INTRODUCTION
Disruption of normal sagittal alignment resulting in global sagittal imbalance can cause significant pain and impair ambulation directly and indirectly through compensatory mechanisms. When global sagittal imbalance arises because a long spinal segment is fixed or fused in a hyperkyphotic and/or hypolordotic position, the patient is said to have fixed sagittal imbalance. The causes of sagittal deformity are myriad, but most commonly include post-traumatic kyphosis, iatrogenic flat back syndrome, postlaminectomy kyphosis, degenerative lumbar kyphosis, and ankylosing spondylitis[1-3]. For many years, posterior column osteotomies, such as Smith-Petersen osteotomy and Ponte osteotomy, with or without release of the anterior aspect of the spine, were the procedure of choice to shorten the posterior column and thereby reduce fixed kyphosis[4]. A surgeon can usually achieve 5°-10° of sagittal correction with each Smith-Petersen osteotomy; additional correction is limited by anatomical constraints of the anterior column.
In 1985, Thomasen[5] first described the three-column posterior wedge osteotomy for the management of fixed sagittal plane deformities in patients with ankylosing spondylitis. The pedicle subtraction osteotomy (PSO) is typically performed at L3 as these vertebrae are the normal apex of lumbar lordosis. It is also safer to perform the osteotomy at one of these levels, as they are caudad to the conus medullaris[6]. The technique involves a transpedicular vertebral wedge resection extending from the posterior elements through the pedicles and into the anterior cortex of the vertebral body. A PSO can usually produce 30°-40° of lordosis at each level at which osteotomy is performed and, when complete, results in bone-on-bone contact throughout all three columns of the spine[7-9]. Unfortunately, the procedure is technically much more demanding than the posterior column osteotomies, so PSO is not commonly performed despite the fact that PSO can better restore lumbar lordosis and improve sagittal balance. Several studies have reported outcomes and complications associated with the procedure[10-14]. The literature focusing on PSO for fixed sagittal imbalance patients is reviewed. The long-term overall outcomes, surgical tips to reduce the complications and suggestions for their proper application are also provided.
Normal sagittal balance and imbalance
In a patient with normal sagittal balance, the center of the C7 vertebral body is in line with the posterior superior corner of S1 and the longitudinal axis of the femur. This normal relationship is demonstrated by the C7 plumb line, also known as the sagittal vertical axis (SVA), which is extended from the center of the C7 body down to the posterosuperior corner of the sacrum at the L5-S1 disc, and the longitudinal axis of femur. In patients with positive sagittal imbalance, the C7 center is shifted forward in relationship to the femoral axis line.
There are two general types of spinal imbalance in the sagittal plane: type 1 and type 2[1]. A type 1 imbalance refers to a condition in which the patient has a segmental or regional imbalance in the sagittal plane of the spine but still has a balanced spine as defined by a plumb line from C7 that falls over the L5-S1 disc. These patients typically have a short segment that is hyperkyphotic and results in the more cephalad or caudad vertebrae having to compensate with lordosis.
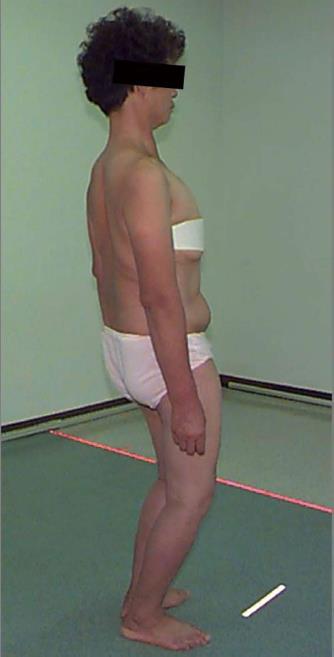
A type 2 imbalance refers to a global imbalance whereby the plumb line falls > 5 cm in front of the L5-S1 disc. A spine with a type 2 imbalance is unable to compensate for the deformity and the patient tends to flex the hips and knees to maintain a proper balance and horizontal gaze (Figure 1). Therefore, during an examination, it is important for these patients to stand with the hips and knees straight so that the uncompensated spinal deformity can be assessed.
