Published online Dec 27, 2021. doi: 10.4254/wjh.v13.i12.2013
Peer-review started: May 18, 2021
First decision: June 15, 2021
Revised: June 23, 2021
Accepted: November 27, 2021
Article in press: October 27, 2021
Published online: December 27, 2021
Processing time: 222 Days and 9.7 Hours
Coronavirus disease 2019 has a wide range of clinical spectrum from asymptomatic infection to severe infection resulting in death within a short time. Currently, it is known that severe acute respiratory syndrome coronavirus 2 (SARS-CoV-2) does not only cause a respiratory tract infection but a more complicated disease that can lead to multiple system involvement including the liver. Herein, we evaluate the epidemiology, the impact of liver injury/ dysfunction on disease prognosis, the pathophysiological mechanisms and management of liver injury. More than one-fourth of the patients have abnormal liver function tests, mostly a mild-to-moderate liver dysfunction. Liver injury is significantly associated with a poor clinical outcome. Direct cytotoxic effect of SARS-CoV-2, the immune response (“cytokine storm”), the complications related to the disease, and drugs used in the treatments are the pathophysiological mechanisms responsible for liver injury. However, the exact mechanism is not yet clearly explained. The binding of SARS-CoV-2 to the angiotensin-converting enzyme 2 receptors and entering the hepatocyte and cholangiocytes can cause cytotoxic effects on the liver. Excessive immune response has an important role in disease progression and causes acute respiratory distress syndrome and multi-organ failures accompanied by liver injury. Treatment drugs, particularly lopinavir/ritonavir, remdesivir and antibiotics are a frequent reason for liver injury. The possible reasons should be meticulously investigated and resolved.
Core Tip: The study evaluated the incidence of liver injury in coronavirus disease 2019 (COVID-19) patients and its impact on clinical outcomes and pathophysiological mechanism of liver injury. More than one-fourth of COVID-19 patients had suffered from liver injury, mostly a mild-to-moderate liver dysfunction. Liver involvement is independently associated with adverse clinical outcomes. Direct viral cytotoxic effect, complications of the disease, and drugs used in the treatments are the pathophy
- Citation: Kayaaslan B, Guner R. COVID-19 and the liver: A brief and core review. World J Hepatol 2021; 13(12): 2013-2023
- URL: https://www.wjgnet.com/1948-5182/full/v13/i12/2013.htm
- DOI: https://dx.doi.org/10.4254/wjh.v13.i12.2013
The emergence of the novel coronavirus disease 2019 (COVID-19) pandemic was a breaking point that deeply affected the whole world and changed medical priorities in daily practice. From the early time of the pandemic, it has been understood that severe acute respiratory syndrome coronavirus 2 (SARS-CoV-2) is not only a respiratory system virus that causes severe lung disease but a systemic disease agent that can affect all systems. Numerous studies from around the world have shown that the liver is damaged in varying degrees in patients with SARS-CoV-2 infection[1]. Recent studies have shown that a considerable part of the COVID-19 patients showed abnormality in liver function tests[2-5]. Liver injury causes a poorer outcome in affected patients, however, its effect on the disease may be more profound than it appears. Herein, we aimed to evaluate the epidemiological characteristics and impact of the liver injury on the clinical outcome, the interaction between pre-existing chronic liver diseases (CLDs) and COVID-19, the pathophysiology of liver involvement and hepatic histopathological findings, and management of liver injury.
The liver is a vital organ that is mainly responsible for protein synthesis, storage of glycogen and regulation of blood glucose levels, metabolism of toxic substances, and many other physiological processes[1]. A great majority of studies revealed that a mild-to-moderate liver involvement was present in a considerable part of COVID-19 patients. However, what liver damage means has not been clearly defined. Zheng et al[6] pointed out that there is no clarity on what liver damage means in their letter to the editors. There are no standardized diagnostic criteria to be considered as a liver injury. The cut-off value of liver function tests varies among studies. The World Health Organization defined the severity of acute COVID-19 as mild, moderate, severe, and critical illness based on respiratory and other systemic findings using technical guidelines[7]. However, the degree of liver and other organ involvement has not been defined yet. There is no standard for cut-off values of liver function tests established by the consensus of researchers. Researchers usually have used different cut-off values, as Zheng Ye et al[6] emphasized. Most of them defined any elevated value above the upper limit of normal (ULN) as liver injury, others preferred values 2-3 times higher than UNL[6,8]. Cai et al[8] defined liver test abnormalities as two groups, elevations of liver enzymes (higher than ULN) and liver injury. Aspartate transaminase (AST)/alanine transaminase (ALT) values above 3 times ULN, or alkaline phosphatase (ALP), gamma-glutamyl transferase (GGT), and total bilirubin values above 2 times ULN were accepted as liver injury.
Lv et al[9] stated concern about the possible misinterpretation of AST data. Determining liver injury incidences based on AST may have led to overestimation. It is believed that ALT is more specific for liver disease and reflects the real hepatic injury. AST is a less specific marker for the liver due to being produced by other tissue such as kidneys, cardiac, and skeletal muscles rather than the liver. Therefore, to be sure of the source of AST, isoform analysis should be done that is not available in routine practice. In addition, antibiotics and antivirals used during the disease also contribute frequently to the elevation of the AST value[5]. A recent study showed that the first rising enzyme is AST followed by ALT[10]. These raise the question of whether the increase in AST may have been caused by other tissues or causes. On the other hand, the studies reported the association between AST level and the disease severity regardless of its source.
In addition, previous diagnosed or undiagnosed CLDs such as chronic viral hepatitis, alcoholic liver disease, non-alcoholic fatty liver disease (NAFLD), may also result in abnormal liver tests. The use of an established set of standards for liver dysfunction/liver injury by researchers is essential in terms of comparability of study results. Therefore, there is an urgent need to define clearly what liver dysfunction/ injury means.
Numerous studies have reported liver injury and varying levels of liver dysfunctions in SARS-CoV-2 infection[3,5]. Most infections manifested as mild to moderate liver disorders presented with abnormal liver function tests [AST/ALT elevations, GGT/ALP elevations, and in some cases hypoproteinemia and prolonged prothrombin time (PT)][2-4,11-15]. In their meta-analysis, Kulkarni et al[5] reported liver function test abnormality in 19% of 1290 non-severe COVID-19 patients from nine articles. Cai et al[8] reported liver injury in 24.9% of non-severe cases. Emerging data from cohort studies have pointed out that liver dysfunction is a commonly encountered entity, usually in more than usually in more than one-third of hospitalized COVID-19 patients[11,16,17]. However, as pointed out above, the incidence of liver injury varies between cohorts, sometimes due to reasons such as differences among study and patient populations, the variety of the drug treatments, and their usage rates. Herein, we mostly addressed several meta-analyses and reviews which evaluated and summarized liver involvements in SARS-CoV-2 infections. A meta-analysis reported the pooled incidence of liver dysfunction as 23.1% at early presentation and 24.1% through the disease course among 15407 patients[5]. The incidence of abnormal levels of liver function was also reported as 29% in another meta-analysis evaluating a total of 38 studies with 3062 COVID-19 patients[17].
