Published online Nov 14, 2022. doi: 10.3748/wjg.v28.i42.6034
Peer-review started: August 27, 2022
First decision: September 30, 2022
Revised: October 6, 2022
Accepted: November 2, 2022
Article in press: November 2, 2022
Published online: November 14, 2022
Processing time: 74 Days and 19 Hours
The disease burden related to hepatocellular carcinoma (HCC) is increasing. Most HCC patients are diagnosed at the advanced stage and multikinase inhibitors have been the only treatment choice for them. Recently, the approval of immune checkpoint inhibitors (ICIs) has provided a new therapeutic strategy for HCC. It is noteworthy that the positive outcomes of the phase III clinical trial IMBrave150 [atezolizumab (anti-programmed cell death ligand 1 antibody) combined with bevacizumab (anti-vascular endothelial growth factor monoclonal antibody)], showed that overall survival and progression-free survival were significantly better with sorafenib. This combination therapy has become the new standard therapy for advanced HCC and has also attracted more attention in the treatment of HCC with anti-angiogenesis-immune combination therapy. Currently, the synergistic antitumor efficacy of this combination has been shown in many preclinical and clinical studies. In this review, we discuss the mechanism and clinical application of anti-angiogenics and immunotherapy in HCC, outline the relevant mechanism and rationality of the combined application of anti-angiogenics and ICIs, and point out the existing challenges of the combination therapy.
Core Tip: Anti-angiogenesis combined with immunotherapy in hepatocellular carcinoma (HCC) has attracted extensive attention. Although the mechanism of these combinations is largely understood, the biomarkers for predicting treatment response are still lacking. Thus, we outline the relevant mechanism and rationality of the combined application of anti-angiogenics and immune checkpoint inhibitors, and further explore the biomarkers that are associated with treatment response, which are critical in studies of HCC.
- Citation: Li SQ, Yang Y, Ye LS. Angiogenesis and immune checkpoint dual blockade: Opportunities and challenges for hepatocellular carcinoma therapy. World J Gastroenterol 2022; 28(42): 6034-6044
- URL: https://www.wjgnet.com/1007-9327/full/v28/i42/6034.htm
- DOI: https://dx.doi.org/10.3748/wjg.v28.i42.6034
According to the statistics from the National Cancer Center of China, the incidence and mortality of hepatocellular carcinoma (HCC) are increasing annually[1]. The World Health Organization (WHO) estimates that, from 2015 to 2030, approximately 10 million people in China will die due to liver cirrhosis and HCC. Although early-stage disease can be cured by surgical removed, transplantation, or interventional therapy, most patients have unresectable disease at the time of diagnosis[2], and current treatments are insufficient to prevent the high metastasis and recurrence rates after HCC treatment.
Currently, immunotherapy is receiving a great deal of attention in the treatment of tumors. Among the immunotherapy options, immune checkpoint blockade (ICB)-based immunotherapy which reactivates dysfunctional or exhausted T cells has shown excellent efficacy in a variety of solid cancers and hematological tumors[3-7]. However, 50%-80% of cancer patients still do not benefit from immunotherapy, and many of them suffer serious adverse events (AEs) during treatment[8]. In fact, there is still no clear mechanism to explain the tolerance of many cancers to immune checkpoint inhibitors (ICIs). HCC is a solid tumor with complex pathophysiological barriers. It is difficult for external lymphocytes to penetrate and infiltrate into tumor tissue. In addition, the rapidly growing tumor cells release immunosuppressive factors, which result in HCC forming an immunosuppressive immune microenvironment, which greatly limits the efficacy of immunotherapy[9]. In addition, the rapidly growing tumor cells release several factors, which result in HCC forming an immunosuppressive immune microenvironment, that greatly limits the efficacy of immunotherapy[10,11]. Therefore, normalizing tumor vasculature and improving the tumor hypoxic microenvironment is expected to reverse the immun
The TME is mainly composed of the vasculature, resident or infiltrating immune cells and various stromal cells. Previous studies have shown that abnormal tumor vasculature promotes the formation of an immunosuppressive TME[11]. Therefore, therapies that promote normalization of the vasculature are of great significance for enhancing immunotherapy of HCC. This review outlines measures to normalize the vasculature of HCC and the common immunotherapy regimens for HCC, and further describes and discusses how to normalize the tumor vasculature to improve the efficacy of immunotherapy in HCC (especially ICB). Additionally, we discuss the challenges associated with emerging combinations of vascular normalization therapy and immunotherapy for HCC.
The excessive growth and abnormal proliferation of tumor cells depend on rapid tumor angiogenesis. Tumor angiogenesis not only provides tumor cells with oxygen, nutrients and removes waste, but also serves as a channel for metastasis of tumor cells and immune cell infiltration[12,13]. However, compared with vessels in normal tissues, tumor neo-vessels have obvious aberrations in both structure and function[11]. Leakage is one of the most notable features of tumor vessels. On the one hand, this property can lead to tumor hypoxia and decreased intra-tumoral pH by impairing perfusion, and on the other hand, leakage will increase interstitial pressure in the TME[10]. Tumor cells overcome these harsh conditions through multiple mechanisms to gain a survival advantage[11]. Abnormal vessels limit the circulation of drugs and immune cells into the tumor, thereby limiting anti-tumor activity[11]. The hypoxia and pH reduction in the tumor caused by abnormal vessels will further lead to abnormal neovascularization, forming a vicious circle.
In the field of cancer research, most studies on angiogenesis has focused on the increased expression of angiogenesis factors [such as vascular endothelial growth factor (VEGF), platelet-derived growth factor (PDGF), fibroblast growth factor (FGF), angiopoietin, hepatocyte growth factor, endoglin (CD105, etc)] and decreased expression of anti-angiogenesis factors, such as angiostatin, endostatin, and thrombospondin 1, which is mainly caused by upregulation of the hypoxia-inducible factor protein. Among these, VEGF/VEGF receptor (VEGFR) axis are widely known to play a major role in vascular abnormalities and are crucial for the occurrence and progression of HCC[14-16]. The VEGF family consists of VEGFA, VEGFB, VEGFC, VEGFD and placental growth factor (PlGF) 1-4, which are involved in tumor angiogenesis (VEGFA, PlGF), maintenance of new blood vessels (VEGFB), lymph-angiogenesis and angiogenesis (VEGFC/D), vascular permeability (VEGFA/C), chemotaxis (VEGFB), migration (VEGFA, PlGF), differentiation (VEGFD) and survival (VEGFA/B/C, PlGF)[17,18]. VEGFR mainly includes VEGFR1, VEGFR2, and VEGFR3[15]. Of these, VEGFR2 is the critical receptor of this family, which is expressed on almost all endothelial cells and is activated by binding to VEGFA, VEGFC or VEGFD, and VEGFA is its major ligand[19]. This binding results in the phosphorylation cascade that triggers downstream cellular pathways, ultimately leading to endothelial cell proliferation and migration, and the formation and branching of new tumor blood vessels[19]. These neo-vessels often manifest as abnormal leaky vasculature, resulting in high interstitial pressure and severe hypoxia or necrosis in tissue regions, further promoting the malignant potential of tumor cells[20]. Previous studies have shown that circulating VEGF levels are increased in HCC, and increased VEGFA has been shown to be associated with angiogenesis in HCC[21,22]. In addition, related studies also observed a positive association between increased local and circulating VEGF and high tumor micro-vessel density with rapid disease progression and decreased survival. These findings support the application of therapies that target the VEGF/VEGFR pathway in HCC[21,22].
Various molecular and physical mechanisms have been reported to contribute to tumor vascular dysfunction, the most prominent of which is the imbalance of angiogenic signaling mediated by pro- and anti-angiogenic molecules[11,23]. In normal tissue, this balance is precisely maintained to ensure normal vascular morphology and function[24]. However, during the process of carcinogenesis, this balance usually tends to angiogenesis, and the generated neo-vessels are immature abnormal vessels without complete structure[24]. In view of the key role of the VEGF/VEGFR axis in abnormal angiogenesis of HCC, rational targeting of this axis can promote the normalization of tissue vessels and limit the occurrence and development of HCC.
