Published online Sep 15, 2022. doi: 10.4251/wjgo.v14.i9.1758
Peer-review started: February 18, 2022
First decision: May 9, 2022
Revised: May 22, 2022
Accepted: July 27, 2022
Article in press: July 27, 2022
Published online: September 15, 2022
Processing time: 202 Days and 22.3 Hours
There is no remedial strategy other than definitive chemoradiotherapy for patients with advanced esophageal squamous cell carcinoma (ESCC) who are not eligible to undergo surgical treatment.
To introduce a novel therapy called endoscopic debulking resection (EdR) followed by additive chemoradiotherapy (CRT) and evaluate its efficacy and safety.
Advanced, inoperable ESCC patients between 1 January 2015 and 30 December 2019 were investigated retrospectively. Patients who received EdR followed by CRT were deemed the EdR + CRT group and those without CRT were deemed the EdR group. Overall survival (OS), progression-free survival (PFS), and adverse events were evaluated.
A total of 41 patients were enrolled. At a median follow-up of 36 mo (range: 1-83), the estimated 1-, 2-, and 3-year cumulative OS rates of patients who underwent EdR plus additive CRT were 92.6%, 85.2%, and 79.5%, respectively, which were higher than those of patients who underwent EdR alone (1-year OS, 83.3%; 2-year OS, 58.3%; 3-year OS, 50%; P = 0.05). The estimated 2-year cumulative PFS rate after EdR + CRT was 85.7%, while it was 61.5% after EdR (P = 0.043). Ac
EdR plus CRT is an alternative strategy for selective advanced inoperable ESCC patients.
Core Tip: Forty-one advanced esophageal squamous cell carcinoma (ESCC) patients were retrospectively enrolled, including 28 patients who underwent endoscopic debulking resection (EdR) plus chemoradiotherapy (CRT) and 13 who received EdR without CRT. Clinicopathological characteristics, perioperative outcomes, cumulative overall survival (OS), and progression-free survival (PFS) rates were analyzed. Our results confirm that EdR is safe and feasible for advanced ESCC patients and that EdR + CRT showed better OS and PFS than EdR alone.
- Citation: Ren LH, Zhu Y, Chen R, Shrestha Sachin M, Lu Q, Xie WH, Lu T, Wei XY, Shi RH. Endoscopic debulking resection with additive chemoradiotherapy: Optimal management of advanced inoperable esophageal squamous cell carcinoma. World J Gastrointest Oncol 2022; 14(9): 1758-1770
- URL: https://www.wjgnet.com/1948-5204/full/v14/i9/1758.htm
- DOI: https://dx.doi.org/10.4251/wjgo.v14.i9.1758
Esophageal carcinoma (EC) is the sixth leading cause of cancer-related death worldwide[1]. The in
Neoadjuvant chemoradiotherapy (nCRT) followed by esophagectomy is currently recommended as the standard therapy for advanced ESCC[4-6]. Advanced ESCC patients who decline to receive surgical treatment or have high surgical risks must choose definitive CRT (dCRT)[7-9]. However, locoregional failure of dCRT is usually unavoidable[10,11]. Patients undergoing dCRT who develop recurrent cancer often have a poor prognosis, with a reported median survival of 4 mo to 28 mo[11]. A series of studies have reported that salvage ER is a promising strategy for locally recurrent lesions after dCRT[12-15], which is good news for recurrent patients. However, salvage ER was deemed to be applicable to superficial lesions only. Furthermore, radiation-induced fibrosis in the submucosa increases the incidence of perforation and bleeding during ER. Therefore, novel strategies that are minimally invasive for advanced inoperable ESCC are urgently needed.
A single-arm prospective study reported by Minashi et al[16] concluded that the combination of ER and selective CRT was comparable to surgery, being regarded as a minimally invasive therapy for T1b(SM1-2)N0M0 patients[16]. Subsequent studies also showed that ER plus CRT had equivalent OS potential to that of esophagectomy for early ESCC patients[16-18], further confirming its high therapeutic value for noncurative ER. However, there are no reports on whether ER plus CRT is suitable for patients with deeper than SM3 invasion.
In this study, we used a new therapy called endoscopic debulking resection (EdR) to treat selected patients diagnosed with advanced ESCC who were unable to undergo surgery, and we extended this treatment option to patients with deeper than T1b (≥ SM3) invasion who were unwilling to receive additional esophagectomy in an attempt to evaluate its efficacy and safety when performed along with additive CRT.
From 1 January 2015 to 30 December 2019, patients diagnosed with clinical stage T1b (SM3)-T4N0/+M0/+ inoperable ESCC in our institution were retrospectively included. The inclusion criteria of patients who underwent EdR were as follows: (1) Protruding tumor growth; (2) Tumor invasion ≥ SM3; and (3) Cervical inoperable ESCC or unwillingness to or unable to receive esophagectomy. Patients who had other concurrent malignancies and needed extra therapies were excluded. Patients who received EdR in our study were all suggested to undergo additional selective CRT. The choice of different CRT strategies was made based on the pathological diagnosis and the patients’ physical tolerance.
All patients were staged with 18fludeoxyglucose positron emission tomography combined with computed tomography (18FDG-PET/CT) or computed tomography (CT). Magnification endoscopy (ME) and endoscopic ultrasound (EUS) were used to assess the T- and N-stage of each patient. The grading of tumors was performed according to the 2010 WHO classification of tumors of the digestive system. The TNM stage of the tumor was determined according to the American Joint Commission on Cancer (AJCC) and Union of International Cancer Control (UICC), 8th edition.