Figure 1 A clinical photograph of a 62-year-old woman who presented with severe lower back pain.
To compensate for the lumbar kyphotic deformity, the patient tends to flex the hips and knees to maintain a proper balance and horizontal gaze.
PSO
Surgical decision making for PSO
The PSO is useful for treating patients with ankylosing spondylitis and an imbalance in the sagittal plane of the spine[5,6,14-16]. Unlike the Smith-Petersen osteotomy, the PSO is mainly useful for deformities with an apex in the lumbar spine. The PSO is historically performed at L2 or L3 and an ideal candidate for the procedure typically has a positive sagittal imbalance of > 12 cm[6,14-17]. The PSO is also indicated for patients who have had a circumferential fusion along multiple vertebrae, which prevents the performance of a Smith-Petersen osteotomy since osteoclasis cannot be obtained through a fused intervertebral disc.
Surgical decision making for PSO includes: (1) thoracic vs lumbar PSO at L2 vs L3 vs L4 vs L5; (2) stop at upper thoracic vs thoracolumbar junction; (3) interbody fusion or not for virgin spine vs PSO through previous fusion; and (4) consideration of operating table: OSI vs Maquett.
Operative technique
Our decision policy of PSO level depended on the site of the most significant pathological entity. For instance, lumbar PSO was indicated for the treatment of iatrogenic lumbar flat back syndrome and degenerative global sagittal imbalance, whereas thoracic PSO was indicated for isolated thoracic kyphosis such as posttraumatic kyphosis. The PSO was performed on patients for whom posterior column osteotomies such as Ponte/Smith-Petersen osteotomies alone would be inadequate, as at least 30° of corrective lordosis was required to correct their sagittal imbalances. Osteotomy was performed on the lumbar spine in most cases, typically at L3. The osteotomy size was based on preoperative standing radiograph measurements of sagittal imbalance, with the surgical goal being restoration of normal sagittal balance with the C7 plumb line falling at or within 5 cm of the posterior-superior corner of S1. A multi-segmental pedicle screw technique was used for posterior spinal instrumentation. All patients were positioned prone on a Jackson frame and were neurophysiologically monitored using somatosensory and motor evoked potentials[18].
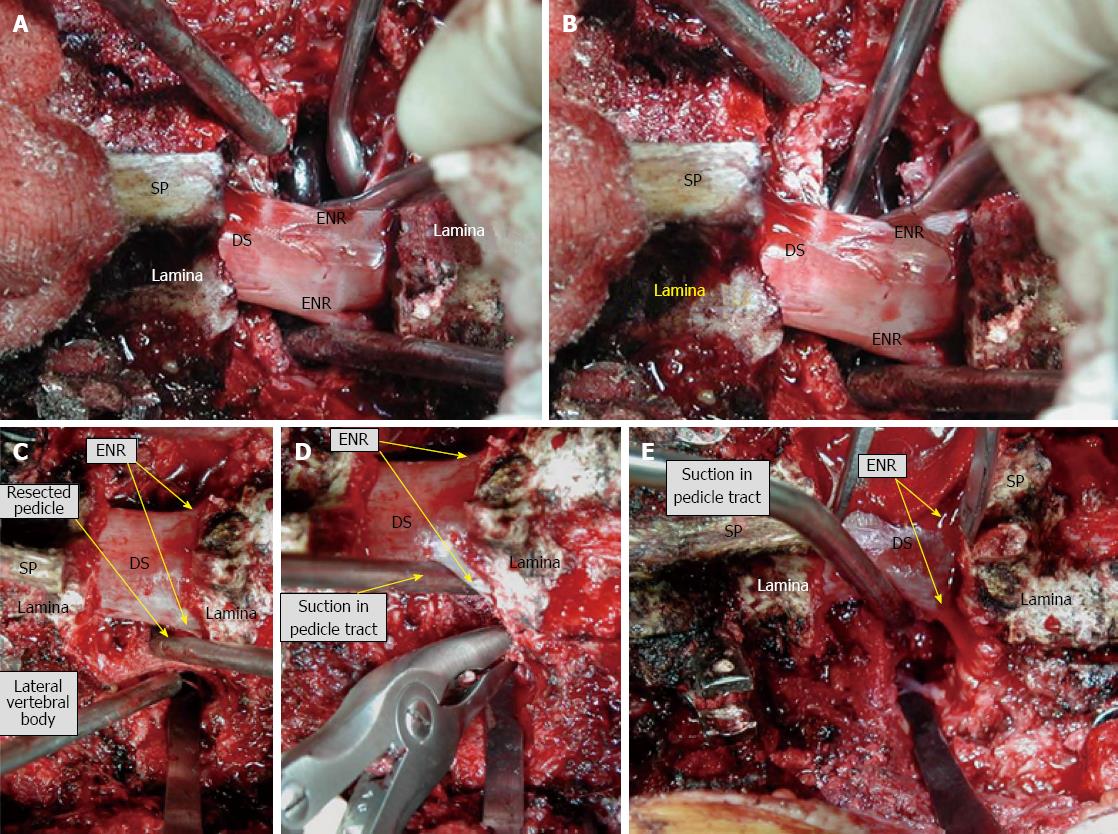