In the past few decades, the development of anti-angiogenesis therapy has mainly focused on blocking VEGF[17,18]. Several studies have also focused on blocking VEGF signaling by silencing VEGFA expression at the transcriptional and post-transcriptional levels. For example, Zou et al[25] identified emodin that could greatly increase seryl-tRNA synthetase expression in tripe-negative breast cancer (TNBC) cells, consequently reducing VEGFA transcription, and emodin potently inhibited vascular development of zebrafish and blocked tumor angiogenesis in TNBC-bearing mice, greatly improving the survival. Li et al[26] and Ding et al[27] raised that VEGF small interference RNA can precisely and efficiently silence VEGF expression and block VEGF signal pathway, leading to a significant decrease in tumor blood vessels and suppression of tumor growth and metastasis. However, these studies have only been tested in animals. Preclinical evidence suggests that monotherapy which blocks VEGF reduces micro-vessel density, inhibits tumor growth in many cancerous subcutaneous xenografts, and even inhibits tumor cell metastasis[28,29]. Ferrara et al[30] researched and developed the first anti-angiogenesis inhibitor (bevacizumab), a recombinant humanized monoclonal antibody that blocks VEGFA. Bevacizumab binds to VEGF in the bloodstream, thereby inhibiting the interaction between VEGF and VEGFR. In clinical trials of combination therapy for HCC, multiple lines of evidence suggest that bevacizumab has a potential therapeutic effect[31,32].
On the other hand, many anti-angiogenesis therapies in HCC focus on targeting VEGFR. Multikinase inhibitors (MKIs) and monoclonal antibodies (mAbs) were developed to inhibit VEGFR and its downstream targets to inhibit endothelial cell proliferation, thereby reducing the nutrient and oxygen supply required by tumors. Currently, the VEGFR-targeted MKIs and mAbs used in advanced HCC mainly include sorafenib, regorafenib, lenvatinib, cabozantinib, and ramucirumab (Table 1).
| Therapy | Type | Line of therapy | Target | Study name | Primary outcome in the study (mo) (95%CI) | Adverse events in the study [affected/at risk (%)] |
| Anti-VEGFA | ||||||
| Bevacizumab (combination with atezolizumab) | mAb | First line (plus atezolizumab) | VEGF-A | IMBrave150 | OS at CCOD 30 mo: 19.2 (17.0-23.7); PFS: 6.8 (5.6-8.3) | Gastrointestinal perforation [1/329 (0.30%)], haemorrhage [40/329 (12.15%)], hypertension [2/329 (0.61%)] and proteinuria [100/329 (30.40%)], etc. |
| Anti-VEGFR | ||||||
| Sorafenib | TKIs | First line | VEGFR1-3, c-Kit, PDGFR-b, and FLT-3 | SHARP | OS: 10.8 (9.5-13.5); TTSP: 4.2 (3.5-4.2) | Diarrhea [166/297 (55.89%)], hand-foot skin reactions [2/297 (0.67%)], fatigue [145/297 (48.82%)], etc. |
| Lenvatinib | TKIs | First line | VEGFR1-3, FGFR1-4, PDGFRa, RET and c-Kit | REFLECT | OS: 13.6 (12.1–14.9); PFS: 7.4 (6.9–8.8) | Diarrhea [154/468 (33%)], decreased appetite [204/468 (44%)], etc. |
| Regorafenib | TKIs | Second line | VEGFR, RET, c-Kit, B-Raf, FGFR1 and PDGFR | RESORCE | OS: 10.7 (9.2-12.3); TTP:3.9 (2.9-4.26) | Toxicities were manageable this sorafenib- tolerant population and were similar to those observed with sorafenib, including diarrhea [163/374 (43.58%)], etc. |
| Cabozantinib | TKIs | Second and third line | VEGFR2, c-Kit, RET, FLT-3, Tie2, and Axl | CELESTIAL | OS: 10.2 (9.1-12.0) | Hypertension [137/467 (29.34%)]. Pneumonia [16/467 (3.43%)], etc. |
| Ramucirumab | mAb | Second line and AFP ≥ 400 ng/mL | VEGFR2 | REACH-2 | OS: 8.5 (7.0-10.6) | Hypertension [48/197 (24.37%)], hyponatremia [11/197 (5.58%)], etc. |
Sorafenib is an oral MKI that blocks VEGFR1-3, c-Kit, PDGF receptor (PDGFR)-b, and FMS-like tyrosine kinase-3 (FLT-3)[33]. The phase III clinical trial SHARP showed a 2.8-mo survival advantage for sorafenib over placebo in patients with advanced HCC. This recent study also showed that sorafenib can benefit patients with HCC regardless of etiology, and patients with hepatitis C appeared to experience greater benefit[34]. Treatment-related AEs were more common in the sorafenib group than in the placebo group (80% vs 52%), and the incidence of dose reductions and interruptions was high during treatment. The MKI regorafenib also targets VEGFR, RET, c-Kit, B-Raf, FGFR1 and PDGFR[33]. It is the first therapy to demonstrate a survival benefit in advanced HCC patients who have progressed on sorafenib[35]. Fatigue, hypertension, diarrhea and hand-foot skin reactions were common AEs in the regorafenib-treated group. Other analyses showed that the survival benefit of first line sorafenib and second line regorafenib was more than 24 mo[35]. The targets of lenvatinib include VEGFR1-3, FGFR1-4, PDGFRa, RET and c-Kit. Recently, a phase III study of lenvatinib vs sorafenib in patients with unresectable HCC showed that overall survival (OS) with lenvatinib was non-inferior to sorafenib[36]. The most common AEs in the lenvatinib group were diarrhea, fatigue, etc[36]. It should be noted that patients with tumors with more than 50% hepatic masses or involvement of branches of the main portal vein were excluded from the trial (NCT01761266); thus, further clinical trials should be conducted. Despite this problem, lenvatinib remains the only drug in first-line clinical trials that was positive against the proven active control, sorafenib. In addition to targeting VEGFR2, c-Kit, RET, FLT-3, Tie2, and Axl, cabozantinib has the unique property of inhibiting c-Met, and its potential activity was observed in phase II trials[37]. The subsequent phase III clinical trial, which compared cabozantinib to placebo in advanced HCC, met its primary endpoint of improved OS after up to two prior treatments, one of which included sorafenib[38]. Hypertension, pneumonia were common AEs in the cabozantinib group[38]. Ramucirumab, the mAb that antagonizes VEGFR2, improved OS in a phase III study in patients with sorafenib progression or intolerance with baseline alpha-fetoprotein (AFP) ≥ 400 ng/mL[39]. Hypertension and hyponatremia were the only over grade 3 AEs in patients of the test group[39]. Based on the results of the previous phase III study, patients can be selected for treatment based on baseline AFP values. A survival benefit was observed with ramucirumab in a subgroup of patients with higher baseline AFP (400 ng/mL), which is the first positive clinical trial in the biomarker-selected HCC population[39].
