to expand on one under-discussed clinical issue for each entity.
HEPATOPULMONARY SYNDROME - THE UNRECOGNIZED DECOMPENSATION
Definition and diagnostic criteria
Hepatopulmonary syndrome is defined by the presence of two key elements encountered in a patient with cirrhosis, portal hypertension, or congenital portosystemic shunts: abnormal arterial oxygenation and the presence of intrapulmonary vascular dilations (IPVDs) in the absence of underlying lung disease. The complete diagnostic criteria are depicted in Figure 1.
Figure 1 Pathophysiology and diagnostic criteria of the hepatopulmonary syndrome.
A-a: Alveolo-arterial; US: Ultrasonography.
Context and a brief history
Lung involvement in cirrhosis has long been recognized, as cyanosis and finger clubbing were among the earliest clinical signs associated with longstanding liver disease, with reports dating back to the late 19th century by Austrian physician and researcher M. Fluckinger[2]. Yet, almost one hundred years had passed before the term “Hepatopulmonary Syndrome” was first coined by Timothy Kennedy and Ronald Knudson in 1977 in an illustrative case report and literature review[3]. In their paper, the authors attempt to crayon the basic clinical profile of “a syndrome characterized by hypoxemia aggravated by exercise, orthodeoxia, hypocapnia, and evidence of hyperdynamic circulation” in patients with cirrhosis and no underlying pulmonary disease. They suggest that HPS might be determined by the “presence of a shunt or shunt-like mechanism consisting of low-resistance vascular communication within the lung,” suggesting a common pathophysiological pathway with hepatorenal syndrome. In the subsequent years, along with the emergence of liver transplantation (LT) as a definite curative solution for patients with cirrhosis and the lack of therapeutic solutions for HPS, research has begun on the impact of HPS on pre-and post-LT outcomes. Initially regarded as an absolute contraindication, evidence has shown the resolution of HPS post-LT. In 1997, Michael J. Krowka and his Mayo Clinic team suggested that HPS might even be considered as a primary indication for LT[4], paving the way towards MELD exceptions in 2006[5], with favorable outcomes recently reported both in the United States[6] and Europe[7]. Since, most of the research has been split between basic science (typically on portal hypertension animal models) and LT, disregarding the largest proportion of patients with ACLD not on LT waitlist.
Current treatment strategies
According to the most recent guidelines on the diagnosis and management of HPS and PoPH, the therapeutic options for HPS are extremely limited[8]. Currently, there is no effective pharmacological therapy approved for HPS, although there have been some tentative attempts with methylene blue, somatostatin, pentoxifylline, propranolol, antibiotics, sorafenib, or garlic with modest or null effects[9]. Although the evidence is primarily anecdotal, there appears to be little or no benefit in alleviating portal pressure by transjugular intrahepatic portosystemic shunt (TIPS) placement[10,11]. Therefore, the only valid strategies are supportive oxygen therapy to maintain O2 saturations above 88% and consideration for LT as the sole definite curative solution with excellent long-term results[7,8].
The (non)diagnostic challenge
HPS is theoretically encountered in up to one-third of the patients listed for LT, according to current estimates. The reported prevalence of HPS is highly heterogeneous, ranging between 5 and 32%, depending on the diagnostic criteria[12]. However, these estimates raise some concerns. All the figures are reported on LT candidates, theoretically skewing the estimates towards the more severe liver disease spectrum. On the other hand, it appears that the presence of HPS does not correlate with liver disease severity[13], which should support the presumption that HPS is also frequently encountered in patients not currently enlisted for LT. Yet, there seems to be little to no evidence of HPS in the literature beyond the realm of LT. Still, the main issue is not the inexact prevalence of HPS among patients with ACLD, but rather its implications of being under- or misdiagnosed. HPS appears to double the risk for waitlist mortality compared to non-HPS candidates[13], which might bear increasing relevance given the scarcity of therapeutic options.
