Published online Aug 27, 2025. doi: 10.4254/wjh.v17.i8.109444
Revised: May 25, 2025
Accepted: July 24, 2025
Published online: August 27, 2025
Processing time: 108 Days and 21.2 Hours
Skeletal muscles perform important metabolic functions. Muscle mass wasting in sarcopenia is an urgent problem of modern medicine, the interest in which is related to its prognostic significance. The liver has numerous direct and indirect metabolic and immune connections with skeletal muscle and disruptions of these connections in liver disease are of clinical interest. A recent article by Liang et al emphasized potential biomarkers of sarcopenia in liver cirrhosis. Identification of biomarkers of sarcopenia in patients with cirrhosis has important diagnostic value. Common pathophysiologic mechanisms of sarcopenia and liver cirrhosis include disorders of protein and energy metabolism, disturbances in the structure of gut microbiota, inflammation and oxidative stress.
Core Tip: Sarcopenia is characterized by loss of mass, strength, and skeletal muscle function. Skeletal muscle has numerous direct and indirect metabolic and immune links with the liver, which are disrupted in diseases such as metabolic dysfunction-associated steatotic liver disease and cirrhosis. Disruption of these links contributes to the prema
- Citation: Kotlyarov SN. Metabolic and immune links between sarcopenia and liver disease. World J Hepatol 2025; 17(8): 109444
- URL: https://www.wjgnet.com/1948-5182/full/v17/i8/109444.htm
- DOI: https://dx.doi.org/10.4254/wjh.v17.i8.109444
Sarcopenia, i.e. age-related loss of skeletal muscle mass, strength and function, is of growing interest to clinicians and researchers as it leads to serious health problems including the development of frailty syndrome, increased risk of falls and premature death[1-3]. Skeletal muscles are considered the largest organ in the human body, but their mass depends on various factors and changes with age[4,5]. In the process of natural aging there is a decrease in skeletal muscle mass, but the rate of this decrease is not so significant and allows to maintain physiological muscle functions until deep old age. A number of diseases can cause premature development of sarcopenia up to cachexia[6]. In patients with liver cirrhosis, the prevalence of sarcopenia, for example, ranges from 40% to 68%[7]. The presence of sarcopenia is an unfavorable prognostic factor in chronic diseases[8,9]. It is also associated with a decrease in the level of physical activity of patients with sarcopenia due to weakness and rapid fatigue and due to impaired biological muscle function. In this regard, improving the early diagnosis of sarcopenia is an important research objective. A recent article by Liang et al[10] emp
Various biomarkers are known to assess the risk of sarcopenia in patients with cirrhosis. It has been previously shown that serum levels of irisin and myostatin (and myostatin in combination with creatine phosphokinase or albumin) can be used as a potential biomarker to predict sarcopenia in patients with cirrhosis. In addition, hemoglobin and ferritin levels and low testosterone levels also predict sarcopenia in these patients[11-15].
Research in recent decades has greatly increased the understanding of the function of skeletal muscles and their links to other organs. In addition to the mechanical function of maintaining body position in space and locomotion, skeletal muscle plays a key role in thermoregulation and energy metabolism. Skeletal muscles also produce a large number of different bioactive substances such as myokines, as well as vesicles containing micro ribonucleic acids (microRNAs) and other regulatory molecules that link muscles and other organs[16-19].
Skeletal muscles are predominantly represented by type I and type II fibers (subtypes IIa and IIx), which have biochemical and physiological differences[20-22]. Type I muscle fibers are known as “slow-twitch” fibers because they are characterized by a slower contraction speed but greater resistance to fatigue due to a greater number of mitochondria. Type II muscle fibers are known as “fast-twitch” fibers. Type 2A muscle fibers primarily use oxidative metabolism, and type IIx fibers rely predominantly on glycolysis[23,24]. However, type I fibers express higher levels of the insulin-dependent glucose transporter 4 and glycolytic enzymes as a consequence of which they are highly sensitive to insulin[25-27].
Given the total mass of skeletal muscle, they are a key insulin-dependent consumer of glucose. They account for the utilization of more than 70% of all glucose in the postprandial period[28,29]. In doing so, insulin regulates muscle glucose uptake both by enhancing glucose entry into the cell and by improving delivery to glucose to muscle fibers through improved muscle microcirculation[30,31]. In addition, insulin has an anabolic effect on skeletal muscle by stimulating protein synthesis[32]. In this regard, insulin resistance has a major impact on skeletal muscle structure and function[33,34].
In addition to glucose, skeletal muscles can utilize fatty acids for energy. Fatty acids enter skeletal muscle cells by both passive diffusion and via transfer proteins. Key transfer proteins include fatty acid translocase/CD36, plasma membrane associated fatty acid binding protein, and fatty acid transfer proteins (FATP1-FATP6). Exercise enhances the expression and movement of these transfer proteins, thereby increasing fatty acid uptake and oxidation[35-37]. But excessive dietary fat intake promotes the accumulation of intramyocellular lipids (IMCL) in muscle[38]. IMCL serves as an intracellular energy source during prolonged submaximal exercise. Excessive amounts of IMCL can lead to lipotoxic phenomena by impairing signaling and insulin action, contributing to metabolic disorders. In addition, accumulation of lipids such as diacylglycerol and ceramide play a role in the development of lipid-induced insulin resistance[38-42]. The presence of ceramides in muscle cells activates inflammatory processes, such as nuclear factor kappa B (NF-κB) signaling, and suppresses anabolic processes, including protein kinase B (AKT) and mammalian target of rapamycin (mTOR) signaling. This dual action results in decreased muscle protein synthesis and increased muscle protein breakdown, accelerating sarcopenia[43,44].
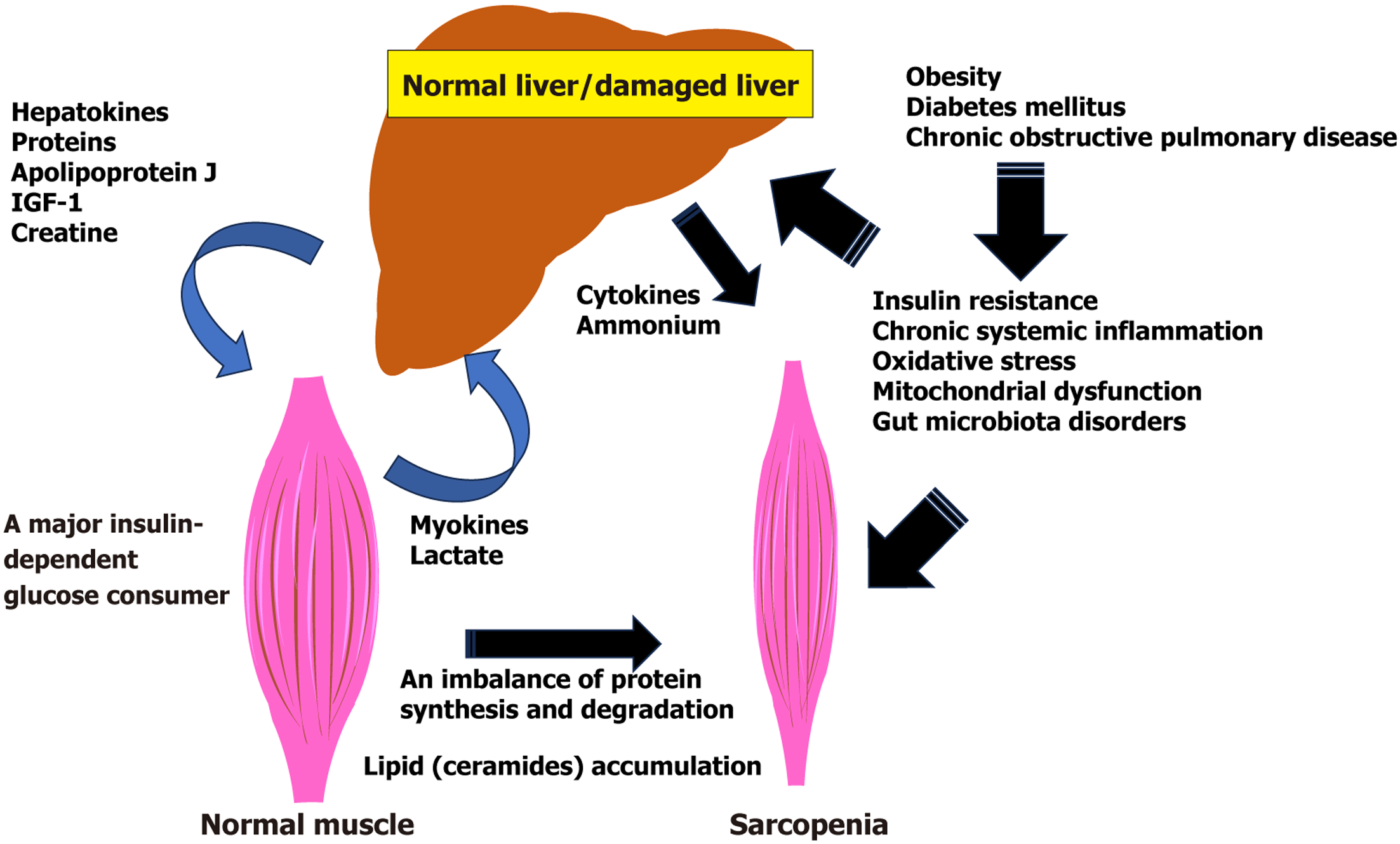
Thus, skeletal muscle is an important metabolic and energy regulator in the body. At the same time, skeletal muscle has numerous metabolic connections with the liver, another important metabolic regulator in the body (Figure 1)[45,46].
Identification and better study of these relationships may improve the understanding of the pathogenesis of sarcopenia in liver disease and enhance its early diagnosis and treatment.
