Published online Oct 27, 2023. doi: 10.4240/wjgs.v15.i10.2331
Peer-review started: July 9, 2023
First decision: August 10, 2023
Revised: August 14, 2023
Accepted: August 21, 2023
Article in press: August 21, 2023
Published online: October 27, 2023
Processing time: 110 Days and 1.6 Hours
Colorectal cancer ranks third in global cancer prevalence and stands as the second leading cause of cancer-related mortalities. With obesity recognized as a pivotal risk factor for colorectal cancer, the potential protective role of bariatric surgery, especially laparoscopic Roux-en-Y gastric bypass and laparoscopic sleeve gas
To investigate the Roux-en-Y gastric bypass (RYGB) vs sleeve gastrectomy (SG) effect on colorectal cancer incidence in obese individuals.
A systematic review and meta-analysis of the literature was conducted following the Preferred Reporting Items for Systematic Reviews and Meta-Analyses. Seven
The meta-analysis revealed a 54% reduction in colorectal cancer risk in morbidly obese patients who underwent bariatric surgery compared to those who did not. A significant 46% reduction in colorectal cancer risk was observed among female patients. However, no significant differences were found in the meta-analysis for various types of bariatric surgery, such as SG and RYGB.
This meta-analysis reveals weight loss surgery, regardless of type, reduces colorectal cancer risk, especially in women, as indicated by RR and hazard ratio assessments. Further validation is essential.
Core Tip: This comprehensive meta-analysis evaluates the impact of bariatric surgery on colorectal cancer (CRC) risk using 17 studies with over 12 million patients. Results show a 54% reduction in CRC risk after surgery, with women experiencing a 46% decrease. Interestingly, the type of bariatric surgery, whether Sleeve Gastrectomy or Roux-en-Y Gastric Bypass, did not influence this risk reduction. Bariatric procedures underscore a pivotal role in managing CRC risk in the obese. The study highlights the surgery's protective effect, especially for women, and the need for further research on potential confounding factors.
- Citation: Liu YN, Gu JF, Zhang J, Xing DY, Wang GQ. Bariatric surgery reduces colorectal cancer incidence in obese individuals: Systematic review and meta-analysis. World J Gastrointest Surg 2023; 15(10): 2331-2342
- URL: https://www.wjgnet.com/1948-9366/full/v15/i10/2331.htm
- DOI: https://dx.doi.org/10.4240/wjgs.v15.i10.2331
Obesity is a multifaceted disease influenced by genetic, behavioral, socioeconomic, and environmental factors. These factors contribute to an elevated risk of morbidity and mortality from several debilitating conditions. Obesity increases the risk of a wide range of chronic diseases, from diabetes and dyslipidemia to mental health conditions. It has a significant impact on the risk of stroke, cardiovascular disease, certain cancers, and osteoarthritis. Worldwide, the rising prevalence of obesity presents substantial challenges to both chronic disease prevention and health promotion[1]. Given its global impact, the World Health Organization (WHO) defines obesity in Western populations as a body mass index (BMI) of ≥ 30 kg/m², while a BMI of ≥ 25 kg/m² is considered overweight. In the United States, 64.5% of adults are categorized as overweight and 30.5% as obese[2]. Data indicates that, relative to individuals with a normal BMI range (18.5-24.9 kg/m²), the mortality rate from various causes is markedly higher among the obese population, with cardiovascular diseases as the leading cause[3]. Although the overall BMI of Asian populations is lower than that of Western populations, Asians tend to exhibit central obesity. Thus, using a BMI ≥ 30 kg/m² as the threshold for diagnosing obesity in Asian populations may underestimate the risks associated with obesity, leading the WHO expert committee to define a BMI ≥ 25 kg/m² as the threshold for obesity in Asian populations[4]. Female obesity rates are generally higher than male rates, and there are gender differences in body fat distribution[5].
Previous reviews have indicated that, compared to individuals with normal weight, obesity increases the risk of developing colorectal cancer by 7% to 60%, with some studies suggesting a stronger association between obesity and colon cancer than rectal cancer[6]. According to research by Moghaddam et al[7], there is a direct and independent relationship between obesity and colorectal cancer. For every 2 kg/m² increase in BMI, the risk of colorectal cancer increases by 7%. A 2 cm increase in waist circumference is associated with a 4% increase in colorectal cancer risk. The association between obesity and colorectal cancer accounts for approximately 20% of the total risk. The risk for men is 1.41 (95%CI: 1.3-1.54), and for women, it is 1.08 (95%CI: 0.98-1.18) (P heterogeneity < 0.001). It is estimated that 6% of all cancer diagnoses in 2007 (4% for men and 7% for women) can be attributed to obesity. Obesity is not only a major risk factor for diabetes but also for most cancers. For a long time, it has been believed that obesity is associated with an increased risk of esophageal, colon, pancreatic, postmenopausal breast, endometrial, and kidney cancers[8].
Weight loss surgery can reduce the incidence of obesity-related tumors, particularly in female patients. An Australian study with over 65000 patients and a follow-up of up to 15 years found that the risk of colorectal cancer in male obese patients decreased by nearly 50% after weight loss surgery. Numerous clinical studies have confirmed that the reduction of insulin levels after weight loss surgery can reduce the risk of developing colorectal tumors[9].
Common weight loss surgeries include laparoscopic sleeve gastrectomy (LSG), Roux-en-Y laparoscopic gastric bypass (LRYGB), laparoscopic adjustable gastric banding, and other novel procedures. RYGB, originally proposed by Wittgrove et al[10], alters the digestive tracts pathway to reduce absorption and is a complex, high-risk procedure with numerous postoperative complications, involving the alteration of gastrointestinal anatomy. RYGB is more complex and has more complications than LSG, but for patients with type 2 diabetes mellitus unresponsive to medical treatment, RYGB is the preferred option. RYGB is also the gold standard for revision surgery in patients with postoperative weight regain or complications, such as high gastric leaks and severe reflux esophagitis. LRYGB is safe and effective in treating obesity and type 2 diabetes, and there is a correlation between BMI, blood glucose levels, and quality of life before and after surgery[11,12].
Due to its significant effects on weight loss and metabolic improvement, LSG has gained widespread application in the field of bariatric surgery. This surgical procedure is known for its simplicity, reduced surgical risks, and lower incidence of postoperative complications. LSG has remarkable weight loss and metabolic improvement effects.
The findings from these studies prompt an essential inquiry: Is the incidence of colorectal cancer lower in obese patients who undergo weight loss surgeries like LRYGB and LSG compared to those who opt against such procedures? Furthermore, do gender differences play a significant role in this issue, and are there any differences when choosing between RYGB and sleeve gastrectomy (SG) procedures? This meta-analysis seeks to discern the influence of weight loss surgeries on the colorectal cancer risk among obese individuals. Earlier meta-analytic studies have documented a decreased cancer risk following weight loss surgery. However, those studies encompassed research with considerable variances in follow-up durations and lacked specific analyses to address this bias. Therefore, our research objective is to conduct a new meta-analysis to elucidate the specific impact of weight loss surgery on the risk of colorectal cancer in obese patients.
We conducted a systematic review of the literature following the guidelines for the Preferred Reporting Items for Systematic Reviews and Meta-Analyses (PRISMA) and Assessing the Methodological Quality of Systematic Reviews (AMSTAR)[13] and Meta-analysis of Observational Studies in Epidemiology (MOOSE) recommendations. This study does not require ethical approval or informed consent, as all data were retrieved from published literature. The search, eligibility assessment, data extraction, and quality evaluation were independently performed by four researchers. Any disagreements were resolved through discussion, with consensus required among the four researchers.
We searched PubMed, Embase, Web of Science, and Cochrane Library up to April 30, 2023, without imposing any time restrictions. We specifically adapted vocabulary and syntax for each database. We used computerized searches of the PubMed database with the following keywords: Obesity, obesity and cancer, colorectal cancer, weight loss surgery, gastric bypass, and sleeve gastrectomy. We did not impose any language restrictions. We also manually screened the reference lists of relevant articles to identify other potential records. In our search strategy, we included studies and previous reviews on the topic, screening their references according to the selection criteria to find more related studies.
In adherence to PRISMA guidelines, we employed the Population, Intervention, Comparison, Outcome, and Study design framework to determine study eligibility: Population (P): Adult individuals (≥ 18 years old) diagnosed with morbid obesity, followed up for at least three years to investigate the incidence of colorectal cancer. Intervention (I): Weight loss surgery. Comparison (C): Simple observation or any behavioral or pharmacological treatment. Outcome (O): Risk of developing colorectal cancer during the follow-up period. Study design (S): Retrospective and prospective comparative studies with at least ten patients in each group.
