Published online Aug 14, 2022. doi: 10.3748/wjg.v28.i30.4044
Peer-review started: January 9, 2022
First decision: March 8, 2022
Revised: March 30, 2022
Accepted: July 19, 2022
Article in press: July 19, 2022
Published online: August 14, 2022
Processing time: 213 Days and 3.7 Hours
Langerhans cell histiocytosis (LCH) is a malignant disease of the histiocytes involving various organ systems. The spectrum of liver involvement in LCH ranges from mild transaminitis to end-stage liver disease. The hallmark of hepatic LCH is secondary sclerosing cholangitis, which manifests due to a progressive destruction of the biliary tree by malignant histiocytes. Chemotherapy remains the mainstay of treatment for active LCH. Early recognition, diagnosis and a systematic approach to the management of LCH can ameliorate the disease process. Nonetheless, the liver involvement in these patients may progress despite the LCH being in remission. Liver transplantation (LT) remains central in the management of such patients. Various facets of the management of LCH, espe
Core Tip: Involvement of the liver in Langerhans cell histiocytosis (LCH) is considered a high-risk disease and the management algorithm needs to factor in the malignancy and the severity of liver disease. Liver transplantation (LT) is usually offered to LCH patients in remission with decompensated liver disease. However, given the paucity of currently available literature, the role of LT in LCH remains undefined. There is hence a need for large collaborative, multicentre studies to provide recommendations on the management algorithm for LCH.
- Citation: Menon J, Rammohan A, Vij M, Shanmugam N, Rela M. Current perspectives on the role of liver transplantation for Langerhans cell histiocytosis: A narrative review. World J Gastroenterol 2022; 28(30): 4044-4052
- URL: https://www.wjgnet.com/1007-9327/full/v28/i30/4044.htm
- DOI: https://dx.doi.org/10.3748/wjg.v28.i30.4044
Langerhans cell histiocytosis (LCH) is a malignant disease of the histiocytes involving various organ systems including the liver, spleen, skin, bone, lymph nodes, lung, and the gastrointestinal tract[1]. Involvement of the liver in LCH occurs in many ways. The spectrum ranges from mild transaminitis to end-stage liver disease (ESLD). The hallmark of hepatic LCH is secondary sclerosing cholangitis (SSC), which manifests due to a progressive destruction of the biliary tree by malignant histiocytes[2]. Chemotherapy remains the mainstay of treatment for active LCH[3]. However, conventionally used vinblastine-based chemotherapy may not be tolerated by patients with concomitant liver involvement and modified regimens which balance the risk of liver decompensation with the oncological efficacy need to be instituted[3,4].
By the time the bile duct injury presents clinically, the primary disease is usually burnt out and the characteristic histiocytes are absent. ESLD and portal hypertension develop as a sequalae of SSC[5]. These patients are offered liver transplantation (LT) with an intent to cure the liver damage[6,7]. Various facets of the management of LCH, especially those with a liver involvement remain unclear. Furthermore, aspects of LT in LCH with regards to the indication, timing and post-LT management, including immunosuppression and adjuvant therapy, remain undefined. This review summaries the current evidence and discuss the practical aspects of the role of LT in the management of LCH.
Liver is involved in 20%-60% of patients with LCH and is more common when there is multiorgan involvement[2]. LCH-related liver disease is conventionally divided into two stages: Early infiltrative and late sclerosis[8]. The early stage is characterised by reactions elicited by the malignant histiocytes and other inflammatory cells, manifesting as periportal infiltration and hepatic nodular lesions. In the late fibrous stage, progressive destruction of biliary tree occurs resulting in cirrhosis and portal hypertension[8,9].
Hepatic involvement as defined by the European Consortium for Histiocytosis includes a palpable liver 3 cm below the coastal margin confirmed by ultrasound and liver dysfunction as defined by hyperbilirubinemia (at least 3 times the upper limit of normal) hypoalbuminemia (< 30 g/dL) , alanine transaminase (ALT) and/or aspartate transaminase (AST) (more than 3 times the upper limit of normal), -glutamyl transpeptidase (GGT) over twice the upper limit of normal, ascites, oedema, or intrahepatic nodular mass[3]. Sclerosing cholangitis in LCH is defined as involvement of extrahepatic/intrahepatic biliary tree with strictures, dilatation, pruning detected on imaging (computed tomography or magnetic resonance imaging) and/or on liver biopsy with or without elevated GGT[10,11]. The characteristic features of bile duct injury can be detected by magnetic resonance cholangiopancreatography. It is noteworthy that despite resolution of active malignancy, the biliary injury continues to progress. The major site of LCH involvement is the large bile duct, and hence sites of active disease may be missed on liver biopsy[8].
