Published online May 26, 2023. doi: 10.12998/wjcc.v11.i15.3637
Peer-review started: February 5, 2023
First decision: March 14, 2023
Revised: April 3, 2023
Accepted: April 18, 2023
Article in press: April 18, 2023
Published online: May 26, 2023
Processing time: 109 Days and 9.2 Hours
After vaccination was mandated worldwide, various adverse effects associated with the coronavirus disease 2019 (COVID-19) vaccination, including shoulder pain, have been reported. Here, we report a case of new-onset shoulder pain after BNT162b2 (Comirnaty, Pfizer-BioNTech) mRNA vaccination.
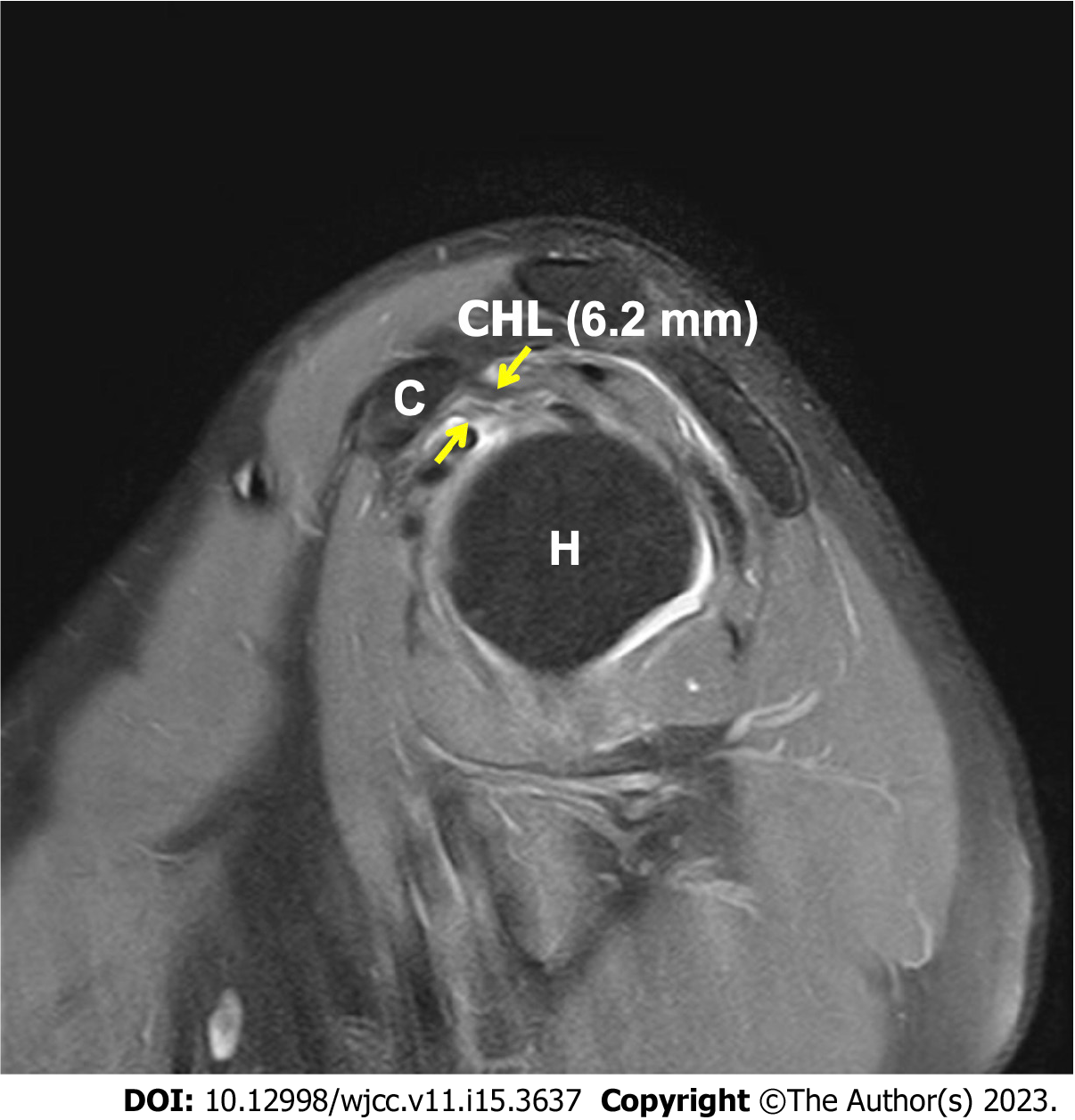
A 50-year-old man visited our rehabilitation center with left shoulder range of motion (ROM) limitation that had persisted for more than 5 mo. The history included no specific noteworthy events, except vaccination. The pain in the patient’s left deltoid muscle appeared 1 day after the second BNT162b2 vaccination and intensified to severe pain. The patient self-administered aspirin, with which the pain subsided immediately, whereas ROM limitation persisted. At the first visit, the patient complained of dull pain and ROM restriction of the left shoulder (flexion 130°, abduction 110°, and external rotation 40°). Among the diagnostic studies conducted for the evaluation of the shoulder, magnetic resonance imaging showed a thickened coracohumeral ligament. Nerve conduction studies and needle electromyography showed no electrodiagnostic abnormalities. The patient received comprehensive rehabilitation for 7 mo and had an overall improvement in pain and ROM of the left shoulder.
