Published online May 26, 2021. doi: 10.12998/wjcc.v9.i15.3546
Peer-review started: January 5, 2021
First decision: January 17, 2021
Revised: January 21, 2021
Accepted: March 6, 2021
Article in press: March 6, 2021
Published online: May 26, 2021
Processing time: 125 Days and 20.4 Hours
The effectiveness of adjunctive corticosteroid use in patients with coronavirus disease 2019 (COVID-19) remains inconclusive.
To investigate the effectiveness of adjunctive corticosteroid therapy in patients with severe COVID-19.
We conducted a retrospective analysis of the difference in several outcomes between patients with severe COVID-19 who received corticosteroid therapy (the corticosteroid group) and patients with severe COVID-19 who did not receive corticosteroid therapy (the non-corticosteroid group).
Seventy-five patients were included in this study. Of these, 47 patients were in the corticosteroid group and 28 patients were in the non-corticosteroid group. There were no differences between the two groups in the total length of hospital stay, the length of intensive care unit stay, high-flow oxygen days, non-invasive ventilator days, invasive ventilation days, and mortality rate. Total lesion volume ratio, consolidation volume ratio and ground-glass opacity volume ratio in the corticosteroid group decreased significantly on day 14, while those in the non-corticosteroid group did not show a significant decrease.
Our results show that adjunctive corticosteroid use did not significantly improve clinical outcomes in severe COVID-19 patients, but might promote the absorption of pulmonary lesions. Larger multicenter randomized controlled studies may be needed to confirm this.
Core Tip: Corticosteroids have been used in the treatment of severe acute respiratory syndrome, Middle East respiratory syndrome and coronavirus disease 2019 (COVID-19). Many studies believe that corticosteroids have an inhibitory effect on inflammatory factors caused by viruses. In this study, 75 patients with severe COVID-19 were studied and divided into either the treatment group or the control group according to corticosteroid use. We found that adjunctive corticosteroid use did not significantly improve clinical outcomes in severe COVID-19 patients, but might promote the absorption of pulmonary lesions.
- Citation: Xiong B, He LM, Qin YY, Du H, Zhan Z, Zhou YH, Chen YK, Zhang A. Effectiveness of adjunctive corticosteroid therapy in patients with severe COVID-19: A retrospective cohort study. World J Clin Cases 2021; 9(15): 3546-3558
- URL: https://www.wjgnet.com/2307-8960/full/v9/i15/3546.htm
- DOI: https://dx.doi.org/10.12998/wjcc.v9.i15.3546
The outbreak of coronavirus disease 2019 (COVID-19) is currently a serious global public health challenge. COVID-19 is caused by the novel coronavirus, severe acute respiratory syndrome coronavirus 2 (SARS-CoV-2), which is an enveloped RNA virus and is mainly transmitted through the respiratory tract, causing damage to the human respiratory system, systemic inflammatory reactions, and multiple organ failure in certain cases[1,2]. Prior to the COVID-19 outbreak, two similar coronavirus outbreaks had already occurred in Asia, including the outbreak of SARS-CoV in 2003[3] and the outbreak of Middle East respiratory syndrome (MERS) coronavirus in 2012[4,5].
According to existing literature, the symptoms of patients with COVID-19 are non-specific, and include fever, cough and myalgia, accompanied by diarrhea, with or without the subsequent development of dyspnea. Severe cases involve acute respiratory distress syndrome (ARDS), sepsis, and septic shock[6-10]. While symptomatic supportive treatment may be administered, there is currently no specific antiviral treatment for COVID-19. Different organizations, authoritative institutions, and scholars have successively released a variety of COVID-19 diagnosis or treatment plans or opinions, but drug treatment recommendations have been inconsistent. Corticosteroids have an inhibitory effect on inflammatory factors, and are often used as an adjuvant treatment for viral pneumonia. Studies have shown that the use of corticosteroids can help suppress excessive inflammatory reactions; nevertheless, the use of corticosteroids may delay the elimination of the virus and may result in serious adverse effects[11]. To date, the effectiveness of systemic corticosteroid use in patients with COVID-19 remains unknown. The aim of the present study was to investigate the effectiveness of adjunctive corticosteroid administration in patients with severe COVID-19.
The Ethics Commission of the Chongqing Public Health Medical Center approved this study (No. 2020-025-KY). Written informed consent was waived due to the rapid emergence of this infectious disease.
The present study is a retrospective cohort study, which included all severe COVID-19 patients admitted to Chongqing Public Health Medical Center and Chongqing Three Gorges Central Hospital from January to March, 2020.
The diagnosis of severe COVID-19 in subjects had to meet the following criteria: (1) Identification of COVID-19 via real-time reverse transcription-polymerase chain reaction and/or next-generation sequencing; and (2) Having at least one of the following conditions: (a) Respiratory distress (≥ 30 times/min); (b) Oxygen saturation ≤ 93% at rest; (c) Oxygenation index (PaO2/FiO2) ≤ 300 mmHg; (d) Respiratory failure requiring mechanical ventilation; (e) Septic shock development; and (f) Critical organ failure requiring intensive care unit (ICU) care. Shock was defined according to the World Health Organization guidelines for COVID-19[12]. Acute kidney injury was identified on the basis of serum creatinine[13]. Cardiac injury was diagnosed if the serum concentration of hypersensitive cardiac troponin I was above the upper limit of the reference range (> 28 pg/mL), measured in the laboratory of the designated hospital.
All patients received similar conventional COVID-19 treatment according to the Chinese Clinical Guidance for COVID-19 Pneumonia Diagnosis and Treatment (Trial Version 5)[14]. We stratified the included patients into two groups according to whether they received corticosteroids (methylprednisolone via intravenous injection at a dose of 1-2 mg/kg/d for 3-5 d) in their treatment regimens or not, namely, the corticosteroid group and the non-corticosteroid group.
We reviewed clinical electronic medical records, nursing records, laboratory findings, and radiological examinations for all patients with the diagnosis of severe COVID-19. The admission data of these patients from day 0 (D 0), which was defined as the day of severe COVID-19 diagnosis to discharge or death were collected. Data were evaluated and collated, using case record forms. We collected data on gender, age, smoking, vital signs (heart rate, respiratory rate, mean arterial pressure), chronic medical history (hypertension, diabetes, coronary heart disease (CAD), chronic obstructive pulmonary disease (COPD), cerebrovascular disease, viral hepatitis, neoplastic disease, chronic kidney disease (CKD), symptoms from onset to hospital admission (fever, cough, sputum, dyspnea, weakness, headache, diarrhea), treatment (oxygen therapy, antibiotic agents, antifungal agents, immunoglobulin, thymopentin (or thymalfasin), and extracorporeal membrane oxygenation (ECMO), acute physiology and chronic health assessment (APACHE) II score, as well as total length of hospital stay (LOS), and length of ICU stay.