In a review, Alqahtani et al[18] analyzed more than thirty published, ahead of print and preprint reports which consisted of mostly case series. They summarized the details of the study types, patients’ numbers, hepatobiliary function markers, inflammatory markers, and proposed possible mechanisms of liver injury. More than 20 publications included in the review had reported abnormal levels of aminotransferase, up to 61.1% of cases. Almost all cases had a modest liver injury except one who had an AST reaching a maximum of 1263 U/L and ALT reaching 2093 U/L. Another retrospective study by Chen et al[19], included in the review, reported that one case had experienced severe hepatitis with an AST of 1445 and ALT of 7590 U/L. A negligible part of patients had pre-existing liver disease. COVID-19 causes usually mild-to-moderate liver injury presented with modest abnormality in liver function tests, and it occasionally resulted in severe hepatitis.
In a comprehensive review evaluating the incidence of hepatic abnormalities in SARS, the Middle East respiratory syndrome, and SARS-CoV-2, Kukla et al[20] analyzed 2541 patients infected with SARS CoV-2 in 11 studies reported from China and reported that liver involvement had occurred with predominantly mild to moderately high transaminases, hypoalbuminemia, and prolongation of PT. A large-scale study of 5700 patients hospitalized with COVID-19 reported elevations of ALT and AST in 39.0% and 58.4% of the patient population, respectively[21]. Cai et al[8] reported 76.3% abnormal liver function tests (higher than ULN) and 21.5% liver injury (defined higher than 3 × AST/ALT or 2 × ALP/GGT/total bilirubin) at admission.
A slight hyperbilirubinemia is accompanied by elevated transaminase in COVID-19. Its incidence was reported as 13.4% in Kulkarni et al[3]’s study. The studies also reported the increase in other liver function tests (ALP, GGT), prolonged PT and decrease in albumin level. Cai et al[8] reported GGT elevation in more than 15% of the patients at admission and in approximately half of the patients during hospitalization. The pooled incidence of prolonged PT was 9.7% in adults with a meta-analysis[5]. As a result, although the incidence rates are in a wide range in studies, the incidence of liver injury was present in at least one-fourth of patients or more.
Accumulated data since the beginning of the pandemic shows that liver dysfunction is significantly associated with a poor outcome in SARS-CoV-2 infection[3,8,11,16,17,22]. Cai et al[8] reported that patients with liver injury had a 9-fold-greater risk of severe COVID-19. A meta-analysis involving 3722 cases in 13 studies revealed that mortality and clinical severity were associated with liver injury in COVID-19 patients[3]. Fu et al[16] reported a higher mortality rate in patients with abnormal liver function tests compared to those with normal liver function tests (29.6% vs 6.5%, P < 0.001), especially AST elevation and total bilirubin elevation groups. Serum AST level was higher in deceased patients and severe COVID-19 cases than in surviving patients and non-severe cases [odds ratio (OR) = 4.48, 95% confidence interval (CI): 3.24-7.21, P < 0.001][3]. A comprehensive meta-analysis investigating the incidence of elevated liver functions, and the association of the patients' outcomes with liver dysfunction and CLDs upon 15407 patients revealed that COVID-19 patients with elevated liver functions had an increased risk for mortality (OR = 3.46, 95%CI: 2.42-4.95, P <0.001) and severe disease (OR = 2.87, 95%CI: 2.29-3.6, P <0.001) compared to patients without elevated liver functions[5]. In another meta-analysis, a higher level of AST, ALT, and bilirubin values, prolonged PT, and a lower level of serum albumin value were found to be associated with severe COVID-19[23]. In consequence, the elevated transaminase and abnormality of other liver function tests were common in COVID-19 patients and independently associated with adverse clinical outcomes.
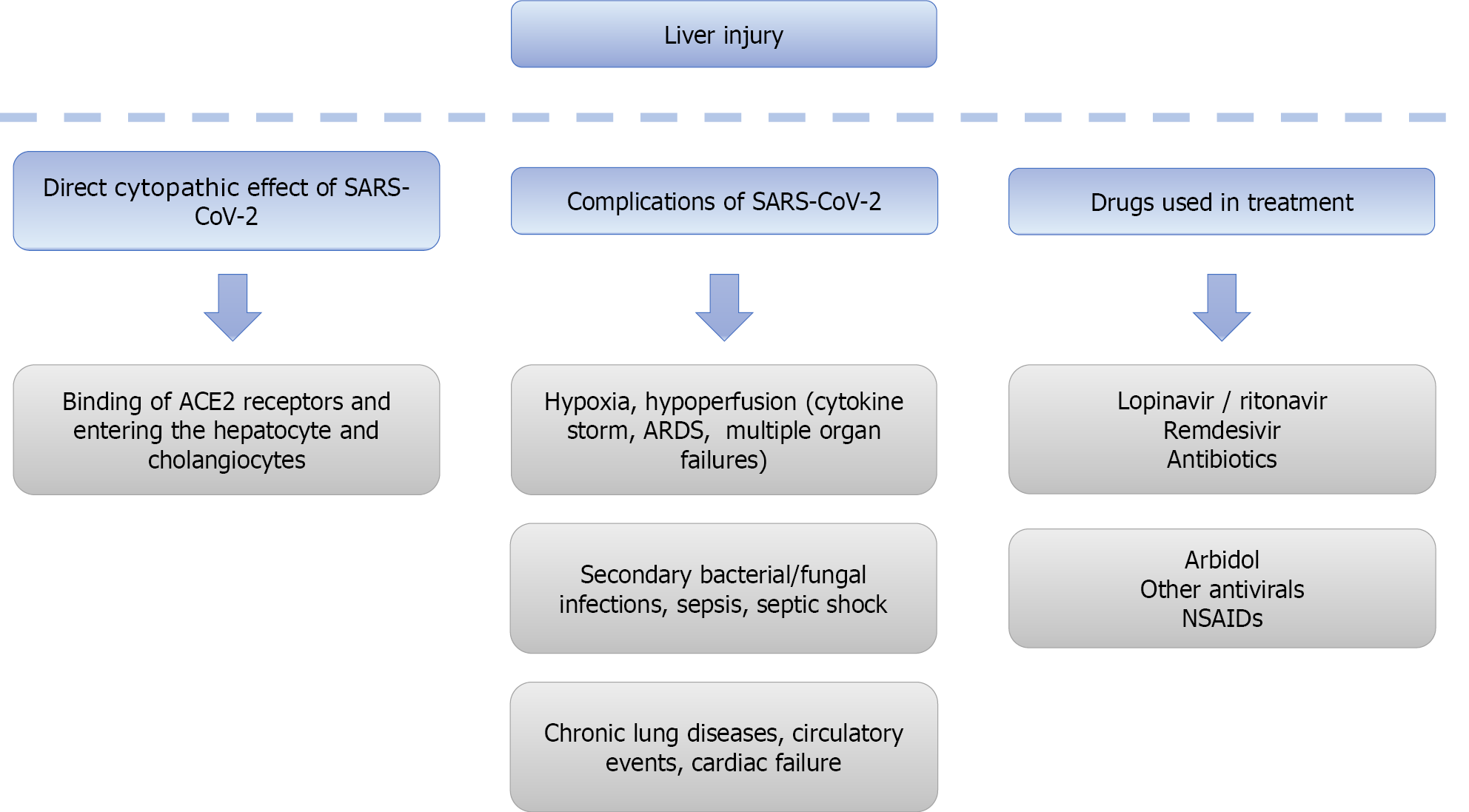
Although much has been learned about SARS-CoV-2 in the elapsed time since the beginning of the pandemic, there remain many points that need to be clarified, particularly its pathogenesis. There is still a dilemma about whether SARS-CoV-2 increases transaminases directly by viral cytotoxic effect or by the consequences of the disease such as hyperinflammation, sepsis, and drugs[24]. Although not yet fully clarified, the pathogenesis of COVID-19 associated liver injury appears to be related to direct viral hepatitis, or the disease-induced complications such as severe respiratory involvement related to hypoxia [e.g., acute respiratory distress syndrome (ARDS)], sepsis, cytokine storm, or drug-related liver enzyme elevations during the infection[9,20,25]. Possible mechanism of liver injury is given in Figure 1.
