Published online Aug 15, 2019. doi: 10.4251/wjgo.v11.i8.642
Peer-review started: May 21, 2019
First decision: July 16, 2019
Revised: July 18, 2019
Accepted: August 3, 2019
Article in press: August 3, 2019
Published online: August 15, 2019
Processing time: 94 Days and 22.4 Hours
The Borrmann classification system is used to describe the macroscopic appearance of advanced gastric cancer, and Borrmann type IV disease is independently associated with a poor prognosis.
To evaluate the prognostic significance of lymphatic and/or blood vessel invasion (LBVI) combined with the Borrmann type in advanced proximal gastric cancer (APGC).
The clinicopathological and survival data of 440 patients with APGC who underwent curative surgery between 2005 and 2012 were retrospectively analyzed.
In these 440 patients, LBVI+ status was associated with Borrmann type IV, low histological grade, large tumor size, and advanced pT and pN status. The 5-year survival rate of LBVI+ patients was significantly lower than that of LBVI– patients, although LBVI was not an independent prognostic factor in the multivariate analysis. No significant difference in the prognosis of patients with Borrmann type III/LBVI+ disease and patients with Borrmann type IV disease was observed. Therefore, we proposed a revised Borrmann type IV (r-Bor IV) as Borrmann type III plus LBVI+, and found that r-Bor IV was associated with poor prognosis in patients with APGC, which outweighed the prognostic significance of pT status.
LBVI is related to the prognosis of APGC, but is not an independent prognostic factor. LBVI status can be used to differentiate Borrmann types III and IV, and the same approach can be used to treat r-Bor IV and Borrmann type IV.
Core tip: In this study, we found that lymphatic and/or blood vessel invasion (LBVI) + status was associated with Borrmann type IV, low histological grade, large tumor size, and advanced pT and pN status. The 5-year survival rate of LBVI+ patients was significantly lower than that of LBVI– patients. No significant difference was observed in the prognosis of Borrmann type III/LBVI+ disease and Borrmann type IV disease. Therefore, we proposed a revised Borrmann type IV (r-Bor IV) as Borrmann type III plus LBVI+, and found that r-Bor IV was associated with poor prognosis in patients with advanced proximal gastric cancer, which outweighed the prognostic significance of pT status.
- Citation: Gao S, Cao GH, Ding P, Zhao YY, Deng P, Hou B, Li K, Liu XF. Retrospective evaluation of lymphatic and blood vessel invasion and Borrmann types in advanced proximal gastric cancer. World J Gastrointest Oncol 2019; 11(8): 642-651
- URL: https://www.wjgnet.com/1948-5204/full/v11/i8/642.htm
- DOI: https://dx.doi.org/10.4251/wjgo.v11.i8.642
Gastric cancer is a significant public health problem and is associated with substantial rates of morbidity and mortality[1,2]. Although radical D2 gastrectomy is the main treatment for gastric cancer, the high incidence of recurrence and metastasis postoperatively leads to a high mortality rate, even after curative resection[3-5]. Proximal-third gastric cancers (PGCs) have a poorer prognosis than distal gastric cancers, as they are characterized by a more aggressive biological behavior and are typically diagnosed at an advanced stage[6-8]. Furthermore, although the overall incidence of gastric cancer has decreased, the incidence of PGC has increased in recent decades[9]. Therefore, it is important to identify prognostic factors for PGC and to adopt more aggressive adjuvant treatment strategies, particularly for patients with advanced PGC (APGC).
Lymphatic and/or blood vessel invasion (LBVI) is defined as the presence of tumor cells in the lymphovascular system or in the small veins of tumor tissue, under microscope. In addition, LBVI plays an important role in tumor cell dissemination and metastasis during progression, which contributes to the poor prognosis of patients with gastric cancer[10-12]. The Borrmann classification system is used to describe the macroscopic appearance of advanced gastric cancer, and Borrmann type IV gastric cancer is independently associated with poor prognosis. Moreover, Borrmann type IV is more likely to be diagnosed in patients with PGC than in patients with distal gastric cancers[13-15]. However, few studies have examined the prognostic significance of LBVI status combined with Borrmann type in patients with APGC. Therefore, in this retrospective study, the clinicopathological characteristics of APGC patients with LBVI (LBVI+) or without LBVI (LBVI–) were compared, and LBVI status was examined to determine whether it could be used to further enhance the Borrmann classification system.
All patients underwent curative proximal or total gastrectomy with standard lymph node dissection at the First Affiliated Hospital of the China Medical University between January 2005 and October 2012. A total of 494 patients with APGC and complete clinicopathological and surgical data, including age, sex, tumor size, Borrmann classification, LBVI status, pT status, pN status, histological grade, resection type, and extent of resection, were included in this study. We defined APGC as: (1) A tumor located at the proximal third of the stomach, including the gastric cardia, esophageal-gastric junction, and fundus; (2) A tumor histologically diagnosed as gastric adenocarcinoma; and (3) A tumor with a depth of invasion beyond the mucous layer. We excluded the following patients: (1) Those with distant metastasis, peritoneal dissemination, or pathological T4b stage; (2) Those who underwent palliative surgery; (3) Those who died due to postoperative non-tumor-related events; (4) Those who had undergone preoperative chemotherapy or radiotherapy; (5) Those with carcinoma in the remnant stomach; and (6) Those who were lost to follow-up. Therefore, 440 patients were included in the final analyses.