Routine blood test results were also collected, including blood gas analysis, complete blood count, and serum biochemical tests, including electrolytes, lactate dehydrogenase, liver and kidney function, coagulation function, cardiac enzymes, interleukin-6 (IL-6), cluster of differentiation (CD)-4-positive T-cell count, CD-8-positive T-cell count, C-reactive protein (CRP), D-dimer, and procalcitonin (PCT). Chest radiographs or computed tomography (CT) scans were also performed for all hospitalized patients, and the frequency of examinations was determined by the attending physician.
We assessed the changes in APACHE II score and oxygenation index (PaO2/FiO2), the changes in lymphocyte count, CD4+ T-cell count, CD8+ T-cell count, pulmonary lesion volume, LOS, and length of ICU stay, proportions of patients using mechanical ventilation, and mortality rate.
The measurement of the extent of pneumonia was performed with the FACT Medical Imaging System (Dexin Medical Imaging Technology Co., Ltd.), in which Pulmonary Infection Assisted Diagnosis (V1.7.0.1) automatically segmented the entire lung and lesions depicted on CT images using the “break and repair” shape analysis strategy, and then calculated the volumes of the entire lung and all lesions. Based on the initial automatic segmentation, the boundaries of each lesion were further precisely adjusted by manual tools to avoid the influence of non-inflammatory lesions such as large blood vessels, pleural effusion, pleural thickening, and calcification. In addition, according to the definitions of ground-glass opacity (GGO) and consolidation in the Fleischner Society Recommendations[15,16], we further measured the volume occupied by GGOs and consolidation components in the lesions. After completing all adjustments, the system automatically calculated the volumes of the lesions, GGOs, consolidation, and that of the whole lung. Finally, the volume ratio was calculated by the following formulas: total lesion volume ratio = (volume of all lesions)/(volume of whole lung) × 100%; GGO volume ratio = (volume of GGOs)/(volume of whole lung) × 100%; and consolidation volume ratio = (volume of consolidation)/(volume of whole lung) × 100%.
The data were analyzed with IBM SPSS software version 26 (IBM-SPSS Inc., Chicago, IL, United States). The corticosteroid group and the non-corticosteroid group were initially analyzed by descriptive statistical methods. We performed the Kolmogorov-Smirnov test to evaluate the normality of the data distributions. Continuous variables are presented as the mean ± SD, and independent samples were analyzed by the t-test. Categorical variables were analyzed using the Chi-squared test or the Fisher's exact test. Differences were statistically significant at P < 0.05.
The demographic and clinical characteristics of the patients are summarized in Table 1. Seventy-five patients with severe COVID-19 were included in this study. The mean age was 58.92 ± 14.29 years, and 39 (52%) patients were male. Of the 75 patients, 47 (62.7%) received corticosteroids and were therefore classified into the corticosteroid group, whereas 28 (37.3%) did not receive corticosteroids and were therefore classified into the non-corticosteroid group. All 47 patients in the corticosteroid group received corticosteroids within 72 h of severe COVID-19 diagnosis. The symptoms of COVID-19 in the included patients were fever (61.33%), cough (76%), dyspnea (53.33%), sputum production (34.67%), and weakness (29.33%). There were no statistically calculated differences between the two groups of patients in terms of gender, age, smoking, APACHE II score, hypertension, diabetes, CAD, COPD, cerebrovascular disease, viral hepatitis, neoplastic disease, CKD, fever, cough, sputum, weakness, headache, diarrhea, shock, leukopenia, thrombocytopenia, liver dysfunction, acute kidney injury, or cardiac injury, as displayed in Table 1.
| Variables | Total (n = 75) | Corticosteroid group (n = 47) | Non-corticosteroid group (n = 28) | P value |
| Demographic factors | ||||
| Male (%) | 39 (52.00) | 25 (53.19) | 14 (50.00) | 0.789 |
| Age (%) | 58.92 ± 14.29 | 57.09 ± 13.74 | 62.00 ± 14.90 | 0.151 |
| Smoking (%) | 9 (12.00) | 8 (17.02) | 1 (3.57) | 0.083 |
| APACHE II | 7.97 ± 3.37 | 8.24 ± 3.57 | 7.52 ± 3.02 | 0.382 |
| Comorbidity | ||||
| Hypertension (%) | 13 (17.33) | 7 (14.89) | 6 (21.43) | 0.470 |
| Diabetes (%) | 19 (25.33) | 13 (27.66) | 6 (21.43) | 0.548 |
| CHD (%) | 4 (5.33) | 2 (4.26) | 2 (7.14) | 0.590 |
| COPD (%) | 7 (9.33) | 3 (6.38) | 4 (14.28) | 0.437 |
| Cerebrovascular disease (%) | 1 (1.33) | 1 (2.13) | 0 | 1.000 |
| Viral hepatitis (%) | 1 (1.33) | 1 (2.13) | 0 | 1.000 |
| Tumor (%) | 2 (2.67) | 1 (2.13) | 1 (3.57) | 0.707 |
| CKD (%) | 1 (1.33) | 1 (2.13) | 0 | 1.000 |
| Symptoms | ||||
| Fever (%) | 46 (61.33) | 31 (65.96) | 15 (53.57) | 0.287 |
| Cough (%) | 57 (76.00) | 37 (78.72) | 20 (71.43) | 0.474 |
| Sputum (%) | 26 (34.67) | 15 (31.91) | 11 (39.29) | 0.516 |
| Dyspnea (%) | 40 (53.33) | 30 (63.83) | 10 (35.71) | 0.018 |
| Weakness (%) | 22 (29.33) | 12 (25.53) | 10 (35.71) | 0.349 |
| Headache (%) | 8 (10.67) | 4 (8.51) | 4 (14.28) | 0.433 |
| Diarrhea (%) | 6 (8.00) | 3 (6.38) | 3 (10.71) | 0.819 |
| Complication | ||||
| Shock (%) | 3 (4.00) | 3 (6.38) | 0 | 0.450 |
| Leukopenia (%) | 4 (5.33) | 3 (6.38) | 1 (3.57) | 0.600 |
| Thrombocytopenia (%) | 2 (2.67) | 2 (4.26) | 0 | 0.715 |
| Liver dysfunction (%) | 3 (4.00) | 2 (4.26) | 1 (3.57) | 0.884 |
| AKI (%) | 2 (2.67) | 1 (2.13) | 1 (3.57) | 0.707 |
| Cardiac injury (%) | 2 (2.67) | 2 (4.26) | 0 | 0.715 |
Approximately 83% of patients (62/75) received antibiotic therapy, and 12 (16%) patients received antifungal therapy. Sixty-three patients (84%) received thymopentin or thymalfasin, and 30 patients (42.76%) received intravenous (IV) injection of immunoglobulin. Twenty-eight patients (37.33%) received non-invasive ventilation (NIV), 7 patients (9.33%) received invasive ventilation, and three patients (4%) received ECMO therapy.
