Published online Apr 26, 2015. doi: 10.4252/wjsc.v7.i3.649
Peer-review started: November 29, 2014
First decision: December 12, 2014
Revised: December 18, 2014
Accepted: January 18, 2015
Article in press: January 20, 2015
Published online: April 26, 2015
Processing time: 145 Days and 10.9 Hours
Normal mouse pluripotent stem cells were originally derived from the inner cell mass (ICM) of blastocysts and shown to be the in vitro equivalent of those pre-implantation embryonic cells, and thus were called embryonic stem cells (ESCs). More than a decade later, pluripotent cells were isolated from the ICM of human blastocysts. Despite being called human ESCs, these cells differ significantly from mouse ESCs, including different morphology and mechanisms of control of pluripotency, suggesting distinct embryonic origins of ESCs from the two species. Subsequently, mouse pluripotent stem cells were established from the ICM-derived epiblast of post-implantation embryos. These mouse epiblast stem cells (EpiSCs) are morphological and epigenetically more similar to human ESCs. This raised the question of whether cells from the human ICM are in a more advanced differentiation stage than their murine counterpart, or whether the available culture conditions were not adequate to maintain those human cells in their in vivo state, leading to a transition into EpiSC-like cells in vitro. More recently, novel culture conditions allowed the conversion of human ESCs into mouse ESC-like cells called naïve (or ground state) human ESCs, and the derivation of naïve human ESCs from blastocysts. Here we will review the characteristics of each type of pluripotent stem cells, how (and whether) these relate to different stages of embryonic development, and discuss the potential implications of naïve human ESCs in research and therapy.
Core tip: Mouse embryonic stem cells (ESCs) are the in vitro equivalent of the epiblast of preimplantation embryos. Human ESCs on the other hand, although also pluripotent, appear to be in a more advanced developmental stage than their murine counterpart. Recently, several reports described culture conditions able to support mouse ESC-like human ESCs, called naïve human ESCs. Here we will review the search for human naïve ESCs, discussing their biological and practical relevance.
- Citation: Fonseca SAS, Costas RM, Pereira LV. Searching for naïve human pluripotent stem cells. World J Stem Cells 2015; 7(3): 649-656
- URL: https://www.wjgnet.com/1948-0210/full/v7/i3/649.htm
- DOI: https://dx.doi.org/10.4252/wjsc.v7.i3.649
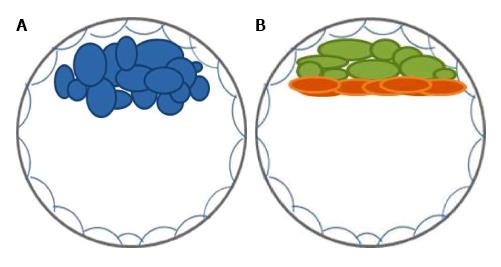
The mammalian preimplatation development is a process of cell multiplication and differentiation from the zygote to the totipotent blastomeres at the cleavage stage, and then to the pluripotent cells at the blastocyst stage. During preimplantation development, cell lineage specification and spatial segregation result in two cell fate decisions, which are believed to be sequential. The first segregation is into the trophectoderm, involved in the formation of the fetal portion of the placenta, and the inner cell mass (ICM) at the early blastocyst stage. The second segregation divides the ICM into the primitive endoderm, that origins parietal and visceral endoderm of the yolk sac[1], and the epiblast, which will give rise to the embryo proper[2] (Figure 1).
Mouse embryonic stem cells (ESCs) are derived from the ICM of blastocysts and represent the epiblast lineage in vitro[3,4]. They are able to maintain an undifferentiated state (self-renewal) and to differentiate into tissues from all three germ layers in vitro and in vivo. In addition, when reintroduced in preimplantation embryos, mouse ESCs are able to get incorporated and give rise to all tissues of the resulting chimera.
In the pluripotent state, mouse ESCs are characterized by the presence of transcription factors such as OCT3/4[5] and Nanog[6], expression of alkaline phosphatase and specific cell surface glycoproteins such as stage-specific embryonic antigen 1[7], and high telomerase activity[8]. In addition, like the epiblast lineage in vivo, female mouse ESCs have not undergone random X chromosome inactivation (XCI), and will do so upon differentiation in vitro[9] .
Maintenance of the undifferentiated state of ESC in vitro is greatly dependent on exogenous factors. The first mESC derived were maintained in medium conditioned by teratocarcinoma cells[4], or co-cultured with irradiated mouse embryonic fibroblasts (MEF) in an enriched culture medium which guaranteed self-renewal[3,10]. Later, maintenance of pluripotency was shown to be dependent on myeloid leukemia inhibitory factor (LIF), a molecule which induces differentiation of M1 myeloid leukemia cells[11]. LIF belongs to the interleukin-6 cytokine family and binds to a heterodimeric receptor consisting of the LIF-receptor and gp130[12]. LIF acts on mESC self-renewal mainly by activating Stat3 (signal transducer and activator of transcription 3) through the LIF/Stat3 pathway[13]. In addition, different matrices and scaffolds are also important for maintenance and/or determination of cell fate[14].
The first lines of human ESCs were established in 1998 from the ICM of human blastocysts, in culture conditions equivalent to those of mouse ESCs[15,16]. Subsequently, it was shown that instead of LIF, bFGF was required for the prolonged culture of human ESCs in the pluripotent state[17].