Driven by the success of ICB therapy in melanoma, ICB has been extensively studied in a variety of malignancies including HCC[40,41]. Long-term liver injury or chronic hepatitis infection often leaves the liver in a state of chronic inflammation[42]. Moderate inflammation can defend against pathogens and repair tissue damage, whereas persistent liver inflammation can disturb the microenvironment, thus favoring carcinogenesis. On the one hand, hepatic endocrine cytokines play a key role in tumorigenesis through regulating regulatory T cells (Tregs) that inhibit CD8+ T cell activation[43]. It was previously reported that Tregs derived from hepatitis B virus-positive HCC tumors exhibited higher programmed cell death protein 1 (PD-1) expression and superior inhibitory capacity against CD8+ T cells. On the other hand, cytotoxic immune populations frequently express markers of exhaustion such as PD-1, cytotoxic t-lymphocyte associated antigen 4 (CTLA-4) and lymphocyte activating gene 3[44]. Under normal conditions, these molecules inhibit T cell activation to maintain inflammatory homeostasis, protect tissue integrity, and prevent unnecessary autoimmunity[45]. However, in tumors, expression of these markers of exhaustion is inversely correlated with their function, making them a prime target for ICBs to revitalize and restore the cytotoxic capacity of CD8+ T cells[43]. At the same time, the expression of PD-1 and its ligand (PD-L1) in tumor cells is upregulated, and when it binds to PD-1 expressed by T cells activated by tumor infiltration, it induces T cell exhaustion and suppresses the anti-tumor immune activity of these immune cells, thereby enabling tumor cells to evade immunity[46]. ICBs generate robust multitarget immune responses and can even induce long-lasting tumor remissions in some patients. Inhibition of the PD-1/PD-L1 interaction reverses the depleted state of these cytotoxic immune cells and reactivates their antitumor activity[46,47]. In HCC, the mAbs pembrolizumab and nivolumab that target PD-1, and nivolumab combined with ipilimumab (a mAb directed against CTLA-4), has been approved in the United States for sorafenib-treated patients, based on promising results from clinical trials[48-50]. A clinical trial confirmed the efficacy and safety of PD-1-targeting immunotherapy in HCC[51]. However, subsequent phase III trials of nivolumab vs sorafenib in first-line therapy failed to meet the primary survival endpoints[50,52]. The combination of phase III nivolumab and ipilimumab is currently under evaluation (NCT04039607).
In the current phase III clinical treatment of HCC, immune combination therapy has attracted considerable attention, especially the combination of ICIs and anti-angiogenic inhibitors (Table 2). Lenvatinib combined with pembrolizumab, bevacizumab combined with atezolizumab (PD-L1-targeting mAb) (T+A combination), and cabozantinib combined with atezolizumab have all obtained encouraging results in clinical studies[53-55]. Of these, the combination of bevacizumab and atezolizumab has been successful in phase III clinical trials of first-line treatment of HCC (IMbrave150). Compared with sorafenib, this combination improved the primary endpoints: OS and progression-free survival, and this combination was shown to be safe and improved quality of life[56]. Hypertension and proteinuria were typical side effects of bevacizumab and were the top two AEs in this test group. Upper gastrointestinal bleeding, another known side effect of bevacizumab, occurred in 7% of patients in this group, and was within the range of previous evaluations of bevacizumab AEs for the treatment of HCC[57,58]. Elevated transaminases and pruritus are common side effects of atezolizumab[32]. As this study applied the usual inclusion criteria in HCC clinical trials and included only Child-Pugh A patients, further clinical trials are pending[32].
| Trial name | Treatment | Setting | Primary endpoints | Current status | Enrollment, n |
| NCT03434379 | Atezolizumab + bevacizumab | Advanced HCC; first line | OS, PFS | Active, not recruiting | 558 |
| NCT04770896 | Atezolizumab + lenvatinib/sorafenib versus lenvatinib/sorafenib | Unresectable HCC; second line | OS | Recruiting | 554 |
| NCT03713593 | Pembrolizumab + lenvatinib versus lenvatinib | Advanced HCC; first line | OS, PFS | Active, not recruiting | 794 |
| NCT03755791 | Atezolizumab + cabozantinib versus sorafenib | Advanced HCC; first line | OS, PFS | Recruiting | 740 |
As shown above, the success of the phase III trial (IMbrave150) with the combination of atezolizumab and bevacizumab in advanced HCC is groundbreaking as it is the first treatment with a better survival rate than sorafenib since the approval of sorafenib in 2007 and is also the only successful first-line immunotherapy combination therapy for HCC in the world[32]. Antibodies targeting VEGF not only inhibit tumor growth but also reprogram the TME from immunosuppressive to immune activation[8,59]. Based on these findings, PD-1/PD-L1 inhibitors combined with VEGF/VEGFR inhibitors have attracted extensive attention in the treatment of HCC. Next, we will outline the mechanism and rationality of PD-1/PD-L1 inhibitors combined with VEGF/VEGFR inhibitor therapy, and the biomarkers of response to targeted immune combination therapy.
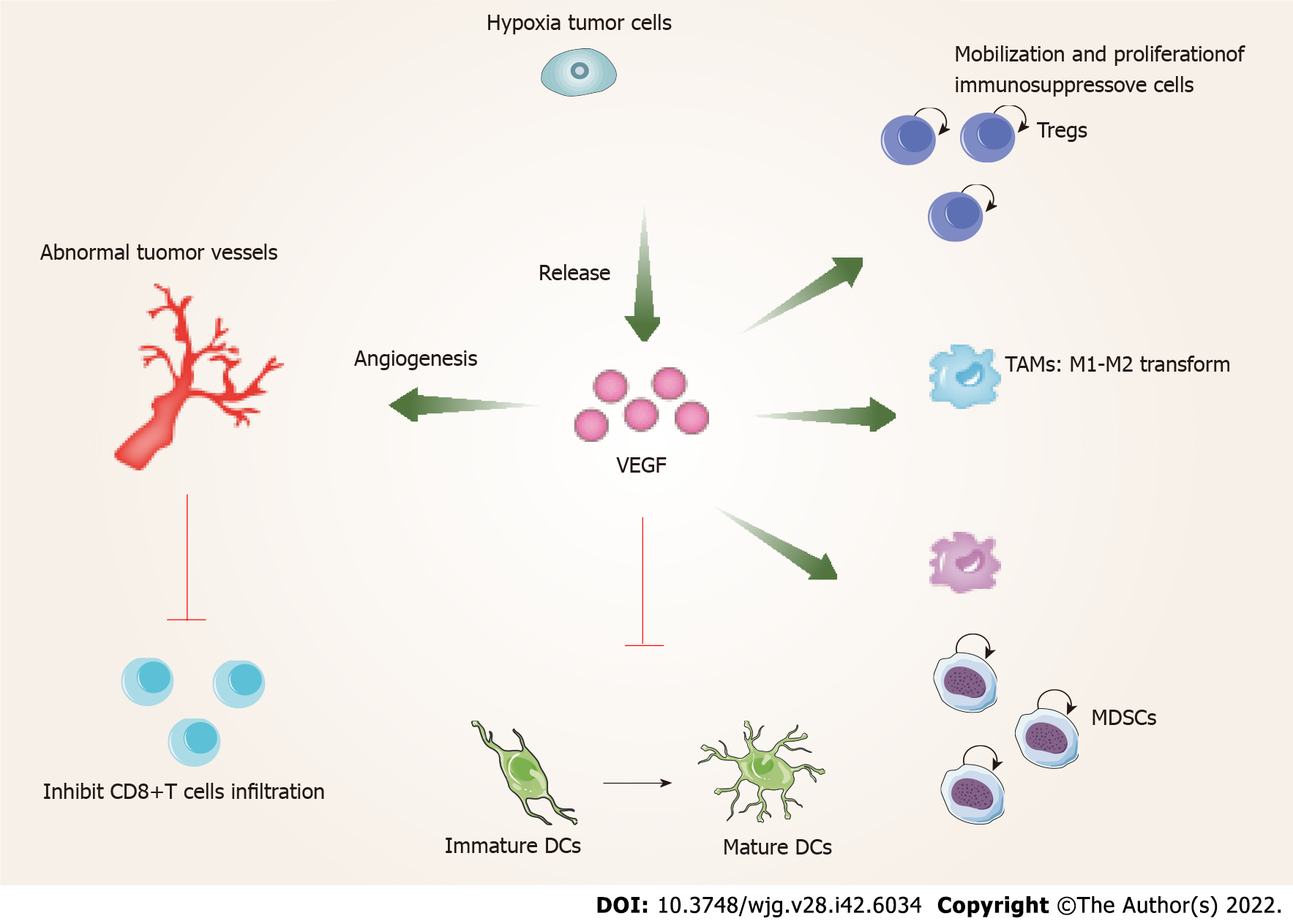
In the tumor area, VEGF released by hypoxic cancer cells promotes tumor cell growth and metastasis by angiogenesis[8]. On the other hand, VEGF can also promote the malignant progression of tumors by affecting the TME (Figure 1). The tumor immune cycle mainly includes seven steps, the release of tumor antigens, the uptake and presentation of tumor antigens by dendritic cells (DCs), the initiation and activation of T cells, the migration of T cells to tumors, the invasion of T cells against tumors, the recognition of tumor cells by T cells, and the attack of tumor cells by T cells[60]. VEGF is involved in almost every step of the tumor immune cycle and finally promotes tumor immune escape[61-64]. VEGF enhances the mobilization and proliferation of immunosuppressive cells, including Tregs, tumor-associated macrophages (TAMs), and myeloid-derived suppressor cells (MDSCs) etc, enhances the release of immunosuppressive cytokines[8,59], and promotes M1-TAMs to M2 type polarization. In addition, Tregs and TAMs release immunosuppressive factors, such as VEGF and angiopoietin 2, which form positive feedback to further promote tumor progression[65]. Furthermore, VEGF inhibits the maturation and antigen presentation of DCs. Thus, even in the presence of neoantigens, VEGF can reduce the proliferation and activation of naive CD8+ cells by inhibiting DCs[65]. VEGF prevents antigen activated CD8+ T cells from infiltrating tumor tissue by promoting the formation of abnormal tumor vessels. In addition, VEGF forms an immunosuppressive TME that inhibits the function of T cells in tumors during the effector phase of the immune response[65]. Therefore, inhibition of VEGF/VEGFR interaction not only normalizes vessels but also enhances antitumor immunity.