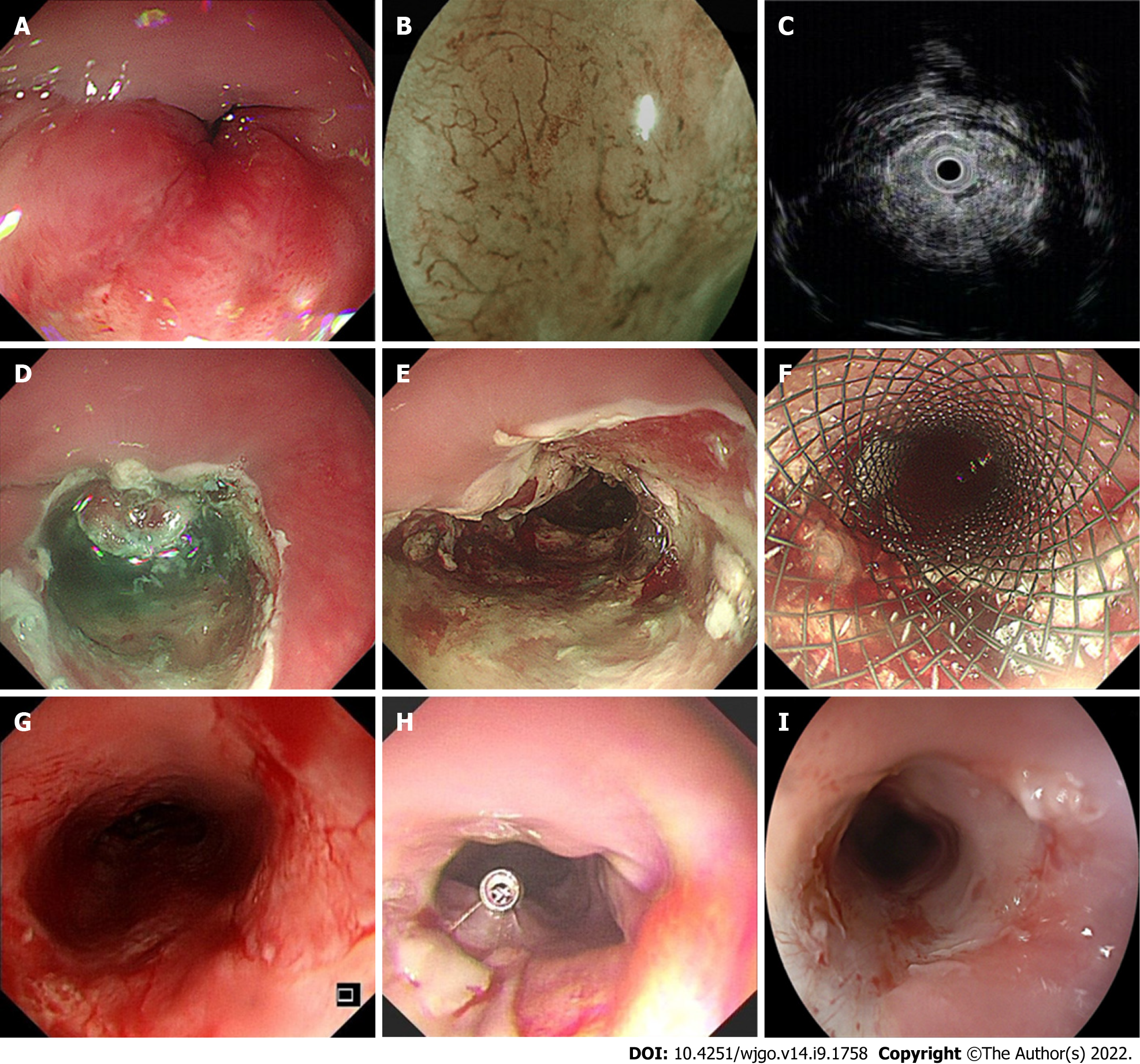
EdR was performed by experienced endoscopists in our center (Figure 1). All patients underwent operation under intubation anesthesia. Carbon dioxide insufflation and a GIF-H260 endoscope (Olympus, Tokyo, Japan) fitted with a transparent cap (Tokyo, Japan) were used during the therapy. We handled a VIO-300D electrosurgical generator (ERBE, Tübingen, Germany), set to Endocut I mode, with Effect 2 for incision and coagulation and Effect 3 (40 W) for dissection. The lesion border was marked by making spots around it with a Hybrid Knife (ERBE). A mixture of saline solution diluted with methylthionine chloride and epinephrine was injected into the fundus of the lesion. Sometimes, hyaluronic acid is used for its efficiency and persistency; however, as the lesions in our study were always deep in the submucosa, it was difficult to create submucosal fluid cushion and lift the lesion completely. In these cases, we did the separation along the stripping imaginary line and dissected lesions carefully step by step. The tumor was removed with a snare by fragment resection. Bleeding vessels were coagulated by hemostatic forceps (FD-410LR; Olympus, Japan). A fully covered esophageal stent (Micro-Tech Co., Ltd., Nanjing, China) was chosen depending on the postoperative wound, which was resected to the muscularis propria. After the operation, all patients fasted for at least 24 h and were treated with acid suppression, hemostasis, and anti-infection agents. The specimens were examined by experienced pathologists who referred to the Japanese Classification of Esophageal Cancer, 11th edition.
Radiotherapy was administered 2 mo after EdR. A megavoltage photon beam (16-18 MV), a CT simulator, and a radiation treatment planning system were used at our institution. Tumor bed volume (GTVtb) was defined as the volume of the primary tumor. GTVtb was expanded to the planning target volume (PGTVtb) by extending 1 cm in all three dimensions. The clinical tumor volume (CTV) included the tumor bed and some optional areas of the regional lymph nodes (bilateral supraclavicular, periesophageal, mediastinal, and perigastric). The planning target volume (PTV) included the CTV plus a margin of 0.5 cm. Three-dimensional radiotherapy treatment planning was performed to reduce the dose to the normal organs. A total dose of 40 Gy to 46 Gy in 20 fractions was delivered with intensity-modulated radiotherapy or anterior/posterior opposed portals according to the normal organs. A tumor boost of 4-6 Gy was delivered to the tumor bed after EdR. All patients were treated 5 d a week.
Based on the patient’s physical state, different chemotherapy regimens were administered based on the pathological diagnosis and the patient’s physical condition. The chemotherapy regimens in our study comprised (1) Cisplatin plus 5-fluorouracil (5-FU): Two cycles of cisplatin (70 mg/m2/d) on day 1 and 5-FU (700 mg/m2/d) on days 1-4 at an interval of 4 wk; (2) Nedaplatin plus 5-FU: the dosage and administration schedule were the same as those for cisplatin plus 5-FU; and (3) Docetaxel plus 5-FU: docetaxel (7.5 mg/m2/d, days 1, 8, 22, and 29) and continuous infusion of 5-FU (250 mg/m2) on days 1-5, 8-12, 15-19, 22-26, 29-33, 36-40, and 43-45 (Supplementary Table 1).
Patients were monitored with weekly hematological examinations, including blood cell counts, liver and kidney function tests, tumor marker tests, electrocardiography, esophagogastroduodenoscopy (EGD), and neck-to-abdominal CT every 3 mo. Local recurrence and metastatic recurrence were defined as a positive biopsy at endoscopy, metastatic lesions to distant organs, and/or local lymph nodes enlarged inside of the irradiation area on 18FDG-PET/CT or CT. The follow-up cutoff date was 31 December 2021.
The primary end point was overall survival (OS), defined as the time from the date of the initial treatment to the date of death from any cause or the date of the last contact. The key secondary end point was progression-free survival (PFS), which was measured as the time from treatment to either progression or death from any cause or the date of the last follow-up. Other secondary endpoints were adverse events (AEs) of treatments, according to the National Cancer Institute Common Terminology Criteria (NCI-CTCAE ver. 4.0).
Categorical data were compared between groups by Fisher’s exact test or the chi-square test. Quantitative data with a nonnormal distribution were compared with the nonparametric Mann-Whitney U test. Quantitative data are expressed as mean ± SD or median (range). Kaplan-Meier survival analysis was performed using SPSS 26.0 statistical software (IBM Corp., Armonk, NY, United States). Univariate and multivariate analyses using the Cox proportional hazards regression model were run to evaluate the influence of covariates on OS and PFS. P < 0.05 was considered significant.
