Published online Feb 26, 2025. doi: 10.4252/wjsc.v17.i2.102091
Revised: December 16, 2024
Accepted: February 7, 2025
Published online: February 26, 2025
Processing time: 138 Days and 16.9 Hours
Burn wound management is challenging, and while mesenchymal stem cell-derived exosomes show therapeutic potential, optimal delivery methods are unclear.
To study chitosan (CS)-αβ-glycerophosphate (CS-αβ-GP) hydrogel crosslinked with adipose-derived stem cell exosomes (ASC-Exos) for healing deep burn injuries.
Rats with deep burn injuries were divided into the CS + ASCs-Exos group, the ASCs-Exos group, the CS group, and the control group. The healing rates on days 4, 7, and 14 after treatment were analyzed using ImageJ software. On day 14, the tissues were stained with hematoxylin and eosin staining, Masson’s trichrome staining, and immunohistochemical analysis to evaluate tumor necrosis factor α, interleukin-6 (IL-6), IL-1α, IL-10, transforming growth factor β, and epidermal growth factor. The mRNA levels of IL-1α, CD86, C-C motif chemokine ligand 22, and CD163 were evaluated through quantitative polymerase chain reaction.
The CS + ASC-Exos group exhibited enhanced healing, reduced lymphocyte infiltration, blood vessels, and muscle fiber distribution. Increased IL-10, transforming growth factor β, and epidermal growth factor and decreased tumor necrosis factor α, IL-1α, and IL-6 expression were observed. Quantitative polymerase chain reaction revealed reduced IL-1α and CD86 and increased C-C motif chemokine ligand 22 and CD163 expression. Protein analysis showed downregulation of phosphorylated inhibitor of kappa Balpha and P65 in the nuclear factor κB (NF-κB) pathway. ASC-Exos crosslinked with CS-αβ-GP hydrogel demonstrates superior effects in anti-inflammation, wound healing promotion, and promotion of M1 macrophage transformation to M2 macrophage by blocking the NF-κB pathway compared to ASC-Exos alone.
Our research demonstrates that the ASC-Exos cross-linked CS-αβ-GP hydrogel represents an advanced therapeutic approach for treating deep burn wounds. It has anti-inflammatory effects, promotes wound healing, and facilitates the transition of M1 macrophages to M2 macrophages by blocking the NF-κB pathway.
Core Tip: Studies indicate that the exosome hydrogel composite demonstrates exceptional efficacy in facilitating the healing of deep burn wounds and managing infection. It has anti-inflammatory effects, promotes wound healing, and facilitates the transition of M1 macrophages to M2 macrophages by blocking the nuclear factor κB pathway, thereby offering a novel strategy for the clinical utilization of mesenchymal stem cell-derived exosomes.
- Citation: Xu L, Liu D, Yun HL, Zhang W, Ren L, Li WW, Han C. Effect of adipose-derived stem cells exosomes cross-linked chitosan-αβ-glycerophosphate thermosensitive hydrogel on deep burn wounds. World J Stem Cells 2025; 17(2): 102091
- URL: https://www.wjgnet.com/1948-0210/full/v17/i2/102091.htm
- DOI: https://dx.doi.org/10.4252/wjsc.v17.i2.102091
In clinical practice, improving burn wound management remains a critical issue due to the increasing incidence of burns resulting from both natural disasters and human activities. Mesenchymal stem cell (MSC) derived exosomes (MSC-Exos), referred to as MSC-Exos, have emerged as a highly promising therapeutic modality for the management of chronic wounds, as revealed by recent developments in regenerative medicine and tissue engineering.
Extensive research has demonstrated that MSC-Exos can significantly enhance the healing of burn wounds, offering several advantages compared to MSCs alone. These benefits encompass superior safety profiles, increased storage stability, and simplified collection operations[1-3]. The present applications of MSC-Exos in wound repair predominantly depend on subcutaneous or intravenous delivery. But these methods have significant limitations due to rapid clearance and burst release patterns, which could potentially compromise their therapeutic efficacy across the complex stages of wound healing.
Several different delivery technologies, such as patches, injectable microcarriers, and hydrogels, are being investigated by researchers in order to solve these constraints. The goal of these systems is to preserve Exos function at wound sites while simultaneously enhancing both efficiency and safety[4-6]. Chitosan (CS) has demonstrated a great deal of promise among the many artificial injectable hydrogels that promote sustained Exo release and enhance local retention. This is mostly owing to the fact that it possesses natural antibacterial qualities and the capacity to maintain appropriate wound moisture[7]. This study seeks to evaluate if this approach serves as an optimal vehicle for MSC-Exos in burn wound healing applications.
Sprague Dawley rat adipose-derived stem cells (ASC) (product code: RASMD-01001) were obtained from OriCell with approval from the Ethics Committee (approval number: XBZQZYY2021-046). Cell culture was initiated by diluting 1 mL of cells in 9 mL basic medium, followed by centrifugation at 1000 rpm for 5 minutes. Following the removal of the supernatant, the cells were resuspended in 4 mL of medium supplemented with 10% serum and subsequently transferred to a 25T culture flask.
Cultures were maintained at 37 °C with 5% CO2, with initial media change after 24 hours following confirmation of cell adherence. Cells were passaged at 90%-100% confluence using standard protocols. For passaging, cells were rinsed with phosphate buffered saline (PBS), detached, and split between new culture flasks supplemented with complete medium. For cryopreservation, cells showing optimal viability (approximately 70% density) were collected, centrifuged, and resuspended in cryopreservation solution at a density of 5 × 106 cells/mL to 1 × 107 cells/mL. The cell suspension was divided between cryopreservation tubes, gradually frozen at -80 °C overnight, and transferred to liquid nitrogen for long-term storage.
Electron microscopy observation: The morphology of Exos was carefully analyzed using transmission electron microscopy, a powerful tool for examining nanoscale structures. Exo suspensions were first prepared by dissolving the Exos at a concentration of 0.5 g/L in PBS. To preserve the structural integrity of the Exos, the suspension was fixed with 2.5% glutaraldehyde, which stabilizes the particles by crosslinking proteins and other biomolecules. A 20-30 μL aliquot of the fixed Exo suspension was then applied to carbon-coated copper grids, which serve as a stable substrate for the Exos during imaging. These grids were briefly dried under an infrared lamp for 5 minutes to remove excess solution, ensuring that the sample adhered well to the surface.