Despite being derived from preimplatation embryos in apparently the same stage of development, human ESCs differ from their murine counterparts in many ways, including morphology (flat vs domed shaped colonies), passage in culture (clusters vs single cell passage), growth factors dependence (bFGF and TGFB/ACTIVIN/NODAL vs LIF/STAT3 signaling)[16], genomic stability[18], and the epigenetic state of the X chromosome[19,20]. Furthermore, control of induced specific differentiation of mouse ESCs was shown to differ from human ESCs in many cases[21], where protocols established with mouse ESCs do not necessarily yield the same differentiated cell population from their human counterpart. This suggests distinct epigenetic states and/or developmental pathways between ESCs from the two species.
Regarding the X chromosome, although female human ESCs in a pre-XCI status equivalent to that of mouse ESCs have been described[22], this appears to be an unstable epigenetic state, which is lost with time in culture leading to pluripotent cells with one active and one inactive X chromosome[19,23,24]. This suggested that human ESCs may be in a different developmental stage than mouse ESCs. However, the kinetics of XCI during human pre-implantation development is still not clear, and thus it is not known whether human ESCs are indeed the in vitro equivalent of the epiblast lineage, or whether they are altered during establishment/culture by the current in vitro conditions[25,26].
A novel source of pluripotent stem cells was established in 2006: those derived through nuclear reprograming of somatic cells by exogenous expression of Oct4, Klf4, c-Myc and Sox2[27]. These induced pluripotent stem cells (iPSCs) were first established from mouse fibroblasts, and the same strategy was able to convert human fibroblasts also into a pluripotent state[28].
Human and mouse iPSCs are equivalent to their embryo-derived counterparts human and mouse ESCs, respectively[27,28], regarding morphology, growth factor dependence and XCI state. Thus, human and mouse pluripotent stem cells derived from developmentally equivalent cells (adult fibroblasts) are still distinct in several important aspects. This may suggest that the differences between pluripotent stem cells from the two species are not due to distinct origins of the starting embryonic cell, but rather to distinct pathways defining and controlling pluripotency.
Indirect evidence that human ESCs are not the developmental equivalent of mouse ESCs came from the establishment of pluripotent SCs derived from the epiblast of post-implantation mouse embryo-epiblast stem cells (EpiSCs)[29,30]. Although these cells form teratomas when injected in SCID mice, when injected into preimplantation embryos EpiSCs do not integrate to form chimeras like mouse ESCs do. EpiSCs keep their pluripotency in vitro when cultured under human ESCs’ conditions, present the same flat colony-morphology of the human cells, and are in a post-XCI state. In addition, EpiSC and human ESCs present expression profiles characteristic of the epiblast, suggesting that indeed the human pluripotent cells have embryonic origin distinct of mouse ESCs, and similar to mouse EpiSC.
Therefore, two distinct classes of pluripotent SCs were defined: ground state, or naïve-pluripotent SCs, like mouse ESCs/iPSCs; and primed-pluripotent SC, like human ESCs/iPSCs and mouse EpiSCs, which are in a latter developmental stage[31]. And thus, the question was rephrased: can human naïve ESCs be established?
In 2009, Bao et al[32] demonstrated that mouse EpiSC were able to revert to the more primitive epigenetic state of ESCs just by culture in LIF/MEFs. After few passages, ESC-like cells started appearing among EpiSCs, and eventually a sub-line of reprogrammed epiblast ESC-like cells was derived. As part of the reprogramming, the cells reactivated the inactive X chromosome, changing into a pre-XCI state like mouse ESCs and iPSCs. Thus, if mouse EpiSCs could be reverted into ESC-like cells, could human ESCs also be reverted into a naïve pluripotent state by simply changing media conditions?
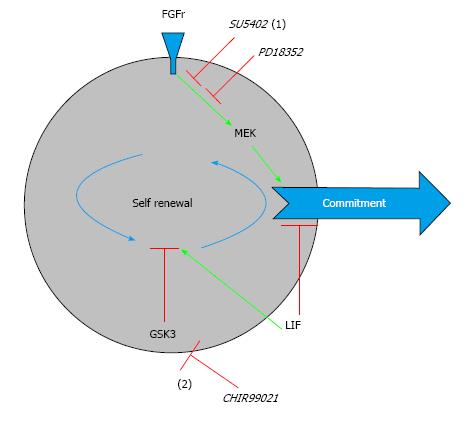
One important improvement in the dissection of the pluripotent state was the demonstration that it was not dependent on external stimuli like serum and MEFs, but instead that pluripotency could be maintained by inhibition of endogenous signaling by mitogen-activated protein kinase (MEK), fibroblast growth factor (FGF) receptor tyrosine kinase and glycogen synthase kinase 3 (GSK3). This lead to the development of the 3i culture condition, which involves small molecules-inhibition of GSK3, MEK and FGF receptor tyrosine kinase signaling by small molecules CHIR99021, PD18352 and SU5402, respectively[33]. While inhibition of GSK3 promotes cell growth and viability, inhibition of FGF receptor tyrosine kinase blocks phosphorylation of MEK, thus hampering its action in promoting cell commitment (Figure 2). In comparison, in the original culture conditions of mouse ESCs, LIF acts only in blocking cell commitment, which would be one step later that ERK phosphorilation, and other factors present in serum and secreted from MEFs promote cell growth and viability[13,34].
Subsequently it was found that inhibition of MEK by PD0325901 was more potent and could substitute both PD18352 and SU5402, in what was called 2i media (CHIR99021 + PD0325901)[33,35].The 3i/2i culture conditions allowed the derivation of germ line competent pluripotent stem cells from other species like rat[36,37] and pigs[38-43].
