Published online May 7, 2023. doi: 10.3748/wjg.v29.i17.2551
Peer-review started: December 26, 2022
First decision: March 8, 2023
Revised: March 10, 2023
Accepted: April 13, 2023
Article in press: April 13, 2023
Published online: May 7, 2023
Processing time: 132 Days and 0.9 Hours
Alcohol-related hepatitis (ARH) is a unique type of alcohol-associated liver disease characterized by acute liver inflammation caused by significant alcohol use. It ranges in severity from mild to severe and carries significant morbidity and mortality. The refinement of scoring systems has enhanced prognostication and guidance of clinical decision-making in the treatment of this complex disease. Although treatment focuses on supportive care, steroids have shown benefit in select circumstances. There has been a recent interest in this disease process, as coronavirus disease 2019 pandemic led to substantial rise in cases. Although much is known regarding the pathogenesis, prognosis remains grim due to limited treatment options. This article summarizes the epidemiology, genetics, pathogenesis, diagnosis and treatment of ARH.
Core Tip: The aim of this article is to review alcohol-related hepatitis (ARH). Despite the increased understanding of the pathogenesis of this disease process, treatment options remain limited. Our review article focuses on epidemiology, genetics, pathogenesis, diagnosis and treatment. We also discuss complications of ARH along with their optimal management and ongoing clinical trials. Further research evaluating therapeutic targets for its management are warranted.
- Citation: Chaudhry H, Sohal A, Iqbal H, Roytman M. Alcohol-related hepatitis: A review article. World J Gastroenterol 2023; 29(17): 2551-2570
- URL: https://www.wjgnet.com/1007-9327/full/v29/i17/2551.htm
- DOI: https://dx.doi.org/10.3748/wjg.v29.i17.2551
The burden of alcohol-associated liver disease continues to grow with rising cases during and even after the coronavirus disease 2019 (COVID-19) pandemic[1-2]. Some experts believe that the rise in cases during the COVID-19 pandemic will have long lasting effects on society and healthcare. Alcohol is an integral part of American culture and its widespread use cuts across socioeconomic and racial lines. Alcohol-related liver disease (ALD) can range from hepatic steatosis to end-stage liver disease. Alcohol-related hepatitis (ARH), a form of ALD, is an acute inflammatory syndrome of jaundice and liver injury occurring in patients with significant alcohol use. The severity can range from subclinical to acute severe illness, which is associated with high mortality. The recent trends in ARH hospitalizations in the United States suggest its importance in the current realm of clinical practice[1-4].
The prevalence of ARH has been on a rise even prior to COVID-19 pandemic[4]. Studies have shown a 53% increase in ARH from 2019 to 2020 with a 64% increase in the latter phase of the pandemic[2]. Multiple other studies have shown that the societal disruptions associated with the pandemic have further intensified the rise in cases of ARH. ARH can resolve with abstinence and supportive therapy; however, these measures do not guarantee recovery in all patients. Acute-on-chronic liver failure (ACLF) and development of cirrhosis are the most feared complications of ARH and is associated with significant morbidity and mortality. Thus, identifying patients early during the course of the disease is critical.
The true prevalence of ARH is difficult to assess as patients can be asymptomatic and remain undiagnosed. A study by Jinjuvadia et al[5] evaluating the temporal trends in ARH hospitalizations reported an increase in patients with ARH from 249884 in 2002 to 326403 in 2010. Their study included hospitalizations with a primary and secondary diagnosis of ARH. To estimate true prevalence, Ali et al[4] included patients only with a primary diagnosis of ARH and reported an increase in the total hospitalizations from 67070 in 2009 to 125540 in 2019. They also reported an increase in inpatient mortality from 2.48% in 2009 to 3.78% in 2019. The increase in the cases of ARH has been further intensified during the COVID-19 pandemic, especially in younger patients and in women.
ARH is also associated with a significant risk of mortality. Mortality has been estimated to be around 15% at 30 d and 39% at 1 year and has been linked to the severity of the disease[5]. Patients with mild liver injury have a 20% mortality, while patients with severe liver injury have an overall 40% mortality[6]. Mortality in patients with higher Maddrey’s discriminant function (MDF) score (> 30), a surrogate marker of severity, is estimated to be between 30%-50%[7-8].
In previous years, there has also been an increase in the healthcare burden of ARH hospitalizations. An increase was noted in the mean inpatient cost for ARH hospitalizations from $31189 in 2009 to $62229 in 2019[4]. A study by Thompson et al[9], using commercial insurance claims, examined hospitalized patients between 2006-2013 and reported the average cost per patient is $145000 in their cohort of 15546 patients. They also reported the cost surrounding death of ARH was 200%-300% higher than the average cost surrounding death.
Alcohol intake: The amount of alcohol consumption that places patients at risk of ARH is largely unknown. The estimates of the amount of alcohol consumed by patients may not be accurate as they are based on interviewing patients and family members[9]. The majority of patients with ARH consume more than 100 g/d of alcohol[10]. American Association for the Study of Liver Disease (AASLD) practice guidelines recommend suspecting ARH in women who consume > 40 g of alcohol and in men who consume > 60 g/d for ≥ 6 mo[11-12]. It is also pertinent to note that not all patients who drink heavily develop ALD. In a study by Friedman et al[13], 35% of the patients developed steatohepatitis and ARH, while only 10% of the patient’s developed cirrhosis.
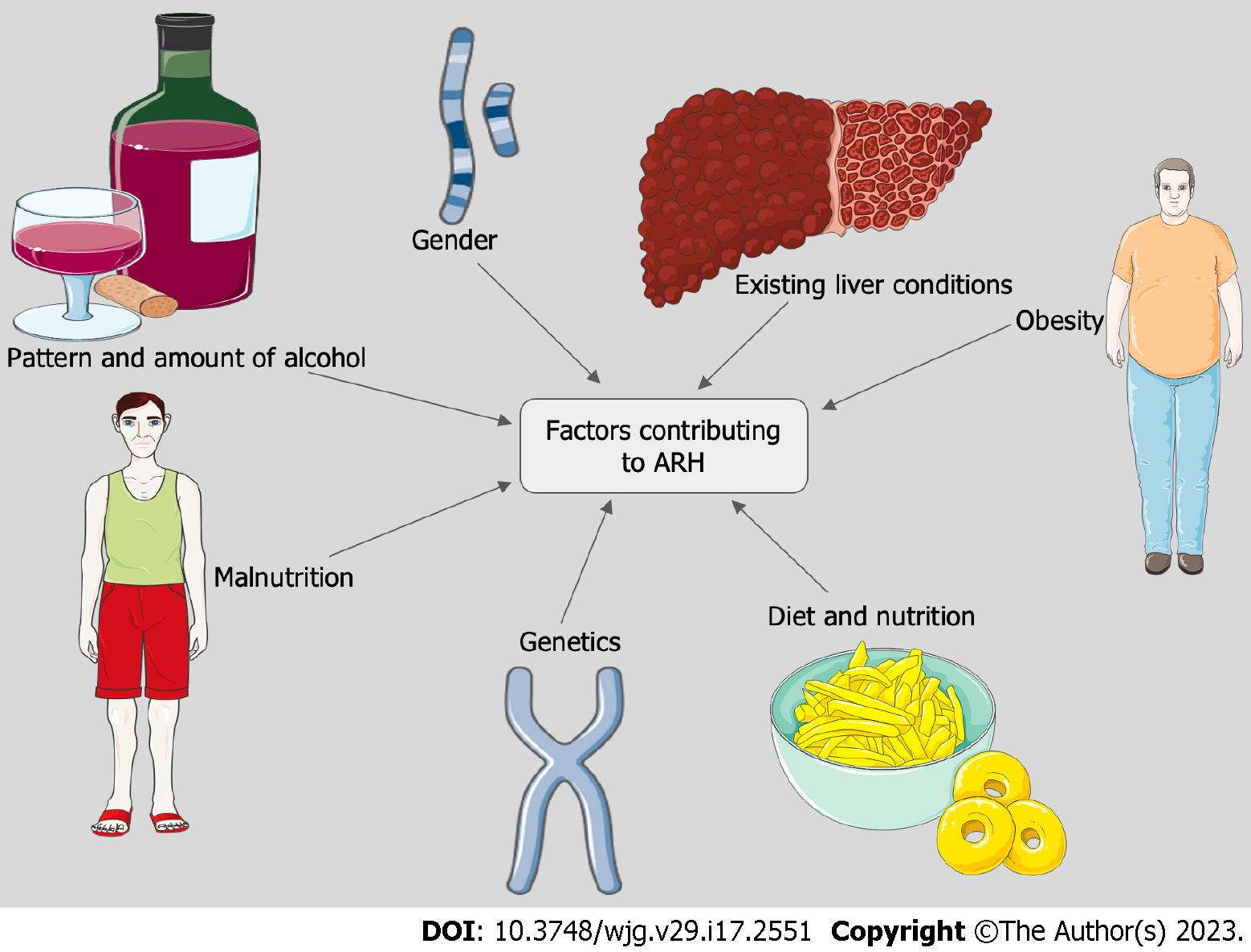
The pattern of drinking as well as the type of alcohol consumed also contributes to the risk of developing ARH. Drinking spirits or beer, binge drinking as well as drinking outside of the typical mealtimes has been associated with a higher risk of developing ALD[13-15] (Figure 1).
Gender: There are significant differences based on gender. Women are at a higher risk of developing ARH at a lower threshold compared to men. This is attributed to differences in the variability of alcohol dehydrogenase activity, sex hormones, body fat distribution and liver volume between the two genders[16-18]. There has been an increase in the cases of ARH in women in recent years. A study conducted between 2009-2019 showed an increase in the proportion of women being admitted with ARH from 29.1% to 34.1%[4].
Malnutrition: Patients who consume alcohol excessively are often malnourished and can suffer from vitamin deficiencies[19]. Mortality has been closely related to the severity of protein-energy malnutrition. Patients with severe malnutrition and ARH have an estimated mortality of 80%[20]. Additionally, depletion of hepatic vitamin A and vitamin E can aggravate liver disease[21]. Zinc deficiency in patients with excessive alcohol can lead to disruption of mucosal permeability which may play a role in the pathogenesis of ARH.
Concurrent liver disease: It is well established that the combination of alcohol misuse and chronic hepatitis C (HCV) increases the incidence of cirrhosis and hepatocellular carcinoma (HCC) and is associated with reduced survival compared to patients with either HCV or alcohol use alone[22]. Consuming upward of 50 g/d of alcohol increases the relative risk of liver disease compared to patients with HCV who do not consume alcohol[23]. Patients with chronic HCV have been noted to have worse outcomes and higher in-hospital mortality when admitted with ARH[24].
While the literature clearly demonstrated worse outcomes in patients with concomitant alcohol misuse and chronic HCV, the data regarding the effect of alcohol use on patients with chronic hepatitis B (HBV) remains understudied. A study by Iida-Ueno et al[25] revealed that patients with light to moderate alcohol consumption had a 1.5-fold increase of chronic HBV infection while patients with heavy alcohol consumption had significantly accelerated progression to cirrhosis and HCC with 1.3 to 8.4-fold increased risk. Although the mechanism by which alcohol enhances disease progression is less studied in patients with chronic HBV in comparison to patients with HCV, patients with either chronic HCV and HBV should be counseled to avoid alcohol consumption to minimize chances of liver disease progression.
Obesity, diabetes mellitus and metabolic syndrome-underlying non-alcoholic fatty liver disease, a hepatic manifestation of metabolic syndrome, can be additive to the damage sustained through alcohol misuse and lead to worse outcomes. A study by Siddiqui et al[26] reported obesity to be an independent predictor of mortality in ARH.
Seasonality: Studies have shown that there might be a seasonal trend to admissions with alcohol use. We have previously reported that in the United States, ARH was highest in the summer months, as compared to the common opinion that ARH hospitalizations are higher in the winter season[27].
Significant variability has been noted among patients in regard to the susceptibility of developing ARH as well as the severity of symptoms. This variability has prompted several studies exploring a possible genetic basis for individual response to ARH. Studies have found an alteration in the expression of claudins, osteopontin, CD209, selenoprotein, and bile duct proliferation genes in patients with ARH compared to patients with alcoholic steatosis and healthy controls without liver disease[28]. Colmenero et al[29] identified several genes that were upregulated in ARH including extracellular matrix proteins, fibrogenesis mediators, inflammatory cytokines, and apoptosis regulators. Cytochrome p450 2E1 and angiotensinogen were found to be downregulated. Additionally, certain genes were found to be correlated with the severity of disease features including tissue inhibitors of metalloproteinases-1 and growth-related oncogene α. Epigenetic studies have also shown that a liver affected by ARH has significant alterations in HNF4α-dependent genes, which play a role in impairing metabolic and synthetic function[30]. A cohort study by Beaudoin et al[31] demonstrated an increased risk for developing ARH with a higher total bilirubin in patients with variants in the patatin-like phospholipase domain-containing protein 3 (PNPLA3) and the haptoglobin genes as compared to other genetic variants associated with chronic liver disease states[32].
The PNPLA3 gene in particular has been identified to play a significant role in the pathogenesis of several liver diseases, as it belongs to a group of lipid-metabolizing enzymes. The I148M variant, a single-nucleotide polymorphism of the gene, is associated with an increased risk for developing hepatic steatosis, non-alcoholic steatohepatitis (NASH), ALD, cirrhosis, and HCC[33]. Despite these correlations, the precise function of the PNPLA3 gene is yet to be fully understood. Palmer et al[33] demonstrated that variants in the PNPLA3 gene in Hispanic-American and African-American patients is significantly associated with the development of hepatic steatosis, and other studies have shown that these groups suffer from higher mortality rates due to ALD. Levy et al[34] reported that patients of Hispanic ancestry presented with ALD at a significantly earlier age than patients of Caucasian or African-American background, though the mechanisms remain unclear and may be related to the gene variants described by Palmer et al[33] as well as health care disparities. Future studies can help to elucidate the underlying mechanisms of these discrepancies and the potential of gene-targeted therapy in the management of ARH.
