Published online Jul 26, 2019. doi: 10.4252/wjsc.v11.i7.398
Peer-review started: February 22, 2019
First decision: June 4, 2019
Revised: June 18, 2019
Accepted: June 27, 2019
Article in press: June 27, 2019
Published online: July 26, 2019
Processing time: 155 Days and 19.6 Hours
In recent decades, cancer stem cells (CSCs) have been increasingly identified in many malignancies. CSC-related signaling pathways and their functions provide new strategies for treating cancer. The aberrant activation of related signaling pathways (e.g., Wnt, Notch, and Hedgehog pathways) has been linked to multiple types of malignant tumors, which makes these pathways attractive targets for cancer therapy. CSCs display many characteristic features, such as self-renewal, differentiation, high tumorigenicity, and drug resistance. Therefore, there is an urgent need to develop new therapeutic strategies to target these pathways to control stem cell replication, survival, and differentiation. Notable crosstalk occurs among different signaling pathways and potentially leads to compensatory escape. Therefore, multitarget inhibitors will be one of the main methods to overcome the drug resistance of CSCs. Many small molecule inhibitors of components of signaling pathways in CSCs have entered clinical trials, and some inhibitors, such as vismodegib, sonidegib, and glasdegib, have been approved. Tumor cells are susceptible to sonidegib and vismodegib resistance due to mutations in the Smo protein. The signal transducers and activators of transcription 3 (STAT3) inhibitor BBI608 is being evaluated in a phase III trial for a variety of cancers. Structural derivatives of BBI608 are the main focus of STAT3 inhibitor development, which is another strategy for CSC therapy. In addition to the potential pharmacological inhibitors targeting CSC-related signaling pathways, other methods of targeting CSCs are available, such as nano-drug delivery systems, mitochondrion targeting, autophagy, hyperthermia, immunotherapy, and CSC microenvironment targeting. In addition, we summarize the latest advances in the clinical development of agents targeting CSC-related signaling pathways and other methods of targeting CSCs.
Core tip: The review aims to introduce the field of cancer stem cells (CSCs) and the important signaling pathways in CSCs and present approved inhibitors as well as candidate drugs. Due to the complexity of the crosstalk among various signaling pathways, current strategies involve the development of multitarget inhibitors, combination therapy, and precision treatments based on the genetic characteristics of patients. Other methods of targeting CSCs are introduced as well, including nano-drug delivery systems, mitochondrion targeting, hyperthermia, immunotherapy, and CSC microenvironment targeting. However, this field remains in its infancy, and considerable research will be required to produce mature products that can contribute to curing cancer.
- Citation: Du FY, Zhou QF, Sun WJ, Chen GL. Targeting cancer stem cells in drug discovery: Current state and future perspectives. World J Stem Cells 2019; 11(7): 398-420
- URL: https://www.wjgnet.com/1948-0210/full/v11/i7/398.htm
- DOI: https://dx.doi.org/10.4252/wjsc.v11.i7.398
The concept of cancer stem cells (CSCs) was first proposed in 1983[1], and CSC self-renewal was posited as the core of tumor growth. Therefore, irreversible inactivation of stem cells is the key to killing tumor cells. In 1994, Lapidot et al[2] identified and isolated human acute myeloid leukemia (AML) cells expressing the stem cell surface marker phenotype CD34+/CD38-[3]. Cell transplantation experiments showed that compared with common tumor cells, CD34+/CD38- cells were tumorigenic and the surface antigens of these cells were similar to those of normal hematopoietic stem cells. In 2006, the American Association for Cancer Research clearly defined CSCs as a reservoir of self-sustaining cells with the exclusive ability to self-renew and maintain tumor growth[4].
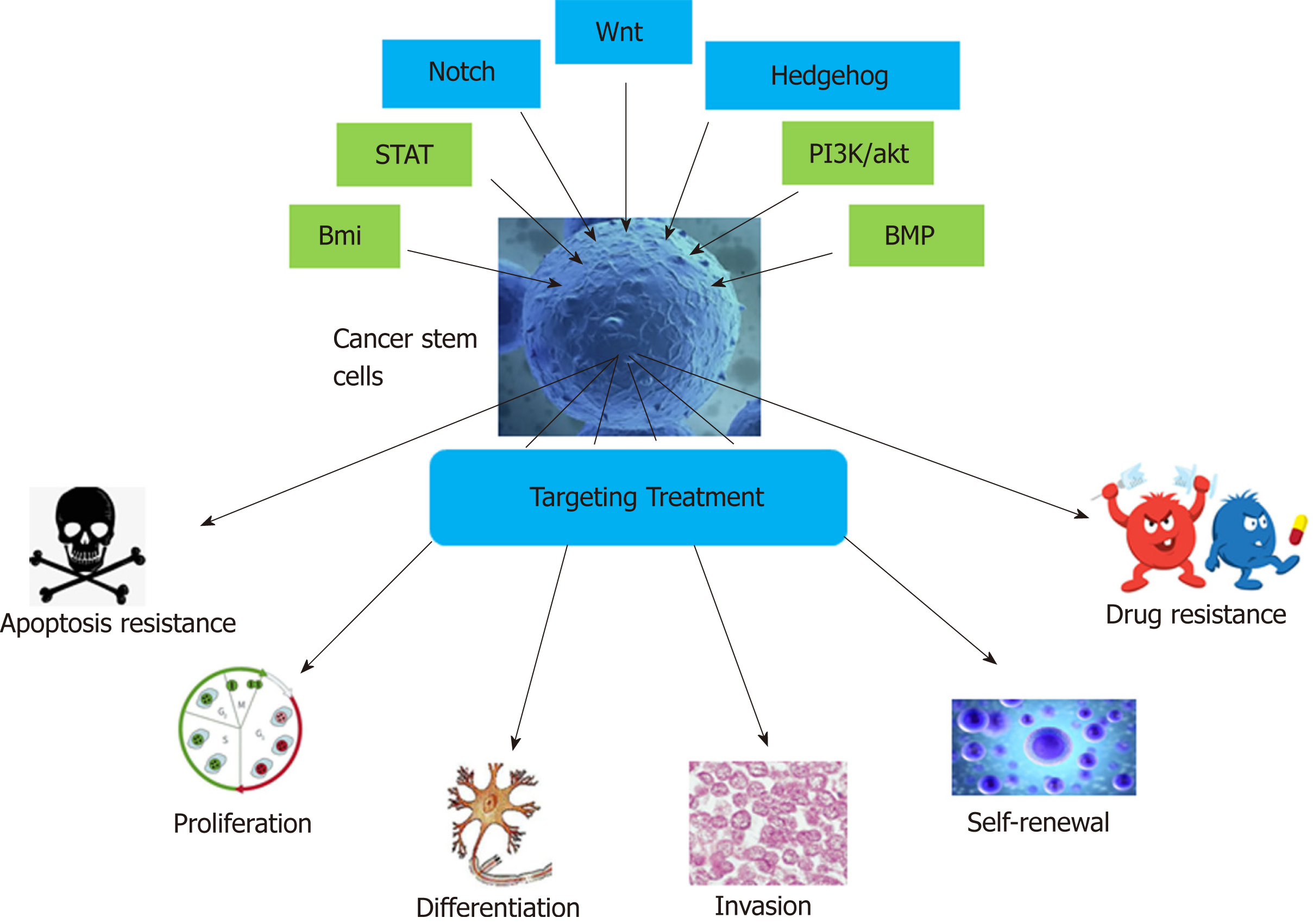
In recent years, the existence of CSCs in solid tumors, including breast cancer[5,6], central nervous system tumors[7,8], prostate cancer[9,10], pancreatic cancer[11], liver cancer[12], lung cancer[13], colon cancer[14,15], melanoma[16,17], and nasopharyngeal carcinoma[18], has been confirmed, and the CSC model has been successfully established. CSCs generally have the following features (Figure 1): (1) Unlimited self-renewal[19]: CSCs are able to produce progeny cells that are identical to the parental cells and maintain continuous tumors through self-renewal; (2) Differentiation potential[20]: CSCs can produce different lineages of differentiated progeny tumor cells; (3) High tumorigenicity[5]: A small number of CSCs cultured in vitro form colonies, and few CSCs are required to form tumors in vivo upon injection into experimental animals; this property is not shared by general tumor cells; and (4) Drug resistance[21]. The main factors for the development of drug resistance are as follows. First, most CSCs are in a resting or dormant state and are not undergoing cell division[22]. Second, CSCs mostly express ATP-binding cassette family membrane transporters[23], which are responsible for the transport and efflux of metabolites, drugs, toxic substances, endogenous lipids, peptides, nucleotides, and sterols and can render these cells resistant to many chemotherapeutic drugs. In addition, CSCs have a powerful DNA repair ability[24]. Finally, the abnormal expression of signaling pathway components and the diversification of the CSC microenvironment are also related to drug resistance. At present, the presence of CSCs is believed to be the main cause of chemotherapy and radiotherapy failure[25].
Currently, researchers have not only confirmed the existence of CSCs but also identified the specific surface markers of many types of CSCs[26,27] and their signal transduction pathways. Many chemical agents of different classes targeting the Wnt, Notch, Hedgehog, signal transducers and activators of transcription (STAT), bone morphogenetic protein (BMP), Bmi, and PI3K/Akt pathways have entered clinical trials (Figure 1). In this review, the approval status and progress of these investigational agents are summarized. In addition to the potential pharmacological inhibitors targeting CSC-related signaling pathways, other methods of targeting CSCs, such as nano-drug delivery systems (NDDSs), mitochondrion targeting, autophagy, hyperthermia, immunotherapy, and CSC microenvironment targeting, are also summarized.
There are two main theories about the possible formation of CSCs: From normal stem cells and from non-stem cells. Studies have shown that CSCs are formed by the transformation of adult stem cells caused by genetic mutations. Normal stem cells have activated self-renewal mechanisms, have longer survival time, and can accumulate more mutations; thus, they have more opportunities to mutate into CSCs[28-30]. Therefore, we hypothesize that gene mutations in normal adult stem cells are caused by endogenous or exogenous stimuli, and then they enter the cell cycle, rapidly divide, and transform into CSCs. Moreover, some differentiated cells may also regain self-renewal capacity before canceration and mutate into CSCs[31,32]. Because CSCs are derived from normal cells, their signaling pathways are similar to those of normal cells. The main pathways affecting CSCs include the Wnt, Hedgehog, Notch, BMP, Bmi, PI3K/Akt, and STAT pathways[33], which regulate CSC self-renewal and differentiation. Among them, the Wnt, Hedgehog, and Notch pathways are the most thoroughly studied[34].
The Wnt signaling pathway regulates cell proliferation, differentiation, and apoptosis and cell-cell interactions and plays an important role in processes involved in embryogenesis and tissue repair[35,36]. Extracellular Wnt protein can trigger different intracellular signal transduction pathways, which are classified as Wnt/β-catenin dependent (canonical pathway) or β-catenin independent (noncanonical pathway). The canonical pathway is activated by the binding of Wnt ligands to the low-density lipoprotein receptor (LRP)-5/6 receptor and the Frizzled (Fzd) receptor. Subsequently, this complex activates the cytoplasmic protein disheveled (Dvl), resulting in the recruitment of protein complexes (axin, GSK-3β, casein kinase 1, and adenomatosis polyposis coli protein) to the receptor[37-39]. The Wnt-Fzd-axin-LRP-5/6 complex sequesters cytosolic GSK-3β, blocking it from phosphorylating β-catenin. As a result, unphosphorylated β-catenin accumulates in the cytoplasm and migrates to the nucleus, thus leading to the transcription of target genes such as c-Myc and cyclin D1[39], which promote the abnormal proliferation of tumor cells.
Examples of the β-catenin-independent pathway include the Wnt/Ca2+ pathway and the planar cell polarity (PCP) pathway[40]. In the Wnt/Ca2+ pathway, Wnt5a and Wnt11 bind to and activate the Fzd receptor to activate Dvl, which in turn inhibits cGMP-dependent protein kinase and activates phospholipase C (PLC), resulting in increased Ca2+ release; moreover, PLC promotes the accumulation of Ca2+ through the generation of inositol 1,4,5-trisphosphate (IP3). Increased Ca2+ levels activate the protein kinases calmodulin-dependent protein kinase II (CaMK-II), protein kinase C, and calcineurin[41]. CaMK-II can phosphorylate T-cell factor (TCF) via transcriptional growth factor β-activated kinase 1 and NEMO kinase, and phosphorylated TCF loses the ability to bind β-catenin. Therefore, this pathway antagonizes the Wnt/β-catenin-dependent pathway[42]. The PCP pathway can be initiated by the Wnt interaction with Fzd receptors or the coreceptors receptor related to tyrosine kinase and retinoic acid-related orphan receptor, which leads to Dvl activation[43]. Myosin and Rho-associated kinase are activated by Rho GTPase and alter actin activity and the cytoskeletal arrangement. Tandem activation of Rac GTPase and Rac stimulates c-Jun N-terminal kinase (JNK) activity[44]. In addition to its regulation of cell proliferation, JNK is involved in a series of physiological processes, such as cell growth, differentiation, migration, and cancer[45].
The canonical Wnt signaling pathway may be involved in the development of malignant breast cancer, which is manifested by elevated β-catenin levels in the nucleus and cytoplasm. In breast cancer patients, elevated β-catenin often indicates a poor prognosis, which may be due to β-catenin mutations[46]. Cyclin Y (CCNY) promotes the proliferation of ovarian cancer cells through the Wnt/β-catenin signaling pathway. Overexpression of CCNY enhances the expression of the Wnt downstream target genes c-Myc, cyclin D1, PFTK1, and N-acetylglucosamine transferase[47]. Downregulation of tumor necrosis factor α-induced protein 2 can inhibit the proliferation of esophageal squamous cell carcinoma by downregulating the downstream target genes of β-catenin (e.g., c-Myc and cyclin D1) and upregulating E-cadherin and p-GSK-3β levels[48]. Silencing of the Golgi phosphoprotein 3 (GOLPH3) gene can inhibit the proliferation of SW620 colon cancer cells, in which β-catenin is downregulated and Wnt pathway activity is decreased. Conversely, overexpression of the GOLPH3 gene promotes colon cancer cell proliferation[49]. Reishi inhibits the proliferation and migration of breast cancer cells by inhibiting the Wnt signaling pathway, and the main mechanism of action is to block the Wnt pathway by inhibiting the phosphorylation of the Wnt coreceptor low-density lipoprotein receptor-related protein 6 (LRP6). Reishi can significantly reduce LRP6 phospho-rylation and inhibit the Wnt3a-mediated expression of the target gene Axin2 in human and mouse breast cancer cell lines[50]. Breast cancer, melanoma, prostate cancer, lung cancer, and other cancers can be generated by altering the expression of the CTNNB1 gene, which encodes β-catenin.
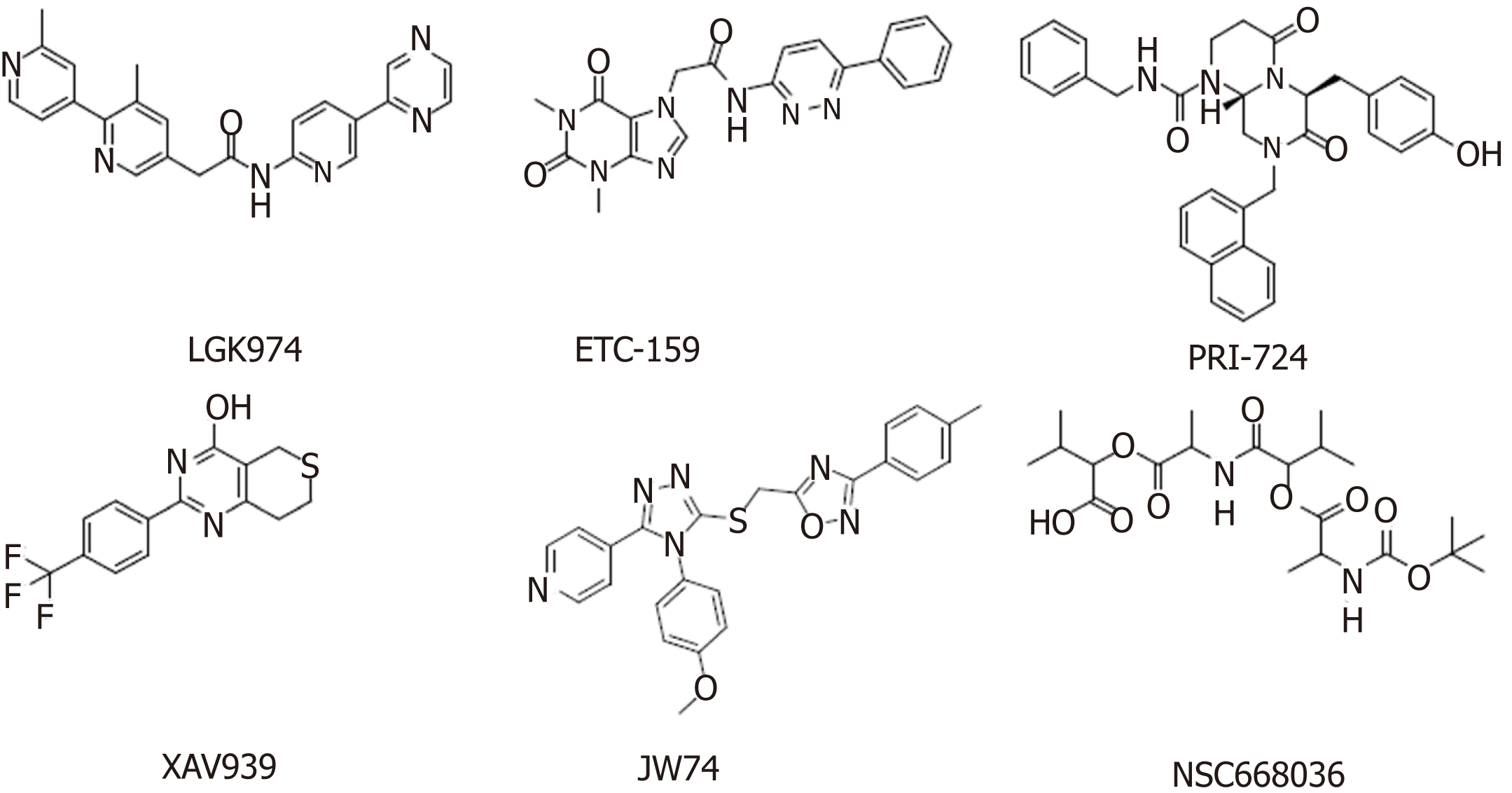
The increased expression of Wnt ligands, such as Wnt1, Wnt2, and Wnt7a, leads to glioblastoma, esophageal cancer, and ovarian cancer, respectively[51]. Deregulation of the Wnt signaling pathway has been found to be associated with many cancers, making it an attractive target for anticancer therapies. The developed Wnt signaling pathway inhibitors are mainly monoclonal antibodies, β-catenin inhibitors, and various small molecule inhibitors. The discovery of small molecule inhibitors has been a research focus, with the main goal of finding small molecules that affect nuclear β-catenin and Wnt signaling pathways through high-throughput screening of small molecule compound libraries. Agents targeting the Wnt signaling pathway that are in clinical trials are summarized below (Table 1, Figure 2).
| Compound | Target/mechanism | Tumor type | Highest phase | Organization |
| LGK974[53] | Porcupine inhibitor | Melanoma; breast cancer; pancreatic cancer | Phase I | Novartis |
| ETC-159[54] | Porcupine inhibitor | Solid tumors | Phase I | ETC/Duke-NUS |
| PRI-724[55] | β-catenin/CBP | Myeloid leukemia | Phase I/II | Prism/Eisai Pharmaceuticals |
| XAV939[59] | Tankyrase 1 and 2 inhibitor | - | Preclinical | Novartis |
| IWR1[61] | Tankyrase 1 and 2 inhibitor | - | Preclinical | Tocris Bioscience |
| JW74[70] | Tankyrase 1 and 2 inhibitor | - | Preclinical | Tocris Bioscience |
| NSC668036[71] | Disheveled | - | Preclinical | Tocris Bioscience |
| OMP-18R5[64] (Vantictumab) | Frizzled receptor | Solid tumors; breast cancer; non-small cell lung cancer; pancreatic cancer | Phase I/Ib | OncoMed Pharmaceuticals |
| OMP-54F28[65] (Ipafricept) | Fzd8-Fc fusion protein | Solid tumors; hepatocellular carcinoma; ovarian cancer; pancreatic cancer | Phase I/Ib | OncoMed Pharmaceuticals |
Porcupine is an acyltransferase that plays an important role in the formation and secretion of Wnt ligands[52]. Among the currently reported small molecule inhibitors of porcupine, LGK974[53] and ETC-159[54] have successfully entered phase I clinical trials. Studies have shown that LGK974 can inhibit Wnt signaling by reducing LRP6 phosphorylation and Wnt target gene expression both in vivo and in vitro, and LGK974 has significant effects in both tumor models and human cancer cell lines in mice. However, ETC-159, the first small molecule for the treatment of colorectal cancer, was subsequently discovered[54]. PRI-724 is a potent inhibitor of the Wnt signaling pathway that inhibits the binding and recruitment of β-catenin and its coactivator CBP[55]. PRI-724 specifically binds to CBP, disrupting the interaction between CBP and β-catenin. PRI-724 selectively induces apoptosis and inhibits the growth of colon cancer cell lines in vitro but has no effect on normal colonic epithelial cells[56,57].