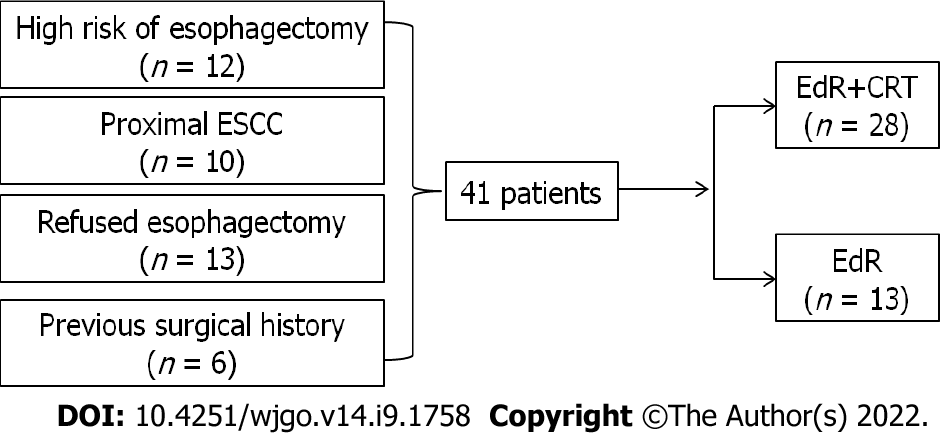
A total of 41 eligible patients were included retrospectively, the study flow diagram is shown in Figure 2. Among them, 12 patients had a high surgical risk of esophagectomy, 10 patients suffered proximal esophageal carcinoma, 13 patients were unwilling to receive esophagectomy, and the other 6 patients were unable to undergo esophagectomy due to a previous surgical history (n = 1, massive small intestine resection; n = 1, lung cancer resection; n = 2, gastric cancer resection; n = 2, cardia cancer resection). Twenty-eight patients underwent EdR plus CRT (EdR + CRT group), while 13 received EdR without CRT (EdR group). Among the 13 patients in the EdR group, 2 preferred not to undergo CRT due to poor physical condition, 9 due to older age (> 70 years), and 2 due to complications with fistulas.
The median age of the 41 enrolled patients was 69 years (range: 38-91). The median follow-up period was 36 mo (range: 1-83). Among the 41 cases, there were 22 (54%) primary tumors located less than 25 cm from the incisors, while 19 (46%) were located more than 25 cm from the incisors. There were 17 (42%), 23 (56%), and 1 (2%) patients diagnosed with clinical stage T1b-SM3, T2, and T3 disease, respectively. Seven (17%) patients had lymph node metastases, while 2 (5%) patients had M1 metastases. The clinical characteristics of the enrolled patients are listed in Table 1. There were no significant differences in the baseline clinical characteristics between the two groups.
| Characteristics | Total (41) | EdR + CRT (28) | EdR (13) | P value |
| Age, median (range), yr | 69 (38-91) | 67 (38-87) | 74 (61-91) | 0.519 |
| Sex, n (%) | 0.524 | |||
| Male | 25 (61) | 18 (43.9) | 7 (17.1) | |
| Female | 16 (39) | 10 (24.4) | 6 (14.6) | |
| Location, n (%) | 0.184 | |||
| ≤ 25 cm | 22 (54) | 17 (41.5) | 5 (12.5) | |
| > 25 cm | 19 (46) | 11 (26.8) | 8 (19.2) | |
| TNM stage, n (%) | 0.400 | |||
| IB | 16 (39) | 10 (24) | 6 (15) | |
| IIA | 18 (43.9) | 14 (34.1) | 4 (9.8) | |
| IIB | 1 (2.4) | 1 (2.4) | 0 (0) | |
| IIIA | 1 (2.4) | 0 (0) | 1 (2.4) | |
| IIIB | 3 (7.3) | 2 (4.9) | 1 (2.4) | |
| IVA | 0 (0) | 0 (0) | 0 (0) | |
| IVB | 2 (4.9) | 1 (2.4) | 1 (2.4) | |
| T stage, n (%) | 0.348 | |||
| T1b | 17 (41.5) | 11 (26.8) | 6 (14.7) | |
| T2 | 23 (56.1) | 17 (41.5) | 6 (14.6) | |
| T3 | 1 (2.4) | 0 (0) | 1 (2.4) | |
| T4 | 0 (0) | 0 (0) | 0 (0) | |
| N stage, n (%) | 0.52 | |||
| N0 | 34 (82.9) | 24 (58.5) | 10 (24.4) | |
| N1 | 2 (4.9) | 1 (2.45) | 1 (2.45) | |
| N2 | 5 (12.2) | 3 (7.3) | 2 (4.9) | |
| M stage, n (%) | 0.539 | |||
| M0 | 39 (95.1) | 27 (65.9) | 12 (29.2) | |
| M1 | 2 (4.9) | 1 (2.45) | 1 (2.45) |
The EdR was performed successfully in all 41 patients (Table 2). Ten (24%) received R0 resection, while 31 (76%) patients received R1 resection (deemed positive horizontal/vertical margins or unjudged margins). The procedure time, measured from the start of marking the lesions to the end of treatment, was 65 ± 29 min (range: 25-150 min). No intraoperative adverse events were observed except for one (1/41) case of mild subcutaneous emphysema, whose symptoms were relieved after 2 d of conservative treatment. Two patients (2/41) suffered delayed bleeding 7 d after the procedure but recovered with anti-acid therapy. Two patients (2/41) developed tracheoesophageal fistula within 2 mo after EdR, of which one died at 24 mo and the other was lost to follow-up at 25 mo. A total of 19 patients (19/41) developed degrees of esophageal stenosis: 2 patients were lost to follow-up at 25 mo and 30 mo, 16 had alleviated dysphagia after receiving retrievable stenting or bougie dilation, and 1 died due to an tracheoesophageal fistula at 24 mo.
| Procedure details and outcomes of EdR | |
| Technical success, n (%) | |
| Success | 41 (100) |
| Failure | 0 (0) |
| R0 resection, n (%) | |
| Yes | 10 (24.4) |
| No | 31 (75.6) |
| Procedure time [mean ± SD (range), min] | 65 ± 29 (25-150) |
| Intraoperative complications, n (%) | |
| Mild subcutaneous emphysema | 1 (2.4) |
| None | 40 (97.6) |
| Post-operative complications, n (%) | |
| Delayed bleeding | 2 (4.9) |
| Esophageal stenosis | 19 (46.3) |
| Tracheoesophageal fistula | 2 (4.9) |
| None | 18 (43.9) |
| Complications of CRT after EdR, n (%) | |
| Myelosuppression | 7 (25) |
| Radiation pneumonia | 3 (10.7) |
| Mucous toxicity | 3 (10.7) |
| None | 15 (53.6) |
| Follow-up period [median (range), mo] | 36 (1-83) |
After EdR, 28 patients received additive CRT. Complications such as myelosuppression were observed in 7 patients, including 5 cases of Grade I, 1 of Grade II, and 1 of Grade III. Three patients developed Grade I radiation pneumonia and 3 patients suffered Grade II mucous toxicity. No severe adverse events were observed during the CRT procedure.