The Exo samples were subsequently stained with 1% phosphotungstic acid at room temperature for 5 minutes. Phosphotungstic acid is commonly used as a negative stain to enhance contrast in electron microscopy, as it binds to the surface of the Exos and helps to highlight their structure. After staining, the samples were dried again for 10 minutes to remove any excess staining solution. The final samples were then carefully examined under a transmission electron microscope, which allowed for detailed observation of the Exo size, shape, and surface characteristics.
Western blot detection of protein expression level: Protein expression levels were assessed using western blotting, a widely used technique for detecting specific proteins within a complex mixture. The first step in the process involved lysing the samples in a buffer containing phenylmethylsulfonyl fluoride, a protease inhibitor, at a ratio of 100:1. The mixture was incubated on ice for 10-15 minutes to ensure complete lysis of the cells or Exos, followed by centrifugation at 12000 rpm for 10 minutes at 4 °C. This step allowed for the separation of cellular debris from the soluble proteins, with the supernatant containing the proteins of interest.
The protein solution was then heat-treated at 100 °C for 20 minutes with loading buffer to denature the proteins and break down any secondary structures, ensuring they were in their linear forms for electrophoresis. The samples were stored at -20 °C until further analysis. After electrophoresis, proteins were transferred to a membrane for immunostaining, where primary antibodies specific to the target proteins were used to bind to the proteins of interest. Secondary antibodies conjugated with an enzyme were then applied to facilitate detection. The results were quantified using ImageJ software, which allowed for precise measurement of the intensity of protein bands, enabling comparison of protein expression levels across different samples and treatment conditions.
The preparation of the cross-linked ASC-Exos hydrogel involved a careful synthesis process to ensure the successful incorporation of Exos into the hydrogel matrix. CS, a biopolymer with excellent biocompatibility and gelation properties, was first dissolved at a concentration of 2 g in 100 mL of acetic acid/sodium acetate buffer, adjusted to a pH of 4.6. CS has a degree of deacetylation of 75%, which contributes to its ability to form gels in specific conditions. The solution was filtered to remove any particulates and sterilized to ensure that the hydrogel would be safe for use in biological applications.
Next, the CS solution was cooled in an ice bath to prepare it for the addition of αβ-glycerophosphate (αβ-GP) (a gel-forming agent). A 50% weight/volume (W/V) αβ-GP solution was prepared separately and added dropwise to the chilled CS solution. The CS/αβ-GP ratio was carefully controlled at 8.8/1.2 to achieve the desired gel strength and thermosensitive properties. The mixture was kept in the ice bath for 20 minutes to promote the crosslinking process and to ensure a homogenous blend. The resulting hydrogel was then stored at 4 °C for further use.
To incorporate Exos into the hydrogel, ASC-Exos were added at a concentration of 1 g/L. This allowed for the controlled release of Exos upon application, providing sustained therapeutic effects at the wound site. The incorporation of Exos into the hydrogel matrix enhances the overall functionality of the system, allowing for both localized delivery and prolonged release of therapeutic Exos. The final formulation of the CS-αβ-GP hydrogel loaded with ASC-Exos is expected to offer a promising therapeutic strategy for wound healing, combining the advantages of biocompatible materials and bioactive molecules.
Porosity of the CS-αβ-GP hydrogel: The porosity of the hydrogel was calculated using the anhydrous ethanol displacement method by measuring the change in ethanol volume. Anhydrous ethanol was poured into a 50 mL volumetric flask, and the initial volume of ethanol (V0) was recorded. The dried hydrogel sample was immersed in anhydrous ethanol, and the volume of ethanol at this point was measured as V1. After removing the hydrogel sample from the ethanol, the remaining volume of ethanol was recorded as V2. Each sample was subjected to at least three replicate experiments, and the average was taken as the final porosity, as per the equation: Porosity = (V0 - V2)/(V1 - V2) × 100%.
The cumulative release rate of Exos after crosslinking the CS-αβ-GP hydrogel with ASC-Exos: A 200 μL hydrogel-Exos complex containing 200 μg of Exos was immersed in PBS solution at 37 °C. The supernatant was analyzed at 1, 2, 3, 4, 6, 8, 10, 12, 24, 36, and 72 hours using a BCA protein assay kit (Pierce, Rockford, IL, United States). The cumulative release rate of Exos = (amount in supernatant/initial amount in hydrogel) × 100%.
Rheological testing of CS-αβ-GP hydrogel: The elastic modulus (G’) and viscous modulus (G’’) of the samples were assessed utilizing a rotational rheometer (Physician MCR301, Anton Paar, Austria) across a temperature range of 10 °C to 50 °C to analyze their rheological characteristics.
Degradation rate of CS-αβ-GP hydrogel: The initial quantity of hydrogel, W0, was documented. The hydrogel was incubated in PBS solution for 72 hours, with its weight measured at 1, 2, 3, 4, 6, 8, 10, 12, 24, 36, and 72 hours, recorded as Wt. The degradation of the hydrogels in PBS was assessed over a period of 72 hours, as the equation: Degradation rate = (W0 - Wt)/W0 × 100%.
Based on previous literature and experience with animal sample sizes, 40 rats were purchased and divided into four groups with ten rats in each group[8]. The procedure began with anesthesia using intravenous thiopental sodium (40 mg/kg), administered through the tail vein to ensure the rats were fully sedated. The fur on the rats’ backs was then shaved to expose the skin for the burn induction. To create the burn, 80 °C water was applied to the skin for 8 seconds, resulting in a burn wound with a diameter of 16 mm. After the burn was induced, sterile saline-soaked gauze was placed over the wound for 6 minutes to control immediate post-burn effects and minimize thermal damage. Once the burn was established, the rats were assigned to one of four groups for further treatment: Group A (CS + ASC-Exos), which received the application of a cross-linked hydrogel containing 200 μg of Exos; group B (ASC-Exos), which received 200 μg of Exos in 200 μL of PBS; group C (CS), which received the equivalent volume of the hydrogel without Exos; and group D (control), which received only a standard transparent dressing. All groups had their dressings changed every two days during the study period to maintain a controlled healing environment.
