Published online Jul 6, 2025. doi: 10.12998/wjcc.v13.i19.102212
Revised: January 19, 2025
Accepted: February 28, 2025
Published online: July 6, 2025
Processing time: 158 Days and 13.8 Hours
Hepatitis B virus (HBV) infection can lead to renal involvement, commonly manifested as HBV-associated glomerulonephritis (HBV-GN), which typically presents as nephrotic or nephritic syndrome. Antineutrophil cytoplasmic anti
This manuscript presents the case of an older male patient who presented with persistent foamy urine lasting over two weeks. Initial clinical findings included nephrotic syndrome and renal insufficiency, which subsequently progressed to involve the lungs, immune system, hematologic system, and other organ systems. The patient was diagnosed with HBV-GN complicated by AAV, a rare and complex condition. Despite receiving comprehensive treatment, including corti
HBV infection may trigger nephropathy with AAV. Early recognition and intervention are crucial for improving patient prognosis.
Core Tip: This case highlights a 63-year-old man diagnosed with hepatitis B virus-related nephropathy complicated by antineutrophil cytoplasmic antibody-associated vasculitis. Despite undergoing treatments such as plasma exchange, protein A immunoadsorption, and immune modulation, the patient suffered dual renal insults, necessitating maintenance dialysis and resulting in a poor prognosis. Chronic hepatitis B virus infection may contribute to antineutrophil cytoplasmic antibody production, emphasizing the importance of early diagnosis and proactive clearance of immune complexes to improve outcomes in similar cases.
- Citation: Li MR, Li LY, Tang J, Sun J. Chronic hepatitis B triggering antineutrophil cytoplasmic antibody-associated vasculitis complicated by glomerulonephritis: A case report. World J Clin Cases 2025; 13(19): 102212
- URL: https://www.wjgnet.com/2307-8960/full/v13/i19/102212.htm
- DOI: https://dx.doi.org/10.12998/wjcc.v13.i19.102212
Chronic hepatitis B virus (HBV) infection affects an estimated 250-300 million individuals worldwide, with approximately 3%-5% of these patients developing renal complications. Among these, membranous nephropathy is the most common glomerular injury, although HBV infection is also associated with IgA nephropathy, cryoglobulinemia, and polyarteritis nodosa (Kussmaul-Maier disease)[1]. The HBV-related kidney involvement, termed HBV-associated glomerulonephritis (HBV-GN), commonly presents as nephrotic or nephritic syndrome. Currently, two hepatitis viruses have been linked to systemic vasculitis: HBV, which is primarily associated with classic polyarteritis nodosa, and hepatitis C virus, which is related to cryoglobulinemic vasculitis[2]. The HBV infection has a recognized tendency to induce antineutrophil cytoplasmic antibody (ANCA) production, resulting in clinical manifestations that mimic ANCA-associated vasculitis (AAV)[3]. Both HBV and AAV significantly impact renal function, increasing the likelihood of misdiagnosis or oversight of one condition during clinical evaluation. Simultaneous occurrence of HBV-GN and AAV is infrequent[2-4], with limited data available on diagnosis, management, clinical outcomes, and prognosis. Here, we report a 63-year-old Chinese man who was diagnosed with AAV complicated by glomerulonephritis. After comprehensive treatment with antiviral, immunotherapy, and plasma exchange, the patient’s prognosis was still poor. This case demonstrates a complex clinical scenario at the intersection of nephrology and infectious diseases, where concurrent HBV-GN and AAV complicate the diagnostic process. It underscores the need for heightened clinical awareness and offers a practical diagnostic approach to aid physicians in identifying and managing such rare cases.
The patient reported foamy urine persisting over two weeks and generalized swelling lasting 4 days.
Approximately two weeks prior, the patient experienced the onset of foamy urine without an identifiable cause, accompanied by bilateral lumbar and abdominal distension, and increased nocturia (5-6 times/night). Over the last 4 days, the patient developed facial and bilateral lower limb swelling, which progressively worsened.
The patient had a longstanding history of gouty arthritis, managed intermittently over several years.
The patient had unremarkable familial or personal history, with no notable medical conditions or genetic predispositions.
On admission, physical examination revealed facial and bilateral lower extremity edema. Blood pressure was elevated at 181/97 mmHg. No significant abnormalities were noted in pulmonary auscultation or findings.
