Published online Aug 14, 2022. doi: 10.3748/wjg.v28.i30.4182
Peer-review started: March 12, 2022
First decision: April 10, 2022
Revised: April 24, 2022
Accepted: July 16, 2022
Article in press: July 16, 2022
Published online: August 14, 2022
Processing time: 151 Days and 0.5 Hours
Prospective studies of the long-term outcomes of patients with hepatitis C virus (HCV) infection after treatment with interferon-based therapy (IBT) or direct-acting antivirals (DAA) are limited in many Asian countries.
To elucidate the incidences of hepatocellular carcinoma (HCC) and death/transplantation based on treatment with IBT or DAA, to compare the outcomes of the sustained virologic response (SVR) to IBT and DAA, and to investigate outcome-determining factors after SVR.
This cohort included 2054 viremic patients (mean age, 57 years; 46.5% male; 27.4% with cirrhosis) prospectively enrolled at seven hospitals between 2007 and 2019. They were classified as the untreated group (n = 619), IBT group (n = 578), and DAA group (n = 857). Outcomes included the incidences of HCC and death/transplantation. The incidences of the outcomes for each group according to treatment were calculated using an exact method based on the Poisson distribution. A multivariate Cox regression analysis was performed to determine the factors associated with HCC or death/transplantation, followed by propensity score matching to confirm the results.
During a median of 4.1 years of follow-up, HCC and death/transplantation occurred in 113 and 206 patients, respectively, in the entire cohort. Compared with the untreated group, the incidences of HCC and death/transplantation were significantly lower in the IBT group [adjusted hazard ratio (aHR) 0.47, 95%CI: 0.28-0.80 and aHR 0.28, 95%CI: 0.18-0.43, respectively] and the DAA group (aHR 0.58, 95%CI: 0.35-0.96, and aHR 0.19, 95%CI: 0.20-0.68, respectively). Among 1268 patients who attained SVR with IBT (n = 451) or DAA (n = 816), the multivariable-adjusted analysis showed no differences in the risks of HCC (HR 2.03; 95%CI: 0.76-5.43) and death/transplantation (HR 1.38; 95%CI: 0.55-3.49) between the two groups. This was confirmed by a propensity score-matching analysis. Independent factors for HCC after SVR were age, genotype 1, and the presence of cirrhosis.
Treatment and achieving SVR with either IBT or DAA significantly reduced the incidences of HCC and mortality in the Asian patients with HCV infection. The risks of HCC and mortality were not significantly different regardless of whether SVR was induced by IBT or DAA.
Core Tip: Treatment and sustained virologic response (SVR) with either interferon-based treatment (IBT) or direct-acting antiviral (DAA) significantly reduced the incidences of hepatocellular carcinoma and mortality in our Asian prospective cohort. The risks of HCC and all-cause of mortality were not significantly different regardless of whether SVR was induced by IBT or DAA. After achieving SVR, age, the presence of cirrhosis, and genotype 1 hepatitis C virus infection were indicators of worse clinical outcomes.
- Citation: Choi GH, Jang ES, Kim YS, Lee YJ, Kim IH, Cho SB, Lee HC, Jang JW, Ki M, Choi HY, Baik D, Jeong SH. Hepatocellular carcinoma, decompensation, and mortality based on hepatitis C treatment: A prospective cohort study. World J Gastroenterol 2022; 28(30): 4182-4200
- URL: https://www.wjgnet.com/1007-9327/full/v28/i30/4182.htm
- DOI: https://dx.doi.org/10.3748/wjg.v28.i30.4182
Hepatitis C virus (HCV) is a major cause of liver cirrhosis, hepatocellular carcinoma (HCC), and liver-related and overall mortality. In 2019, the World Health Organization estimated that 58 million people worldwide had chronic HCV infection[1]. Currently, this substantial public health burden can be reduced by active screening and treatment with highly tolerable direct-acting antivirals (DAA), which result in a cure rate of more than 95% in terms of the sustained virologic response (SVR)[2-4].
From the identification of HCV to the introduction of DAA therapy, interferon (IFN)-based therapy (IBT) was the only option for HCV treatment, with an approximate SVR rate of 50%[5]. Although IBT-induced SVR includes a wide variety of adverse effects and narrow indications, it significantly reduces the incidence of HCC and long-term mortality[6,7]. Furthermore, DAA-induced SVR has resulted in reductions in hepatic fibrosis[8,9], portal hypertension[10], hepatic decompensation[11], HCC incidence[11,12], and liver-related and overall mortality[11,13]; however, the follow-up durations were relatively short.
To reach the HCV elimination goal of 2030, it is imperative to understand the regional epidemiology and outcomes of HCV. However, prospective studies of the long-term outcomes after treatment with IBT or DAA are limited in many Asian countries, where hepatitis B virus (HBV) is the major cause of liver-related complications. Additionally, comparative studies of the outcomes of SVR induced by IBT and DAA are scarce.