Studies were excluded if they did not involve a comparison of colorectal cancer risk between obese patients who underwent or did not undergo weight loss surgery, did not provide specific indicators or metrics for evaluating colorectal cancer risk, the full text was not accessible, or the study was not in English.
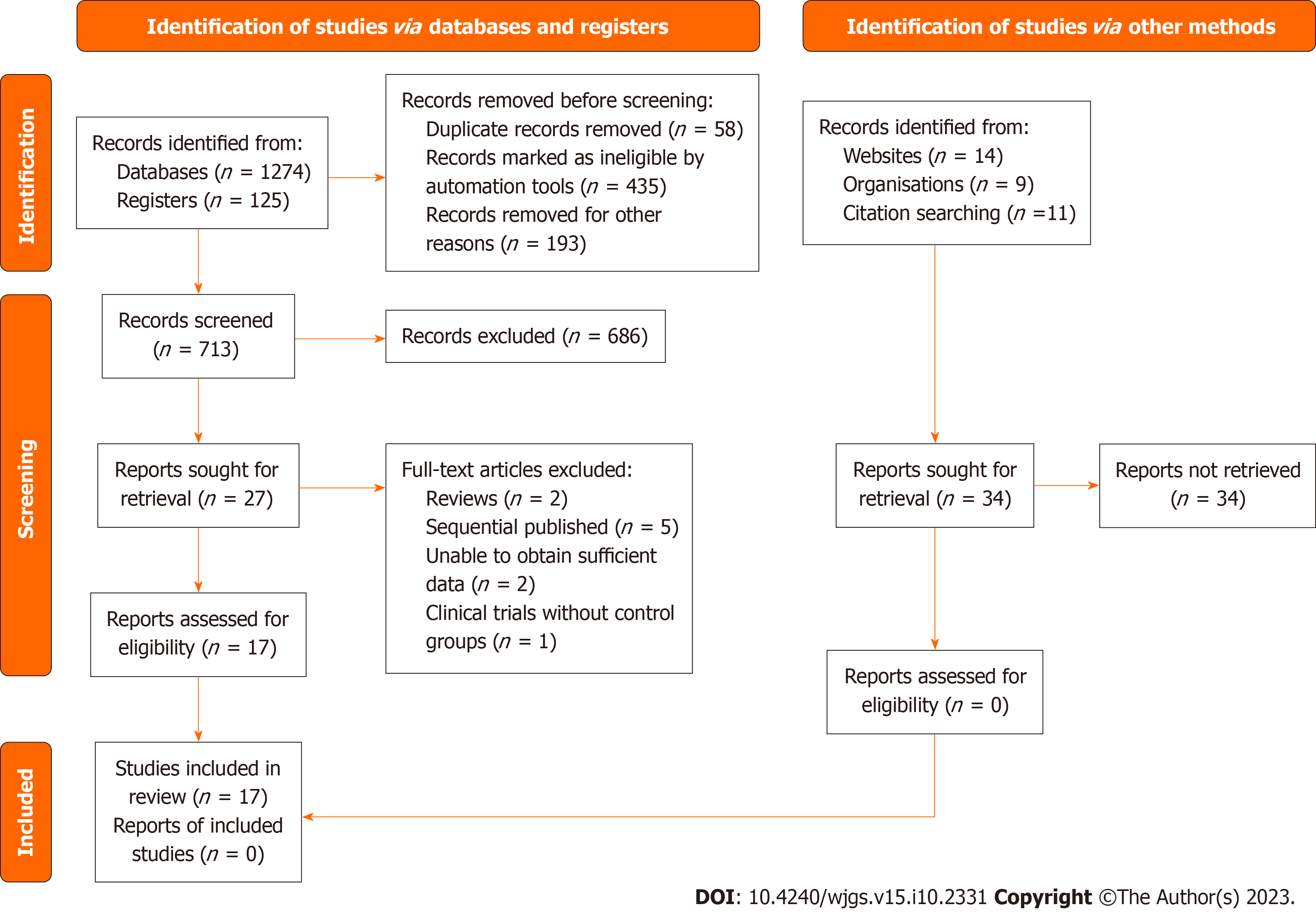
In the first step, 1399 articles were identified through literature searches. Duplicate entries found across different databases were removed using Mendeley reference software (Mendeley Ltd., London, United Kingdom). Then, the titles and abstracts of 713 records were screened using a filtering table created to guide study selection.
We assessed the quality of the included studies using the Newcastle-Ottawa Scale (NOS)[14]. This scale consists of three domains: selection, comparability, and outcome/exposure, with a maximum score of 9 points. Two evaluators assessed the following nine NOS sections: Representativeness of the exposed cohort, selection of the non-exposed cohort (selection bias); ascertainment of exposure, demonstration of the outcome, comparability of cohorts (comparability bias); assessment of the outcome, sufficient follow-up time, adequacy of cohort follow-up (outcome bias). Scores of 0-3, 4-6, and 7-9 represent low, medium, and high study quality, respectively.
A computerized electronic spreadsheet (Microsoft Excel 2021; Microsoft Corporation, Redmond, Washington, United Kingdom) was used to collect information regarding study design and methods, participant demographics and baseline characteristics, obesity treatment details, colorectal cancer outbreak risk, and survival.
The primary outcome was expressed as the relative risk (RR) of developing colorectal cancer in obese patients who underwent weight loss surgery compared to those who did not. In the same subset, secondary outcomes included determining the RR for colon cancer and rectal cancer separately. Additionally, subgroup analyses were performed, considering the RR of colorectal cancer (CRC) for males and females separately, and depending on the type of weight loss surgery performed (LSG vs RYGB). Finally, we determined the hazard ratio (HR) for developing colorectal cancer in obese individuals who underwent or did not undergo weight loss surgery.
We assessed study heterogeneity using chi-square statistics and quantified it with the I2 statistic. An I2 value of 0% suggests no observed heterogeneity, whereas an I2 value greater than 50% points to significant heterogeneity, while an I2 value > 50% indicates significant heterogeneity. We standardized the relative risk in each article and combined them using a random-effects model. We conducted sensitivity analyses to assess the robustness of our findings and to pinpoint any individual studies that might influence the overall effect size. This analysis involved sequentially removing each study from the meta-analysis and recalculating the overall effect size, checking whether the point estimate of the overall effect remained within the 95% confidence interval of the initial combined effect. Publication bias was examined using funnel plot symmetry and Egger's test in meta-analyses with ten or more eligible articles. If the funnel plot appeared asymmetrical, we evaluated if the assumption of unpublished negative studies, attributable to publication bias, had a significant impact on the effect estimate. In all statistical tests, two-sided P values < 0.05 were considered statistically significant. We used STATA version 17 (StataCorp, College Station, TX, United States) to analyze the data from randomized controlled trials that met our inclusion criteria.
As depicted in Figure 1, our quantitative and qualitative analyses encompassed 17 studies[15-31]. A total of 12497322 patients were analyzed in the meta-analysis (Table 1).
