Published online Apr 6, 2024. doi: 10.12998/wjcc.v12.i10.1810
Peer-review started: November 20, 2023
First decision: January 17, 2024
Revised: January 31, 2024
Accepted: March 7, 2024
Article in press: March 7, 2024
Published online: April 6, 2024
Processing time: 133 Days and 17.8 Hours
Idiopathic mesenteric phlebosclerosis (IMP) is a rare type of ischemic colitis cha
A 77-year-old woman was admitted to the hospital due to chronic diarrhea for over 2 months. She had been consuming Chinese patent medicines (CPM) containing fructus gardeniae for more than 15 years. Colonoscopy revealed an edematous mucosa, bluish-purple discoloration, erosions, and ulcerations throughout the colorectal area. Abdominal computed tomography (CT) showed diffuse mural thickening of the entire colorectum, with tortuous thread-like calcifications in the right hemicolon, left hemicolon, and rectum. Most of the calcifications were located in the mesenteric vein. The diagnosis of IMP was established based on medical history, colonoscopy, CT findings, and histopathological examination. The patient was treated conservatively with papaverine and rifaximin, and CPM was stopped. Her diarrhea symptoms improved, indicating the effectiveness of the treatment. Over the next several years, she took opium alkaloids for an extended period and did not require hospitalization for the aforementioned gastrointestinal disorder.
IMP is a rare gastrointestinal disease affecting Asian populations, possibly related to long-term herbal medicine intake. Accurate imaging analysis is crucial for diagnosis, but insufficient understanding of the disease can lead to misdiagnosis or missed diagnosis. Treatment strategies should be personalized.
Core Tip: Idiopathic mesenteric phlebosclerosis (IMP) is a rare type of ischemic colitis characterized by thickening of the wall of the right hemicolon and calcification, sclerosis, and fibrosis of mesenteric veins. The diagnosis is based on typical clinical features and abdominal imaging. We report a case of IMP that was initially missed by the radiologist. Finally, IMP was diagnosed by a clinically experienced endoscopist.
- Citation: Wang M, Wan YX, Liao JW, Xiong F. Idiopathic mesenteric phlebosclerosis missed by a radiologist at initial diagnosis: A case report. World J Clin Cases 2024; 12(10): 1810-1816
- URL: https://www.wjgnet.com/2307-8960/full/v12/i10/1810.htm
- DOI: https://dx.doi.org/10.12998/wjcc.v12.i10.1810
Idiopathic mesenteric phlebosclerosis (IMP) is a rare form of ischemic colitis characterized by thickening of the wall of the right hemicolon and calcification, sclerosis, and fibrosis of mesenteric veins[1]. This clinical entity was first described by Yao et al[2] in 1989, and it was proposed to be named as IMP in 2003[3]. In the early stages of the disease, patients may be asymptomatic or have atypical symptoms. As the disease progresses, patients may present with abdominal pain, abnormal stool, or intestinal obstruction[4]. The diagnosis of IMP is based on the characteristic clinical features and abdominal imaging findings[5]. However, we encountered a case of IMP that was initially missed by the radiologist.
A 77-year-old woman was admitted to the hospital with chronic diarrhea for more than 2 months.
Symptoms started more than 2 months before presentation with chronic diarrhea.
The patient had a history of taking Chinese patent medicines (CPM) containing fructus gardeniae for more than 15 years for treatment of constipation. Four years back, she was diagnosed with chronic obstructive pulmonary disease. There was no history of infectious diseases (such as hepatitis or tuberculosis), hypertension, coronary heart disease, or diabetes.
There was no family history of malignant tumors.
On physical examination, there was severe edema in the patient’s lower legs and her abdomen was distended and mildly tender.
Initial laboratory investigations showed hypoalbuminemia (albumin: 30.8 g/L), electrolyte imbalance (K+: 2.38 mmol/L; Na+: 128.4 µmol/L; Cl-: 90.8 mmol/L), and anemia (hemoglobin: 78 g/L). Other laboratory indices were normal. Polymerase chain reaction for Mycobacterium tuberculosis, Epstein-Barr virus-encoded RNA, cytomegalovirus (CMV)-BD, and CMV-KT were all negative. The enzyme immunoassay for Clostridium difficile toxins A and B were also negative. The stool bacterium culture was negative.
