Published online Jul 15, 2003. doi: 10.3748/wjg.v9.i7.1550
Revised: March 4, 2003
Accepted: March 10, 2003
Published online: July 15, 2003
AIM: To modify the hamster-to-rat liver xenotransplant technique to prevent postoperative complications, and to study the inhibiting effect of multiglycosides tripterygium wilfordii (TII) on immune rejection.
METHODS: Female golden hamsters and inbred male Wistar rats were used as donors and recipients, respectively. One hundred and twelve orthotopic liver xenotransplants were performed by Kamada’s cuff technique with modifications. Over 72 hour survival of the animal after operation was considered as a successful operation. When the established surgical model became stable, 30 of the latest 42 cases were divided into untreated control group (n = 15) and TII group (n = 15) at random. Survival of recipients was observed. Liver specimens were collected at 2 and 72 h from the operated animals and postmortem, respectively, for histological study.
RESULTS: The successfully operative rate of the 30 operations was 80%, and the survival of the control and TII group was 7.1 ± 0.35 was days and 7.2 ± 0.52 d, respectively (t = 0.087, P = 0.931). The rate of conjunctival hyperemia in control group (100%) differed significantly from that (31%) in TII group (P = 0.001). Rejection did not occur in both groups within 2 h postoperatively, but became obvious in control group at 72 h after surgery and mild in TII group. Although rejections were obvious in both groups at death of recipients, it was less severe in TII group than in control group.
CONCLUSION: This modified Kamada’s technique can be used to establish a stable hamster-to-rat liver xenotransplant model. Monotherapy with multiglycosides tripteryguiumwilfordii (30 mg•kg-1•l-1) suppresses the rejection mildly, but fails to prolong survival of recipients.
- Citation: Guo H, Wu YJ, Zheng SS, Wang WL, Yu J. Application of modified two-cuff technique and multiglycosides tripterygium wilfordii in hamster-to-rat liver xenotransplant model. World J Gastroenterol 2003; 9(7): 1550-1553
- URL: https://www.wjgnet.com/1007-9327/full/v9/i7/1550.htm
- DOI: https://dx.doi.org/10.3748/wjg.v9.i7.1550
The hamster-to-rat liver transplant model is useful to study immunology and physiology of liver xenotransplantation (XT)[1-11], especially for the extended host response to long-surviving xenografts[12]. As it is very difficult to establish this surgical model[13], we summarized our surgical experience in producing for performing this model.
The model could serve as a tool to evaluate immunosuppressive drugs[14,15]. Tripterygium wilfordii hook F (TWHF) has been used for more than two thousand years as a traditional Chinese herb. TII is extracted and refined from the root of TWHF. In recent years, TII and other extracts of TWHF have been applied to allotransplantation and cardiac XT as immunosuppressive drugs with successful results[16-27]. However, the effects of TII in liver XT are unknown and deserve study.
Female golden hamsters (weighing 80-130 g) and inbred male Wistar rats (weighing 130-180 g) were used as donors and recipients, respectively. One hundred and twelve orthotopic liver xenotransplants were performed by Kamada’s cuff technique[28] with modifications, which were outlined as follows.
Donor operation Ligaments and vessels around the liver were partially dissected while the liver was protected by wrapping film. The common bile duct was entered from anterior wall and a Teflon catheter was inserted into the lumen proximally. The proper hepatic artery was dissected but not ligated. The infrahepatic vena cava (VC) was clamped at the level of left renal vein. The liver was perfused through the abdominal aorta with 10-20 mL of cold (4 °C) lactated Ringer’s solution containing 5 U/mL of heparin. Meanwhile the thoracic cavity was opened with the thoracic aorta clamped, and the thoracic inferior VC was opened to release the perfusate. Then, the infrahepatic VC was transected at the upper part of the clamp. So the perfusate could escape from the two ends of VC. Deep ligaments around the liver were dissected while perfusing and the right suprarenal vein was ligated. At the end of perfusion, the proper hepatic artery and portal vein (PV) at the level of splenic vein were ligated and transected. The free liver was stored in lactated Ringer’s solution at 0-4 °C.