| Ref. | Country | Bariatric surgery1 | Control2 | Follow-up | Risk estimate (95%CI)3 | ||
| Total | Events | Total | Events | ||||
| Adams et al[17], 2009 | United States | 6596 | 25 (0.004) | 9442 | 52 (0.006) | 12.3 | HR: 0.7 (0.43-1.15) |
| Christou et al[20], 2008 | Canada | 1035 | 2 (0.002) | 5746 | 35 (0.006) | 5 | RR: 0.32 (0.076-1.313) |
| McCawley et al[28], 2009 | United States | 1482 | 1 (0.0007) | 3495 | 11 (0.003) | - | |
| Derogar et al[21], 2013 | Sweden | 15095 | 70 (0.005) | 62016 | 373 (0.006) | 10 | |
| Aravani et al[19], 2018 | United States | 39747 | 43 (0.001) | 962860 | 3237 (0.003) | 3 | |
| Mackenzie et al[27], 2018 | Sweden/United States | 8794 | 16 (0.002) | 8794 | 35 (0.004) | 4.6 | HR: 2.19 (1.21-3.96)a |
| Khalid et al[24], 2022 | United States | 19272 | 66 (0.003) | 9636 | 55 (0.006) | 5 | |
| Kwak et al[23], 2019 | United States | 2231 | 5 (0.002) | 2231 | 6 (0.002) | 7.8 | |
| Schauer et al[29], 2019 | United States | 22198 | 105 (0.005) | 66427 | 533 (0.008) | 3.5 | HR: 0.59 (0.36-0.97)a |
| Tao et al[15], 2020 | Denmark/Sweden/Norway/Finland/Iceland | 49931 | 155 (0.003) | 492427 | 3158 (0.006) | 3.1 | |
| Bailly et al[16], 2020 | France | 74131 | 423 (0.006) | 971217 | 12629 (0.013) | 5.7 | HR: 0.68 (0.6-0.77)a |
| Tsui et al[31], 2020 | United States | 71000 | 340 (0.005) | 323197 | 1334 (0.004) | - | |
| Taube et al[30], 2021 | Sweden | 2006 | 58 (0.03) | 2038 | 67 (0.03) | 22.2 | HR: 0.89 (0.62-1.28) |
| Aminian et al[18], 2022 | United States | 5053 | 16 (0.003) | 25265 | 86 (0.003) | 6.1 | |
| Desai et al[22], 2022 | United States | 279145 | 19 (0.0001) | 7398104 | 32276 (0.004) | - | |
| Hussan et al[23], 2022 | United States | 88630 | 88 (0.001) | 327734 | 325 (0.001) | 3 | HR: 1.02 (0.76-1.37) |
| Lazzati et al[26], 2022 | France | 288604 | 329 (0.0001) | 851743 | 4434 (0.005) | 5.7 | HR: 0.93 (0.79-1.08) |
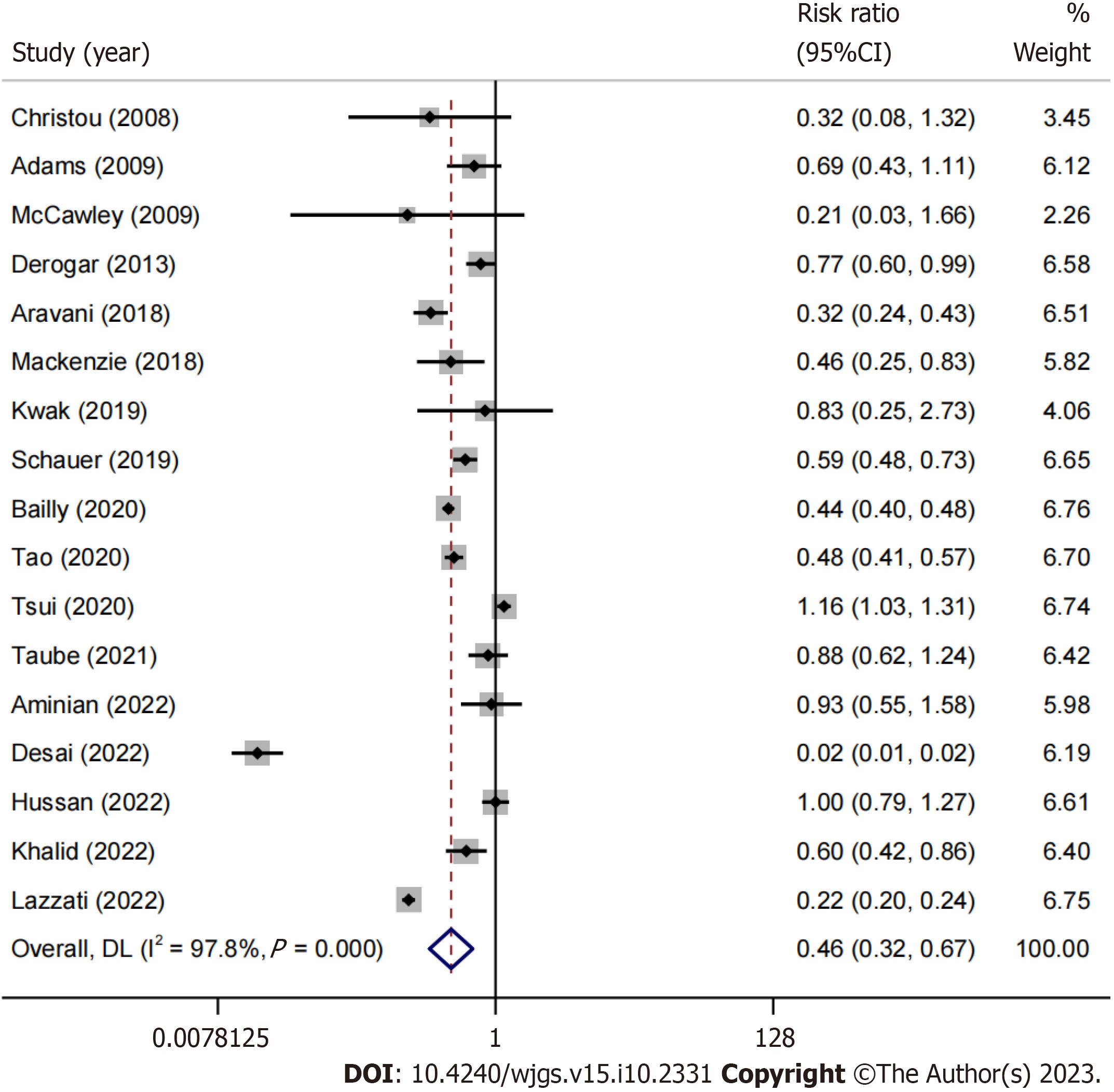
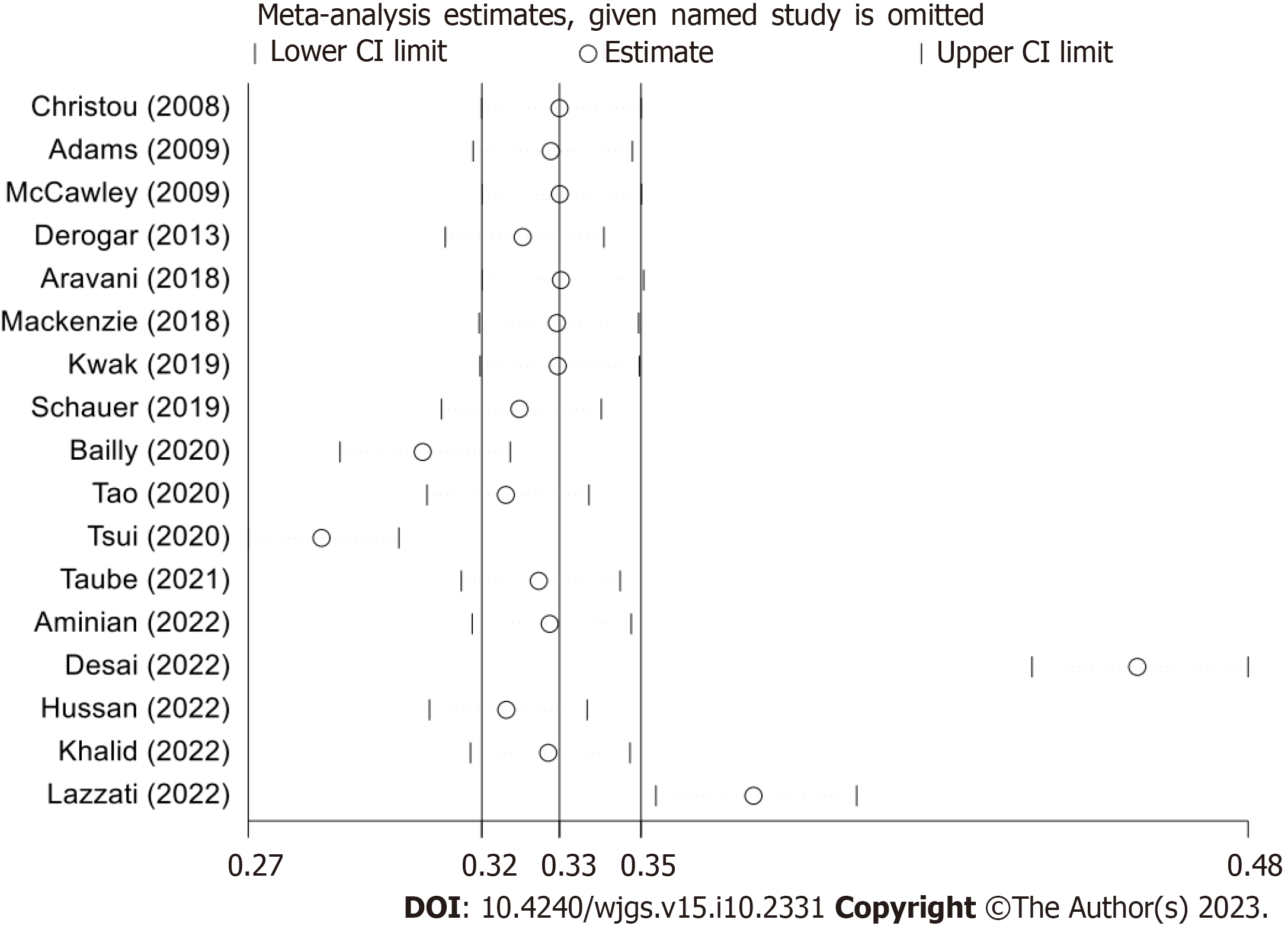
Preliminary results were sourced from 17 studies[15-31]. The risk of developing colorectal cancer during the follow-up period was reduced by 54% (RR: 0.46, 95%CI: 0.32-0.67, P < 0.01, I2 = 97.8%) in morbidly obese patients who underwent bariatric surgery (BS) (Figure 2). Due to the high degree of heterogeneity, a sensitivity analysis was performed (Figure 3). Four studies[22,23,26,31] were identified as primarily responsible for this heterogeneity, but their exclusion did not consistently reduce the heterogeneity; the advantage remained prominent for obese individuals with a history of BS (RR: 0.57, 95%CI: 0.47-0.69, P = 0.0001, I2 =75%). Only three studies separately reported data on colon and rectal cancer[15,23,30]; for colon cancer, the meta-analysis showed a trend favoring BS patients, though not significant (RR: 0.75, 95%CI: 0.46-1.21, P = 0.2444, I2 = 89%). Similarly, considering rectal cancer, there was no apparent trend in favor of BS patients (RR: 0.74, 95%CI: 0.4-1.39, P = 0.3523, I2 = 87%).