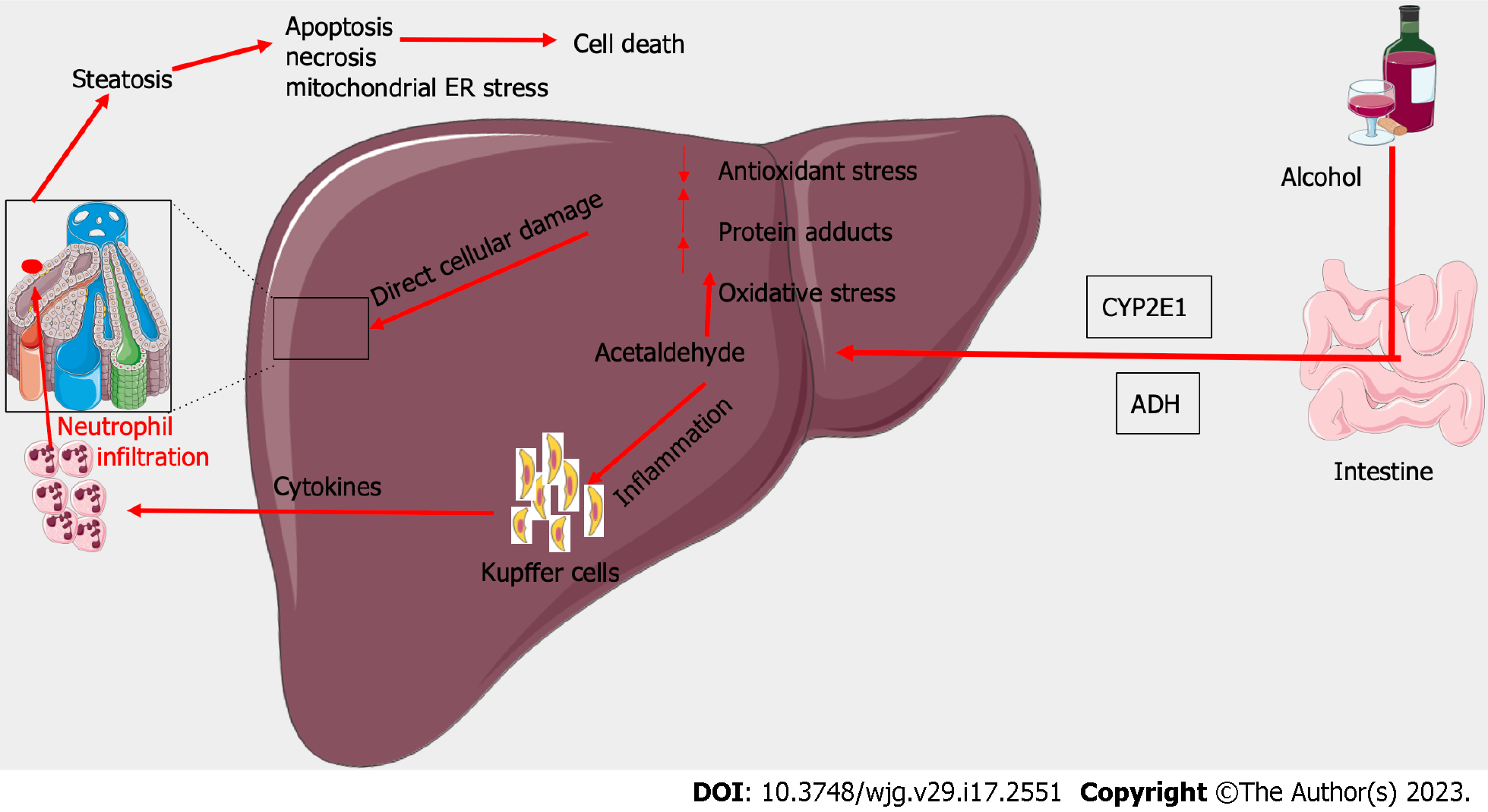
The pathogenesis of ARH involves acute inflammation superimposed on chronic ALD. Initial hepatic injury results from the metabolism of ethanol into acetaldehyde by alcohol dehydrogenase and cytochrome P450 2E1 (CYP2E1). As ethanol builds up as a result of increased consumption, acetaldehyde also accumulates in the liver and begins to exert toxic effects[35]. Acetaldehyde leads to the formation of adducts, in the form of bonds with proteins, lipids, and DNA. This in turn impairs the normal function of the affected proteins and lipids and leads to DNA damage. CYP2E1 further contributes to liver injury by the production of reactive oxygen species which leads to hepatocyte injury[36]. This is thought to occur as a result of lipid peroxidation and interaction with proteins and nucleic acids, leading to hepatocyte necrosis[37] (Figure 2).
The oxidation of ethanol and acetaldehyde leads to an increase in the levels of nicotinamide adenine dinucleotide, which enhances lipogenesis in the liver[38]. Fatty infiltration of the liver is also a result of increased gut permeability due to alcohol ingestion, leading to higher levels of lipopolysaccharides present in the portal circulation[39]. Endotoxin, a lipopolysaccharide present in cell walls of gut microbiota, forms a protein complex and binds to the CD14 receptor on Kupffer cells in the liver[40]. The activation of Kupffer cells leads to the release of tumor necrosis factor-α (TNF-α), a pro-inflammatory cytokine[41]. TNF-α exerts its hepatotoxic effects by causing hepatocyte apoptosis and stimulating the release of other cytokines[42]. Higher levels of TNF-α are correlated with increased severity of ARH, demonstrating the significance of the cytokine in the pathogenesis of ARH[43].
The stimulation of Kupffer cells also lead to an increase in polymorphonuclear leukocyte infiltrate in the liver, as a result of chemotactic factors such as interleukin-8[44]. The multifactorial process of inflammation superimposed on alcohol-induced hepatic damage leads to the impairment of liver function observed as systemic illness and laboratory abnormalities in ARH. A thorough understanding of the pathogenesis of ARH can aid in its treatment, and further studies elucidating the precise mechanisms involved may improve treatment modalities and outcomes.
Physical examination can range from benign to profoundly abnormal[45]. Patients may have signs of malnutrition/sarcopenia manifested as temporal or thenar muscle wasting. Jaundice and scleral icterus may be apparent in patients with more severe disease while subtle signs of jaundice can be noted sublingually or in the tympanic membranes in patients with less severe disease. The presence of spider angiomas, gynecomastia in men and caput medusa is uncommon in patients with ARH in the absence of cirrhosis. Right upper quadrant tenderness to palpation along the enlarged hepatic border is common, with the liver edge extending into the pelvis and crossing the midline in severe cases. Some studies have reported a bruit over the liver as a feature of severe ARH in > 50% of patients[46]. Splenomegaly can often be appreciated as well even in the absence of cirrhosis. Fluid wave, shifting dullness and frank abdominal distention signifying large-volume ascites can be seen in severe cases. Lower extremity swelling progressing to anasarca can also be seen in severe cases. Finally, asterixis can be seen in patients with significant hepatic dysfunction.
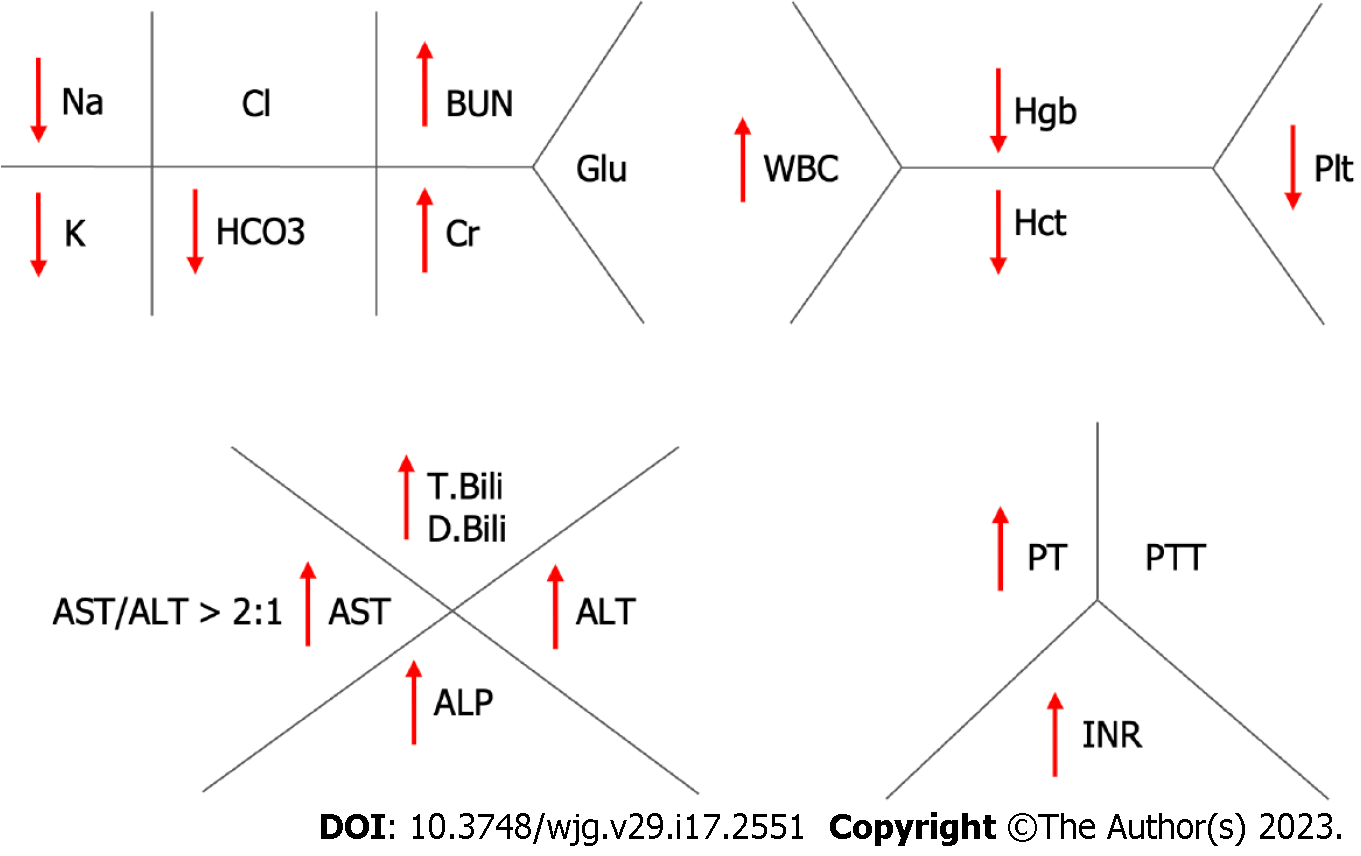
Patients with suspected ARH should undergo a broad laboratory workup including comprehensive metabolic panel, complete blood count, international normalized ratio (INR), and gamma glutamyl transferase (GGT). Serologies for HAV, HBV and HCV should also be obtained to rule out alternative/concomitant infectious causes. Typical laboratory findings in ARH consist of elevated white blood cell count, thrombocytopenia, low sodium, potassium and magnesium. Other findings include total bilirubin > 3 mg/dL, elevated alanine transaminase (ALT) and aspartate transaminase (AST) with levels usually < 400 U/L, AST:ALT ratio > 2, GGT > 100 U/mL, INR > 1.5, and albumin < 3.0 g/L[47]. Other sequelae of chronic alcohol use such as macrocytic anemia can also be seen (Figure 3).
It is pertinent to rule out alternative/concomitant causes of liver disease such as Wilson’s disease, autoimmune liver diseases and iron overload syndromes. This is especially important in patients with severe disease who may be considered for liver transplantation, as these diagnoses will have significant implications on patient’s ability to qualify for an urgent transplant as well as pre-and post-transplant management. Making these diagnoses can be challenging as patients with ARH may have multiple metabolic derangements. For example, patients with ARH may have decreased ceruloplasmin levels in the setting of hepatic dysfunction leading to suspicion of Wlison’s disease. If there is a significant clinical concern for Wilson’s disease, patients should undergo a slit lamp examination for Kayser-Fleischer rings as well as a 24-h urine copper collection. Ferritin levels as well as iron saturations in patients with ARH can be markedly elevated in ARH therefore genetic testing for HFE gene mutation should be considered[48]. A clinical suspicion of autoimmune liver disease, especially in the setting of elevated autoimmune markers [antinuclear antibody (ANA), anti-smooth muscle antibody (SMA), antimitochondrial antibody, anti-liver kidney microsomal antibody], should prompt consideration of liver biopsy. Alpha-1-antitrypsin levels can be artificially elevated in the setting of acute illness; genetic testing should be considered if clinical suspicion is sufficiently high.
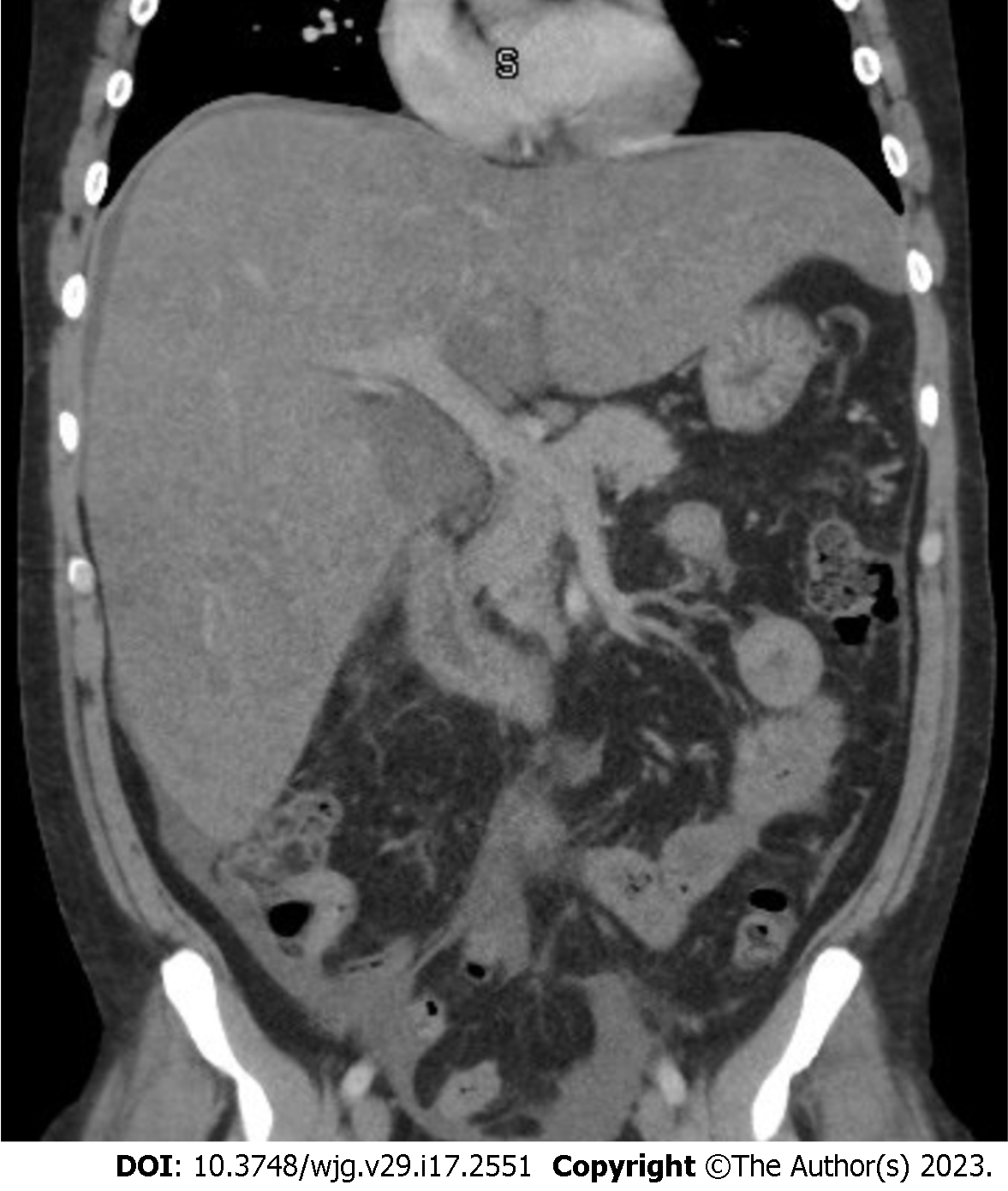
Abdominal imaging can be effective in the initial evaluation of suspected ARH however, it cannot serve as the sole basis for the diagnosis. Abdominal ultrasound (US) computed tomography scan and/or magnetic resonance imaging will likely reveal and enlarged liver with smooth contours and decreased attenuation, and in more severe cases, an enlarged spleen, ascites and other sequelae of portal hypertension. The presence of smooth hepatic borders, as opposed to the nodular contour, can help to differentiate ARH from cirrhosis[49] (Figure 4).
