Published online Feb 6, 2022. doi: 10.12998/wjcc.v10.i4.1140
Peer-review started: May 14, 2021
First decision: June 2, 2021
Revised: June 28, 2021
Accepted: December 23, 2021
Article in press: December 23, 2021
Published online: February 6, 2022
Processing time: 254 Days and 21.5 Hours
Coronavirus disease 2019 (COVID-19) has caused a pandemic that affected all countries with nearly 270 million patients and 5 million deaths, as of as of December, 2021. The severe acute respiratory syndrome coronavirus 2 virus targets the receptor, angiotensin-converting enzyme 2, which is frequently found in human intestinal epithelial cells, bile duct epithelial cells, and liver cells, and all gastrointestinal system organs are affected by COVID-19 infection. The aim of this study is to review the gastrointestinal manifestations and liver damage of COVID-19 infection and investigate the severe COVID-19 infection risk in patients that have chronic gastrointestinal disease, along with current treatment guidelines. A literature search was conducted on electronic databases of PubMed, Scopus, and Cochran Library, consisting of COVID-19, liver injury, gastrointestinal system findings, and treatment. Liver and intestinal involvements are the most common manifestations. Diarrhea, anorexia, nausea/vomiting, abdominal pain are the most frequent symptoms seen in intestinal involvement. Mild hepatitis occurs with elevated levels of transaminases. Gastrointestinal involvement is associated with long hospital stay, severity of the disease, and intensive care unit necessity. Treatments and follow-up of patients with inflammatory bowel diseases, cirrhosis, hepatocellular carcinoma, or liver transplant have been negatively affected during the pandemic. Patients with cirrhosis, hepatocellular carcinoma, auto-immune diseases, or liver transplantation may have a greater risk for severe COVID-19. Diagnostic or therapeutic procedures should be restricted with specific conditions. Telemedicine should be used in non-urgent periodic patient follow up. COVID-19 treatment should not be delayed in patients at the risk group. COVID-19 vaccination should be prioritized in this group.
Core Tip: Coronavirus disease 2019 is a pandemic viral infection. The virus binds to the angiotensin-converting enzyme 2 receptor, therefore all tissues and systems are affected by the infection. This review highlighted pathogenesis, clinical and laboratory features of gastrointestinal manifestations and liver injury. Change of gut microbiota was also discussed in this study. Furthermore, risk situations, treatment, and the follow-up of chronic liver diseases, irritable bowel syndrome, and digestive system cancers during the pandemic period with recommendations were discussed.
- Citation: Ozkurt Z, Çınar Tanrıverdi E. COVID-19: Gastrointestinal manifestations, liver injury and recommendations. World J Clin Cases 2022; 10(4): 1140-1163
- URL: https://www.wjgnet.com/2307-8960/full/v10/i4/1140.htm
- DOI: https://dx.doi.org/10.12998/wjcc.v10.i4.1140
Coronavirus disease 2019 (COVID-19) has caused a pandemic that affected all countries with more than 238 million patients and nearly 5 million deaths, as of October, 2021[1]. The clinical outcome of COVID-19 infection is ranged from asymptomatic to death. Patients who are older, male, and have comorbidities such as diabetes mellitus, hypertension and cardiovascular diseases are regarded as the high-risk group for COVID-19 infection. Severe acute respiratory syndrome coronavirus 2 (SARS-CoV-2) is spread throughout the system by angiotensin-converting enzyme 2 (ACE2) receptors found in endothelial cells[2]. Thus, COVID-19 infection is not limited to the respiratory system and spreads to all tissue of the body, and causes multi-systemic diseases.
During COVID-19 infection, the gastrointestinal (GI) system is affected on varying degrees. Liver and intestinal involvements are the most common manifestations. Because of the SARS-CoV-2 virus found in both the oral mucosa and the intestinal tract endoscopy procedures have become risk procedures for nosocomial transmission from patients to healthcare professionals.
In another aspect, patients with chronic diseases such as chronic hepatitis, cirrhosis, irritable bowel syndrome, ulcerative colitis, and liver transplant have been affected by the pandemic transformation of hospitals. Treatment and control visits were canceled or postponed for an unknown period due to this extraordinary situation.
The aim of this study is to review the GI manifestations and liver damage of COVID-19 infection and investigate the severe COVID-19 infection risk in patients that have chronic GI disease, along with current treatment guidelines.
SARS-CoV-2 virus enters into the target cell and tissue via ACE2 binding receptor[2]. ACE2 receptors have been found and expressed in intestinal enterocyte cells. Gastric and intestinal epithelial surfaces cilia of a glandular cell include ACE2 mainly in the cytoplasm. It has been rarely found in esophageal epithelial surfaces. Thus, viral nucleocapsid proteins are found in the gastric, duodenal, rectal glandular epithelial cells, but not in the esophagus[3,4]. Zou et al[5] described that if tissue ACE2 ex
After receptor binding, S protein enters the host cell by host proteases such as mucosa-specific transmembrane serine protease type 2 and 4 (TMPRSS2 and TMPRSS4), trypsin, elastase, cathepsin L, cathepsin B, factor X, and furin[6]. Then, viral replication of SARS-CoV-2 occurs[2].
Zang et al[6] showed SARS-CoV-2 intestinal replication in human small intestinal enteroids. They found that TMPRSS2 and TMPRSS4 are important to virus entry into the host cell. Interestingly they showed also that when the virus is present in the intestinal lumen, it can be inactivated by colonic fluid, thus the virus has not been recovered from stool specimens.
In addition, the SARS-CoV-2 virus is also disrupting the normal intestinal flora, leading to GI symptoms, especially diarrhea[4].
During COVID-19 infection lung-derived CCR9+ CD4+ T cells increase and into the small intestine by increased CCL25[7]. Thus, intestinal inflammation occurs. Balance of the intestinal flora is disturbed. Interleukin (IL)-17A increases and it causes neut
Besides, ACE2 receptor is found in bile duct epithelial cell and liver. Therefore, virus replication occurs in the liver and bile ducts, and manifests as elevated liver enzymes, decrease albumin, and prolongation of prothrombin time.
The rates of GI symptoms are from 11% to 53%, and nearly half of the patients have at least one of the symptoms such as loss of appetite, nausea, vomiting, diarrhea, and abdominal pain[3]. GI manifestations rate was reported as 17.6% in a large series including 60 studies and 4243 patients[10]. In a meta-analysis that enrolled 47 studies and including 10890 patients, the most frequent GI manifestations reported as nausea/vomiting (7.8%), diarrhea (7.7%) and abdominal pain (2.7%)[11]. In a meta-analysis of 43 reports, including 18246 patients, the most frequent GI symptoms were diarrhea (11.5%), nausea, vomiting (6.3%), and abdominal pain (2.3%)[12]. Ferm et al[13] reported GI symptoms of 892 patients as diarrhea (19.8%), nausea (16.6%), vomiting (10.2%), loss of appetite (11.8%), abdominal pain (7.8%), and loss of taste (2.4%). At least one GI manifestation such as diarrhea, nausea, vomiting, or abdominal pain was found in patients with COVID-19 infection. The mean duration of GI symptoms was found to be 4 (3-7) d. In our series, which includes 430 cases, the prevalence of GI symptoms was 19.8%, and the most common GI symptoms were nausea/vomiting, abdominal pain, and diarrhea (10.4%, 6.1%, and 2.9%, respectively)[14]. The rates of GI manifestations in COVID-19 infection are shown in Table 1.
| Gastrointestinal manifestations | Frequency % |
| Gastrointestinal involvement | 11-79 |
| Anorexia | 34-67 |
| Diarrhea | 2-49.5 |
| Vomiting | 1-16 |
| Nausea | 1-16 |
| Abdominal pain | 2.7-9.2 |
| Liver injury | 15-53 |
GI symptoms frequently start at an early period of the disease and 1-2 d before respiratory symptoms. GI manifestations were also reported more frequently in severe cases (17.1%) than that in non-severe cases (11.8%)[4,10].
Hospitalization rates of the patients who had GI involvement were found five times higher than others. It has increased 8 times in patients with diarrhea, as compared to without GI symptoms[15].
This finding supports the theory of SARS-CoV-2 gastro-intestinal entry and infection via the ACE2 receptor[2].
ACE2 is frequently found in human small intestinal epithelial cells, and it is more strongly expressed in type II epithelial cells[5,16,17]. It is a key enzyme for the renin-angiotensin system that regulating intestinal inflammation and diarrhea[16,18]. Virus and/or pro-inflammatory cytokines may affect host cells. They may cause changing bowel flora. Thus watery diarrhea occurs. Lymphocytic infiltration in the esophageal epithelium, abundant infiltrating plasma cells and lymphocytes, and interstitial edema in the stomach, duodenum, and rectum lamina propria were found.
The most common symptoms of COVID-19 GI involvement are diarrhea and anorexia. Anorexia is present in most infectious diseases. The loss of taste and smell seen in COVID-19 infection contributes to anorexia. Patients become more anorectic and worse if cacosmia and/or cacousi are present.
Diarrhea is another common GI system symptom of COVID 19. At the onset of the pandemic, respiratory symptoms were dominant, and watery diarrhea was reported only 2%-10% in cases. However, in the summer season, diarrhea was observed more frequently up to 49.5%[19]. Diarrhea may be the first symptom in some patients and lasted for an average of 4-5 d, and even in some patients for 8-14 d[11].
Approximately 2%-19.5% of patients complained of diarrhea as the first symptom of the infection and in such cases, the virus genome could be isolated from stool and blood samples[20]. It is estimated that about half of admitted patients had at least one GI symptom, with these symptoms becoming more pronounced as the severity of the disease progressed. GI symptoms were found as high as 79.1%[16,19]. Patients with GI symptoms were also found to have a longer symptom time from onset to admission[4].
Viral gastroenteritis is characterized by non-bloody, watery diarrhea with abdo
A recent case report also documented SARS-CoV-2 GI infection-causing hem
SARS-CoV-2 invades the GI system and excretes in the stool. SARS-CoV-2 nucleic acid was detected in the stool of up to nearly 54% of patients[22,23]. The results of these studies indicate that the GI tract is a site where SARS-CoV-2 invades and it is released[23].
The incidence of GI symptoms in patients with COVID-19 in Wuhan was signi
SARS-CoV-2 virus RNA was present in stool samples 35.7%-54.5% in the patients with diarrhea and even in those who have not diarrhea. But this positivity found was not correlated with the disease symptoms and the presence of diarrhea[24]. The viral load in stool was reached to a very high levels (mean viral load 4.7 log10 copies per mL) in patients with diarrhea[10]. Cheung et al[10] also found SARS-CoV-2 positivity 48.1% at the same time both respiratory secretions and stool samples, and 70.3% in only stool samples. Meanwhile, the rate of viral RNA positivity in stool sample were found longer than respiratory sample[22,25]. The virus can be found positive in the stool even after it has been cleared from the respiratory system. Gupta et al[22] reported that 53.9% of stool samples were RNA positive, and fecal shedding time ranged from 1 d to 33 d, even after negative respiratory results. Thus the rate of stool viral RNA positive and respiratory secretion negative was found as 70% in patients with COVID-19. Fecal virus shedding may continue up to 47 d, and 13 d after from respiratory system becomes negative[22]. Interestingly, it was found that steroid usage causes longer time viral shedding from stool[10].
Fecal viral shedding causes environmental contamination and mechanical vectors may be contributing to virus distribution[26]. Although SARS-CoV-2 is an envelope virus, it was found viable 14 d at 4 °C, and 2 d at 20 °C in sewage[25,27]. Additionally, the virus can be stable on surface and viable for up to 7 d on stainless steel, plastic, cardboard[28].
These findings have shown that SARS-CoV-2 may be transmitted by direct fecal-oral route or indirect contamination of hands through the contaminated household environmental surface[29].
There is no specific treatment for viral diarrhea. Hydration is the main treatment of diarrhea and vomiting. Tian et al[30] reported that The National Health Commission of the People's Republic of China showed that symptomatic treatment and adequate hydration were essential to prevent electrolyte imbalance. Some medications such as dioctahedral montmorillonite powder and loperamide can be used for diarrhea. Probiotics may be given for intestinal dysbiosis and antispasmodics may be given for abdominal pain[30]. Dioctahedral montmorillonite powder and loperamide may be used to treated diarrhea. Antispasmodics may be given for abdominal pain. Besides, probiotics may be useful for gut dysbiosis.
Enteral nutrition and digestive tract function are important for therapy. Enteral nutrition is needed for providing energy, restoring and maintaining the normal physiological function of the GI tract, and microecology with mucosal immunity[16]. Parenteral nutrition is used in only patients who have been intubated, have GI system lesions, and intolerant to enteral nutrition. Enteral nutrition should be reversed immediately.
SARS-CoV-2 virus in the stool may be transmitted through contact with hands. Additionally, feces particles widespread air and may be inhaled by other persons. Therefore, when cleaning digestive tract secretions of COVID-19, patients should be attentive and all protective equipment should be worn, hand hygiene should be provided, and hospital or house environmental surfaces should be also properly disinfected[16].
The number of ACE2 receptors is at high levels in patients with inflammatory bowel diseases (IBD) such as ulcerative colitis and Crohn's disease. Burgueño et al[31] found that ACE2 and TMPRSS2 expression were the highest in ileum and colon respectively. The drugs given to the patients lead to immunosuppression, and they predispose them to SARS-CoV-2. Both their immunosuppressive therapy and increased ACE2 receptors make this group of patients susceptible to COVID-19 infection. Thus, it is estimated that patients who have been given biological agents are at-risk group. But clinical experience showed that the risk of the patient IBD diseases is similar to that of other persons[11]. IBD patients with comorbidity, and/or who are elderly (> 70), use prednisolone per day (> 20 mg), biologic agents and have used steroid in the last 6 wk, who have moderately or severely active diseases, short gut syndrome, and who need parenteral nutrition are at the high-risk group.
During the COVID-19 pandemic, patients with IBD can be treated. The International Organization for the Study of Inflammatory Bowel Diseases, Crohn's and Colitis UK, and Crohn's and Colitis Foundation recommended ongoing medical treatment for IBD[32]. Thiopurines therapy has increased the risk of viral infection, but mesalamine has not such an effect. Steroids can be used in the pandemic period, because steroids are not damaged clinical course of COVID-19. Vedolizumab can be continued. Uste
Patients should be questioned or screened in terms of SARS-CoV-2 infection before beginning the immunosuppressive therapy.
Elective endoscopy and surgery of IBD patients should be postponed. Indications of the endoscopy of IBD patients are given as follows: (1) Confirmation of a new diagnosis in moderate to severe IBD; (2) Evaluation of severe acute flare up of ulcerative colitis; (3) Evaluation of partial or subacute bowel obstruction; and (4) Developing cholangitis in patients with IBS who has primary sclerosing cholangitis[4].
