Published online Oct 25, 2015. doi: 10.4253/wjge.v7.i15.1157
Peer-review started: May 13, 2015
First decision: July 10, 2015
Revised: August 24, 2015
Accepted: September 7, 2015
Article in press: September 8, 2015
Published online: October 25, 2015
Processing time: 174 Days and 20.3 Hours
Recent advances in imaging technology have resulted in an increase in incidental discoveries of pancreatic cystic lesions. Pancreatic cysts comprise a wide variety of lesions and include non-neoplastic cysts and neoplastic cysts. Because some pancreatic cysts have more of a malignant potential than others, it is absolutely essential that an accurate diagnosis is rendered so that effective care can be given to each patient. In many centers, endoscopic ultrasound (EUS)-guided fine-needle aspiration (FNA) has emerged as the modality of choice that enables one to distinguish between mucinous and non-mucinous lesion, diagnose malignancy and collect cyst fluid for further diagnostic studies, such as pancreatic enzyme levels, molecular analysis and other tumor biomarkers. The current review will focus on EUS-guided FNA and the cytological diagnosis for pancreatic cysts.
Core tip: Pancreatic cysts comprise non-neoplastic cysts and neoplastic cysts. It is absolutely essential that an accurate diagnosis is rendered so that effective care can be given to each patient. In many centers, endoscopic ultrasound (EUS)-guided fine-needle aspiration (FNA) has emerged as the modality of choice that enables one to distinguish between mucinous and non-mucinous lesion, diagnose malignancy and collect cyst fluid for further diagnostic studies, such as pancreatic enzyme levels, molecular analysis, and other tumor biomarkers. The current review will focus on EUS-guided FNA and the cytological diagnosis and new classification for pancreatic cysts.
- Citation: Martin AK, Zhou Z. Endoscopic ultrasound-guided fine-needle aspiration for the diagnosis of pancreatic cysts by combined cytopathology and cystic content analysis. World J Gastrointest Endosc 2015; 7(15): 1157-1169
- URL: https://www.wjgnet.com/1948-5190/full/v7/i15/1157.htm
- DOI: https://dx.doi.org/10.4253/wjge.v7.i15.1157
The overall prevalence for cystic lesions of the pancreas has been estimated to be no more than 1% of all pancreatic neoplasms[1]. However, the advent of high-resolution imaging modalities has led to the increased frequency of incidentally discovered pancreatic cysts. In the United States, a prevalence of incidental pancreatic cysts estimates about 2.6% to 13.5% of adults[2-4]. The increasing incidence of pancreatic cystic lesions has been directly linked to increasing age[5]. Pancreatic cystic lesions are also being detected sooner rather than later as reflected in the decreasing median sizes of pancreatic cysts both in the United States and in other countries[6]. Although a recent study suggests that only 2% of pancreatic cysts are malignant at diagnosis[7], the trend of increasing discovery of pancreatic cysts is significant because some types of pancreatic cystic lesions carry an augmented risk for malignant transformation.
Pancreatic cysts comprise a wide variety of lesions and include non-neoplastic cysts and neoplastic cysts. The classification and nomenclature of pancreatic cysts are very important for pathologic and clinical diagnosis. The non-neoplastic cysts include pseudocysts, retention cysts, lymphoepithelial cysts, benign epithelial cysts, and congenital cysts. Non-neoplastic cysts are believed to have low to no malignant potential. Neoplastic cysts are typically categorized as mucinous and non-mucinous based on the type of epithelium they possess[8]. The mucinous cysts consist of mucinous cystic neoplasms (MCN) and intraductal papillary mucinous neoplasms (IPMN). The non-mucinous cysts include serous cystadenomas, solid pseudopapillary neoplasms (SPN), cystic pancreatic neuroendocrine tumors (PNET), cystic pancreatic ductal adenocarcinomas (PDA) and its variants, cholangiocarcinoma, acinar cell carcinoma, high-grade neuroendocrine carcinoma (small cell and large cell), pancreatoblastoma, lymphomas, sarcomas, and metastatic tumors. The neoplastic cysts are categorized as being malignant (i.e., PDA, PNET) or having malignant potential (i.e., MCN, IPMN, SPN). Among mucinous subtypes of cysts, it has also been suggested that branch duct IPMN (BD-IPMN), while having malignant potential, may exhibit more indolent behavior compared to main duct-IPMN[9,10].
The management options for pancreatic cystic lesions are as varied as the lesions they are designed to diagnose and treat. Endoscopic ultrasound (EUS)-guided fine-needle aspiration (FNA) is a major technique used in many institutions to sample pancreatic cystic lesions. As will be described in this review, there are multiple ways available to further study these lesions, including cytologic diagnosis and cystic content analysis by chemical and molecular tests. A new histologic classification system for pancreatic lesions has also been introduced to help clinicians and patients understand the malignant potential of each type of pancreatic lesion. Based on new diagnosis and classification of the cystic lesions, most patients need no further treatment. However, many patients require surveillance or other more invasive therapies (i.e., surgical resection) depending upon the risk of malignant transformation. Hence, obtaining an accurate differential diagnosis is of utmost importance in properly managing these patients in such a way that minimizes risk of complications[11].
The initial clinical workup for incidentally discovered pancreatic cysts involves the use of radiologic imaging to further characterize the lesion[12]. MRI with magnetic resonance cholangiopancreatography (MRCP) is the preferred method over pancreatic protocol multidetector (MD) CT because MRCP is able to evaluate the presence of septa, nodules, main-duct involvement, and branch duct involvement. In many cases, it is also able to accurately distinguish between mucinous and non-mucinous cysts[13,14]. Studies have also found that, within the proper clinical context, MRI and CT are capable of determining which pancreatic cystic lesions are more likely to be malignant[15,16]. This is especially true if the features pathognomonic for a given lesion are present. However, in many instances, the combined clinical and radiologic picture is unable to elucidate the type of lesion or its likelihood of harboring malignancy, thus making definitive treatment difficult to achieve. Much of this has to do with the fact that morphologic features of many pancreatic cystic lesions frequently overlap and can appear similar on imaging studies[17]. In this regard, cytologic diagnosis with EUS-FNA is a good means to arriving at a more definitive diagnosis.
EUS-guided FNA is a safe procedure that employs the use of an image guidance system and an endoscope that is passed through the esophagus and into the stomach and/or duodenum. Because the importance of obtaining a good sample as well as adequate sample preparation cannot be overemphasized, many centers perform EUS-guided FNA in conjunction with rapid on-site evaluation (ROSE) by a cytopathologist or cytotechnologist. ROSE has been shown to improve the diagnostic yield of specimens and turnaround time obtained by EUS-guided FNA[18-21]. During ROSE, a cytopathologist or cytotechnologist screens air-dried smears that are first stained with rapid-Romanowsky method, such as Diff-Quik® and Hemacolor®, in order to determine specimen adequacy and to give a preliminary diagnosis, if possible. The rest smears can be alcohol-fixed and stained with the Papanicolaou stain. Additional FNA samples are collected in saline or other alcohol-rich preservative solutions for liquid-based preparations (i.e., ThinPrep®, SurePath®), cytocentrifuge preparations, or cell blocks. Cell blocks are especially helpful in the event that immunohistochemistry is needed to differentiate between the different types of pancreatic lesions. Specimens obtained by EUS-guided FNA can also be used for further diagnostic studies, such as enzymatic testing and molecular testing (to be discussed below in detail in this review).
The advantages of EUS-guided FNA are numerous and include direct real-time visualization of the needle, identification of smaller lesions that can be missed by imaging studies, as well as identification of local metastases and invasion of structures[22]. One study recently showed that the incremental increase in diagnostic yield of EUS and fluid analysis over CT and MRI for prediction of a neoplastic cyst is 36% and 54%, respectively. Complication rates were also low, with pancreatitis being the most common complication (1.1%)[23]. One large prospective multicenter study revealed a complication rate of 6%; bleeding was the most common complication[24]. An extremely rare complication associated with EUS-guided FNA is tumor seeding, especially with IPMN[25-27].
Despite the high specificity of EUS-guided FNA, the main disadvantage that comes with EUS-guided FNA is that samples obtained are often hypocellular. The study by de Jong et al[28] showed that a cytopathologic diagnosis was only possible in one-third to one-half of all cases examined. However, it has also been suggested that the sensitivity, which can range from 60% to 100%, often depends upon an institution’s experience with the technique[29]. One way to potentially overcome the low sensitivity of this procedure is to do cystic wall puncture (CWP), a procedure in which a targeted FNA of the cyst wall is performed after removal of cyst fluid. One study utilizing this method reported adequate cytologic material in 81% of all cases. Complication rate was minimal with only one patient developing mild pancreatitis post-CWP[30]. The study by Rogart et al[31] also showed that CWP may also be helpful in increasing the diagnostic yield of mucinous cystic lesions of the pancreas. Furthermore, there are some important diagnostic pitfalls. For example, GI contamination can cause one to interpret an inadequate specimen as adequate, thus leading to a false-negative diagnosis. Conversely, markedly reactive epithelial cells can be mistaken for malignancy[32,33]. Fortunately, it is possible to avoid these diagnostic pitfalls by making sure cytopathologists have a working knowledge of normal, reactive, and neoplastic pancreatic conditions as well as being sure to correlate all cytologic findings with each patient’s clinical history and imaging studies.