All patients underwent standardized follow-up every 3 mo for the first 2 years after surgery, every 6 mo during the third year, and annually thereafter. The follow-up period was from surgery until the patient’s death or October 2017. The median follow-up period was 65 mo (range: 2–122 mo).
All resected specimens were fixed using 10% formalin, dehydrated using ethanol, embedded in paraffin, stained with hematoxylin and eosin (HE), and evaluated using an optical microscope. Each patient’s LBVI status and Borrmann type were independently evaluated by two pathologists, and disagreements were resolved by a third pathologist. The Borrmann classification and LBVI status were determined based on the Japanese Classification of Gastric Carcinoma[16]. Borrmann type I (limited mass) was defined as a tumor that protruded toward the gastric lumen and had a distinct border separating it from the surrounding mucosa. Borrmann type II (limited ulcerative) was defined as an ulcerated tumor with its dam-shaped margin protruding and surrounded by a thickened gastric wall with clear margins. Borrmann type III (infiltrative ulcerative) was defined as a Borrmann type II tumor that was surrounded by a thickened gastric wall without clear margins. Borrmann type IV (diffuse infiltrative) was defined as a tumor with diffuse infiltration through each layer of the gastric wall, without marked ulceration or raised margins, and without clear margins separating the tumor from normal tissue. The presence of LBVI was identified based on the presence of tumor cells in the lymphovascular system or in the small veins of tumor tissue. Pathological tumor staging was based on the TNM system of the American Joint Committee on Cancer and the International Union Against Cancer (eighth edition).
Differences between the LBVI– and LBVI+ groups were evaluated using the Chi-square test for categorical variables and the independent t test for continuous variables. The Kaplan-Meier method and log-rank test were used to identify univariate differences in survival. A multivariate analysis was performed using the Cox proportional hazards model. All statistical analyses were performed using SPSS software (version 22.0; IBM Corp., Armonk, NY, USA), and differences were considered statistically significant at two-sided P values of < 0.05.
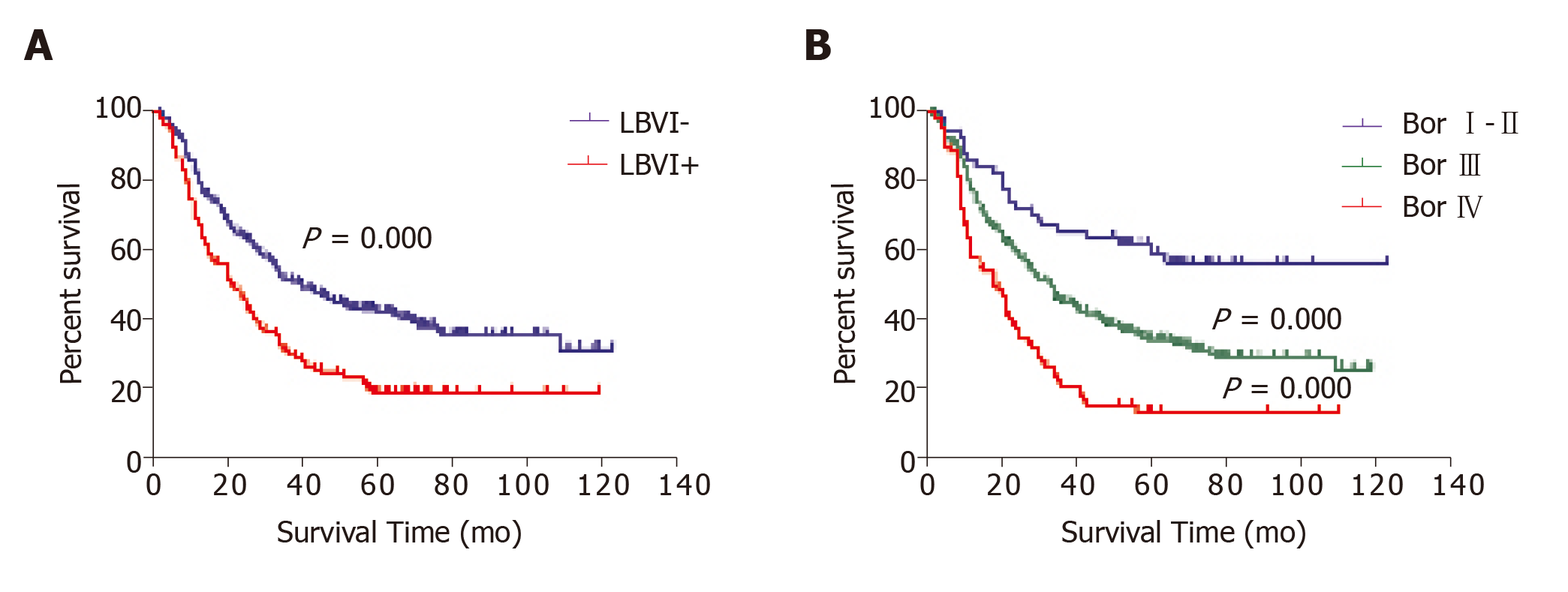
Of the 440 eligible patients with APGC, 97 patients (22.1%) underwent proximal gastrectomy and 343 patients (77.9%) underwent total gastrectomy. All patients achieved radical resection (R0), and D2 lymphadenectomy was performed in 436 patients (98.2%). Among the 440 patients, 151 (34.3%) had LBVI and 72 (16.4%) had Borrmann type IV disease. The incidence of LBVI+ was 15.5% (9/58) in Borrmann type I–II disease, 35.8% (111/310) in Borrmann type III disease, and 43.1% (31/72) in Borrmann type IV disease.