Screening for COVID-19 infection is recommended if patients need endoscopy or surgery.
Dysbiosis may lead to epithelial inflammation and increase ACE2 expression by changing dietary amino acid homeostasis. ACE2 regulates the intestinal uptake of tryptophan. Tryptophan increases antimicrobial peptides (AMPs) through the mammalian Target of Rapamycin (mTOR) pathway. AMPs change the composition of gut microbiota. Butyrate-producing bacteria stimulate the immune system cells (macrophages, dendritic cells, and T cells), IFN-I and anti-inflammatory ILs[33]. SARS-CoV-2 binds to ACE2 and inhibits balance in microflora. In the intestinal system, ACE2 expression has regulated by gut bacteria. Bacteroides species downregulate ACE2 expression in the murine gut[33,34]. Proinflammatory gut microbiome increases ACE2 expression and may cause favorable conditions in the gut epithelium for SARS-CoV-2 infection[8,33]. SARS-CoV-2 may prevent nutrient absorption and disrupt intestinal homeostasis by binding to ACE2. Diarrhea in COVID-19 infection may be explained by ACE2-dependent regulation of the gut microbiota. Alterations in the gut and/or lung microbiome by ACE2 might also be associated with cardiopulmonary pathology[35]. Downregulation of ACE2 decreases AMPs secretion, and causes increased pathogen survival and gut dysbiosis.
Zuo et al[36] investigated changing of gut microbiota of patients with COVID-19 during hospitalization. They found the depletion of beneficial commensals and increasing opportunistic pathogens. Gut dysbiosis continued after viral clearance from the respiratory tract. In this study, Coprobacillus, Clostridium ramosum, and Clostridium hathewayi were found to correlate with the disease severity. Opposite Faecalibacterium prausnitzii (an anti-inflammatory bacterium) was found inverse correlated with severe outcome. Bacteroides species down-regulate ACE2 expression in murine guts, and correlated inversely with viral load in fecal samples from patients. Fecal microbiota alterations were found to be associated with both fecal viral load and the severity of the disease[36].
Yeoh et al[37] found that gut microbiome was significantly altered in patients with COVID-19 compared with non-COVID-19 individuals whether patients had been given medication or not. In stool samples, Faecalibacterium prausnitzii, Eubacterium rectale, and bifidobacteria remained low at 30 d after disease improving. These changes were found to correlate with disease severity and elevated cytokines and other blood markers. Therefore, they suggest that the gut microbiome has played important role in the severity of COVID-19 infection by modulating host immune responses. Additionally, dysbiosis after improving the disease may contribute to persistent symptoms in long COVID-19[37].
Prolonged gut microbiome dysbiosis in COVID-19 is associated with fecal virus shedding and disease severity. These data provide a new therapeutic target and modulation of the gut microbiota may be useful to supportive therapy of COVID-19.
Additionally, some drugs used in COVID-19 treatment such as chloroquine and corticosteroids have been shown to interact with gut microbiome[2].
The treatment strategies improving microbiota may be useful to decrease disease severity. Unless secondary bacterial pathogens present, antibiotic usage should be avoided in COVID-19 viral pneumonia. Microbiota and host cytokine pathway interactions are best understood and should be considered as developing new treatment approaches[38].
The microbiome also affects the immune response to the vaccine. Sufficient antibody levels after vaccine would not occur if the human has dysbiosis[39]. Dietary changes, giving probiotics and prebiotics, and avoiding antibiotic usage are mandatory to improve microbiota. Probiotics may improve diarrhea by intestinal micro ecological balance and prevent secondary bacterial infections[16]. Probiotics may have potential use for reducing symptoms of upper respiratory tract infections in overweight and obese people, and older age[40].
Although, SARS-CoV-2 is a respiratory virus, liver involvement is frequently seen in the COVID-19 infection. SARS-CoV-2 enters into the cell by binding ACE2 receptors. ACE2 receptors are found in many systems of the body such as lungs, liver, heart, kidney, and blood vessels[36,41]. There is more ACE2 receptor expression in bile duct epithelial cells (cholangiocytes 57.7%) markedly higher than in liver cells (hepatocytes 2.6%)[42]. Overexpression of ACE2 receptors in cholangiocytes causes liver injury. Liver damage probably occurs secondary to bile duct cell injury[4].
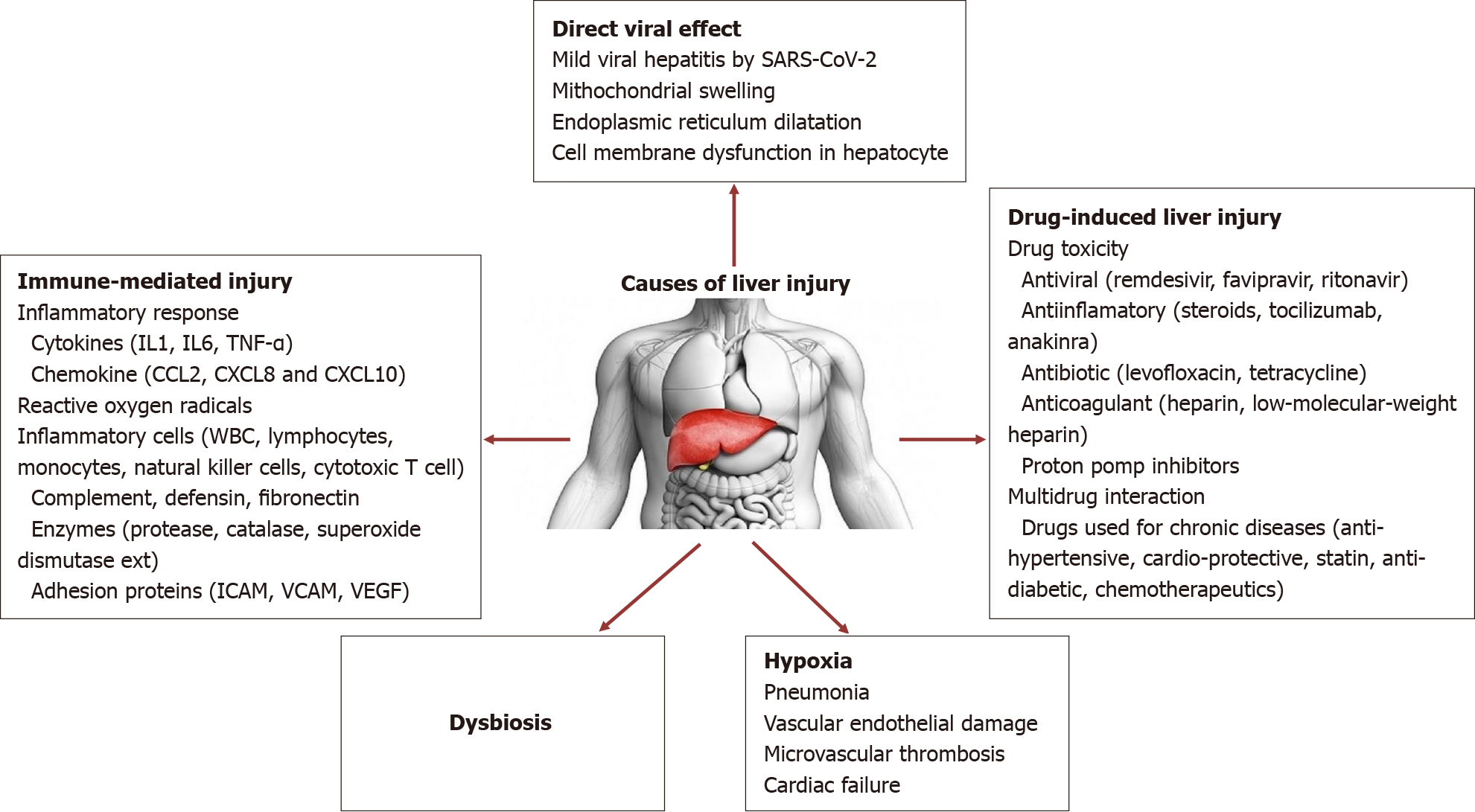
Cholestatic hepatitis is not the usual liver involvement pattern in COVID-19 infection[41,42]. Hepatic ACE2 receptor expression was highly elevated in females. This may explain the better clinical outcome of COVID-19 infection in females[41-45]. Multifactorial causes lead to liver damage during COVID-19 infection, such as direct virus cytopathic effect, inflammation, intrahepatic immune activation, microvascular thrombosis, hepatic congestion, disruption of the gut liver axis, drug toxicity, multidrug interactions[46-48]. Causes of liver injury are summarized in Figure 1.
Liver injury may occur by direct virus cytopathic effect by lysis or by inducing apoptosis [49-53]. The virus-specific protein 7a has been induced a caspase-dependent apoptosis pathway that presence in cell lines of different organs such as lung, kidney, and liver. The virus was detected by the low viral load in liver tissue[54]. Moreover, it was showed that the virus can be replicated in hepatocytes, and spike proteins found cytoplasm[55].
These findings suggest that SARS-CoV-2 could directly affect liver tissue and may cause cytopathic effect in hepatocytes[41,55]. Wang et al[56] showed that SARS-CoV-2 caused mitochondrial swelling, endoplasmic reticulum dilatation, glycogen granule decrease, and cell membrane dysfunction in hepatocytes. Massive hepatic apoptosis and some binuclear hepatocytes were reported in the same study. Both ultrastructural and histological evidence indicated a typical lesion of viral infection. Immunohistochemical results showed scarce CD4+ and CD8+ lymphocytes. No obvious eosinophil infiltration, cholestasis, fibrin deposition, granuloma, massive central necrosis, or interface hepatitis were observed[56].
Secondly, immune-mediated liver injury, source from pro-inflammatory cytokines (IL-1, IL-6, tumor necrosis factor), chemokine, and inflammatory cells produced against the SARS-CoV-2, are another important causes of liver damage[3,17,55,57]. Cytokine storm and mass syndrome also create liver injury. Elevated levels of all inflammatory and coagulopathy markers such as IL-2, IL6, chemokines, CRP, ferritin, D-dimer, and lactate dehydrogenase (LDH) correlated the severity of disease and poor outcome[3,57].
Viral-induced cytotoxic T cells (CD8) are also another important pathogenesis for viral infections[3,58]. Coagulation dysfunction and endothelial damage are found to be the main cause of liver injury in COVID-19 infection. Lagana et al[59] describe mitochondrial swelling, endoplasmic reticulum dilatation, and cell membrane dysfunction in liver biopsy samples of 40 COVID-19 patient autopsies. They found viral RNA and viral replication in hepatocytes. Polymerase chain reaction (PCR) positivity was found in 55% of patients in liver tissue[59]. PCR positivity and viral load were found correlated with histological results or liver enzymes. Mild and moderate micro-vesicular steatosis (75%), mild lobular necroinflammation (mild acute hepatitis) (50%) and portal inflammation (50%), and sinusoidal microthrombi (15%) were reported as the most common findings[59]. Mild lobular and portal activity showed in post-mortem liver autopsy specimen examinations[60].
Water degeneration and infiltrates (neutrophils, Kupffer cells and plasmocytes) were detected in lobules, sinusoidal, and portal areas[3,61,62]. Histopathological examination of 48 COVID-19 positive patients, acute vascular changes such as terminal vessel dilations, thrombosis and luminal ectasia, and chronic changes manifest fibrous thickening of the vascular wall observed in the portal and sinusoidal vessels were reported from post-mortem examination[59,62].
Intranuclear or intracytoplasmic viral inclusions were seen in patients with COVID-19 infection. Bile duct damage and liver failure signs were not seen in liver samples of severe COVID-19 patients[63].
Histopathological changes may be occurred due to direct viral effect, hypoxia or drugs.
Drugs such as antivirals, anti-inflammatory drugs, anticoagulants, antibiotics, and which are used underlying chronic diseases that are used during the COVID-19 infection contribute to liver injury[41,55]. Multi-drug usage and their interactions have also caused hepatotoxicity.
Another important cause of liver involvement is hypoxia that is a result of pneumonia. Even under oxygen therapy, hypoxic liver injury may occur.
Gut vascular barrier and dysbiosis (microbiota alterations) may be other causes contributing to liver damage due to indirect effect of toxic compound of opportunistic microorganisms.
The liver injury might develop more frequently in patients with chronic liver disease (CLD). CLD was found as a risk factor for prolonged hospitalization, and fatal course of the COVID-19[3].
In the COVID-19 infection, except CLD, usually mild hepatitis has been seen[3]. Pathologic liver function test results have been found to increase in nearly half of the patients with COVID-19[3,41]. Liver injury is also seen particularly in severe cases[4,63].
Hepatic involvement usually manifests elevated liver enzymes, sometimes decreasing albumin and rarely increasing bilirubin levels. Elevated levels of aspartate transaminase (AST), alanine transaminase (ALT), alkaline phosphatase (ALP), gamma-glutamyl transferase (GGT), LDH, hyperbilirubinemia, prolonged prothrombin time, and hypoalbuminemia may be found as laboratory findings of liver injury. Alteration of liver function tests may be described as parenchymal, cholestatic, and mixed type. ALT and/or AST 3-fold increase in the values above the upper limit of normal (ULN) in parenchymal form; ALP or GGT values 2-fold higher according to the ULN in cholestatic, and with both parenchymal and cholestatic pathologies present in the mixed type. The rates of parenchymal, cholestatic, and mixed form liver injuries were reported as 75%, 29.2%, and 43.4%, respectively in COVID-19 infection. Most of all were found the hepatocellular pattern, and ALT-AST levels rarely increase > 5 × ULN[48]. In a meta-analysis investigating 47 studies and including 10890 patients, elevation of ALT and AST were found as 15.0% and 20% in countries other than China[11].
The rates and ranges of ALT and AST elevations were 2.5%-50.0% to 2.5%-61.1% respectively[55]. In a case series including 1100 patients with COVID-19 infection, AST and ALT levels were (56% and 28%) higher in a severe case than that of mild-moderate ones (18% and 29%)[42] AST was found correlated with mortality[56]. In another study, AST/ALT, LDH, and bilirubin were found to increase by 25%, 20%, and 3% respectively. But, ALP was reported as normal in nearly all patients in the same study. Bilirubin and ALP levels did not markedly increase in COVID-19. Elevation rates of bilirubin were reported between 0%-35.3%[55]. Elevated total bilirubin was found in 16.7% COVID-19 patients from China[11]. The peak of increased bilirubin was reported at 5 d (4 to 12) after discharge. Hyperbilirubinemia and elevated ALT, ALP, and GGT levels were especially found in male patients, but hypoalbuminemia was frequently seen in males[3].