Although various descriptions of PSO exist, the first step in the technique consisted of removing all posterior elements at the level of planned correction, including the spinous process, the lamina, and the superior and inferior facets adjacent to the pedicle. In addition, the cephalad and caudal laminae were undercut to avoid iatrogenic canal stenosis or neural impingement during osteotomy closure. Next, pedicles were taken down to the level of the posterior vertebral body. The vertebral body was then decancellated through each pedicle. In most instances, a temporary rod was placed across the osteotomy site to prevent subluxation or premature osteotomy closure. A wedge-shaped portion of the lateral vertebral body walls was removed through the pedicle (Figure 2). A reverse-angled curette or Woodson elevator was used to thin and fracture the posterior cortex of the vertebral body underlying the spinal canal into the wedge-shaped defect. A posterior compression force was applied to the spine facilitating spinal hyperextension across the osteotomy site hinging on the anterior margin of the vertebral body. Typically, this maneuver was performed by changing the bolsters on the Jackson frame to allow extension across the osteotomy, with further compression and correction obtained using compression techniques across the posterior instrumentation. In this step, a central hook/rod construct can be helpful[19]. It not only adds fixation strength to the overall construct but avoids placement of undue stress on pedicle screws that can lead to screw loosening and potential fixation failure. When the middle and posterior column bone defects are closed, the length of the anterior vertebral cortex remains unchanged (Figure 3).
Figure 2 Intraoperative photographs of pedicle subtraction osteotomy.
To summarize, exposure transverse to transverse/laminectomy/resection of transverse process/pedicle resection (A and B)/decancellation through pedicle or osteotomy of the pedicle after exposure of the lateral border (C and D)/resect the posterior vertebral body wall (E) and lateral wall if you did not take it/check the mobility and closure using gravity/instrument/operation table. SP: Spinous process; DS: Dural sac; ENR: Exiting nerve root.
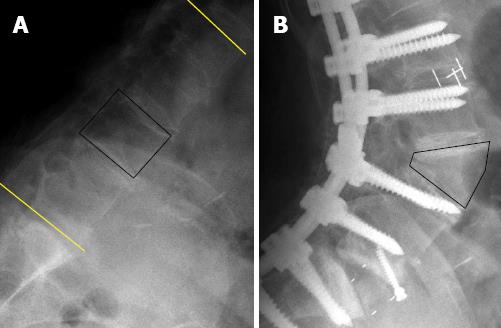
Figure 3 Pre- and post-operative lateral plain radiographs of a 70-year-old woman who underwent pedicle subtraction osteotomy at the L3 vertebra.
Kyphotic lumbar spine (A) and hyperlordotic lumbar spine after pedicle subtraction osteotomy (B). Note that the length of the anterior vertebral cortex remains unchanged even after the shortening of the middle and posterior spinal columns.
