Published online Sep 28, 2020. doi: 10.3748/wjg.v26.i36.5450
Peer-review started: May 18, 2020
First decision: July 29, 2020
Revised: August 7, 2020
Accepted: September 8, 2020
Article in press: September 8, 2020
Published online: September 28, 2020
Processing time: 128 Days and 22.1 Hours
The undifferentiated-type (UDT) component profoundly affects the clinical course of early gastric cancers (EGCs). However, an accurate preoperative diagnosis of the histological types is unsatisfactory. To date, few studies have investigated whether the UDT component within mixed-histological-type (MT) EGCs can be recognized preoperatively.
To clarify the histopathological characteristics of the endoscopically-resected MT EGCs for investigating whether the UDT component could be recognized preoperatively.
This was a single-center retrospective study. First, we attempted to clarify the histopathological characteristics of the endoscopically-resected MT EGCs with emphasis on the UDT component. Histopathological examination investigated each lesion’s UDT component: (1) Whole mucosal layer occupation of the UDT component; (2) UDT component exposure to the surface of the mucosa; and (3) existence of a clear border between the differentiated-type and UDT components. Then, preoperative endoscopic images with magnifying endoscopy with narrow-band imaging (ME-NBI) were examined to identify whether the endoscopic UDT component finding was recognizable within the area where it was present in the histopathological examination. The preoperative biopsy results and comparative relationships between endoscopic and histopathological findings were also examined.
In the histopathological examination, the whole mucosal layer occupation of the UDT component and exposure of the UDT component to the mucosal surface were observed in 67.3% (33/49) and 79.6% (39/49) of samples, respectively. A clear distinction of the border between the differentiated-type and UDT components could not be drawn in 65.3% (32/49) of MT lesions. In the endoscopic examination, the preoperative endoscopic images showed that only 24.5% (12/49) of MT EGCs revealed the UDT component within the area where it was present histopathologically. Histopathological UDT predominance was the single significant factor associated with the presence of the endoscopic UDT component finding (61.5% vs 11.1%, P = 0.0009). Only 26.5% (13/49) of the lesions were diagnosed from the pretreatment biopsy as having a UDT component. Combined results of the pretreatment biopsy and ME-NBI showed the preoperative presence of the UDT component in 40.8% (20/49) of MT EGCs.
Recognition of a UDT component within MT EGCs is difficult even when pretreatment biopsy and ME-NBI are combined. Endoscopic resection plays a significant role in both treatment and diagnosis.
Core Tip: Accurate pretreatment diagnosis of the undifferentiated-type (UDT) component was hardly achieved even when pretreatment biopsy and diagnostic endoscopy were combined. Histopathological UDT predominance was the single significant factor associated with the presence of the endoscopic UDT component finding. The possibility of UDT predominance should be suspected when a lesion shows an endoscopic finding of the UDT component. Endoscopic resection plays a significant role in both the diagnosis and treatment of mixed-histological-type early gastric cancers.
- Citation: Ozeki Y, Hirasawa K, Kobayashi R, Sato C, Tateishi Y, Sawada A, Ikeda R, Nishio M, Fukuchi T, Makazu M, Taguri M, Maeda S. Histopathological validation of magnifying endoscopy for diagnosis of mixed-histological-type early gastric cancer. World J Gastroenterol 2020; 26(36): 5450-5462
- URL: https://www.wjgnet.com/1007-9327/full/v26/i36/5450.htm
- DOI: https://dx.doi.org/10.3748/wjg.v26.i36.5450
Endoscopic submucosal dissection (ESD) has made it possible to treat early gastric cancers less invasively[1,2]. ESD is currently performed according to the guidelines issued by the Japanese Gastric Cancer Association[3]. Principal indications for endoscopic treatment are tumors with a very low possibility of lymph node metastasis, established by estimation with a large number of surgical cases[4-6]. The Japanese Classification of Gastric Carcinoma[7] classifies gastric cancers into two major histological types, differentiated-type (DT) and undifferentiated-type (UDT). Gastric cancers that contain a mixture of DT and UDT components are called mixed-histological-type (MT) and are classified according to the predominant histological type in the above classification and guidelines[3,7]. For UDT early gastric cancers, the resection is classified as curative with en bloc resection, tumor size ≤ 2 cm, pT1a, without ulceration, negative horizontal margin, negative vertical margin, and no lymphovascular infiltration. Since UDT-predominant MT gastric cancers are regarded as UDT gastric cancers and are, hence, subject to stricter criteria for endoscopic curative resection[8], it is important to accurately diagnose the UDT component before making treatment decisions. Moreover, the presence of the UDT component is an independent risk factor for non-curative endoscopic resection, even in DT-predominant MT gastric cancers[9-11]. However, few studies have investigated whether the UDT component within MT early gastric cancers can be recognized preoperatively. Thus, the present study investigated the feasibility of preoperative diagnosis of the UDT component within MT early gastric cancers. First, we attempted to clarify the histopathological characteristics of the endoscopically-resected MT early gastric cancers with emphasis on the UDT component. Then, we reviewed preoperative endoscopic images, including magnifying endoscopy with narrow-band imaging (ME-NBI), emphasizing the area where the UDT component was present in the histopathological examination. Finally, we analyzed the comparative relationships between the preoperative endoscopic findings and the histopathological findings to investigate whether the UDT component could be recognized preoperatively.