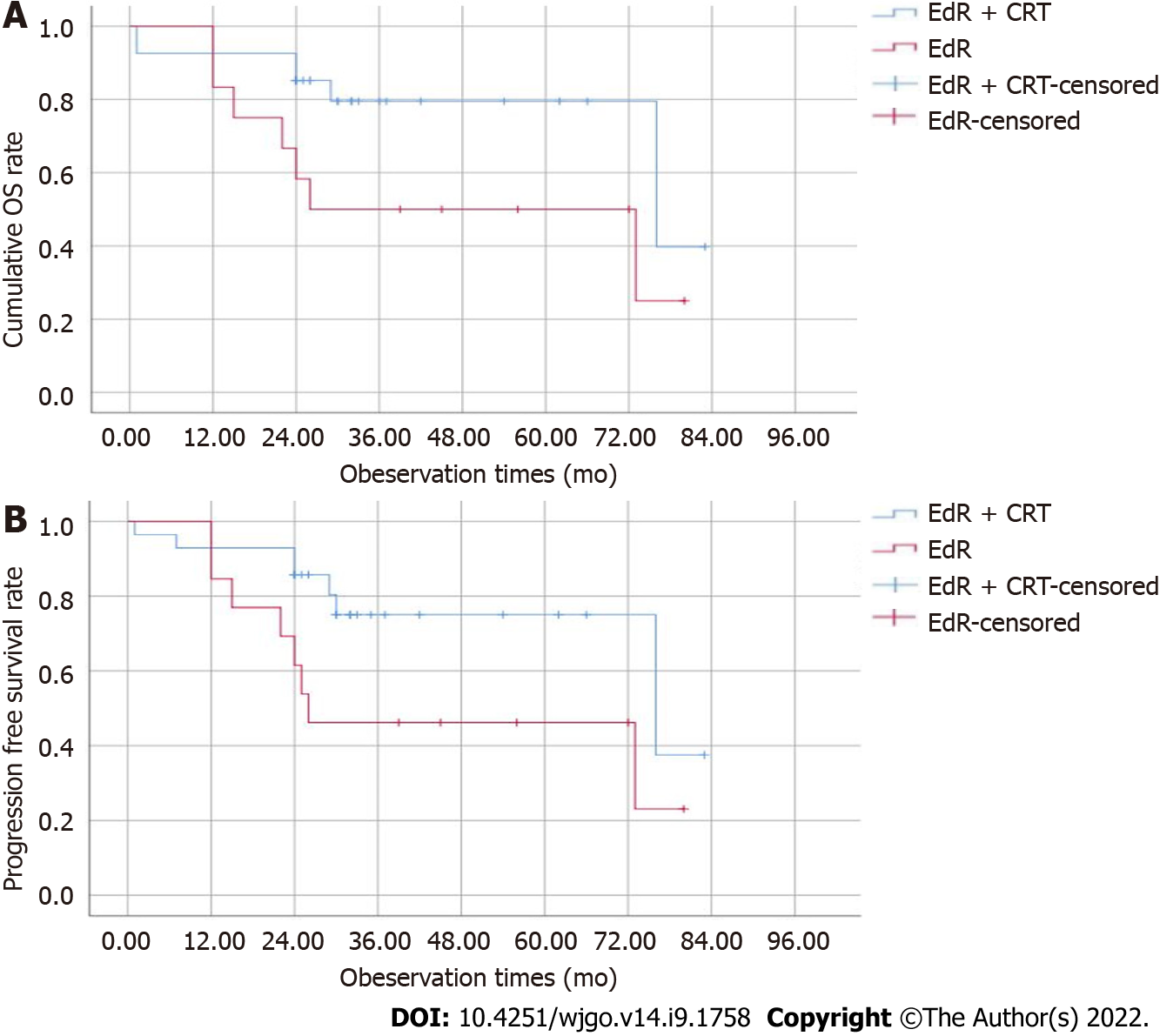
The median follow-up period was 36 (1-83) mo, and 2 patients were lost to follow-up at 25 mo and 30 mo. The estimated 1-, 2-, and 3-year cumulative OS rates of the EdR + CRT group were 92.6%, 85.2%, and 79.5%, respectively, which were higher than those of the EdR group (1-year OS, 83.3%; 2-year OS, 58.3%; 3-year OS, 50%; P = 0.05) (Figure 3A). As shown in Figure 3B, the estimated 2-year PFS rate of the EdR + CRT group was 85.7%, higher than that of the EdR group (61.5%, P = 0.043). Univariate Cox regression analyses showed that clinical stage, additive CRT, lymphoid metastasis and distant metastasis were potential influencing factors of cumulative OS. These variables were included in a multivariate Cox regression analysis, which identified clinical stage as the only factor affecting OS. Similarly, early clinical stage and no lymphoid or distant metastasis were independent protective prognostic indicators for PFS in univariate Cox analyses, but multivariate analysis found only early clinical stage was a protective factor associated with PFS (Table 3).
| Characteristics | OS, n = 41 | PFS, n = 41 | ||||||
| Univariate analysis | Multivariate analysis | Univariate analysis | Multivariate analysis | |||||
| HR (95%CI) | P value | HR (95%CI) | P value | HR (95%CI) | P value | HR (95%CI) | P value | |
| Clinical stage (> IIB vs ≤ IIB) | 18.908 (4.629-77.235) | 0.000 | 18.908 (4.629-77.235) | 0.000 | 11.311 (3.397-37.622) | 0.000 | 11.311 (3.397-37.622) | 0.000 |
| Intervention (EdR vs EdR + CRT) | 4.861 (1.213-19.487) | 0.026 | 0.198 | 0.063 | 0.411 | |||
| T stage (≥ 2 vs < 2) | 68.037 (0.304-15204.04) | 0.126 | 66.824 (0.505-8840.4) | 0.092 | ||||
| N stage (≥ 1 vs 0) | 13.329 (3.309-53.7) | 0.000 | 0.737 | 4.937 (1.621-15.031) | 0.005 | 0.318 | ||
| M stage (≥ 1 vs 0) | 9.13 (1.82-45.775) | 0.007 | 0.876 | 7.035 (1.481-33.418) | 0.014 | 0.906 | ||
| Margin (positive/vague vs negative) | 7.281 (0.84-63.144) | 0.072 | 0.258 | |||||
Patients who undergo noncurative R1 resection or have deeper than SM2 invasion always need additional esophagectomy. However, this concept faced challenges due to the possibility of no residual tumor present in the supplementary surgical specimen. Furthermore, esophagectomy is not a sensible choice for patients who are at high risk from surgery. Patients with advanced cervical or upper ESCC and have a history of lower esophageal or cardiac surgery usually cannot have an esophagectomy. Here, we tested a new treatment, EdR, on selected inoperable advanced ESCCs and extended it to patients with SM3 invasion who were unwilling to receive additional esophagectomy. The results revealed an encouraging short-term OS and low AE rate. Patients who received additive CRT after EdR had a better survival prognosis than those who received only EdR.