Initial laboratory testing identified significant abnormalities (Table 1), including proteinuria (3 +), hematuria (3 +), hypoalbuminemia (22.9 g/L), elevated serum creatinine (Scr) (387 μmol/L), increased blood urea nitrogen (16.8 mmol/L), and hypercholesterolemia (9.24 mmol/L). Complement analysis showed a reduced C4 level (0.4 g/L). Immunological studies revealed positive perinuclear ANCA and an elevated anti-myeloperoxidase antibody IgG level of 380 AU/mL (Table 2).
| Items | Result | Reference values |
| Sedimentation (mm/hour) | 91 | 0-28 |
| CRP (mg/L) | 27.66 | 0-6 |
| WBC (× 109/L) | 8.95 | 3.5-9.5 |
| Neutrophils | 6.97 | 1.8-6.3 |
| PLT (× 109/L) | 167 | 125-350 |
| Hb (g/L) | 106 | 130-175 |
| D-dimer (mg/L) | 5.65 | < 0.55 |
| PCT (ng/mL) | 0.215 | < 0.045 |
| Albumin (g/L) | 22.9 | 40-55 |
| Serum creatinine (mol/L) | 387 | 57-111 |
| Blood urea nitrogen (mmol/L) | 16.8 | 3.1-9.5 |
| 24-hour urine protein (g/L) | 12.397 | <0.15 |
| Proteinuria | +++ | Negative |
| Hematuria | +++ | Negative |
| Cholesterol (mmol/L) | 9.24 | < 6.22 |
| Triglyceride (mmol/L) | 2.74 | < 1.7 |
| AFP (ng/mL) | 7.98 | < 7 |
| Items | Result | Reference values |
| ANA antibody | 1:160 (+) | Negative |
| Anti-SS-A | ± | Negative |
| Anti-dsDNA | Negative | Negative |
| Anti-SM | Negative | Negative |
| Anticardiolipin IgG (GPLU/mL) | 57.7 | 0-8 |
| C3 (g/L) | 0.88 | 0.79-1.52 |
| C4 (g/L) | 0.4 | 0.16-0.38 |
| p-ANCA | + | Negative |
| Anti-MPO | + | Negative |
| IgG4 (g/L) | 4.52 | 0.03-2.01 |
| Lupus anticoagulant | Negative | Negative |
| Anti-β2 glycoprotein 1 antibody | Negative | Negative |
| Serum anti-phospholipase A2 receptor antibody | Negative | Negative |
| HBsAg(ng/mL) | 0.01 | 0-0.5 |
| HBsAb (MIU/mL) | 99.23 | 0-10 |
| HBeAg (PEIU/mL) | 0.01 | < 0.1 |
| HBeAb (PEIU/mL) | 0.02 | < 0.15 |
| HBcAb (PEIU/mL) | 3.06 | < 0.7 |
| HBV-DNA (IU/mL) | < 20 | < 20 |
Ultrasound of the urinary system showed standard kidney dimensions with enhanced parenchymal echogenicity and reduced blood flow signals.
HBV triggering AAV complicated by glomerulonephritis.
The patient was started on a treatment regimen that included methylprednisolone (40 mg/day) for inflammation control, cyclophosphamide (0.6 g) for immunosuppression, ceftriaxone (1 g/day) for infection control, and enoxaparin (4000 IU/day) for anticoagulation at the beginning of the hospitalization. Furthermore, supportive treatments included renal protection, detoxification, antihypertensive therapy, gastric protection, calcium supplementation, albumin infusion, and intermittent diuretics. This comprehensive treatment approach reduced Scr to 228 mmol/L and significantly improved systemic edema.
