Published online Jan 6, 2020. doi: 10.12998/wjcc.v8.i1.217
Peer-review started: September 30, 2019
First decision: October23, 2019
Revised: November 16, 2019
Accepted: November 30, 2019
Article in press: November 30, 2019
Published online: January 6, 2020
Processing time: 99 Days and 0.5 Hours
We analyzed the main features of an infant diagnosed with temporary neonatal cyanosis in order to strengthen our understanding of the disease.
We report a patient diagnosed with temporary neonatal cyanosis. The main clinical characteristics, gene mutation and treatment are discussed and a review of related literature was conducted. The neonate aged 1 d and 5 h was admitted to hospital due to cyanosis after birth. The main clinical manifestation was cyanosis, which was not improved by auxiliary ventilation and the patient showed no obvious shortness of breath or methemoglobinemia. Gene mutation analysis showed a heterozygous c.190C>T mutation in the HBG2 gene associated with transient neonatal cyanosis, which was derived from his mother. Symptomatic supportive treatment was given for 2 mo. The neonate was discharged and gradually improved with oral administration of vitamin C and vitamin B2 for 2 wk.
There is no special treatment for temporary neonatal cyanosis caused by heterozygous mutation of the HBG2 gene.
Core tip: We report a case diagnosed with temporary neonatal cyanosis. The main characteristics of the clinical manifestations and gene mutation, and treatments were assessed and a review of the related literature was performed, which may help to improve the understanding of the disease.
- Citation: Yuan J, Zhu XP. Clinical characteristics on manifestation and gene mutation of a transient neonatal cyanosis: A case report. World J Clin Cases 2020; 8(1): 217-221
- URL: https://www.wjgnet.com/2307-8960/full/v8/i1/217.htm
- DOI: https://dx.doi.org/10.12998/wjcc.v8.i1.217
Temporary neonatal cyanosis[1] is characterized by varying degrees of bruising on the body, around the mouth, or at the extremities, with or without other symptoms or signs. Fetal hemoglobin (Hb) chains are defective, and the affinity for oxygen is weakened, resulting in neonatal cyanosis. In some patients, anemia occurs due to abnormal red blood cells (RBCs). Cyanosis is usually self-healing after 5 to 6 mo or earlier when the adult β-globin chain replaces the fetal γ-globin chain. There are few similar reports both at home and abroad, and scholars such as Celeste Bento reported that the disease is gene-related[2]. This article reports the clinical features, diagnosis and treatment of a child with temporary neonatal cyanosis associated with HBG2 gene mutation. The child was diagnosed in the neonatal intensive care unit of the Children’s Hospital of Soochow University, and the purpose of this article is to improve our understanding of this disease.
Discovering bruises for 12 h.
Proband Male, born 29 h at the time of the visit. He was treated for “discovering bruises for 12 h”.
G1P1, gestational age 38+4 wk, born by cesarean section at Taicang First People’s Hospital on October 18, 2017, birth weight 3140 g, Apgar score at 1 min 10 points, Apgar score at 5 min 10 points, amniotic fluid clear; however, the quantity is unknown. The placenta status is unknown, and a history of rescue suffocation is denied.
Parents are not close relatives, and his mother had similar symptoms when she was a baby.
His body temperature was 36.5 °C, pulse 147 beats/min, respiratory rate 56 breaths/min, the oxygen pulse of hood oxygen was 81%, body weight 3.02 kg, length 48 cm, head circumference 34 cm, and chest circumference 32 cm. The neonate had full-term appearance, color cyan, scalp edema, anterior sputum soft, size approximately 1.5 cm × 1.5 cm tension was not high, perioral cyanosis, no spitting, soft neck, no polypnea, no obvious inspiratory three concave sign, the lungs showed thick breath sounds, no rales were heard in the lungs, heart rhythm showed no obvious murmur in the precordium, abdomen soft, liver and spleen under the ribs, cold extremities, nerve reflex could be induced.