Although endoscopic resection (ER) is usually indicated for submucosal lesions, especially SM1-2 invasion, it is not recommended to perform ER for SM3 invasion because of the higher metastasis risk. The literature is unclear regarding additional esophagectomy following noncurative ER. ER can obtain accurate T staging while remove the primary lesion, and adjuvant CRT therapy can further reduce the potential of metastasis or recurrence. Therefore, ER plus CRT is considered an alternative strategy of esophagectomy for clinical stage I ESCC[19-21]. Follow-up studies also showed that ER followed by CRT displayed comparable outcomes of esophagectomy for T1b (SM1-2) cancer[22]. However, it is unclear whether ER followed by CRT is applicable to SM3 invasion. Here, we tentatively performed EdR plus CRT in patients with SM3 invasion. Among the 41 patients, 17 patients had T1b-SM3 cancer, and 5 out of 17 patients underwent R1 resection. Five patients with R1 resection received additional CRT, except 1 due to a history of severe emphysema. For these 17 patients, follow-up lasted 24 to 83 mo, and a favorable prognosis was found, except for 1 failed follow-up at 30 mo post-EdR. It is worth noting that 4 patients in our hospital with SM3 invasion who underwent R1 resection plus supplemental esophagectomy showed negative residual tumors and negative nodal metastases in the surgical specimens. Whether additional surgery or additive CRT be adopted for patients with lesions deeper than SM3 requires a large prospective study.
For advanced, inoperable ESCC, dCRT is the only choice. Previous studies reported the 5-year OS of ESCC patients who received dCRT was only 20%-27%, with a median survival of 14 mo[23,24]. Furthermore, the incidence of local failure of dCRT was up to nearly 50% with poor life quality[25]. Another randomized phase III trial enrolled 267 unresectable ESCC patients who received dCRT; these data showed a median PFS was merely 9.7 mo[26]. There is an urgent need for a new strategy that is more effective than dCRT for unresectable ESCCs. Salvage ER, a complementary treatment after dCRT, has exhibited decent results in recent studies. Yano et al[13] showed that the 5-year survival rate of salvage EMR of stage I-III esophageal cancer patients after dCRT was 49.1%. Another retrospective study, reported by Nakajo et al[25], concluded that the 1-year local relapse-free survival (LRFS) rates of salvage ESD were 86%-100%, confirming the role of salvage ER in patients with dCRT failure[25,27,28]. Nevertheless, these patients all had localized and superficial lesions with no lymph node or distant metastasis. In addition, radiation-induced fibrosis and vessel vulnerability lead to a high risk of acute AEs, such as bleeding or perforation.
In this study, we enrolled 41 patients, including 17 with T1b tumors, 23 with T2, and 1 with T3. Seven (17%) patients had lymph node metastases while 2 (5%) patients had M1 metastases. The primary tumor was partially or completely removed from all enrolled 41 patients, with a mean procedure time of 65 ± 29 min. Only two patients suffered delayed bleeding, and one suffered mild subcutaneous emphysema, with no severe intra- or postoperative AEs observed. All of the patients were cured by conservative therapy. It is recognized that lesions with a circumferential extension of > 3/4 of the esophageal lumen, depth of invasion above M2, and mucosal defects longer than 3 cm are independent risk factors for esophageal stricture[29,30]. Since the lesions in our study were mostly deeper than SM2 and had muscularis propria injuries, esophageal stents were implanted intraoperatively in 15 patients (15/41) to prevent postoperative stricture and delayed bleeding or perforation. Reassuringly, 7 of the 15 patients have no esophageal stenosis during follow-up, while the remaining 8 intake semi-fluid smoothly. It is well known that esophageal stenosis usually occurred late in the radiotherapy. Once the radiation esophagitis and stenosis occurred, the bleeding or perforation risks of endoscopic therapy were extremely high. Although the complication of stenosis in our study was 46% (19/41), it was manageable, as all of these patients intake semi-fluid smoothly. In spite of this, patients who have a high risk of stenosis and choose EdR should fully understand and accept this likely complication. Clinicians must also be cautious when choosing EdR for those with high stricture risk.
Patients who receive dCRT always suffer complications, such as hemorrhage, perforation, radiation esophagitis, pericarditis, pneumonia, and tracheal stenosis. The fatal complication of tracheoesophageal fistula occasionally occurred, especially under the conditions of a high RT dose[31,32]. One study reported that 6 of 49 patients (12%) with T1 or T2 esophageal cancer developed tracheoesophageal fistula, and 3 of them died. A list of studies reporting the rates of esophageal fistula in locally advanced ESCC patients who received dCRT varied from 3.7% to 24%. In our study, there were no fistulas in the EdR + CRT group, while 2 patients suffered fistulas in the single EdR group. We think that incomplete tumor resection and stent mechanical compression were the main reasons for the fistulas. Patients suffering from fistulas always have a poor prognosis due to the increased risk of severe infection and malnutrition. We usually plant a fully covered esophageal stent to plug the fistula, but as a residual necrotic tumor, the fistula cannot be completely cured. To our delight, patients who received EdR plus CRT had no fistulas up to our last follow-up. Our clinical experience tells us that the time point of additive CRT after EdR is extremely important. We implemented CRT at 2 mo after EdR, leaving sufficient time for esophageal mucosa repair. Furthermore, we reduced the ordinary radiation dose and reduced the scope of radiation treatment, relieving the toxicity of radiation. There were no severe adverse events in the EdR + CRT group. Complications, including mild myelosuppression, radiation pneumonia, and mucous toxicity, were observed in 25%, 11%, and 11% of patients, respectively.
One study reported that the 5-year relative survival rate of ESCC patients treated with surgery is only 19%-24%[33]. Zhang et al[34] showed that the 5-year OS of advanced ESCC patients who received adjuvant radiotherapy after surgery was 62.2%, which was much higher than the 5-year OS of patients who underwent surgery alone. The survival benefit of postoperative chemotherapy has also been confirmed[35]. A randomized phase II trial reported by Liu et al[36] reported that the 3-year OS rate of advanced ESCC patients in the CRT group was 38.1% while that in the induction chemotherapy group was 41.8%. Other cohort studies that included stage II-III ESCC patients reported a median DFS of 13 mo in the CRT group[37] and a median OS in the CRT group of 14.1 mo[38]. In our study, the estimated 1-, 2-, and 3-year cumulative OS rates after EdR + CRT were 92.6%, 85.2%, and 79.5%, respectively, and the estimated 2-year PFS rate after EdR + CRT was 85.7%, both satisfactory outcomes. The median survival time of the EdR + CRT group from Kaplan–Meier survival analysis was 76 mo. It is encouraging that 13 patients who received EdR alone also had fair outcomes, with a calculated median survival time of 26 mo. Although the number of EdR was small, the cumulative OS and PFS still were relatively good. According to univariate and multivariate Cox regression analyses, early clinical stage (stage ≤ IIB) and additive CRT after EdR were potential protective factors.
The initial aim of our study was to remove the primary lesion, reduce tumor burden, and enhance the effect of CRT. This strategy was only a daring attempt, and the conclusions in our study need to be treated with caution. As mentioned above, our study has several limitations. First, this was a small, retrospective, short-follow-up study. It is clinically preferable to evaluate the 5-year OS, but we deemed it important to obtain results as soon as possible, so we ultimately designated the primary endpoint as the 3-year OS. Due to the special and strict eligibility criteria of patients, the number of patients in our study was small. Second, the study was conducted at a single institution, which may limit its external generalizability. Large, multicenter, long-term follow-up studies are needed to validate the endoscopic advantages.