With a mortality risk of over three times that of patients without liver involvement, hepatic LCH is considered a high-risk disease. In a study of patients with LCH, liver involvement drastically reduced survival from 97% to 52%[12]. SSC may be observed in up to 20%, which invariably progress to ESLD requiring LT[13]. It is noteworthy that an acute hepatic involvement by active LCH may be reversible with timely instituted chemotherapy[12]. Later stages and/or burnt out disease on the other hand may continue to have worsening liver injury. Furthermore, this progression of SSC is more rapid than what is observed in primary sclerosing cholangitis[14]. In a series of adults with LCH, nearly 30% died due to complications of SSC and ESLD[14].
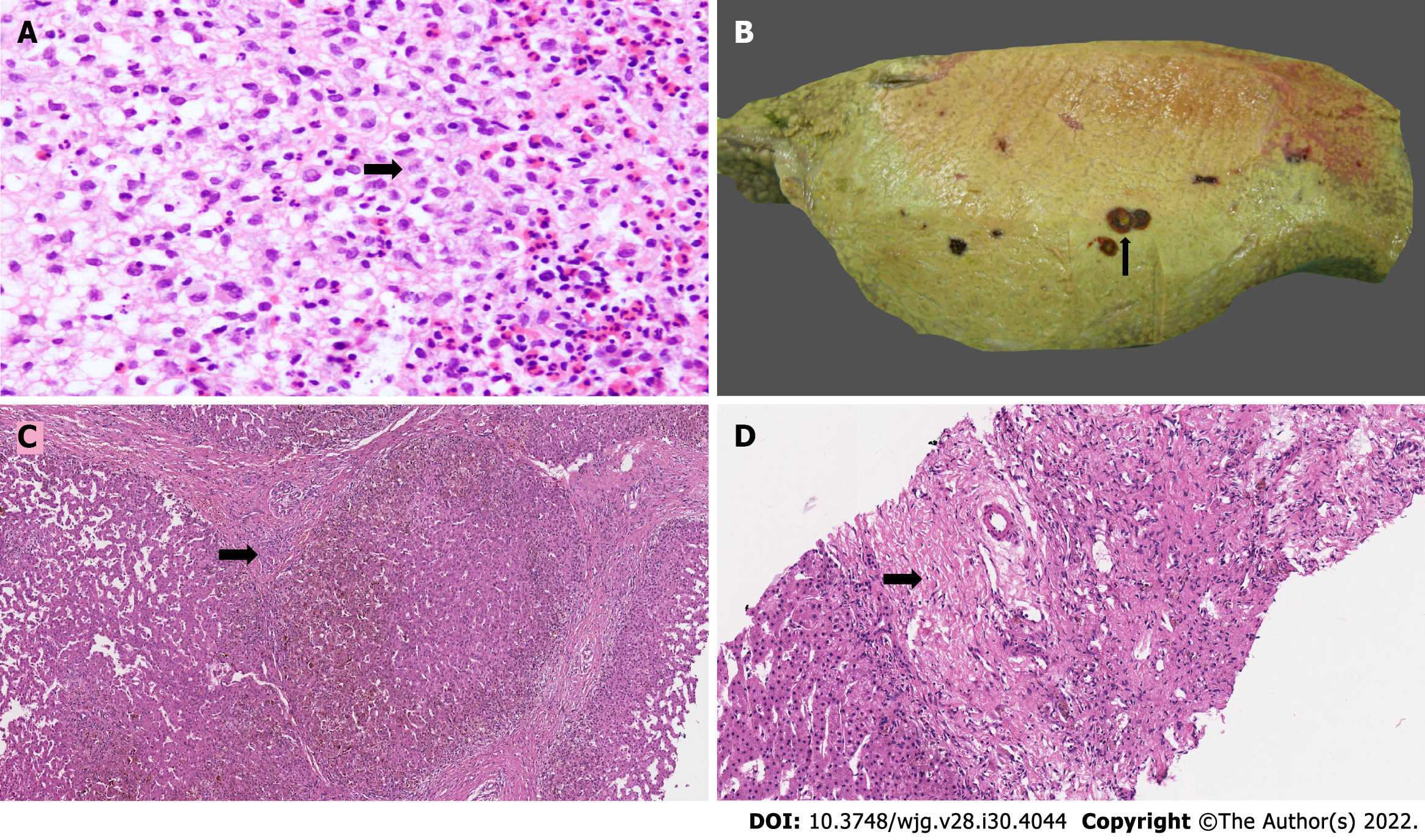
LCH exhibit diverse morphological features in the liver, which vary depending on the phase, stage and activity of the disease. Aggregates of LCs having lobulated, coffee-bean-shaped or contorted nuclei with a fine chromatin pattern and abundant eosinophilic cytoplasm admixed with eosinophils, polymorphs, lymphocytes, plasma cells, non-Langerhans histiocytes and rare multinucleated giant cells forming masses of variable sizes have been described (Figure 1A). Most cases with hepatic infiltration show marked tropism of the bile ducts. Active infiltrations of the bile ducts cause mucosal injury and fibrosis that lead to a progressive pattern of SSC, ultimately resulting in biliary cirrhosis[11,15,16]. There is destruction of the walls of bile ducts which may lead to cystic dilatation with intraluminal biliary sludge and rupture (Figure 1B). LCs can be identified within the basement membrane of the bile ducts, displacing the epithelial cells off the wall. Many cases demonstrate concentric periductal fibrosis. Cholangiopathy causes periportal bile ductular proliferation (Figure 1C), duct loss (Figure 1D), and features of chronic cholestasis with periportal cholestasis and stainable granules of copper and copper-binding protein. It is important to note that, despite evidence of biliary damage, the characteristic LCs may not be detected on liver biopsy. The diagnosis then rests on the presence of a concomitant infiltrate in other sites. Reassuringly, examination of liver explants in patients with LCH-induced biliary cirrhosis usually shows no evidence of active disease[17].