White blood cell count 7.75 × 109/L, RBC 4.00 × 1012/L, Hb 136 g/L, platelet distribution width 9.40%, blood gas analysis showed Ca2+ 1.13 mmol/L, Cl- 111 mmol/L, BE- 3.5 mmol/L, lactic acid 6.0 mmol/L, total bilirubin 182 mmol/L, whole blood oxygen content 6.5 mmol/L, oxygen partial pressure 140 mmHg, blood pH 7.438, carbon dioxide partial pressure 21.6 mmHg, methemoglobin (MetHb) fluctuation between 2.6% and 5.7%. Glucose-6-phosphate dehydrogenase activity was 1.45, which was within the normal range. Complete hemolysis examination showed Hb F > 40%, Hb A2 0%, Hb electrophoresis showed no abnormal zone, and a small abnormal S window peak was observed by HPLC.
Echocardiography demonstrated an atrial septal defect (2.7 mm) and patent ductus arteriosus. Chest and abdominal imaging showed that the texture of both lungs was darkened and blurred. Hepatobiliary, pancreatic, spleen, kidney and cranial ultrasound were normal, and scrotum ultrasound showed a right testicular hydrocele.
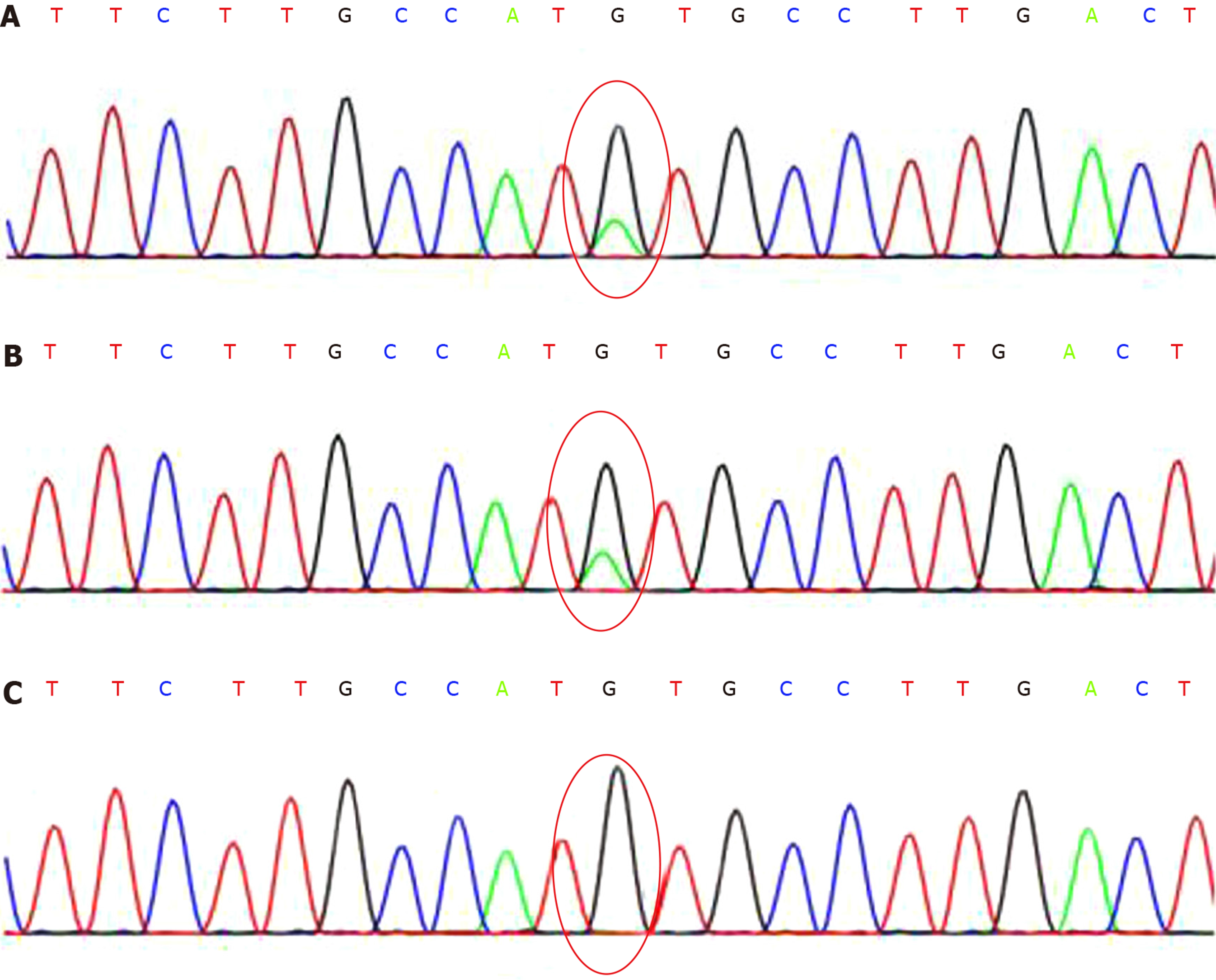
The patient (SH171214132), chr11:5275647, showed a heterozygous mutation of c.190C>T; the father (SH171214133), chr11:5275647 showed no mutation; the mother (SH171214134), chr11:5275647 showed a heterozygous mutation of c.190C>T (Figure 1).