Given the limitations above, the results should be interpreted with caution. However, to the best of our knowledge, this is the first study to expand the ER indicator of lesions deeper than SM3, and it is the first study to provide evidence regarding the efficacy and safety of EdR followed by CRT for advanced inoperable ESCC, which might become an attractive therapeutic strategy for selected ESCC patients.
EdR is an alternative strategy for selected advanced inoperable ESCC patients. Additive CRT was not associated with more adverse events but showed better prognosis than EdR alone.
Advanced esophageal squamous cell carcinoma (ESCC) patients who decline surgery or have high surgical risks have no treatment option but definitive chemoradiotherapy (dCRT). However, the complications from high doses of radiation and local recurrence result in a poor prognosis.
To explore a new therapy to treat patients diagnosed with advanced ESCC who were unable to undergo surgery and to extend this therapy to patients with deeper than T1b (≥ SM3) invasion who were unwilling or unable to receive additional esophagectomy.
To evaluate efficacy and safety of the strategy of endoscopic debulking resection (EdR) with additive chemoradiotherapy (CRT) for selected advanced ESCC patients.
Patients who received (EdR) followed by CRT were deemed the EdR + CRT group and those without CRT were deemed the EdR group. Outcomes of overall survival (OS), progression-free survival (PFS), and adverse events were evaluated.
This study showed promising short-term overall and cancer-specific survival after EdR plus additive CRT, with estimated 1-, 2-, and 3-year cumulative OS rates of 92.6%, 85.2%, and 79.5%, respectively, and a 2-year cumulative PFS rate of 85.7%. Early clinical stage (stage ≤ IIB) and additive CRT were potential protective factors for cumulative OS.
EdR plus CRT is relatively safe and feasible for selected advanced inoperable ESCC patients.
The authors will continue to follow up the enrolled patients and increase the sample size to validate the endoscopic advantages and disadvantages.
We wish to thank Xie WH for help with statistics and Sachin Mulmi Shrestha for language editing.
Provenance and peer review: Unsolicited article; Externally peer reviewed.
Peer-review model: Single blind
Specialty type: Gastroenterology and hepatology
Country/Territory of origin: China
Peer-review report’s scientific quality classification
Grade A (Excellent): 0
Grade B (Very good): 0
Grade C (Good): C, C, C
Grade D (Fair): D
Grade E (Poor): 0
P-Reviewer: Bove A, Italy; Elsayed MO, United Kingdom; Man-i M, Japan; Oguma J, Japan S-Editor: Ma YJ L-Editor: Filipodia P-Editor: Ma YJ
| 1. | Bray F, Ferlay J, Soerjomataram I, Siegel RL, Torre LA, Jemal A. Global cancer statistics 2018: GLOBOCAN estimates of incidence and mortality worldwide for 36 cancers in 185 countries. CA Cancer J Clin. 2018;68:394-424. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 53206] [Cited by in RCA: 55833] [Article Influence: 7976.1] [Reference Citation Analysis (132)] |
| 2. | He Y, Liang D, Li D, Shan B, Zheng R, Zhang S, Wei W, He J. Incidence and mortality of laryngeal cancer in China, 2015. Chin J Cancer Res. 2020;32:10-17. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 17] [Cited by in RCA: 35] [Article Influence: 7.0] [Reference Citation Analysis (0)] |
| 3. | Ishihara R, Arima M, Iizuka T, Oyama T, Katada C, Kato M, Goda K, Goto O, Tanaka K, Yano T, Yoshinaga S, Muto M, Kawakubo H, Fujishiro M, Yoshida M, Fujimoto K, Tajiri H, Inoue H; Japan Gastroenterological Endoscopy Society Guidelines Committee of ESD/EMR for Esophageal Cancer. Endoscopic submucosal dissection/endoscopic mucosal resection guidelines for esophageal cancer. Dig Endosc. 2020;32:452-493. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 127] [Cited by in RCA: 262] [Article Influence: 52.4] [Reference Citation Analysis (0)] |
| 4. | Ando N, Iizuka T, Ide H, Ishida K, Shinoda M, Nishimaki T, Takiyama W, Watanabe H, Isono K, Aoyama N, Makuuchi H, Tanaka O, Yamana H, Ikeuchi S, Kabuto T, Nagai K, Shimada Y, Kinjo Y, Fukuda H; Japan Clinical Oncology Group. Surgery plus chemotherapy compared with surgery alone for localized squamous cell carcinoma of the thoracic esophagus: a Japan Clinical Oncology Group Study--JCOG9204. J Clin Oncol. 2003;21:4592-4596. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 491] [Cited by in RCA: 519] [Article Influence: 24.7] [Reference Citation Analysis (0)] |
| 5. | Ando N, Kato H, Igaki H, Shinoda M, Ozawa S, Shimizu H, Nakamura T, Yabusaki H, Aoyama N, Kurita A, Ikeda K, Kanda T, Tsujinaka T, Nakamura K, Fukuda H. A randomized trial comparing postoperative adjuvant chemotherapy with cisplatin and 5-fluorouracil versus preoperative chemotherapy for localized advanced squamous cell carcinoma of the thoracic esophagus (JCOG9907). Ann Surg Oncol. 2012;19:68-74. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 788] [Cited by in RCA: 1052] [Article Influence: 75.1] [Reference Citation Analysis (0)] |
| 6. | Shapiro J, van Lanschot JJB, Hulshof MCCM, van Hagen P, van Berge Henegouwen MI, Wijnhoven BPL, van Laarhoven HWM, Nieuwenhuijzen GAP, Hospers GAP, Bonenkamp JJ, Cuesta MA, Blaisse RJB, Busch ORC, Ten Kate FJW, Creemers GM, Punt CJA, Plukker JTM, Verheul HMW, Bilgen EJS, van Dekken H, van der Sangen MJC, Rozema T, Biermann K, Beukema JC, Piet AHM, van Rij CM, Reinders JG, Tilanus HW, Steyerberg EW, van der Gaast A; CROSS study group. Neoadjuvant chemoradiotherapy plus surgery versus surgery alone for oesophageal or junctional cancer (CROSS): long-term results of a randomised controlled trial. Lancet Oncol. 2015;16:1090-1098. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 1292] [Cited by in RCA: 1825] [Article Influence: 182.5] [Reference Citation Analysis (0)] |
| 7. | Faiz Z, van Putten M, Verhoeven RHA, van Sandick JW, Nieuwenhuijzen GAP, van der Sangen MJC, Lemmens VEPP, Wijnhoven BPL, Plukker JTM. Impact of Age and Comorbidity on Choice and Outcome of Two Different Treatment Options for Patients with Potentially Curable Esophageal Cancer. Ann Surg Oncol. 2019;26:986-995. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 17] [Cited by in RCA: 18] [Article Influence: 3.0] [Reference Citation Analysis (0)] |