Therefore, could this media support derivation of human naïve ESCs? Although one report described the generation of apparently naïve hiPSCs in 3i media[44], those results were not replicated by any other group, nor were the human cell lines derived ever used in other reports. Thus, by the lack of publications at that time on the derivation of human naïve ESCs, it became obvious that those relatively simple conditions that worked for many mammalian species were not enough for human cells.
Nevertheless, small molecules took the spot light in pluripotent stem cell research, including epi-drugs like inhibitors of DNA methyltransferases and histone deacetylases, shown to have important roles on cell reprograming and maintenance of pluripotency[45].
Indeed, it was subsequently shown that naïve human pluripotent SCs could only be obtained in 2i when expression of exogenous reprogramming factors was maintained in the cells[46,47]. Generation of hiPSCs with constant ectopic expression of Oct4, Sox2, Klf4, c-Myc, and Nanog in the presence of LIF lead to human pluripotent SCs with behavior in culture and ease to be genetically manipulated equivalent to those of mouse ESCs[46]. Similarly, ectopic induction of Oct4, Klf4 and Klf2 in human ESCs in 2i/LIF media reprogrammed the cells into a naïve state transcriptionally similar to that of mouse ESCs, including the reactivation of the inactive X[47]. Nevertheless, both strategies required continuous expression of the exogenous factors for maintenance of the naïve state.
The next year, Wang et al[48] reported the efficient generation of exogenous factor-independent naïve hiPSC by transient expression of Oct4, c-Myc, Klf4, Sox2, Rarg and Lrh-1 in adult fibroblasts, demonstrating a pivotal role of retinoic acid receptor signaling in reprogramming. However, to our knowledge their results were not replicated by any other group.
After a hiatus of almost two years on reports of naïve human PSCs, within 3 mo four articles described the identification of culture conditions adequate for culture and/or derivation of human naïve ESCs and iPSCs without expression of external factors[49-52] (Table 1). The first report performed a functional screen of 16 small molecules able to support OCT4 expression in hiPSCs in LIF/2i. This lead to the identification of a combination of 7 factors (including bFGF) able to maintain hiPSCs in a naïve state and to support derivation of naïve human ESCs from blastocysts[49]. When introduced to mouse embryos, the naïve cells were able to contribute to the formation of E10.5 chimeras, a hallmark of mouse ESCs and not EpiSCs.
| Ref. | Compounds (inhibitor of) | Conversion | Derivation | XCI status |
| Gafni et al[49] | PD0325901 (MEKi)1, CHIR99021 (GSKi)1, SP600125 (JNKi), SB203580 (MAPKi), LIF1, IGF1, FGF2, TGFB1 | hESC/hiPSC | hESC/hiPSC | XIST RNA (-) H3K27me3 (-) XaXa |
| Chan et al[50] | PD0325901 (MEKi)1, BIO (GSKi)1, Dorsomorphin (BMPi), LIF12 | hESC/hiPSCs | hiPSCs | nt |
| Ware et al[51] | PD0325901 (MEKi)1, CHIR99021 (GSKi)1, FGF2, LIF1, Activin A, NSC74859 (STAT3i), SU5402 (FGFRi) | hESC/hiPSC | hESC | XIST RNA (-) XaXa |
| Valamehr et al[52] | PD0325901 (MEKi)1, CHIR99021 (GSKi)1, Thiazovivin (ROCKi), FGF2, LIF1, SB431542 (ALKi/TGFB1R) | - | hiPSC | XIST RNA (downreg.) H3K27me3 (< 10%) XaXa |
| Theunissen et al[55] | PD0325901 (MEKi)1, CHIR99021 (GSKi)1, SB590885 (BRAFi), Y27632 (Rocki), WH-4-023 (pan-RTKi), LIF1, FGF, Activin A2 | hESC | hESC/hiPSC | XIST RNA (+) XaXi |
In another report, based on the finding that NANOG expression is higher in the human pre-implantation epiblast than in human ESCs[53], Chan et al[50] tested 11 small molecules that would increase NANOG expression in hESCs. They identified a combination of 3 factors that rewired human ESCs into a naïve state responsive to LIF, which was required for self-renewal of the cells (Table 1). In addition, the authors showed that the naïve cells had an expression profile more similar to that of human blastocyst cells than conventional (or primed) human ESCs.
A third report of conditions for naïve human PSCs derivation and culture described a simpler strategy, where human ESCs were exposed to histone deacetylase inhibitors for a few passages before being transitioned to 2i media with bFGF[51] (Table 1). The 2i + bFGF also supported the derivation of a new line of naïve human ESC that could then be transitioned in to 3i media with LIF. Nevertheless, the efficiency of derivation was very low, and cells in 3i + LIF showed significantly higher rates of differentiation than those kept in 2i + bFGF. Furthermore, although cloning efficiency of the naïve human ESC line established was twice of the primed cell line, it was still relatively low (20%) when compared to other reports, whereas there was no improvement of cloning efficiency of naïve H1 compared to the original primed H1. Thus, the strategy appeared to generate cells in an intermediate naïve state.
Another alternative media for maintenance of transgene-expression free naïve hiPSCs was described by Valamehr et al[52] (Table 1). While developing culture conditions for high throughput generation of hiPSCs, the group developed a media containing inhibitors of ROCK, GSK3 and MEK pathways, LIF and bFGF that allowed long term culturing of naïve hiPSCs derived in a previously described cocktail of small molecules[54]. Although those naïve hiPSCs did not have mouse ESC-like morphology, they showed high survival rates in single-cell dissociation, and appeared to be in a pre-XCI state.
