Published online Dec 15, 2019. doi: 10.4251/wjgo.v11.i12.1182
Peer-review started: February 26, 2019
First decision: July 31, 2019
Revised: August 29, 2019
Accepted: November 4, 2019
Article in press: November 4, 2019
Published online: December 15, 2019
Processing time: 288 Days and 14.8 Hours
Malnourishment and sarcopenia are well documented phenomena in oesophageal cancer. Patients undergoing neo-adjuvant chemotherapy prior to oesophagectomy have complex nutritional needs.
To examine the effect of regular nutritional support via feeding jejunostomy on overall body composition in patients undergoing neo-adjuvant chemotherapy prior to oesophagectomy for oesophageal cancer.
Retrospective data were collected for 15 patients before and after neo-adjuvant chemotherapy. All patients had feeding jejunostomies inserted at staging laparoscopy prior to neo-adjuvant chemotherapy and underwent regular jejunostomy feeding. Changes in body composition were determined by analysis of computed tomography imaging.
Patient age was 61.3 ± 12.8 years, and 73% of patients were male. The time between start of chemotherapy and surgery was 107 ± 21.6 d. There was no change in weight (74.5 ± 14.1 kg to 74.8 ± 13.1 kg) and body mass index (26.0 ± 3.8 kg/m2 to 26.1 ± 3.4 kg/m2). Body composition analysis revealed a statistically significant decrease in lumbar skeletal muscle index despite regular feeding (45.8 ± 8.0 cm2/m2 to 43.5 ± 7.3 cm2/m2; P = 0.045). The proportion of sarcopenic patients increased (33.3% to 60%). Six patients (40%) experienced dose-limiting toxicity during chemotherapy.
Regular jejunostomy feeding during neo-adjuvant chemotherapy can maintain weight and adipose tissue. Feeding alone is not sufficient to maintain muscle mass. Further insight into the underlying processes causing reduced muscle mass in cancer patients may help to provide targeted interventions.
Core tip: Patients undergoing neo-adjuvant chemotherapy prior to oesophagectomy have complex nutritional needs. Retrospective data were collected for 15 patients before and after neo-adjuvant chemotherapy to examine the effect of regular nutritional support via feeding jejunostomy on overall body composition. There was no change in weight and body mass index. Body composition analysis revealed a statistically significant decrease in lumbar skeletal muscle index despite regular feeding. Regular jejunostomy feeding during neo-adjuvant chemotherapy can maintain weight and adipose tissue. Further insight into processes causing reduced muscle mass in cancer patients may provide targeted interventions.
- Citation: Mohamed IM, Whiting J, Tan BH. Impact of regular enteral feeding via jejunostomy during neo-adjuvant chemotherapy on body composition in patients with oesophageal cancer. World J Gastrointest Oncol 2019; 11(12): 1182-1192
- URL: https://www.wjgnet.com/1948-5204/full/v11/i12/1182.htm
- DOI: https://dx.doi.org/10.4251/wjgo.v11.i12.1182
Oesophageal cancer is the sixth most common cause of cancer death in the United States[1]. Neo-adjuvant chemotherapy is now the mainstay of treatment prior to oesophagectomy and is associated with improved survival compared to surgery alone[2]. However, use of such therapy places additional nutritional burden on patients and has an adverse impact on nutritional status and body composition as measured by the proportions of skeletal muscle and fat[3]. Continued improvement in overall survival has led to increased focus on optimising patients’ nutritional status throughout the whole continuum of their management.
Patients with oesophageal cancer have complex nutritional needs. Such patients often experience the onset of a catabolic state induced by the malignancy as well as tumour-associated dysphagia. The use of multiple strategies to manage disease progression including radiation, chemotherapy and surgical resection present an additional nutritional burden to these patients who are often malnourished at presentation. Adequate nutritional reserves are essential to allow patients to tolerate neo-adjuvant chemotherapy as well as subsequent surgery. Malnourishment and sarcopenia are well documented phenomena in oesophageal cancer. Local tumour effects, dysphagia, loss of appetite, physical inactivity as well as psychological factors all impair dietary intake and serve to compound cancer cachexia[2-4]. Malnourishment reduces the potential response of the malignancy to chemoradiotherapy and impairs the patient’s ability to tolerate the full course of treatment. Nutritional deficiencies may also contribute to increased perioperative morbidity and mortality.
There are various indicators of nutritional status that predict an individual’s ability to tolerate neo-adjuvant chemotherapy and surgery for oesophageal cancer. Lower body mass index (BMI), skeletal muscle depletion, sarcopenia and sarcopenic obesity are associated with dose-limiting toxicity (DLT) during treatment with neo-adjuvant chemotherapy[4-6]. Treatment with neo-adjuvant chemotherapy is also associated with changes in body composition. Indeed, Awad et al[7] showed a decrease in fat free mass and increased prevalence of sarcopenia in their observational study after treatment with neo-adjuvant chemotherapy. Tsujimoto et al[8] recorded statistically significant decreases in body weight and serum total protein in patients who received neo-adjuvant chemotherapy. Toxicity from neo-adjuvant chemotherapy can also lead to further malnutrition and weight loss. Thus, further changes in body composition can affect tolerability of subsequent treatment, which in turn relates to poorer patient outcomes[3,9].
The period preceding surgery is the ideal time to ensure patients are in the best possible state for surgery. Optimisation of nutrition through nutritional screening and supplementation is an important consideration in the overall peri-operative management of oesophageal cancer, including during treatment with neo-adjuvant chemotherapy.
The use of enteral feeding via jejunostomy, particularly in oesophageal cancer, is a reliable method to optimise nutrition. The associated risks of insertion do not significantly outweigh the conferred benefits[8,10,11]. Previous studies of feeding via laparoscopically inserted jejunostomy prior to neo-adjuvant chemotherapy demonstrated an increase in weight ranging from 0.4 to 11.8 kg[2]. The present study is the first to our knowledge to examine the changes of body composition from regular enteral feeding via jejunostomy during neo-adjuvant therapy for oesophageal cancer[2].