Preparation of donor liver Preparation of the donor liver was done in lactated Ringer’s solution at 0-4 °C. Two cuffs with different diameters were mounted on the PV and infrahepatic VC, respectively. The cystic duct near the common bile duct was ligated and followed by removal of the gallbladder. The suprahepatic VC was trimmed and a stitch was left on each side for suture.
Recipient operation Vessels, the common bile duct and ligaments around the recipient liver were divided, respectively. The right suprarenal vein behind the papilla lobe was ligated, the recipient liver was removed and covered with fresh film orthotopically. The suprahepatic VC was sutured, the PV was anastomosed with cuff method. The infrahepatic VC was anastomosed as the method for suprahepatic VC. Finally, the distal part of Teflon catheter in donor bile duct was inserted into the recipient bile duct and secured by a silk suture. The greater omentum was wrapped around the bile duct. The abdominal incision was closed with continuous suture.
When the model became stable, 30 recipients of the latest 42 cases were divided into untreated controls (n = 15) and treatment group with TII 30 mg•kg-1•l-1 (n = 15) at random by gavages three days prior to operation till the end of experiment.
Liver specimens were collected at 2 and 72 h after operation or at death of the recipients, respectively, fixed in 10% formalin, embedded in paraffin, sectioned and stained with hematoxylin and eosin for light microscopy to determine the grade of rejection.
The results were analyzed by t test and Fisher’s exact test, respectively. Statistical significance was defined as a P value less than 0.05.
One hundred and twelve operations were performed from October 2000 to March 2001. The successful rate of the latest 30 operations was 80% (24/30). The causes of death were anesthesia accident in one case, chronic hemorrhage from the surface of donor liver in 3 cases, thrombosis in infrahepatic VC in one case and suppurate peritonitis in one case. The 6 recipients died within 72 h after operation were excluded from the experiment[14]. The average survival of 24 cases was 7.2 ± 0.44 d.
The average survival of the untreated controls and TII group was 7.1 ± 0.35 d and 7.2 ± 0.52 d, respectively (t = 0.087, P = 0.931). Four cases in controls and 2 in TII group died within 72 h after operation were eliminated from statistic study.
The morbidity of conjunctival hyperemia was 100% (11/11) in controls and 31% (4/13) in TII group (P = 0.001). The commence time of conjunctival hyperemia in controls and TII group were 4.73 ± 0.47 d and 5.75 ± 0.5 d, respectively (t = 3.688, P = 0.003).
Pus in conjunctiva was obvious in controls with conjunctival hyperemia, and three cases had ablepsia. However, no pus and ablepsia were observed in TII group.
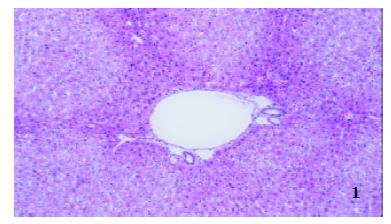
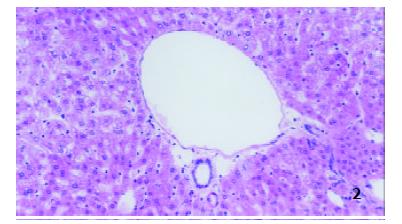
Two hours after operation The structures of hepatic lobule were normal and no inflammatory cells in portal area in both groups were observed (Figure 1 and Figure 2).
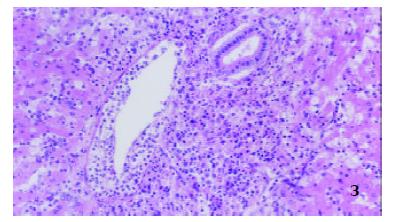
Seventy-two hours after operation In controls, hepatic lobules were normal, numerous inflammatory cells consisting mainly of mononuclear cells and neutrophilic granulocytes infiltrated in portal area, edema could be seen in the tunica intima of interlobular veins and inflammatory cells infiltrated into subintima. However, epithelia of interlobular bile ducts were not injured (Figure 3).