Tankyrase belongs to the poly(ADP-ribose) polymerase (PARP) family and has two isoforms, tankyrase 1 (PARP5a) and tankyrase 2 (PARP5b), which are associated with Wnt/β-catenin signaling. Both of these isoforms increase axin degradation through the ubiquitin-proteasome pathways[58]. XAV939 specifically inhibits tankyrase PARP activity[59]. Treatment with 1.0 μM XAV939 for 12 h reduced DNA-PKcs protein levels to a minimum level, and the relative expression was less than 25% of that in the control group[60]. IWR-1 and XAV939 act as reversible inhibitors of the Wnt pathway and have similar pharmacological effects in vivo and in vitro. However, IWR-1 functions through interacting with axin and XAV939 directly binds to TNKS[61].
Monoclonal antibodies against Wnt-1 and Wnt-2 have proven that Wnt inhibition leads to tumor suppression in melanoma, sarcoma, colorectal cancer, non-small cell lung cancer (NSCLC), and mesothelioma[62,63]. OMP-18R5 (vantictumab), developed by OncoMed Pharmaceuticals/Bayer, is a monoclonal antibody that targets five of the ten Fzd receptors. The safety and efficacy of these antibody therapeutics are being assessed separately or in combination with chemotherapy in NSCLC, pancreatic cancer, and breast cancer[64]. The first-in-class recombinant fusion protein ipafricept (OMP-54F28) blocks Wnt signaling by binding to Wnt ligands[65]. Among this class of therapeutics, for example, vantictumab[66], ipafricept[67], and ETC-159[68], which show anti-CSC effects in preclinical model experiments, are in clinical trials to treat cancer patients[69].
The Notch signaling pathway is widespread and highly conserved in mammals, and it has various functions and a relatively simple molecular structure[72-75]. The Notch pathway is mainly composed of Notch receptors (Notch 1-4) and Notch ligands (Jagged 1, Jagged 2, Delta-like ligand (DLL)-1, DLL-3, and DLL-4). The DNA binding protein CSL [CBF1/RBP, Su(H)/Lag-1] is the primary effector of the Notch signaling pathway. CSL binds to activated Notch to form the Notch intracellular domain (NICD)-CSL complex.
The Notch signaling pathway is divided into the non-CSL-dependent pathway (noncanonical Notch signaling pathway) and the CSL-dependent pathway (canonical Notch signaling pathway)[76], and these pathways maintain the existence of stem cells and initiate embryonic or fetal cell differentiation[77]. The Notch pathway affects the normal growth and development of cells and tissues by regulating cell differentiation, proliferation, and apoptosis, which play core roles in the development of malignant tumors[78,79].
The canonical Notch signaling pathway involves the binding of receptor and ligand, which releases the NICD via metalloproteinase- and γ-secretase-mediated protease hydrolysis. Then, the NICD enters the nucleus and forms a complex with RBPJ, which mediates the transcription and expression of its downstream target genes Hes and Hey, thus affecting cell differentiation, proliferation, and apoptosis. During the activation and transduction of the Notch signaling pathway, enzymatic cleavage by γ-secretase plays an extremely important role. The inhibition of γ-secretase prevents the release of NICD, thereby blocking the Notch signaling pathway[80]. Treatment strategies directly related to the Notch pathway involve γ-secretase inhibitors (GSIs) and inhibitors of Notch receptors, ligands, or other pathway elements.
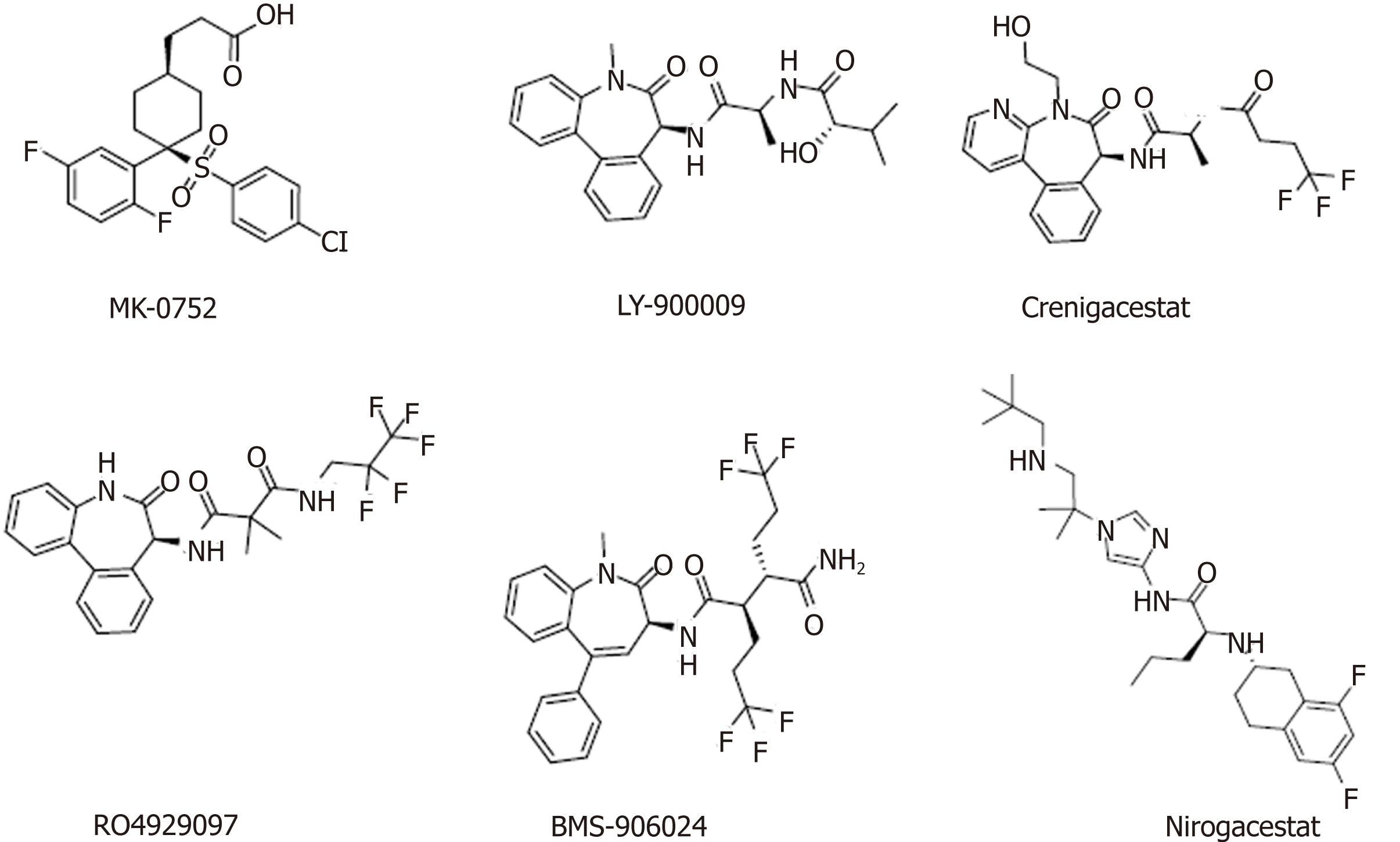
Currently, several classes of Notch signaling inhibitors are being clinically researched, and they have significant differences in targets, mechanisms, and tumor types (Table 2, Figure 3). A major class of agents targeting the Notch pathway is GSIs, which are the first Notch inhibitors to enter clinical studies[81]. Favorable tissue penetration, low cost, ease of administration, and potential pan-Notch inhibitory activity are the main advantages of GSIs[82,83]. However, the toxicity of these agents, especially the serious toxicity in the gastrointestinal tract, may affect the further development of this class of inhibitors[84]. Moreover, GSIs have a variety of substrates, such as Notch receptors, cadherin, ERBB4, CD44, and amyloid precursor protein, and some scholars believe that GSIs are not druggable[85]. RO4929097 showed insufficient activity as a single agent against metastatic melanoma and platinum-resistant ovarian cancer in clinical trials[86,87], which indicated that it has potential in combination with other drugs to treat related diseases[88,89].
| Compound | Target/mechanism | Tumor type | Highest phase | Organization |
| MK-0752 | γ-secretase inhibitor | Breast cancer | Phase I | Merck |
| LY-900009 | γ-secretase inhibitor | Metastatic cancer/lymphoma | Phase I | Perrigo and Lilly |
| Crenigacestat | Notch signaling inhibitor | T-cell acute lymphoblastic leukemia | Phase I/II | Lilly |
| CB-103 | Notch signaling inhibitor | Solid tumors; hematologic cancers | Phase I/II | Cellestia Biotech |
| LY-3056480 | Notch signaling inhibitor | Sensorineural hearing loss | Phase I/II | Lilly |
| RO4929097 | γ-secretase inhibitor | Kidney cancer; pancreatic cancer; metastatic cancer; prostate cancer; glioblastoma multiforme; metastatic melanoma | Phase II | SpringWorks Therapeutics |
| BMS-906024 | Notch signaling pathway | Adenoid cystic carcinoma | Phase II | Bristol-Myers Squibb |
| Nirogacestat | γ-secretase inhibitor | Desmoid tumors | Phase II | SpringWorks Therapeutics |
| Enoticumab | Anti-DLL4 | Advanced solid malignancies | Phase I | Regeneron and Sanofi |
Nirogacestat (PF-3084014) is another GSI in phase II clinical trials for the treatment of desmoid tumors. Long-term follow-up of patients with desmoid fibromatosis treated with nirogacestat showed that it had promising activity (objective response rate of 71.4%) at relatively low doses and high tolerability, thus leading to prolonged disease control[90,91]. Nirogacestat was also reported to show antitumor and antimetastatic effects in hepatocellular carcinoma[92]. Pretreatment of multiple myeloma (MM) CSCs with the γ-secretase inhibitor RO4929097, a Notch pathway inhibitor, can reverse bruceantin-induced effects on MM-CSC proliferation[93].
BMS-906024, a potent pan-Notch inhibitor, showed robust efficacy in vivo at tolerated doses in Notch-driven leukemia and solid tumors[94]. In one study, BMS-906024 enhanced the efficacy of paclitaxel in lung adenocarcinoma and improved the patient’s response to combination therapy[34]. Notably, whether a specific chemical class of GSIs is preferable with regard to safety and efficacy remains unclear, and the ongoing clinical and preclinical studies will provide valuable data.
DLL4 is the only Notch ligand specifically expressed in endothelial cells. The DLL4-mediated Notch pathway plays a vital role in regulating angiogenesis, especially in regulating embryonic vascular development and tumor angiogenesis[95,96]. The DLL4/Notch pathway in adult vasculature mainly inhibits the activity, migration, and differentiation of endothelial cells, thereby impeding angiogenesis and maintaining vascular stability[97,98]. Generally, vascular endothelial growth factor (VEGF) induces DLL4 production in endothelial cells, which promotes the activation of the Notch signaling pathway, downregulates VEGF3, and promotes endothelial cell sprouting[99]. Therefore, targeting DLL4 with monoclonal antibodies is another strategy to block Notch signaling, and this concept is being tested in the clinic. Enoticumab is a fully human IgG1 antibody to DLL4 in phase I clinical development for the treatment of advanced solid malignancies. Intravenous enoticumab was found to have promising clinical activity in phase I safety, dose-limiting toxicity, and pharmacokinetics studies in patients with solid tumors[100]. However, it should be noted that chronic treatment with anti-DLL4 monoclonal antibodies causes hemangiomas in animal models[101].
The Hedgehog signaling pathway plays a significant role in mammalian development, is involved in the regulation of cell proliferation, survival, and differentiation[102,103], and is responsible for the formation of the neural tube, axial bone, hair, and teeth[104-106]. The Hedgehog gene is inhibited in most adult tissues and involved in the maintenance and repair of only certain tissues. Abnormally activated Hedgehog signaling directly regulates various growth factors and promotes the proliferation and survival of tumor cells.
When the Hedgehog ligand (Hh) is absent, Patched (Ptch) inhibits the activation of Smoothened (Smo), glioma-associated oncogene homolog (Gli) is suppressed by a protein complex mainly composed of suppressor of fused (Sufu), and Gli is phosphorylated and unable to enter the nucleus. In the presence of Hh, Ptch is bound by Hh, which relieves Ptch-mediated Smo inhibition. Activated Smo promotes the dissociation of the Sufu and Gli complex, allowing Gli to enter the nucleus and trigger downstream target gene expression, which results in the activation of the canonical Hedgehog signaling pathway[107]. To date, the most successful drugs targeting the Hedgehog signaling pathway are the three Smo inhibitors vismodegib, sonidegib, and glasdegib, which have been approved by the Food and Drug Administration (FDA) for the treatment of basal cell carcinoma (BCC). Meanwhile, many other Smo inhibitors are being clinically researched and have therapeutic effects on a variety of tumors. In addition, Gli inhibitors and Shh ligand inhibitors are in the preliminary stage of research.
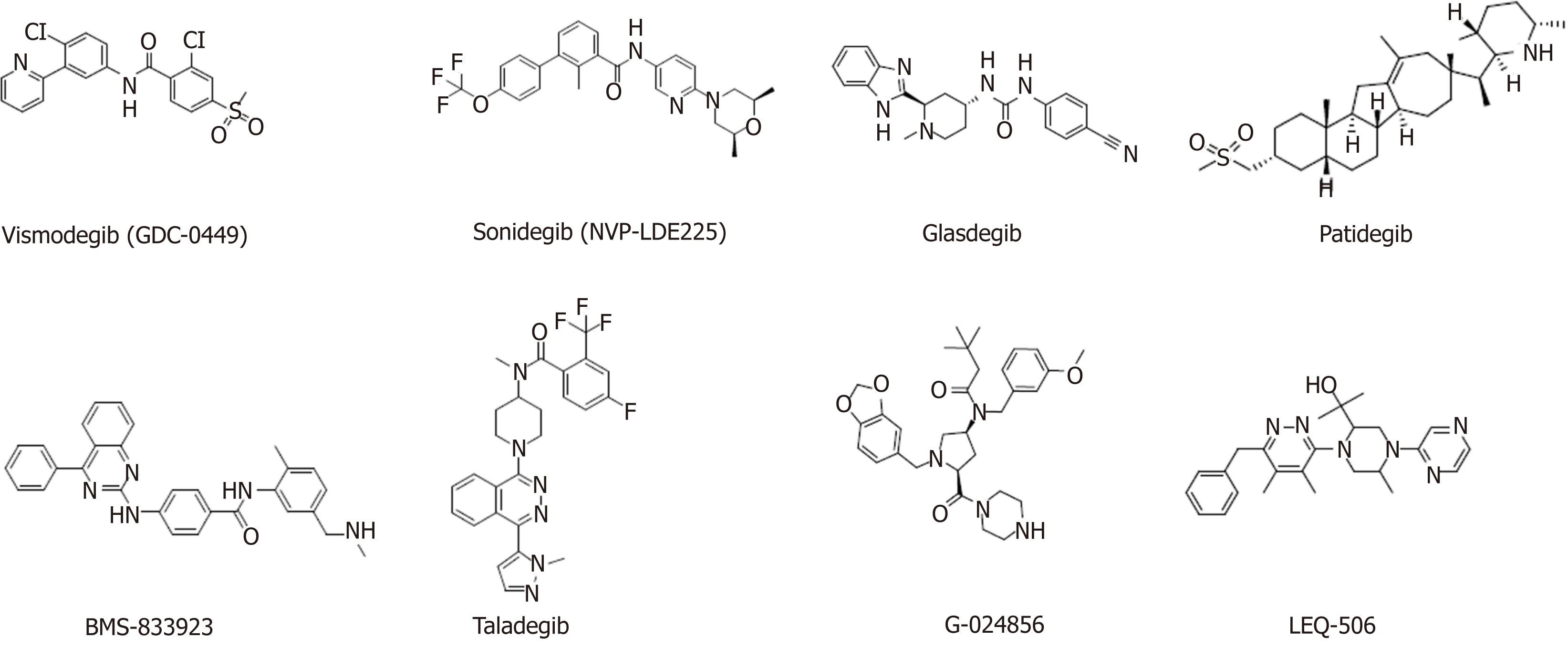
Smo inhibitors: Smo is a major target for Hedgehog signaling pathway inhibitors. Inhibiting Smo activity can directly block the activation of the downstream transcription factor Gli and thus inhibit the expression of genes downstream of the Hedgehog signaling pathway. However, Smo-specific inhibitors are prone to drug resistance. Currently, resistance to two launched Smo inhibitors has been observed, and ubiquitous toxic side effects limit their application. The following agents targeting the Smo protein are under development in clinical trials (Table 3, Figure 4).
| Compound | Target/mechanism | Cancer type | Highest phase | Organization |
| Vismodegib (GDC-0449) | Smo | Basal cell carcinoma | Launched | Roche |
| Sonidegib (NVP-LDE225) | Smo | Basal cell carcinoma | Launched | Novartis |
| Glasdegib | Smo | AML | Launched | Pfizer |
| Patidegib | Smo | Basal cell nevus syndrome; skin cancer | Phase II | PellePharm |
| BMS-833923 | Smo | Leukemia | Phase II | Bristol-Myers Squibb |
| Taladegib | Smo | Esophageal cancer; gastroesophageal junction cancer; solid tumors; small cell lung carcinoma | Phase I/II | Ignyta |
| G-024856 | Smo | Basal cell carcinoma | Phase I | Roche |
| LEQ-506 | Smo | Advanced solid tumors | Phase I | Novartis |
Applications for the approval of vismodegib, a first-in-class small molecule inhibitor of Hedgehog signaling, were filed by Genentech in the United States and the E.U. in 2011 for the treatment of advanced BCC. In 2012, vismodegib was approved by the FDA and launched. Phase II clinical development is ongoing for the treatment of breast cancer, gastrointestinal cancer, pancreatic cancer, chondrosarcoma, keratocystic odontogenic tumor, medulloblastoma, and meningioma. This drug is also in phase I/II clinical trials for the treatment of prostate cancer and sarcoma. Treatment of HCC38 cells, a triple-negative breast cancer (TNBC) stem cell line, with vismodegib significantly decreased TNBC cell proliferation, cell invasion, and mammosphere formation while inducing cell apoptosis by inhibiting the protein expression or phosphorylation of downstream signaling molecules. Tumor formation and growth of HCC1806 cells (another TNBC stem cell line) pretreated with vismodegib are effectively suppressed in xenograft mouse models. Treatment with vismodegib may provide a novel alternative therapeutic strategy against TNBC that targets breast CSCs (BCSCs), and it could provide promising insights for clinical applications in patients with TNBC[108].