In this case of severe shoulder pain after COVID-19 vaccination that subsided immediately with aspirin treatment, the exact cause and mechanism of pain are unclear. However, the clinical symptoms and diagnostic workups in our report suggest the possibility that the COVID-19 vaccination triggered an immunochemical response that resulted in shoulder pathology.
Core Tip: In this case, the patient experienced severe pain immediately after coronavirus disease 2019 (COVID-19) vaccination and was diagnosed with a frozen shoulder. Pain resolved immediately after taking aspirin. The pain of frozen shoulder is associated with inflammatory mediators, such as cytokines. Although the pathogenetic mechanisms of frozen shoulder after COVID-19 vaccination are unknown, an association between some cytokines and frozen shoulder after COVID-19 vaccination likely exists.
- Citation: Jo HS, Kim HM, Han JY, Park HK. Atypical progress of frozen shoulder after COVID-19 vaccination: A case report. World J Clin Cases 2023; 11(15): 3637-3642
- URL: https://www.wjgnet.com/2307-8960/full/v11/i15/3637.htm
- DOI: https://dx.doi.org/10.12998/wjcc.v11.i15.3637
Vaccination to prevent the coronavirus disease (COVID-19) has been administered worldwide, and various adverse effects have been reported for the different types of COVID-19 vaccines, for which the most common injection site is the deltoid muscle. Post-vaccination side effects include pain, redness, itching, edema, allergic reactions, fever, local soreness, rash, and others[1] and most of these symptoms are usually mild and transitory, and disappear within 2 to 3 d after vaccination[2]. However, shoulder pathology, including frozen shoulder that is associated with chronic persistent symptoms, can occur after vaccination, including the COVID-19 vaccination[3]. The exact pathogenesis of frozen shoulder remains unclear. Frozen shoulder is a severe dysfunction that takes more than a year to resolve with treatment. In 30%-40% of patients, mild to moderate symptoms can persist at 2 to 3 years of follow-up, and many patients can experience symptoms for even longer than 6 years[4].
The treatment includes surgery or nonoperative treatments, such as physiotherapy and intraarticular or subacromial corticosteroid injections. However, the data on management strategies and outcomes vary[5]. Here, we report an atypical case of frozen shoulder that occurred after BNT162b2 (Comirnaty, Pfizer-BioNTech) mRNA vaccination that differed in terms of the clinical course.
A 51-year-old Korean man presented with stiffness and limitation of active and passive range of motion (ROM) in the non-dominant left shoulder for more than five months.
Approximately 5 mo earlier, the patient received the second BNT162b2 vaccination dose into the left deltoid muscle. The next day onward, the patient experienced left shoulder pain (VAS; Visual Analogue Scale, 7-8) and decreased range of motion. There were no adverse effects reported about people who vaccinated on the same day with the same lot number. Owing to persistent pain and impaired overhead activities, the patient visited another local hospital and received conservative treatment, including transcutaneous electrical nerve stimulation, superficial heat therapy, ultrasound as pain modalities and oral NSAIDs (Ibuprofen, three capsules per day, each containing 400 mg ibuprofen) for 1 mo. However, the left shoulder pain persisted and the ROM decreased further. Therefore, the patient self-administered aspirin 500 mg per day for 2 d without a doctor’s prescription following which the pain resolved immediately although the shoulder ROM limitation persisted. After 3 mo, the patient visited our department with stiffness and limitation of active and passive ROM in left shoulder.