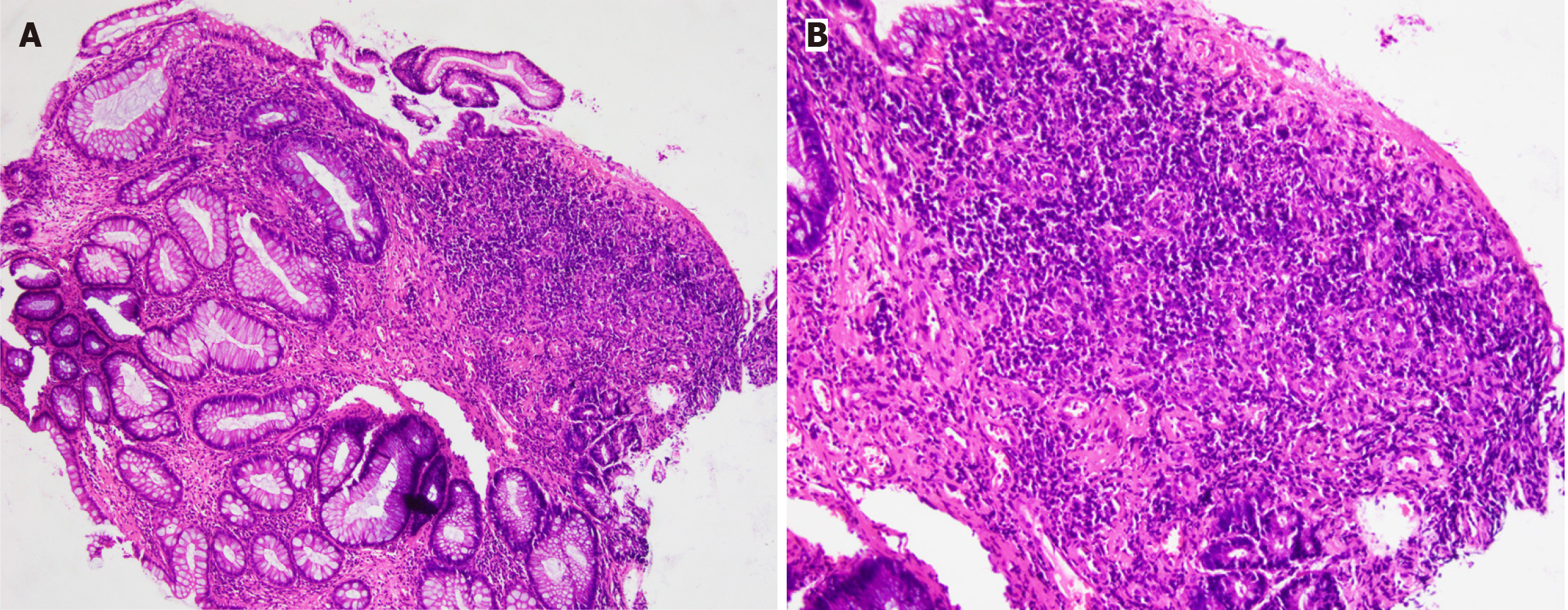
Carotid color Doppler flow imaging showed carotid atherosclerosis plaque. Abdominal sonography showed the presence of ascites. The initial abdominal computed tomography (CT) scan did not clearly depict numerous thread-like calcifications within the right-side mesenteric veins and their branches. These calcifications appeared denser in intramural tributaries, marginal veins, and vena recti peripherally. Colonoscopy revealed an edematous mucosa with bluish-purple discoloration, erosions, and ulcerations throughout the colorectal region (Figure 1). Histopathological examination showed mild inflammation and granulomatous reactions (Figure 2).
After reevaluating the abdominal CT images in conjunction with colonoscopy findings, a tentative diagnosis of IMP was considered. Further observation of the abdominal CT images exhibited diffuse mural thickening of the entire colon and rectum, with tortuous thread-like calcifications in the right hemicolon, left hemicolon, and rectum. Notably, most of these calcifications were located within the mesenteric vein (Figure 3). Arterial calcification usually occurs in the major arteries. However, it exhibits an irregular and patchy appearance, and usually there are no large areas of point-like fine linear calcifications. Our patient had a plaque in the abdominal aorta and showed stratified calcification in the intestinal wall (Figure 4A and B). The number of point-like intensifications in the arterial and venous phases in similar areas at the same level varied. There was more punctate reinforcement in the arterial phase than in the venous phase, and the punctate reinforcement in the venous phase indicates the foci of calcification (Figure 4C and D). Mesenteric venous calcification points, usually compared to the same level of the venous phase, can be found in the relatively dense occurrence of punctate and linear enhanced areas. In contrast to the arterial phase, the point of disappearance and the position of linear enhancement in the venous phase are usually near the calcification point, and some of the linear enhancement sites become point-like locations, suggesting that there may be arterial enhancement near the calcification point at the same time, which makes the imaging turn linear. It becomes punctate calcification only after the venous phase, relatively in a nearby concomitant position, similar to the relative position of the arteriovenous mesentery. In the arterial phase, the vanishing points in the venous phase will appear randomly, and there will be no similar concomitant relationship between the vanishing points and the calcification points in the same area, and there will be no possibility of linear calcification becoming punctate calcification.