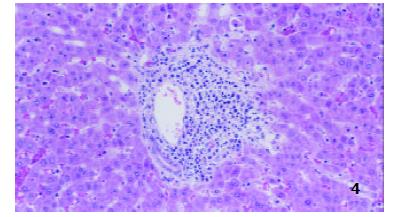
In TII group, the structures of hepatic lobules and interlobular veins were normal, moderate amounts of inflammatory cells were found in portal area, and epithelium of interlobular bile ducts was intact (Figure 4).
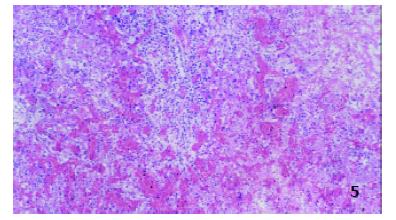
Postmortem specimens on day 7 postoperation In controls, hepatic lobules were damaged severely, inflammatory cells consisting mainly of mononuclear cells and neutrophilic granulocytes in portal area as well as massive hemorrhage were observed, interlobular vessels were disappeared and damaged epithelia of interlobular bile ducts were observed (Figure 5).
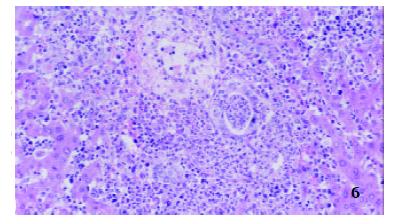
In TII group, many mixed cells could also be seen in portal area, but most of the interlobular vessels and bile ducts were existed with inflammatory intima, and variable injured epithelia, hepatocytes around portal area were swollen, degenerated and partially necrotic and some liver plates were destroyed (Figure 6).
Sun et al[29] firstly advocated that perfusate should enter the liver via dual vascular systems of the hepatic artery and the PV, then the liver and its structures around were divided. The authors performed 300 liver transplantations in rats, and the successful rate of operation and survival rate in one week were 92.7% and 88.4%, respectively. So we adopted this dual perfusion except for perfusion by PV in initial trials of 20 cases. We found that perfusion by PV could often result in incomplete perfusion in some small areas and undesirable pressure. Incomplete perfusion did not occur with dual perfusion, and due to the invariant pressure of intestinal circulation, the liver was not mechanically injured by fluctuant pressure and warm ischemia and mechancial injuries were avoided. If the donor liver was dissected at the end of complete perfusion, the vessels became pale and were difficult to be identified and were easily injured accidentally. After modification of the technique, the quality of the liver in donor was excellent in the trials and the survival time of recipient animals was similar to that in the literature[14,30,31].
We used to ligate the cystic duct, and then to remove the gallbladder and electrocauterize the bed of the gallbladder. However, it usually rendered slow hemorrhage in the bed of the gallbladder and the recipients died in hours after transplantation. So we modified the method as follows: ligating cystic duct near the common bile duct, then ligating cervix of the gallbladder and cutting gallbladder at the distal side of the ligation. No postoperative hemorrhage was observed after the modifications.
For the sake of exposing and ligating suprahepatic VC of the donor and the recipient, papilla lobe and right lobe of the liver were turned over frequently and thus were injured easily, and the recipient could die from mild hemorrhage from the surface of the graft. Sometimes, ligation of the infra-branch of suprarenal vein was mistaken for main stem with left supra-branch unligated, which rendered hemorrhage at recipient liver’s cutting and posttransplantation. Therefore, we ligated the right suprarenal vein at the end of perfusion, the structures around the donor liver were divided completely and the right suprarenal vein was exposed easily, so that liver injury and false ligation were reduced. Loop-ligation behind papilla lobe method was used to ligate the suprarenal vein of the recipient. The right suprarenal vein and part of ligaments behind the recipient liver were ligated with 5-0 silk sutures behind the papilla lobe and near the infrahepatic VC. This ligation was reliable, simple, and shortened the anhepatic phase directly.