There are two major regulatory mechanisms of skeletal muscle growth. First, the insulin-like growth factor 1 (IGF-1) pathway, which regulates muscle growth through the AKT and mTOR pathways. IGF-1 also stimulates muscle cell proliferation via MAPK and ERK. The second pathway in the regulation of skeletal muscle growth is the myostatin/Smad pathway. Myostatin belongs to the transforming growth factor (TGF)-β superfamily and negatively regulates muscle growth through activation of the myostatin/Smad pathway by inhibiting transcription of muscle regulatory factors that regulate muscle cell differentiation and proliferation. Myostatin also negatively affects muscle growth by inhibiting the AKT/mTOR pathway and thereby inhibiting protein synthesis[47-51].
The imbalance of protein synthesis and degradation is one of the most important mechanisms of sarcopenia. This imbalance is influenced by factors such as endoplasmic reticulum stress and unfolded protein response, which play an important role in muscle atrophy[52-54].
Insulin resistance is an important mechanism associated with the development of sarcopenia. Given the extensive role of skeletal muscles in glucose utilization, insulin resistance leads to impaired glucose uptake and utilization by muscles, impairing the maintenance of their energy balance on the one hand, and on the other hand increasing hyperglycemia and liver burden[55,56]. The importance of impaired carbohydrate metabolism is evidenced by the high prevalence of sarcopenia in patients with diabetes mellitus, which according to various studies is more than 18%[57-59].
Systemic inflammation is another important factor contributing to the development of sarcopenia. Tumor necrosis factor alpha (TNF-α), interleukin-6 (IL-6) and other pro-inflammatory molecules, activate NF-κB in muscle by stimulating proteolysis via the ubiquitin-proteasome system (UPS). On the other hand, muscles are active participants in immune processes through the production of myokines[60-63].
Mitochondrial dysfunction is considered another important mechanism of sarcopenia. Mitochondrial dysfunction leads to impaired oxidative phosphorylation and adenosine triphosphate (ATP) production, which are critical for muscle function and maintenance[64]. Oxidative stress may contribute to the development of mitochondrial dysfunction. Accu
The links between mitochondrial dysfunction and inflammation are of interest. Damaged mitochondria release mitochondrial DNA and other signals that activate inflammatory pathways such as NF-κB, leading to chronic inflammation[70,71]. On the other hand, chronic inflammation can impair mitochondrial function by disrupting mitochondrial biogenesis, leading to further mitochondrial damage and dysfunction. In conditions such as insulin resistance, a vicious cycle occurs in which mitochondrial dysfunction leads to increased lipid accumulation and oxidative stress, which in turn promotes inflammation and exacerbates mitochondrial damage[72-74]. One pathway by which inflammation may affect mitochondrial function in muscle is through nitric oxide (NO) signaling. TNF-α is a potent inducer of inducible NO synthase, which promotes excess NO production, resulting in extensive inhibition of the mitochondrial electron transport chain[75]. TNF-α can also reduce mitochondrial respiration via NF-κB. TNF-α-induced activation of NF-κB decreased promoter transactivation and transcriptional activity of regulators of mitochondrial biogenesis and muscle oxidative phenotype (oxphen)[76]. In this regard, the role of chronic systemic inflammation and its links with sarcopenia is of con
Skeletal muscle and liver are important metabolic and energy regulators. There are direct and indirect functional connections between these organs. Examples of this linkage include creatine, which is synthesized endogenously in the liver, kidney, and pancreas by a two-step process involving the enzymes (1) L-Arginine: Glycine amidinotransferase; and (2) Guanidinoacetate methyltransferase[77,78]. After synthesis, creatine is transported to target cells, including muscle cells, by a specific creatine transporter 1[79]. Creatine and its phosphorylated form, phosphocreatine (PCr), act as an energy buffer. The enzyme creatine kinase catalyzes the reversible phosphorylation of creatine to PCr, which can rapidly recover ATP from ADP during periods of high energy demand. Creatine increases muscle strength and bulk, allowing for faster muscle mass gain and improved muscle performance[80]. Creatine supplementation is being considered as a way to correct sarcopenia in liver cirrhosis[81].
During intense anaerobic exercise, muscles produce lactate as a byproduct of glycolysis because of the high demand for ATP and limited oxygen availability. This lactate accumulates in the muscle and enters the bloodstream[82]. Lactate can be used as an energy substrate by both type I and type II muscle fibers, providing adequate energy levels during exercise. During the Cori cycle, lactate produced by anaerobic glycolysis in muscle is transported to the liver. In the liver, lactate is converted back to glucose via gluconeogenesis[83-85]. This glucose is then released back into the bloodstream and can be taken up by the muscles to be used as an energy source, allowing sustained physical activity[83-85]. The Cori cycle is critical for endurance performance because it helps maintain blood glucose levels during prolonged exercise, preventing hypoglycemia and allowing continuous muscle activity[83-85]. Efficient excretion of lactate and its conversion to glucose in the liver supports sustained aerobic metabolism and slows the onset of fatigue.
Skeletal muscle is an important regulatory organ. Muscles secrete myokines (e.g., irisin, IL-6) that affect liver metabolism by stimulating gluconeogenesis or fat oxidation[86-89]. Skeletal muscle production of IL-6 is stimulated by exercise and muscle loading[90,91]. Short-term exposure to IL-6 is known to enhance insulin-stimulated glucose utilization and fatty acid oxidation in skeletal muscle via AMP-activated protein kinase (AMPK)[92]. In addition, muscle-produced IL-6 can have systemic effects on the liver, adipose tissue, and immune system, as well as provide interactions between intestinal L-cells and pancreatic islets of Langerhans[93].
It is important to note that the role of IL-6 in skeletal muscle is more multifaceted, including those related to negative effects on skeletal muscle, as IL-6 can act in both anti-inflammatory and pro-inflammatory ways. Through its pro-inflammatory action, IL-6 causes muscle atrophy. Continuous administration of IL-6 to muscle tissue has been shown to cause significant muscle atrophy by activating immune receptors and decreasing energy metabolism[94]. Elevated IL-6 Levels are also associated with increased protein ubiquitination and decreased markers of muscle regeneration. This suggests that IL-6 promotes muscle atrophy by enhancing protein degradation pathways, particularly the UPS[95]. In this regard, IL-6 is a promising target for further research.
Irisin has been shown to attenuate liver fibrosis by inhibiting hepatic stellate cell activation and improving mito
The liver in turn produces hepatokines [e.g., fibroblast growth factor 21 (FGF21), angiopoietin-like protein 4 (ANGPTL4)] that regulate energy metabolism in muscle. Regular physical activity increases the secretion of some hepatokines such as FGF21, follistatin, ANGPTL4 and IGF binding protein 1. These hepatokines help exert the positive effects of exercise on whole body metabolism, including increasing insulin sensitivity and reducing the risk of metabolic diseases such as type 2 diabetes and cardiovascular disease[102,103]. In addition, liver-produced apolipoprotein J improves muscle glucose metabolism and insulin sensitivity through mechanisms involving low-density lipoprotein receptor-related protein-2 and the insulin receptor signaling cascade. This interaction is critical for maintaining normal glucose homeostasis and preventing insulin resistance[104]. The altered profile of hepatokine secretion in hepatic steatosis, may cause skeletal muscle insulin resistance and muscle atrophy. This occurs due to increased protein break
Thus, the liver and skeletal muscle have numerous reciprocal relationships based on different metabolic and immune mechanisms. These mechanisms may be impaired in various liver diseases, which is of clinical interest.
Sarcopenia, characterized by loss of muscle mass and strength, is closely related to MASLD, a common liver disease. The relationship between these two conditions is bidirectional and involves several common pathophysiologic mechanisms. Insulin resistance is a major mechanism of both sarcopenia and MASLD. Skeletal muscle plays a key role in glucose utilization, and loss of muscle mass in sarcopenia exacerbates insulin resistance, which in turn contributes to the development of MASLD[109-111]. A sedentary lifestyle contributes to both sarcopenia and MASLD. Lack of physical activity leads to muscle atrophy and impairs insulin resistance, which further promotes fat accumulation in the liver[109,110]. Eating disorders play an important role in both diseases. Excessive dietary fat intake leads to fat accumulation in both skeletal muscle and liver.
Chronic low-intensity inflammation is another common feature of both diseases[109,112]. Inflammatory cytokines contribute to muscle atrophy and liver fibrosis, linking systemic inflammation to the progression of both sarcopenia and MASLD. Oxidative stress is another common mechanism. It causes damage to muscle cells and hepatocytes, leading to muscle atrophy and liver fibrosis[109,113]. Dysregulation of hormones such as myostatin and adiponectin plays an important role in the pathogenesis of sarcopenia and MASLD. Myostatin suppresses muscle growth and adiponectin has anti-inflammatory properties. An imbalance of these hormones can lead to muscle atrophy and fat accumulation in the liver[109].