A Smo mutation (D473H) was identified in tumor samples from a medulloblastoma patient who relapsed after an initial response to vismodegib. Acquired mutations in a serpentine receptor with features of a G protein–coupled receptor can lead to vismodegib resistance in human cancer[109]. Overall, vismodegib could represent an important treatment option for patients with advanced BCC.
Sonidegib, the second Smo antagonist developed by Novartis, was launched in the U.S. in 2015 for the treatment of advanced BCC. Phase II clinical trials are ongoing for the treatment of MM, and phase I/II clinical trials are underway for the treatment of pancreatic adenocarcinoma, myelofibrosis, and essential thrombocythemia. A variety of murine Smo mutations that lead to sonidegib resistance have been found in mouse models of medulloblastoma[110].
Glasdegib, developed by Pfizer, is a Hedgehog pathway inhibitor that was approved and launched in the U.S. in 2018 for the oral treatment of newly diagnosed AML. Pfizer is also conducting phase II clinical trials on the treatment of myelo-dysplastic syndrome and chronic myelomonocytic leukemia.
As single-target agents, the approved Smo inhibitors have common drug resistance problems. For example, Smo mutations in the tumor tissue of patients with medulloblastoma cause vismodegib to lose efficacy. A variety of drug-resistant Smo mutants were found in a mouse tumor model treated with sonidegib, which further indicates the limitations of sonidegib for cancer treatment. To overcome drug resistance and the side effects of traditional Smo inhibitors, novel Hedgehog signaling pathway inhibitors must be developed.
Gli inhibitors: The Gli transcription factor is the terminal component of the Hedgehog signaling pathway, and it is also regulated by other important signaling pathways. Activation of Gli1 and Gli2 promotes tumor growth, survival, angio-genesis, and drug resistance[111].
Arsenic trioxide (ATO) is an FDA-approved drug for the treatment of acute promyelocytic leukemia, and ATO was later discovered to be a Gli transcription factor inhibitor and entered as such into phase II clinical trials[112]. Studies have shown that ATO inhibits the Hedgehog signaling pathway by directly binding to Gli1 and Gli2[113]. GANT-61 is another Gli inhibitor developed by the U.S. National Cancer Institute that significantly inhibits DNA binding to Gli1 and Gli2[114]. GANT-61 can induce the apoptosis of AML cells, inhibit cell proliferation, and enhance the cytotoxic effect of cytarabine on cancer cells[115]. This drug is currently under preclinical study.
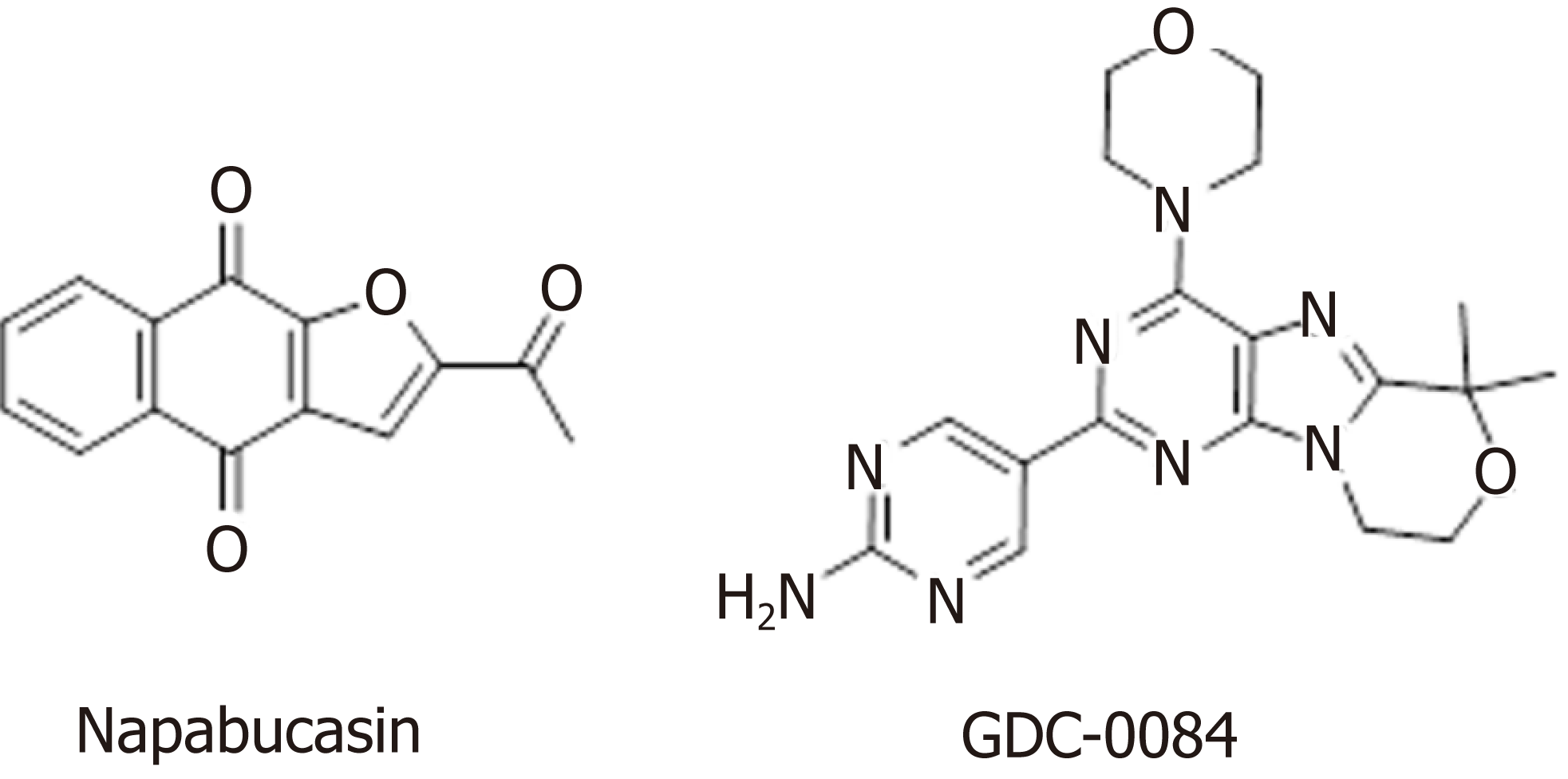
In addition to the well-studied Wnt, Hedgehog, and Notch signaling pathways, the BMP, Bmi, PI3K/Akt, and STAT signaling pathways also harbor abnormalities that can lead to tumor development. The STAT family is a group of cytoplasmic proteins that are activated by cytokine receptors, and they act as a transducer in the process of receptor-mediated cytokine signaling, and subsequently, extracellular signals are transmitted to the nucleus to ultimately regulate downstream gene transcription[116]. There are multiple subfamilies of the STAT family; for example, STAT3 signaling regulates the self-renewal, differentiation, and apoptosis of CSCs[117]. Napabucasin (BBI608) is a first-in-class STAT3 inhibitor in phase III clinical trials sponsored by Sumitomo Dainippon Pharma for the treatment of metastatic colorectal carcinoma and pancreatic cancer (Figure 5). In one study, BBI608 inhibited stemness gene expression, depleted CSCs, and overcame cisplatin resistance in NSCLC[118]. Napabucasin is effective in mice as a monotherapy or in combination with other agents.
In 2015, Li et al[119] demonstrated that BBI608 has strong anti-CSC effects in vitro and in vivo in a broad range of cancer types. Zhang et al[120] confirmed that BBI608 is able to kill prostate CSCs, inhibit CSC properties, and inhibit the expression of stemness-related genes. Currently, BBI608 is being assessed in clinical trials, and the results suggest that BBI608 is a potent anti-tumorigenic and anti-CSC drug in different tumor types[118,121-123]. To further improve the pharmacokinetic properties of BBI608 and its antitumor activity, our research team designed and synthesized a series of BBI608 derivatives that display stronger inhibitory activity than BBI608 in HepG2 cells[124].
B-cell-specific Moloney murine leukemia virus integration site 1 (Bmi-1) is a member of the PcG family and a nuclear chromatin regulatory factor. The encoded protein plays an important role in tumorigenesis[125]. The Bmi-1 gene is highly expressed in many malignant tumors and closely related to tumor invasion, metastasis, and recurrence. Decreased Bmi-1 gene expression in embryos and adults leads to defects in stem cell proliferation and self-renewal[126]. Bmi-1 is required for the self-renewal and maintenance of acute myelocytic leukemia stem cells[127]. PTC-596 is an oral Bmi-1 inhibitor in phase I clinical development for the treatment of female reproductive system cancer. Studies using mouse xenograft models demonstrated that PTC-596 inhibited leukemia cell growth in vivo while sparing normal hematopoietic cells[128].
BMP, an important member of the transforming growth factor-β (TGF-β) family, represents a class of secreted glycoproteins that induce ectopic bone and cartilage formation, and it is involved in many important biological processes, such as embryonic development, cell differentiation, and organ formation[129]. BMP-7 has been shown to reduce macrophage infiltration and tissue damage in animal models of acute and chronic renal failure[130]. BMP-7 was originally approved in 2001 for the treatment of the nonunion of long bone fractures secondary to trauma, and it has also been studied in phase II/III trials for the treatment of periodontal disease.
The PI3K/Akt signaling pathway is important in various types of cells, in which it regulates cell growth, proliferation, differentiation, apoptosis, and cytoskeletal rearrangement. Akt, a downstream molecule of PI3K, is a serine/threonine protein kinase. The PI3K/Akt signaling pathway is both a convergence and divergence point for many cell signals that regulate the phosphorylation of signaling molecules, and this pathway engages in crosstalk with the BMP/Smad and Wnt/β-catenin pathways, among others. Then, downstream signaling affects the expression of nuclear transcription factors, ultimately regulating cell proliferation, self-renewal, and multidirectional differentiation potential[130,131]. GDC-0084 (Figure 5), a small molecule inhibitor of the PI3K/AKT/mTOR pathway, is being studied in a phase II clinical trial for the treatment of glioblastoma multiforme (GBM). GDC-0084, which is distributed throughout the brain and intracranial tumors, potently inhibits the PI3K pathway[132]. In vitro and in vivo experiments showed that GDC-0084 can inhibit human cutaneous squamous cell carcinoma cell growth by blocking PI3K-Akt-mTOR and DNA-PKcs signaling[133].
ETC-159 has shown dose-limiting toxicities, including hyperbilirubinemia and skeletal fragility fractures[134]. The preliminary results of the clinical phase I trial are available, and they showed that PRI-724 results in tertiary reversible hyperbil-irubinemia with dose-limiting toxicity. An open phase I/II clinical study involving PRI-724 dose escalation in patients with advanced malignant myeloid blood diseases is still ongoing[51].
For enoticumab, the common severe adverse effects (AEs) are fatigue, headache, hypertension, and nausea[135]. However, six treatment-related serious adverse events were reported in four patients: Brain natriuretic peptide increase, troponin I increase, right ventricular dysfunction and pulmonary hypertension, and left ventricular dysfunction and pulmonary hypertension[135]. One patient had a dose-limiting toxicity of grade 3 vomiting and diarrhea[136]. Fragility fractures were reported at an ipafricept dose of 20 mg/kg. The hypophosphatemia and decrease in weight are grade III AEs associated with ipafricept treatment[137]. A dose-limiting toxicity of grade III mucosal inflammation was observed at the 30 mg dose level of LY-900009[138]. When patients were treated with crenigacestat, the most frequent related AEs included diarrhea, vomiting, nausea, decreased appetite, fatigue, asthenia, and hypophosphataemia[139]. In the phase I clinical trial, dose escalation of LY3056480 confirmed that trans-tympanic injection at the highest dose of 250 μg is safe and well tolerated and safety issues were not observed[140]. Patients with glioma were treated with RO-4929097 in combination with radiotherapy and temozolomide, and the results were positive, safe, and effective[141]. Treatment with BMS-906024 was found to be relatively well tolerated, with minimal diarrhea observed in the subjects[142].
Moreover, the AEs commonly observed in vismodegib-treated patients, including muscle spasms, ageusia/dysgeusia, alopecia, weight loss, and fatigue, lead to poor clinical outcomes because of the decreased quality of life and treatment discontinuation[143], and the more severe AEs can lead to death[144]. In some cases, treatment with vismodegib resulted in the development of squamous cell carcinoma[145], amenorrhea[146], and persistent alopecia[147]. Patient treated with vismodegib for one month can develop severe nausea, jaundice, and cholestasis with significantly elevated BUN, creatinine, and liver enzymes[148].
Sonidegib shows the same issues of drug resistance and AEs as vismodegib. However, a phase I trial demonstrated that sonidegib treatment is well tolerated and effective. The maximum tolerated dose is 800 mg administered once daily[149]. Common grade 3/4 hematological AEs are thrombocytopenia (91%), followed by neutropenia (84%) and anemia (77%). The ClinicalTrials.gov Identifier is NCT02129101, and the treatment is under examination. Patients treated with glasdegib develop some adverse events, such as dysgeusia, muscle spasms, alopecia, decreased appetite, increased blood creatinine phosphokinase, constipation, and diarrhea[147].
Treatment with patidegib results in fatigue, muscle cramps, and rash[150]. A phase I study demonstrated that BMS-833923 administered to patients with cancer was safe and well tolerated at all doses[151]. Adverse events related to napabucasin administration have been mostly mild, although some patients have experienced grade 3 gastrointestinal adverse events. More severe adverse events can be alleviated by dose reduction, discontinuation of napabucasin, or medication to reverse or manage symptoms[122]. Patients developed fatigue, hyperglycemia, nausea, rash hypertriglyceridemia, mucositis, hypophosphatemia, decreased appetite, and diarrhea after treatment with GDC-0084[152].
In addition to the potential pharmacological inhibitors targeting CSC-related signaling pathways, other methods of targeting CSCs are available, such as nanoparticles, mitochondria targeting, autophagy, hyperthermia, and antibodies.
A NDDS with a variety of properties is often used for targeted delivery and sustained release drugs[153]. Due to the strong phagocytic ability of tumor cells, abundant blood vessels in diseased tissues, and large gaps in vascular endothelial cells, NDDSs are more likely to enter the capillary wall of a tumor and become enriched in tumor tissues, which provides a new method for eradicating CSCs. The nanocarriers are small sized, have a large specific surface area, and present high surface activity, degradation in vivo, good biocompatibility, and no immunogenicity; moreover, the drug loading and pharmacokinetic properties can be improved by surface modifications to achieve targeted delivery and sustained release of drugs[154,155].
Liu et al[156] prepared the paclitaxel-loaded nanoparticles modified by cell-penetrating peptide R8, which specifically target CSCs, block angiogenesis, and reduce nutrient and blood supplement, thus leading to the suppressed proliferation of glioma CSCs. Han et al[157] delivered gemcitabine to BCSCs using hyaluronic acid (HA)-modified liposomes, and this modification improved the drug stability, prolonged the half-life, enhanced cell toxicity and anti-tumor metastasis, and inhibited cell clone formation.
Cui et al[158] synthesized a gelatinase-sensitive polymer nanocarrier that was loaded with miR-200c and acted on gastric cancer cells, and their results indicated that gastric cancer cells were significantly sensitized to radiotherapy. In addition, the expression of CD44 was down-regulated, the number of BGC823 cells (CD44+) was decreased, the cell invasion, metastasis, and anti-apoptosis ability were weakened, and the resistance characteristics of CSCs were decreased[158]. Moreover, other NDDSs have been used for targeted therapy of CSCs, such as nano-loaded genes, nano-loaded siRNA[159], and nano-compound both drugs[160].
Changes in the mitochondria in CSCs, including morphological changes, abnormal activation of signaling pathways, dysfunction, reactive oxygen species (ROS) generation, and mitochondrial autophagy, are key to regulating the proliferation and apoptosis of CSCs and represent one of the causes of anticancer therapy failure. CSCs exhibited significant anaerobic glycolysis characteristics, such as increased expression of glycolytic enzymes, increased production of lactic acid, and decreased or rested mitochondrial function[161]. These characteristics are similar to those of stem cells, suggesting that the mitochondrial glycolysis pathway plays a key role in regulating the proliferation and apoptosis of CSCs.
Liu et al[162] reported that CSCs could utilize the glycolytic pathway to supply energy induced by glucose and increase the expression of hexokinase 1 (HK-1), HK-2 and pyruvate dehydrogenase kinase 1 (PDK-1), which can prolong the life of CSCs. Dichloroethyl ester can inhibit PDK phosphorylation of pyruvate dehydrogenase and promote the conversion of pyruvate to Acyl-CoA, which converts mitochondrial metabolism from glycolytic to oxidative phosphorylation, thereby reducing cell proliferation, promoting apoptosis, and inhibiting tumor growth[163]. In addition to producing ATP, mitochondria are the main site for ROS production. BCSCs have been reported to have a higher ability to scavenge ROS than the corresponding tumor cells, thereby maintaining ROS at a relatively low level, although BCSCs die when the ROS levels are too high[164]. Kawano et al[165] found that knocking out the CD44 variant gene could reduce the defense ability of CSCs against ROS and enhance the killing ability of CD44 variant strongly positive cells treated with cisplatin but not CD44 variant weakly positive cells. Increasing the ROS level and destroying the protective effect of ROS on CSCs may also offer an alternative method for cancer treatment[166].
Over the past several years, autophagy has emerged as a requirement for the maintenance of stemness in both normal tissue stem cells[167] and CSCs[168,169]. Autophagy plays a vital role in promoting and inhibiting two different effects in different stages of tumor and tumor development; therefore, activation and inhibition of autophagy could improve the therapeutic effects for tumors[170]. Autophagy maintains the function of different types of stem cells, such as microenvironmental homeostasis and stem cell characteristics[171].
Some autophagy-related genes promote the survival of CSCs in the autophagy process. Gong et al[172] discovered that Beclin1 and autophagy are also essential for CSC maintenance and tumorigenesis in vivo. A key role for autophagy in maintaining BCSCs has been identified according to two different shRNA screens, with Beclin-1/ATG6 identified from an shRNA screen for genes that modulate the plasticity of BCSCs[173] and ATG4A identified from a screen for genes required for mammosphere formation[174]. Damage-regulated autophagy modulator 1 (DRAM1) and P62 are abundantly expressed in adult glioblastoma, and glioma stem cells are regulated in biological metabolism, tumor migration and invasion by specific siRNA acting on DRAM1 and P62 proteins[175]. When knocking out LC3 and ATG12 genes, the CD44+/CD24-/low BCSCs are inhibited. The autophagy flux of non-adherent cells (CD44+/CD24-/low) in TNBC is higher than that of adherent cells, and the expression of CD44+/CD24-/low in TNBC is decreased after treatment with the autophagy inhibitor chloroquine[176]. Jiang et al[177] found that CSCs from malignant gliomas cause cell death due to the accumulation of autophagy-related proteins and autophagosomes. Similarly, the activation of autophagy under hypoxic conditions promotes the dedifferentiation of non-pancreatic CSCs into stem-like cells, suggesting that autophagy can affect the source transformation process of CSC[178].