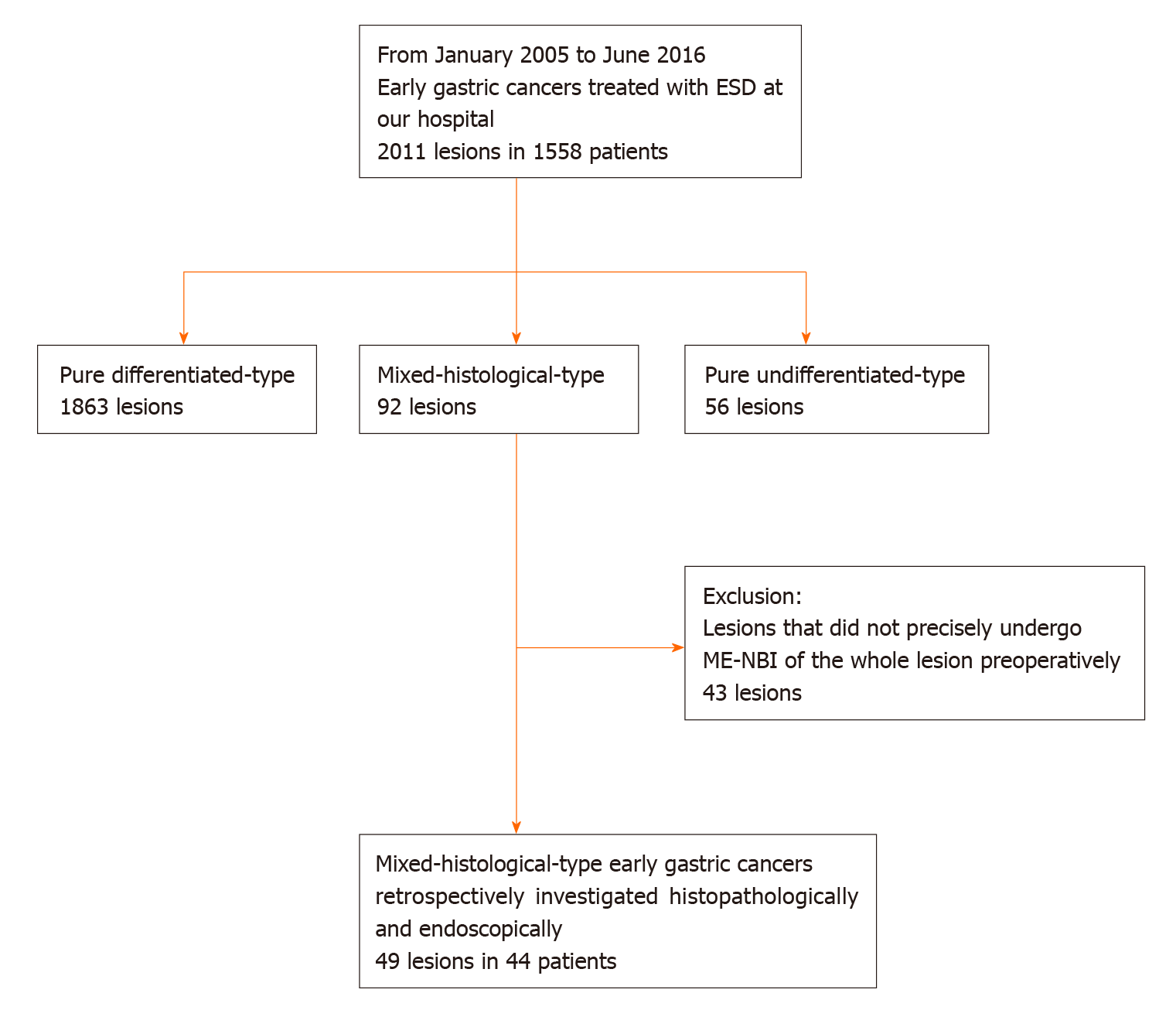
This was a single-center retrospective study. Figure 1 shows the flowchart of patient recruitment. We treated 2011 consecutive early gastric cancers in 1558 patients with ESD between January 2005 and June 2016 at the Yokohama City University Medical Center. Of these, 92 lesions in 87 patients were histopathologically diagnosed as MT gastric cancers after ESD. After excluding lesions that did not precisely undergo ME-NBI of the whole lesion before treatment, we analyzed 49 MT gastric cancers in 44 patients. Preoperative endoscopic images and postoperative histopathological slides were reviewable for all lesions. All patients were fully informed about the treatment and histopathological examination and provided consent. Our institutional review board approved the study, which was performed in accordance with the 1964 Declaration of Helsinki and later revisions.
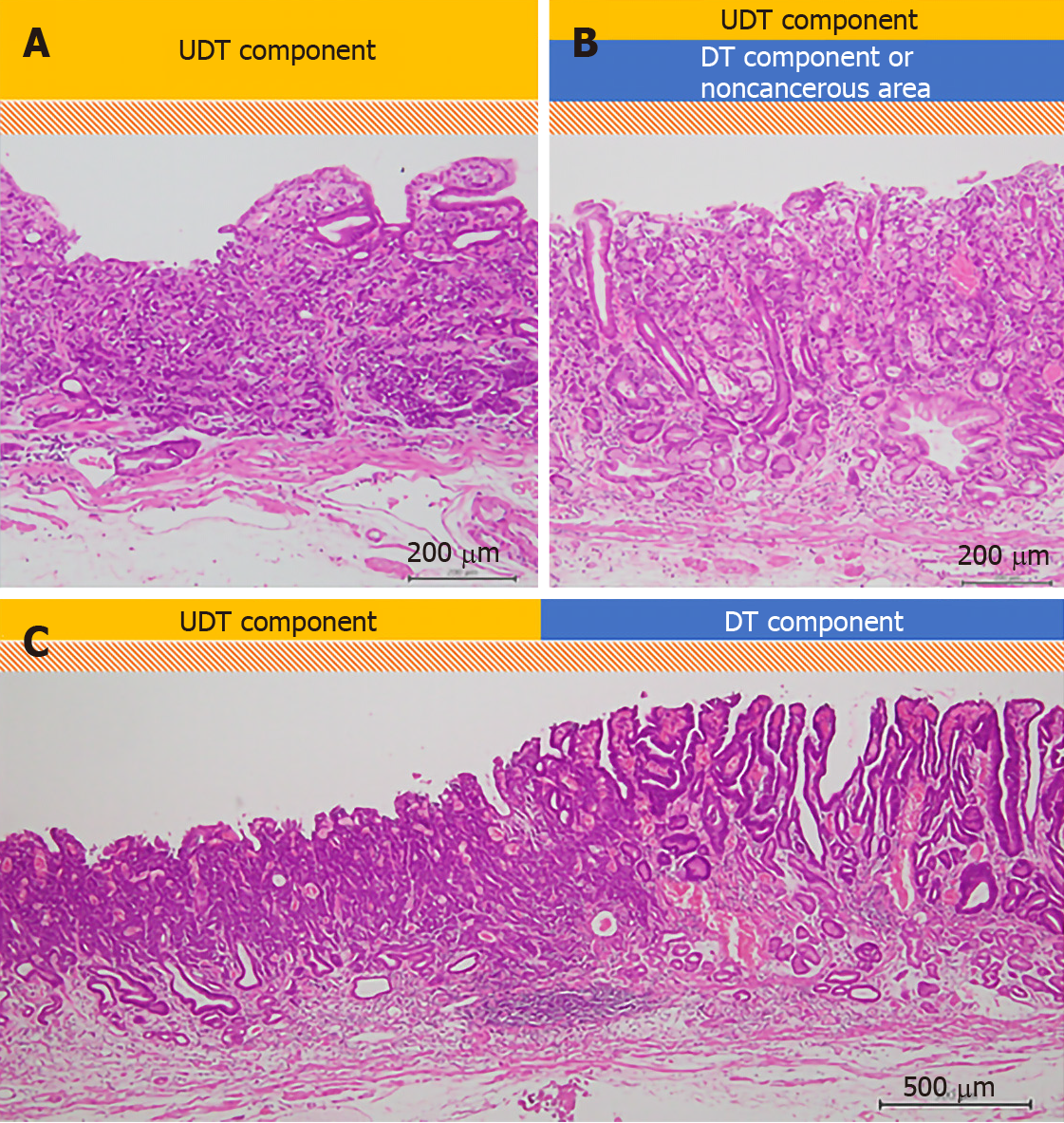
All specimens resected with ESD were “pinned out” onto polystyrene receivers to facilitate subsequent histopathological sectioning before immediate fixation in 10% buffered formalin solution, then cut into 2-mm-thick slices. The slices were embedded in paraffin, cut into 3 µm sections, stained with hematoxylin and eosin, and microscopically examined for histopathological findings by two or more expert pathologists. In addition to the items that are stipulated in guidelines issued by the Japanese Gastric Cancer Association[3,7], we investigated the UDT component within a lesion with regard to the presence or absence of the following three factors that we hypothesized could potentially be associated with recognition using ME-NBI: (1) The whole mucosal layer occupation of the UDT component; (2) Exposure of the UDT component to the surface of the mucosa; and (3) Existence of a clear border between the DT and UDT components (Figure 2). Cases that met category (1) criteria included cases that met category (2) criteria. However, we distinguished category (1) from category (2) by the UDT cancer status, since category (1) had more progressed cancer compared to category (2). UDT early gastric cancers are thought to progress from the intermediate-layer type to the superficial type and finally to the whole-layer type[12].