Wound healing progression was evaluated using standardized digital photography and quantitative analysis. Images were captured immediately post-injury and at designated timepoints (4, 7, and 14 days) across all treatment groups. Wound areas were precisely measured using ImageJ software calibrated with a standardized scale marker in each image. The healing rate was calculated using the formula: Wound healing rate = (1 - unhealed wound area/original wound area) × 100%.
After the 14-day treatment period, full-thickness tissue samples were collected from both the wound sites and corresponding uninjured areas, which served as internal controls. The tissue specimens were processed by first fixing them in 40 g/L paraformaldehyde solution at room temperature for 24 hours. Subsequent to fixation, the samples were subjected to a dehydration procedure utilising graded alcohols, after which they were embedded in paraffin. Subsequent to this, serial sections with a thickness of 4 μm were generated utilising a rotary microtome.
The histological analysis involved staining protocols to evaluate tissue characteristics. Hematoxylin and eosin staining was used to assess the cellular architecture and inflammatory infiltrate, while Masson’s Trichrome staining allowed for the evaluation of collagen deposition and organization. To ensure quantitative analysis, inflammatory cell infiltration was assessed by counting the lymphocytes per high-power field using ImageJ software. Additionally, the collagen volume fraction (CVF) was calculated by digitally analyzing Masson’s stained sections using standardized thresholding methods.
Immunohistochemical analysis was employed to evaluate the expression of pro-inflammatory markers at the wound site. Specific markers such as tumor necrosis factor α (TNF-α), interleukin-6 (IL-6), and other cytokines were assessed to determine the inflammatory status of the wound. These markers are crucial for understanding the immune response and inflammation dynamics during the healing process, as they play significant roles in the regulation of tissue repair and regeneration. By quantifying these markers, the study aimed to assess the effect of the different treatments on modulating inflammation and promoting tissue regeneration.
Macrophage polarization was assessed by analyzing the mRNA expression of key markers for M1 and M2 macrophages from wound tissues collected on day 14. To evaluate the polarization of macrophages, we focused on pro-inflammatory M1 markers, such as IL-1α and CD86, which are associated with the promotion of inflammation and tissue damage, and anti-inflammatory M2 markers, including C-C motif chemokine ligand 22 (CCL22) and CD163, which play a role in tissue repair and resolution of inflammation. RNA was extracted from the wound tissues of rats in the different treatment groups and analyzed using quantitative polymerase chain reaction. The expression levels of the selected markers were normalized to GAPDH as a housekeeping gene to account for any variations in RNA quantity and quality across the samples. This normalization allowed for a precise comparison of M1 and M2 polarization between the treatment groups.
Statistical analyses were conducted utilising SPSS Statistics (version 22.0; IBM, Armonk, NY, United States) and GraphPad Prism (version 8.0, San Diego, CA, United States). One-way analysis of variance (ANOVA) accompanied by Bonferroni’s test was employed to assess the differences among groups. All experimental outcomes are shown as mean ± SD. Statistical significance was established at P < 0.05.
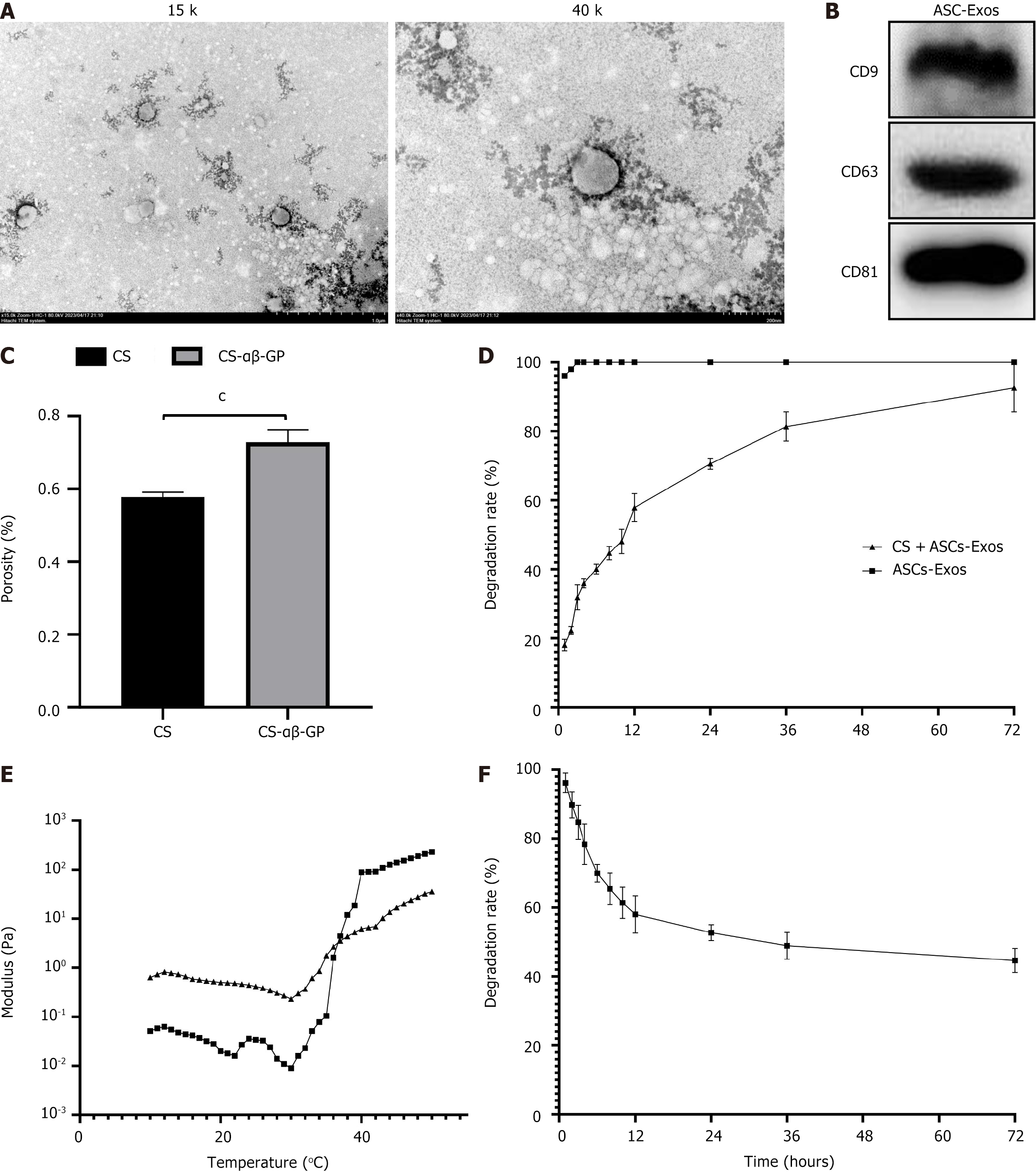
Comprehensive biophysical characterization confirmed the successful isolation and purification of ASC-Exos. Transmission electron microscopy analysis revealed distinctive nanostructures exhibiting characteristic morphology of extracellular vesicles. These vesicular structures displayed the expected double-membrane architecture with diameters consistently falling within the 30-150 nm range typical of Exos (Figure 1A). Molecular analysis through western blotting confirmed the presence of canonical Exo surface markers, including strong expression of CD9, CD63, and CD81 proteins, validating the identity and purity of the isolated vesicles (Figure 1B).