In the corticosteroid group, the vast majority of enrolled patients (95.74%) received antibiotics, and 25.53% of patients received antifungal therapy; the proportion of patients receiving antibiotics and antifungals was significantly higher in the corticosteroid group compared to the non-corticosteroid group (P < 0.05). Nevertheless, we found no differences between the two groups in terms of total LOS, length of ICU stay, high-flow oxygen days, NIV days, invasive ventilator days, and mortality rate, as indicated in Table 2.
| Variables | Total (n = 75) | Corticosteroid group (n = 47) | Non-corticosteroid group (n = 28) | P value |
| Treatments | ||||
| Antibiotics (%) | 62 (82.67) | 45 (95.74) | 17 (60.71) | < 0.001 |
| Antifungal (%) | 12 (16.00) | 12 (25.53) | 0 | 0.004 |
| IV immunoglobulin (%) | 30 (42.76) | 27 (57.45) | 3 (10.71) | < 0.001 |
| Thymopentin or thymalfasin (%) | 63 (84.00) | 43 (91.49) | 20 (71.43) | 0.022 |
| High-flow oxygen (%) | 40 (53.33) | 28 (59.57) | 12 (42.86) | 0.160 |
| Noninvasive ventilator (%) | 28 (37.33) | 24 (51.06) | 4 (14.28) | 0.001 |
| Invasive ventilator (%) | 7 (9.33) | 7 (14.89) | 0 | 0.032 |
| ECMO (%) | 3 (4.00) | 3 (6.38) | 0 | 0.450 |
| Outcomes | ||||
| Total length of hospital stay | 21.05 ± 8.96 | 21.21 ± 9.57 | 20.79 ± 7.98 | 0.843 |
| Length of ICU stay | 14.64 ± 6.67 | 15.23 ± 6.72 | 13.64 ± 6.58 | 0.321 |
| High-flow oxygen days | 5.58 ± 4.19 | 5.32 ± 3.58 | 6.17 ± 5.49 | 0.565 |
| Noninvasive ventilator days | 6.69 ± 5.37 | 7.54 ± 5.46 | 2.60 ± 2.30 | 0.060 |
| Invasive ventilator days | 9.00 ± 6.14 | 9.00 ± 6.14 | - | - |
| Mortality rate (%) | 4 (5.33) | 4 (8.51) | 0 | 0.291 |
There were no significant differences in APACHE II scores between the corticosteroid group and the non-corticosteroid group on D 0 or D 14. Moreover, the APACHE II score significantly decreased in each of the two groups from D 0 to D14 (P < 0.05), as shown in Table 3.
| Variables | Corticosteroid group | Non-corticosteroid group | ||
| D 0 | D 14 | D 0 | D 14 | |
| Heart rate (bpm) | 84 ± 16 | 80 ± 16 | 88 ± 15 | 74 ± 6b |
| Respiratory rate | 24 ± 12 | 20 ± 2 | 20 ± 2 | 20 ± 2 |
| MAP (mmHg) | 90 ± 10 | 87 ± 11 | 91 ± 12 | 82 ± 21b |
| Body temperature (℃) | 37.4 ± 1.0 | 36.8 ± 0.5b | 37.3 ± 0.8 | 36.5 ± 0.3b |
| APACHE II | 8.24 ± 3.57 | 6.72 ± 4.87b | 7.52 ± 3.02 | 6.08 ± 3.39b |
| P/F ratio (mmHg) | 196.68 ± 105.70 | 297.47 ± 135.68b | 239.00 ± 107.76 | 308.20 ± 102.54b |
| WBC (× 109/L) | 6.06 ± 3.17 | 6.87 ± 2.70 | 6.24 ± 2.72 | 6.36 ± 1.68 |
| N (× 109/L) | 4.91 ± 3.11 | 5.46 ± 2.67 | 4.75 ± 2.57 | 4.11 ± 1.11 |
| L (× 109/L) | 0.79 ± 0.34a | 0.84 ± 0.54a | 1.02 ± 0.39 | 1.13 ± 0.31 |
| PCT (ng/mL) | 0.14 ± 0.13 | 0.12 ± 0.11 | 0.07 ± 0.05 | 0.05 ± 0.04b |
| ALT (U/L) | 53.86 ± 45.69 | 56.18 ± 52.36 | 27.06 ± 14.50 | 38.42 ± 26.58b |
| AST (U/L) | 52.39 ± 44.33 | 60.15 ± 49.77 | 31.78 ± 10.39 | 29.49 ± 17.07 |
| LDH (U/L) | 365.63 ± 169.73 | - | 321.50 ± 85.82 | - |
| Albumin (g/L) | 36.27 ± 4.25 | 34.12 ± 3.28 | 37.49 ± 3.93 | 33.21 ± 3.64b |
| TB (μmol/L) | 18.22 ± 6.59 | - | 14.43 ± 6.96 | - |
| CRP (mg/L) | 83.19 ± 63.09 | 27.15 ± 20.05b | 70.92 ± 61.73 | 18.64 ± 11.09b |
| D-dimer (mg/L) | 1.86 ± 1.42 | 4.03 ± 5.64b | 0.58 ± 0.47 | 1.01 ± 0.89 |
| IL 10 (pg/mL) | 4.51 ± 2.37 | 6.99 ± 6.35b | 4.68 ± 1.37 | 3.47 ± 0.78b |
| IL 6 (pg/mL) | 43.35 ± 34.19 | 40.16 ± 28.61 | 33.03 ± 23.69 | 31.86 ± 27.13 |
| CD 4+ T (cells/μL) | 253.20 ± 144.84 | 374.76 ± 195.80b | 298.31 ± 164.52 | 430.20 ± 161.40b |
| CD 8+ T (cells/μL) | 157.55 ± 103.76a | 285.95 ± 248.60b | 220.55 ± 110.87 | 275.29 ± 108.06b |
| CD 4/CD 8 Ratio | 1.70 ± 0.84 | 1.91 ± 1.39b | 1.51 ± 0.63 | 1.68 ± 0.61 |
| PT (s) | 11.70 ± 1.30 | 11.41 ± 1.76 | 12.03 ± 2.16 | 11.42 ± 0.74 |
| Cr (μmol/L) | 64.51 ± 17.13 | 66.99 ± 24.80 | 68.94 ± 30.99 | 73.91 ± 50.68 |
We did not observe a significant difference in the oxygenation index between the corticosteroid group and the non-corticosteroid group on D 0 or D 14. In addition, the oxygenation index in each of the two groups significantly increased from D 0 to D 14 (P < 0.05).
