INTRODUCTION
The clinical presentation of immunoglobulin (Ig) A nephropathy was initially delineated by Berger and Hinglais[1] in 1968 in a patient exhibiting inter-capillary IgA deposition. It is regarded as the most common type of primary glomerulonephritis worldwide. Although it is usually considered a benign disease, a significant proportion of cases may progress to end-stage kidney disease[2]. Here, we provide a brief summary of the clinical presentation and pathophysiology followed by a literature review focusing on novel treatment approaches.
CLINICAL PRESENTATION AND PROGNOSIS
The clinical presentation may vary considerably, from incidental microscopic hematuria to nephrotic syndrome or rapidly progressive glomerulonephritis. However, in almost all cases, there is a history of a triggering infection, the majority of which are upper respiratory tract infections. The highest incidence of IgA nephropathy (IgAN) is observed in the Asia-Pacific region, while the lowest prevalence is seen in Africa and India[2]. The clinical presentation and prognosis of IgAN significantly varies across age groups and ethnicities. It is predominantly diagnosed between the second and fourth decades of life. In younger patients, the predominant findings are gross hematuria accompanied by urine sediment abnormalities. In contrast, elderly cases commonly present with acute kidney injury, reduced glomerular filtration, hypertension, and prominent proteinuria[3,4].
Previously, IgAN was regarded as a relatively benign disease. However, longitudinal studies have demonstrated that 25%-50% of patients ultimately progress to end-stage renal disease (ESRD) over time. Additionally, IgAN has been linked to an elevated mortality risk and a reduced life expectancy of approximately six years[5,6]. A number of defined predictors of worse outcomes in IgAN have been identified, including: (1) Heavy proteinuria; (2) Male gender; (3) Lower serum albumin level; (4) Diastolic hypertension; (5) Age below 30 years; and (6) Presence of renal dysfunction at the time of diagnosis and active urinary sediment[7]. Therefore, IgAN should not be considered as a benign course, and preventive measures should be implemented as early as possible to avoid disease progression.
PATHOPHYSIOLOGY
The deposition of immune complexes in the mesangium represents the primary mechanism underlying IgAN. It is well established that the level of galactose-deficient IgA1 (Gd-IgA1) is increased in patients with IgAN. Furthermore, Gd-IgA1 is proposed as a marker for diagnosis and assessment of the treatment response. IgG autoantibodies recognize gd-IgA1 antibodies and form circulating immune complexes that subsequently deposit in the mesangium. The deposition of immune complexes in the mesangium induces mesangial cell proliferation and extracellular matrix protein production, which is mediated by increased levels of cytokines and growth factors. This in turn results in impaired podocyte and filtration barrier functions, leading to proteinuria and hematuria[8]. Gut dysbiosis is also shown to contribute to the pathogenesis of IgAN by stimulating the production of Gd-IGA1[9].