Liver, bone marrow and spleen are defined as the high-risk organs and their involvement makes the prognosis worse in patients with LCH[3]. The standard chemotherapy regimen for high-risk LCH as per the LCH IV protocol includes an induction phase of 6 wk with vinblastine (6 mg/m2) once weekly for 6 wk and prednisolone (40 mg/m2/d) daily for 4 wk, tapered over the next 2 wk[3,18]. LCH is invariably avid on positron emission tomography (PET), and presence of active disease can easily be ascertained. At the end of 6 wk, a PET scan is recommended, and the maintenance phase of therapy is commenced only after confirmed remission of LCH. Nonresponders conventionally receive another 6-wk cycle of the same regimen. If after the second cycle, remission is not achieved, second-line salvage chemotherapeutic agents are used[3,18].
Maintenance-phase therapy consists of vinblastine (6 mg/m2) given once in 3 wk along with prednisolone (40 mg/m2/d) for 5 d in the same week for 1 year. A few other drugs have been tried with doubtful benefit. These include methotrexate, fludarabine, 6-mercaptopurine, cyclophosphamide and etoposide[17,19]. Clinical trials have shown that newer targeted agents like BRAF inhibitors (see below) have shown promise in achieving remission in patients with refractory disease[20].
In contrast to other hepatic malignancies like hepatoblastomas or haematological cancers, hepatic involvement in LCH is unique. While the latter result in cirrhosis progressing to ESLD, the former do not. Hence management of LCH with liver involvement needs a meticulous balance of two major decision domains, i.e., malignancy and ESLD.
In patients who have normal liver function, standard chemotherapeutic regimens (vinca alkaloids and steroids) can be safely used. However, in those with features of decompensated liver disease (DCLD) and/or synthetic failure, these drugs can result in serious adverse effects. Vinblastine is inherently hepatotoxic and can worsen liver dysfunction[16,21,22]. Also, since vinblastine is metabolised in the liver, a failing liver could lead to severe drug-related toxicity (peripheral neuropathy, myelosuppression, etc.)[16,21,22]. Nonetheless, a delay in initiating chemotherapy for the fear of compromising liver function potentially worsens the malignant process. Deferring LT may result in morbidity and morbidity from cirrhosis and decompensation[13]. However, premature LT in the presence of active malignancy is not a standard recommended practice, and risks recurrent LCH in the graft.
The oncologist and the transplant clinician are faced with four clinical scenarios in patients who have LCH with liver involvement. These include patients with CLD and LCH in remission, patients with DCLD and LCH in remission, those with CLD and active LCH, and lastly, those with DCLD and active LCH.
These are a subset of patients who have completed chemotherapy and have the disease in remission. As mentioned above, they could still have progressive liver disease as sequalae of LCH. It could be many years before they present with symptoms of liver disease, which may be in the form of cirrhosis and/portal hypertension[23]. LT is offered to these patients independent of their previous history of LCH. Apart from decompensation, other indications for LT in these patients include, portal hypertension, intractable pruritus and growth retardation. Nonetheless, irrespective of the lag-time between LCH remission and LT, prior to LT, a PET scan is performed to confirm the absence of active LCH.
When indicated, LT may be performed in these patients. Evidence shows that the post-LT outcomes including disease-free survival in these patients is similar to those who have undergone LT for other indications[7].
Standard chemotherapeutic regimens (vinca alkaloid based) directed at LCH treatment should be initiated after appropriate staging workup for the disease. If the liver does not show signs of decompensation, patients should complete the full course of chemotherapy (induction and maintenance)[3]. They should however remain on close follow-up for LCH remission, signs of worsening of the liver disease and consequently the need for LT. The total duration of 12 mo chemotherapy is recommended in patients with LCH and an LT is offered after a minimum interval of 3 wk, when indicated[3]. The need for adjuvant chemotherapy in these patients is debatable, and a decision is usually made on a case-by-case basis. There may be a situation wherein the liver decompensates after initiating chemotherapy. In these patients, the severity of liver decompensation and the state of LCH remission guides further course of management.