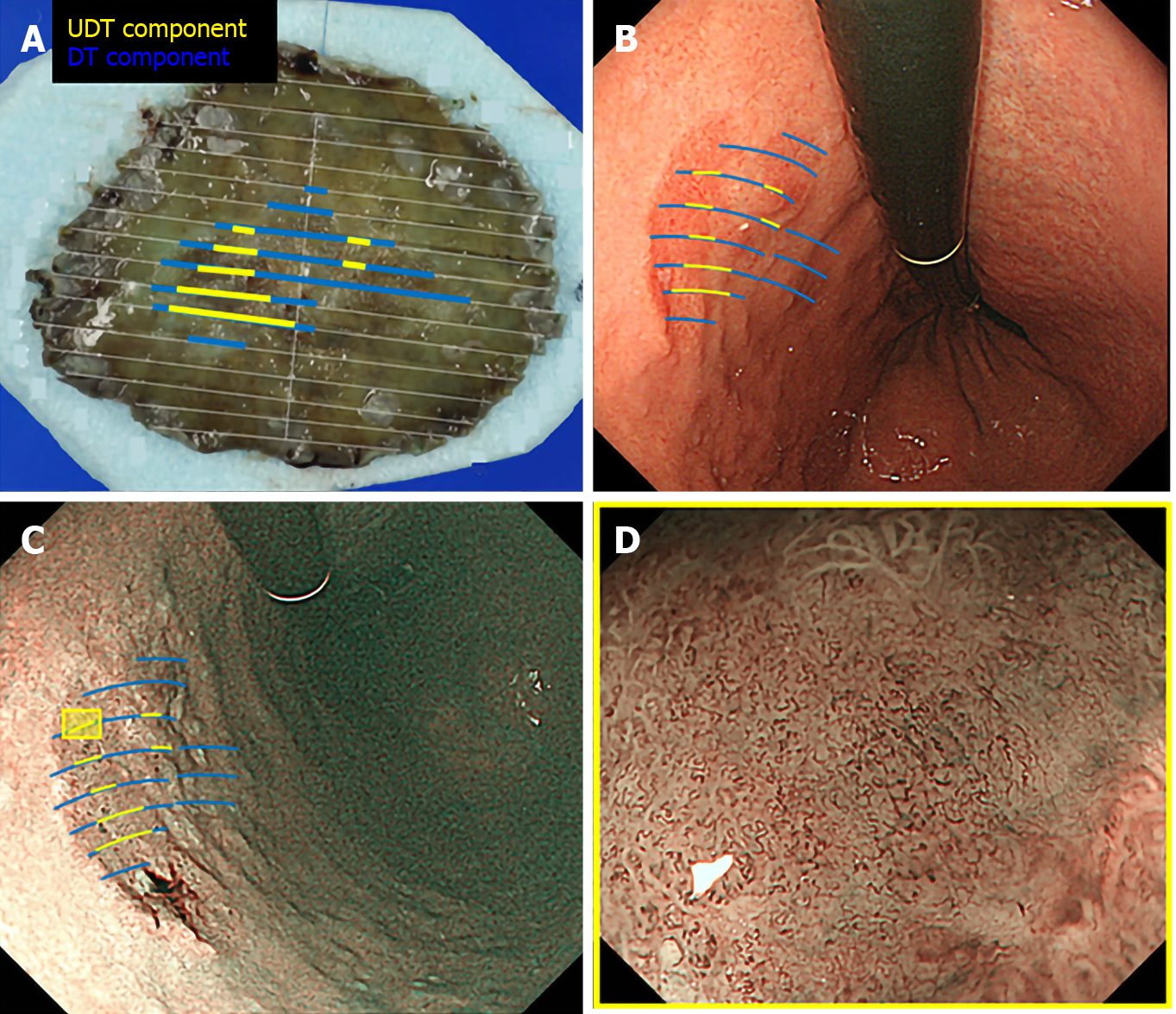
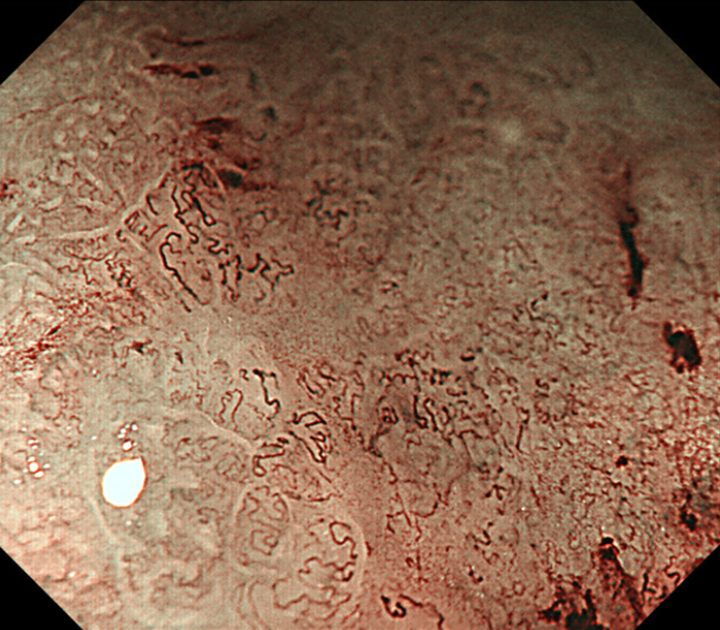
We used the Evis Lucera Spectrum endoscopy system from January 2005 to October 2012 and the Evis Lucera Elite system from November 2012 onwards (Olympus Medical Systems, Tokyo, Japan), along with high-resolution optical magnifying endoscopies (GIF-H260Z; Olympus Optical, Tokyo, Japan) for NBI. We used a black soft hood attachment (MB46; Olympus) on the tip of the scope in all cases. All patients were examined under conscious sedation, as described in our previous report[13]. Three specialists certified by the Japan Gastroenterological Endoscopy Society retrospectively reviewed the preoperative endoscopic images of all lesions. In the review, we first mapped the histological type diagnosed in the histopathological examination onto the corresponding endoscopic images (Figure 3A and B). Then, we focused on the area where the UDT component was histopathologically present and investigated whether the endoscopic finding of the UDT component could be identified (Figure 3C and D). In this study, the endoscopic finding of the UDT component was evaluated by the ME-NBI finding because we aimed to focus on the exact area where the UDT component existed histopathologically, even if the area was limited in size. The ME-NBI finding of the UDT component was defined by two elements: a microvascular pattern, which has an isolated and disordered quality, and a microstructure pattern, which lacks visibility or even disappears (Figure 4). This complies with the ME-NBI finding described as a corkscrew pattern in the study by Nakayoshi et al[14], who reported that 85.7% of UDT early gastric cancers showed a corkscrew pattern in ME-NBI. Kishino et al[15] recently reported that the positive predictive value of ME-NBI for UDT early gastric cancer was 88.9% when a lesion showed both an unclear surface pattern and severe vascular irregularity in accordance with the corkscrew pattern. The results of the preoperative biopsies were also investigated.