A diagnosis of IMP was established based on the patient’s medical history, colonoscopy and CT findings.
The patient was immediately advised to discontinue the use of CPM. Intravenous papaverine (30 mg twice a day) was administered for 3 d, but there was no significant improvement in diarrhea. Although fecal occult blood test was positive and fecal flora was normal, the levels of C-reactive protein (25.03 mg/L) and procalcitonin (0.13 ng/mL) were raised. Therefore, oral rifaximin 0.2 g three times a day was initiated to cover potential intestinal infection. The patient’s diarrhea significantly improved, with 1-2 bowel movements per day and improved stool consistency. After discharge, oral rifaximin was continued for 2 wk, while oral papaverine 30 mg is being continued till date, without any side effects.
Nearly 5 years after discharge, the patient has not been readmitted to the hospital for the above disease.
IMP is a rare chronic disease characterized by non-thrombotic, non-inflammatory stenosis or occlusion of the mesenteric veins[6]. It primarily affects the right hemicolon, often involving the proximal colon, and may extend to the terminal ileum or the distal colon[7]. According to a literature review, IMP is more prevalent in middle-aged and elderly men [median age at onset: 61 years (range, 22-87 years)][8]. Guo et al[9] observed that the majority of patients were Asians, predominantly from Japan, Taiwan, and mainland China. The exact etiology and pathogenesis of IMP remain elusive but long-term exposure to toxins and biochemical substances may play a role[10]. Nearly all reported cases of IMP had a history of prolonged use of medicinal herbs containing geniposide, such as oldenlandia herbacea, plantaginis semen, eucommiae cortex, herba paederiae scandentis, radix scrophulariae, and fructus gardeniae[11]. Notably, licorice has also been reported as a possible causative agent of IMP[11]. Our patient was a 77-year-old Chinese woman who had a history of taking CPM containing fructus gardeniae for over 15 years, consistent with previous literature reports, and ultimately developed IMP.
Some scholars have proposed that geniposide can be transformed into genipin in the intestinal tract through hydrolysis under the influence of various microenvironmental factors such as the intestinal flora, amino acids, and sulfuric acid. The absorbed genipin then reacts with proteins in the mesenteric vein plasma[6,8]. Additionally, the gradual submucosal accumulation of collagen leads to intimal hyperplasia, venous wall thickening, and fibrosis, resulting in a condition known as “mummification”[6,12]. This process leads to obstruction of venous lumen, impedes venous return, and induces intestinal wall thickening, edema, gliosis, and sclerosis[8,12]. However, geniposide undergoes hydrolysis only after entering the colon and is then transformed to its metabolite, genipin. Genipin then permeates the enterocyte membrane under the influence of various colon microenvironments[6]. This explains why the characteristic lesion sites of IMP are the right-sided colon and transverse colon, which can spread to the entire colorectal area as the disease progresses.
In the early stage, many patients with IMP (10%-27.7%) are asymptomatic[8,13-15]. However, as the disease progresses, patients may present with nonspecific symptoms such as abdominal pain, diarrhea, constipation, nausea, vomiting, abdominal distension, fever, and bloody stool[8,15]. These symptoms complicate the diagnosis of IMP, necessitating the use of abdominal CT and colonoscopy for diagnostic purposes, with histopathological examination ultimately confirming the diagnosis[8]. The typical abdominal CT findings are colonic wall thickening and diffuse calcifications in small mesenteric veins and their intramural branches[16]. Although the right-sided colon is more commonly involved, the entire colon may be involved in severe cases[16]. The typical colonoscopy findings are a blue or bluish-purple mucosa, which may also display hyperemia, edema, erosion, or ulceration[6]. Histopathologically, IMP is characterized by venous wall thickening, fibrosis, and calcification, venous narrowing, atrophy, and submucosal fibrosis with intrinsic myometrium thickening[10]. Our patient presented with chronic diarrhea. Abdominal CT showed diffuse mural thickening of the entire colorectum and tortuous thread-like calcifications in the right and left hemicolon and rectum, with most calcifications located in the mesenteric veins. Colonoscopy revealed lesions involving the entire colorectal region. Histopathological examination revealed mild inflammation and granulomatous changes. These findings were consistent with the diagnosis of IMP.