Although ALP and GGT tests usually normal values, GGT elevation found 37.6% of patients with non-alcoholic fatty liver disease (NAFLD)[55,64]. Despite elevated GGT level was reported in 41% of patients in another study, GGT usually increase in the severe patient[41,55,64]. The GGT elevation develops from drug toxicity more than obstruction and usually is not associated with ALP elevation[55]. Although GGT is a cholangiocyte injury marker, its levels did not increase in all COVID-19 patients. It was only found high in severe patients.
Hypoalbuminemia was found correlated with the disease severity and fatal course[13,41,65,66].
AST levels usually increase more than AST in liver diseases and other viral infections, but AST levels higher ATL in COVID-19 infection[63]. Elevated AST could reflect zone 3 injuries of the hepatocyte cells. Zone 3 is the biggest reservoir of AST and susceptible to hypoxia. AST is found in the cytosol and the mitochondria. AST release increases due to the mitochondrial damage by the virus. The relationship between SARS-CoV-2 and mitochondrial proteins may be causing AST elevation. AST also produced from skeletal muscle, cardiac, kidney and lung tissue[67]. Thus, an elevated AST reflects not only the liver but also a multi-organ damage. Severe pneumonia causes liver injury through a hypoxic situation. Cytokine storm and endothelial activation cause multiorgan and liver damage in COVID-19 infection.
It was found that AST/ALT ratio > 1 provides predict mortality, severe pneumonia and intensive care unit (ICU) required in patients with ICU and severe pneumonia[68]. AST/ALT ratio, total bilirubin, and ALT/ALP ratio may estimate outcomes in cirrhotic COVID-19 patients[69].
The incidence of liver injury ranges from 15% to 53%[4]. The rates alteration of liver function test having been reported in 19%-76% of COVID-19 cases[48]. In a meta-analysis, hepatic injury developed in 23.70% total, 31.66% in non-severe and 44.63% in severe patients with COVID-19 infection[70]. ALT and AST elevations were reported as 39.58% and 49.68% in severe and 24.15% and 19.40% in non-severe disease. AST elevation was more than ALT in the severe COVID disease[70]. ALP and GGT ele
In another meta-analysis screening 15407 COVID-19 patients, liver injury was reported 23.1% at an early period, and 24.4% at a course of the disease[71]. Liver injury was found 48.5% at the first two weeks of admission of hospitalization, and it was reported peaked 10 d (7-12) after being discharged[3,72]. Liver injury has been seen 26.7% in patients with severe pneumonia[3]. Elevation of ALP levels is rare and it is reported only in patients with multi-organ and liver failure or death from COVID-19 infection[73].
In meta-analysis, elevated ALT, AST, and bilirubin levels were found correlated with the severity of the disease[74]. Liver injury is associated with 9-fold greater risk of severe disease, an increased intensive care need, intubation, and fatality.
Liver damage presence at admission is suggested as an independent prognostic factor for COVID-19[75]. It was reported that patients with liver damage had a higher mortality rate (28.9% vs 9.0%), male gender (65.1% vs 40.8%), and systemic inflammatory response syndrome (53.5% vs 41.3%). Grade-2 liver damage was reported more in males than females. Grade of liver damage was also reported as a predictor of death (hazard ratio: 1.377). The proportion of liver injury of ICU patients (61.5%) was higher than non-ICU patients (25.0%)[75]. Alteration of liver functions rates was reported between in 58.06% to 78% fatal cases[13,41,76].
Liver injury was found correlated with prolonged hospitalization, the severity of the disease, GGT, ferritin, lower albumin, and CD4+ T cells and B lymphocyte[56,77]. Serum albumin levels were also significantly lower in patients who died due to the infection. Increased CRP, procalcitonin, IL-6, and ferritin with decreased albumin and platelet have been correlated with liver injury[42,78]. Decreased lymphocyte count and male gender was also found important for liver injury and poor outcome. CT imaging may be a predictor of liver damage and predict a severe outcome. Acute liver failure due to COVID-19 infection has been rare[78,79]. Thus SARS-CoV-2 should not be considered as a hepatotropic virus.
Multiorgan failure and other systemic complications may be developed from a secondary nosocomial infection acquired from the ICU. In our series, we found albumin and AST levels to be correlated with the severity of the disease, presence of pneumonia, intensive care requirement, and prolonged hospitalization times. Intensive care needs was in 9.6% of patients with liver damage[14].
Drug usage is another cause of elevation of liver function tests. In a meta-analysis that included 20874 COVID-19 patients, the drug toxicity rate was reported as 25.4%[71].
Antivirals (favipiravir, remdesivir, lopinavir/ritonavir, chloroquine, oseltamivir, and ribavirin) drugs used anti-inflammatory effect (tocilizumab), antipyretics (acetaminophen), and many other drugs used during the treatment during COVID-19 infection may lead to hepatotoxicity[3]. The hepatotoxic effect has developed in severe patients and/or in the presence CLD[3,63,80,81].
The hepatotoxic drug should be excluded in patients with liver injury. Because most drugs used treatment of COVID-19 (e.g., oseltamivir, lopinavir/ritonavir, and chloroquines) are metabolized in the liver.
In a study, the drugs (antivirals, antibiotics, antipyretics) used the previous hospitalization were found unrelated to liver injury[41]. GGT and bilirubin elevation are usually seen in the usage of antiviral against SARS-CoV-2.
Lopinavir/ritonavir, an antiretroviral protease inhibitor, can cause transient and usually mild elevations in liver enzymes. Lopinavir plasma levels increase in advanced liver diseases, therefore it should be carefully used. The liver injury developed in 57.8% of patients given lopinavir or ritonavir. Most hepatotoxicity effect developed in COVID-19 patients giver lopinavir/ritonavir or ribavirin[82]. A restart of the drug is not recommended[11]. Nearly 2.5 fold increase of liver enzyme reported in patients given remdesivir[83].
Favipiravir may cause a hepatotoxic effect and there are no data available on favipiravir usage in patients with CLD[84].
Azithromycin-induced liver injury, that hepatocellular type, was rare and has been developed 1-3 wk after initiation. It was spontaneously recovered[41,85].
Chloroquine rarely causes hepatotoxicity possibly due to hypersensitivity. Two acute liver failures were reported by hydroxychloroquine[41]. It should be used carefully in patients with CLD because hydroxychloroquine can concentrate in the liver tissue[11]. Although dose adjustments are not needed in patients with hepatic failure, it should be carefully used because of both cardiac and hepatic adverse effect[41,85,86].
In a randomized-controlled double-blind study, remdesivir found responsible hepatotoxicity in 23% of patients[87]. Serum bilirubin and aminotransferases had been elevated in remdesivir group[56,87,88]. Liver injury and encephalopathy were reported by remdesivir. It was reported that acetyl cysteine had successfully improved a patient's encephalopathy and liver failure due to remdesivir[3,88]. Remdesivir has not experienced liver cirrhosis. Increased transaminase levels have been reported in up to 22.6% of the patients[87].
Prednisolone is one of the most frequent drugs used in severe COVID-19 patients for anti-inflammatory effect. But if it is used for a long time at a high dose, it may cause hepatitis B virus (HBV) reactivation and spontaneous bacterial peritonitis in patients with decompensated liver cirrhosis[41].
Another anti-inflammatory drug tocilizumab (an IL-6 inhibitor) should not be used in decompensated cirrhosis[41]. Tocilizumab may cause mild serum elevations of aminotransferase and bilirubin levels, which are usually short and asymptomatic periods[41]. Tocilizumab may cause reactivation of HBV, in the patient given HBV should be screened and antiviral therapy should be given according to the guidelines[41,89].
In patients receiving lopinavir/ritonavir, remdesivir, or tocilizumab, liver functions should be monitored. Usage of these drugs is not contraindicated in liver injury. But in patients who have AST or ALT levels > 5 × ULN, these drugs may be excluded[88].
ACE inhibitors and ACE2 receptor blockers usage was not found correlated with the disease severity[3]. Other anti-hypertensive drugs and ACE-related drugs were found not different with respect to the disease outcome. Antibiotics, nonsteroidal anti-inflammatory drugs, ribavirin, or interferon were not found associated with liver functions. But lopinavir and ritonavir may cause 4.4-5-fold increase by dose-depended in liver function tests[3].
Cytokine release syndrome in severe COVID-19; IL-6 receptor antagonist tocili
Ivermectin may cause a minor, self-limiting liver enzyme elevation. Dose adju
Most liver injuries in COVID-19 are mild and spontaneously recover and it requires no special treatment. If liver injury persists, chronic underlying diseases such as hepatitis B and C should be considered[90]. Although there was no evidence of hepatitis reactivation or decompensation, patients with underlying chronic hepatitis or other CLDs should be closely monitored[4]. Drug interactions and drug-induced hepatotoxicity should be kept in mind. Multidrug interaction must be regularly monitored[4]. American Association for the Study of Liver Diseases (AASLD) recommends that unnecessary imaging should be avoided[90].
Because of its anti-inflammatory and immunomodulation activity, ursodeoxycholic acid may be added treatment especially in patients with liver injury. Recommended dose is 13-15 mg/kg per day[91]. Further detailed analyses are required. European Association for the Study of the Liver (EASL) and APASL recommendations related to hepatic care in the COVID-19 pandemic have been published[48,92]. The use of diagnostic procedures, supervision of patients with CLDs, and liver transplantation have been described[48].
COVID-19 infection may lead to liver damage by several routes in patients with existing CDL. Additional hepatic injury induced by SARS-CoV-2 might lead to hepatic failure in patients with compromised hepatic reserves[48,68]. Immunosuppressive effect of drugs or COVID-19 may lead to viral reactivation in patients with chronic viral hepatitis[48,68]. Drugs used for infection such as antivirals, anti-inflammatory, antipyretic, and antibiotics may lead to hepatotoxicity.
Although it seems that patients with CLD are not at greater risk for acquiring the infection, those with cirrhosis, hepatocellular carcinoma (HCC), NAFLD, autoimmune liver diseases or liver transplant may have a greater risk for developing liver injury and severe COVID-19[93]. Disease severity and liver damage develop depending on CLD, presence of advanced fibrosis or cirrhosis[47]. Similarly, other infections in the liver, hepatocytes are probably more affected in patients with underlying CLD.
Kim et al[94] reported that 867 patients with CLDs acute liver disease (ALD), decompensated cirrhosis, and HCC have higher mortality during COVID-19 infection.
The severity of COVID-19 infection, male gender, and low liver CT density were found causative factors that are strongly related to liver injury (odds ratios: 6.543, 3.387, 2.936, respectively)[4,95].
It is estimated that pre-existing cirrhosis is a risk factor for higher severity and mortality in COVID-19[48,96].
Patients with cirrhosis have an increased risk for both decompensation and development acute or chronic liver failure during COVID-19 infection[55]. Cirrhosis-associated immune dysfunction may cause severe disease course[41,48]. Increased Child-Pugh class was correlated with severe course. But whether the increase of complications is related to cirrhosis such as liver failure, hepatic encephalopathy, and upper GI bleeding during COVID-19 has not been known.
Acute or chronic liver failure may develop in patients with cirrhosis due to ACE2 overexpression in bile duct cells and inflammatory response. The increased inflammatory response may cause liver failure and death in cirrhotic patients[55]. These patients have a higher bacterial infection risk.
An international multicenter study investigated the role of pre-existing liver disease in COVID-19. A total of 745 patients from the United Kingdom hospital network with COVID-19 (386 patients with cirrhosis and 359 without cirrhosis), enrolled in this study. Mortality was found very high (32%) in cirrhotic patients and low (8%) in non-cirrhotic cases. In addition, mortality and other prognostic markers were associated with the degree of liver decompensation, and mortality was found as 19% in Child-Pugh class A, 35% in B, and 51% in C. Child-Pugh class A, B, C found also correlated with ICU (40%, 62%, 79%) and invasive ventilation requirement (52%, 74%, 90%)[97]. The main cause of death was reported as respiratory failure (71%). Acute liver failure had developed in 46% of patients with cirrhosis, and 21% of these patients had no respiratory symptoms. Age, liver disease stage, and alcohol-induced liver disease (ALD) were found to be risk factors for mortality in COVID-19[97]. It was reported that 5 of 21 (23%) pre-existing cirrhotic patients died during COVID-19 infection[94]. Thus cirrhotic patients have increased the risk of COVID-19, higher risk for severe disease, and increased risk for hepatic decompensation[98].
Regular screenings and monitoring procedures for patients with compensated cirrhosis should be postponed. In patients who have decompensated cirrhosis, particular measures should be carried out for those patients with cirrhosis admitted to non-COVID-19 hospital[48,99]. Patients with cirrhosis should receive the best standard of care according to the guideline. Patients with cirrhosis who developed SARS-CoV-2 infection are at high risk of hepatic decompensation, severe COVID-19 and death. To prevent decompensation and decrease the risk of hospitalization, guidelines on prophylaxis of spontaneous bacterial peritonitis, GI hemorrhage, and hepatic encephalopathy should be closely followed. Particular effort should be made to patients with cirrhosis who are admitted, and these patients manage in a designated non-COVID-19 ward. Patients with new or worsening hepatic decompensation or acute-on-chronic liver should be tested for SARS-CoV-2 even in the absence of respiratory symptoms. Early admission should be considered for patients with cirrhosis who developed COVID-19 infection. Vasoconstrictor therapy should be given with a considerable caution among critically ill patients with cirrhosis and COVID-19. Because they may lead to pulmonary pressure and decrease cardiac output[48]. All patients should receive vaccination for COVID-19, Streptococcus pneumonia, and influenza.
There was no emergent situation for chronic viral hepatitis at COVID-19 pandemic. However, despite both studies demonstrating associations between severity of liver disease and poor outcome, it remains unknown whether the presence of chronic viral hepatitis influences prognosis.
In a case series including 1099 patients with COVID-19, the rate of chronic HBV (CHB) was found to be 2.1%. Only one patient with CHB had severe disease, thus it was considered CHB has not a negative effect on the outcome of COVID-19[55].
Drugs of the patients with chronic HBV and chronic hepatitis C virus (HCV) should be continued if they have been receiving treatment. To follow, local laboratory test results should be sending and telemedicine should be applied. Prescriptions should be sent to the patients by mail or telephone to minimize disease transmission[48].
Interferon-alpha usage has an unknown impact, therefore, alternative agents should be used for patients with HBV during the COVID-19 pandemic[48].
In chronic HBV and HCV patients with COVID-19 infection, hepatitis treatment should be postponed until recovery. Patients with both COVID-19 and HBV flare-up, should be made on a case basis. IL antagonists such as tocilizumab and baricitinib used to mass syndrome might cause reactivation of HBV. In such a case antiviral prophylaxis should be given to prevent reactivation of HBV[4,48].