Aspirates obtained from EUS-guided FNA are graded in much the same way as aspirates obtained for other non-gynecological specimens. Specimens that lack sufficient cytologic material to render a diagnosis are designated as “unsatisfactory”. Specimens that have adequate cytologic material and that are helpful in explaining the presence of a radiologically detected lesion are designated as “satisfactory”. Satisfactory specimens are further characterized as “negative for malignancy”, “atypical”, “benign neoplasm”, “suspicious for malignancy” or “positive for malignancy” depending upon the degree of cytologic atypia, cellularity (or lack thereof) and other background features present. Wherever possible, more descriptive terms are also used if a specific pathologic diagnosis can be made. However, there is variable, if not conflicting terminology, used in different institutes and even by individual pathologists. Therefore, tremendous effort has been made to develop a standardized system of classification for pancreatic cytopathology. Recently, Pitman et al[34] published a “standardized terminology and nomenclature for pancreatobiliary cytology: The Papanicolaou Society of Cytopathology Guidelines” (see modified guideline in Table 1). In their categorization, “Non-Diagnostic” lesions are in Category I, lesions classified as “Negative for Malignancy” are in Category II, “Atypical” lesions are in Category III, lesions classified as “Suspicious for Malignancy” are in Category V, and lesions that are “Malignant” are in Category VI. Category IV consists of Category IVA for “Neoplastic: Benign” and Category IVB for “Neoplastic: Other”. Serous cystadenoma is the main neoplasm in Category IVA. In Category IVB, they include both mucinous neoplasms, such as IPMN and MCN, and non-mucinous neoplasms, such as pancreatic endocrine tumor. However, as detailed below, the morphologic, molecular, and immunohistochemical features are very different for these lesions, with mucinous neoplasms having a greater potential to become malignant than non-mucinous neoplasms. Therefore, we suggest that Category IVB should be further separated into Category IVB1 as “Neoplastic: Mucinous neoplasm” and Category IVB2 as “Neoplastic: Non-mucinous neoplasm”. IPMN, MCN, and intraductal papillary neoplasm of the bile ducts should be included in Category IVB1, and pancreatic endocrine neoplasm, SPN, and the rare gastrointestinal stromal tumor should be in Category IVB2. Nevertheless, this classification system serves a significant step towards a much needed uniform categorization of these lesions. Ultimately, the authors hope that each category of pancreatic tumor will be further discussed with gastroenterologists, GI surgeons, and GI oncologists. The following section will now describe the cytologic features of some of the more common pancreatic cystic lesions in accordance with the current classification described by Pitman et al[34].
| Terminology category | Definition | Example interpretations |
| Category I: | No diagnostic or useful information about the solid or cystic lesion sampled | Gastrointestinal contamination only; |
| Non-diagnostic | Non-specific cyst contents with insufficient cyst fluid volume for ancillary testing; | |
| Evaluation limited by scant cellularity | ||
| Category II: | Adequate cellular and/or extracellular tissue to evaluate | Benign pancreatobiliary tissue in the setting of vague fullness and no discrete mass |
| Negative (for malignancy) | Acute pancreatitis | |
| Chronic pancreatitis | ||
| Autoimmune pancreatitis | ||
| Pseudocysts | ||
| Lymph epithelial cyst | ||
| Spleenful/accessory spleen | ||
| Category III: | Cells present with cytoplasmic, nuclear, or architectural features that are not consistent with normal or reactive cellular changes of the pancreas or bile ducts and are insufficient to classify them as a neoplasm or suspicious for a high-grade malignancy | Atypical ductal cells obscured by crush artifact |
| Atypical | Scant population of small monomorphic polygonal cells of unclear origin: Normal cigar cells vs endocrine proliferation | |
| Atypical bile duct epithelium with nuclear features suggestive of repair in a background of acute inflammation | ||
| Atypical bile duct epithelium with mucinous metaplasia and mild nuclear atypia | ||
| Category IVA: | The presence of a cytological specimen sufficiently cellular and representative, with or without the context of clinical, imaging and ancillary studies, to be diagnostic of a benign neoplasm | Scant non-mucinous cuboidal epithelium and scant hemosiderin-laden macrophages in a non-mucinous cyst fluid consistent with the clinical impression of a serous cystadenoma |
| Neoplastic: Benign | ||
| Category IVB1: | Premalignant such as intraductal papillary neoplasm of the bile ducts (IPN-B), IPMN or MCN with low, intermediate or high-grade dysplasia by cytological criteria | MCN: Typically a multiloculated, mucin-producing epithelial neoplasm with sub epithelial ovarian-type stroma that in almost all cases does not communicate with the pancreatic ductal system and in almost all cases occurs in women; located in the body or tail; easily removed comparing life-long surveillance |
| Neoplastic: | IPMN: Primarily intraductal proliferations of ductal epithelium creating a macroscopic lesion resulting in ductal dilatation, cyst formation and/or a mass lesion | |
| Mucinous neoplasm | 1 Main-duct IPMN: Associated with diffuse dilatation of any portion of the main pancreatic duct or the entire pancreas | |
| 2 BD-IPMN: Cysts adjacent to a non-dilated main pancreatic duct | ||
| IPN-B: A papillary proliferation of mucin containing neoplastic cells that may occur anywhere in the ductal system; similar to IPMN | ||
| Category IVB2: | A low-grade malignant neoplasm such as well-differentiated PanNET, SPN or rare GIST | PanNET (pancreatic endocrine tumor and pancreatic endocrine neoplasm): A well-differentiated proliferation of the pancreatic endocrine cells creating a mass lesion greater than 0.5 cm that may or may not be functional by producing inappropriate levels of various hormones and that may or may not demonstrate aggressive features on histological examination |
| Neoplastic: | SPN: A solid, secondarily cystic low-grade epithelial neoplasm with established clonal mutations in cancer-associated genes and an ability to metastasize | |
| Non-mucinous neoplasm | GIST: Spindle cell and/or epithelioid mesenchymal neoplasms with differentiation along the lines of the interstitial cell of Cajal that usually expression c-kit protein (CD117), DOG1 and CD34 by immunohistochemistry; located in a peripanreatic location | |
| Category V: | when some, but an insufficient number of the typical features of a specific malignant neoplasm are present, mainly pancreatic adenocarcinoma | Rare markedly atypical epithelial cells suspicious for adenocarcinoma |
| Suspicious (for malignancy) | Mucinous cyst with high-grade epithelial atypia and abundant coagulate necrosis suspicious for invasive carcinoma | |
| Solid cellular neoplasm with features suspicious for acinar cell carcinoma. Tissue for confirmatory ancillary studies is not available | ||
| Category VIA: | A group of neoplasms that unequivocally display malignant cytological characteristics and include PDAC and its variants, cholangiocarcinoma, acinar cell carcinoma, high-grade neuroendocrine carcinoma (small cell and large cell), pancreatoblastoma, lymphomas, sarcomas and metastases to the pancreas | PDAC: A malignant invasive gland (duct) forming epithelial neoplasm typically composed of classic tubular glands; 85%-90% of all pancreatic malignancies |
| PDAC and variants | Colloid carcinoma (mucinous, non-cystic): Abundant extracellular mucin production, with at least 80% of the tumor on histology demonstrating large pools of extracellular mucin and cuboidal epithelial cells "floating" in the mucin | |