At the same time, we found that inflammatory indicators at baseline [e.g., CRP, PCT, white blood cell (WBC), D-dimer, IL-6, and IL-10] and some serum biochemical indicators were not significantly different between the two groups on D 0 or D 14.
Some inflammatory factors in the corticosteroid group, including D-dimer and IL-10, increased over time, while CRP decreased significantly from D 0 to D 14 (P < 0.05). In addition, some inflammatory factors in the non-corticosteroid group, including PCT, CRP, and IL-10, decreased significantly from D 0 to D 14 (P < 0.05).
Subsequently, compared with the non-corticosteroid group, we found that CD4+, but not CD8+ T-cell counts were significantly decreased in both groups from D 0 to D 14 (P < 0.05). These findings are illustrated in detail in Table 3.
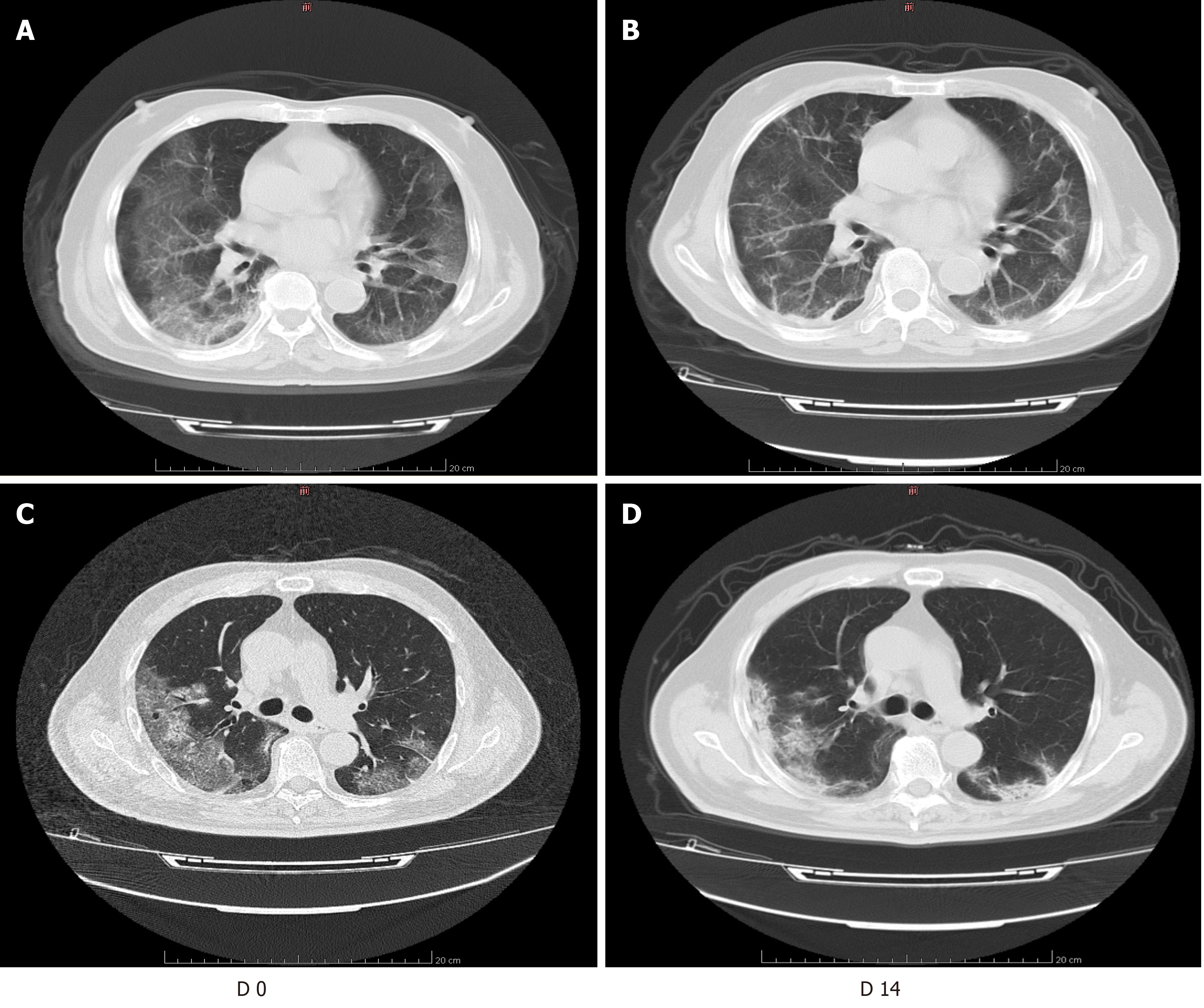
Table 4 shows the results of imaging evaluations in each group of patients with severe COVID-19. We found that common chest CT features of COVID-19 patients mainly included consolidation, GGOs, and linear opacities. The total lesion volume ratio, consolidation volume ratio, and GGO volume ratio were significantly higher in the corticosteroid group than in the non-corticosteroid group on D 0 (P < 0.05). However, there was no significant difference in these indicators between the two groups on D 14 (P > 0.05). These findings are illustrated in Figure 1.
We found that total lesion volume ratio and GGO volume ratio in both groups decreased over time, while total lesion volume ratio and GGO volume ratio in the corticosteroid group decreased significantly on D 14, which was different to that in the non-corticosteroid group. However, unlike patients in the corticosteroid group, whose consolidation volume ratio decreased significantly on D 14, we found that the consolidation volume ratio in the non-corticosteroid group increased significantly on D 14.
In this study, we found that the levels of inflammatory indicators (e.g., CRP, PCT, WBC, D-dimer, IL-6, and IL-10) were increased in patients with COVID-19, lymphocyte, CD4+, and CD8+ T-cell counts were decreased in patients with COVID-19, and common chest CT features in COVID-19 patients were consolidation and GGOs, compared with the normal reference value. The lymphocyte, CD4+, and CD8+ T-cell counts were significantly reduced in the adjunctive corticosteroid group compared with those in the conventional corticosteroid group, but there were no significant differences in the APACHE II scores, oxygenation indices (PaO2/FiO2), and LOS between these groups. The proportion of patients receiving mechanical ventilation was significantly higher in the corticosteroid group than in the non-corticosteroid group. Among the other parameters, only the duration of NIV was significantly different between the groups. In addition, 4 patients died in the corticosteroid group and no patients died in the non-corticosteroid group.