The European Association for the Study of the Liver clinical practice guideline in 2014 stated that the “presence of acute steatohepatitis can be suspected on the clinical and biochemical grounds, but a definitive diagnosis of acute steatohepatitis requires liver biopsy”[50]. AASLD practice guidelines in 2019 recommend performing biopsy for confirmation if there are potential confounding factors such as[11]: (1) Ischemic hepatitis (e.g. severe upper GI bleed, cocaine use within 7 d or hypotension); (2) Suspected drug-induced liver disease; (3) Uncertain alcohol use assessment; and (4) Atypical laboratory tests (AST < 50 or > 400 IU/L, AST/ALT < 1.5), ANA > 1:160 or SMA > 1:80. Liver biopsy is controversial, especially in patients with coagulopathy and ascites. Percutaneous biopsy may be performed in most patients, but transjugular biopsy is preferred in patients with severe thrombocytopenia and prolonged INR. Liver biopsy is also recommended in patients suspected of having additional etiology for ACLF.
Histology in patients with ALD can range from steatosis to steatofibrosis[51-52]. Steatofibrosis is defined as steatosis with fibrosis, with or without hepatitis. Histological features include steatosis, hepatocyte ballooning, neutrophil-rich inflammation in the lobular parenchyma, necrosis, apoptosis and Mallory-Denk bodies (MDB)[53]. The predominant mode of hepatocyte injury is via ballooning degeneration, followed by lytic necrosis and apoptosis. The hepatocytes become swollen, with clumping of intermediate filaments and loss of cytokeratin 8 and 18[51]. The swelling in hepatocytes can be attributed to severe ATP depletion and an increase in intra-cellular calcium, resulting in loss of plasma membrane volume control, oncotic necrosis and disruption of the intermediate filament network. The majority of changes in ALD are seen in the centrilobular region of hepatic lobe as they contain enzymes such as alcohol dehydrogenase, which are critical in alcohol metabolism[54].
It is pertinent to note there are histological similarities between NASH and ARH[53]. The differentiation is based on clinical information regarding alcohol intake; however some features are seen more commonly in ARH as compared to NASH (Table 1).
| Histological features | NASH | ARH |
| Canalicular cholestasis | Less common | More common |
| Mallory denk body | Less common | More common |
| Ductular reaction | Less common | More common |
| Fibrosis in portal tract | Less common | More common |
| Sclerosing hyaline necrosis | Less common | More common |
| Veno-occlusive lesions | Less common | More common |
| Severe steatosis | More common | Less common |
| Glycogenated nuclei | More common | Less common |
| Lipogranulomas | More common | Less common |
| Fibrosis | Lattice pattern | Solid pattern |
Apoptosis can be triggered by oxidative stress. Apoptotic hepatocytes, also known as councilman bodies or acidophil bodies, are visualized as shrunken cells, chromatin condensation and cellular fragmentation[55]. MDB contain eosinophilic material, located primarily in the perinuclear location in the cytoplasm of hepatocytes[56]. They are formed due to the misfolding of aggregated keratin filaments. MDB demonstrate immunoreactivity with antibodies to keratin 8, 18 and p62. Portal inflammation is milder as compared to other etiologies of liver disease. Portal inflammation, when present, is accompanied by ductular reaction and periportal inflammation[57]. Other features of this disease include glycogenated nuclei, megamitochondria, cholestasis and hemosiderin deposits[58]. Iron deposition can also occur in patients with ALD[59-60]. Based on the index of suspicion, concomitant hereditary hemochromatosis should be ruled out.
As the disease progresses, fibrosis begins in perivenular region (Zone 3) and extends to the perisinusoidal region in patients giving classic “chicken-wire fibrosis”. Trichrome stain visualizes the fibrosis. If the injury continues, fibrosis progresses resulting in nodule formation and finally cirrhosis. An orcein stain, can be beneficial in later stages to differentiate broad bands of fibrosis from areas of collapse, which can occur in superimposed ARH[54].
A histological severity score including cytokeratin 8 and 18, has shown good accuracy in predicting 90-d survival[61]. Alcoholic hepatitis histologic score was created using parameters such as degree of fibrosis, degree of neutrophil inflammation, type of bilirubin stasis, and presence of mitochondria[62]. This score has been shown to be predictive of 90-d mortality. Trépo et al[63] combined the expression patterns of 123 genes with the model for end-stage liver disease (MELD) score to create gene-signature plus MELD scoring system. This helped in discriminating patients with poor and good 90-d survival with an area under the curve of 0.86. This score outperformed other models including MELD plus Lille. This is not yet commercially available for use, however, may hold promise for the future.
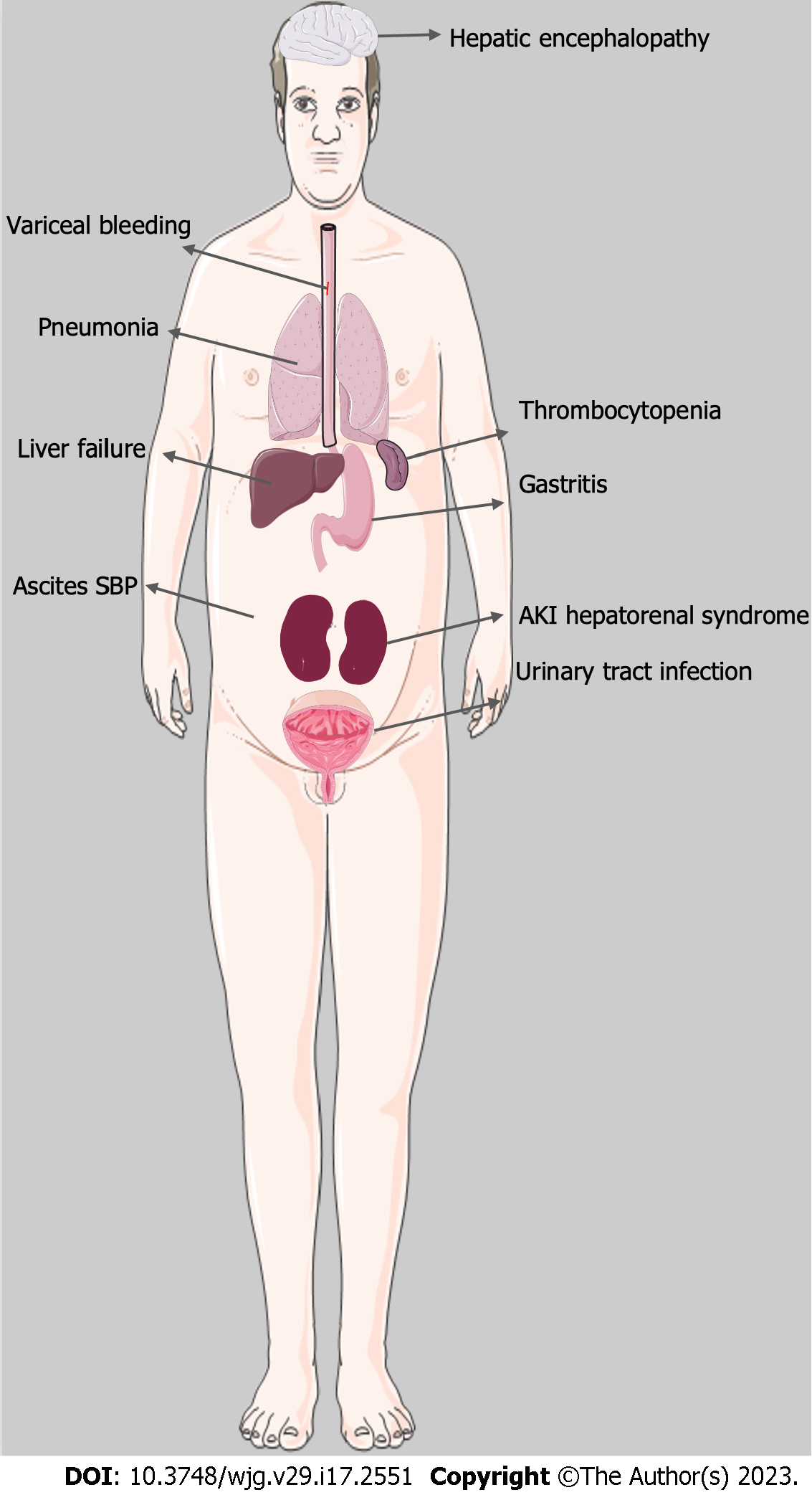
Complications of ARH include infection, kidney injury, gastrointestinal bleeding, and acute liver failure amongst others (Figure 5).
Patients with ascites are at risk for developing spontaneous bacterial peritonitis (SBP), a life-threatening infection of the peritoneal cavity. SBP is almost exclusively seen in patients with ascites and is a result of bacterial translocation of enteric microflora into the ascitic fluid with concurrent failure of liver-related defensive mechanisms due to cirrhosis[64]. Additionally, patients with ARH are often treated with corticosteroids, further suppressing the immune response and increasing the risk of serious infection. Treatment of SBP is usually with a third-generation cephalosporin, such as ceftriaxone, which can also be used prophylactically in patients with decompensated cirrhosis or known patient with cirrhosis who presents with gastrointestinal bleeding.
Despite the well-known association of SBP with ALD, several studies demonstrate that infection due to pneumonia and urinary tract infection (UTI) is more common in severe ARH than SBP[65]. Infection overall is a major cause of mortality in severe ARH, with the majority being bacterial in nature[66-67]. A recent meta-analysis[68] found pneumonia to be the most common infection in severe ARH at 23%, followed by UTI at 10%. In comparison, SBP accounted for only 7% of infections. Patients with acute severe ARH have also been shown to have a greater level of immunosuppression compared to patients with cirrhosis, as they were found to have reduced T-lymphocyte and neutrophil activity[69]. Furthermore, obesity has been associated with increased susceptibility to infection in patients with ARH[70]. A high index of suspicion is needed in monitoring infectious signs and symptoms in patients with ARH as these patients often lack typical signs of infection such as fever or abdominal pain and may present instead with progressive hepatic decompensation. A low threshold for initiation of antibiotic therapy is advised and can be initiated empirically while infectious work-up is in progress.
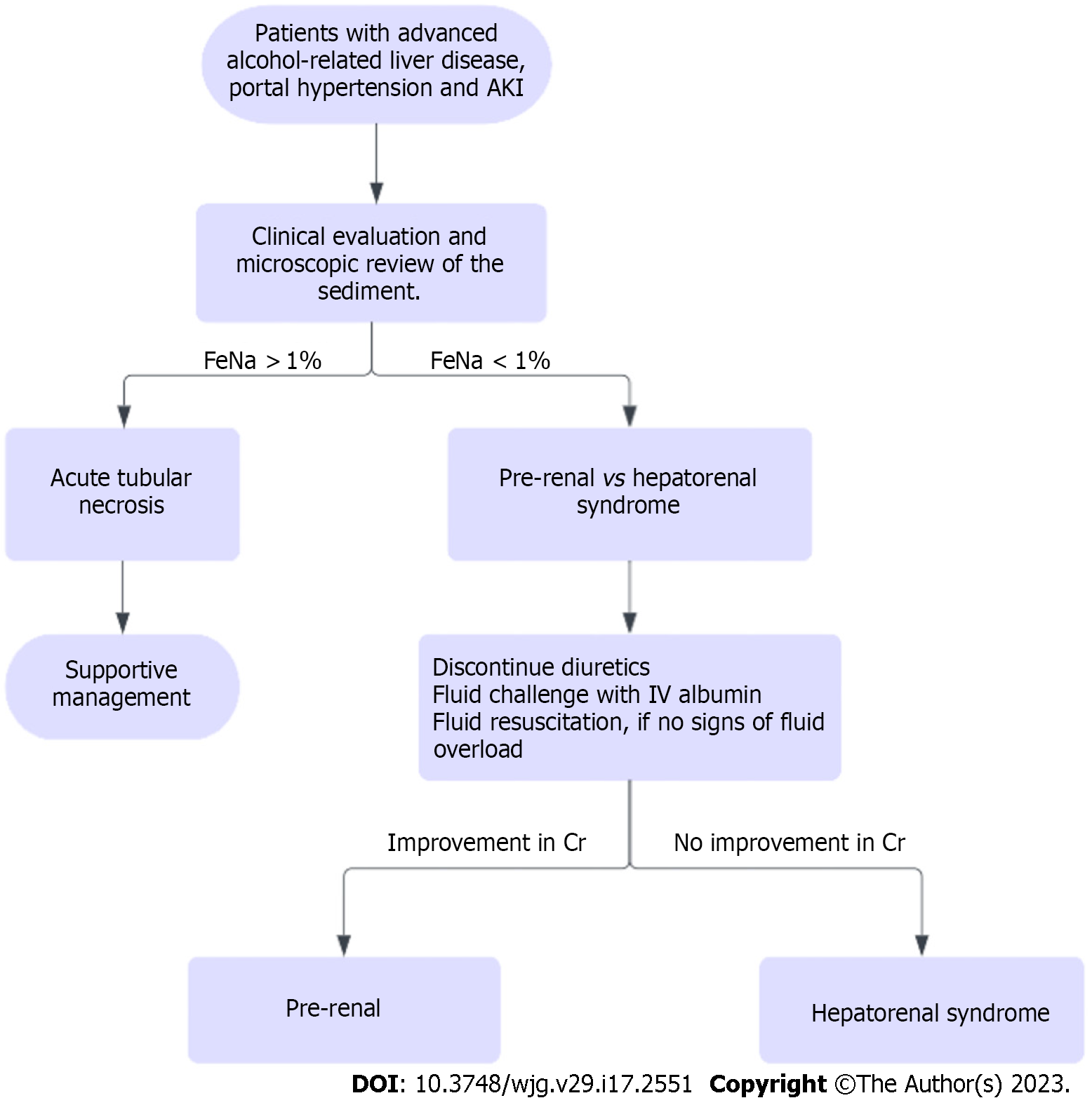
Acute kidney injury (AKI) is a common complication in hospitalized patients with ARH and can be due to pre-renal, intrinsic renal and post-renal causes. Pre-renal AKI continues to be the most common etiology, while the most feared etiology is hepatorenal syndrome (HRS). These entities can be differentiated via volume expansion challenge (25% albumin 1 g/kg for 48 h). Renal US can be used to rule out post-renal causes. Diuretics should be discontinued in patients with AKI.