With every step of correction/compression, careful attention was paid to ensure the central canal and exiting nerve roots were not compressed. After ensuring that exiting nerve roots were free, the final spinal contour was maintained with segmental rod fixation for which cantilever forces were employed. After completing the fixation procedure, autografts and allografts were placed over the laminae, facet joints and transverse processes. Anterior-column support is known to decrease the pseudarthrosis rate associated with long-segment posterior fusions. In all cases where fusions extended to the sacrum, anterior column support was provided at L5-S1 by a transforaminal lumbar interbody fusion or an anterior lumbar interbody fusion (Figure 4). In patients with severe osteopenia/osteoporosis or in those with histories of pseudarthrosis, a demineralized bone matrix or bone morphogenetic protein was considered to supplement fusion. A 3-6 mo course of thoracolumbosacral orthosis or lumbosacral orthosis with a thoracic extension pad was typically prescribed for any patient in whom the PSO was supported only by segmental posterior instrumentation.
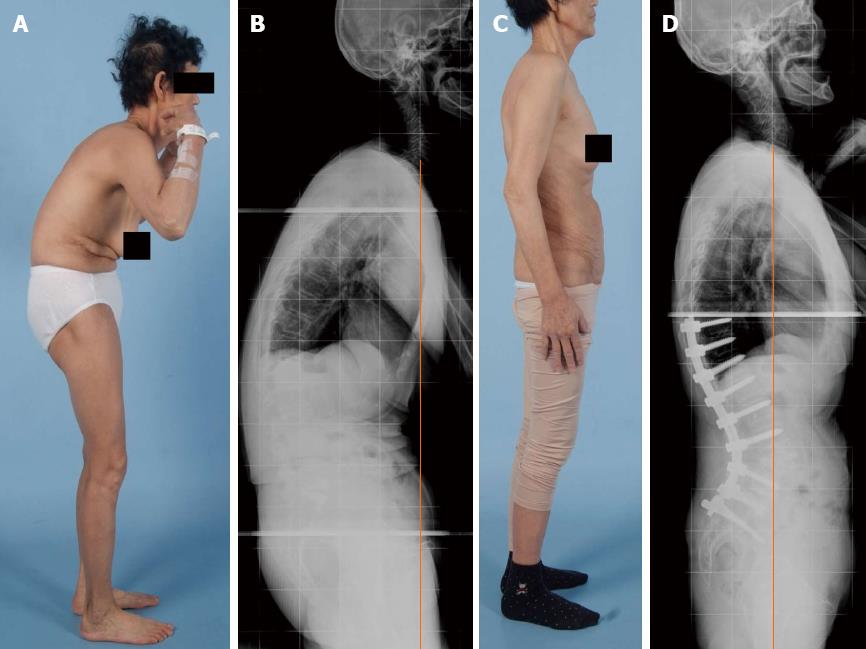
Figure 4 Pre- and post-operative clinical photographs (A)/radiographs (B) of a 65-year-old woman, revealing degenerative lumbar kyphosis with global sagittal imbalance.
The patient presented with intolerable lower back pain and stooping symptoms. She underwent L4 pedicle subtraction osteotomy with anterior column support, and the plain film obtained at her most recent follow-up examination shows dramatic improvement in global sagittal alignment as well as the lumbar kyphosis (C and D).
Sagittal rebalancing after PSO
For ideal preoperative surgical planning, spine surgeons should consider sagittal spinopelvic alignment change after lumbar PSO. Kim et al[20] reported the change with an analysis of 114 patients who had undergone lumbar PSO. The authors found a single level lumbar PSO 34° increase in lumbar lordosis, 15° contribution on thoracic kyphosis, 9° increase in sacral slope, 8° decrease in pelvic tilt and 8.9 cm shortening in SVA at 4.4 years after surgery. Because sagittal rebalancing such as an increase in thoracic kyphosis occurs after lumbar PSO, spine surgeons usually need more correction angle to achieve optimal sagittal balance.