Statistical analyses were performed using JMP Pro 12 (SAS Institute, Cary, NC, United States). Continuous variables were expressed as the mean ± SD, and categorical parameters were expressed as numbers and frequencies. We used the Chi-square test or Fisher’s exact test for the categorical parameters, and t-test to compare the mean values of the continuous variables for two-group comparisons. P < 0.05 was considered statistically significant. The statistical methods and analyses of this study were reviewed by Professor Masataka Taguri of the Yokohama City University School of Data Science.
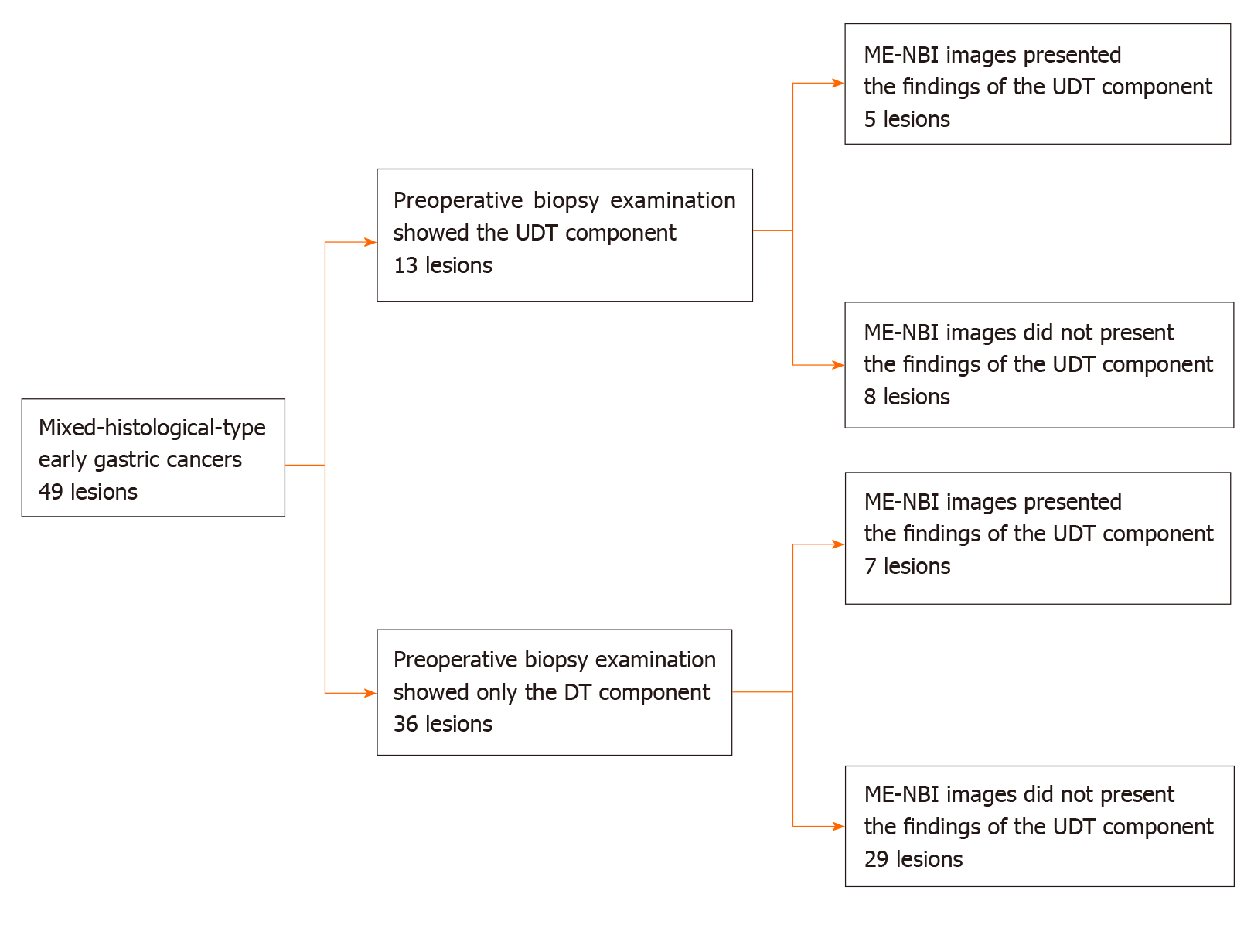
Among 2011 early gastric cancers treated at our hospital with ESD between January 2005 and June 2016, 92 lesions (4.6%) were histopathologically diagnosed as MT gastric cancers after ESD (Figure 1). Table 1 shows the clinicopathological characteristics and treatment outcomes of MT gastric cancers compared to pure DT gastric cancers treated with ESD during the same period. Outcomes of pure UDT gastric cancers are also shown in Table 1 for reference. Treatment outcome revealed that 45.7% (42/92) of MT early gastric cancers were histopathologically identified as submucosal cancers. Lymphovascular invasion was present in 22.8% (21/92) of the MT lesions. Thus, the overall endoscopic curative resection rate of MT gastric cancers was only 38.0% (35/92), which was significantly lower than that of pure DT gastric cancers. After excluding 43 lesions that did not precisely undergo preoperative ME-NBI of the whole lesion, we included 49 MT gastric cancers in 44 patients for additional analysis. Table 2 shows the clinicopathological characteristics and treatment outcomes of the 49 lesions. In the histopathological examination, the whole mucosal layer occupation of the UDT component and exposure of the UDT component to the mucosal surface were seen in 67.3% (33/49) and 71.4% (35/49) of the MT gastric cancers, respectively. A clear distinction of the border between the DT and UDT components could not be drawn in 65% (32/49) of MT lesions. In the endoscopic examination, a review of pretreatment ME-NBI images revealed that 24.5% (12/49) of the MT lesions showed an endoscopic finding of the UDT component within the area where it was present in the histopathological examination. Only 26.5% (13/49) of the lesions were diagnosed from the pretreatment biopsy as having a UDT component. Table 3 shows the comparative relationships between the presence or absence of an ME-NBI UDT component finding and histopathological UDT component findings. Tumor diameter did not differ significantly between the lesions with an ME-NBI UDT component finding and those without the ME-NBI UDT component finding. Fourteen of 15 lesions (93%) with submucosal invasion did not show an ME-NBI UDT component finding. The UDT component did not expose to the mucosal surface in 10 lesions histopathologically and none of them showed an ME-NBI UDT component finding. In this analysis, histopathological UDT predominance was the single significant factor associated with an ME-NBI UDT component finding, with 62% (8/13) of UDT-predominant MT gastric cancers showing an ME-NBI UDT component. Of the 36 lesions that were diagnosed from the pretreatment biopsy as having only a DT component, seven showed an ME-NBI UDT component finding. Therefore, the addition of ME-NBI improved the pretreatment diagnosis rate of a UDT component by 14%. Consequently, combined results of the pretreatment biopsy and an ME-NBI finding showed the presence of a UDT component within the lesion in 40% (20/49) of MT gastric cancers in this study (Figure 5).