Patients with non-alcoholic hepatosteastosis (NASH) have elevated liver enzymes particularly ALT. Obesity usually co-exists in patients with NASH. Thus they may be at more risk for severe infection. Elevation of liver enzymes and longer viral clearance time were reported in patients with NASH[41,64]. Other underlying diseases source by chronic inflammation such as diabetes and chronic cardiac disease that is of high risk for the severe and fatal course of COVID-19 infection, may be also found in these patients[41,48]. NASH was found to be a high risk for severity of COVID-19 even in young patients who have not underlying diseases[100].
It was estimated that the balance between inflammation-promoting and inflammation-suppressing macrophage (M1 and M2) is insufficient in NADFL patients, it may cause the progression of COVID-19[41,101].
In patients with CLD and/or NASH, ACE2 expression has increased, and cytokine production has been exaggerated associated with COVID-19. During the COVID-19 infection, it was shown that NASH patients have a high risk for complication progre
In other studies, it was reported that NASH was not associated with severe disease[48,103,104]. Further research is needed to understand the impact of COVID-19 in NASH.
During the COVID-19 pandemic, patients should be aware that a sedentary lifestyle has a negative effect on their metabolic conditions and contributes to increasing NASH degree. To prevent disease progression, lifestyle changes should be recommended in relation with nutritional guidance, weight loss advice, and diabetes management[48].
The mortality rate of COVID-19 is high in cancer patients. Chemotherapy within the last 14 d significantly increased the severity of COVID-19 infection. Liver cancer is one of the most frequent cancers with 782 thousand deaths in 2018[105,106]. More than 90% of liver cancer is HCC. HCC usually develops as a result of underlying CLD, such as chronic HBV or HCV infection, ALD, and NAFLD. The majority of cases of HCC are found in the Asian-Pacific region[106]. Since Asia-Pacific region has more experience both in HCC and in COVID-19 than anywhere else. Thus experts prepare some recommendations of the management of HCC in the era of COVID-19[106].
HCC is a highly malignant and aggressive tumor, therefore its diagnosis and treatment should not be delayed due to the COVID-19 pandemic. Because patients may develop poor outcomes, HCC patients should be closely monitored during COVID-19 infection. To minimalize the exposure to the SARS-CoV-2, telemedicine may be used for screening HCC patients[48]. More intensive surveillance and early admission of HCC patients have been recommended. Systemic treatments and evaluation for liver transplant should be continued according to the guideline. Although 2 mo delay is reasonable, ADDSL recommended that HCC patients at-risk group (cirrhosis and chronic hepatitis B) continue both being monitored and therapy. The treatment should be continued and curative treatments should be periodized but other treatments such as vascular interventions and systemic therapy should be postponed. The risks and benefits of delaying surveillance should be told to the patients and documented. Images of patients with liver masses should be evaluated with an expert radiologist before the visit. Digital patient visits may be planned to discuss the diagnosis and management of liver tumors. If COVID-19 infection present, diagnosis and treatment of HCC patients should be postponed until the viral clearance[106].
HCC treatments should not be delayed[90]. HCC risk stratification scores should be used. Patients at an increasingly higher risk group (elevated alpha-fetoprotein levels, advanced cirrhosis, chronic hepatitis B, NASH/diabetes) should be prioritized for liver transplant[48,99]. In patients with COVID-19, immunosuppressive therapies should be temporarily withdrawn and kinase inhibitors should be taken on a case basis[48]. Imaging examination to detect recurrence at the early stage should not be postponed[106].
In patients with autoimmune hepatitis (AIH), immunosuppressive therapy makes those patients at greater risk for severe infection and should be prioritized for early testing. If elevated liver enzymes developed in AIH patients during COVID-19 infection flare-up should be considered and confirmation through biopsy should be made[90]. EASL-European Society of Clinical Microbiology and Infectious Diseases (ESCMID) advises withdrawn immunosuppressive therapy in such patients[48]. Withdrawing especially for antimetabolites, should only be considered after consultation of an expert and under special situation (such as in severe COVID-19 with medication-induced lymphopenia, or presence of other superinfection)[48].
There are some observational studies related to corticosteroid usage and predisposition for severe COVID-19 infection[48,107]. The World Health Organization suggests minimizing high-dose prednisolone but to continue small doses to avoid adrenal insufficiency[48,107].
Patients with autoimmune CLD have a high risk for COVID-19 infection because of their suppressed immune function[69]. Reducing immunosuppressive therapy is not recommended in patients with autoimmune liver disease, to prevent SARS-CoV-2 infection[48]. Reductions should only be considered in only some conditions such as drug-associated lymphopenia and bacterial/fungal superinfection in cases of severe COVID-19. To decrease systemic glucocorticoid exposure budesonide is recommended as a first-line agent to induce remission in patients without cirrhosis who have a flare of AIH.
In patients who developed COVID-19, corticosteroid dosing should be decreased. Change with dexamethasone may be considered in only hospitalized patients. There is not a specific recommendation for patients with primary biliary cholangitis, primary sclerosing cholangitis, or immunoglobulin G4-related disease.
Patients should be vaccinated for Streptococcus pneumonia, influenza and COVID-19.
Data related to liver transplant recipient patients with COVID-19 infection are still limited. However, patients with liver transplants have a high risk for COVID-19 infection due to their suppressed immune function by immunosuppressive therapy[108]. The clinical course of COVID-19 infection is different in immunosuppressed transplant recipients from that in non-immunosuppressed patients.
An Italian center reported no increase in the hospitalization among > 300 children who followed for liver transplantation, AIH, or hepatoblastoma. SARS-CoV-2 positivity developed only in 3 of 13 hospitalized children, who have liver trans
In a large liver transplant series including 151 patients, 6 patients became infected SARS-CoV-2, and 2.7%of long term recipients died, but all 7.5% of short-term recipients survived[110]. Immunosuppressive regimen discontinued three cases, acute respiratory distress syndrome (ARDS) rapidly developed in and caused death. Becchetti et al[111] reported that fever and dyspnea were found more frequent in the long-term than that of short-term recipients (91% vs 63%, and 59% vs 29% respectively). Computed tomography scan and pulmonary radiography showed typical features of the disease in 24 (43%) and 23 (40%) cases, respectively. These pathologies can be attributed to the long-term use of immunosuppressant medications[112]. In a European study conducted 19-transplant centers enrolled 57 liver transplant recipients with COVID-19 (16 outpatients and 41 hospitalized), mortality was found as 17%. ARDS developed 19% and hospitalization was needed in 72% of patients. Total fatality rates found were 12% and 17% in hospitalized patients, most of whom had cancer[111].
In an Italian study (13%) of 24 liver transplant recipients with COVID 19 were admitted to the ICU and 5 (21%) of them died[113]. A Swiss Transplant Cohort Study (a prospective observational multicenter study) reported 21 solid organs transplant patients. In five patients had liver transplant, steroids and calcineurin inhibitors continued the same dose. They reported that the clinical course was not different from other people[114].
In transplant recipients with COVID-19, liver injury is relatively seen less frequent, but acute kidney injury is more common probably due to calcineurin inhibitors usage[58,102,103].
Qin et al[115] observed a case of COVID-19 in a patient who underwent liver transplantation. Tacrolimus and glucocorticoids continued but decreased to lower doses. Another post-transplant patient recovered from severe COVID-19 pneumonia after gradual withdrawal from immunosuppression therapy and given low-dose corticosteroid[111].
EASL-ESCMID recommended that liver transplantation recipients should be restricted to patient with poor prognosis with acute/acute-on-chronic liver failure, end-stage liver disease (elevated MELD score), and HCC. Hospital stay time should be short and consultation from other clinics should be restricted. Complications such as peritonitis or encephalopathy should be followed to prevent hospitalization. Streptococcus pneumonia and influenza vaccination should be given[48]. COVID-19 vaccine should be applied. EASL-ESCMID recommendations on liver transplant recipients and candidates are shown in Table 2[48].
| EASL-ECCMID recommendations |
| Liver transplant recipients |
| Reduction of immunosuppressive therapy should only be considered under special circumstances such as drug-induced lymphopenia, superinfection in case of severe COVID-19 |
| LT recipients have high anxiety for COVID-19, and therefore their follow-up and treatment compliance may be impaired |
| Drug levels of calcineurin inhibitors and rapamycin inhibitors should be closely monitored. Because drugs used COVID-10 treatment such as hydroxychloroquine or protease inhibitors may interact them |
| Early admission should be made for LT recipients with COVID-19 infection |
| LT recipients, who have underlying malignancy, sarcopenia, graft dysfunction and metabolic disease are at-risk group for a severe COVID-19 infection |
| All patients should receive vaccination for Streptococcus pneumonia, influenza and COVID-19 |
| Liver transplant candidates |
| Patients on the LT waiting list with decompensated cirrhosis are at high risk of severe COVID-19 |
| LT should be prioritized for patients with poor short-term prognosis including those with acute liver failure, ACLF, high MELD score (including exceptional MELD points), and HCC at the upper limits of the Milan criteria |
| All donors for should be screening for SARS-CoV-2 infection by PCR and recommend |
| Both LT donors and recipients should be questioned clinical history, performed chest radiology, and SARS-CoV-2 testing |
| To reduce the risk of SARS-CoV-2 infection in the peri-transplantation period, protection measures should be strictly applied. Inward of high disease burden, a COVID-19 free pathway through transplantation should be implemented, including strict social isolation for waiting list patients, telephone screening for symptoms and exposures before admission, and perioperative management in a designated clean intensive care unit and post-LT ward |
| Consent for transplantation should include the potential risk of nosocomial COVID-19 |
| LT candidates should be informed that infection with SARS-CoV-2 in patients undergoing major surgery is associated with an increased risk of severe COVID-19 and death |
| Living-donor transplantations should be considered on a case-by-case basis and include careful risk stratification of donor and recipient, incorporating a combination of clinical history, chest radiology, and SARS-CoV-2 testing |
SARS-CoV-2 screening should be used both hospitalized patients with acute decompensation or acute liver failure. COVID-19 screening should be applied to both donors and recipients before transplantation[113]. COVID-19 screening is recomme
EASL-ESCMID suggested decreasing immunosuppressive therapy in post-transplant patients with severe COVID-19 only under special circumstances (e.g., medication-induced lymphopenia, bacterial/fungal superinfection)[48].
During the COVID-19 pandemic, all transplant centers are serving with limited operation similarly and more than other surgeries.
AASLD recommends delay liver transplant and screening both donors and recipients. Additionally, they suggest that a negative result does not exclude the COVID-19 infection. All informed consents provide for hospitalization and surgery have nosocomial COVID-19 transmission risk. Drug levels of mTOR inhibitors and calcineurin inhibitors should be monitored in patients who used drugs for COVID-19 therapy such as hydroxychloroquine or azithromycin[99]. Reduction in immunosuppressive therapy should be evaluated case basis.
A COVID-19-free transplant plan must include strict social isolation for patients on a list, wireless screening for signs and exposures to SARS-CoV-2 virus before admission, safe anesthesia procedures with PPE and N95, clean intensive care facility, and post-liver transplant care unit[99].
The COVID-19 pandemic negatively affected diagnosis and treatments of other patients with various diseases, including GI system cancer. Brazilian Gastrointestinal Tumors Group published evidence-based recommendations for GI cancers during the COVID-19 pandemic[116].
These recommendations were based on scientific evidence, such as randomized clinical trials, meta-analyses, and large cohort studies. However, some recommendations performed an expert opinion. There are detailed recommendations and aimed to prioritize curative-intent cancer treatments during the pandemic, to support the treatment of aggressive tumors when effective therapies are available, to decrease the number of or delay oncological non-priority surgeries, to decrease hospital visits, to minimize anticancer therapy-related immunosuppression in a specific high-risk group[116]. Recommendations related to GI cancers are showed in Table 3.
| Recommendations for gastrointestinal system tumors[116] |
| Social distancing mandates that every in-person interaction between patients and the health care system be scrutinized and only essential physical contacts between patients and health care professionals occur to diminish the risk of viral exposure to patients. Thus, minimize blood tests, scans and routine tests. Telephone and telemedicine visits should replace routine face-to-face clinic visits whenever possible |
| Whenever COVID-19 is clinically suspected or confirmed, systemic treatments should be suspended, and surgery should be postponed unless an urgent procedure is necessary (EOR) |
| Whenever surgery is indicated, SARS-CoV-2 testing should be considered |
| There are insufficient data to recommend in favor or against an open versus minimally invasive approach. Proven benefits of minimally invasive surgeries of reduced length of stay and complications should be considered individually. Nevertheless, whenever minimally invasive surgeries are indicated, the use of devices to filter released CO2 for aerosolized particles or techniques to treat the intra-abdominal gas whenever it should be emptied, is strongly advised |
| Central venous catheter flushing intervals should be increased to every 60 (younger and fit patients) or every 90 (older, frail patients with multiple comorbidities) days (EOR) |
| For early stage (cT1/2 cN0) colorectal, biliary, hepatocellular, esophagus and gastric tumors, where neoadjuvant treatment is not standard, consider deferring surgical resection to up to 8 weeks. If delays beyond 8 wk are expected, repeat staging exams (EOR) |
| Radiation schedules should be hypofractionated, whenever possible |
| Follow-up imaging and appointments should be reserved for those with symptoms suggestive of disease relapse. Asymptomatic patients not on active treatment should avoid imaging and follow up appointments, delaying tumor markers and colonoscopies, for example, for until the pandemic is over (EOR). In such cases, if possible, telemedicine or telephone consultation is indicated |
| DYPD screening is indicated whenever possible, before the use of fluoropyrimidines |
| Adjuvant treatment for colon and other gastrointestinal tumors, when recommended, should start in 4 wk to 8 wk after primary tumor resection. Monitoring blood counts at every cycle can be done by telemedicine if patients are asymptomatic |
| Infusional 5FU should be substituted for capecitabine in the following regimens: FOLFOX, cisplatin and 5FU, monotherapy, or when combined with radiotherapy. Exceptions are patients with severe renal dysfunction (creatinine clearance ≤ 30 mL/min); in patients with moderate (30 mL/min to 50 mL/min) renal dysfunction when upfront dose reduction of 25% is recommended |
| In curative-intent treatments, we encourage to maintain dose-intensity with the use of colony-stimulating growth factor (CSGF), if needed (EOR) |
| In the metastatic setting, consider dose-reduce chemotherapy instead of adding CSGF, if the latter requires more hospital visits (EOR) |
| In the metastatic setting, omit bolus 5FU in FOLFOX or FOLFIRI regimens to minimize toxicity (EOR) |
| Whenever possible, chemotherapy holidays may be considered in patients with low-volume metastatic disease, who are responding or experiencing tumor stabilization and when there is no major risk of complications for site-specific progression (e.g., peritoneum, biliary obstruction). If maintenance is considered to be beneficial instead of chemo holidays (e.g., more aggressive disease), prefer capecitabine alone, without bevacizumab[116] |
| Standard second or further lines of anticancer therapies should be recommended for ECOG 0 or 1 patient. Preferably, when there is clinically relevant overall survival gain demonstrated by randomized phase III trials (e.g., second-line for colorectal cancer) |
| Anti-PD1 immune checkpoint inhibitors are recommended in second or further lines of treatment for all gastrointestinal malignancies with microsatellite instability, regardless of the diagnostic method |
| For those in which immunotherapy monotherapy is indicated, we recommend the 6 wks’ schedule with pembrolizumab |
| Multidisciplinary team discussions (MDT) by web conferencing systems are highly encouraged. We think MDT are key to help with decisions about risks and benefits of cancer-directed therapies during the COVID-19 pandemic |
| In all cases, clinical individual judgment is advised and decisions should be shared with patients. Additionally, the anticipated survival benefit for each patient versus the risks of exposure to the virus should be discussed with patients, taking into consideration the individual’s comorbidities and degree of frailty, as well as caregivers and family members at home |
| Clinical trial enrolment |
| Patients who are candidates for clinical trials should be encouraged to enroll in the following situations: studies testing orphan drug indications, experimental treatments where benefits are very likely to outweigh the risks (e.g., immunotherapy combo of ipilimumab and nivolumab for microsatellite unstable metastatic colorectal cancer (CheckMate 8HW-NCT 04008030) or rare tumors. However, institutions and principal investigators should discuss and align with sponsors and Institutional Research Ethical Boards about how to minimize hospital visits (e.g., all lab and image tests performed in one single day), implement telemedicine in certain moments of trial conduction (lab checks for fit patients who are tolerating well the trial therapy, for example), extend intervals between hospital visits, if possible |
| For patients already on trial, treatment should continue based on clinical judgement that should balance tolerance versus benefit |
| The same principles cited above to decrease hospital visits should be sought |
GI symptoms are common in patients with COVID-19. During the pandemic, SARS-CoV-2 infection should be kept in mind in patients who have acute GI symptoms. COVID-19 infection is usually associated with mild hepatic involvement. All with CLD appear to be at great risk of contracting an infection. However, it appears that patients with cirrhosis, HCC, autoimmune diseases, or liver transplantation may have a greater risk for severe COVID-19. COVID-19 treatment should not be delayed in patients at these high-risk groups. Nutrition can be continued to improve important GI functions and clinical outcomes. Vaccines against SARS-CoV-2 have also been developed and are being widely used around the world. Like all chronic patients, COVID-19 vaccination should be prioritized in this group.
