Published online Jan 28, 2023. doi: 10.4329/wjr.v15.i1.10
Peer-review started: September 30, 2022
First decision: October 29, 2022
Revised: November 12, 2022
Accepted: January 9, 2023
Article in press: January 9, 2023
Published online: January 28, 2023
Processing time: 109 Days and 6.5 Hours
Despite the recent progress of medical technology in the diagnosis and treatment of tumors, pancreatic carcinoma remains one of the most malignant tumors, with extremely poor prognosis partly due to the difficulty in early and accurate imaging evaluation. This paper focuses on the research progress of magnetic resonance imaging, nuclear medicine molecular imaging and radiomics in the diagnosis of pancreatic carcinoma. We also briefly described the achievements of our team in this field, to facilitate future research and explore new technologies to optimize diagnosis of pancreatic carcinoma.
Core Tip: Pancreatic carcinoma remains high incidence and poor prognosis. Molecular imaging enables early and precise diagnosis, efficient assessment, non-invasive pathological classification. This paper aims to review the recent research progress of nuclear medicine, magnetic resonance imaging and radiomics in the diagnosis of pancreatic carcinoma, and also briefly describe our team's work in this field.
- Citation: Pang XX, Xie L, Yao WJ, Liu XX, Pan B, Chen N. Advancements of molecular imaging and radiomics in pancreatic carcinoma. World J Radiol 2023; 15(1): 10-19
- URL: https://www.wjgnet.com/1949-8470/full/v15/i1/10.htm
- DOI: https://dx.doi.org/10.4329/wjr.v15.i1.10
In the past decade, significant progress has been made in the medical technology for the treatment of cancers. However, the prognosis of pancreatic carcinoma remains extremely poor due to its insidious location, high malignancy, easy metastasis and rapid progression, which increases the difficulty of early and accurate assessment. Radical surgical resection rate of pancreatic cancer patients is less than 20%. Pancreatic carcinoma is also resistant to radiotherapy and chemotherapy. Moreover, targeted drug therapy, and cytotoxic T-lymphocyte-associated protein 4 and programmed death-1/programmed death-ligand 1 antibody immunotherapy are ineffective. The five-year survival rate of patients remains below 5%-9%, and the number of deaths is the fourth highest among malignant tumors[1]. Early and accurate diagnosis as well as efficacious assessment of pancreatic carcinoma have important clinical significance.
Conventional imaging techniques makes important significance in theranostic of pancreatic cancer; however, these technologies are still deficiencies yet. First of all, magnetic resonance (MR) and computed tomography (CT) only detects limited range with regional scan in clinical routine diagnosis of pancreatic carcinoma, therefore, many patients with distant metastasis are misdiagnosed or never diagnosed. Secondly, the rate of misdiagnosis was high in lymphatic metastasis by MR and CT scan. By reason of no static image provided, ultrasound examination is very unfavorable for reading in clinical work, even this method has the double advantage of real time imaging and radiation lessness. What's more, ultrasound is affected greatly by operators. Finally, some patients who cannot have a proper assessment in regional lymphatic metastasis, especially patients after chemotherapy, even whole body
Molecular imaging has advanced rapidly in recent years. It enables early and precise diagnosis, efficacy assessment, non-invasive pathological classification, and acts as an important "bridge" to achieve precise diagnosis and treatment[2]. It can meet clinical demands and better protect patient privacy compared to genetic testing. This paper aimed to review the recent research progress of nuclear medicine, magnetic resonance imaging, molecular imaging and radiomics in the diagnosis and treatment of pancreatic carcinoma, and also briefly describe our team's work in this field.
Nuclear medicine molecular imaging is based on the principle of injecting microscopic molecular probes into the body and selectively targeting them to appropriate sites based on different properties, in order to qualify or quantify organs, tissues or lesions for assessing diseases at the molecular level. Molecular imaging in nuclear medicine has made significant advances in the treatment of pancreatic carcinoma in recent years.
18F-FDG is a glucose analogue, which is rapidly taken up by the glucose-transporter on the cell surface after intravenous injection. Various tumor cells, including pancreatic carcinoma, and inflammatory cells in the tumor microenvironment absorb a large amount of 18F-FDG, but the uptake is influenced by various conditions and the underlying mechanisms are complex[3].
18F-FDG PET/CT has high specificity, accuracy and sensitivity in the diagnosis of pancreatic carcinoma, and has important clinical value in the diagnosis, staging, surgical indication and evaluation efficacy of pancreatic carcinoma[4]. 18F-FDG PET/CT is more sensitive and accurate than CT in detecting tumor metastasis, and its whole-body scan is beneficial for tumor staging[5]. This technique detected distant metastases in about one-third of pancreatic carcinoma patients and changed the staging of approximately 26.8% of patients[6]. Its standardized uptake value (SUV) quantification and the rate of change were significantly correlated with tumor size[7], malignancy[8], vascular invasion[9], and lymph node metastasis. In addition, 18F-FDG PET has significant value in efficacy assessment[10] and survival prediction[11]. For example, the patients with baseline SUV < 3.5 (and/or) SUV decrease ≥ 60% had better overall survival (OS) and progression-free survival (PFS)[12]. In locally advanced or metastatic pancreatic carcinoma, the PFS of patients with SUVmax < 6.8 was significantly longer than that of patients with SUVmax ≥ 6.8[13]. 18F-FDG PET/CT-guided radiotherapy with metabolic tumor volume and total lesion glycolysis (TLG) can be used as independent factors affecting prognosis[14]. Yamamoto et al[15] found that the early postoperative recurrence rate of pancreatic carcinoma in patients with SUVmax ≥ 6.0 was higher than that of patients with SUVmax < 6.0, and median OS of the former was lower than the latter (Table 1)[16].
| Year | Study type | Pancreatic cancer–all (n) | 18F-FDG PET-CT | CT | MRI | EUS | ||||
| Sens | Spec | Sens | Spec | Sens | Spec | Sens | Spec | |||
| 2016 | Retrospective study | 139-139 | 0.78 | – | 0.76 | – | – | – | – | – |
| 2014 | Retrospective study | 33–52 | 1 | 0.9 | 0.92 | 0.5 | 0.89 | 0.75 | 1 | 0.88 |
| 2009 | Meta-analysis | 38571 | 0.9 | 0.8 | – | – | – | – | 0.81 | 0.93 |
| 2017 | Meta-analysis | 3567–5399 | 0.89 | 0.7 | 0.9 | 0.87 | 0.93 | 0.89 | 0.91 | 0.86 |
| 2014 | Retrospective study | 80–91 | 0.68 | 0.73 | – | – | – | – | – | – |
| 2018 | Prospective study | 278–583 | 0.93 | 0.76 | 0.89 | 0.71 | – | – | – | – |
| 2015 | Retrospective study | 50–70 | 0.92 | 0.65 | 0.82 | 0.65 | – | – | – | – |
With the increasing application of 18F-FDG PET/CT in recent years, several shortcomings have been gradually revealed. First, as a non-tumor-specific imaging agent, 18F-FDG PET reflects glucose metabolism and is not directly related to the biological properties of the tumor. So non-neoplastic lesions such as inflammation, tuberculosis, or even non-specific uptake with increased glucose metabolism can lead to false positive results. Second, if the patient has high blood glucose levels, uses short-acting insulin or exercises, 18F-FDG can also lead to reduced sensitivity due to increased background uptake. In order to address these problems, nuclear medicine researchers have developed a series of more specific imaging agents for different targets.
