Published online Dec 16, 2021. doi: 10.12998/wjcc.v9.i35.11050
Peer-review started: June 9, 2021
First decision: July 15, 2021
Revised: July 26, 2021
Accepted: October 27, 2021
Article in press: October 27, 2021
Published online: December 16, 2021
Processing time: 183 Days and 20.8 Hours
Colchicine has been widely used as an anti-gout medication over the past decades. However, it is less commonly used due to its narrow therapeutic range, meaning that its lethal dose is close to its therapeutic dose. The lethal dose of colchicine is considered to be 0.8 mg/kg. As chronic colchicine poisoning has multiple manifestations, it poses a challenge in the clinician’s differential diagnosis. Historically, the drug was important in treating gout; however, clinical studies are currently underway regarding the use of colchicine in patients with coronavirus disease 2019 as well as its use in coronary artery disease, making this drug more important in clinical practice.
A 61-year-old male with a history of gout and chronic colchicine intake was admitted to our Emergency Department due to numbness and weakness of the lower limbs. The patient reported a history of colchicine intake for 23 years. After thorough examination, he was diagnosed with colchicine poisoning, manifesting as neuromyopathy, multiple gastric ulcers and myelosuppression. We advised him to stop taking colchicine and drinking alcohol. We also provided a prescription of lansoprazole and mecobalamin, and then asked him to return to the clinic for re-examination. The patient was followed up for 3-mo during which time his gout symptoms were controlled to the point where he was asymptomatic.
Colchicine overdose can mimic the clinical manifestations of several conditions. Physicians easily pay attention to the disease while ignoring the cause of the disease. Thus, the patient’s medication history should never be ignored.
Core Tip: With ongoing clinical trials on colchicine use in more generalized diseases, the probability of chronic colchicine poisoning is on the rise. Chronic colchicine poisoning has multiple manifestations. In this case, it presented with neuromyopathy, multiple gastric ulcers and myelosuppression, demonstrating its clinical significance.
- Citation: Li MM, Teng J, Wang Y. Chronic colchicine poisoning with neuromyopathy, gastric ulcers and myelosuppression in a gout patient: A case report. World J Clin Cases 2021; 9(35): 11050-11055
- URL: https://www.wjgnet.com/2307-8960/full/v9/i35/11050.htm
- DOI: https://dx.doi.org/10.12998/wjcc.v9.i35.11050
Colchicine is known for its anti-inflammatory effects, and was historically used for gout. However, due to its narrow therapeutic range, there have been numerous reports regarding acute and chronic colchicine poisoning. Therefore, it has seldom been used in the clinical setting in recent years. Due to more generalized studies on colchicine, chronic colchicine poisoning has raised concerns among physicians. The ongoing GRECCO-19[1] study aims to identify whether colchicine may positively intervene in the clinical course of coronavirus disease 2019. At the same time, the LoDoCo2[2] and COLCOT[3] trials have demonstrated the effects of colchicine on the secondary prevention of coronary artery disease. These studies have made the use of colchicine more generalized, while long-term use of colchicine increases the risk of chronic colchicine poisoning. Thus, it is essential for clinicians to realize chronic colchicine poisoning.
A 61-year-old male was admitted to the emergency department (ED) due to numbness and weakness of the lower limbs, which was his second hospital admission.