| Pure DT (n = 1863) | Pure UDT (n = 56) | MT (n = 92) | P value (Pure DT vs MT) | |
| Age (yr), mean ± SD | 71.7 ± 8.5 | 66.0 ± 10.3 | 71.0 ± 9.1 | < 0.0001 |
| Male | 1409 (75.6) | 30 (53.6) | 62 (67.4) | 0.0739 |
| Location | 0.5385 | |||
| Upper third | 340 (18.3) | 6 (10.7) | 14 (15.2) | |
| Middle third | 536 (28.8) | 28 (50.0) | 31 (33.7) | |
| Lower third | 987 (53.0) | 22 (39.3) | 47 (51.1) | |
| Macroscopic type | 0.0002 | |||
| Elevated | 759 (40.7) | 3 (5.4) | 18 (19.6) | |
| Flat | 34 (1.83) | 0 | 1 (1.1) | |
| Depressed | 1070 (57.4) | 53 (94.6) | 73 (79.4) | |
| Tumor diameter (mm), mean ± SD | 16.1 ± 11.4 | 14.6 ± 9.6 | 24.4 ± 13.9 | < 0.0001 |
| Depth | < 0.0001 | |||
| Mucosal | 1635 (87.8) | 45 (80.4) | 50 (54.4) | |
| Submucosal | 228 (12.2) | 11 (19.6) | 42 (45.7) | |
| Ulcerative findings | 150 (8.1) | 9 (16.1) | 18 (19.6) | 0.0001 |
| Lympovascular invasion | 57 (3.1) | 4 (7.1) | 21 (22.8) | < 0.0001 |
| En bloc resection | 1853 (99.5) | 55 (98.2) | 92 (100) | 0.4811 |
| R0 resection | 1835 (98.5) | 53 (94.6) | 81 (88.0) | < 0.0001 |
| Curative resection | 1686 (90.5) | 27 (48.2) | 35 (38.0) | < 0.0001 |
| Additional surgery | 97 (5.2) | 13 (23.2) | 36 (39.1) | < 0.0001 |
| Patient characteristics | |
| Age (yr), mean ± SD | 71.7 ± 9.5 |
| Male | 34 (69.4) |
| Endoscopic examination | |
| Location | |
| Upper third | 4 (8.2) |
| Middle third | 12 (24.5) |
| Lower third | 33 (67.3) |
| Macroscopic types | |
| Elevated | 5 (10.2) |
| Flat | 0 |
| Depressed | 44 (89.8) |
| Preoperative biopsy results | |
| Only DT component (tub1, tub2 or pap) | 36 (73.5) |
| UDT component (por1, por2, sig or muc) with or without DT component | 13 (26.5) |
| ME-NBI findings of UDT gastric cancer | 12 (24.5) |
| Histopathological examination | |
| Tumor diameter (mm), mean ± SD | 23.3 ± 14.4 |
| Depth | |
| Mucosal | 34 (69.4) |
| Submucosal, < 500 um | 7 (14.3) |
| Submucosal, ≥ 500 um | 8 (16.3) |
| Lymphovascular invasion | |
| Lymphoid invasion | 10 (20.4) |
| Vascular invasion | 2 (4.1) |
| Vertical margin positive | 2 (4.1) |
| Horizontal margin positive | 1 (2.0) |
| Ulcerative finding | 10 (20.4) |
| Predominant histological type | |
| Differentiated | 36 (73.5) |
| Undifferentiated | 13 (26.5) |
| The whole mucosal layer extension of UDT component | 33 (67.3) |
| Exposure of UDT cancer to the surface of the mucosa | 39 (79.6) |
| Clear border between DT and UDT components | 17 (34.7) |
| Treatment outcome | |
| Curative resection | 24 (49.0) |
| Additional surgery | 19 (38.8) |
| Lymph node metastasis | 1 (2.0) |
| Recurrence | 0 |
| ME-NBI findings of UDT component | P value | ||
| Present (n = 12) | Absent (n = 37) | ||
| Tumor diameter (mm), mean ± SD | 20.6 ± 10.2 | 24.2 ± 15.5 | 0.450 |
| Preoperative biopsy results | 0.259 | ||
| Only DT component | 7 (19.4) | 29 (80.6) | |
| UDT component with or without DT component | 5 (38.5) | 8 (61.5) | |
| Ulcerative findings | 1 | ||
| Present | 2 (20) | 8 (80) | |
| Absent | 10 (26) | 29 (74) | |
| Depth | 0.0754 | ||
| Mucosal | 11 (32) | 23 (68) | |
| Submucosal | 1 (7) | 14 (93) | |
| Predominant histological type | 0.0009 | ||
| Differentiated | 4 (11) | 32 (89) | |
| Undifferentiated | 8 (62) | 5 (38) | |
| The whole mucosal layer extension of UDT component | 0.290 | ||
| Present | 10 (30) | 23 (70) | |
| Absent | 2 (12) | 14 (88) | |
| Exposure of UDT cancer to the surface of the mucosa | 0.0926 | ||
| Present | 12 (31) | 27 (69) | |
| Absent | 0 (0) | 10 (100) | |
| Clear border between DT and UDT components | 0.175 | ||
| Present | 2 (12) | 15 (88) | |
| Absent | 10 (31) | 22 (69) | |
In this retrospective study, we investigated whether a UDT component within MT early gastric cancers could be recognized preoperatively, since the presence of a UDT component profoundly affects the clinical course and treatment decision of early gastric cancers. Some retrospective studies of surgical cases reported a higher probability of lymph node metastasis[16-20] and even poorer prognosis[21-24] in MT gastric cancers, while other studies have reported that these relationships may not apply to MT gastric cancers that meet the criteria for endoscopic curative resection[25,26]. However, retrospective studies of surgical cases tend to ignore the uncertainty of a preoperative diagnosis. A previous study of endoscopically treated cases reported that the presence of a UDT component was an independent risk factor for non-curative endoscopic resection in early gastric cancers[9-11,27]. The present study included cases that underwent ESD as an initial treatment, and only 38% (35/92) of them turned out to fulfill criteria for endoscopic curative resection. Therefore, preoperative recognition of a UDT component may reduce the incidence of cases that need additional gastrectomy after undergoing ESD.