Clinical outcomes of PSO
The PSO is advantageous in that it can produce substantial correction at a single level, it results in successful bone union due to the three columns of bony contact, and it can be done without the use of a supplemental anterior approach[21]. In a prospective study in which thirty patients underwent a Smith-Petersen osteotomy and forty-one patients underwent a PSO, only 39% of the patients treated with the PSO required a concomitant anterior arthrodesis compared with 87% of those treated with the Smith-Petersen osteotomy[17]. Kim et al[16] retrospectively analyzed their results at a minimum of five years following PSOs in thirty-five patients. Between two and five years postoperatively, the authors did not see any significant radiographic changes in thoracic kyphosis or lumbar lordosis (P = 0.38 and 0.84, respectively). Eight patients (22.8%) subsequently underwent revision procedures for treatment of pseudarthrosis. The Oswestry Disability Index (ODI) and Scoliosis Research Society outcome scores between two and five years postoperatively also did not change significantly. A sagittal vertical axis of < 8 cm at the time of final follow-up was significantly associated with a better Scoliosis Research Society outcome score (P = 0.038). The authors concluded that PSO can provide satisfactory clinical and radiographic outcomes at a minimum of five years postoperatively.
In a retrospective study comparing anterior-posterior circumferential fusion to PSO in twenty-six patients with posttraumatic kyphosis who were followed for a mean of 3.5 years, Suk et al[22] found that the PSO had a shorter operative time (215 min compared with 351 min), less intraoperative bleeding, and more correction of the kyphosis between the preoperative and postoperative examinations (25.7° compared with 11.2°). In our recent study with a long-term follow-up data, we analyzed 13 consecutive PSO-treated patients presenting with fixed sagittal imbalances from 1999 to 2006[14]. The median follow-up period was 73 mo (range 41-114 mo). The average preoperative ODI score was 55.4 ± 13.6, and the mean postoperative ODI score 30.2 ± 16.5. Improvement after surgery was statistically significant (P < 0.001). Subjective evaluation of back pain showed that nine patients perceived improvement, three no change, and one an aggravation. Outcome of leg pain demonstrated that seven patients perceived improvement, five no change, and one an aggravation. The patients who experienced suboptimal outcome of leg pain had arthralgic rather than neuralgic pain. Subjective assessment of stooping symptoms showed that no patient perceived a result as aggravated even if the patient experienced proximal junctional kyphosis (PJK) or rod fracture. Statistical analysis revealed that ODI score reduction was significantly related to the postoperative C7 plumb line value (P = 0.003), but not to lumbar lordosis, thoracic kyphosis or PJK.
Complications of PSO
Postoperative sagittal decompensation following PSO is a problematic event. Kim et al[23] found that the prevalence of sagittal decompensation following PSO for adult patients with sagittal imbalance was 29% and associated risk factors were immediate postoperative SVA > 8 cm, the sum of TK, LL and pelvic incidence Cobb angles > 45°, the sum of TK and LL Cobb angles < 25°, T12 horizontal angle > 15°, LL Cobb angle increase ≥ 40°, associated comorbidities, age at surgery > 55 years, uppermost instrumented vertebra below T8, and preoperative SVA > 15 cm. Multiple factors should be considered to obtain optimal correction while minimizing the risk of developing sagittal decompensation.
Pedicle subtraction osteotomies are technically demanding and involve substantial mobilization of the neural tissue, and the blood loss is greater than that associated with the Smith-Petersen osteotomy[24,25]. A retrospective analysis of data obtained prospectively in a study of forty-six patients who were sixty years of age or older showed that patients who underwent a PSO were seven times more likely to have at least one major complication compared with patients who underwent a different spinal procedure (OR = 6.96; 95%CI: 1.10-79)[26]. Major complications included neurological deficits, deep wound infection, pulmonary embolus, pneumonia and myocardial infarction. Increasing age was a significant predictor of a complication (P < 0.05). The researchers concluded that the age at which patients are able to tolerate a major procedure such as a PSO may be lower than the age at which they can tolerate other common spinal procedures. Buchowski et al[27] reported the prevalence of intraoperative and postoperative neurological deficits to be 11.1% and the prevalence of permanent deficits to be 2.8% in a study of 108 patients who had undergone a PSO. In a study by Bridwell et al[11], five (15%) of thirty-three patients who had undergone a PSO for the treatment of an imbalance in the sagittal plane experienced a transient neurological deficit. In another study, Yang et al[13] found the prevalence of intraoperative or postoperative neurological deficits to be 4% (one of twenty-eight patients) after lumbar or thoracic PSO for the treatment of an imbalance in the sagittal plane. This single deficit was thought to be most likely due to nerve root compression.
