Published online Jul 6, 2025. doi: 10.12998/wjcc.v13.i19.99311
Revised: January 27, 2025
Accepted: February 26, 2025
Published online: July 6, 2025
Processing time: 242 Days and 21.4 Hours
Esophageal squamous papilloma (ESP) is a rare benign tumor of the esophagus, typically characterized by an exophytic and warty appearance, with a diameter of less than 5 mm. Endoscopic resection is considered the most effective treatment for these tumors.
In this paper, we discussed the pathogenesis, clinical characteristics, and therapeutic options of ESP based on our experience with a case presenting a depressed appearance and treated with interferon-alpha 2a (IFNα2a) injection.
This paper reports the first successful clinical case of using the human IFNα2a for the treatment of ESP with a depressed endoscopic appearance. However, the efficacy of interferon treatment requires to validation in a large number of subse
Core Tip: Esophageal squamous papilloma (ESP) is a rare benign epithelial tumor of the esophagus, typically presenting with an exophytic and warty appearance, often measuring less than 5 mm in diameter. Endoscopic resection is regarded as the most effective treatment for these tumors. However, the complete resection rate remains low. In this paper, we reported a case of ESP located in the lower esophagus that exhibited an unusual depressed endoscopic appearance with a diameter approaching 2 cm, which showed significant therapeutic outcomes by recombinant human interferon-alpha 2a. Nevertheless, further validation of the efficacy of interferon treatment is necessary through larger-scale studies involving subsequent cases.
- Citation: Jiang J, Shi HT, Wu J, Sha SM, Cai SX, Liu X. Successful treatment of depressed esophageal squamous papilloma with interferon- alpha 2a: A case report. World J Clin Cases 2025; 13(19): 99311
- URL: https://www.wjgnet.com/2307-8960/full/v13/i19/99311.htm
- DOI: https://dx.doi.org/10.12998/wjcc.v13.i19.99311
Esophageal squamous papilloma (ESP) is a rare benign tumor of the esophagus. The incidence of ESP reported ranges from 0.01% to 0.45%[1]. ESP can occur throughout the esophagus, with the lower and middle esophagus being the most commonly affected regions. Endoscopically, ESP usually appears as white or grayish-white, wart-like lesion with a diameter of less than 5 mm. It is often solitary but can occasionally present as multiple lesions, and in rare cases, may involve the entire esophagus[2,3]. ESP is believed to result from a chronic inflammatory response triggered by chemical or mechanical irritants, including gastroesophageal reflux disease, smoking, alcohol consumption, caustic injury, food impaction, or human papillomavirus (HPV) infection[4]. Currently, endoscopic treatment is the primary clinical app
Intermittent retrosternal discomfort for more than one month.
A 40-year-old man presented to our hospital on December 11, 2015, with intermittent retrosternal discomfort persisting for more than one month. His symptoms were accompanied by acid reflux and heartburn but without significant nausea, vomiting, palpitations, shortness of breath, chest tightness, chest pain, fever, or night sweats. The patient had been taking oral omeprazole 20 mg once daily; however, his symptoms did not significantly improve, and he subsequently developed adverse swallowing/ dysphagia and retrosternal pain.
The patient reported no history of previous illness, medication use, or exposure to toxic substances.
The patient denied any family history of malignant tumors.
Upon physical examination, his vital signs were stable, and no significant abnormalities were observed in the cardiac, pulmonary, or abdominal examinations.
Routine blood and urine laboratory test results were within normal limits. Biochemical tests, tumor markers, viral and mycobacterium tuberculosis DNA and antibody screening, and T-SPOT.TB results were all normal. Autoantibody screening revealed a positive Jo-1 antibody, while other markers were negative. The erythrocyte sedimentation rate was elevated at 58 mm/h, and C-reactive protein level was 2.09 mg/dL. Stool occult blood was positive. The electrocardiogram showed no significant abnormalities.