The aim of this study is to examine the effect of regular enteral feeding via jejunostomy on overall body composition in a cohort of patients with oesophageal cancer undergoing neo-adjuvant chemotherapy prior to oesophagectomy. The effect of regular jejunostomy feeding on the development of DLT during neo-adjuvant chemotherapy was also examined.
Patients having potentially curative, locally advanced oesophageal and oesophago-gastric junctional cancer without evidence of metastasis presenting to the Queen Elizabeth Hospital, Birmingham Upper Gastrointestinal Multidisciplinary Team between March 2014 and June 2017 were considered for this study. All patients were routinely staged with a combination of computed tomography (CT), endoscopic ultrasound and laparoscopy according to the International Union Against Cancer system[12]. Those with locally advanced disease (T2 and greater and/or locoregional lymphadenopathy) and with sufficient physiological reserve to tolerate neo-adjuvant chemotherapy were further examined retrospectively for suitability for inclusion into the present study.
All patients included in this study were reviewed by a Consultant Oesophago-gastric Surgeon after diagnosis and were assessed to have a dysphagia score of 3 (the ability to swallow liquids only) or 4 (complete dysphagia)[13]. These patients had a feeding jejunostomy inserted at the time of staging laparoscopy prior to commencement of neo-adjuvant chemotherapy.
All patients were assessed by a dietician prior to starting regular jejunostomy feeding. After assessment by the dietitian, each patient had the feeding regimen tailored to their individual nutritional needs. The Henry equation[14] was used to estimate basal metabolic rate with 10% added as a stress factor during chemotherapy. Each individuals’ activity factor was also considered. Based on these, the calorie intake required per day was calculated. The feeding regiment aims to provide 0.17-0.2 g nitrogen/kg/d. Each individuals’ feeding regiment takes into account the amount of oral diet achieved. The feeding regimen aimed to provide 10-12 hr overnight continuous feeding if possible, to limit disruption of daytime activities. All patients were maintained on regular feeding daily throughout their period of receiving neo-adjuvant chemotherapy.
Weight and height were recorded according to standard methods. Weight was measured with a medical balance beam scale, and height was measured with a stadiometer. BMI was calculated [weight (kg)/height (m2)].
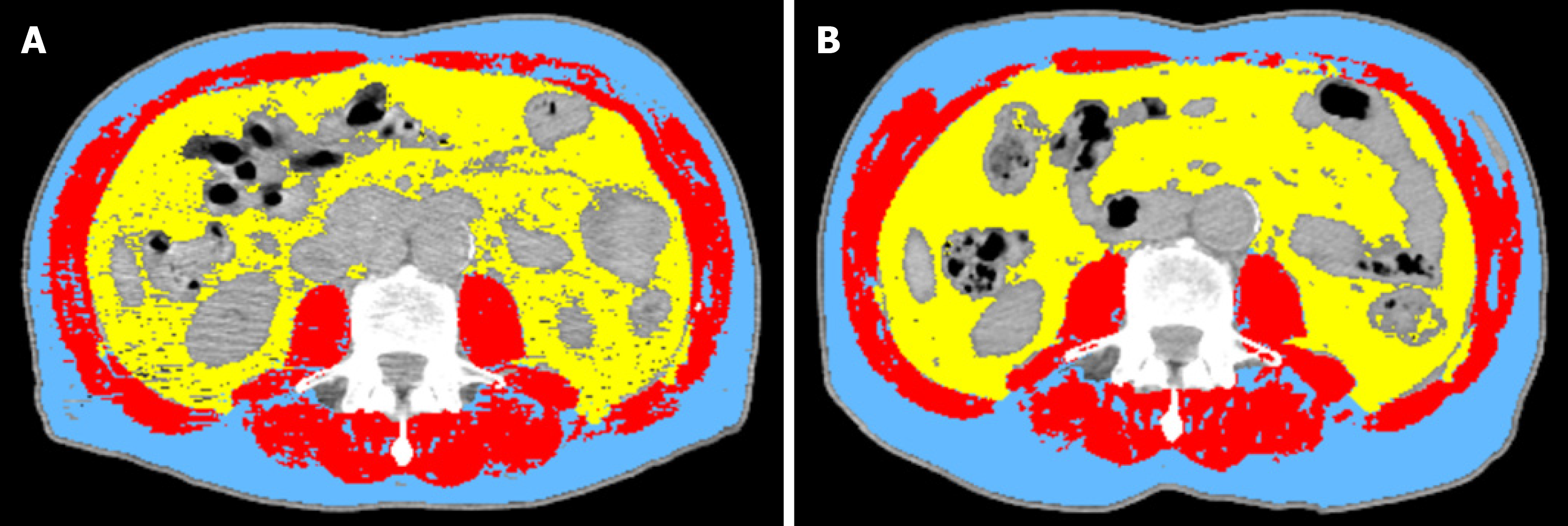
CT has proven to be accurate for measuring human body composition[15,16]. Regional muscle tissue was measured by CT from electronically stored images, which had been done previously for diagnostic purposes. CT scans were performed at two time points: The first at diagnosis prior to commencement of chemotherapy and the second after completion of neo-adjuvant chemotherapy prior to surgery (Figure 1).
The third lumbar vertebra (L3) was chosen as a landmark, and two consecutive slices were assessed to measure cross-sectional area of muscle and adipose tissue as described[17]. The average value of two images was computed for each patient. Images were analysed using Slice-O-Matic software V4.3 (Tomovision). Cross sectional area for muscle and adipose tissue was normalized for stature (cm2/m2) and reported. Patients were classified as sarcopenic according to established cut offs: L3 muscle index < 41 cm2/m2 for women and < 43 cm2/m2 for men with a BMI < 25 or L3 muscle index < 53 cm2/m2 for men with a BMI > 25[18]. The mean Hounsfield unit measurement of all skeletal muscle within the L3 cross-section was recorded as a measure of myosteatosis, which was defined operationally as a mean skeletal muscle radiodensity of < 33 Hounsfield unit in those with a BMI > 25; and < 41 Hounsfield unit in those with a BMI < 25 across the axial orthogonal view[18].