Histopathological examination of ESD resected specimens showed that in 67.3% (33/49) of MT gastric cancers, the UDT component occupied the whole mucosal layer, and 79.6% (39/49) showed the UDT component exposed to the surface of the mucosa. Therefore, it is reasonable to assume that preoperative ME-NBI may contribute to diagnosing a UDT component within MT gastric cancers. However, a thorough review of preoperative ME-NBI images by three specialists certified by the Japan Gastroenterological Endoscopy Society revealed that only 24.5% (12/49) of MT gastric cancers showed an endoscopic finding of a UDT component within the area where the UDT component existed histopathologically. Taking the results of pretreatment biopsy into account, only 40% (20/49) of MT early gastric cancers in this study showed preoperative findings suggestive of a UDT component. These results indicate that the accurate pretreatment diagnosis of a UDT component is difficult even when the pretreatment biopsy and ME-NBI are combined.
One reason why pretreatment recognition of a UDT component in MT early gastric cancers using ME-NBI is difficult is that histopathologically, a clear distinction between DT and UDT components within a lesion could not be drawn in 65% (32/49) of cases where the DT and UDT components were mixed at a glandular structure level such as tub2+por. Thus, they did not show an endoscopic finding of the UDT component, such as a corkscrew pattern. A second reason is that even though there was an area where the UDT component occupied the whole mucosal layer or was exposed to the mucosal surface histopathologically, it could not be recognized endoscopically using ME-NBI when the area was not large enough. In other words, an area with a certain amount of UDT component may be necessary for the endoscopic finding to appear. This might be why histopathological UDT predominance was the only significant factor associated with an ME-NBI finding of a UDT component in this study (P = 0.0009). Therefore, the possibility of UDT predominance should be suspected when a lesion shows an endoscopic finding of the UDT component. Third, MT gastric cancers with submucosal invasion were less likely to show an ME-NBI finding of a UDT component compared to mucosal cancers, although the difference was not statistically significant. In lesions with submucosal invasion, microstructure and microvascular patterns were occasionally destructed, making the ME-NBI finding hard to interpret. Moreover, some lesions harbor a UDT component only in the submucosa. Previous studies have shown that MT gastric cancers tend to be glandular in the mucosa and then grow diffusely in the deeper layers[28].
Few reports have dealt with the pretreatment diagnosis of MT early gastric cancer. Horiuchi et al[29] reported that accuracy in diagnosing the predominant histological type in MT early gastric cancers was 177/192 (92.2%) when using a combination of biopsy and ME-NBI. A possible explanation for the discrepancy between previous results and our finding is that we focused on the preoperative recognition of the UDT component within MT early gastric cancers regardless of the UDT component amount, while the previous study distinguished the predominant histological type of MT early gastric cancers. It is considered more difficult to preoperatively detect a UDT component when the UDT component amount is small or when the UDT and DT components are mixed at a glandular level. Given that the presence of a UDT component adversely affects the treatment outcome of even DT-predominant MT early gastric cancers[9-11,27], we need to pay attention to even minuscule amounts of UDT component. To the best of our knowledge, the present study is the first to address the feasibility of preoperative recognition of a UDT component within early gastric cancers, regardless of histological predominance. By investigating the comparative relationships between the histopathological and endoscopic findings, the present study demonstrated the difficulty in preoperative recognition of a UDT component in MT early gastric cancers. Our results suggest that endoscopic resection plays a significant role not only in treatment but also in diagnosis.
Our study has several limitations. First, there was an inherent selection bias because this was a single-center retrospective study. Notably, we excluded early gastric cancers that had not undergone preoperative ME-NBI of the whole lesion and that had undergone gastrectomy instead of ESD as the first therapy. Thus, this study may have eliminated the MT early gastric cancers that had been easily diagnosed with a predominant UDT component, which, in turn, could have lowered the preoperative diagnosis rate of UDT components. However, we believe that the results of this study accurately reflect actual clinical practice, given that the proportion of UDT-predominant MT gastric cancers in this study was similar to that of previous studies[11,29]. Second, although we investigated 2011 consecutive early gastric cancers in 1558 patients treated with ESD over a decade in a tertiary referral center, the sample size was small and may have been insufficient. This was mainly a result of the rarity of MT early gastric cancer. Multicenter studies with larger samples are needed. Finally, we retrospectively reviewed the stored preoperative endoscopic images knowing that the final diagnoses were MT gastric cancers, which may have influenced the diagnosis rate of the UDT component. Thus, the accurate pretreatment diagnosis rate of a UDT component using ME-NBI in actual clinical practice is likely to be lower than that seen in this study.
The present study is the first to address the feasibility of preoperative UDT component recognition in early gastric cancers regardless of histological predominance. Accurate pretreatment diagnosis of a UDT component within MT early gastric cancers is difficult even when pretreatment biopsy and ME-NBI are combined. Considering our results, endoscopic resection plays a significant role in both treatment and diagnosis.
Endoscopic resection has become a mainstay therapy for early gastric cancers (EGCs). EGCs with very low risk of lymph node metastasis are indications for endoscopic resection. The risk of lymph node metastasis markedly differs according to the histological types.
Although the undifferentiated-type (UDT) component adversely affects the clinical course of EGCs, whether the UDT component within histological-mixed-type (MT) EGCs can be recognized preoperatively is not known. This is particularly important because accurate pretreatment diagnosis of the UDT component can help make the right treatment decision for EGC.
We investigated the feasibility of the preoperative diagnosis of the UDT component within MT EGCs.
This is a retrospective study using clinical data and samples from patients with EGCs treated by endoscopic submucosal dissection. Through histopathological examination, we investigated each lesion’s UDT component: (1) Whole mucosal layer occupation; (2) Exposure to the mucosal surface; and (3) Existence of a clear border between the differentiated-type and UDT components. We examined endoscopic images using magnifying endoscopy with narrow-band imaging to identify whether the endoscopic UDT component finding was recognizable within the area where it was present in the histopathological examination. The preoperative biopsy results and comparative relationships between endoscopic and histopathological findings were also examined.
Whole mucosal layer occupation of the UDT component and exposure of the UDT component to the mucosal surface were observed in 67.3% and 79.6% of samples, respectively. However, the preoperative endoscopic images showed that only 24.5% of MT EGCs revealed the UDT component within the area where it was present histopathologically. Histopathological UDT predominance was the single significant factor associated with the presence of the endoscopic UDT component finding (61.5% vs 11.1%, P = 0.0009). Combined results of the pretreatment biopsy and magnifying endoscopy with narrow-band imaging showed the preoperative presence of the UDT component in 40% of MT EGCs.