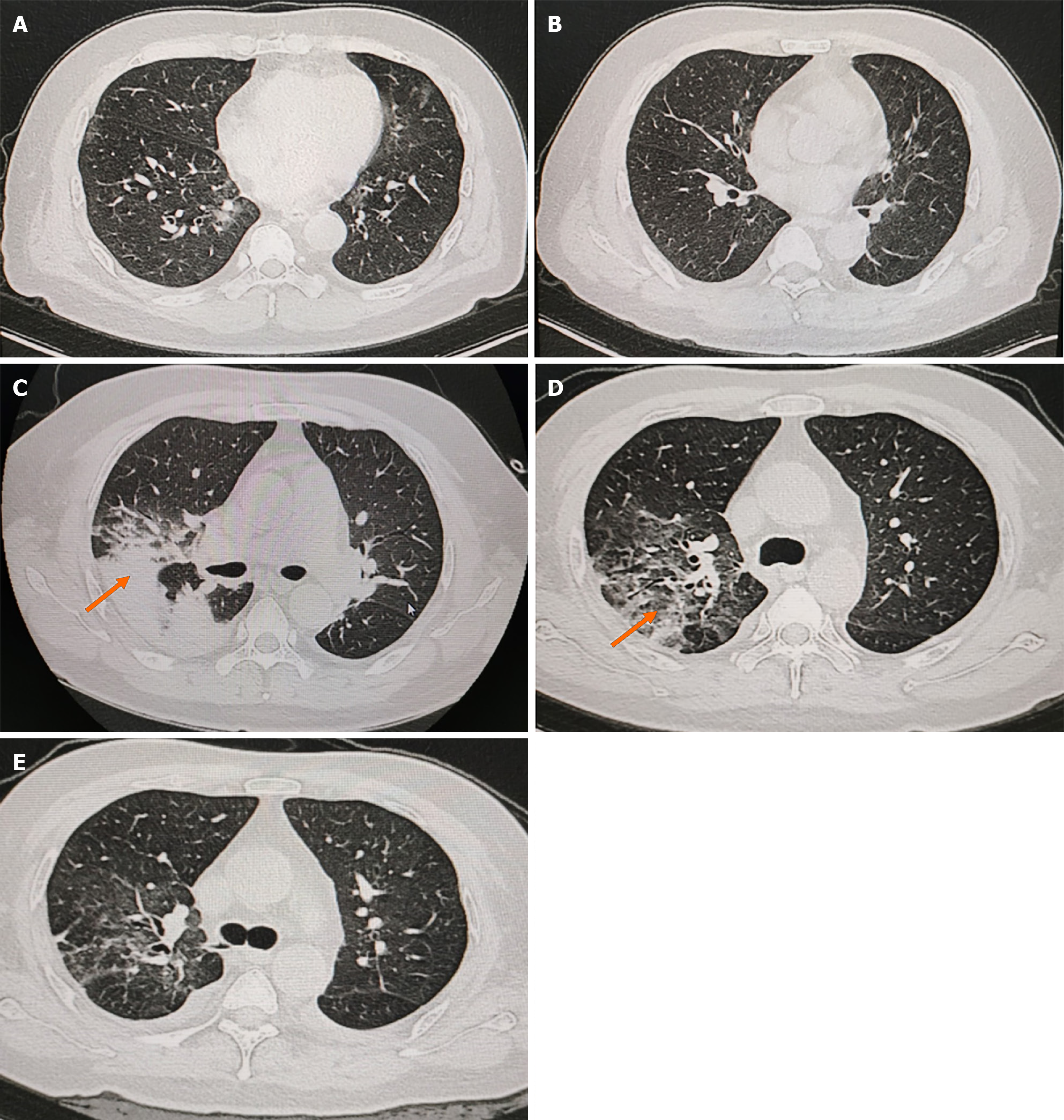
During hospitalization, the patient developed complications, including fever, dyspnea, and pulmonary hemorrhage (Figure 1). These symptoms necessitated transfer to the intensive care unit, where noninvasive ventilatory support was initiated to manage respiratory distress. According to the result of next-generation sequencing of alveolar lavage fluid, the treatment regimen was revised to include methylprednisolone (20 mg/day), meropenem (1 g/8 hour) combined with moxifloxacin (0.4 g/day) for antimicrobial therapy, and trimethoprim-sulfamethoxazole (0.48 g/day) for prophylaxis against Pneumocystis jirovecii. Entecavir (0.5 mg/5 day) was administered for antiviral treatment. Additional interventions included plasma transfusions to improve coagulation, intravenous immunoglobulin to enhance immune function, and plasmapheresis combined with six sessions of protein A immunoadsorption, performed between March 29 and April 12. Supportive care measures were also implemented, including renal protection and symptomatic management. The patient showed clinical improvement following treatment.