| Medullary carcinoma: Poor histologic differentiation, syncytial growth pattern, pushing borders and an intense lymphoplasmacytic response | ||
| Undifferentiated carcinoma with osteoclast-like giant cells: Distinctive type of sarcomatoid carcinoma with the striking and unique cytohistologic features characterized by a prominent component of reactive osteoclast-like giant cells in a background of spindle cells. | ||
| Undifferentiated carcinoma: A high-grade carcinoma composed of large, undifferentiated, markedly pleomorphic cells; 2%-7% of PDAC | ||
| Category VIA: | A group of neoplasms that unequivocally display malignant cytologic characteristics excluding PDAC and its variants; including acinar cell carcinoma, high-grade neuroendocrine carcinoma (small cell and large cell), cholangiocarcinoma, pancreatoblastoma, lymphomas, sarcomas and metastases to the pancreas | Cholangiocarcinoma: The diagnostic criteria for invasive cholangiocarcinoma are the same as for ductal adenocarcinoma; usually diagnosis by bile duct brushings with high false negative rate due to overlying benign epithelium, insufficient sampling, reactive change with stent; degeneration due to bile |
| Malignancy: | Acinar cell carcinoma: A rare malignant epithelial neoplasm with exocrine acinar differentiation | |
| Others | Poorly-differentiated neuroendocrine carcinoma (small cell carcinoma or large cell neuroendocrine carcinoma): Rare high-grade neuroendocrine tumor with < 1% of pancreatic tumor and 2%-3% of PanNETs | |
| Pancreatoblastoma: A rare neoplasm, primarily of childhood, characterized by acinar differentiation, endocrine differentiation and distinctive squamoid nests | ||
| Non-Hodgkin lymphoma: Rare and usually involve the pancreas secondarily | ||
| Metastatic tumors: Secondary neoplasms involving the pancreas are rare; most common: Renal cell carcinoma |
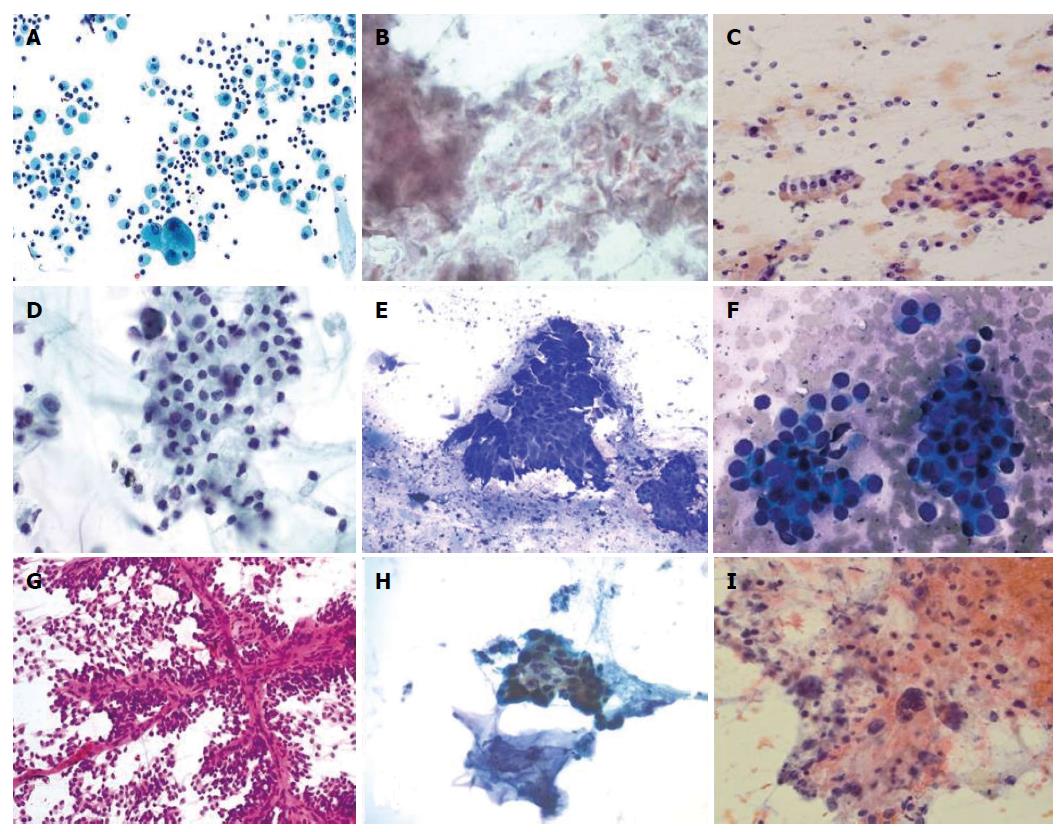
Pseudo cyst (category II: Negative): Pseudocysts are the most common type of pancreatic cysts, accounting for at least 75% of all pancreatic cystic lesions. They generally arise in the setting of acute pancreatitis and are due to autodigestion of the pancreatic parenchyma. By definition, pseudocysts lack an epithelial lining and are instead composed of an inflammatory, fibrous capsule surrounding a region of necrosis. Aspirates are typically paucicellular and consist of granular debris, hemosiderin-laden macrophages, and bile (Figure 1A).
Lymphangiomas and lymphoepithelial cysts (category II: Negative): Lymphangiomas and lymphoepithelial cysts are both very rare benign lesions of the pancreas. The former is characterized cytologically by a uniform population of small, round lymphocytes accompanied by histiocytes, plasma cells, centrocytes, and centroblasts, whereas the latter is characterized by numerous anucleated squamous cells and amorphous debris with rare to no lymphocytes present. Aspirates from lymphangiomas tend to be very cellular[35] (Figure 1B); however, aspirates from lymphoepithelial cysts are largely acellular. Although EUS-guided FNA may have a limited role in identifying lymphoepithelial cysts, it has been proposed that paying attention to signal intensity on MRI may be helpful in identifying these lesions pre-operatively[36].
Serous cystadenoma (category IVA: Neoplastic: benign): Serous cystadenomas comprise 1% to 2% of all pancreatic neoplasms. There are two types that are named based on the number and size of its cysts. Serous microcystic adenomas, which are the more common of the two types, have numerous small cysts, whereas serous oligocystic adenomas have fewer but larger cysts. Serous cystadenomas occur most frequently in older women, with the preferred sites being the body and tail of the pancreas. Aspirates of serous cystadenomas are sparsely cellular and may contain rare fragments of flat sheets and/or loose clusters of cuboidal cells with glycogenated cytoplasm and indistinct cytoplasmic borders (Figure 1C).
Mucinous neoplasm (category IVB: Neoplastic: others): There are two distinctive types of mucinous tumors, namely MCN and IPMN. Because both of these entities share many morphologic features, it is almost impossible to tell the difference between the two based on cytomorphologic features alone. In these cases, direct correlation with clinical and imaging studies is required. In general, MCNs occur almost exclusively in middle-aged women, with most being located in the body or tail of the pancreas. Of note, these lesions are closed cysts that do not communicate with the ductal system. A defining histologic feature of these lesions is the presence of ovarian-type stroma directly beneath mucinous epithelium that is positive for estrogen and progesterone receptors. On the other hand, IPMN is seen more commonly in men and are typically seen in the head of the pancreas. Unlike MCN, IPMN is radiologically shown to communicate with the ductal system (typically involving the main pancreatic duct) and has the ability to grow along the entire length of the pancreatic duct and its branches. Aspirates of MCN and IPMN are hypocellular specimens that contain thick mucin and, if present, columnar mucinous sheets (Figure 1D and E). Cytologic interpretation is somewhat less problematic if nuclear and architectural atypia indicative of dysplasia or malignancy is identified. The WHO uses a three-tier classification based on degree of dysplasia present: benign, borderline, or malignant. Recent years, molecular tests such as KRAS and GNAS mutation are developed for differentiating MCN and IPMN. Nevertheless, given the malignant potential of these lesions, surgical resection is the most often utilized treatment of choice. Since cystic mucinous neoplasms have unique cytopathologic, molecular features and high risk for malignancy compared to non-mucinous neoplasms, we suggest that the Category IVB should be separated into two subcategories: Category IVB1: Neoplastic: Mucinous and IVB2: Neoplastic: Non-mucinous.
Cystic PNET (category IVB: Neoplastic: others): PNET represent approximately 1% to 2% of pancreatic neoplasms. Most PNET are small, functional solid tumors, but cystic pancreatic neuroendocrine tumors (cPanNETs) account for 13% to 17% of PanNETs[37]. PNET can secrete a variety of hormones, including insulin, glucagon, and somatostatin, and adrenocorticotrophic hormone. Although they can occur in any age group, they most commonly occur in adults. Aspirates of well-differentiated PNETs can range from sparsely cellular to highly cellular specimens consisting predominantly of abundant isolated cells and numerous bare nuclei. However, loosely cohesive clusters of cells and pseudorosette formation are not uncommon. The cells are characterized by uniformly round, eccentrically placed nuclei with “salt-and-pepper” chromatin and moderate amounts of cytoplasm (Figure 1F). More poorly differentiated PNETs display more nuclear pleomorphic and higher mitotic activity. In doubtful cases, immunohistochemical stains for chromogranin and synaptophysin can be extremely helpful in confirming the diagnosis if the cell block is available. The surgical resection is the first line treatment. Enucleation or cytoreductive surgery is also recommended for patients with locoregional recurrences or hepatic metastases. Regional adjuvants such as radiofrequency ablation, transarterial chemoembolization, and others are often employed in an attempt to palliate symptoms and prolong survival[38]. Again, because cystic PNETs can be both functional and non-functional tumors with special morphological and immunohistochemical features, it should be separated from MCN into category IVB2: Neoplastic: Non-mucinous.
