Published online Apr 16, 2015. doi: 10.12998/wjcc.v3.i4.389
Peer-review started: October 21, 2014
First decision: December 12, 2014
Revised: January 4, 2015
Accepted: February 4, 2015
Article in press: February 9, 2015
Published online: April 16, 2015
Processing time: 174 Days and 6 Hours
Malignant peripheral nerve sheath tumor (MPNST) with rhabdomyoblastic differentiation is called as malignant triton tumor (MTT). It is highly aggressive soft tissue tumor with higher local recurrence rate. MTT has poor prognosis than MPNST. MTT seems to be more aggressive in patients with neurofibromatosis (NF-1). We herein, reporting an interesting case of 55 years male with multiple neurofibromas all over the body since 30 years and multiple café-au-lait spots, diagnosed as NF-1. Since 6 years, he had an enlarged mass in left thigh. Wide excision of mass was done. On histopathological examination revealed the diagnosis of MTT and diagnosis of which was confirmed on immunohistochemistry.
Core tip: Meticulous histopathological examination along with immunohistochemistry is the mainstay to arrive at such rare histological diagnosis. The surgical pathologist should keep in mind such rare entity while dealing with such kind of patients.
- Citation: Shete S, Bolde S, Pandit G, Matkari P, Ingle SB. Unusual histological variant of malignant peripheral nerve sheath tumor with rhabdomyoblastic differentiation. World J Clin Cases 2015; 3(4): 389-392
- URL: https://www.wjgnet.com/2307-8960/full/v3/i4/389.htm
- DOI: https://dx.doi.org/10.12998/wjcc.v3.i4.389
Malignant peripheral nerve sheath tumor (MPNST) is an unusual type of soft tissue sarcomas. It accounts for about 5%-10% of all soft tissue sarcomas [1]. Amongst them malignant triton tumor (MTT) constitutes about 5% of all MPNSTs[2]. MTT is a subtype of MPNST that has real diagnostic challenge for surgical pathologists with regard to its cell of origin and its relatively aggressive course. It is comprised of malignant Schwann cells coexisting with characteristic malignant rhabdomyoblasts[3]. It occurs in two forms, one is sporadic form and the other is neurofibromatosis type 1 (NF-1) associated form. Majority of the reported cases are of later variety. Immunohistochemically the rhabdomyoblastic elements are typically positive for skeletal muscle markers such as desmin, myoglobin or muscle actin. MTT has a poorer prognosis than MPNST[3]. This poor outcome is mainly attributed to the high frequency of grade III histology in this sarcoma[4].
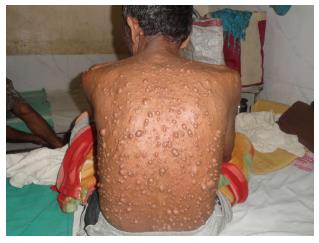
A 55-year-old male patient presented with multiple swellings all over the body ranging from 1-3 cm since 30 years (Figure 1). These nodules were histopathologically reported as neurofibromas. A subcutaneous nodule in left thigh was enlarged to attain a present size of 8 cm × 4 cm × 3 cm. He had café-au-lait spots on the trunk ranging from 0.5-1.8 mm. On family history, patient’s mother, sister and brother had similar complaints. So he was diagnosed as a case of NF-1. Ultrasonography was suggestive of neoplastic lesion mostly soft tissue sarcoma.
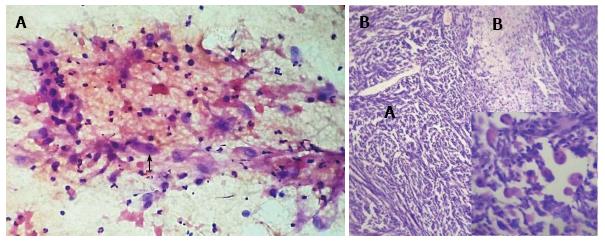
Fine needle aspiration cytology was done and reported as malignant soft tissue tumor with possibilities of malignant fibrous histiocytoma and rhabdomyosarcoma (Figure 2A). Complete resection of the mass was done and the specimen was sent for histopathological evaluation.