| 8. | Koëter M, van Putten M, Verhoeven RHA, Lemmens VEPP, Nieuwenhuijzen GAP. Definitive chemoradiation or surgery in elderly patients with potentially curable esophageal cancer in the Netherlands: a nationwide population-based study on patterns of care and survival. Acta Oncol. 2018;57:1192-1200. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 20] [Cited by in RCA: 25] [Article Influence: 3.6] [Reference Citation Analysis (0)] |
| 9. | Yamamoto S, Ishihara R, Motoori M, Kawaguchi Y, Uedo N, Takeuchi Y, Higashino K, Yano M, Nakamura S, Iishi H. Comparison between definitive chemoradiotherapy and esophagectomy in patients with clinical stage I esophageal squamous cell carcinoma. Am J Gastroenterol. 2011;106:1048-1054. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 75] [Cited by in RCA: 86] [Article Influence: 6.1] [Reference Citation Analysis (0)] |
| 10. | Versteijne E, van Laarhoven HW, van Hooft JE, van Os RM, Geijsen ED, van Berge Henegouwen MI, Hulshof MC. Definitive chemoradiation for patients with inoperable and/or unresectable esophageal cancer: locoregional recurrence pattern. Dis Esophagus. 2015;28:453-459. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 57] [Cited by in RCA: 71] [Article Influence: 7.1] [Reference Citation Analysis (0)] |
| 11. | Welsh JW, Seyedin SN, Allen PK, Hofstetter WL, Ajani JA, Chang JY, Gomez DR, Amini A, Swisher SG, Blum MA, Younes AI, Nguyen QN, Minsky BD, Erasmus JJ, Lee JH, Bhutani M, Komaki RU. Local Control and Toxicity of a Simultaneous Integrated Boost for Dose Escalation in Locally Advanced Esophageal Cancer: Interim Results from a Prospective Phase I/II Trial. J Thorac Oncol. 2017;12:375-382. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 39] [Cited by in RCA: 53] [Article Influence: 5.9] [Reference Citation Analysis (0)] |
| 12. | Hattori S, Muto M, Ohtsu A, Boku N, Manabe T, Doi T, Ishikura S, Yoshida S. EMR as salvage treatment for patients with locoregional failure of definitive chemoradiotherapy for esophageal cancer. Gastrointest Endosc. 2003;58:65-70. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 59] [Cited by in RCA: 54] [Article Influence: 2.5] [Reference Citation Analysis (0)] |
| 13. | Yano T, Muto M, Hattori S, Minashi K, Onozawa M, Nihei K, Ishikura S, Ohtsu A, Yoshida S. Long-term results of salvage endoscopic mucosal resection in patients with local failure after definitive chemoradiotherapy for esophageal squamous cell carcinoma. Endoscopy. 2008;40:717-721. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 77] [Cited by in RCA: 80] [Article Influence: 4.7] [Reference Citation Analysis (0)] |
| 14. | Hombu T, Yano T, Hatogai K, Kojima T, Kadota T, Onozawa M, Yoda Y, Hori K, Oono Y, Ikematsu H, Fujii S. Salvage endoscopic resection (ER) after chemoradiotherapy for esophageal squamous cell carcinoma: What are the risk factors for recurrence after salvage ER? Dig Endosc. 2018;30:338-346. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 12] [Cited by in RCA: 14] [Article Influence: 2.0] [Reference Citation Analysis (0)] |
| 15. | Al-Kaabi A, Schoon EJ, Deprez PH, Seewald S, Groth S, Giovannini M, Braden B, Berr F, Lemmers A, Hoare J, Bhandari P, van der Post RS, Verhoeven RHA, Siersema PD. Salvage endoscopic resection after definitive chemoradiotherapy for esophageal cancer: a Western experience. Gastrointest Endosc. 2021;93:888-898.e1. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 20] [Cited by in RCA: 16] [Article Influence: 4.0] [Reference Citation Analysis (0)] |
| 16. | Minashi K, Nihei K, Mizusawa J, Takizawa K, Yano T, Ezoe Y, Tsuchida T, Ono H, Iizuka T, Hanaoka N, Oda I, Morita Y, Tajika M, Fujiwara J, Yamamoto Y, Katada C, Hori S, Doyama H, Oyama T, Nebiki H, Amagai K, Kubota Y, Nishimura K, Kobayashi N, Suzuki T, Hirasawa K, Takeuchi T, Fukuda H, Muto M. Efficacy of Endoscopic Resection and Selective Chemoradiotherapy for Stage I Esophageal Squamous Cell Carcinoma. Gastroenterology. 2019;157:382-390.e3. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 149] [Cited by in RCA: 139] [Article Influence: 23.2] [Reference Citation Analysis (0)] |
| 17. | Tanaka T, Ueno M, Iizuka T, Hoteya S, Haruta S, Udagawa H. Comparison of long-term outcomes between esophagectomy and chemoradiotherapy after endoscopic resection of submucosal esophageal squamous cell carcinoma. Dis Esophagus. 2019;32. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 6] [Cited by in RCA: 11] [Article Influence: 1.8] [Reference Citation Analysis (0)] |
| 18. | Suzuki G, Yamazaki H, Aibe N, Masui K, Sasaki N, Shimizu D, Kimoto T, Shiozaki A, Dohi O, Fujiwara H, Ishikawa T, Konishi H, Naito Y, Otsuji E, Yamada K. Endoscopic submucosal dissection followed by chemoradiotherapy for superficial esophageal cancer: choice of new approach. Radiat Oncol. 2018;13:246. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 33] [Cited by in RCA: 49] [Article Influence: 7.0] [Reference Citation Analysis (0)] |
| 19. | Kawaguchi G, Sasamoto R, Abe E, Ohta A, Sato H, Tanaka K, Maruyama K, Kaizu M, Ayukawa F, Yamana N, Liu J, Takeuchi M, Kobayashi M, Aoyama H. The effectiveness of endoscopic submucosal dissection followed by chemoradiotherapy for superficial esophageal cancer. Radiat Oncol. 2015;10:31. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 77] [Cited by in RCA: 80] [Article Influence: 8.0] [Reference Citation Analysis (0)] |
| 20. | Hisano O, Nonoshita T, Hirata H, Sasaki T, Watanabe H, Wakiyama H, Ono M, Ohga S, Honda H. Additional radiotherapy following endoscopic submucosal dissection for T1a-MM/T1b-SM esophageal squamous cell carcinoma improves locoregional control. Radiat Oncol. 2018;13:14. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 19] [Cited by in RCA: 34] [Article Influence: 4.9] [Reference Citation Analysis (0)] |
| 21. | Huang B, Xu MC, Pennathur A, Li Z, Liu Z, Wu Q, Wang J, Luo K, Bai J, Wei Z, Xiang J, Fang W, Zhang J. Endoscopic resection with adjuvant treatment versus esophagectomy for early-stage esophageal cancer. Surg Endosc. 2022;36:1868-1875. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 2] [Cited by in RCA: 5] [Article Influence: 1.3] [Reference Citation Analysis (0)] |