As there are currently no standardized treatment guidelines or specific therapeutic agents for IMP, discontinuation of herbal medicine is considered the first-line therapy for early-stage IMP, which, when combined with proper treatment, generally leads to a favorable prognosis[17,18]. According to a large-scale nationwide survey, discontinuation of herbal medicines led to alleviation of symptoms in 84.3% of patients, with none of the cases requiring surgery post-discontinuation[13]. However, the choice between conservative and surgical treatment in patients with IMP is challenging. Surgical treatment will be necessary in case of severe complications, such as colonic obstruction, necrosis, perforation, or massive intestinal bleeding[19]. Sze et al[20] reported a 45-year-old Chinese woman with IMP who had stopped con
IMP is a rare intestinal disease that is more prevalent among Asians. The pathogenesis of IMP remains unclear, but it may be associated with the long-term intake of medicinal herbs. While it is crucial to analyze the imaging features on CT or colonoscopy, a lack of awareness about the disease can easily lead to missed or misdiagnoses. The treatment strategy for IMP should be individualized to achieve the best possible outcome.
Provenance and peer review: Unsolicited article; Externally peer reviewed.
Peer-review model: Single blind
Specialty type: Gastroenterology and hepatology
Country/Territory of origin: China
Peer-review report’s scientific quality classification
Grade A (Excellent): A
Grade B (Very good): 0
Grade C (Good): 0
Grade D (Fair): 0
Grade E (Poor): 0
P-Reviewer: Sharma D, India S-Editor: Zhang H L-Editor: Wang TQ P-Editor: Zhao S
| 1. | Zheng Q, Zhang B, Cao Z, Wang L. Idiopathic mesenteric phlebosclerosis associated with long-term oral intake of herb drug occurring in a mother and her daughter. Clin Res Hepatol Gastroenterol. 2021;45:101467. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 1] [Cited by in RCA: 3] [Article Influence: 0.8] [Reference Citation Analysis (0)] |
| 2. | Yao K, Iwashita A. Diagnosis of Early Gastric Cancer: Endoscopic Diagnosis and Classification: VS Classification System for the Diagnosis of Early Gastric Cancer by Magnifying Endoscopy. In: Chiu PWY, Sano Y, Uedo N, Singh R. Endoscopy in Early Gastrointestinal Cancers, Volume 1. Singapore: Springer. [DOI] [Full Text] |
| 3. | Iwashita A, Yao T, Schlemper RJ, Kuwano Y, Iida M, Matsumoto T, Kikuchi M. Mesenteric phlebosclerosis: a new disease entity causing ischemic colitis. Dis Colon Rectum. 2003;46:209-220. [RCA] [PubMed] [DOI] [Full Text] [Cited by in RCA: 28] [Reference Citation Analysis (0)] |
| 4. | Ding J, Zhang W, Wang L, Zhu Z, Wang J, Ma J. Idiopathic mesenteric phlebosclerosis: clinical and CT imaging characteristics. Quant Imaging Med Surg. 2021;11:763-771. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 3] [Cited by in RCA: 15] [Article Influence: 3.8] [Reference Citation Analysis (0)] |
| 5. | Takasaki T, Motomura Y, Shioya K. Idiopathic Mesenteric Phlebosclerosis. Clin Gastroenterol Hepatol. 2022;20:A19. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 1] [Cited by in RCA: 1] [Article Influence: 0.3] [Reference Citation Analysis (0)] |
| 6. | Wen Y, Chen YW, Meng AH, Zhao M, Fang SH, Ma YQ. Idiopathic mesenteric phlebosclerosis associated with long-term oral intake of geniposide. World J Gastroenterol. 2021;27:3097-3108. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in CrossRef: 7] [Cited by in RCA: 18] [Article Influence: 4.5] [Reference Citation Analysis (0)] |