Because autophagy changes the sensitivity of CSCs to conventional treatment or directly destroys cells using the toxicity produced by CSC autophagy, autophagy could be used as a treatment for CSCs. Conventional therapy presents difficulty in the specific killing of tumor cells and shows many negative effects in patients. Although autophagy therapy has milder effects on normal cells than conventional therapy, it may also have unknown effects that remain to be explored.
As a curative treatment for cancer, hyperthermia is attracting increasing attention and recognition from doctors and patients. In hyperthermia treatment, tumor tissue is directly heated through ultrasound, microwave, radiofrequency, infrared, visible light, alternating magnetic fields, or heat-generating substances. This therapy does not include ionizing radiation and thus avoids the damage from radiation to patients and operators and pollution to the environment.
Tumor cells are more sensitive to heat, and the tumor area radiates more slowly; therefore, the temperature in the tumor area can be changed by hyperthermia, which leads to the death of tumor cells. At present, the temperature range of the hyperthermia method is 42-45 °C and the temperature of the thermal ablation is above 65 °C[179,180]. Hyperthermia can enhance the expression of apoptotic genes, such as P53 and FAS, thereby blocking the cell cycle, inhibiting tumor cell proliferation, and leading to tumor cell apoptosis[181]. The expression of heat shock protein is increased during hyperthermia, which stimulates the body's anti-tumor immune response[182]. The endothelial cells of the tumor microvessels proliferate and are more sensitive under heat; thus, the microvessels in the tumor tissue are more susceptible to heat damage than those of normal tissues.
Hyperthermia enhances the responses of the immune system to cancer, such as by up-regulating the homing of immune cells and the function of adhesion molecules on both immune cells and endothelial cells, activating cytotoxic T cells, dendritic cells, and natural killer cell, and inhibiting immune suppression[183]. As an adjunct to cancer therapy, hyperthermia plays an increasingly important role in the treatment of tumors[184]. By using hyperthermia in combination with chemotherapy[185], radiotherapy[186], immunotherapy[187], or surgery[188], the dose of these therapies may be reduced to ease their side effects without reducing their therapeutic effects.
The AEs of acute or chronic periods of regional hyperthermia do not develop often and are usually minor, and they include skin burns and pain; however, these events usually heal spontaneously[189]. Overall, hyperthermia is considered an alternative therapeutic method when it is used appropriately.
The CD44 receptor is a specific tumor antigen located on the surface of CSCs, and hyaluronan (HA) is the ligand for all CD44 receptors or subtypes. CD44 receptor-specific antigens play an important role in CSCs. Utilizing the monoclonal antibody MEN-85 that binds to the C-terminus of the hyaluronate-binding domain of CD44 causes a conformational rearrangement that results in the CD44 receptor detaching from the surface of CSCs, thereby blocking the signaling pathway of HA-CD44[190]. CD44 is a known marker of CSCs, and the CD44 gene splice isoforms are CD44s and CD44v. Li et al[191] reported that the CSC marker CD44s is up-regulated in human pancreatic tumors and associated with patient survival time. CD44s is necessary for the initiation, growth, metastasis, and postradiation recurrence of xenograft tumors in mice. Antibodies targeting the CD44 receptor can eliminate bulk tumor cells and CSCs from the tumors. CD44s is the predominant isoform expressed in BCSCs. Elimination of the CD44s isoform impairs CSC traits. However, manipulating the splicing regulator epithelial splicing regulatory protein 1 to shift alternative splicing from CD44v to CD44s leads to the induction of CSC properties[192]. These results suggest that alternative splicing provides functional gene versatility that is essential for different cancer cell states and thus cancer phenotypes. CSCs can also express a variety of specific antigens; for example, the CD44 receptor can also be expressed in lung cancer cells as well as BCSCs[193]. BCSCs not only express CD44 receptor but also express aldehyde dehydrogenase (ALDH)[194].
The Notch signaling pathway is abnormally active in the tumor microenvironment (TME). Inhibitor targeting that blocks signal transmission can reduce the number of CSCs and inhibit the development of tumors[195]. Blocking Wnt signaling can reduce the expression of CD44 and ALDH on the CSC surface and inhibit tumor self-renewal and metastasis[196]. ALDH1 is widely distributed in humans and highly expressed in stem cells of normal tissues as a marker of normal stem cells. Notably, the activity of ALDH1 is increased in MM and myeloid leukemia[197]. Moreover, ALDH1 is also expressed in most cancer tissues at different levels, such as breast cancer, lung cancer, and colon cancer[198].
In 2019, Chen et al[199] demonstrated that the depletion of ubiquitin-specific protease 9X (USP9X) markedly downregulated ALDH1A3, thereby resulting in the loss of the self-renewal and tumorigenic capacity of mesenchymal (MES) glioblastoma stem cells (GSCs). Furthermore, the USP9X inhibitor WP1130 induced ALDH1A3 degradation and showed marked therapeutic efficacy in MES GSC-derived orthotopic xenograft models.
Humanized anti-CD47 antibody Hu5F9-G4 can safely and effectively treat five invasive childhood brain tumors in mice[200]. Hu5F9-G4 demonstrated therapeutic efficacy in vitro and in vivo in patient-derived orthotopic xenograft models; notably, Hu5F9-G4 showed minimal activity against normal human neural cells. Advani et al[201] confirmed that the macrophage checkpoint inhibitor Hu5F9-G4 synergized with rituximab can safely and effectively eliminate aggressive and indolent lymphoma, and no clinically significant safety events were observed in this study. Liu and coworkers indicated that CpG oligodeoxynucleotide, a toll-like receptor 9 agonist, combined with CD47 inhibitors can rapidly induce tumor shrinkage and prolong survival in mice[202]. It is worth noting that a number of new drugs targeting CD47 are undergoing clinical trials, such as AO-176, CC-90002, NI-1701, IBI118, and TI-063. Therefore, macrophage immunological checkpoint blocking therapy (e.g., CD47 antibody) is expected to provide a new cancer immunotherapy strategy.
Although tumors initiate from oncogenic changes in a cancer cell, subsequent tumor progression and therapeutic responses depend on interactions between the cancer cells and the TME[203,204]. The cells and molecules in the TME are in a dynamic process that leads to a large number of immunosuppressive cells (e.g., myeloid-derived suppressor cells, regulatory cells, and tumor-associated macrophage), and a large number of inflammatory related factors (e.g., chemokines, TGF-β, and interleukins) assemble together in the TME. Then, they jointly promote tumor immune escape, tumor growth, and metastasis[205].
Among the identified CXC chemokine receptors (CXCRs), CXCR4 is most closely related to tumor cells, and it was first discovered as a co-receptor of HIV. Until now, CXCR4 has been found in a variety of cancers, including breast cancer, prostate cancer, lung cancer, colon cancer, and MM[206]. The overexpression of CXCR4 is associated with a poor prognosis in GBM. Wu et al[207] indicated that anti-CXCR4 and anti-programmed cell death protein 1 (PD-1) combination immunotherapy can modulate tumor-infiltrating populations of the glioma microenvironment. Some CXCR4 antagonists (e.g., BL-8040 and X4P-001-IO) are used in combination with PD-(L)1 drugs, such as Keytruda, Tecentri, and Opdivo. CXCL12/CXCR4-mediated desmoplasia in metastatic breast cancers (mBCs) promotes immunosuppression; therefore, it is a potential target for overcoming therapeutic resistance to immune checkpoint blockade in mBC patients[208,209].
Neutrophils are first-responders to sites of infection and tissue damage[210]. Powell et al[211] demonstrated that the chemokine receptor CXCR1 promotes neutrophil recruitment, CSC proliferation, and neoplastic mass formation. Therefore, neutrophil recruitment signaling pathways, such as CXCL8-CXCR1, have the potential for use as targets for anti-cancer therapies. Danhier et al[212] utilized the acidic TME as a target of nano-theranostics to enable cancer-specific imaging and therapy, and this approach showed advantages over conventional tumor targeting strategies. However, the acidic TME not only plays an essential role in the initiation, progression, and metastasis of tumors but also participates in the induction of treatment resistance. The TME is related with oncogenesis, host genetics, the microbiome, and immune cell activity[213]. More novel therapies targeting CSCs and the TME require further exploration, comprehensive analysis, and consolidation of both clinical and experimental data.
In recent decades, numerous new agents and methods for treating patients with cancer, such as CSC-related singling pathway inhibitors, monoclonal antibodies, hyperthermia, or NDDSs, have become available or are under clinical trial. The CSC hypothesis has provided an explanation for the failure of traditional cancer treatments. Targeting CSCs via the Wnt, Hedgehog, and Notch signaling pathways has promise in preventing disease recurrence. However, the development of such agents is hindered by many challenges. It is now clear that all signaling pathways do not function in isolation but rather as a coordinated network. The output from the entire signaling network regulates the phenotype of CSCs. Therefore, the development of CSC inhibitors will require an understanding of the stem cell signaling network. For example, combination therapy could pave the way for new and innovative strategies for the pharmacological treatment of CSCs to increase the efficacy and decrease the toxicity of individual agents.
However, NDDSs can effectively utilize the microenvironmental properties of CSCs and efficiently deliver anti-tumor drugs to tumors, which can improve the chemotherapy effect, reduce the toxicity and AEs, and improve patient compliance. Therefore, NDDSs may provide a novel alternative therapeutic strategy targeting CSCs. In addition, immunotherapy that targets the surface markers expressed by CSCs (e.g., CD47, ALDH1, and CD44) demonstrates a remarkably inhibiting effect on CSCs. Notably, a number of new drugs targeting CD47 are undergoing clinical trials, such as AO-176, CC-90002, NI-1701, IBI118, and TI-063. Hence, macrophage immunological checkpoint blocking therapy is expected to provide a new method for cancer immunotherapy.
As for the optimal time for the use of a CSC-specific therapy, there is no related reference to clarify this key issue. However, once the related cancer is identified, the therapy targeting CSCs can be concurrently proceeded with neoadjuvant chemo/radiotherapy. Generally, conventional chemotherapy can only inhibit tumor growth and lead to drug resistance, but cannot kill CSCs. However, the key to cancer treatment is how to deracinate CSCs. Therapy targeting CSCs can effectively remove CSCs and avoid drug resistance. Moreover, synergy therapy with chemo/ radiotherapy may reduce the dosage and AEs while not changing the physiological effect. However, when and how to use targeting CSCs to treat related cancers requires clinical trials to determine.
Research on CSC-related signaling pathways provides new avenues for the development of related drugs. With further developments in science and technology, CSCs will eventually be fully understood and cancer development and progression and the underlying signal regulatory mechanisms will be thoroughly clarified, thereby laying the foundation for the treatment of malignant tumors. In addition, clarifying the considerable crosstalk between cell signaling pathways is critical for designing effective therapeutic approaches. The focus of future trials should be on the use of combination therapies that affect multiple pathways in the tumor. However, this field remains in its infancy, and considerable research will be required to produce mature products. However, for the first time in medical history, we have the tools to understand the fate of tumor cells and target them therapeutically.
Manuscript source: Invited manuscript
Specialty type: Cell and tissue engineering
Country of origin: China
Peer-review report classification
Grade A (Excellent): A
Grade B (Very good): B
Grade C (Good): C, C
Grade D (Fair): 0
Grade E (Poor): 0
P-Reviewer: Andrisani OM, Labusca L, Luo WR, Valenti MT S-Editor: Yan JP L-Editor: Wang TQ E-Editor: Ma YJ
| 1. | Mackillop WJ, Ciampi A, Till JE, Buick RN. A stem cell model of human tumor growth: Implications for tumor cell clonogenic assays. J Natl Cancer Inst. 1983;70:9-16. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 2] [Cited by in RCA: 1] [Article Influence: 0.0] [Reference Citation Analysis (0)] |
| 2. | Lapidot T, Sirard C, Vormoor J, Murdoch B, Hoang T, Caceres-Cortes J, Minden M, Paterson B, Caligiuri MA, Dick JE. A cell initiating human acute myeloid leukaemia after transplantation into SCID mice. Nature. 1994;367:645-648. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 3316] [Cited by in RCA: 3381] [Article Influence: 109.1] [Reference Citation Analysis (0)] |
| 3. | Bonnet D, Dick JE. Human acute myeloid leukemia is organized as a hierarchy that originates from a primitive hematopoietic cell. Nat Med. 1997;3:730-737. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 4851] [Cited by in RCA: 4852] [Article Influence: 173.3] [Reference Citation Analysis (1)] |
| 4. | Clarke MF, Dick JE, Dirks PB, Eaves CJ, Jamieson CH, Jones DL, Visvader J, Weissman IL, Wahl GM. Cancer stem cells--perspectives on current status and future directions: AACR Workshop on cancer stem cells. Cancer Res. 2006;66:9339-9344. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 2176] [Cited by in RCA: 2198] [Article Influence: 115.7] [Reference Citation Analysis (0)] |
| 5. | Dick JE. Breast cancer stem cells revealed. Proc Natl Acad Sci U S A. 2003;100:3547-3549. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 218] [Cited by in RCA: 216] [Article Influence: 9.8] [Reference Citation Analysis (0)] |
| 6. | Ponti D, Costa A, Zaffaroni N, Pratesi G, Petrangolini G, Coradini D, Pilotti S, Pierotti MA, Daidone MG. Isolation and in vitro propagation of tumorigenic breast cancer cells with stem/progenitor cell properties. Cancer Res. 2005;65:5506-5511. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 1325] [Cited by in RCA: 1371] [Article Influence: 68.6] [Reference Citation Analysis (0)] |
| 7. | Singh SK, Clarke ID, Hide T, Dirks PB. Cancer stem cells in nervous system tumors. Oncogene. 2004;23:7267-7273. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 502] [Cited by in RCA: 486] [Article Influence: 23.1] [Reference Citation Analysis (0)] |
| 8. | Singh SK, Hawkins C, Clarke ID, Squire JA, Bayani J, Hide T, Henkelman RM, Cusimano MD, Dirks PB. Identification of human brain tumour initiating cells. Nature. 2004;432:396-401. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 5422] [Cited by in RCA: 5549] [Article Influence: 264.2] [Reference Citation Analysis (0)] |
| 9. | Richardson GD, Robson CN, Lang SH, Neal DE, Maitland NJ, Collins AT. CD133, a novel marker for human prostatic epithelial stem cells. J Cell Sci. 2004;117:3539-3545. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 562] [Cited by in RCA: 551] [Article Influence: 26.2] [Reference Citation Analysis (0)] |
| 10. | Collins AT, Berry PA, Hyde C, Stower MJ, Maitland NJ. Prospective identification of tumorigenic prostate cancer stem cells. Cancer Res. 2005;65:10946-10951. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 1998] [Cited by in RCA: 2033] [Article Influence: 107.0] [Reference Citation Analysis (0)] |
| 11. | Jung J, Yoon SM, Park JH, Seo DW, Lee SS, Kim MH, Lee SK, Park DH, Song TJ, Ryoo BY, Chang HM, Kim KP, Yoo C, Jeong JH, Kim SC, Hwang DW, Lee JH, Song KB, Jo YY, Park J, Kim JH. Stereotactic body radiation therapy for locally advanced pancreatic cancer. PLoS One. 2019;14:e0214970. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 23] [Cited by in RCA: 37] [Article Influence: 6.2] [Reference Citation Analysis (0)] |
| 12. | Fiegel HC, Glüer S, Roth B, Rischewski J, von Schweinitz D, Ure B, Lambrecht W, Kluth D. Stem-like cells in human hepatoblastoma. J Histochem Cytochem. 2004;52:1495-1501. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 52] [Cited by in RCA: 55] [Article Influence: 2.6] [Reference Citation Analysis (0)] |
| 13. | Kim CF, Jackson EL, Woolfenden AE, Lawrence S, Babar I, Vogel S, Crowley D, Bronson RT, Jacks T. Identification of bronchioalveolar stem cells in normal lung and lung cancer. Cell. 2005;121:823-835. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 1571] [Cited by in RCA: 1595] [Article Influence: 79.8] [Reference Citation Analysis (0)] |
| 14. | O'Brien CA, Pollett A, Gallinger S, Dick JE. A human colon cancer cell capable of initiating tumour growth in immunodeficient mice. Nature. 2007;445:106-110. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 2977] [Cited by in RCA: 3045] [Article Influence: 160.3] [Reference Citation Analysis (0)] |
| 15. | Dalerba P, Dylla SJ, Park IK, Liu R, Wang X, Cho RW, Hoey T, Gurney A, Huang EH, Simeone DM, Shelton AA, Parmiani G, Castelli C, Clarke MF. Phenotypic characterization of human colorectal cancer stem cells. Proc Natl Acad Sci U S A. 2007;104:10158-10163. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 1541] [Cited by in RCA: 1660] [Article Influence: 92.2] [Reference Citation Analysis (0)] |
| 16. | Monzani E, Facchetti F, Galmozzi E, Corsini E, Benetti A, Cavazzin C, Gritti A, Piccinini A, Porro D, Santinami M, Invernici G, Parati E, Alessandri G, La Porta CA. Melanoma contains CD133 and ABCG2 positive cells with enhanced tumourigenic potential. Eur J Cancer. 2007;43:935-946. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 404] [Cited by in RCA: 406] [Article Influence: 22.6] [Reference Citation Analysis (0)] |