The patient engaged in office work and had not experienced any significant shoulder pain, stiffness, and trauma prior to the administration of the COVID-19 vaccine.
The patient had no specific family history and no allergic reactions prior to the vaccination.
At the first visit, the ROM of shoulder was ascertained as shoulder flex 130, abduction 110, internal rotation 30, and external rotation 40. The strength at the shoulder was normal. The patient had a negative O’Brien test and a positive Hawkins test, without any local tenderness, swelling, sensory disturbance, and apparent trauma or shoulder pathology. There was neither atrophy around the deltoid or shoulder girdle nor any scapular winging.
All laboratory test results were within the reference values.
Standard anteroposterior and lateral radiographs of the affected shoulder were checked and found to be within normal limits. Magnetic resonance imaging (MRI) was conducted and showed no rotator cuff tear; however, coracoacromial ligament thickening (6.2 mm, whereas normal value < 4 mm; Figure 1) was noted.
For facilitating the differential diagnosis, an electrodiagnostic study was conducted and revealed results that were within normal limits.
The patient was accordingly diagnosed with frozen shoulder.
An intra-articular shoulder joint injection of saline and lidocaine (0.5% lidocaine) was administered once, but did not improve the ROM immediately nor after the 1-week follow-up. Moreover, the patient underwent a rehabilitation program that included ROM exercises and pain modalities such as transcutaneous electrical nerve stimulation, superficial heat therapy, ultrasound for improving shoulder ROM at least 2 times per week. The exercise types included finger to wall climbing exercise, both forward and sideways to improve forward flexion and abduction, respectively, until the symptoms had subsided. Pendulum exercises and anterior posterior capsular stretching were also undertaken.
After 5-mo rehabilitation, the shoulder ROM improved as follows: Shoulder flex 150, abduction 140, external rotation 65, and internal rotation 15, with no complaints of pain (Table 1). And there was no adverse event.
| Before rehabilitation | After rehabilitation | |
| Numerical rating scale | 7 | 3 |
| Range of motion | ||
| Flexion | 130° | 150° |
| Abduction | 110° | 110° |
| External rotation | 40° | 65° |
| MMT (Rt. U/E/Lt. U/E) | (5/5/5)/(4/5/5) | (5/5/5)/(4/5/5) |
In general, vaccines are frequently injected intramuscularly into the deltoid muscle, and the guidelines for vaccination, including the needle length and depth of injection, have been specified[6,7]. However, injection too high in the deltoid muscle can result in shoulder stiffness[1]. Another mechanism of injury that can occur after vaccination includes an antigen–antibody reaction that can occur in the muscle. Nonetheless, in general, the injury is transitory and progresses moderately. However, in some cases, antibodies from previous infections or vaccinations can lead to considerably longer-lasting inflammatory reactions[8,9] and can cause adhesive capsulitis of the shoulder[10]. Furthermore, vaccination in the shoulder can induce localized autoimmune reactions in the shoulder that can lead to a frozen shoulder[3].
Historically, vaccination is generally administered as an intramuscular injection into the deltoid muscle. However, the patient did not complain of severe pain at the injection site after the vaccination and, after vaccination, there was no erythema, swelling, burning sensation, and other complications.
Any painful condition of the shoulder joint after a vaccination was termed shoulder injury related to vaccine administration, by Atanasoff in 2010[8]. Shoulder injury after or related to vaccine administration have been defined as any pain and dysfunction of the shoulder that occur within 48 h of vaccination and lasts for more than 7 d. However, the presentation and prognosis of the COVID-19 vaccination-related shoulder dysfunction, including frozen shoulder, is unclear[3]. Age-related degenerative change can be considered in this case. However, the symptoms, including pain and ROM limitation, occurred immediately post-vaccination in this case, as reported in prior research.
A frozen shoulder is diagnosed based on the clinical presentation of shoulder stiffness and pain as well as the absence of radiological, laboratory, or other pathologies, as in the present case[1].
Some patients typically present with severe pain immediately after vaccination, especially when the needle enters the subacromial bursa or injures the peripheral nerves[11]. Furthermore, there may be an increase in fluid within the subacromial or subdeltoid bursae that is evident on imaging[12]. However, in the present case, the MRI and electrodiagnostic studies showed no bursitis and nerve injury.