In this light, one key question arises: Is HPS hiding in plain sight? The available evidence seems to point towards an affirmative response. A US-based study group has evaluated the rate of HPS diagnosis in a sizeable cirrhotic cohort, which included over 40000 patients from 28 medical centers, of which only two were LT centers. Moreover, using the available medical records, the authors have also analyzed whether the patients were correctly diagnosed, having both contrast-enhanced trans-thoracic heart ultrasonography with evidence of delayed shunting and abnormal arterial oxygenation (A-a gradient exceeding 15 mmHg). The results were in stark contrast with prior estimates. Of the 42749 unique patients, only 194 (0.45%) had a diagnosis of HPS. Furthermore, few met the current diagnostic criteria among these patients, as only 54% of them had evidence of delayed shunting, and 26% had documented abnormal oxygenation. Thus, only 41 patients ultimately fulfilled the criteria of HPS (22.5% of the patients initially diagnosed, and less than 0.1% of the entire cohort)[14]. The findings support the conclusion that HPS is grossly underdiagnosed outside LT centers, and, even when suspected, the diagnostic workup tends to be incomplete. This reflects a critical knowledge gap between transplantation hepatologists and their peers who work in non-LT centers and might provide sufficient ground to increase the visibility, education, and, not least, active screening for HPS.
Screening for HPS using pulse oximetry is highly convenient, as it takes less than 10 s to perform and is typically included within the regular clinical examination. However, screening for HPS appears to be less straightforward. In theory, the direct result of shunting is hypoxemia, which should be easily diagnosed using pulse oximetry and the current practice guidelines recommend a cut-off value of SpO2 < 96% for further testing[8]. Yet, recent evidence suggests that the area under the receiving operating characteristic (AUROC) curve is only 