Table 1 shows the patients’ clinicopathological characteristics according to LBVI status. The presence of LVBI was significantly associated with Borrmann type IV (P = 0.003), low histological grade (P = 0.001), large tumor size (P = 0.013), advanced pT status (P = 0.002), and advanced pN status (P < 0.001).
| Variable | n | Lymphatic vessel invasion | P value | |
| Absent (n = 289, 65.7%) | Present (n = 151, 34.3%) | |||
| Age (yr) | 440 | 59.86 ± 10.4 | 58.55 ± 11.128 | 0.426 |
| Gender | 0.765 | |||
| Male | 333 (75.7) | 220 (66.1) | 113 (33.9) | |
| Female | 107 (24.3) | 69 (64.5) | 38 (35.5) | |
| Tumor size (cm) | 0.013 | |||
| ≤ 5 | 169 (38.5) | 123 (72.8) | 46 (27.2) | |
| > 5 | 231 (52.5) | 166 (60.4) | 105 (39.6) | |
| Macroscopic type | 0.003 | |||
| Borrmann I-II | 58 (13.2) | 49 (84.5) | 9 (15.5) | |
| Borrmann III | 310 (70.4) | 199 (64.2) | 111 (35.8) | |
| Borrmann IV | 72 (16.4) | 41 (56.9) | 31 (43.1) | |
| Adjacent organs invaded1 | 0.815 | |||
| With | 105 (23.9) | 70 (66.7) | 36 (33.3) | |
| Without | 335 (76.1) | 219 (65.4) | 116 (34.6) | |
| Histologic grade | 0.001 | |||
| Well | 203 (46.1) | 149 (73.4) | 54 (26.6) | |
| Poor | 237 (53.9) | 140 (59.1) | 97 (40.9) | |
| AJCC pT category | 0.002 | |||
| pT2 | 53 (12.0) | 37 (69.8) | 16 (30.2) | |
| pT3 | 136 (31.0) | 104 (76.5) | 32 (23.5) | |
| pT4a | 251 (57.0) | 148 (59.0) | 103 (41.0) | |
| AJCC pN category | 0.000 | |||
| pN0 | 102 (23.2) | 96 (94.1) | 6 (5.9) | |
| pN1 | 71 (16.2) | 51 (71.8) | 20 (28.2) | |
| pN2 | 98 (22.2) | 65 (66.3) | 33 (33.7) | |
| pN3a | 89 (20.2) | 47 (52.8) | 42 (47.2) | |
| pN3b | 80 (18.2) | 30 (37.5) | 50 (62.5) | |
| Resection type | 0.426 | |||
| Proximal | 97 (22.1) | 67 (69.1) | 30 (30.1) | |
| Total | 343 (77.9) | 222 (64.7) | 121 (35.3) | |
| Combined other organ resection | 0.815 | |||
| No | 396 (90.0) | 259 (65.4) | 137 (34.6) | |
| Yes | 44 (10.0) | 30 (68.2) | 14 (31.8) | |
| Survival at 5 yr | 34.2% | 42.5% | 25.1% | 0.000 |
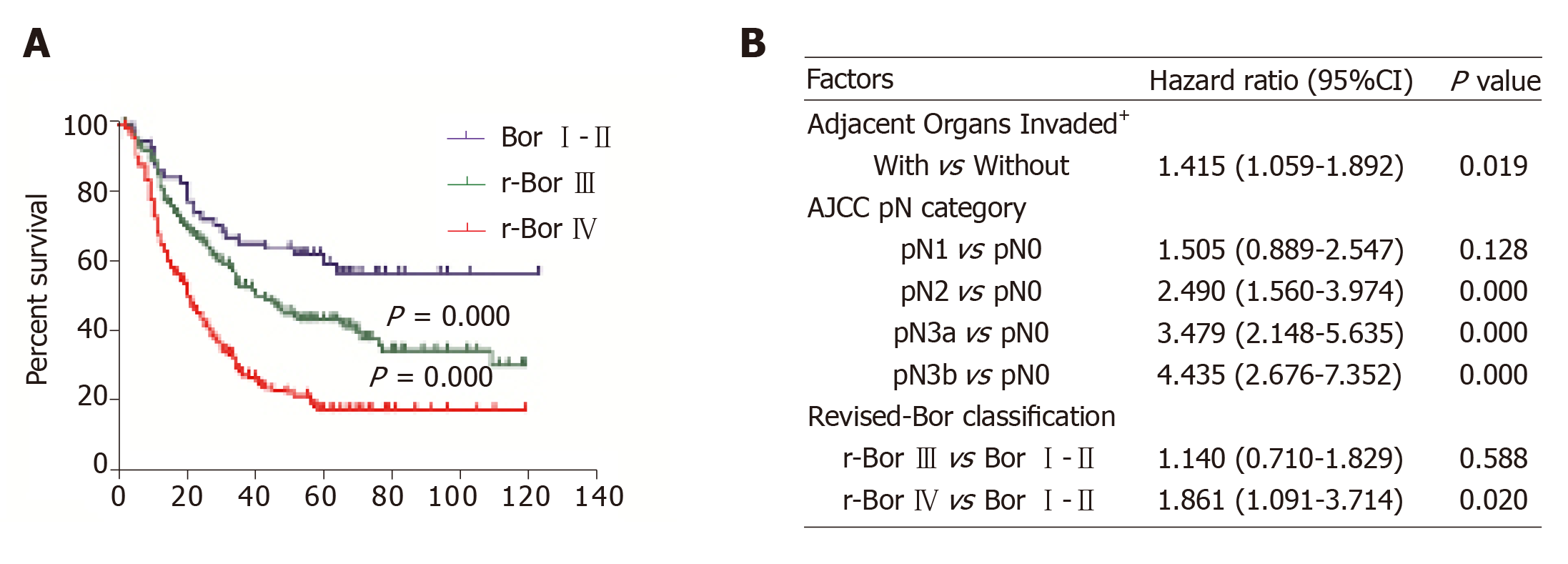
In the univariate survival analyses, the 5-year overall survival rate of LBVI+ patients was significantly lower than that of LBVI– patients (19.1% vs 25.1%, P < 0.001; Figure 1A). In addition, significant differences in the 5-year survival rates were observed when patients with Borrmann type I–II (59.4%), type III (34.3%), and type IV disease (13.4%) were compared (P < 0.001; Figure 1B). Multivariate Cox regression analysis revealed that poor survival was independently associated with lymph node metastasis (P < 0.001), Borrmann type IV (P = 0.016), and invasion of adjacent organs (P = 0.015), but was not independently associated with LBVI+ status (P = 0.197) or advanced pT status (P = 0.996; Table 2).