In our recent investigation, no patient died or became permanently paraplegic as a result of surgery[14]. The incidence of perioperative transient neurological deficits was 7.6% (1 of 13 patients). Surgery was associated with 16 complications in 8 patients (61%); there were 3 intraoperative complications (dural tear, massive bleeding > 5000 mL), 3 perioperative complications (hypotension, cerebrospinal fluid leakage and spinal cord compression) and 10 late-onset postoperative complications (PJK with or without adjacent segment collapse, pseudoarthrosis including rod fracture and screw loosening). Complication frequencies were significantly higher in patients who rated clinical outcomes as aggravated or unchanged than in patients who rated outcomes as improved (P = 0.012).
Surgical tips to reduce the complications
Surgical and medical complications in earlier reports have included hypotension resulting from intraoperative massive bleeding, cerebrospinal fluid leakage, neurological injury, wound-related problems and nosocomial infections[13,27]. In our series, massive bleeding occurred in two patients with preoperative normal laboratory findings. An option to reduce the large blood loss known to be associated with all PSOs is vertebral body embolization or intraoperative administration of tranexamic acid, but this strategy awaits further clinical validation[28]. For safe and uneventful surgery, intraoperative neurophysiological monitoring should be performed by an experienced neurophysiologist or technician. Nevertheless, in our practice, one patient experienced transient paraplegia because a bone fragment caused spinal cord compression that was not detected by intraoperative neuromonitoring. In a previous analysis of 45 patients with ankylosing spondylitis, Kim et al[29] found transient postoperative radiculopathy in four patients and spinal cord compression caused by a bone fragment at T12 in one patient. Although it is unclear what mechanisms were responsible for development of neurological deficits in most previous reports, the problems were thought to arise from a combination of subluxation, residual dorsal impingement, dural buckling and spinal cord ischemia[10,13,27]. To reduce the risk of intraoperative or postoperative neurological deficits, some authors recommend central canal enlargement, careful osteotomy closure to prevent subluxation across the osteotomy site, limited osteotomy at the level of the spinal cord or conus medullaris, a wake-up test after osteotomy closure, and examination of all motor groups following surgery[10,11,13,27].
Most late-onset complications are related to kyphosis progression, pseudarthrosis and instrumentation failure. There was a higher incidence of complications in patients with compression fractures, caused mainly by osteoporosis, compared with patients with degenerative changes. Correcting kyphosis after a compression fracture is associated with a high risk of additional compression fractures in adjacent vertebrae. Some researchers have studied complications after PSO using the combined anterior-posterior approach and have emphasized the need for anterior reconstruction[11]. We found the PJK prevalence on median 6 year follow-up to be 23% and clinical outcomes were not adversely affected by PJK[14]. Kim et al[30] reported that 62 (39%) of 161 adult spinal deformity patients with segmental posterior spinal instrumented fusions were diagnosed with PJK at an average of 7.8 years postoperative follow-up. Concordant to our results, self-reported patient satisfaction was not adversely affected by PJK.
Several investigators have noted that titanium rods have the advantages of magnetic resonance imaging compatibility and tolerable rigidity, but are prone to microfracture propagation and are notch-sensitive in bending[13,31]. Stainless steel or cobalt chrome rods are thus used in most patients with spinal deformities, except for those with infections or tumors. Recently, we found that the use of a multi-rod construct is a safe, simple and effective method to provide increased stability across 3-column osteotomy sites in order to significantly prevent implant failure and symptomatic pseudarthrosis vs a standard 2-rod construct[32]. Thus, we strongly recommend using a multi-rod construct to stabilize 3-column osteotomy of the thoracic and lumbar spine.