Disturbances in the composition of the gut microbiota are of growing interest due to its multifaceted role in many immune and metabolic processes not only in the gut but also in other organs through a number of bioactive substances such as short-chain fatty acids (SCFAs). SCFAs, including acetate, propionate and butyrate, are produced by the intestinal microbiota as a result of fermentation of dietary fiber in the colon. SCFAs serve as a direct source of energy for muscle cells and influence glucose metabolism. SCFAs enhance insulin sensitivity in skeletal muscle by increasing histone acetylation, which promotes glucose uptake. Acetate improves mitochondrial function, which is critical for energy production and muscle function[114]. Butyrate reduces inflammation in skeletal muscle by decreasing the expression of inflammatory mediators such as IL-6, monocyte chemoattractant protein-1, and C-C motif chemokine ligand 5. This effect is mediated by inhibition of NF-κB and signal transducer and activator of transcription 3 signaling pathways[115,116]. SCFAs activate AMPK, promoting fatty acid uptake, lipid catabolism, and mitochondrial biogenesis, thereby inhibiting lipid anabolism in skeletal muscle[117-119]. SCFAs also act in an anti-inflammatory manner[114]. Transplantation of gut microbiota from healthy mice to mice reared without microbes has been shown to improve muscle mass and muscle function, indicating a critical role for gut microbiota in muscle health[120]. The connection between the gut and liver, involving the gut microbiota and their metabolites, affects both muscle and liver health. Dysbiosis can lead to increased gut permeability, systemic inflammation and subsequent muscle and liver damage. Disturbances in the gut microbiota are considered an important contributor to the development of MASLD, for example through increased bacterial lipo
The bacterium Akkermansia muciniphila (A. muciniphila) has been shown to play an important role in intestinal and liver function. In patients with cirrhosis, lower levels of A. muciniphila are associated with sarcopenia, a condition characterized by decreased muscle mass and function. This bacterium is part of the gut microbiota that supports physical activity and muscle health[124,125]. Supplementation of A. muciniphila in mice has been shown to reduce metabolic inflammation and improve muscle function by reducing endoplasmic reticulum stress and inflammation in muscle tissues[126].
The pathogenesis of sarcopenia in cirrhosis is also multifactorial and involves several interrelated mechanisms. Altered metabolism, decreased protein synthesis, and increased protein degradation are important contributors to sarcopenia in cirrhosis. Cirrhosis leads to a catabolic state in which muscle protein synthesis is decreased and protein degradation is increased[127,128]. This imbalance is caused by metabolic changes including hyperammonemia and chronic inflammation[127]. As previously mentioned, IGF-1, is a peptide hormone that is mainly produced in the liver and plays a critical role in growth, metabolism and cellular functions such as proliferation, differentiation and apoptosis. In liver cirrhosis, IGF-1 Levels are significantly decreased, which affects disease progression and patient prognosis[129-131].
A key factor contributing to sarcopenia in patients with chronic liver disease may be the hyperammonemia-induced increased production of myostatin, which causes muscle atrophy through the expression of atrophy-related genes[132]. Ammonia, which is produced during protein catabolism and is an important component of nucleic acid and protein biosynthesis, is mainly neutralized in the liver. In patients with liver disease, especially cirrhosis, insufficient detoxification of ammonia leads to an increase in its concentration in the blood, resulting in severe skeletal muscle atrophy, increased apoptosis and decreased protein synthesis. Ammonia induces the expression of myostatin, a negative regulator of muscle growth, and promotes autophagy, leading to muscle atrophy[132-134]. In liver cirrhosis, skeletal muscle replaces the liver as the primary ammonia excreting organ. Thus, a vicious cycle occurs in which hyperammonemia causes severe muscle damage and sarcopenia, which in turn limits the muscle's ability to excrete excess ammonia from the blood[134]. The use of the hypoammonemic agent L-ornithine-L-aspartate in the treatment of sarcopenia in cirrhosis has the potential for positive effects on muscle mass, strength, and function, given the pathogenetic basis of sarcopenia in cirrhosis[134,135].
Liver cirrhosis is also associated with decreased levels of testosterone and growth factors, which are essential for the maintenance and growth of muscle mass[7,136]. Cirrhosis suppresses the hypothalamic-pituitary-gonadal axis, resulting in hypogonadism, high estrogen levels, and decreased testosterone levels[137]. Testosterone, a key anabolic hormone, plays a critical role in the maintenance of muscle mass, and its decline with age is associated with the development and progression of sarcopenia. Testosterone interacts with androgen receptors in muscle cells, affecting muscle metabolism and promoting muscle growth. Its deficiency can lead to muscle atrophy and weakness, characteristic features of sarcopenia[138-140]. In a recent review, decreased testosterone levels have been shown to be an important factor in the development of sarcopenia in cirrhosis[141].
Dietary restrictions, loss of appetite, absorption disorders, and metabolic alterations in patients with cirrhosis result in inadequate intake of calories and protein needed to maintain muscle mass[128,142].
Persistent inflammation in cirrhosis also contributes to muscle breakdown through activation of proteolytic pathways[127,132]. Increased ROS in cirrhosis leads to mitochondrial dysfunction and further muscle protein degradation[133].
Alterations in the gut microbiota of patients with cirrhosis may exacerbate inflammation and metabolic disorders, contributing to sarcopenia[143]. Patients with cirrhosis have a significant decrease in the alpha diversity of the gut mic
Thus, the metabolic and immune connections between the liver and skeletal muscles are impaired in liver diseases such as MASLD and cirrhosis, entailing the development and progression of sarcopenia. The severity of sarcopenia may indicate the severity of liver disease.
Thus, sarcopenia, characterized by progressive loss of muscle mass and function, is a serious clinical problem. The presence of sarcopenia in liver disease is a natural outcome of impaired multiple bilateral metabolic and immune con
| 1. | Cruz-Jentoft AJ, Bahat G, Bauer J, Boirie Y, Bruyère O, Cederholm T, Cooper C, Landi F, Rolland Y, Sayer AA, Schneider SM, Sieber CC, Topinkova E, Vandewoude M, Visser M, Zamboni M; Writing Group for the European Working Group on Sarcopenia in Older People 2 (EWGSOP2), and the Extended Group for EWGSOP2. Sarcopenia: revised European consensus on definition and diagnosis. Age Ageing. 2019;48:16-31. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 6646] [Cited by in RCA: 7801] [Article Influence: 1300.2] [Reference Citation Analysis (1)] |
| 2. | Wu J, Ding P, Wu H, Yang P, Guo H, Tian Y, Meng L, Zhao Q. Sarcopenia: Molecular regulatory network for loss of muscle mass and function. Front Nutr. 2023;10:1037200. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in RCA: 26] [Reference Citation Analysis (0)] |
| 3. | Wiedmer P, Jung T, Castro JP, Pomatto LCD, Sun PY, Davies KJA, Grune T. Sarcopenia - Molecular mechanisms and open questions. Ageing Res Rev. 2021;65:101200. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 55] [Cited by in RCA: 249] [Article Influence: 62.3] [Reference Citation Analysis (0)] |
| 4. | Choi Y, Kim D, Kim SK. Effects of Physical Activity on Body Composition, Muscle Strength, and Physical Function in Old Age: Bibliometric and Meta-Analyses. Healthcare (Basel). 2024;12:197. [RCA] [PubMed] [DOI] [Full Text] [Cited by in RCA: 7] [Reference Citation Analysis (0)] |
| 5. | Forbes GB. Longitudinal changes in adult fat-free mass: influence of body weight. Am J Clin Nutr. 1999;70:1025-1031. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 105] [Cited by in RCA: 102] [Article Influence: 3.9] [Reference Citation Analysis (0)] |
| 6. | Rogeri PS, Zanella R Jr, Martins GL, Garcia MDA, Leite G, Lugaresi R, Gasparini SO, Sperandio GA, Ferreira LHB, Souza-Junior TP, Lancha AH Jr. Strategies to Prevent Sarcopenia in the Aging Process: Role of Protein Intake and Exercise. Nutrients. 2021;14:52. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 6] [Cited by in RCA: 78] [Article Influence: 19.5] [Reference Citation Analysis (0)] |
| 7. | Geladari E, Alexopoulos T, Kontogianni MD, Vasilieva L, Mani I, Alexopoulou A. Mechanisms of sarcopenia in liver cirrhosis and the role of myokines. Ann Gastroenterol. 2023;36:392-404. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in RCA: 6] [Reference Citation Analysis (0)] |