We identified five studies from the literature[16,19,21,23,30] that reported CRC incidence exclusively in males across both the BS and non-surgical groups. Meta-analysis showed a trend toward a reduced risk of CRC in males with a history of BS, although not significant (RR: 0.74, 95%CI: 0.43-1.28, P = 0.2798, I2 = 96%). We found a significant reduction in CRC risk (46%) in females with a history of BS (RR: 0.54, 95%CI: 0.37-0.79, P = 0.0014, I2 = 90%). We noted six studies[16,19,21,23,28,30] that detailed female-specific cancer incidence. Regarding the type of weight loss surgery, SG and RYGB were the most common. The meta-analysis of binary outcomes from 4 studies[16,19,27,31] showed no difference in CRC risk for patients with a history of SG or RYGB (RR: 1.02, 95%CI: 0.71-1.45, P = 0.8708, I2 = 43%). Only three studies[16,27,31] reported data following laparoscopic adjustable gastric banding, and thus this technique was not included in the meta-analysis.
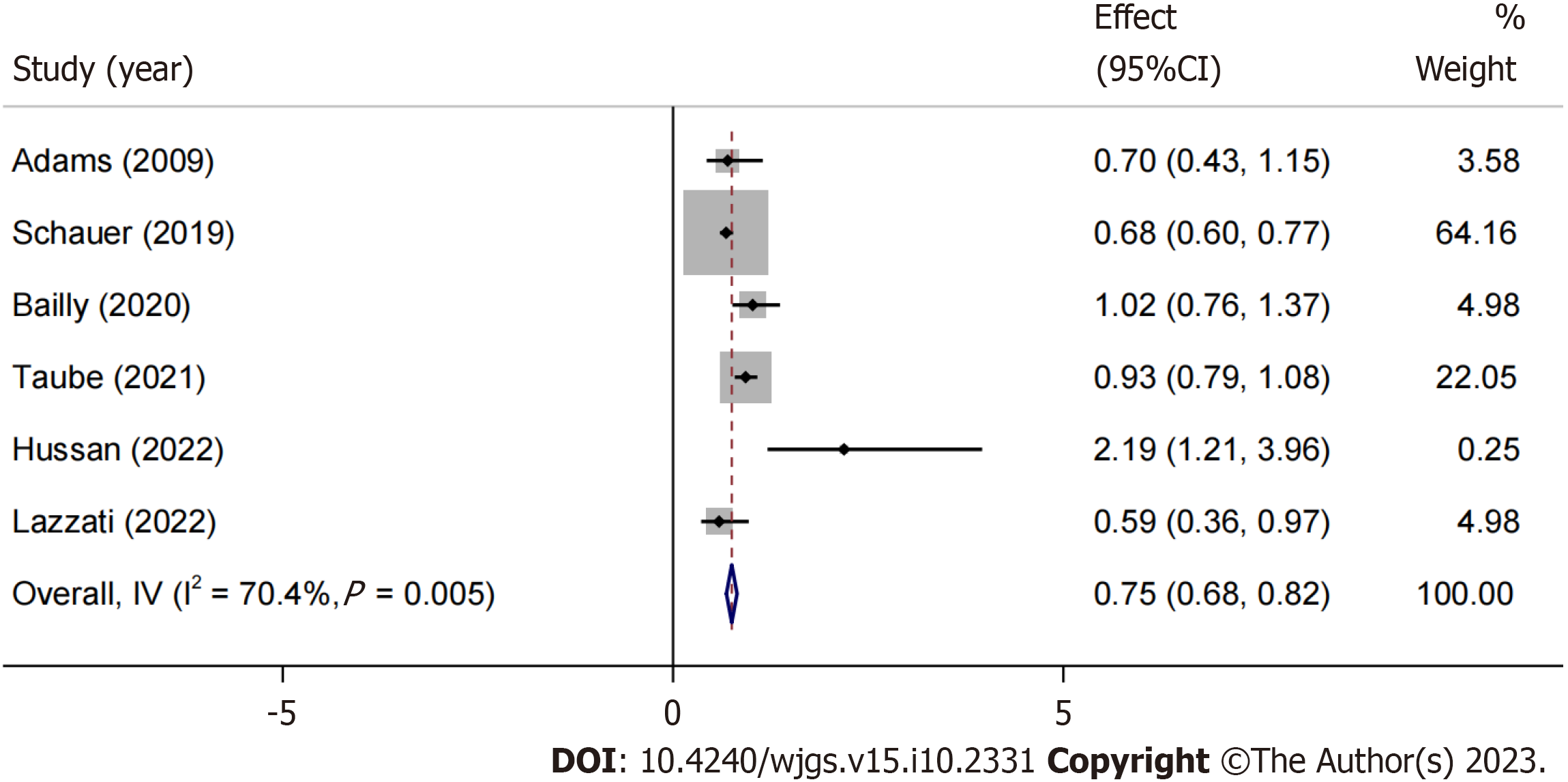
We focused on articles that reported HR estimates for CRC development in obese individuals, irrespective of their history of undergoing BS[16,17,23,26,27,29,30]. Although there was a trend favoring the BS group (HR: 0.88, 95%CI: 0.69-1.12, P = 0.2974, I2 = 77%), the meta-analysis of HR did not find significant estimates favoring either the BS or non-surgical groups. However, after sensitivity analysis and exclusion of the study by Mackenzie et al[27], a significant reduction of 25% in HR was observed for obese patients with a history of BS (HR: 0.75, 95%CI: 0.68-0.82, P = 0.005, I2 = 70.4%) (Figure 4).