The highly specific non-FDG molecular probes with different targets achieve accurate diagnosis of pancreatic carcinoma, and also enable non-invasive visualization of the expression of different receptors in tumors, facilitating individualized precision medicine. These imaging agents have been particularly successful in imaging of integrin receptor, somatostatin receptor, tumor-associated fibroblasts, etc. Our team has also conducted in-depth research on PD-L1-targeted imaging, non-radioactive molecular imaging and highly specific targeted radiotherapy.
Somatostatin receptor imaging is mainly used in pancreatic neuroendocrine tumors, with sensitivity of 86%-100% and specificity of 79%-100%[17]. The precursors of somatostatin receptor (SSTR) imaging agents are mainly Tyr(3)-octreotate, 1-Nal(3)-octreotide and D-Phe1-Tyr(3)-octreotide, which have different affinities for different somatostatin receptor subtypes[17]. The neuroendocrine tumors with high differentiation (G1-G2, Ki-67 < 10%) generally showed high expression of SSTR and positive SSTR imaging. Moreover, the degree of malignancy was low, the level of glycolysis was decreased, and the metabolism of FGD was only slightly increased or defective, which led to low sensitivity of 18F-FDG PET[18,19]. In contrast, due to the loss of SSTR and negative SSTR imaging, the increase of malignant degree led to increased glycolysis[20], high metabolism of FGD and increased sensitivity of 18F-FDG PET/CT. In addition to the above three SSTR agonists, SSTR inhibitors have other advantages such as several binding sites, low degradation rate and longer retention in tumors[21] (Table 2)[17].
| Clinical key points | |||
| Radiopharmaceuticals | Main indication | Diagnostic accuracy | False positive findings |
| 68Ga-TATE, TOC, NOC | Staging and restaging any non-insulinoma panNET case; detection of the unknown primary tumour site or early relapse; evaluation in-vivo SRE; selection for PRRT and/or cold SSA | Sensitivity: 86% to 100%; specificity: from 79% to 100% | Pancreatic uncinate process, accessory spleens (including intra-pancreatic, splenules, infectious/inflammatory findings, non-neuroendocrine tumours) |
| 18F-FDG | High grade G2, G3 and NEC; prognosis; rapid tumour progression in earlier diagnosed G1–G2 tumours | Sensitivity: 40% in G1, 60% in G2; 95% in G3 patients | Infectious/inflammatory findings, non-neuroendocrine tumours |
Cancer-associated fibroblasts (CAFs) are a major component of the mesenchyme surrounding epithelial cancer cells. Fibroblast activating protein (FAP) is a marker of CAFs. It is highly expressed in tumor stromal fibroblasts of most common human epithelial carcinomas, and has lower expression in normal tissues[22]. CAFs can form physical and metabolic barriers, which is partly responsible for the resistance of pancreatic carcinoma to chemotherapy and radiotherapy, by reducing the therapeutic effect of combined chemotherapy on pancreatic carcinoma[23]. High expression of CAFs in pancreatic carcinoma is associated with shorter OS and disease-free survival[24,25].
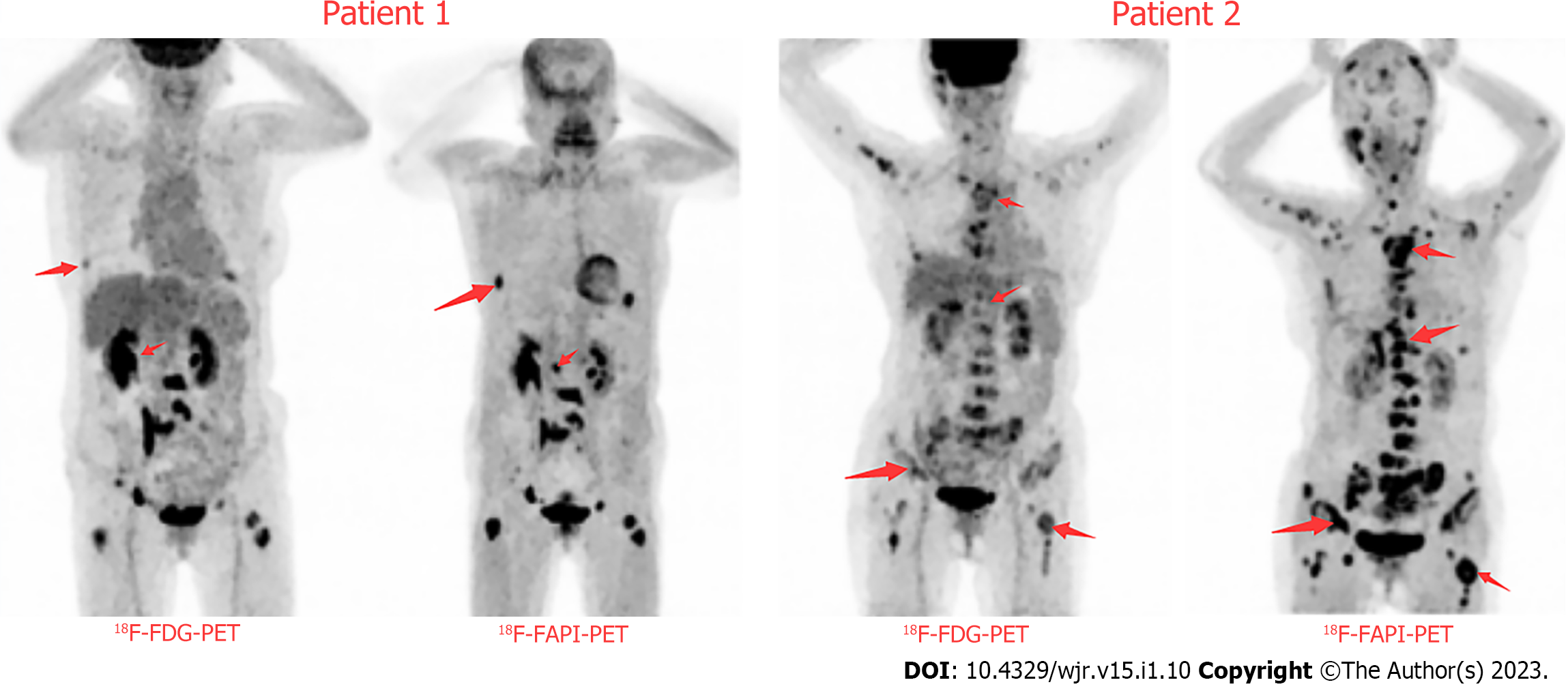
At present, the commonly used FAP-targeted imaging agents are various radionuclide-labeled small molecular FAP inhibitors (FAPIs), mainly FAPI-04, FAPI-21 and FAPI-46. The commonly used imaging agent 68Ga/18F-labeled FAPI-04 shows a significantly high uptake in pancreatic carcinoma, which has a good diagnostic efficacy for the primary focus of pancreatic carcinoma. In a comparative study of pancreatic carcinoma and pancreatitis, 68Ga-FAPI-04 PET/MR and 18F-FDG PET/CT positive rates were both 100%, but the former SUVmax was significantly higher than the latter SUVmax (P < 0.05). In addition,
Our team conducted a comparative study of FAPI and FDG imaging of pancreatic cancer (Figure 1), and redesigned FAPI based on new ideas, which is expected to exceed the existing FAPI-04, FAPI-21 and FAPI-46 in imaging and therapeutic effects. At present, chemical synthesis has been completed and radionuclides such as iodine and technetium have been labeled, and further cellular and animal experiments will be conducted soon.