| 8. | Choi YJ, Kim T, Park HJ, Cho JH, Byun MK. Long-Term Clinical Outcomes of Patients with Chronic Obstructive Pulmonary Disease with Sarcopenia. Life (Basel). 2023;13:1628. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 1] [Cited by in RCA: 7] [Article Influence: 3.5] [Reference Citation Analysis (0)] |
| 9. | Costa TM, Costa FM, Moreira CA, Rabelo LM, Boguszewski CL, Borba VZ. Sarcopenia in COPD: relationship with COPD severity and prognosis. J Bras Pneumol. 2015;41:415-421. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 74] [Cited by in RCA: 83] [Article Influence: 8.3] [Reference Citation Analysis (0)] |
| 10. | Liang QY, Wang J, Yang YF, Zhao K, Luo RL, Tian Y, Li FX. Machine learning to identify potential biomarkers for sarcopenia in liver cirrhosis. World J Hepatol. 2025;17:105332. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in RCA: 1] [Reference Citation Analysis (0)] |
| 11. | Moctezuma-Velázquez C, Low G, Mourtzakis M, Ma M, Burak KW, Tandon P, Montano-Loza AJ. Association between Low Testosterone Levels and Sarcopenia in Cirrhosis: A Cross-sectional Study. Ann Hepatol. 2018;17:615-623. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 26] [Cited by in RCA: 44] [Article Influence: 6.3] [Reference Citation Analysis (0)] |
| 12. | Boga S, Yildirim AE, Ucbilek E, Koksal AR, Sisman ST, Durak I, Sen I, Dogu B, Serin E, Ucbilek AB, Yildirim MO, Erturk SM, Alkim H, Alkim C. The effect of sarcopenia and serum myokines on prognosis and survival in cirrhotic patients: a multicenter cross-sectional study. Eur J Gastroenterol Hepatol. 2022;34:1261-1268. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 2] [Cited by in RCA: 8] [Article Influence: 2.7] [Reference Citation Analysis (1)] |
| 13. | Alexopoulos T, Vasilieva L, Kontogianni MD, Tenta R, Georgiou A, Stroumpouli E, Mani I, Alexopoulou A. Myostatin in combination with creatine phosphokinase or albumin may differentiate patients with cirrhosis and sarcopenia. Am J Physiol Gastrointest Liver Physiol. 2021;321:G543-G551. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 6] [Cited by in RCA: 22] [Article Influence: 5.5] [Reference Citation Analysis (0)] |
| 14. | Shibamoto A, Namisaki T, Suzuki J, Kubo T, Iwai S, Tomooka F, Takeda S, Fujimoto Y, Enomoto M, Murata K, Inoue T, Tsuji Y, Fujinaga Y, Nishimura N, Kitagawa K, Takaya H, Kaji K, Kawaratani H, Akahane T, Mitoro A, Yoshiji H. Hemoglobin levels as a surrogate marker of sarcopenia in patients with liver cirrhosis. Hepatol Res. 2023;53:713-722. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 4] [Cited by in RCA: 6] [Article Influence: 3.0] [Reference Citation Analysis (0)] |
| 15. | Shibamoto A, Namisaki T, Suzuki J, Kubo T, Iwai S, Tomooka F, Takeda S, Fujimoto Y, Inoue T, Tanaka M, Koizumi A, Yorioka N, Matsuda T, Asada S, Tsuji Y, Fujinaga Y, Nishimura N, Sato S, Takaya H, Kitagawa K, Kaji K, Kawaratani H, Akahane T, Mitoro A, Yoshiji H. Hemoglobin and Endotoxin Levels Predict Sarcopenia Occurrence in Patients with Alcoholic Cirrhosis. Diagnostics (Basel). 2023;13:2218. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 3] [Cited by in RCA: 5] [Article Influence: 2.5] [Reference Citation Analysis (0)] |
| 16. | Jia J, Wang L, Zhou Y, Zhang P, Chen X. Muscle-derived extracellular vesicles mediate crosstalk between skeletal muscle and other organs. Front Physiol. 2024;15:1501957. [RCA] [PubMed] [DOI] [Full Text] [Cited by in RCA: 3] [Reference Citation Analysis (0)] |
| 17. | Hoffmann C, Weigert C. Skeletal Muscle as an Endocrine Organ: The Role of Myokines in Exercise Adaptations. Cold Spring Harb Perspect Med. 2017;7:a029793. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 140] [Cited by in RCA: 233] [Article Influence: 29.1] [Reference Citation Analysis (0)] |
| 18. | Severinsen MCK, Pedersen BK. Muscle-Organ Crosstalk: The Emerging Roles of Myokines. Endocr Rev. 2020;41:594-609. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 243] [Cited by in RCA: 617] [Article Influence: 123.4] [Reference Citation Analysis (0)] |
| 19. | Ma S, Xing X, Huang H, Gao X, Xu X, Yang J, Liao C, Zhang X, Liu J, Tian W, Liao L. Skeletal muscle-derived extracellular vesicles transport glycolytic enzymes to mediate muscle-to-bone crosstalk. Cell Metab. 2023;35:2028-2043.e7. [RCA] [PubMed] [DOI] [Full Text] [Cited by in RCA: 46] [Reference Citation Analysis (0)] |
| 20. | Bourdeau Julien I, Sephton CF, Dutchak PA. Metabolic Networks Influencing Skeletal Muscle Fiber Composition. Front Cell Dev Biol. 2018;6:125. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 26] [Cited by in RCA: 53] [Article Influence: 7.6] [Reference Citation Analysis (0)] |
| 21. | Scott W, Stevens J, Binder-Macleod SA. Human skeletal muscle fiber type classifications. Phys Ther. 2001;81:1810-6. [PubMed] |
| 22. | Murgia M, Nogara L, Baraldo M, Reggiani C, Mann M, Schiaffino S. Protein profile of fiber types in human skeletal muscle: a single-fiber proteomics study. Skelet Muscle. 2021;11:24. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 63] [Cited by in RCA: 95] [Article Influence: 23.8] [Reference Citation Analysis (0)] |
| 23. | Wang Y, Pessin JE. Mechanisms for fiber-type specificity of skeletal muscle atrophy. Curr Opin Clin Nutr Metab Care. 2013;16:243-250. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 263] [Cited by in RCA: 328] [Article Influence: 27.3] [Reference Citation Analysis (0)] |
| 24. | Talbot J, Maves L. Skeletal muscle fiber type: using insights from muscle developmental biology to dissect targets for susceptibility and resistance to muscle disease. Wiley Interdiscip Rev Dev Biol. 2016;5:518-534. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 161] [Cited by in RCA: 270] [Article Influence: 30.0] [Reference Citation Analysis (0)] |
| 25. | Garvey WT, Maianu L, Hancock JA, Golichowski AM, Baron A. Gene expression of GLUT4 in skeletal muscle from insulin-resistant patients with obesity, IGT, GDM, and NIDDM. Diabetes. 1992;41:465-475. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 148] [Cited by in RCA: 152] [Article Influence: 4.6] [Reference Citation Analysis (0)] |
| 26. | Sylow L, Tokarz VL, Richter EA, Klip A. The many actions of insulin in skeletal muscle, the paramount tissue determining glycemia. Cell Metab. 2021;33:758-780. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 56] [Cited by in RCA: 188] [Article Influence: 47.0] [Reference Citation Analysis (0)] |
| 27. | Albers PH, Pedersen AJ, Birk JB, Kristensen DE, Vind BF, Baba O, Nøhr J, Højlund K, Wojtaszewski JF. Human muscle fiber type-specific insulin signaling: impact of obesity and type 2 diabetes. Diabetes. 2015;64:485-497. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 126] [Cited by in RCA: 158] [Article Influence: 15.8] [Reference Citation Analysis (0)] |
| 28. | Jensen J, Rustad PI, Kolnes AJ, Lai YC. The role of skeletal muscle glycogen breakdown for regulation of insulin sensitivity by exercise. Front Physiol. 2011;2:112. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 198] [Cited by in RCA: 267] [Article Influence: 19.1] [Reference Citation Analysis (0)] |
| 29. | Baron AD, Brechtel G, Wallace P, Edelman SV. Rates and tissue sites of non-insulin- and insulin-mediated glucose uptake in humans. Am J Physiol. 1988;255:E769-E774. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 144] [Cited by in RCA: 294] [Article Influence: 7.9] [Reference Citation Analysis (0)] |
| 30. | Meijer RI, Serné EH, Korkmaz HI, van der Peet DL, de Boer MP, Niessen HW, van Hinsbergh VW, Yudkin JS, Smulders YM, Eringa EC. Insulin-induced changes in skeletal muscle microvascular perfusion are dependent upon perivascular adipose tissue in women. Diabetologia. 2015;58:1907-1915. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 38] [Cited by in RCA: 42] [Article Influence: 4.2] [Reference Citation Analysis (0)] |
| 31. | Keske MA, Premilovac D, Bradley EA, Dwyer RM, Richards SM, Rattigan S. Muscle microvascular blood flow responses in insulin resistance and ageing. J Physiol. 2016;594:2223-2231. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 43] [Cited by in RCA: 55] [Article Influence: 5.5] [Reference Citation Analysis (0)] |
| 32. | Bennet WM, Rennie MJ. Protein anabolic actions of insulin in the human body. Diabet Med. 1991;8:199-207. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 17] [Cited by in RCA: 16] [Article Influence: 0.5] [Reference Citation Analysis (0)] |
| 33. | Abdul-Ghani MA, DeFronzo RA. Pathogenesis of insulin resistance in skeletal muscle. J Biomed Biotechnol. 2010;2010:476279. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 292] [Cited by in RCA: 403] [Article Influence: 26.9] [Reference Citation Analysis (0)] |
| 34. | Marušić M, Paić M, Knobloch M, Liberati Pršo AM. NAFLD, Insulin Resistance, and Diabetes Mellitus Type 2. Can J Gastroenterol Hepatol. 2021;2021:6613827. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 17] [Cited by in RCA: 91] [Article Influence: 22.8] [Reference Citation Analysis (2)] |