Using the NOS, we evaluated the methodological quality of each randomized controlled trial. Overall, 1 study scored 7 points, 15 studies scored 8 points, and 1 study scored 9 points. No studies were blinded, and there was no evidence of concealed allocation. No apparent funding bias was found in any of the studies. We found no studies with incomplete outcome data, premature stopping bias, or imbalances at baseline. The risks of bias and corresponding ratios are summarized in Table 2.
| Ref. | Selection | Comparability | Outcome | Total score | |||||
| Representativeness of the exposed cohort | Selection of the non -exposed cohort | Ascertainment of exposure | Demonstration that outcome | Comparability of cohorts | Assessment of outcome | Was follow-up long enough | Adequacy of follow up of cohorts | ||
| Christou et al[20], 2008 | ★ | ★ | ★ | ★★ | ★ | ★ | ★ | 8 | |
| Adams et al[17], 2009 | ★ | ★ | ★ | ★ | ★★ | ★ | ★ | ★ | 9 |
| McCawley et al[28], 2009 | ★ | ★ | ★ | ★ | ★ | ★ | ★ | ★ | 8 |
| Derogar et al[21], 2013 | ★ | ★ | ★ | ★ | ★ | ★ | ★ | ★ | 8 |
| Aravani et al[19], 2018 | ★ | ★ | ★ | ★ | ★★ | ★ | ★ | 8 | |
| Mackenzie et al[27], 2018 | ★ | ★ | ★ | ★ | ★ | ★ | ★ | 7 | |
| Kwak et al[25], 2019 | ★ | ★ | ★ | ★★ | ★ | ★ | ★ | 8 | |
| Schauer et al[29], 2019 | ★ | ★ | ★ | ★ | ★ | ★ | ★ | ★ | 8 |
| Bailly et al[16], 2020 | ★ | ★ | ★ | ★ | ★ | ★ | ★ | ★ | 8 |
| Tao et al[15], 2020 | ★ | ★ | ★ | ★ | ★★ | ★ | ★ | 8 | |
| Tsui et al[31], 2020 | ★ | ★ | ★ | ★ | ★ | ★ | ★ | ★ | 8 |
| Taube et al[30], 2021 | ★ | ★ | ★ | ★ | ★ | ★ | ★ | ★ | 8 |
| Aminian et al[18], 2022 | ★ | ★ | ★ | ★ | ★★ | ★ | ★ | 8 | |
| Desai et al[22], 2022 | ★ | ★ | ★ | ★★ | ★ | ★ | ★ | 8 | |
| Hussan et al[23], 2022 | ★ | ★ | ★ | ★ | ★ | ★ | ★ | ★ | 8 |
| Khalid et al[24], 2022 | ★ | ★ | ★ | ★★ | ★ | ★ | ★ | 8 | |
| Lazzati et al[26], 2022 | ★ | ★ | ★ | ★ | ★★ | ★ | ★ | 8 | |
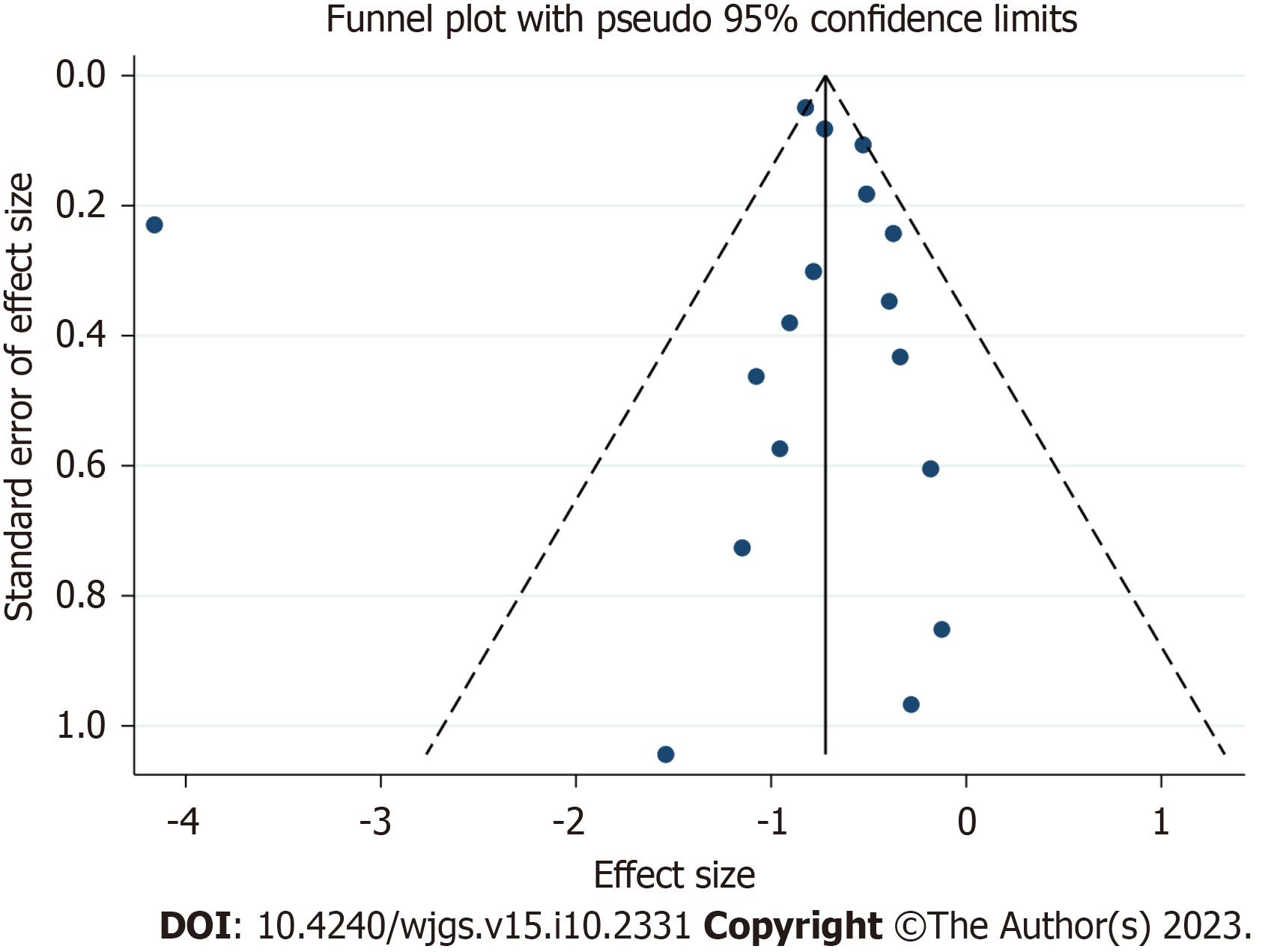
The funnel plot constructed from the observed studies showed symmetry, and no significant publication bias was detected in the funnel plot (Figure 5).
Colorectal cancer is the third most common cancer worldwide and the second leading cause of cancer-related deaths. By 2020, approximately 1.9 million new cases and around 935000 deaths were related to colorectal cancer[32]. Obesity is a well-established risk factor for colorectal cancer. For every 5 kg/m² increase in BMI, the risk of colorectal cancer due to obesity rises by 24% in men and 9% in women. Additionally, obesity is linked to a 47% heightened risk of colorectal adenoma, implying its potential role in the initial stages of colorectal cancer progression. Postoperative risk of CRC is anticipated to decrease following BS, a procedure recognized for its ability to reduce inflammatory markers, mitigate genomic damage, and enhance anti-tumor response[33]. With sustained weight loss and alleviation or improvement of obesity-related comorbidities, BS is considered the most effective method for treating morbid obesity and its associated medical issues.
In this study, we conducted a meta-analysis of 17 studies, encompassing 12497322 patients. Our findings highlight that morbidly obese patient who underwent BS experienced a 54% reduction in CRC risk throughout the follow-up duration. Due to high heterogeneity, a sensitivity analysis was performed. Although no significant differences were found in the subgroup analysis of colon and rectal cancers, a 46% reduction in CRC risk was observed among female patients. However, no significant differences were found in the meta-analysis for various types of bariatric surgery, such as SG and RYGB. Lastly, a meta-analysis of articles reporting HR estimates for colorectal cancer in obese patients showed a significant 25% reduction in HR for those with a history of BS after sensitivity analysis.
The results of this study demonstrate that bariatric surgery has a significant impact on reducing the risk of colorectal cancer in morbidly obese patients. This aligns with earlier research, underscoring the risk-mitigating effect of bariatric surgery on colorectal cancer in those with obesity. Regarding gender disparities, our study pinpointed a marked decrease in CRC risk in female patients’ post-bariatric surgery, potentially stemming from the varying impacts of obesity on colorectal cancer risk between the sexes. Renehan et al[34] found that for every 5 kg/m² increase in BMI, the risk of obesity-related colorectal cancer increased by 24% in men and 9% in women. This suggests that female patients might derive greater benefits from bariatric surgery. Regarding the types of bariatric surgery, no significant differences were found between SG and RYGB in reducing the risk of colorectal cancer. This may suggest that both procedures have similar effects on reducing the risk of colorectal cancer in obese patients. In sum, our findings reinforce the beneficial role of bariatric surgery in curtailing the risk of colorectal cancer for morbidly obese individuals, particularly emphasizing its pronounced impact on females. This finding may help clinicians develop more personalized treatment strategies to reduce the risk of colorectal cancer in obese patients. Nonetheless, subsequent studies should delve deeper into the enduring effects of varied bariatric surgical methods and investigate other potential determinants.