| Univariate analysis | Multivariate analysis | |||
| Hazard ratio (95%CI) | P value | Hazard ratio (95%CI) | P value | |
| Age (yr) | ||||
| > 60 vs ≤ 60 | 1.295 (1.031-1.628) | 0.026 | 1.206 (0.928-1.567) | 0.161 |
| Tumor size (cm) | ||||
| > 5 vs ≤ 5 | 2.118 (1.648-2.722) | 0.000 | 1.246 (0.922-1.684) | 0.153 |
| Adjacent organs invaded1 | ||||
| With vs Without | 1.630 (1.264-2.102) | 0.000 | 1.436 (1.074-1.919) | 0.015 |
| Histologic differentiation | ||||
| Poor vs well | 1.370 (1.088-1.725) | 0.007 | 1.278 (0.980-1.666) | 0.070 |
| AJCC pT category | 0.996 | |||
| pT3 vs pT2 | 1.707 (1.093-2.666) | 0.019 | 1.019 (0.604-1.718) | 0.943 |
| pT4a vs pT2 | 2.405 (1.582-3.656) | 0.000 | 1.024 (0.614-1.707) | 0.927 |
| AJCC pN category | 0.000 | |||
| pN1 vs pN0 | 1.758 (1.110-2.783) | 0.016 | 1.487 (0.878-2.517) | 0.140 |
| pN2 vs pN0 | 2.996 (1.995-4.500) | 0.000 | 2.501 (1.567-3.990) | 0.000 |
| pN3a vs pN0 | 4.531 (3.034-6.765) | 0.000 | 3.406 (2.106-5.509) | 0.000 |
| pN3b vs pN0 | 6.377 (4.240-9.592) | 0.000 | 4.245 (2.551-7.064) | 0.000 |
| Resection type | ||||
| Total vs Proximal | 1.772 (1.279-2.456) | 0.001 | 1.103 (0.773-1.578) | 0.590 |
| Combined other organ resection | ||||
| Yes vs No | 1.579 (1.109-2.248) | 0.011 | 1.189 (0.812-1.740) | 0.374 |
| LBVI | ||||
| Positive vs Negative | 1.750 (1.387-2.207) | 0.000 | 1.200 (0.910-1.583) | 0.197 |
| Borrmann classification | 0.019 | |||
| Bor III vs Bor I-II | 2.016 (1.321-3.077) | 0.001 | 1.273 (0.807-2.006) | 0.299 |
| Bor IV vs Bor I-II | 3.686 (2.295-5.922) | 0.000 | 1.935 (1.131-3.311) | 0.016 |
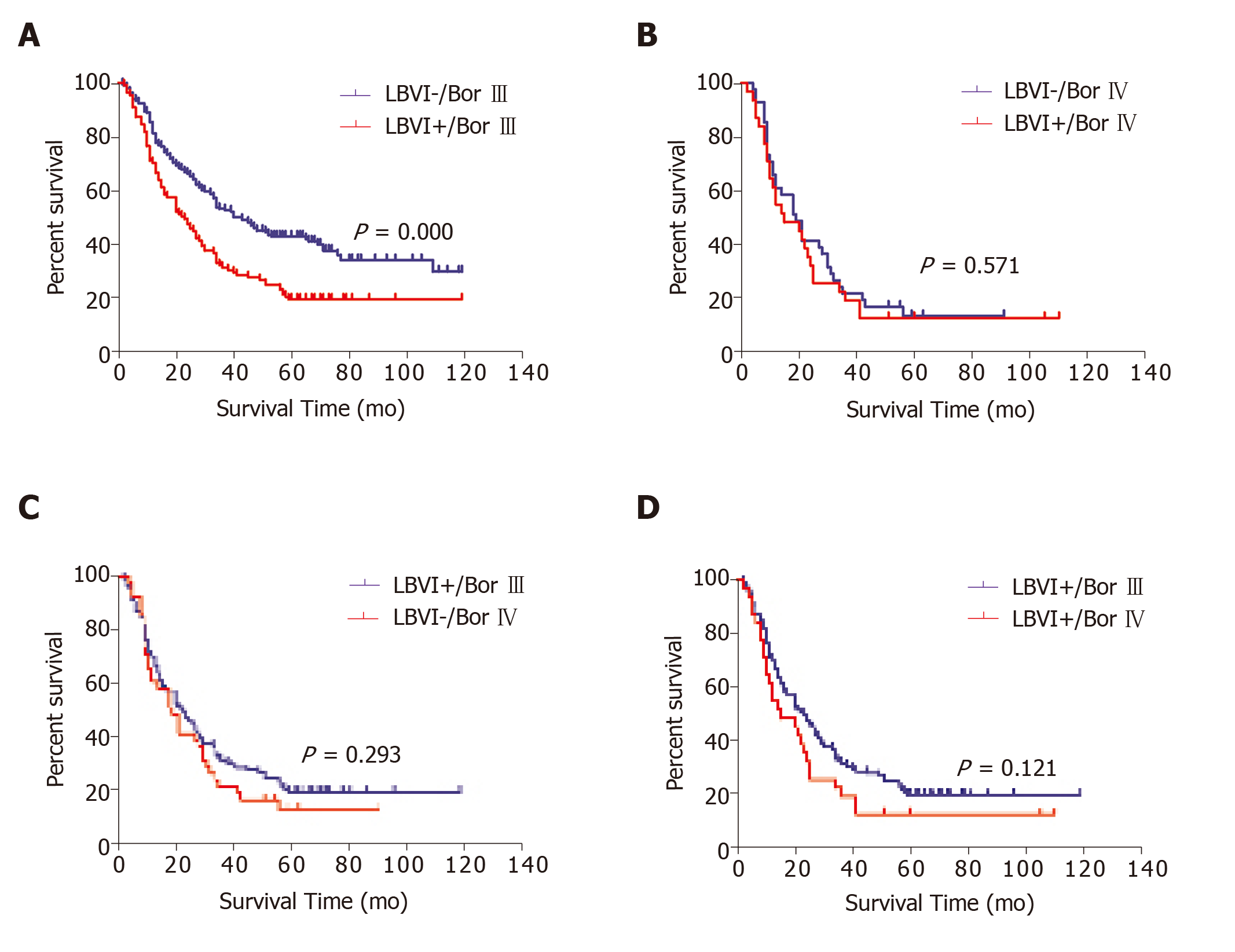
Borrmann type III or IV disease was present in 382 of the 440 patients with APGC (86.8%), and the prognostic significance of LBVI status in this subgroup was evaluated. Among the patients with Borrmann type III disease, LBVI+ status was associated with significantly poorer survival (5-year survival: 19.8% vs 43.1%, P < 0.001; Figure 2A). However, no significant difference in survival was observed according to LBVI status in patients with Borrmann type IV disease (5-year survival: 13.0% vs 13.7%, P = 0.571; Figure 2B). Patients with LBVI+ and Borrmann type III disease had a similar prognosis to patients with LBVI– and Borrmann type IV disease (5-year survival: 19.8% vs 13.7%, P = 0.293; Figure 2C) and to patients with LBVI+ and Borrmann type IV disease (5-year survival: 19.8% vs 12.9%, P = 0.121; Figure 2D).