| 35. | Harasim E, Kalinowska A, Chabowski A, Stepek T. [The role of fatty-acid transport proteins (FAT/CD36, FABPpm, FATP) in lipid metabolism in skeletal muscles]. Postepy Hig Med Dosw (Online). 2008;62:433-441. [PubMed] |
| 36. | Turcotte LP, Fisher JS. Skeletal muscle insulin resistance: roles of fatty acid metabolism and exercise. Phys Ther. 2008;88:1279-1296. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 110] [Cited by in RCA: 135] [Article Influence: 7.9] [Reference Citation Analysis (0)] |
| 37. | Kiens B, Roepstorff C. Utilization of long-chain fatty acids in human skeletal muscle during exercise. Acta Physiol Scand. 2003;178:391-396. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 12] [Cited by in RCA: 12] [Article Influence: 0.5] [Reference Citation Analysis (0)] |
| 38. | Umek N, Horvat S, Cvetko E. Skeletal muscle and fiber type-specific intramyocellular lipid accumulation in obese mice. Bosn J Basic Med Sci. 2021;21:730-738. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 6] [Cited by in RCA: 21] [Article Influence: 5.3] [Reference Citation Analysis (0)] |
| 39. | Corpeleijn E, Saris WH, Blaak EE. Metabolic flexibility in the development of insulin resistance and type 2 diabetes: effects of lifestyle. Obes Rev. 2009;10:178-193. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 164] [Cited by in RCA: 168] [Article Influence: 10.5] [Reference Citation Analysis (0)] |
| 40. | Bosma M, Kersten S, Hesselink MK, Schrauwen P. Re-evaluating lipotoxic triggers in skeletal muscle: relating intramyocellular lipid metabolism to insulin sensitivity. Prog Lipid Res. 2012;51:36-49. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 93] [Cited by in RCA: 99] [Article Influence: 7.6] [Reference Citation Analysis (0)] |
| 41. | Schrauwen P. High-fat diet, muscular lipotoxicity and insulin resistance. Proc Nutr Soc. 2007;66:33-41. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 82] [Cited by in RCA: 97] [Article Influence: 5.4] [Reference Citation Analysis (0)] |
| 42. | van Loon LJ, Goodpaster BH. Increased intramuscular lipid storage in the insulin-resistant and endurance-trained state. Pflugers Arch. 2006;451:606-616. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 153] [Cited by in RCA: 161] [Article Influence: 8.1] [Reference Citation Analysis (0)] |
| 43. | Rivas DA, McDonald DJ, Rice NP, Haran PH, Dolnikowski GG, Fielding RA. Diminished anabolic signaling response to insulin induced by intramuscular lipid accumulation is associated with inflammation in aging but not obesity. Am J Physiol Regul Integr Comp Physiol. 2016;310:R561-R569. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 67] [Cited by in RCA: 89] [Article Influence: 9.9] [Reference Citation Analysis (0)] |
| 44. | Rivas DA, Morris EP, Haran PH, Pasha EP, Morais Mda S, Dolnikowski GG, Phillips EM, Fielding RA. Increased ceramide content and NFκB signaling may contribute to the attenuation of anabolic signaling after resistance exercise in aged males. J Appl Physiol (1985). 2012;113:1727-1736. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 68] [Cited by in RCA: 78] [Article Influence: 6.0] [Reference Citation Analysis (0)] |
| 45. | Chakravarthy MV, Siddiqui MS, Forsgren MF, Sanyal AJ. Harnessing Muscle-Liver Crosstalk to Treat Nonalcoholic Steatohepatitis. Front Endocrinol (Lausanne). 2020;11:592373. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 37] [Cited by in RCA: 57] [Article Influence: 11.4] [Reference Citation Analysis (0)] |
| 46. | Chen C, Xie L, Zhang M, Shama, Cheng KKY, Jia W. The interplay between the muscle and liver in the regulation of glucolipid metabolism. J Mol Cell Biol. 2024;15:mjad073. [RCA] [PubMed] [DOI] [Full Text] [Cited by in RCA: 7] [Reference Citation Analysis (0)] |
| 47. | Watts R, McAinch AJ, Dixon JB, O'Brien PE, Cameron-Smith D. Increased Smad signaling and reduced MRF expression in skeletal muscle from obese subjects. Obesity (Silver Spring). 2013;21:525-528. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 33] [Cited by in RCA: 35] [Article Influence: 2.9] [Reference Citation Analysis (0)] |
| 48. | Yoshida T, Delafontaine P. Mechanisms of IGF-1-Mediated Regulation of Skeletal Muscle Hypertrophy and Atrophy. Cells. 2020;9:1970. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 83] [Cited by in RCA: 381] [Article Influence: 76.2] [Reference Citation Analysis (0)] |
| 49. | Miramontes E, Mozdziak P, Petitte JN, Kulus M, Wieczorkiewicz M, Kempisty B. Skeletal Muscle and the Effects of Ammonia Toxicity in Fish, Mammalian, and Avian Species: A Comparative Review Based on Molecular Research. Int J Mol Sci. 2020;21:4641. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 5] [Cited by in RCA: 10] [Article Influence: 2.0] [Reference Citation Analysis (0)] |
| 50. | Timmer LT, Hoogaars WMH, Jaspers RT. The Role of IGF-1 Signaling in Skeletal Muscle Atrophy. Adv Exp Med Biol. 2018;1088:109-137. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 42] [Cited by in RCA: 60] [Article Influence: 8.6] [Reference Citation Analysis (0)] |
| 51. | Carnac G, Vernus B, Bonnieu A. Myostatin in the pathophysiology of skeletal muscle. Curr Genomics. 2007;8:415-422. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 39] [Cited by in RCA: 68] [Article Influence: 4.9] [Reference Citation Analysis (0)] |
| 52. | Wang Y, Le WD. Autophagy and Ubiquitin-Proteasome System. Adv Exp Med Biol. 2019;1206:527-550. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 52] [Cited by in RCA: 129] [Article Influence: 21.5] [Reference Citation Analysis (0)] |
| 53. | Kim IY, Park S, Jang J, Wolfe RR. Understanding Muscle Protein Dynamics: Technical Considerations for Advancing Sarcopenia Research. Ann Geriatr Med Res. 2020;24:157-165. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 18] [Cited by in RCA: 29] [Article Influence: 5.8] [Reference Citation Analysis (0)] |
| 54. | Barclay RD, Burd NA, Tyler C, Tillin NA, Mackenzie RW. The Role of the IGF-1 Signaling Cascade in Muscle Protein Synthesis and Anabolic Resistance in Aging Skeletal Muscle. Front Nutr. 2019;6:146. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 51] [Cited by in RCA: 101] [Article Influence: 16.8] [Reference Citation Analysis (0)] |
| 55. | Cleasby ME, Jamieson PM, Atherton PJ. Insulin resistance and sarcopenia: mechanistic links between common co-morbidities. J Endocrinol. 2016;229:R67-R81. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 286] [Cited by in RCA: 386] [Article Influence: 42.9] [Reference Citation Analysis (0)] |
| 56. | Widajanti N, Srinowati H, Firdausi H, Thaha M. The Relationship between Insulin Resistance and the Degree of Sarcopenia in Elderly Patients on Maintenance Hemodialysis. J Nat Sc Biol Med. 2021;12:338. [DOI] [Full Text] |
| 57. | Du Y, Wang Y, Zhang P, Zhong X, Pan T. Analysis of Risk Factors for the Association of Sarcopenia in Patients with Type 2 Diabetes Mellitus. Diabetes Metab Syndr Obes. 2024;17:1455-1466. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in RCA: 1] [Reference Citation Analysis (0)] |
| 58. | Feng L, Gao Q, Hu K, Wu M, Wang Z, Chen F, Mei F, Zhao L, Ma B. Prevalence and Risk Factors of Sarcopenia in Patients With Diabetes: A Meta-analysis. J Clin Endocrinol Metab. 2022;107:1470-1483. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 8] [Cited by in RCA: 105] [Article Influence: 35.0] [Reference Citation Analysis (0)] |
| 59. | Ai Y, Xu R, Liu L. The prevalence and risk factors of sarcopenia in patients with type 2 diabetes mellitus: a systematic review and meta-analysis. Diabetol Metab Syndr. 2021;13:93. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 18] [Cited by in RCA: 53] [Article Influence: 13.3] [Reference Citation Analysis (0)] |
| 60. | Wang T. Searching for the link between inflammaging and sarcopenia. Ageing Res Rev. 2022;77:101611. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 7] [Cited by in RCA: 51] [Article Influence: 17.0] [Reference Citation Analysis (0)] |
| 61. | Dalle S, Rossmeislova L, Koppo K. The Role of Inflammation in Age-Related Sarcopenia. Front Physiol. 2017;8:1045. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 371] [Cited by in RCA: 449] [Article Influence: 56.1] [Reference Citation Analysis (0)] |
| 62. | Xie S, Wu Q. Association between the systemic immune-inflammation index and sarcopenia: a systematic review and meta-analysis. J Orthop Surg Res. 2024;19:314. [RCA] [PubMed] [DOI] [Full Text] [Cited by in RCA: 10] [Reference Citation Analysis (0)] |
| 63. | Wang J, Xiang Y, Wu L, Zhang C, Han B, Cheng Y, Tong Y, Yan D, Wang L. The association between inflammatory cytokines and sarcopenia-related traits: a bi-directional Mendelian randomization study. Eur J Clin Nutr. 2024;78:1032-1040. [RCA] [PubMed] [DOI] [Full Text] [Cited by in RCA: 4] [Reference Citation Analysis (0)] |