It is well known that in the treatment of viral pneumonia, the intermediate-acting corticosteroid methylprednisolone sodium succinate is commonly used due to its rapid onset, short biological half-life, good safety profile, and weak inhibition of the hypothalamus-pituitary-adrenal axis; in addition, it is the only steroidal drug available for shock therapy[11]. In the treatment of COVID-19 patients, clinicians use corticosteroids mainly because of its anti-inflammatory effects[17]. Corticosteroids can act on various stages and suppress multiple links of the immune response. In the early stages of inflammation, corticosteroids can reduce capillary dilation, inflammatory cell exudation, leukocyte infiltration, and phagocytosis. In later phases of the inflammatory response, corticosteroids can inhibit the excessive proliferation of capillaries and fibroblasts[18]. Corticosteroids can act on corticosteroid receptors, inhibit nuclear transcription factor nuclear factor-kappa B signaling, and further inhibit the transcription and translation of inflammatory factors. They may also play a role by inhibiting IL-2 gene transcription and then inhibiting T-cell clonal proliferation. Furthermore, corticosteroids can inhibit the induction of the immune response and affect the expression of interferon-γ, tumor necrosis factor-α, IL-1 and other cytokines[19].
Pathological reports of patients who died from COVID-19 indicate that the lungs displayed alveolar injury with fibro-mucosal exudation, alveolar epithelial detachment, and pulmonary hyaline membrane formation, consistent with ARDS; inflammatory infiltration of mononuclear cells, mainly lymphocytes, in the interstitium was seen in the lungs, and the overall pathological changes were similar to those observed in patients who died from SARS and MERS[20]. Pathological findings of patients with SARS showed significant increases in T and B lymphocyte apoptosis, and mononuclear and macrophage numbers in immune organs. Similarly, in patients who died of COVID-19, there was a decrease in peripheral blood lymphocytes, but lymphocytes were also over-activated, while the numbers of highly pro-inflammatory CCR4+, CCR6+, and Th17 cells increased. The above results suggest that there is a severe reduction in anti-inflammatory immune cells, and an increase in immune response-related damage in the lungs[20]. Similarly, the results of previous studies showed that 44.4% (12/27) of patients with mild COVID-19 and 84.6% (11/13) of patients with severe COVID-19 had lymphopenia. The absolute count of peripheral blood lymphocytes in patients with severe COVID-19 was significantly lower than that in patients with mild COVID-19; the counts of CD3+, CD4+, and CD8+ T-cell subsets were lower in patients with severe COVID-19 than in patients with mild COVID-19[21]. Recent studies have shown that the numbers of total T-cells, CD4+ T-cells, and CD8+ T-cells were significantly reduced in COVID-19 patients[22], which is consistent with our findings. We also found that compared with patients in the conventional corticosteroid group, COVID-19 patients treated with adjunctive methylprednisolone had significantly lower absolute counts of CD4+ and CD8+ T-cells. This is similar to previous reports by Ghoneim et al[23], who found that the early use of corticosteroids (dexamethasone) to treat flu in rats significantly reduced the counts of CD4+ and CD8+ T-cells on day 7. A possible reason for this is that the use of corticosteroids may cause systemic immunosuppression[24,25], which has a cytotoxic effect on CD4+ T-cells[26] and is not conducive to viral elimination, resulting in delayed virus clearance and accelerated disease progression.
In the early course of corticosteroid treatment of viral pneumonia, studies have shown that the adverse effects of glucocorticoid use are significantly greater than the anti-inflammatory effects. In patients with SARS infections, long-term high-dose corticosteroids were used, and there were serious adverse reactions such as invasive fungal infections and femoral head necrosis[27]. Brun-Buisson et al[28] studied the use of corticosteroids for the treatment of ARDS caused by influenza A (H1N1) virus and found that the incidence of acquired pneumonia and death was higher in patients with the early use of corticosteroids than in those who did not receive corticosteroid therapy. In addition, the duration of mechanical ventilation in severely ill patients using corticosteroids was significantly prolonged[28]. In reports of adverse reactions in patients with SARS treated with corticosteroids, the adverse reactions were often positively correlated with the dose and timing of administration of corticosteroids[29]. Earlier use of corticosteroids to treat mild SARS and MERS patients was found to increase SARS viral load in patients, and to delay the clearance of MERS virus RNA[30,31]. A meta-analysis published in 2019 that included ten observational studies on influenza found that the mortality rate, LOS in the ICU, and incidence of secondary bacterial or fungal infections were increased in patients receiving corticosteroids[32]. However, it is worth noting that a series of clinical trials have shown that when low-dose or physiological-dose corticosteroids are used to treat patients with septic shock caused by lung infection, the use of corticosteroids can significantly reduce the mortality of such patients, and can reverse shock, shorten the ICU LOS, and reduce the use of mechanical ventilation[33,34]. However, our study found that adjunctive corticosteroids did not significantly improve the oxygenation index (PaO2/FiO2 ratio), shorten the LOS (including ICU stay), reduce mechanical ventilation usage time, or decrease mortality. At the same time, Wang et al[35] found that no positive effects on outcomes were observed after the use of methylprednisolone. However, recent studies have found that systemic corticosteroid treatment in the initial 3 to 5 d can enhance the ratio of blood oxygen saturation and arterial oxygen tension to the oxygen fraction in patients with severe COVID-19, and ventilation can further enhance these two indicators[36]. Unlike COVID-19 patients with ARDS, COVID-19 patients with shock or multiple organ injury derive no additional survival benefit from corticosteroid therapy. However, the use of corticosteroids can significantly inhibit the inflammatory cytokine storm that occurs in the ARDS stage in COVID-19 patients, giving patients valuable time to control the infection, and prevent secondary multiple organ damage and septic shock[36].
It is well known that corticosteroids do not directly inhibit viral replication. Their main role is to reduce inflammation and suppress the immune response. The anti-inflammatory effect is mainly manifested in the reduction of alveolar exudation and capillary permeability. The use of corticosteroids can delay the progression of mild pneumonia to ARDS, which can alleviate the occurrence of pulmonary fibrosis[37]. Studies have shown that when SARS patients have increased shadows on lung imaging and increased dyspnea, the early and appropriate use of corticosteroids can significantly improve the patients' clinical symptoms, reduce the degree of disease progression, and accelerate the absorption of lung lesions[38,39]. Our study found that the addition of methylprednisolone can significantly improve lung lesions in patients with severe COVID-19 on D 14. We found that clinicians prefer to use corticosteroids in patients with more extensive lung lesions. This may be related to the fact that pulmonary lesions were significantly worse in the corticosteroid group than in the non-corticosteroid group at baseline. These patients were critically ill, progressed rapidly, were difficult to treat, and required enhanced immunotherapy.
To a certain extent, our findings have several biases, which are inherent flaws of retrospective studies. In addition, due to the low numbers of patients with severe COVID-19 in Chongqing, a limited number of critically ill patients were included in this study. Studies utilizing a larger sample size are warranted.
In summary, we found that adjunctive corticosteroids do not significantly improve clinical outcomes in patients with severe COVID-19, but might promote the absorption of pulmonary lesions. Larger multicenter randomized controlled studies may be needed to confirm this.