We established a prospective, nationwide, multicenter HCV cohort (the Korea HCV cohort study) funded by the Korean National Institute of Health in 2007. Using these data, we aimed to elucidate the clinical outcomes, including HCC, hepatic decompensation, and all-cause death, among Korean patients with chronic HCV infection. We compared the outcomes based on the antiviral treatments (untreated, IBT, and DAA) and analyzed patients after achieving SVR (IBT-SVR and DAA-SVR groups). Additionally, we investigated outcome-determining factors after SVR.
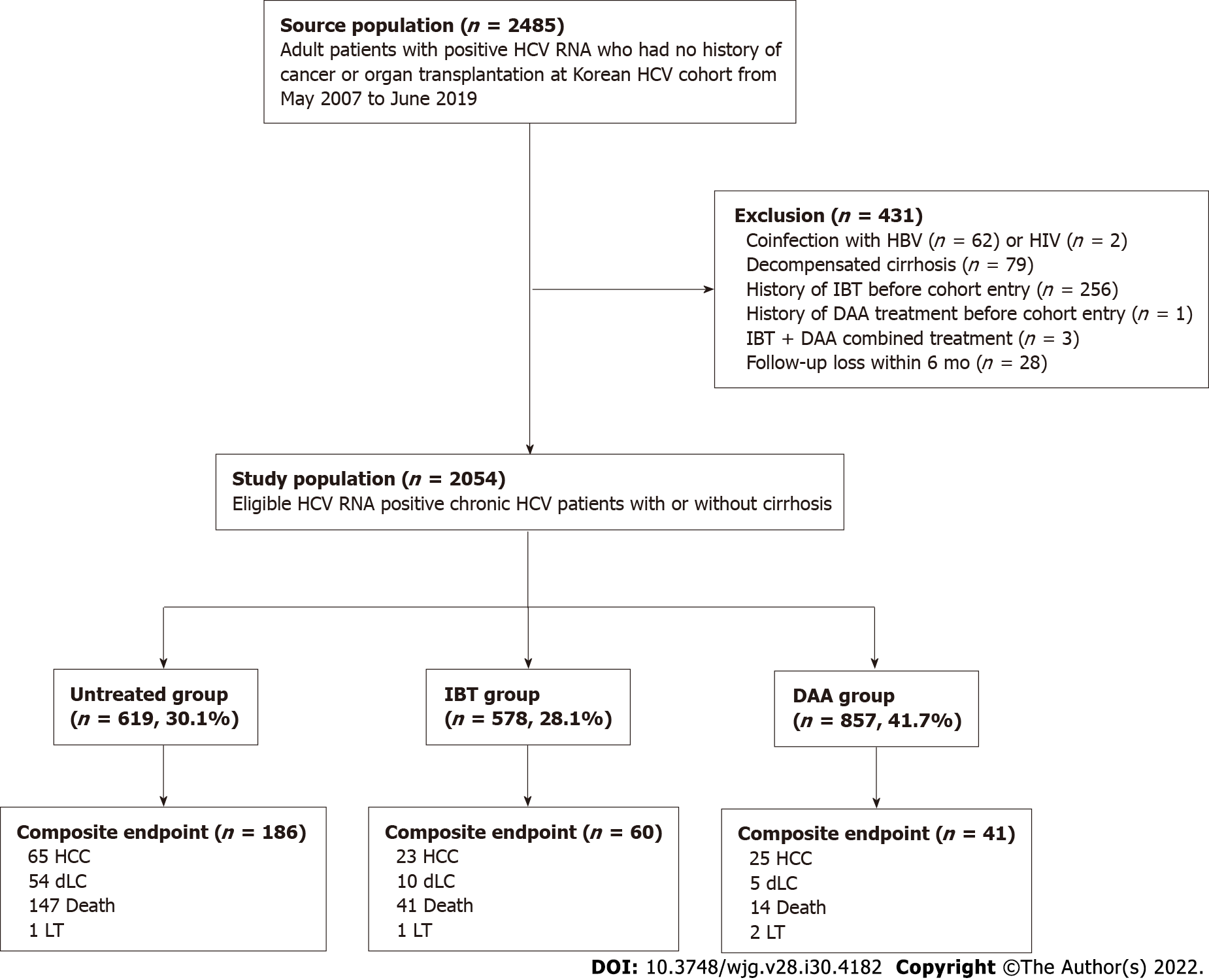
The Korea HCV cohort is a prospective cohort of 2485 adult patients with HCV RNA positivity at seven tertiary centers nationwide enrolled from January 2007 to June 2019 in South Korea. Patients who met any of the following criteria were excluded: Positive serology for HBV surface antigen (n = 62) or HIV (n = 2); decompensated cirrhosis at enrollment (n = 79); previous antiviral treatment before cohort entry (n = 260); and less than 6 mo of follow-up (n = 28). Additionally, patients who had HCC before or at the time of cohort entry were excluded.