| 64. | Brunetta HS, Holwerda AM, van Loon LJC, Holloway GP. Mitochondrial ROS and Aging: Understanding Exercise as a Preventive Tool. J of Sci in Sport and Exercise. 2020;2:15-24. [DOI] [Full Text] |
| 65. | Kamarulzaman NT, Makpol S. The link between Mitochondria and Sarcopenia. J Physiol Biochem. 2025;81:1-20. [RCA] [PubMed] [DOI] [Full Text] [Cited by in RCA: 2] [Reference Citation Analysis (0)] |
| 66. | Ferri E, Marzetti E, Calvani R, Picca A, Cesari M, Arosio B. Role of Age-Related Mitochondrial Dysfunction in Sarcopenia. Int J Mol Sci. 2020;21:5236. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 29] [Cited by in RCA: 114] [Article Influence: 22.8] [Reference Citation Analysis (0)] |
| 67. | Bellanti F, Lo Buglio A, Vendemiale G. Mitochondrial Impairment in Sarcopenia. Biology (Basel). 2021;10:31. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 46] [Cited by in RCA: 49] [Article Influence: 12.3] [Reference Citation Analysis (0)] |
| 68. | Chen X, Ji Y, Liu R, Zhu X, Wang K, Yang X, Liu B, Gao Z, Huang Y, Shen Y, Liu H, Sun H. Mitochondrial dysfunction: roles in skeletal muscle atrophy. J Transl Med. 2023;21:503. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in RCA: 116] [Reference Citation Analysis (0)] |
| 69. | Coen PM, Musci RV, Hinkley JM, Miller BF. Mitochondria as a Target for Mitigating Sarcopenia. Front Physiol. 2018;9:1883. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 58] [Cited by in RCA: 108] [Article Influence: 18.0] [Reference Citation Analysis (0)] |
| 70. | Irazoki A, Gordaliza-Alaguero I, Frank E, Giakoumakis NN, Seco J, Palacín M, Gumà A, Sylow L, Sebastián D, Zorzano A. Disruption of mitochondrial dynamics triggers muscle inflammation through interorganellar contacts and mitochondrial DNA mislocation. Nat Commun. 2023;14:108. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in RCA: 46] [Reference Citation Analysis (0)] |
| 71. | Zampino M, Brennan NA, Kuo PL, Spencer RG, Fishbein KW, Simonsick EM, Ferrucci L. Poor mitochondrial health and systemic inflammation? Test of a classic hypothesis in the Baltimore Longitudinal Study of Aging. Geroscience. 2020;42:1175-1182. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 11] [Cited by in RCA: 28] [Article Influence: 5.6] [Reference Citation Analysis (0)] |
| 72. | Li Y, Guo W, Li H, Wang Y, Liu X, Kong W. The Change of Skeletal Muscle Caused by Inflammation in Obesity as the Key Path to Fibrosis: Thoughts on Mechanisms and Intervention Strategies. Biomolecules. 2024;15:20. [RCA] [PubMed] [DOI] [Full Text] [Cited by in RCA: 2] [Reference Citation Analysis (0)] |
| 73. | Passaro A, Sanz JM, Naumovski N, Sergi D. The complex interplay between oxinflammation, mitochondrial dysfunction and lipotoxicity: Focus on their role in the pathogenesis of skeletal muscle insulin resistance and modulation by dietary fatty acids. Adv Redox Res. 2024;11:100100. [DOI] [Full Text] |
| 74. | Warfel JD, Bermudez EM, Mendoza TM, Ghosh S, Zhang J, Elks CM, Mynatt R, Vandanmagsar B. Mitochondrial fat oxidation is essential for lipid-induced inflammation in skeletal muscle in mice. Sci Rep. 2016;6:37941. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 24] [Cited by in RCA: 35] [Article Influence: 3.9] [Reference Citation Analysis (0)] |
| 75. | Marzetti E, Calvani R, Cesari M, Buford TW, Lorenzi M, Behnke BJ, Leeuwenburgh C. Mitochondrial dysfunction and sarcopenia of aging: from signaling pathways to clinical trials. Int J Biochem Cell Biol. 2013;45:2288-2301. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 439] [Cited by in RCA: 436] [Article Influence: 36.3] [Reference Citation Analysis (0)] |
| 76. | Remels AH, Gosker HR, Schrauwen P, Hommelberg PP, Sliwinski P, Polkey M, Galdiz J, Wouters EF, Langen RC, Schols AM. TNF-alpha impairs regulation of muscle oxidative phenotype: implications for cachexia? FASEB J. 2010;24:5052-5062. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 73] [Cited by in RCA: 85] [Article Influence: 5.7] [Reference Citation Analysis (0)] |
| 77. | Ireland Z, Russell AP, Wallimann T, Walker DW, Snow R. Developmental changes in the expression of creatine synthesizing enzymes and creatine transporter in a precocial rodent, the spiny mouse. BMC Dev Biol. 2009;9:39. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 48] [Cited by in RCA: 56] [Article Influence: 3.5] [Reference Citation Analysis (0)] |
| 78. | Barcelos RP, Stefanello ST, Mauriz JL, Gonzalez-Gallego J, Soares FA. Creatine and the Liver: Metabolism and Possible Interactions. Mini Rev Med Chem. 2016;16:12-18. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 36] [Cited by in RCA: 55] [Article Influence: 5.5] [Reference Citation Analysis (0)] |
| 79. | Balestrino M, Gandolfo C, Perasso L. Controlling the Flow of Energy: Inhibition and Stimulation of the Creatine Transporter. Curr Enzym Inhib. 2009;5:223-233. [RCA] [DOI] [Full Text] [Cited by in Crossref: 5] [Cited by in RCA: 6] [Article Influence: 0.4] [Reference Citation Analysis (0)] |
| 80. | Kraemer WJ, Beeler MK, Post EM, Luk H, Lombard JR, Dunn-Lewis C, Volek JS. Physiological Basis for Creatine Supplementation in Skeletal Muscle and the Central Nervous System. In: Bagchi D, Nair S, Sen CK, editors. Nutrition and Enhanced Sports Performance. Netherlands: Elsevier, 2019. [DOI] [Full Text] |
| 81. | Casciola R, Leoni L, Cuffari B, Pecchini M, Menozzi R, Colecchia A, Ravaioli F. Creatine Supplementation to Improve Sarcopenia in Chronic Liver Disease: Facts and Perspectives. Nutrients. 2023;15:863. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 14] [Cited by in RCA: 14] [Article Influence: 7.0] [Reference Citation Analysis (0)] |
| 82. | Nalbandian M, Takeda M. Lactate as a Signaling Molecule That Regulates Exercise-Induced Adaptations. Biology (Basel). 2016;5:38. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 86] [Cited by in RCA: 100] [Article Influence: 11.1] [Reference Citation Analysis (0)] |
| 83. | Manoj KM, Nirusimhan V, Parashar A, Edward J, Gideon DA. Murburn precepts for lactic-acidosis, Cori cycle, and Warburg effect: Interactive dynamics of dehydrogenases, protons, and oxygen. J Cell Physiol. 2022;237:1902-1922. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 3] [Cited by in RCA: 18] [Article Influence: 4.5] [Reference Citation Analysis (0)] |
| 84. | Rubin RP. Carl and Gerty Cori: A collaboration that changed the face of biochemistry. J Med Biogr. 2021;29:143-148. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 7] [Cited by in RCA: 12] [Article Influence: 3.0] [Reference Citation Analysis (0)] |
| 85. | Horikoshi M, Harada K, Tsuno S, Kitaguchi T, Hirai MY, Matsumoto M, Terada S, Tsuboi T. Distinct lactate metabolism between hepatocytes and myotubes revealed by live cell imaging with genetically encoded indicators. Biochem Biophys Res Commun. 2024;694:149416. [RCA] [PubMed] [DOI] [Full Text] [Cited by in RCA: 3] [Reference Citation Analysis (0)] |
| 86. | Luo J, Pu Q, Wu X. Recent Advances of Exosomes Derived from Skeletal Muscle and Crosstalk with Other Tissues. Int J Mol Sci. 2024;25:10877. [RCA] [PubMed] [DOI] [Full Text] [Cited by in RCA: 5] [Reference Citation Analysis (0)] |
| 87. | Guo L, Quan M, Pang W, Yin Y, Li F. Cytokines and exosomal miRNAs in skeletal muscle-adipose crosstalk. Trends Endocrinol Metab. 2023;34:666-681. [RCA] [PubMed] [DOI] [Full Text] [Cited by in RCA: 17] [Reference Citation Analysis (0)] |
| 88. | Ni P, Yang L, Li F. Exercise-derived skeletal myogenic exosomes as mediators of intercellular crosstalk: a major player in health, disease, and exercise. J Physiol Biochem. 2023;79:501-510. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 11] [Cited by in RCA: 13] [Article Influence: 6.5] [Reference Citation Analysis (0)] |
| 89. | Mytidou C, Koutsoulidou A, Katsioloudi A, Prokopi M, Kapnisis K, Michailidou K, Anayiotos A, Phylactou LA. Muscle-derived exosomes encapsulate myomiRs and are involved in local skeletal muscle tissue communication. FASEB J. 2021;35:e21279. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 63] [Cited by in RCA: 55] [Article Influence: 13.8] [Reference Citation Analysis (0)] |
| 90. | Wu H, Ballantyne CM. Skeletal muscle inflammation and insulin resistance in obesity. J Clin Invest. 2017;127:43-54. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 279] [Cited by in RCA: 470] [Article Influence: 58.8] [Reference Citation Analysis (0)] |
| 91. | Fischer CP. Interleukin-6 in acute exercise and training: what is the biological relevance? Exerc Immunol Rev. 2006;12:6-33. [PubMed] |
| 92. | Carey AL, Steinberg GR, Macaulay SL, Thomas WG, Holmes AG, Ramm G, Prelovsek O, Hohnen-Behrens C, Watt MJ, James DE, Kemp BE, Pedersen BK, Febbraio MA. Interleukin-6 increases insulin-stimulated glucose disposal in humans and glucose uptake and fatty acid oxidation in vitro via AMP-activated protein kinase. Diabetes. 2006;55:2688-2697. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 576] [Cited by in RCA: 644] [Article Influence: 33.9] [Reference Citation Analysis (0)] |