Another etiology that should be considered in patients with ARH is acute tubular necrosis (ATN) secondary to cholemic nephrosis, or bile salt nephropathy and should be suspected in patients with prolonged severe cholestasis. Hyperbilirubinemia can also lead to formation of tubular casts, which can lead to ATN[71]. Evaluation of fractional excretion of sodium/urea as well as urine microscopy is beneficial to identify the etiology of kidney disease. Workup for AKI in patients with ARH is presented in Figure 6.
The most feared complication, HRS, results from renal artery vasoconstriction secondary to splanchnic vasodilation seen in liver disease[72]. Patients with ARH and other ALD often develop dilation of the splanchnic vasculature in response to increased hepatic resistance, which in turn leads to activation of the renin-angiotensin-aldosterone system, sympathetic nervous system, and arginine vasopressin system. The activation of these vasoconstrictor systems leads to constriction of the renal arteries and subsequent AKI due to decreased renal blood flow often in the setting of decreased cardiac output[73]. The development of AKI in patients with ARH is associated with a significantly higher mortality rate compared to patients without AKI, 65% vs 7% (P < 0.0001) respectively[74].
Treatment of HRS is complex, with most studies supporting the use of vasoconstrictors in combination with albumin administration. Terlipressin, a vasopressin analogue, has shown promising results in the treatment of HRS as it was found to be superior to both placebo and octreotide[75]. It has recently been approved in the United States by the Food and Drug Administration for the management of HRS. Liver transplantation (LT) is the treatment of choice in patients with HRS as renal dysfunction is potentially reversible with the correction of hepatic failure[76]. Interestingly, obesity has been found to be associated with decreased survival in acute ARH, and this is thought to be due to an increased risk of developing renal failure[77].
Severe ARH can lead to ACLF, a condition that is associated with increased mortality in patients with ARH and is commonly seen during the course of the disease[78]. ACLF manifests as worsening jaundice and coagulopathy and is usually triggered by a precipitating event. Current literature shows that ARH serves as a common precipitating event for the development of ACLF[76]. Studies have demonstrated that 65% of patients with severe ARH had ACLF at the time of diagnosis or within a 6-mo follow-up period[77]. Given the poor prognosis of ACLF, early liver transplantation is becoming more commonplace in the treatment of severe ARH, with recent literature supporting its use with stringent patient selection criteria[79-80].
Many patients with ARH also have portal hypertension or will develop portal hypertension at some point during their disease course potentially leading to variceal hemorrhage. Overall one-month mortality in moderate to severe ARH is 23%, with the most common causes of death being liver failure, gastrointestinal bleeding, and infection[81] The one-month mortality in non-severe ARH is 6%, however the rate increases to 13% after one year[82].
Long-term follow-up in patients with ARH over 4 years revealed the probability of developing cirrhosis to be 10%-20% per year and up to 70% of patients will ultimately develop cirrhosis[83]. In another study, about 40% of the patients were diagnosed with cirrhosis on biopsy, 5 years after the episode of ARH[84]. The probability of developing cirrhosis is associated with continued alcohol use, as Rehm et al[85] demonstrated in a meta-analysis that there is a close dose-dependent relationship between ongoing alcohol use and the risk of progression of ALD to liver cirrhosis. Parés et al[86] found an 18% risk of progression to cirrhosis in patients who abstained from alcohol after 20 mo compared to 23% in patients with continued alcohol consumption.
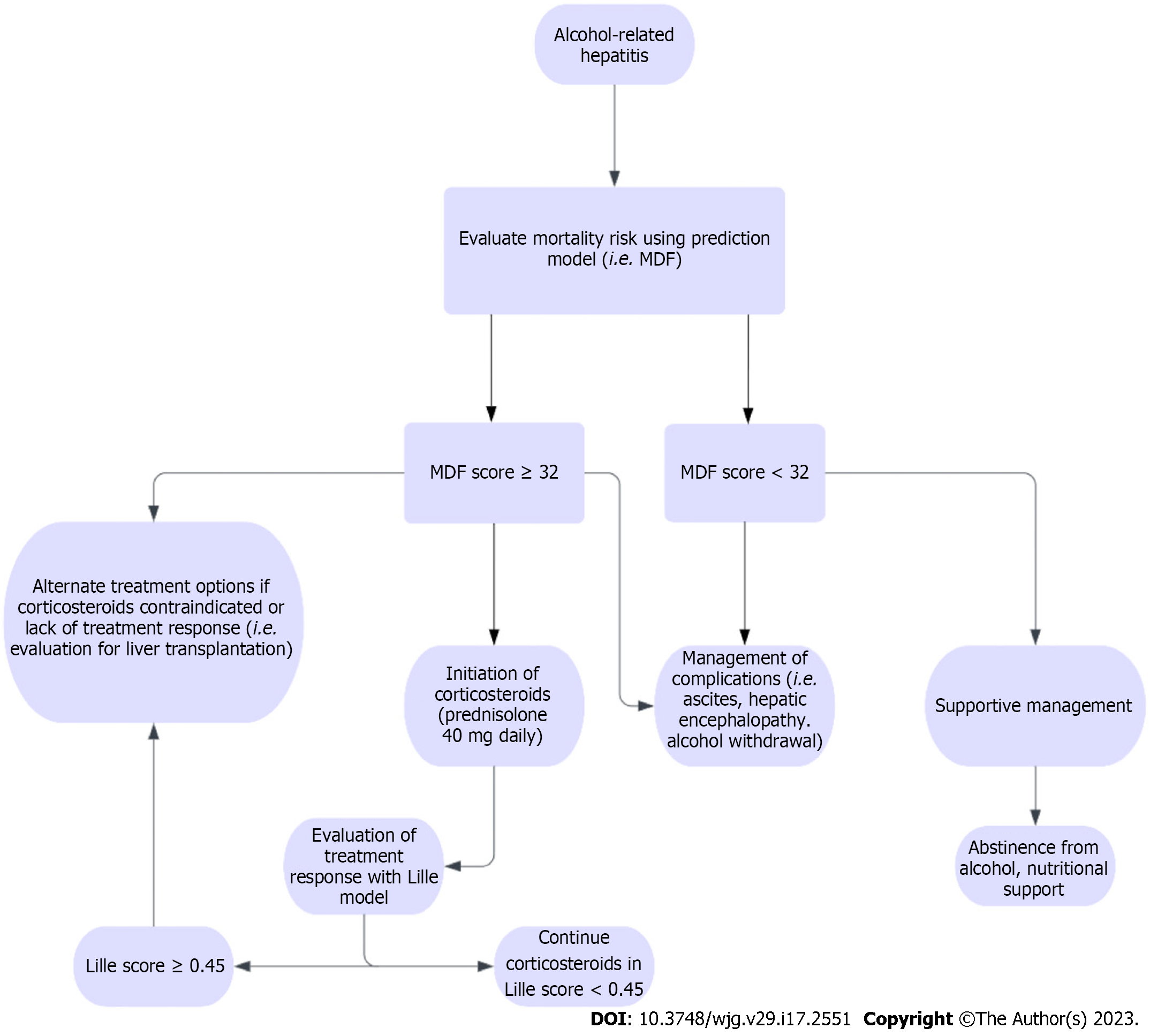
MDF is a commonly used prognostic indicator in the management of ARH as it is specific for ARH and has shown to be relatively accurate in predicting short-term outcomes[87]. The MDF score incorporates two of the major indices of liver function, prothrombin time (PT) and total bilirubin with the formula 4.6 × (prolongation of PT in seconds) + bilirubin (µmol/L)/17. MDF score of < 32 signifies mild to moderate ARH with 30-d survival estimated at 80%-100%. MDF score ≥ 32 is considered to be severe ARH with a 30-d survival of only 50%, and these patients warrant corticosteroid administration for treatment of their disease[87] (Table 2).
| Score | Variables | Mortality rate |
| MDF | PT, total bilirubin | 50% at 30 d for score ≥ 32 |
| Lille model | Age, albumin, change in total bilirubin, renal insufficiency, MDF score | 75% at 6 mo for score ≥ 0.45 |
| Model for end-stage liver disease | Creatinine, total bilirubin, INR | 20% at 90 d for score > 21 |
| Glasgow alcoholic hepatitis score | Age, white blood cell count, BUN, total bilirubin, PT | 48% at 28 d for score ≥ 9 |
| ABIC score | Age, total bilirubin, creatinine, INR | 75% at 90 d for score > 9 |
Lille Model is used to evaluate treatment response to corticosteroids and includes age, initial serum albumin, change in total serum bilirubin at day 7 of treatment, renal insufficiency, and initial MDF score[88]. Patients with a Lille score of < 0.45 had an average 6-mo survival rate of 85%, compared to just 25% in patients who scored ≥ 0.45 after 7 d of treatment (P < 0.0001). The Lille model is primarily used in conjunction with the MDF to guide treatment of ARH. Patients who score ≥ 0.45 after receiving corticosteroid therapy for 7 d are considered for discontinuation of steroids and initiation of alternative treatment modalities, as continuation of steroids is of little benefit in these patients.
MELD was initially created to assess the mortality risk in patients with portal hypertension undergoing transjugular intrahepatic portosystemic shunts procedure however, it has now become a validated tool in assessing patients with a variety of liver pathologies[89]. MELD differs from MDF in that it incorporates serum creatinine and uses INR rather than PT. The formula for MELD is 9.57 × loge (creatinine) + 3.78 × loge (total bilirubin) + 11.2 × loge (INR) + 6.43. In comparison to the MDF, the MELD has been shown to be superior in predicting outcomes of patients with ARH. Several studies have demonstrated this effect, further supporting the use of MELD in patients with ARH[90-94]. Patients with a MELD score ≤ 11 have mild disease associated with a very low mortality rate. Patients who score > 21 are considered to have severe ARH.
Glasgow alcoholic hepatitis score (GAHS) was developed in order to present a model with increased specificity than MDF and a more accurate cutoff score than MELD. The GAHS incorporates many of the same parameters as the MDF and MELD, but differs in its inclusion of the peripheral white blood cell count[95]. The inclusion of an inflammatory component is thought to provide a more accurate predictor of mortality in ARH and has been shown to be superior to MDF in predicting 28-d and 84-d mortality. The cutoff point delineating severe ARH from non-severe is a GAHS score of ≥ 9.
Age, serum bilirubin, INR, serum creatinine (ABIC) score is another scoring system that was developed to predict mortality in patients with ARH[96]. The ABIC score includes liver function as well as kidney function and has a formula of (age × 0.1) + (serum bilirubin × 0.08) + (serum creatinine × 0.3) + (INR × 0.8). This model is validated for predicting 3-mo survival rates in ARH based on stratification of the severity of disease. Patients are classified based on their ABIC score into low (< 6.71), intermediate (6.71-8.99), and high (> 9.0) mortality risk. These classes correspond to a 90-d mortality of 0%, 30%, and 75%, respectively. Despite its efficacy in death risk stratification, the ABIC score does not play a major role in treatment or in determining whether corticosteroid administration is indicated.
The management of ARH involves reducing hepatic inflammation by targeting pro-inflammatory and immunologic substances acutely and reversing alcohol-induced hepatotoxicity in the long term. The mainstay of treatment in ALD is abstinence from alcohol. A retrospective study by Lackner et al[97] demonstrated a 5-year mortality rate of 13% in early ALD and 43% in decompensated ALD. Abstaining from alcohol showed an improvement in survival in patients with both early and decompensated ALD. A meta-analysis by Xie et al[98] determined that patients with ALD see an improvement in survival rates after approximately 1.5 years of abstinence. However, studies have also shown that histological improvements in ALD can be observed in patients as soon as 2 wk after discontinuation of alcohol consumption[99].
Many of the traditional pharmacologic agents used to promote abstinence from alcohol such as disulfiram, naltrexone, acamprosate, and topiramate are of limited use in ALD as they may be hepatotoxic and lack data supporting their usage[100]. Baclofen, a selective gamma-aminobutyric acid B receptor agonist, has been studied as a potential agent of choice in patients with ALD as it has minimal hepatic clearance. A recent multi-site randomized control trial[101] of patients with and without ALD demonstrated that low-medium dose baclofen at 30-75 mg daily led to a significant improvement in alcohol abstinence.
Nutritional supplementation is a vital component in the treatment of ALD. Malnutrition is common in patients with ALD, and the severity of malnourishment is correlated with the severity of liver disease[102]. The AASLD recommends a diet of at least 2000 kcal/d with at least 1.5 g protein/kg/d in patients with ALD[103]. These patients suffer from nutritional deficiencies and appropriate nutritional supplementation should be provided as nutritional deficiencies have been shown to be associated with worse outcomes[104].
Treatment of mild to moderate ARH, as defined by an MDF score < 32, consists mainly of supportive care and management of complications such as ascites, hepatic encephalopathy, and acute alcohol withdrawal syndrome[105]. Ascites is managed primarily through sodium restriction and diuresis. Patients with ascites generally have decreased sodium excretion, warranting a daily sodium restriction of 2-4 g/d. AASLD guidelines recommend that for the first episode of ascites, aldosterone antagonists alone can generate an adequate response with few side effects[106]. On the contrary, if the patient has long-standing ascites, anasarca or hepatic hydrothorax, the recommended regimen is a combination of an aldosterone antagonist and loop diuretic.