| 17. | Frank NY, Margaryan A, Huang Y, Schatton T, Waaga-Gasser AM, Gasser M, Sayegh MH, Sadee W, Frank MH. ABCB5-mediated doxorubicin transport and chemoresistance in human malignant melanoma. Cancer Res. 2005;65:4320-4333. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 413] [Cited by in RCA: 413] [Article Influence: 20.7] [Reference Citation Analysis (0)] |
| 18. | Wu A, Luo W, Zhang Q, Yang Z, Zhang G, Li S, Yao K. Aldehyde dehydrogenase 1, a functional marker for identifying cancer stem cells in human nasopharyngeal carcinoma. Cancer Lett. 2013;330:181-189. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 61] [Cited by in RCA: 61] [Article Influence: 5.1] [Reference Citation Analysis (0)] |
| 19. | Al-Hajj M, Clarke MF. Self-renewal and solid tumor stem cells. Oncogene. 2004;23:7274-7282. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 639] [Cited by in RCA: 625] [Article Influence: 29.8] [Reference Citation Analysis (0)] |
| 20. | Koźlik M, Wójcicki P. The use of stem cells in plastic and reconstructive surgery. Adv Clin Exp Med. 2014;23:1011-1017. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 23] [Cited by in RCA: 32] [Article Influence: 2.9] [Reference Citation Analysis (0)] |
| 21. | Rosen JM, Jordan CT. The increasing complexity of the cancer stem cell paradigm. Science. 2009;324:1670-1673. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 551] [Cited by in RCA: 523] [Article Influence: 32.7] [Reference Citation Analysis (0)] |
| 22. | Houghton J, Stoicov C, Nomura S, Rogers AB, Carlson J, Li H, Cai X, Fox JG, Goldenring JR, Wang TC. Gastric cancer originating from bone marrow-derived cells. Science. 2004;306:1568-1571. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 861] [Cited by in RCA: 837] [Article Influence: 39.9] [Reference Citation Analysis (0)] |
| 23. | Staud F, Pavek P. Breast cancer resistance protein (BCRP/ABCG2). Int J Biochem Cell Biol. 2005;37:720-725. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 147] [Cited by in RCA: 152] [Article Influence: 7.6] [Reference Citation Analysis (0)] |
| 24. | Leslie EM, Deeley RG, Cole SP. Multidrug resistance proteins: Role of P-glycoprotein, MRP1, MRP2, and BCRP (ABCG2) in tissue defense. Toxicol Appl Pharmacol. 2005;204:216-237. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 996] [Cited by in RCA: 1034] [Article Influence: 51.7] [Reference Citation Analysis (0)] |
| 25. | Wong ALA, Bellot GL, Hirpara JL, Pervaiz S. Understanding the cancer stem cell phenotype: A step forward in the therapeutic management of cancer. Biochem Pharmacol. 2019;162:79-88. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 8] [Cited by in RCA: 12] [Article Influence: 2.0] [Reference Citation Analysis (0)] |
| 26. | Panaccione A, Zhang Y, Ryan M, Moskaluk CA, Anderson KS, Yarbrough WG, Ivanov SV. MYB fusions and CD markers as tools for authentication and purification of cancer stem cells from salivary adenoid cystic carcinoma. Stem Cell Res. 2017;21:160-166. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 20] [Cited by in RCA: 22] [Article Influence: 2.8] [Reference Citation Analysis (0)] |
| 27. | Li W, Reeb AN, Sewell WA, Elhomsy G, Lin RY. Phenotypic characterization of metastatic anaplastic thyroid cancer stem cells. PLoS One. 2013;8:e65095. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 32] [Cited by in RCA: 39] [Article Influence: 3.3] [Reference Citation Analysis (0)] |
| 28. | Miyamoto T, Weissman IL, Akashi K. AML1/ETO-expressing nonleukemic stem cells in acute myelogenous leukemia with 8;21 chromosomal translocation. Proc Natl Acad Sci U S A. 2000;97:7521-7526. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 361] [Cited by in RCA: 341] [Article Influence: 13.6] [Reference Citation Analysis (0)] |
| 29. | Cozzio A, Passegué E, Ayton PM, Karsunky H, Cleary ML, Weissman IL. Similar MLL-associated leukemias arising from self-renewing stem cells and short-lived myeloid progenitors. Genes Dev. 2003;17:3029-3035. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 507] [Cited by in RCA: 477] [Article Influence: 22.7] [Reference Citation Analysis (0)] |
| 30. | Savage P. Chemotherapy curable malignancies and cancer stem cells: A biological review and hypothesis. BMC Cancer. 2016;16:906. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 13] [Cited by in RCA: 14] [Article Influence: 1.6] [Reference Citation Analysis (0)] |
| 31. | Prasad S, Ramachandran S, Gupta N, Kaushik I, Srivastava SK. Cancer cells stemness: A doorstep to targeted therapy. Biochim Biophys Acta Mol Basis Dis. 2019;pii:S0925-4439(19)30071-7. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 108] [Cited by in RCA: 103] [Article Influence: 20.6] [Reference Citation Analysis (0)] |
| 32. | Na HK, Wilson MR, Kang KS, Chang CC, Grunberger D, Trosko JE. Restoration of gap junctional intercellular communication by caffeic acid phenethyl ester (CAPE) in a ras-transformed rat liver epithelial cell line. Cancer Lett. 2000;157:31-38. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 47] [Cited by in RCA: 48] [Article Influence: 1.9] [Reference Citation Analysis (0)] |
| 33. | An SM, Ding Q, Zhang J, Xie J, Li L. Targeting stem cell signaling pathways for drug discovery: Advances in the Notch and Wnt pathways. Sci China Life Sci. 2014;57:575-580. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 13] [Cited by in RCA: 13] [Article Influence: 1.2] [Reference Citation Analysis (0)] |
| 34. | Takebe N, Miele L, Harris PJ, Jeong W, Bando H, Kahn M, Yang SX, Ivy SP. Targeting Notch, Hedgehog, and Wnt pathways in cancer stem cells: Clinical update. Nat Rev Clin Oncol. 2015;12:445-464. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 916] [Cited by in RCA: 991] [Article Influence: 99.1] [Reference Citation Analysis (2)] |
| 35. | Murdoch B, Chadwick K, Martin M, Shojaei F, Shah KV, Gallacher L, Moon RT, Bhatia M. Wnt-5A augments repopulating capacity and primitive hematopoietic development of human blood stem cells in vivo. Proc Natl Acad Sci U S A. 2003;100:3422-3427. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 176] [Cited by in RCA: 164] [Article Influence: 7.5] [Reference Citation Analysis (0)] |
| 36. | Ferrer-Mayorga G, Niell N, Cantero R, González-Sancho JM, Del Peso L, Muñoz A, Larriba MJ. Vitamin D and Wnt3A have additive and partially overlapping modulatory effects on gene expression and phenotype in human colon fibroblasts. Sci Rep. 2019;9:8085. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 26] [Cited by in RCA: 25] [Article Influence: 4.2] [Reference Citation Analysis (0)] |
| 37. | Wu D, Pan W. GSK3: A multifaceted kinase in Wnt signaling. Trends Biochem Sci. 2010;35:161-168. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 559] [Cited by in RCA: 674] [Article Influence: 42.1] [Reference Citation Analysis (0)] |
| 38. | Bilic J, Huang YL, Davidson G, Zimmermann T, Cruciat CM, Bienz M, Niehrs C. Wnt induces LRP6 signalosomes and promotes dishevelled-dependent LRP6 phosphorylation. Science. 2007;316:1619-1622. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 763] [Cited by in RCA: 720] [Article Influence: 40.0] [Reference Citation Analysis (0)] |
| 39. | Gordon MD, Nusse R. Wnt signaling: Multiple pathways, multiple receptors, and multiple transcription factors. J Biol Chem. 2006;281:22429-22433. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 969] [Cited by in RCA: 1030] [Article Influence: 54.2] [Reference Citation Analysis (0)] |
| 40. | MacDonald BT, Tamai K, He X. Wnt/beta-catenin signaling: Components, mechanisms, and diseases. Dev Cell. 2009;17:9-26. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 4613] [Cited by in RCA: 4499] [Article Influence: 281.2] [Reference Citation Analysis (0)] |
| 41. | Kim J, Kim DW, Chang W, Choe J, Kim J, Park CS, Song K, Lee I. Wnt5a is secreted by follicular dendritic cells to protect germinal center B cells via Wnt/Ca2+/NFAT/NF-κB-B cell lymphoma 6 signaling. J Immunol. 2012;188:182-189. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 49] [Cited by in RCA: 57] [Article Influence: 4.1] [Reference Citation Analysis (0)] |
| 42. | De A. Wnt/Ca2+ signaling pathway: A brief overview. Acta Biochim Biophys Sin (Shanghai). 2011;43:745-756. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 302] [Cited by in RCA: 384] [Article Influence: 27.4] [Reference Citation Analysis (0)] |
| 43. | Kohn AD, Moon RT. Wnt and calcium signaling: Beta-catenin-independent pathways. Cell Calcium. 2005;38:439-446. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 504] [Cited by in RCA: 558] [Article Influence: 32.8] [Reference Citation Analysis (0)] |
| 44. | Topol L, Jiang X, Choi H, Garrett-Beal L, Carolan PJ, Yang Y. Wnt-5a inhibits the canonical Wnt pathway by promoting GSK-3-independent beta-catenin degradation. J Cell Biol. 2003;162:899-908. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 573] [Cited by in RCA: 592] [Article Influence: 26.9] [Reference Citation Analysis (0)] |
| 45. | Richards T, Modarage K, Dean C, McCarthy-Boxer A, Hilton H, Esapa C, Norman J, Wilson P, Goggolidou P. Atmin modulates Pkhd1 expression and may mediate Autosomal Recessive Polycystic Kidney Disease (ARPKD) through altered non-canonical Wnt/Planar Cell Polarity (PCP) signalling. Biochim Biophys Acta Mol Basis Dis. 2019;1865:378-390. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 15] [Cited by in RCA: 17] [Article Influence: 2.8] [Reference Citation Analysis (0)] |
| 46. | Minde DP, Radli M, Forneris F, Maurice MM, Rüdiger SG. Large extent of disorder in Adenomatous Polyposis Coli offers a strategy to guard Wnt signalling against point mutations. PLoS One. 2013;8:e77257. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 38] [Cited by in RCA: 45] [Article Influence: 3.8] [Reference Citation Analysis (0)] |
| 47. | Liu H, Shi H, Fan Q, Sun X. Cyclin Y regulates the proliferation, migration, and invasion of ovarian cancer cells via Wnt signaling pathway. Tumour Biol. 2016;37:10161-10175. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 19] [Cited by in RCA: 16] [Article Influence: 1.8] [Reference Citation Analysis (0)] |
| 48. | Xie Y, Wang B. Downregulation of TNFAIP2 suppresses proliferation and metastasis in esophageal squamous cell carcinoma through activation of the Wnt/β-catenin signaling pathway. Oncol Rep. 2017;37:2920-2928. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 19] [Cited by in RCA: 31] [Article Influence: 3.9] [Reference Citation Analysis (0)] |
| 49. | Wang Q, Wang X, Zhang CB. Lentivirus mediated GOLPH3 shRNA inhibits growth and metastasis of esophageal squamous cancer. Asian Pac J Cancer Prev. 2013;14:5391-5396. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 8] [Cited by in RCA: 9] [Article Influence: 0.8] [Reference Citation Analysis (0)] |
| 50. | Zhang Y. Ganoderma lucidum (Reishi) suppresses proliferation and migration of breast cancer cells via inhibiting Wnt/β-catenin signaling. Biochem Biophys Res Commun. 2017;488:679-684. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 32] [Cited by in RCA: 39] [Article Influence: 4.9] [Reference Citation Analysis (0)] |
| 51. | Anastas JN, Moon RT. WNT signalling pathways as therapeutic targets in cancer. Nat Rev Cancer. 2013;13:11-26. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 1384] [Cited by in RCA: 1523] [Article Influence: 126.9] [Reference Citation Analysis (0)] |
| 52. | Wang X, Moon J, Dodge ME, Pan X, Zhang L, Hanson JM, Tuladhar R, Ma Z, Shi H, Williams NS, Amatruda JF, Carroll TJ, Lum L, Chen C. The development of highly potent inhibitors for porcupine. J Med Chem. 2013;56:2700-2704. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 78] [Cited by in RCA: 89] [Article Influence: 7.4] [Reference Citation Analysis (0)] |
| 53. | Liu J, Pan S, Hsieh MH, Ng N, Sun F, Wang T, Kasibhatla S, Schuller AG, Li AG, Cheng D, Li J, Tompkins C, Pferdekamper A, Steffy A, Cheng J, Kowal C, Phung V, Guo G, Wang Y, Graham MP, Flynn S, Brenner JC, Li C, Villarroel MC, Schultz PG, Wu X, McNamara P, Sellers WR, Petruzzelli L, Boral AL, Seidel HM, McLaughlin ME, Che J, Carey TE, Vanasse G, Harris JL. Targeting Wnt-driven cancer through the inhibition of Porcupine by LGK974. Proc Natl Acad Sci U S A. 2013;110:20224-20229. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 510] [Cited by in RCA: 657] [Article Influence: 54.8] [Reference Citation Analysis (0)] |
| 54. | Madan B, Ke Z, Harmston N, Ho SY, Frois AO, Alam J, Jeyaraj DA, Pendharkar V, Ghosh K, Virshup IH, Manoharan V, Ong EH, Sangthongpitag K, Hill J, Petretto E, Keller TH, Lee MA, Matter A, Virshup DM. Wnt addiction of genetically defined cancers reversed by PORCN inhibition. Oncogene. 2016;35:2197-2207. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 204] [Cited by in RCA: 263] [Article Influence: 26.3] [Reference Citation Analysis (0)] |
| 55. | Sasaki T, Kahn M. Inhibition of β-catenin/p300 interaction proximalizes mouse embryonic lung epithelium. Transl Respir Med. 2014;2:8. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 15] [Cited by in RCA: 15] [Article Influence: 1.4] [Reference Citation Analysis (0)] |
| 56. | Ma J, Li TF, Han XW, Yuan HF. Downregulated MEG3 contributes to tumour progression and poor prognosis in oesophagal squamous cell carcinoma by interacting with miR-4261, downregulating DKK2 and activating the Wnt/β-catenin signalling. Artif Cells Nanomed Biotechnol. 2019;47:1513-1523. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 22] [Cited by in RCA: 31] [Article Influence: 5.2] [Reference Citation Analysis (0)] |
| 57. | Osawa Y, Oboki K, Imamura J, Kojika E, Hayashi Y, Hishima T, Saibara T, Shibasaki F, Kohara M, Kimura K. Inhibition of Cyclic Adenosine Monophosphate (cAMP)-response Element-binding Protein (CREB)-binding Protein (CBP)/β-Catenin Reduces Liver Fibrosis in Mice. EBioMedicine. 2015;2:1751-1758. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 50] [Cited by in RCA: 75] [Article Influence: 7.5] [Reference Citation Analysis (0)] |
| 58. | Arqués O, Chicote I, Puig I, Tenbaum SP, Argilés G, Dienstmann R, Fernández N, Caratù G, Matito J, Silberschmidt D, Rodon J, Landolfi S, Prat A, Espín E, Charco R, Nuciforo P, Vivancos A, Shao W, Tabernero J, Palmer HG. Tankyrase Inhibition Blocks Wnt/β-Catenin Pathway and Reverts Resistance to PI3K and AKT Inhibitors in the Treatment of Colorectal Cancer. Clin Cancer Res. 2016;22:644-656. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 111] [Cited by in RCA: 149] [Article Influence: 14.9] [Reference Citation Analysis (0)] |
| 59. | Dregalla RC, Zhou J, Idate RR, Battaglia CL, Liber HL, Bailey SM. Regulatory roles of tankyrase 1 at telomeres and in DNA repair: Suppression of T-SCE and stabilization of DNA-PKcs. Aging (Albany NY). 2010;2:691-708. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 47] [Cited by in RCA: 60] [Article Influence: 4.3] [Reference Citation Analysis (0)] |
| 60. | Huang SM, Mishina YM, Liu S, Cheung A, Stegmeier F, Michaud GA, Charlat O, Wiellette E, Zhang Y, Wiessner S, Hild M, Shi X, Wilson CJ, Mickanin C, Myer V, Fazal A, Tomlinson R, Serluca F, Shao W, Cheng H, Shultz M, Rau C, Schirle M, Schlegl J, Ghidelli S, Fawell S, Lu C, Curtis D, Kirschner MW, Lengauer C, Finan PM, Tallarico JA, Bouwmeester T, Porter JA, Bauer A, Cong F. Tankyrase inhibition stabilizes axin and antagonizes Wnt signalling. Nature. 2009;461:614-620. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 1480] [Cited by in RCA: 1659] [Article Influence: 103.7] [Reference Citation Analysis (0)] |
| 61. | Lu J, Ma Z, Hsieh JC, Fan CW, Chen B, Longgood JC, Williams NS, Amatruda JF, Lum L, Chen C. Structure-activity relationship studies of small-molecule inhibitors of Wnt response. Bioorg Med Chem Lett. 2009;19:3825-3827. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 124] [Cited by in RCA: 115] [Article Influence: 7.2] [Reference Citation Analysis (0)] |
| 62. | He B, You L, Uematsu K, Xu Z, Lee AY, Matsangou M, McCormick F, Jablons DM. A monoclonal antibody against Wnt-1 induces apoptosis in human cancer cells. Neoplasia. 2004;6:7-14. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 193] [Cited by in RCA: 205] [Article Influence: 9.8] [Reference Citation Analysis (0)] |
| 63. | Katoh M, Katoh M. Molecular genetics and targeted therapy of WNT-related human diseases (Review). Int J Mol Med. 2017;40:587-606. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 57] [Cited by in RCA: 109] [Article Influence: 13.6] [Reference Citation Analysis (0)] |
| 64. | Pavlovic Z, Adams JJ, Blazer LL, Gakhal AK, Jarvik N, Steinhart Z, Robitaille M, Mascall K, Pan J, Angers S, Moffat J, Sidhu SS. A synthetic anti-Frizzled antibody engineered for broadened specificity exhibits enhanced anti-tumor properties. MAbs. 2018;10:1157-1167. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 21] [Cited by in RCA: 50] [Article Influence: 7.1] [Reference Citation Analysis (0)] |
| 65. | Fischer MM, Cancilla B, Yeung VP, Cattaruzza F, Chartier C, Murriel CL, Cain J, Tam R, Cheng CY, Evans JW, O'Young G, Song X, Lewicki J, Kapoun AM, Gurney A, Yen WC, Hoey T. WNT antagonists exhibit unique combinatorial antitumor activity with taxanes by potentiating mitotic cell death. Sci Adv. 2017;3:e1700090. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 85] [Cited by in RCA: 116] [Article Influence: 14.5] [Reference Citation Analysis (0)] |
| 66. | Gurney A, Axelrod F, Bond CJ, Cain J, Chartier C, Donigan L, Fischer M, Chaudhari A, Ji M, Kapoun AM, Lam A, Lazetic S, Ma S, Mitra S, Park IK, Pickell K, Sato A, Satyal S, Stroud M, Tran H, Yen WC, Lewicki J, Hoey T. Wnt pathway inhibition via the targeting of Frizzled receptors results in decreased growth and tumorigenicity of human tumors. Proc Natl Acad Sci U S A. 2012;109:11717-11722. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 378] [Cited by in RCA: 483] [Article Influence: 37.2] [Reference Citation Analysis (0)] |