| 93. | Pedersen BK, Febbraio MA. Muscles, exercise and obesity: skeletal muscle as a secretory organ. Nat Rev Endocrinol. 2012;8:457-465. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 1664] [Cited by in RCA: 1906] [Article Influence: 146.6] [Reference Citation Analysis (0)] |
| 94. | Sun H, Sun J, Li M, Qian L, Zhang L, Huang Z, Shen Y, Law BY, Liu L, Gu X. Transcriptome Analysis of Immune Receptor Activation and Energy Metabolism Reduction as the Underlying Mechanisms in Interleukin-6-Induced Skeletal Muscle Atrophy. Front Immunol. 2021;12:730070. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 11] [Cited by in RCA: 21] [Article Influence: 5.3] [Reference Citation Analysis (0)] |
| 95. | Chen F, Fu J, Feng H. IL-6 Promotes Muscle Atrophy by Increasing Ubiquitin-Proteasome Degradation of Muscle Regeneration Factors After Cerebral Infarction in Rats. Neuromolecular Med. 2025;27:3. [RCA] [PubMed] [DOI] [Full Text] [Cited by in RCA: 2] [Reference Citation Analysis (0)] |
| 96. | Li F, Liu X, Bai N, Li Y, Hou M, Hou Y, Liu Y, Wang X, He Q, Li J. Irisin attenuates liver fibrosis by regulating energy metabolism and HMGB1/β-catenin signaling in hepatic stellate cells. Eur J Pharmacol. 2025;998:177519. [RCA] [PubMed] [DOI] [Full Text] [Cited by in RCA: 2] [Reference Citation Analysis (0)] |
| 97. | Lai TL, Park SY, Nguyen G, Pham PTM, Kang SM, Hong J, Lee JH, Im SS, Choi DH, Cho EH. Irisin Attenuates Hepatic Stellate Cell Activation and Liver Fibrosis in Bile Duct Ligation Mice Model and Improves Mitochondrial Dysfunction. Endocrinol Metab (Seoul). 2024;39:908-920. [RCA] [PubMed] [DOI] [Full Text] [Cited by in RCA: 1] [Reference Citation Analysis (0)] |
| 98. | Liao X, Zhan W, Li R, Tian T, Yu L, Yang Q. Irisin ameliorates endoplasmic reticulum stress and liver fibrosis through inhibiting PERK-mediated destabilization of HNRNPA1 in hepatic stellate cells. Biol Chem. 2021;402:703-715. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 3] [Cited by in RCA: 18] [Article Influence: 4.5] [Reference Citation Analysis (0)] |
| 99. | So WY, Leung PS. Irisin ameliorates hepatic glucose/lipid metabolism and enhances cell survival in insulin-resistant human HepG2 cells through adenosine monophosphate-activated protein kinase signaling. Int J Biochem Cell Biol. 2016;78:237-247. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 46] [Cited by in RCA: 55] [Article Influence: 6.1] [Reference Citation Analysis (0)] |
| 100. | Medhat D, El-Bana MA, El-Daly SM, Ashour MN, Elias TR, Mohamed RA, Yassen NN, Abdel-Monem MA, Hussein J. Influence of irisin on diet-induced metabolic syndrome in experimental rat model. J Complement Integr Med. 2021;18:347-354. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 3] [Cited by in RCA: 9] [Article Influence: 2.3] [Reference Citation Analysis (0)] |
| 101. | Zhao Y, Li J, Ma A, Wang Z, Ni Y, Wu D, Zhou Y, Zhang N, Zhang L, Chang Y, Wang Q. Irisin alleviates hepatic steatosis by activating the autophagic SIRT3 pathway. Chin Med J (Engl). 2025;. [RCA] [PubMed] [DOI] [Full Text] [Cited by in RCA: 2] [Reference Citation Analysis (0)] |
| 102. | Weigert C, Hoene M, Plomgaard P. Hepatokines-a novel group of exercise factors. Pflugers Arch. 2019;471:383-396. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 28] [Cited by in RCA: 38] [Article Influence: 5.4] [Reference Citation Analysis (0)] |
| 103. | Seo DY, Park SH, Marquez J, Kwak HB, Kim TN, Bae JH, Koh JH, Han J. Hepatokines as a Molecular Transducer of Exercise. J Clin Med. 2021;10:385. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 9] [Cited by in RCA: 21] [Article Influence: 5.3] [Reference Citation Analysis (0)] |
| 104. | Seo JA, Kang MC, Yang WM, Hwang WM, Kim SS, Hong SH, Heo JI, Vijyakumar A, Pereira de Moura L, Uner A, Huang H, Lee SH, Lima IS, Park KS, Kim MS, Dagon Y, Willnow TE, Aroda V, Ciaraldi TP, Henry RR, Kim YB. Apolipoprotein J is a hepatokine regulating muscle glucose metabolism and insulin sensitivity. Nat Commun. 2020;11:2024. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 22] [Cited by in RCA: 45] [Article Influence: 9.0] [Reference Citation Analysis (0)] |
| 105. | Meex RCR, Watt MJ. Hepatokines: linking nonalcoholic fatty liver disease and insulin resistance. Nat Rev Endocrinol. 2017;13:509-520. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 336] [Cited by in RCA: 459] [Article Influence: 57.4] [Reference Citation Analysis (2)] |
| 106. | Pasmans K, Adriaens ME, Olinga P, Langen R, Rensen SS, Schaap FG, Olde Damink SWM, Caiment F, van Loon LJC, Blaak EE, Meex RCR. Hepatic Steatosis Contributes to the Development of Muscle Atrophy via Inter-Organ Crosstalk. Front Endocrinol (Lausanne). 2021;12:733625. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in RCA: 4] [Reference Citation Analysis (0)] |
| 107. | Kurosawa T, Goto M, Kaji N, Aikiyo S, Mihara T, Ikemoto-Uezumi M, Toyoda M, Kanazawa N, Nakazawa T, Hori M, Uezumi A. Liver fibrosis-induced muscle atrophy is mediated by elevated levels of circulating TNFα. Cell Death Dis. 2021;12:11. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 4] [Cited by in RCA: 16] [Article Influence: 4.0] [Reference Citation Analysis (0)] |
| 108. | Culver A, Hamang M, Wang Y, Jiang H, Yanum J, White E, Gawrieh S, Vuppalanchi RK, Chalasani NP, Dai G, Yaden BC. GDF8 Contributes to Liver Fibrogenesis and Concomitant Skeletal Muscle Wasting. Biomedicines. 2023;11:1909. [RCA] [PubMed] [DOI] [Full Text] [Cited by in RCA: 3] [Reference Citation Analysis (0)] |
| 109. | Joo SK, Kim W. Interaction between sarcopenia and nonalcoholic fatty liver disease. Clin Mol Hepatol. 2023;29:S68-S78. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 2] [Cited by in RCA: 40] [Article Influence: 20.0] [Reference Citation Analysis (0)] |
| 110. | Akhmedov VA, Marinenko VS. Sarcopenia and non-alcoholic fatty liver disease-complex pathogenetic relationships. Eksp Klin Gastroenterol. 2025;. [DOI] [Full Text] |
| 111. | Kim JA, Choi KM. Sarcopenia and fatty liver disease. Hepatol Int. 2019;13:674-687. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 39] [Cited by in RCA: 86] [Article Influence: 14.3] [Reference Citation Analysis (0)] |
| 112. | Petrescu M, Vlaicu SI, Ciumărnean L, Milaciu MV, Mărginean C, Florea M, Vesa ȘC, Popa M. Chronic Inflammation-A Link between Nonalcoholic Fatty Liver Disease (NAFLD) and Dysfunctional Adipose Tissue. Medicina (Kaunas). 2022;58:641. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 1] [Cited by in RCA: 74] [Article Influence: 24.7] [Reference Citation Analysis (0)] |
| 113. | Jiang S, Kim TM, Park SY, Jin EJ. ROS-responsive MnO(2) mesoporous hydrogel to modulate liver-muscle crosstalk and mitigate NAFLD-associated sarcopenia via exosomal miR-582-5p delivery. Theranostics. 2025;15:4579-4592. [RCA] [PubMed] [DOI] [Full Text] [Cited by in RCA: 2] [Reference Citation Analysis (0)] |
| 114. | Liu X, Xu M, Wang H, Zhu L. Role and Mechanism of Short-Chain Fatty Acids in Skeletal Muscle Homeostasis and Exercise Performance. Nutrients. 2025;17:1463. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in RCA: 2] [Reference Citation Analysis (0)] |
| 115. | Van K, Burns JL, Monk JM. Effect of Short-Chain Fatty Acids on Inflammatory and Metabolic Function in an Obese Skeletal Muscle Cell Culture Model. Nutrients. 2024;16:500. [RCA] [PubMed] [DOI] [Full Text] [Cited by in RCA: 9] [Reference Citation Analysis (0)] |
| 116. | Martin JLA, Cartwright NM, Hutchinson AL, Robinson LE, Ma DWL, Monk JM. Differential Effects of Short-Chain Fatty Acids on L6 Myotube Inflammatory Mediator Production in Response to Lipopolysaccharide- or Palmitic Acid-Stimulation. Nutrients. 2022;14:2826. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in RCA: 5] [Reference Citation Analysis (0)] |
| 117. | He J, Zhang P, Shen L, Niu L, Tan Y, Chen L, Zhao Y, Bai L, Hao X, Li X, Zhang S, Zhu L. Short-Chain Fatty Acids and Their Association with Signalling Pathways in Inflammation, Glucose and Lipid Metabolism. Int J Mol Sci. 2020;21:6356. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 127] [Cited by in RCA: 549] [Article Influence: 109.8] [Reference Citation Analysis (0)] |
| 118. | Frampton J, Murphy KG, Frost G, Chambers ES. Short-chain fatty acids as potential regulators of skeletal muscle metabolism and function. Nat Metab. 2020;2:840-848. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 128] [Cited by in RCA: 277] [Article Influence: 55.4] [Reference Citation Analysis (0)] |
| 119. | den Besten G, van Eunen K, Groen AK, Venema K, Reijngoud DJ, Bakker BM. The role of short-chain fatty acids in the interplay between diet, gut microbiota, and host energy metabolism. J Lipid Res. 2013;54:2325-2340. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 2408] [Cited by in RCA: 3285] [Article Influence: 273.8] [Reference Citation Analysis (3)] |