Previous studies have shown that inhibition of VEGF/VEGFR can enhance antitumor immunity. Gabrilovich et al[63] found that the targeted drugs which inhibit VEGF/VEGFR lead to an enhanced antigen-presenting capacity of DCs. In addition, studies also found that these drugs inhibit the production of Tregs, TAMs and MDSCs at tumor sites, and negatively regulate the expression of immunosuppressive cytokines such as transforming growth factor-beta and interleukin-10[66]. Therefore, blocking VEGF/VEGFR reprograms the immunosuppressive TME[67]. At the same time, the combination of PD-1/PD-L1 antibody can further enhance the antitumor activity of T cells. First, by reversing the VEGF-mediated suppression of DCs maturation resulting in efficient priming and activation of T cells[67,68]; second, by normalizing tumor vasculature and promoting efficient T cell infiltration into tumors[69]; and third, VEGF/VEGFR inhibitors inhibit the activity of MDSCs, Tregs, and TAMs, leading to the reprogramming of the immunosuppressive microenvironment to immune activation[61]. Finally, PD-1/PD-L1 inhibitors enhance the ability of T cells to attack tumor cells. These four aspects can lead to effective antitumor immunity and tumor growth inhibition. As described above, the use of molecularly targeted drugs against VEGF/VEGFR reactivates the aberrant immunosuppressive TME caused by VEGF, and finally allows the tumor cells to be effectively attacked[60,62]. A recent study of T+A therapy showed that the improved outcome of the combination of bevacizumab and atezolizumab compared with atezolizumab alone was mainly related to higher Tregs expression, suggesting that bevacizumab inhibits VEGF-enhanced antitumor immunity mainly related to inhibiting the function of Tregs[70]. Notably, previous studies have shown that anti-PD-1 therapy can increase tumor blood perfusion by normalizing blood vessels in breast and colorectal cancer models, which is closely related to its antitumor efficacy[71]. These studies form the rationale for the combination of VEGF/VEGFR inhibitors and PD-1/PD-L1 inhibitors.
A major factor limiting the benefit of angiogenesis and immune checkpoint dual blockade for HCC is the lack of biomarkers to predict patients who can benefit from treatment. PD-L1 expression in tumor specimens was not associated with prognosis in HCC patients treated with nivolumab[48]. This study demonstrated that further comprehensive tumor and stromal immune scores, or tumor gene signatures should be explored. A recent study showed that pre-existing immunity (high expression of CD274, T-effector signature and intra-tumoral CD8+ T cell density) was positively associated with better clinical outcomes with the T+A combination and reduced clinical benefit was associated with a high Treg to effector T cell (Teff) ratio and expression of oncofetal genes (GPC3, AFP)[70]. Hatanaka et al[72] reported that C-reactive protein and AFP could be useful for predicting therapeutic outcomes and treatment-related AEs in HCC patients treated with the T+A combination. These results indicate that the clinical studies of bevacizumab combined with atezolizumab, lenvatinib combined with pembrolizumab, cabozantinib combined with atezolizumab and other combination therapies are a valuable platform for the analysis of other potential biomarkers of response to targeted immunotherapy and offer several new possibilities for identifying subpopulations of patients who benefit from these treatments. In several cancers, tumor mutational burden (TMB) and microsatellite instability (MSI) are associated with longer OS after ICB therapy. Considering the low frequency of TMB and MSI in HCC, their predictive applications in HCC are limited. However, it is worth noting that studies have shown that HCC with high TMB and low MSI responded to nivolumab for more than 2 years[73]. Thus, much more research is needed to determine the biomarkers of targeted immunotherapy in HCC.
The global disease burden of HCC is increasing year by year. According to statistics, the annual incidence of HCC may exceed 1 million cases in the near future, and most patients are in advanced stages at diagnosis. Currently, only reasonable systemic treatment can effectively delay the progression of HCC. This article describes the characteristics and treatment strategies of abnormal angiogenesis in HCC, and briefly reviews the immunotherapy of HCC. The strategy and rationality of angiogenesis and immune checkpoint dual blockade are further discussed. Among these combinatorial strategies, the success of the IMBrave150 clinical trial demonstrated that bevacizumab altered the tumor immune microenvironment, enabling greater responses to ICB, successfully transforming the immunosuppressive TME to an immune-activated microenvironment. Therefore, the efficacy achieved by the combination of anti-PD-1/PD-L1 antibody and VEGF/VEGFR inhibitor may be due to normalization of the TME. In addition to the combination of atezolizumab and bevacizumab, other combination therapies targeting the same mechanism have also received attention. While the clinical development of VEGF/VEGFR-targeted drugs is due to their anti-angiogenesis inhibitory effects, the potential of this class of drugs is as immunomodulators in combination with immunotherapy.
In the post-sorafenib era of advanced HCC treatment, a great number of combination therapies are being studied. However, one of the biggest challenges with combination therapy is the discovery of predictive biomarkers to accurately identify patients most likely to respond to treatment. In the study of HCC, anti-angiogenesis therapy has been used for more than a decade and ICB has been approved for several years, but these two therapies still lack convincing biomarkers. Therefore, for combination therapy, a better understanding of the mechanism of synergistic therapeutic effect and the discovery of predictive biomarkers will help to design more precise treatment regimens.
Provenance and peer review: Invited article; Externally peer reviewed.