Based on these results, we proposed revised versions of Borrmann type III (r-Bor III) as Borrmann type III and LBVI– and type IV (r-Bor IV) as Borrmann type III plus LBVI+ or Borrmann type IV. Figure 3 shows that patients with r-Bor IV APGC had a significantly poorer prognosis based on the univariate analysis (P < 0.001) and multivariate analysis (hazard ratio: 1.861, 95% CI: 1.091-3.714, P = 0.02).
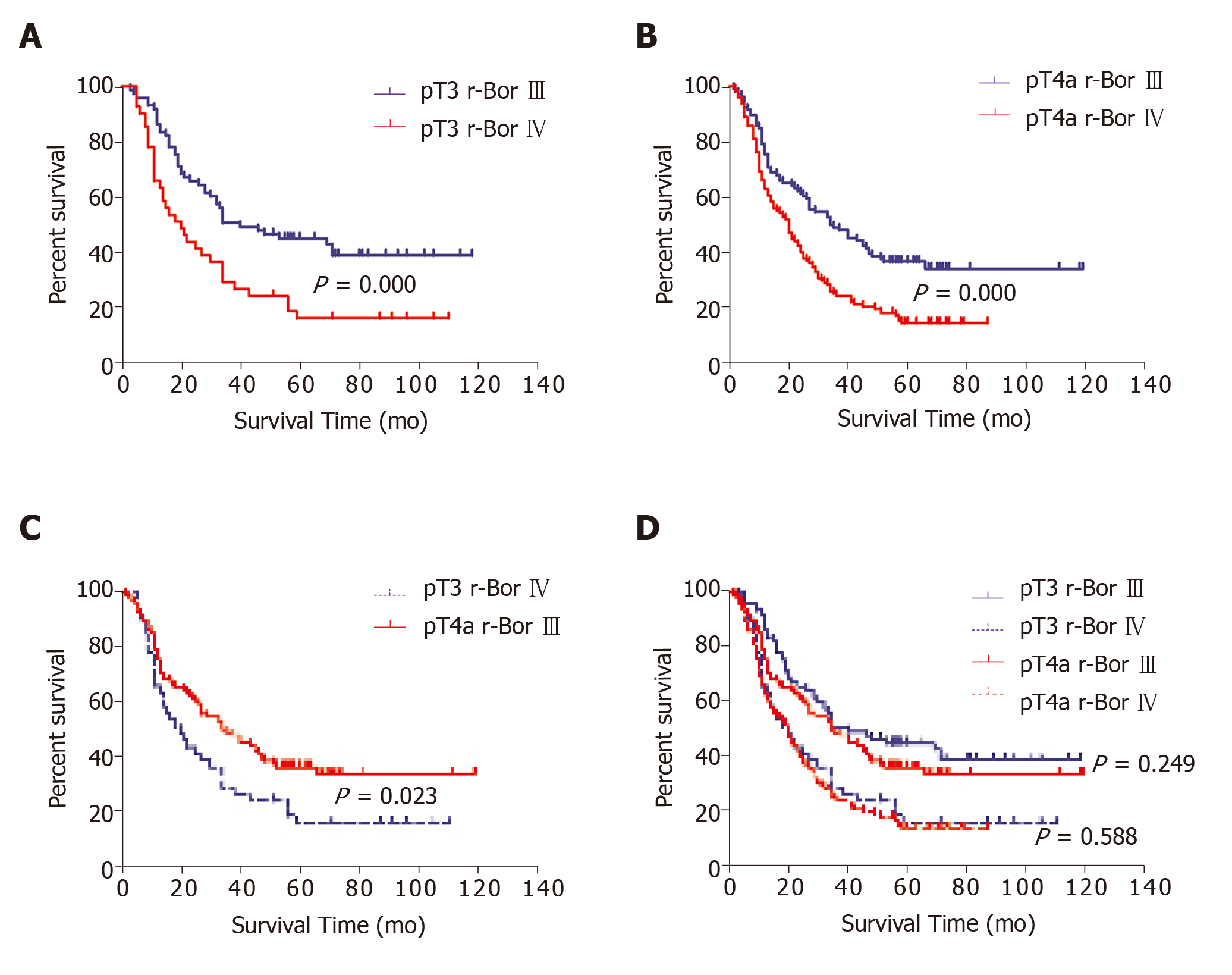
We further examined our r-Bor III and r-Bor IV subgroups to identify survival differences after radical surgery according to pT3 and pT4a status. Patients with pT3 and r-Bor IV disease had a significantly poorer prognosis than patients with pT3 and r-Bor III disease (P = 0.002; Figure 4A) and those with pT4a disease (P < 0.001; Figure 4B). Moreover, patients with pT3 and r-Bor IV disease had a significantly poorer prognosis than patients with pT4a and r-Bor III disease (P = 0.023; Figure 4C). No significant differences in prognosis were observed when we compared pT3 and pT4a status in patients with r-Bor III disease (P = 0.249; Figure 4D), or when we compared pT3 and pT4a status in patients with r-Bor IV disease (P = 0.588; Figure 4D).
Compared to patients with distal gastric cancer, patients with APGC are more likely to have adverse clinicopathological characteristics that affect their prognosis, such as lymph node metastasis, serosa invasion, LBVI+ status, and larger tumor size[8,17]. The multivariate analysis in the present study confirmed that lymph node metastasis, Borrmann type IV, and invasion of adjacent organs were independent risk factors for patients with APGC. The present study also revealed that LBVI+ status was related to poor prognosis in this setting, although it was not an independent prognostic factor. Previous studies revealed that LBVI+ status is associated with poor prognosis after radical surgery for gastric cancer, and is an independent predictor of poor prognosis in patients with negative lymph nodes[18-20].
The Borrmann classification is generally assigned based on macroscopic examination of the tumor, although some Borrmann type III tumors have a similar appearance and prognosis to Borrmann type IV tumors, which has led to the term “limited Borrmann type IV”[21]. Previous studies have indicated that Borrmann type IV disease has a very poor prognosis[22,23], and we also found that Borrmann type IV disease was an independent prognostic factor in APGC patients (5-year survival: 13.3%). We incorporated LBVI in an attempt to better differentiate Borrmann type III and IV