INTRODUCTION
Leiomyosarcomas are rare soft tissue tumors that arise from smooth muscle cells[1]. They account for about 5%-7% of all soft tissue sarcomas[2] and are frequently found in retroperitoneal tissue, the gastrointestinal tract, the urinary tract, and the uterus[3,4]. Several histological subtypes have been identified. Pleomorphic leiomyosarcomas make up around 8.6% of all leiomyosarcomas[5]. These tumors behave aggressively and are often associated with poor patient prognoses[5]. Pleomorphic leiomyosarcomas can be identified by the presence of cells with heterogeneous morphology, including smooth muscle, spindle, and distinctly atypical pleomorphic cells[5,6]. Diagnosis of such tumors is difficult because their clinical presentation is dependent on the organ affected. Histopathological analysis of tissue specimens is the only conclusive method of diagnosis[7]. Immunohistochemistry is required to distinguish leiomyosarcoma from other mesenchymal tumors and to determine histological subtypes[7]. The present report describes a 44-year-old man with a history of pleomorphic leiomyosarcoma of the left maxilla. His most recent positron emission tomography-computed tomography (PET-CT) scan showed uptake in the ascending and transverse colons. A colonoscopy identified a 5.0 cm × 3.5 cm × 3.0 cm pedunculated polyp in the ascending colon. Histopathologic examination of the polyp following its retrieval showed a metastatic pleomorphic leiomyosarcoma.
CASE PRESENTATION
Chief complaints
A 44-year-old man who was not current taking medications but was a chronic smoker presented at our center with painful and firm left-sided infraorbital swelling.
History of present illness
The infraorbital swelling was first observed four months earlier, had progressively increased in size, and was associated with severe pain and numbness. The patient thought that the pain and swelling were tooth related, but tooth extraction provided minimal relief. His vision and sense of smell were intact, and there was no evidence of