Peer-review model: Single blind
Specialty type: Gastroenterology and hepatology
Country/Territory of origin: China
Peer-review report’s scientific quality classification
Grade A (Excellent): 0
Grade B (Very good): B, B
Grade C (Good): C
Grade D (Fair): 0
Grade E (Poor): 0
P-Reviewer: Kwee S, United States; Liang Y, China; Sehrawat A, India S-Editor: Wang JJ L-Editor: A P-Editor: Wang JJ
| 1. | Xu XF, Xing H, Han J, Li ZL, Lau WY, Zhou YH, Gu WM, Wang H, Chen TH, Zeng YY, Li C, Wu MC, Shen F, Yang T. Risk Factors, Patterns, and Outcomes of Late Recurrence After Liver Resection for Hepatocellular Carcinoma: A Multicenter Study From China. JAMA Surg. 2019;154:209-217. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 375] [Cited by in RCA: 394] [Article Influence: 65.7] [Reference Citation Analysis (0)] |
| 2. | Lau WY, Leung TW, Lai BS, Liew CT, Ho SK, Yu SC, Tang AM. Preoperative systemic chemoimmunotherapy and sequential resection for unresectable hepatocellular carcinoma. Ann Surg. 2001;233:236-241. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 65] [Cited by in RCA: 85] [Article Influence: 3.5] [Reference Citation Analysis (0)] |
| 3. | Topalian SL, Taube JM, Anders RA, Pardoll DM. Mechanism-driven biomarkers to guide immune checkpoint blockade in cancer therapy. Nat Rev Cancer. 2016;16:275-287. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 1659] [Cited by in RCA: 2093] [Article Influence: 232.6] [Reference Citation Analysis (0)] |
| 4. | Mellman I, Coukos G, Dranoff G. Cancer immunotherapy comes of age. Nature. 2011;480:480-489. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 2348] [Cited by in RCA: 2898] [Article Influence: 207.0] [Reference Citation Analysis (0)] |
| 5. | Chen X, Xu R, He D, Zhang Y, Chen H, Zhu Y, Cheng Y, Liu R, Zhu R, Gong L, Xiao M, Wang Z, Deng L, Cao K. CD8+ T effector and immune checkpoint signatures predict prognosis and responsiveness to immunotherapy in bladder cancer. Oncogene. 2021;40:6223-6234. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 14] [Cited by in RCA: 61] [Article Influence: 15.3] [Reference Citation Analysis (0)] |
| 6. | Zaremba A, Eggermont AMM, Robert C, Dummer R, Ugurel S, Livingstone E, Ascierto PA, Long GV, Schadendorf D, Zimmer L. The concepts of rechallenge and retreatment with immune checkpoint blockade in melanoma patients. Eur J Cancer. 2021;155:268-280. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 13] [Cited by in RCA: 54] [Article Influence: 13.5] [Reference Citation Analysis (0)] |
| 7. | Keenan TE, Tolaney SM. Role of Immunotherapy in Triple-Negative Breast Cancer. J Natl Compr Canc Netw. 2020;18:479-489. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 119] [Cited by in RCA: 370] [Article Influence: 74.0] [Reference Citation Analysis (0)] |
| 8. | Fukumura D, Kloepper J, Amoozgar Z, Duda DG, Jain RK. Enhancing cancer immunotherapy using antiangiogenics: opportunities and challenges. Nat Rev Clin Oncol. 2018;15:325-340. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 745] [Cited by in RCA: 1389] [Article Influence: 198.4] [Reference Citation Analysis (0)] |
| 9. | Hanahan D, Weinberg RA. Hallmarks of cancer: the next generation. Cell. 2011;144:646-674. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 51728] [Cited by in RCA: 47023] [Article Influence: 3358.8] [Reference Citation Analysis (5)] |
| 10. | Jain RK. Normalizing tumor microenvironment to treat cancer: bench to bedside to biomarkers. J Clin Oncol. 2013;31:2205-2218. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 833] [Cited by in RCA: 892] [Article Influence: 74.3] [Reference Citation Analysis (0)] |
| 11. | Jain RK. Antiangiogenesis strategies revisited: from starving tumors to alleviating hypoxia. Cancer Cell. 2014;26:605-622. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 1120] [Cited by in RCA: 1189] [Article Influence: 108.1] [Reference Citation Analysis (0)] |
| 12. | Pinho SS, Reis CA. Glycosylation in cancer: mechanisms and clinical implications. Nat Rev Cancer. 2015;15:540-555. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 1664] [Cited by in RCA: 2180] [Article Influence: 218.0] [Reference Citation Analysis (0)] |
| 13. | Jain RK, Koenig GC, Dellian M, Fukumura D, Munn LL, Melder RJ. Leukocyte-endothelial adhesion and angiogenesis in tumors. Cancer Metastasis Rev. 1996;15:195-204. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 77] [Cited by in RCA: 76] [Article Influence: 2.6] [Reference Citation Analysis (0)] |
| 14. | Xiong XX, Qiu XY, Hu DX, Chen XQ. Advances in Hypoxia-Mediated Mechanisms in Hepatocellular Carcinoma. Mol Pharmacol. 2017;92:246-255. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 82] [Cited by in RCA: 83] [Article Influence: 10.4] [Reference Citation Analysis (0)] |
| 15. | Kim KR, Moon HE, Kim KW. Hypoxia-induced angiogenesis in human hepatocellular carcinoma. J Mol Med (Berl). 2002;80:703-714. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 79] [Cited by in RCA: 81] [Article Influence: 3.5] [Reference Citation Analysis (0)] |
| 16. | von Marschall Z, Cramer T, Höcker M, Finkenzeller G, Wiedenmann B, Rosewicz S. Dual mechanism of vascular endothelial growth factor upregulation by hypoxia in human hepatocellular carcinoma. Gut. 2001;48:87-96. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 130] [Cited by in RCA: 144] [Article Influence: 6.0] [Reference Citation Analysis (0)] |
| 17. | Ferrara N. VEGF and the quest for tumour angiogenesis factors. Nat Rev Cancer. 2002;2:795-803. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 1091] [Cited by in RCA: 1108] [Article Influence: 48.2] [Reference Citation Analysis (0)] |
| 18. | Ferrara N, Gerber HP, LeCouter J. The biology of VEGF and its receptors. Nat Med. 2003;9:669-676. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 6747] [Cited by in RCA: 6941] [Article Influence: 315.5] [Reference Citation Analysis (0)] |
| 19. | Amini A, Masoumi Moghaddam S, Morris DL, Pourgholami MH. The critical role of vascular endothelial growth factor in tumor angiogenesis. Curr Cancer Drug Targets. 2012;12:23-43. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 66] [Cited by in RCA: 82] [Article Influence: 6.3] [Reference Citation Analysis (0)] |
| 20. | Zhu AX, Duda DG, Sahani DV, Jain RK. HCC and angiogenesis: possible targets and future directions. Nat Rev Clin Oncol. 2011;8:292-301. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 429] [Cited by in RCA: 440] [Article Influence: 31.4] [Reference Citation Analysis (0)] |
| 21. | Poon RT, Fan ST, Wong J. Clinical significance of angiogenesis in gastrointestinal cancers: a target for novel prognostic and therapeutic approaches. Ann Surg. 2003;238:9-28. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 39] [Cited by in RCA: 54] [Article Influence: 2.5] [Reference Citation Analysis (0)] |
| 22. | Poon RT, Fan ST, Wong J. Clinical implications of circulating angiogenic factors in cancer patients. J Clin Oncol. 2001;19:1207-1225. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 428] [Cited by in RCA: 426] [Article Influence: 17.8] [Reference Citation Analysis (0)] |