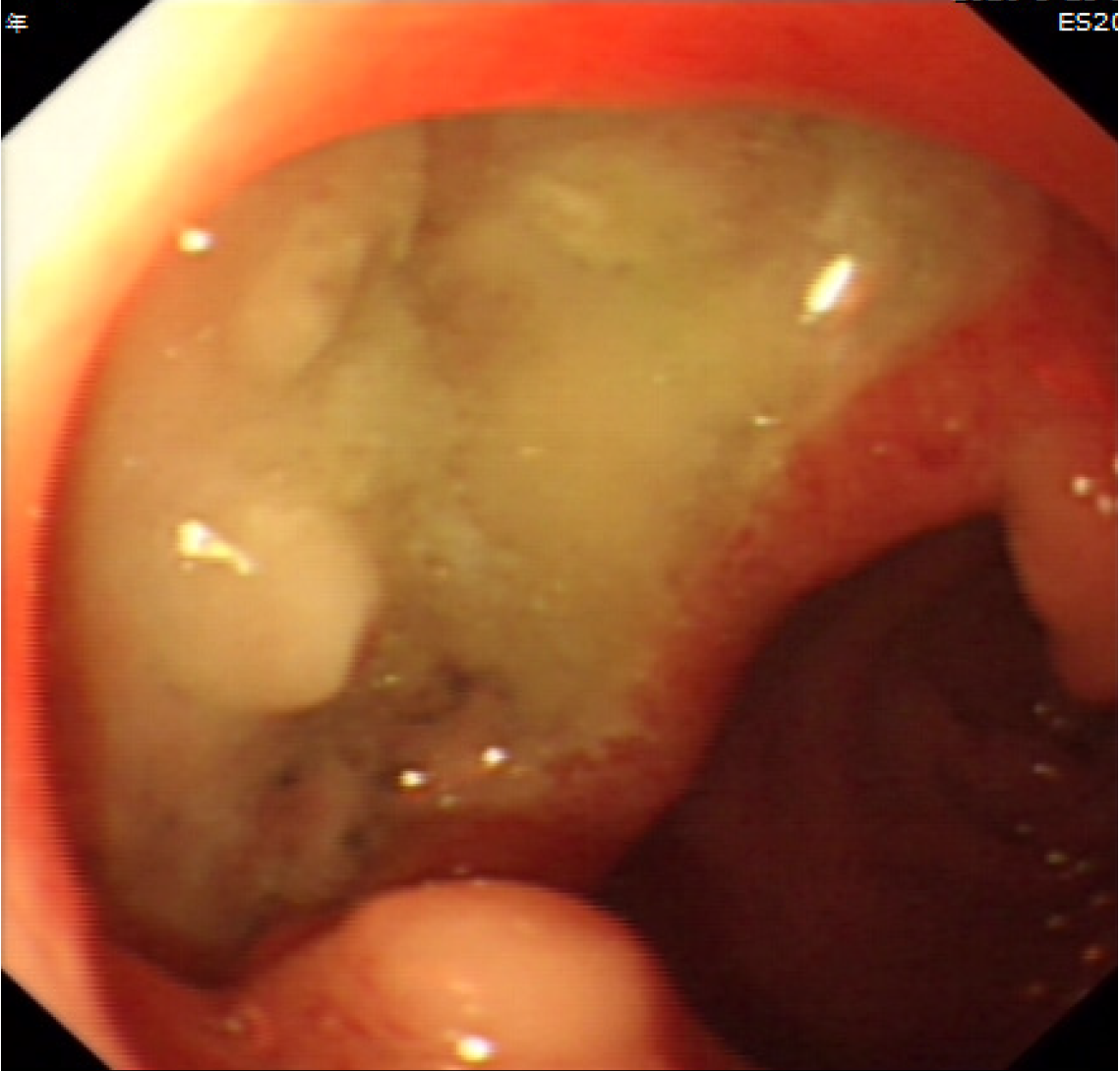
The patient reported a history of colchicine consumption for gout. He initially only took colchicine temporally for joint pain control or preventive purposes before drinking alcohol. He then started taking high doses of colchicine over the recent five years as his gout progressed. He reported discontinuously taking 30 mg monthly on average over the past five years. He also complained of diarrhea after taking colchicine. He was diagnosed with multiple gastric ulcers by gastroscopy (Figure 1) and pathology, for which he received a subtotal gastrectomy approximately 6 mo ago.
The patient had hypertension for over 10 years.
He had a history of alcohol abuse.