The physical and mechanical properties of the CS-αβ-GP hydrogel were comprehensively evaluated through various characterization methods. As shown in Figure 1C, the porosity of the CS-αβ-GP thermosensitive hydrogel is significantly higher than that of the CS hydrogel, suggesting that the CS-αβ-GP thermosensitive hydrogel is more suitable for cell adhesion and growth compared to the CS hydrogel. As illustrated in Figure 1D, after crosslinking ASCs-Exos with the CS-αβ-GP thermosensitive hydrogel, the cumulative release rate of exosomes reached only about 70% after 24 hours, indicating that this material is suitable for the sustained release of ASCs-Exos. As shown in Figure 1E, the G’ and G’’ values of the CS-αβ-GP thermosensitive hydrogel rapidly increased at temperatures 30 °C and 40 °C, completing the sol-gel transition process at 37 °C. The results indicate that the CS-αβ-GP thermosensitive hydrogel possesses a phase transition temperature that is well-suited for wound administration. An effective wound dressing needs to have a certain degree of degradability. As depicted in Figure 1F, the weight of the CS-αβ-GP thermosensitive hydrogel decreased to about 40% after 72 hours, demonstrating its degradability.
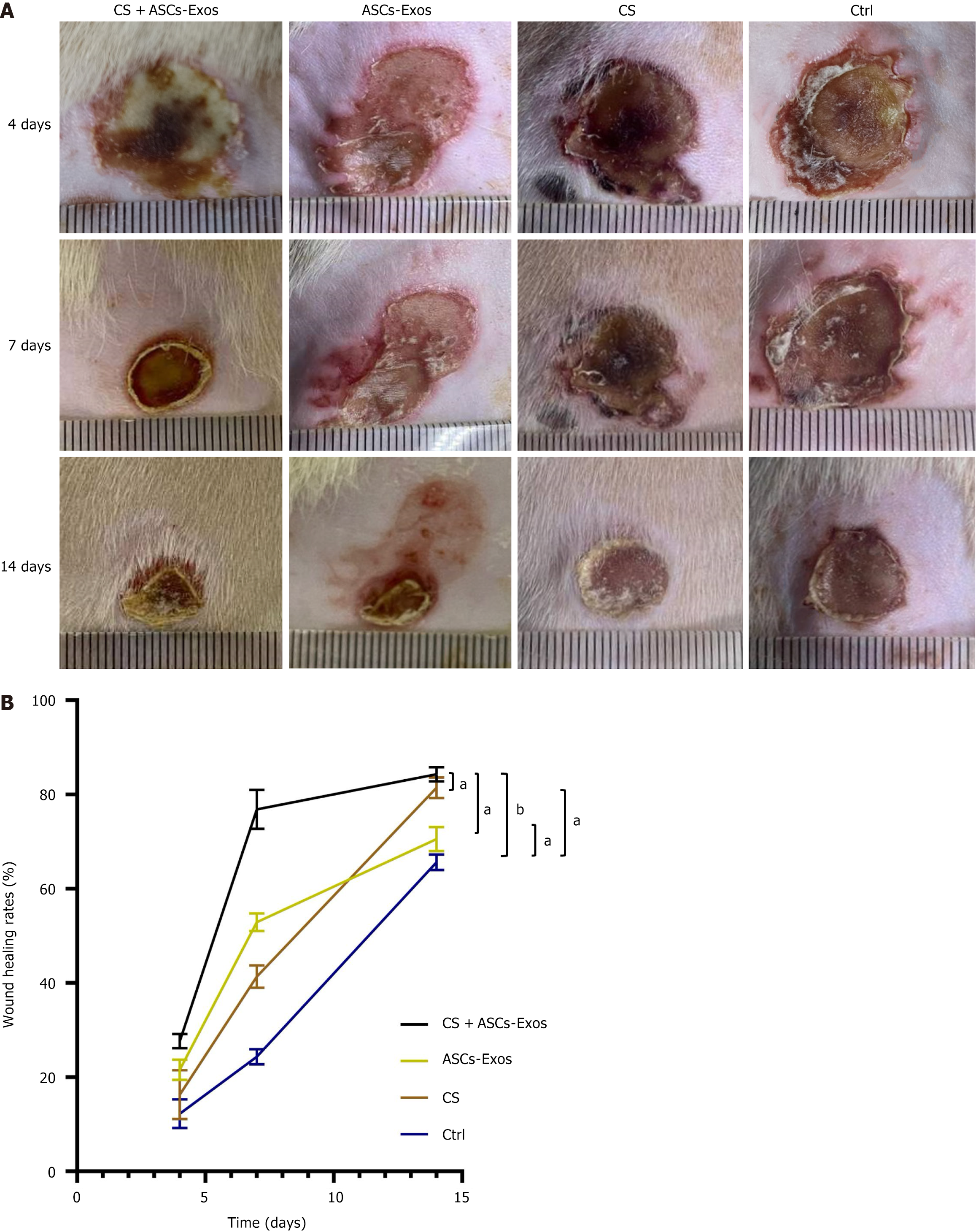
To establish therapeutic efficacy, we evaluated the Exos hydrogel composite in a rat burn model. As depicted in Figure 2, the CS + ASC-Exos group demonstrated superior healing kinetics, with significant acceleration beginning on day 4 and continuing to show improved outcomes through day 14 compared to other groups. This enhanced performance validates the Exos hydrogel composite’s therapeutic potential.