SPN (category IVB: Neoplastic: others): SPN are uncommon tumors of unknown malignant potential that predominantly occur in young women. Aspirates of these lesions are highly cellular, with the most characteristic features being myxoid or hyalinized vascular stalks lined by single or multiple layers of cells exhibiting round to oval nuclei, nuclear grooves, and indistinct cell borders (Figure 1G). Immunostain for β-catenin with nuclear positivity has emerged as a helpful attribute in diagnosing SPN. Other immunohistochemical stains that are helpful in confirming the diagnosis include CD10, CD56, vimentin and SMAD4. Surgical resection of these tumors leads to a good prognosis. With the special morphological features and immunohistochemical features of these lesions, SPN should be classified as Category IVB2: Neoplastic: non-mucinous.
Pancreatic ductal adenocarcinoma with cystic neoplasm (Category VI: Malignant): Pancreatic ductal adenocarcinomas (PDAC) with cystic neoplasm is the most common malignant cystic neoplasm of the pancreas and usually arises from MCN and IPMN. PDAC typically occur in older individuals, with smoking and alcohol abuse being major risk factors. Despite being able to detect these lesions at earlier stages, long term survival remains abysmal, with 90% of all patients dying within one year of diagnosis. Cytologically, aspirates are usually very cellular and consist of atypical ductal cells with irregular nuclear contours and prominent, centrally placed nucleoli arranged singly or in clusters and sheets (Figure 1H). Mitotic figures can also be seen.
Although it has been established that EUS-guided FNA has a valuable role in the multidisciplinary approach to the management of pancreatic cystic lesions, much controversy remains in regards to its ability to accurately triage patients with incidentally discovered lesions that appear benign on imaging. In one of the early studies performed by Frossard et al[39] in 2003, it was determined that EUS-guided FNA successfully identified the lesion of interest in 65 cases (97%). The overall sensitivity, specificity, positive predictive value, and negative predictive value for EUS-guided FNA in this study were 97%, 100%, 100%, and 95%, respectively[39]. The cytologic diagnosis of cystic lesions with EUS-FNA has been studied extensively with widely variable sensitivity[40-44]. The sensitivity has been reported to range from 23% to 100% and specificity has been reported to range from 71% to 100%[40,45,46]. One meta-analysis showed that the pooled sensitivity and specificity in diagnosing mucinous cystic lesions were 63% and 88%, respectively, in 11 studies and 54% and 92%, respectively, in 4 prospective studies[45]. In one recently published meta-analysis study, the sensitivity and specificity of cytology was 0.42 and 0.99; the sensitivity and specificity of K-RAS was 0.39 and 0.95; and the sensitivity and specificity of the combined test of cytology and K-RAS was 0.71 and 0.88, respectively[47]. The sensitivity in our study (47%) was between two meta-analysis results[46]. We further studied the false negative rate of EUS-FNA, and we found that the false-negative rate (3%) caused by an interpretative error was significantly lower than that caused by a sampling error (23%) (P = 0.003). This finding suggests that sampling error, rather than interpretative error by cytology, is a major cause of high false-negative rates. We further examined the false-negative rate for solid lesions and cystic lesions. The false-negative rate for cystic lesions was significantly higher than that for solid lesions (53% vs 15%; P = 0.005). Recently, Rogart et al[31] reported that cyst wall puncture performed during FNA improved the diagnostic yield for mucinous cysts[31]. In addition, cytologic classification with high-grade epithelial atypia in cystic lesion FNA specimens demonstrated a higher prediction for malignancy and added value for the clinical evaluation of cystic lesions[42,48]. One study also found that certain factors, such as the identification of a solid component and performing more than one pass, resulted in significant increases in sensitivity (as high as 78%)[49]. In light of these issues with sensitivity, a newer series has suggested that EUS-guided FNA, when used in conjunction with other “screening” tests, contributes to a triple-negative screening test (i.e., no high-risk stigmata, no worrisome features, and no high grade atypia on cytology) that has a negative predictive value for malignancy of 99%[50]. In general, EUS-guided FNA has a low sensitivity, but good specificity[45]. More sensitive and specific techniques are needed and should be developed as new technologies emerge, such as cystic fluid analysis by chemical or molecular tests and confocal laser endomicroscopy.
Recently, cystic PNET diagnosis and management received a lot of attention. In one study, cytology made a specific diagnosis of a cystic PNET in 71% of the biopsies compared with a specific diagnosis by EUS in 38% of cases[37]. All cysts but one revealed low carcinoembryonic antigen (CEA) levels (range, 0.2 to > 500 ng/mL; mean, 29.5 ng/mL), and amylase levels were < 500 U/L in all but 2 cases (range, 16-1493 U/L; mean, 205 U/L). In another study, cystic PNETs were found to be larger than solid PNETs (mean 26.8 mm vs 20.1 mm, P = 0.05) and more frequently nonfunctional (96 % vs 80 %, P = 0.03). With histology as the reference standard, EUS-FNA accuracies for malignancy of cystic and solid PNETs were 89.3 % and 90 %, respectively; cystic PNETs were less associated with metastatic adenopathy (22 % vs 42 %, P = 0.03) and liver metastasis (0 % vs 26 %, P < 0.001). Cystic fluid analysis (n = 13), showed benign cystic PNETs had low CEA, Ki-67 ≤ 2 %, and no loss of heterozygosity (LOH). Patients with cystic and solid PNETs had similar recurrence risk up to 5 years after complete resection[51]. In one review which compared EUS and EUS-FNA for cystic PNET, they found that EUS-FNA cytology and cyst fluid analysis is a useful adjunct to abdominal imaging in the diagnosis of pancreatic cystic lesions. They hypothesize that cyst fluid characteristics, including cytomorphological features, is the most accurate test to achieve a preoperative diagnosis and to provide a basis for prognostic prediction[52].
Another technique that shows promise in improving the sensitivity and specificity of detecting and diagnosing pancreatic cystic lesions is confocal laser endomicroscopy. Confocal laser endomicroscopy is a novel imaging technology in which a low-power laser illuminates and scans a single focal plane of the tissue[53-56]. This technique allows for the detection of the microscopic detail of the surface epithelium in pancreatic cysts. Needle-based confocal laser endomicroscopy (nCLE) utilizes a sub-millimeter probe that is compatible with an EUS needle and enables real-time imaging with microscopic detail of pancreatic cystic lesions[56]. The presence of epithelial villous structures based on nCLE was associated with pancreatic cystic neoplasm (P = 0.004) and provided a sensitivity of 59%, specificity of 100%, positive predictive value of 100%, and negative predictive value of 50%. This technique is rather new for evaluating the pancreatic cystic lesions. However, we believe that the development of this new technique may facilitate sampling the most suspicious area of a cyst in the future.
As mentioned previously, pancreatic cystic neoplasms represent a diagnostic challenge for EUS-guided FNA because lining cells may or may not be adequately sampled, thus precluding further classification. The current way of solving this dilemma relies upon a combination of methods and includes visual cyst fluid evaluation at the time of immediate assessment, chemical analysis of cyst fluid, and molecular testing. All of these tests can be utilized to help to differentiate between mucinous and non-mucinous cysts.
The possibility of a mucinous cyst can be strongly suggested by looking for the “string sign”. This can be assessed by stretching out a drop of cyst fluid between the thumb and index finger and subsequently measuring the length of the string of cyst fluid. A length of at least 3.5 mm is believed to be consistent with that of a mucinous cyst. Studies have also shown that mucinous cysts consistently have a higher relative viscosity compared to serum, whereas the opposite is true for non-mucinous cysts, which a lower relative viscosity compared to serum[57].
Amylase: The chemical analysis of cyst fluid relies upon examining pancreatic enzyme levels as well as the presence of tumor markers. Pancreatic enzyme levels are typically used to differentiate between pseudocysts and neoplastic cysts. One of the most important enzymes studied in making this distinction is amylase. Pseudocysts and other non-neoplastic cysts consistently show elevated levels of amylase. In fact, one study showed that an amylase level below 250 U/L virtually excludes pseudocysts from the differential diagnosis[58]. Conversely, amylase is low in neoplastic cysts.
CEA: A variety of tumor markers have been studied for their ability to discriminate between mucinous and non-mucinous cysts[57-60]. According to many studies, CEA levels are the most accurate[60,61]. Although CEA cutoff values of > 192 ng/mL have been shown to have an accuracy of 79%[60], levels > 800 ng/mL have been shown to be highly predictive of mucinous neoplasms with a specificity of 98%. Unfortunately, the sensitivity, even at these markedly elevated levels, is still less than 50%[58]. It is also important to note that CEA cannot be used to distinguish between benign and malignant lesions[62,63]. Amylase, however, may be helpful in this regard[64]. In contrast, a very low CEA level ≤ 5 is 95% specific for pseudocysts, neuroendocrine tumors, and serous cystadenoma[58].
Other markers: Multiple biomarkers have also been studied to identify pancreatic ductal carcinoma (PDAC). Plectin-1, a marker related to PDAC, was found to be a potentially promising biomarker for the detection of malignancy in IPMNs[65]. Plectin-1 expression was assayed using immunohistochemistry in cyst fluid and tissue sample from benign and malignant IPMN, as well as lymph node metastasis from carcinoma arising from IPMN. The sensitivity and specificity were 84% and 83%, respectively. In animal models, Cathepsin E is specifically and highly expressed in PDAC and pancreatic intraepithelial neoplasias (PanINs), A prospective double-blind control study was performed to evaluate the accuracy of this method in diagnosing PDAC and PanINs of all grades (> 82.7%)[66].