Physical examination showed that the patient was afebrile, with no abnormal findings on heart and lung and abdominal examinations. However, multiple joint tenderness was present, consistent with gout. The patient’s muscle strength scores were 4 and 3 bilaterally in the upper and lower limbs, respectively.
After admission, we ordered a complete blood count (CBC), liver and kidney function tests, C-reactive protein, erythrocyte sedimentation rate (ESR), tumor markers and other blood tests. Additionally, we performed an electromyogram, a positron emission tomography-computed tomography (PET-CT) scan, and brain nuclear magnetic resonance imaging (MRI). The CBC revealed the following: Leukocytes 2.72 × 109/L, neutrophils 1.75 × 109/L, hemoglobin 76 g/L, reticulocyte ratio 1.43%, indicating microcytic hypochromic anemia and leukopenia; carbohydrate antigen 724 (CA724) > 500 U/mL, which was highly increased; the ESR was 107 mm/h, which was also increased, for which we considered gout and infection as the most probable etiologic factors. Serum uric acid level was 449 µmol/L, which was also elevated. The patient’s serum ferritin, vitamin B12 and folic acid levels were all within the normal ranges. The patient’s potassium level was 3.2 mmol/L, calcium level was 2.1 mmol/L, and phosphate level was 0.71 mmol/L, suggesting hypokalemia, hypocalcemia and hypophosphatemia. After consulting a neurologist, we considered Guillain-Barre syndrome (GBS) as a preliminary diagnosis. In order to reach a precise diagnosis, we performed a lumbar puncture, bone marrow puncture and biopsy. The biopsy results demonstrated myelodysplasia. No abnormalities were detected in the cerebrospinal fluid, which excluded GBS.
The PET-CT scan did not reveal any structural lesions such as malignant tumors, but showed an infection of the right inferior molar teeth. Brain MRI was also normal. The electromyogram delineated neurogenic damage of the upper and lower limbs.
The examination results led to a dilemma: What is the correct diagnosis in this patient? Considering the history of chronic colchicine intake, we suspected that all of the patient’s clinical manifestations might be side effects of colchicine therapy. Colchicine poisoning can manifest as myelosuppression, neuromyopathy, and gastrointestinal symptoms, which were all present in this patient. Based on these facts, we eventually diagnosed the patient with chronic colchicine poisoning manifested as myelosuppression, neuromyopathy and multiple gastric ulcers combined with right inferior molar teeth infection.
Following admission, the patient suddenly developed a fever, his numbness and weakness progressed to both upper limbs, and he was unable to ambulate normally. Given the lack of a precise protocol for colchicine overdose, we counseled the patient to stop taking colchicine, and then administered recombinant human granulocyte factor to increase his leukocyte count. Ceftizoxime was also given to treat any concurrent infection. Lansoprazole was prescribed to treat his gastric ulcers. However, the patient still developed chalkstones, which is a manifestation of gout. Based on the PET-CT scans, we suspected that bacterial infection secondary to myelosuppression was the cause of his fever. After consulting a rheumatologist, we administered benzbromarone to reduce the patient’s serum uric acid levels. Considering his history of gastric ulcer, we did not administer nonsteroidal anti-inflammatory drugs (NSAIDS). After 7 d of hospitalization, the patient’s infection was controlled, he was afebrile and his leukocyte count was back to normal. However, his hemoglobin level did not change and his symptoms of neuromyopathy persisted. The patient was now in a stable condition, the recovery from colchicine poisoning was a slow process and many complications such as gastric ulcers were irreversible. He was then discharged and transferred to the rehabilitation ward for further recovery. We counseled him to stop taking colchicine and drinking alcohol, and prescribed lansoprazole, mecobalamin, cefdinir (for 7 more days) and advised him to return to the clinic for follow-up after a month.