nasal obstruction, discharge or bleeding. He sought advice at his local hospital for persistent symptoms. An incisional biopsy of the mass at his local hospital revealed pleomorphic leiomyosarcoma. The patient did not have any pertinent gastrointestinal symptoms, such as abdominal pain, alterations in bowel habits, or per rectal bleeding.
History of past illness
The patient did not have any relevant past medical or surgical history.
Personal and family history
The patient did not have any relevant family history.
Physical examination
Physical examination showed left-sided infraorbital swelling measuring 2 cm × 2 cm. There was no evidence of intraoral extension.
Laboratory examinations
Laboratory workup was unremarkable.
Imaging examinations
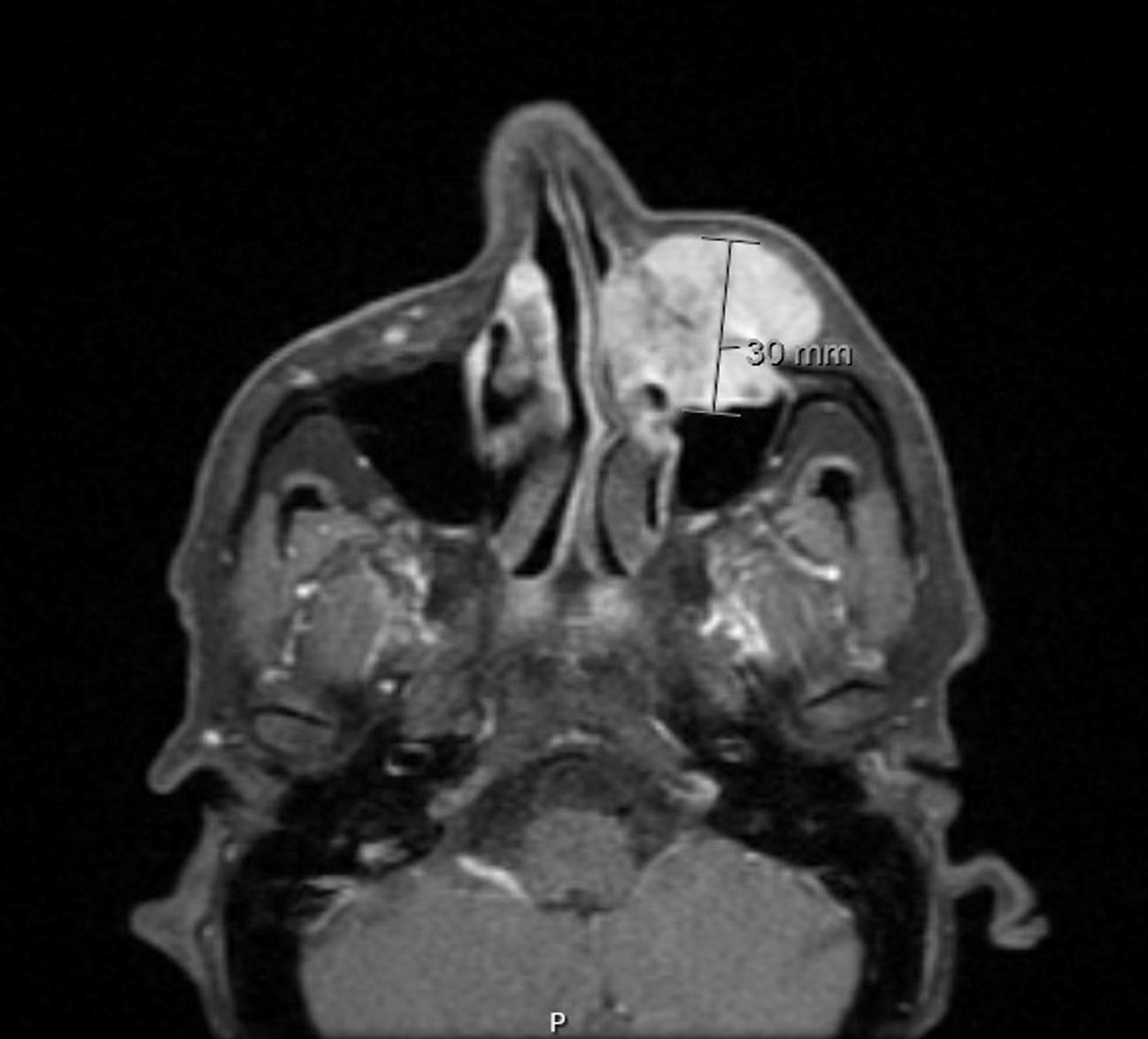
Initial magnetic resonance imaging of the face and neck showed a left maxillary mass, measuring 23 mm × 29 mm × 30 mm in the anteroposterior, mediolateral, and craniocaudal dimensions, respectively (Figure 1). Postcontrast sequences showed intense homogeneous enhancement and invasion of the left maxillary sinus through the anterior bony wall, resulting in bone destruction, and subsequent obstruction of the left nasal cavity. PET-CT scan after neoadjuvant radiotherapy, surgical excision, and three cycles of gemictabine chemotherapy showed a mild fludeoxyglucose avid, ill-defined, semisolid nodule in the right upper lobe, measuring 1.2 cm, and focal moderate to intense activity at the lateral aspect of the left clavicle. The patient was administered six cycles of gemcitabine and docetaxel chemotherapy. The latest follow-up PET-CT scan in May 2023 showed focal intense FDG uptake in the ascending colon and transverse colon, as well as liver metastasis. He was diagnosed with a pleomorphic leiomyosarcoma of the left maxilla with metastasis to the colon.
Figure 1 Magnetic resonance imaging of the face and neck showing a left maxillary mass.
FINAL DIAGNOSIS
Pleomorphic leiomyosarcoma of the left maxilla with metastasis to the colon.