The outbreak of coronavirus disease 2019 (COVID-19), caused by severe acute respiratory syndrome coronavirus 2 (SARS-CoV-2), is currently a serious global public health challenge. Severe cases involve acute respiratory distress syndrome, sepsis, and septic shock. At present, there is no significant and effective drug for severe COVID-19 patients. Corticosteroids have been used in the treatment of virus pneumonia, such as SARS, Middle East respiratory syndrome, and influenza A, but their efficacy is still inconclusive.
In clinical practice, some severe COVID-19 patients will benefit from the application of corticosteroids, but there are also adverse reactions. Therefore, whether corticosteroids should be used in COVID-19 patients and how to use them are issues worthy of discussion.
The aim of the present study was to investigate the effectiveness of adjunctive corticosteroid administration in patients with severe COVID-19.
Seventy-five patients with severe COVID-19 were divided into the corticosteroid group (47, 62.7%) and the non-corticosteroid group (28, 37.3%). In the corticosteroid group, methylprednisolone was administered via intravenous injection at a dose of 1-2 mg/kg/day for 3-5 d. We assessed the changes in APACHE II score and oxygenation index (PaO2/FiO2), the changes in lymphocyte count, CD4+ T-cell count, CD8+ T-cell count, pulmonary lesion volume, LOS, and length of ICU stay, proportions of patients using mechanical ventilation, and mortality rate. We measured the extent of pneumonia with the FACT Medical Imaging System, and calculated the volumes of the lesions, ground-glass opacity (GGO), consolidation, and that of the whole lung. Finally, the volume ratio was calculated by formulas.
Seventy-five patients were included in this study. Of these, 47 patients were in the corticosteroid group and 28 patients were in the non-corticosteroid group. There were no differences between the two groups in the total length of hospital stay, the length of ICU stay, high-flow oxygen days, non-invasive ventilator days, invasive ventilation days, and mortality rate. Total lesion volume ratio, consolidation volume ratio and GGO volume ratio in the corticosteroid group decreased significantly on day 14, while those in the non-corticosteroid group did not show a significant decrease.
Adjunctive corticosteroid use did not significantly improve clinical outcomes in severe COVID-19 patients, but might promote the absorption of pulmonary lesions.
Adjunctive corticosteroid treatment in severe COVID-19 patients should be comprehensively evaluated and used with caution.
We thank all the patients involved in the study.
Manuscript source: Unsolicited manuscript
Corresponding Author's Membership in Professional Societies: Chinese Society of Critical Care Medicine, 2021-2025.
Specialty type: Infectious diseases
Country/Territory of origin: China
Peer-review report’s scientific quality classification
Grade A (Excellent): 0
Grade B (Very good): B
Grade C (Good): 0
Grade D (Fair): 0
Grade E (Poor): 0
P-Reviewer: Mohammadi M S-Editor: Zhang H L-Editor: Webster JR P-Editor: Xing YX
| 1. | Gao GF. From "A"IV to "Z"IKV: Attacks from Emerging and Re-emerging Pathogens. Cell. 2018;172:1157-1159. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 155] [Cited by in RCA: 187] [Article Influence: 31.2] [Reference Citation Analysis (0)] |
| 2. | Heymann DL, Shindo N; WHO Scientific and Technical Advisory Group for Infectious Hazards. COVID-19: what is next for public health? Lancet. 2020;395:542-545. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 663] [Cited by in RCA: 581] [Article Influence: 116.2] [Reference Citation Analysis (0)] |
| 3. | Zhong NS, Zheng BJ, Li YM, Poon, Xie ZH, Chan KH, Li PH, Tan SY, Chang Q, Xie JP, Liu XQ, Xu J, Li DX, Yuen KY, Peiris, Guan Y. Epidemiology and cause of severe acute respiratory syndrome (SARS) in Guangdong, People's Republic of China, in February, 2003. Lancet. 2003;362:1353-1358. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 1047] [Cited by in RCA: 1001] [Article Influence: 45.5] [Reference Citation Analysis (0)] |
| 4. | Zaki AM, van Boheemen S, Bestebroer TM, Osterhaus AD, Fouchier RA. Isolation of a novel coronavirus from a man with pneumonia in Saudi Arabia. N Engl J Med. 2012;367:1814-1820. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 4030] [Cited by in RCA: 4029] [Article Influence: 309.9] [Reference Citation Analysis (0)] |
| 5. | Liu J, Zheng X, Tong Q, Li W, Wang B, Sutter K, Trilling M, Lu M, Dittmer U, Yang D. Overlapping and discrete aspects of the pathology and pathogenesis of the emerging human pathogenic coronaviruses SARS-CoV, MERS-CoV, and 2019-nCoV. J Med Virol. 2020;92:491-494. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 411] [Cited by in RCA: 373] [Article Influence: 74.6] [Reference Citation Analysis (0)] |
| 6. | Rodriguez-Morales AJ, Cardona-Ospina JA, Gutiérrez-Ocampo E, Villamizar-Peña R, Holguin-Rivera Y, Escalera-Antezana JP, Alvarado-Arnez LE, Bonilla-Aldana DK, Franco-Paredes C, Henao-Martinez AF, Paniz-Mondolfi A, Lagos-Grisales GJ, Ramírez-Vallejo E, Suárez JA, Zambrano LI, Villamil-Gómez WE, Balbin-Ramon GJ, Rabaan AA, Harapan H, Dhama K, Nishiura H, Kataoka H, Ahmad T, Sah R; Latin American Network of Coronavirus Disease 2019-COVID-19 Research (LANCOVID-19). Electronic address: https://www.lancovid.org. Clinical, laboratory and imaging features of COVID-19: A systematic review and meta-analysis. Travel Med Infect Dis. 2020;34:101623. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 1647] [Cited by in RCA: 1432] [Article Influence: 286.4] [Reference Citation Analysis (0)] |
| 7. | Huang C, Wang Y, Li X, Ren L, Zhao J, Hu Y, Zhang L, Fan G, Xu J, Gu X, Cheng Z, Yu T, Xia J, Wei Y, Wu W, Xie X, Yin W, Li H, Liu M, Xiao Y, Gao H, Guo L, Xie J, Wang G, Jiang R, Gao Z, Jin Q, Wang J, Cao B. Clinical features of patients infected with 2019 novel coronavirus in Wuhan, China. Lancet. 2020;395:497-506. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 35178] [Cited by in RCA: 30106] [Article Influence: 6021.2] [Reference Citation Analysis (3)] |