| 22. | Hamada K, Ishihara R, Yamasaki Y, Hanaoka N, Yamamoto S, Arao M, Suzuki S, Iwatsubo T, Kato M, Tonai Y, Shichijo S, Matsuura N, Nakahira H, Kanesaka T, Akasaka T, Takeuchi Y, Higashino K, Uedo N, Iishi H, Kanayama N, Hirata T, Kawaguchi Y, Konishi K, Teshima T. Efficacy and Safety of Endoscopic Resection Followed by Chemoradiotherapy for Superficial Esophageal Squamous Cell Carcinoma: A Retrospective Study. Clin Transl Gastroenterol. 2017;8:e110. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 42] [Cited by in RCA: 41] [Article Influence: 5.1] [Reference Citation Analysis (0)] |
| 23. | Herskovic A, Martz K, al-Sarraf M, Leichman L, Brindle J, Vaitkevicius V, Cooper J, Byhardt R, Davis L, Emami B. Combined chemotherapy and radiotherapy compared with radiotherapy alone in patients with cancer of the esophagus. N Engl J Med. 1992;326:1593-1598. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 1514] [Cited by in RCA: 1445] [Article Influence: 43.8] [Reference Citation Analysis (0)] |
| 24. | Ochi M, Murakami Y, Nishibuchi I, Kubo K, Imano N, Takeuchi Y, Kimura T, Hamai Y, Emi M, Okada M, Nagata Y. Long-term results of definitive chemoradiotherapy for unresectable locally advanced esophageal squamous cell carcinoma. J Radiat Res. 2021;62:142-148. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 6] [Cited by in RCA: 15] [Article Influence: 3.8] [Reference Citation Analysis (0)] |
| 25. | Nakajo K, Yoda Y, Hori K, Takashima K, Sinmura K, Oono Y, Ikematsu H, Yano T. Technical feasibility of endoscopic submucosal dissection for local failure after chemoradiotherapy or radiotherapy for esophageal squamous cell carcinoma. Gastrointest Endosc. 2018;88:637-646. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 25] [Cited by in RCA: 30] [Article Influence: 4.3] [Reference Citation Analysis (0)] |
| 26. | Cooper JS, Guo MD, Herskovic A, Macdonald JS, Martenson JA Jr, Al-Sarraf M, Byhardt R, Russell AH, Beitler JJ, Spencer S, Asbell SO, Graham MV, Leichman LL. Chemoradiotherapy of locally advanced esophageal cancer: long-term follow-up of a prospective randomized trial (RTOG 85-01). Radiation Therapy Oncology Group. JAMA. 1999;281:1623-1627. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 1261] [Cited by in RCA: 1372] [Article Influence: 52.8] [Reference Citation Analysis (0)] |
| 27. | Koizumi S, Jin M, Matsuhashi T, Tawaraya S, Watanabe N, Sawaguchi M, Kanazawa N, Yamada Y, Onochi K, Kimura Y, Ohba R, Kataoka J, Hatakeyma N, Mashima H, Ohnishi H. Salvage endoscopic submucosal dissection for the esophagus-localized recurrence of esophageal squamous cell cancer after definitive chemoradiotherapy. Gastrointest Endosc. 2014;79:348-353. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 23] [Cited by in RCA: 30] [Article Influence: 2.7] [Reference Citation Analysis (0)] |
| 28. | Saito Y, Takisawa H, Suzuki H, Takizawa K, Yokoi C, Nonaka S, Matsuda T, Nakanishi Y, Kato K. Endoscopic submucosal dissection of recurrent or residual superficial esophageal cancer after chemoradiotherapy. Gastrointest Endosc. 2008;67:355-359. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 31] [Cited by in RCA: 29] [Article Influence: 1.7] [Reference Citation Analysis (0)] |
| 29. | Shi Q, Ju H, Yao LQ, Zhou PH, Xu MD, Chen T, Zhou JM, Chen TY, Zhong YS. Risk factors for postoperative stricture after endoscopic submucosal dissection for superficial esophageal carcinoma. Endoscopy. 2014;46:640-644. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 112] [Cited by in RCA: 110] [Article Influence: 10.0] [Reference Citation Analysis (0)] |
| 30. | Katada C, Muto M, Manabe T, Boku N, Ohtsu A, Yoshida S. Esophageal stenosis after endoscopic mucosal resection of superficial esophageal lesions. Gastrointest Endosc. 2003;57:165-169. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 217] [Cited by in RCA: 222] [Article Influence: 10.1] [Reference Citation Analysis (0)] |
| 31. | Gaspar LE, Winter K, Kocha WI, Coia LR, Herskovic A, Graham M. A phase I/II study of external beam radiation, brachytherapy, and concurrent chemotherapy for patients with localized carcinoma of the esophagus (Radiation Therapy Oncology Group Study 9207): final report. Cancer. 2000;88:988-995. [PubMed] |
| 32. | Ishikura S, Nihei K, Ohtsu A, Boku N, Hironaka S, Mera K, Muto M, Ogino T, Yoshida S. Long-term toxicity after definitive chemoradiotherapy for squamous cell carcinoma of the thoracic esophagus. J Clin Oncol. 2003;21:2697-2702. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 281] [Cited by in RCA: 285] [Article Influence: 13.0] [Reference Citation Analysis (0)] |
| 33. | Siegel RL, Miller KD, Jemal A. Cancer statistics, 2019. CA Cancer J Clin. 2019;69:7-34. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 13300] [Cited by in RCA: 15474] [Article Influence: 2579.0] [Reference Citation Analysis (2)] |
| 34. | Zhang Z, Xu L, Di X, Zhang C, Ge X, Sun X. A retrospective study of postoperative radiotherapy for locally advanced esophageal squamous cell carcinoma. Ann Palliat Med. 2019;8:708-716. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 4] [Cited by in RCA: 5] [Article Influence: 1.0] [Reference Citation Analysis (0)] |
| 35. | Zhao P, Yan W, Fu H, Lin Y, Chen KN. Efficacy of postoperative adjuvant chemotherapy for esophageal squamous cell carcinoma: A meta-analysis. Thorac Cancer. 2018;9:1048-1055. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 12] [Cited by in RCA: 24] [Article Influence: 3.4] [Reference Citation Analysis (0)] |
| 36. | Liu S, Luo L, Zhao L, Zhu Y, Liu H, Li Q, Cai L, Hu Y, Qiu B, Zhang L, Shen J, Yang Y, Liu M, Xi M. Induction chemotherapy followed by definitive chemoradiotherapy versus chemoradiotherapy alone in esophageal squamous cell carcinoma: a randomized phase II trial. Nat Commun. 2021;12:4014. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 8] [Cited by in RCA: 29] [Article Influence: 7.3] [Reference Citation Analysis (0)] |
| 37. | Sakin A, Sahin S, Aldemir MN, Iliklerden UH, Kotan MC. Chemoradiotherapy followed by surgery versus observation in esophageal squamous cell carcinoma. J BUON. 2021;26:1509-1516. [PubMed] |
| 38. | Duarte MBO, Pereira EB, Lopes LR, Andreollo NA, Carvalheira JBC. Chemoradiotherapy With or Without Surgery for Esophageal Squamous Cancer According to Hospital Volume. JCO Glob Oncol. 2020;6:828-836. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 5] [Cited by in RCA: 5] [Article Influence: 1.0] [Reference Citation Analysis (0)] |