Five months post-discharge, the patient’s edema had resolved; however, renal function showed no significant recovery, necessitating ongoing maintenance hemodialysis.
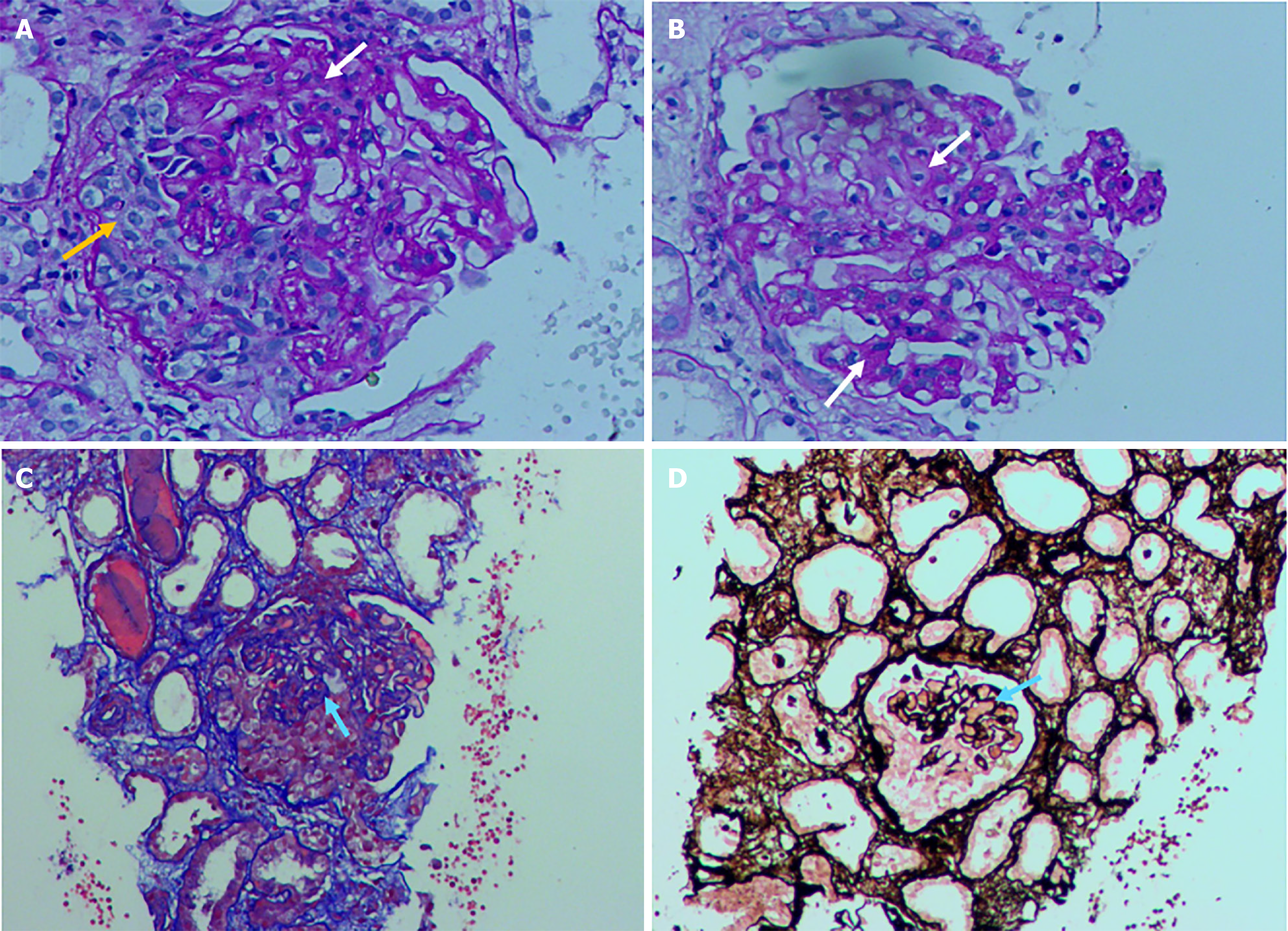
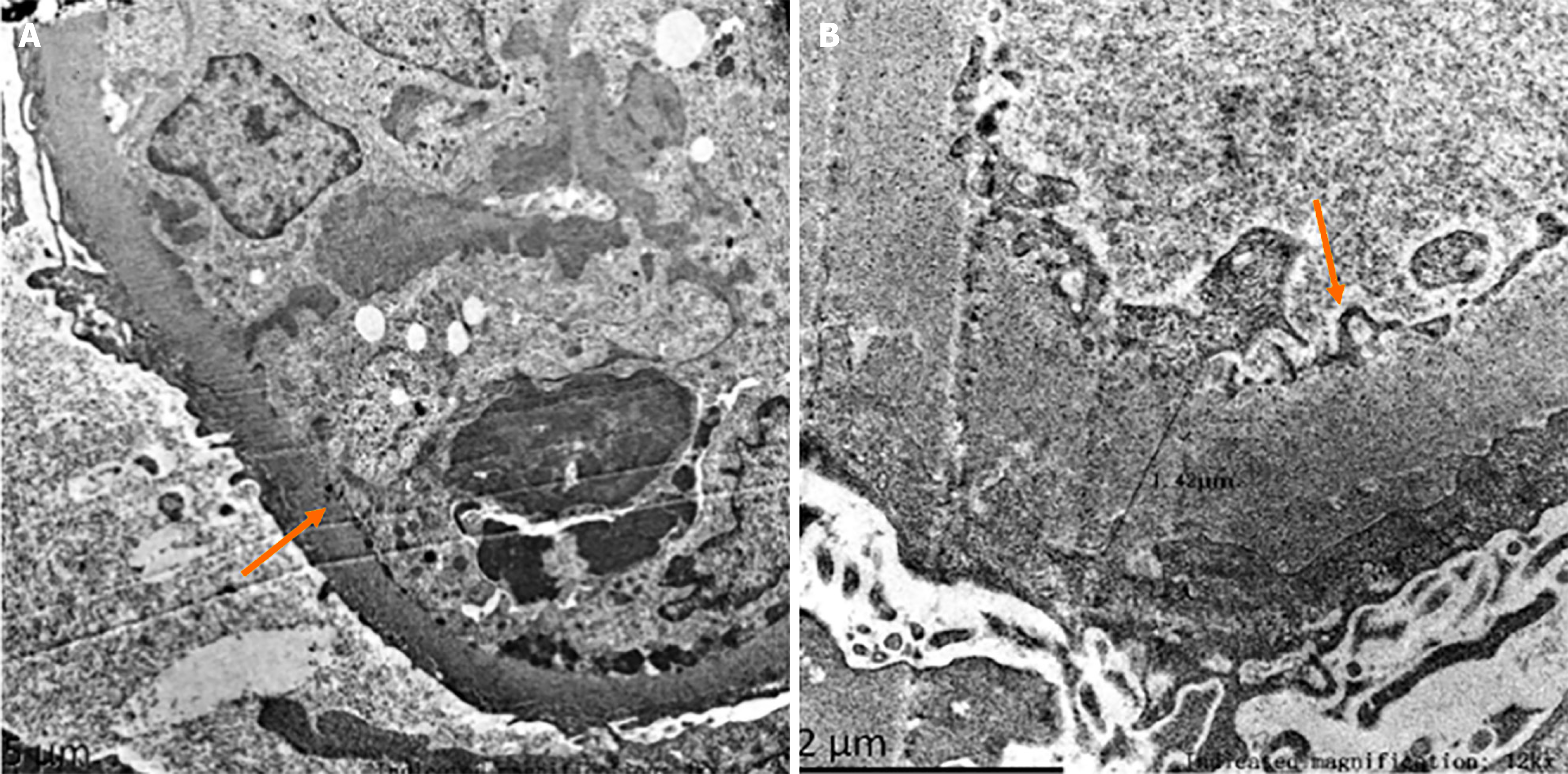
The underlying mechanisms of nephropathy in chronic HBV infection remain unclear. The condition is thought to result from the deposition of immune complexes (ICs)[5], with genetic predisposition potentially contributing to susceptibility[6]. The patient presented with multiorgan involvement, and renal pathology revealed crescent formation, suggesting a combination of HBV-GN and AAV-related renal damage. However, AAV typically manifests as necrotizing and crescentic glomerulonephritis without IC deposition[7]. However, renal biopsy findings (Figure 2) in this case revealed membranous changes accompanied by crescent formation. Granular deposition of IgA, IgM, C3, C1q, and IgG was observed in the mesangium and capillary loops alongside the granular presence of HBV markers (HBsAg, HBcAg, HBeAg) within the same regions. Electron microscopy (Figure 3) further confirmed the presence of IC deposits in subepithelial, intramembranous, subendothelial, and mesangial locations. These findings suggest that, in addition to neutrophil-mediated endothelial cell injury induced by ANCA[7], HBV infection contributes to IC formation and deposition within blood vessels, leading to progressive vascular wall damage over time. This process may trigger vasculitis and associated renal manifestations. The underlying pathogenesis likely involves a combination of direct viral invasion of vascular endothelial cells, IC-mediated vascular injury, and autoimmunity-mediated activation of autoreactive B cells and regulatory T cells[5]. This cascade of events, coupled with local complement activation and recruitment of inflammatory cells[8], results in blood vessel damage. A retrospective study involving 153 patients with AAV reported that those with positive HBcAb levels had a substantially greater risk of relapse and poorer survival rates, particularly in cases of eosinophilic granulomatosis with polyangiitis[9]. Chronic HBV infection induces ANCA production, resulting in clinical manifestations resembling