In most cases of vaccination-related injury, patients suffer persistent symptoms, including pain and chronic ROM limitation[8]. Furthermore, the pain of the frozen shoulder may be nociceptive, as a result of peripheral abnormalities associated with capsular structures. In addition, inflammatory mediators can mediate the process of peripheral sensitization[13]. Several studies of frozen shoulder have revealed immune cells and inflammatory mediators, including cytokines such as interleukins, tumor necrosis factor, and immune mediators[14]. Moreover, cytokines such as the Alarmins high mobility group box 1 (HMGB1) are associated with pain in the frozen shoulder and peripheral nerve ingrowth[15]. In addition, cytokines such as HMGB1 are associated with the severe acute respiratory syndrome coronavirus 2 infections[16]. In the present case, the pain resolved with aspirin intake, which can suppress endogenous prostaglandin synthesis via inhibition of cyclooxygenase activity and its effects on the concentration of interleukins[17]. In addition, aspirin decreases the levels of cytokines such as HMGB1[18]. Accordingly, the pain in the present case may have improved immediately. Frozen shoulder after vaccination has been previously reported. But in this case, the progress was somewhat different from the general course including progress of pain.
We report the case of a patient who experienced severe shoulder pain after BNT162b2 vaccination and whose pain resolved immediately after taking aspirin. Physicians usually recommend injections, pain modalities, or physiotherapy for frozen shoulders. There are reports that the HMGB1 signaling pathway is associated with acute lung injury and acute respiratory distress syndrome in COVID-19 patients. However, studies of the association of cytokines, such as HMGB1, and frozen shoulder after COVID-19 vaccination have not been reported. Nonetheless, the exact cause of pain is unclear although the clinical symptoms and diagnostic workup in our case suggest the possibility of an association between cytokines, such as HMGB1, and shoulder pathology. Further studies are needed to understand the pathophysiology of shoulder pain after COVID-19 vaccination and to ascertain the relationship between cytokines, pain, frozen shoulder, and aspirin.
The authors would like to thank the patient and the patient’s family for agreeing to participate in this research.
Provenance and peer review: Unsolicited article; Externally peer reviewed.
Peer-review model: Single blind
Specialty type: Medicine, general and internal
Country/Territory of origin: South Korea
Peer-review report’s scientific quality classification
Grade A (Excellent): 0
Grade B (Very good): B
Grade C (Good): C
Grade D (Fair): 0
Grade E (Poor): 0
P-Reviewer: Su C, China; Trevisan A, Italy S-Editor: Liu JH L-Editor: A P-Editor: Zhang XD
| 1. | Saleh ZM, Faruqui S, Foad A. Onset of Frozen Shoulder Following Pneumococcal and Influenza Vaccinations. J Chiropr Med. 2015;14:285-289. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 14] [Cited by in RCA: 15] [Article Influence: 1.5] [Reference Citation Analysis (0)] |
| 2. | Chuaychoosakoon C, Parinyakhup W, Tanutit P, Maliwankul K, Klabklay P. Shoulder injury related to Sinovac COVID-19 vaccine: A case report. Ann Med Surg (Lond). 2021;68:102622. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 19] [Cited by in RCA: 32] [Article Influence: 8.0] [Reference Citation Analysis (0)] |
| 3. | Sahu D, Shetty G. Frozen shoulder after COVID-19 vaccination. JSES Int. 2022;6:682-685. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 2] [Cited by in RCA: 14] [Article Influence: 4.7] [Reference Citation Analysis (0)] |
| 4. | Hand C, Clipsham K, Rees JL, Carr AJ. Long-term outcome of frozen shoulder. J Shoulder Elbow Surg. 2008;17:231-236. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 291] [Cited by in RCA: 302] [Article Influence: 16.8] [Reference Citation Analysis (0)] |
| 5. | Challoumas D, Biddle M, McLean M, Millar NL. Comparison of Treatments for Frozen Shoulder: A Systematic Review and Meta-analysis. JAMA Netw Open. 2020;3:e2029581. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 165] [Cited by in RCA: 117] [Article Influence: 23.4] [Reference Citation Analysis (0)] |