More recently, Theunissen et al[55] performed an alternative functional screen of small molecules able to support growth of human naïve ESCs based on the finding that naïve pluripotent stem cells use the distal while primed cells used the proximal enhancer of OCT4. Using a GFP-reporter gene under the control of the distal enhancer of OCT4 in human ESCs, the authors screened a kinase inhibitor library in the presence of 2i/LIF, identifying a combination of 5 compounds which, together with Activin A and FGF, supports conversion of primed into naïve human ESCs and the derivation of new naïve human ESCs and iPSCs (Table 1). It is important to note that, although these converted or newly established naïve cells had the expected dome-shaped morphology and expression profile more similar to mouse ESCs than to primed human ESCs, they presented a few undesired characteristics, namely, (1) high frequency of aneuploidies, and (2) XCI. The authors suggest that, rather than limitations of their “naïve” cells, these may represent differences between naïve human and murine ESCs.
Which brings us to a crucial point: what is a human naïve ESC? When searching for the human naïve pluripotent state, are we looking for the in vitro equivalent of the human epiblast, or for a human pluripotent stem cell as amenable to manipulation in culture as mouse ESCs? Depending on the goal, the characteristics of the resulting naïve human cells can be very different.
The methods for culturing human primed ESCs have significantly improved in the last 10 years with the development of defined and feeder-free culture conditions and of solutions to substitute manual passage. Nevertheless, these cells are still harder to culture and to genetically manipulate than their mouse counterpart. Thus, the development of culture conditions that make human ESCs behave like mouse ESCs may facilitate their use in research and therapy, regardless of whether this naïve state has any relevance as a model for human development. In that case, we know that these human naïve ESCs should have enhanced proliferation, be passaged as single cells (high cloning efficiency), be genetically stable and amenable to efficient genetic manipulation in vitro. In addition, being in a pre-XCI state and undergoing random XCI upon differentiation would be desirable for the generation of naïve hiPSCs to model X-linked diseases[56].
On the other hand, if the goal is to obtain cells that model the pluripotent epiblast from human blastocysts, then we must step back and look at what we know about these cells in vivo. Despite the difficulties in studying the human embryo, enough has been done to know that there are significant differences between human and mouse blastocysts so that one cannot completely extrapolate data from one species to the other[57]. For instance, at the blastocyst stage, while Oct4 expression is restricted to epiblast cells in mouse[58], it is also found in the human trophectoderm[59]. Thus, the organization and/or the markers of pluripotent cells in the human blastocyst may differ from mouse.
Recent advances in single-cell RNA sequencing (RNAseq) have allowed the study of expression profiles of individual cells during human pre-implantation development[53,60]. Analysis of RNAseq data from 30 single-cells derived from human blastocysts identified three distinct clusters corresponding to cells from the epiblast, primitive endoderm and throphectoderm. Furthermore, comparison of the expression profile of human epiblast cells with human ESCs at different stages of derivation showed that, although similar, there are significant changes in gene expression in the embryo-derived cells at the initial primary outgrow of hESCs[53]. That is not surprising given that the cells are adapting to the in vitro conditions, but how distinct can they be while still modeling the epiblast in vitro? It will be interesting to do the same comparison in mice in order to evaluate whether these changes between murine epiblast and ES cells are less accentuated. Nevertheless, it would be reasonable to use the single-cell RNASeq data from human blastocysts as a signature of “true” naïve human ESCs. Indeed, Chan et al[50] showed that the expression profile of their naïve human ESCs was more similar to those of single cells from human blastocysts. The same analysis should be performed with the other reported naïve human ESCs for comparison.
Regarding XCI, the kinetics of the process is very well characterized during mouse preimplatation development, starting at the two-cell stage with the imprinted inactivation of the paternal X (pX) in all cells of the embryo. Then, at the blastocyst stage, cells of the ICM reactivate the pX, and go through a second wave of XCI, where each cell will randomly choose the Xi. In contrast, much less is known about the XCI status of the human preimplatation embryo, and results are conflicting[23]. It would be extremely valuable if “true” naïve human ESCs were in a pre-XCI state like their murine counterpart in order to study the mechanisms of this epigenetic phenomenon in vitro. However, we still don’t know whether this would represent the in vivo situation, and thus, whether the pre-XCI state is a prerequisite for being a “true” naïve human ESC.
While this manuscript was in revision, Wang et al[61] showed that small populations of naïve human pluripotent SCs exist within cultures of primed human ESCs and iPSCs. These naïve cells can be purified and maintained in 2i/LIF media. In addition, the authors identified increased expression of the primate-specific endogenous retrovirus HERVH as a genetic signature of naïve human pluripotent SCs, which can be used for the development of culture conditions to support human naïve PSC culture. More importantly, the study of HERVH-associated transcription during human pre-implantation embryonic development may provide more insights into this still much-unknown process.
In conclusion, in the last year different culture conditions were developed that support the derivation and growth of human ESCs more similar to mouse ESCs. Whether the distinct methods generate similar or distinct epigenetic states of human pluripotent SCs, and more importantly, which of these potentially naïve human pluripotent SCs are more similar to the cells of the human epiblast remain to be determined. These studies will have great impact on the establishment of a good in vitro model for human embryonic development.