DLT was defined by intolerable toxicities requiring the postponement of treatment, a drug dose reduction or definitive interruption of drug administration.
Data were recorded as mean ± standard deviation unless otherwise stated. Comparison between values at diagnosis and post neo-adjuvant chemotherapy were calculated using the paired t-test or McNemar’s test. A P-value less than 0.05 was considered to be statistically significant. Statistical analysis was done using SPSS 15.0 statistical package (SPSS Inc.).
During the study period, 15 patients underwent regular jejunostomy feeding during neo-adjuvant chemotherapy prior to surgery. Demographic data of the 15 patients are shown in Table 1. The mean age was 61.3 ± 12.8 years, and 73% of patients were male.
| No. of patients, n = 15 | |
| Age in yr, mean ± SD | 61.3 ± 12.8 |
| Sex | |
| M | 11 (73.3) |
| F | 4 (26.7) |
| Tumour site | |
| Mid oesophagus | 1 (6.6) |
| Lower oesophagus | 4 (26.7) |
| Oesophageal-gastric junction | 10 (66.7) |
| Histology | |
| Adenocarcinoma | 11 (73.3) |
| Squamous cell cancer | 3 (20.0) |
| Other | 1 (6.7) |
| Stage | |
| I | 1 (6.6) |
| II | 4 (26.7) |
| III | 10 (66.7) |
| Neo-adjuvant chemotherapy protocol | |
| ECX | 7 (46.7) |
| ECF | 3 (20) |
| EOX | 1 (6.6) |
| CX | 2 (13.3) |
| CF | 2 (13.3) |
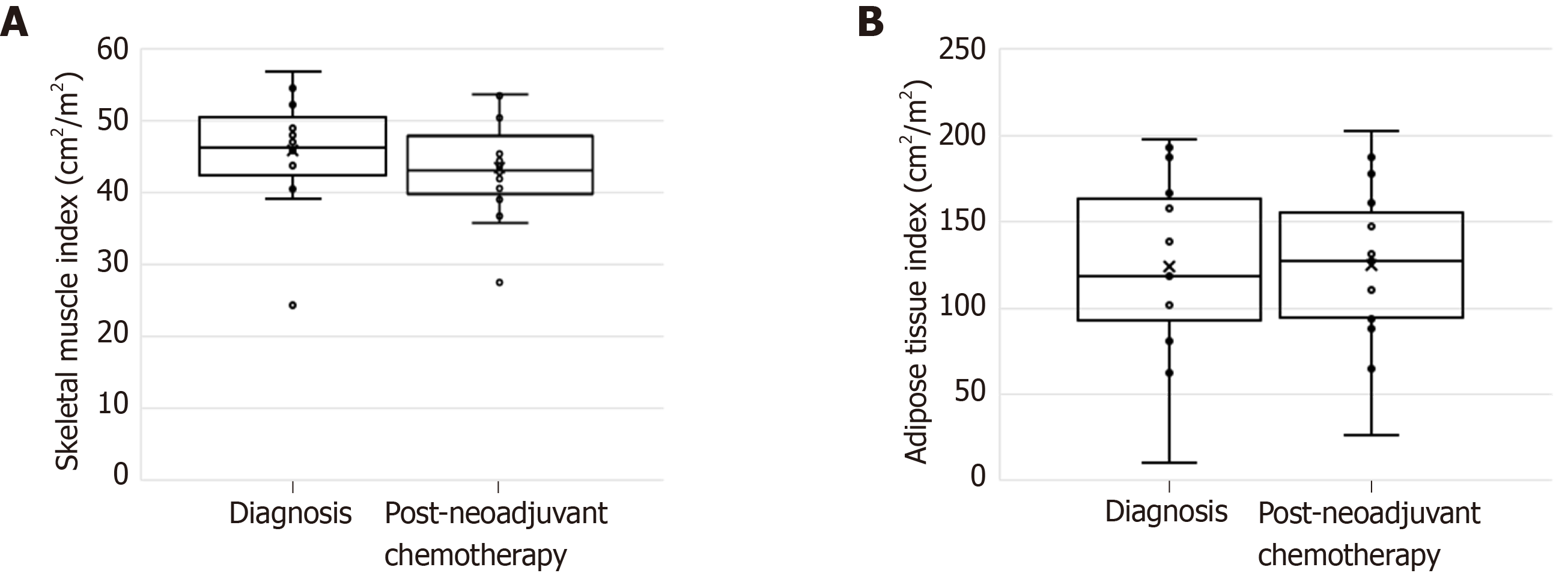
The time interval between start of chemotherapy and surgery was 107 ± 21.6 d. During this period, there was no change in weight and BMI (Table 2). However, body composition analysis revealed a statistically significant loss of skeletal muscle despite regular feeding during neo-adjuvant chemotherapy. Lumbar skeletal muscle index decreased from 45.8 ± 8.0 cm2/m2 to 43.5 ± 7.3 cm2/m2 (P = 0.045). Adipose tissue index remained largely unchanged (Figure 2). The number of sarcopenic patients increased from five (33.3%) at diagnosis to nine (60%) after completion of neo-adjuvant chemotherapy. There was no change in the number of myosteatotic patients before and after neo-adjuvant chemotherapy (Table 2).