Recent studies show that SARS-CoV-2 uses angiotensin-converting enzyme 2 (ACE2) receptors, mainly expressed in type 2 alveolar cells of the lungs, to enter the body[26,27]. ACE2 receptors are also mainly localized in the heart, kidney, testes, and other tissues[8]. The liver is a potential target organ for the virus due to its containing high levels of ACE2 receptors[28]. The direct cytotoxic effect and/or inflammatory response of the body to SARS-CoV-2 may be responsible for liver injury. It has been suggested that the binding of SARS-CoV-2 to the ACE2 receptors and entering the hepatocyte and cholangiocytes can cause a direct viral cytotoxic effect on the liver[5], a suggestion that is supported by the findings of a previous study where SARS-CoV-2 RNA was detected in a liver sample[29]. Nardo et al[30] reviewed the pathological findings of COVID-19 patients and proposed that the pathological findings of COVID-19 might be caused by hepatocellular infection with direct cytopathic effect of SARS-CoV-2 and cytokine storm, hypoxic conditions due to ARDS and drug-induced liver injury (DILI) may contribute to these findings. Previous studies had extensively investigated the cell entry mechanism of SARS-CoV-2, and reported that viral entry is triggered by the binding of receptor-binding domain of ACE receptors to the target cells such as alveolar type 2 cells, hepatocytes or cholangiocytes and activated by human proteases such as TMPRSS2[31-33]. However, more data is required to assess the relevance between virus and liver damage. Interestingly, ACE2 expression in cholangiocytes is at similar levels to the lungs, and higher than in the hepatocytes[28]. This may explain the increase in ALP, GGT, and total bilirubin levels. However, COVID-19 patients do not commonly denote a cholestatic pattern of hepatic dysfunction; increased transaminase levels are more predominant. This can be explained by the possibility that hepatic dysfunction predominantly results from secondary causes such as hypoxia and cytokine storm than the direct viral cytopathic effect of the virus[28,34]. Further studies are required to explain why serum transaminases are elevated more than ALP and bilirubin, and to assess the relevance between virus and liver injury.
COVID-19 has a wide range of clinical spectrum from asymptomatic infection to severe infection resulting in death within a short time. COVID-19 patients particularly with severe illness suffer from various degrees of respiratory system involvement and multiple organ failure. Its pathogenesis is complicated and mainly based on immune system dysfunction, at local and systemic levels[35]. Accumulated data on COVID-19 pathogenesis indicates that SARS-CoV-2 induces an excessive cytokine release, known as cytokine storm in some patients, and causes ARDS and multiple organ failures including heart, liver, and kidney[35-37]. Cytokine storm is the life-threatening overactivation of immune cells and dysregulated inflammatory cytokine and chemical production in relation to a triggering factor such as bacterial, fungal and viral pathogens, and is accepted as the main cause of multiple organ injury. It was confirmed that a high level of inflammatory markers such as C-reactive protein, cytokines [interleukin (IL) 1, IL-6, IL-18, tumor necrosis factor, granulocyte-colony stimulating factor], and chemokines are associated with severe infection[11,34,35,38-43]. Cytokines and chemokines stimulate both the innate and adoptive immune system resulting in apoptosis of the infected cells and immune cell hyperstimulation. Therefore, cytokine storm may play a role in the appearance of abnormal liver function tests.
Thromboembolic events are frequent in COVID-19 patients, and another possible explanation of liver involvement is endothelial injury and hyper-coagulability[44]. In a preliminary study, the signs of acute (thrombosis, luminal ectasia) and chronic (fibrous thickening of the vascular wall or phlebosclerosis, presence of abnormal portal intrahepatic system) hepatic vascular involvement was found in all specimens in varying degrees among the main pathological findings[45].
Multiple organ dysfunction induced by other COVID-19-related complications probably contribute to elevated liver function tests. COVID-19 patients, particularly with a severe and critical illness, are at risk for secondary bacterial and fungal infections[46]. Sepsis is a common condition in COVID-19 patients, especially those who are followed up in the intensive care unit and can cause multiple organ dysfunction, including the liver. Besides, the development of septic shock increases the risk of hepatotoxicity through hypoperfusion[47]. Hypoxia and cardiac failure in affected COVID-19 patients can lead to liver injury[34]. Circulatory events, underlying CLD disorders are other secondary reasons for liver injury[11,28,34].
Liver injury may be partially attributed to the drugs used in COVID-19 treatment[5,11]. Liver damage has been reported with the use of lopinavir/ritonavir as an antiviral in SARS-CoV-2 infection[5,8,11]. Cai et al[8] did not detect any significant evidence for increased risk for liver injury in patients using suspected drugs (including antibiotics, non-steroidal anti-inflammatory drugs (NSAIDs), ribavirin, herbal medication used in Chinese medicine, and interferon), except for lopinavir/ritonavir. Patients who used lopinavir/ritonavir had a higher GGT and ALP level. Similarly, Cichoż-Lach et al[11] reported that they did not find any association between the use of antibiotics, NSAIDs, ribavirin, and interferon, and hepatic complications. Only lopinavir/ritonavir had provoked the deterioration of liver function. In a study, the rate of lopinavir/ritonavir use had been detected higher in the patients with hepatic dysfunction than in those without hepatic dysfunction[48]. Kulkarni et al[5] also reported that drug-induced liver injury due to the use of lopinavir/ritonavir, remdesivir, and arbidol is common, but not resulting in life-threatening conditions. The incidence of abnormal liver function tests with lopinavir/ritonavir ranges from 22.7% to 54.6%. Remdesivir is another drug that causes frequent increases (15.2%) in liver function tests. Elevated liver function tests were reported at a rate of 18.7% with the use of arbidol.
Hydroxychloroquine, an antimalarial drug, is one of the most used and studied as immunomodulatory drugs in the treatment of COVID-19[49,50]. Although there is conflicting information about its effectiveness in COVID-19, hepatotoxicity is not a common side effect of hydroxychloroquine. Hydroxychloroquine has been used in the treatment of systemic lupus erythematosus, rheumatoid arthritis, and related diseases for over 70 years[51]. There are only a few case reports of hepatoxicity with hydroxychloroquine[34,52].