TREATMENT
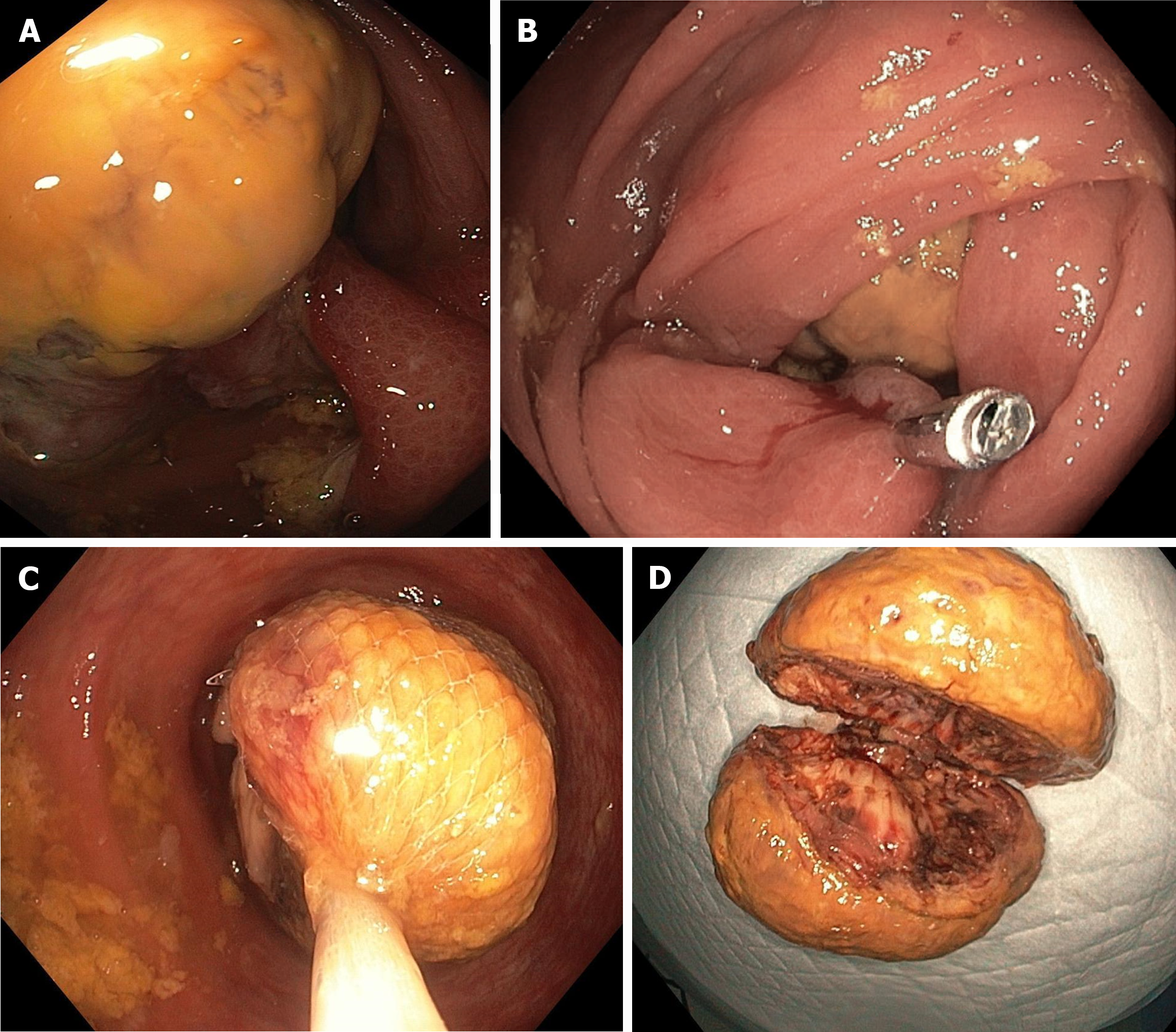
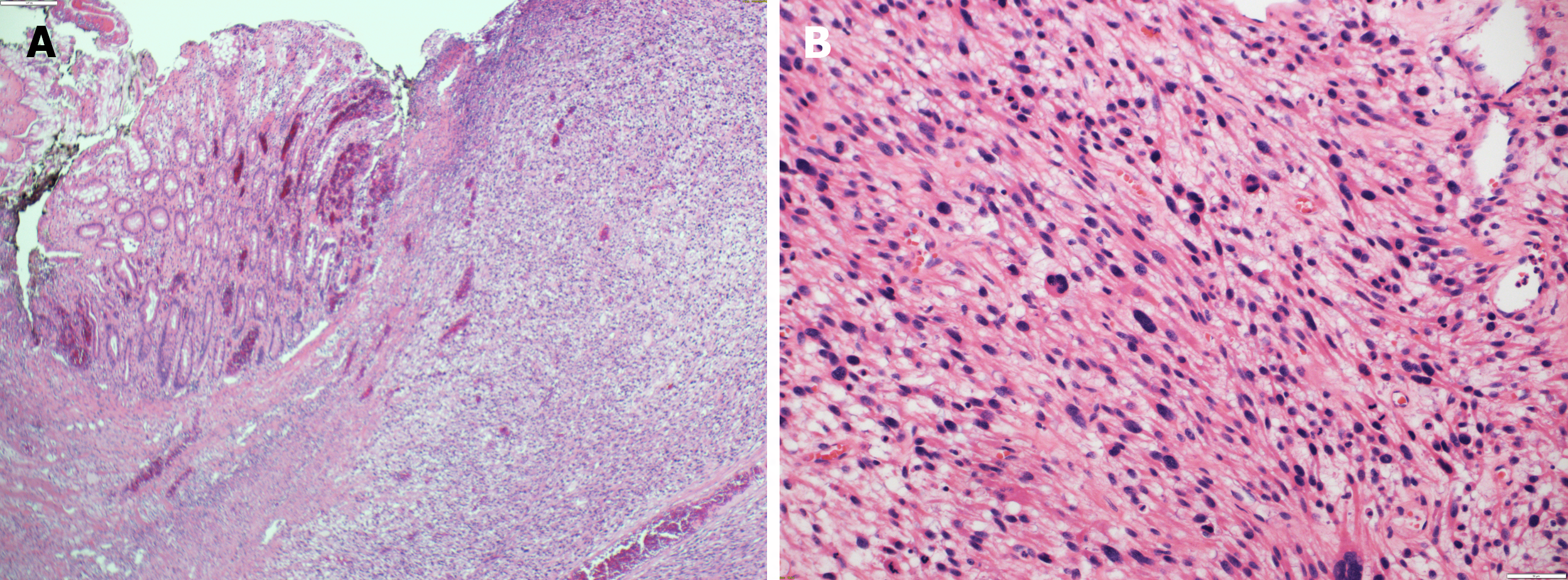
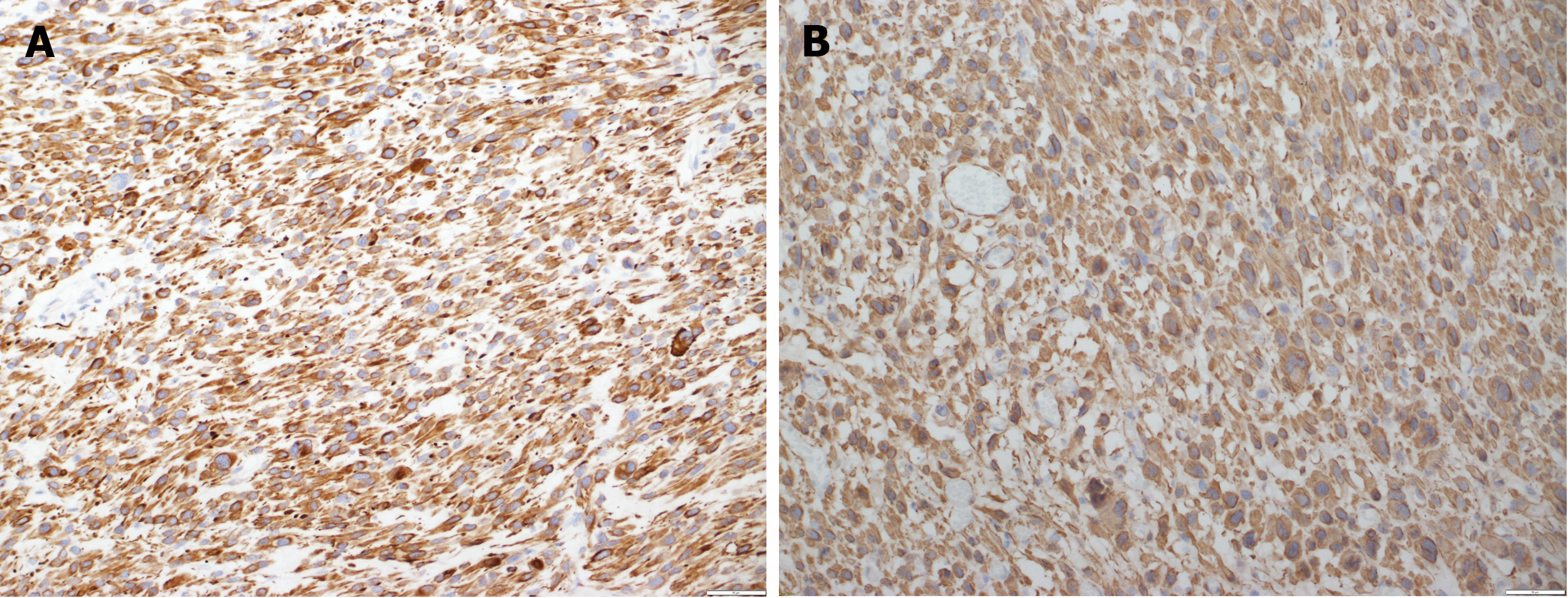
The patient was administered neoadjuvant radiation, followed by a left anterior maxillectomy. Pathologic examination confirmed that the tumor was a pleomorphic leiomyosarcoma of infraorbital soft tissue. Intraneural invasion was observed, but there was no evidence of lymphovascular invasion. The patient completed three cycles of doxorubicin chemotherapy. A follow-up PET-CT scan showed a mild FDG-avid, ill-defined semisolid nodule in the right upper lobe, measuring 1.2 cm, and focal moderate to intense activity at the lateral aspect of the left clavicle. Six cycles of gemcitabine and docetaxel chemotherapy were administered. The latest follow-up PET-CT scan in May 2023 showed focal intense FDG uptake in the ascending colon and transverse