We followed the patient for 3 mo during which time his gout symptoms were controlled. His hemoglobin level rose to 110 g/L, he was afebrile, and his weakness and numbness were almost completely alleviated. The patient was still unable to walk. His leukocyte count was within the normal range. The patient was admitted to the rehabilitation ward for further recovery.
Colchicine has been reported to be rapidly absorbed from the gastrointestinal tract[4]. The serum concentration of colchicine has been shown to peak 0.5-3 h after ingestion[4]. Colchicine poisoning consists of three phases. First, the gastrointestinal phase: 0–24 h post-ingestion. Second, the multi-organ failure phase: 1-7 d post-ingestion. Third, the recovery phase: 7-21 d post-ingestion[4]. This patient experienced diarrhea after taking colchicine, which was consistent with the gastrointestinal phase. The patient presented to the ED with neuromyopathy, bone marrow suppression, hypokalemia, hypocal
Multiple gastric ulcers are a rare and seldom reported manifestation of colchicine poisoning. According to the instructions on the drug’s manual, gastric changes can occur in patients with long-term intake. Therefore, the patient’s multiple gastric ulcers were a manifestation of chronic colchicine poisoning. Colchicine poisoning manifesting as neuromyopathy has been reported several times in patients with long-term intake[5,6]. This patient with chronic colchicine intake developed neurogenic damage of the upper and lower limbs, consistent with previous reports. Nowadays, more knowledge on colchicine is available; thus, there have been fewer reports of neuromyopathy in recent years. However, this clinical manifestation of colchicine poisoning cannot be ignored.
Bone marrow suppression has also been reported as a clinical manifestation of colchicine poisoning in some patients. It has been reported that granulocyte colony-stimulating factor is effective in treating leukopenia[7]. This patient had leukopenia and anemia, and the bone marrow biopsy confirmed the diagnosis of myelodysplasia. We treated his bone marrow suppression with recombinant human granulocyte factor, a type of granulocyte colony-stimulating factor (G-CSF). Following treatment with G-CSF, the patient’s leukocyte count returned to normal. The patient also developed a fever after admission, and the PET-CT scan confirmed the diagnosis of infection. We thought that this infection was secondary to bone marrow suppression. As tooth infections are usually responsive to third-generation cephalosporins, we managed his infection with ceftizoxime. The patient was afebrile after 7 days of antibiotic therapy.
Colchicine blocks microtubule polymerization by an anti-inflammatory mechanism[8]. Recently the American College of Rheumatology (ACR)[9] and European League Against Rheumatism (EULAR)[10] recommended the initiation of colchicine for acute gout treatment at a dosing regimen of 1.2 mg once (1.0 mg in the EULAR recommendations), followed by the administration of 0.6 mg 1 h later (0.5 mg in the EULAR recommendations). ACR[9] guidelines recommend colchicine 0.5-0.6 mg once or twice daily as first-line prophylactic therapy in patients who are initiated on urate-lowering therapy. They recommend using colchicine for 3 mo after achieving the target serum urate concentration in a patient without tophi on physical examination or 6 mo after achieving the target serum urate concentration in patients with resolution of tophi previously seen on physical examination. In addition, EULAR[10] guidelines recommend using colchicine for the first 6 mo after urate-lowering therapy, with a recommended dose of 0.5-1 mg/d. Our patient did not use colchicine correctly according to the latest guidelines. He took colchicine at a dose higher than the prescribed dose. However, urate-lowering therapy was not initiated. This is why his gout was not controlled and he was diagnosed with chronic colchicine poisoning. According to the newest ACR[9] guidelines, urate-lowering therapy is recommended as the anchor medicine for gout and anti-inflammatory drugs are recommended for gout flare treatment. Allopurinol is recommended as the first-line treatment for urate-lowering therapy over other urate-lowering drugs. Moreover, colchicine and NSAIDs are recommended for gout flare treatment. The patient did not use colchicine correctly, as he used it as a prophylactic drug instead of gout flare treatment. He received urate-lowering drugs after discharge and his gout symptoms were controlled.