AAV[3]. Moreover, patients with concurrent HBV infection have a heightened risk of poor prognosis compared with those without HBV infection. The patient's HBV DNA concentration was reported as < 20 IU/mL, however, it could trigger rapidly progressive and severe pathology. Although such low levels of viral replication may not cause significant hepatitis, they can stimulate the immune system, resulting in a chronic inflammatory state that may contribute to the development of vasculitis and glomerulonephritis. Moreover, an individual's genetic predisposition and immune response may influence the clinical manifestations of HBV infection[6]. Therefore, when a rapid decline in renal function occurs, the possibility of HBV-associated rapidly progressive glomerulonephritis should be considered, renal biopsy should be promptly carried out to find the basis, and antiviral and immunotherapy should be initiated promptly. This case exemplifies the critical importance of early renal function monitoring in patients with HBV infection.
However, in patients with concurrent HBV infection, the use of immunosuppressants poses the risk of rapid viral replication[8]. Elevated viral loads or persistent infection can facilitate immune complexes (IC) formation, with subsequent deposition in medium and small arteries[10], creating a self-perpetuating cycle of inflammation and tissue damage. Antiviral therapy is pivotal in addressing this issue. In the present case, treatment with entecavir, corticosteroids, and cytotoxic agents improved systemic edema, although no significant recovery in renal function was observed. Generally, clearing HBsAg from the bloodstream mitigates liver and kidney involvement[11]. However, a subset of patients may experience disease progression, resulting in chronic renal insufficiency[12]. Emerging evidence suggests that a combination of antiviral and immunosuppressive therapies can reduce proteinuria in patients with HBV-GN without altering HBV replication or impairing liver and kidney function[13]. However, the short duration of observation in these studies highlights the need for further evaluation of long-term outcomes. Patients with elevated Scr, alanine aminotransferase levels, and reduced estimated glomerular