disease, which revealed that patients with r-Bor IV disease had a similar prognosis to those with Borrmann type IV disease, but had significantly poorer prognosis than patients with r-Bor III disease. Thus, LBVI status can help differentiate “limited Borrmann type IV” from standard Borrmann type III. Interestingly, some studies have reported a lower rate of liver metastasis after surgical resection in Borrmann type IV patients than in Borrmann type III patients, which raises the question of whether LBVI might also be useful in that setting. Further studies are needed to examine this hypothesis.
Although pT status is not an independent prognostic factor for APGC, it is an important factor that influences the prognosis of advanced gastric cancer. In this context, pT1 and pT2 tumors are generally smaller and more accurately classified in a comprehensive pathological examination, while pT3 and pT4 tumors are generally larger, and it is more difficult to accurately determine the depth of invasion using conventional methods. Moreover, there is some overlap between the pT system and the Borrmann classification system in patients with advanced gastric cancer. Therefore, we examined the influence of our revised Borrmann classification in patients with pT3 and pT4 APGC, which revealed that r-Bor IV was an independent predictor of poor prognosis in all patients with APGC. In patients with infiltrative gastric cancer (pT3 and pT4a), r-Bor IV was still associated with significantly poorer survival than r-Bor III, and the magnitude of the survival difference was large enough to outweigh the prognostic value of pT status. Thus, a more aggressive postoperative adjuvant treatment strategy should be adopted for patients with pT3 or pT4a r-Bor IV.
This study has several limitations. First, we only included patients with advanced cancers who underwent radical surgery, and excluded patients with pT4b disease to more accurately evaluate the influence of LBVI status. However, we reviewed the patients’ surgical records, and identified 105 patients who were diagnosed with sT4b disease (intraoperative identification of adjacent organ invasion), and only 10 patients who were postoperatively diagnosed with pT4b disease. Thus, we included sT4b as a covariate in the Cox regression model. Another limitation is that LBVI was identified based on routine histological H&E staining, and the tumor sections were examined at the largest specimen width which may have limited the sensitivity of LBVI detection.