| 120. | Lahiri S, Kim H, Garcia-Perez I, Reza MM, Martin KA, Kundu P, Cox LM, Selkrig J, Posma JM, Zhang H, Padmanabhan P, Moret C, Gulyás B, Blaser MJ, Auwerx J, Holmes E, Nicholson J, Wahli W, Pettersson S. The gut microbiota influences skeletal muscle mass and function in mice. Sci Transl Med. 2019;11:eaan5662. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 153] [Cited by in RCA: 374] [Article Influence: 74.8] [Reference Citation Analysis (0)] |
| 121. | Tokuhara D. Role of the Gut Microbiota in Regulating Non-alcoholic Fatty Liver Disease in Children and Adolescents. Front Nutr. 2021;8:700058. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 11] [Cited by in RCA: 56] [Article Influence: 14.0] [Reference Citation Analysis (0)] |
| 122. | Ferenc K, Jarmakiewicz-Czaja S, Filip R. What Does Sarcopenia Have to Do with Nonalcoholic Fatty Liver Disease? Life (Basel). 2023;14:37. [RCA] [PubMed] [DOI] [Full Text] [Cited by in RCA: 4] [Reference Citation Analysis (0)] |
| 123. | Yaghmaei H, Bahanesteh A, Soltanipur M, Takaloo S, Rezaei M, Siadat SD. The Role of Gut Microbiota Modification in Nonalcoholic Fatty Liver Disease Treatment Strategies. Int J Hepatol. 2024;2024:4183880. [RCA] [PubMed] [DOI] [Full Text] [Cited by in RCA: 4] [Reference Citation Analysis (0)] |
| 124. | Efremova I, Alieva A, Maslennikov R, Poluektova E, Zharkova M, Kudryavtseva A, Krasnov G, Zharikov Y, Nerestyuk Y, Karchevskaya A, Ivashkin V. Akkermansia muciniphila is associated with normal muscle mass and Eggerthella is related with sarcopenia in cirrhosis. Front Nutr. 2024;11:1438897. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in RCA: 1] [Reference Citation Analysis (0)] |
| 125. | Ponziani FR, Picca A, Marzetti E, Calvani R, Conta G, Del Chierico F, Capuani G, Faccia M, Fianchi F, Funaro B, Josè Coelho-Junior H, Petito V, Rinninella E, Paroni Sterbini F, Reddel S, Vernocchi P, Cristina Mele M, Miccheli A, Putignani L, Sanguinetti M, Pompili M, Gasbarrini A; GuLiver study group. Characterization of the gut-liver-muscle axis in cirrhotic patients with sarcopenia. Liver Int. 2021;41:1320-1334. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 22] [Cited by in RCA: 66] [Article Influence: 16.5] [Reference Citation Analysis (0)] |
| 126. | Zhao S, Liu W, Wang J, Shi J, Sun Y, Wang W, Ning G, Liu R, Hong J. Akkermansia muciniphila improves metabolic profiles by reducing inflammation in chow diet-fed mice. J Mol Endocrinol. 2017;58:1-14. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 156] [Cited by in RCA: 196] [Article Influence: 24.5] [Reference Citation Analysis (0)] |
| 127. | Allen SL, Quinlan JI, Dhaliwal A, Armstrong MJ, Elsharkawy AM, Greig CA, Lord JM, Lavery GG, Breen L. Sarcopenia in chronic liver disease: mechanisms and countermeasures. Am J Physiol Gastrointest Liver Physiol. 2021;320:G241-G257. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 11] [Cited by in RCA: 53] [Article Influence: 13.3] [Reference Citation Analysis (0)] |
| 128. | Meyer F, Bannert K, Wiese M, Esau S, Sautter LF, Ehlers L, Aghdassi AA, Metges CC, Garbe LA, Jaster R, Lerch MM, Lamprecht G, Valentini L. Molecular Mechanism Contributing to Malnutrition and Sarcopenia in Patients with Liver Cirrhosis. Int J Mol Sci. 2020;21:5357. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 34] [Cited by in RCA: 51] [Article Influence: 10.2] [Reference Citation Analysis (0)] |
| 129. | Conchillo M, Prieto J, Quiroga J. [Insulin-like growth factor I (IGF-I) and liver cirrhosis]. Rev Esp Enferm Dig. 2007;99:156-164. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 6] [Cited by in RCA: 11] [Article Influence: 0.6] [Reference Citation Analysis (0)] |
| 130. | Li BW, Feng LL, Tian ZJ. [Research progress of the role of IGF-1 in metabolic diseases and the effect of exercise intervention]. Sheng Li Xue Bao. 2021;73:342-352. [PubMed] [DOI] [Full Text] |
| 131. | Saeki C, Kanai T, Ueda K, Nakano M, Oikawa T, Torisu Y, Saruta M, Tsubota A. Insulin-like growth factor 1 predicts decompensation and long-term prognosis in patients with compensated cirrhosis. Front Med (Lausanne). 2023;10:1233928. [RCA] [PubMed] [DOI] [Full Text] [Cited by in RCA: 9] [Reference Citation Analysis (0)] |
| 132. | Kim Y. Emerging Treatment Options for Sarcopenia in Chronic Liver Disease. Life (Basel). 2021;11:250. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 4] [Cited by in RCA: 16] [Article Influence: 4.0] [Reference Citation Analysis (0)] |
| 133. | Jindal A, Jagdish RK. Sarcopenia: Ammonia metabolism and hepatic encephalopathy. Clin Mol Hepatol. 2019;25:270-279. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 35] [Cited by in RCA: 75] [Article Influence: 12.5] [Reference Citation Analysis (0)] |
| 134. | Butterworth RF. L-Ornithine L-Aspartate for the Treatment of Sarcopenia in Chronic Liver Disease: The Taming of a Vicious Cycle. Can J Gastroenterol Hepatol. 2019;2019:8182195. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 15] [Cited by in RCA: 21] [Article Influence: 3.5] [Reference Citation Analysis (0)] |
| 135. | Ostrovskaya AS, Maevskaya MV, Loban KM, Zharkova MS, Shapka MP, Vasiltsova EA, Chvileva YO, Ivashkin VT. Possibilities of treating sarcopenia in liver cirrhosis with hypoammonemic agents. Med Counc. 2024;. [RCA] [DOI] [Full Text] [Cited by in RCA: 1] [Reference Citation Analysis (0)] |
| 136. | Liu J, Huang H. [Pathogenesis of sarcopenia in liver cirrhosis and current status of its diagnosis and treatment]. Linchuang Gandanbing Zazhi. 2020;36:895-899. [DOI] [Full Text] |
| 137. | Sinclair M, Grossmann M, Gow PJ, Angus PW. Testosterone in men with advanced liver disease: abnormalities and implications. J Gastroenterol Hepatol. 2015;30:244-251. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 120] [Cited by in RCA: 89] [Article Influence: 8.9] [Reference Citation Analysis (0)] |
| 138. | Saad F, Röhrig G, von Haehling S, Traish A. Testosterone Deficiency and Testosterone Treatment in Older Men. Gerontology. 2017;63:144-156. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 69] [Cited by in RCA: 72] [Article Influence: 8.0] [Reference Citation Analysis (0)] |
| 139. | Basualto-Alarcón C, Varela D, Duran J, Maass R, Estrada M. Sarcopenia and Androgens: A Link between Pathology and Treatment. Front Endocrinol (Lausanne). 2014;5:217. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 21] [Cited by in RCA: 33] [Article Influence: 3.0] [Reference Citation Analysis (0)] |
| 140. | Shigehara K, Kato Y, Izumi K, Mizokami A. Relationship between Testosterone and Sarcopenia in Older-Adult Men: A Narrative Review. J Clin Med. 2022;11:6202. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in RCA: 29] [Reference Citation Analysis (0)] |
| 141. | Chrysavgis L, Adamantou M, Angelousi A, Cholongitas E. The association of testosterone with sarcopenia and frailty in chronic liver disease. Eur J Clin Invest. 2024;54:e14108. [RCA] [PubMed] [DOI] [Full Text] [Cited by in RCA: 7] [Reference Citation Analysis (0)] |
| 142. | Fox R, Stenning K, Slee A, Macnaughtan J, Davies N. Sarcopenia in liver cirrhosis: Prevalence, pathophysiology and therapeutic strategies. Anal Biochem. 2022;647:114581. [RCA] [PubMed] [DOI] [Full Text] [Cited by in RCA: 11] [Reference Citation Analysis (0)] |
| 143. | Xu JQ, Pan YK, Zhang JX, Dai SX, Xu LS. Sarcopenia in liver cirrhosis: perspectives from epigenetics and microbiota. Front Med (Lausanne). 2023;10:1264205. [RCA] [PubMed] [DOI] [Full Text] [Cited by in RCA: 4] [Reference Citation Analysis (0)] |
| 144. | Maslennikov R, Ivashkin V, Efremova I, Poluektova E, Shirokova E. Gut-liver axis in cirrhosis: Are hemodynamic changes a missing link? World J Clin Cases. 2021;9:9320-9332. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in CrossRef: 21] [Cited by in RCA: 26] [Article Influence: 6.5] [Reference Citation Analysis (3)] |
| 145. | Wu Z, Zhou H, Liu D, Deng F. Alterations in the gut microbiota and the efficacy of adjuvant probiotic therapy in liver cirrhosis. Front Cell Infect Microbiol. 2023;13:1218552. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 16] [Cited by in RCA: 7] [Article Influence: 3.5] [Reference Citation Analysis (0)] |
| 146. | Wang FC, Li ZY, Zhang WJ, Mao XR, Li JF. [The significance of gut microbiota in acute-on-chronic liver failure]. Linchuang Gandanbing Zazhi. 2022;38:1667-1670. [DOI] [Full Text] |
| 147. | Aliwa B, Horvath A, Traub J, Feldbacher N, Habisch H, Fauler G, Madl T, Stadlbauer V. Altered gut microbiome, bile acid composition and metabolome in sarcopenia in liver cirrhosis. J Cachexia Sarcopenia Muscle. 2023;14:2676-2691. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 6] [Cited by in RCA: 22] [Article Influence: 11.0] [Reference Citation Analysis (0)] |
| 148. | Lee PC, Lee KC, Yang TC, Lu HS, Cheng TY, Chen YJ, Chiou JJ, Huang CW, Yang UC, Chia-Hui Tan E, Chou SH, Kuo YL, Schnabl B, Huang YH, Hou MC. Sarcopenia-related gut microbial changes are associated with the risk of complications in people with cirrhosis. JHEP Rep. 2023;5:100619. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in RCA: 12] [Reference Citation Analysis (0)] |