| 23. | Jain RK, Martin JD, Stylianopoulos T. The role of mechanical forces in tumor growth and therapy. Annu Rev Biomed Eng. 2014;16:321-346. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 549] [Cited by in RCA: 671] [Article Influence: 67.1] [Reference Citation Analysis (0)] |
| 24. | Jain RK. Normalization of tumor vasculature: an emerging concept in antiangiogenic therapy. Science. 2005;307:58-62. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 4319] [Cited by in RCA: 3955] [Article Influence: 197.8] [Reference Citation Analysis (0)] |
| 25. | Zou G, Zhang X, Wang L, Li X, Xie T, Zhao J, Yan J, Ye H, Jiao S, Xiang R, Shi Y. Herb-sourced emodin inhibits angiogenesis of breast cancer by targeting VEGFA transcription. Theranostics. 2020;10:6839-6853. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 69] [Cited by in RCA: 94] [Article Influence: 18.8] [Reference Citation Analysis (0)] |
| 26. | Li F, Wang Y, Chen WL, Wang DD, Zhou YJ, You BG, Liu Y, Qu CX, Yang SD, Chen MT, Zhang XN. Co-delivery of VEGF siRNA and Etoposide for Enhanced Anti-angiogenesis and Anti-proliferation Effect via Multi-functional Nanoparticles for Orthotopic Non-Small Cell Lung Cancer Treatment. Theranostics. 2019;9:5886-5898. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 39] [Cited by in RCA: 78] [Article Influence: 13.0] [Reference Citation Analysis (0)] |
| 27. | Ding X, Su Y, Wang C, Zhang F, Chen K, Wang Y, Li M, Wang W. Synergistic Suppression of Tumor Angiogenesis by the Co-delivering of Vascular Endothelial Growth Factor Targeted siRNA and Candesartan Mediated by Functionalized Carbon Nanovectors. ACS Appl Mater Interfaces. 2017;9:23353-23369. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 44] [Cited by in RCA: 52] [Article Influence: 6.5] [Reference Citation Analysis (0)] |
| 28. | Ferrara N, Carver-Moore K, Chen H, Dowd M, Lu L, O'Shea KS, Powell-Braxton L, Hillan KJ, Moore MW. Heterozygous embryonic lethality induced by targeted inactivation of the VEGF gene. Nature. 1996;380:439-442. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 2693] [Cited by in RCA: 2564] [Article Influence: 88.4] [Reference Citation Analysis (0)] |
| 29. | Lieu CH, Tran H, Jiang ZQ, Mao M, Overman MJ, Lin E, Eng C, Morris J, Ellis L, Heymach JV, Kopetz S. The association of alternate VEGF ligands with resistance to anti-VEGF therapy in metastatic colorectal cancer. PLoS One. 2013;8:e77117. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 73] [Cited by in RCA: 105] [Article Influence: 8.8] [Reference Citation Analysis (0)] |
| 30. | Ferrara N, Hillan KJ, Novotny W. Bevacizumab (Avastin), a humanized anti-VEGF monoclonal antibody for cancer therapy. Biochem Biophys Res Commun. 2005;333:328-335. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 649] [Cited by in RCA: 701] [Article Influence: 35.1] [Reference Citation Analysis (0)] |
| 31. | Cheng AL, Qin S, Ikeda M, Galle PR, Ducreux M, Kim TY, Lim HY, Kudo M, Breder V, Merle P, Kaseb AO, Li D, Verret W, Ma N, Nicholas A, Wang Y, Li L, Zhu AX, Finn RS. Updated efficacy and safety data from IMbrave150: Atezolizumab plus bevacizumab vs. sorafenib for unresectable hepatocellular carcinoma. J Hepatol. 2022;76:862-873. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 309] [Cited by in RCA: 946] [Article Influence: 315.3] [Reference Citation Analysis (0)] |
| 32. | Finn RS, Qin S, Ikeda M, Galle PR, Ducreux M, Kim TY, Kudo M, Breder V, Merle P, Kaseb AO, Li D, Verret W, Xu DZ, Hernandez S, Liu J, Huang C, Mulla S, Wang Y, Lim HY, Zhu AX, Cheng AL; IMbrave150 Investigators. Atezolizumab plus Bevacizumab in Unresectable Hepatocellular Carcinoma. N Engl J Med. 2020;382:1894-1905. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 2542] [Cited by in RCA: 4659] [Article Influence: 931.8] [Reference Citation Analysis (2)] |
| 33. | Cheng AL, Kang YK, Chen Z, Tsao CJ, Qin S, Kim JS, Luo R, Feng J, Ye S, Yang TS, Xu J, Sun Y, Liang H, Liu J, Wang J, Tak WY, Pan H, Burock K, Zou J, Voliotis D, Guan Z. Efficacy and safety of sorafenib in patients in the Asia-Pacific region with advanced hepatocellular carcinoma: a phase III randomised, double-blind, placebo-controlled trial. Lancet Oncol. 2009;10:25-34. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 3854] [Cited by in RCA: 4645] [Article Influence: 273.2] [Reference Citation Analysis (0)] |
| 34. | Bruix J, Cheng AL, Meinhardt G, Nakajima K, De Sanctis Y, Llovet J. Prognostic factors and predictors of sorafenib benefit in patients with hepatocellular carcinoma: Analysis of two phase III studies. J Hepatol. 2017;67:999-1008. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 360] [Cited by in RCA: 452] [Article Influence: 56.5] [Reference Citation Analysis (0)] |
| 35. | Bruix J, Qin S, Merle P, Granito A, Huang YH, Bodoky G, Pracht M, Yokosuka O, Rosmorduc O, Breder V, Gerolami R, Masi G, Ross PJ, Song T, Bronowicki JP, Ollivier-Hourmand I, Kudo M, Cheng AL, Llovet JM, Finn RS, LeBerre MA, Baumhauer A, Meinhardt G, Han G; RESORCE Investigators. Regorafenib for patients with hepatocellular carcinoma who progressed on sorafenib treatment (RESORCE): a randomised, double-blind, placebo-controlled, phase 3 trial. Lancet. 2017;389:56-66. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 2160] [Cited by in RCA: 2705] [Article Influence: 338.1] [Reference Citation Analysis (0)] |
| 36. | Llovet JM, Hernandez-Gea V. Hepatocellular carcinoma: reasons for phase III failure and novel perspectives on trial design. Clin Cancer Res. 2014;20:2072-2079. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 272] [Cited by in RCA: 323] [Article Influence: 29.4] [Reference Citation Analysis (0)] |
| 37. | Schöffski P, Gordon M, Smith DC, Kurzrock R, Daud A, Vogelzang NJ, Lee Y, Scheffold C, Shapiro GI. Phase II randomised discontinuation trial of cabozantinib in patients with advanced solid tumours. Eur J Cancer. 2017;86:296-304. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 49] [Cited by in RCA: 61] [Article Influence: 7.6] [Reference Citation Analysis (0)] |
| 38. | Abou-Alfa GK, Meyer T, Cheng AL, El-Khoueiry AB, Rimassa L, Ryoo BY, Cicin I, Merle P, Chen Y, Park JW, Blanc JF, Bolondi L, Klümpen HJ, Chan SL, Zagonel V, Pressiani T, Ryu MH, Venook AP, Hessel C, Borgman-Hagey AE, Schwab G, Kelley RK. Cabozantinib in Patients with Advanced and Progressing Hepatocellular Carcinoma. N Engl J Med. 2018;379:54-63. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 1630] [Cited by in RCA: 1763] [Article Influence: 251.9] [Reference Citation Analysis (0)] |