Therefore, 2054 viremic patients with or without compensated cirrhosis were analyzed as the entire cohort, which was further classified into three groups based on their treatment: untreated group (n = 619; 30.2%); IBT group (n = 578; 28.1%); and DAA group (n = 857; 41.7%). Patients in the untreated group did not receive IBT or DAA treatment during the entire follow-up period. Subject selection, classification, and overall outcomes are summarized in Figure 1.
In the entire cohort, 1267 patients achieved SVR (61.7%) with IBT (n = 451) or DAA (n = 816); these patients comprised the SVR cohort. Diagnostic criteria for liver cirrhosis were based on histology or at least one clinical sign of portal hypertension, such as cirrhotic features on radiological images, platelet count less than 100,000/mm3, and documented gastroesophageal varices without hemorrhage. The study protocol was approved by the Institutional Review Board of seven hospitals, and each enrolled patient provided written informed consent.
At the time of enrollment, trained research coordinators at the seven hospitals interviewed the patients using a standardized questionnaire that included demographic and socioeconomic factors (age, sex, body mass index, education level, and occupation), health behaviors (smoking and alcohol consumption), comorbidities (extrahepatic cancers, thyroid disease, psychiatric disease, diabetes, kidney disease, cerebrovascular disease, and cardiovascular disease), and lifetime exposure to risk factors for HCV infection.
Laboratory data at baseline and follow-up visits were collected from the medical records, including the anti-HCV antibody, serum HCV RNA level, HCV genotype, HBV surface antigen, HBV core antibody immunoglobulin G (anti-HBc IgG), white blood cell count, hemoglobin, platelet count, aspartate aminotransferasel (AST), alanine aminotransferase, alkaline phosphatase, total bilirubin, albumin, creatinine, prothrombin time (PT) and alpha-fetoprotein (AFP). The following three non-invasive serum fibrosis assessment scores were calculated at the index date: The fibrosis-4 (FIB-4) index[14], the AST-to-platelet ratio index (APRI) score[15], and the albumin-bilirubin (ALBI) score[16].
The results of imaging studies, such as abdominal ultrasonography or computed tomography, liver pathology, and transient elastography (FibroScan®, Echosens, Paris, France), were collected when available. Detailed information about antiviral treatment, including therapeutic regimens, duration, and achievement of SVR, was collected from the patients’ medical records. These data were entered into the established electronic case report form on the authorized website of the Korean Centers for Disease Control Korea HCV cohort study (http://is.cdc.go.kr/) by the research coordinators. All input data were quality-controlled by independent statistical researchers (Baik D, Choi HY, and Ki M) at least four times per year.
All patients underwent regular clinical assessments every 3 to 12 mo, and HCV treatment was recommended by their attending physicians according to the treatment guidelines for HCV infection unless there were contraindications or patient's refusal. If the patients were treated, then SVR was evaluated, and regular follow-up visits every 6 to 12 mo after SVR were encouraged. HCC surveillance using abdominal ultrasonography and serum AFP tests every 6 to 12 mo were recommended according to the pretreatment fibrosis stage. If the patients did not adhere to the regular follow-up schedule, then research coordinators called the patients or their families to encourage them to attend the clinic and to check their survival status, disease progression to hepatic decompensation, or development of HCC. The end of follow-up was defined as the date of death/liver transplantation (LT), or the last follow-up date (June 30, 2020).
Outcomes included the incidences of HCC, hepatic decompensation, and death/LT. HCC was diagnosed according to pathology or typical imaging criteria observed on dynamic computed tomography and/or magnetic resonance imaging in accordance with the Korean Liver Cancer Study Group guidelines (similar to major international guidelines)[17]. Decompensated liver cirrhosis was defined as the presence of ascites, jaundice, variceal bleeding, encephalopathy, or a combination of these[18]. All-cause mortality or death/LT was directly documented or indirectly indicated as disqualification from the National Health Insurance status provided in the electronic medical records. In Korea, enrollment in the National Health Insurance is compulsory for all individuals; therefore, disqualification from the National Health Insurance indicates death or emigration in most cases[19]. To verify the survival status, physician-confirmed death certificate data, including the date and cause of death, were obtained from the Statistics Korea mortality database, which was established in 1981.
To calculate the outcomes of the entire cohort, the index date of the entire cohort was defined as the date of cohort entry when HCV RNA positivity was confirmed by the referred clinics. To compare the outcomes of patients with SVR induced by IBT and patients with SVR induced by DAA, the index date for the SVR cohort was defined as the initiation day of the antiviral treatment. SVR was evaluated using an intention-to-treat analysis; therefore, patients who received at least one dose of IBT or DAA were included.