| 67. | Le PN, McDermott JD, Jimeno A. Targeting the Wnt pathway in human cancers: Therapeutic targeting with a focus on OMP-54F28. Pharmacol Ther. 2015;146:1-11. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 143] [Cited by in RCA: 191] [Article Influence: 17.4] [Reference Citation Analysis (0)] |
| 68. | D'Amico L, Mahajan S, Capietto AH, Yang Z, Zamani A, Ricci B, Bumpass DB, Meyer M, Su X, Wang-Gillam A, Weilbaecher K, Stewart SA, DeNardo DG, Faccio R. Dickkopf-related protein 1 (Dkk1) regulates the accumulation and function of myeloid derived suppressor cells in cancer. J Exp Med. 2016;213:827-840. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 86] [Cited by in RCA: 128] [Article Influence: 14.2] [Reference Citation Analysis (0)] |
| 69. | Katoh M. Canonical and non-canonical WNT signaling in cancer stem cells and their niches: Cellular heterogeneity, omics reprogramming, targeted therapy and tumor plasticity (Review). Int J Oncol. 2017;51:1357-1369. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 216] [Cited by in RCA: 342] [Article Influence: 42.8] [Reference Citation Analysis (0)] |
| 70. | Stratford EW, Daffinrud J, Munthe E, Castro R, Waaler J, Krauss S, Myklebost O. The tankyrase-specific inhibitor JW74 affects cell cycle progression and induces apoptosis and differentiation in osteosarcoma cell lines. Cancer Med. 2014;3:36-46. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 41] [Cited by in RCA: 49] [Article Influence: 4.1] [Reference Citation Analysis (0)] |
| 71. | Fujii N, You L, Xu Z, Uematsu K, Shan J, He B, Mikami I, Edmondson LR, Neale G, Zheng J, Guy RK, Jablons DM. An antagonist of dishevelled protein-protein interaction suppresses beta-catenin-dependent tumor cell growth. Cancer Res. 2007;67:573-579. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 201] [Cited by in RCA: 179] [Article Influence: 9.9] [Reference Citation Analysis (0)] |
| 72. | Nichols AM, Pan Y, Herreman A, Hadland BK, De Strooper B, Kopan R, Huppert SS. Notch pathway is dispensable for adipocyte specification. Genesis. 2004;40:40-44. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 45] [Cited by in RCA: 47] [Article Influence: 2.4] [Reference Citation Analysis (0)] |
| 73. | Gazave E, Lapébie P, Richards GS, Brunet F, Ereskovsky AV, Degnan BM, Borchiellini C, Vervoort M, Renard E. Origin and evolution of the Notch signalling pathway: An overview from eukaryotic genomes. BMC Evol Biol. 2009;9:249. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 157] [Cited by in RCA: 194] [Article Influence: 12.1] [Reference Citation Analysis (0)] |
| 74. | Andersson ER, Sandberg R, Lendahl U. Notch signaling: Simplicity in design, versatility in function. Development. 2011;138:3593-3612. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 671] [Cited by in RCA: 729] [Article Influence: 52.1] [Reference Citation Analysis (0)] |
| 75. | Hernandez Tejada FN. Galvez Silva JR, Zweidler-McKay PA. The challenge of targeting notch in hematologic malignancies. Front Pediatr. 2014;2:54. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 18] [Cited by in RCA: 20] [Article Influence: 1.8] [Reference Citation Analysis (0)] |
| 76. | Fiúza UM, Arias AM. Cell and molecular biology of Notch. J Endocrinol. 2007;194:459-474. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 256] [Cited by in RCA: 262] [Article Influence: 14.6] [Reference Citation Analysis (0)] |
| 77. | Nakata T, Shimizu H, Nagata S, Ito G, Fujii S, Suzuki K, Kawamoto A, Ishibashi F, Kuno R, Anzai S, Murano T, Mizutani T, Oshima S, Tsuchiya K, Nakamura T, Hozumi K, Watanabe M, Okamoto R. Indispensable role of Notch ligand-dependent signaling in the proliferation and stem cell niche maintenance of APC-deficient intestinal tumors. Biochem Biophys Res Commun. 2017;482:1296-1303. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 7] [Cited by in RCA: 11] [Article Influence: 1.2] [Reference Citation Analysis (0)] |
| 78. | Wang Y, Lu E, Bao R, Xu P, Feng F, Wen W, Dong Q, Hu C, Xiao L, Tang M, Li G, Wang J, Zhang C. Notch signalling regulates steroidogenesis in mouse ovarian granulosa cells. Reprod Fertil Dev. 2019;. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 11] [Cited by in RCA: 10] [Article Influence: 1.7] [Reference Citation Analysis (0)] |
| 79. | Li Y, Ma J, Qian X, Wu Q, Xia J, Miele L, Sarkar FH, Wang Z. Regulation of EMT by Notch signaling pathway in tumor progression. Curr Cancer Drug Targets. 2013;13:957-962. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 59] [Cited by in RCA: 70] [Article Influence: 6.4] [Reference Citation Analysis (0)] |
| 80. | Tolcher AW, Messersmith WA, Mikulski SM, Papadopoulos KP, Kwak EL, Gibbon DG, Patnaik A, Falchook GS, Dasari A, Shapiro GI, Boylan JF, Xu ZX, Wang K, Koehler A, Song J, Middleton SA, Deutsch J, Demario M, Kurzrock R, Wheler JJ. Phase I study of RO4929097, a gamma secretase inhibitor of Notch signaling, in patients with refractory metastatic or locally advanced solid tumors. J Clin Oncol. 2012;30:2348-2353. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 179] [Cited by in RCA: 208] [Article Influence: 16.0] [Reference Citation Analysis (0)] |
| 81. | Cook N, Basu B, Smith DM, Gopinathan A, Evans J, Steward WP, Palmer D, Propper D, Venugopal B, Hategan M, Anthoney DA, Hampson LV, Nebozhyn M, Tuveson D, Farmer-Hall H, Turner H, McLeod R, Halford S, Jodrell D. A phase I trial of the γ-secretase inhibitor MK-0752 in combination with gemcitabine in patients with pancreatic ductal adenocarcinoma. Br J Cancer. 2018;118:793-801. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 80] [Cited by in RCA: 91] [Article Influence: 13.0] [Reference Citation Analysis (0)] |
| 82. | Kongkavitoon P, Butta P, Sanpavat A, Bhattarakosol P, Tangtanatakul P, Wongprom B, Tangkijvanich P, Hirankarn N, Palaga T. Regulation of periostin expression by Notch signaling in hepatocytes and liver cancer cell lines. Biochem Biophys Res Commun. 2018;506:739-745. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 11] [Cited by in RCA: 11] [Article Influence: 1.6] [Reference Citation Analysis (0)] |
| 83. | Andersson ER, Lendahl U. Therapeutic modulation of Notch signalling--are we there yet? Nat Rev Drug Discov. 2014;13:357-378. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 335] [Cited by in RCA: 364] [Article Influence: 33.1] [Reference Citation Analysis (0)] |
| 84. | Takebe N, Nguyen D, Yang SX. Targeting notch signaling pathway in cancer: Clinical development advances and challenges. Pharmacol Ther. 2014;141:140-149. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 274] [Cited by in RCA: 325] [Article Influence: 27.1] [Reference Citation Analysis (0)] |
| 85. | Strosberg JR, Yeatman T, Weber J, Coppola D, Schell MJ, Han G, Almhanna K, Kim R, Valone T, Jump H, Sullivan D. A phase II study of RO4929097 in metastatic colorectal cancer. Eur J Cancer. 2012;48:997-1003. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 117] [Cited by in RCA: 115] [Article Influence: 8.8] [Reference Citation Analysis (0)] |
| 86. | Diaz-Padilla I, Wilson MK, Clarke BA, Hirte HW, Welch SA, Mackay HJ, Biagi JJ, Reedijk M, Weberpals JI, Fleming GF, Wang L, Liu G, Zhou C, Blattler C, Ivy SP, Oza AM. A phase II study of single-agent RO4929097, a gamma-secretase inhibitor of Notch signaling, in patients with recurrent platinum-resistant epithelial ovarian cancer: A study of the Princess Margaret, Chicago and California phase II consortia. Gynecol Oncol. 2015;137:216-222. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 50] [Cited by in RCA: 61] [Article Influence: 6.1] [Reference Citation Analysis (0)] |
| 87. | Lee SM, Moon J, Redman BG, Chidiac T, Flaherty LE, Zha Y, Othus M, Ribas A, Sondak VK, Gajewski TF, Margolin KA. Phase 2 study of RO4929097, a gamma-secretase inhibitor, in metastatic melanoma: SWOG 0933. Cancer. 2015;121:432-440. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 70] [Cited by in RCA: 76] [Article Influence: 7.6] [Reference Citation Analysis (0)] |
| 88. | Pan E, Supko JG, Kaley TJ, Butowski NA, Cloughesy T, Jung J, Desideri S, Grossman S, Ye X, Park DM. Phase I study of RO4929097 with bevacizumab in patients with recurrent malignant glioma. J Neurooncol. 2016;130:571-579. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 33] [Cited by in RCA: 41] [Article Influence: 4.6] [Reference Citation Analysis (0)] |
| 89. | Richter S, Bedard PL, Chen EX, Clarke BA, Tran B, Hotte SJ, Stathis A, Hirte HW, Razak AR, Reedijk M, Chen Z, Cohen B, Zhang WJ, Wang L, Ivy SP, Moore MJ, Oza AM, Siu LL, McWhirter E. A phase I study of the oral gamma secretase inhibitor R04929097 in combination with gemcitabine in patients with advanced solid tumors (PHL-078/CTEP 8575). Invest New Drugs. 2014;32:243-249. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 56] [Cited by in RCA: 67] [Article Influence: 5.6] [Reference Citation Analysis (0)] |
| 90. | Villalobos VM, Hall F, Jimeno A, Gore L, Kern K, Cesari R, Huang B, Schowinsky JT, Blatchford PJ, Hoffner B, Elias A, Messersmith W. Long-Term Follow-Up of Desmoid Fibromatosis Treated with PF-03084014, an Oral Gamma Secretase Inhibitor. Ann Surg Oncol. 2018;25:768-775. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 29] [Cited by in RCA: 43] [Article Influence: 5.4] [Reference Citation Analysis (0)] |
| 91. | Kummar S, O'Sullivan Coyne G, Do KT, Turkbey B, Meltzer PS, Polley E, Choyke PL, Meehan R, Vilimas R, Horneffer Y, Juwara L, Lih A, Choudhary A, Mitchell SA, Helman LJ, Doroshow JH, Chen AP. Clinical Activity of the γ-Secretase Inhibitor PF-03084014 in Adults With Desmoid Tumors (Aggressive Fibromatosis). J Clin Oncol. 2017;35:1561-1569. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 139] [Cited by in RCA: 139] [Article Influence: 17.4] [Reference Citation Analysis (0)] |
| 92. | Wu CX, Xu A, Zhang CC, Olson P, Chen L, Lee TK, Cheung TT, Lo CM, Wang XQ. Notch Inhibitor PF-03084014 Inhibits Hepatocellular Carcinoma Growth and Metastasis via Suppression of Cancer Stemness due to Reduced Activation of Notch1-Stat3. Mol Cancer Ther. 2017;16:1531-1543. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 47] [Cited by in RCA: 57] [Article Influence: 7.1] [Reference Citation Analysis (0)] |
| 93. | Issa ME, Berndt S, Carpentier G, Pezzuto JM, Cuendet M. Bruceantin inhibits multiple myeloma cancer stem cell proliferation. Cancer Biol Ther. 2016;17:966-975. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 17] [Cited by in RCA: 24] [Article Influence: 2.7] [Reference Citation Analysis (0)] |
| 94. | Morgan KM, Fischer BS, Lee FY, Shah JJ, Bertino JR, Rosenfeld J, Singh A, Khiabanian H, Pine SR. Gamma Secretase Inhibition by BMS-906024 Enhances Efficacy of Paclitaxel in Lung Adenocarcinoma. Mol Cancer Ther. 2017;16:2759-2769. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 33] [Cited by in RCA: 46] [Article Influence: 5.8] [Reference Citation Analysis (0)] |
| 95. | Krebs LT, Xue Y, Norton CR, Shutter JR, Maguire M, Sundberg JP, Gallahan D, Closson V, Kitajewski J, Callahan R, Smith GH, Stark KL, Gridley T. Notch signaling is essential for vascular morphogenesis in mice. Genes Dev. 2000;14:1343-1352. [PubMed] |
| 96. | Yang J, Xing H, Lu D, Wang J, Li B, Tang J, Gu F, Hong L. Role of Jagged1/STAT3 signalling in platinum-resistant ovarian cancer. J Cell Mol Med. 2019;23:4005-4018. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 16] [Cited by in RCA: 29] [Article Influence: 4.8] [Reference Citation Analysis (0)] |
| 97. | Chen Z, Zuo X, Pu L, Zhang Y, Han G, Zhang L, Wu Z, You W, Qin J, Dai X, Shen H, Wang X, Wu J. Hypomethylation-mediated activation of cancer/testis antigen KK-LC-1 facilitates hepatocellular carcinoma progression through activating the Notch1/Hes1 signalling. Cell Prolif. 2019;52:e12581. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 14] [Cited by in RCA: 34] [Article Influence: 5.7] [Reference Citation Analysis (0)] |
| 98. | Liu X, Luo Q, Zheng Y, Liu X, Hu Y, Liu W, Luo M, Zhao Y, Zou L. NOTCH4 signaling controls EFNB2-induced endothelial progenitor cell dysfunction in preeclampsia. Reproduction. 2016;152:47-55. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 11] [Cited by in RCA: 13] [Article Influence: 1.4] [Reference Citation Analysis (0)] |
| 99. | Tammela T, Zarkada G, Wallgard E, Murtomäki A, Suchting S, Wirzenius M, Waltari M, Hellström M, Schomber T, Peltonen R, Freitas C, Duarte A, Isoniemi H, Laakkonen P, Christofori G, Ylä-Herttuala S, Shibuya M, Pytowski B, Eichmann A, Betsholtz C, Alitalo K. Blocking VEGFR-3 suppresses angiogenic sprouting and vascular network formation. Nature. 2008;454:656-660. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 617] [Cited by in RCA: 635] [Article Influence: 37.4] [Reference Citation Analysis (0)] |
| 100. | Chiorean EG, LoRusso P, Strother RM, Diamond JR, Younger A, Messersmith WA, Adriaens L, Liu L, Kao RJ, DiCioccio AT, Kostic A, Leek R, Harris A, Jimeno A. A Phase I First-in-Human Study of Enoticumab (REGN421), a Fully Human Delta-like Ligand 4 (Dll4) Monoclonal Antibody in Patients with Advanced Solid Tumors. Clin Cancer Res. 2015;21:2695-2703. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 116] [Cited by in RCA: 110] [Article Influence: 11.0] [Reference Citation Analysis (0)] |
| 101. | Yan M. Therapeutic promise and challenges of targeting DLL4/NOTCH1. Vasc Cell. 2011;3:17. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 28] [Cited by in RCA: 30] [Article Influence: 2.1] [Reference Citation Analysis (0)] |
| 102. | Nüsslein-Volhard C, Wieschaus E. Mutations affecting segment number and polarity in Drosophila. Nature. 1980;287:795-801. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 2958] [Cited by in RCA: 2889] [Article Influence: 64.2] [Reference Citation Analysis (0)] |
| 103. | Singh AN, Sharma N. Epigenetic Modulators as Potential Multi-targeted Drugs Against Hedgehog Pathway for Treatment of Cancer. Protein J. 2019;. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 11] [Cited by in RCA: 11] [Article Influence: 2.2] [Reference Citation Analysis (0)] |
| 104. | Guan Z, Dong B, Huang C, Hu X, Zhang Y, Lin C. Expression patterns of genes critical for SHH, BMP, and FGF pathways during the lumen formation of human salivary glands. J Mol Histol. 2019;50:217-227. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 4] [Cited by in RCA: 4] [Article Influence: 0.7] [Reference Citation Analysis (0)] |
| 105. | Babu D, Fanelli A, Mellone S, Muniswamy R, Wasniewska M, Prodam F, Petri A, Bellone S, Salerno MC, Giordano M. Novel GLI2 mutations identified in patients with Combined Pituitary Hormone Deficiency (CPHD): Evidence for a pathogenic effect by functional characterization. Clin Endocrinol (Oxf). 2019;90:449-456. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 9] [Cited by in RCA: 11] [Article Influence: 1.8] [Reference Citation Analysis (1)] |
| 106. | Hashimoto H, Jiang W, Yoshimura T, Moon KH, Bok J, Ikenaka K. Strong sonic hedgehog signaling in the mouse ventral spinal cord is not required for oligodendrocyte precursor cell (OPC) generation but is necessary for correct timing of its generation. Neurochem Int. 2018;119:178-183. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 7] [Cited by in RCA: 8] [Article Influence: 1.0] [Reference Citation Analysis (0)] |
| 107. | Wu C, Zhu X, Liu W, Ruan T, Tao K. Hedgehog signaling pathway in colorectal cancer: Function, mechanism, and therapy. Onco Targets Ther. 2017;10:3249-3259. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 39] [Cited by in RCA: 61] [Article Influence: 7.6] [Reference Citation Analysis (0)] |
| 108. | Li W, Yang H, Li X, Han L, Xu N, Shi A. Signaling pathway inhibitors target breast cancer stem cells in triple-negative breast cancer. Oncol Rep. 2019;41:437-446. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 14] [Cited by in RCA: 18] [Article Influence: 2.6] [Reference Citation Analysis (0)] |
| 109. | Yauch RL, Dijkgraaf GJ, Alicke B, Januario T, Ahn CP, Holcomb T, Pujara K, Stinson J, Callahan CA, Tang T, Bazan JF, Kan Z, Seshagiri S, Hann CL, Gould SE, Low JA, Rudin CM, de Sauvage FJ. Smoothened mutation confers resistance to a Hedgehog pathway inhibitor in medulloblastoma. Science. 2009;326:572-574. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 720] [Cited by in RCA: 697] [Article Influence: 43.6] [Reference Citation Analysis (0)] |
| 110. | Minami H, Ando Y, Ma BB, Hsiang Lee J, Momota H, Fujiwara Y, Li L, Fukino K, Ito K, Tajima T, Mori A, Lin CC. Phase I, multicenter, open-label, dose-escalation study of sonidegib in Asian patients with advanced solid tumors. Cancer Sci. 2016;107:1477-1483. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 13] [Cited by in RCA: 16] [Article Influence: 1.8] [Reference Citation Analysis (0)] |
| 111. | Wickström M, Dyberg C, Shimokawa T, Milosevic J, Baryawno N, Fuskevåg OM, Larsson R, Kogner P, Zaphiropoulos PG, Johnsen JI. Targeting the hedgehog signal transduction pathway at the level of GLI inhibits neuroblastoma cell growth in vitro and in vivo. Int J Cancer. 2013;132:1516-1524. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 83] [Cited by in RCA: 98] [Article Influence: 7.5] [Reference Citation Analysis (0)] |
| 112. | List A, Beran M, DiPersio J, Slack J, Vey N, Rosenfeld CS, Greenberg P. Opportunities for Trisenox (arsenic trioxide) in the treatment of myelodysplastic syndromes. Leukemia. 2003;17:1499-1507. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 95] [Cited by in RCA: 79] [Article Influence: 3.6] [Reference Citation Analysis (0)] |
| 113. | Beauchamp EM, Ringer L, Bulut G, Sajwan KP, Hall MD, Lee YC, Peaceman D, Ozdemirli M, Rodriguez O, Macdonald TJ, Albanese C, Toretsky JA, Uren A. Arsenic trioxide inhibits human cancer cell growth and tumor development in mice by blocking Hedgehog/GLI pathway. J Clin Invest. 2011;121:148-160. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 255] [Cited by in RCA: 281] [Article Influence: 18.7] [Reference Citation Analysis (0)] |