In patients where the liver decompensation can be managed supportively, chemotherapy is continued with an aim to achieve remission. In this case however, the chemotherapy needs to be switched to relatively nonhepatoxic agents like cytarabine[24]. Once remission is confirmed following induction chemotherapy, these patients may be offered LT. It is nonetheless imperative to complete the full chemotherapeutic course. A chemotherapy-free interval of at least 3 wk is recommended prior to LT. This helps reduce the risk of chemotherapy-related damage to the liver allograft. Evidence is divisive with regards to the timing, dosing and regimen of post-LT chemotherapy. It is nonetheless unequivocally clear that the maintenance phase of chemotherapy needs to be completed. The agents are reintroduced usually 3 wk following LT. This allows for the transplanted liver to cope with the toxic agents better. Furthermore, it is advisable to reintroduce vinca alkaloids when the bilirubin is near normal (< 3 mg/dL)[22].
Striking the balance between achieving remission with chemotherapy, abating worsening liver dysfunction and postponing LT remains a challenge in the management of these patients. The prudent approach would be to attempt achieving remission with relatively nonhepatotoxic, low-dose chemotherapy. Should this be achieved, as mentioned above, LT may be offered following a negative PET scan. A rarer and unfortunate situation is when a patient with active LCH requires LT. While LT may not be justified as per protocol, these are exceptional situations requiring a multi-disciplinary team decision.
LCH is a malignancy; therefore, before offering these patients LT, the ethical aspects need to be debated. Immunosuppressants mute the native immune system’s antitumour action, potentially resulting in a higher risk of recurrence. Furthermore, the ideal latency period between disease remission and LT remains undefined. There are also data to suggest that in patients with DCLD, delaying LT may result in worse outcomes[9]. Hence, for selected patients with DCLD, potential benefit and risk must be evaluated on an individual basis[25]. It does however, seem reasonable to indicate LT only when the oncological probability of survival disregarding immunosuppression is at least 50% at 5 years. The caveat in these cases is that the LCH should be amenable to sustained remission prior to LT and there should always remain the option of post-LT adjuvant therapy. Living donor LT (LDLT), especially in regions where deceased donor LT (DDLT) is uncommonly performed, permits optimal timing of the LT and avoidance of delays between the end of chemotherapy and LT.
LCH is a relatively rare oncological condition, and despite advances in medical oncology, there are few approved chemotherapeutic agents and even fewer clinical trials in this regard. An ethical dilemma also arises when despite chemotherapy, disease remission cannot be achieved in patients with DCLD. This subgroup of patients along with those who have active LCH and urgently require LT are outliers who need to have their management tailored according to the disease state; ideally in a clinical trial setting.
Nonetheless, prior to offering LT, it is imperative to satisfy the time-tested tenets of justice, utility and normal feasance for DDLT, and that of balancing the donor risk versus recipient benefit (principle of double equipoise) in living donor LT.
Paediatric transplant oncology is a niche area that aims at optimising post-LT oncological outcomes. Given that LCH is a rare oncological condition, the literature remains sparse. Earlier series of LCH noted a recurrence of 30%-55% with a patient survival of 60%–67%[26,27]. With advances in immunosuppression, chemotherapeutic agents, and a better understanding of the oncopathology, more recent series including ours, have shown remarkably improved outcomes which are on par with LT for other indications[7,28] (Table 1). In a recent meta-analysis of 60 LT recipients with LCH, the 1-year, 3-year and 5-year patient survival rates were 86.6%, 82.4%, and 82.4%, respectively[25]. Furthermore, a cumulative recurrence of LCH in the grafts was 8%[25].