colon, as well as liver metastasis. The patient denied any pertinent symptoms, such as abdominal pain, alternating bowel habits, and PR bleeding. Colonoscopy showed a sizeable pedunculated polyp with a thick stalk in the proximal ascending colon, partially obstructing the lumen on the way to the cecum (Figure 2A). Endoscopic examination showed that the polyp was about 5 cm in size. Although polypectomy using an EndoLoop 30 mm was attempted, it was unsuccessful due to the large size of the polyp. Therefore, a hemoclip measuring 20 mm was applied to the stalk (Figure 2B). The polyp was removed by hot snare polypectomy, with two rounds required owing to the large size and thickness of the polyp. The first half was retrieved using a Roth net (Figure 2C). The second half was initially retrieved using a Roth net, with a scope and suctioning used when it reached the rectum. Direct examination showed that the polyp was 5.0 cm × 3.5 cm × 3.0 cm in size (Figure 2D). Pathological examination revealed a pleomorphic spindle cell malignancy with a high mitotic rate (Figure 3). Immunohistochemical examination showed that the tumor cells were positive for desmin and smooth muscle actin (Figure 4), but negative for markers of gastrointestinal stromal tumor (GIST), such as DOG-1 and CD 117.
Figure 2 Colonoscopy.
A: Ascending colon polyp; B: Hemoclip applied to ascending colon polyp; C: Roth net covering ascending colon polyp; D: Retrieved ascending colon polyp.
Figure 3 Histopathological examination.
A: Submucosal diffuse spindle cell neoplasm [hematoxylin and eosin (H&E) × 10]; B: Higher magnification of the spindle cell neoplasm showed high mitotic figures and prominent nuclear pleomorphism (H&E × 20).
Figure 4 Immunohistochemistry.