Hepatic encephalopathy is managed primarily with lactulose and rifaximin in order to promote the excretion of toxic metabolites via stool and improve the biodiversity of gut flora. Acute alcohol withdrawal is treated with benzodiazepines such as lorazepam on a scheduled regimen or symptom-triggered dose in accordance with protocols such as the Clinical Institute Withdrawal Assessment. Lorazepam has been shown to be the safest agent in its class in patients with liver disease due to minimal hepatotoxicity and is therefore the drug of choice[107]. Use of long-acting benzodiazepines, such as chlordiazepoxide, should be avoided in patients with significant hepatic dysfunction due to decreased rate of hepatic metabolism and potential for rapid accumulation leading to progressive decline in mental status[108].
The most well-studied treatment modality in severe ARH, classified as an MDF score ≥ 32, is the use of corticosteroids. Its use was validated in the “Steroids or pentoxifylline for alcoholic hepatitis” (STOPAH)[109] trial, which included 1053 subjects with ARH. There was a 28-d mortality benefit in patients treated with prednisolone. On the contrary, no mortality benefit was observed in the prednisolone group after 90 d or 1 year, adding further evidence that the positive effects of steroids in severe ARH are limited to short-term (Figure 7).
A meta-analysis of 5 randomized control trials[110] revealed a significant improvement in 28-d survival among patients with severe ARH treated with corticosteroids (79.97% ± 2.8% vs 65.7% ± 3.4%, P = 0.0005). A more recent meta-analysis[111] of 11 randomized control trials found similar results in short-term mortality benefit, however also demonstrated that corticosteroid treatment did not decrease mortality rate after 6 mo. The primary mechanism by which corticosteroids exert their effect in severe ARH is by blunting the immune response and inflammation that contributes to mortality[112]. The standard regimen for treatment of severe ARH is administration of prednisolone 40 mg daily[113]. Contraindications to steroid administration include active infection, gastrointestinal bleeding, acute pancreatitis, and renal failure, as steroids can acutely exacerbate these conditions[114].
STOPAH109 trial reported that incidence of serious infections occurred in 13% of patients in the prednisolone group compared to 7% in patients who were not treated with prednisolone (P = 0.002). However, a recent meta-analysis[115] found that corticosteroid administration in severe ARH reduced 28-d mortality from liver-related death with no significant change in mortality from bacterial infection or gastrointestinal bleeding. The study also found a higher incidence of fungal infection in steroid-treated patients. Pentoxifylline was previously used in the management of ARH. Its use has been limited based on the results of STOPAH trial, in which use of pentoxifylline was not associated with mortality benefit.
LT has been controversial in the treatment of ARH, as patients with ALD have historically been considered poor candidates for transplantation due to concern for continued alcohol use after transplantation. However, recent trends have shown an opportunity for LT to gain a larger role in the treatment of ARH with strict selection criteria for candidates, as demonstrated in several studies[116]. A prospective trial by Mathurin et al[117] showed a significantly improved 6-mo survival rate in patients with severe ARH (MDF > 32, Lillie > 0.45) who received LT compared to controls without LT (77% and 23% respectively, P < 0.001). However, selection criteria were remarkably strict, as only patients with no prior episodes of hepatic decompensation, strong family support, complete commitment to abstinence, and consensus among all care providers were accepted for LT, which was less than 2% of all patients admitted with ARH during the study period. A more recent trial by Weeks et al[118] demonstrated a 97% 1-year survival rate in patients who received LT for severe ARH, however had an alcohol use relapse rate at 17% due to less strict inclusion requirements (including patients with recent hepatic decompensation). A recent large multi-center retrospective analysis (ACCELERATE-AH)[119] found a 94% 1-year survival rate and 84% 3-year survival rate with a 11% relapse rate. Of note, median MELD score was 39 and median Lille score was 0.82 for patients who received LT.
In summary, LT should be considered for patients with life-threatening ARH who have failed medical therapy and who have a low likelihood of alcohol relapse based on predetermined medical and social criteria. The proportion of candidates meeting these criteria may be relatively low, however stringent patient selection can aid in avoiding improper use of donor organs while dramatically improving survival rates. A study reported that the rates of relapse-free survival and hazardous relapse-free survival did not differ between the patients who received early transplantation compared to those who were transplanted after 6 mo of abstinence[120].
Multiple therapies are currently under clinical trial for the management of ARH. While the discussion regarding all the therapies under investigation is beyond the scope of this article, information regarding the various mechanisms and clinical trials currently targeting this disease is presented in Table 3 below.
| Drugs | Mechanisms | Clinical trials |
| Anakinra and Canakinumab | Reduce hepatic inflammation | NCT01809132[121], NCT04072822[122], NCT03775109[123] |
| N-acetyl cysteine, S-adenosyl methionine (SAM-E) | Reduce oxidative stress | NCT03069300[124], NCT00851981[125], NCT02024295[126] |
| DUR-928 | Epigenetic modulation of lipid homeostasis, inflammation, cell survival, tissue regeneration | NCT03432260[127], NCT03917407[128] |
| Granulocyte colony stimulating growth factors, interleukin-22 and obeticholic acid | Boosts liver regeneration | NCT02442180[129], NCT03703674[130], NCT04066179[131], NCT02655510[132], NCT01918462[133], NCT02039219[134] |
| Rifaximin, zinc supplementation, bovine colostrum, probiotics, and fecal microbiota transplant | Targets the gut-liver axis | NCT02116556[135], NCT02485106[136], NCT01968382[137], NCT02473341[138], NCT01922895[139], NCT02335632[140], NCT02458079[141], NCT03091010[142], NCT03827772[143] |
ARH remains a disease process with high morbidity and mortality. Despite research advances in pathogenesis of this disease process, limited progress has been made in management of this condition. Abstinence remains critical in maximizing the chances of hepatic recovery; corticosteroids continue to play a role in select populations. Given the grim prognosis and increasing prevalence of ARH, research efforts are aiming at inhibiting inflammatory cytokine pathways that lead to progressive hepatic damage. In the meantime, many centers around the world are resorting to liver transplantation as salvage therapy. The COVID-19 pandemic exacerbated the pre-existing trend of rising cases of ARH. Urgent societal and government interventions are needed to prevent the ongoing rise in ARH cases in addition to intensifying research efforts in identifying and testing therapeutic targets for this complex disease process.
We thank Dino Dukovic for his expertise and assistance in writing the manuscript.
Provenance and peer review: Invited article; Externally peer reviewed.
Peer-review model: Single blind
Specialty type: Gastroenterology and hepatology
Country/Territory of origin: United States
Peer-review report’s scientific quality classification
Grade A (Excellent): 0
Grade B (Very good): 0
Grade C (Good): C, C, C
Grade D (Fair): 0
Grade E (Poor): 0
P-Reviewer: Li M, China; Liu W, China S-Editor: Li L L-Editor: A P-Editor: Zhang XD
| 1. | Moon AM, Curtis B, Mandrekar P, Singal AK, Verna EC, Fix OK. Alcohol-Associated Liver Disease Before and After COVID-19-An Overview and Call for Ongoing Investigation. Hepatol Commun. 2021;5:1616-1621. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 41] [Cited by in RCA: 43] [Article Influence: 10.8] [Reference Citation Analysis (0)] |
| 2. | Sohal A, Khalid S, Green V, Hagino J, Chaudhry H, Gulati A, Roytman M. Soaring rates of alcohol-related hepatitis in the latter phase of the COVID-19 pandemic: A new normal? JGH Open. 2023;7:148-151. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 3] [Cited by in RCA: 2] [Article Influence: 1.0] [Reference Citation Analysis (0)] |
| 3. | Gonzalez HC, Zhou Y, Nimri FM, Rupp LB, Trudeau S, Gordon SC. Alcohol-related hepatitis admissions increased 50% in the first months of the COVID-19 pandemic in the USA. Liver Int. 2022;42:762-764. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 9] [Cited by in RCA: 28] [Article Influence: 9.3] [Reference Citation Analysis (0)] |
| 4. | Ali H, Pamarthy R, Bolick NL, Farooq MF. Ten-year trends and prediction model of 30-day inpatient mortality for alcoholic hepatitis in the United States. Ann Gastroenterol. 2022;35:427-433. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Reference Citation Analysis (2)] |
| 5. | Jinjuvadia R, Liangpunsakul S; Translational Research and Evolving Alcoholic Hepatitis Treatment Consortium. Trends in Alcoholic Hepatitis-related Hospitalizations, Financial Burden, and Mortality in the United States. J Clin Gastroenterol. 2015;49:506-511. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 106] [Cited by in RCA: 112] [Article Influence: 11.2] [Reference Citation Analysis (1)] |
| 6. | Mathurin P, Duchatelle V, Ramond MJ, Degott C, Bedossa P, Erlinger S, Benhamou JP, Chaput JC, Rueff B, Poynard T. Survival and prognostic factors in patients with severe alcoholic hepatitis treated with prednisolone. Gastroenterology. 1996;110:1847-1853. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 214] [Cited by in RCA: 199] [Article Influence: 6.9] [Reference Citation Analysis (0)] |
| 7. | Morgan MY. The prognosis and outcome of alcoholic liver disease. Alcohol Alcohol Suppl. 1994;2:335-343. [PubMed] |
| 8. | Shah NJ, Royer A, John S. Alcoholic Hepatitis. 2023. In: StatPearls [Internet]. Treasure Island (FL): StatPearls Publishing; 2023. [PubMed] |
| 9. | Thompson JA, Martinson N, Martinson M. Mortality and costs associated with alcoholic hepatitis: A claims analysis of a commercially insured population. Alcohol. 2018;71:57-63. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 15] [Cited by in RCA: 30] [Article Influence: 4.3] [Reference Citation Analysis (0)] |
| 10. | Midanik L. The validity of self-reported alcohol consumption and alcohol problems: a literature review. Br J Addict. 1982;77:357-382. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 388] [Cited by in RCA: 350] [Article Influence: 8.1] [Reference Citation Analysis (0)] |
| 11. | Naveau S, Giraud V, Borotto E, Aubert A, Capron F, Chaput JC. Excess weight risk factor for alcoholic liver disease. Hepatology. 1997;25:108-111. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 449] [Cited by in RCA: 436] [Article Influence: 15.6] [Reference Citation Analysis (0)] |
| 12. | Crabb DW, Im GY, Szabo G, Mellinger JL, Lucey MR. Diagnosis and Treatment of Alcohol-Associated Liver Diseases: 2019 Practice Guidance From the American Association for the Study of Liver Diseases. Hepatology. 2020;71:306-333. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 281] [Cited by in RCA: 567] [Article Influence: 113.4] [Reference Citation Analysis (0)] |
| 13. | Friedman SL, Neuschwander-Tetri BA, Rinella M, Sanyal AJ. Mechanisms of NAFLD development and therapeutic strategies. Nat Med. 2018;24:908-922. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 1376] [Cited by in RCA: 2914] [Article Influence: 416.3] [Reference Citation Analysis (1)] |
| 14. | Barrio E, Tomé S, Rodríguez I, Gude F, Sánchez-Leira J, Pérez-Becerra E, González-Quintela A. Liver disease in heavy drinkers with and without alcohol withdrawal syndrome. Alcohol Clin Exp Res. 2004;28:131-136. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 62] [Cited by in RCA: 60] [Article Influence: 2.9] [Reference Citation Analysis (0)] |
| 15. | Wechsler H, Austin SB. Binge drinking: the five/four measure. J Stud Alcohol. 1998;59:122-124. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 119] [Cited by in RCA: 112] [Article Influence: 4.1] [Reference Citation Analysis (0)] |
| 16. | Mezey E, Kolman CJ, Diehl AM, Mitchell MC, Herlong HF. Alcohol and dietary intake in the development of chronic pancreatitis and liver disease in alcoholism. Am J Clin Nutr. 1988;48:148-151. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 71] [Cited by in RCA: 54] [Article Influence: 1.5] [Reference Citation Analysis (0)] |
| 17. | Bellentani S, Saccoccio G, Costa G, Tiribelli C, Manenti F, Sodde M, Saveria Crocè L, Sasso F, Pozzato G, Cristianini G, Brandi G. Drinking habits as cofactors of risk for alcohol induced liver damage. The Dionysos Study Group. Gut. 1997;41:845-850. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 437] [Cited by in RCA: 405] [Article Influence: 14.5] [Reference Citation Analysis (0)] |
| 18. | Lu XL, Luo JY, Tao M, Gen Y, Zhao P, Zhao HL, Zhang XD, Dong N. Risk factors for alcoholic liver disease in China. World J Gastroenterol. 2004;10:2423-2426. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in CrossRef: 49] [Cited by in RCA: 45] [Article Influence: 2.1] [Reference Citation Analysis (1)] |
| 19. | Mendenhall CL, Moritz TE, Roselle GA, Morgan TR, Nemchausky BA, Tamburro CH, Schiff ER, McClain CJ, Marsano LS, Allen JI. Protein energy malnutrition in severe alcoholic hepatitis: diagnosis and response to treatment. The VA Cooperative Study Group #275. JPEN J Parenter Enteral Nutr. 1995;19:258-265. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 120] [Cited by in RCA: 113] [Article Influence: 3.8] [Reference Citation Analysis (2)] |