On, gross examination showed a mass of 8 cm × 4 cm × 3 cm in size, covered with skin. Cut surface was soft, pale grey in colour with areas of necrosis.
Histopathological examination showed dense areas of malignant spindle shaped cells with oval nuclei with prominent mitoses, marked nuclear pleomorphism along with foci of necrosis. Many nuclei were bizarre and hyperchromatic. Alternating with hypercellular Antoni A areas, there were hypocellular myxoid areascalled as- Antoni B areas. Thick walled congested blood vessels were found. Interspersed within it are seen many Scattered round cells with abundant eosinophilic cytoplasm with atypical nuclei, which were recognized as rhabdomyoblasts (Figure 2B).
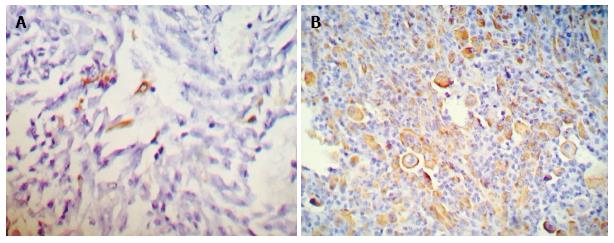
The surgical cut margins margins were free of tumor invasion. On Immunohistochemical evaluation, spindle cells showed focal S-100 positivity (Figure 3A) and cells with deeply eosinophilic cytoplasm (rhabdomyoblasts) showed positivity for Desmin (Figure 3B).
After histopathological confirmation patient was under treatment with radiotherapy and doing well without any recurrence on follow up since last 6 mo till date.
Peripheral nerve malignant lesions are displaying differentiation towards Schwann cells, perineural cells, fibroblasts are labelled as MPNST. This term now replacing the earlier terminologies malignant schwannoma, neurofibrosarcoma and neurogenic sarcoma[1]. The diagnostic criteria for NF-1, is presence of two or more of the following signs[1]: (1) Six or more cafe-au-lait macules; (2) Two or more neurofibromas of any type or one plexiform neurofibroma; (3) Freckling in the axillary or inguinal region; (4) Optic Glioma; (5) Two or more Lisch nodules; (6) Osseous Lesion; and (7) First degree relative (parent, sibling, offspring) with NF-1.
MPNST constitutes 5%-10% of all soft tissue sarcomas, about one fourth to one half occur in the setting of neurofibromatosis[1]. Patients with NF-1 have propensity to transform in to sarcoma after a prolonged latent period (10-20 years)[5]. Our patient was diagnosed as having NF-1 and after long latency period of 30 years, he developed MPSNT with rhabdomyoblastic differentiation. MPNST can also arise spontaneously without association of NF-1[4].
MTT is a rare tumor arising from peripheral nerves. It is an autosomal dominant disorder. It has a strong association with neurofibromatosis (type 1). The common sites of occurrence are head, neck, extremities and trunk[4]. The symptoms are mainly attributed to mass effect giving rise to neurological signs and symptoms[4].
In 1973 Woodnelf et al[6] proposed the classification of MTT by establishing three criteria for diagnosis: (1) Tumor with peripheral nerve involvement in a patient with NF-1; (2) Majority of the cells in the tumor are Schwann cells; and (3) Presence of Rhabdomyoblasts[6].
Our patient had NF-1 and histology showed all the above mentioned criteria. The pathognomic feature of this tumor is the presence of rhabdomyoblasts. The number of rhabdomyoblasts varies from area to area in the same tumor. They are having abundant eosinophilic cytoplasm. Desmin is demonstrated in the rhabdomyoblasts.