Tumor neovascularization (angiogenesis) is necessary for maintaining the growth of malignant tumors, which plays a key role in tumor growth, invasion and metastasis, it is an important target for tumor diagnosis and treatment.
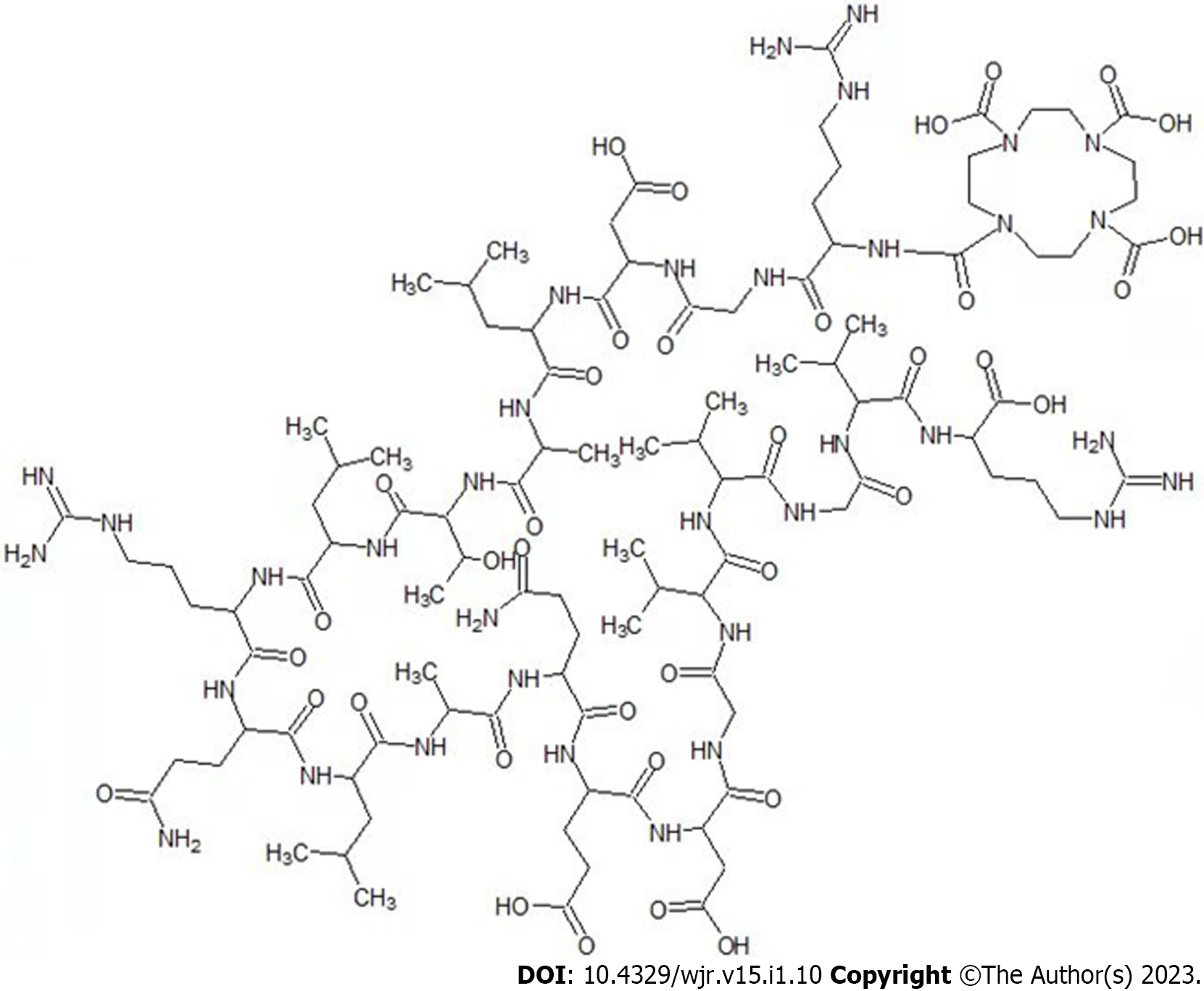
Integrin αvβ3 receptor is an important component of the 24 integrins, which is highly expressed on the cell surface of tumor neovascular endothelial cells and many solid tumors. However, it has low or no expression in mature vascular endothelial cells and most normal tissues and organs in healthy people, and plays an important role in angiogenesis, metastasis and tumor invasion[30,31]. The integrin αvβ3 receptor is highly expressed in about 60% of invasive pancreatic carcinomas, and the small polypeptide arginine glycine aspartic acid sequence (RGD) can be targeted to bind to αvβ3 receptor. Using radionuclide-labeled RGD peptides, such as 111In, 18F, 68Ga-labeled RGD, can be used to visualize and treat pancreatic carcinoma. Our research team has also studied RII and RIT based on molecular probes constructed by different radionuclide-labeled RGD and RRL peptides[32].
In addition to the integrin αvβ3 receptor, integrin αVβ6 is also highly expressed in pancreatic carcinoma[33]. Radiation molecular pancreatic probes constructed from the radionuclide 99mTc and 111In-labeled HHK can target αvβ6 with high specificity to achieve early diagnosis of pancreatic carcinoma and its metastases[34,35]. Based on previous studies, our research team redesigned the HHK peptide (Figure 2). The Gd-DOTA-HHK compound was obtained by chelating Gd3+, which can achieve high specific enhancement of tumor αvβ6 receptor during MRI T1WI scanning. Single photon emission CT imaging with high sensitivity and MRI with high soft tissue resolution combine perfectly to achieve high sensitivity and non-invasive visualization of αvβ6 targets at high resolution.