| 20. | Mendenhall C, Roselle GA, Gartside P, Moritz T. Relationship of protein calorie malnutrition to alcoholic liver disease: a reexamination of data from two Veterans Administration Cooperative Studies. Alcohol Clin Exp Res. 1995;19:635-641. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 158] [Cited by in RCA: 134] [Article Influence: 4.5] [Reference Citation Analysis (0)] |
| 21. | Leevy CM, Moroianu SA. Nutritional aspects of alcoholic liver disease. Clin Liver Dis. 2005;9:67-81. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 58] [Cited by in RCA: 46] [Article Influence: 2.3] [Reference Citation Analysis (0)] |
| 22. | Singal AK, Anand BS. Mechanisms of synergy between alcohol and hepatitis C virus. J Clin Gastroenterol. 2007;41:761-772. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 90] [Cited by in RCA: 98] [Article Influence: 5.4] [Reference Citation Analysis (0)] |
| 23. | Poynard T, Bedossa P, Opolon P. Natural history of liver fibrosis progression in patients with chronic hepatitis C. The OBSVIRC, METAVIR, CLINIVIR, and DOSVIRC groups. Lancet. 1997;349:825-832. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 2199] [Cited by in RCA: 2159] [Article Influence: 77.1] [Reference Citation Analysis (0)] |
| 24. | Singal AK, Sagi S, Kuo YF, Weinman S. Impact of hepatitis C virus infection on the course and outcome of patients with acute alcoholic hepatitis. Eur J Gastroenterol Hepatol. 2011;23:204-209. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 18] [Cited by in RCA: 21] [Article Influence: 1.5] [Reference Citation Analysis (0)] |
| 25. | Iida-Ueno A, Enomoto M, Tamori A, Kawada N. Hepatitis B virus infection and alcohol consumption. World J Gastroenterol. 2017;23:2651-2659. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in CrossRef: 56] [Cited by in RCA: 56] [Article Influence: 7.0] [Reference Citation Analysis (0)] |
| 26. | Siddiqui T, Trieu J, Bilal M, Mehta D, Gollapudi LA, Nabors C, Wolf DC. Obesity Has Significant Impact on Mortality in Patients With Acute Alcoholic Hepatitis. Am. J. Gastroenterol. 113:S591-S593. [DOI] [Full Text] |
| 27. | Sohal A, Bains K, Dhaliwal A, Chaudhry H, Sharma R, Singla P, Gupta G, Dukovic D, Sandhu S, Roytman M, Tringali S. Seasonal Variations of Hospital Admissions for Alcohol-Related Hepatitis in the United States. Gastroenterology Res. 2022;15:75-81. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 1] [Cited by in RCA: 2] [Article Influence: 0.7] [Reference Citation Analysis (0)] |
| 28. | Seth D, Gorrell MD, Cordoba S, McCaughan GW, Haber PS. Intrahepatic gene expression in human alcoholic hepatitis. J Hepatol. 2006;45:306-320. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 60] [Cited by in RCA: 58] [Article Influence: 3.1] [Reference Citation Analysis (0)] |
| 29. | Colmenero J, Bataller R, Sancho-Bru P, Bellot P, Miquel R, Moreno M, Jares P, Bosch J, Arroyo V, Caballería J, Ginès P. Hepatic expression of candidate genes in patients with alcoholic hepatitis: correlation with disease severity. Gastroenterology. 2007;132:687-697. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 90] [Cited by in RCA: 95] [Article Influence: 5.3] [Reference Citation Analysis (0)] |
| 30. | Argemi J, Latasa MU, Atkinson SR, Blokhin IO, Massey V, Gue JP, Cabezas J, Lozano JJ, Van Booven D, Bell A, Cao S, Vernetti LA, Arab JP, Ventura-Cots M, Edmunds LR, Fondevila C, Stärkel P, Dubuquoy L, Louvet A, Odena G, Gomez JL, Aragon T, Altamirano J, Caballeria J, Jurczak MJ, Taylor DL, Berasain C, Wahlestedt C, Monga SP, Morgan MY, Sancho-Bru P, Mathurin P, Furuya S, Lackner C, Rusyn I, Shah VH, Thursz MR, Mann J, Avila MA, Bataller R. Defective HNF4alpha-dependent gene expression as a driver of hepatocellular failure in alcoholic hepatitis. Nat Commun. 2019;10:3126. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 130] [Cited by in RCA: 150] [Article Influence: 25.0] [Reference Citation Analysis (0)] |
| 31. | Beaudoin JJ, Liang T, Tang Q, Banini BA, Shah VH, Sanyal AJ, Chalasani NP, Gawrieh S. Role of candidate gene variants in modulating the risk and severity of alcoholic hepatitis. Alcohol Clin Exp Res. 2021;45:709-719. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 13] [Cited by in RCA: 11] [Article Influence: 2.8] [Reference Citation Analysis (0)] |
| 32. | Bruschi FV, Tardelli M, Claudel T, Trauner M. PNPLA3 expression and its impact on the liver: current perspectives. Hepat Med. 2017;9:55-66. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 46] [Cited by in RCA: 58] [Article Influence: 7.3] [Reference Citation Analysis (0)] |
| 33. | Palmer ND, Musani SK, Yerges-Armstrong LM, Feitosa MF, Bielak LF, Hernaez R, Kahali B, Carr JJ, Harris TB, Jhun MA, Kardia SL, Langefeld CD, Mosley TH Jr, Norris JM, Smith AV, Taylor HA, Wagenknecht LE, Liu J, Borecki IB, Peyser PA, Speliotes EK. Characterization of European ancestry nonalcoholic fatty liver disease-associated variants in individuals of African and Hispanic descent. Hepatology. 2013;58:966-975. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 119] [Cited by in RCA: 122] [Article Influence: 10.2] [Reference Citation Analysis (0)] |
| 34. | Levy R, Catana AM, Durbin-Johnson B, Halsted CH, Medici V. Ethnic differences in presentation and severity of alcoholic liver disease. Alcohol Clin Exp Res. 2015;39:566-574. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 44] [Cited by in RCA: 74] [Article Influence: 7.4] [Reference Citation Analysis (0)] |
| 35. | Setshedi M, Wands JR, Monte SM. Acetaldehyde adducts in alcoholic liver disease. Oxid Med Cell Longev. 2010;3:178-185. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 182] [Cited by in RCA: 237] [Article Influence: 15.8] [Reference Citation Analysis (0)] |
| 36. | Lieber CS. Cytochrome P-4502E1: its physiological and pathological role. Physiol Rev. 1997;77:517-544. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 649] [Cited by in RCA: 613] [Article Influence: 21.9] [Reference Citation Analysis (0)] |
| 37. | Gao B, Bataller R. Alcoholic liver disease: pathogenesis and new therapeutic targets. Gastroenterology. 2011;141:1572-1585. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 1244] [Cited by in RCA: 1495] [Article Influence: 106.8] [Reference Citation Analysis (0)] |
| 38. | Osna NA, Donohue TM Jr, Kharbanda KK. Alcoholic Liver Disease: Pathogenesis and Current Management. Alcohol Res. 2017;38:147-161. [PubMed] |
| 39. | Abenavoli L, Masarone M, Federico A, Rosato V, Dallio M, Loguercio C, Persico M. Alcoholic Hepatitis: Pathogenesis, Diagnosis and Treatment. Rev Recent Clin Trials. 2016;11:159-166. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 24] [Cited by in RCA: 26] [Article Influence: 2.9] [Reference Citation Analysis (0)] |
| 40. | Su GL, Klein RD, Aminlari A, Zhang HY, Steinstraesser L, Alarcon WH, Remick DG, Wang SC. Kupffer cell activation by lipopolysaccharide in rats: role for lipopolysaccharide binding protein and toll-like receptor 4. Hepatology. 2000;31:932-936. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 197] [Cited by in RCA: 210] [Article Influence: 8.4] [Reference Citation Analysis (0)] |
| 41. | Morgan TR, McClain CJ. Pentoxifylline and alcoholic hepatitis. Gastroenterology. 2000;119:1787-1791. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 14] [Cited by in RCA: 15] [Article Influence: 0.6] [Reference Citation Analysis (0)] |
| 42. | Bradham CA, Plümpe J, Manns MP, Brenner DA, Trautwein C. Mechanisms of hepatic toxicity. I. TNF-induced liver injury. Am J Physiol. 1998;275:G387-G392. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 64] [Cited by in RCA: 102] [Article Influence: 3.8] [Reference Citation Analysis (0)] |
| 43. | McClain C, Hill D, Schmidt J, Diehl AM. Cytokines and alcoholic liver disease. Semin Liver Dis. 1993;13:170-182. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 209] [Cited by in RCA: 182] [Article Influence: 5.7] [Reference Citation Analysis (0)] |
| 44. | Bird G. Interleukin-8 in alcoholic liver disease. Acta Gastroenterol Belg. 1994;57:255-259. [PubMed] |
| 45. | Dugum M, Zein N, McCullough A, Hanouneh I. Alcoholic hepatitis: Challenges in diagnosis and management. Cleve Clin J Med. 2015;82:226-236. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 3] [Cited by in RCA: 6] [Article Influence: 0.7] [Reference Citation Analysis (0)] |
| 46. | Hardison JE. Auscultation of the Liver. In: Clinical Methods: The History, Physical, and Laboratory Examinations. Boston: Butterworths; 1990. [PubMed] |
| 47. | Keating M, Lardo O, Hansell M. Alcoholic Hepatitis: Diagnosis and Management. Am Fam Physician. 2022;105:412-420. [PubMed] |
| 48. | Atkinson SR, Hamesch K, Spivak I, Guldiken N, Cabezas J, Argemi J, Theurl I, Zoller H, Cao S, Mathurin P, Shah VH, Trautwein C, Bataller R, Thursz MR, Strnad P. Serum Transferrin Is an Independent Predictor of Mortality in Severe Alcoholic Hepatitis. Am J Gastroenterol. 2020;115:398-405. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 17] [Cited by in RCA: 14] [Article Influence: 2.8] [Reference Citation Analysis (0)] |
| 49. | Grillet F, Calame P, Cervoni JP, Weil D, Thevenot T, Ronot M, Delabrousse E. Non-invasive diagnosis of severe alcoholic hepatitis: Usefulness of cross-sectional imaging. Diagn Interv Imaging. 2021;102:247-254. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 3] [Cited by in RCA: 6] [Article Influence: 1.2] [Reference Citation Analysis (0)] |
| 50. | European Association for the Study of the Liver. EASL Clinical Practice Guidelines: Management of alcohol-related liver disease. J Hepatol. 2018;69:154-181. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 636] [Cited by in RCA: 585] [Article Influence: 83.6] [Reference Citation Analysis (0)] |
| 51. | Crawford JM. Histologic findings in alcoholic liver disease. Clin Liver Dis. 2012;16:699-716. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 35] [Cited by in RCA: 45] [Article Influence: 3.5] [Reference Citation Analysis (1)] |
| 52. | Lefkowitch JH. Morphology of alcoholic liver disease. Clin Liver Dis. 2005;9:37-53. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 139] [Cited by in RCA: 139] [Article Influence: 7.0] [Reference Citation Analysis (0)] |
| 53. | Sakhuja P. Pathology of alcoholic liver disease, can it be differentiated from nonalcoholic steatohepatitis? World J Gastroenterol. 2014;20:16474-16479. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in CrossRef: 66] [Cited by in RCA: 68] [Article Influence: 6.2] [Reference Citation Analysis (1)] |
| 54. | Theise ND. Histopathology of alcoholic liver disease. Clin Liver Dis (Hoboken). 2013;2:64-67. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 28] [Cited by in RCA: 44] [Article Influence: 3.7] [Reference Citation Analysis (0)] |
| 55. | Feldstein AE, Gores GJ. Apoptosis in alcoholic and nonalcoholic steatohepatitis. Front Biosci. 2005;10:3093-3099. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 137] [Cited by in RCA: 143] [Article Influence: 7.2] [Reference Citation Analysis (0)] |
| 56. | Denk H, Stumptner C, Zatloukal K. Mallory bodies revisited. J Hepatol. 2000;32:689-702. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 142] [Cited by in RCA: 134] [Article Influence: 5.4] [Reference Citation Analysis (0)] |
| 57. | Yeh MM, Brunt EM. Pathology of nonalcoholic fatty liver disease. Am J Clin Pathol. 2007;128:837-847. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 211] [Cited by in RCA: 200] [Article Influence: 11.1] [Reference Citation Analysis (1)] |
| 58. | Tiniakos DG. Liver biopsy in alcoholic and non-alcoholic steatohepatitis patients. Gastroenterol Clin Biol. 2009;33:930-939. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 22] [Cited by in RCA: 21] [Article Influence: 1.3] [Reference Citation Analysis (0)] |
| 59. | Kowdley KV. Iron Overload in Patients With Chronic Liver Disease. Gastroenterol Hepatol (N Y). 2016;12:695-698. [PubMed] |
| 60. | Eng SC, Taylor SL, Reyes V, Raaka S, Berger J, Kowdley KV. Hepatic iron overload in alcoholic end-stage liver disease is associated with iron deposition in other organs in the absence of HFE-1 hemochromatosis. Liver Int. 2005;25:513-517. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 25] [Cited by in RCA: 23] [Article Influence: 1.2] [Reference Citation Analysis (0)] |
| 61. | Mookerjee RP, Lackner C, Stauber R, Stadlbauer V, Deheragoda M, Aigelsreiter A, Jalan R. The role of liver biopsy in the diagnosis and prognosis of patients with acute deterioration of alcoholic cirrhosis. J Hepatol. 2011;55:1103-1111. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 110] [Cited by in RCA: 110] [Article Influence: 7.9] [Reference Citation Analysis (0)] |
| 62. | Altamirano J, Miquel R, Katoonizadeh A, Abraldes JG, Duarte-Rojo A, Louvet A, Augustin S, Mookerjee RP, Michelena J, Smyrk TC, Buob D, Leteurtre E, Rincón D, Ruiz P, García-Pagán JC, Guerrero-Marquez C, Jones PD, Barritt AS 4th, Arroyo V, Bruguera M, Bañares R, Ginès P, Caballería J, Roskams T, Nevens F, Jalan R, Mathurin P, Shah VH, Bataller R. A histologic scoring system for prognosis of patients with alcoholic hepatitis. Gastroenterology. 2014;146:1231-9.e1. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 320] [Cited by in RCA: 333] [Article Influence: 30.3] [Reference Citation Analysis (0)] |
| 63. | Trépo E, Goossens N, Fujiwara N, Song WM, Colaprico A, Marot A, Spahr L, Demetter P, Sempoux C, Im GY, Saldarriaga J, Gustot T, Devière J, Thung SN, Minsart C, Sersté T, Bontempi G, Abdelrahman K, Henrion J, Degré D, Lucidi V, Rubbia-Brandt L, Nair VD, Moreno C, Deltenre P, Hoshida Y, Franchimont D. Combination of Gene Expression Signature and Model for End-Stage Liver Disease Score Predicts Survival of Patients With Severe Alcoholic Hepatitis. Gastroenterology. 2018;154:965-975. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 32] [Cited by in RCA: 45] [Article Influence: 6.4] [Reference Citation Analysis (0)] |