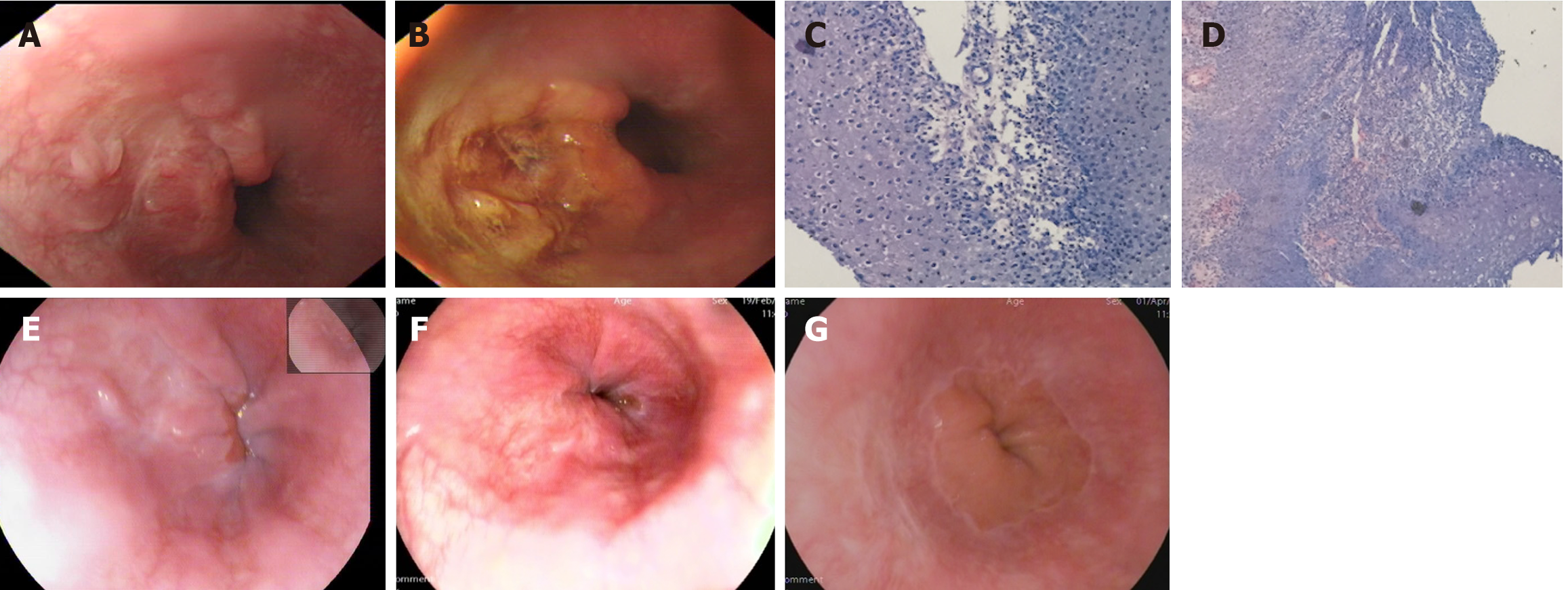
Chest and abdominal computed tomography revealed striae in the right middle lobe lung lobe, gallbladder stones, and calcified foci on the right liver lobe. Upper gastrointestinal endoscopy showed an irregular, depressed lesion in the lower esophagus near the cardia (Figure 1A), measuring approximately 2.0 cm in diameter, with 1.2% Lugol's iodine staining and no obvious unstained areas (Figure 1B). Several biopsies were obtained, and pathological findings revealed esophageal squamous epithelial papilloma proliferation with necrosis and severe acute and chronic inflammation, with numerous koilocytes (Figure 1C and D).
The depressed lesion in the lower esophagus was diagnosed as an HPV infection-associated esophageal papilloma.
The patient received an intramuscular injection of recombinant human IFNα2a at a dose of 300 IU once every other day for 1 month.
