Copyright
©2009 Baishideng.
World J Gastrointest Oncol. Oct 15, 2009; 1(1): 47-54
Published online Oct 15, 2009. doi: 10.4251/wjgo.v1.i1.47
Published online Oct 15, 2009. doi: 10.4251/wjgo.v1.i1.47
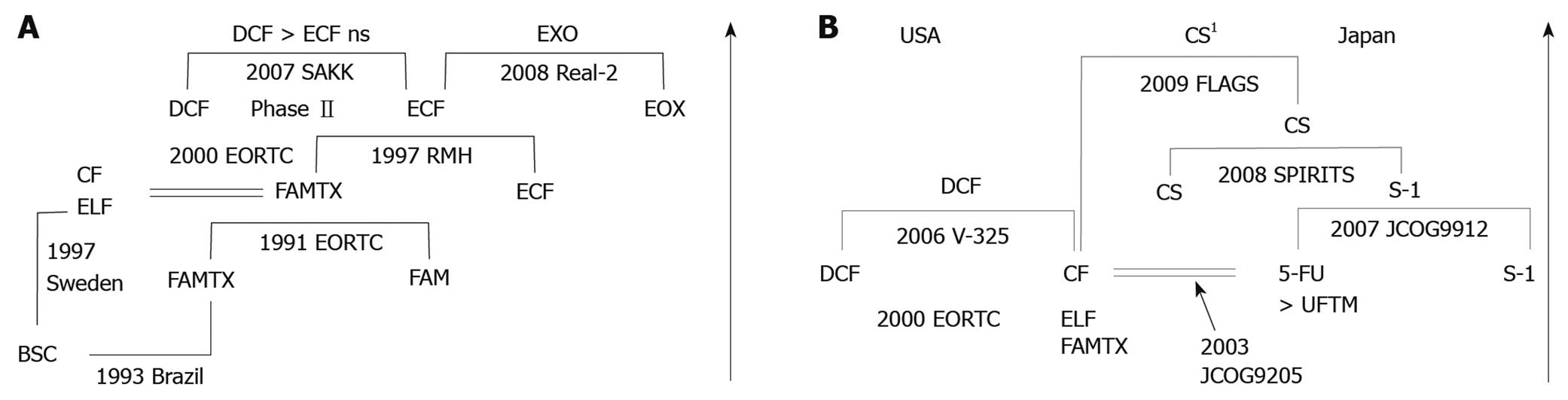
Figure 1 Phase III trials are the most successful for far advanced gastric cancer in Europe (A) and in both United States and Japan (B).
Arrows show chronological direction. BSC: Best supportive care; FAMTX: 5-FU/Adriamycin/Methotrexate; CF: Cisplatin/5-FU; ELF: Etoposide/Leucovorin/5-FU; FAM: 5-FU/Adriamycin/Mitomycin-C; ECF: Epirubicin/Cisplatin/5-FU; EOX: Epirubicin/Oxaliplatin/Capecitabine; DCF: Docetaxel/Cisplatin/5-FU; EORTC: European Organization for Research and Treatment of Cancer; RMH: Royal Marsden Hospital; SAKK: Swiss Group for Clinical Cancer Research; ns: Not significant. UFTM: UFT/Mitomycin-C; CS: S-1/Cisplatin; 1CS regimens in the FLAGS study used 25 mg/m2 of S-1 differently from the SPILITS trial (40 mg/m2 of S-1).
- Citation: Sakuramoto S, Yamashita K, Watanabe M. Newly emerging standard chemotherapies for gastric cancer and clinical potential in elderly patients. World J Gastrointest Oncol 2009; 1(1): 47-54
- URL: https://www.wjgnet.com/1948-5204/full/v1/i1/47.htm
- DOI: https://dx.doi.org/10.4251/wjgo.v1.i1.47