| 39. | Zhu AX, Kang YK, Yen CJ, Finn RS, Galle PR, Llovet JM, Assenat E, Brandi G, Pracht M, Lim HY, Rau KM, Motomura K, Ohno I, Merle P, Daniele B, Shin DB, Gerken G, Borg C, Hiriart JB, Okusaka T, Morimoto M, Hsu Y, Abada PB, Kudo M; REACH-2 study investigators. Ramucirumab after sorafenib in patients with advanced hepatocellular carcinoma and increased α-fetoprotein concentrations (REACH-2): a randomised, double-blind, placebo-controlled, phase 3 trial. Lancet Oncol. 2019;20:282-296. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 1027] [Cited by in RCA: 1242] [Article Influence: 207.0] [Reference Citation Analysis (0)] |
| 40. | Yang W, Feng Y, Zhou J, Cheung OK, Cao J, Wang J, Tang W, Tu Y, Xu L, Wu F, Tan Z, Sun H, Tian Y, Wong J, Lai PB, Chan SL, Chan AW, Tan PB, Chen Z, Sung JJ, Yip KY, To KF, Cheng AS. A selective HDAC8 inhibitor potentiates antitumor immunity and efficacy of immune checkpoint blockade in hepatocellular carcinoma. Sci Transl Med. 2021;13. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 25] [Cited by in RCA: 102] [Article Influence: 25.5] [Reference Citation Analysis (0)] |
| 41. | Llovet JM, Castet F, Heikenwalder M, Maini MK, Mazzaferro V, Pinato DJ, Pikarsky E, Zhu AX, Finn RS. Immunotherapies for hepatocellular carcinoma. Nat Rev Clin Oncol. 2022;19:151-172. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 206] [Cited by in RCA: 1002] [Article Influence: 334.0] [Reference Citation Analysis (2)] |
| 42. | Medzhitov R. Origin and physiological roles of inflammation. Nature. 2008;454:428-435. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 3548] [Cited by in RCA: 4367] [Article Influence: 256.9] [Reference Citation Analysis (0)] |
| 43. | Farhood B, Najafi M, Mortezaee K. CD8+ cytotoxic T lymphocytes in cancer immunotherapy: A review. J Cell Physiol. 2019;234:8509-8521. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 535] [Cited by in RCA: 1115] [Article Influence: 159.3] [Reference Citation Analysis (0)] |
| 44. | Barber DL, Wherry EJ, Masopust D, Zhu B, Allison JP, Sharpe AH, Freeman GJ, Ahmed R. Restoring function in exhausted CD8 T cells during chronic viral infection. Nature. 2006;439:682-687. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 2815] [Cited by in RCA: 3158] [Article Influence: 157.9] [Reference Citation Analysis (0)] |
| 45. | Pardoll DM. The blockade of immune checkpoints in cancer immunotherapy. Nat Rev Cancer. 2012;12:252-264. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 9936] [Cited by in RCA: 10321] [Article Influence: 793.9] [Reference Citation Analysis (34)] |
| 46. | Xie F, Xu M, Lu J, Mao L, Wang S. The role of exosomal PD-L1 in tumor progression and immunotherapy. Mol Cancer. 2019;18:146. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 124] [Cited by in RCA: 250] [Article Influence: 41.7] [Reference Citation Analysis (0)] |
| 47. | Yao H, Lan J, Li C, Shi H, Brosseau JP, Wang H, Lu H, Fang C, Zhang Y, Liang L, Zhou X, Wang C, Xue Y, Cui Y, Xu J. Inhibiting PD-L1 palmitoylation enhances T-cell immune responses against tumours. Nat Biomed Eng. 2019;3:306-317. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 173] [Cited by in RCA: 358] [Article Influence: 59.7] [Reference Citation Analysis (0)] |
| 48. | El-Khoueiry AB, Sangro B, Yau T, Crocenzi TS, Kudo M, Hsu C, Kim TY, Choo SP, Trojan J, Welling TH Rd, Meyer T, Kang YK, Yeo W, Chopra A, Anderson J, Dela Cruz C, Lang L, Neely J, Tang H, Dastani HB, Melero I. Nivolumab in patients with advanced hepatocellular carcinoma (CheckMate 040): an open-label, non-comparative, phase 1/2 dose escalation and expansion trial. Lancet. 2017;389:2492-2502. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 3278] [Cited by in RCA: 3300] [Article Influence: 412.5] [Reference Citation Analysis (1)] |
| 49. | Yau T, Kang YK, Kim TY, El-Khoueiry AB, Santoro A, Sangro B, Melero I, Kudo M, Hou MM, Matilla A, Tovoli F, Knox JJ, Ruth He A, El-Rayes BF, Acosta-Rivera M, Lim HY, Neely J, Shen Y, Wisniewski T, Anderson J, Hsu C. Efficacy and Safety of Nivolumab Plus Ipilimumab in Patients With Advanced Hepatocellular Carcinoma Previously Treated With Sorafenib: The CheckMate 040 Randomized Clinical Trial. JAMA Oncol. 2020;6:e204564. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 908] [Cited by in RCA: 960] [Article Influence: 192.0] [Reference Citation Analysis (0)] |
| 50. | Finn RS, Ryoo BY, Merle P, Kudo M, Bouattour M, Lim HY, Breder V, Edeline J, Chao Y, Ogasawara S, Yau T, Garrido M, Chan SL, Knox J, Daniele B, Ebbinghaus SW, Chen E, Siegel AB, Zhu AX, Cheng AL; KEYNOTE-240 investigators. Pembrolizumab As Second-Line Therapy in Patients With Advanced Hepatocellular Carcinoma in KEYNOTE-240: A Randomized, Double-Blind, Phase III Trial. J Clin Oncol. 2020;38:193-202. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 1365] [Cited by in RCA: 1337] [Article Influence: 267.4] [Reference Citation Analysis (0)] |
| 51. | Scheiner B, Kirstein MM, Hucke F, Finkelmeier F, Schulze K, von Felden J, Koch S, Schwabl P, Hinrichs JB, Waneck F, Waidmann O, Reiberger T, Müller C, Sieghart W, Trauner M, Weinmann A, Wege H, Trojan J, Peck-Radosavljevic M, Vogel A, Pinter M. Programmed cell death protein-1 (PD-1)-targeted immunotherapy in advanced hepatocellular carcinoma: efficacy and safety data from an international multicentre real-world cohort. Aliment Pharmacol Ther. 2019;49:1323-1333. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 67] [Cited by in RCA: 109] [Article Influence: 18.2] [Reference Citation Analysis (0)] |
| 52. | Yau T, Hsu C, Kim TY, Choo SP, Kang YK, Hou MM, Numata K, Yeo W, Chopra A, Ikeda M, Kuromatsu R, Moriguchi M, Chao Y, Zhao H, Anderson J, Cruz CD, Kudo M. Nivolumab in advanced hepatocellular carcinoma: Sorafenib-experienced Asian cohort analysis. J Hepatol. 2019;71:543-552. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 191] [Cited by in RCA: 189] [Article Influence: 31.5] [Reference Citation Analysis (0)] |
| 53. | Finn RS, Ikeda M, Zhu AX, Sung MW, Baron AD, Kudo M, Okusaka T, Kobayashi M, Kumada H, Kaneko S, Pracht M, Mamontov K, Meyer T, Kubota T, Dutcus CE, Saito K, Siegel AB, Dubrovsky L, Mody K, Llovet JM. Phase Ib Study of Lenvatinib Plus Pembrolizumab in Patients With Unresectable Hepatocellular Carcinoma. J Clin Oncol. 2020;38:2960-2970. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 661] [Cited by in RCA: 871] [Article Influence: 174.2] [Reference Citation Analysis (0)] |
| 54. | Castet F, Willoughby CE, Haber PK, Llovet JM. Atezolizumab plus Bevacizumab: A Novel Breakthrough in Hepatocellular Carcinoma. Clin Cancer Res. 2021;27:1827-1829. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 16] [Cited by in RCA: 12] [Article Influence: 3.0] [Reference Citation Analysis (0)] |
| 55. | Llovet JM, Montal R, Sia D, Finn RS. Molecular therapies and precision medicine for hepatocellular carcinoma. Nat Rev Clin Oncol. 2018;15:599-616. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 1458] [Cited by in RCA: 1377] [Article Influence: 196.7] [Reference Citation Analysis (0)] |