In addition, some researchers have explored the application of radionuclide labeling dopa, Exendin-4, CXCR4, and PSMA in pancreatic carcinoma.
MRI utilizes magnetic resonance to obtain electromagnetic signals from the human body, which can be reconstructed by computer to show the different chemical components in the same tissue. Because MRI has the advantages of high soft tissue resolution, non-radiation, unrestricted imaging depth and multi-sequence imaging, and with the development of MRI-specific imaging agents, it is possible to evaluate lesions from multiple dimensions of functional and molecular images by MRI.
Diffusion-weighted imaging (DWI) is the most widely used conventional MRI technique in addition to T1WI and T2WI. The diffusion movement of water molecules reflects the microstructure of the tumor, such as internal cell density, extracellular space and heterogeneity. When the cell density increases, edema, fibrosis, etc., affect the cell membrane function, which can be detected due to enhanced signal.
Increased b-value (>1000 s/mm2) DWI can increase lesion detection, but high b-value DWI images often exhibit low signal noise ratio, large diffusion-sensitive gradients that tend to distort images and longer scan times. The emergence of computed DWI has partially solved the above problem[36]. Liang et al[37] explored the value of computed DWI (cDWI) technique in the diagnosis of pancreatic carcinoma, and the results initially showed that a b-value of c1000-c1500 s/mm2 at cDWI technique could effectively display pancreatic carcinoma as well as maintain the image quality. Compared to DWI, intravoxel incoherent motion imaging is based on a biexponential model, which can quantify the diffusion and perfusion motions of water molecules separately. It can reflect the diffusion and perfusion characteristics of tissue cells, respectively, and has the advantages of fast-imaging, low-noise, and multi-parameters[38].
MR enhanced or perfusion imaging facilitates T-staging of pancreatic carcinoma by observing the relationship between the lesion, its surrounding tissue and vascular invasion. Both dynamic contrast-enhanced MRI (DCE-MRI) and perfusion MRI can provide quantitative information on blood flow perfusion of lesions (such as tumor tissue)[39]. The most common forms include T2*-weighted dynamic susceptibility contrast (DSC) perfusion and T1-weighted DCE perfusion[40]. However, there are significant differences between the two imaging methods, PWI (DCE and DSC) can reflect the tumor microenvironment such as blood vessel density and blood flow state by quantitative and functional parameters, such as first transit time, mean transit time, time to peak, etc. while dynamic enhancement can only obtain time perfusion curves through multi-phase dynamic enhancement, but they are not molecular imaging per se.
The basic principle of MR targeted molecular imaging is similar to nuclear medicine molecular imaging. The first step is to construct a specific molecular probe, and then introduce it into the body. After the probe actively and specifically binds to the imaging target, the lesions containing specific molecular targets in the body will be imaged by MRI[41]. Due to the high specificity of the molecular probe, delayed scan time, continuous enhancement within the tumor and relatively high signal on T1WI during the delayed scan, the specificity of the diagnosis is greatly improved[42,43], which helps to improve the detection rate of early microscopic pancreatic carcinoma lesions.
MR molecular probes meet the requirements of high specificity, affinity and signal elements that can be detected by MRI, such as T1 contrast agent represented by gadolinium (Gd), manganese, zinc chelates (Positive) and T2 contrast agent represented by MNP (Negative). Gd is used as a signal component to synthesize paramagnetic molecular imaging probes, mainly to shorten the longitudinal relaxation time of hydrogen protons, increase the T1 relaxation rate and produce positive T1WI contrast[43]. The traditional Gd agent enhanced MRI is diagnosed by the hemodynamic characteristics of lack of blood supply in pancreatic carcinoma, with low relaxation rate and lack of tissue specificity[44]. In recent years, MNP, as represented by SPION, has been applied in MR molecular imaging studies, mainly to shorten the transverse relaxation time, improve the T2 relaxation rate and produce negative T2WI contrast. Compared with Gd and SPION, it has better magnetization and biocompatibility, and no risk of nephrogenic systemic fibrosis[45,46].
In recent years, MR molecular imaging of pancreatic carcinoma is mainly based on basic scientific research. At present, the main targets involved are SSTR[47], urokinase-type plasminogen activator receptor[48], insulin-like growth factor-1 receptor, αVβ6, epidermal growth factor receptor, vascular endothelial growth factor receptor-2[49], etc., but their prospect of clinical application requires further study. In addition, targets such as reticulin-1 (plectin-1)[50], mucin-1[51], MUC4, carcinoembryonic antigen-related cell adhesion molecule 6[52], γ-glutamyltransferase 5[53], P32 protein[54], mesothelin[55], thymus fine cell differentiation antigen-1, cathepsin E, neutrophil gelatinase-associated lipid transport protein[56] were also examined, which could lead to a new imaging target for pancreatic carcinoma.
The slow progress of MR targeted molecular imaging compared to nuclear medicine molecular imaging is mainly due to its own limitations. First, the specificity of the above-mentioned types of targets is poor, which affects the specificity of MR molecular imaging[56]. Second, high concentrations of Gd molecular probes are required for imaging, which is difficult to achieve when some molecular targets are expressed at low levels. Finally, factors such as low blood supply, low perfusion in pancreatic carcinoma, denser stromal components in the tumor, and excessive uptake of the molecular probe by the reticuloendothelial system such as liver and spleen in vivo decrease the aggregation dose in the tumor, thus affecting the effect of MR molecular imaging[57]. It can be optimized from the following aspects: (1) Improve the biocompatibility of molecular probes and appropriately prolong their blood circulation time to promote more molecular probes to bind to the tumor; (2) The molecular probe simultaneously combines more Gd ions to obtain higher relaxation rate[43]; (3) Multi-target molecular imaging facilitates specific imaging of lesions[42]; and (4) Reduce the volume and molecular weight of molecular probe to achieve better penetration efficiency, paramagnetic resonance effect, reduce immunity and reticuloendothelial system uptake in vivo[42].
Different imaging techniques have their advantages and disadvantages, and their combined application can achieve complementary advantages and improve the value of clinical applications[58]. In addition, the management of radiopharmaceuticals is extremely strict in some countries, and very few radiopharmaceuticals are clinically approved. Therefore, in addition to the research and development of various imaging agents with high specificity and high sensitivity mentioned above, AI technology to improve diagnostic performance or complement existing technologies may be worth exploring.
AI, artificial intelligence, can be used to mine various medical images for biometric information and imaging features that are not easily perceived by physicians. In recent years, the application of AI-based radiomics has been used for lesion detection, pathological diagnosis, radiotherapy target delineation and curative effect prediction, so as to improve effective treatment decision-making for cancer patients. Based on radiomics, the cross-validated support vector machine classification diagnostic model can automatically extract quantitative features from MDCT[59]. Liu et al[60] used the AI system of R-CNN depth neural network to verify the diagnosis of CT images of pancreatic carcinoma in 100 cases, and established an AI diagnosis system of pancreatic carcinoma based on enhanced CT images. The system can assist doctors to identify pancreatic carcinoma, normal pancreatic tissue, chronic pancreatitis or benign pancreatic tumors. Mori et al[61] constructed 18F-FDG-PET/CT radiomic model to predict the recurrence survival value of patients with LAPC after radiotherapy for locally advanced pancreatic cancer, which could significantly improve treatment outcome while avoiding over-treatment of patients with poorer expected outcomes.