| Diagnosis | Post-chemotherapy | P value | |
| Weight, mean ± SD, kg | 74.5 ± 14.1 | 74.8 ± 13.1 | 0.6591 |
| BMI, mean ± SD, kg/m2) | 26.0 ± 3.8 | 26.1 ± 3.4 | 0.6231 |
| Lumbar skeletal muscle index as cm2/m2, mean ± SD | 45.8 ± 8.0 | 43.5 ± 7.3 | 0.0481 |
| Lumbar adipose tissue index as cm2/m2, mean ± SD, | 124.0 ± 53.7 | 124.6 ± 47.9 | 0.8781 |
| Sarcopenic | 5 (33.3) | 9 (60.0) | 0.2192 |
| Myosteatotic | 8 (53.3) | 8 (53.3) | 1.0002 |
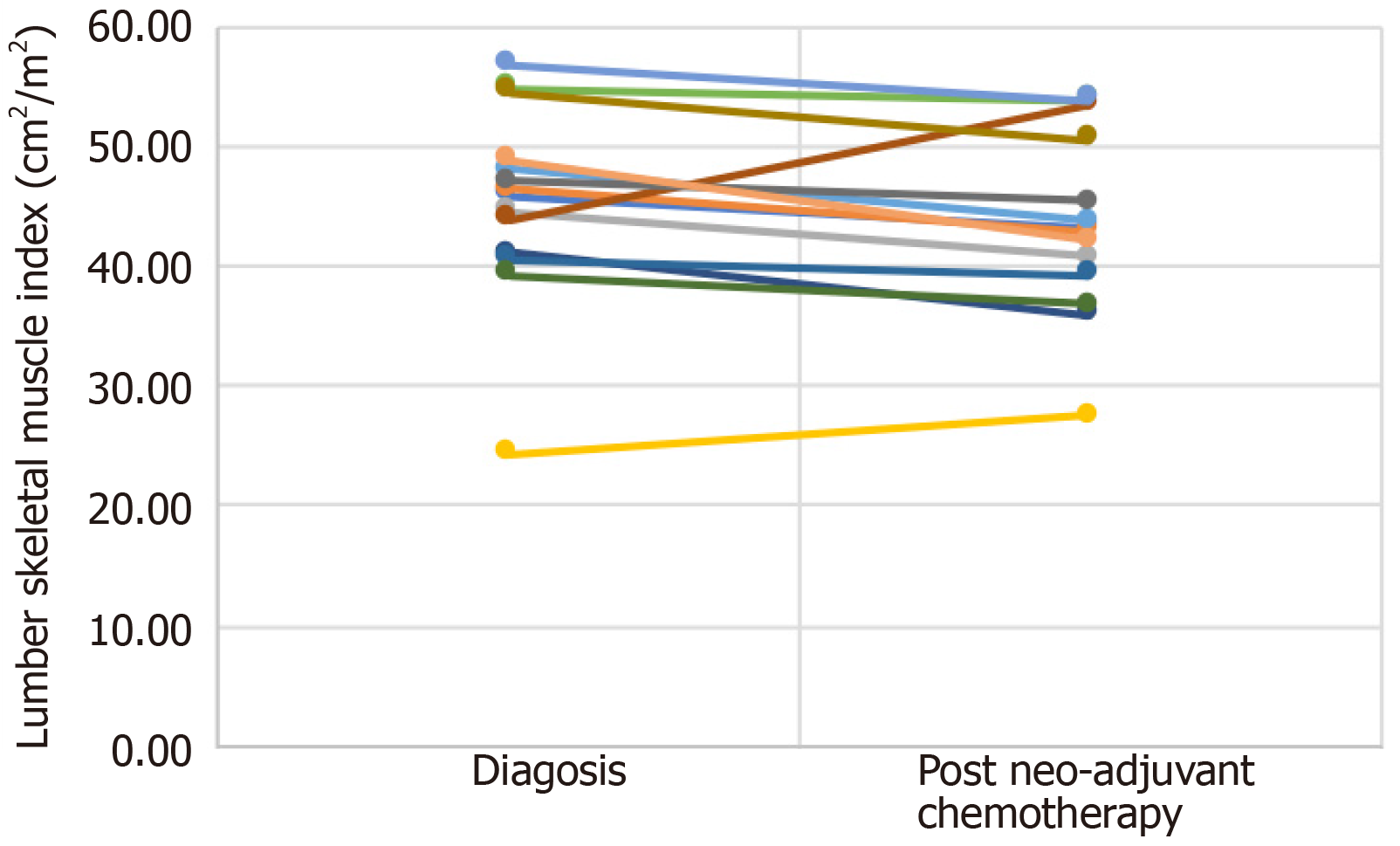
Table 3 and Figure 3 show the individual values for changes in lumbar skeletal muscle index (cm2/m2) from diagnosis to after neo-adjuvant chemotherapy following regular enteral feeding via jejunostomy. Patients 4 and 8 showed an increase in lumbar skeletal muscle index between diagnosis and after neo-adjuvant chemotherapy. It is interesting to note that both patients were sarcopenic at diagnosis but were no longer sarcopenic after neo-adjuvant chemotherapy.