P- Reviewer: Bartova E, Chen Y S- Editor: Tian YL L- Editor: A E- Editor: Lu YJ
| 1. | Kwon GS, Viotti M, Hadjantonakis AK. The endoderm of the mouse embryo arises by dynamic widespread intercalation of embryonic and extraembryonic lineages. Dev Cell. 2008;15:509-520. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 361] [Cited by in RCA: 323] [Article Influence: 19.0] [Reference Citation Analysis (0)] |
| 2. | Schrode N, Xenopoulos P, Piliszek A, Frankenberg S, Plusa B, Hadjantonakis AK. Anatomy of a blastocyst: cell behaviors driving cell fate choice and morphogenesis in the early mouse embryo. Genesis. 2013;51:219-233. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 76] [Cited by in RCA: 85] [Article Influence: 7.1] [Reference Citation Analysis (0)] |
| 3. | Evans MJ, Kaufman MH. Establishment in culture of pluripotential cells from mouse embryos. Nature. 1981;292:154-156. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 5956] [Cited by in RCA: 5431] [Article Influence: 123.4] [Reference Citation Analysis (0)] |
| 4. | Martin GR. Isolation of a pluripotent cell line from early mouse embryos cultured in medium conditioned by teratocarcinoma stem cells. Proc Natl Acad Sci USA. 1981;78:7634-7638. [PubMed] |
| 5. | Schöler HR. Octamania: the POU factors in murine development. Trends Genet. 1991;7:323-329. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 246] [Cited by in RCA: 265] [Article Influence: 7.8] [Reference Citation Analysis (0)] |
| 6. | Mitsui K, Tokuzawa Y, Itoh H, Segawa K, Murakami M, Takahashi K, Maruyama M, Maeda M, Yamanaka S. The homeoprotein Nanog is required for maintenance of pluripotency in mouse epiblast and ES cells. Cell. 2003;113:631-642. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 2410] [Cited by in RCA: 2328] [Article Influence: 105.8] [Reference Citation Analysis (0)] |
| 7. | Solter D, Knowles BB. Monoclonal antibody defining a stage-specific mouse embryonic antigen (SSEA-1). Proc Natl Acad Sci USA. 1978;75:5565-5569. [PubMed] |
| 8. | Armstrong L, Saretzki G, Peters H, Wappler I, Evans J, Hole N, von Zglinicki T, Lako M. Overexpression of telomerase confers growth advantage, stress resistance, and enhanced differentiation of ESCs toward the hematopoietic lineage. Stem Cells. 2005;23:516-529. [RCA] [PubMed] [DOI] [Full Text] [Cited by in RCA: 5] [Reference Citation Analysis (0)] |
| 9. | Payer B, Lee JT. X chromosome dosage compensation: how mammals keep the balance. Annu Rev Genet. 2008;42:733-772. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 353] [Cited by in RCA: 376] [Article Influence: 23.5] [Reference Citation Analysis (0)] |
| 10. | Wobus AM, Holzhausen H, Jäkel P, Schöneich J. Characterization of a pluripotent stem cell line derived from a mouse embryo. Exp Cell Res. 1984;152:212-219. [PubMed] |
| 11. | Williams RL, Hilton DJ, Pease S, Willson TA, Stewart CL, Gearing DP, Wagner EF, Metcalf D, Nicola NA, Gough NM. Myeloid leukaemia inhibitory factor maintains the developmental potential of embryonic stem cells. Nature. 1988;336:684-687. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 1387] [Cited by in RCA: 1346] [Article Influence: 36.4] [Reference Citation Analysis (0)] |
| 12. | Gearing DP, Thut CJ, VandeBos T, Gimpel SD, Delaney PB, King J, Price V, Cosman D, Beckmann MP. Leukemia inhibitory factor receptor is structurally related to the IL-6 signal transducer, gp130. EMBO J. 1991;10:2839-2848. [PubMed] |
| 13. | Niwa H, Ogawa K, Shimosato D, Adachi K. A parallel circuit of LIF signalling pathways maintains pluripotency of mouse ES cells. Nature. 2009;460:118-122. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 677] [Cited by in RCA: 690] [Article Influence: 43.1] [Reference Citation Analysis (0)] |
| 14. | Lambshead JW, Meagher L, O’Brien C, Laslett AL. Defining synthetic surfaces for human pluripotent stem cell culture. Cell Regen (Lond). 2013;2:7. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 23] [Cited by in RCA: 25] [Article Influence: 2.1] [Reference Citation Analysis (0)] |
| 15. | Thomson JA, Itskovitz-Eldor J, Shapiro SS, Waknitz MA, Swiergiel JJ, Marshall VS, Jones JM. Embryonic stem cell lines derived from human blastocysts. Science. 1998;282:1145-1147. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 11399] [Cited by in RCA: 10427] [Article Influence: 386.2] [Reference Citation Analysis (0)] |
| 16. | Reubinoff BE, Pera MF, Fong CY, Trounson A, Bongso A. Embryonic stem cell lines from human blastocysts: somatic differentiation in vitro. Nat Biotechnol. 2000;18:399-404. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 2078] [Cited by in RCA: 1873] [Article Influence: 74.9] [Reference Citation Analysis (0)] |
| 17. | Amit M, Carpenter MK, Inokuma MS, Chiu CP, Harris CP, Waknitz MA, Itskovitz-Eldor J, Thomson JA. Clonally derived human embryonic stem cell lines maintain pluripotency and proliferative potential for prolonged periods of culture. Dev Biol. 2000;227:271-278. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 1117] [Cited by in RCA: 1014] [Article Influence: 40.6] [Reference Citation Analysis (0)] |