| 7. | Jiang Y, Huang S, Chen K, Chen X. [Idiopathic mesenteric phlebosclerosis: one case report and literature review]. Zhong Nan Da Xue Xue Bao Yi Xue Ban. 2017;42:117-120. [RCA] [PubMed] [DOI] [Full Text] [Reference Citation Analysis (0)] |
| 8. | Wang J, Shao J, Lu H, Wang B, Chen J. Idiopathic mesenteric phlebosclerosis: one case report and systematic literature review of 240 cases. Am J Transl Res. 2021;13:13156-13166. [PubMed] |
| 9. | Guo F, Zhou YF, Zhang F, Yuan F, Yuan YZ, Yao WY. Idiopathic mesenteric phlebosclerosis associated with long-term use of medical liquor: two case reports and literature review. World J Gastroenterol. 2014;20:5561-5566. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in CrossRef: 24] [Cited by in RCA: 29] [Article Influence: 2.6] [Reference Citation Analysis (0)] |
| 10. | Wen Y, Zhao M, Huang W, Fang S, Lin C. Idiopathic mesenteric phlebosclerosis associated with use of Chinese herbal medicine: Two case reports. Medicine (Baltimore). 2020;99:e22813. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 4] [Cited by in RCA: 3] [Article Influence: 0.6] [Reference Citation Analysis (0)] |
| 11. | Jin YR, Zhou H, Liu ZZ, Li XF, Liang J. Idiopathic mesenteric phlebosclerosis occurring after long-term medication with licorice: A case report. J Dig Dis. 2022;23:183-185. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 1] [Cited by in RCA: 1] [Article Influence: 0.3] [Reference Citation Analysis (0)] |
| 12. | Young Chuah Y, Yeh Lee Y. Geniposide Causes Idiopathic Mesenteric Phlebosclerosis. Turk J Gastroenterol. 2023;34:890-891. [RCA] [PubMed] [DOI] [Full Text] [Cited by in RCA: 3] [Reference Citation Analysis (1)] |
| 13. | Shimizu S, Kobayashi T, Tomioka H, Ohtsu K, Matsui T, Hibi T. Involvement of herbal medicine as a cause of mesenteric phlebosclerosis: results from a large-scale nationwide survey. J Gastroenterol. 2017;52:308-314. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 25] [Cited by in RCA: 32] [Article Influence: 4.0] [Reference Citation Analysis (0)] |
| 14. | Chen W, Zhu H, Chen H, Shan G, Xu G, Chen L, Dong F. Phlebosclerotic colitis: Our clinical experience of 25 patients in China. Medicine (Baltimore). 2018;97:e12824. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 11] [Cited by in RCA: 18] [Article Influence: 2.6] [Reference Citation Analysis (0)] |
| 15. | Chou JW, Chang CH, Wu YH, Chang KC, Cheng KS, Huang PJ. Idiopathic Mesenteric Phlebosclerosis: A Single-Institute Experience in Taiwan. Turk J Gastroenterol. 2023;34:483-489. [RCA] [PubMed] [DOI] [Full Text] [Cited by in RCA: 5] [Reference Citation Analysis (1)] |
| 16. | Li H, Liu H. Idiopathic Mesenteric Phlebosclerosis. Radiology. 2023;306:76. [RCA] [PubMed] [DOI] [Full Text] [Cited by in RCA: 1] [Reference Citation Analysis (0)] |
| 17. | Shan J, Chen F, Yu P. Intestinal obstruction due to idiopathic mesenteric phlebosclerosis colitis: A case report. Front Surg. 2022;9:969154. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in RCA: 4] [Reference Citation Analysis (0)] |
| 18. | Kawashima A, Shimomura A, Inagaki T. Idiopathic Mesenteric Phlebosclerosis Secondary to Chinese Herbal Medicine Intake in an Older Adult. Am J Trop Med Hyg. 2023;109:715-716. [RCA] [PubMed] [DOI] [Full Text] [Reference Citation Analysis (0)] |
| 19. | Washington C, Carmichael JC. Management of ischemic colitis. Clin Colon Rectal Surg. 2012;25:228-235. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 81] [Cited by in RCA: 81] [Article Influence: 6.8] [Reference Citation Analysis (0)] |
| 20. | Sze SF, Lam PWY, Lam JTW, Chung CCC, Lee YT. Idiopathic mesenteric phlebosclerosis: A rare cause of chronic diarrhea. JGH Open. 2020;4:769-770. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in RCA: 5] [Reference Citation Analysis (0)] |