Provenance and peer review: Invited article; Externally peer reviewed.
Peer-review model: Single blind
Specialty type: Medicine, research and experimental
Country/Territory of origin: Turkey
Peer-review report’s scientific quality classification
Grade A (Excellent): 0
Grade B (Very good): 0
Grade C (Good): C
Grade D (Fair): 0
Grade E (Poor): 0
P-Reviewer: Zhuang ZH S-Editor: Gao CC L-Editor: A P-Editor: Gao CC
| 1. | World Health Organization. WHO Coronavirus (COVID-19) Dashboard. [cited 12 June 2021]. In: World Health Organization [Internet]. Available from: https://covid19.who.int/. |
| 2. | Viana SD, Nunes S, Reis F. ACE2 imbalance as a key player for the poor outcomes in COVID-19 patients with age-related comorbidities - Role of gut microbiota dysbiosis. Ageing Res Rev. 2020;62:101123. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 77] [Cited by in RCA: 121] [Article Influence: 24.2] [Reference Citation Analysis (0)] |
| 3. | Cichoż-Lach H, Michalak A. Liver injury in the era of COVID-19. World J Gastroenterol. 2021;27:377-390. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in CrossRef: 53] [Cited by in RCA: 51] [Article Influence: 12.8] [Reference Citation Analysis (2)] |
| 4. | Aguila EJT, Cua IHY, Dumagpi JEL, Francisco CPD, Raymundo NTV, Sy-Janairo MLL, Cabral-Prodigalidad PAI, Lontok MAD. COVID-19 and its effects on the digestive system and endoscopy practice. JGH Open. 2020;4:324-331. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 29] [Cited by in RCA: 31] [Article Influence: 6.2] [Reference Citation Analysis (0)] |
| 5. | Zou X, Chen K, Zou J, Han P, Hao J, Han Z. Single-cell RNA-seq data analysis on the receptor ACE2 expression reveals the potential risk of different human organs vulnerable to 2019-nCoV infection. Front Med. 2020;14:185-192. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 1286] [Cited by in RCA: 1528] [Article Influence: 305.6] [Reference Citation Analysis (0)] |
| 6. | Zang R, Gomez Castro MF, McCune BT, Zeng Q, Rothlauf PW, Sonnek NM, Liu Z, Brulois KF, Wang X, Greenberg HB, Diamond MS, Ciorba MA, Whelan SPJ, Ding S. TMPRSS2 and TMPRSS4 promote SARS-CoV-2 infection of human small intestinal enterocytes. Sci Immunol. 2020;5. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 809] [Cited by in RCA: 765] [Article Influence: 153.0] [Reference Citation Analysis (0)] |
| 7. | Papadakis KA, Prehn J, Nelson V, Cheng L, Binder SW, Ponath PD, Andrew DP, Targan SR. The role of thymus-expressed chemokine and its receptor CCR9 on lymphocytes in the regional specialization of the mucosal immune system. J Immunol. 2000;165:5069-5076. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 262] [Cited by in RCA: 252] [Article Influence: 10.1] [Reference Citation Analysis (0)] |
| 8. | Wang J, Li F, Wei H, Lian ZX, Sun R, Tian Z. Respiratory influenza virus infection induces intestinal immune injury via microbiota-mediated Th17 cell-dependent inflammation. J Exp Med. 2014;211:2397-2410. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 256] [Cited by in RCA: 366] [Article Influence: 33.3] [Reference Citation Analysis (0)] |
| 9. | Tripathi A, Debelius J, Brenner DA, Karin M, Loomba R, Schnabl B, Knight R. The gut-liver axis and the intersection with the microbiome. Nat Rev Gastroenterol Hepatol. 2018;15:397-411. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 872] [Cited by in RCA: 970] [Article Influence: 138.6] [Reference Citation Analysis (0)] |
| 10. | Cheung KS, Hung IFN, Chan PPY, Lung KC, Tso E, Liu R, Ng YY, Chu MY, Chung TWH, Tam AR, Yip CCY, Leung KH, Fung AY, Zhang RR, Lin Y, Cheng HM, Zhang AJX, To KKW, Chan KH, Yuen KY, Leung WK. Gastrointestinal Manifestations of SARS-CoV-2 Infection and Virus Load in Fecal Samples From a Hong Kong Cohort: Systematic Review and Meta-analysis. Gastroenterology. 2020;159:81-95. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 1113] [Cited by in RCA: 1131] [Article Influence: 226.2] [Reference Citation Analysis (1)] |
| 11. | Sultan S, Altayar O, Siddique SM, Davitkov P, Feuerstein JD, Lim JK, Falck-Ytter Y, El-Serag HB; AGA Institute. Electronic address: ewilson@gastro.org. AGA Institute Rapid Review of the Gastrointestinal and Liver Manifestations of COVID-19, Meta-Analysis of International Data, and Recommendations for the Consultative Management of Patients with COVID-19. Gastroenterology. 2020;159:320-334.e27. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 309] [Cited by in RCA: 293] [Article Influence: 58.6] [Reference Citation Analysis (1)] |
| 12. | Silva FAFD, Brito BB, Santos MLC, Marques HS, Silva Júnior RTD, Carvalho LS, Vieira ES, Oliveira MV, Melo FF. COVID-19 gastrointestinal manifestations: a systematic review. Rev Soc Bras Med Trop. 2020;53:e20200714. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 52] [Cited by in RCA: 48] [Article Influence: 9.6] [Reference Citation Analysis (0)] |
| 13. | Ferm S, Fisher C, Pakala T, Tong M, Shah D, Schwarzbaum D, Cooley V, Hussain S, Kim SH. Analysis of Gastrointestinal and Hepatic Manifestations of SARS-CoV-2 Infection in 892 Patients in Queens, NY. Clin Gastroenterol Hepatol. 2020;18:2378-2379.e1. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 46] [Cited by in RCA: 60] [Article Influence: 12.0] [Reference Citation Analysis (0)] |
| 14. | Alay H, Yılmaz S, Albayrak A, Ozkurt Z, Ozden K, Parlak E, Tekin E, Kocak AO. Gastrointestinal Symptoms and Liver Damage in Patients with COVID-19. FLORA. 2021;26:249-256. [RCA] [DOI] [Full Text] [Cited by in Crossref: 1] [Cited by in RCA: 1] [Article Influence: 0.3] [Reference Citation Analysis (0)] |
| 15. | Cholankeril G, Podboy A, Aivaliotis VI, Pham EA, Spencer SP, Kim D, Ahmed A. Association of Digestive Symptoms and Hospitalization in Patients With SARS-CoV-2 Infection. Am J Gastroenterol. 2020;115:1129-1132. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 35] [Cited by in RCA: 32] [Article Influence: 6.4] [Reference Citation Analysis (0)] |
| 16. | Ye Q, Wang B, Zhang T, Xu J, Shang S. The mechanism and treatment of gastrointestinal symptoms in patients with COVID-19. Am J Physiol Gastrointest Liver Physiol. 2020;319:G245-G252. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 120] [Cited by in RCA: 113] [Article Influence: 22.6] [Reference Citation Analysis (2)] |
| 17. | Hamming I, Timens W, Bulthuis ML, Lely AT, Navis G, van Goor H. Tissue distribution of ACE2 protein, the functional receptor for SARS coronavirus. A first step in understanding SARS pathogenesis. J Pathol. 2004;203:631-637. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 3643] [Cited by in RCA: 4149] [Article Influence: 197.6] [Reference Citation Analysis (0)] |
| 18. | Hashimoto T, Perlot T, Rehman A, Trichereau J, Ishiguro H, Paolino M, Sigl V, Hanada T, Hanada R, Lipinski S, Wild B, Camargo SM, Singer D, Richter A, Kuba K, Fukamizu A, Schreiber S, Clevers H, Verrey F, Rosenstiel P, Penninger JM. ACE2 Links amino acid malnutrition to microbial ecology and intestinal inflammation. Nature. 2012;487:477-481. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 787] [Cited by in RCA: 987] [Article Influence: 75.9] [Reference Citation Analysis (0)] |
| 19. | Fang D, Ma JD, Guan JL, Wang MR, Song Y, Tian DA, Li PY. A single-center, descriptive study of the digestive system performance of hospitalized patients with novel coronavirus pneumonia in Wuhan area. Zhonghua Xiaohua Zazhi. 2020;40:151-156. [DOI] [Full Text] |
| 20. | Han C, Duan C, Zhang S, Spiegel B, Shi H, Wang W, Zhang L, Lin R, Liu J, Ding Z, Hou X. Digestive Symptoms in COVID-19 Patients With Mild Disease Severity: Clinical Presentation, Stool Viral RNA Testing, and Outcomes. Am J Gastroenterol. 2020;115:916-923. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 413] [Cited by in RCA: 383] [Article Influence: 76.6] [Reference Citation Analysis (0)] |
| 21. | Carvalho A, Alqusairi R, Adams A, Paul M, Kothari N, Peters S, DeBenedet AT. SARS-CoV-2 Gastrointestinal Infection Causing Hemorrhagic Colitis: Implications for Detection and Transmission of COVID-19 Disease. Am J Gastroenterol. 2020;115:942-946. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 139] [Cited by in RCA: 143] [Article Influence: 28.6] [Reference Citation Analysis (0)] |
| 22. | Gupta S, Parker J, Smits S, Underwood J, Dolwani S. Persistent viral shedding of SARS-CoV-2 in faeces - a rapid review. Colorectal Dis. 2020;22:611-620. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 147] [Cited by in RCA: 142] [Article Influence: 28.4] [Reference Citation Analysis (0)] |
| 23. | da Silva FAF, de Brito BB, Santos MLC, Marques HS, da Silva Júnior RT, de Carvalho LS, de Sousa Cruz S, Rocha GR, Correa Santos GL, de Souza KC, Maciel RGA, Lopes DS, Silva NOE, Oliveira MV, de Melo FF. Transmission of severe acute respiratory syndrome coronavirus 2 via fecal-oral: Current knowledge. World J Clin Cases. 2021;9:8280-8294. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in CrossRef: 4] [Cited by in RCA: 4] [Article Influence: 1.0] [Reference Citation Analysis (1)] |
| 24. | Zhang Y, Chen C, Song Y, Zhu S, Wang D, Zhang H, Han G, Weng Y, Xu J, Yu P, Jiang W, Yang X, Lang Z, Yan D, Wang Y, Song J, Gao GF, Wu G, Xu W. Excretion of SARS-CoV-2 through faecal specimens. Emerg Microbes Infect. 2020;9:2501-2508. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 31] [Cited by in RCA: 41] [Article Influence: 8.2] [Reference Citation Analysis (0)] |
| 25. | Wu Y, Guo C, Tang L, Hong Z, Zhou J, Dong X, Yin H, Xiao Q, Tang Y, Qu X, Kuang L, Fang X, Mishra N, Lu J, Shan H, Jiang G, Huang X. Prolonged presence of SARS-CoV-2 viral RNA in faecal samples. Lancet Gastroenterol Hepatol. 2020;5:434-435. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 1050] [Cited by in RCA: 1150] [Article Influence: 230.0] [Reference Citation Analysis (0)] |
| 26. | Chen Y, Chen L, Deng Q, Zhang G, Wu K, Ni L, Yang Y, Liu B, Wang W, Wei C, Yang J, Ye G, Cheng Z. The presence of SARS-CoV-2 RNA in the feces of COVID-19 patients. J Med Virol. 2020;92:833-840. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 505] [Cited by in RCA: 570] [Article Influence: 114.0] [Reference Citation Analysis (0)] |
| 27. | Chin AWH, Chu JTS, Perera MRA, Hui KPY, Yen HL, Chan MCW, Peiris M, Poon LLM. Stability of SARS-CoV-2 in different environmental conditions. Lancet Microbe. 2020;1:e10. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 984] [Cited by in RCA: 1180] [Article Influence: 236.0] [Reference Citation Analysis (0)] |
| 28. | van Doremalen N, Bushmaker T, Morris DH, Holbrook MG, Gamble A, Williamson BN, Tamin A, Harcourt JL, Thornburg NJ, Gerber SI, Lloyd-Smith JO, de Wit E, Munster VJ. Aerosol and Surface Stability of SARS-CoV-2 as Compared with SARS-CoV-1. N Engl J Med. 2020;382:1564-1567. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 5894] [Cited by in RCA: 5665] [Article Influence: 1133.0] [Reference Citation Analysis (0)] |
| 29. | Amirian ES. Potential fecal transmission of SARS-CoV-2: Current evidence and implications for public health. Int J Infect Dis. 2020;95:363-370. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 218] [Cited by in RCA: 197] [Article Influence: 39.4] [Reference Citation Analysis (0)] |
| 30. | Tian Y, Rong L, Nian W, He Y. Review article: gastrointestinal features in COVID-19 and the possibility of faecal transmission. Aliment Pharmacol Ther. 2020;51:843-851. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 594] [Cited by in RCA: 583] [Article Influence: 116.6] [Reference Citation Analysis (0)] |
| 31. | Burgueño JF, Reich A, Hazime H, Quintero MA, Fernandez I, Fritsch J, Santander AM, Brito N, Damas OM, Deshpande A, Kerman DH, Zhang L, Gao Z, Ban Y, Wang L, Pignac-Kobinger J, Abreu MT. Expression of SARS-CoV-2 Entry Molecules ACE2 and TMPRSS2 in the Gut of Patients With IBD. Inflamm Bowel Dis. 2020;26:797-808. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 128] [Cited by in RCA: 138] [Article Influence: 27.6] [Reference Citation Analysis (0)] |
| 32. | Abreu MT, Peyrin-Biroulet L. Providing Guidance During a Global Viral Pandemic for the Care of Patients With Inflammatory Bowel Disease. J Crohns Colitis. 2020;14:S767-S768. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 5] [Cited by in RCA: 5] [Article Influence: 1.0] [Reference Citation Analysis (0)] |