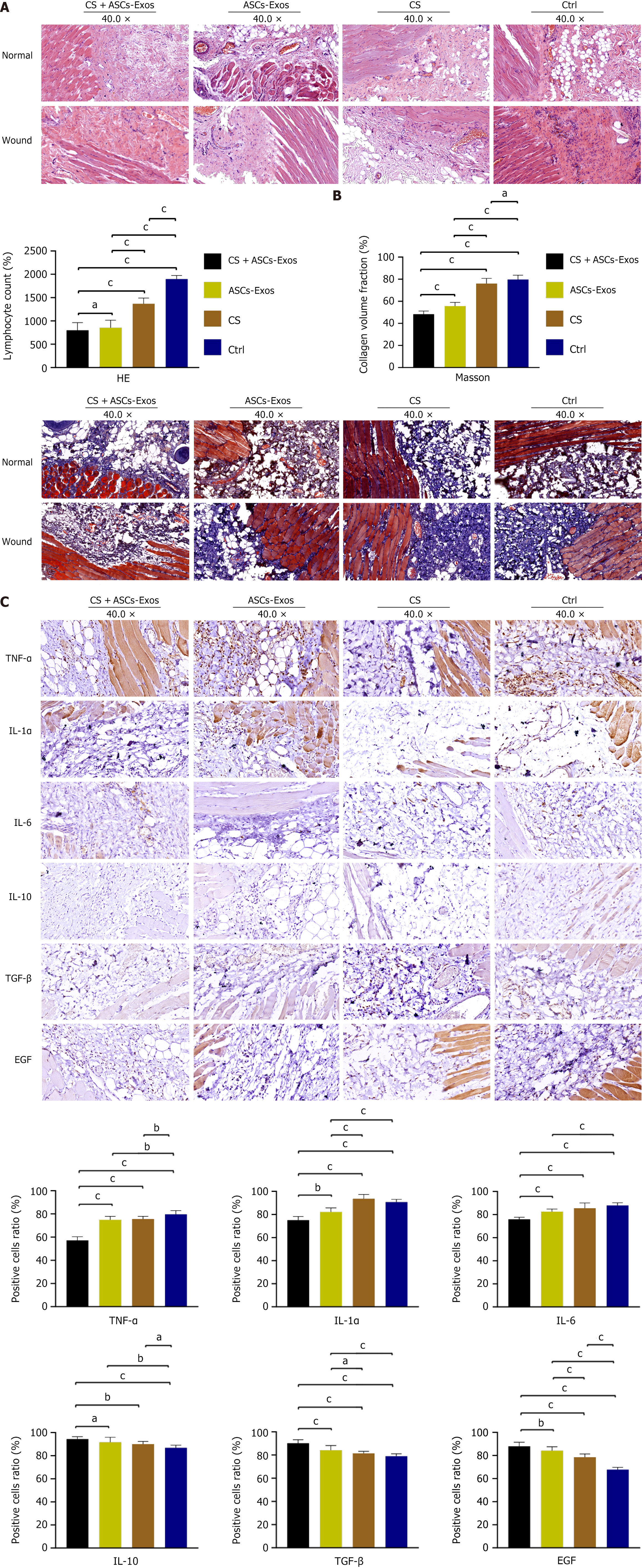
Histological analysis was conducted on full-thickness skin samples obtained after 14 days of treatment, with normal tissue used as a control for comparison. As depicted in Figure 3A, hematoxylin and eosin staining revealed significant differences between the treatment groups. The CS + ASC-Exos group demonstrated notably reduced lymphocyte infiltration and blood vessel density in the wound area compared to the other groups. This indicates that the Exo-loaded hydrogel effectively controlled inflammation, reducing the cellular inflammatory response and promoting a more favorable wound healing environment.
As shown in Figure 3B, Masson’s Trichrome staining revealed that, in the CS + ASC-Exos group, after 14 days, normal tissue exhibited a small amount of collagen fibers interspersed between the abundant muscle fibers. In the wound tissue, the amount and distribution of collagen and muscle fibers were consistent with those in the normal tissues under a microscope. This suggests that the Exo-loaded hydrogel facilitated tissue regeneration by promoting a well-organized matrix that resembled the natural tissue structure. The collagen deposition was well-structured, contributing to improved tissue integrity and function. The lower CVF observed in the CS + ASC-Exos group suggests that the treatment promoted better wound healing with reduced scar formation.
Immunohistochemical analysis of important inflammatory markers confirmed that CS + ASC-Exos treatment affects wound healing. In the CS + ASC-Exos group, anti-inflammatory and growth factors including IL-10, transforming growth factor β, and epidermal growth factor were upregulated, whereas pro-inflammatory cytokines like TNF-α, IL-1α, and IL-6 were considerably decreased. These data suggest that CS + ASC-Exos treatment promotes tissue regeneration and regulates inflammation to improve wound healing (Figure 3C).