| 8. | Wang C, Horby PW, Hayden FG, Gao GF. A novel coronavirus outbreak of global health concern. Lancet. 2020;395:470-473. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 4848] [Cited by in RCA: 4386] [Article Influence: 877.2] [Reference Citation Analysis (1)] |
| 9. | Chan JF, Yuan S, Kok KH, To KK, Chu H, Yang J, Xing F, Liu J, Yip CC, Poon RW, Tsoi HW, Lo SK, Chan KH, Poon VK, Chan WM, Ip JD, Cai JP, Cheng VC, Chen H, Hui CK, Yuen KY. A familial cluster of pneumonia associated with the 2019 novel coronavirus indicating person-to-person transmission: a study of a family cluster. Lancet. 2020;395:514-523. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 6483] [Cited by in RCA: 5422] [Article Influence: 1084.4] [Reference Citation Analysis (0)] |
| 10. | Chen N, Zhou M, Dong X, Qu J, Gong F, Han Y, Qiu Y, Wang J, Liu Y, Wei Y, Xia J, Yu T, Zhang X, Zhang L. Epidemiological and clinical characteristics of 99 cases of 2019 novel coronavirus pneumonia in Wuhan, China: a descriptive study. Lancet. 2020;395:507-513. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 14869] [Cited by in RCA: 12971] [Article Influence: 2594.2] [Reference Citation Analysis (1)] |
| 11. | Liu H, Li J, Chen M, Su J. Glucocorticoid treatment of suspected organizing pneumonia after H7N9 infection: A case report. Medicine (Baltimore). 2019;98:e16839. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 10] [Cited by in RCA: 11] [Article Influence: 1.8] [Reference Citation Analysis (0)] |
| 12. | World Health Organization. Clinical management of severe acute respiratory infection when novel coronavirus (nCoV) infection is suspected: interim guidance, 25 January 2020. Geneva: World Health Organization, 28 January 2020. |
| 13. | Kidney disease: improving global outcomes (KDIGO) acute kidney injury work group. In: KDIGO clinical practice guideline for acute kidney injury. March 2012. [cited February 8, 2020]. Available from: https://kdigo.org/wp-content/uploads/2016/10/KDIGO-2012-AKI-Guideline-English.pdf. |
| 14. | National Health Commission of the People’s Republic of China. Guidelines for the Diagnosis and Treatment of Novel Coronavirus (2019-nCoV) Infection by the National Health Commission (Trial Version 5) 2020. [cited February 8, 2020]. Available from: http://www.chinacdc.cn/jkzt/crb/zl/szkb_11803/jszl_11815/202001/W020200128207842237479.pdf. |
| 15. | Hansell DM, Bankier AA, MacMahon H, McLoud TC, Müller NL, Remy J. Fleischner Society: glossary of terms for thoracic imaging. Radiology. 2008;246:697-722. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 2471] [Cited by in RCA: 2674] [Article Influence: 157.3] [Reference Citation Analysis (0)] |
| 16. | Schoen K, Horvat N, Guerreiro NFC, de Castro I, de Giassi KS. Spectrum of clinical and radiographic findings in patients with diagnosis of H1N1 and correlation with clinical severity. BMC Infect Dis. 2019;19:964. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 115] [Cited by in RCA: 119] [Article Influence: 19.8] [Reference Citation Analysis (0)] |
| 17. | Shang L, Zhao J, Hu Y, Du R, Cao B. On the use of corticosteroids for 2019-nCoV pneumonia. Lancet. 2020;395:683-684. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 424] [Cited by in RCA: 448] [Article Influence: 89.6] [Reference Citation Analysis (0)] |
| 18. | Cruz-Topete D, Cidlowski JA. One hormone, two actions: anti- and pro-inflammatory effects of glucocorticoids. Neuroimmunomodulation. 2015;22:20-32. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 263] [Cited by in RCA: 310] [Article Influence: 31.0] [Reference Citation Analysis (0)] |
| 19. | Vandewalle J, Luypaert A, De Bosscher K, Libert C. Therapeutic Mechanisms of Glucocorticoids. Trends Endocrinol Metab. 2018;29:42-54. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 236] [Cited by in RCA: 354] [Article Influence: 50.6] [Reference Citation Analysis (0)] |
| 20. | Xu Z, Shi L, Wang Y, Zhang J, Huang L, Zhang C, Liu S, Zhao P, Liu H, Zhu L, Tai Y, Bai C, Gao T, Song J, Xia P, Dong J, Zhao J, Wang FS. Pathological findings of COVID-19 associated with acute respiratory distress syndrome. Lancet Respir Med. 2020;8:420-422. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 5228] [Cited by in RCA: 5781] [Article Influence: 1156.2] [Reference Citation Analysis (2)] |
| 21. | Liu J, Li S, Liu J, Liang B, Wang X, Wang H, Li W, Tong Q, Yi J, Zhao L, Xiong L, Guo C, Tian J, Luo J, Yao J, Pang R, Shen H, Peng C, Liu T, Zhang Q, Wu J, Xu L, Lu S, Wang B, Weng Z, Han C, Zhu H, Zhou R, Zhou H, Chen X, Ye P, Zhu B, Wang L, Zhou W, He S, He Y, Jie S, Wei P, Zhang J, Lu Y, Wang W, Zhang L, Li L, Zhou F, Wang J, Dittmer U, Lu M, Hu Y, Yang D, Zheng X. Longitudinal characteristics of lymphocyte responses and cytokine profiles in the peripheral blood of SARS-CoV-2 infected patients. EBioMedicine. 2020;55:102763. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 1171] [Cited by in RCA: 1207] [Article Influence: 241.4] [Reference Citation Analysis (0)] |
| 22. | Diao B, Wang C, Tan Y, Chen X, Liu Y, Ning L, Chen L, Li M, Wang G, Yuan Z, Feng Z, Zhang Y, Wu Y, Chen Y. Reduction and Functional Exhaustion of T Cells in Patients With Coronavirus Disease 2019 (COVID-19). Front Immunol. 2020;11:827. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 1687] [Cited by in RCA: 1757] [Article Influence: 351.4] [Reference Citation Analysis (0)] |
| 23. | Ghoneim HE, McCullers JA. Adjunctive corticosteroid therapy improves lung immunopathology and survival during severe secondary pneumococcal pneumonia in mice. J Infect Dis. 2014;209:1459-1468. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 34] [Cited by in RCA: 37] [Article Influence: 3.1] [Reference Citation Analysis (0)] |