Radiomics based on AI has the potential to supplement information for clinical diagnosis and treatment and help solve certain clinical problems, but there are some limitations, such as incorrect tumor screening, insufficient design of database, case number and sensitive feature algorithm.
Nuclear medicine molecular imaging is based on the principle of injecting microscopic molecular probes into the body and selectively targeting them to appropriate sites based on different properties, in order to qualify or quantify organs, tissues or lesions for assessing diseases at the molecular level. Molecular imaging in nuclear medicine has made significant advances in the assessment of pancreatic carcinoma in recent years.
Provenance and peer review: Invited article; Externally peer reviewed.
Peer-review model: Single blind
Specialty type: Oncology
Country/Territory of origin: China
Peer-review report’s scientific quality classification
Grade A (Excellent): 0
Grade B (Very good): B
Grade C (Good): C
Grade D (Fair): 0
Grade E (Poor): 0
P-Reviewer: Chisthi MM, India; Wu CQ, China S-Editor: Wang JL L-Editor: A P-Editor: Chen YX
| 1. | Siegel RL, Miller KD, Jemal A. Cancer statistics, 2020. CA Cancer J Clin. 2020;70:7-30. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 12667] [Cited by in RCA: 15184] [Article Influence: 3036.8] [Reference Citation Analysis (4)] |
| 2. | Sellmyer MA, Lee IK, Mankoff DA. Building the Bridge: Molecular Imaging Biomarkers for 21(st) Century Cancer Therapies. J Nucl Med. 2021;62:1672-1676. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 6] [Cited by in RCA: 18] [Article Influence: 4.5] [Reference Citation Analysis (0)] |
| 3. | Peppicelli S, Andreucci E, Ruzzolini J, Bianchini F, Calorini L. FDG uptake in cancer: a continuing debate. Theranostics. 2020;10:2944-2948. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 11] [Cited by in RCA: 26] [Article Influence: 5.2] [Reference Citation Analysis (0)] |
| 4. | Ghaneh P, Hanson R, Titman A, Lancaster G, Plumpton C, Lloyd-Williams H, Yeo ST, Edwards RT, Johnson C, Abu Hilal M, Higginson AP, Armstrong T, Smith A, Scarsbrook A, McKay C, Carter R, Sutcliffe RP, Bramhall S, Kocher HM, Cunningham D, Pereira SP, Davidson B, Chang D, Khan S, Zealley I, Sarker D, Al Sarireh B, Charnley R, Lobo D, Nicolson M, Halloran C, Raraty M, Sutton R, Vinjamuri S, Evans J, Campbell F, Deeks J, Sanghera B, Wong WL, Neoptolemos JP. PET-PANC: multicentre prospective diagnostic accuracy and health economic analysis study of the impact of combined modality 18fluorine-2-fluoro-2-deoxy-d-glucose positron emission tomography with computed tomography scanning in the diagnosis and management of pancreatic cancer. Health Technol Assess. 2018;22:1-114. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 75] [Cited by in RCA: 80] [Article Influence: 11.4] [Reference Citation Analysis (0)] |
| 5. | Daamen LA, Groot VP, Goense L, Wessels FJ, Borel Rinkes IH, Intven MPW, van Santvoort HC, Molenaar IQ. The diagnostic performance of CT versus FDG PET-CT for the detection of recurrent pancreatic cancer: a systematic review and meta-analysis. Eur J Radiol. 2018;106:128-136. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 29] [Cited by in RCA: 46] [Article Influence: 6.6] [Reference Citation Analysis (0)] |
| 6. | Topkan E, Parlak C, Yapar AF. FDG-PET/CT-based restaging may alter initial management decisions and clinical outcomes in patients with locally advanced pancreatic carcinoma planned to undergo chemoradiotherapy. Cancer Imaging. 2013;13:423-428. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 17] [Cited by in RCA: 16] [Article Influence: 1.3] [Reference Citation Analysis (0)] |
| 7. | Xi Y, Guo R, Hu J, Zhang M, Zhang X, Li B. 18F-fluoro-2-deoxy-D-glucose retention index as a prognostic parameter in patients with pancreatic cancer. Nucl Med Commun. 2014;35:1112-1118. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 10] [Cited by in RCA: 12] [Article Influence: 1.2] [Reference Citation Analysis (0)] |
| 8. | Ahn SJ, Park MS, Lee JD, Kang WJ. Correlation between 18F-fluorodeoxyglucose positron emission tomography and pathologic differentiation in pancreatic cancer. Ann Nucl Med. 2014;28:430-435. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 20] [Cited by in RCA: 24] [Article Influence: 2.2] [Reference Citation Analysis (0)] |
| 9. | Myssayev A, Myssayev A, Ideguchi R, Eguchi S, Adachi T, Sumida Y, Tobinaga S, Uetani M, Kudo T. Usefulness of FDG PET/CT derived parameters in prediction of histopathological finding during the surgery in patients with pancreatic adenocarcinoma. PLoS One. 2019;14:e0210178. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 4] [Cited by in RCA: 8] [Article Influence: 1.3] [Reference Citation Analysis (0)] |
| 10. | Barreto SG, Loveday B, Windsor JA, Pandanaboyana S. Detecting tumour response and predicting resectability after neoadjuvant therapy for borderline resectable and locally advanced pancreatic cancer. ANZ J Surg. 2019;89:481-487. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 37] [Cited by in RCA: 30] [Article Influence: 5.0] [Reference Citation Analysis (1)] |
| 11. | Hyun SH, Kim HS, Choi SH, Choi DW, Lee JK, Lee KH, Park JO, Kim BT, Choi JY. Intratumoral heterogeneity of (18)F-FDG uptake predicts survival in patients with pancreatic ductal adenocarcinoma. Eur J Nucl Med Mol Imaging. 2016;43:1461-1468. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 70] [Cited by in RCA: 82] [Article Influence: 9.1] [Reference Citation Analysis (0)] |
| 12. | Chang JS, Choi SH, Lee Y, Kim KH, Park JY, Song SY, Cho A, Yun M, Lee JD, Seong J. Clinical usefulness of ¹⁸F-fluorodeoxyglucose-positron emission tomography in patients with locally advanced pancreatic cancer planned to undergo concurrent chemoradiation therapy. Int J Radiat Oncol Biol Phys. 2014;90:126-133. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 33] [Cited by in RCA: 37] [Article Influence: 3.4] [Reference Citation Analysis (0)] |