K-RAS mutation: Molecular analysis of cyst fluid shows promise in distinguishing not only between mucinous and non-mucinous cysts, but also in the diagnosis of malignant cysts. One study that examined surgically resected specimens showed that the identification of the K-RAS mutation had a sensitivity of 54% and a specificity of 100% for mucinous cysts. The combined used of CEA and K-RAS improved the sensitivity to 83% while decreasing the specificity to 85%[67]. However, a smaller study found that there was no increase in sensitivity when combining CEA and K-RAS[68]. LOH and increased DNA quantity have also been tried as a means of accurately predicting the presence of a mucinous lesion, but the sensitivity for each method is less than 11%. However, the detection of any molecular changes (i.e., K-RAS mutation, LOH, or increased DNA quantity) has been found to be 90% specific for mucinous cysts[12]. Recently, one meta-analysis study found that the sensitivity and specificity of K-RAS was 0.39 and 0.95; and the sensitivity and specificity of the combined test of cytology and K-RAS was 0.71 and 0.88, respectively[47]. The K-RAS mutation combined with cytology test greatly increases the sensitivity of EUS-FNA. K-RAS mutation analysis may also prove to be a powerful ancillary for testing cystic samples with scant cellularity.
GNAS mutation: Another diagnostic marker that has received considerable interest is the presence of GNAS mutations. Recent studies have shown that GNAS mutations can be detected in IPMNs[69,70]. It has also been shown that the combination of GNAS and KRAS mutations in cyst fluid is very specific for IPMNs. One study found GNAS mutations to be significantly more prevalent in IPMNs (42%) than in SCAs (10%), adenocarcinomas (0%), and MCNs (0%). This same study also showed that double GNAS and KRAS mutations only occur in IPMNs (P = 0.006) and that mutations in either gene equated to a sensitivity of 98% and a specificity of 84%[71]. GNAS mutations are rare to absent in MCN, SCA, PNET, or PDAC.
MicroRNA change: MicroRNA (miRNA) expression profiles have also received considerable interest and are currently being studied as another way to characterize pancreatic lesions. miRNA is nineteen to twenty-four nucleotide long single-stranded, non-coding regions of RNA that are highly stable and which may be useful in diagnosing various malignancies as well as pancreatic cystic neoplasms. In a recent study, together with IPMN surgical specimens, 65 cyst fluid samples were examined for differential selective miRNA candidate expression. A subset of 18 miRNAs separated high-grade from low-grade lesions. A logistic regression model using nine miRNAs allowed prediction of high-grade IPMNs, PNET and SPN vs low-grade IPMNs and SCA with a sensitivity of 89%, a specificity of 100% and area under ROC curve of 1[72]. Another study evaluated miRNA in 69 pathology specimens and identified several miRNA panels that enabled them to differentiate SCA from MCN and IPMN, and MCN from BD-IPMN with a sensitivity ranging from 85%-100% and a specificity of 100%[73].
Integrated molecular pathology: Perhaps the greatest dilemma in managing pancreatic cysts is the fact that none of the currently recommended guidelines can accurately predict the malignant potential of pancreatic cysts. For example, the current IAP 2012 criteria risk stratifies patients into two categories: “surveillance” criteria (low malignant potential) and “surgery” criteria (high malignant potential). Symptomatic patients with mucinous cysts and at least one other “high-risk stigmata” (i.e., obstructive jaundice with a cyst located in the pancreatic head, a post-contrast enhancing solid component, a main pancreatic duct diameter ≥ 1 cm, abrupt change in duct caliber, cyst size ≥ 3 cm, presumptive diagnosis of MCN, and “suspicious” cytology) as detailed by the 2012 International Association of Pancreatology (IAP) guidelines should be referred for surgery[74]. Patients with cysts less than 1 cm and no concerning radiologic features can be monitored with periodic imaging studies. If more worrisome features are detected, the patients are subsequently referred for EUS-guided FNA to help determine the nature of the cyst (i.e., mucinous versus non-mucinous) and whether malignancy is present. Nevertheless, given the high mortality rate for pancreatic cancer, the IAP ultimately recommends that any patient with “worrisome” features associated with malignancy undergo surgery. However, it has been shown that approximately 60%-80% of patients undergoing surgery often have non-malignant disease. Therefore, other methods that prevent overtreatment of benign disease while providing early detection of cancer are needed.
Integrated molecular pathology (IMP) testing addresses this need it that incorporates all of the testing methods mentioned above (i.e., cytology, imaging studies, fluid chemistry, and molecular analysis). Unlike other guidelines, it utilizes four different diagnostic categories of “benign”, “statistically indolent”, “statistically higher risk”, or SHR, and “aggressive” based on both the number of molecular criterion met and other clinical features, if applicable (Table 2)[75]. In one study, 492 patients were categorized using IMP. Follow up for at least three years was available for 46% of patients. The overall accuracy was found to be 90%, and the specificity and negative predictive value were 91% and 97%, respectively. The sensitivity for malignant outcome with this cohort of patients was 83%, and the positive predictive value was 58%. When compared to the 2012 IAP criteria, it was found that the IAP criteria and IMP showed similar sensitivity and negative likelihood ratios. However, there was a statistically significant difference between the IAP guidelines and the IMP in that the specificity and positive likelihood ratios were higher using IMP criteria. These findings suggest that IMP is very useful in not only risk stratifying patients, but also in preventing patients with indolent disease from undergoing unnecessary surgeries[75].
| Diagnostic category | Molecular criteria1 | Co-existing concerning clinical features2 |
| Benign | DNA lacks molecular criteria | Not considered for this diagnosis |
| Statistically indolent | DNA meets 1 molecular criterion | None |
| SHR | DNA meets 1 molecular criterion | 1 or more |
| Aggressive | DNA meets at least 2 molecular criteria | Not considered for this diagnosis |
EUS-guided FNA serves a pivotal role in the accurate diagnosis of incidentally discovered pancreatic cysts. Its advantages over imaging alone include the ability to confirm the presence or absence of suspicious features identified on radiologic imaging, determine whether a lesion is malignant, and monitor for changes in cystic lesions. The new classification schema, while not perfect, goes hand-in-hand with the role of EUS-guided FNA in that it helps clinicians and patients to have a better understanding of which lesions need to be treated as opposed to those which do not, thus sparing patients from undergoing procedures that may result in increased morbidity and/or mortality. Despite these advantages, arriving at a proper diagnosis still requires the integrated use of clinical, radiologic, and cytological findings. Newer chemical and molecular studies show promise in improving the ability of clinicians to effectively diagnose and treat these lesions.