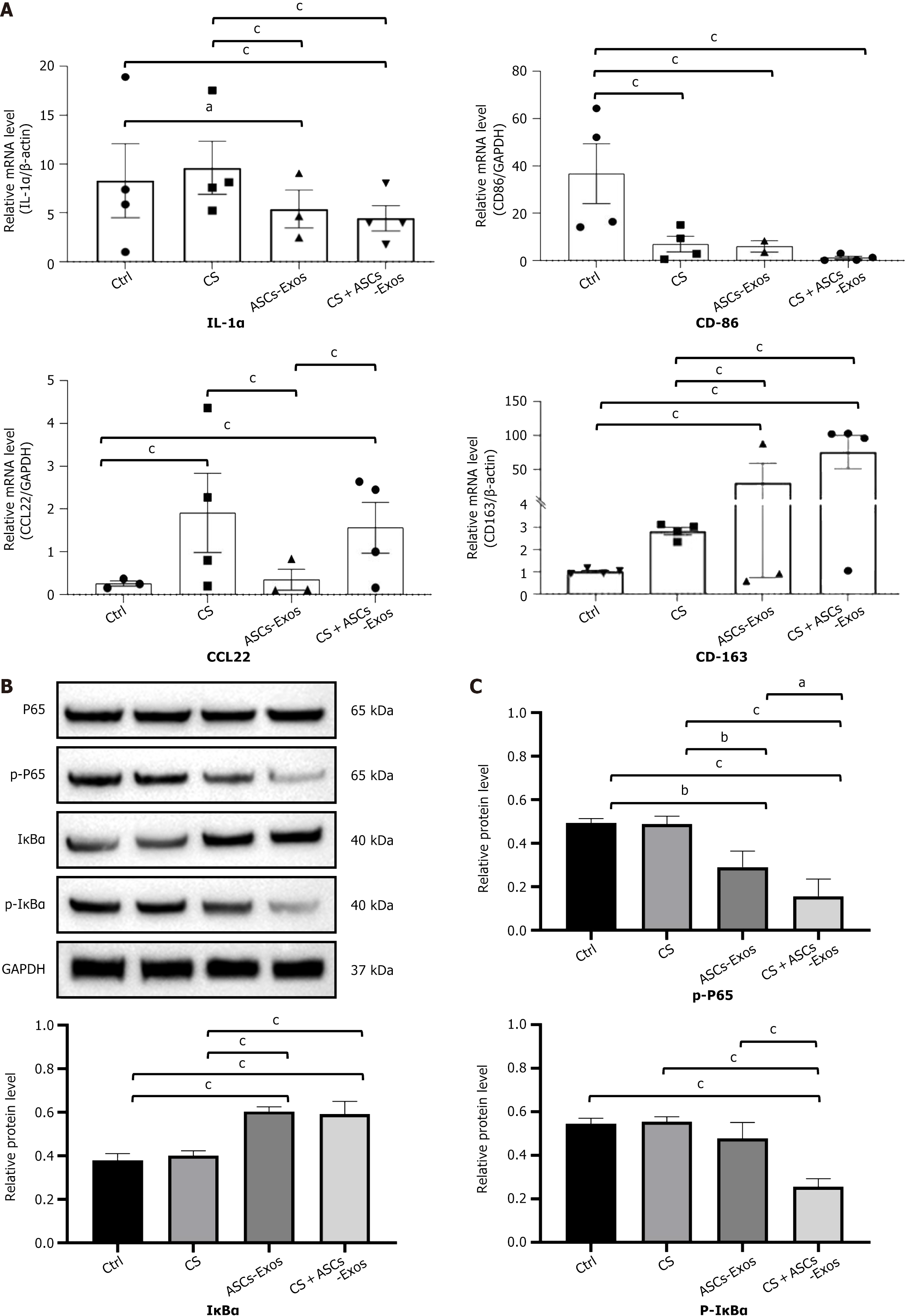
The CS + ASC-Exos therapy drastically affected macrophage polarization, as indicated by alterations in the expression of essential macrophage markers. As depicted in Figure 4A, the therapy group exhibited a significant reduction in the pro-inflammatory M1 markers IL-1α and CD86, which are commonly linked to inflammation and tissue damage. Concurrently, M2 macrophage markers CCL22 and CD163 were increased, signifying a transition towards an anti-inflammatory phenotype that facilitates tissue repair and regeneration. This shift in macrophage polarization indicates that CS + ASC-Exos therapy not only diminishes inflammation but also promotes a more conducive wound healing environment by facilitating macrophage polarization towards the M2 phenotype.
To investigate the immunomodulatory effect of Exos hydrogel composite, nuclear factor κB (NF-κB) inflammatory pathway protein levels were assessed. Figure 4B and C demonstrate that the CS + ASC-Exos group had considerably lower p-inhibitor of kappa Balpha (p-IκBα) protein expression relative to other treatment groups. A significant decrease in p-P65 protein levels in the inflammatory NF-κB pathway was seen in the CS + ASC-Exos group. The study found that CS + ASC-Exos can modulate macrophage polarisation from M1 to M2 by blocking the NF-κB pathway.
MSCs are multipotent progenitor cells capable of differentiating into many cell types under specific conditions. Approximately 5000 ASCs can be obtained from one gramme of adipose tissue. Despite the existence of various sources of MSCs, ASCs have attracted significant interest due to their availability and yield[8]. Research has shown that MSCs respond to tissue damage by migrating to affected regions and engaging with the immune and inflammatory systems throughout their presence there[9]. MSCs release beneficial chemicals via paracrine pathways, facilitating wound repair and tissue regeneration in response to inflammatory conditions[9].
When it comes to therapeutic applications, however, MSCs encounter a number of hurdles, such as the potential for tumorigenicity, concerns over immunological rejection, and complex storage needs. MSC-Exos have a number of advantages over their parent cells, including the following: (1) High stability: Despite being approximately one millionth the size of Exos are able to sustain their activity even when stored for an extended period of time at a temperature of -80 degrees Celsius[1]; (2) Efficient production: Each MSC has the ability to generate thousands of Exos, which can be recovered using conventional techniques such as ultracentrifugation[2]; and (3) Safety: Exos have a low membrane-bound protein content and are non-proliferative, which means that they have small risk of immune rejection or tumour formation[3]. This is one of the reasons why their safety profile has been enhanced.
According to the findings of a number of studies, MSC-Exos are effective in the process of wound healing. Zhang et al[10] demonstrated that induced pluripotent stem cell-MSC-Exos sped up the process of epithelial reconstruction, decreased the width of scars, and enhanced collagen maturation while simultaneously stimulating angiogenesis. Similarly, research conducted using umbilical cord-MSC-Exos showed their capacity to stimulate wound re-epithelial by activating the Wnt/β-catenin pathway[11]. Li et al[12] established that umbilical cord-MSC-Exos containing miR-181c could control inflammation by the Toll-like receptor 4 signaling pathway. Several studies explicitly focused on ASC-Exos have produced encouraging findings. In their study, Hu et al[13] proved that the treatment with ASC-Exos, either locally or systemically, enhanced fibroblast function and maximized collagen production. Using Wnt/β-catenin signaling, Ma et al[14] demonstrated that ASC-Exos could promote keratinocyte proliferation and migration while simultaneously suppressing. It has also been widely examined how macrophage polarization plays a role, with M1 macrophages responsible for producing pro molecules and M2 macrophages responsible for secreting anti-inflammatory mediators[15]. By focusing on pknox1, He et al[16] demonstrated that bone marrow-MSC-Exos containing miR-223 could enhance M2 polarization.