In conclusion, APGC patients with LBVI had significantly shorter survival than patients without LBVI, although LBVI+ status was not an independent prognostic factor in the multivariate analysis. Our data indicate that LBVI status may help clarify the difference between Borrmann type III and IV tumors. Based on our revised system, we suggest that patients with r-Bor IV APGC should be treated the same as patients with standard Borrmann type IV APGC. Furthermore, in patients with pT3 and pT4a APGC, r-Bor IV could be used to guide prognostication and follow-up treatment.
Lymphatic and/or blood vessel invasion (LBVI) plays an important role in tumor cell dissemination and metastasis during tumor progression, which contributes to the poor prognosis of patients with gastric cancer.
The Borrmann classification is generally assigned based on macroscopic examination of the tumor, although some Borrmann type III tumors have a similar appearance and prognosis to Borrmann type IV tumors, which has led to the term “limited Borrmann type IV”. Previous studies have indicated that Borrmann type IV disease has a very poor prognosis.
In this study, the authors aim to evaluate the prognostic significance of LBVI combined with the Borrmann type in advanced proximal gastric cancer (APGC).
The clinicopathological and survival data of 440 patients with APGC who underwent curative surgery between 2005 and 2012 were retrospectively analyzed.
In these 440 patients, LBVI+ status was associated with Borrmann type IV, low histological grade, large tumor size, and advanced pT and pN status. The 5-year survival rate of LBVI+ patients was significantly lower than that of LBVI– patients, although LBVI was not an independent prognostic factor in the multivariate analysis. No significant difference in the prognosis of patients with Borrmann type III/LBVI+ disease and patients with Borrmann type IV disease was observed. Therefore, we proposed a revised Borrmann type IV (r-Bor IV) as Borrmann type III plus LBVI+, and found that r-Bor IV was associated with poor prognosis in patients with APGC, which outweighed the prognostic significance of pT status.
LBVI status may help clarify the difference between Borrmann type III and IV tumors. Based on our revised classification, we suggest that patients with (r-Bor IV APGC should be treated the same as patients with standard Borrmann type IV APGC.
In patients with pT3 and pT4a APGC, r-Bor IV could be used to guide prognostication and follow-up treatment.
Manuscript source: Unsolicited manuscript
Specialty type: Oncology
Country of origin: China
Peer-review report classification
Grade A (Excellent): 0
Grade B (Very good): B, B
Grade C (Good): C
Grade D (Fair): 0
Grade E (Poor): 0
P-Reviewer: Reich R, Satya R, Manuel C S-Editor: Wang JL L-Editor: A E-Editor: Qi LL
| 1. | Jemal A, Bray F, Center MM, Ferlay J, Ward E, Forman D. Global cancer statistics. CA Cancer J Clin. 2011;61:69-90. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 23762] [Cited by in RCA: 25540] [Article Influence: 1824.3] [Reference Citation Analysis (7)] |
| 2. | Lin Y, Ueda J, Kikuchi S, Totsuka Y, Wei WQ, Qiao YL, Inoue M. Comparative epidemiology of gastric cancer between Japan and China. World J Gastroenterol. 2011;17:4421-4428. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in CrossRef: 119] [Cited by in RCA: 133] [Article Influence: 9.5] [Reference Citation Analysis (0)] |
| 3. | Deng J, Liang H, Wang D, Sun D, Pan Y, Liu Y. Investigation of the recurrence patterns of gastric cancer following a curative resection. Surg Today. 2011;41:210-215. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 75] [Cited by in RCA: 75] [Article Influence: 5.4] [Reference Citation Analysis (0)] |
| 4. | Shen L, Shan YS, Hu HM, Price TJ, Sirohi B, Yeh KH, Yang YH, Sano T, Yang HK, Zhang X, Park SR, Fujii M, Kang YK, Chen LT. Management of gastric cancer in Asia: resource-stratified guidelines. Lancet Oncol. 2013;14:e535-e547. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 326] [Cited by in RCA: 382] [Article Influence: 31.8] [Reference Citation Analysis (0)] |
| 5. | Kang WM, Meng QB, Yu JC, Ma ZQ, Li ZT. Factors associated with early recurrence after curative surgery for gastric cancer. World J Gastroenterol. 2015;21:5934-5940. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in CrossRef: 60] [Cited by in RCA: 69] [Article Influence: 6.9] [Reference Citation Analysis (2)] |
| 6. | Gulmann C, Hegarty H, Grace A, Leader M, Patchett S, Kay E. Differences in proximal (cardia) versus distal (antral) gastric carcinogenesis via the retinoblastoma pathway. World J Gastroenterol. 2004;10:17-21. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in CrossRef: 10] [Cited by in RCA: 11] [Article Influence: 0.5] [Reference Citation Analysis (0)] |
| 7. | Ishikawa S, Shimada S, Miyanari N, Hirota M, Takamori H, Baba H. Pattern of lymph node involvement in proximal gastric cancer. World J Surg. 2009;33:1687-1692. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 21] [Cited by in RCA: 23] [Article Influence: 1.4] [Reference Citation Analysis (0)] |