| Patient ID | Diagnosis | After neo-adjuvant chemotherapy |
| Patient 1 | 45.86 | 43.20 |
| Patient 2 | 46.27 | 42.87 |
| Patient 3 | 44.44 | 40.65 |
| Patient 4 | 24.25 | 27.50 |
| Patient 5 | 47.97 | 43.77 |
| Patient 6 | 54.85 | 53.80 |
| Patient 7 | 41.05 | 35.91 |
| Patient 8 | 43.79 | 53.48 |
| Patient 9 | 47.00 | 45.39 |
| Patient 10 | 54.47 | 50.53 |
| Patient 11 | 40.50 | 39.20 |
| Patient 12 | 39.15 | 36.71 |
| Patient 13 | 52.26 | 44.59 |
| Patient 14 | 56.74 | 53.76 |
| Patient 15 | 48.84 | 42.05 |
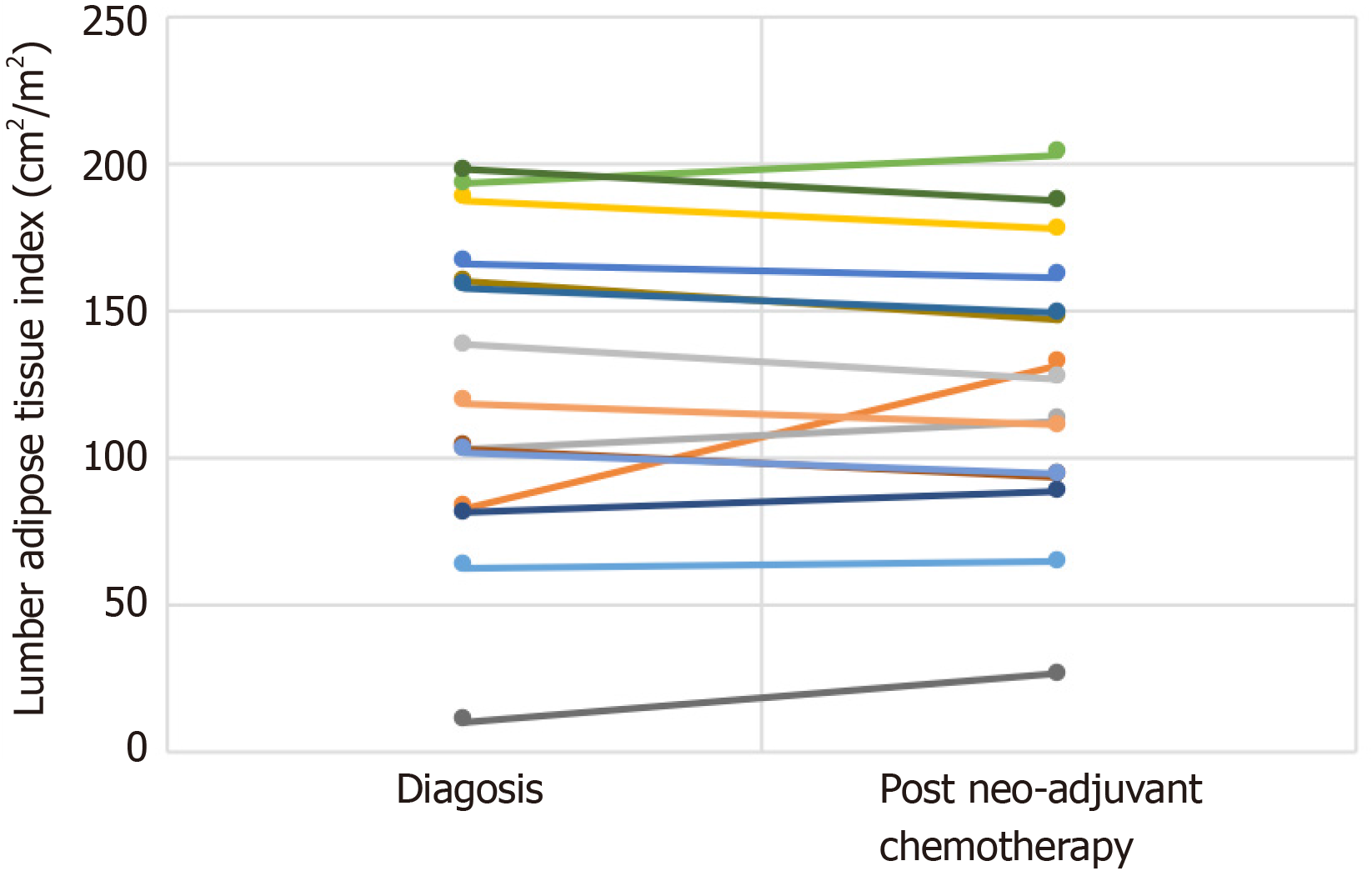
Table 4 and Figure 4 show individual values for changes in lumbar adipose tissue index (cm2/m2) from diagnosis to after neo-adjuvant chemotherapy following regular enteral feeding via jejunostomy. Patients 2, 3, 5, 6, 7 and 9 showed an increase in lumbar adipose tissue index from diagnosis to after neo-adjuvant chemotherapy. The greatest increases were seen in patients 2 and 9. None of these patients were sarcopenic at diagnosis, however patients 2, 3, 6 and 7 became sarcopenic after neo-adjuvant chemotherapy.
| Patient ID | Diagnosis | After neo-adjuvant chemotherapy |
| Patient 1 | 166.10 | 160.91 |
| Patient 2 | 82.71 | 131.25 |
| Patient 3 | 102.27 | 112.29 |
| Patient 4 | 187.18 | 177.17 |
| Patient 5 | 62.08 | 64.25 |
| Patient 6 | 192.85 | 202.49 |
| Patient 7 | 80.79 | 88.04 |
| Patient 8 | 102.87 | 93.52 |
| Patient 9 | 10.03 | 26.22 |
| Patient 10 | 159.49 | 146.65 |
| Patient 11 | 157.76 | 148.82 |
| Patient 12 | 197.29 | 186.82 |
| Patient 13 | 101.62 | 93.82 |
| Patient 14 | 118.47 | 110.45 |
| Patient 15 | 137.98 | 126.63 |
Six patients (40%) experienced DLT during chemotherapy. Three patients required a dose reduction, two patients stopped chemotherapy early and one patient experienced a delay in treatment.
This observational study of 15 patients undergoing neo-adjuvant chemotherapy prior to oesophagectomy confirmed that formal nutritional intervention provided through the use of feeding jejunostomy had a positive effect on maintaining mean overall weight and BMI.
In concordance with previous studies, this study revealed a small amount of weight gain (0.35 ± 3.0 kg) after regular jejunostomy feeding, though this was negligible in the current study. Interestingly, adipose tissue was maintained but a significant loss in muscle mass remained. The incidence of sarcopenia increased from 33% of patients to 60% of patients after completion of neo-adjuvant chemotherapy.