Interpreting the data on whether antibiotics, NSAIDs, and other drugs used to treat COVID-19 patients cause hepatotoxic effects is a complicated issue. As discussed above, elevated AST and ALT levels are seen in severe cases or occur during the disease course even if it is normal on admission. These cases stay longer in hospital and combat unfavorable conditions such as secondary bacterial and fungal infections, sepsis, and cytokine storm which require the administration of certain other medications. Rather than thinking that liver enzyme elevation is related solely to the drugs used, it seems more plausible to account that all factors contribute.
Understanding histopathological findings of COVID-19 has an important role in elucidating the pathogenesis of the disease and how liver damage develops. The most common finding in histopathology is steatosis. In a review that involved 9 biopsies and 226 autopsies, histopathology findings of COVID-19 cases in the published studies were evaluated and the most important histopathological findings of lung, heart, liver, and kidney were summarized[53]. Although a limited number of samples was performed in biopsy/autopsy, the most remarkable findings have been detected as steatosis and inflammation. Similarly, Díaz et al[24] reported detecting hepatic steatosis and vascular thrombosis as major and prevalent histological liver findings. Portal and lobular inflammation and Kupffer cell hyperplasia or proliferation were other frequent findings. Steatosis was higher than the normal population. It should be noted that these findings may lead to a bias since patients with more severe illnesses are included in the autopsy or biopsy studies. Besides, it can also be explained by the co-existence of other common causes of steatosis (e.g., diabetes, obesity, NAFLD, hypertension, and heart diseases) in severe COVID-19 patients[9,24].
The prevalence of CLDs among COVID-19 patients is low. Kulkarni et al[5] reported the pooled prevalence of underlying CLDs as 3.6% (95%CI: 2.5-5.1) among 15407 patients in 50 articles, and as 3.9% among 1587 severely ill patients in 15 articles that reported it. However, there are higher rates of its prevalence in different studies. Oyelade et al[54] reported its prevalence as 3%-11% in their meta-analysis. Fu et al[16] reported the prevalence of CLDs as 19.9% (viral hepatitis 8.9% and NAFLD 1%) in their study population and did not find any significant associations between CLDs and elevated liver function tests. Certain studies reported that underlying CLDs are associated with higher mortality[55-57]. Contrary to this, in the comprehensive meta-analysis by Kulkarni et al[5], the presence of CLDs was not associated with severe COVID-19 (OR = 0.8, 95%CI: 0.31-2.09, P = 0.67). Similar to Kulkarni, Lippi et al[58] could not find any association between CLDs and COVID-19 severity (OR = 0.96, 95%CI: 0.36–2.52) and its mortality (OR = 2.33, 95%CI: 0.77–7.04). Conflicting results in the literature about the relation between SARS-CoV-2 infection and pre-existing liver disease may be associated with the heterogeneity of the study populations and the type (e.g., alcoholic liver disease, NAFLD, viral hepatitis) and severity of the underlying liver diseases (e.g., cirrhosis, decompensated disease or hepatocellular carcinoma), and further investigation is needed to clearly understand.
An observational study found the presence of alcohol-related liver disease, decompensated cirrhosis, and hepatocellular carcinoma as independent risk factors for higher mortality in patients with CLDs[55]. In APCOLIS study (APASL COVID-19 Liver Injury Spectrum Study), patients with obesity (in cirrhotic) and diabetes mellitus (in non-cirrhotic) were vulnerable to liver injury[59]. In fact, it appears that chronic liver patients in advanced stages, rather than all chronic liver patients, have a higher risk of severe infection and mortality[56].
The individual risk to being infected with COVID-19 in patients with CLDs depends on several factors including comorbidity, etiology of chronic disease, and baseline liver disease stage[56,60]. Controlled viral hepatitis B and C was not accepted as an exact predisposing factor to SARS-CoV-2 infection[25]. Patients with cirrhosis or hepatocellular carcinoma may be more vulnerable to SARS-CoV-2 infection because of the impairment of patients’ immune systems[61]. However, many more studies are needed to clarify the issue of whether chronic viral hepatitis creates a predisposition to SARS-CoV-2 infection.
In mild cases of COVID-19, liver injury usually resolves spontaneously[61]. If liver injury develops during the COVID-19 clinical course, it should first be investigated whether the abnormal liver function tests are related to the drugs including antivirals, antibiotics, NSAIDs used in the treatment, and if necessary, the drug held responsible for liver damage should be discontinued[34]. However, severe liver injury may require a more meticulous evaluation and careful treatment. The actual cause of liver injury should be investigated, and appropriate treatment provided for possible factors. If present, hypoxia and hypoperfusion should be regulated. Timely control of immune-mediated systemic inflammation and cytokine storm improve the prognosis and reduce respiratory cell infiltration and hypoxia. Anti-inflammatory treatments such as dexamethasone or other corticosteroids that have been found to reduce mortality by suppression of inflammation are used. Dexamethasone 6 mg IV or orally for 10 d (or until discharge if earlier), is recommended in severe cases of COVID-19 particularly with end organ dysfunction. Alternatively, methylprednisolone 32 mg and prednisone 40 mg which are equivalent doses to dexamethasone 6 mg can also be used[62-64]. Corticosteroids are also one of the treatment options in hemophagocytic lymphohistiocytosis, a type of cytokine storm associated with deepening laboratory abnormalities including elevated liver function tests and seen in COVID-19 patients[35]. Other immunomodulatory and cytokine antagonists can be used in the treatment[35]. Adding tocilizumab to standard of care is recommended for progressive severe and critical cases of COVID-19 who have elevated markers of systemic inflammation[62]. Thus, liver damage due to hypoxia or hyperinflammation can be reduced with appropriate and on-time treatment.
To prevent the risks that may arise with COVID-19 infection, EASL recommends SARS-CoV-2 vaccination as early as possible in patients with CLDs, hepatocellular carcinoma, and candidates for liver transplantations as the potential benefits of the vaccine outweigh the risks associated with the vaccine. In transplanted patients, the optimal time of vaccination is 3-6 mo after transplantation[60].
In conclusion, we summarized the epidemiological characteristics of liver involvement in COVID-19 infection and the effects of liver dysfunction on the COVID-19 prognosis. We also evaluated the data on the pathophysiology of liver injury. Abnormal liver function tests have been detected in more than one-fourth of patients with COVID-19 and were associated with poorer outcomes. Abnormal liver function tests in COVID-19 need to be carefully investigated. The detection of real mechanisms on liver injury is a complicated and concurrent condition. Direct viral cytotoxic effect, the disease-induced complications and drugs used in COVID-19 treatment can cause singular or joined liver injury. Appropriate treatment should be provided for the possible reasons of liver injury.
Provenance and peer review: Invited article; Externally peer reviewed.