By elucidating the role of bariatric surgery in reducing the risk of colorectal cancer in morbidly obese patients, this study emphasizes the importance of proactive intervention for obese individuals. Firstly, this research reveals the positive impact of bariatric surgery on reducing the risk of colorectal cancer in obese patients, particularly among female patients. This robustly advocates for the formulation of tailored treatment strategies that cater to individual genders. Secondly, the study found that SG and RYGB have similar effects on reducing the risk of colorectal cancer. This finding helps clinicians make more informed decisions when selecting the most suitable bariatric surgery for obese patients.
Lastly, the results of this study hold significant implications for clinicians and patients. Grasping the influence of bariatric surgery on colorectal cancer risk enables doctors and patients to judiciously evaluate the risks and benefits during treatment planning, facilitating tailored medical interventions for obese patients. This study's findings are pivotal in deciphering risk management for obese patients prone to colorectal cancer, bolstering both the efficacy and personalization of clinical treatments. Subsequent studies should delve deeper into the enduring impacts of varied bariatric surgeries and explore other potential determinants affecting colorectal cancer risk among obese patients.
Data from Hussan et al[23] indicates that men have an increased risk of colorectal cancer following weight loss surgery compared to women. In female patients, the risk of CRC decreased following RYGB compared to the control group, but not following SG. Our study found a significant 46% reduction in colorectal cancer risk among female patients, but the incidence rate was not associated with either RYGB or SG weight loss surgery. Bustamante-Lopez et al[35] specifically analyzed the impact of bariatric surgery on early-onset colorectal neoplasia, concluding no significant impact of bariatric surgery on Early-Onset Colorectal Cancer risk. In contrast, our study provided a broader perspective, revealing a significant 54% reduction in colorectal cancer risk among morbidly obese patients undergoing bariatric surgery, especially highlighting a 46% risk reduction in females. Research highlighted in Pararas et al[36] suggests that type 2 diabetes is a component of metabolic syndrome in morbidly obese patients and is an independent prognostic factor associated with increased colorectal cancer risk. The varying degrees of metabolic improvement and alleviation of type 2 diabetes may be the underlying cause for the differences in colorectal cancer risk among different weight loss surgery types. SG outperformed RYGB in warding off colorectal cancer, suggesting that malabsorptive surgeries exert a gentler effect, which might lead to unfavorable alterations in the colonic and rectal microenvironments. Tao et al[15] suggests that alterations in the gut microbiota may play a key role in the initiation and progression of colorectal cancer. It is worth noting that the gut microbiota in RYGB patients shares similarities with that of cancer patients, but our study found no association between RYGB or SG weight loss surgery and the incidence rate.
Recently, Davey et al[37] unveiled evidence pointing to a diminished CRC risk following bariatric surgery among obese subjects. Our research aligns with these findings but further expands upon them. We drew from a comprehensive dataset sourced from 17 studies, involving a significant sample size of 12497322 patients. This expansive dataset furnishes a comprehensive perspective on the interplay between bariatric surgery and CRC. Unlike Davey et al[37], who solely employed odds ratios, our study incorporates both RR and HR metrics, enhancing the depth of our risk assessment. Additionally, where Davey et al[37] focused predominantly on RYGB and SG surgeries, we identified a generalized protective effect of diverse bariatric procedures against CRC. Importantly, our analysis indicates a 46% reduction in CRC risk among female patients, hinting at potential gender-specific mechanisms. Building on the foundational work of Wilson et al[38], we delved deeper into the intricacies of the CRC-obesity relationship and the protective role of bariatric interventions. We recognized a significant 46% decrease in CRC risk in females, an aspect less emphasized in previous literature. From a methodological standpoint, our adherence to PRISMA, AMSTAR, and MOOSE standards vouched for the accuracy and reproducibility of our study. In comparison to Pararas et al[36], our extensive sample provides further support for the protective association between bariatric surgery and CRC. Our study illuminates the 46% CRC risk reduction in females, a perspective not central to Pararas et al's research[36]. Chierici et al's contributions were foundational in our inquiry[39]. We shed light on the global prevalence of CRC in relation to obesity. Our conclusions, backed by our vast sample, distinguish between the risks associated with colon and rectal cancer and underscore the pronounced 46% risk reduction in female patients.
Research by heavy-c2 underscores obesity as a risk factor for colorectal cancer. While some investigations hint at an escalated risk of colorectal cancer post weight loss surgery, the link remains neither significant nor robust. Our study involved 1.2 million participants and employed a population-based cohort research method, studying morbidly obese patients who underwent weight loss surgery in Denmark, Finland, Iceland, Norway, and Sweden between 1980 and 2015. The outcomes failed to offer compelling evidence linking weight loss surgery to a notable increase in rectal cancer risk. This could be attributed to limited sample size, inconsistent data, among other potential reasons. Our findings show that the risk of colorectal cancer significantly decreased during the follow-up period for morbidly obese patients who underwent weight loss surgery. Notably, three of the studies in our analysis exhibited abbreviated follow-up durations. This insinuates a potential limitation in these studies regarding insights into the protracted risk of colorectal cancer. Nevertheless, they provide crucial data regarding immediate risks, which contribute substantially to the overall effect estimate. The aforementioned studies with shorter follow-up durations might introduce some degree of bias. This could slightly overestimate or underestimate the risk of colorectal cancer. However, our sensitivity analysis revealed that omitting these three studies did not result in a significant change in the overall effect size.
Treatments for obesity include lifestyle changes (nutrition education, behavioral counseling, physical exercise), medications, and weight loss surgery for severe obesity. Weight loss surgery is evidenced to markedly trim down enduring weight among severely obese individuals, simultaneously diminishing mortality rates. When studying the relationship between weight loss surgery and cancer incidence, it is challenging to separate the effects of surgery from the multiple associated changes. It is essential to consider that weight loss surgery is more commonly performed in younger individuals, while cancer is more frequently observed in older individuals[40]. Our investigation might not have sufficiently addressed the age distribution variances, potentially affecting our final conclusions. Our study may not have fully considered the time-varying analysis, meaning that the cancer risk in obese patients may change over time during the follow-up period. Our chief focus was on gauging the impact of weight loss surgery on colorectal cancer risk, sidelining other treatment modalities like lifestyle alterations and medications. Such treatments could influence the colorectal cancer risk and warrant exploration in subsequent studies. Our investigation might not have comprehensively catered to every confounding element, including dietary habits, genetic factors, and environmental influences. These factors may also influence colorectal cancer risk and warrant more detailed consideration in future studies. Chronic inflammation stemming from a compromised barrier can disturb the balance between beneficial and deleterious bacteria in the GI tract, amplifying the likelihood of CRC and T2D onset. Surgery stands as a frontline treatment approach for both primary and metastatic colorectal cancer, whereas probiotics aren't recognized as a viable treatment alternative[41].
Weight loss surgery stands validated in its efficacy to curtail weight, mitigate obesity-associated complications, and lower cancer occurrence rates. This meta-analysis confirms the protective effect of weight loss surgery on colorectal cancer through the assessment of RR and HR, particularly in women. Moreover, this assessment was independent of the type of surgery performed. Further research is needed to confirm these findings.
Obesity, with varying global definitions due to body fat disparities between Western and Asian populations, elevates colorectal cancer risk by 7% to 60%. Weight loss surgeries, especially Roux-en-Y laparoscopic gastric bypass (LRYGB) and laparoscopic sleeve gastrectomy (LSG), reduce this risk. Our meta-analysis addresses the varying post-surgery outcomes and research gaps from prior studies.
LRYGB and LSG surgeries exhibit potential impacts on colorectal cancer, but current studies show inconsistencies. Our research aims to bridge these gaps, informing clinical choices, shaping policies, and directing future investigations.
To quantitatively analyze the link between obesity and colorectal cancer, assess the impact of LRYGB and LSG surgeries, discern gender-specific risks, and refine methodological approaches in meta-analyses.
We conducted a comprehensive search on PubMed, Embase, Web of Science, and Cochrane Library using terms including "obesity," "colorectal cancer," "gastric bypass," and "sleeve gastrectomy." We then assessed the retrieved articles based on the PRISMA-guided the Population, Intervention, Comparison, Outcome, and Study design framework: Population (adults with morbid obesity), Intervention (weight loss surgery), Comparison (observation or other treatments), Outcome (risk of colorectal cancer), and Study design (comparative studies with minimum ten patients/group).