| 56. | Galle PR, Finn RS, Qin S, Ikeda M, Zhu AX, Kim TY, Kudo M, Breder V, Merle P, Kaseb A, Li D, Mulla S, Verret W, Xu DZ, Hernandez S, Ding B, Liu J, Huang C, Lim HY, Cheng AL, Ducreux M. Patient-reported outcomes with atezolizumab plus bevacizumab versus sorafenib in patients with unresectable hepatocellular carcinoma (IMbrave150): an open-label, randomised, phase 3 trial. Lancet Oncol. 2021;22:991-1001. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 58] [Cited by in RCA: 231] [Article Influence: 57.8] [Reference Citation Analysis (0)] |
| 57. | Pinter M, Ulbrich G, Sieghart W, Kölblinger C, Reiberger T, Li S, Ferlitsch A, Müller C, Lammer J, Peck-Radosavljevic M. Hepatocellular Carcinoma: A Phase II Randomized Controlled Double-Blind Trial of Transarterial Chemoembolization in Combination with Biweekly Intravenous Administration of Bevacizumab or a Placebo. Radiology. 2015;277:903-912. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 48] [Cited by in RCA: 64] [Article Influence: 6.4] [Reference Citation Analysis (0)] |
| 58. | Siegel AB, Cohen EI, Ocean A, Lehrer D, Goldenberg A, Knox JJ, Chen H, Clark-Garvey S, Weinberg A, Mandeli J, Christos P, Mazumdar M, Popa E, Brown RS Jr, Rafii S, Schwartz JD. Phase II trial evaluating the clinical and biologic effects of bevacizumab in unresectable hepatocellular carcinoma. J Clin Oncol. 2008;26:2992-2998. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 381] [Cited by in RCA: 393] [Article Influence: 23.1] [Reference Citation Analysis (0)] |
| 59. | Chouaib S, Messai Y, Couve S, Escudier B, Hasmim M, Noman MZ. Hypoxia promotes tumor growth in linking angiogenesis to immune escape. Front Immunol. 2012;3:21. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 116] [Cited by in RCA: 138] [Article Influence: 10.6] [Reference Citation Analysis (0)] |
| 60. | Chen DS, Mellman I. Oncology meets immunology: the cancer-immunity cycle. Immunity. 2013;39:1-10. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 3297] [Cited by in RCA: 4749] [Article Influence: 395.8] [Reference Citation Analysis (1)] |
| 61. | Hegde PS, Wallin JJ, Mancao C. Predictive markers of anti-VEGF and emerging role of angiogenesis inhibitors as immunotherapeutics. Semin Cancer Biol. 2018;52:117-124. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 181] [Cited by in RCA: 332] [Article Influence: 41.5] [Reference Citation Analysis (0)] |
| 62. | Ferrara N, Hillan KJ, Gerber HP, Novotny W. Discovery and development of bevacizumab, an anti-VEGF antibody for treating cancer. Nat Rev Drug Discov. 2004;3:391-400. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 1788] [Cited by in RCA: 1890] [Article Influence: 90.0] [Reference Citation Analysis (0)] |
| 63. | Gabrilovich DI, Chen HL, Girgis KR, Cunningham HT, Meny GM, Nadaf S, Kavanaugh D, Carbone DP. Production of vascular endothelial growth factor by human tumors inhibits the functional maturation of dendritic cells. Nat Med. 1996;2:1096-1103. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 1375] [Cited by in RCA: 1402] [Article Influence: 48.3] [Reference Citation Analysis (0)] |
| 64. | Gabrilovich D, Ishida T, Oyama T, Ran S, Kravtsov V, Nadaf S, Carbone DP. Vascular endothelial growth factor inhibits the development of dendritic cells and dramatically affects the differentiation of multiple hematopoietic lineages in vivo. Blood. 1998;92:4150-4166. [PubMed] |
| 65. | Voron T, Marcheteau E, Pernot S, Colussi O, Tartour E, Taieb J, Terme M. Control of the immune response by pro-angiogenic factors. Front Oncol. 2014;4:70. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 237] [Cited by in RCA: 262] [Article Influence: 23.8] [Reference Citation Analysis (0)] |
| 66. | Elovic AE, Ohyama H, Sauty A, McBride J, Tsuji T, Nagai M, Weller PF, Wong DT. IL-4-dependent regulation of TGF-alpha and TGF-beta1 expression in human eosinophils. J Immunol. 1998;160:6121-6127. [PubMed] |
| 67. | Tamura R, Tanaka T, Akasaki Y, Murayama Y, Yoshida K, Sasaki H. The role of vascular endothelial growth factor in the hypoxic and immunosuppressive tumor microenvironment: perspectives for therapeutic implications. Med Oncol. 2019;37:2. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 96] [Cited by in RCA: 155] [Article Influence: 25.8] [Reference Citation Analysis (0)] |
| 68. | Zhao H, Wu L, Yan G, Chen Y, Zhou M, Wu Y, Li Y. Inflammation and tumor progression: signaling pathways and targeted intervention. Signal Transduct Target Ther. 2021;6:263. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 272] [Cited by in RCA: 1389] [Article Influence: 347.3] [Reference Citation Analysis (2)] |
| 69. | Goel S, Duda DG, Xu L, Munn LL, Boucher Y, Fukumura D, Jain RK. Normalization of the vasculature for treatment of cancer and other diseases. Physiol Rev. 2011;91:1071-1121. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 1259] [Cited by in RCA: 1203] [Article Influence: 85.9] [Reference Citation Analysis (0)] |
| 70. | Zhu AX, Abbas AR, de Galarreta MR, Guan Y, Lu S, Koeppen H, Zhang W, Hsu CH, He AR, Ryoo BY, Yau T, Kaseb AO, Burgoyne AM, Dayyani F, Spahn J, Verret W, Finn RS, Toh HC, Lujambio A, Wang Y. Molecular correlates of clinical response and resistance to atezolizumab in combination with bevacizumab in advanced hepatocellular carcinoma. Nat Med. 2022;28:1599-1611. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 3] [Cited by in RCA: 339] [Article Influence: 113.0] [Reference Citation Analysis (0)] |
| 71. | Zheng X, Fang Z, Liu X, Deng S, Zhou P, Wang X, Zhang C, Yin R, Hu H, Chen X, Han Y, Zhao Y, Lin SH, Qin S, Kim BY, Jiang W, Wu Q, Huang Y. Increased vessel perfusion predicts the efficacy of immune checkpoint blockade. J Clin Invest. 2018;128:2104-2115. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 95] [Cited by in RCA: 182] [Article Influence: 26.0] [Reference Citation Analysis (0)] |
| 72. | Hatanaka T, Kakizaki S, Hiraoka A, Tada T, Hirooka M, Kariyama K, Tani J, Atsukawa M, Takaguchi K, Itobayashi E, Fukunishi S, Tsuji K, Ishikawa T, Tajiri K, Ochi H, Yasuda S, Toyoda H, Ogawa C, Nishimura T, Shimada N, Kawata K, Kosaka H, Tanaka T, Ohama H, Nouso K, Morishita A, Tsutsui A, Nagano T, Itokawa N, Okubo T, Arai T, Imai M, Naganuma A, Koizumi Y, Nakamura S, Joko K, Kaibori M, Iijima H, Hiasa Y, Kumada T; Real-life Practice Experts for HCC (RELPEC) Study Group, and HCC 48 Group (hepatocellular carcinoma experts from 48 clinics in Japan). Prognostic impact of C-reactive protein and alpha-fetoprotein in immunotherapy score in hepatocellular carcinoma patients treated with atezolizumab plus bevacizumab: a multicenter retrospective study. Hepatol Int. 2022;16:1150-1160. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 47] [Cited by in RCA: 45] [Article Influence: 15.0] [Reference Citation Analysis (0)] |
| 73. | Ang C, Klempner SJ, Ali SM, Madison R, Ross JS, Severson EA, Fabrizio D, Goodman A, Kurzrock R, Suh J, Millis SZ. Prevalence of established and emerging biomarkers of immune checkpoint inhibitor response in advanced hepatocellular carcinoma. Oncotarget. 2019;10:4018-4025. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 64] [Cited by in RCA: 131] [Article Influence: 21.8] [Reference Citation Analysis (0)] |