Peer-review model: Single blind
Specialty type: Infectious Diseases
Country/Territory of origin: Turkey
Peer-review report’s scientific quality classification
Grade A (Excellent): A
Grade B (Very good): B
Grade C (Good): C
Grade D (Fair): 0
Grade E (Poor): 0
P-Reviewer: Fan Y, Khan MKA, Valencia GA S-Editor: Chang KL L-Editor: Filipodia P-Editor: Chang KL
| 1. | Trefts E, Gannon M, Wasserman DH. The liver. Curr Biol. 2017;27:R1147-R1151. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 340] [Cited by in RCA: 902] [Article Influence: 128.9] [Reference Citation Analysis (0)] |
| 2. | Cheong J, Bartell N, Peeraphatdit T, Mosli M, Al-Judaibi B. Gastrointestinal and liver manifestations of COVID-19. Saudi J Gastroenterol. 2020;26:226-232. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 19] [Cited by in RCA: 16] [Article Influence: 3.2] [Reference Citation Analysis (0)] |
| 3. | Wu ZH, Yang DL. A meta-analysis of the impact of COVID-19 on liver dysfunction. Eur J Med Res. 2020;25:54. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 53] [Cited by in RCA: 48] [Article Influence: 9.6] [Reference Citation Analysis (0)] |
| 4. | Kullar R, Patel AP, Saab S. Hepatic Injury in Patients With COVID-19. J Clin Gastroenterol. 2020;54:841-849. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 14] [Cited by in RCA: 20] [Article Influence: 4.0] [Reference Citation Analysis (0)] |
| 5. | Kulkarni AV, Kumar P, Tevethia HV, Premkumar M, Arab JP, Candia R, Talukdar R, Sharma M, Qi X, Rao PN, Reddy DN. Systematic review with meta-analysis: liver manifestations and outcomes in COVID-19. Aliment Pharmacol Ther. 2020;52:584-599. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 199] [Cited by in RCA: 183] [Article Influence: 36.6] [Reference Citation Analysis (0)] |
| 6. | Ye Z, Song B. Liver injury in COVID-19: Diagnosis and associated factors. Liver Int. 2020;40:2040-2041. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 9] [Cited by in RCA: 7] [Article Influence: 1.4] [Reference Citation Analysis (0)] |
| 7. | World Health Organization. Clinical management of COVID-19: Interim Guidance. 27 May, 2020. [cited 18 May 2021]. Available from: https://www.who.int/publications-detail/clinical-management-of-covid-19. |
| 8. | Cai Q, Huang D, Yu H, Zhu Z, Xia Z, Su Y, Li Z, Zhou G, Gou J, Qu J, Sun Y, Liu Y, He Q, Chen J, Liu L, Xu L. COVID-19: Abnormal liver function tests. J Hepatol. 2020;73:566-574. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 623] [Cited by in RCA: 661] [Article Influence: 132.2] [Reference Citation Analysis (0)] |
| 9. | Lv XH, Yang JL, Deng K. Letter to the Editor: COVID-19-Related Liver Injury: The Interpretation for Aspartate Aminotransferase Needs to Be Cautious. Hepatology. 2021;73:874. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 3] [Cited by in RCA: 4] [Article Influence: 1.0] [Reference Citation Analysis (0)] |
| 10. | Lei F, Liu YM, Zhou F, Qin JJ, Zhang P, Zhu L, Zhang XJ, Cai J, Lin L, Ouyang S, Wang X, Yang C, Cheng X, Liu W, Li H, Xie J, Wu B, Luo H, Xiao F, Chen J, Tao L, Cheng G, She ZG, Zhou J, Wang H, Lin J, Luo P, Fu S, Ye P, Xiao B, Mao W, Liu L, Yan Y, Chen G, Huang X, Zhang BH, Yuan Y. Longitudinal Association Between Markers of Liver Injury and Mortality in COVID-19 in China. Hepatology. 2020;72:389-398. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 318] [Cited by in RCA: 309] [Article Influence: 61.8] [Reference Citation Analysis (0)] |
| 11. | Cichoż-Lach H, Michalak A. Liver injury in the era of COVID-19. World J Gastroenterol. 2021;27:377-390. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in CrossRef: 53] [Cited by in RCA: 52] [Article Influence: 13.0] [Reference Citation Analysis (2)] |
| 12. | Deidda S, Tora L, Firinu D, Del Giacco S, Campagna M, Meloni F, Orrù G, Chessa L, Carta MG, Melis A, Spolverato G, Littera R, Perra A, Onali S, Zorcolo L, Restivo A. Gastrointestinal coronavirus disease 2019: epidemiology, clinical features, pathogenesis, prevention, and management. Expert Rev Gastroenterol Hepatol. 2021;15:41-50. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 26] [Cited by in RCA: 20] [Article Influence: 5.0] [Reference Citation Analysis (0)] |
| 13. | Napodano C, Pocino K, Stefanile A, Marino M, Miele L, Gulli F, Basile V, Pandolfi F, Gasbarrini A, Rapaccini GL, Basile U. COVID-19 and hepatic involvement: The liver as a main actor of the pandemic novel. Scand J Immunol. 2021;93:e12977. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 9] [Cited by in RCA: 10] [Article Influence: 2.0] [Reference Citation Analysis (0)] |
| 14. | Wong YJ, Tan M, Zheng Q, Li JW, Kumar R, Fock KM, Teo EK, Ang TL. A systematic review and meta-analysis of the COVID-19 associated liver injury. Ann Hepatol. 2020;19:627-634. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 44] [Cited by in RCA: 45] [Article Influence: 9.0] [Reference Citation Analysis (0)] |
| 15. | Ghoda A, Ghoda M. Liver Injury in COVID-19 Infection: A Systematic Review. Cureus. 2020;12:e9487. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 23] [Cited by in RCA: 25] [Article Influence: 5.0] [Reference Citation Analysis (0)] |
| 16. | Li X, Xu S, Yu M, Wang K, Tao Y, Zhou Y, Shi J, Zhou M, Wu B, Yang Z, Zhang C, Yue J, Zhang Z, Renz H, Liu X, Xie J, Xie M, Zhao J. Risk factors for severity and mortality in adult COVID-19 inpatients in Wuhan. J Allergy Clin Immunol. 2020;146:110-118. [RCA] [PubMed] [DOI] [Full Text] [Cited by in RCA: 1] [Reference Citation Analysis (0)] |
| 17. | Zhu J, Ji P, Pang J, Zhong Z, Li H, He C, Zhang J, Zhao C. Clinical characteristics of 3062 COVID-19 patients: A meta-analysis. J Med Virol. 2020;92:1902-1914. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 422] [Cited by in RCA: 377] [Article Influence: 75.4] [Reference Citation Analysis (0)] |
| 18. | Alqahtani SA, Schattenberg JM. Liver injury in COVID-19: The current evidence. United European Gastroenterol J. 2020;8:509-519. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 176] [Cited by in RCA: 166] [Article Influence: 33.2] [Reference Citation Analysis (0)] |