| Author (year) | No. of patients | Age at diagnosis (mo) | Age at transplant (mo) | Immunosuppression (No. of patients) | Surgical complications (No. of patients) | Medical complications (No. of patients) | Survival (follow-up in mo) |
| Stieber (1990) | 3 (1adult/2 paediatric) | - | - | Cyclosporine, steroid | - | Recurrent rejection (3); retransplant (2) | 66% (1 adult died of long bone fracture) |
| Whitington (1992) | 2 (paediatric) | 36 & 19 | 60 & 30 | Cyclosporine, steroid | Roux-en Y anastomotic leak | CMV hepatitis (2); rejection (2); GI bleed (1) | 100% (30-34) |
| Zandi (1995) | 5 (paediatric) | 23 ± 13 | 151 ± 43 | Cyclosporine, steroid, azathioprine (3); cyclosporine, steroid (1); OKT3, steroid (1) | - | Rejection (4); CMV infection (2); GI bleed (1); Kidney injury (1) | 60% (0.25-88.00) |
| Newell (1997) | 6 (paediatric) | 15 (12-30) | 36 | Cyclosporine, steroid, azathioprine (6) | Nil | PTLD (4); rejection (6); retransplantation (4); recurrence (2) | 67% (24-74) |
| Hazdic (2000) | 2 (paediatric) | 16 & 17 | 34 & 14 | Cyclosporine, steroid, MMF (1); tacrolimus, steroid (1) | Bowel perforation due to PTLD (1); PVT (1) | Recurrence (2); PTLD (1); rejection (2) | 100% (5 & 60) |
| Braier (2002) | 5 (paediatric) | - | - | Cyclosporine, steroid, azathioprine (5) | HAT & Retransplant (1) | CMV (1); rejection (1) | 60% (14-37) |
| Chen (2020) | 5 (paediatric) | 15 (13-28) | 53 (24-81) | Tacrolimus, steroid, MMF (5) | HAT (1) | EBV (4), CMV (1); LCH recurrence (1); DILI (1) | 100%: 32 (2-67) |
| Our experience | 6 (paediatric) | 25 (9-48) | 52.5 (33-204) | Tacrolimus, steroid (6) | Nil | Nil | 100%: 36 (18-80) |
We offer LT in patients with acute decompensation, portal hypertension with sclerosing cholangitis or growth retardation or intractable pruritus. If any patient decompensates in between chemotherapy, a LT is offered, following which the chemotherapy cycles are completed. A 18-fluorodeoxyglucose (FDG)-PET scan is mandatorily done before considering a LT in these patients. Patients who do not tolerate the full course of chemotherapy are also considered for LT during mid-cycle if the FDG-PET shows disease in remission. An interval of 2-3 wk is considered between the chemotherapy cycle and LT. This is followed by restarting of chemotherapy (if the cycles are not completed prior to surgery) after 3 wk.
Our unit’s protocol includes the use of standard chemotherapy regimen in patients who are likely to withstand the full course. We use a modified chemotherapy in patients with liver dysfunction. This regimen includes low-dose cytarabine (100 mg/m2) every 3 wk along with prednisolone (40 mg/m2/d) daily for 4 wk tapered over the next 2 wk (each cycle is of 6 wk duration). Maintenance chemotherapy includes the same dose of cytarabine and prednisolone as the standard regimen, administered for 1 year. In our experience of six patients who underwent a LDLT, all are alive after mean follow up 36 mo (18-80 mo).
Patients transplanted for LCH are known to have a higher incidence of rejections and post-transplant lymphoproliferative disorder (PTLD) (0%-67%)[7,8,27]. A purported theory is that of increased release of proinflammatory cytokines from the neoplastic cells predisposing to rejection. There remain concerns that immunosuppression could potentially increase risk an early recurrence of the index cancer, making LT futile. Hence the relative hesitancy at increasing immunosuppression in the early experiences of LT in LCH. There is no evidence till for the role of the mechanistic target of rapamycin inhibitors like sirolimus for preventing the occurrence of PTLD in these patients.
There is some encouraging evidence on the use of targeted therapy in LCH refractory to standard chemotherapy. BRAF gene mutations are observed in up to 67% of patients with LCH[29]. Vemurafenib and dabrafenib are two such agents that have shown potential in the clinical trial settings. Vemurafenib showed 83% response rate in refractory LCH[30]. In another trial of children with refractory LCH, 65% showed a remission with dabrafenib[31]. While vemurafenib may be hepatotoxic, dabrafenib is safe in patients with liver disease[32]. The authors concluded, that these agents may be used as the second- or third-line therapy for patients with refractory LCH and DCLD.
Another target for such novel therapy is the MEK1 mutation. It is observed in 19% of patients with LCH. Cobimetinib and trametinib are MEK1 inhibitors, and have shown some promise in clinical trials[33]. There however, remains a need for further research especially in those with concomitant liver disease to assess the true efficacy of these novel agents.
There have been studies evaluating the role of PD-1 (programmed cell death-1)/PD-L1 (programmed cell death ligand-1) check points in pathogenesis of LCH, especially when it involves the musculoskeletal system[34]. Hence anti PD-1 immunotherapy (pembrolizumab/nivolumab) may have a role in treating patients of LCH if there is an active musculoskeletal disease[35,36]. The role of anti-PD-1-based immunotherapy in the post-LT population remains a matter of intense debate. Given that these drugs act primarily by potentiating the native immune response, there have been studies demonstrating a high likelihood of rejections and allograft loss[37]. Therefore, the indications of anti PDL-1 therapy in the LCH patients who are likely to need LT remains undefined.