The latest meta-analysis confirmed that colchicine increases the rate of diarrhea and gastrointestinal adverse events but does not increase the rate of liver, sensory, muscle, infectious or hematologic adverse events or death[11]. Therefore, this patient’s clinical manifestations were rare. Contrary to chronic colchicine poisoning, acute colchicine poisoning is sometimes seen in clinical practice. Chronic colchicine poisoning is usually seen in patients taking colchicine prophylactically. This patient had a history of chronic colchicine intake for prophylaxis. Chronic colchicine poisoning is seldom seen and it has been associated with a high mortality rate. Only a minority of patients recover from chronic colchicine poisoning. Hence, patients’ medication history should never be ignored. Moreover, to prevent similar cases, we should also educate patients on the side effects of colchicine and counsel them to take drugs as indicated by their healthcare provider.
With ongoing clinical trials on colchicine use in more generalized diseases, the probability of chronic colchicine poisoning is on the rise. Colchicine poisoning has multiple clinical manifestations, and is usually misdiagnosed as physicians usually do not inquire about colchicine intake while taking the patient’s history.
Provenance and peer review: Unsolicited article; Externally peer reviewed.
Specialty type: Emergency medicine
Country/Territory of origin: China
Peer-review report’s scientific quality classification
Grade A (Excellent): 0
Grade B (Very good): B, B
Grade C (Good): C
Grade D (Fair): 0
Grade E (Poor): 0
P-Reviewer: Anghelescu A, Jatuworapruk K S-Editor: Gong ZM L-Editor: Webster JR P-Editor: Gong ZM
| 1. | Deftereos SG, Siasos G, Giannopoulos G, Vrachatis DA, Angelidis C, Giotaki SG, Gargalianos P, Giamarellou H, Gogos C, Daikos G, Lazanas M, Lagiou P, Saroglou G, Sipsas N, Tsiodras S, Chatzigeorgiou D, Moussas N, Kotanidou A, Koulouris N, Oikonomou E, Kaoukis A, Kossyvakis C, Raisakis K, Fountoulaki K, Comis M, Tsiachris D, Sarri E, Theodorakis A, Martinez-Dolz L, Sanz-Sánchez J, Reimers B, Stefanini GG, Cleman M, Filippou D, Olympios CD, Pyrgakis VN, Goudevenos J, Hahalis G, Kolettis TM, Iliodromitis E, Tousoulis D, Stefanadis C. The Greek study in the effects of colchicine in COvid-19 complications prevention (GRECCO-19 study): Rationale and study design. Hellenic J Cardiol. 2020;61:42-45. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 100] [Cited by in RCA: 104] [Article Influence: 20.8] [Reference Citation Analysis (0)] |
| 2. | Nidorf SM, Fiolet ATL, Mosterd A, Eikelboom JW, Schut A, Opstal TSJ, The SHK, Xu XF, Ireland MA, Lenderink T, Latchem D, Hoogslag P, Jerzewski A, Nierop P, Whelan A, Hendriks R, Swart H, Schaap J, Kuijper AFM, van Hessen MWJ, Saklani P, Tan I, Thompson AG, Morton A, Judkins C, Bax WA, Dirksen M, Alings M, Hankey GJ, Budgeon CA, Tijssen JGP, Cornel JH, Thompson PL; LoDoCo2 Trial Investigators. Colchicine in Patients with Chronic Coronary Disease. N Engl J Med. 2020;383:1838-1847. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 653] [Cited by in RCA: 1319] [Article Influence: 263.8] [Reference Citation Analysis (0)] |