Neonatal methemoglobinemia? Neonatal pneumonia; congenital foramen ovale; congenital atrial septal defect; neonatal hyperbilirubinemia; right testicular hydrocele.
When the patient was admitted to hospital, the oxygen pulse of hood oxygen was maintained at 75%-78%, perioral and facial region cyanosis was observed, lowest blood pressure was 54/37 mmHg, saline was expanded, assisted ventilation was administered, and the patient was actively treated. The percutaneous oxygen pulse still fluctuated around 72%-85%, and the blood oxygen saturation following arterial blood gas analysis was 96.0%-100%, and the oxygen partial pressure was maintained in the normal range. After hospitalization, the MetHb content was high, fluctuating between 2.6% and 5.7%. After clinical diagnosis, vitamin C and vitamin B2 treatment was given, but there was no significant improvement in percutaneous oxygen pulse saturation, and Hb-related gene detection was performed. The patient was diagnosed with temporary neonatal cyanosis. The clinical phenotype showed anemia, cyanosis, and methemoglobinemia. The oxygen pulse during assisted ventilation did not increase significantly. The child did not have breathing difficulties, no obvious inspiratory three concave sign, breathing was stable, and arterial blood gas analysis showed that oxygen partial pressure and oxygen saturation were maintained in the normal range. The existence of genetic problems was considered. Gene examination was carried out and a transfusion to correct anemia was administered; however, percutaneous oxygen saturation showed no obvious increase. After 3 d, assisted ventilation was discontinued and replaced with hood oxygen. The percutaneous oxygen pulse was maintained at approximately 75%, and his respiration rate was 47 breaths/min. When hood oxygen was stopped, percutaneous oxygen saturation was maintained between 70% and 80%, with no obvious dyspnea, no dysphoria or other clinical manifestations. Seventeen days after admission, he was discharged on vitamin C and vitamin B2 therapy. Hyperbilirubinemia occurred during the disease course, and phototherapy was administered due to jaundice, and the ganglioside nutrient nervous system was used for rehabilitation. Vitamin C and vitamin B2 were discontinued 2 wk after discharge. Perioral and facial cyanosis gradually subsided. After 189 d of follow-up, the child’s growth and development were normal, and his body weight was 9 kg. No recurrence was observed.
Gene mutation analysis identified a heterozygous mutation in the exon region of the HBG2 gene: c.190C>T (nucleotide number 190 in the coding region was changed from cytosine to thymine), resulting in amino acid change p.H64Y (No. 64) in the patient. The amino acid was changed from histidine to tyrosine, which is a missense mutation[3]. This mutation does not belong to the polymorphic site, and the frequency of occurrence in the population is low. The Human Gene Mutation Database Professional Edition has reported an association with the Hb variant[4]. After verification by the family, the father of the subject showed no variation at this site, and the mother of the subject showed a heterozygous variation at the same site[5]. This site is a suspected mutation. If the mother had similar symptoms, it should theoretically cause illness; if the mother had no similar symptoms, the possibility of the site causing mutation is very small. In this case, no specific disease-related copy number variation was found at the exon level.