Hamster’s liver is more fragile than that of rat. Slight press or attrition on its surface could result in contuse and chronic hemorrhage. Moreover, it has poor coagulating mechanism. Thus, fresh film has been used to protect the donor liver in operation, and the pressure on donor liver could be dispersed, the accident stab to the donor liver by instruments could be relieved to some extent and the donor liver could be kept from drying and crisping. Moreover, careful and delicate operation should not be ignored. Nevertheless, in our 30 trials, 3 of 6 failed cases died due to chronic hemorrhage on the surface of the donor liver.
We found that TII at a dosage of 30 mg•kg-1•l-1 could not prolong the survival of the operated animal. But the rejection in TII group was less severe in comparison with that of controls. We speculated that this dosage of TII had certain limited immunosuppressive effect. Li et al[32] found that the extracts of TWHF could inhibit human peripheral blood mononuclear cells (PBMC) in a concentration-dependent manner and the best inhibitory rate could be achieved at 72 h. Another research suggested that TII at a dosage of 30 mg•kg-1•l-1 had no effects on the liver, kidney, heart and the total number of WBC in rat, but could inhibit the transforming of spleen lymphocytes significantly. TII at this dosage could prolong the survival time of renal allotransplantation in Wistar-to-SD combination significantly. When it was combined with CsA, the survival time could be further prolonged[19]. Nevertheless, in other organ transplantations such as small intestine transplantation, whose rejection was relatively strong, and the monotherapy with TII was demonstrated to have limited effects[21]. Wang et al[16] applied PG27 (extract of TWHF) combined with a low dosage of CsA to hamster-to-rat cardiac xenotransplantation and all xenografts survived more than 100 d. However, they found that either PG27 or CsA had no such effects. Therefore, although a high dosage of TII was given to the recipient 3 d before operation in our trials as monotherapy, the survival time of the recipient was not prolonged due to the strong rejection in this model.
In this study, conjunctival hyperemia occurred in all untreated control recipients on day 4-5 after operation, and deteriorated rapidly, even resulted in ablepsia. However, in the recipients treated with TII, the sign occurred only in 31% rats (on day 5-6 after transplantation) and ablepsia did not occur. The differences between the two groups were highly significant and the similar report has not been found in literature. We speculated that conjunctival hyperemia could be a local representation of rejection in this model. Relieving the sign with TII treatment supported the hypothesis, but the exact mechanism of the sign needs to be further studied.
Edited by Ren SY and Wang XL
| 1. | Tandin A, Goller AL, Miki T, Lee YH, Kovscek AM, Fung JJ, Starzl TE, Valdivia LA. Concordant hamster-to-rat liver xenotransplantation leads to hyperlipidemia. Transplant Proc. 2000;32:1109. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 1] [Cited by in RCA: 1] [Article Influence: 0.0] [Reference Citation Analysis (0)] |
| 2. | Celli S, Marto JA, Falchetto R, Shabanowitz J, Valdivia LA, Fung JJ, Hunt DF, Kelly RH. Serum protein immunogenicity: implications for liver xenografting. Electrophoresis. 2000;21:965-975. [RCA] [PubMed] [DOI] [Full Text] [Cited by in RCA: 1] [Reference Citation Analysis (0)] |