TREATMENT
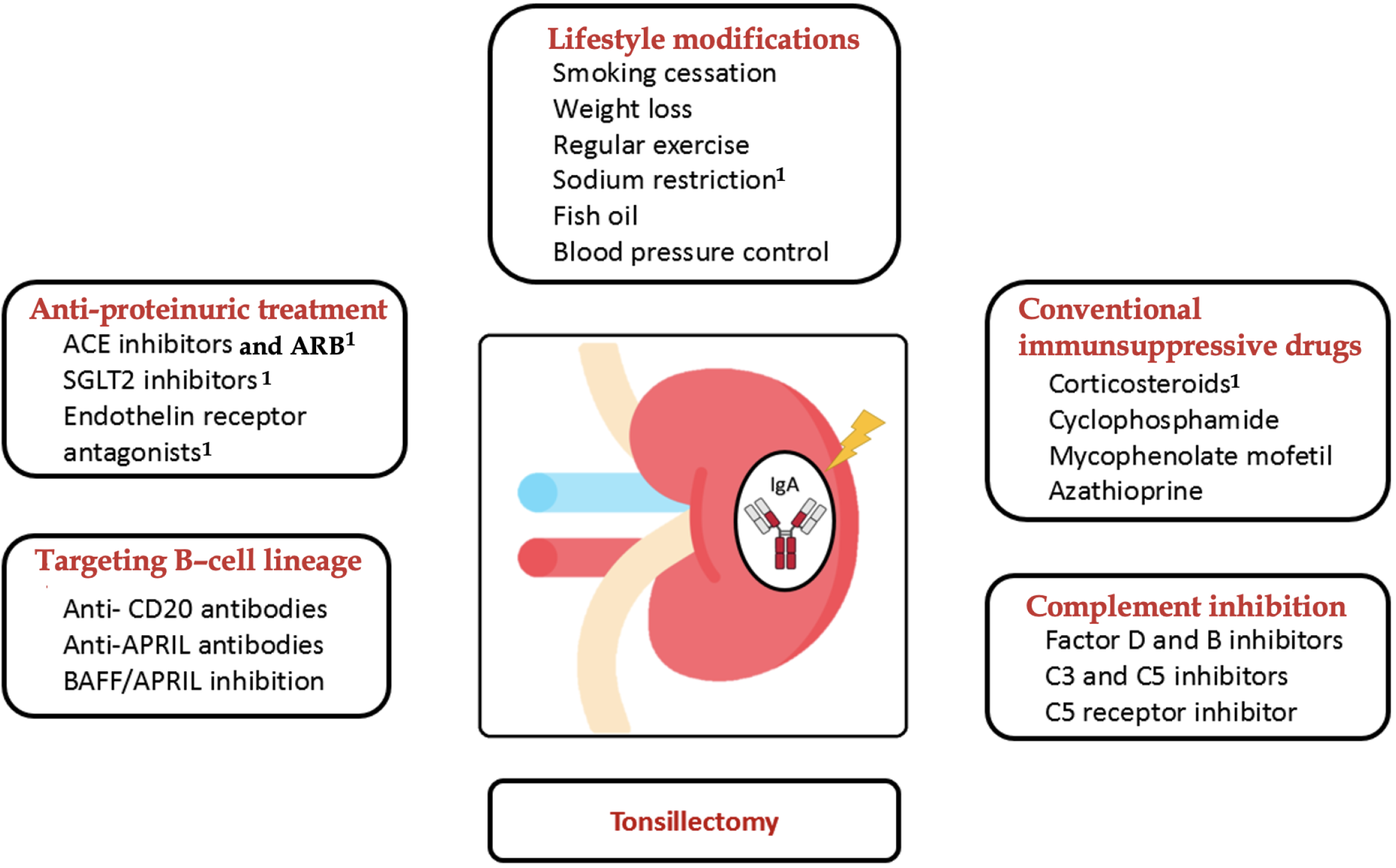
The management of IgAN should be determined based on the clinical presentation, the presence of renal dysfunction and the amount of proteinuria. The treatment of IgAN comprises lifestyle modifications, anti-proteinuric therapy and immunosuppressive treatment. Novel agents targeting B-cell lineage and the complement system have yielded promising results in early phase trials[3,10]. However, there is a paucity of consensus regarding the efficacy of tonsillectomy, with current evidence suggesting its potential benefit in the Japanese population. The treatment approach for IgAN, including novel therapies, is illustrated in Figure 1.
Figure 1 Treatment options in immunoglobulin A nephropathy.
ACE: Angiotensin converting enzyme; ARB: Angiotensin receptor blockers; APRIL: A proliferation-induced ligand; BAFF: B-cell-activating factor; SGLT2: Sodium-glucose cotransporter-2. 1Treatment options with proven certain clinical benefits by clinical trials.