Neonatal cyanosis is a common sign in the early neonatal period, and it is one of the most serious clinical manifestations[6]. Whether it is a gas exchange disorder caused by cardiopulmonary disease or a localized reduction in Hb around the tissue capillaries which can cause cyanosis is still unknown[7]. Skin mucosa in a child after birth which shows persistent cyanosis and percutaneous oxygen saturation less than 90%, with the lowest at 60%, is classified as pathological cyanosis. The current reasons for cyanosis are complicated. After hospitalization, the patient received oxygen, continuous positive airway pressure assisted ventilation, and mechanical ventilation. Repeated blood gas analysis showed that arterial oxygen saturation and oxygen partial pressure were similar to non-oxygenation, indicating that assisted ventilation did not improve hypoxia, and heart color Doppler ultrasound suggested congenital heart disease; however, there was no obvious pulmonary hypertension, so central cyanosis was excluded. The child's body temperature was normal, the temperature of the extremities was normal, the Hb content was not high, clinical manifestations of shock or heart failure were not obvious, and there was no obvious local circulation failure; thus, peripheral cyanosis was excluded. Therefore, considering other factors which may cause bruising, repeated blood gas analysis was performed after admission and showed that the MetHb concentration was significantly higher than the normal value, the highest being 8.2%. Blood tests showed that the child’s blood color was brownish red, indicating that the heart status, B-ultrasound, chest X-ray and other related examinations, in addition to congenital cyanosis, low oxygen pulse and other factors, led to the diagnosis of neonatal methemoglobinemia. MetHb is a genetic factor and a toxic compound produced by RBCs. Hb is composed of globin and heme, which combine with oxygen to form oxygenated Hb. MetHb is a Hb derivative in which the iron ion of the heme group in deoxygenated or oxygenated Hb is oxidized from ferrous to ferric iron. The MetHb reduction process mainly depends on NADH-dependent Cytochrome 65 reductase, and the absence of this enzyme leads to abnormal accumulation of MetHb. When MetHb is overproduced, the ratio of MetHb and Hb is unbalanced and methemoglobinemia is formed[8]. Following hospitalization for active oxygen therapy, the condition of the child improved and the diagnosis was clinically supported. Methylene blue is recommended for methemoglobinemia, but the child was also thought to have an Hb disease and possible genetic problems. Blood gas analysis after treatment showed that the content of MetHb was still high. So, the parents of the child were asked about the history of pregnancy. The mother of the child likes to eat spicy food, and was also born with a similar condition to the child; thus, it was considered that the child's cyanosis was closely related to MetHb content.
In this case of suspected methemoglobinemia, a gene mutation was identified and the patient was diagnosed with temporary neonatal cyanosis, and clinical follow-up showed that the condition gradually improved. The recurrence of unexplained cyanosis and the repeated rise in MetHb can be diagnosed by genetic testing. The HBG2 gene is associated with transient neonatal cyanosis[9], and is reported to occur via autosomal dominant inheritance. Theoretically, chromogenic pathogenic mutations can cause the disease[10], and the clinical manifestations are similar to this case. In this patient, genetic testing detected a heterozygous mutation in the transient neonatal cyanosis-related gene HBG2, which was derived from his mother and is X-linked. The genetic characteristic, in this case, temporary neonatal cyanosis, was treated with vitamin C and vitamin B2, and after active oxygen therapy and anti-oxidation therapy, perioral and facial cyanosis gradually subsided. Usually cyanosis can take 5 to 6 mo to resolve with possible earlier self-healing, as the adult β-globin chain replaces the fetal γ-globin chain. In this case, although early treatment was administered, early genetic testing was not carried out. Temporary neonatal cyanosis should be diagnosed early with early treatment to avoid other diseases and improve the prognosis.
Transient neonatal cyanosis caused by heterozygous mutation of the HBG2 gene has no specific treatment and can resolve itself. With the development of genetic diagnosis, early diagnosis of neonatal diseases can be achieved relatively easily. The formation of a neonatal disease is a complex pathological process, and its prognosis will also be determined by the progress of pathological changes. The general condition of the patient should be comprehensively evaluated and early conservative treatment is recommended.