The percentage of patients (40%) experiencing DLT whilst undergoing neo-adjuvant chemotherapy was similar to a previous study despite regular jejunostomy feeding[5]. This can perhaps be explained by the high prevalence of sarcopenia in oesophageal cancer patients despite regular feeding. Whilst the mechanism that links sarcopenia with increased chemotherapy toxicity in patients is unknown, this association is described in the literature[3,6,7].
Sarcopenia is prevalent in patients diagnosed with oesophageal cancer, and the incidence increases following neo-adjuvant chemotherapy. This is coupled with an overall decrease in body weight, BMI, fat mass and fat-free mass. BMI is often used as an objective marker to assess patients’ nutritional status. However, patients can be acutely malnourished and still have a normal or elevated BMI, especially with the increasing prevalence of obesity. Therefore, assessment of specific changes in body composition is perhaps more important than overall changes in weight or BMI.
One of the strengths of this study was the use of the standardised technique of using CT imaging to measure muscle mass using the skeletal muscle index at the level of the L3 vertebra. This has been proven to be a well-established technique for this purpose. Although magnetic resonance imaging and dual energy x-ray absorptiometry are also considered extremely useful to evaluate body composition in clinical practice, CT is performed in all patients with oesophageal cancer. Therefore, analysis of these images is easily available with no additional cost or patient burden[9].
Sarcopenia, myosteatosis and loss of skeletal muscle during neo-adjuvant treatment have been shown to be associated with worse oncological outcomes in surgically treated oesophageal cancer patients[3,7,9]. In sarcopenic patients, the doses of neo-adjuvant therapy may have to be adjusted and even reduced in order to compensate for toxicity associated complications. If the development of sarcopenia can be minimised, patients may benefit not only from longer courses of neo-adjuvant therapy, but the documented poorer outcomes may also be averted. It would therefore seem logical to attempt to maintain or improve skeletal mass during neo-adjuvant chemotherapy.
The use of feeding jejunostomy tubes in this cohort of patients has been shown to be safe. Although the current study did not specifically examine patient tolerance of feeding jejunostomy or complications associated with insertion, a systematic review examining nutritional optimisation in these patients reported no 30-d postprocedural complications in patients undergoing laparoscopically inserted feeding jejunostomy tubes. All such patients showed an increase in overall weight, and available evidence suggests that greater than 90% of patients completed their neo-adjuvant treatment[2]. In light of the relatively few and minor complications associated with jejunostomy, it would make sense to employ this as a feeding technique over nasogastric tubes, which are prone to easily dislodge. The question of whether placement of jejunostomy would increase difficulty of the oesophagectomy is valid. However, with laparoscopic insertion of jejunostomy tubes, intra-abdominal adhesions should be kept to a minimum, and the benefit of the tube to the patient would lend further support to placement of these tubes. Indeed, the jejunostomy tube may aid with early nutrition post-oesophagectomy.
A randomised study has suggested that enteral feed enriched with eicosapentaenoic acid, a compound that modulates the immune system and reduces catabolism in advanced cancer, may contribute to maintaining fat-free mass[19]. However, further research is required to determine the effect of such immuno-enhanced nutrition on overall clinical outcomes in patients with oesophageal cancer.
There are several limitations in the current study. One being the relatively small sample size. There is also heterogeneity in the type of chemotherapy used, which may be a contributory factor towards the changes in body composition. Other factors that may affect changes in weight such as hypoalbuminaemia, oedema and other comorbidities were not specifically examined in this study population. However, by the time of diagnosis, patients with oesophageal cancer are often already suffering from malnutrition and involuntary weight loss with sarcopenia being a major marker of frailty. Thus, addressing malnutrition pre-operatively is a major area to target for pre-optimisation before surgery[4].
It would also be useful to examine data on patients who did not receive feeding via feeding jejunostomy and especially those who were maintained on an oral diet. This would allow comparison with the current study group in order to measure any differences in outcome between the two groups and therefore would contribute to more detailed assessment of the effect of feeding in this group of patients. Nevertheless, this study has shown that regular jejunostomy feeding during neo-adjuvant chemotherapy can attenuate the previously shown loss in overall weight and adipose tissue. It is clear, however, that feeding alone is not sufficient to maintain muscle mass in this group of patients. Further insight into the underlying processes causing reduced muscle mass in cancer patients may help to provide targeted interventions.
In the meantime, there is evidence that physical exercise and resistance training has proven efficacy in catabolic conditions including sarcopenia and is advocated as a nonpharmacological intervention in cancer-related skeletal muscle wasting. Interestingly, resistance training during adjuvant breast cancer treatment has been reported to reverse sarcopenic status and lead to higher chemotherapy completion rates[20,21]. Therefore, the potential for such interventions to attenuate DLT in oesophageal cancer warrants further investigation.
This study has shown that nutritional supplementation does serve to maintain overall weight and BMI. Looking more specifically at body composition, skeletal muscle mass decreased despite this nutritional support, and the proportion of sarcopenic patients increased. It is the authors’ opinion that multimodal intervention incorporating a combination of regular nutritional support and exercise during the period of neo-adjuvant chemotherapy may lead to improvement in treatment tolerance and optimising surgical candidacy in patients with oesophageal cancer. This would be best investigated by means of a prospective multi-centre, multi-arm randomised controlled trial.
Malnourishment and sarcopenia are well documented phenomena in oesophageal cancer. Neo-adjuvant chemotherapy is now the mainstay of treatment prior to oesophagectomy and is associated with improved survival compared to surgery alone. Patients undergoing neo-adjuvant chemotherapy prior to oesophagectomy have complex nutritional needs. The use of multiple strategies to manage disease progression including radiation, chemotherapy and surgical resection present an additional nutritional burden to these patients who are often malnourished at presentation.