| 19. | Chen N, Zhou M, Dong X, Qu J, Gong F, Han Y, Qiu Y, Wang J, Liu Y, Wei Y, Xia J, Yu T, Zhang X, Zhang L. Epidemiological and clinical characteristics of 99 cases of 2019 novel coronavirus pneumonia in Wuhan, China: a descriptive study. Lancet. 2020;395:507-513. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 14869] [Cited by in RCA: 12977] [Article Influence: 2595.4] [Reference Citation Analysis (1)] |
| 20. | Kukla M, Skonieczna-Żydecka K, Kotfis K, Maciejewska D, Łoniewski I, Lara LF, Pazgan-Simon M, Stachowska E, Kaczmarczyk M, Koulaouzidis A, Marlicz W. COVID-19, MERS and SARS with Concomitant Liver Injury-Systematic Review of the Existing Literature. J Clin Med. 2020;9. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 69] [Cited by in RCA: 78] [Article Influence: 15.6] [Reference Citation Analysis (0)] |
| 21. | Richardson S, Hirsch JS, Narasimhan M, Crawford JM, McGinn T, Davidson KW; the Northwell COVID-19 Research Consortium, Barnaby DP, Becker LB, Chelico JD, Cohen SL, Cookingham J, Coppa K, Diefenbach MA, Dominello AJ, Duer-Hefele J, Falzon L, Gitlin J, Hajizadeh N, Harvin TG, Hirschwerk DA, Kim EJ, Kozel ZM, Marrast LM, Mogavero JN, Osorio GA, Qiu M, Zanos TP. Presenting Characteristics, Comorbidities, and Outcomes Among 5700 Patients Hospitalized With COVID-19 in the New York City Area. JAMA. 2020;323:2052-2059. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 6024] [Cited by in RCA: 6518] [Article Influence: 1303.6] [Reference Citation Analysis (0)] |
| 22. | Kovalic AJ, Huang G, Thuluvath PJ, Satapathy SK. Elevated Liver Biochemistries in Hospitalized Chinese Patients With Severe COVID-19: Systematic Review and Meta-analysis. Hepatology. 2021;73:1521-1530. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 31] [Cited by in RCA: 40] [Article Influence: 10.0] [Reference Citation Analysis (0)] |
| 23. | Youssef M, H Hussein M, Attia AS, M Elshazli R, Omar M, Zora G, S Farhoud A, Elnahla A, Shihabi A, Toraih EA, S Fawzy M, Kandil E. COVID-19 and liver dysfunction: A systematic review and meta-analysis of retrospective studies. J Med Virol. 2020;92:1825-1833. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 54] [Cited by in RCA: 56] [Article Influence: 11.2] [Reference Citation Analysis (0)] |
| 24. | Díaz LA, Idalsoaga F, Cannistra M, Candia R, Cabrera D, Barrera F, Soza A, Graham R, Riquelme A, Arrese M, Leise MD, Arab JP. High prevalence of hepatic steatosis and vascular thrombosis in COVID-19: A systematic review and meta-analysis of autopsy data. World J Gastroenterol. 2020;26:7693-7706. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in CrossRef: 61] [Cited by in RCA: 57] [Article Influence: 11.4] [Reference Citation Analysis (1)] |
| 25. | Amin M. COVID-19 and the liver: overview. Eur J Gastroenterol Hepatol. 2021;33:309-311. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 53] [Cited by in RCA: 44] [Article Influence: 11.0] [Reference Citation Analysis (0)] |
| 26. | Hoffmann M, Kleine-Weber H, Schroeder S, Krüger N, Herrler T, Erichsen S, Schiergens TS, Herrler G, Wu NH, Nitsche A, Müller MA, Drosten C, Pöhlmann S. SARS-CoV-2 Cell Entry Depends on ACE2 and TMPRSS2 and Is Blocked by a Clinically Proven Protease Inhibitor. Cell. 2020;181:271-280.e8. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 11946] [Cited by in RCA: 14274] [Article Influence: 2854.8] [Reference Citation Analysis (0)] |
| 27. | Yan R, Zhang Y, Li Y, Xia L, Guo Y, Zhou Q. Structural basis for the recognition of SARS-CoV-2 by full-length human ACE2. Science. 2020;367:1444-1448. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 3274] [Cited by in RCA: 3678] [Article Influence: 735.6] [Reference Citation Analysis (0)] |
| 28. | Jothimani D, Venugopal R, Abedin MF, Kaliamoorthy I, Rela M. COVID-19 and the liver. J Hepatol. 2020;73:1231-1240. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 364] [Cited by in RCA: 358] [Article Influence: 71.6] [Reference Citation Analysis (1)] |
| 29. | Remmelink M, De Mendonça R, D'Haene N, De Clercq S, Verocq C, Lebrun L, Lavis P, Racu ML, Trépant AL, Maris C, Rorive S, Goffard JC, De Witte O, Peluso L, Vincent JL, Decaestecker C, Taccone FS, Salmon I. Unspecific post-mortem findings despite multiorgan viral spread in COVID-19 patients. Crit Care. 2020;24:495. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 215] [Cited by in RCA: 209] [Article Influence: 41.8] [Reference Citation Analysis (0)] |
| 30. | Nardo AD, Schneeweiss-Gleixner M, Bakail M, Dixon ED, Lax SF, Trauner M. Pathophysiological mechanisms of liver injury in COVID-19. Liver Int. 2021; 41 :20-32. [RCA] [PubMed] [DOI] [Full Text] [Cited by in RCA: 1] [Reference Citation Analysis (1)] |
| 31. | Shang J, Wan Y, Luo C, Ye G, Geng Q, Auerbach A, Li F. Cell entry mechanisms of SARS-CoV-2. Proc Natl Acad Sci U S A. 2020;117:11727-11734. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 1930] [Cited by in RCA: 2378] [Article Influence: 475.6] [Reference Citation Analysis (0)] |
| 32. | Belouzard S, Millet JK, Licitra BN, Whittaker GR. Mechanisms of coronavirus cell entry mediated by the viral spike protein. Viruses. 2012;4:1011-1033. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 824] [Cited by in RCA: 929] [Article Influence: 71.5] [Reference Citation Analysis (0)] |
| 33. | Heald-Sargent T, Gallagher T. Ready, set, fuse! Viruses. 2012;4:557-580. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 284] [Cited by in RCA: 256] [Article Influence: 19.7] [Reference Citation Analysis (0)] |
| 34. | Metawea MI, Yousif WI, Moheb I. COVID 19 and liver: An A-Z literature review. Dig Liver Dis. 2021;53:146-152. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 62] [Cited by in RCA: 60] [Article Influence: 15.0] [Reference Citation Analysis (2)] |
| 35. | Ye Q, Wang B, Mao J. The pathogenesis and treatment of the `Cytokine Storm' in COVID-19. J Infect. 2020;80:607-613. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 1858] [Cited by in RCA: 1960] [Article Influence: 392.0] [Reference Citation Analysis (0)] |
| 36. | Nile SH, Nile A, Qiu J, Li L, Jia X, Kai G. COVID-19: Pathogenesis, cytokine storm and therapeutic potential of interferons. Cytokine Growth Factor Rev. 2020;53:66-70. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 286] [Cited by in RCA: 301] [Article Influence: 60.2] [Reference Citation Analysis (0)] |