MSC-Exos have shown significant potential in promoting burn wound healing, yet traditional delivery methods often face challenges, such as rapid clearance and burst release, which limit their effectiveness[17]. Recent advancements in injectable hydrogels have addressed these issues, offering a more controlled and sustained release, thereby enhancing the local retention of Exos and improving therapeutic outcomes. Among various hydrogel systems, CS-based hydrogels have garnered particular attention due to their intrinsic antibacterial properties, biocompatibility, and ability to maintain optimal moisture levels at wound sites, which are crucial for healing[7]. The CS-αβ-GP hydrogel, which exhibits temperature-dependent gelation and long-term stability at 4 °C, stands out as an excellent candidate for Exo delivery, providing a favorable environment for burn wound regeneration[18].
In our study, the CS + ASC-Exos hydrogel system demonstrated superior therapeutic efficacy. Notably, the system accelerated the healing process starting from day 4, with significantly better final healing rates at day 14 compared to the control groups. Histological analysis revealed that the CS + ASC-Exos treatment reduced inflammatory cell infiltration and vascularization, which suggests effective modulation of the inflammatory response. Moreover, the collagen and muscle fiber distribution in the wound area showed remarkable normalization, with the CVF notably lower in the CS + ASC-Exos group, indicating improved tissue regeneration and better scar formation outcomes.
The immunomodulatory effects of the CS + ASC-Exos system were also evident, as demonstrated by the increased expression of anti-inflammatory and growth factors, such as IL-10, transforming growth factor β, and epidermal growth factor. In contrast, pro-inflammatory markers, including TNF-α, IL-6, and IL-1α, were significantly reduced. Additionally, the treatment promoted macrophage polarization toward the anti-inflammatory M2 phenotype, evidenced by higher levels of M2 markers like CCL22 and CD163, while M1 markers such as IL-1α and CD86 were reduced. This shift in mac
It has been reported that MSC-derived Exos contain a large number of active molecules, such as lipids, proteins, and microRNAs, especially miR-199a, which regulates the innate immune system by blocking factors in the NF-κB pathway[19-22]. Phosphorylated IκBα, a key regulatory factor in the NF-κB signaling pathway, positively regulates the expression of pro-inflammatory molecules[21,22]. Our study found that, compared to other treatment groups, the protein expression of p-IκBα was significantly downregulated in the CS + ASC-Exos group. Consistently, the protein level of the down
However, while this study presents promising results, several aspects require further investigation to fully understand and optimize the therapeutic potential of this composite system. One key area for future research is the detailed exami
Our research demonstrates that the ASC-Exos cross-linked CS-αβ-GP hydrogel represents an advanced therapeutic approach for treating deep burn wounds. It has anti-inflammatory effects, promotes wound healing, and facilitates the transition of M1 macrophages to M2 macrophages by blocking the NF-κB pathway.
We thank Dr. Xian-Hui Li from the Department of Burns at the General Hospital of the Western Theater Command and Dr. Hao Yao from the Department of Hematology at the General Hospital of the Western Theater Command for their contributions to the study.
| 1. | Yu B, Zhang X, Li X. Exosomes derived from mesenchymal stem cells. Int J Mol Sci. 2014;15:4142-4157. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 624] [Cited by in RCA: 589] [Article Influence: 53.5] [Reference Citation Analysis (0)] |
| 2. | Mendt M, Kamerkar S, Sugimoto H, McAndrews KM, Wu CC, Gagea M, Yang S, Blanko EVR, Peng Q, Ma X, Marszalek JR, Maitra A, Yee C, Rezvani K, Shpall E, LeBleu VS, Kalluri R. Generation and testing of clinical-grade exosomes for pancreatic cancer. JCI Insight. 2018;3:e99263. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 346] [Cited by in RCA: 578] [Article Influence: 82.6] [Reference Citation Analysis (0)] |
| 3. | Marote A, Teixeira FG, Mendes-Pinheiro B, Salgado AJ. MSCs-Derived Exosomes: Cell-Secreted Nanovesicles with Regenerative Potential. Front Pharmacol. 2016;7:231. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 154] [Cited by in RCA: 212] [Article Influence: 23.6] [Reference Citation Analysis (0)] |
| 4. | Shi Q, Qian Z, Liu D, Sun J, Wang X, Liu H, Xu J, Guo X. GMSC-Derived Exosomes Combined with a Chitosan/Silk Hydrogel Sponge Accelerates Wound Healing in a Diabetic Rat Skin Defect Model. Front Physiol. 2017;8:904. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 151] [Cited by in RCA: 268] [Article Influence: 33.5] [Reference Citation Analysis (0)] |
| 5. | Shafei S, Khanmohammadi M, Heidari R, Ghanbari H, Taghdiri Nooshabadi V, Farzamfar S, Akbariqomi M, Sanikhani NS, Absalan M, Tavoosidana G. Exosome loaded alginate hydrogel promotes tissue regeneration in full-thickness skin wounds: An in vivo study. J Biomed Mater Res A. 2020;108:545-556. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 91] [Cited by in RCA: 173] [Article Influence: 28.8] [Reference Citation Analysis (0)] |
| 6. | Mol EA, Lei Z, Roefs MT, Bakker MH, Goumans MJ, Doevendans PA, Dankers PYW, Vader P, Sluijter JPG. Injectable Supramolecular Ureidopyrimidinone Hydrogels Provide Sustained Release of Extracellular Vesicle Therapeutics. Adv Healthc Mater. 2019;8:e1900847. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 34] [Cited by in RCA: 67] [Article Influence: 11.2] [Reference Citation Analysis (0)] |