| 33. | Vasieva O, Goryanin I. The Gut Microbiome versus COVID-19. J Comput Sci Syst Biol. 2021;14. |
| 34. | Lu CC, Ma KL, Ruan XZ, Liu BC. Intestinal dysbiosis activates renal renin-angiotensin system contributing to incipient diabetic nephropathy. Int J Med Sci. 2018;15:816-822. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 32] [Cited by in RCA: 35] [Article Influence: 5.0] [Reference Citation Analysis (0)] |
| 35. | Cole-Jeffrey CT, Liu M, Katovich MJ, Raizada MK, Shenoy V. ACE2 and Microbiota: Emerging Targets for Cardiopulmonary Disease Therapy. J Cardiovasc Pharmacol. 2015;66:540-550. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 78] [Cited by in RCA: 81] [Article Influence: 9.0] [Reference Citation Analysis (0)] |
| 36. | Zuo T, Zhang F, Lui GCY, Yeoh YK, Li AYL, Zhan H, Wan Y, Chung ACK, Cheung CP, Chen N, Lai CKC, Chen Z, Tso EYK, Fung KSC, Chan V, Ling L, Joynt G, Hui DSC, Chan FKL, Chan PKS, Ng SC. Alterations in Gut Microbiota of Patients With COVID-19 During Time of Hospitalization. Gastroenterology. 2020;159:944-955.e8. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 739] [Cited by in RCA: 1064] [Article Influence: 212.8] [Reference Citation Analysis (0)] |
| 37. | Yeoh YK, Zuo T, Lui GC, Zhang F, Liu Q, Li AY, Chung AC, Cheung CP, Tso EY, Fung KS, Chan V, Ling L, Joynt G, Hui DS, Chow KM, Ng SSS, Li TC, Ng RW, Yip TC, Wong GL, Chan FK, Wong CK, Chan PK, Ng SC. Gut microbiota composition reflects disease severity and dysfunctional immune responses in patients with COVID-19. Gut. 2021;70:698-706. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 995] [Cited by in RCA: 863] [Article Influence: 215.8] [Reference Citation Analysis (0)] |
| 38. | Mendes V, Galvão I, Vieira AT. Mechanisms by Which the Gut Microbiota Influences Cytokine Production and Modulates Host Inflammatory Responses. J Interferon Cytokine Res. 2019;39:393-409. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 36] [Cited by in RCA: 66] [Article Influence: 11.0] [Reference Citation Analysis (0)] |
| 39. | Hagan T, Cortese M, Rouphael N, Boudreau C, Linde C, Maddur MS, Das J, Wang H, Guthmiller J, Zheng NY, Huang M, Uphadhyay AA, Gardinassi L, Petitdemange C, McCullough MP, Johnson SJ, Gill K, Cervasi B, Zou J, Bretin A, Hahn M, Gewirtz AT, Bosinger SE, Wilson PC, Li S, Alter G, Khurana S, Golding H, Pulendran B. Antibiotics-Driven Gut Microbiome Perturbation Alters Immunity to Vaccines in Humans. Cell. 2019;178:1313-1328.e13. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 467] [Cited by in RCA: 425] [Article Influence: 70.8] [Reference Citation Analysis (0)] |
| 40. | Mullish BH, Marchesi JR, McDonald JAK, Pass DA, Masetti G, Michael DR, Plummer S, Jack AA, Davies TS, Hughes TR, Wang D. Probiotics reduce self-reported symptoms of upper respiratory tract infection in overweight and obese adults: should we be considering probiotics during viral pandemics? Gut Microbes. 2021;13:1-9. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 26] [Cited by in RCA: 28] [Article Influence: 7.0] [Reference Citation Analysis (0)] |
| 41. | Metawea MI, Yousif WI, Moheb I. COVID 19 and liver: An A-Z literature review. Dig Liver Dis. 2021;53:146-152. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 62] [Cited by in RCA: 60] [Article Influence: 15.0] [Reference Citation Analysis (2)] |
| 42. | Yao N, Wang SN, Lian JQ, Sun YT, Zhang GF, Kang WZ, Kang W. [Clinical characteristics and influencing factors of patients with novel coronavirus pneumonia combined with liver injury in Shaanxi region]. Zhonghua Gan Zang Bing Za Zhi. 2020;28:234-239. [RCA] [PubMed] [DOI] [Full Text] [Cited by in RCA: 39] [Reference Citation Analysis (0)] |
| 43. | Chai X, Hu L, Zhang Y, Han W, Lu Z, Ke A, Zhou J, Shi G, Fang N, Fan J. Specific ACE2 expression in cholangiocytes may cause liver damage after 2019-nCoV infection. 2020 Preprint. Available from: bioRxiv: 2020.02.03.931766. [DOI] [Full Text] |
| 44. | Tukiainen T, Villani AC, Yen A, Rivas MA, Marshall JL, Satija R, Aguirre M, Gauthier L, Fleharty M, Kirby A, Cummings BB, Castel SE, Karczewski KJ, Aguet F, Byrnes A; GTEx Consortium; Laboratory, Data Analysis &Coordinating Center (LDACC)—Analysis Working Group; Statistical Methods groups—Analysis Working Group; Enhancing GTEx (eGTEx) groups; NIH Common Fund; NIH/NCI; NIH/NHGRI; NIH/NIMH; NIH/NIDA; Biospecimen Collection Source Site—NDRI; Biospecimen Collection Source Site—RPCI; Biospecimen Core Resource—VARI; Brain Bank Repository—University of Miami Brain Endowment Bank; Leidos Biomedical—Project Management; ELSI Study; Genome Browser Data Integration &Visualization—EBI; Genome Browser Data Integration &Visualization—UCSC Genomics Institute, University of California Santa Cruz, Lappalainen T, Regev A, Ardlie KG, Hacohen N, MacArthur DG. Landscape of X chromosome inactivation across human tissues. Nature. 2017;550:244-248. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 741] [Cited by in RCA: 760] [Article Influence: 95.0] [Reference Citation Analysis (0)] |
| 45. | Jin JM, Bai P, He W, Wu F, Liu XF, Han DM, Liu S, Yang JK. Gender Differences in Patients With COVID-19: Focus on Severity and Mortality. Front Public Health. 2020;8:152. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 1408] [Cited by in RCA: 1330] [Article Influence: 266.0] [Reference Citation Analysis (0)] |
| 46. | Morgan K, Samuel K, Vandeputte M, Hayes PC, Plevris JN. SARS-CoV-2 Infection and the Liver. Pathogens. 2020;9. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 37] [Cited by in RCA: 36] [Article Influence: 7.2] [Reference Citation Analysis (0)] |
| 47. | Sonzogni A, Previtali G, Seghezzi M, Grazia Alessio M, Gianatti A, Licini L, Morotti D, Zerbi P, Carsana L, Rossi R, Lauri E, Pellegrinelli A, Nebuloni M. Liver histopathology in severe COVID 19 respiratory failure is suggestive of vascular alterations. Liver Int. 2020;40:2110-2116. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 162] [Cited by in RCA: 218] [Article Influence: 43.6] [Reference Citation Analysis (0)] |
| 48. | Boettler T, Marjot T, Newsome PN, Mondelli MU, Maticic M, Cordero E, Jalan R, Moreau R, Cornberg M, Berg T. Impact of COVID-19 on the care of patients with liver disease: EASL-ESCMID position paper after 6 mo of the pandemic. JHEP Rep. 2020;2:100169. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 103] [Cited by in RCA: 121] [Article Influence: 24.2] [Reference Citation Analysis (0)] |
| 49. | Feng G, Zheng KI, Yan QQ, Rios RS, Targher G, Byrne CD, Poucke SV, Liu WY, Zheng MH. COVID-19 and Liver Dysfunction: Current Insights and Emergent Therapeutic Strategies. J Clin Transl Hepatol. 2020;8:18-24. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 289] [Cited by in RCA: 278] [Article Influence: 55.6] [Reference Citation Analysis (0)] |
| 50. | Bourgonje AR, Abdulle AE, Timens W, Hillebrands JL, Navis GJ, Gordijn SJ, Bolling MC, Dijkstra G, Voors AA, Osterhaus AD, van der Voort PH, Mulder DJ, van Goor H. Angiotensin-converting enzyme 2 (ACE2), SARS-CoV-2 and the pathophysiology of coronavirus disease 2019 (COVID-19). J Pathol. 2020;251:228-248. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 661] [Cited by in RCA: 738] [Article Influence: 147.6] [Reference Citation Analysis (0)] |
| 51. | Gomez Cifuentes JD, Sparkman J, Choi K, Sealock RJ. Regarding: Lui RN, Wong SH, Sánchez-Luna SA, et al Overview of guidance for endoscopy during the coronavirus disease 2019 pandemic. J. Gastroenterol. Hepatol. 2020;35(5):749-759. https://doi.org/10.1111/jgh.15053. J Gastroenterol Hepatol. 2020;35:1654. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 3] [Cited by in RCA: 4] [Article Influence: 0.8] [Reference Citation Analysis (0)] |
| 52. | Li J, Fan JG. Characteristics and Mechanism of Liver Injury in 2019 Coronavirus Disease. J Clin Transl Hepatol. 2020;8:13-17. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 199] [Cited by in RCA: 206] [Article Influence: 41.2] [Reference Citation Analysis (2)] |
| 53. | Machhi J, Herskovitz J, Senan AM, Dutta D, Nath B, Oleynikov MD, Blomberg WR, Meigs DD, Hasan M, Patel M, Kline P, Chang RC, Chang L, Gendelman HE, Kevadiya BD. The Natural History, Pathobiology, and Clinical Manifestations of SARS-CoV-2 Infections. J Neuroimmune Pharmacol. 2020;15:359-386. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 357] [Cited by in RCA: 355] [Article Influence: 71.0] [Reference Citation Analysis (0)] |
| 54. | Tan YJ, Fielding BC, Goh PY, Shen S, Tan TH, Lim SG, Hong W. Overexpression of 7a, a protein specifically encoded by the severe acute respiratory syndrome coronavirus, induces apoptosis via a caspase-dependent pathway. J Virol. 2004;78:14043-14047. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 157] [Cited by in RCA: 166] [Article Influence: 7.9] [Reference Citation Analysis (0)] |
| 55. | Garrido I, Liberal R, Macedo G. Review article: COVID-19 and liver disease-what we know on 1st May 2020. Aliment Pharmacol Ther. 2020;52:267-275. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 125] [Cited by in RCA: 118] [Article Influence: 23.6] [Reference Citation Analysis (0)] |
| 56. | Wang Y, Zhang D, Du G, Du R, Zhao J, Jin Y, Fu S, Gao L, Cheng Z, Lu Q, Hu Y, Luo G, Wang K, Lu Y, Li H, Wang S, Ruan S, Yang C, Mei C, Wang Y, Ding D, Wu F, Tang X, Ye X, Ye Y, Liu B, Yang J, Yin W, Wang A, Fan G, Zhou F, Liu Z, Gu X, Xu J, Shang L, Zhang Y, Cao L, Guo T, Wan Y, Qin H, Jiang Y, Jaki T, Hayden FG, Horby PW, Cao B, Wang C. Remdesivir in adults with severe COVID-19: a randomised, double-blind, placebo-controlled, multicentre trial. Lancet. 2020;395:1569-1578. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 2337] [Cited by in RCA: 2487] [Article Influence: 497.4] [Reference Citation Analysis (0)] |
| 57. | Fara A, Mitrev Z, Rosalia RA, Assas BM. Cytokine storm and COVID-19: a chronicle of pro-inflammatory cytokines. Open Biol. 2020;10:200160. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 205] [Cited by in RCA: 229] [Article Influence: 45.8] [Reference Citation Analysis (0)] |
| 58. | Shi H, Wang W, Yin J, Ouyang Y, Pang L, Feng Y, Qiao L, Guo X, Shi H, Jin R, Chen D. The inhibition of IL-2/IL-2R gives rise to CD8+ T cell and lymphocyte decrease through JAK1-STAT5 in critical patients with COVID-19 pneumonia. Cell Death Dis. 2020;11:429. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 62] [Cited by in RCA: 66] [Article Influence: 13.2] [Reference Citation Analysis (0)] |
| 59. | Lagana SM, Kudose S, Iuga AC, Lee MJ, Fazlollahi L, Remotti HE, Del Portillo A, De Michele S, de Gonzalez AK, Saqi A, Khairallah P, Chong AM, Park H, Uhlemann AC, Lefkowitch JH, Verna EC. Hepatic pathology in patients dying of COVID-19: a series of 40 cases including clinical, histologic, and virologic data. Mod Pathol. 2020;33:2147-2155. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 130] [Cited by in RCA: 181] [Article Influence: 36.2] [Reference Citation Analysis (0)] |
| 60. | Xu Z, Shi L, Wang Y, Zhang J, Huang L, Zhang C, Liu S, Zhao P, Liu H, Zhu L, Tai Y, Bai C, Gao T, Song J, Xia P, Dong J, Zhao J, Wang FS. Pathological findings of COVID-19 associated with acute respiratory distress syndrome. Lancet Respir Med. 2020;8:420-422. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 5228] [Cited by in RCA: 5781] [Article Influence: 1156.2] [Reference Citation Analysis (2)] |
| 61. | Brito CA, Barros FM, Lopes EP. Mechanisms and consequences of COVID-19 associated liver injury: What can we affirm? World J Hepatol. 2020;12:413-422. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 15] [Cited by in RCA: 20] [Article Influence: 4.0] [Reference Citation Analysis (0)] |
| 62. | Sonzogni A, Previtali G, Seghezzi M, Alessio MG, Gianatti A, Licini L, Zerbi P, Carsana L, Rossi R, Lauri E. Liver and COVID 19 infection: a very preliminary lesson learnt from histological post-mortem findings in 48 patients. 2020 Preprint. Available from: Preprints2020040438. [DOI] [Full Text] |