Accurate pretreatment diagnosis of the UDT component was hardly achieved even when pretreatment biopsy and diagnostic endoscopy were combined. However, the possibility of UDT predominance should be suspected when a lesion shows an endoscopic finding of the undifferentiated-type component.
Currently, endoscopic resection plays a significant role in both the diagnosis and treatment of MT EGCs. Future studies should seek novel UDT findings to distinguish MT EGCs from pure differentiated-type EGCs.
We thank all the members of the Department of Pathology at Yokohama City University Medical Center for their devotion to the work and excellent evaluation of the samples.
Manuscript source: Unsolicited manuscript
Specialty type: Gastroenterology and hepatology
Country/Territory of origin: Japan
Peer-review report’s scientific quality classification
Grade A (Excellent): 0
Grade B (Very good): B
Grade C (Good): C
Grade D (Fair): 0
Grade E (Poor): 0
P-Reviewer: Ekpenyong CE, Hassan A S-Editor: Zhang L L-Editor: A P-Editor: Ma YJ
| 1. | Gotoda T, Yamamoto H, Soetikno RM. Endoscopic submucosal dissection of early gastric cancer. J Gastroenterol. 2006;41:929-942. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 485] [Cited by in RCA: 507] [Article Influence: 26.7] [Reference Citation Analysis (0)] |
| 2. | Oka S, Tanaka S, Kaneko I, Mouri R, Hirata M, Kawamura T, Yoshihara M, Chayama K. Advantage of endoscopic submucosal dissection compared with EMR for early gastric cancer. Gastrointest Endosc. 2006;64:877-883. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 487] [Cited by in RCA: 527] [Article Influence: 27.7] [Reference Citation Analysis (0)] |
| 3. | Japanese Gastric Cancer Association. Japanese gastric cancer treatment guidelines 2014 (ver. 4). Gastric Cancer. 2017;20:1-19. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 1575] [Cited by in RCA: 1915] [Article Influence: 239.4] [Reference Citation Analysis (1)] |
| 4. | Gotoda T, Yanagisawa A, Sasako M, Ono H, Nakanishi Y, Shimoda T, Kato Y. Incidence of lymph node metastasis from early gastric cancer: estimation with a large number of cases at two large centers. Gastric Cancer. 2000;3:219-225. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 1308] [Cited by in RCA: 1328] [Article Influence: 53.1] [Reference Citation Analysis (0)] |
| 5. | Hirasawa T, Gotoda T, Miyata S, Kato Y, Shimoda T, Taniguchi H, Fujisaki J, Sano T, Yamaguchi T. Incidence of lymph node metastasis and the feasibility of endoscopic resection for undifferentiated-type early gastric cancer. Gastric Cancer. 2009;12:148-152. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 344] [Cited by in RCA: 368] [Article Influence: 24.5] [Reference Citation Analysis (0)] |
| 6. | Takizawa K, Ono H, Yamamoto Y, Katai H, Hori S, Yano T, Umegaki E, Sasaki S, Iizuka T, Kawagoe K, Shimoda T, Muto M, Sasako M. Incidence of lymph node metastasis in intramucosal gastric cancer measuring 30 mm or less, with ulceration; mixed, predominantly differentiated-type histology; and no lymphovascular invasion: a multicenter retrospective study. Gastric Cancer. 2016;19:1144-1148. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 17] [Cited by in RCA: 20] [Article Influence: 2.2] [Reference Citation Analysis (0)] |
| 7. | Japanese Gastric Cancer Association. Japanese classification of gastric carcinoma: 3rd English edition. Gastric Cancer. 2011;14:101-112. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 2390] [Cited by in RCA: 2873] [Article Influence: 205.2] [Reference Citation Analysis (0)] |
| 8. | Takizawa K, Ono H, Kakushima N, Tanaka M, Hasuike N, Matsubayashi H, Yamagichi Y, Bando E, Terashima M, Kusafuka K, Nakajima T. Risk of lymph node metastases from intramucosal gastric cancer in relation to histological types: how to manage the mixed histological type for endoscopic submucosal dissection. Gastric Cancer. 2013;16:531-536. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 80] [Cited by in RCA: 91] [Article Influence: 7.6] [Reference Citation Analysis (0)] |
| 9. | Shim CN, Chung H, Park JC, Lee H, Shin SK, Lee SK, Lee YC. Early gastric cancer with mixed histology predominantly of differentiated type is a distinct subtype with different therapeutic outcomes of endoscopic resection. Surg Endosc. 2015;29:1787-1794. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 13] [Cited by in RCA: 17] [Article Influence: 1.5] [Reference Citation Analysis (0)] |
| 10. | Han JP, Hong SJ, Kim HK. Long-term outcomes of early gastric cancer diagnosed as mixed adenocarcinoma after endoscopic submucosal dissection. J Gastroenterol Hepatol. 2015;30:316-320. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 30] [Cited by in RCA: 34] [Article Influence: 3.4] [Reference Citation Analysis (0)] |
| 11. | Horiuchi Y, Fujisaki J, Yamamoto N, Ishizuka N, Omae M, Ishiyama A, Yoshio T, Hirasawa T, Yamamoto Y, Nagahama M, Takahashi H, Tsuchida T. Undifferentiated-type component mixed with differentiated-type early gastric cancer is a significant risk factor for endoscopic non-curative resection. Dig Endosc. 2018;30:624-632. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 31] [Cited by in RCA: 35] [Article Influence: 5.0] [Reference Citation Analysis (0)] |
| 12. | Okada K, Fujisaki J, Kasuga A, Omae M, Hirasawa T, Ishiyama A, Inamori M, Chino A, Yamamoto Y, Tsuchida T, Nakajima A, Hoshino E, Igarashi M. Diagnosis of undifferentiated type early gastric cancers by magnification endoscopy with narrow-band imaging. J Gastroenterol Hepatol. 2011;26:1262-1269. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 53] [Cited by in RCA: 58] [Article Influence: 4.1] [Reference Citation Analysis (0)] |
| 13. | Makazu M, Hirasawa K, Sato C, Ikeda R, Fukuchi T, Ishii Y, Kobayashi R, Kaneko H, Taguri M, Tateishi Y, Inayama Y, Maeda S. Histological verification of the usefulness of magnifying endoscopy with narrow-band imaging for horizontal margin diagnosis of differentiated-type early gastric cancers. Gastric Cancer. 2018;21:258-266. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 9] [Cited by in RCA: 11] [Article Influence: 1.6] [Reference Citation Analysis (0)] |