Early recognition, diagnosis and a systematic approach to the management of LCH can ameliorate the disease process. Nonetheless, the liver involvement in these patients may progress despite the LCH in remission. LT remains the mainstay in the management of such patients. Over the last decade significant advances in immunosuppression protocols, and availability of effective chemotherapeutic agents, have translated into better long-term allograft and recurrence-free patient survival. The post-LT outcomes are now comparable with those for other indications. However, given the rarity of the disease and the paucity of currently available literature, there is a need for large collaborative international, multicentre, society-based studies to provide recommendations on evidence-based algorithm for LCH, especially in patients with liver involvement.
Provenance and peer review: Invited article; Externally peer reviewed.
Peer-review model: Single blind
Specialty type: Transplantation
Country/Territory of origin: India
Peer-review report’s scientific quality classification
Grade A (Excellent): A
Grade B (Very good): 0
Grade C (Good): C, C
Grade D (Fair): 0
Grade E (Poor): 0
P-Reviewer: Guo WZ, China; Hashimoto K, Japan; Saito R, Japan S-Editor: Chen YL L-Editor: Kerr C P-Editor: Chen YL
| 1. | Allen CE, Merad M, McClain KL. Langerhans-Cell Histiocytosis. N Engl J Med. 2018;379:856-868. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 250] [Cited by in RCA: 348] [Article Influence: 49.7] [Reference Citation Analysis (0)] |
| 2. | Yi X, Han T, Zai H, Long X, Wang X, Li W. Liver involvement of Langerhans' cell histiocytosis in children. Int J Clin Exp Med. 2015;8:7098-7106. [PubMed] |
| 3. | Haupt R, Minkov M, Astigarraga I, Schäfer E, Nanduri V, Jubran R, Egeler RM, Janka G, Micic D, Rodriguez-Galindo C, Van Gool S, Visser J, Weitzman S, Donadieu J; Euro Histio Network. Langerhans cell histiocytosis (LCH): guidelines for diagnosis, clinical work-up, and treatment for patients till the age of 18 years. Pediatr Blood Cancer. 2013;60:175-184. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 394] [Cited by in RCA: 417] [Article Influence: 34.8] [Reference Citation Analysis (0)] |
| 4. | Floyd J, Mirza I, Sachs B, Perry MC. Hepatotoxicity of chemotherapy. Semin Oncol. 2006;33:50-67. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 179] [Cited by in RCA: 187] [Article Influence: 9.8] [Reference Citation Analysis (0)] |
| 5. | Fu Z, Li H, Arslan ME, Ells PF, Lee H. Hepatic Langerhans cell histiocytosis: A review. World J Clin Oncol. 2021;12:335-341. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in CrossRef: 4] [Cited by in RCA: 14] [Article Influence: 3.5] [Reference Citation Analysis (0)] |
| 6. | Stieber AC, Sever C, Starzl TE. Liver transplantation in patients with Langerhans' cell histiocytosis. Transplantation. 1990;50:338-340. [PubMed] |
| 7. | Chen C, Gu G, Zhou T, Huang M, Xia Q. Combination of Neoadjuvant Therapy and Liver Transplantation in Pediatric Multisystem Langerhans Cell Histiocytosis With Liver Involvement. Front Oncol. 2020;10:566987. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 2] [Cited by in RCA: 3] [Article Influence: 0.6] [Reference Citation Analysis (0)] |
| 8. | Jaffe R. Liver involvement in the histiocytic disorders of childhood. Pediatr Dev Pathol. 2004;7:214-225. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 46] [Cited by in RCA: 48] [Article Influence: 2.3] [Reference Citation Analysis (0)] |
| 9. | Braier J, Ciocca M, Latella A, de Davila MG, Drajer M, Imventarza O. Cholestasis, sclerosing cholangitis, and liver transplantation in Langerhans cell Histiocytosis. Med Pediatr Oncol. 2002;38:178-182. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 87] [Cited by in RCA: 72] [Article Influence: 3.1] [Reference Citation Analysis (0)] |
| 10. | Haas S, Theuerkauf I, Kühnen A, Wickesberg A, Fischer HP. [Langerhans' cell histiocytosis of the liver. Differential diagnosis of a rare chronic destructive sclerosing cholangitis]. Pathologe. 2003;24:119-123. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 5] [Cited by in RCA: 5] [Article Influence: 0.2] [Reference Citation Analysis (0)] |
| 11. | Kaplan KJ, Goodman ZD, Ishak KG. Liver involvement in Langerhans' cell histiocytosis: a study of nine cases. Mod Pathol. 1999;12:370-378. [PubMed] |