Lifestyle modifications
It is recommended that the general recommendations for reducing cardiovascular risk be applied to patients with IgAN. The following measures are advised: (1) Smoking cessation; (2) Regular exercise; and (3) Maintaining weight within normal ranges. In addition, sodium intake should be restricted to below 2 g per day, as this is the only dietary recommendation that has been associated with outcomes[10,11]. Furthermore, blood pressure should be controlled, with the target systolic blood pressure set at below 120 mmHg. While some studies have indicated beneficial effects, there is nevertheless a paucity of consensus regarding the use of fish oil[10,11].
Anti-proteinuric treatment
Anti-proteinuric treatment comprises three main categories of pharmacological agents: (1) Renin-angiotensin-aldosterone system (RAAS) inhibitors; (2) Sodium-glucose cotransporter-2 (SGLT2) inhibitors; and (3) Endothelin receptor antagonists (ERAs). The RAAS inhibitors comprise angiotensin converting enzyme (ACE) inhibitors and angiotensin receptor blockers (ARB). RAAS inhibitors should be administered and the dose should be uptitrated to the maximally tolerated dose, particularly in patients with more than 500 mg/d proteinuria[11]. RAAS inhibitors are the only treatment that has demonstrated a clinical benefit among non-immunosuppressive therapies[12]. Combination of ACE inhibitors and ARBs results in a greater reduction in proteinuria levels. However, it is not recommended due to increased risk of adverse events including acute kidney injury and hyperkalemia[13].
SGLT2 inhibitors were initially developed as antidiabetic agents, and it was subsequently demonstrated that they exert beneficial effects on the treatment of heart failure and chronic kidney disease (CKD), irrespective of their anti-diabetic properties. “Dapagliflozin in patients with CKD” (DAPA-CKD) and “empagliflozin in patients with CKD” trials provided evidence that the addition of dapagliflozin or empagliflozin to standard care slowed CKD progression and resulted in a reduction in cardiovascular mortality in patients with or without diabetes[14,15]. A post-hoc subgroup analysis of IgAN patients enrolled in the DAPA-CKD trial revealed a significant benefit in this population[16]. Therefore, SGLT2 inhibitors should be considered in patients with persistent proteinuria despite maximally tolerated RAAS inhibition or as an adjunctive therapy on top of RAAS inhibition to improve outcomes. However, it should be noted that concomitant use of steroids and SGLT2 inhibitors might increase the risk of urinary tract infections[11].
Endothelin-1 plays a significant role in the pathophysiology of IgAN by inducing inflammation, vasoconstriction, podocyte injury and cell proliferation. The PROTECT study revealed a 41% greater reduction in proteinuria with sparsentan [a dual endothelin receptor A (ETA) and angiotensin receptor blocker] compared to irbesartan[17]. There are some concerns about ETAs in relation to salt and water intake, but there is growing evidence that the combined use of SGLT2 inhibitors and ETAs is an effective and safe treatment option[18]. Combined use of RAAS inhibitors, SGLT2 inhibitors and ERAs is proposed to be a standard supportive treatment of IgAN as similar to 4-pillar treatment of heart failure[19].
Conventional immunosuppressive therapy
Conventional immunosuppressive therapy comprises corticosteroids, cyclophosphamide, mycophenolate mofetil and azathioprine. The use of corticosteroids has been demonstrated to reduce proteinuria levels in patients with persistent proteinuria despite long-term anti-proteinuric therapy, as evidenced by the findings of the "supportive vs immunosuppressive therapy for the treatment of progressive IgAN" trial[20]. The “therapeutic effects of steroids in IgAN global” trial, in which 75% of participants are Chinese patients, also exhibited a notable reduction in proteinuria levels following corticosteroid treatment. However, the trial was terminated prematurely due to concerns regarding the potential for increased adverse effects associated with corticosteroid therapy[21]. The current guidelines recommend a short course of corticosteroid treatment (6 mo) for patients with persistent proteinuria despite maximal supportive care[10].