Gastroscopy performed one month after treatment showed a significant reduction in the lesion (Figure 1E). After two months, scar formation was observed at the lesion site (Figure 1F). No recurrence was noted during a follow-up gastroscopy conducted 4 months later (Figure 1G).
ESP is a rare benign epithelial tumor of the esophagus. Most ESP cases lack specific clinical manifestations and are often discovered incidentally during upper gastrointestinal endoscopy performed for symptoms such as epigastric discomfort and acid reflux. Endoscopically, ESP typically presents as a single proliferative papillary bulge of approximately 2-8 mm in diameter. Studies have shown that exophytic growth, a warty appearance, and crossed vessels on the lesion surface observed under narrow-band imaging are three important features of ESP, with a diagnostic accuracy up to 88.2%[5] when all three features were present. However, there have also been reports of ESP cases with diameters exceeding 3 cm or with a flat appearance[6-9].
In this paper, we report a case of ESP in the lower esophagus near the cardia, consistent with the common site of ESP occurrence, but presenting with an unusual flat, depressed, ulcer-like appearance. The lesion measured nearly 2 cm in diameter and was difficult to distinguish from early esophageal cancer under conventional white-light endoscopy. No unstained areas were observed following local spraying with 1.2% Lugol's iodine, and subsequent pathological analysis confirmed the diagnosis of ESP. To date, no other reports have described ESP with a depressed growth pattern.
The etiology of ESP is not fully understood, and two hypotheses have been proposed[10]. The first suggests that ESP results from chronic irritation of the lower esophageal mucosa due to gastroesophageal reflux, esophagitis, esophageal hiatal hernia, or other physicochemical or mechanical injury. The second hypothesis suggests a strong association between ESP and HPV infection. Syrjänen et al[11] conducted a meta-analysis of 427 ESP cases from various studies, reporting that 132 cases (30%) were HPV DNA-positive, with diverse HPV serotypes identified. A study from Turkey detected HPV DNA in 7 of 38 ESP patients, including 4 cases of low-risk HPV (types 6 and 81) and 3 cases of high-risk HPV (types 16, 18, and 31)[12]. Another smaller study conducted in Mexico found that 10 out of the 14 HPV-positive ESP patients were infected with low-risk HPV (types 6 and 11) and 2 patients with high-risk HPV (type 16)[13]. Some studies have shown that the presence of koilocytes in HPV-positive tissues is significantly higher than in HPV-negative tissues, suggesting that koilocytes may have diagnostic value for HPV infection[14]. In the ESP case reported in this paper, histological examination of the lesion revealed thickened squamous epithelium with visible papillary structures and central visible fibrovascularity, without cellular anisotropy, consistent with ESP pathology. Although histological HPV DNA testing was performed, the presence of numerous koilocytes suggests a potential correlation with HPV infection.
It is generally accepted that pharmacological treatment by itself has limited efficacy, and endoscopic resection is the preferred treatment due to its effectiveness and lower recurrence rates. Depending on the lesion’s size, morphology, and location, different endoscopic treatment measures can be selected. Lesions measuring 1-5 mm are typically selected for cold forceps, thermal biopsy, or argon plasma coagulation. Lesions measuring 0.5-1.0 cm can be treated with cold forceps polypectomy or endoscopic mucosal resection, which allows intact lesion retrieval for detailed pathologic evaluation. Larger lesions, particularly those exceeding 2 cm in diameter pose challenges for complete endoscopic resection, with lower success rates and a higher likelihood of recurrence. Better outcomes have been reported with alternative methods such as radiofrequency ablation or cryotherapy[15,16].