Baseline characteristics of the patients were compared using the chi-square test for categorical variables, and a one-way analysis of variance (ANOVA) or t-test was used for continuous variables. For multiple comparisons, a one-way ANOVA was used followed by a Bonferroni correction. The total follow-up (in person-years) of each group was calculated by multiplying the cohort population size by the average follow-up in years. Survival time was calculated as the time from cohort entry (the entire cohort) or the start of the first treatment (SVR cohort) until the date of death/LT or the last available follow-up date. A few patients who received a second course of DAA treatment because of failure of the first DAA treatment were censored at the time of retreatment.
The incidences and 95% confidence intervals (CI) of the outcomes for each group based on treatment were calculated using an exact method based on the Poisson distribution. Cumulative incidence curves for outcome development were estimated using the pseudo-Kaplan–Meier method with a clock reset procedure for patients treated with IBT or DAA during follow-up and compared using the log-rank test. Therefore, patients who had received IBT and subsequent DAA treatment were considered as the DAA group, but the period between IBT and DAA treatment was included in the IBT group with no event.
A time-varying Cox regression model was used to determine the factors associated with outcomes, and the adjusted hazard ratio was estimated for the entire cohort and the SVR cohort. In the models, baseline variables (sex, body mass index, alcohol, smoking, HCV genotype) were adjusted, and age, antiviral treatment, laboratory data, achievement of SVR, and presence of cirrhosis were considered time-dependent variables. A multivariate Cox regression analysis was performed to determine the factors associated with HCC or death/LT, and the adjusted hazard ratio was estimated for the SVR cohort at the time of IBT or DAA initiation. Covariates with P < 0.05 in the univariate Cox regression model were used as covariates for the multivariate Cox regression analyses. To confirm the multivariate analysis results for the SVR cohort, significant differences in characteristics at the time of initiation of each treatment were adjusted by propensity score (PS) matching for all possible variables, including the time from cohort entry to treatment. We used nearest-neighbor matching with a caliper size of 0.1 and matched the patients using a 1:1 ratio. The covariate balance was considered to be achieved if the absolute standardized difference between the two groups was ≤ 0.1.
All P-values were two-sided, and P < 0.05 was considered significant. SPSS (version 21; IBC Corp., Armonk, NY, United States) and R (version 4.0.4; http://cran.r-project.org/) software were used for statistical analyses. The R package of MatchIt was used for matching analyses.
The demographic, clinical, and laboratory characteristics of the entire cohort (n = 2054), untreated group (n = 619; 30.1%), IBT group (n = 578; 28.1%), and DAA group (n = 857; 41.7%) at the index date are provided in Table 1. The mean age of the patients was 57 years; 46.5% were men and 27.4% had compensated cirrhosis.
| Variable, n (%) | Untreated group (n = 619) | IBT group (n = 578) | DAA group (n = 857) | P value (3 Gr.) | P value (U vs I) | P value (U vs D) | P value (I vs D) |
| Age (yr) | 60.4 ± 13.4 | 51.4 ± 10.9 | 59.1±11.4 | < 0.001 | < 0.001 | 0.086 | < 0.001 |
| Male sex | 280 (45.2) | 277 (47.9) | 412 (48.1) | 0.511 | 0.351 | 0.281 | 0.955 |
| HCV RNA, log10 IU/mL | 5.89 (5.08-6.45) | 5.83 (4.87-6.49) | 6.04 (5.27-6.53) | < 0.001 | 0.979 | 0.017 | < 0.001 |