| 13. | Moon SY, Joo KR, So YR, Lim JU, Cha JM, Shin HP, Yang YJ. Predictive value of maximum standardized uptake value (SUVmax) on 18F-FDG PET/CT in patients with locally advanced or metastatic pancreatic cancer. Clin Nucl Med. 2013;38:778-783. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 30] [Cited by in RCA: 38] [Article Influence: 3.5] [Reference Citation Analysis (0)] |
| 14. | Zhang A, Ren S, Yuan Y, Li X, Zhu X, Jiang L, Li D, Zuo C. Prognostic values of 18F-FDG PET/CT metabolic parameters and clinical figures in locally advanced pancreatic cancer underwent chemotherapy combined with stereotactic body radiation therapy. Medicine (Baltimore). 2019;98:e15064. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 7] [Cited by in RCA: 7] [Article Influence: 1.2] [Reference Citation Analysis (0)] |
| 15. | Yamamoto T, Sugiura T, Mizuno T, Okamura Y, Aramaki T, Endo M, Uesaka K. Preoperative FDG-PET predicts early recurrence and a poor prognosis after resection of pancreatic adenocarcinoma. Ann Surg Oncol. 2015;22:677-684. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 50] [Cited by in RCA: 55] [Article Influence: 5.0] [Reference Citation Analysis (0)] |
| 16. | Pu Y, Wang C, Zhao S, Xie R, Zhao L, Li K, Yang C, Zhang R, Tian Y, Tan L, Li J, Li S, Chen L, Sun H. The clinical application of (18)F-FDG PET/CT in pancreatic cancer: a narrative review. Transl Cancer Res. 2021;10:3560-3575. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 2] [Cited by in RCA: 2] [Article Influence: 0.5] [Reference Citation Analysis (0)] |
| 17. | Calabrò D, Argalia G, Ambrosini V. Role of PET/CT and Therapy Management of Pancreatic Neuroendocrine Tumors. Diagnostics (Basel). 2020;10. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 23] [Cited by in RCA: 23] [Article Influence: 4.6] [Reference Citation Analysis (0)] |
| 18. | Carideo L, Prosperi D, Panzuto F, Magi L, Pratesi MS, Rinzivillo M, Annibale B, Signore A. Role of Combined [(68)Ga]Ga-DOTA-SST Analogues and [(18)F]FDG PET/CT in the Management of GEP-NENs: A Systematic Review. J Clin Med. 2019;8. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 61] [Cited by in RCA: 57] [Article Influence: 9.5] [Reference Citation Analysis (0)] |
| 19. | Zhang P, Yu J, Li J, Shen L, Li N, Zhu H, Zhai S, Zhang Y, Yang Z, Lu M. Clinical and Prognostic Value of PET/CT Imaging with Combination of (68)Ga-DOTATATE and (18)F-FDG in Gastroenteropancreatic Neuroendocrine Neoplasms. Contrast Media Mol Imaging. 2018;2018:2340389. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 41] [Cited by in RCA: 65] [Article Influence: 9.3] [Reference Citation Analysis (0)] |
| 20. | Evangelista L, Ravelli I, Bignotto A, Cecchin D, Zucchetta P. Ga-68 DOTA-peptides and F-18 FDG PET/CT in patients with neuroendocrine tumor: A review. Clin Imaging. 2020;67:113-116. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 19] [Cited by in RCA: 27] [Article Influence: 5.4] [Reference Citation Analysis (0)] |
| 21. | Fani M, Nicolas GP, Wild D. Somatostatin Receptor Antagonists for Imaging and Therapy. J Nucl Med. 2017;58:61S-66S. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 131] [Cited by in RCA: 178] [Article Influence: 22.3] [Reference Citation Analysis (0)] |
| 22. | Liu Y, Watabe T, Kaneda-Nakashima K, Shirakami Y, Naka S, Ooe K, Toyoshima A, Nagata K, Haberkorn U, Kratochwil C, Shinohara A, Hatazawa J, Giesel F. Fibroblast activation protein targeted therapy using [(177)Lu]FAPI-46 compared with [(225)Ac]FAPI-46 in a pancreatic cancer model. Eur J Nucl Med Mol Imaging. 2022;49:871-880. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 18] [Cited by in RCA: 112] [Article Influence: 28.0] [Reference Citation Analysis (0)] |
| 23. | Sunami Y, Häußler J, Kleeff J. Cellular Heterogeneity of Pancreatic Stellate Cells, Mesenchymal Stem Cells, and Cancer-Associated Fibroblasts in Pancreatic Cancer. Cancers (Basel). 2020;12. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 16] [Cited by in RCA: 29] [Article Influence: 5.8] [Reference Citation Analysis (0)] |
| 24. | Ji X, Qian J, Rahman SMJ, Siska PJ, Zou Y, Harris BK, Hoeksema MD, Trenary IA, Heidi C, Eisenberg R, Rathmell JC, Young JD, Massion PP. xCT (SLC7A11)-mediated metabolic reprogramming promotes non-small cell lung cancer progression. Oncogene. 2018;37:5007-5019. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 130] [Cited by in RCA: 227] [Article Influence: 32.4] [Reference Citation Analysis (0)] |
| 25. | Zhang L, Huang Y, Ling J, Zhuo W, Yu Z, Luo Y, Zhu Y. Overexpression of SLC7A11: a novel oncogene and an indicator of unfavorable prognosis for liver carcinoma. Future Oncol. 2018;14:927-936. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 26] [Cited by in RCA: 40] [Article Influence: 5.7] [Reference Citation Analysis (0)] |
| 26. | Zhang Z, Jia G, Pan G, Cao K, Yang Q, Meng H, Yang J, Zhang L, Wang T, Cheng C, Zuo C. Comparison of the diagnostic efficacy of (68) Ga-FAPI-04 PET/MR and (18)F-FDG PET/CT in patients with pancreatic cancer. Eur J Nucl Med Mol Imaging. 2022;49:2877-2888. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 27] [Cited by in RCA: 43] [Article Influence: 14.3] [Reference Citation Analysis (0)] |
| 27. | Röhrich M, Naumann P, Giesel FL, Choyke PL, Staudinger F, Wefers A, Liew DP, Kratochwil C, Rathke H, Liermann J, Herfarth K, Jäger D, Debus J, Haberkorn U, Lang M, Koerber SA. Impact of (68)Ga-FAPI PET/CT Imaging on the Therapeutic Management of Primary and Recurrent Pancreatic Ductal Adenocarcinomas. J Nucl Med. 2021;62:779-786. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 47] [Cited by in RCA: 141] [Article Influence: 28.2] [Reference Citation Analysis (0)] |
| 28. | Gallo A, Dedionigi C, Civitelli C, Panzeri A, Corradi C, Squizzato A. Optimal Management of Cirrhotic Ascites: A Review for Internal Medicine Physicians. J Transl Int Med. 2020;8:220-236. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 6] [Cited by in RCA: 9] [Article Influence: 1.8] [Reference Citation Analysis (0)] |