| 18. | Amps K, Andrews PW, Anyfantis G, Armstrong L, Avery S, Baharvand H, Baker J, Baker D, Munoz MB, Beil S. Screening ethnically diverse human embryonic stem cells identifies a chromosome 20 minimal amplicon conferring growth advantage. Nat Biotechnol. 2011;29:1132-1144. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 489] [Cited by in RCA: 438] [Article Influence: 31.3] [Reference Citation Analysis (0)] |
| 19. | Silva SS, Rowntree RK, Mekhoubad S, Lee JT. X-chromosome inactivation and epigenetic fluidity in human embryonic stem cells. Proc Natl Acad Sci USA. 2008;105:4820-4825. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 226] [Cited by in RCA: 225] [Article Influence: 13.2] [Reference Citation Analysis (0)] |
| 20. | Shen Y, Matsuno Y, Fouse SD, Rao N, Root S, Xu R, Pellegrini M, Riggs AD, Fan G. X-inactivation in female human embryonic stem cells is in a nonrandom pattern and prone to epigenetic alterations. Proc Natl Acad Sci USA. 2008;105:4709-4714. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 159] [Cited by in RCA: 163] [Article Influence: 9.6] [Reference Citation Analysis (0)] |
| 21. | Schnerch A, Cerdan C, Bhatia M. Distinguishing between mouse and human pluripotent stem cell regulation: the best laid plans of mice and men. Stem Cells. 2010;28:419-430. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 54] [Cited by in RCA: 64] [Article Influence: 4.3] [Reference Citation Analysis (0)] |
| 22. | Lengner CJ, Gimelbrant AA, Erwin JA, Cheng AW, Guenther MG, Welstead GG, Alagappan R, Frampton GM, Xu P, Muffat J. Derivation of pre-X inactivation human embryonic stem cells under physiological oxygen concentrations. Cell. 2010;141:872-883. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 298] [Cited by in RCA: 310] [Article Influence: 20.7] [Reference Citation Analysis (0)] |
| 23. | van den Berg IM, Galjaard RJ, Laven JS, van Doorninck JH. XCI in preimplantation mouse and human embryos: first there is remodelling…. Hum Genet. 2011;130:203-215. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 22] [Cited by in RCA: 25] [Article Influence: 1.8] [Reference Citation Analysis (0)] |
| 24. | Bruck T, Benvenisty N. Meta-analysis of the heterogeneity of X chromosome inactivation in human pluripotent stem cells. Stem Cell Res. 2011;6:187-193. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 55] [Cited by in RCA: 60] [Article Influence: 4.0] [Reference Citation Analysis (0)] |
| 25. | Hoffman LM, Hall L, Batten JL, Young H, Pardasani D, Baetge EE, Lawrence J, Carpenter MK. X-inactivation status varies in human embryonic stem cell lines. Stem Cells. 2005;23:1468-1478. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 96] [Cited by in RCA: 100] [Article Influence: 5.0] [Reference Citation Analysis (0)] |
| 26. | Dvash T, Lavon N, Fan G. Variations of X chromosome inactivation occur in early passages of female human embryonic stem cells. PLoS One. 2010;5:e11330. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 47] [Cited by in RCA: 49] [Article Influence: 3.3] [Reference Citation Analysis (0)] |
| 27. | Takahashi K, Yamanaka S. Induction of pluripotent stem cells from mouse embryonic and adult fibroblast cultures by defined factors. Cell. 2006;126:663-676. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 17989] [Cited by in RCA: 18178] [Article Influence: 956.7] [Reference Citation Analysis (0)] |
| 28. | Takahashi K, Tanabe K, Ohnuki M, Narita M, Ichisaka T, Tomoda K, Yamanaka S. Induction of pluripotent stem cells from adult human fibroblasts by defined factors. Cell. 2007;131:861-872. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 14327] [Cited by in RCA: 14305] [Article Influence: 841.5] [Reference Citation Analysis (0)] |
| 29. | Brons IG, Smithers LE, Trotter MW, Rugg-Gunn P, Sun B, Chuva de Sousa Lopes SM, Howlett SK, Clarkson A, Ahrlund-Richter L, Pedersen RA. Derivation of pluripotent epiblast stem cells from mammalian embryos. Nature. 2007;448:191-195. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 1711] [Cited by in RCA: 1518] [Article Influence: 84.3] [Reference Citation Analysis (0)] |
| 30. | Tesar PJ, Chenoweth JG, Brook FA, Davies TJ, Evans EP, Mack DL, Gardner RL, McKay RD. New cell lines from mouse epiblast share defining features with human embryonic stem cells. Nature. 2007;448:196-199. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 1687] [Cited by in RCA: 1637] [Article Influence: 90.9] [Reference Citation Analysis (0)] |
| 31. | Nichols J, Smith A. Naive and primed pluripotent states. Cell Stem Cell. 2009;4:487-492. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 1599] [Cited by in RCA: 1377] [Article Influence: 86.1] [Reference Citation Analysis (0)] |
| 32. | Bao S, Tang F, Li X, Hayashi K, Gillich A, Lao K, Surani MA. Epigenetic reversion of post-implantation epiblast to pluripotent embryonic stem cells. Nature. 2009;461:1292-1295. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 314] [Cited by in RCA: 298] [Article Influence: 18.6] [Reference Citation Analysis (0)] |