A: The malignant cells are positive with desmin [immunohistochemistry (IHC) × 20]; B: The malignant cells are positive with SMA (IHC × 20).
OUTCOME AND FOLLOW-UP
The patient was followed-up in the clinic afterward. He did not report any gastrointestinal-related symptoms, but mostly complained of left clavicular and shoulder pain. He was treated with six cycles of dacarbazine chemotherapy, but the most recent CT scan revealed disease progression, as evidenced by increases in sizes of pulmonary nodules and liver lesions. He was then shifted to pazopanib.
DISCUSSION
Pleomorphic leiomyosarcoma can involve the retroperitoneum; the gastrointestinal tract, including the liver and pancreas; the adrenal glands; the mesenteric vessels; the skin; the heart; and the intrascrotum area[7-16]. Clinical manifestations have included abdominal discomfort, lower gastrointestinal bleeding, cardiac tamponade, back pain, and enlargement of the testicles or scrotum[7-15]. Oral cavity leiomyosarcomas are rare, with about eighty cases described in the literature[17]. More than half of these tumors involved the mandible[18]. Unlike leiomyosarcomas of other organs, intraoral leiomyosarcomas exhibit nodal metastasis, particularly in the cervical lymph nodes[19]. They also metastasize to the lungs and liver[19]. The present patient had metastases not only to the lungs and liver but also to the colon. To our knowledge, there have been no reports of other patients with pleomorphic leiomyosarcoma of the maxilla who showed metastasis to the colon. To date, only nine patents with pleomorphic leiomyosarcoma involving the mesocolon have been described[20], with the most common sites of involvement being the sigmoid and descending mesocolons. Mass, followed by pain, were the most common complaints. One case study described a patient who presented with lower GI bleeding secondary to mesenteric pleomorphic leiomyosarcoma that was inseparable from the descending colon[8]. The present patient presented with luminal involvement of the ascending colon with pleomorphic leiomyosarcoma, as evidenced by imaging and histopathologic examination of the polyp. Pleomorphic leiomyosarcomas are diagnosed through histological and immunohistochemical examination[7]. Typical histological features include smooth muscle cells, neoplastic spindle cells, and distinctly atypical pleomorphic cells[5,6]. Immunohistochemical staining has shown that most cells are positive for smooth muscle actin (90%-95%) and desmin (70%-90%)[21], with some being positive for caldesmon[5]. In contrast, pleomorphic leiomyosarcomas are negative for the GIST markers c-kit, CD117 and CD34, allowing differentiation of these two tumor types[6,8,22]. Although surgical resection with complete resection margins is the mainstay of management of pleomorphic leiomyosarcomas[8], local recurrence has been observed in 44%-85% of patients[23,24]. These findings point to the need for a comprehensive treatment plan, including chemotherapy and radiation. Neoadjuvant chemotherapy can reduce tumor size before surgical resection[25]. Although several randomized clinical trials have shown that adjuvant chemotherapy can minimize metastasis in patients with pleomorphic leiomyosarcomas, but further research is required to assess its survival benefit[26]. Radiotherapy is effective in treating pleomorphic leiomyosarcomas of the trunk and extremities[27]. Despite treatment, however, recurrence and metastasis are still common, with Metastatic pleomorphic leiomyosarcoma being associated with poor survival[8].
CONCLUSION
In summary, pleomorphic leiomyosarcoma is a rare and aggressive form of leiomyosarcoma that can affect any organ. Although few such patients have been described to date, their prognosis remains poor. Further studies are needed to understand this malignancy and explore newer treatment options.
Provenance and peer review: Unsolicited article; Externally peer reviewed.
Peer-review model: Single blind
Specialty type: Gastroenterology and hepatology
Country of origin: Saudi Arabia
Peer-review report’s classification
Scientific Quality: Grade C
Novelty: Grade C
Creativity or Innovation: Grade C
Scientific Significance: Grade C
P-Reviewer: Pattarajierapan S, Thailand S-Editor: Chen YL L-Editor: A P-Editor: Cai YX