| 29. | Deng M, Chen Y, Cai L. Comparison of 68Ga-FAPI and 18F-FDG PET/CT in the Imaging of Pancreatic Cancer With Liver Metastases. Clin Nucl Med. 2021;46:589-591. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 19] [Cited by in RCA: 16] [Article Influence: 4.0] [Reference Citation Analysis (0)] |
| 30. | Xiong J, Balcioglu HE, Danen EH. Integrin signaling in control of tumor growth and progression. Int J Biochem Cell Biol. 2013;45:1012-1015. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 45] [Cited by in RCA: 42] [Article Influence: 3.5] [Reference Citation Analysis (0)] |
| 31. | Herter J, Zarbock A. Integrin Regulation during Leukocyte Recruitment. J Immunol. 2013;190:4451-4457. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 140] [Cited by in RCA: 143] [Article Influence: 11.9] [Reference Citation Analysis (0)] |
| 32. | Huo Y, Kang L, Pang X, Shen H, Yan P, Zhang C, Liao X, Chen X, Wang R. Noninvasive PET Imaging of a Ga-68-Radiolabeled RRL-Derived Peptide in Hepatocarcinoma Murine Models. Mol Imaging Biol. 2019;21:286-296. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 3] [Cited by in RCA: 2] [Article Influence: 0.3] [Reference Citation Analysis (0)] |
| 33. | Reader CS, Vallath S, Steele CW, Haider S, Brentnall A, Desai A, Moore KM, Jamieson NB, Chang D, Bailey P, Scarpa A, Lawlor R, Chelala C, Keyse SM, Biankin A, Morton JP, Evans TJ, Barry ST, Sansom OJ, Kocher HM, Marshall JF. The integrin αvβ6 drives pancreatic cancer through diverse mechanisms and represents an effective target for therapy. J Pathol. 2019;249:332-342. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 61] [Cited by in RCA: 83] [Article Influence: 13.8] [Reference Citation Analysis (0)] |
| 34. | Liu Z, Liu H, Ma T, Sun X, Shi J, Jia B, Sun Y, Zhan J, Zhang H, Zhu Z, Wang F. Integrin αvβ₆-Targeted SPECT Imaging for Pancreatic Cancer Detection. J Nucl Med. 2014;55:989-994. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 45] [Cited by in RCA: 42] [Article Influence: 3.8] [Reference Citation Analysis (0)] |
| 35. | Koay EJ, Hall W, Park PC, Erickson B, Herman JM. The role of imaging in the clinical practice of radiation oncology for pancreatic cancer. Abdom Radiol (NY). 2018;43:393-403. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 6] [Cited by in RCA: 4] [Article Influence: 0.6] [Reference Citation Analysis (0)] |
| 36. | Higaki T, Nakamura Y, Tatsugami F, Kaichi Y, Akagi M, Akiyama Y, Baba Y, Iida M, Awai K. Introduction to the Technical Aspects of Computed Diffusion-weighted Imaging for Radiologists. Radiographics. 2018;38:1131-1144. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 19] [Cited by in RCA: 20] [Article Influence: 2.9] [Reference Citation Analysis (0)] |
| 37. | Liang L, Gu JY, Liu K, Fu CX, Rao SX, Zeng MS. [Application of cDWI in diagnosis of pancreatic cancer: a preliminary study]. Fangshexue Shijian. 2019;34:1081-1087. [DOI] [Full Text] |
| 38. | Kaandorp MPT, Barbieri S, Klaassen R, van Laarhoven HWM, Crezee H, While PT, Nederveen AJ, Gurney-Champion OJ. Improved unsupervised physics-informed deep learning for intravoxel incoherent motion modeling and evaluation in pancreatic cancer patients. Magn Reson Med. 2021;86:2250-2265. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 39] [Cited by in RCA: 52] [Article Influence: 13.0] [Reference Citation Analysis (0)] |
| 39. | Ottens T, Barbieri S, Orton MR, Klaassen R, van Laarhoven HWM, Crezee H, Nederveen AJ, Zhen X, Gurney-Champion OJ. Deep learning DCE-MRI parameter estimation: Application in pancreatic cancer. Med Image Anal. 2022;80:102512. [RCA] [PubMed] [DOI] [Full Text] [Reference Citation Analysis (0)] |
| 40. | Valenzuela RF, Kundra V, Madewell JE, Costelloe CM. Advanced Imaging in Musculoskeletal Oncology: Moving Away From RECIST and Embracing Advanced Bone and Soft Tissue Tumor Imaging (ABASTI) - Part I - Tumor Response Criteria and Established Functional Imaging Techniques. Semin Ultrasound CT MR. 2021;42:201-214. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 1] [Cited by in RCA: 2] [Article Influence: 0.4] [Reference Citation Analysis (0)] |
| 41. | Chen X, Zhou H, Li X, Duan N, Hu S, Liu Y, Yue Y, Song L, Zhang Y, Li D, Wang Z. Plectin-1 Targeted Dual-modality Nanoparticles for Pancreatic Cancer Imaging. EBioMedicine. 2018;30:129-137. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 29] [Cited by in RCA: 43] [Article Influence: 6.1] [Reference Citation Analysis (0)] |
| 42. | Wang Q, Yan H, Jin Y, Wang Z, Huang W, Qiu J, Kang F, Wang K, Zhao X, Tian J. A novel plectin/integrin-targeted bispecific molecular probe for magnetic resonance/near-infrared imaging of pancreatic cancer. Biomaterials. 2018;183:173-184. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 25] [Cited by in RCA: 27] [Article Influence: 3.9] [Reference Citation Analysis (0)] |
| 43. | Huang X, Fan C, Zhu H, Le W, Cui S, Chen X, Li W, Zhang F, Huang Y, Sh D, Cui Z, Shao C, Chen B. Glypican-1-antibody-conjugated Gd-Au nanoclusters for FI/MRI dual-modal targeted detection of pancreatic cancer. Int J Nanomedicine. 2018;13:2585-2599. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 17] [Cited by in RCA: 18] [Article Influence: 2.6] [Reference Citation Analysis (0)] |
| 44. | Lee S, Kim SH, Park HK, Jang KT, Hwang JA, Kim S. Pancreatic Ductal Adenocarcinoma: Rim Enhancement at MR Imaging Predicts Prognosis after Curative Resection. Radiology. 2018;288:456-466. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 25] [Cited by in RCA: 25] [Article Influence: 3.6] [Reference Citation Analysis (0)] |
| 45. | Zhang C, Yan Y, Zou Q, Chen J, Li C. Superparamagnetic iron oxide nanoparticles for MR imaging of pancreatic cancer: Potential for early diagnosis through targeted strategies. Asia Pac J Clin Oncol. 2016;12:13-21. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 18] [Cited by in RCA: 17] [Article Influence: 1.7] [Reference Citation Analysis (0)] |