Manuscript source: Unsolicited manuscript
Specialty type: Medicine, Research and Experimental
Country of origin: China
Peer-review report classification
Grade A (Excellent): 0
Grade B (Very good): 0
Grade C (Good): C, C
Grade D (Fair): 0
Grade E (Poor): 0
P-Reviewer:Akbulut S, Skok PS-Editor:Dou Y L-Editor: Webster JR E-Editor: Liu JH
| 1. | Dainer E, Shell R, Miller R, Atkin JF, Pastore M, Kutlar A, Zhuang L, Holley L, Davis DH, Kutlar F. Neonatal cyanosis due to a novel fetal hemoglobin: Hb F-Circleville [Ggamma63(E7)His-->Leu, CAT>CTT]. Hemoglobin. 2008;32:596-600. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 15] [Cited by in RCA: 14] [Article Influence: 0.9] [Reference Citation Analysis (0)] |
| 2. | Bento C, Magalhães Maia T, Carvalhais I, Moita F, Abreu G, Relvas L, Pereira A, Farela Neves J, Ribeiro ML. Transient neonatal cyanosis associated with a new Hb F variant: Hb F viseu. J PediatrHematol Oncol. 2013;35:e77-e80. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 10] [Cited by in RCA: 9] [Article Influence: 0.8] [Reference Citation Analysis (0)] |
| 3. | Kedar PS, Gupta V, Warang P, Chiddarwar A, Madkaikar M. Novel mutation (R192C) in CYB5R3 gene causing NADH-cytochrome b5 reductase deficiency in eight Indian patients associated with autosomal recessive congenital methemoglobinemia type-I. Hematology. 2018;23:567-573. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 6] [Cited by in RCA: 5] [Article Influence: 0.7] [Reference Citation Analysis (0)] |
| 4. | Adachi K, Asakura T, Schwartz E. Aggregation of hemoglobin S and hemoglobin CHarlem with nonsickle hemoglobin in concentrated phosphate buffer. Blood. 1980;55:494-500. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 1] [Cited by in RCA: 1] [Article Influence: 0.0] [Reference Citation Analysis (0)] |
| 5. | Giordano SJ, Kaftory A, Steggles AW. A splicing mutation in the cytochrome b5 gene from a patient with congenital methemoglobinemia and pseudohermaphrodism. Hum Genet. 1994;93:568-570. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 49] [Cited by in RCA: 46] [Article Influence: 1.5] [Reference Citation Analysis (0)] |
| 6. | Qureshi AU, Latiff HA, Sivalingam S. Persistent valve of systemic venous sinus: a cause of neonatal cyanosis. Cardiol Young. 2014;24:756-759. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 5] [Cited by in RCA: 5] [Article Influence: 0.5] [Reference Citation Analysis (0)] |
| 7. | Ko HS, Chen MR, Lin YC. A huge Chiari network presenting with persistent cyanosis in a neonate. PediatrCardiol. 2011;32:239-240. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 4] [Cited by in RCA: 3] [Article Influence: 0.2] [Reference Citation Analysis (0)] |
| 8. | Wright RO, Lewander WJ, Woolf AD. Methemoglobinemia: etiology, pharmacology, and clinical management. Ann Emerg Med. 1999;34:646-656. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 485] [Cited by in RCA: 498] [Article Influence: 19.2] [Reference Citation Analysis (0)] |
| 9. | Koga M, Inada S, Yoshino K, Miyazaki A. IFCC-HbA1c May Be More Useful than NGSP-HbA1c in the Comparison between HbA1c Values in Variant Hemoglobin with a Mutation on α Chain and β Chain. Ann Clin Lab Sci. 2019;49:650-655. [PubMed] |
| 10. | Yang L, Liu X, Li Z, Zhang P, Wu B, Wang H, Hu L, Cheng G, Wang L, Zhou W. Genetic aetiology of early infant deaths in a neonatal intensive care unit. J Med Genet. 2019;. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 18] [Cited by in RCA: 18] [Article Influence: 3.0] [Reference Citation Analysis (0)] |