| 24. | Schäcke H, Döcke WD, Asadullah K. Mechanisms involved in the side effects of glucocorticoids. Pharmacol Ther. 2002;96:23-43. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 1237] [Cited by in RCA: 1328] [Article Influence: 57.7] [Reference Citation Analysis (0)] |
| 25. | Rhen T, Cidlowski JA. Antiinflammatory action of glucocorticoids--new mechanisms for old drugs. N Engl J Med. 2005;353:1711-1723. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 2099] [Cited by in RCA: 2099] [Article Influence: 105.0] [Reference Citation Analysis (1)] |
| 26. | Wilkinson TM, Li CK, Chui CS, Huang AK, Perkins M, Liebner JC, Lambkin-Williams R, Gilbert A, Oxford J, Nicholas B, Staples KJ, Dong T, Douek DC, McMichael AJ, Xu XN. Preexisting influenza-specific CD4+ T cells correlate with disease protection against influenza challenge in humans. Nat Med. 2012;18:274-280. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 713] [Cited by in RCA: 810] [Article Influence: 62.3] [Reference Citation Analysis (0)] |
| 27. | Li YM, Wang SX, Gao HS, Wang JG, Wei CS, Chen LM, Hui WL, Yuan SL, Jiao ZS, Yang Z, Su B. [Factors of avascular necrosis of femoral head and osteoporosis in SARS patients' convalescence]. Zhonghua Yi Xue Za Zhi. 2004;84:1348-1353. [PubMed] |
| 28. | Brun-Buisson C, Richard JC, Mercat A, Thiébaut AC, Brochard L; REVA-SRLF A/H1N1v 2009 Registry Group. Early corticosteroids in severe influenza A/H1N1 pneumonia and acute respiratory distress syndrome. Am J Respir Crit Care Med. 2011;183:1200-1206. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 249] [Cited by in RCA: 259] [Article Influence: 18.5] [Reference Citation Analysis (0)] |
| 29. | Stockman LJ, Bellamy R, Garner P. SARS: systematic review of treatment effects. PLoS Med. 2006;3:e343. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 861] [Cited by in RCA: 861] [Article Influence: 45.3] [Reference Citation Analysis (0)] |
| 30. | Lee N, Allen Chan KC, Hui DS, Ng EK, Wu A, Chiu RW, Wong VW, Chan PK, Wong KT, Wong E, Cockram CS, Tam JS, Sung JJ, Lo YM. Effects of early corticosteroid treatment on plasma SARS-associated Coronavirus RNA concentrations in adult patients. J Clin Virol. 2004;31:304-309. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 413] [Cited by in RCA: 455] [Article Influence: 22.8] [Reference Citation Analysis (0)] |
| 31. | Arabi YM, Mandourah Y, Al-Hameed F, Sindi AA, Almekhlafi GA, Hussein MA, Jose J, Pinto R, Al-Omari A, Kharaba A, Almotairi A, Al Khatib K, Alraddadi B, Shalhoub S, Abdulmomen A, Qushmaq I, Mady A, Solaiman O, Al-Aithan AM, Al-Raddadi R, Ragab A, Balkhy HH, Al Harthy A, Deeb AM, Al Mutairi H, Al-Dawood A, Merson L, Hayden FG, Fowler RA; Saudi Critical Care Trial Group. Corticosteroid Therapy for Critically Ill Patients with Middle East Respiratory Syndrome. Am J Respir Crit Care Med. 2018;197:757-767. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 720] [Cited by in RCA: 791] [Article Influence: 131.8] [Reference Citation Analysis (0)] |
| 32. | Ni YN, Chen G, Sun J, Liang BM, Liang ZA. The effect of corticosteroids on mortality of patients with influenza pneumonia: a systematic review and meta-analysis. Crit Care. 2019;23:99. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 242] [Cited by in RCA: 271] [Article Influence: 45.2] [Reference Citation Analysis (0)] |
| 33. | Rhodes A, Evans LE, Alhazzani W, Levy MM, Antonelli M, Ferrer R, Kumar A, Sevransky JE, Sprung CL, Nunnally ME, Rochwerg B, Rubenfeld GD, Angus DC, Annane D, Beale RJ, Bellinghan GJ, Bernard GR, Chiche JD, Coopersmith C, De Backer DP, French CJ, Fujishima S, Gerlach H, Hidalgo JL, Hollenberg SM, Jones AE, Karnad DR, Kleinpell RM, Koh Y, Lisboa TC, Machado FR, Marini JJ, Marshall JC, Mazuski JE, McIntyre LA, McLean AS, Mehta S, Moreno RP, Myburgh J, Navalesi P, Nishida O, Osborn TM, Perner A, Plunkett CM, Ranieri M, Schorr CA, Seckel MA, Seymour CW, Shieh L, Shukri KA, Simpson SQ, Singer M, Thompson BT, Townsend SR, Van der Poll T, Vincent JL, Wiersinga WJ, Zimmerman JL, Dellinger RP. Surviving Sepsis Campaign: International Guidelines for Management of Sepsis and Septic Shock: 2016. Crit Care Med. 2017;45:486-552. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 1784] [Cited by in RCA: 1986] [Article Influence: 248.3] [Reference Citation Analysis (1)] |
| 34. | Marik PE. Steroids for sepsis: yes, no or maybe. J Thorac Dis. 2018;10:S1070-S1073. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 10] [Cited by in RCA: 13] [Article Influence: 1.9] [Reference Citation Analysis (0)] |
| 35. | Wang D, Hu B, Hu C, Zhu F, Liu X, Zhang J, Wang B, Xiang H, Cheng Z, Xiong Y, Zhao Y, Li Y, Wang X, Peng Z. Clinical Characteristics of 138 Hospitalized Patients With 2019 Novel Coronavirus-Infected Pneumonia in Wuhan, China. JAMA. 2020;323:1061-1069. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 14113] [Cited by in RCA: 14764] [Article Influence: 2952.8] [Reference Citation Analysis (0)] |
| 36. | Zhou W, Liu Y, Tian D, Wang C, Wang S, Cheng J, Hu M, Fang M, Gao Y. Potential benefits of precise corticosteroids therapy for severe 2019-nCoV pneumonia. Signal Transduct Target Ther. 2020;5:18. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 153] [Cited by in RCA: 168] [Article Influence: 33.6] [Reference Citation Analysis (0)] |
| 37. | Xie LX, Liu YN, Hao FY, Dong J, Cao L, Xu HM, Tian Q, Fan BX, Li YP, Ma L, Su YF. [Prognostic analysis of lung function and chest X-ray changes of 258 patients with severe acute respiratory syndrome in rehabilitation after discharge]. Zhonghua Jie He He Hu Xi Za Zhi. 2004;27:147-150. [PubMed] |
| 38. | Meng QH, Dong PL, Guo YB, Zhang K, Liang LC, Hou W, Dong JL. [Use of glucocorticoid in treatment of severe acute respiratory syndrome cases]. Zhonghua Yu Fang Yi Xue Za Zhi. 2003;37:233-235. [PubMed] |
| 39. | Zhao ZW, Zhang FC, Xu M, Huang K, Zhong WN, Cai WP, Yin CB, Huang SD, Deng ZT, Wei M. [Clinical analysis of 190 cases of outbreak with atypical pneumonia in Guangzhou in spring, 2003]. Zhonghua Yi Xue Za Zhi. 2003;83:713-718. [PubMed] |