| 114. | Lauth M, Bergström A, Shimokawa T, Toftgård R. Inhibition of GLI-mediated transcription and tumor cell growth by small-molecule antagonists. Proc Natl Acad Sci U S A. 2007;104:8455-8460. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 601] [Cited by in RCA: 642] [Article Influence: 35.7] [Reference Citation Analysis (0)] |
| 115. | Long B, Wang LX, Zheng FM, Lai SP, Xu DR, Hu Y, Lin DJ, Zhang XZ, Dong L, Long ZJ, Tong XZ, Liu Q. Targeting GLI1 Suppresses Cell Growth and Enhances Chemosensitivity in CD34+ Enriched Acute Myeloid Leukemia Progenitor Cells. Cell Physiol Biochem. 2016;38:1288-1302. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 40] [Cited by in RCA: 47] [Article Influence: 5.2] [Reference Citation Analysis (0)] |
| 116. | Bromberg JF, Wrzeszczynska MH, Devgan G, Zhao Y, Pestell RG, Albanese C, Darnell JE. Stat3 as an oncogene. Cell. 1999;98:295-303. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 2236] [Cited by in RCA: 2344] [Article Influence: 90.2] [Reference Citation Analysis (0)] |
| 117. | Yu H, Lee H, Herrmann A, Buettner R, Jove R. Revisiting STAT3 signalling in cancer: New and unexpected biological functions. Nat Rev Cancer. 2014;14:736-746. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 1338] [Cited by in RCA: 1653] [Article Influence: 150.3] [Reference Citation Analysis (0)] |
| 118. | MacDonagh L, Gray SG, Breen E, Cuffe S, Finn SP, O'Byrne KJ, Barr MP. BBI608 inhibits cancer stemness and reverses cisplatin resistance in NSCLC. Cancer Lett. 2018;428:117-126. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 46] [Cited by in RCA: 72] [Article Influence: 10.3] [Reference Citation Analysis (0)] |
| 119. | Li Y, Rogoff HA, Keates S, Gao Y, Murikipudi S, Mikule K, Leggett D, Li W, Pardee AB, Li CJ. Suppression of cancer relapse and metastasis by inhibiting cancer stemness. Proc Natl Acad Sci U S A. 2015;112:1839-1844. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 300] [Cited by in RCA: 376] [Article Influence: 37.6] [Reference Citation Analysis (0)] |
| 120. | Zhang Y, Jin Z, Zhou H, Ou X, Xu Y, Li H, Liu C, Li B. Suppression of prostate cancer progression by cancer cell stemness inhibitor napabucasin. Cancer Med. 2016;5:1251-1258. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 52] [Cited by in RCA: 66] [Article Influence: 7.3] [Reference Citation Analysis (0)] |
| 121. | Beyreis M, Gaisberger M, Jakab M, Neureiter D, Helm K, Ritter M, Kiesslich T, Mayr C. The Cancer Stem Cell Inhibitor Napabucasin (BBI608) Shows General Cytotoxicity in Biliary Tract Cancer Cells and Reduces Cancer Stem Cell Characteristics. Cancers (Basel). 2019;11:pii: E276. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 9] [Cited by in RCA: 17] [Article Influence: 2.8] [Reference Citation Analysis (0)] |
| 122. | Hubbard JM, Grothey A. Napabucasin: An Update on the First-in-Class Cancer Stemness Inhibitor. Drugs. 2017;77:1091-1103. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 83] [Cited by in RCA: 120] [Article Influence: 15.0] [Reference Citation Analysis (0)] |
| 123. | Zuo D. Shogren KL, Zang J, Jewison DE, Waletzki BE, Miller AL 2nd, Okuno SH, Cai Z, Yaszemski MJ, Maran A. Inhibition of STAT3 blocks protein synthesis and tumor metastasis in osteosarcoma cells. J Exp Clin Cancer Res. 2018;37:244. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 27] [Cited by in RCA: 52] [Article Influence: 7.4] [Reference Citation Analysis (0)] |
| 124. | Zhou Q, Peng C, Du F, Zhou L, Shi Y, Du Y, Liu D, Sun W, Zhang M, Chen G. Design, synthesis and activity of BBI608 derivatives targeting on stem cells. Eur J Med Chem. 2018;151:39-50. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 27] [Cited by in RCA: 31] [Article Influence: 4.4] [Reference Citation Analysis (0)] |
| 125. | Haupt Y, Alexander WS, Barri G, Klinken SP, Adams JM. Novel zinc finger gene implicated as myc collaborator by retrovirally accelerated lymphomagenesis in E mu-myc transgenic mice. Cell. 1991;65:753-763. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 415] [Cited by in RCA: 433] [Article Influence: 12.7] [Reference Citation Analysis (0)] |
| 126. | Mihara K, Chowdhury M, Nakaju N, Hidani S, Ihara A, Hyodo H, Yasunaga S, Takihara Y, Kimura A. Bmi-1 is useful as a novel molecular marker for predicting progression of myelodysplastic syndrome and patient prognosis. Blood. 2006;107:305-308. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 103] [Cited by in RCA: 102] [Article Influence: 5.1] [Reference Citation Analysis (0)] |
| 127. | Nishida Y, Maeda A, Kim MJ, Cao L, Kubota Y, Ishizawa J, AlRawi A, Kato Y, Iwama A, Fujisawa M, Matsue K, Weetall M, Dumble M, Andreeff M, Davis TW, Branstrom A, Kimura S, Kojima K. The novel BMI-1 inhibitor PTC596 downregulates MCL-1 and induces p53-independent mitochondrial apoptosis in acute myeloid leukemia progenitor cells. Blood Cancer J. 2017;7:e527. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 55] [Cited by in RCA: 72] [Article Influence: 9.0] [Reference Citation Analysis (0)] |
| 128. | Urist MR. Bone: Formation by autoinduction. Science. 1965;150:893-899. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 4079] [Cited by in RCA: 3689] [Article Influence: 61.5] [Reference Citation Analysis (0)] |
| 129. | Liao WJ, Lin H, Cheng CF, Ka SM, Chen A, Yang RB. SCUBE1-enhanced bone morphogenetic protein signaling protects against renal ischemia-reperfusion injury. Biochim Biophys Acta Mol Basis Dis. 2019;1865:329-338. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 4] [Cited by in RCA: 6] [Article Influence: 0.9] [Reference Citation Analysis (0)] |
| 130. | Altman AM, Mahmud J, Nikolovska-Coleska Z, Chan G. HCMV modulation of cellular PI3K/AKT/mTOR signaling: New opportunities for therapeutic intervention? Antiviral Res. 2019;163:82-90. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 31] [Cited by in RCA: 30] [Article Influence: 5.0] [Reference Citation Analysis (0)] |
| 131. | Wang X, Wang X, Zhang S, Sun H, Li S, Ding H, You Y, Zhang X, Ye SD. The transcription factor TFCP2L1 induces expression of distinct target genes and promotes self-renewal of mouse and human embryonic stem cells. J Biol Chem. 2019;294:6007-6016. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 22] [Cited by in RCA: 27] [Article Influence: 4.5] [Reference Citation Analysis (0)] |
| 132. | Salphati L, Alicke B, Heffron TP, Shahidi-Latham S, Nishimura M, Cao T, Carano RA, Cheong J, Greve J, Koeppen H, Lau S, Lee LB, Nannini-Pepe M, Pang J, Plise EG, Quiason C, Rangell L, Zhang X, Gould SE, Phillips HS, Olivero AG. Brain Distribution and Efficacy of the Brain Penetrant PI3K Inhibitor GDC-0084 in Orthotopic Mouse Models of Human Glioblastoma. Drug Metab Dispos. 2016;44:1881-1889. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 42] [Cited by in RCA: 40] [Article Influence: 4.4] [Reference Citation Analysis (0)] |
| 133. | Ding LT, Zhao P, Yang ML, Lv GZ, Zhao TL. GDC-0084 inhibits cutaneous squamous cell carcinoma cell growth. Biochem Biophys Res Commun. 2018;503:1941-1948. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 13] [Cited by in RCA: 16] [Article Influence: 2.3] [Reference Citation Analysis (0)] |
| 134. | EDDC (Experimental Drug Development Centre). [accessed 2019 May 16]. In: ClinicalTrials.gov [Internet]. Bethesda (MD): U.S. National Library of Medicine, Identifier: NCT02521844. Available from: URL: https://clinicaltrials.gov/ct2/show/NCT02521844 ClinicalTrials.gov. |
| 135. | Huang J, Hu W, Hu L, Previs RA, Dalton HJ, Yang XY, Sun Y, McGuire M, Rupaimoole R, Nagaraja AS, Kang Y, Liu T, Nick AM, Jennings NB, Coleman RL, Jaffe RB, Sood AK. Dll4 Inhibition plus Aflibercept Markedly Reduces Ovarian Tumor Growth. Mol Cancer Ther. 2016;15:1344-1352. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 37] [Cited by in RCA: 39] [Article Influence: 4.3] [Reference Citation Analysis (0)] |
| 136. | Vantictumab and OMP-54F78 trials on hold to assess adverse events. React Wkly. 2014;1509:2-2. [RCA] [DOI] [Full Text] [Cited by in Crossref: 2] [Cited by in RCA: 1] [Article Influence: 0.1] [Reference Citation Analysis (0)] |
| 137. | Jimeno A, Gordon M, Chugh R, Messersmith W, Mendelson D, Dupont J, Stagg R, Kapoun AM, Xu L, Uttamsingh S, Brachmann RK, Smith DC. A First-in-Human Phase I Study of the Anticancer Stem Cell Agent Ipafricept (OMP-54F28), a Decoy Receptor for Wnt Ligands, in Patients with Advanced Solid Tumors. Clin Cancer Res. 2017;23:7490-7497. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 110] [Cited by in RCA: 154] [Article Influence: 19.3] [Reference Citation Analysis (0)] |
| 138. | Pant S, Jones SF, Kurkjian CD, Infante JR, Moore KN, Burris HA, McMeekin DS, Benhadji KA, Patel BKR, Frenzel MJ, Kursar JD, Zamek-Gliszczynski MJ, Yuen ESM, Chan EM, Bendell JC. A first-in-human phase I study of the oral Notch inhibitor, LY900009, in patients with advanced cancer. Eur J Cancer. 2016;56:1-9. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 56] [Cited by in RCA: 67] [Article Influence: 7.4] [Reference Citation Analysis (0)] |
| 139. | Mir O, Azaro A, Merchan J, Chugh R, Trent J, Rodon J, Ohnmacht U, Diener JT, Smith C, Yuen E, Oakley GJ, Le Cesne A, Soria JC, Benhadji KA, Massard C. Notch pathway inhibition with LY3039478 in soft tissue sarcoma and gastrointestinal stromal tumours. Eur J Cancer. 2018;103:88-97. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 15] [Cited by in RCA: 22] [Article Influence: 3.1] [Reference Citation Analysis (0)] |
| 140. | PHARMABIZ. com. Audion, REGAIN Consortium announce positive results from phase I study of notch inhibitor LY3056480 in patients with sensorineural hearing loss [cited 17 March 2019]. Amsterdam, Netherlands. Available from: http://www.pharmabiz.com/NewsDetails.aspx?aid=114010sid=2. |
| 141. | LoConte NK, Razak AR, Ivy P, Tevaarwerk A, Leverence R, Kolesar J, Siu L, Lubner SJ, Mulkerin DL, Schelman WR, Deming DA, Holen KD, Carmichael L, Eickhoff J, Liu G. A multicenter phase 1 study of γ -secretase inhibitor RO4929097 in combination with capecitabine in refractory solid tumors. Invest New Drugs. 2015;33:169-176. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 28] [Cited by in RCA: 32] [Article Influence: 2.9] [Reference Citation Analysis (0)] |
| 142. | Zweidler-McKay PA, DeAngelo DJ, Douer D, Dombret H, Ottmann OG, Vey N, Thomas DA, Zhu LL, Huang F, Bajaj G, Fischer BS. The safety and activity of BMS-906024, a gamma secretase inhibitor (GSI) with anti-Notch activity, in patients with relapsed T-cell acute lymphoblastic leukemia (T-ALL): initial results of a phase 1 trial. Blood. 2014;124:968. |
| 143. | Lacouture ME, Dréno B, Ascierto PA, Dummer R, Basset-Seguin N, Fife K, Ernst S, Licitra L, Neves RI, Peris K, Puig S, Sokolof J, Sekulic A, Hauschild A, Kunstfeld R. Characterization and Management of Hedgehog Pathway Inhibitor-Related Adverse Events in Patients With Advanced Basal Cell Carcinoma. Oncologist. 2016;21:1218-1229. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 89] [Cited by in RCA: 113] [Article Influence: 12.6] [Reference Citation Analysis (0)] |
| 144. | Sekulic A, Migden MR, Oro AE, Dirix L, Lewis KD, Hainsworth JD, Solomon JA, Yoo S, Arron ST, Friedlander PA, Marmur E, Rudin CM, Chang AL, Low JA, Mackey HM, Yauch RL, Graham RA, Reddy JC, Hauschild A. Efficacy and safety of vismodegib in advanced basal-cell carcinoma. N Engl J Med. 2012;366:2171-2179. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 947] [Cited by in RCA: 1029] [Article Influence: 79.2] [Reference Citation Analysis (0)] |
| 145. | Saintes C, Saint-Jean M, Brocard A, Peuvrel L, Renaut JJ, Khammari A, Quéreux G, Dréno B. Development of squamous cell carcinoma into basal cell carcinoma under treatment with Vismodegib. J Eur Acad Dermatol Venereol. 2015;29:1006-1009. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 31] [Cited by in RCA: 34] [Article Influence: 3.1] [Reference Citation Analysis (0)] |
| 146. | Strasswimmer J, Latimer B, Ory S. Amenorrhea secondary to a vismodegib-induced blockade of follicle-stimulating hormone-receptor activation. Fertil Steril. 2014;102:555-557. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 10] [Cited by in RCA: 10] [Article Influence: 0.9] [Reference Citation Analysis (0)] |
| 147. | Alkeraye S, Maire C, Desmedt E, Templier C, Mortier L. Persistent alopecia induced by vismodegib. Br J Dermatol. 2015;172:1671-1672. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 20] [Cited by in RCA: 21] [Article Influence: 2.1] [Reference Citation Analysis (0)] |
| 148. | Ash MM, Jolly PS. Cholestatic hepatic injury associated with vismodegib, aspirin, and naproxen use: A case study and review of vismodegib safety. Int J Dermatol. 2015;54:370-374. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 13] [Cited by in RCA: 14] [Article Influence: 1.3] [Reference Citation Analysis (0)] |
| 149. | Rodon J, Tawbi HA, Thomas AL, Stoller RG, Turtschi CP, Baselga J, Sarantopoulos J, Mahalingam D, Shou Y, Moles MA, Yang L, Granvil C, Hurh E, Rose KL, Amakye DD, Dummer R, Mita AC. A phase I, multicenter, open-label, first-in-human, dose-escalation study of the oral smoothened inhibitor Sonidegib (LDE225) in patients with advanced solid tumors. Clin Cancer Res. 2014;20:1900-1909. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 172] [Cited by in RCA: 185] [Article Influence: 16.8] [Reference Citation Analysis (0)] |
| 150. | Bowles DW, Keysar SB, Eagles JR, Wang G, Glogowska MJ, McDermott JD, Le PN, Gao D, Ray CE, Rochon PJ, Roop DR, Tan AC, Serracino HS, Jimeno A. A pilot study of cetuximab and the hedgehog inhibitor IPI-926 in recurrent/metastatic head and neck squamous cell carcinoma. Oral Oncol. 2016;53:74-79. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 23] [Cited by in RCA: 31] [Article Influence: 3.1] [Reference Citation Analysis (0)] |
| 151. | Siu LL, Papadopoulos K, Alberts SR, Kirchoff-Ross R, Vakkalagadda B, Lang L, Ahlers CM, Bennett KL, Van Tornout JM. A first-in-human, phase I study of an oral hedgehog (HH) pathway antagonist, BMS-833923(XL139), in subjects with advanced or metastatic solid tumors. Mol Cancer Ther. 2009;8:A55-A55. [DOI] [Full Text] |
| 152. | Genentech, Inc; Genentech, Inc. [accessed 2016 Nov 2]. In: ClinicalTrials.gov [Internet]. Bethesda (MD): U.S. National Library of Medicine. Identifier: NCT01547546. Available from: URL: https://clinicaltrials.gov/ct2/show/NCT01547546 ClinicalTrials.gov. |
| 153. | Li SJ, Liao YF, Du Q. [Research and application of quercetin-loaded nano drug delivery system]. Zhongguo Zhong Yao Za Zhi. 2018;43:1978-1984. [RCA] [PubMed] [DOI] [Full Text] [Cited by in RCA: 3] [Reference Citation Analysis (0)] |
| 154. | Kanapathipillai M, Brock A, Ingber DE. Nanoparticle targeting of anti-cancer drugs that alter intracellular signaling or influence the tumor microenvironment. Adv Drug Deliv Rev. 2014;79-80:107-118. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 162] [Cited by in RCA: 166] [Article Influence: 15.1] [Reference Citation Analysis (0)] |
| 155. | Peng C, Xu J, Yu M, Ning X, Huang Y, Du B, Hernandez E, Kapur P, Hsieh JT, Zheng J. Tuning the In Vivo Transport of Anticancer Drugs Using Renal-Clearable Gold Nanoparticles. Angew Chem Int Ed Engl. 2019;58:8479-8483. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 58] [Cited by in RCA: 69] [Article Influence: 11.5] [Reference Citation Analysis (0)] |
| 156. | Liu Y, Mei L, Yu Q, Xu C, Qiu Y, Yang Y, Shi K, Zhang Q, Gao H, Zhang Z, He Q. Multifunctional Tandem Peptide Modified Paclitaxel-Loaded Liposomes for the Treatment of Vasculogenic Mimicry and Cancer Stem Cells in Malignant Glioma. ACS Appl Mater Interfaces. 2015;7:16792-16801. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 54] [Cited by in RCA: 53] [Article Influence: 5.3] [Reference Citation Analysis (0)] |
| 157. | Han NK, Shin DH, Kim JS, Weon KY, Jang CY, Kim JS. Hyaluronan-conjugated liposomes encapsulating gemcitabine for breast cancer stem cells. Int J Nanomedicine. 2016;11:1413-1425. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 49] [Cited by in RCA: 42] [Article Influence: 4.7] [Reference Citation Analysis (0)] |
| 158. | Cui FB, Liu Q, Li RT, Shen J, Wu PY, Yu LX, Hu WJ, Wu FL, Jiang CP, Yue GF, Qian XP, Jiang XQ, Liu BR. Enhancement of radiotherapy efficacy by miR-200c-loaded gelatinase-stimuli PEG-Pep-PCL nanoparticles in gastric cancer cells. Int J Nanomedicine. 2014;9:2345-2358. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 4] [Cited by in RCA: 12] [Article Influence: 1.1] [Reference Citation Analysis (0)] |
| 159. | Kim SS, Rait A, Kim E, Pirollo KF, Nishida M, Farkas N, Dagata JA, Chang EH. A nanoparticle carrying the p53 gene targets tumors including cancer stem cells, sensitizes glioblastoma to chemotherapy and improves survival. ACS Nano. 2014;8:5494-5514. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 108] [Cited by in RCA: 116] [Article Influence: 10.5] [Reference Citation Analysis (0)] |
| 160. | Wei X, He J, Wang J, Wang W. MPEG-CS/Bmi-1RNAi Nanoparticles Synthesis and Its Targeted Inhibition Effect on CD133+ Laryngeal Stem Cells. J Nanosci Nanotechnol. 2018;18:1577-1584. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 6] [Cited by in RCA: 6] [Article Influence: 0.9] [Reference Citation Analysis (0)] |
| 161. | Ciavardelli D, Rossi C, Barcaroli D, Volpe S, Consalvo A, Zucchelli M, De Cola A, Scavo E, Carollo R, D'Agostino D, Forlì F, D'Aguanno S, Todaro M, Stassi G, Di Ilio C, De Laurenzi V, Urbani A. Breast cancer stem cells rely on fermentative glycolysis and are sensitive to 2-deoxyglucose treatment. Cell Death Dis. 2014;5:e1336. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 177] [Cited by in RCA: 219] [Article Influence: 4.2] [Reference Citation Analysis (0)] |
| 162. | Liu PP, Liao J, Tang ZJ, Wu WJ, Yang J, Zeng ZL, Hu Y, Wang P, Ju HQ, Xu RH, Huang P. Metabolic regulation of cancer cell side population by glucose through activation of the Akt pathway. Cell Death Differ. 2014;21:124-135. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 147] [Cited by in RCA: 181] [Article Influence: 15.1] [Reference Citation Analysis (0)] |