0.59, which is sub-par for a screening method. Moreover, SpO2 had a very low sensitivity (28%)[15]. Therefore, the path towards a more accurate HPS diagnosis using an efficient and easy-to-use screening method might face a serious roadblock, and its disentanglement should provide the basis for further research, as both contrast-enhanced heart ultrasonography and arterial blood gas analysis are less than ideal screening tools.
Summing up, our opinion is that beyond exact cut-offs and perfect diagnostic criteria, one must think of HPS each time when facing a patient with portal hypertension, as the silent clinical appearance of this entity is not detached from its proven prognostic significance.
PORTO-PULMONARY HYPERTENSION - THE UNTREATED DECOMPENSATION
Definition and diagnostic criteria
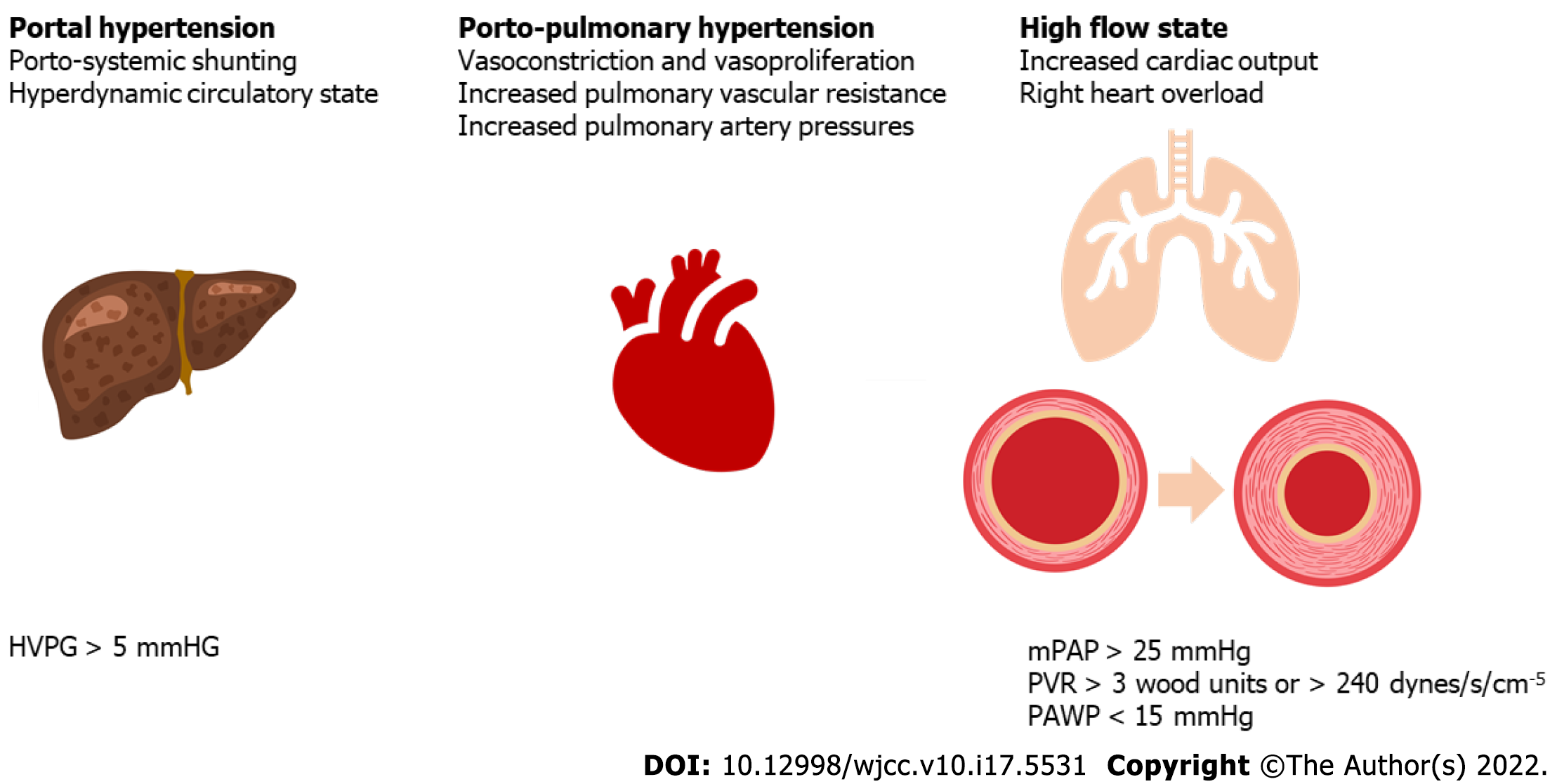
Porto-pulmonary hypertension is a pulmonary vascular complication defined by the presence of pulmonary arterial hypertension (PAH) as a direct consequence of PHT in a patient with no underlying pulmonary disease or left ventricular failure[9]. The gold standard for PoPH diagnosis is cardio-pulmonary hemodynamic assessment via right heart catheterization (RHC). The hemodynamic diagnostic triad for PoPH consists of increased mean pulmonary artery pressure (mPAP), due to increased pulmonary vascular resistance (PVR), in the setting of a normal pulmonary artery wedged pressure (PAWP)[8]. The hemodynamic criteria for POPH are depicted in Figure 2.
Figure 2 Hemodynamic definition criteria for porto-pulmonary hypertension.
HVPG: Hepatic venous pressure gradient; mPAP: Mean pulmonary artery pressure; PVR: Pulmonary vascular resistance; PAWP: Pulmonary artery wedged pressure.
Given that RHC is an invasive diagnostic method, transthoracic Doppler heart ultrasonography is recommended as a less invasive surrogate method to provide the grounds for further testing[8]. However, its effectiveness as a screening method is disputable, being more accurate at ruling out rather than ruling in PAH[16].