| 12. | Henter JI, Tondini C, Pritchard J. Histiocyte disorders. Crit Rev Oncol Hematol. 2004;50:157-174. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 117] [Cited by in RCA: 116] [Article Influence: 5.5] [Reference Citation Analysis (0)] |
| 13. | Khanna R, Pawaria A, Alam S, Rawat D. Liver Transplantation in LCH: Risk Reactivation or Wait Till Decompensation? J Pediatr Hematol Oncol. 2016;38:664-665. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 1] [Cited by in RCA: 1] [Article Influence: 0.1] [Reference Citation Analysis (0)] |
| 14. | Abdallah M, Généreau T, Donadieu J, Emile JF, Chazouillères O, Gaujoux-Viala C, Cabane J. Langerhans' cell histiocytosis of the liver in adults. Clin Res Hepatol Gastroenterol. 2011;35:475-481. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 35] [Cited by in RCA: 39] [Article Influence: 2.8] [Reference Citation Analysis (0)] |
| 15. | Li H, Ells P, Arslan ME, Robstad KA, Lee H. Hepatic Langerhans Cell Histiocytosis (LCH) Presenting as a Harbinger of Multisystem LCH. Cureus. 2020;12:e8591. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 2] [Cited by in RCA: 5] [Article Influence: 1.0] [Reference Citation Analysis (0)] |
| 16. | Tang Y, Zhang Z, Chen M, Ju W, Wang D, Ji F, Ren Q, Guo Z, He X. Severe sclerosing cholangitis after Langerhans cell histiocytosis treated by liver transplantation: An adult case report. Medicine (Baltimore). 2017;96:e5994. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 9] [Cited by in RCA: 13] [Article Influence: 1.6] [Reference Citation Analysis (0)] |
| 17. | Jezierska M, Stefanowicz J, Romanowicz G, Kosiak W, Lange M. Langerhans cell histiocytosis in children - a disease with many faces. Recent advances in pathogenesis, diagnostic examinations and treatment. Postepy Dermatol Alergol. 2018;35:6-17. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 50] [Cited by in RCA: 74] [Article Influence: 10.6] [Reference Citation Analysis (0)] |
| 18. | Allen CE, Ladisch S, McClain KL. How I treat Langerhans cell histiocytosis. Blood. 2015;126:26-35. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 119] [Cited by in RCA: 135] [Article Influence: 13.5] [Reference Citation Analysis (0)] |
| 19. | Aricò M. Langerhans cell histiocytosis in children: from the bench to bedside for an updated therapy. Br J Haematol. 2016;173:663-670. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 38] [Cited by in RCA: 40] [Article Influence: 4.4] [Reference Citation Analysis (0)] |
| 20. | Abla O, Weitzman S. Treatment of Langerhans cell histiocytosis: role of BRAF/MAPK inhibition. Hematology Am Soc Hematol Educ Program. 2015;2015:565-570. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 24] [Cited by in RCA: 31] [Article Influence: 3.1] [Reference Citation Analysis (0)] |
| 21. | Grigorian A, O'Brien CB. Hepatotoxicity Secondary to Chemotherapy. J Clin Transl Hepatol. 2014;2:95-102. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 32] [Cited by in RCA: 75] [Article Influence: 6.8] [Reference Citation Analysis (0)] |
| 22. | Bahirwani R, Reddy KR. Drug-induced liver injury due to cancer chemotherapeutic agents. Semin Liver Dis. 2014;34:162-171. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 38] [Cited by in RCA: 53] [Article Influence: 4.8] [Reference Citation Analysis (0)] |
| 23. | Bansal D, Marwaha RK, Trehan A, Poddar U, Radotra BD. Portal hypertension secondary to Langerhans cell histiocytosis. Indian J Gastroenterol. 2001;20:201-202. [PubMed] |
| 24. | Simko SJ, McClain KL, Allen CE. Up-front therapy for LCH: is it time to test an alternative to vinblastine/prednisone? Br J Haematol. 2015;169:299-301. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 30] [Cited by in RCA: 37] [Article Influence: 3.4] [Reference Citation Analysis (0)] |
| 25. | Ziogas IA, Kakos CD, Wu WK, Montenovo MI, Matsuoka LK, Zarnegar-Lumley S, Alexopoulos SP. Liver Transplantation for Langerhans Cell Histiocytosis: A US Population-Based Analysis and Systematic Review of the Literature. Liver Transpl. 2021;27:1181-1190. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 4] [Cited by in RCA: 4] [Article Influence: 1.0] [Reference Citation Analysis (0)] |
| 26. | Zandi P, Panis Y, Debray D, Bernard O, Houssin D. Pediatric liver transplantation for Langerhans' cell histiocytosis. Hepatology. 1995;21:129-133. [PubMed] |