| 163. | Michelakis ED, Webster L, Mackey JR. Dichloroacetate (DCA) as a potential metabolic-targeting therapy for cancer. Br J Cancer. 2008;99:989-994. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 469] [Cited by in RCA: 512] [Article Influence: 30.1] [Reference Citation Analysis (0)] |
| 164. | Diehn M, Cho RW, Lobo NA, Kalisky T, Dorie MJ, Kulp AN, Qian D, Lam JS, Ailles LE, Wong M, Joshua B, Kaplan MJ, Wapnir I, Dirbas FM, Somlo G, Garberoglio C, Paz B, Shen J, Lau SK, Quake SR, Brown JM, Weissman IL, Clarke MF. Association of reactive oxygen species levels and radioresistance in cancer stem cells. Nature. 2009;458:780-783. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 2102] [Cited by in RCA: 1928] [Article Influence: 120.5] [Reference Citation Analysis (0)] |
| 165. | Kawano Y, Iwama E, Tsuchihashi K, Shibahara D, Harada T, Tanaka K, Nagano O, Saya H, Nakanishi Y, Okamoto I. CD44 variant-dependent regulation of redox balance in EGFR mutation-positive non-small cell lung cancer: A target for treatment. Lung Cancer. 2017;113:72-78. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 8] [Cited by in RCA: 8] [Article Influence: 1.0] [Reference Citation Analysis (0)] |
| 166. | Moloney JN, Cotter TG. ROS signalling in the biology of cancer. Semin Cell Dev Biol. 2018;80:50-64. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 754] [Cited by in RCA: 1305] [Article Influence: 163.1] [Reference Citation Analysis (0)] |
| 167. | García-Prat L, Martínez-Vicente M, Perdiguero E, Ortet L, Rodríguez-Ubreva J, Rebollo E, Ruiz-Bonilla V, Gutarra S, Ballestar E, Serrano AL, Sandri M, Muñoz-Cánoves P. Autophagy maintains stemness by preventing senescence. Nature. 2016;529:37-42. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 791] [Cited by in RCA: 1005] [Article Influence: 111.7] [Reference Citation Analysis (0)] |
| 168. | Sharif T, Martell E, Dai C, Kennedy BE, Murphy P, Clements DR, Kim Y, Lee PW, Gujar SA. Autophagic homeostasis is required for the pluripotency of cancer stem cells. Autophagy. 2017;13:264-284. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 81] [Cited by in RCA: 111] [Article Influence: 12.3] [Reference Citation Analysis (0)] |
| 169. | Whelan KA, Chandramouleeswaran PM, Tanaka K, Natsuizaka M, Guha M, Srinivasan S, Darling DS, Kita Y, Natsugoe S, Winkler JD, Klein-Szanto AJ, Amaravadi RK, Avadhani NG, Rustgi AK, Nakagawa H. Autophagy supports generation of cells with high CD44 expression via modulation of oxidative stress and Parkin-mediated mitochondrial clearance. Oncogene. 2017;36:4843-4858. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 54] [Cited by in RCA: 78] [Article Influence: 9.8] [Reference Citation Analysis (0)] |
| 170. | Liu J, Debnath J. The Evolving, Multifaceted Roles of Autophagy in Cancer. Adv Cancer Res. 2016;130:1-53. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 40] [Cited by in RCA: 55] [Article Influence: 6.1] [Reference Citation Analysis (0)] |
| 171. | Smith AG, Macleod KF. Autophagy, cancer stem cells and drug resistance. J Pathol. 2019;247:708-718. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 160] [Cited by in RCA: 281] [Article Influence: 46.8] [Reference Citation Analysis (0)] |
| 172. | Gong C, Bauvy C, Tonelli G, Yue W, Deloménie C, Nicolas V, Zhu Y, Domergue V, Marin-Esteban V, Tharinger H, Delbos L, Gary-Gouy H, Morel AP, Ghavami S, Song E, Codogno P, Mehrpour M. Beclin 1 and autophagy are required for the tumorigenicity of breast cancer stem-like/progenitor cells. Oncogene. 2013;32:2261-2272, 2272e.1-2272e11. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 242] [Cited by in RCA: 281] [Article Influence: 21.6] [Reference Citation Analysis (0)] |
| 173. | Gupta PB, Fillmore CM, Jiang G, Shapira SD, Tao K, Kuperwasser C, Lander ES. Stochastic state transitions give rise to phenotypic equilibrium in populations of cancer cells. Cell. 2011;146:633-644. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 1079] [Cited by in RCA: 1110] [Article Influence: 79.3] [Reference Citation Analysis (0)] |
| 174. | Wolf J, Dewi DL, Fredebohm J, Müller-Decker K, Flechtenmacher C, Hoheisel JD, Boettcher M. A mammosphere formation RNAi screen reveals that ATG4A promotes a breast cancer stem-like phenotype. Breast Cancer Res. 2013;15:R109. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 83] [Cited by in RCA: 107] [Article Influence: 8.9] [Reference Citation Analysis (0)] |
| 175. | Galavotti S, Bartesaghi S, Faccenda D, Shaked-Rabi M, Sanzone S, McEvoy A, Dinsdale D, Condorelli F, Brandner S, Campanella M, Grose R, Jones C, Salomoni P. The autophagy-associated factors DRAM1 and p62 regulate cell migration and invasion in glioblastoma stem cells. Oncogene. 2013;32:699-712. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 179] [Cited by in RCA: 215] [Article Influence: 16.5] [Reference Citation Analysis (0)] |
| 176. | Cufí S, Vazquez-Martin A, Oliveras-Ferraros C, Martin-Castillo B, Vellon L, Menendez JA. Autophagy positively regulates the CD44(+) CD24(-/low) breast cancer stem-like phenotype. Cell Cycle. 2011;10:3871-3885. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 138] [Cited by in RCA: 154] [Article Influence: 11.0] [Reference Citation Analysis (0)] |
| 177. | Jiang H, Gomez-Manzano C, Aoki H, Alonso MM, Kondo S, McCormick F, Xu J, Kondo Y, Bekele BN, Colman H, Lang FF, Fueyo J. Examination of the therapeutic potential of Delta-24-RGD in brain tumor stem cells: Role of autophagic cell death. J Natl Cancer Inst. 2007;99:1410-1414. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 209] [Cited by in RCA: 222] [Article Influence: 12.3] [Reference Citation Analysis (0)] |
| 178. | Zhu H, Wang D, Liu Y, Su Z, Zhang L, Chen F, Zhou Y, Wu Y, Yu M, Zhang Z, Shao G. Role of the Hypoxia-inducible factor-1 alpha induced autophagy in the conversion of non-stem pancreatic cancer cells into CD133+ pancreatic cancer stem-like cells. Cancer Cell Int. 2013;13:119. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 92] [Cited by in RCA: 102] [Article Influence: 8.5] [Reference Citation Analysis (0)] |
| 179. | Spriggs DR, Zivanovic O. Ovarian Cancer Treatment - Are We Getting Warmer? N Engl J Med. 2018;378:293-294. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 25] [Cited by in RCA: 29] [Article Influence: 4.1] [Reference Citation Analysis (0)] |
| 180. | Lee Titsworth W, Murad GJ, Hoh BL, Rahman M. Fighting fire with fire: The revival of thermotherapy for gliomas. Anticancer Res. 2014;34:565-574. [PubMed] |
| 181. | Guan J, Stavridi E, Leeper DB, Iliakis G. Effects of hyperthermia on p53 protein expression and activity. J Cell Physiol. 2002;190:365-374. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 23] [Cited by in RCA: 26] [Article Influence: 1.1] [Reference Citation Analysis (0)] |
| 182. | Sottile ML, Losinno AD, Fanelli MA, Cuello-Carrión FD, Montt-Guevara MM, Vargas-Roig LM, Nadin SB. Hyperthermia effects on Hsp27 and Hsp72 associations with mismatch repair (MMR) proteins and cisplatin toxicity in MMR-deficient/proficient colon cancer cell lines. Int J Hyperthermia. 2015;31:464-475. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 20] [Cited by in RCA: 20] [Article Influence: 2.0] [Reference Citation Analysis (0)] |
| 183. | Zhang HG, Mehta K, Cohen P, Guha C. Hyperthermia on immune regulation: A temperature's story. Cancer Lett. 2008;271:191-204. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 94] [Cited by in RCA: 86] [Article Influence: 5.1] [Reference Citation Analysis (0)] |
| 184. | Skitzki JJ, Repasky EA, Evans SS. Hyperthermia as an immunotherapy strategy for cancer. Curr Opin Investig Drugs. 2009;10:550-558. [PubMed] |
| 185. | Correale P, Del Vecchio MT, La Placa M, Montagnani F, Di Genova G, Savellini GG, Terrosi C, Mannucci S, Giorgi G, Francini G, Cusi MG. Chemotherapeutic drugs may be used to enhance the killing efficacy of human tumor antigen peptide-specific CTLs. J Immunother. 2008;31:132-147. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 46] [Cited by in RCA: 42] [Article Influence: 2.5] [Reference Citation Analysis (0)] |
| 186. | Mallory M. Gogineni E, Jones GC, Greer L, Simone CB 2nd. Therapeutic hyperthermia: The old, the new, and the upcoming. Crit Rev Oncol Hematol. 2016;97:56-64. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 145] [Cited by in RCA: 134] [Article Influence: 13.4] [Reference Citation Analysis (0)] |
| 187. | Kleef R, Moss R, Szasz AM, Bohdjalian A, Bojar H, Bakacs T. Complete Clinical Remission of Stage IV Triple-Negative Breast Cancer Lung Metastasis Administering Low-Dose Immune Checkpoint Blockade in Combination With Hyperthermia and Interleukin-2. Integr Cancer Ther. 2018;17:1297-1303. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 21] [Cited by in RCA: 28] [Article Influence: 4.0] [Reference Citation Analysis (0)] |
| 188. | Gnanamony M, Gondi CS. Chemoresistance in pancreatic cancer: Emerging concepts. Oncol Lett. 2017;13:2507-2513. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 44] [Cited by in RCA: 51] [Article Influence: 6.4] [Reference Citation Analysis (0)] |
| 189. | Feldmann HJ, Seegenschmiedt MH, Molls M. Hyperthermia--its actual role in radiation oncology. Part III: Clinical rationale and results in deep seated tumors. Strahlenther Onkol. 1995;171:251-264. [PubMed] |
| 190. | Škerlová J, Král V, Kachala M, Fábry M, Bumba L, Svergun DI, Tošner Z, Veverka V, Řezáčová P. Molecular mechanism for the action of the anti-CD44 monoclonal antibody MEM-85. J Struct Biol. 2015;191:214-223. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 10] [Cited by in RCA: 13] [Article Influence: 1.3] [Reference Citation Analysis (0)] |
| 191. | Li L, Hao X, Qin J, Tang W, He F, Smith A, Zhang M, Simeone DM, Qiao XT, Chen ZN, Lawrence TS, Xu L. Antibody against CD44s inhibits pancreatic tumor initiation and postradiation recurrence in mice. Gastroenterology. 2014;146:1108-1118. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 105] [Cited by in RCA: 121] [Article Influence: 11.0] [Reference Citation Analysis (0)] |
| 192. | Zhang H, Brown RL, Wei Y, Zhao P, Liu S, Liu X, Deng Y, Hu X, Zhang J, Gao XD, Kang Y, Mercurio AM, Goel HL, Cheng C. CD44 splice isoform switching determines breast cancer stem cell state. Genes Dev. 2019;33:166-179. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 81] [Cited by in RCA: 158] [Article Influence: 26.3] [Reference Citation Analysis (0)] |
| 193. | Thapa R, Wilson GD. The Importance of CD44 as a Stem Cell Biomarker and Therapeutic Target in Cancer. Stem Cells Int. 2016;2016:2087204. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 106] [Cited by in RCA: 201] [Article Influence: 22.3] [Reference Citation Analysis (0)] |
| 194. | Peiris-Pagès M, Martinez-Outschoorn UE, Pestell RG, Sotgia F, Lisanti MP. Cancer stem cell metabolism. Breast Cancer Res. 2016;18:55. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 271] [Cited by in RCA: 361] [Article Influence: 6.3] [Reference Citation Analysis (0)] |
| 195. | Mamaeva V, Niemi R, Beck M, Özliseli E, Desai D, Landor S, Gronroos T, Kronqvist P, Pettersen IK, McCormack E, Rosenholm JM, Linden M, Sahlgren C. Inhibiting Notch Activity in Breast Cancer Stem Cells by Glucose Functionalized Nanoparticles Carrying γ-secretase Inhibitors. Mol Ther. 2016;24:926-936. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 78] [Cited by in RCA: 90] [Article Influence: 10.0] [Reference Citation Analysis (0)] |
| 196. | Jiang L, Bai L. Immunotherapy of targeting at tumor stem cells. Basic Clin Med. 2018;38:246-250. [DOI] [Full Text] |
| 197. | Li K, Guo X, Wang Z, Li X, Bu Y, Bai X, Zheng L, Huang Y. The prognostic roles of ALDH1 isoenzymes in gastric cancer. Onco Targets Ther. 2016;9:3405-3414. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 10] [Cited by in RCA: 21] [Article Influence: 2.3] [Reference Citation Analysis (0)] |
| 198. | Zhou Y, Wang Y, Ju X, Lan J, Zou H, Li S, Qi Y, Jia W, Hu J, Liang W, Zhang W, Pang L, Li F. Clinicopathological significance of ALDH1A1 in lung, colorectal, and breast cancers: A meta-analysis. Biomark Med. 2015;9:777-790. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 13] [Cited by in RCA: 15] [Article Influence: 1.5] [Reference Citation Analysis (0)] |
| 199. | Chen Z, Wang HW, Wang S, Fan L, Feng S, Cai X, Peng C, Wu X, Lu J, Chen D, Chen Y, Wu W, Lu D, Liu N, You Y, Wang H. USP9X deubiquitinates ALDH1A3 and maintains mesenchymal identity in glioblastoma stem cells. J Clin Invest. 2019;130:2043-2055. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 52] [Cited by in RCA: 72] [Article Influence: 12.0] [Reference Citation Analysis (0)] |
| 200. | Gholamin S. Mitra SS, Feroze AH, Liu J, Kahn SA, Zhang M, Esparza R, Richard C, Ramaswamy V, Remke M, Volkmer AK, Willingham S, Ponnuswami A, McCarty A, Lovelace P, Storm TA, Schubert S, Hutter G, Narayanan C, Chu P, Raabe EH, Harsh G 4th, Taylor MD, Monje M, Cho YJ, Majeti R, Volkmer JP, Fisher PG, Grant G, Steinberg GK, Vogel H, Edwards M, Weissman IL, Cheshier SH. Disrupting the CD47-SIRPα anti-phagocytic axis by a humanized anti-CD47 antibody is an efficacious treatment for malignant pediatric brain tumors. Sci Transl Med. 2017;9:pii: eaaf2968. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 206] [Cited by in RCA: 330] [Article Influence: 41.3] [Reference Citation Analysis (0)] |
| 201. | Advani R, Flinn I, Popplewell L, Forero A, Bartlett NL, Ghosh N, Kline J, Roschewski M, LaCasce A, Collins GP, Tran T, Lynn J, Chen JY, Volkmer JP, Agoram B, Huang J, Majeti R, Weissman IL, Takimoto CH, Chao MP, Smith SM. CD47 Blockade by Hu5F9-G4 and Rituximab in Non-Hodgkin's Lymphoma. N Engl J Med. 2018;379:1711-1721. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 599] [Cited by in RCA: 851] [Article Influence: 121.6] [Reference Citation Analysis (0)] |
| 202. | Liu M, O'Connor RS, Trefely S, Graham K, Snyder NW, Beatty GL. Metabolic rewiring of macrophages by CpG potentiates clearance of cancer cells and overcomes tumor-expressed CD47-mediated 'don't-eat-me' signal. Nat Immunol. 2019;20:265-275. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 102] [Cited by in RCA: 209] [Article Influence: 34.8] [Reference Citation Analysis (0)] |
| 203. | Liu H, Kiseleva AA, Golemis EA. Ciliary signalling in cancer. Nat Rev Cancer. 2018;18:511-524. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 102] [Cited by in RCA: 123] [Article Influence: 17.6] [Reference Citation Analysis (0)] |
| 204. | Ma B, Cheng H, Mu C, Geng G, Zhao T, Luo Q, Ma K, Chang R, Liu Q, Gao R, Nie J, Xie J, Han J, Chen L, Ma G, Zhu Y, Chen Q. The SIAH2-NRF1 axis spatially regulates tumor microenvironment remodeling for tumor progression. Nat Commun. 2019;10:1034. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 35] [Cited by in RCA: 63] [Article Influence: 10.5] [Reference Citation Analysis (0)] |
| 205. | Jurcak N, Zheng L. Signaling in the microenvironment of pancreatic cancer: Transmitting along the nerve. Pharmacol Ther. 2019;pii:S0163-7258(19)30073-7. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 25] [Cited by in RCA: 42] [Article Influence: 7.0] [Reference Citation Analysis (0)] |
| 206. | Li X, Bu W, Meng L, Liu X, Wang S, Jiang L, Ren M, Fan Y, Sun H. CXCL12/CXCR4 pathway orchestrates CSC-like properties by CAF recruited tumor associated macrophage in OSCC. Exp Cell Res. 2019;378:131-138. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 60] [Cited by in RCA: 124] [Article Influence: 20.7] [Reference Citation Analysis (0)] |
| 207. | Wu A, Maxwell R, Xia Y, Cardarelli P, Oyasu M, Belcaid Z, Kim E, Hung A, Luksik AS, Garzon-Muvdi T, Jackson CM, Mathios D, Theodros D, Cogswell J, Brem H, Pardoll DM, Lim M. Combination anti-CXCR4 and anti-PD-1 immunotherapy provides survival benefit in glioblastoma through immune cell modulation of tumor microenvironment. J Neurooncol. 2019;143:241-249. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 64] [Cited by in RCA: 94] [Article Influence: 15.7] [Reference Citation Analysis (0)] |
| 208. | Chen IX, Chauhan VP, Posada J, Ng MR, Wu MW, Adstamongkonkul P, Huang P, Lindeman N, Langer R, Jain RK. Blocking CXCR4 alleviates desmoplasia, increases T-lymphocyte infiltration, and improves immunotherapy in metastatic breast cancer. Proc Natl Acad Sci U S A. 2019;pii:201815515. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 177] [Cited by in RCA: 293] [Article Influence: 48.8] [Reference Citation Analysis (0)] |
| 209. | Sledge GW. Targeting CXCR4-induced desmoplasia to improve checkpoint inhibition in breast cancer. Proc Natl Acad Sci U S A. 2019;116:4769-4771. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 4] [Cited by in RCA: 5] [Article Influence: 0.8] [Reference Citation Analysis (0)] |
| 210. | Fricker SP, Sprott K, Spyra M, Uhlig P, Lange N, David K, Wang Y. Characterization and Validation of Antibodies for Immunohistochemical Staining of the Chemokine CXCL12. J Histochem Cytochem. 2019;67:257-266. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 2] [Cited by in RCA: 2] [Article Influence: 0.3] [Reference Citation Analysis (0)] |
| 211. | Powell D, Lou M, Barros Becker F, Huttenlocher A. Cxcr1 mediates recruitment of neutrophils and supports proliferation of tumor-initiating astrocytes in vivo. Sci Rep. 2018;8:13285. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 29] [Cited by in RCA: 49] [Article Influence: 7.0] [Reference Citation Analysis (0)] |
| 212. | Danhier F, Feron O, Préat V. To exploit the tumor microenvironment: Passive and active tumor targeting of nanocarriers for anti-cancer drug delivery. J Control Release. 2010;148:135-146. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 1824] [Cited by in RCA: 1826] [Article Influence: 121.7] [Reference Citation Analysis (0)] |
| 213. | Feng L, Dong Z, Tao D, Zhang Y, Liu Z. The acidic tumor microenvironment: a target for smart cancer nano-theranostics. Nat Sci Rev. 2017;5:269-286. [RCA] [DOI] [Full Text] [Cited by in Crossref: 156] [Cited by in RCA: 160] [Article Influence: 20.0] [Reference Citation Analysis (0)] |