| 63. | Zhang C, Shi L, Wang FS. Liver injury in COVID-19: management and challenges. Lancet Gastroenterol Hepatol. 2020;5:428-430. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 1348] [Cited by in RCA: 1295] [Article Influence: 259.0] [Reference Citation Analysis (4)] |
| 64. | Ji D, Qin E, Xu J, Zhang D, Cheng G, Wang Y, Lau G. Non-alcoholic fatty liver diseases in patients with COVID-19: A retrospective study. J Hepatol. 2020;73:451-453. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 336] [Cited by in RCA: 408] [Article Influence: 81.6] [Reference Citation Analysis (2)] |
| 65. | Liu W, Tao ZW, Wang L, Yuan ML, Liu K, Zhou L, Wei S, Deng Y, Liu J, Liu HG, Yang M, Hu Y. Analysis of factors associated with disease outcomes in hospitalized patients with 2019 novel coronavirus disease. Chin Med J (Engl). 2020;133:1032-1038. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 562] [Cited by in RCA: 636] [Article Influence: 127.2] [Reference Citation Analysis (0)] |
| 66. | Trevenzoli M, Guarnaccia A, Alberici I, Fassan M, Di Meco E, Farinati F, Cattelan AM. SARS-CoV-2 and hepatitis. J Gastrointestin Liver Dis. 2020;29:473-475. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 15] [Cited by in RCA: 12] [Article Influence: 2.4] [Reference Citation Analysis (0)] |
| 67. | Gordon DE, Jang GM, Bouhaddou M, Xu J, Obernier K, White KM, O'Meara MJ, Rezelj VV, Guo JZ, Swaney DL, Tummino TA, Hüttenhain R, Kaake RM, Richards AL, Tutuncuoglu B, Foussard H, Batra J, Haas K, Modak M, Kim M, Haas P, Polacco BJ, Braberg H, Fabius JM, Eckhardt M, Soucheray M, Bennett MJ, Cakir M, McGregor MJ, Li Q, Meyer B, Roesch F, Vallet T, Mac Kain A, Miorin L, Moreno E, Naing ZZC, Zhou Y, Peng S, Shi Y, Zhang Z, Shen W, Kirby IT, Melnyk JE, Chorba JS, Lou K, Dai SA, Barrio-Hernandez I, Memon D, Hernandez-Armenta C, Lyu J, Mathy CJP, Perica T, Pilla KB, Ganesan SJ, Saltzberg DJ, Rakesh R, Liu X, Rosenthal SB, Calviello L, Venkataramanan S, Liboy-Lugo J, Lin Y, Huang XP, Liu Y, Wankowicz SA, Bohn M, Safari M, Ugur FS, Koh C, Savar NS, Tran QD, Shengjuler D, Fletcher SJ, O'Neal MC, Cai Y, Chang JCJ, Broadhurst DJ, Klippsten S, Sharp PP, Wenzell NA, Kuzuoglu-Ozturk D, Wang HY, Trenker R, Young JM, Cavero DA, Hiatt J, Roth TL, Rathore U, Subramanian A, Noack J, Hubert M, Stroud RM, Frankel AD, Rosenberg OS, Verba KA, Agard DA, Ott M, Emerman M, Jura N, von Zastrow M, Verdin E, Ashworth A, Schwartz O, d'Enfert C, Mukherjee S, Jacobson M, Malik HS, Fujimori DG, Ideker T, Craik CS, Floor SN, Fraser JS, Gross JD, Sali A, Roth BL, Ruggero D, Taunton J, Kortemme T, Beltrao P, Vignuzzi M, García-Sastre A, Shokat KM, Shoichet BK, Krogan NJ. A SARS-CoV-2 protein interaction map reveals targets for drug repurposing. Nature. 2020;583:459-468. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 3145] [Cited by in RCA: 3185] [Article Influence: 637.0] [Reference Citation Analysis (0)] |
| 68. | Medetalibeyoglu A, Catma Y, Senkal N, Ormeci A, Cavus B, Kose M, Bayramlar OF, Yildiz G, Akyuz F, Kaymakoglu S, Tukek T. The effect of liver test abnormalities on the prognosis of COVID-19. Ann Hepatol. 2020;19:614-621. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 42] [Cited by in RCA: 53] [Article Influence: 10.6] [Reference Citation Analysis (0)] |
| 69. | Sarin SK, Choudhury A, Lau GK, Zheng MH, Ji D, Abd-Elsalam S, Hwang J, Qi X, Cua IH, Suh JI, Park JG, Putcharoen O, Kaewdech A, Piratvisuth T, Treeprasertsuk S, Park S, Wejnaruemarn S, Payawal DA, Baatarkhuu O, Ahn SH, Yeo CD, Alonzo UR, Chinbayar T, Loho IM, Yokosuka O, Jafri W, Tan S, Soo LI, Tanwandee T, Gani R, Anand L, Esmail ES, Khalaf M, Alam S, Lin CY, Chuang WL, Soin AS, Garg HK, Kalista K, Batsukh B, Purnomo HD, Dara VP, Rathi P, Al Mahtab M, Shukla A, Sharma MK, Omata M; APASL COVID Task Force, APASL COVID Liver Injury Spectrum Study (APCOLIS Study-NCT 04345640). Pre-existing liver disease is associated with poor outcome in patients with SARS CoV2 infection; The APCOLIS Study (APASL COVID-19 Liver Injury Spectrum Study). Hepatol Int. 2020;14:690-700. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 217] [Cited by in RCA: 216] [Article Influence: 43.2] [Reference Citation Analysis (1)] |
| 70. | Kumar-M P, Mishra S, Jha DK, Shukla J, Choudhury A, Mohindra R, Mandavdhare HS, Dutta U, Sharma V. Coronavirus disease (COVID-19) and the liver: a comprehensive systematic review and meta-analysis. Hepatol Int. 2020;14:711-722. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 117] [Cited by in RCA: 111] [Article Influence: 22.2] [Reference Citation Analysis (0)] |
| 71. | Kulkarni AV, Kumar P, Tevethia HV, Premkumar M, Arab JP, Candia R, Talukdar R, Sharma M, Qi X, Rao PN, Reddy DN. Systematic review with meta-analysis: liver manifestations and outcomes in COVID-19. Aliment Pharmacol Ther. 2020;52:584-599. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 199] [Cited by in RCA: 183] [Article Influence: 36.6] [Reference Citation Analysis (0)] |
| 72. | Wang D, Hu B, Hu C, Zhu F, Liu X, Zhang J, Wang B, Xiang H, Cheng Z, Xiong Y, Zhao Y, Li Y, Wang X, Peng Z. Clinical Characteristics of 138 Hospitalized Patients With 2019 Novel Coronavirus-Infected Pneumonia in Wuhan, China. JAMA. 2020;323:1061-1069. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 14113] [Cited by in RCA: 14764] [Article Influence: 2952.8] [Reference Citation Analysis (0)] |
| 73. | Holshue ML, DeBolt C, Lindquist S, Lofy KH, Wiesman J, Bruce H, Spitters C, Ericson K, Wilkerson S, Tural A, Diaz G, Cohn A, Fox L, Patel A, Gerber SI, Kim L, Tong S, Lu X, Lindstrom S, Pallansch MA, Weldon WC, Biggs HM, Uyeki TM, Pillai SK; Washington State 2019-nCoV Case Investigation Team. First Case of 2019 Novel Coronavirus in the United States. N Engl J Med. 2020;382:929-936. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 4155] [Cited by in RCA: 3820] [Article Influence: 764.0] [Reference Citation Analysis (1)] |
| 74. | Parohan M, Yaghoubi S, Seraji A. Liver injury is associated with severe coronavirus disease 2019 (COVID-19) infection: A systematic review and meta-analysis of retrospective studies. Hepatol Res. 2020;50:924-935. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 103] [Cited by in RCA: 92] [Article Influence: 18.4] [Reference Citation Analysis (0)] |
| 75. | Chen LY, Chu HK, Bai T, Tu SJ, Wei Y, Li ZL, Hu LL, Zhu R, Zhang L, Han CQ, Xiao L, He Q, Song J, Liu WH, Zhu QJ, Chen H, Yang L, Hou XH. Liver damage at admission is an independent prognostic factor for COVID-19. J Dig Dis. 2020;21:512-518. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 33] [Cited by in RCA: 32] [Article Influence: 6.4] [Reference Citation Analysis (0)] |
| 76. | Zhang B, Zhou X, Qiu Y, Song Y, Feng F, Feng J, Song Q, Jia Q, Wang J. Clinical characteristics of 82 cases of death from COVID-19. PLoS One. 2020;15:e0235458. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 269] [Cited by in RCA: 324] [Article Influence: 64.8] [Reference Citation Analysis (0)] |
| 77. | Fierro NA. COVID-19 and the liver: What do we know after six months of the pandemic? Ann Hepatol. 2020;19:590-591. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 23] [Cited by in RCA: 23] [Article Influence: 4.6] [Reference Citation Analysis (0)] |
| 78. | Xie H, Zhao J, Lian N, Lin S, Xie Q, Zhuo H. Clinical characteristics of non-ICU hospitalized patients with coronavirus disease 2019 and liver injury: A retrospective study. Liver Int. 2020;40:1321-1326. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 198] [Cited by in RCA: 213] [Article Influence: 42.6] [Reference Citation Analysis (0)] |
| 79. | Sobotka LA, Esteban J, Volk ML, Elmunzer BJ, Rockey DC; North American Alliance for the Study of Digestive Manifestation of COVID-19*. Acute Liver Injury in Patients Hospitalized with COVID-19. Dig Dis Sci. 2021;. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 4] [Cited by in RCA: 17] [Article Influence: 5.7] [Reference Citation Analysis (0)] |
| 80. | Bertolini A, van de Peppel IP, Bodewes FAJA, Moshage H, Fantin A, Farinati F, Fiorotto R, Jonker JW, Strazzabosco M, Verkade HJ, Peserico G. Abnormal Liver Function Tests in Patients With COVID-19: Relevance and Potential Pathogenesis. Hepatology. 2020;72:1864-1872. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 219] [Cited by in RCA: 193] [Article Influence: 38.6] [Reference Citation Analysis (0)] |
| 81. | Li Y, Hu Y, Yu J, Ma T. Retrospective analysis of laboratory testing in 54 patients with severe- or critical-type 2019 novel coronavirus pneumonia. Lab Invest. 2020;100:794-800. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 57] [Cited by in RCA: 67] [Article Influence: 13.4] [Reference Citation Analysis (0)] |
| 82. | Chu H, Bai T, Chen L, Hu L, Xiao L, Yao L, Zhu R, Niu X, Li Z, Zhang L, Han C, Song S, He Q, Zhao Y, Zhu Q, Chen H, Schnabl B, Yang L, Hou X. Multicenter Analysis of Liver Injury Patterns and Mortality in COVID-19. Front Med (Lausanne). 2020;7:584342. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 23] [Cited by in RCA: 22] [Article Influence: 4.4] [Reference Citation Analysis (0)] |
| 83. | Li Y, Xie Z, Lin W, Cai W, Wen C, Guan Y, Mo X, Wang J, Wang Y, Peng P, Chen X, Hong W, Xiao G, Liu J, Zhang L, Hu F, Li F, Zhang F, Deng X, Li L. An exploratory randomized, controlled study on the efficacy and safety of lopinavir/ritonavir or arbidol treating adult patients hospitalized with mild/moderate COVID-19 (ELACOI). 2020 Preprint. Available from: medRxiv:2020.03.19.20038984. [DOI] [Full Text] |
| 84. | Kumar P, Kulkarni A, Sharma M, Rao PN, Reddy DN. Favipiravir-induced Liver Injury in Patients with Coronavirus Disease 2019. J Clin Transl Hepatol. 2021;9:276-278. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 6] [Cited by in RCA: 6] [Article Influence: 1.5] [Reference Citation Analysis (0)] |
| 85. | Martinez MA, Vuppalanchi R, Fontana RJ, Stolz A, Kleiner DE, Hayashi PH, Gu J, Hoofnagle JH, Chalasani N. Clinical and histologic features of azithromycin-induced liver injury. Clin Gastroenterol Hepatol. 2015;13:369-376.e3. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 85] [Cited by in RCA: 85] [Article Influence: 8.5] [Reference Citation Analysis (0)] |
| 86. | Oyelade T, Alqahtani J, Canciani G. Prognosis of COVID-19 in Patients with Liver and Kidney Diseases: An Early Systematic Review and Meta-Analysis. Trop Med Infect Dis. 2020;5. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 113] [Cited by in RCA: 110] [Article Influence: 22.0] [Reference Citation Analysis (0)] |
| 87. | Grein J, Ohmagari N, Shin D, Diaz G, Asperges E, Castagna A, Feldt T, Green G, Green ML, Lescure FX, Nicastri E, Oda R, Yo K, Quiros-Roldan E, Studemeister A, Redinski J, Ahmed S, Bernett J, Chelliah D, Chen D, Chihara S, Cohen SH, Cunningham J, D'Arminio Monforte A, Ismail S, Kato H, Lapadula G, L'Her E, Maeno T, Majumder S, Massari M, Mora-Rillo M, Mutoh Y, Nguyen D, Verweij E, Zoufaly A, Osinusi AO, DeZure A, Zhao Y, Zhong L, Chokkalingam A, Elboudwarej E, Telep L, Timbs L, Henne I, Sellers S, Cao H, Tan SK, Winterbourne L, Desai P, Mera R, Gaggar A, Myers RP, Brainard DM, Childs R, Flanigan T. Compassionate Use of Remdesivir for Patients with Severe Covid-19. N Engl J Med. 2020;382:2327-2336. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 1926] [Cited by in RCA: 1884] [Article Influence: 376.8] [Reference Citation Analysis (0)] |
| 88. | Carothers C, Birrer K, Vo M. Acetylcysteine for the Treatment of Suspected Remdesivir-Associated Acute Liver Failure in COVID-19: A Case Series. Pharmacotherapy. 2020;40:1166-1171. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 45] [Cited by in RCA: 50] [Article Influence: 10.0] [Reference Citation Analysis (0)] |
| 89. | Chen X, Jiang Q, Ma Z, Ling J, Hu W, Cao Q, Mo P, Yao L, Yang R, Gao S, Gui X, Hou W, Xiong Y, Li J, Zhang Y. Clinical Characteristics of Hospitalized Patients with SARS-CoV-2 and Hepatitis B Virus Co-infection. Virol Sin. 2020;35:842-845. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 34] [Cited by in RCA: 64] [Article Influence: 12.8] [Reference Citation Analysis (0)] |