Context and a brief history
The concept of PoPH gained traction in hepatology in the early 1990s, prior reports being few and far between[17]. In the largest available series of patients with PHT and RHC, the reported prevalence of PoPH was 2%[18], although higher figures were reported in patients evaluated for LT, even up to 8.5%[19]. Similar to HPS, clinical research in PoPH is closely tied to LT. In contrast to HPS, though, the relationship between PoPH and LT is multifaceted. On the one hand, an mPAP < 35 mmHg appears to pose no additional risk for LT outcomes and, along with patients with mPAP > 35 mmHg who respond adequately to PAH-specific treatment, can benefit from MELD exceptions. On the other hand, severe PoPH, with mPAP exceeding 45-50 mmHg, is associated with severe postoperative complications and thus, is regarded as an absolute contraindication to LT[8]. Therefore, LT patients with PoPH walk on a tightrope between favorable LT odds if PAH is mild or adequately controlled and extremely low LT probability if PAH is unstable or progressive.
Therapeutic options - the role of PAH-specific treatment
The outcome of patients with PoPH is highly dependent on the therapeutic approach: patients with PoPH and no medical treatment had a dismal 14% 5-year survival[20], while data from the US Registry to Evaluate Early and Long-term Pulmonary Arterial Hypertension Disease Management (REVEAL) has shown that patients with pharmacological-only treatment, who did not benefit from LT had a 40% 5-year survival[21]. The statistics vastly improve for patients who benefited from both medical therapy and liver transplantation, with 5-year survival rates of 81% according to recent data from the large-scale French PoPH registry[22].
The staples of PoPH pharmacological treatment are phosphodiesterase 5-inhibitors (sildenafil), endothelin receptor antagonists (bosentan, ambrisentan, and macitentan), and prostanoids (epoprostenol, treprostinil, and inhaled iloprost). However, most of their therapeutic prowess is extrapolated from studies on PAH, with available data on PoPH barely exceeding a pooled 100 patients for each drug class, mostly comprised of small, non-controlled studies, typically including patients on LT waiting lists[9]. Yet when and how to treat patients with PoPH remains poorly defined and is largely up to the latitude of the LT center in which the patient is followed.
To this point, the PORTICO study is the only randomized controlled trial published on the pharmacological treatment of PoPH[23]. In this multicentric study, 85 patients were enrolled and randomly assigned to receive either macitentan or placebo. Following a 12-week treatment course, patients receiving macitentan had a 35% decline in PVR, a significant reduction in mPAP (-6.4 mmHg vs +0.4 mmHg in the placebo arm), and an improved cardiac index. The rate of adverse events was slightly higher in the active arm (84% vs 79% in the placebo arm), but event severity was low, with less than 10% of patients requiring treatment interruption. The second consistent batch of data comes from the French Pulmonary Hypertension Registry[22]. While the prospective design was not controlled and treatment selection was deliberate (non-random), the study's strength resides in the long-term follow-up (up to 10 years), a large number of patients included (n = 637), and the focus on mortality as a major outcome. Most patients were initially treated with oral monotherapy with phosphodiesterase 5-inhibitors (sildenafil or tadalafil) or endothelin receptor antagonists (bosentan, ambrisentan, or macitentan), while some were on double or, exceptionally (n = 5) triple therapy. Regardless of therapeutic regimens, all patients had significant improvements after 4.5 mo in PVR, mPAP, cardiac output, and functional capacity. In most variables, gains were augmented in dual therapy. Overall survival rates at 1, 3, and 5 years were significantly higher for patients who also benefited LT (92% vs 84%, 83% vs 69%, and 81% vs 51%, respectively).
The un/mistreated complication
The evidence appears to be relatively straightforward regarding the efficacy of PAH-specific therapy in PoPH. However, translation to clinical practice seems far from ideal despite proven benefits. Krowka et al. have analyzed the PoPH-related data from the REVEAL registry and found that even though patients with PoPH had a worse outcome than PAH etiologies, they were less likely to be on pharmacological therapy. This fact is even more striking given that patients with PoPH were in a worse cardiac functional class and, consequently, had a higher symptom burden[21]. Moreover, a recent prognostic analysis, REVEAL 2.0, designed to redefine the prior risk score, has attributed an additional risk point for patients with PoPH. This marks PoPH as the single most important predictor for high-risk PAH[24]. Another critical aspect is defining the optimal timing for PAH-therapy commencement. Analyzing recently published data from the Spanish REHAP registry, patients with PoPH appear to have a better hemodynamic profile than other patients with PAH, yet have a worse outcome, even when strictly considering PAH-related deaths. Therefore, an educated guess would suggest that the indication for pharmacological treatment commencement should start at a lower PVR or mPAP threshold. Furthermore, data from REHAP further reinforce the significant discrepancy in treatment between POPH and other cases of PAH, as patients with PoPH were both less likely to be treated and more likely to be on monotherapy rather than combination therapy[25]. A potential explanation might reside in hepatologists’ lack of awareness and familiarity with PAH and its corresponding therapeutic options outside large LT centers and a lack of pulmonologist referrals for patients with PHT to provide an adequate, personalized, multidisciplinary approach to care.