| 46. | Ze X, Zou W, Li Z. Translational research in anti-pancreatic fibrosis drug discovery and development. J Transl Int Med. 2021;9:225-227. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 1] [Cited by in RCA: 1] [Article Influence: 0.3] [Reference Citation Analysis (0)] |
| 47. | Ahmadi Y, Kostenich G, Oron-Herman M, Wadsak W, Mitterhauser M, Orenstein A, Mirzaei S, Knoll P. In vivo magnetic resonance imaging of pancreatic tumors using iron oxide nanoworms targeted with PTR86 peptide. Colloids Surf B Biointerfaces. 2017;158:423-430. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 7] [Cited by in RCA: 7] [Article Influence: 0.9] [Reference Citation Analysis (0)] |
| 48. | Li H, Wang P, Gong W, Wang Q, Zhou J, Zhu WH, Cheng Y. Dendron-Grafted Polylysine-Based Dual-Modal Nanoprobe for Ultra-Early Diagnosis of Pancreatic Precancerosis via Targeting a Urokinase-Type Plasminogen Activator Receptor. Adv Healthc Mater. 2018;7. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 24] [Cited by in RCA: 18] [Article Influence: 2.6] [Reference Citation Analysis (0)] |
| 49. | Morin E, Sjöberg E, Tjomsland V, Testini C, Lindskog C, Franklin O, Sund M, Öhlund D, Kiflemariam S, Sjöblom T, Claesson-Welsh L. VEGF receptor-2/neuropilin 1 trans-complex formation between endothelial and tumor cells is an independent predictor of pancreatic cancer survival. J Pathol. 2018;246:311-322. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 20] [Cited by in RCA: 22] [Article Influence: 3.1] [Reference Citation Analysis (0)] |
| 50. | Zhang W, Liang X, Zhu L, Zhang X, Jin Z, Du Y, Tian J, Xue H. Optical magnetic multimodality imaging of plectin-1-targeted imaging agent for the precise detection of orthotopic pancreatic ductal adenocarcinoma in mice. EBioMedicine. 2022;80:104040. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 11] [Cited by in RCA: 14] [Article Influence: 4.7] [Reference Citation Analysis (0)] |
| 51. | Zou Q, Zhang CJ, Yan YZ, Min ZJ, Li CS. MUC-1 aptamer targeted superparamagnetic iron oxide nanoparticles for magnetic resonance imaging of pancreatic cancer in vivo and in vitro experiment. J Cell Biochem. 2019;120:18650-18658. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 13] [Cited by in RCA: 15] [Article Influence: 2.5] [Reference Citation Analysis (0)] |
| 52. | Zou J, Chen S, Li Y, Zeng L, Lian G, Li J, Huang K, Chen Y. Nanoparticles modified by triple single chain antibodies for MRI examination and targeted therapy in pancreatic cancer. Nanoscale. 2020;12:4473-4490. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 17] [Cited by in RCA: 13] [Article Influence: 2.6] [Reference Citation Analysis (0)] |
| 53. | Ramsay EE, Decollogne S, Joshi S, Corti A, Apte M, Pompella A, Hogg PJ, Dilda PJ. Employing pancreatic tumor γ-glutamyltransferase for therapeutic delivery. Mol Pharm. 2014;11:1500-1511. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 13] [Cited by in RCA: 12] [Article Influence: 1.1] [Reference Citation Analysis (0)] |
| 54. | Jiang Y, Liu S, Zhang Y, Li H, He H, Dai J, Jiang T, Ji W, Geng D, Elzatahry AA, Alghamdi A, Fu D, Deng Y, Zhao D. Magnetic mesoporous nanospheres anchored with LyP-1 as an efficient pancreatic cancer probe. Biomaterials. 2017;115:9-18. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 46] [Cited by in RCA: 38] [Article Influence: 4.8] [Reference Citation Analysis (0)] |
| 55. | Nahm CB, Turchini J, Jamieson N, Moon E, Sioson L, Itchins M, Arena J, Colvin E, Howell VM, Pavlakis N, Clarke S, Samra JS, Gill AJ, Mittal A. Biomarker panel predicts survival after resection in pancreatic ductal adenocarcinoma: A multi-institutional cohort study. Eur J Surg Oncol. 2019;45:218-224. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 17] [Cited by in RCA: 25] [Article Influence: 3.6] [Reference Citation Analysis (0)] |
| 56. | Tummers WS, Willmann JK, Bonsing BA, Vahrmeijer AL, Gambhir SS, Swijnenburg RJ. Advances in Diagnostic and Intraoperative Molecular Imaging of Pancreatic Cancer. Pancreas. 2018;47:675-689. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 25] [Cited by in RCA: 27] [Article Influence: 3.9] [Reference Citation Analysis (0)] |
| 57. | Dadfar SM, Roemhild K, Drude NI, von Stillfried S, Knüchel R, Kiessling F, Lammers T. Iron oxide nanoparticles: Diagnostic, therapeutic and theranostic applications. Adv Drug Deliv Rev. 2019;138:302-325. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 717] [Cited by in RCA: 644] [Article Influence: 107.3] [Reference Citation Analysis (0)] |
| 58. | Feng L, Guo J, Wang S, Liu X, Ge N, Wang G, Sun S. Endoscopic Transmural Drainage and Necrosectomy in Acute Necrotizing Pancreatitis: A Review. J Transl Int Med. 2021;9:168-176. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 13] [Cited by in RCA: 1] [Article Influence: 0.3] [Reference Citation Analysis (0)] |
| 59. | Wei R, Lin K, Yan W, Guo Y, Wang Y, Li J, Zhu J. Computer-Aided Diagnosis of Pancreas Serous Cystic Neoplasms: A Radiomics Method on Preoperative MDCT Images. Technol Cancer Res Treat. 2019;18:1533033818824339. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 51] [Cited by in RCA: 52] [Article Influence: 8.7] [Reference Citation Analysis (0)] |
| 60. | Liu SL, Li S, Guo YT, Zhou YP, Zhang ZD, Lu Y. Establishment and application of an artificial intelligence diagnosis system for pancreatic cancer with a faster region-based convolutional neural network. Chin Med J (Engl). 2019;132:2795-2803. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 54] [Cited by in RCA: 48] [Article Influence: 8.0] [Reference Citation Analysis (0)] |
| 61. | Mori M, Passoni P, Incerti E, Bettinardi V, Broggi S, Reni M, Whybra P, Spezi E, Vanoli EG, Gianolli L, Picchio M, Di Muzio NG, Fiorino C. Training and validation of a robust PET radiomic-based index to predict distant-relapse-free-survival after radio-chemotherapy for locally advanced pancreatic cancer. Radiother Oncol. 2020;153:258-264. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 9] [Cited by in RCA: 21] [Article Influence: 4.2] [Reference Citation Analysis (0)] |