| 7. | Goy RC, Morais ST, Assis OB. Evaluation of the antimicrobial activity of chitosan and its quaternized derivative on E. coli and S. aureus growth. Rev Bras Farmacogn. 2016;26:122-127. [RCA] [DOI] [Full Text] [Cited by in Crossref: 266] [Cited by in RCA: 254] [Article Influence: 28.2] [Reference Citation Analysis (0)] |
| 8. | Rangatchew F, Vester-Glowinski P, Rasmussen BS, Haastrup E, Munthe-Fog L, Talman ML, Bonde C, Drzewiecki KT, Fischer-Nielsen A, Holmgaard R. Mesenchymal stem cell therapy of acute thermal burns: A systematic review of the effect on inflammation and wound healing. Burns. 2021;47:270-294. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 31] [Cited by in RCA: 27] [Article Influence: 6.8] [Reference Citation Analysis (0)] |
| 9. | Tamama K, Kerpedjieva SS. Acceleration of Wound Healing by Multiple Growth Factors and Cytokines Secreted from Multipotential Stromal Cells/Mesenchymal Stem Cells. Adv Wound Care (New Rochelle). 2012;1:177-182. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 48] [Cited by in RCA: 63] [Article Influence: 4.8] [Reference Citation Analysis (0)] |
| 10. | Zhang J, Guan J, Niu X, Hu G, Guo S, Li Q, Xie Z, Zhang C, Wang Y. Exosomes released from human induced pluripotent stem cells-derived MSCs facilitate cutaneous wound healing by promoting collagen synthesis and angiogenesis. J Transl Med. 2015;13:49. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 381] [Cited by in RCA: 557] [Article Influence: 55.7] [Reference Citation Analysis (0)] |
| 11. | Zhang B, Wang M, Gong A, Zhang X, Wu X, Zhu Y, Shi H, Wu L, Zhu W, Qian H, Xu W. HucMSC-Exosome Mediated-Wnt4 Signaling Is Required for Cutaneous Wound Healing. Stem Cells. 2015;33:2158-2168. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 406] [Cited by in RCA: 575] [Article Influence: 57.5] [Reference Citation Analysis (0)] |
| 12. | Li X, Liu L, Yang J, Yu Y, Chai J, Wang L, Ma L, Yin H. Exosome Derived From Human Umbilical Cord Mesenchymal Stem Cell Mediates MiR-181c Attenuating Burn-induced Excessive Inflammation. EBioMedicine. 2016;8:72-82. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 356] [Cited by in RCA: 346] [Article Influence: 38.4] [Reference Citation Analysis (0)] |
| 13. | Hu L, Wang J, Zhou X, Xiong Z, Zhao J, Yu R, Huang F, Zhang H, Chen L. Exosomes derived from human adipose mensenchymal stem cells accelerates cutaneous wound healing via optimizing the characteristics of fibroblasts. Sci Rep. 2016;6:32993. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 296] [Cited by in RCA: 459] [Article Influence: 51.0] [Reference Citation Analysis (0)] |
| 14. | Ma T, Fu B, Yang X, Xiao Y, Pan M. Adipose mesenchymal stem cell-derived exosomes promote cell proliferation, migration, and inhibit cell apoptosis via Wnt/β-catenin signaling in cutaneous wound healing. J Cell Biochem. 2019;120:10847-10854. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 95] [Cited by in RCA: 198] [Article Influence: 33.0] [Reference Citation Analysis (0)] |
| 15. | Xiu C, Zheng H, Jiang M, Li J, Zhou Y, Mu L, Liu W. MSCs-Derived miR-150-5p-Expressing Exosomes Promote Skin Wound Healing by Activating PI3K/AKT Pathway through PTEN. Int J Stem Cells. 2022;15:359-371. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 1] [Cited by in RCA: 19] [Article Influence: 6.3] [Reference Citation Analysis (0)] |
| 16. | He X, Dong Z, Cao Y, Wang H, Liu S, Liao L, Jin Y, Yuan L, Li B. MSC-Derived Exosome Promotes M2 Polarization and Enhances Cutaneous Wound Healing. Stem Cells Int. 2019;2019:7132708. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 99] [Cited by in RCA: 260] [Article Influence: 43.3] [Reference Citation Analysis (0)] |
| 17. | Yi H, Wang Y, Yang Z, Xie Z. Efficacy assessment of mesenchymal stem cell transplantation for burn wounds in animals: a systematic review. Stem Cell Res Ther. 2020;11:372. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 10] [Cited by in RCA: 13] [Article Influence: 2.6] [Reference Citation Analysis (0)] |
| 18. | Zhou HY, Chen XG, Kong M, Liu CS. Preparation of chitosan‐based thermosensitive hydrogels for drug delivery. J Appl Polym Sci. 2009;112:1509-1515. [RCA] [DOI] [Full Text] [Cited by in Crossref: 25] [Cited by in RCA: 22] [Article Influence: 1.4] [Reference Citation Analysis (0)] |
| 19. | Guan P, Liu C, Xie D, Mao S, Ji Y, Lin Y, Chen Z, Wang Q, Fan L, Sun Y. Exosome-loaded extracellular matrix-mimic hydrogel with anti-inflammatory property Facilitates/promotes growth plate injury repair. Bioact Mater. 2022;10:145-158. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 16] [Cited by in RCA: 78] [Article Influence: 19.5] [Reference Citation Analysis (0)] |
| 20. | Fan L, Liu C, Chen X, Zheng L, Zou Y, Wen H, Guan P, Lu F, Luo Y, Tan G, Yu P, Chen D, Deng C, Sun Y, Zhou L, Ning C. Exosomes-Loaded Electroconductive Hydrogel Synergistically Promotes Tissue Repair after Spinal Cord Injury via Immunoregulation and Enhancement of Myelinated Axon Growth. Adv Sci (Weinh). 2022;9:e2105586. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 156] [Cited by in RCA: 201] [Article Influence: 67.0] [Reference Citation Analysis (0)] |
| 21. | Yang Q, Su S, Liu S, Yang S, Xu J, Zhong Y, Yang Y, Tian L, Tan Z, Wang J, Yu Z, Shi Z, Liang F. Exosomes-loaded electroconductive nerve dressing for nerve regeneration and pain relief against diabetic peripheral nerve injury. Bioact Mater. 2023;26:194-215. [RCA] [PubMed] [DOI] [Full Text] [Cited by in RCA: 33] [Reference Citation Analysis (0)] |
| 22. | Fan L, Guan P, Xiao C, Wen H, Wang Q, Liu C, Luo Y, Ma L, Tan G, Yu P, Zhou L, Ning C. Exosome-functionalized polyetheretherketone-based implant with immunomodulatory property for enhancing osseointegration. Bioact Mater. 2021;6:2754-2766. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 30] [Cited by in RCA: 87] [Article Influence: 21.8] [Reference Citation Analysis (0)] |