| 3. | Bouabdallaoui N, Tardif JC, Waters DD, Pinto FJ, Maggioni AP, Diaz R, Berry C, Koenig W, Lopez-Sendon J, Gamra H, Kiwan GS, Blondeau L, Orfanos A, Ibrahim R, Grégoire JC, Dubé MP, Samuel M, Morel O, Lim P, Bertrand OF, Kouz S, Guertin MC, L'Allier PL, Roubille F. Time-to-treatment initiation of colchicine and cardiovascular outcomes after myocardial infarction in the Colchicine Cardiovascular Outcomes Trial (COLCOT). Eur Heart J. 2020;41:4092-4099. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 170] [Cited by in RCA: 202] [Article Influence: 40.4] [Reference Citation Analysis (0)] |
| 4. | Finkelstein Y, Aks SE, Hutson JR, Juurlink DN, Nguyen P, Dubnov-Raz G, Pollak U, Koren G, Bentur Y. Colchicine poisoning: the dark side of an ancient drug. Clin Toxicol (Phila). 2010;48:407-414. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 440] [Cited by in RCA: 388] [Article Influence: 25.9] [Reference Citation Analysis (0)] |
| 5. | De Deyn PP, Ceuterick C, Saxena V, Crols R, Chappel R, Martin JJ. Chronic colchicine-induced myopathy and neuropathy. Acta Neurol Belg. 1995;95:29-32. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 3] [Cited by in RCA: 2] [Article Influence: 0.3] [Reference Citation Analysis (0)] |
| 6. | Choi SS, Chan KF, Ng HK, Mak WP. Colchicine-induced myopathy and neuropathy. Hong Kong Med J. 1999;5:204-207. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 2] [Cited by in RCA: 2] [Article Influence: 0.4] [Reference Citation Analysis (0)] |
| 7. | Harris R, Marx G, Gillett M, Kark A, Arunanthy S. Colchicine-induced bone marrow suppression: treatment with granulocyte colony-stimulating factor. J Emerg Med. 2000;18:435-440. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 44] [Cited by in RCA: 44] [Article Influence: 1.8] [Reference Citation Analysis (0)] |
| 8. | Pascart T, Richette P. Colchicine in Gout: An Update. Curr Pharm Des. 2018;24:684-689. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 24] [Cited by in RCA: 39] [Article Influence: 6.5] [Reference Citation Analysis (0)] |
| 9. | FitzGerald JD, Dalbeth N, Mikuls T, Brignardello-Petersen R, Guyatt G, Abeles AM, Gelber AC, Harrold LR, Khanna D, King C, Levy G, Libbey C, Mount D, Pillinger MH, Rosenthal A, Singh JA, Sims JE, Smith BJ, Wenger NS, Bae SS, Danve A, Khanna PP, Kim SC, Lenert A, Poon S, Qasim A, Sehra ST, Sharma TSK, Toprover M, Turgunbaev M, Zeng L, Zhang MA, Turner AS, Neogi T. 2020 American College of Rheumatology Guideline for the Management of Gout. Arthritis Care Res (Hoboken). 2020;72:744-760. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 335] [Cited by in RCA: 474] [Article Influence: 94.8] [Reference Citation Analysis (0)] |
| 10. | Richette P, Doherty M, Pascual E, Barskova V, Becce F, Castañeda-Sanabria J, Coyfish M, Guillo S, Jansen TL, Janssens H, Lioté F, Mallen C, Nuki G, Perez-Ruiz F, Pimentao J, Punzi L, Pywell T, So A, Tausche AK, Uhlig T, Zavada J, Zhang W, Tubach F, Bardin T. 2016 updated EULAR evidence-based recommendations for the management of gout. Ann Rheum Dis. 2017;76:29-42. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 1106] [Cited by in RCA: 976] [Article Influence: 122.0] [Reference Citation Analysis (0)] |
| 11. | Stewart S, Yang KCK, Atkins K, Dalbeth N, Robinson PC. Adverse events during oral colchicine use: a systematic review and meta-analysis of randomised controlled trials. Arthritis Res Ther. 2020;22:28. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 66] [Cited by in RCA: 120] [Article Influence: 24.0] [Reference Citation Analysis (0)] |