P- Reviewer: Swan MP S- Editor: Ji FF L- Editor: A E- Editor: Jiao XK
| 1. | Moparty B, Logroño R, Nealon WH, Waxman I, Raju GS, Pasricha PJ, Bhutani MS. The role of endoscopic ultrasound and endoscopic ultrasound-guided fine-needle aspiration in distinguishing pancreatic cystic lesions. Diagn Cytopathol. 2007;35:18-25. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 46] [Cited by in RCA: 47] [Article Influence: 2.6] [Reference Citation Analysis (0)] |
| 2. | Gardner TB, Glass LM, Smith KD, Ripple GH, Barth RJ, Klibansky DA, Colacchio TA, Tsapakos MJ, Suriawinata AA, Tsongalis GJ. Pancreatic cyst prevalence and the risk of mucin-producing adenocarcinoma in US adults. Am J Gastroenterol. 2013;108:1546-1550. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 95] [Cited by in RCA: 101] [Article Influence: 8.4] [Reference Citation Analysis (0)] |
| 3. | Laffan TA, Horton KM, Klein AP, Berlanstein B, Siegelman SS, Kawamoto S, Johnson PT, Fishman EK, Hruban RH. Prevalence of unsuspected pancreatic cysts on MDCT. AJR Am J Roentgenol. 2008;191:802-807. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 724] [Cited by in RCA: 658] [Article Influence: 38.7] [Reference Citation Analysis (0)] |
| 4. | Lee KS, Sekhar A, Rofsky NM, Pedrosa I. Prevalence of incidental pancreatic cysts in the adult population on MR imaging. Am J Gastroenterol. 2010;105:2079-2084. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 420] [Cited by in RCA: 436] [Article Influence: 29.1] [Reference Citation Analysis (0)] |
| 5. | de Jong K, Bruno MJ, Fockens P. Epidemiology, diagnosis, and management of cystic lesions of the pancreas. Gastroenterol Res Pract. 2012;2012:147465. [PubMed] |
| 6. | Chung JW, Chung MJ, Park JY, Bang S, Song SY, Chung JB, Park SW. Clinicopathologic features and outcomes of pancreatic cysts during a 12-year period. Pancreas. 2013;42:230-238. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 19] [Cited by in RCA: 23] [Article Influence: 1.9] [Reference Citation Analysis (0)] |
| 7. | Wu BU, Sampath K, Berberian CE, Kwok KK, Lim BS, Kao KT, Giap AQ, Kosco AE, Akmal YM, Difronzo AL. Prediction of malignancy in cystic neoplasms of the pancreas: a population-based cohort study. Am J Gastroenterol. 2014;109:121-129; quiz 130. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 61] [Cited by in RCA: 61] [Article Influence: 5.5] [Reference Citation Analysis (0)] |
| 8. | Brugge WR, Lauwers GY, Sahani D, Fernandez-del Castillo C, Warshaw AL. Cystic neoplasms of the pancreas. N Engl J Med. 2004;351:1218-1226. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 537] [Cited by in RCA: 490] [Article Influence: 23.3] [Reference Citation Analysis (0)] |
| 9. | Sahani DV, Kambadakone A, Macari M, Takahashi N, Chari S, Fernandez-del Castillo C. Diagnosis and management of cystic pancreatic lesions. AJR Am J Roentgenol. 2013;200:343-354. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 100] [Cited by in RCA: 92] [Article Influence: 7.7] [Reference Citation Analysis (0)] |
| 10. | Pelaez-Luna M, Chari ST, Smyrk TC, Takahashi N, Clain JE, Levy MJ, Pearson RK, Petersen BT, Topazian MD, Vege SS. Do consensus indications for resection in branch duct intraductal papillary mucinous neoplasm predict malignancy? A study of 147 patients. Am J Gastroenterol. 2007;102:1759-1764. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 219] [Cited by in RCA: 205] [Article Influence: 11.4] [Reference Citation Analysis (0)] |
| 11. | But DY, Poley JW. To fine needle aspiration or not? An endosonographer’s approach to pancreatic cystic lesions. Endosc Ultrasound. 2014;3:82-90. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 4] [Cited by in RCA: 9] [Article Influence: 0.8] [Reference Citation Analysis (0)] |
| 12. | Kadiyala V, Lee LS. Endosonography in the diagnosis and management of pancreatic cysts. World J Gastrointest Endosc. 2015;7:213-223. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in CrossRef: 30] [Cited by in RCA: 31] [Article Influence: 3.1] [Reference Citation Analysis (1)] |
| 13. | Waters JA, Schmidt CM, Pinchot JW, White PB, Cummings OW, Pitt HA, Sandrasegaran K, Akisik F, Howard TJ, Nakeeb A. CT vs MRCP: optimal classification of IPMN type and extent. J Gastrointest Surg. 2008;12:101-109. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 172] [Cited by in RCA: 158] [Article Influence: 9.3] [Reference Citation Analysis (0)] |
| 14. | Sainani NI, Saokar A, Deshpande V, Fernández-del Castillo C, Hahn P, Sahani DV. Comparative performance of MDCT and MRI with MR cholangiopancreatography in characterizing small pancreatic cysts. AJR Am J Roentgenol. 2009;193:722-731. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 123] [Cited by in RCA: 128] [Article Influence: 8.0] [Reference Citation Analysis (0)] |
| 15. | Visser BC, Yeh BM, Qayyum A, Way LW, McCulloch CE, Coakley FV. Characterization of cystic pancreatic masses: relative accuracy of CT and MRI. AJR Am J Roentgenol. 2007;189:648-656. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 125] [Cited by in RCA: 121] [Article Influence: 6.7] [Reference Citation Analysis (0)] |
| 16. | Kim YC, Choi JY, Chung YE, Bang S, Kim MJ, Park MS, Kim KW. Comparison of MRI and endoscopic ultrasound in the characterization of pancreatic cystic lesions. AJR Am J Roentgenol. 2010;195:947-952. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 61] [Cited by in RCA: 61] [Article Influence: 4.1] [Reference Citation Analysis (0)] |
| 17. | Al-Haddad M, Schmidt MC, Sandrasegaran K, Dewitt J. Diagnosis and treatment of cystic pancreatic tumors. Clin Gastroenterol Hepatol. 2011;9:635-648. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 46] [Cited by in RCA: 38] [Article Influence: 2.7] [Reference Citation Analysis (0)] |
| 18. | Leiman G. My approach to pancreatic fine needle aspiration. J Clin Pathol. 2007;60:43-49. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 14] [Cited by in RCA: 16] [Article Influence: 0.8] [Reference Citation Analysis (0)] |
| 19. | Jhala NC, Eltoum IA, Eloubeidi MA, Meara R, Chhieng DC, Crowe DR, Jhala D. Providing on-site diagnosis of malignancy on endoscopic-ultrasound-guided fine-needle aspirates: should it be done? Ann Diagn Pathol. 2007;11:176-181. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 34] [Cited by in RCA: 41] [Article Influence: 2.3] [Reference Citation Analysis (0)] |
| 20. | Matynia AP, Schmidt RL, Barraza G, Layfield LJ, Siddiqui AA, Adler DG. Impact of rapid on-site evaluation on the adequacy of endoscopic-ultrasound guided fine-needle aspiration of solid pancreatic lesions: a systematic review and meta-analysis. J Gastroenterol Hepatol. 2014;29:697-705. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 67] [Cited by in RCA: 97] [Article Influence: 8.8] [Reference Citation Analysis (0)] |
| 21. | Layfield LJ, Esebua M, Witt BL. Cytotechnologist screening of fine-needle aspiration specimens: impact on turnaround time and diagnostic accuracy. Diagn Cytopathol. 2014;42:606-608. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 1] [Cited by in RCA: 2] [Article Influence: 0.2] [Reference Citation Analysis (0)] |
| 22. | Brugge WR. Advances in the endoscopic management of patients with pancreatic and biliary malignancies. South Med J. 2006;99:1358-1366. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 13] [Cited by in RCA: 10] [Article Influence: 0.6] [Reference Citation Analysis (0)] |
| 23. | Wang KX, Ben QW, Jin ZD, Du YQ, Zou DW, Liao Z, Li ZS. Assessment of morbidity and mortality associated with EUS-guided FNA: a systematic review. Gastrointest Endosc. 2011;73:283-290. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 329] [Cited by in RCA: 293] [Article Influence: 20.9] [Reference Citation Analysis (0)] |
| 24. | Tarantino I, Fabbri C, Di Mitri R, Pagano N, Barresi L, Mocciaro F, Maimone A, Curcio G, Repici A, Traina M. Complications of endoscopic ultrasound fine needle aspiration on pancreatic cystic lesions: final results from a large prospective multicenter study. Dig Liver Dis. 2014;46:41-44. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 34] [Cited by in RCA: 42] [Article Influence: 3.8] [Reference Citation Analysis (0)] |
| 25. | Virgilio E, Mercantini P, Ferri M, Cunsolo G, Tarantino G, Cavallini M, Ziparo V. Is EUS-FNA of solid-pseudopapillary neoplasms of the pancreas as a preoperative procedure really necessary and free of acceptable risks? Pancreatology. 2014;14:536-538. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 16] [Cited by in RCA: 18] [Article Influence: 1.6] [Reference Citation Analysis (0)] |