| 33. | Ying QL, Wray J, Nichols J, Batlle-Morera L, Doble B, Woodgett J, Cohen P, Smith A. The ground state of embryonic stem cell self-renewal. Nature. 2008;453:519-523. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 2986] [Cited by in RCA: 2653] [Article Influence: 156.1] [Reference Citation Analysis (0)] |
| 34. | Kunath T, Saba-El-Leil MK, Almousailleakh M, Wray J, Meloche S, Smith A. FGF stimulation of the Erk1/2 signalling cascade triggers transition of pluripotent embryonic stem cells from self-renewal to lineage commitment. Development. 2007;134:2895-2902. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 653] [Cited by in RCA: 592] [Article Influence: 32.9] [Reference Citation Analysis (0)] |
| 35. | Bain J, Plater L, Elliott M, Shpiro N, Hastie CJ, McLauchlan H, Klevernic I, Arthur JS, Alessi DR, Cohen P. The selectivity of protein kinase inhibitors: a further update. Biochem J. 2007;408:297-315. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 2164] [Cited by in RCA: 2127] [Article Influence: 118.2] [Reference Citation Analysis (0)] |
| 36. | Meek S, Wei J, Sutherland L, Nilges B, Buehr M, Tomlinson SR, Thomson AJ, Burdon T. Tuning of β-catenin activity is required to stabilize self-renewal of rat embryonic stem cells. Stem Cells. 2013;31:2104-2115. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 34] [Cited by in RCA: 38] [Article Influence: 3.5] [Reference Citation Analysis (0)] |
| 37. | Hirabayashi M, Goto T, Tamura C, Sanbo M, Hara H, Kato-Itoh M, Sato H, Kobayashi T, Nakauchi H, Hochi S. Derivation of embryonic stem cell lines from parthenogenetically developing rat blastocysts. Stem Cells Dev. 2014;23:107-114. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 5] [Cited by in RCA: 6] [Article Influence: 0.5] [Reference Citation Analysis (0)] |
| 38. | Telugu BP, Ezashi T, Roberts RM. Porcine induced pluripotent stem cells analogous to naïve and primed embryonic stem cells of the mouse. Int J Dev Biol. 2010;54:1703-1711. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 86] [Cited by in RCA: 81] [Article Influence: 5.8] [Reference Citation Analysis (0)] |
| 39. | Telugu BP, Ezashi T, Sinha S, Alexenko AP, Spate L, Prather RS, Roberts RM. Leukemia inhibitory factor (LIF)-dependent, pluripotent stem cells established from inner cell mass of porcine embryos. J Biol Chem. 2011;286:28948-28953. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 86] [Cited by in RCA: 79] [Article Influence: 5.6] [Reference Citation Analysis (0)] |
| 40. | Haraguchi S, Kikuchi K, Nakai M, Tokunaga T. Establishment of self-renewing porcine embryonic stem cell-like cells by signal inhibition. J Reprod Dev. 2012;58:707-716. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 35] [Cited by in RCA: 40] [Article Influence: 3.1] [Reference Citation Analysis (0)] |
| 41. | Gao Y, Guo Y, Duan A, Cheng D, Zhang S, Wang H. Optimization of culture conditions for maintaining porcine induced pluripotent stem cells. DNA Cell Biol. 2014;33:1-11. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 21] [Cited by in RCA: 22] [Article Influence: 1.8] [Reference Citation Analysis (0)] |
| 42. | Hall VJ, Hyttel P. Breaking down pluripotency in the porcine embryo reveals both a premature and reticent stem cell state in the inner cell mass and unique expression profiles of the naive and primed stem cell states. Stem Cells Dev. 2014;23:2030-2045. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 32] [Cited by in RCA: 32] [Article Influence: 2.9] [Reference Citation Analysis (0)] |
| 43. | Petkov S, Hyttel P, Niemann H. The small molecule inhibitors PD0325091 and CHIR99021 reduce expression of pluripotency-related genes in putative porcine induced pluripotent stem cells. Cell Reprogram. 2014;16:235-240. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 17] [Cited by in RCA: 18] [Article Influence: 1.6] [Reference Citation Analysis (0)] |
| 44. | Li W, Wei W, Zhu S, Zhu J, Shi Y, Lin T, Hao E, Hayek A, Deng H, Ding S. Generation of rat and human induced pluripotent stem cells by combining genetic reprogramming and chemical inhibitors. Cell Stem Cell. 2009;4:16-19. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 432] [Cited by in RCA: 413] [Article Influence: 24.3] [Reference Citation Analysis (0)] |
| 45. | David L, Samavarchi-Tehrani P, Golipour A, Wrana JL. Looking into the black box: insights into the mechanisms of somatic cell reprogramming. Genes (Basel). 2011;2:81-106. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 4] [Cited by in RCA: 5] [Article Influence: 0.4] [Reference Citation Analysis (0)] |
| 46. | Buecker C, Chen HH, Polo JM, Daheron L, Bu L, Barakat TS, Okwieka P, Porter A, Gribnau J, Hochedlinger K. A murine ESC-like state facilitates transgenesis and homologous recombination in human pluripotent stem cells. Cell Stem Cell. 2010;6:535-546. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 181] [Cited by in RCA: 167] [Article Influence: 11.1] [Reference Citation Analysis (0)] |
| 47. | Hanna J, Cheng AW, Saha K, Kim J, Lengner CJ, Soldner F, Cassady JP, Muffat J, Carey BW, Jaenisch R. Human embryonic stem cells with biological and epigenetic characteristics similar to those of mouse ESCs. Proc Natl Acad Sci USA. 2010;107:9222-9227. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 649] [Cited by in RCA: 645] [Article Influence: 43.0] [Reference Citation Analysis (0)] |