There are various indicators of nutritional status that predict an individual’s ability to tolerate neo-adjuvant chemotherapy and surgery for oesophageal cancer. Lower body mass index (BMI), skeletal muscle depletion, sarcopenia and sarcopenic obesity are associated with dose-limiting toxicity during treatment with neo-adjuvant chemotherapy. The period preceding surgery is the ideal time to ensure patients are in the best possible state for surgery. The use of enteral feeding via jejunostomy, particularly in oesophageal cancer, is a reliable method to optimise nutrition. The associated risks of insertion do not significantly outweigh the conferred benefits. The present study is the first to our knowledge to examine the changes of body composition from regular enteral feeding via jejunostomy during neo-adjuvant therapy for oesophageal cancer.
The aim of this study is to examine the effect of regular enteral feeding via jejunostomy on overall body composition in a cohort of patients with oesophageal cancer undergoing neo-adjuvant chemotherapy prior to oesophagectomy. The effect of regular jejunostomy feeding on the development of dose-limiting toxicity during neo-adjuvant chemotherapy was also examined.
Patients having potentially curative, locally advanced oesophageal and oesophago-gastric junctional cancer without evidence of metastasis were considered for this study. All patients were routinely staged with a combination of computed tomography (CT), endoscopic ultrasound and laparoscopy according to the International Union Against Cancer system. All patients were assessed by a dietician prior to starting regular jejunostomy feeding. After assessment by the dietitian, each patient had the feeding regimen tailored to their individual nutritional needs. Weight and height were recorded according to standard methods. CT has proven to be accurate for measuring human body composition. Regional muscle tissue was measured by CT from electronically stored images, which had been done previously for diagnostic purposes. CT scans were performed at two time points: The first at diagnosis prior to commencement of chemotherapy and the second after completion of neo-adjuvant chemotherapy prior to surgery. The third lumbar vertebra was chosen as a landmark, and two consecutive slices were assessed to measure cross-sectional area of muscle and adipose tissue as described. The average value of two images was computed for each patient.
During the study period, 15 patients underwent regular jejunostomy feeding during neo-adjuvant chemotherapy prior to surgery. The time interval between the start of chemotherapy and surgery was 107 ± 21.6 d. During this period, there was no change in weight and BMI. However, body composition analysis revealed a statistically significant loss of skeletal muscle despite regular feeding during neo-adjuvant chemotherapy. Lumbar skeletal muscle index decreased. Adipose tissue index remained largely unchanged.
This observational study of 15 patients undergoing neo-adjuvant chemotherapy prior to oesophagectomy confirmed that formal nutritional intervention provided through the use of feeding jejunostomy had a positive effect on maintaining mean overall weight and BMI.
This study has shown that nutritional supplementation does serve to maintain overall weight and BMI. Looking more specifically at body composition, skeletal muscle mass decreased despite this nutritional support, and the proportion of sarcopenic patients increased. It is the authors’ opinion that multimodal intervention incorporating a combination of regular nutritional support and exercise during the period of neo-adjuvant chemotherapy may lead to improvement in treatment tolerance and optimising surgical candidacy in patients with oesophageal cancer. This would be best investigated by means of a prospective multi-centre, multi-arm randomised controlled trial.
Manuscript source: Invited manuscript
Specialty type: Oncology
Country of origin: United Kingdom
Peer-review report classification
Grade A (Excellent): 0
Grade B (Very good): B
Grade C (Good): C, C
Grade D (Fair): 0
Grade E (Poor): 0
P-Reviewer: Aykan NF, Merrett ND, Shi H S-Editor: Ma YJ L-Editor: Filipodia E-Editor: Qi LL
| 1. | Cancer Research UK. Cancer Research UK. Oesophageal Cancer Statistics. 2018;[Internet]. [cited 2019 Jan 12] Available from: http://www.cancerresearchuk.org/health-professional/cancer-statistics/statistics-by-cancer-type/oesophageal-cancer - heading-Zero. |
| 2. | Huddy JR, Huddy FMS, Markar SR, Tucker O. Nutritional optimization during neoadjuvant therapy prior to surgical resection of esophageal cancer-a narrative review. Dis Esophagus. 2018;31:1-11. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 28] [Cited by in RCA: 48] [Article Influence: 6.9] [Reference Citation Analysis (0)] |
| 3. | Guinan EM, Doyle SL, Bennett AE, O'Neill L, Gannon J, Elliott JA, O'Sullivan J, Reynolds JV, Hussey J. Sarcopenia during neoadjuvant therapy for oesophageal cancer: characterising the impact on muscle strength and physical performance. Support Care Cancer. 2018;26:1569-1576. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 15] [Cited by in RCA: 34] [Article Influence: 4.3] [Reference Citation Analysis (0)] |
| 4. | Steenhagen E, van Vulpen JK, van Hillegersberg R, May AM, Siersema PD. Nutrition in peri-operative esophageal cancer management. Expert Rev Gastroenterol Hepatol. 2017;11:663-672. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 39] [Cited by in RCA: 59] [Article Influence: 7.4] [Reference Citation Analysis (0)] |
| 5. | Tan BH, Brammer K, Randhawa N, Welch NT, Parsons SL, James EJ, Catton JA. Sarcopenia is associated with toxicity in patients undergoing neo-adjuvant chemotherapy for oesophago-gastric cancer. Eur J Surg Oncol. 2015;41:333-338. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 168] [Cited by in RCA: 204] [Article Influence: 18.5] [Reference Citation Analysis (0)] |