| HCV genotype | < 0.001 | < 0.001 | < 0.001 | < 0.001 | |||
| 1 | 256 (41.4) | 242 (41.9) | 491 (57.3) | ||||
| 2 | 299 (48.3) | 323 (55.9) | 355 (41.4) | ||||
| Others/missing | 64 (10.3) | 13 (2.2) | 11 (1.3) | ||||
| Diagnosis status | < 0.001 | < 0.001 | 0.002 | 0.058 | |||
| Chronic hepatitis | 417 (67.4) | 456 (78.9) | 639 (74.6) | ||||
| Compensated cirrhosis | 202 (32.6) | 122 (21.1) | 218 (25.4) | ||||
| FIB-4 index | < 0.001 | < 0.001 | 0.142 | < 0.001 | |||
| ≤ 1.45 | 112 (18.9) | 191 (33.3) | 164 (19.9) | ||||
| 1.45-3.25 | 218 (36.8) | 200 (34.8) | 337 (40.9) | ||||
| ≥ 3.25 | 263 (44.3) | 183 (31.9) | 323 (39.2) | ||||
| APRI score | 0.15 | 0.09 | 0.094 | 0.853 | |||
| ≤ 2.0 | 480 (80.3) | 485 (84.1) | 708 (83.7) | ||||
| > 2.0 | 118 (19.7) | 92 (15.9) | 138 (16.3) | ||||
| ALBI score | 0.004 | 0.04 | 0.001 | 0.313 | |||
| ≤ -2.60 (Grade 1) | 446 (73.0) | 450 (78.1) | 674 (80.3) | ||||
| > -2.60 (Grade 2 or 3) | 165 (27.0) | 126 (21.9) | 165 (19.7) | ||||
| Ever smoker | 264 (42.9) | 283 (49.5) | 380 (44.5) | 0.058 | 0.022 | 0.519 | 0.068 |
| Alcohol intake | < 0.001 | < 0.001 | < 0.001 | < 0.001 | |||
| None | 326 (53.0) | 226 (39.4) | 419 (48.9) | ||||
| Social | 227 (36.9) | 308 (53.7) | 229 (26.7) | ||||
| Significant1 | 61 (9.9) | 40 (7.0) | 209 (24.4) | ||||
| Fatty liver disease on imaging study, n = 1389 | 71 (19.3), n = 367 | 86 (21.1), n = 408 | 140 (22.8), n = 614 | 0.435 | 0.549 | 0.202 | 0.516 |
| Anti-HBcIgG (+), n = 1442 | 47 (11.0), n = 429 | 33 (8.4), n = 391 | 64 (10.3), n = 622 | 0.46 | 0.225 | 0.73 | 0.33 |
| BMI, kg/m2 | 23.4 ± 3.2 | 23.8 ± 3.0 | 23.8 ± 3.1 | 0.02 | 0.143 | 0.02 | 1 |
| Diabetes mellitus | 120 (19.4) | 77 (13.3) | 128 (14.9) | 0.01 | 0.005 | 0.024 | 0.391 |
| Hypertension | 145 (23.4) | 107 (18.5) | 243 (28.4) | < 0.001 | 0.037 | 0.034 | < 0.001 |
| Cardiovascular disease | 16 (2.6) | 3 (0.5) | 25 (2.9) | 0.006 | 0.004 | 0.701 | 0.001 |
| Cerebrovascular disease | 17 (2.7) | 4 (0.7) | 20 (2.3) | 0.026 | 0.007 | 0.617 | 0.017 |
| Laboratory study | |||||||
| WBC, × 103/mm3 | 5.09 (4.29-6.50) | 5.05 (3.91-6.16) | 5.40 (4.40-6.61) | < 0.001 | 0.145 | 0.164 | < 0.001 |
| Hemoglobin, g/dL | 13.5 (12.3-14.5) | 13.7 (12.7-14.8) | 13.8 (12.7-14.8) | < 0.001 | 0.004 | < 0.001 | 1 |
| Platelet, × 103/mm3 | 162 (118-212) | 174 (130-220) | 172 (129-215) | 0.229 | 0.236 | 0.317 | 1 |
| Albumin, g/dL | 4.2 (3.9-4.4) | 4.2 (4.0-4.5) | 4.2 (4.0-4.4) | < 0.001 | < 0.001 | 0.001 | 1 |
| Total bilirubin, mg/dL | 0.8 (0.5-1.0) | 0.7 (0.6-1.0) | 0.7 (0.6-1.0) | 0.784 | 1 | 1 | 1 |
| ALP, IU/L | 87 (68-136) | 79 (63-112) | 121 (78-250) | < 0.001 | 0.001 | < 0.001 | < 0.001 |
| AST, IU/L | 50 (30-85) | 50 (31-83) | 46 (30-76) | 0.026 | 1 | 0.139 | 0.038 |
| ALT, IU/L | 42 (23-80) | 52 (29-100) | 36 (22-70) | < 0.001 | 0.013 | 0.176 | < 0.001 |
| Creatinine, mg/dL | 0.9 (0.7-1.0) | 0.8 (0.7-1.0) | 0.8 (0.7-1.0) | 0.857 | 1 | 1 | 1 |
| PT, INR | 1.06 (1.01-1.12) | 1.03 (0.98-1.08) | 1.03 (0.97-1.09) | < 0.001 | < 0.001 | 0.007 | 0.119 |
| AFP, ng/dL | 4.3 (2.5-9.7) | 3.5 (2.3-7.1) | 3.8 (2.4-6.6) | 0.868 | 1 | 1 | 1 |
| Cohort entry time | < 0.001 | < 0.001 | < 0.001 | < 0.001 | |||
| January 2007–June 2015 | 488 (78.8) | 561 (97.1) | 49 (5.7) | ||||
| July 2015–June 2019 | 131 (21.2) | 17 (2.9) | 808 (94.3) | ||||
| SVR rate | - | 67.5 (451/668) | 95.3 (817/857) | - | - | < 0.001 | - |
| Follow-up duration (yr) | 5.6 (3.4-8.2) | 7.9 (6.1-9.7) | 3.5 (2.0-5.5) | < 0.001 | < 0.001 | < 0.001 | < 0.001 |
Compared with the untreated and DAA groups, the IBT group was significantly younger, had a higher proportion of genotype 2, lower rates of alcohol consumption, cirrhosis and high FIB-4 index values (≥ 3.25), and fewer comorbidities.