| 48. | Wang W, Yang J, Liu H, Lu D, Chen X, Zenonos Z, Campos LS, Rad R, Guo G, Zhang S. Rapid and efficient reprogramming of somatic cells to induced pluripotent stem cells by retinoic acid receptor gamma and liver receptor homolog 1. Proc Natl Acad Sci USA. 2011;108:18283-18288. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 208] [Cited by in RCA: 204] [Article Influence: 14.6] [Reference Citation Analysis (0)] |
| 49. | Gafni O, Weinberger L, Mansour AA, Manor YS, Chomsky E, Ben-Yosef D, Kalma Y, Viukov S, Maza I, Zviran A. Derivation of novel human ground state naive pluripotent stem cells. Nature. 2013;504:282-286. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 764] [Cited by in RCA: 834] [Article Influence: 69.5] [Reference Citation Analysis (0)] |
| 50. | Chan YS, Göke J, Ng JH, Lu X, Gonzales KA, Tan CP, Tng WQ, Hong ZZ, Lim YS, Ng HH. Induction of a human pluripotent state with distinct regulatory circuitry that resembles preimplantation epiblast. Cell Stem Cell. 2013;13:663-675. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 309] [Cited by in RCA: 317] [Article Influence: 28.8] [Reference Citation Analysis (1)] |
| 51. | Ware CB, Nelson AM, Mecham B, Hesson J, Zhou W, Jonlin EC, Jimenez-Caliani AJ, Deng X, Cavanaugh C, Cook S. Derivation of naive human embryonic stem cells. Proc Natl Acad Sci USA. 2014;111:4484-4489. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 355] [Cited by in RCA: 372] [Article Influence: 33.8] [Reference Citation Analysis (0)] |
| 52. | Valamehr B, Robinson M, Abujarour R, Rezner B, Vranceanu F, Le T, Medcalf A, Lee TT, Fitch M, Robbins D. Platform for induction and maintenance of transgene-free hiPSCs resembling ground state pluripotent stem cells. Stem Cell Reports. 2014;2:366-381. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 114] [Cited by in RCA: 141] [Article Influence: 12.8] [Reference Citation Analysis (0)] |
| 53. | Yan L, Yang M, Guo H, Yang L, Wu J, Li R, Liu P, Lian Y, Zheng X, Yan J. Single-cell RNA-Seq profiling of human preimplantation embryos and embryonic stem cells. Nat Struct Mol Biol. 2013;20:1131-1139. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 1095] [Cited by in RCA: 1230] [Article Influence: 102.5] [Reference Citation Analysis (0)] |
| 54. | Valamehr B, Abujarour R, Robinson M, Le T, Robbins D, Shoemaker D, Flynn P. A novel platform to enable the high-throughput derivation and characterization of feeder-free human iPSCs. Sci Rep. 2012;2:213. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 44] [Cited by in RCA: 60] [Article Influence: 4.6] [Reference Citation Analysis (0)] |
| 55. | Theunissen TW, Powell BE, Wang H, Mitalipova M, Faddah DA, Reddy J, Fan ZP, Maetzel D, Ganz K, Shi L. Systematic identification of culture conditions for induction and maintenance of naive human pluripotency. Cell Stem Cell. 2014;15:471-487. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 535] [Cited by in RCA: 619] [Article Influence: 56.3] [Reference Citation Analysis (0)] |
| 56. | Mekhoubad S, Bock C, de Boer AS, Kiskinis E, Meissner A, Eggan K. Erosion of dosage compensation impacts human iPSC disease modeling. Cell Stem Cell. 2012;10:595-609. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 251] [Cited by in RCA: 272] [Article Influence: 20.9] [Reference Citation Analysis (0)] |
| 57. | Niakan KK, Han J, Pedersen RA, Simon C, Pera RA. Human pre-implantation embryo development. Development. 2012;139:829-841. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 227] [Cited by in RCA: 259] [Article Influence: 19.9] [Reference Citation Analysis (0)] |
| 58. | Dietrich JE, Hiiragi T. Stochastic patterning in the mouse pre-implantation embryo. Development. 2007;134:4219-4231. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 385] [Cited by in RCA: 395] [Article Influence: 21.9] [Reference Citation Analysis (0)] |
| 59. | Chen AE, Egli D, Niakan K, Deng J, Akutsu H, Yamaki M, Cowan C, Fitz-Gerald C, Zhang K, Melton DA. Optimal timing of inner cell mass isolation increases the efficiency of human embryonic stem cell derivation and allows generation of sibling cell lines. Cell Stem Cell. 2009;4:103-106. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 158] [Cited by in RCA: 150] [Article Influence: 9.4] [Reference Citation Analysis (0)] |
| 60. | Xue Z, Huang K, Cai C, Cai L, Jiang CY, Feng Y, Liu Z, Zeng Q, Cheng L, Sun YE. Genetic programs in human and mouse early embryos revealed by single-cell RNA sequencing. Nature. 2013;500:593-597. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 687] [Cited by in RCA: 733] [Article Influence: 61.1] [Reference Citation Analysis (0)] |
| 61. | Wang J, Xie G, Singh M, Ghanbarian AT, Raskó T, Szvetnik A, Cai H, Besser D, Prigione A, Fuchs NV. Primate-specific endogenous retrovirus-driven transcription defines naive-like stem cells. Nature. 2014;516:405-409. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 292] [Cited by in RCA: 324] [Article Influence: 29.5] [Reference Citation Analysis (0)] |