| 6. | Rier HN, Jager A, Sleijfer S, Maier AB, Levin MD. The Prevalence and Prognostic Value of Low Muscle Mass in Cancer Patients: A Review of the Literature. Oncologist. 2016;21:1396-1409. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 98] [Cited by in RCA: 156] [Article Influence: 17.3] [Reference Citation Analysis (0)] |
| 7. | Awad S, Tan BH, Cui H, Bhalla A, Fearon KC, Parsons SL, Catton JA, Lobo DN. Marked changes in body composition following neoadjuvant chemotherapy for oesophagogastric cancer. Clin Nutr. 2012;31:74-77. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 175] [Cited by in RCA: 210] [Article Influence: 15.0] [Reference Citation Analysis (0)] |
| 8. | Tsujimoto H, Hiraki S, Takahata R, Nomura S, Ito N, Kanematsu K, Horiguchi H, Aosasa S, Yamamoto J, Hase K. Laparoscopic jejunostomy for obstructing upper gastrointestinal malignancies. Mol Clin Oncol. 2015;3:1307-1310. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 10] [Cited by in RCA: 12] [Article Influence: 1.2] [Reference Citation Analysis (0)] |
| 9. | Yip C, Goh V, Davies A, Gossage J, Mitchell-Hay R, Hynes O, Maisey N, Ross P, Gaya A, Landau DB, Cook GJ, Griffin N, Mason R. Assessment of sarcopenia and changes in body composition after neoadjuvant chemotherapy and associations with clinical outcomes in oesophageal cancer. Eur Radiol. 2014;24:998-1005. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 160] [Cited by in RCA: 180] [Article Influence: 16.4] [Reference Citation Analysis (0)] |
| 10. | Jenkinson AD, Lim J, Agrawal N, Menzies D. Laparoscopic feeding jejunostomy in esophagogastric cancer. Surg Endosc. 2007;21:299-302. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 38] [Cited by in RCA: 43] [Article Influence: 2.3] [Reference Citation Analysis (0)] |
| 11. | Ben-David K, Kim T, Caban AM, Rossidis G, Rodriguez SS, Hochwald SN. Pre-therapy laparoscopic feeding jejunostomy is safe and effective in patients undergoing minimally invasive esophagectomy for cancer. J Gastrointest Surg. 2013;17:1352-1358. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 32] [Cited by in RCA: 26] [Article Influence: 2.2] [Reference Citation Analysis (0)] |
| 12. | Brierley JD, Gospodarowicz MK, Wittekind C. TNM classification of malignant tumours, 8th Edition. Wiley-Blackwell. 2016;272. |
| 13. | Knyrim K, Wagner HJ, Bethge N, Keymling M, Vakil N. A controlled trial of an expansile metal stent for palliation of esophageal obstruction due to inoperable cancer. N Engl J Med. 1993;329:1302-1307. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 543] [Cited by in RCA: 499] [Article Influence: 15.6] [Reference Citation Analysis (0)] |
| 14. | Henry CJ. Basal metabolic rate studies in humans: measurement and development of new equations. Public Health Nutr. 2005;8:1133-1152. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 580] [Cited by in RCA: 655] [Article Influence: 34.5] [Reference Citation Analysis (0)] |
| 15. | Mitsiopoulos N, Baumgartner RN, Heymsfield SB, Lyons W, Gallagher D, Ross R. Cadaver validation of skeletal muscle measurement by magnetic resonance imaging and computerized tomography. J Appl Physiol (1985). 1998;85:115-122. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 963] [Cited by in RCA: 1127] [Article Influence: 41.7] [Reference Citation Analysis (0)] |
| 16. | Heymsfield SB, Wang Z, Baumgartner RN, Ross R. Human body composition: advances in models and methods. Annu Rev Nutr. 1997;17:527-558. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 329] [Cited by in RCA: 323] [Article Influence: 11.5] [Reference Citation Analysis (0)] |
| 17. | Prado CM, Heymsfield SB. Lean tissue imaging: a new era for nutritional assessment and intervention. JPEN J Parenter Enteral Nutr. 2014;38:940-953. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 336] [Cited by in RCA: 413] [Article Influence: 37.5] [Reference Citation Analysis (0)] |
| 18. | Martin L, Birdsell L, Macdonald N, Reiman T, Clandinin MT, McCargar LJ, Murphy R, Ghosh S, Sawyer MB, Baracos VE. Cancer cachexia in the age of obesity: skeletal muscle depletion is a powerful prognostic factor, independent of body mass index. J Clin Oncol. 2013;31:1539-1547. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 1436] [Cited by in RCA: 1512] [Article Influence: 126.0] [Reference Citation Analysis (0)] |
| 19. | Ryan AM, Reynolds JV, Healy L, Byrne M, Moore J, Brannelly N, McHugh A, McCormack D, Flood P. Enteral nutrition enriched with eicosapentaenoic acid (EPA) preserves lean body mass following esophageal cancer surgery: results of a double-blinded randomized controlled trial. Ann Surg. 2009;249:355-363. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 198] [Cited by in RCA: 227] [Article Influence: 14.2] [Reference Citation Analysis (0)] |
| 20. | Dieli-Conwright CM, Courneya KS, Demark-Wahnefried W, Sami N, Lee K, Buchanan TA, Spicer DV, Tripathy D, Bernstein L, Mortimer JE. Effects of Aerobic and Resistance Exercise on Metabolic Syndrome, Sarcopenic Obesity, and Circulating Biomarkers in Overweight or Obese Survivors of Breast Cancer: A Randomized Controlled Trial. J Clin Oncol. 2018;36:875-883. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 142] [Cited by in RCA: 238] [Article Influence: 34.0] [Reference Citation Analysis (0)] |
| 21. | Adams SC, Segal RJ, McKenzie DC, Vallerand JR, Morielli AR, Mackey JR, Gelmon K, Friedenreich CM, Reid RD, Courneya KS. Impact of resistance and aerobic exercise on sarcopenia and dynapenia in breast cancer patients receiving adjuvant chemotherapy: a multicenter randomized controlled trial. Breast Cancer Res Treat. 2016;158:497-507. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 88] [Cited by in RCA: 116] [Article Influence: 12.9] [Reference Citation Analysis (0)] |