Among the DAA group, 14.0% had been treated with an IFN-based regimen after cohort enrollment, and 86.0% were treatment-naive. DAA treatments administered were sofosbuvir plus ribavirin (32.1%), daclatasvir plus asunaprevir (26.3%), elbasvir plus grazoprevir (13.7%), glecaprevir plus pibrentasvir (13.7%), ledipasvir plus sofosbuvir (9.1%), and ombitasvir plus paritaprevir plus ritonavir plus dasabuvir (3.9%).
The median time periods between cohort entry (index date) and the end of follow-up were 5.6 years [interquartile range (IQR), 3.4-8.2] for the untreated group, 7.9 years (IQR, 6.1-9.7) for the IBT group, and 3.5 years (IQR, 2.0-5.5) for the DAA group (Table 1). During this period, 113 patients developed HCC, 69 patients experienced hepatic decompensation, 202 patients died (119 Liver related and 83 non-liver related), and four patients underwent LT.
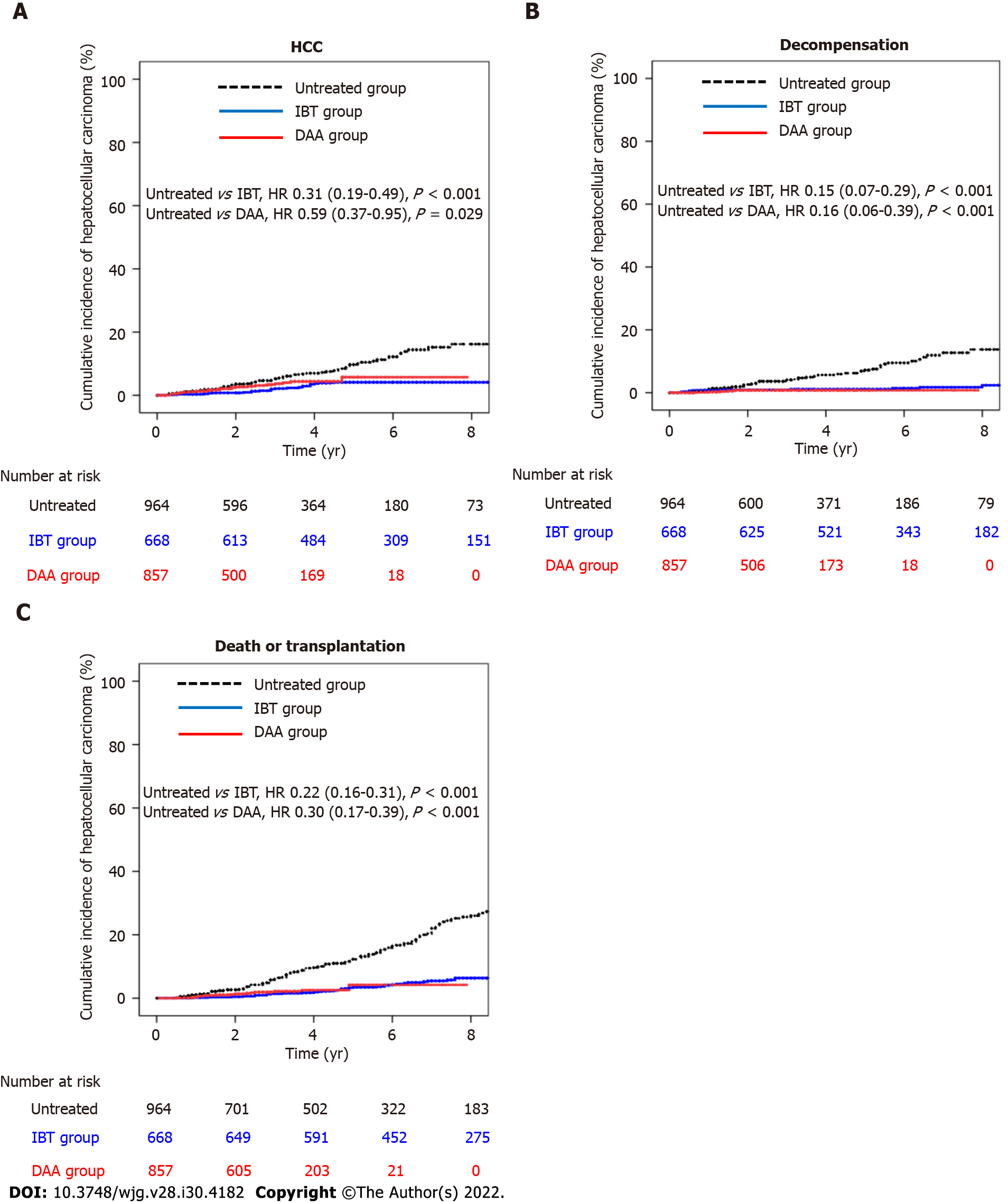
As shown in Table 2, the estimated HCC incidence rates per 100 person-years for the untreated group, IBT group, and DAA group were 1.98 (95%CI: 1.56-2.52), 0.59 (95%CI: 0.39-0.89), and 1.16 (95%CI: 0.78-1.71), respectively (Table 2). The incidence rates of hepatic decompensation per 100 person-years for the untreated group, IBT group, and DAA group were 1.62 (95%CI: 1.24-2.11), 0.25 (95%CI: 0.14-0.47), and 0.23 (95%CI: 0.10-0.55), respectively. The incidence rates of death/LT per 100 person-years for the untreated group, IBT group, and DAA group were 3.33 (95%CI: 2.85-3.91), 0.87 (95%CI: 0.64-1.18), and 0.65 (95%CI: 0.40-1.06), respectively. The incidence rates of all three outcomes were significantly lower in the IBT and DAA groups compared with the untreated group. However, the incidence of HCC in the non-cirrhotic group was not significantly different between the untreated and DAA groups, while the incidences of decompensation and death/LT were significantly lower in the DAA group compared with the untreated group. The cumulative outcome incidences for the untreated, IBT, and DAA groups are shown in Figure 2, and those for the non-cirrhotic and cirrhotic subgroups are shown in Supplementary Figures 1 and 2, respectively.
| Group | PY | No. of events | No./100 PY (95%CI) | HR (95%CI) | P value | |
| Entire cohort | Hepatocellular carcinoma | |||||
| Untreated group | 3285.4 | 65 | 1.98 (1.56-2.52) | Reference | - | |
| IBT group | 3888.9 | 23 | 0.59 (0.39-0.89) | 0.31 (0.19-0.49) | < 0.001 | |