| 37. | Chousterman BG, Swirski FK, Weber GF. Cytokine storm and sepsis disease pathogenesis. Semin Immunopathol. 2017;39:517-528. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 554] [Cited by in RCA: 864] [Article Influence: 108.0] [Reference Citation Analysis (0)] |
| 38. | Qin C, Zhou L, Hu Z, Zhang S, Yang S, Tao Y, Xie C, Ma K, Shang K, Wang W, Tian DS. Dysregulation of Immune Response in Patients With Coronavirus 2019 (COVID-19) in Wuhan, China. Clin Infect Dis. 2020;71:762-768. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 2495] [Cited by in RCA: 3341] [Article Influence: 668.2] [Reference Citation Analysis (0)] |
| 39. | Soy M, Keser G, Atagündüz P, Tabak F, Atagündüz I, Kayhan S. Cytokine storm in COVID-19: pathogenesis and overview of anti-inflammatory agents used in treatment. Clin Rheumatol. 2020;39:2085-2094. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 575] [Cited by in RCA: 552] [Article Influence: 110.4] [Reference Citation Analysis (0)] |
| 40. | Li H, Liu L, Zhang D, Xu J, Dai H, Tang N, Su X, Cao B. SARS-CoV-2 and viral sepsis: observations and hypotheses. Lancet. 2020;395:1517-1520. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 772] [Cited by in RCA: 887] [Article Influence: 177.4] [Reference Citation Analysis (0)] |
| 41. | Huang C, Wang Y, Li X, Ren L, Zhao J, Hu Y, Zhang L, Fan G, Xu J, Gu X, Cheng Z, Yu T, Xia J, Wei Y, Wu W, Xie X, Yin W, Li H, Liu M, Xiao Y, Gao H, Guo L, Xie J, Wang G, Jiang R, Gao Z, Jin Q, Wang J, Cao B. Clinical features of patients infected with 2019 novel coronavirus in Wuhan, China. Lancet. 2020;395:497-506. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 35178] [Cited by in RCA: 30123] [Article Influence: 6024.6] [Reference Citation Analysis (3)] |
| 42. | Liu J, Li S, Liu J, Liang B, Wang X, Wang H, Li W, Tong Q, Yi J, Zhao L, Xiong L, Guo C, Tian J, Luo J, Yao J, Pang R, Shen H, Peng C, Liu T, Zhang Q, Wu J, Xu L, Lu S, Wang B, Weng Z, Han C, Zhu H, Zhou R, Zhou H, Chen X, Ye P, Zhu B, Wang L, Zhou W, He S, He Y, Jie S, Wei P, Zhang J, Lu Y, Wang W, Zhang L, Li L, Zhou F, Wang J, Dittmer U, Lu M, Hu Y, Yang D, Zheng X. Longitudinal characteristics of lymphocyte responses and cytokine profiles in the peripheral blood of SARS-CoV-2 infected patients. EBioMedicine. 2020;55:102763. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 1171] [Cited by in RCA: 1208] [Article Influence: 241.6] [Reference Citation Analysis (0)] |
| 43. | Satış H, Özger HS, Aysert Yıldız P, Hızel K, Gulbahar Ö, Erbaş G, Aygencel G, Guzel Tunccan O, Öztürk MA, Dizbay M, Tufan A. Prognostic value of interleukin-18 and its association with other inflammatory markers and disease severity in COVID-19. Cytokine. 2021;137:155302. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 107] [Cited by in RCA: 107] [Article Influence: 26.8] [Reference Citation Analysis (0)] |
| 44. | Klok FA, Kruip MJHA, van der Meer NJM, Arbous MS, Gommers DAMPJ, Kant KM, Kaptein FHJ, van Paassen J, Stals MAM, Huisman MV, Endeman H. Incidence of thrombotic complications in critically ill ICU patients with COVID-19. Thromb Res. 2020;191:145-147. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 3488] [Cited by in RCA: 3408] [Article Influence: 681.6] [Reference Citation Analysis (0)] |
| 45. | Sonzogni A, Previtali G, Seghezzi M, Alessio MG, Nebuloni M. Liver and COVID 19 infection: a very preliminary lesson learnt from histological post-mortem findings in 48 patients. 2020 Preprint. Available from: Preprints:2020040438. [DOI] [Full Text] |
| 46. | Lai CC, Wang CY, Hsueh PR. Co-infections among patients with COVID-19: The need for combination therapy with non-anti-SARS-CoV-2 agents? J Microbiol Immunol Infect. 2020;53:505-512. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 341] [Cited by in RCA: 311] [Article Influence: 62.2] [Reference Citation Analysis (0)] |
| 47. | Wu J, Song S, Cao HC, Li LJ. Liver diseases in COVID-19: Etiology, treatment and prognosis. World J Gastroenterol. 2020;26:2286-2293. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in CrossRef: 105] [Cited by in RCA: 97] [Article Influence: 19.4] [Reference Citation Analysis (1)] |
| 48. | Fan Z, Chen L, Li J, Cheng X, Yang J, Tian C, Zhang Y, Huang S, Liu Z, Cheng J. Clinical Features of COVID-19-Related Liver Functional Abnormality. Clin Gastroenterol Hepatol. 2020;18:1561-1566. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 559] [Cited by in RCA: 556] [Article Influence: 111.2] [Reference Citation Analysis (0)] |
| 49. | Guner R, Hasanoglu I, Kayaaslan B, Aypak A, Akinci E, Bodur H, Eser F, Kaya Kalem A, Kucuksahin O, Ates I, Bastug A, Tezer Tekce Y, Bilgic Z, Gursoy FM, Akca HN, Izdes S, Erdem D, Asfuroglu E, Hezer H, Kilic H, Cıvak M, Aydogan S, Buzgan T. Comparing ICU admission rates of mild/moderate COVID-19 patients treated with hydroxychloroquine, favipiravir, and hydroxychloroquine plus favipiravir. J Infect Public Health. 2021;14:365-370. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 13] [Cited by in RCA: 10] [Article Influence: 2.5] [Reference Citation Analysis (0)] |
| 50. | RECOVERY Collaborative Group. , Horby P, Mafham M, Linsell L, Bell JL, Staplin N, Emberson JR, Wiselka M, Ustianowski A, Elmahi E, Prudon B, Whitehouse T, Felton T, Williams J, Faccenda J, Underwood J, Baillie JK, Chappell LC, Faust SN, Jaki T, Jeffery K, Lim WS, Montgomery A, Rowan K, Tarning J, Watson JA, White NJ, Juszczak E, Haynes R, Landray MJ. Effect of Hydroxychloroquine in Hospitalized Patients with Covid-19. N Engl J Med. 2020;383:2030-2040. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 878] [Cited by in RCA: 854] [Article Influence: 170.8] [Reference Citation Analysis (0)] |
| 51. | Rainsford KD, Parke AL, Clifford-Rashotte M, Kean WF. Therapy and pharmacological properties of hydroxychloroquine and chloroquine in treatment of systemic lupus erythematosus, rheumatoid arthritis and related diseases. Inflammopharmacology. 2015;23:231-269. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 320] [Cited by in RCA: 373] [Article Influence: 37.3] [Reference Citation Analysis (0)] |