HEPATIC HYDROTHORAX - THE HIDDEN LATE DECOMPENSATION
Definition and diagnostic criteria
HH is an uncommon, understudied complication of cirrhosis and portal hypertension, occurring in 5%-15% of patients[26]. It is defined as a transudative pleural effusion, usually larger than 500 mL occurring in a cirrhotic patient in the absence of underlying cardiac, pulmonary, or pleural disease.
Context and a brief history
Morrow first coined the term hepatic hydrothorax in 1958 in an illustrative case report aimed to acknowledge the relationship between the occurrence of a pleural transudate and advanced liver disease[27]. In the subsequent decades, the relatively few papers published in the field have focused on epidemiological data. Yet, some notable attempts to characterize the pathophysiology using radioiodinated albumin and India ink in select cases were published[28]. The exact mechanism through which HH develops is not yet fully known. However, it seems to stem from an imbalance between hydrostatic and osmotic pressures. During inspiration, the negative intrathoracic pressure allows the fluid to pass from the abdomen to the pleural space through minor parietal defects of less than 1 cm, situated in the tendinous part of the diaphragm, more common on the right side[9]. Consequently, 85% of patients develop HH on the right side, while only 2% of cases are bilateral[29].
As opposed to patients with ascites, who can tolerate large volumes of fluid with only mild symptoms, HH can become symptomatic at a buildup of only 500 mL. Clinical manifestations may include cough, shortness of breath, pleuritic chest pain, hypoxemia, and respiratory failure. Most patients have progressive dyspnea and a reduced tolerance for physical effort.
The diagnosis is based on the presence of liver cirrhosis and lack of any primary pleural, cardiac, or pulmonary disease that could account for the buildup of fluid. HH can be complicated by spontaneous bacterial empyema (SBEM), the infection of a preexisting HH that can appear in approximately 15% of patients and should be evaluated through fluid analysis[29]. The work-up includes serum and fluid protein, albumin, lactate dehydrogenase levels, cell count, Gram stain, and culture. Other tests may be required depending on the clinical setting. Pharmacological treatment of HH closely resembles the treatment of ascites. Sodium restriction and diuretics, with a combination of furosemide 40 mg and spironolactone 100 mg daily, are the cornerstones for therapy. If there is a lack of response, the doses may be doubled stepwise every three to five days up to a maximum of 160 mg furosemide and 400 mg spironolactone daily. In uncomplicated HH, the polymorphonuclear cell (PMN) count is low (< 250 cells/mm3). In contrast, in the setting of SBEM, the PMN count is elevated, with a diagnostic cutoff of > 250 cells/mm3 if cultures are positive or > 500 cells/mm3 if cultures are negative. The etiologic agents in most cases are Escherichia coli, Streptococcus, Enterococcus, Klebsiella, or Pseudomonas, which typically respond well to a 7 d to 10 d course of third-generation cephalosporins.
Treatment strategies - evidence beyond diuretics
Akin to patients with other decompensating events, there is a stepwise approach to treating HH beyond diuretics and salt restriction. However, in contrast to other, more established complications of cirrhosis, the quality of evidence is typically low, consisting of single-center, low-volume, and non-controlled data.