For ESP associated with HPV infection that recurs after endoscopic treatment or cannot be treated endoscopically, pharmacological treatment, particularly interferon therapy, may be considered as an adjuvant treatment option. Interferon therapy has been reported to be effective in patients with papillomas of the respiratory tract or pharynx[17,18], and successful cases of bronchial papillomas treated with recombinant human IFNα2a have also been reported[19]. In this paper, we report an ESP case that exhibited an ulcer-like appearance endoscopically, measured nearly 2 cm in diameter, and was difficult to manage with curative endoscopic resection. Given the histological findings suggestive of HPV infection, the patient was treated with intramuscular injections of recombinant human IFNα2a at a dose of 300 IU once every other day for 1 month. Follow-up gastroscopy after two months showed scar formation at the lesion site, and no signs of recurrence were observed after four months, suggesting the potential effectiveness of interferon therapy.
ESP is a benign lesion that can occur at any location throughout the esophagus. Most cases present endoscopically as small, solitary, exophytic, white lesions with wart-like elevations. To date, there have been no reported cases of ESP exhibiting a depressed growth pattern. The exact etiology of ESP is not fully known, although HPV infection is considered one of the possible triggering factors. Endoscopic resection remains the primary treatment method. Currently, there are no documented cases in the literature of successful treatment of ESP using only interferon injection. In this paper, we report a case of ESP in the lower esophagus with a previously unreported depressed endoscopic appearance, which was treated with recombinant human IFNα2a, echieving significant results. However, the efficacy of interferon treatment requires further validation through larger case studies.
| 1. | Jideh B, Weltman M, Wu Y, Chan CHY. Esophageal squamous papilloma lacks clear clinicopathological associations. World J Clin Cases. 2017;5:134-139. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in CrossRef: 7] [Cited by in RCA: 11] [Article Influence: 1.4] [Reference Citation Analysis (0)] |
| 2. | Lee K, Lee O, Lee S. Gastrointestinal: diffuse esophageal papillomatosis involving the entire esophagus. J Gastroenterol Hepatol. 2014;29:1951. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 2] [Cited by in RCA: 2] [Article Influence: 0.2] [Reference Citation Analysis (0)] |
| 3. | Gençdal G, Degirmencioglu S, Akyıldız M. Diffuse Esophageal Squamous Papillomatosis Covering the Entire Esophagus. Clin Gastroenterol Hepatol. 2018;16:A28. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 11] [Cited by in RCA: 4] [Article Influence: 0.6] [Reference Citation Analysis (0)] |
| 4. | Mahajan R, Kurien RT, Joseph AJ, Dutta AK, Chowdhury SD. Squamous papilloma of esophagus. Indian J Gastroenterol. 2016;35:151. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 1] [Cited by in RCA: 1] [Article Influence: 0.1] [Reference Citation Analysis (0)] |
| 5. | Wong MW, Bair MJ, Shih SC, Chu CH, Wang HY, Wang TE, Chang CW, Chen MJ. Using typical endoscopic features to diagnose esophageal squamous papilloma. World J Gastroenterol. 2016;22:2349-2356. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in CrossRef: 16] [Cited by in RCA: 12] [Article Influence: 1.3] [Reference Citation Analysis (1)] |
| 6. | Inomata S, Aoyagi K, Eguchi K, Sakisaka S. Giant esophageal papilloma. Gastrointest Endosc. 2004;60:430. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 5] [Cited by in RCA: 6] [Article Influence: 0.3] [Reference Citation Analysis (0)] |
| 7. | Tabatabaei SA, Moghadam NA, Ahmadinejad M, Mirmohammadsadeghi A, Masoudpour H, Adibi P. Giant esophageal squamous papilloma: a case report. J Dig Dis. 2009;10:228-230. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 11] [Cited by in RCA: 12] [Article Influence: 0.8] [Reference Citation Analysis (0)] |