| 64. | Koulaouzidis A, Bhat S, Saeed AA. Spontaneous bacterial peritonitis. World J Gastroenterol. 2009;15:1042-1049. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in CrossRef: 74] [Cited by in RCA: 66] [Article Influence: 4.1] [Reference Citation Analysis (2)] |
| 65. | Karakike E, Moreno C, Gustot T. Infections in severe alcoholic hepatitis. Ann Gastroenterol. 2017;30:152-160. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 5] [Cited by in RCA: 9] [Article Influence: 1.0] [Reference Citation Analysis (0)] |
| 66. | Orntoft NW, Sandahl TD, Jepsen P, Vilstrup H. Short-term and long-term causes of death in patients with alcoholic hepatitis in Denmark. Clin Gastroenterol Hepatol. 2014;12:1739-44.e1. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 31] [Cited by in RCA: 42] [Article Influence: 3.8] [Reference Citation Analysis (0)] |
| 67. | Markwick LJ, Riva A, Ryan JM, Cooksley H, Palma E, Tranah TH, Manakkat Vijay GK, Vergis N, Thursz M, Evans A, Wright G, Tarff S, O'Grady J, Williams R, Shawcross DL, Chokshi S. Blockade of PD1 and TIM3 restores innate and adaptive immunity in patients with acute alcoholic hepatitis. Gastroenterology. 2015;148:590-602.e10. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 125] [Cited by in RCA: 161] [Article Influence: 16.1] [Reference Citation Analysis (0)] |
| 68. | Falagas ME, Kompoti M. Obesity and infection. Lancet Infect Dis. 2006;6:438-446. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 549] [Cited by in RCA: 606] [Article Influence: 31.9] [Reference Citation Analysis (0)] |
| 69. | Leclerc M, Lanot A, Béchade C, Le Naoures C, Comoz F, Lobbedez T. [Bile salt nephropathy/cholemic nephrosis]. Nephrol Ther. 2016;12:460-462. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 6] [Cited by in RCA: 6] [Article Influence: 0.7] [Reference Citation Analysis (0)] |
| 70. | Ginès P, Solà E, Angeli P, Wong F, Nadim MK, Kamath PS. Hepatorenal syndrome. Nat Rev Dis Primers. 2018;4:23. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 110] [Cited by in RCA: 171] [Article Influence: 24.4] [Reference Citation Analysis (3)] |
| 71. | Francoz C, Durand F, Kahn JA, Genyk YS, Nadim MK. Hepatorenal Syndrome. Clin J Am Soc Nephrol. 2019;14:774-781. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 41] [Cited by in RCA: 82] [Article Influence: 13.7] [Reference Citation Analysis (0)] |
| 72. | Altamirano J, Fagundes C, Dominguez M, García E, Michelena J, Cárdenas A, Guevara M, Pereira G, Torres-Vigil K, Arroyo V, Caballería J, Ginès P, Bataller R. Acute kidney injury is an early predictor of mortality for patients with alcoholic hepatitis. Clin Gastroenterol Hepatol. 2012;10:65-71.e3. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 117] [Cited by in RCA: 136] [Article Influence: 10.5] [Reference Citation Analysis (0)] |
| 73. | Wang H, Liu A, Bo W, Feng X, Hu Y. Terlipressin in the treatment of hepatorenal syndrome: A systematic review and meta-analysis. Medicine (Baltimore). 2018;97:e0431. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 36] [Cited by in RCA: 50] [Article Influence: 7.1] [Reference Citation Analysis (0)] |
| 74. | Parker R, Kim SJ, Im GY, Nahas J, Dhesi B, Vergis N, Sinha A, Ghezzi A, Rink MR, McCune A, Aithal GP, Newsome PN, Weston CJ, Holt A, Gao B. Obesity in acute alcoholic hepatitis increases morbidity and mortality. EBioMedicine. 2019;45:511-518. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 19] [Cited by in RCA: 38] [Article Influence: 6.3] [Reference Citation Analysis (0)] |
| 75. | Liang W, Chikritzhs T, Pascal R, Binns CW. Mortality rate of alcoholic liver disease and risk of hospitalization for alcoholic liver cirrhosis, alcoholic hepatitis and alcoholic liver failure in Australia between 1993 and 2005. Intern Med J. 2011;41:34-41. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 23] [Cited by in RCA: 29] [Article Influence: 2.1] [Reference Citation Analysis (0)] |
| 76. | Jalan R, Gines P, Olson JC, Mookerjee RP, Moreau R, Garcia-Tsao G, Arroyo V, Kamath PS. Acute-on chronic liver failure. J Hepatol. 2012;57:1336-1348. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 447] [Cited by in RCA: 457] [Article Influence: 35.2] [Reference Citation Analysis (1)] |
| 77. | Michelena J, Altamirano J, Abraldes JG, Affò S, Morales-Ibanez O, Sancho-Bru P, Dominguez M, García-Pagán JC, Fernández J, Arroyo V, Ginès P, Louvet A, Mathurin P, Mehal WZ, Caballería J, Bataller R. Systemic inflammatory response and serum lipopolysaccharide levels predict multiple organ failure and death in alcoholic hepatitis. Hepatology. 2015;62:762-772. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 217] [Cited by in RCA: 219] [Article Influence: 21.9] [Reference Citation Analysis (0)] |
| 78. | Arroyo V, Moreau R, Jalan R. Acute-on-Chronic Liver Failure. N Engl J Med. 2020;382:2137-2145. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 219] [Cited by in RCA: 429] [Article Influence: 85.8] [Reference Citation Analysis (2)] |
| 79. | Bodzin AS, Baker TB. Liver Transplantation Today: Where We Are Now and Where We Are Going. Liver Transpl. 2018;24:1470-1475. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 76] [Cited by in RCA: 129] [Article Influence: 18.4] [Reference Citation Analysis (0)] |
| 80. | Jadlowiec CC, Taner T. Liver transplantation: Current status and challenges. World J Gastroenterol. 2016;22:4438-4445. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in CrossRef: 238] [Cited by in RCA: 211] [Article Influence: 23.4] [Reference Citation Analysis (2)] |
| 81. | Yu CH, Xu CF, Ye H, Li L, Li YM. Early mortality of alcoholic hepatitis: a review of data from placebo-controlled clinical trials. World J Gastroenterol. 2010;16:2435-2439. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in CrossRef: 36] [Cited by in RCA: 39] [Article Influence: 2.6] [Reference Citation Analysis (0)] |
| 82. | Bennett K, Enki DG, Thursz M, Cramp ME, Dhanda AD. Systematic review with meta-analysis: high mortality in patients with non-severe alcoholic hepatitis. Aliment Pharmacol Ther. 2019;50:249-257. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 28] [Cited by in RCA: 35] [Article Influence: 5.8] [Reference Citation Analysis (0)] |
| 83. | Bird GL, Williams R. Factors determining cirrhosis in alcoholic liver disease. Mol Aspects Med. 1988;10:97-105. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 40] [Cited by in RCA: 35] [Article Influence: 0.9] [Reference Citation Analysis (0)] |
| 84. | Alexander JF, Lischner MW, Galambos JT. Natural history of alcoholic hepatitis. II. The long-term prognosis. Am J Gastroenterol. 1971;56:515-525. [PubMed] |
| 85. | Rehm J, Taylor B, Mohapatra S, Irving H, Baliunas D, Patra J, Roerecke M. Alcohol as a risk factor for liver cirrhosis: a systematic review and meta-analysis. Drug Alcohol Rev. 2010;29:437-445. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 514] [Cited by in RCA: 439] [Article Influence: 29.3] [Reference Citation Analysis (0)] |
| 86. | Parés A, Caballería J, Bruguera M, Torres M, Rodés J. Histological course of alcoholic hepatitis. Influence of abstinence, sex and extent of hepatic damage. J Hepatol. 1986;2:33-42. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 214] [Cited by in RCA: 189] [Article Influence: 4.8] [Reference Citation Analysis (0)] |
| 87. | Maddrey WC, Boitnott JK, Bedine MS, Weber FL Jr, Mezey E, White RI Jr. Corticosteroid therapy of alcoholic hepatitis. Gastroenterology. 1978;75:193-199. [PubMed] |
| 88. | Louvet A, Naveau S, Abdelnour M, Ramond MJ, Diaz E, Fartoux L, Dharancy S, Texier F, Hollebecque A, Serfaty L, Boleslawski E, Deltenre P, Canva V, Pruvot FR, Mathurin P. The Lille model: a new tool for therapeutic strategy in patients with severe alcoholic hepatitis treated with steroids. Hepatology. 2007;45:1348-1354. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 479] [Cited by in RCA: 512] [Article Influence: 28.4] [Reference Citation Analysis (0)] |
| 89. | Kamath PS, Kim WR; Advanced Liver Disease Study Group. The model for end-stage liver disease (MELD). Hepatology. 2007;45:797-805. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 1075] [Cited by in RCA: 1226] [Article Influence: 68.1] [Reference Citation Analysis (0)] |
| 90. | Morales-Arráez D, Ventura-Cots M, Altamirano J, Abraldes JG, Cruz-Lemini M, Thursz MR, Atkinson SR, Sarin SK, Kim W, Chavez-Araujo R, Higuera-de la Tijera MF, Singal AK, Shah VH, Kamath PS, Duarte-Rojo A, Charles EA, Vargas V, Jager M, Rautou PE, Rincon D, Zamarripa F, Restrepo-Gutiérrez JC, Torre A, Lucey MR, Arab JP, Mathurin P, Louvet A, García-Tsao G, González JA, Verna EC, Brown RS Jr, Argemi J, Fernández-Carrillo C, Clemente A, Alvarado-Tapias E, Forrest E, Allison M, Bataller R. The MELD Score Is Superior to the Maddrey Discriminant Function Score to Predict Short-Term Mortality in Alcohol-Associated Hepatitis: A Global Study. Am J Gastroenterol. 2022;117:301-310. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 50] [Cited by in RCA: 53] [Article Influence: 17.7] [Reference Citation Analysis (0)] |
| 91. | Srikureja W, Kyulo NL, Runyon BA, Hu KQ. MELD score is a better prognostic model than Child-Turcotte-Pugh score or Discriminant Function score in patients with alcoholic hepatitis. J Hepatol. 2005;42:700-706. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 181] [Cited by in RCA: 165] [Article Influence: 8.3] [Reference Citation Analysis (0)] |
| 92. | Dunn W, Jamil LH, Brown LS, Wiesner RH, Kim WR, Menon KV, Malinchoc M, Kamath PS, Shah V. MELD accurately predicts mortality in patients with alcoholic hepatitis. Hepatology. 2005;41:353-358. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 357] [Cited by in RCA: 374] [Article Influence: 18.7] [Reference Citation Analysis (0)] |
| 93. | Sheth M, Riggs M, Patel T. Utility of the Mayo End-Stage Liver Disease (MELD) score in assessing prognosis of patients with alcoholic hepatitis. BMC Gastroenterol. 2002;2:2. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 123] [Cited by in RCA: 118] [Article Influence: 5.1] [Reference Citation Analysis (0)] |
| 94. | Peng Y, Qi X, Guo X. Child-Pugh Versus MELD Score for the Assessment of Prognosis in Liver Cirrhosis: A Systematic Review and Meta-Analysis of Observational Studies. Medicine (Baltimore). 2016;95:e2877. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 344] [Cited by in RCA: 340] [Article Influence: 37.8] [Reference Citation Analysis (0)] |
| 95. | Forrest EH, Evans CD, Stewart S, Phillips M, Oo YH, McAvoy NC, Fisher NC, Singhal S, Brind A, Haydon G, O'Grady J, Day CP, Hayes PC, Murray LS, Morris AJ. Analysis of factors predictive of mortality in alcoholic hepatitis and derivation and validation of the Glasgow alcoholic hepatitis score. Gut. 2005;54:1174-1179. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 268] [Cited by in RCA: 264] [Article Influence: 13.2] [Reference Citation Analysis (0)] |
| 96. | Dominguez M, Rincón D, Abraldes JG, Miquel R, Colmenero J, Bellot P, García-Pagán JC, Fernández R, Moreno M, Bañares R, Arroyo V, Caballería J, Ginès P, Bataller R. A new scoring system for prognostic stratification of patients with alcoholic hepatitis. Am J Gastroenterol. 2008;103:2747-2756. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 217] [Cited by in RCA: 232] [Article Influence: 13.6] [Reference Citation Analysis (0)] |
| 97. | Lackner C, Spindelboeck W, Haybaeck J, Douschan P, Rainer F, Terracciano L, Haas J, Berghold A, Bataller R, Stauber RE. Histological parameters and alcohol abstinence determine long-term prognosis in patients with alcoholic liver disease. J Hepatol. 2017;66:610-618. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 126] [Cited by in RCA: 201] [Article Influence: 25.1] [Reference Citation Analysis (0)] |
| 98. | Xie YD, Feng B, Gao Y, Wei L. Effect of abstinence from alcohol on survival of patients with alcoholic cirrhosis: A systematic review and meta-analysis. Hepatol Res. 2014;44:436-449. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 32] [Cited by in RCA: 44] [Article Influence: 4.0] [Reference Citation Analysis (0)] |
| 99. | Diehl AM. Alcoholic liver disease: natural history. Liver Transpl Surg. 1997;3:206-211. [PubMed] [DOI] [Full Text] |
| 100. | Dugum M, McCullough A. Diagnosis and Management of Alcoholic Liver Disease. J Clin Transl Hepatol. 2015;3:109-116. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 48] [Cited by in RCA: 52] [Article Influence: 5.2] [Reference Citation Analysis (0)] |
| 101. | Morley KC, Baillie A, Fraser I, Furneaux-Bate A, Dore G, Roberts M, Abdalla A, Phung N, Haber PS. Baclofen in the treatment of alcohol dependence with or without liver disease: multisite, randomised, double-blind, placebo-controlled trial. Br J Psychiatry. 2018;212:362-369. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 66] [Cited by in RCA: 93] [Article Influence: 13.3] [Reference Citation Analysis (0)] |
| 102. | McClain CJ, Barve SS, Barve A, Marsano L. Alcoholic liver disease and malnutrition. Alcohol Clin Exp Res. 2011;35:815-820. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 67] [Cited by in RCA: 78] [Article Influence: 5.6] [Reference Citation Analysis (0)] |
| 103. | Bertha M, Choi G, Mellinger J. Diagnosis and Treatment of Alcohol-Associated Liver Disease: A Patient-Friendly Summary of the 2019 AASLD Guidelines. Clin Liver Dis (Hoboken). 2021;17:418-423. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 1] [Cited by in RCA: 8] [Article Influence: 2.0] [Reference Citation Analysis (0)] |