| 26. | Yoon WJ, Daglilar ES, Fernández-del Castillo C, Mino-Kenudson M, Pitman MB, Brugge WR. Peritoneal seeding in intraductal papillary mucinous neoplasm of the pancreas patients who underwent endoscopic ultrasound-guided fine-needle aspiration: the PIPE Study. Endoscopy. 2014;46:382-387. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 66] [Cited by in RCA: 71] [Article Influence: 6.5] [Reference Citation Analysis (0)] |
| 27. | Katanuma A, Maguchi H, Hashigo S, Kaneko M, Kin T, Yane K, Kato R, Kato S, Harada R, Osanai M. Tumor seeding after endoscopic ultrasound-guided fine-needle aspiration of cancer in the body of the pancreas. Endoscopy. 2012;44 Suppl 2 UCTN:E160-E161. [PubMed] |
| 28. | de Jong K, Poley JW, van Hooft JE, Visser M, Bruno MJ, Fockens P. Endoscopic ultrasound-guided fine-needle aspiration of pancreatic cystic lesions provides inadequate material for cytology and laboratory analysis: initial results from a prospective study. Endoscopy. 2011;43:585-590. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 94] [Cited by in RCA: 99] [Article Influence: 7.1] [Reference Citation Analysis (0)] |
| 29. | Pitman MB, Deshpande V. Endoscopic ultrasound-guided fine needle aspiration cytology of the pancreas: a morphological and multimodal approach to the diagnosis of solid and cystic mass lesions. Cytopathology. 2007;18:331-347. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 75] [Cited by in RCA: 70] [Article Influence: 3.9] [Reference Citation Analysis (0)] |
| 30. | Hong SK, Loren DE, Rogart JN, Siddiqui AA, Sendecki JA, Bibbo M, Coben RM, Meckes DP, Kowalski TE. Targeted cyst wall puncture and aspiration during EUS-FNA increases the diagnostic yield of premalignant and malignant pancreatic cysts. Gastrointest Endosc. 2012;75:775-782. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 78] [Cited by in RCA: 71] [Article Influence: 5.5] [Reference Citation Analysis (0)] |
| 31. | Rogart JN, Loren DE, Singu BS, Kowalski TE. Cyst wall puncture and aspiration during EUS-guided fine needle aspiration may increase the diagnostic yield of mucinous cysts of the pancreas. J Clin Gastroenterol. 2011;45:164-169. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 44] [Cited by in RCA: 48] [Article Influence: 3.4] [Reference Citation Analysis (0)] |
| 32. | Kocjan G. Fine needle aspiration cytology of the pancreas: a guide to the diagnostic approach. Coll Antropol. 2010;34:749-756. [PubMed] |
| 33. | Gleeson FC, Kipp BR, Caudill JL, Clain JE, Clayton AC, Halling KC, Henry MR, Rajan E, Topazian MD, Wang KK. False positive endoscopic ultrasound fine needle aspiration cytology: incidence and risk factors. Gut. 2010;59:586-593. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 68] [Cited by in RCA: 68] [Article Influence: 4.5] [Reference Citation Analysis (0)] |
| 34. | Pitman MB, Centeno BA, Ali SZ, Genevay M, Stelow E, Mino-Kenudson M, Castillo CF, Schmidt CM, Brugge WR, Layfield LJ. Standardized terminology and nomenclature for pancreatobiliary cytology: The Papanicolaou Society of Cytopathology Guidelines. Cytojournal. 2014;11:3. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 33] [Cited by in RCA: 57] [Article Influence: 5.2] [Reference Citation Analysis (0)] |
| 35. | Coe AW, Evans J, Conway J. Pancreas cystic lymphangioma diagnosed with EUS-FNA. JOP. 2012;13:282-284. [PubMed] |
| 36. | Martin J, Roberts KJ, Sheridan M, Falk GA, Joyce D, Walsh RM, Smith AM, Morris-Stiff G. Lymphoepithelial cysts of the pancreas: a management dilemma. Hepatobiliary Pancreat Dis Int. 2014;13:539-544. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 5] [Cited by in RCA: 6] [Article Influence: 0.5] [Reference Citation Analysis (0)] |
| 37. | Morales-Oyarvide V, Yoon WJ, Ingkakul T, Forcione DG, Casey BW, Brugge WR, Fernández-del Castillo C, Pitman MB. Cystic pancreatic neuroendocrine tumors: the value of cytology in preoperative diagnosis. Cancer Cytopathol. 2014;122:435-444. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 50] [Cited by in RCA: 42] [Article Influence: 3.8] [Reference Citation Analysis (0)] |
| 38. | Burns WR, Edil BH. Neuroendocrine pancreatic tumors: guidelines for management and update. Curr Treat Options Oncol. 2012;13:24-34. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 72] [Cited by in RCA: 70] [Article Influence: 5.4] [Reference Citation Analysis (0)] |
| 39. | Frossard JL, Amouyal P, Amouyal G, Palazzo L, Amaris J, Soldan M, Giostra E, Spahr L, Hadengue A, Fabre M. Performance of endosonography-guided fine needle aspiration and biopsy in the diagnosis of pancreatic cystic lesions. Am J Gastroenterol. 2003;98:1516-1524. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 268] [Cited by in RCA: 240] [Article Influence: 10.9] [Reference Citation Analysis (0)] |
| 40. | Pitman MB, Yaeger KA, Brugge WR, Mino-Kenudson M. Prospective analysis of atypical epithelial cells as a high-risk cytologic feature for malignancy in pancreatic cysts. Cancer Cytopathol. 2013;121:29-36. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 30] [Cited by in RCA: 35] [Article Influence: 2.7] [Reference Citation Analysis (0)] |
| 41. | Chebib I, Yaeger K, Mino-Kenudson M, Pitman MB. The role of cytopathology and cyst fluid analysis in the preoperative diagnosis and management of pancreatic cysts &gt; 3 cm. Cancer Cytopathol. 2014;122:804-809. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 11] [Cited by in RCA: 15] [Article Influence: 1.4] [Reference Citation Analysis (0)] |
| 42. | Pitman MB, Genevay M, Yaeger K, Chebib I, Turner BG, Mino-Kenudson M, Brugge WR. High-grade atypical epithelial cells in pancreatic mucinous cysts are a more accurate predictor of malignancy than “positive” cytology. Cancer Cytopathol. 2010;118:434-440. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 96] [Cited by in RCA: 88] [Article Influence: 5.9] [Reference Citation Analysis (0)] |
| 43. | Belsley NA, Pitman MB, Lauwers GY, Brugge WR, Deshpande V. Serous cystadenoma of the pancreas: limitations and pitfalls of endoscopic ultrasound-guided fine-needle aspiration biopsy. Cancer. 2008;114:102-110. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 126] [Cited by in RCA: 101] [Article Influence: 5.9] [Reference Citation Analysis (0)] |
| 44. | Moparty B, Brugge WR. Approach to pancreatic cystic lesions. Curr Gastroenterol Rep. 2007;9:130-135. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 9] [Cited by in RCA: 11] [Article Influence: 0.6] [Reference Citation Analysis (0)] |
| 45. | Thosani N, Thosani S, Qiao W, Fleming JB, Bhutani MS, Guha S. Role of EUS-FNA-based cytology in the diagnosis of mucinous pancreatic cystic lesions: a systematic review and meta-analysis. Dig Dis Sci. 2010;55:2756-2766. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 117] [Cited by in RCA: 109] [Article Influence: 7.3] [Reference Citation Analysis (0)] |
| 46. | Woolf KM, Liang H, Sletten ZJ, Russell DK, Bonfiglio TA, Zhou Z. False-negative rate of endoscopic ultrasound-guided fine-needle aspiration for pancreatic solid and cystic lesions with matched surgical resections as the gold standard: one institution’s experience. Cancer Cytopathol. 2013;121:449-458. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 37] [Cited by in RCA: 40] [Article Influence: 3.3] [Reference Citation Analysis (0)] |
| 47. | Gillis A, Cipollone I, Cousins G, Conlon K. Does EUS-FNA molecular analysis carry additional value when compared to cytology in the diagnosis of pancreatic cystic neoplasm? A systematic review. HPB (Oxford). 2015;17:377-386. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 48] [Cited by in RCA: 50] [Article Influence: 5.0] [Reference Citation Analysis (0)] |
| 48. | Genevay M, Mino-Kenudson M, Yaeger K, Konstantinidis IT, Ferrone CR, Thayer S, Castillo CF, Sahani D, Bounds B, Forcione D. Cytology adds value to imaging studies for risk assessment of malignancy in pancreatic mucinous cysts. Ann Surg. 2011;254:977-983. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 115] [Cited by in RCA: 104] [Article Influence: 8.0] [Reference Citation Analysis (0)] |
| 49. | Lim LG, Lakhtakia S, Ang TL, Vu CK, Dy F, Chong VH, Khor CJ, Lim WC, Doshi BK, Varadarajulu S. Factors determining diagnostic yield of endoscopic ultrasound guided fine-needle aspiration for pancreatic cystic lesions: a multicentre Asian study. Dig Dis Sci. 2013;58:1751-1757. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 41] [Cited by in RCA: 46] [Article Influence: 3.8] [Reference Citation Analysis (0)] |
| 50. | Wu RI, Yoon WJ, Brugge WR, Mino-Kenudson M, Pitman MB. Endoscopic ultrasound-guided fine needle aspiration (EUS-FNA) contributes to a triple-negative test in preoperative screening of pancreatic cysts. Cancer Cytopathol. 2014;122:412-419. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 12] [Cited by in RCA: 14] [Article Influence: 1.2] [Reference Citation Analysis (0)] |