| DAA group | 2158.0 | 25 | 1.16 (0.78-1.71) | 0.59 (0.37-0.95) | 0.029 | |
| Decompensation | ||||||
| Untreated group | 3333.6 | 54 | 1.62 (1.24-2.11) | Reference | - | |
| IBT group | 3950.1 | 10 | 0.25 (0.14-0.47) | 0.15 (0.07-0.29) | < 0.001 | |
| DAA group | 2178.7 | 5 | 0.23 (0.10-0.55) | 0.16 (0.06-0.39) | < 0.001 | |
| Death or transplantation | ||||||
| Untreated group | 4438.3 | 148 | 3.33 (2.85-3.91) | Reference | - | |
| IBT group | 4850.1 | 42 | 0.87 (0.64-1.18) | 0.22 (0.16-0.31) | < 0.001 | |
| DAA group | 2467.2 | 16 | 0.65 (0.40-1.06) | 0.30 (0.17-0.51) | < 0.001 | |
| No cirrhosis | Hepatocellular carcinoma | |||||
| Untreated group | 2364.6 | 15 | 0.63 (0.38-1.05) | Reference | - | |
| IBT group | 2971.8 | 6 | 0.20 (0.09-0.45) | 0.30 (0.11-0.77) | 0.012 | |
| DAA group | 1569.4 | 8 | 0.51 (0.26-1.02) | 0.90 (0.37-2.18) | 0.812 | |
| Decompensation | ||||||
| Untreated group | 2369.9 | 6 | 0.25 (0.11-0.56) | Reference | - | |
| IBT group | 2992.3 | 1 | 0.03 (0.00-0.24) | 0.09 (0.01-0.74) | 0.026 | |
| DAA group | 1573.4 | 0 | 0.00 | 0.00 | N/A | |
| Death or transplantation | ||||||
| Untreated group | 3176.3 | 63 | 1.98 (4.15-7.12) | Reference | - | |
| IBT group | 3712.3 | 22 | 0.59 (0.39-0.90) | 0.26 (0.16-0.42) | < 0.001 | |
| DAA group | 1784.6 | 7 | 0.39 (0.19-0.82) | 0.33 (0.15-0.75) | 0.008 | |
| Cirrhosis | Hepatocellular carcinoma | |||||
| Untreated group | 919.9 | 50 | 5.44 (4.15-7.12) | Reference | - | |
| IBT group | 917.1 | 17 | 1.85 (1.16-2.97) | 0.35 (0.20-0.61) | < 0.001 | |
| DAA group | 588.6 | 17 | 2.89 (1.81-4.61) | 0.53 (0.30-0.92) | 0.025 | |
| Decompensation | ||||||
| Untreated group | 962.8 | 48 | 4.99 (3.78-6.57) | Reference | - | |
| IBT group | 957.7 | 9 | 0.84 (0.49-1.80) | 0.18 (0.09-0.37) | < 0.001 | |
| DAA group | 606.3 | 5 | 0.82 (0.34-1.97) | 0.17 (0.07-0.44) | < 0.001 | |
| Death or transplantation | ||||||
| Untreated group | 1262.1 | 85 | 6.73 (5.48-8.27) | Reference | - | |
| IBT group | 1137.8 | 20 | 1.76 (1.14-2.71) | 0.21 (0.13-0.35) | < 0.001 | |
| DAA group | 682.6 | 9 | 1.32 (0.69-2.52) | 0.28 (0.14-0.56) | < 0.001 | |