filtration rate derive more significant benefits from entecavir therapy[14]. This finding underscores the essential role of antiviral treatment in managing HBV-associated renal complications. Combining plasmapheresis with antiviral therapy can significantly expedite the clearance of ICs, providing benefits in case of acute organ failure[15]. In this case, the kidneys experienced a dual hit, and despite receiving interventions such as protein A immunoadsorption, corticosteroids, and cyclophosphamide, targeting AAV antibodies, the renal function could not be restored, necessitating long-term dialysis and resulting in a poor prognosis. An analysis of this case highlights several factors that may influence patient outcomes, including treatment strategies, the extent of renal involvement, the type and titer of ANCA, the level of viral replication control, the timing of therapeutic interventions, and disease activity scores[4]. Early clearance or control of HBV-related ICs may facilitate recovery in patients with HBV-associated nephropathy and reduce the risks of renal insufficiency and vasculitis, ultimately improving patient outcomes[16,17]. However, immunosuppressive therapy carries the risk of secondary infections, as it diminishes immune defenses[18]. These infections can progress rapidly, with atypical clinical presentations, complicating timely diagnosis[19]. Therefore, prompt microbiologic evaluation and immediate treatment of new or worsening symptoms are crucial. This case underscores the challenges of diagnosing and managing co-occurring HBV-GN and AAV. The rarity of this dual pre
The co-occurrence of HBV-GN and AAV is exceedingly rare, and this case serves as a valuable reference for developing practical diagnostic and therapeutic strategies. It highlights the importance of improving physicians' awareness and diagnostic capabilities when managing uncommon clinical scenarios. This case provides a deeper understanding of HBV-associated renal diseases, including HBV-GN and its potential association with AAV. The insights from this case provide a foundation for future research into the pathogenesis, diagnostic techniques, therapeutic measures, and prognostic factors associated with HBV-GN and AAV co-morbidity.