| 6. | Greenberg RN, Gurtman A, Frenck RW, Strout C, Jansen KU, Trammel J, Scott DA, Emini EA, Gruber WC, Schmoele-Thoma B. Sequential administration of 13-valent pneumococcal conjugate vaccine and 23-valent pneumococcal polysaccharide vaccine in pneumococcal vaccine-naïve adults 60-64 years of age. Vaccine. 2014;32:2364-2374. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 114] [Cited by in RCA: 124] [Article Influence: 11.3] [Reference Citation Analysis (0)] |
| 7. | Barnes MG, Ledford C, Hogan K. A "needling" problem: shoulder injury related to vaccine administration. J Am Board Fam Med. 2012;25:919-922. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 35] [Cited by in RCA: 40] [Article Influence: 3.1] [Reference Citation Analysis (0)] |
| 8. | Atanasoff S, Ryan T, Lightfoot R, Johann-Liang R. Shoulder injury related to vaccine administration (SIRVA). Vaccine. 2010;28:8049-8052. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 82] [Cited by in RCA: 101] [Article Influence: 6.7] [Reference Citation Analysis (0)] |
| 9. | Cantarelli Rodrigues T, Hidalgo PF, Skaf AY, Serfaty A. Subacromial-subdeltoid bursitis following COVID-19 vaccination: a case of shoulder injury related to vaccine administration (SIRVA). Skeletal Radiol. 2021;50:2293-2297. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 29] [Cited by in RCA: 49] [Article Influence: 12.3] [Reference Citation Analysis (0)] |
| 10. | Thompson AR, Ensrud ER. Bilateral adhesive capsulitis following influenza vaccination: A case report. Clin Case Rep. 2020;8:2155-2157. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 7] [Cited by in RCA: 3] [Article Influence: 0.6] [Reference Citation Analysis (0)] |
| 11. | Massel DH, Haziza S, Rivera S, Mohile N, Subhawong TK, Hernandez VH. Septic Arthritis of the Shoulder After SARS-CoV-2 Pfizer Vaccination: A Case Report. JBJS Case Connect. 2021;11. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 12] [Cited by in RCA: 13] [Article Influence: 3.3] [Reference Citation Analysis (1)] |
| 12. | Salmon JH, Geoffroy M, Eschard JP, Ohl X. Bone erosion and subacromial bursitis caused by diphtheria-tetanus-poliomyelitis vaccine. Vaccine. 2015;33:6152-6155. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 21] [Cited by in RCA: 11] [Article Influence: 1.1] [Reference Citation Analysis (0)] |
| 13. | Struyf F, Meeus M. Current evidence on physical therapy in patients with adhesive capsulitis: what are we missing? Clin Rheumatol. 2014;33:593-600. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 39] [Cited by in RCA: 32] [Article Influence: 2.7] [Reference Citation Analysis (0)] |
| 14. | Kabbabe B, Ramkumar S, Richardson M. Cytogenetic analysis of the pathology of frozen shoulder. Int J Shoulder Surg. 2010;4:75-78. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 54] [Cited by in RCA: 71] [Article Influence: 5.1] [Reference Citation Analysis (0)] |
| 15. | de la Serna D, Navarro-Ledesma S, Alayón F, López E, Pruimboom L. A Comprehensive View of Frozen Shoulder: A Mystery Syndrome. Front Med (Lausanne). 2021;8:663703. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 7] [Cited by in RCA: 44] [Article Influence: 11.0] [Reference Citation Analysis (0)] |
| 16. | Al-Kuraishy HM, Al-Gareeb AI, Alkazmi L, Habotta OA, Batiha GE. High-mobility group box 1 (HMGB1) in COVID-19: extrapolation of dangerous liaisons. Inflammopharmacology. 2022;30:811-820. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 48] [Cited by in RCA: 54] [Article Influence: 18.0] [Reference Citation Analysis (0)] |
| 17. | Brox R, Hackstein H. Physiologically relevant aspirin concentrations trigger immunostimulatory cytokine production by human leukocytes. PLoS One. 2021;16:e0254606. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 2] [Cited by in RCA: 2] [Article Influence: 0.5] [Reference Citation Analysis (0)] |
| 18. | Zhu D, Zou H, Liu J, Wang J, Ma C, Yin J, Peng X, Li D, Yang Y, Ren Y, Zhang Z, Zhou P, Wang X, Cao Y, Xu X. Inhibition of HMGB1 Ameliorates the Maternal-Fetal Interface Destruction in Unexplained Recurrent Spontaneous Abortion by Suppressing Pyroptosis Activation. Front Immunol. 2021;12:782792. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 2] [Cited by in RCA: 65] [Article Influence: 21.7] [Reference Citation Analysis (0)] |