Analyzing 17 studies with over 12 million patients, we found that bariatric surgery (BS) patients had a 54% reduced colorectal cancer risk. Subgroup insights showed a 46% risk reduction in females post-BS, while male trends were inconclusive. Specific surgical methods like sleeve gastrectomy and RYGB didn't show differential benefits. Rigorous hazard ratio and Newcastle-Ottawa Scale assessments validated our findings, with no detected publication bias. However, distinct outcomes for colon and rectal cancer remained unresolved.
Bariatric surgery notably lowers colorectal cancer risk, especially in females, irrespective of the surgical type. Our expansive meta-analysis, adhering to rigorous standards, offers a nuanced insight into this relationship.
Future studies should unravel the mechanisms behind bariatric surgery's protective effect, explore gut microbiota's role, compare surgery types, assess long-term impacts, and consider confounding factors.
Provenance and peer review: Unsolicited article; Externally peer reviewed.
Peer-review model: Single blind
Specialty type: Gastroenterology and hepatology
Country/Territory of origin: China
Peer-review report’s scientific quality classification
Grade A (Excellent): 0
Grade B (Very good): B
Grade C (Good): C
Grade D (Fair): 0
Grade E (Poor): 0
P-Reviewer: Bustamante-Lopez LA, Brazil; Salimi M, Iran S-Editor: Liu JH L-Editor: A P-Editor: Xu ZH
| 1. | Hruby A, Hu FB. The Epidemiology of Obesity: A Big Picture. Pharmacoeconomics. 2015;33:673-689. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 1738] [Cited by in RCA: 1709] [Article Influence: 170.9] [Reference Citation Analysis (0)] |
| 2. | Lascano CA, Kaidar-Person O, Szomstein S, Rosenthal R, Wexner SD. Challenges of laparoscopic colectomy in the obese patient: a review. Am J Surg. 2006;192:357-365. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 56] [Cited by in RCA: 62] [Article Influence: 3.3] [Reference Citation Analysis (0)] |
| 3. | Flegal KM, Graubard BI, Williamson DF, Gail MH. Cause-specific excess deaths associated with underweight, overweight, and obesity. JAMA. 2007;298:2028-2037. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 1069] [Cited by in RCA: 1102] [Article Influence: 61.2] [Reference Citation Analysis (0)] |
| 4. | WHO Expert Consultation. Appropriate body-mass index for Asian populations and its implications for policy and intervention strategies. Lancet. 2004;363:157-163. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 7065] [Cited by in RCA: 8241] [Article Influence: 392.4] [Reference Citation Analysis (0)] |
| 5. | Lovejoy JC, Sainsbury A; Stock Conference 2008 Working Group. Sex differences in obesity and the regulation of energy homeostasis. Obes Rev. 2009;10:154-167. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 266] [Cited by in RCA: 292] [Article Influence: 18.3] [Reference Citation Analysis (0)] |
| 6. | Larsson SC, Wolk A. Obesity and colon and rectal cancer risk: a meta-analysis of prospective studies. Am J Clin Nutr. 2007;86:556-565. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 442] [Cited by in RCA: 445] [Article Influence: 24.7] [Reference Citation Analysis (0)] |
| 7. | Moghaddam AA, Woodward M, Huxley R. Obesity and risk of colorectal cancer: a meta-analysis of 31 studies with 70,000 events. Cancer Epidemiol Biomarkers Prev. 2007;16:2533-2547. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 431] [Cited by in RCA: 448] [Article Influence: 26.4] [Reference Citation Analysis (0)] |
| 8. | Reeves GK, Pirie K, Beral V, Green J, Spencer E, Bull D; Million Women Study Collaboration. Cancer incidence and mortality in relation to body mass index in the Million Women Study: cohort study. BMJ. 2007;335:1134. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 1125] [Cited by in RCA: 1034] [Article Influence: 57.4] [Reference Citation Analysis (0)] |
| 9. | Rapp K, Klenk J, Ulmer H, Concin H, Diem G, Oberaigner W, Schroeder J. Weight change and cancer risk in a cohort of more than 65,000 adults in Austria. Ann Oncol. 2008;19:641-648. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 71] [Cited by in RCA: 69] [Article Influence: 3.8] [Reference Citation Analysis (0)] |
| 10. | Wittgrove AC, Clark GW, Tremblay LJ. Laparoscopic Gastric Bypass, Roux-en-Y: Preliminary Report of Five Cases. Obes Surg. 1994;4:353-357. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 568] [Cited by in RCA: 458] [Article Influence: 14.8] [Reference Citation Analysis (0)] |
| 11. | Castagneto Gissey L, Casella Mariolo JR, Mingrone G. How to Choose the Best Metabolic Procedure? Curr Atheroscler Rep. 2016;18:43. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 10] [Cited by in RCA: 12] [Article Influence: 1.5] [Reference Citation Analysis (0)] |
| 12. | Gómez-Huelgas R. [Bariatric surgery versus conventional medical therapy for type 2 diabetes]. Rev Clin Esp. 2012;212:461. [RCA] [PubMed] [DOI] [Full Text] [Reference Citation Analysis (0)] |
| 13. | Page MJ, McKenzie JE, Bossuyt PM, Boutron I, Hoffmann TC, Mulrow CD, Shamseer L, Tetzlaff JM, Akl EA, Brennan SE, Chou R, Glanville J, Grimshaw JM, Hróbjartsson A, Lalu MM, Li T, Loder EW, Mayo-Wilson E, McDonald S, McGuinness LA, Stewart LA, Thomas J, Tricco AC, Welch VA, Whiting P, Moher D. The PRISMA 2020 statement: an updated guideline for reporting systematic reviews. Syst Rev. 2021;10:89. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 2603] [Cited by in RCA: 4357] [Article Influence: 1089.3] [Reference Citation Analysis (33)] |
| 14. | Sterne JA, Hernán MA, Reeves BC, Savović J, Berkman ND, Viswanathan M, Henry D, Altman DG, Ansari MT, Boutron I, Carpenter JR, Chan AW, Churchill R, Deeks JJ, Hróbjartsson A, Kirkham J, Jüni P, Loke YK, Pigott TD, Ramsay CR, Regidor D, Rothstein HR, Sandhu L, Santaguida PL, Schünemann HJ, Shea B, Shrier I, Tugwell P, Turner L, Valentine JC, Waddington H, Waters E, Wells GA, Whiting PF, Higgins JP. ROBINS-I: a tool for assessing risk of bias in non-randomised studies of interventions. BMJ. 2016;355:i4919. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 7683] [Cited by in RCA: 10723] [Article Influence: 1191.4] [Reference Citation Analysis (2)] |
| 15. | Tao W, Artama M, von Euler-Chelpin M, Hull M, Ljung R, Lynge E, Ólafsdóttir GH, Pukkala E, Romundstad P, Talbäck M, Tryggvadottir L, Lagergren J. Colon and rectal cancer risk after bariatric surgery in a multicountry Nordic cohort study. Int J Cancer. 2020;147:728-735. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 16] [Cited by in RCA: 36] [Article Influence: 6.0] [Reference Citation Analysis (0)] |
| 16. | Bailly L, Fabre R, Pradier C, Iannelli A. Colorectal Cancer Risk Following Bariatric Surgery in a Nationwide Study of French Individuals With Obesity. JAMA Surg. 2020;155:395-402. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 37] [Cited by in RCA: 77] [Article Influence: 19.3] [Reference Citation Analysis (0)] |
| 17. | Adams TD, Hunt SC. Cancer and obesity: effect of bariatric surgery. World J Surg. 2009;33:2028-2033. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 52] [Cited by in RCA: 48] [Article Influence: 3.0] [Reference Citation Analysis (0)] |
| 18. | Aminian A, Wilson R, Al-Kurd A, Tu C, Milinovich A, Kroh M, Rosenthal RJ, Brethauer SA, Schauer PR, Kattan MW, Brown JC, Berger NA, Abraham J, Nissen SE. Association of Bariatric Surgery With Cancer Risk and Mortality in Adults With Obesity. JAMA. 2022;327:2423-2433. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 40] [Cited by in RCA: 199] [Article Influence: 66.3] [Reference Citation Analysis (0)] |