| 3. | Molleví DG, Jaurrieta E, Ribas Y, Hurtado I, Serrano T, Gómez N, de Oca J, Fiol C, Figueras J. Liver xenotransplantation: changes in lipid and lipoprotein concentration after long-term graft survival. J Hepatol. 2000;32:655-660. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 5] [Cited by in RCA: 5] [Article Influence: 0.2] [Reference Citation Analysis (0)] |
| 4. | Tan JW, Zhang SG, Jiang Y, Yang JM, Qian GX, Wu MC. Apoptosis in acute rejection of hamster-to-rat liver transplantation. Hepatobiliary Pancreat Dis Int. 2002;1:340-344. [PubMed] |
| 5. | Tan JW, Yao HX, Yang JM, Qian GX, Wu MC. The role of spleen in rejection of concordant liver xenograft. Zhonghua Qiguan Yizhi Zazhi. 2000;21:83-85. |
| 6. | Valdivia LA, Fung JJ, Demetris AJ, Celli S, Pan F, Tsugita M, Starzl TE. Donor species complement after liver xenotransplantation. The mechanism of protection from hyperacute rejection. Transplantation. 1994;57:918-922. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 27] [Cited by in RCA: 26] [Article Influence: 0.8] [Reference Citation Analysis (0)] |
| 7. | Schraa EO, Stockmann HB, Broekhuizen AJ, Scheringa M, Schuurman HJ, Marquet RL, IJzermans JN. IgG, but not IgM, mediates hyperacute rejection in hepatic xenografting. Xenotransplantation. 1999;6:110-116. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 12] [Cited by in RCA: 12] [Article Influence: 0.5] [Reference Citation Analysis (0)] |
| 8. | Diao TJ, Yuan TY, Li YL. Immunologic role of nitric oxide in acute rejection of golden hamster to rat liver xenotransplantation. World J Gastroenterol. 2002;8:746-751. [PubMed] |
| 9. | Zhang SG, Wu MC, Tan JW, Chen H, Yang JM, Qian QJ. Expression of perforin and granzyme B mRNA in judgement of immunosuppressive effect in rat liver transplantation. World J Gastroenterol. 1999;5:217-220. [PubMed] |
| 10. | Valdivia LA, Demetris AJ, Fung JJ, Celli S, Murase N, Starzl TE. Successful hamster-to-rat liver xenotransplantation under FK506 immunosuppression induces unresponsiveness to hamster heart and skin. Transplantation. 1993;55:659-661. [PubMed] |
| 11. | Celli S, Valdivia LA, Fung JJ, Kelly RH. Early recipient-donor switch of the complement type after liver xenotransplantation. Immunol Invest. 1997;26:589-600. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 6] [Cited by in RCA: 6] [Article Influence: 0.2] [Reference Citation Analysis (0)] |
| 12. | Molleví DG, Ribas Y, Ginesta MM, Serrano T, Mestre M, Vidal A, Figueras J, Jaurrieta E. Heart and liver xenotransplantation under low-dose tacrolimus: graft survival after withdrawal of immunosuppression. Transplantation. 2001;71:217-223. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 7] [Cited by in RCA: 7] [Article Influence: 0.3] [Reference Citation Analysis (0)] |
| 13. | Tan JW, Zhang SG, Qian GX, Wu MC. The modified for orthotopic liver transplantation model in hamster-to-rat. Zhongguo Puwai Jichu Yu Linchuang Zazhi. 1999;6:201-203. |
| 14. | Murase N, Starzl TE, Demetris AJ, Valdivia L, Tanabe M, Cramer D, Makowka L. Hamster-to-rat heart and liver xenotransplantation with FK506 plus antiproliferative drugs. Transplantation. 1993;55:701-707; discussion 701-707;. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 116] [Cited by in RCA: 128] [Article Influence: 4.0] [Reference Citation Analysis (0)] |
| 15. | Murase N, Demetris AJ, Tanabe M, Miyazawa H, Valdivia LA, Nakamura K, Starzl TE. Effect of FK 506 and antiproliferative agents for heart and liver xenotransplantation from hamster to rat. Transplant Proc. 1993;25:425-426. [PubMed] |
| 16. | Wang J, Xu R, Jin R, Chen Z, Fidler JM. Immunosuppressive activity of the Chinese medicinal plant Tripterygium wilfordii. II. Prolongation of hamster-to-rat cardiac xenograft survival by combination therapy with the PG27 extract and cyclosporine. Transplantation. 2000;70:456-464. [PubMed] |