In cases of occult HBV infection, both the virus itself and unresolved ICs can trigger nephropathy and vasculitis-related clinical manifestations, potentially leading to misdiagnosis or inappropriate treatment. This situation necessitates heightened vigilance and a comprehensive approach during clinical diagnosis and treatment.
| 1. | Kidney Disease: Improving Global Outcomes (KDIGO) Glomerular Diseases Work Group. KDIGO 2021 Clinical Practice Guideline for the Management of Glomerular Diseases. Kidney Int. 2021;100:S1-S276. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 530] [Cited by in RCA: 1173] [Article Influence: 293.3] [Reference Citation Analysis (0)] |
| 2. | Sharma A, Sharma K. Hepatotropic viral infection associated systemic vasculitides-hepatitis B virus associated polyarteritis nodosa and hepatitis C virus associated cryoglobulinemic vasculitis. J Clin Exp Hepatol. 2013;3:204-212. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 27] [Cited by in RCA: 24] [Article Influence: 2.0] [Reference Citation Analysis (0)] |
| 3. | Meng Z, Cui W, Arend L, Mikdashi J. Hepatitis B virus infection associated with polyarteritis nodosa and microscopic polyangiitis. BMJ Case Rep. 2021;14:e240015. [RCA] [PubMed] [DOI] [Full Text] [Reference Citation Analysis (0)] |
| 4. | Guillevin L, Mahr A, Callard P, Godmer P, Pagnoux C, Leray E, Cohen P; French Vasculitis Study Group. Hepatitis B virus-associated polyarteritis nodosa: clinical characteristics, outcome, and impact of treatment in 115 patients. Medicine (Baltimore). 2005;84:313-322. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 317] [Cited by in RCA: 231] [Article Influence: 11.6] [Reference Citation Analysis (0)] |
| 5. | Thomas K, Vassilopoulos D. Infections and vasculitis. Curr Opin Rheumatol. 2017;29:17-23. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 13] [Cited by in RCA: 17] [Article Influence: 2.1] [Reference Citation Analysis (0)] |
| 6. | Thursz MR, Kwiatkowski D, Allsopp CE, Greenwood BM, Thomas HC, Hill AV. Association between an MHC class II allele and clearance of hepatitis B virus in the Gambia. N Engl J Med. 1995;332:1065-1069. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 316] [Cited by in RCA: 317] [Article Influence: 10.6] [Reference Citation Analysis (0)] |
| 7. | Jennette JC, Xiao H, Falk RJ. Pathogenesis of vascular inflammation by anti-neutrophil cytoplasmic antibodies. J Am Soc Nephrol. 2006;17:1235-1242. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 155] [Cited by in RCA: 146] [Article Influence: 7.7] [Reference Citation Analysis (0)] |
| 8. | Cacoub P, Terrier B. Hepatitis B-related autoimmune manifestations. Rheum Dis Clin North Am. 2009;35:125-137. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 59] [Cited by in RCA: 67] [Article Influence: 4.2] [Reference Citation Analysis (0)] |
| 9. | Lee SW, Kim DY, Ahn SH, Park YB, Han KH, Park JY. HBsAg-negative and anti-HBc-positive in eosinophilic granulomatosis with polyangiitis: a retrospective pilot study. Rheumatol Int. 2018;38:1531-1538. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 5] [Cited by in RCA: 5] [Article Influence: 0.7] [Reference Citation Analysis (0)] |