| 27. | Newell KA, Alonso EM, Kelly SM, Rubin CM, Thistlethwaite JR Jr, Whitington PF. Association between liver transplantation for Langerhans cell histiocytosis, rejection, and development of posttransplant lymphoproliferative disease in children. J Pediatr. 1997;131:98-104. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 45] [Cited by in RCA: 37] [Article Influence: 1.3] [Reference Citation Analysis (0)] |
| 28. | Hadzic N, Pritchard J, Webb D, Portmann B, Heaton ND, Rela M, Dhawan A, Baker AJ, Mieli-Vergani G. Recurrence of Langerhans cell histiocytosis in the graft after pediatric liver transplantation. Transplantation. 2000;70:815-819. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 45] [Cited by in RCA: 47] [Article Influence: 1.9] [Reference Citation Analysis (0)] |
| 29. | Feng S, Han L, Yue M, Zhong D, Cao J, Guo Y, Sun Y, Zhang H, Cao Z, Cui X, Liu R. Frequency detection of BRAF V600E mutation in a cohort of pediatric langerhans cell histiocytosis patients by next-generation sequencing. Orphanet J Rare Dis. 2021;16:272. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 5] [Cited by in RCA: 13] [Article Influence: 3.3] [Reference Citation Analysis (0)] |
| 30. | Donadieu J, Larabi IA, Tardieu M, Visser J, Hutter C, Sieni E, Kabbara N, Barkaoui M, Miron J, Chalard F, Milne P, Haroche J, Cohen F, Hélias-Rodzewicz Z, Simon N, Jehanne M, Kolenova A, Pagnier A, Aladjidi N, Schneider P, Plat G, Lutun A, Sonntagbauer A, Lehrnbecher T, Ferster A, Efremova V, Ahlmann M, Blanc L, Nicholson J, Lambilliote A, Boudiaf H, Lissat A, Svojgr K, Bernard F, Elitzur S, Golan M, Evseev D, Maschan M, Idbaih A, Slater O, Minkov M, Taly V, Collin M, Alvarez JC, Emile JF, Héritier S. Vemurafenib for Refractory Multisystem Langerhans Cell Histiocytosis in Children: An International Observational Study. J Clin Oncol. 2019;37:2857-2865. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 128] [Cited by in RCA: 140] [Article Influence: 23.3] [Reference Citation Analysis (0)] |
| 31. | Yang Y, Wang D, Cui L, Ma HH, Zhang L, Lian HY, Zhang Q, Zhao XX, Zhang LP, Zhao YZ, Li N, Wang TY, Li ZG, Zhang R. Effectiveness and Safety of Dabrafenib in the Treatment of 20 Chinese Children with BRAFV600E-Mutated Langerhans Cell Histiocytosis. Cancer Res Treat. 2021;53:261-269. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 6] [Cited by in RCA: 24] [Article Influence: 4.8] [Reference Citation Analysis (0)] |
| 32. | Spengler EK, Kleiner DE, Fontana RJ. Vemurafenib-induced granulomatous hepatitis. Hepatology. 2017;65:745-748. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 19] [Cited by in RCA: 25] [Article Influence: 3.1] [Reference Citation Analysis (0)] |
| 33. | Diamond EL, Durham BH, Ulaner GA, Drill E, Buthorn J, Ki M, Bitner L, Cho H, Young RJ, Francis JH, Rampal R, Lacouture M, Brody LA, Ozkaya N, Dogan A, Rosen N, Iasonos A, Abdel-Wahab O, Hyman DM. Efficacy of MEK inhibition in patients with histiocytic neoplasms. Nature. 2019;567:521-524. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 230] [Cited by in RCA: 259] [Article Influence: 43.2] [Reference Citation Analysis (0)] |
| 34. | Hashimoto K, Nishimura S, Sakata N, Inoue M, Sawada A, Akagi M. Characterization of PD-1/PD-L1 immune checkpoint expression in the pathogenesis of musculoskeletal Langerhans cell histiocytosis: A retrospective study. Medicine (Baltimore). 2021;100:e27650. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 3] [Cited by in RCA: 8] [Article Influence: 2.0] [Reference Citation Analysis (0)] |
| 35. | Hashimoto K, Nishimura S, Sakata N, Inoue M, Sawada A, Akagi M. Treatment Outcomes of Langerhans Cell Histiocytosis: A Retrospective Study. Medicina (Kaunas). 2021;57. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 3] [Cited by in RCA: 10] [Article Influence: 2.5] [Reference Citation Analysis (0)] |
| 36. | Hashimoto K, Nishimura S, Ito T, Akagi M. Characterization of PD-1/PD-L1 immune checkpoint expression in soft tissue sarcomas. Eur J Histochem. 2021;65. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 10] [Cited by in RCA: 11] [Article Influence: 2.8] [Reference Citation Analysis (0)] |
| 37. | Rammohan A, Reddy MS, Farouk M, Vargese J, Rela M. Pembrolizumab for metastatic hepatocellular carcinoma following live donor liver transplantation: The silver bullet? Hepatology. 2018;67:1166-1168. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 42] [Cited by in RCA: 50] [Article Influence: 7.1] [Reference Citation Analysis (0)] |