Therapeutic thoracocentesis is the next logical step in patients with severe symptoms that diuretics cannot control. However, it appears that the need for repeat thoracocentesis is associated with higher rates of acute-on-chronic liver failure and inpatient mortality compared to patients without HH, possibly related to a higher rate of nosocomial infections[30]. Patients who require thoracentesis every two to three weeks should be considered for alternative treatments. They are at increased risk of having adverse events such as pneumothorax, pleural empyema, purulent soft tissue infection of the chest wall, and air embolism. In addition, large-volume thoracentesis may increase microvascular permeability and cause re-expansion pulmonary edema. The complication rates appear to be significantly higher compared to patients with ascites and repeated paracenteses[31].
Chest tubes should not be placed for HH treatment, as this can result in massive protein and electrolyte depletion, infection, renal failure, and bleeding[32-34].
Local therapeutic options for patients who are refractory to the treatments mentioned above include pleurodesis and thoracoscopic surgery for diaphragmatic repair. However, it should be noted that although control of symptoms and resolution of effusion can be achieved in up to 75% of patients, recurrence rates are high, and a significantly increased prevalence of procedure-related morbidity and mortality hinders this procedure’s routine application[35-37].
Another therapeutic option is TIPS placement. Several studies have reported a beneficial effect in patients with HH, with an overall response rate of up to 80%, an average 30 d mortality rate of 18%, and a 1-year survival rate of 52%[33,38-43]. However, TIPS should be avoided in patients older than 60 years, as this seems to correlate inversely with survival rates[38]. Of note, most of the data is relatively old and precedes the covered stent era, which drastically improved shunt patency rates. However, considering the data, it appears sensible that HH is a viable indication for TIPS, especially in patients with concurrent decompensating events, such as ascites or a single episode of variceal bleeding.
Liver transplantation is an excellent definitive therapeutic option for cirrhotic patients with HH and end-stage liver disease. The need for thoracenteses decreases post-transplantation, and the presence of preoperative HH does not seem to have a significant negative influence on postoperative outcomes, with similar long-term survival as in other indications for LT[44,45].
Is hepatic hydrothorax an indicator for late decompensation?
Hepatic hydrothorax is more frequent in patients with higher Child-Pugh scores, potentially serving as a marker of decreased liver function (8%, 26%, and 65% in Child-Pugh class A, B, and C, respectively)[46]. HH seems to be more frequent in patients with cirrhosis and ascites with a recurrent need for paracentesis, higher bilirubin, diabetes, and lack of NSBB therapy[47]. While the MELD score doesn’t seem to predict the risk of HH more accurately than the Child-Pugh score, one might consider HH a prognostic indicator itself, akin to other decompensation events such as ascites or variceal bleeding. As previously shown in a study by our group, patients with HH have a significantly higher long-term mortality rate when compared to patients without HH. Their underlying liver disease is more advanced, based on markers of liver function, decompensation, and prognostic scores. Yet, even after matching patients for age and liver function, HH appears to be an independent mortality predictor[48]. HH tends to develop later in the disease course and increases mortality up to four times, especially in patients with MELD scores higher than 16[47,48]. These data are further reinforced by a study that included 3487 patients. Pleural effusion was associated with 30 d, 90 d, 1-year, and 3-year mortalities of 20.1%, 40.2%, 59.1%, and 75.9%, respectively[49]. Considering this, HH might be viewed as an independent risk factor for disease progression. Therefore, it should be taken into account when selecting patients for tertiary care referral or more expensive and less available procedures, such as TIPS or LT, especially in limited-resource settings. Regarding LT, evidence suggests that it can significantly improve survival rates and should be considered whenever possible. Sersté demonstrated a similar time frame of HH resolution and no significant difference in duration of postoperative mechanical ventilation, providing sufficient ground for a similar post LT survival, compared to other indications[45]. Yet, given that patients with HH are typically in a worse shape compared to non-HH patients with otherwise similar characteristics, the question should be raised whether recurrent HH despite optimal therapy could be considered on an individual basis for a non-standard MELD exception[50], similar to HPS and PoPH.