| 8. | Shimamoto Y, Shichijo S, Ishihara R. Sudden Appearance of Widespread Esophageal Squamous Papilloma With Reflux Esophagitis. Clin Gastroenterol Hepatol. 2021;19:e27. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 3] [Cited by in RCA: 2] [Article Influence: 0.5] [Reference Citation Analysis (0)] |
| 9. | Iwamuro M, Okamoto Y, Kawano S, Okada H. Esophageal Papilloma Detected by Positron Emission Tomography. Intern Med. 2020;59:1003-1004. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 3] [Cited by in RCA: 1] [Article Influence: 0.2] [Reference Citation Analysis (0)] |
| 10. | Mavilia MG, Wu GY. Esophageal squamous papilloma: A case series and literature review. J Dig Dis. 2018;19:254-256. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 2] [Cited by in RCA: 2] [Article Influence: 0.3] [Reference Citation Analysis (0)] |
| 11. | Syrjänen K, Syrjänen S. Detection of human papillomavirus in esophageal papillomas: systematic review and meta-analysis. APMIS. 2013;121:363-374. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 18] [Cited by in RCA: 20] [Article Influence: 1.7] [Reference Citation Analysis (0)] |
| 12. | Tiftikçi A, Kutsal E, Altıok E, İnce Ü, Çicek B, Saruç M, Türkel N, Ersoy Ö, Yenmiş G, Tözün N. Analyzing esophageal squamous cell papillomas for the presence of human papilloma virus. Turk J Gastroenterol. 2017;28:176-178. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 6] [Cited by in RCA: 6] [Article Influence: 0.8] [Reference Citation Analysis (0)] |
| 13. | Bohn OL, Navarro L, Saldivar J, Sanchez-Sosa S. Identification of human papillomavirus in esophageal squamous papillomas. World J Gastroenterol. 2008;14:7107-7111. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in CrossRef: 25] [Cited by in RCA: 26] [Article Influence: 1.5] [Reference Citation Analysis (0)] |
| 14. | Miyahara GI, Simonato LE, Mattar NJ, Camilo DJ Jr, Biasoli ER. Correlation between koilocytes and human papillomavirus detection by PCR in oral and oropharynx squamous cell carcinoma biopsies. Mem Inst Oswaldo Cruz. 2011;106:166-169. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 11] [Cited by in RCA: 12] [Article Influence: 0.9] [Reference Citation Analysis (0)] |
| 15. | Alomari M, Wadhwa V, Bejarano P, Amar P, Erim T. Successful Treatment of Extensive Esophageal Squamous Papillomatosis With Cryotherapy. ACG Case Rep J. 2019;6:1-4. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 1] [Cited by in RCA: 1] [Article Influence: 0.2] [Reference Citation Analysis (0)] |
| 16. | Repici A, Genco C, Bravatà I, Anderloni A. Endoprosthetics in the treatment of benign esophageal strictures. Tech Gastrointest Endosc. 2014;16:71-78. [RCA] [DOI] [Full Text] [Cited by in Crossref: 3] [Cited by in RCA: 3] [Article Influence: 0.3] [Reference Citation Analysis (0)] |
| 17. | Suter-Montano T, Montaño E, Martínez C, Plascencia T, Sepulveda MT, Rodríguez M. Adult recurrent respirator papillomatosis: a new therapeutic approach with pegylated interferon alpha 2a (Peg-IFNα-2a) and GM-CSF. Otolaryngol Head Neck Surg. 2013;148:253-260. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 15] [Cited by in RCA: 16] [Article Influence: 1.2] [Reference Citation Analysis (0)] |
| 18. | Kurita T, Umeno H, Chitose S, Ueda Y, Mihashi R, Nakashima T. [Laryngeal Papillomatosis: A Statistical Analysis of 60 Cases]. Nihon Jibiinkoka Gakkai Kaiho. 2015;118:192-200. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 4] [Cited by in RCA: 5] [Article Influence: 0.5] [Reference Citation Analysis (0)] |
| 19. | Yıldırım F, Türk M, Demircan S, Akyürek N, Yurdakul AS. Tracheal Papilloma Treated with Cryotherapy and Interferon-α: A Case Report and Review of the Literature. Case Rep Pulmonol. 2015;2015:356796. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 3] [Cited by in RCA: 8] [Article Influence: 0.8] [Reference Citation Analysis (0)] |