| 14. | Nakayoshi T, Tajiri H, Matsuda K, Kaise M, Ikegami M, Sasaki H. Magnifying endoscopy combined with narrow band imaging system for early gastric cancer: correlation of vascular pattern with histopathology (including video). Endoscopy. 2004;36:1080-1084. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 328] [Cited by in RCA: 335] [Article Influence: 16.0] [Reference Citation Analysis (0)] |
| 15. | Kishino T, Oyama T, Funakawa K, Ishii E, Yamazato T, Shibagaki K, Miike T, Tanuma T, Kuwayama Y, Takeuchi M, Kitamura Y. Multicenter prospective study on the histological diagnosis of gastric cancer by narrow band imaging-magnified endoscopy with and without acetic acid. Endosc Int Open. 2019;7:E155-E163. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 5] [Cited by in RCA: 5] [Article Influence: 0.8] [Reference Citation Analysis (0)] |
| 16. | Hanaoka N, Tanabe S, Mikami T, Okayasu I, Saigenji K. Mixed-histologic-type submucosal invasive gastric cancer as a risk factor for lymph node metastasis: feasibility of endoscopic submucosal dissection. Endoscopy. 2009;41:427-432. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 87] [Cited by in RCA: 98] [Article Influence: 6.1] [Reference Citation Analysis (0)] |
| 17. | Lee JH, Choi IJ, Han HS, Kim YW, Ryu KW, Yoon HM, Eom BW, Kim CG, Lee JY, Cho SJ, Kim YI, Nam BH, Kook MC. Risk of lymph node metastasis in differentiated type mucosal early gastric cancer mixed with minor undifferentiated type histology. Ann Surg Oncol. 2015;22:1813-1819. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 30] [Cited by in RCA: 38] [Article Influence: 3.5] [Reference Citation Analysis (0)] |
| 18. | Miyamae M, Komatsu S, Ichikawa D, Kosuga T, Kubota T, Okamoto K, Konishi H, Shiozaki A, Fujiwara H, Kishimoto M, Otsuji E. Histological mixed-type as an independent risk factor for nodal metastasis in submucosal gastric cancer. Tumour Biol. 2016;37:709-714. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 19] [Cited by in RCA: 20] [Article Influence: 2.0] [Reference Citation Analysis (0)] |
| 19. | Pyo JH, Lee H, Min BH, Lee JH, Choi MG, Lee JH, Sohn TS, Bae JM, Kim KM, Yeon S, Jung SH, Kim JJ, Kim S. Early gastric cancer with a mixed-type Lauren classification is more aggressive and exhibits greater lymph node metastasis. J Gastroenterol. 2017;52:594-601. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 39] [Cited by in RCA: 41] [Article Influence: 5.1] [Reference Citation Analysis (0)] |
| 20. | Zhong Q, Sun Q, Xu GF, Fan XQ, Xu YY, Liu F, Song SY, Peng CY, Wang L. Differential analysis of lymph node metastasis in histological mixed-type early gastric carcinoma in the mucosa and submucosa. World J Gastroenterol. 2018;24:87-95. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in CrossRef: 11] [Cited by in RCA: 18] [Article Influence: 2.6] [Reference Citation Analysis (0)] |
| 21. | Zheng HC, Li XH, Hara T, Masuda S, Yang XH, Guan YF, Takano Y. Mixed-type gastric carcinomas exhibit more aggressive features and indicate the histogenesis of carcinomas. Virchows Arch. 2008;452:525-534. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 96] [Cited by in RCA: 107] [Article Influence: 6.3] [Reference Citation Analysis (0)] |
| 22. | Shimizu H, Ichikawa D, Komatsu S, Okamoto K, Shiozaki A, Fujiwara H, Murayama Y, Kuriu Y, Ikoma H, Nakanishi M, Ochiai T, Kokuba Y, Kishimoto M, Yanagisawa A, Otsuji E. The decision criterion of histological mixed type in "T1/T2" gastric carcinoma--comparison between TNM classification and Japanese Classification of Gastric Cancer. J Surg Oncol. 2012;105:800-804. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 16] [Cited by in RCA: 18] [Article Influence: 1.3] [Reference Citation Analysis (0)] |
| 23. | Komatsu S, Ichikawa D, Miyamae M, Shimizu H, Konishi H, Shiozaki A, Fujiwara H, Okamoto K, Kishimoto M, Otsuji E. Histological mixed-type as an independent prognostic factor in stage I gastric carcinoma. World J Gastroenterol. 2015;21:549-555. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in CrossRef: 26] [Cited by in RCA: 29] [Article Influence: 2.9] [Reference Citation Analysis (0)] |
| 24. | Ikari N, Taniguchi K, Serizawa A, Yamada T, Yamamoto M, Furukawa T. A primary tumor of mixed histological type is a novel poor prognostic factor for patients undergoing resection of liver metastasis from gastric cancer. J Hepatobiliary Pancreat Sci. 2017;24:281-288. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 5] [Cited by in RCA: 7] [Article Influence: 0.9] [Reference Citation Analysis (0)] |
| 25. | Yoon HJ, Kim YH, Kim JH, Kim H, Kim H, Park JJ, Youn YH, Park H, Kim JW, Hyung WJ, Noh SH, Choi SH. Are new criteria for mixed histology necessary for endoscopic resection in early gastric cancer? Pathol Res Pract. 2016;212:410-414. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 18] [Cited by in RCA: 27] [Article Influence: 3.0] [Reference Citation Analysis (0)] |
| 26. | Choi J, Chung H, Kim J, Kim JL, Kim SG, Jung HC. Determining the current indications for endoscopic submucosal dissection in patients with Lauren mixed-type early gastric cancer. J Gastroenterol Hepatol. 2020;35:586-592. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 5] [Cited by in RCA: 8] [Article Influence: 1.6] [Reference Citation Analysis (0)] |
| 27. | Lee JH, Kim JH, Rhee K, Huh CW, Lee YC, Yoon SO, Youn YH, Park H, Lee SI. Undifferentiated early gastric cancer diagnosed as differentiated histology based on forceps biopsy. Pathol Res Pract. 2013;209:314-318. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 19] [Cited by in RCA: 24] [Article Influence: 2.0] [Reference Citation Analysis (0)] |
| 28. | Kozuki T, Yao T, Nakamura S, Matsumoto T, Tsuneyoshi M. Differences in p53 and cadherin-catenin complex expression between histological subtypes in diffusely infiltrating gastric carcinoma. Histopathology. 2002;41:56-64. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 17] [Cited by in RCA: 18] [Article Influence: 0.8] [Reference Citation Analysis (0)] |
| 29. | Horiuchi Y, Tokai Y, Yamamoto N, Yoshimizu S, Ishiyama A, Yoshio T, Hirasawa T, Yamamoto Y, Nagahama M, Takahashi H, Tsuchida T, Fujisaki J. Additive Effect of Magnifying Endoscopy with Narrow-Band Imaging for Diagnosing Mixed-Type Early Gastric Cancers. Dig Dis Sci. 2020;65:591-599. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 11] [Cited by in RCA: 14] [Article Influence: 2.8] [Reference Citation Analysis (0)] |