| 17. | He X, Verran D, Hu C, Wang C, Li L, Wang L, Huang J, Sun J, Sheil AG. Synergistic effect of Tripterygium wilfordii Hook.F (TWHF) and cyclosporin A in rat liver transplantation. Transplant Proc. 2000;32:2054. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 3] [Cited by in RCA: 3] [Article Influence: 0.1] [Reference Citation Analysis (0)] |
| 18. | Qian YY, Shi BY, Liang CQ, Li N, Shi YC, Xia JM. Effects of total gulcosides of triptergium wilfordii (TII) on the reproductive organs of transplant recipients. Zhonghua Miniao Waike Zazhi. 2002;23:209-211. |
| 19. | Zhang LL, Sun JH. Synergistic effect of triptergium wilfordii hook f and cyclosporine A on hetertopic rat cardiac allograft survival. Zhonghua Shiyan Waike Zazhi. 2001;18:407-408. |
| 20. | Qian YY, Shi BY, Liang CQ, Kang XJ, Shi YC, Li N, Xia JM. Total glucosides of triptergium wilfordii in the management of rats with renal allograft. Zhonghua Miniao Waike Zazhi. 1996;17:338-340. |
| 21. | Li YX, Li N, Wu B, Li JS. The effect of Triptergium wilfordii on acute rejection in small bowel transplantation in rats. Zhonghua Xiaoer Waike Zazhi. 2000;21:49-51. |
| 22. | Wang DH, Wang JP, Wang L. Role of triptergium wilfordii hook f(T¢ò) on pancreaticoduodenal allograft in rats. Zhonghua Qiguan Yizhi Zazhi. 2000;21:220-222. |
| 23. | Shao QX, Yin L, Xu HX, Li LJ, Liu GZ, Cao YQ, Chen M. Using DcsMcAb and tripterygll multi-glycosidorum to prevent the rejection of heart-lung allogeneic transplantation in rats. Zhongguo Mianyixue Zazhi. 1998;14:356-359. |
| 24. | Li N, Li J, Li Y. [Experimental and clinical studies of small bowel allotransplantation]. Zhonghua Waike Zazhi. 1995;33:11-14. [PubMed] |
| 25. | Qian YY, Shi By, Liang CQ. Total glucosides of triptergium wilfordii (TII) as an immuno-suppressant in kidney transplantation. Zhonghua Miniao Waike Zazhi. 1995;16:268-269. |
| 26. | Chen BJ. Triptolide, a novel immunosuppressive and anti-inflammatory agent purified from a Chinese herb Tripterygium wilfordii Hook F. Leuk Lymphoma. 2001;42:253-265. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 224] [Cited by in RCA: 234] [Article Influence: 9.8] [Reference Citation Analysis (0)] |
| 27. | Ramgolam V, Ang SG, Lai YH, Loh CS, Yap HK. Traditional Chinese medicines as immunosuppressive agents. Ann Acad Med Singapore. 2000;29:11-16. [PubMed] |
| 28. | Kamada N, Calne RY. A surgical experience with five hundred thirty liver transplants in the rat. Surgery. 1983;93:64-69. [PubMed] |
| 29. | Sun JH, Zeng QH, Wu MC. Experience with orthotopic rat liver transplantation. Chin Med J (. Engl). 1990;103:142-145. [PubMed] |
| 30. | Valdivia LA, Monden M, Gotoh M, Hasuike Y, Kubota N, Endoh W, Okamura J, Mori T. Hepatic xenografts from hamster to rat. Transplant Proc. 1987;19:1158-1159. [PubMed] |
| 31. | Sankary HN, Yin DP, Chong AS, Ma LL, Blinder L, Shen JK, Foster P, Williams JW. FK506 treatment in combination with leflunomide in hamster-to-rat heart and liver xenograft transplantation. Transplantation. 1998;66:832-837. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 18] [Cited by in RCA: 18] [Article Influence: 0.7] [Reference Citation Analysis (0)] |
| 32. | Li XW, Weir MR. Radix Tripterygium wilfordii--a Chinese herbal medicine with potent immunosuppressive properties. Transplantation. 1990;50:82-86. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 33] [Cited by in RCA: 27] [Article Influence: 0.8] [Reference Citation Analysis (0)] |