Although a small study demonstrated a beneficial impact of cyclophosphamide and azathioprine treatment on outcomes, subsequent studies failed to confirm those beneficial effects[11]. The impact of mycophenolate mofetil on IgAN also remains a topic of debate. Studies conducted in Chinese people suggest beneficial effects of mycophenolate mofetil, while studies reported from North America and European countries did not demonstrate such clear benefits[22-24]. Patients with persisting proteinuria (> 1 g/d) despite at least two rounds of steroids or conventional immunosuppressive therapy are defined as treatment refractory cases, and the novel therapeutic agents discussed below should be considered in those patients[25].
Targeting B–cell lineage
B lymphocytes and plasmocytes are responsible for the overproduction of Gd-IgA1. Thus, targeting B cells appears to be a reasonable approach for treating IgAN. The maturation of B cells is regulated by several factors, including B-cell-activating factor (BAFF) and a proliferation-induced ligand (APRIL), which are produced by macrophages and dendritic cells located in the lamina propria[26]. A variety of agents are used to deplete B cells, including anti-CD20 antibodies (such as Rituximab), anti-APRIL antibodies (such as Sibeprenlimab) and combined BAFF/APRIL inhibitors (such as Telitacicept and Atacicept). Rituximab proved ineffective in treating IgAN. However, sibeprenlimab and telitacicept demonstrated favorable outcomes in reducing proteinuria and inducing clinical remission in phase 2 trials. The results of the ongoing phase 3 trials, which include sibeprenlimab and telitacicept, are anticipated to yield promising results[3].
Complement inhibition
The complement system plays a significant role in the pathogenesis of IgAN. Despite the deposition of IgG and IgM, the alternate pathway is activated instead of the classic pathway. There are several agents targeting numerous components of the complement pathway, and clinical trials are ongoing. The results of these trials will inform the role of complement inhibitors in IgAN[3].
Tonsillectomy
The benefits of tonsillectomy were only demonstrated in Japanese individuals. Tonsillectomy resulted in improved kidney survival and increased remission rates in Japanese patients. However, these findings have not been confirmed in other populations. Therefore, tonsillectomy is not routinely recommended in non-Japanese people[10].
CONCLUSION
IgAN is the most common primary glomerulonephritis worldwide, and in the absence of efficacious treatment, it progresses to ESRD. The fundamental aspect of the treatment plan is supportive care, which encompasses a combination of lifestyle modifications and anti-proteinuric therapy. While RAAS blockers were previously the sole available option, SGLT2 inhibitors and ERAs have recently been identified as efficacious alternative anti-proteinuric regimens. While clinical remission is achieved in most cases with supportive treatment, a considerable proportion of patients require additional intervention to control the disease. Given that the principal mechanism underlying the disease appears to be increased production of Gd-IgA1 by B cells, immunosuppressive treatment is employed in high-risk patients. The clinical benefit was only established with corticosteroids by clinical trials and with mycophenolate mofetil in Chinese patients. B-cell lineage depletion therapies (Telitacicept, etc) and complement system inhibiting agents are promising novel therapies, and their role in the treatment is likely to become more important depending on the results of ongoing trials. It should be noted that pathological findings regarding the severity of inflammation and fibrosis may guide the treatment approach. The impact of novel therapies should be evaluated in diverse ethnicities, as genetic background plays a pivotal role in IgAN.
Provenance and peer review: Invited article; Externally peer reviewed.
Peer-review model: Single blind
Specialty type: Medicine, research and experimental
Country of origin: Netherlands
Peer-review report’s classification
Scientific Quality: Grade A, Grade D, Grade E
Novelty: Grade B, Grade C, Grade C
Creativity or Innovation: Grade B, Grade B, Grade C
Scientific Significance: Grade A, Grade B, Grade D
P-Reviewer: Al-Karawi AS; Bacharaki D; Zhou ZL S-Editor: Luo ML L-Editor: Filipodia P-Editor: Zhang XD