| 52. | Falcão MB, Pamplona de Góes Cavalcanti L, Filgueiras Filho NM, Antunes de Brito CA. Case Report: Hepatotoxicity Associated with the Use of Hydroxychloroquine in a Patient with COVID-19. Am J Trop Med Hyg. 2020;102:1214-1216. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 55] [Cited by in RCA: 49] [Article Influence: 9.8] [Reference Citation Analysis (0)] |
| 53. | Vasquez-Bonilla WO, Orozco R, Argueta V, Sierra M, Zambrano LI, Muñoz-Lara F, López-Molina DS, Arteaga-Livias K, Grimes Z, Bryce C, Paniz-Mondolfi A, Rodríguez-Morales AJ. A review of the main histopathological findings in coronavirus disease 2019. Hum Pathol. 2020;105:74-83. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 106] [Cited by in RCA: 95] [Article Influence: 19.0] [Reference Citation Analysis (0)] |
| 54. | Oyelade T, Alqahtani J, Canciani G. Prognosis of COVID-19 in Patients with Liver and Kidney Diseases: An Early Systematic Review and Meta-Analysis. Trop Med Infect Dis. 2020;5. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 113] [Cited by in RCA: 110] [Article Influence: 22.0] [Reference Citation Analysis (0)] |
| 55. | Kim D, Adeniji N, Latt N, Kumar S, Bloom PP, Aby ES, Perumalswami P, Roytman M, Li M, Vogel AS, Catana AM, Wegermann K, Carr RM, Aloman C, Chen VL, Rabiee A, Sadowski B, Nguyen V, Dunn W, Chavin KD, Zhou K, Lizaola-Mayo B, Moghe A, Debes J, Lee TH, Branch AD, Viveiros K, Chan W, Chascsa DM, Kwo P, Dhanasekaran R. Predictors of Outcomes of COVID-19 in Patients With Chronic Liver Disease: US Multi-center Study. Clin Gastroenterol Hepatol. 2021;19:1469-1479.e19. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 176] [Cited by in RCA: 184] [Article Influence: 46.0] [Reference Citation Analysis (0)] |
| 56. | Marjot T, Moon AM, Cook JA, Abd-Elsalam S, Aloman C, Armstrong MJ, Pose E, Brenner EJ, Cargill T, Catana MA, Dhanasekaran R, Eshraghian A, García-Juárez I, Gill US, Jones PD, Kennedy J, Marshall A, Matthews C, Mells G, Mercer C, Perumalswami PV, Avitabile E, Qi X, Su F, Ufere NN, Wong YJ, Zheng MH, Barnes E, Barritt AS 4th, Webb GJ. Outcomes following SARS-CoV-2 infection in patients with chronic liver disease: An international registry study. J Hepatol. 2021;74:567-577. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 399] [Cited by in RCA: 384] [Article Influence: 96.0] [Reference Citation Analysis (0)] |
| 57. | Chen L, Huang S, Yang J, Cheng X, Shang Z, Lu H, Cheng J. Clinical characteristics in patients with SARS-CoV-2/HBV co-infection. J Viral Hepat. 2020;27:1504-1507. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 36] [Cited by in RCA: 55] [Article Influence: 11.0] [Reference Citation Analysis (0)] |
| 58. | Lippi G, de Oliveira MHS, Henry BM. Chronic liver disease is not associated with severity or mortality in Coronavirus disease 2019 (COVID-19): a pooled analysis. Eur J Gastroenterol Hepatol. 2021;33:114-115. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 39] [Cited by in RCA: 47] [Article Influence: 11.8] [Reference Citation Analysis (0)] |
| 59. | Sarin SK, Choudhury A, Lau GK, Zheng MH, Ji D, Abd-Elsalam S, Hwang J, Qi X, Cua IH, Suh JI, Park JG, Putcharoen O, Kaewdech A, Piratvisuth T, Treeprasertsuk S, Park S, Wejnaruemarn S, Payawal DA, Baatarkhuu O, Ahn SH, Yeo CD, Alonzo UR, Chinbayar T, Loho IM, Yokosuka O, Jafri W, Tan S, Soo LI, Tanwandee T, Gani R, Anand L, Esmail ES, Khalaf M, Alam S, Lin CY, Chuang WL, Soin AS, Garg HK, Kalista K, Batsukh B, Purnomo HD, Dara VP, Rathi P, Al Mahtab M, Shukla A, Sharma MK, Omata M; APASL COVID Task Force, APASL COVID Liver Injury Spectrum Study (APCOLIS Study-NCT 04345640). Pre-existing liver disease is associated with poor outcome in patients with SARS CoV2 infection; The APCOLIS Study (APASL COVID-19 Liver Injury Spectrum Study). Hepatol Int. 2020;14:690-700. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 217] [Cited by in RCA: 216] [Article Influence: 43.2] [Reference Citation Analysis (1)] |
| 60. | Cornberg M, Buti M, Eberhardt CS, Grossi PA, Shouval D. EASL position paper on the use of COVID-19 vaccines in patients with chronic liver diseases, hepatobiliary cancer and liver transplant recipients. J Hepatol. 2021;74:944-951. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 169] [Cited by in RCA: 174] [Article Influence: 43.5] [Reference Citation Analysis (0)] |
| 61. | Zhang C, Shi L, Wang FS. Liver injury in COVID-19: management and challenges. Lancet Gastroenterol Hepatol. 2020;5:428-430. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 1348] [Cited by in RCA: 1295] [Article Influence: 259.0] [Reference Citation Analysis (4)] |
| 62. | Bhimraj A, Morgan RL, Shumaker AH, Lavergne V, Baden L, Cheng VC, Edwards KM, Gandhi R, Muller WJ, O'Horo JC, Shoham S, Murad MH, Mustafa RA, Sultan S, Falck-Ytter Y. Infectious Diseases Society of America Guidelines on the Treatment and Management of Patients with COVID-19. Infectious Diseases Society of America 2021; Version 4.2.0. [Accessed 15 May 2021]. Available from: https://www.idsociety.org/practice-guideline/covid-19-guideline-treatment-and-management/. |
| 63. | WHO Rapid Evidence Appraisal for COVID-19 Therapies (REACT) Working Group; Sterne JAC, Murthy S, Diaz JV, Slutsky AS, Villar J, Angus DC, Annane D, Azevedo LCP, Berwanger O, Cavalcanti AB, Dequin PF, Du B, Emberson J, Fisher D, Giraudeau B, Gordon AC, Granholm A, Green C, Haynes R, Heming N, Higgins JPT, Horby P, Jüni P, Landray MJ, Le Gouge A, Leclerc M, Lim WS, Machado FR, McArthur C, Meziani F, Møller MH, Perner A, Petersen MW, Savovic J, Tomazini B, Veiga VC, Webb S, Marshall JC. Association Between Administration of Systemic Corticosteroids and Mortality Among Critically Ill Patients With COVID-19: A Meta-analysis. JAMA. 2020;324:1330-1341. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 1683] [Cited by in RCA: 1681] [Article Influence: 336.2] [Reference Citation Analysis (0)] |
| 64. | Wu C, Hou D, Du C, Cai Y, Zheng J, Xu J, Chen X, Chen C, Hu X, Zhang Y, Song J, Wang L, Chao YC, Feng Y, Xiong W, Chen D, Zhong M, Hu J, Jiang J, Bai C, Zhou X, Song Y, Gong F. Corticosteroid therapy for coronavirus disease 2019-related acute respiratory distress syndrome: a cohort study with propensity score analysis. Crit Care. 2020;24:643. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 37] [Cited by in RCA: 41] [Article Influence: 8.2] [Reference Citation Analysis (0)] |