| 19. | Aravani A, Downing A, Thomas JD, Lagergren J, Morris EJA, Hull MA. Obesity surgery and risk of colorectal and other obesity-related cancers: An English population-based cohort study. Cancer Epidemiol. 2018;53:99-104. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 37] [Cited by in RCA: 57] [Article Influence: 8.1] [Reference Citation Analysis (0)] |
| 20. | Christou NV, Lieberman M, Sampalis F, Sampalis JS. Bariatric surgery reduces cancer risk in morbidly obese patients. Surg Obes Relat Dis. 2008;4:691-695. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 179] [Cited by in RCA: 185] [Article Influence: 10.9] [Reference Citation Analysis (0)] |
| 21. | Derogar M, Hull MA, Kant P, Östlund M, Lu Y, Lagergren J. Increased risk of colorectal cancer after obesity surgery. Ann Surg. 2013;258:983-988. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 136] [Cited by in RCA: 119] [Article Influence: 9.9] [Reference Citation Analysis (0)] |
| 22. | Desai D, Singhal S, Koka J. Evaluating the Correlation of Bariatric Surgery and the Prevalence of Cancers in Obese Patients: A Study of the National Inpatient Sample (NIS) Database. Cureus. 2022;14:e23976. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in RCA: 5] [Reference Citation Analysis (0)] |
| 23. | Hussan H, Akinyeye S, Mihaylova M, McLaughlin E, Chiang C, Clinton SK, Lieberman D. Colorectal Cancer Risk Is Impacted by Sex and Type of Surgery After Bariatric Surgery. Obes Surg. 2022;32:2880-2890. [RCA] [PubMed] [DOI] [Full Text] [Cited by in RCA: 15] [Reference Citation Analysis (1)] |
| 24. | Khalid SI, Maasarani S, Wiegmann J, Wiegmann AL, Becerra AZ, Omotosho P, Torquati A. Association of Bariatric Surgery and Risk of Cancer in Patients With Morbid Obesity. Ann Surg. 2022;275:1-6. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 24] [Cited by in RCA: 37] [Article Influence: 12.3] [Reference Citation Analysis (0)] |
| 25. | Kwak M, Mehaffey JH, Hawkins RB, Hedrick TL, Slingluff CL Jr, Schirmer B, Hallowell PT, Friel CM. Bariatric surgery is independently associated with a decrease in the development of colorectal lesions. Surgery. 2019;166:322-326. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 6] [Cited by in RCA: 14] [Article Influence: 2.3] [Reference Citation Analysis (0)] |
| 26. | Lazzati A, Epaud S, Ortala M, Katsahian S, Lanoy E. Effect of bariatric surgery on cancer risk: results from an emulated target trial using population-based data. Br J Surg. 2022;109:433-438. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 9] [Cited by in RCA: 25] [Article Influence: 8.3] [Reference Citation Analysis (0)] |
| 27. | Mackenzie H, Markar SR, Askari A, Faiz O, Hull M, Purkayastha S, Møller H, Lagergren J. Obesity surgery and risk of cancer. Br J Surg. 2018;105:1650-1657. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 76] [Cited by in RCA: 118] [Article Influence: 16.9] [Reference Citation Analysis (0)] |
| 28. | McCawley GM, Ferriss JS, Geffel D, Northup CJ, Modesitt SC. Cancer in obese women: potential protective impact of bariatric surgery. J Am Coll Surg. 2009;208:1093-1098. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 73] [Cited by in RCA: 67] [Article Influence: 4.2] [Reference Citation Analysis (0)] |
| 29. | Schauer DP, Feigelson HS, Koebnick C, Caan B, Weinmann S, Leonard AC, Powers JD, Yenumula PR, Arterburn DE. Bariatric Surgery and the Risk of Cancer in a Large Multisite Cohort. Ann Surg. 2019;269:95-101. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 172] [Cited by in RCA: 265] [Article Influence: 44.2] [Reference Citation Analysis (0)] |
| 30. | Taube M, Peltonen M, Sjöholm K, Palmqvist R, Andersson-Assarsson JC, Jacobson P, Svensson PA, Carlsson LMS. Long-term incidence of colorectal cancer after bariatric surgery or usual care in the Swedish Obese Subjects study. PLoS One. 2021;16:e0248550. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 9] [Cited by in RCA: 27] [Article Influence: 6.8] [Reference Citation Analysis (0)] |
| 31. | Tsui ST, Yang J, Zhang X, Docimo S Jr, Spaniolas K, Talamini MA, Sasson AR, Pryor AD. Development of cancer after bariatric surgery. Surg Obes Relat Dis. 2020;16:1586-1595. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 9] [Cited by in RCA: 31] [Article Influence: 6.2] [Reference Citation Analysis (0)] |
| 32. | D'Amato S, Sofia M, Agosta M, Litrico G, Sarvà I, La Greca G, Latteri S. The impact of bariatric surgery on colorectal cancer risk. Surg Obes Relat Dis. 2023;19:144-157. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 2] [Cited by in RCA: 9] [Article Influence: 4.5] [Reference Citation Analysis (1)] |
| 33. | Afshar S, Kelly SB, Seymour K, Lara J, Woodcock S, Mathers JC. The effects of bariatric surgery on colorectal cancer risk: systematic review and meta-analysis. Obes Surg. 2014;24:1793-1799. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 64] [Cited by in RCA: 75] [Article Influence: 6.8] [Reference Citation Analysis (0)] |
| 34. | Renehan AG, Tyson M, Egger M, Heller RF, Zwahlen M. Body-mass index and incidence of cancer: a systematic review and meta-analysis of prospective observational studies. Lancet. 2008;371:569-578. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 3505] [Cited by in RCA: 3673] [Article Influence: 216.1] [Reference Citation Analysis (1)] |
| 35. | Bustamante-Lopez L, Sulbaran M, Changoor NR, Tilahun Y, Garcia-Henriquez N, Albert M, Soliman M, Monson JRT, Pepe J. Impact of bariatric surgery on early-onset colorectal cancer risk: a systematic review and meta-analysis. Updates Surg. 2023;75:1051-1057. [RCA] [PubMed] [DOI] [Full Text] [Reference Citation Analysis (0)] |
| 36. | Pararas N, Pikouli A, Dellaportas D, Nastos C, Charalampopoulos A, Muqresh MA, Bagias G, Pikoulis E, Papaconstantinou D. The Protective Effect of Bariatric Surgery on the Development of Colorectal Cancer: A Systematic Review and Meta-Analysis. Int J Environ Res Public Health. 2023;20. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 7] [Cited by in RCA: 7] [Article Influence: 3.5] [Reference Citation Analysis (0)] |
| 37. | Davey MG, Ryan OK, Ryan ÉJ, Donlon NE, Reynolds IS, Fearon NM, Martin ST, Heneghan HM. The Impact of Bariatric Surgery on the Incidence of Colorectal Cancer in Patients with Obesity-a Systematic Review and Meta-analysis of Registry Data. Obes Surg. 2023;33:2293-2302. [RCA] [PubMed] [DOI] [Full Text] [Cited by in RCA: 2] [Reference Citation Analysis (0)] |
| 38. | Wilson RB, Lathigara D, Kaushal D. Systematic Review and Meta-Analysis of the Impact of Bariatric Surgery on Future Cancer Risk. Int J Mol Sci. 2023;24. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 76] [Cited by in RCA: 65] [Article Influence: 32.5] [Reference Citation Analysis (0)] |
| 39. | Chierici A, Amoretti P, Drai C, De Fatico S, Barriere J, Schiavo L, Iannelli A. Does Bariatric Surgery Reduce the Risk of Colorectal Cancer in Individuals with Morbid Obesity? A Systematic Review and Meta-Analysis. Nutrients. 2023;15. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 18] [Cited by in RCA: 5] [Article Influence: 2.5] [Reference Citation Analysis (0)] |
| 40. | Casagrande DS, Rosa DD, Umpierre D, Sarmento RA, Rodrigues CG, Schaan BD. Incidence of cancer following bariatric surgery: systematic review and meta-analysis. Obes Surg. 2014;24:1499-1509. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 64] [Cited by in RCA: 70] [Article Influence: 7.0] [Reference Citation Analysis (0)] |
| 41. | Benioudakis E, Karlafti E, Bekiaridou A, Didangelos T, Papavramidis TS. Gestational Diabetes, Colorectal Cancer, Bariatric Surgery, and Weight Loss among Diabetes Mellitus Patients: A Mini Review of the Interplay of Multispecies Probiotics. Nutrients. 2021;14. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in RCA: 6] [Reference Citation Analysis (0)] |