| 90. | Fix OK, Hameed B, Fontana RJ, Kwok RM, McGuire BM, Mulligan DC, Pratt DS, Russo MW, Schilsky ML, Verna EC, Loomba R, Cohen DE, Bezerra JA, Reddy KR, Chung RT. Clinical Best Practice Advice for Hepatology and Liver Transplant Providers During the COVID-19 Pandemic: AASLD Expert Panel Consensus Statement. Hepatology. 2020;72:287-304. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 280] [Cited by in RCA: 422] [Article Influence: 84.4] [Reference Citation Analysis (0)] |
| 91. | Abdulrab S, Al-Maweri S, Halboub E. Ursodeoxycholic acid as a candidate therapeutic to alleviate and/or prevent COVID-19-associated cytokine storm. Med Hypotheses. 2020;143:109897. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 24] [Cited by in RCA: 38] [Article Influence: 7.6] [Reference Citation Analysis (0)] |
| 92. | American Society for Gastreointestinal Endoscopy. Guidance For GI Endoscopy and Practice Operations During The Covid-19 Pandemic 2021. [cited 12 April 2021]. In: American Society for Gastreointestinal Endoscopy [Internet]. Avaliable from: https://www.asge.org/docs/default-source/default-document-library/guidance-for-gi-endoscopy-and-practice-operations-during-the-covid-19-pandemic_updated_final-march-2021.pdf. |
| 93. | Mantovani A, Beatrice G, Dalbeni A. Coronavirus disease 2019 and prevalence of chronic liver disease: A meta-analysis. Liver Int. 2020;40:1316-1320. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 107] [Cited by in RCA: 120] [Article Influence: 24.0] [Reference Citation Analysis (0)] |
| 94. | Kim D, Adeniji N, Latt N, Kumar S, Bloom PP, Aby ES, Perumalswami P, Roytman M, Li M, Vogel AS, Catana AM, Wegermann K, Carr RM, Aloman C, Chen VL, Rabiee A, Sadowski B, Nguyen V, Dunn W, Chavin KD, Zhou K, Lizaola-Mayo B, Moghe A, Debes J, Lee TH, Branch AD, Viveiros K, Chan W, Chascsa DM, Kwo P, Dhanasekaran R. Predictors of Outcomes of COVID-19 in Patients With Chronic Liver Disease: US Multi-center Study. Clin Gastroenterol Hepatol. 2021;19:1469-1479.e19. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 176] [Cited by in RCA: 184] [Article Influence: 46.0] [Reference Citation Analysis (0)] |
| 95. | Guo H, Zhang Z, Zhang Y, Liu Y, Wang J, Qian Z, Zou Y, Lu H. Analysis of liver injury factors in 332 patients with COVID-19 in Shanghai, China. Aging (Albany NY). 2020;12:18844-18852. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 13] [Cited by in RCA: 15] [Article Influence: 3.0] [Reference Citation Analysis (0)] |
| 96. | Bajaj JS, Garcia-Tsao G, Biggins SW, Kamath PS, Wong F, McGeorge S, Shaw J, Pearson M, Chew M, Fagan A, de la Rosa Rodriguez R, Worthington J, Olofson A, Weir V, Trisolini C, Dwyer S, Reddy KR. Comparison of mortality risk in patients with cirrhosis and COVID-19 compared with patients with cirrhosis alone and COVID-19 alone: multicentre matched cohort. Gut. 2021;70:531-536. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 115] [Cited by in RCA: 176] [Article Influence: 35.2] [Reference Citation Analysis (0)] |
| 97. | Marjot T, Moon AM, Cook JA, Abd-Elsalam S, Aloman C, Armstrong MJ, Pose E, Brenner EJ, Cargill T, Catana MA, Dhanasekaran R, Eshraghian A, García-Juárez I, Gill US, Jones PD, Kennedy J, Marshall A, Matthews C, Mells G, Mercer C, Perumalswami PV, Avitabile E, Qi X, Su F, Ufere NN, Wong YJ, Zheng MH, Barnes E, Barritt AS 4th, Webb GJ. Outcomes following SARS-CoV-2 infection in patients with chronic liver disease: An international registry study. J Hepatol. 2021;74:567-577. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 399] [Cited by in RCA: 384] [Article Influence: 96.0] [Reference Citation Analysis (0)] |
| 98. | Kushner T, Cafardi J. Chronic Liver Disease and COVID-19: Alcohol Use Disorder/Alcohol-Associated Liver Disease, Nonalcoholic Fatty Liver Disease/Nonalcoholic Steatohepatitis, Autoimmune Liver Disease, and Compensated Cirrhosis. Clin Liver Dis (Hoboken). 2020;15:195-199. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 27] [Cited by in RCA: 38] [Article Influence: 7.6] [Reference Citation Analysis (0)] |
| 99. | Sahu T, Mehta A, Ratre YK, Jaiswal A, Vishvakarma NK, Bhaskar LVKS, Verma HK. Current understanding of the impact of COVID-19 on gastrointestinal disease: Challenges and openings. World J Gastroenterol. 2021;27:449-469. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in CrossRef: 18] [Cited by in RCA: 15] [Article Influence: 3.8] [Reference Citation Analysis (0)] |
| 100. | Zhou YJ, Zheng KI, Wang XB, Yan HD, Sun QF, Pan KH, Wang TY, Ma HL, Chen YP, George J, Zheng MH. Younger patients with MAFLD are at increased risk of severe COVID-19 illness: A multicenter preliminary analysis. J Hepatol. 2020;73:719-721. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 104] [Cited by in RCA: 106] [Article Influence: 21.2] [Reference Citation Analysis (0)] |
| 101. | Lefere S, Tacke F. Macrophages in obesity and non-alcoholic fatty liver disease: Crosstalk with metabolism. JHEP Rep. 2019;1:30-43. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 123] [Cited by in RCA: 190] [Article Influence: 31.7] [Reference Citation Analysis (0)] |
| 102. | Li L, Li S, Xu M, Yu P, Zheng S, Duan Z, Liu J, Chen Y, Li J. Risk factors related to hepatic injury in patients with corona virus disease 2019. 2020 Preprint. Available from: medRxiv:2020.02.28.20028514. [DOI] [Full Text] |
| 103. | Valenti L, Jamialahmadi O, Romeo S. Lack of genetic evidence that fatty liver disease predisposes to COVID-19. J Hepatol. 2020;73:709-711. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 20] [Cited by in RCA: 26] [Article Influence: 5.2] [Reference Citation Analysis (0)] |
| 104. | Severe Covid-19 GWAS Group; Ellinghaus D, Degenhardt F, Bujanda L, Buti M, Albillos A, Invernizzi P, Fernández J, Prati D, Baselli G, Asselta R, Grimsrud MM, Milani C, Aziz F, Kässens J, May S, Wendorff M, Wienbrandt L, Uellendahl-Werth F, Zheng T, Yi X, de Pablo R, Chercoles AG, Palom A, Garcia-Fernandez AE, Rodriguez-Frias F, Zanella A, Bandera A, Protti A, Aghemo A, Lleo A, Biondi A, Caballero-Garralda A, Gori A, Tanck A, Carreras Nolla A, Latiano A, Fracanzani AL, Peschuck A, Julià A, Pesenti A, Voza A, Jiménez D, Mateos B, Nafria Jimenez B, Quereda C, Paccapelo C, Gassner C, Angelini C, Cea C, Solier A, Pestaña D, Muñiz-Diaz E, Sandoval E, Paraboschi EM, Navas E, García Sánchez F, Ceriotti F, Martinelli-Boneschi F, Peyvandi F, Blasi F, Téllez L, Blanco-Grau A, Hemmrich-Stanisak G, Grasselli G, Costantino G, Cardamone G, Foti G, Aneli S, Kurihara H, ElAbd H, My I, Galván-Femenia I, Martín J, Erdmann J, Ferrusquía-Acosta J, Garcia-Etxebarria K, Izquierdo-Sanchez L, Bettini LR, Sumoy L, Terranova L, Moreira L, Santoro L, Scudeller L, Mesonero F, Roade L, Rühlemann MC, Schaefer M, Carrabba M, Riveiro-Barciela M, Figuera Basso ME, Valsecchi MG, Hernandez-Tejero M, Acosta-Herrera M, D'Angiò M, Baldini M, Cazzaniga M, Schulzky M, Cecconi M, Wittig M, Ciccarelli M, Rodríguez-Gandía M, Bocciolone M, Miozzo M, Montano N, Braun N, Sacchi N, Martínez N, Özer O, Palmieri O, Faverio P, Preatoni P, Bonfanti P, Omodei P, Tentorio P, Castro P, Rodrigues PM, Blandino Ortiz A, de Cid R, Ferrer R, Gualtierotti R, Nieto R, Goerg S, Badalamenti S, Marsal S, Matullo G, Pelusi S, Juzenas S, Aliberti S, Monzani V, Moreno V, Wesse T, Lenz TL, Pumarola T, Rimoldi V, Bosari S, Albrecht W, Peter W, Romero-Gómez M, D'Amato M, Duga S, Banales JM, Hov JR, Folseraas T, Valenti L, Franke A, Karlsen TH. Genomewide Association Study of Severe Covid-19 with Respiratory Failure. N Engl J Med. 2020;383:1522-1534. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 1512] [Cited by in RCA: 1389] [Article Influence: 277.8] [Reference Citation Analysis (0)] |
| 105. | World Health Organization. Cancer.2020. [cited 12 April 2021]. In: World Health Organization [Internet]. Available from: https://www.who.int/news-room/fact-sheets/detail/cancer. |
| 106. | Shiina S, Gani RA, Yokosuka O, Maruyama H, Nagamatsu H, Payawal DA, Dokmeci AK, Lesmana LA, Tanwandee T, Lau G, Sarin SK, Omata M. APASL practical recommendations for the management of hepatocellular carcinoma in the era of COVID-19. Hepatol Int. 2020;14:920-929. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 12] [Cited by in RCA: 25] [Article Influence: 5.0] [Reference Citation Analysis (0)] |
| 107. | World Health Organization. Clinical management of severe acute respiratory infection (SARI) when COVID-19 disease is suspected: interim guidance, 13 March 2020. [cited 12 April 2021]. In: World Health Organization [Internet]. Available from: https://apps.who.int/iris/handle/10665/331446. |
| 108. | APASL Covid-19 Task Force, Lau G, Sharma M. Clinical practice guidance for hepatology and liver transplant providers during the COVID-19 pandemic: APASL expert panel consensus recommendations. Hepatol Int. 2020;14:415-428. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 61] [Cited by in RCA: 65] [Article Influence: 13.0] [Reference Citation Analysis (0)] |
| 109. | D'Antiga L. Coronaviruses and Immunosuppressed Patients: The Facts During the Third Epidemic. Liver Transpl. 2020;26:832-834. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 479] [Cited by in RCA: 492] [Article Influence: 98.4] [Reference Citation Analysis (0)] |
| 110. | Bhoori S, Rossi RE, Citterio D, Mazzaferro V. COVID-19 in long-term liver transplant patients: preliminary experience from an Italian transplant centre in Lombardy. Lancet Gastroenterol Hepatol. 2020;5:532-533. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 198] [Cited by in RCA: 205] [Article Influence: 41.0] [Reference Citation Analysis (0)] |
| 111. | Becchetti C, Zambelli MF, Pasulo L, Donato MF, Invernizzi F, Detry O, Dahlqvist G, Ciccarelli O, Morelli MC, Fraga M, Svegliati-Baroni G, van Vlierberghe H, Coenraad MJ, Romero MC, de Gottardi A, Toniutto P, Del Prete L, Abbati C, Samuel D, Pirenne J, Nevens F, Dufour JF; COVID-LT group. COVID-19 in an international European liver transplant recipient cohort. Gut. 2020;69:1832-1840. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 113] [Cited by in RCA: 122] [Article Influence: 24.4] [Reference Citation Analysis (0)] |
| 112. | Sahin TT, Akbulut S, Yilmaz S. COVID-19 pandemic: Its impact on liver disease and liver transplantation. World J Gastroenterol. 2020;26:2987-2999. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in CrossRef: 56] [Cited by in RCA: 48] [Article Influence: 9.6] [Reference Citation Analysis (0)] |
| 113. | Gruttadauria S; Italian Board of Experts in Liver Transplantation (I-BELT) Study Group, The Italian Society of Organ Transplantation (SITO). Preliminary Analysis of the Impact of the Coronavirus Disease 2019 Outbreak on Italian Liver Transplant Programs. Liver Transpl. 2020;26:941-944. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 21] [Cited by in RCA: 21] [Article Influence: 4.2] [Reference Citation Analysis (0)] |
| 114. | Tschopp J, L'Huillier AG, Mombelli M, Mueller NJ, Khanna N, Garzoni C, Meloni D, Papadimitriou-Olivgeris M, Neofytos D, Hirsch HH, Schuurmans MM, Müller T, Berney T, Steiger J, Pascual M, Manuel O, van Delden C; Swiss Transplant Cohort Study (STCS). First experience of SARS-CoV-2 infections in solid organ transplant recipients in the Swiss Transplant Cohort Study. Am J Transplant. 2020;20:2876-2882. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 78] [Cited by in RCA: 87] [Article Influence: 17.4] [Reference Citation Analysis (0)] |
| 115. | Qin J, Wang H, Qin X, Zhang P, Zhu L, Cai J, Yuan Y, Li H. Perioperative Presentation of COVID-19 Disease in a Liver Transplant Recipient. Hepatology. 2020;72:1491-1493. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 85] [Cited by in RCA: 88] [Article Influence: 17.6] [Reference Citation Analysis (0)] |
| 116. | Riechelmann RP, Peixoto RD, Fernandes GDS, Weschenfelder RF, Prolla G, Filho DR, Andrade AC, Crosara M, Rego JFM, Gansl RC, Coimbra F, Aguiar S Jr, Carvalho E, Hoff PM, Coutinho AK. Evidence-based recommendations for gastrointestinal cancers during the COVID-19 pandemic by the Brazilian Gastrointestinal Tumours Group. Ecancermedicalscience. 2020;14:1048. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 7] [Cited by in RCA: 6] [Article Influence: 1.2] [Reference Citation Analysis (0)] |