The histogenesis of this unusual tumor is discussed by Masson. He postulated that both cell lines have similar origin, i.e., from less well differentiated neural crest cells[1,7] . The strong relation between neural tissues and rhabdomyoblastic differentiation has been reported as the development of skeletal muscle differentiation within other neural tumors such as Ocular Medulloblastoma[8], Ganglioneuroblastoma[9,10]. The five years survival rate for MTT is only 11% in contrast to 39% for MPNST[11]. MTT is significantly worse than the usual MPNST. The aggressiveness of MTT is attributed to high grade (grade III) nuclear features with high proliferative capacity[4]. Radical excision followed by high dose radiotherapy is the conventional treatment for this unusual tumor[3].
MTT is an uncommon sarcoma which is having high propensity of local recurrence and distant metastases. Histopathologically, the diagnosis of MPNST with mesenchymal differentiation is difficult. So meticulous histopathological examination and immunohistochemical demonstration of neural markers and skeletal muscle markers help to hit the correct diagnosis. Early diagnosis, complete resection of the tumor followed by radiotherapy can help to increase survival of the patient.
A 55-year-old male patient presented with multiple swellings all over the body ranging from 1-3 cm since 30 years.
Multiple neurofibromas.
Neurofibromas, fibrosarcoma, malignant peripheral nerve sheath tumor (MPNST).
Fine needle aspiration cytology, histopathology with Immunohistochemistry showing Desmin positivity in rhabdomyoblasts.
Ultrasonography was suggestive of neoplastic lesion soft tissue sarcoma.
An unusual histological variant of MPNST with rhabdomyoblastic differentiation.
Careful histological examination along with clinical work up is important to arrive at such unusual diagnosis.
Good overview of the diagnostic challenges in the correct interpretation of this rare tumor.
P- Reviewer: Chaudhry IA S- Editor: Song XX L- Editor: A E- Editor: Lu YJ
| 1. | Weiss SW, Goldbum JR. Malignant tumors of nerves. St Louis: CV Mosby Elsevier 2008; 903-944. |
| 2. | Brooks JSJ. Disorders of soft tissue. Diagnostic Surgical Pathology. 3rd ed. Philadelphia: Lipincott Williams and Wilkins 1999; 131-221. |
| 3. | Mae K, Kato Y, Usui K, Abe N, Tsuboi R. A case of malignant peripheral nerve sheath tumor with rhabdomyoblastic differentiation: malignant triton tumor. Case Rep Dermatol. 2013;5:373-378. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 7] [Cited by in RCA: 8] [Article Influence: 0.7] [Reference Citation Analysis (0)] |
| 4. | Brooks JS, Freeman M, Enterline HT. Malignant “Triton” tumors. Natural history and immunohistochemistry of nine new cases with literature review. Cancer. 1985;55:2543-2549. [PubMed] |
| 5. | Guccion JG, Enzinger FM. Malignant Schwannoma associated with von Recklinghausen’s neurofibromatosis. Virchows Arch A Pathol Anat Histol. 1979;383:43-57. [PubMed] |
| 6. | Woodneff JM, Chernik NL, Smith MC. Peripheral nerve tumors with rhabdomyosarcomatous differentiation (Malignant Triton Tumor). Cancer. 1973;32:626-639. |
| 7. | Masson P. Recklinghausen’s Neurofibromatosis, Sensory Neuromas and Motor Neuromas. Libman Anniversary. New York: International press 1933; 793-802. |
| 8. | Zimmerman LE, Font RL, Andersen SR. Rhabdomyosarcomatous differentiation in malignant intraocular medulloepitheliomas. Cancer. 1972;30:817-835. [PubMed] |
| 9. | Taxy JB, Battifora H, Trujillo Y, Dorfman HD. Electron microscopy in the diagnosis of malignant schwannoma. Cancer. 1981;48:1381-1391. [PubMed] |
| 10. | Naka A, Matsumoto S, Shirai T. Ganglioneuroblastoma associated with malignant mesenchymoma. Cancer. 1976;36:1050-1056. |
| 11. | Stasik CJ, Tawfik O. Malignant peripheral nerve sheath tumor with rhabdomyosarcomatous differentiation (malignant triton tumor). Arch Pathol Lab Med. 2006;130:1878-1881. [PubMed] |