| 51. | Ridtitid W, Halawi H, DeWitt JM, Sherman S, LeBlanc J, McHenry L, Coté GA, Al-Haddad MA. Cystic pancreatic neuroendocrine tumors: outcomes of preoperative endosonography-guided fine needle aspiration, and recurrence during long-term follow-up. Endoscopy. 2015;47:617-625. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 21] [Cited by in RCA: 23] [Article Influence: 2.3] [Reference Citation Analysis (0)] |
| 52. | Caglià P, Cannizzaro MT, Tracia A, Amodeo L, Tracia L, Buffone A, Amodeo C, Cannizzaro MA. Cystic pancreatic neuroendocrine tumors: To date a diagnostic challenge. Int J Surg. 2015;Epub ahead of prin. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 17] [Cited by in RCA: 27] [Article Influence: 2.7] [Reference Citation Analysis (0)] |
| 53. | Humphris J, Swartz D, Egan BJ, Leong RW. Status of confocal laser endomicroscopy in gastrointestinal disease. Trop Gastroenterol. 2012;33:9-20. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 16] [Cited by in RCA: 20] [Article Influence: 1.5] [Reference Citation Analysis (0)] |
| 54. | Konda VJ, Aslanian HR, Wallace MB, Siddiqui UD, Hart J, Waxman I. First assessment of needle-based confocal laser endomicroscopy during EUS-FNA procedures of the pancreas (with videos). Gastrointest Endosc. 2011;74:1049-1060. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 111] [Cited by in RCA: 115] [Article Influence: 8.2] [Reference Citation Analysis (0)] |
| 55. | Samarasena JB, Nakai Y, Chang KJ. Endoscopic ultrasonography-guided fine-needle aspiration of pancreatic cystic lesions: a practical approach to diagnosis and management. Gastrointest Endosc Clin N Am. 2012;22:169-185, vii. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 14] [Cited by in RCA: 17] [Article Influence: 1.3] [Reference Citation Analysis (0)] |
| 56. | Konda VJ, Meining A, Jamil LH, Giovannini M, Hwang JH, Wallace MB, Chang KJ, Siddiqui UD, Hart J, Lo SK. A pilot study of in vivo identification of pancreatic cystic neoplasms with needle-based confocal laser endomicroscopy under endosonographic guidance. Endoscopy. 2013;45:1006-1013. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 152] [Cited by in RCA: 143] [Article Influence: 11.9] [Reference Citation Analysis (0)] |
| 57. | Lewandrowski KB, Southern JF, Pins MR, Compton CC, Warshaw AL. Cyst fluid analysis in the differential diagnosis of pancreatic cysts. A comparison of pseudocysts, serous cystadenomas, mucinous cystic neoplasms, and mucinous cystadenocarcinoma. Ann Surg. 1993;217:41-47. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 216] [Cited by in RCA: 189] [Article Influence: 5.9] [Reference Citation Analysis (0)] |
| 58. | van der Waaij LA, van Dullemen HM, Porte RJ. Cyst fluid analysis in the differential diagnosis of pancreatic cystic lesions: a pooled analysis. Gastrointest Endosc. 2005;62:383-389. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 456] [Cited by in RCA: 383] [Article Influence: 19.2] [Reference Citation Analysis (0)] |
| 59. | Hammel P, Levy P, Voitot H, Levy M, Vilgrain V, Zins M, Flejou JF, Molas G, Ruszniewski P, Bernades P. Preoperative cyst fluid analysis is useful for the differential diagnosis of cystic lesions of the pancreas. Gastroenterology. 1995;108:1230-1235. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 200] [Cited by in RCA: 166] [Article Influence: 5.5] [Reference Citation Analysis (0)] |
| 61. | Nagashio Y, Hijioka S, Mizuno N, Hara K, Imaoka H, Bhatia V, Niwa Y, Tajika M, Tanaka T, Ishihara M. Combination of cyst fluid CEA and CA 125 is an accurate diagnostic tool for differentiating mucinous cystic neoplasms from intraductal papillary mucinous neoplasms. Pancreatology. 2014;14:503-509. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 17] [Cited by in RCA: 26] [Article Influence: 2.4] [Reference Citation Analysis (0)] |
| 62. | Cizginer S, Turner BG, Bilge AR, Karaca C, Pitman MB, Brugge WR. Cyst fluid carcinoembryonic antigen is an accurate diagnostic marker of pancreatic mucinous cysts. Pancreas. 2011;40:1024-1028. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 220] [Cited by in RCA: 210] [Article Influence: 15.0] [Reference Citation Analysis (0)] |
| 63. | Ngamruengphong S, Bartel MJ, Raimondo M. Cyst carcinoembryonic antigen in differentiating pancreatic cysts: a meta-analysis. Dig Liver Dis. 2013;45:920-926. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 48] [Cited by in RCA: 45] [Article Influence: 3.8] [Reference Citation Analysis (0)] |
| 64. | Park WG, Mascarenhas R, Palaez-Luna M, Smyrk TC, O’Kane D, Clain JE, Levy MJ, Pearson RK, Petersen BT, Topazian MD. Diagnostic performance of cyst fluid carcinoembryonic antigen and amylase in histologically confirmed pancreatic cysts. Pancreas. 2011;40:42-45. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 137] [Cited by in RCA: 112] [Article Influence: 8.0] [Reference Citation Analysis (0)] |
| 65. | Bausch D, Mino-Kenudson M, Fernández-Del Castillo C, Warshaw AL, Kelly KA, Thayer SP. Plectin-1 is a biomarker of malignant pancreatic intraductal papillary mucinous neoplasms. J Gastrointest Surg. 2009;13:1948-1954; discussion 1954. [PubMed] |
| 66. | Li H, Li Y, Cui L, Wang B, Cui W, Li M, Cheng Y. Monitoring pancreatic carcinogenesis by the molecular imaging of cathepsin E in vivo using confocal laser endomicroscopy. PLoS One. 2014;9:e106566. [PubMed] |
| 67. | Nikiforova MN, Khalid A, Fasanella KE, McGrath KM, Brand RE, Chennat JS, Slivka A, Zeh HJ, Zureikat AH, Krasinskas AM. Integration of KRAS testing in the diagnosis of pancreatic cystic lesions: a clinical experience of 618 pancreatic cysts. Mod Pathol. 2013;26:1478-1487. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 116] [Cited by in RCA: 109] [Article Influence: 9.1] [Reference Citation Analysis (0)] |
| 68. | Al-Haddad M, DeWitt J, Sherman S, Schmidt CM, LeBlanc JK, McHenry L, Coté G, El Chafic AH, Luz L, Stuart JS. Performance characteristics of molecular (DNA) analysis for the diagnosis of mucinous pancreatic cysts. Gastrointest Endosc. 2014;79:79-87. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 80] [Cited by in RCA: 81] [Article Influence: 7.4] [Reference Citation Analysis (0)] |
| 69. | Kanda M, Knight S, Topazian M, Syngal S, Farrell J, Lee J, Kamel I, Lennon AM, Borges M, Young A. Mutant GNAS detected in duodenal collections of secretin-stimulated pancreatic juice indicates the presence or emergence of pancreatic cysts. Gut. 2013;62:1024-1033. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 129] [Cited by in RCA: 125] [Article Influence: 10.4] [Reference Citation Analysis (0)] |
| 70. | Amato E, Molin MD, Mafficini A, Yu J, Malleo G, Rusev B, Fassan M, Antonello D, Sadakari Y, Castelli P. Targeted next-generation sequencing of cancer genes dissects the molecular profiles of intraductal papillary neoplasms of the pancreas. J Pathol. 2014;233:217-227. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 298] [Cited by in RCA: 249] [Article Influence: 22.6] [Reference Citation Analysis (0)] |
| 71. | Singhi AD, Nikiforova MN, Fasanella KE, McGrath KM, Pai RK, Ohori NP, Bartholow TL, Brand RE, Chennat JS, Lu X. Preoperative GNAS and KRAS testing in the diagnosis of pancreatic mucinous cysts. Clin Cancer Res. 2014;20:4381-4389. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 126] [Cited by in RCA: 148] [Article Influence: 13.5] [Reference Citation Analysis (0)] |
| 72. | Matthaei H, Wylie D, Lloyd MB, Dal Molin M, Kemppainen J, Mayo SC, Wolfgang CL, Schulick RD, Langfield L, Andruss BF. miRNA biomarkers in cyst fluid augment the diagnosis and management of pancreatic cysts. Clin Cancer Res. 2012;18:4713-4724. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 114] [Cited by in RCA: 129] [Article Influence: 9.9] [Reference Citation Analysis (0)] |
| 73. | Lee LS, Szafranska-Schwarzbach AE, Wylie D, Doyle LA, Bellizzi AM, Kadiyala V, Suleiman S, Banks PA, Andruss BF, Conwell DL. Investigating MicroRNA Expression Profiles in Pancreatic Cystic Neoplasms. Clin Transl Gastroenterol. 2014;5:e47. [PubMed] |
| 74. | Tanaka M, Fernández-del Castillo C, Adsay V, Chari S, Falconi M, Jang JY, Kimura W, Levy P, Pitman MB, Schmidt CM. International consensus guidelines 2012 for the management of IPMN and MCN of the pancreas. Pancreatology. 2012;12:183-197. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 1714] [Cited by in RCA: 1614] [Article Influence: 124.2] [Reference Citation Analysis (0)] |
| 75. | Al-Haddad MA, Kowalski T, Siddiqui A, Mertz HR, Mallat D, Haddad N, Malhotra N, Sadowski B, Lybik MJ, Patel SN. Integrated molecular pathology accurately determines the malignant potential of pancreatic cysts. Endoscopy. 2015;47:136-142. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 10] [Cited by in RCA: 23] [Article Influence: 2.3] [Reference Citation Analysis (0)] |