| 104. | DiCecco SR, Francisco-Ziller N. Nutrition in alcoholic liver disease. Nutr Clin Pract. 2006;21:245-254. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 40] [Cited by in RCA: 35] [Article Influence: 1.8] [Reference Citation Analysis (0)] |
| 105. | Mitchell MC, Friedman LS, McClain CJ. Medical Management of Severe Alcoholic Hepatitis: Expert Review from the Clinical Practice Updates Committee of the AGA Institute. Clin Gastroenterol Hepatol. 2017;15:5-12. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 58] [Cited by in RCA: 68] [Article Influence: 8.5] [Reference Citation Analysis (0)] |
| 106. | Biggins SW, Angeli P, Garcia-Tsao G, Ginès P, Ling SC, Nadim MK, Wong F, Kim WR. Diagnosis, Evaluation, and Management of Ascites, Spontaneous Bacterial Peritonitis and Hepatorenal Syndrome: 2021 Practice Guidance by the American Association for the Study of Liver Diseases. Hepatology. 2021;74:1014-1048. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 482] [Cited by in RCA: 459] [Article Influence: 114.8] [Reference Citation Analysis (0)] |
| 107. | Benzodiazepines. 2017. In: LiverTox: Clinical and Research Information on Drug-Induced Liver Injury [Internet]. Bethesda (MD): National Institute of Diabetes and Digestive and Kidney Diseases; 2012 . [PubMed] |
| 108. | Peppers MP. Benzodiazepines for alcohol withdrawal in the elderly and in patients with liver disease. Pharmacotherapy. 1996;16:49-57. [PubMed] |
| 109. | Thursz MR, Richardson P, Allison M, Austin A, Bowers M, Day CP, Downs N, Gleeson D, MacGilchrist A, Grant A, Hood S, Masson S, McCune A, Mellor J, O'Grady J, Patch D, Ratcliffe I, Roderick P, Stanton L, Vergis N, Wright M, Ryder S, Forrest EH; STOPAH Trial. Prednisolone or pentoxifylline for alcoholic hepatitis. N Engl J Med. 2015;372:1619-1628. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 526] [Cited by in RCA: 555] [Article Influence: 55.5] [Reference Citation Analysis (1)] |
| 110. | Mathurin P, O'Grady J, Carithers RL, Phillips M, Louvet A, Mendenhall CL, Ramond MJ, Naveau S, Maddrey WC, Morgan TR. Corticosteroids improve short-term survival in patients with severe alcoholic hepatitis: meta-analysis of individual patient data. Gut. 2011;60:255-260. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 292] [Cited by in RCA: 305] [Article Influence: 21.8] [Reference Citation Analysis (1)] |
| 111. | Louvet A, Thursz MR, Kim DJ, Labreuche J, Atkinson SR, Sidhu SS, O'Grady JG, Akriviadis E, Sinakos E, Carithers RL Jr, Ramond MJ, Maddrey WC, Morgan TR, Duhamel A, Mathurin P. Corticosteroids Reduce Risk of Death Within 28 Days for Patients With Severe Alcoholic Hepatitis, Compared With Pentoxifylline or Placebo-a Meta-analysis of Individual Data From Controlled Trials. Gastroenterology. 2018;155:458-468.e8. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 186] [Cited by in RCA: 171] [Article Influence: 24.4] [Reference Citation Analysis (0)] |
| 112. | Levitsky J, Mailliard ME. Diagnosis and therapy of alcoholic liver disease. Semin Liver Dis. 2004;24:233-247. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 74] [Cited by in RCA: 70] [Article Influence: 3.3] [Reference Citation Analysis (0)] |
| 113. | Sohail U, Satapathy SK. Diagnosis and management of alcoholic hepatitis. Clin Liver Dis. 2012;16:717-736. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 18] [Cited by in RCA: 19] [Article Influence: 1.5] [Reference Citation Analysis (0)] |
| 114. | Sougioultzis S, Dalakas E, Hayes PC, Plevris JN. Alcoholic hepatitis: from pathogenesis to treatment. Curr Med Res Opin. 2005;21:1337-1346. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 24] [Cited by in RCA: 20] [Article Influence: 1.0] [Reference Citation Analysis (0)] |
| 115. | Hmoud BS, Patel K, Bataller R, Singal AK. Corticosteroids and occurrence of and mortality from infections in severe alcoholic hepatitis: a meta-analysis of randomized trials. Liver Int. 2016;36:721-728. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 46] [Cited by in RCA: 71] [Article Influence: 7.9] [Reference Citation Analysis (0)] |
| 116. | Im GY, Cameron AM, Lucey MR. Liver transplantation for alcoholic hepatitis. J Hepatol. 2019;70:328-334. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 73] [Cited by in RCA: 82] [Article Influence: 13.7] [Reference Citation Analysis (0)] |
| 117. | Mathurin P, Moreno C, Samuel D, Dumortier J, Salleron J, Durand F, Castel H, Duhamel A, Pageaux GP, Leroy V, Dharancy S, Louvet A, Boleslawski E, Lucidi V, Gustot T, Francoz C, Letoublon C, Castaing D, Belghiti J, Donckier V, Pruvot FR, Duclos-Vallée JC. Early liver transplantation for severe alcoholic hepatitis. N Engl J Med. 2011;365:1790-1800. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 616] [Cited by in RCA: 658] [Article Influence: 47.0] [Reference Citation Analysis (0)] |
| 118. | Weeks SR, Sun Z, McCaul ME, Zhu H, Anders RA, Philosophe B, Ottmann SE, Garonzik Wang JM, Gurakar AO, Cameron AM. Liver Transplantation for Severe Alcoholic Hepatitis, Updated Lessons from the World's Largest Series. J Am Coll Surg. 2018;226:549-557. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 58] [Cited by in RCA: 85] [Article Influence: 12.1] [Reference Citation Analysis (0)] |
| 119. | Lee BP, Mehta N, Platt L, Gurakar A, Rice JP, Lucey MR, Im GY, Therapondos G, Han H, Victor DW, Fix OK, Dinges L, Dronamraju D, Hsu C, Voigt MD, Rinella ME, Maddur H, Eswaran S, Hause J, Foley D, Ghobrial RM, Dodge JL, Li Z, Terrault NA. Outcomes of Early Liver Transplantation for Patients With Severe Alcoholic Hepatitis. Gastroenterology. 2018;155:422-430.e1. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 184] [Cited by in RCA: 271] [Article Influence: 38.7] [Reference Citation Analysis (0)] |
| 120. | Herrick-Reynolds KM, Punchhi G, Greenberg RS, Strauss AT, Boyarsky BJ, Weeks-Groh SR, Krach MR, Anders RA, Gurakar A, Chen PH, Segev DL, King EA, Philosophe B, Ottman SE, Wesson RN, Garonzik-Wang JM, Cameron AM. Evaluation of Early vs Standard Liver Transplant for Alcohol-Associated Liver Disease. JAMA Surg. 2021;156:1026-1034. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 14] [Cited by in RCA: 54] [Article Influence: 13.5] [Reference Citation Analysis (0)] |
| 121. | Mitchell M. Efficacy study of anakinra, pentoxifylline, and zinc compared to methylprednisolone in severe acute alcoholic hepatitis. [accessed 2022 Dec 16]. In: ClinicalTrials.gov [Internet]. Bethesda (MD): U.S. National Library of Medicine. Available from: https://clinicaltrials.gov/ct2/show/NCT01809132 ClinicalTrials.gov Identifier: NCT01809132. |
| 122. | Gawrieh S. Trial of Anakinra (Plus Zinc) or Prednisone in Patients With Severe Alcoholic Hepatitis. [accessed 2022 Dec 16]. In: ClinicalTrials.gov [Internet]. Bethesda (MD): U.S. National Library of Medicine. Available from: https://clinicaltrials.gov/ct2/show/NCT04072822 ClinicalTrials.gov Identifier: NCT04072822. |
| 123. | Imperial College London. IL-1 Signal Inhibition in Alcoholic Hepatitis [accessed 2022 Dec 16]. In: ClinicalTrials.gov [Internet]. Bethesda (MD): U.S. National Library of Medicine. Available from: https://clinicaltrials.gov/ct2/show/NCT03775109 ClinicalTrials.gov Identifier: NCT03775109. |
| 124. | Imperial College London. N-ACetylcysteine to Reduce Infection and Mortality for Alcoholic Hepatitis [accessed 2022 Dec 16]. In: ClinicalTrials.gov [Internet]. Bethesda (MD): U.S. National Library of Medicine. Available from: https://clinicaltrials.gov/ct2/show/NCT03069300 ClinicalTrials.gov Identifier: NCT03069300. |
| 125. | Mendler MH. Randomized, Controlled Trial of S-adenosylmethionine in Alcoholic Liver Disease. [accessed 2022 Dec 16]. In: ClinicalTrials.gov [Internet]. Bethesda (MD): U.S. National Library of Medicine. Available from: https://clinicaltrials.gov/ct2/show/NCT00851981 ClinicalTrials.gov Identifier: NCT00851981. |
| 126. | Cheng J. Efficacy and Safety of S-adenosyl-l-methionine in Treatment of Alcoholic Hepatitis With Cholestasis. [accessed 2022 Dec 16]. In: ClinicalTrials.gov [Internet]. Bethesda (MD): U.S. National Library of Medicine. Available from: https://clinicaltrials.gov/ct2/show/NCT02024295 ClinicalTrials.gov Identifier: NCT02024295. |
| 127. | Gordon R. A Research Study to Assess the Safety, Pharmacokinetics and Pharmacodynamics of DUR-928 in Patients With Alcoholic Hepatitis. [accessed 2022 Dec 16]. In: ClinicalTrials.gov [Internet]. Bethesda (MD): U.S. National Library of Medicine. Available from: https://clinicaltrials.gov/ct2/show/NCT03432260 ClinicalTrials.gov Identifier: NCT03432260. |
| 128. | McClain CJ. DUR-928 in Patients With Alcoholic Hepatitis. [accessed 2022 Dec 16]. In: ClinicalTrials.gov [Internet]. Bethesda (MD): U.S. National Library of Medicine. Available from: https://clinicaltrials.gov/ct2/show/NCT03917407 ClinicalTrials.gov Identifier: NCT03917407. |
| 129. | Kim DJ. Efficacy and Safety of G-CSF in Patients With Severe Alcoholic Hepatitis With Null or Partial Response to Steroid. [accessed 2022 Dec 16]. In: ClinicalTrials.gov [Internet]. Bethesda (MD): U.S. National Library of Medicine. Available from: https://clinicaltrials.gov/ct2/show/NCT02442180 ClinicalTrials.gov Identifier: NCT02442180. |
| 130. | Singh V. GCSF in Alcoholic Hepatitis. [accessed 2022 Dec 16]. In: ClinicalTrials.gov [Internet]. Bethesda (MD): U.S. National Library of Medicine. Available from: https://clinicaltrials.gov/ct2/show/NCT03703674 ClinicalTrials.gov Identifier: NCT03703674. |
| 131. | Mishra A. Efficacy of Monotherapy vs Combination Therapy of Corticosteroids With GCSF in Severe Alcoholic Hepatitis Patients. [accessed 2022 Dec 16]. In: ClinicalTrials.gov [Internet]. Bethesda (MD): U.S. National Library of Medicine. Available from: https://clinicaltrials.gov/ct2/show/NCT04066179 ClinicalTrials.gov Identifier: NCT04066179. |
| 132. | Shah V. Use of F-652 in Patients With Alcoholic Hepatitis [accessed 2022 Dec 16]. In: ClinicalTrials.gov [Internet]. Bethesda (MD): U.S. National Library of Medicine. Available from: https://clinicaltrials.gov/ct2/show/NCT02655510 ClinicalTrials.gov Identifier: NCT02655510. |
| 133. | Stoy S. Protective Immune Mechanisms in Alcoholic Hepatitis. [accessed 2022 Dec 16]. In: ClinicalTrials.gov [Internet]. Bethesda (MD): U.S. National Library of Medicine. Available from: https://clinicaltrials.gov/ct2/show/NCT01918462 ClinicalTrials.gov Identifier: NCT01918462. |
| 134. | Chalasani N. Trial of Obeticholic Acid in Patients With Moderately Severe Alcoholic Hepatitis (AH). [accessed 2022 Dec 16]. In: ClinicalTrials.gov [Internet]. Bethesda (MD): U.S. National Library of Medicine. Available from: https://clinicaltrials.gov/ct2/show/NCT02039219 ClinicalTrials.gov Identifier: NCT02039219. |
| 135. | Vargas V. Effects of Rifaximin in Patients With Acute Alcoholic Hepatitis. [accessed 2022 Dec 16]. In: ClinicalTrials.gov [Internet]. Bethesda (MD): U.S. National Library of Medicine. Available from: https://clinicaltrials.gov/ct2/show/NCT02116556 ClinicalTrials.gov Identifier: NCT02116556. |
| 136. | Song DS. Rifaximin Use in Severe Alcoholic Hepatitis. [accessed 2022 Dec 16]. In: ClinicalTrials.gov [Internet]. Bethesda (MD): U.S. National Library of Medicine. Available from: https://clinicaltrials.gov/ct2/show/NCT02485106 ClinicalTrials.gov Identifier: NCT02485106. |
| 137. | Sanyal A. Safety and Efficacy of IMM 124-E for Patients With Severe Alcoholic Hepatitis. [accessed 2022 Dec 16]. In: ClinicalTrials.gov [Internet]. Bethesda (MD): U.S. National Library of Medicine. Available from: https://clinicaltrials.gov/ct2/show/NCT01968382 ClinicalTrials.gov Identifier: NCT01968382. |
| 138. | Sidhu S. Comparison of Bovine Colostrum Versus Placebo in Treatment of Severe Alcoholic Hepatitis: A Randomized Double Blind Controlled Trial. [accessed 2022 Dec 16]. In: ClinicalTrials.gov [Internet]. Bethesda (MD): U.S. National Library of Medicine. Available from: https://clinicaltrials.gov/ct2/show/NCT02473341 ClinicalTrials.gov Identifier: NCT02473341. |
| 139. | Mitchell M. Novel Therapies in Moderately Severe Acute Alcoholic Hepatitis. [accessed 2022 Dec 16]. In: ClinicalTrials.gov [Internet]. Bethesda (MD): U.S. National Library of Medicine. Available from: https://clinicaltrials.gov/ct2/show/NCT01922895 ClinicalTrials.gov Identifier: NCT01922895. |
| 140. | Suk KT. Effect of Probiotics on Gut-Liver Axis of Alcoholic Hepatitis. [accessed 2022 Dec 16]. In: ClinicalTrials.gov [Internet]. Bethesda (MD): U.S. National Library of Medicine. Available from: https://clinicaltrials.gov/ct2/show/NCT02335632 ClinicalTrials.gov Identifier: NCT02335632. |
| 141. | Institute of Liver and Biliary Sciences. Pentoxyphilline Versus Fecal Microbiota Therapy in Severe Alcoholic Hepatitis. [accessed 2022 Dec 16]. In: ClinicalTrials.gov [Internet]. Bethesda (MD): U.S. National Library of Medicine. Available from: https://clinicaltrials.gov/ct2/show/NCT02458079 ClinicalTrials.gov Identifier: NCT02458079. |
| 142. | Institute of Liver and Biliary Sciences. A Comparison of Fecal Microbiota Transplantation and Steroid Therapy in Patients With Severe Alcoholic Hepatitis. [accessed 2022 Dec 16]. In: ClinicalTrials.gov [Internet]. Bethesda (MD): U.S. National Library of Medicine. Available from: https://clinicaltrials.gov/ct2/show/NCT03091010 ClinicalTrials.gov Identifier: NCT03091010. |
| 143. | Dhiman RK. Fecal Microbiota Transplantation in Severe Alcoholic Hepatitis- Assessment of Impact on Prognosis and Short-term Outcome. [accessed 2022 Dec 16]. In: ClinicalTrials.gov [Internet]. India: U.S. National Library of Medicine. Available from: https://clinicaltrials.gov/ct2/show/NCT03827772 ClinicalTrials.gov Identifier: NCT03827772. |