| 10. | Cacoub P, Saadoun D, Bourlière M, Khiri H, Martineau A, Benhamou Y, Varastet M, Pol S, Thibault V, Rotily M, Halfon P. Hepatitis B virus genotypes and extrahepatic manifestations. J Hepatol. 2005;43:764-770. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 81] [Cited by in RCA: 81] [Article Influence: 4.1] [Reference Citation Analysis (0)] |
| 11. | Lai KN, Lai FM, Tam JS. IgA nephropathy associated with chronic hepatitis B virus infection in adults: the pathogenetic role of HBsAG. J Pathol. 1989;157:321-327. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 20] [Cited by in RCA: 22] [Article Influence: 0.6] [Reference Citation Analysis (0)] |
| 12. | Hsu HC, Lin GH, Chang MH, Chen CH. Association of hepatitis B surface (HBs) antigenemia and membranous nephropathy in children in Taiwan. Clin Nephrol. 1983;20:121-129. [PubMed] |
| 13. | Zheng XY, Wei RB, Tang L, Li P, Zheng XD. Meta-analysis of combined therapy for adult hepatitis B virus-associated glomerulonephritis. World J Gastroenterol. 2012;18:821-832. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in CrossRef: 40] [Cited by in RCA: 41] [Article Influence: 3.2] [Reference Citation Analysis (0)] |
| 14. | Wang L, Xie B, Zheng Q, Xu L, Ye Z. Efficacy of entecavir in treating hepatitis B virus-associated membranous nephropathy. Rev Esp Enferm Dig. 2020;112:843-849. [RCA] [PubMed] [DOI] [Full Text] [Cited by in RCA: 3] [Reference Citation Analysis (0)] |
| 15. | Tripathi PP, Sharma RR, Kopp CR, Basnet A, Ramakrishnan S, Lamba DS, Hans R, Sharma A. Interesting rare case of polyarteritis nodosa related to hepatitis B virus and plasma exchange role? - A case report and review of the literature. Asian J Transfus Sci. 2023;17:7-12. [RCA] [PubMed] [DOI] [Full Text] [Reference Citation Analysis (0)] |
| 16. | Guo Y, Li S, Wei J, Luo M, Liu H. The relationship between HBV antigens deposition in kidneys and renal prognosis in IgA nephropathy patients infected with HBV. Ren Fail. 2024;46:2417737. [RCA] [PubMed] [DOI] [Full Text] [Reference Citation Analysis (0)] |
| 17. | Liu J, Chen R, Zhou S, Guo Z, Su L, Cao L, Li Y, Zhang X, Luo F, Xu R, Gao Q, Lin Y, Xu X, Nie S; CRDS study Investigators. Acute kidney injury is associated with liver-related outcomes in patients with hepatitis B virus infection: a retrospective cohort study. BMC Nephrol. 2025;26:12. [RCA] [PubMed] [DOI] [Full Text] [Reference Citation Analysis (0)] |
| 18. | Sarikaya ZT, Gucyetmez B, Tuzuner F, Dincer O, Sahan C, Dogan L, Yildirim SA, Zengin R, Kocagoz AS, Telci L, Akinci IO. The usage of immunosuppressant agents and secondary infections in patients with COVID-19 in the intensive care unit: a retrospective study. Sci Rep. 2024;14:20991. [RCA] [PubMed] [DOI] [Full Text] [Cited by in RCA: 2] [Reference Citation Analysis (0)] |
| 19. | Crook P, Logan C, Mazzella A, Wake RM, Cusinato M, Yau T, Ong YE, Planche T, Basarab M, Bicanic T. The impact of immunosuppressive therapy on secondary infections and antimicrobial use in COVID-19 inpatients: a retrospective cohort study. BMC Infect Dis. 2023;23:808. [RCA] [PubMed] [DOI] [Full Text] [Reference Citation Analysis (0)] |
| 20. | Zaltron S, Cambianica A, Di Gregorio M, Colangelo C, Storti S, Tiecco G, Castelli F, Quiros-Roldan E. Case report: An occult hepatitis B virus infection reactivation in an HIV/HCV coinfected patient during an immune reconstitution inflammatory syndrome. Front Cell Infect Microbiol. 2023;13:1143346. [RCA] [PubMed] [DOI] [Full Text] [Reference Citation Analysis (0)] |