| 8. | Saito H, Fukumoto Y, Osaki T, Fukuda K, Tatebe S, Tsujitani S, Ikeguchi M. Distinct recurrence pattern and outcome of adenocarcinoma of the gastric cardia in comparison with carcinoma of other regions of the stomach. World J Surg. 2006;30:1864-1869. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 65] [Cited by in RCA: 76] [Article Influence: 4.0] [Reference Citation Analysis (0)] |
| 9. | Borch K, Jönsson B, Tarpila E, Franzén T, Berglund J, Kullman E, Franzén L. Changing pattern of histological type, location, stage and outcome of surgical treatment of gastric carcinoma. Br J Surg. 2000;87:618-626. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 74] [Cited by in RCA: 75] [Article Influence: 3.0] [Reference Citation Analysis (0)] |
| 10. | del Casar JM, Corte MD, Alvarez A, García I, Bongera M, González LO, García-Muñiz JL, Allende MT, Astudillo A, Vizoso FJ. Lymphatic and/or blood vessel invasion in gastric cancer: relationship with clinicopathological parameters, biological factors and prognostic significance. J Cancer Res Clin Oncol. 2008;134:153-161. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 38] [Cited by in RCA: 37] [Article Influence: 2.2] [Reference Citation Analysis (0)] |
| 11. | Zhao B, Huang X, Zhang J, Luo R, Lu H, Xu H, Huang B. Clinicopathologic factors associated with recurrence and long-term survival in node-negative advanced gastric cancer patients. Rev Esp Enferm Dig. 2019;111:111-120. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 2] [Cited by in RCA: 6] [Article Influence: 1.2] [Reference Citation Analysis (0)] |
| 12. | Lee JH, Kim MG, Jung MS, Kwon SJ. Prognostic significance of lymphovascular invasion in node-negative gastric cancer. World J Surg. 2015;39:732-739. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 31] [Cited by in RCA: 44] [Article Influence: 4.4] [Reference Citation Analysis (0)] |
| 13. | An JY, Kang TH, Choi MG, Noh JH, Sohn TS, Kim S. Borrmann type IV: an independent prognostic factor for survival in gastric cancer. J Gastrointest Surg. 2008;12:1364-1369. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 50] [Cited by in RCA: 58] [Article Influence: 3.4] [Reference Citation Analysis (0)] |
| 14. | Luo Y, Gao P, Song Y, Sun J, Huang X, Zhao J, Ma B, Li Y, Wang Z. Clinicopathologic characteristics and prognosis of Borrmann type IV gastric cancer: a meta-analysis. World J Surg Oncol. 2016;14:49. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 35] [Cited by in RCA: 50] [Article Influence: 5.6] [Reference Citation Analysis (0)] |
| 15. | Zhu YL, Yang L, Sui ZQ, Liu L, Du JF. Clinicopathological features and prognosis of Borrmann type IV gastric cancer. J BUON. 2016;21:1471-1475. [PubMed] |
| 16. | Japanese Gastric Cancer Association. Japanese classification of gastric carcinoma: 3rd English edition. Gastric Cancer. 2011;14:101-112. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 2390] [Cited by in RCA: 2872] [Article Influence: 205.1] [Reference Citation Analysis (0)] |
| 17. | Li F, Zhang R, Liang H, Liu H, Quan J. The pattern and risk factors of recurrence of proximal gastric cancer after curative resection. J Surg Oncol. 2013;107:130-135. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 28] [Cited by in RCA: 29] [Article Influence: 2.2] [Reference Citation Analysis (0)] |
| 18. | Du CY, Chen JG, Zhou Y, Zhao GF, Fu H, Zhou XK, Shi YQ. Impact of lymphatic and/or blood vessel invasion in stage II gastric cancer. World J Gastroenterol. 2012;18:3610-3616. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in CrossRef: 21] [Cited by in RCA: 27] [Article Influence: 2.1] [Reference Citation Analysis (0)] |
| 19. | Ichikawa D, Kubota T, Kikuchi S, Fujiwara H, Konishi H, Tsujiura M, Ikoma H, Nakanishi M, Okamoto K, Sakakura C, Ochiai T, Kokuba Y, Otsuji E. Prognostic impact of lymphatic invasion in patients with node-negative gastric cancer. J Surg Oncol. 2009;100:111-114. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 21] [Cited by in RCA: 25] [Article Influence: 1.6] [Reference Citation Analysis (0)] |
| 20. | Lee EW, Lee WY, Koo HS. Prognostic Factors for Node-Negative Advanced Gastric Cancer after Curative Gastrectomy. J Gastric Cancer. 2016;16:161-166. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 9] [Cited by in RCA: 9] [Article Influence: 1.0] [Reference Citation Analysis (0)] |
| 21. | Pan M, Huang P, Li S, Chen J, Wei S, Zhang Y. Double contrast-enhanced ultrasonography in preoperative Borrmann classification of advanced gastric carcinoma: comparison with histopathology. Sci Rep. 2013;3:3338. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 13] [Cited by in RCA: 15] [Article Influence: 1.3] [Reference Citation Analysis (0)] |
| 22. | Huang JY, Wang ZN, Lu CY, Miao ZF, Zhu Z, Song YX, Xu HM, Xu YY. Borrmann type IV gastric cancer should be classified as pT4b disease. J Surg Res. 2016;203:258-267. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 12] [Cited by in RCA: 14] [Article Influence: 1.6] [Reference Citation Analysis (0)] |
| 23. | Yang B, Wu G, Wang X, Zhang X. Discussion of modifying stage IV gastric cancer based on Borrmann classification. Tumour Biol. 2013;34:1485-1491. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 5] [Cited by in RCA: 6] [Article Influence: 0.5] [Reference Citation Analysis (0)] |