A multivariable time-varying Cox regression analysis with the untreated group as a reference showed that IBT [hazard ratio (HR), 0.47; 95%CI: 0.28-0.80; P = 0.005] and DAA groups (HR, 0.58; 95%CI: 0.35-0.96; P = 0.035) were independently associated with a significantly lower risk of HCC (Table 3, model 1). Other independent HCC risk factors were older age (HR, 1.06; 95%CI: 1.03-1.08; P < 0.001), male sex (HR, 2.50; 95%CI: 1.37-4.55; P = 0.003), genotype 1 (HR, 2.25; 95%CI: 1.45-3.48; P < 0.001), the presence of cirrhosis (HR, 3.81; 95%CI: 2.38-6.10; P < 0.001), significant alcohol consumption (HR, 2.20; 95%CI: 1.14-4.24; P = 0.027), prolonged PT (HR, 2.66; 95%CI: 1.13-6.24; P = 0.025), and higher AFP level (HR, 2.12; 95%CI: 1.48-3.05; P < 0.001) (Table 3, model 1).
| Variable | Hepatocellular carcinoma | Decompensation | Death/transplantation | |||
| aHR (95%CI) | P value | aHR (95%CI) | P value | aHR (95%CI) | P value | |
| Model 1 | ||||||
| Untreated group | Reference | - | Reference | - | Reference | - |
| IBT group | 0.47 (0.28-0.80) | 0.005 | 0.16 (0.08-0.33) | < 0.001 | 0.28 (0.18-0.43) | < 0.001 |
| DAA group | 0.58 (0.35-0.96) | 0.035 | 0.12 (0.03-0.33) | < 0.001 | 0.19 (0.10-0.35) | < 0.001 |
| Age, yr | 1.06 (1.03-1.08) | < 0.001 | 1.00 (0.98-1.03) | 0.805 | 1.05 (1.03-1.06) | < 0.001 |
| Male sex | 2.50 (1.37-4.55) | 0.003 | - | - | 1.70 (1.24-2.33) | < 0.001 |
| HCV genotype | - | - | - | - | ||
| 2 | Reference | - | ||||
| 1 | 2.25 (1.45-3.48) | < 0.001 | ||||
| Others/unknown | 1.72 (0.66-4.48) | 0.266 | ||||
| Cirrhosis | 3.81 (2.38-6.10) | < 0.001 | 9.26 (4.03-21.03) | < 0.001 | 1.88 (1.34-2.64) | < 0.001 |
| Ever smoker | 1.47 (0.82-2.66) | 0.192 | - | - | - | - |
| Alcohol consumption | - | - | - | - | ||
| None | Reference | - | ||||
| Social | 1.20 (0.73-1.98) | 0.473 | ||||
| Significant | 2.20 (1.14-4.24) | 0.027 | ||||
| BMI, kg/m2 | - | - | - | - | 0.94 (0.89-0.99) | 0.019 |
| Diabetes mellitus | 0.96 (0.59-1.56) | 0.872 | - | - | 1.12 (0.77-1.62) | 0.563 |
| Albumin, g/dL | 0.71 (0.44-1.15) | 0.162 | 0.35 (0.20-0.63) | < 0.001 | 0.36 (0.25-0.51) | < 0.001 |
| Total bilirubin, mg/dL | 0.95 (0.80-1.12) | 0.568 | 1.03 (0.98-1.08) | 0.264 | - | - |
| PT, INR | 2.66 (1.13-6.24) | 0.025 | 3.32 (1.08-10.19) | 0.036 | 2.34 (1.06-5.16) | 0.034 |
| AFP, log10 ng/dL | 2.12 (1.48-3.05) | < 0.001 | 1.54 (0.95-2.51) | 0.08 | 1.29 (0.94-1.77) | 0.112 |
| Model 2 | ||||||
| Untreated or no SVR | Reference | - | Reference | - | Reference | - |
| SVR | 0.41 (0.26-0.65) | < 0.001 | 0.10 (0.04-0.29) | < 0.001 | 0.26 (0.17-0.39) | < 0.001 |
| Age, yr | 1.06 (1.04- 1.08) | < 0.001 | 1.02 (0.99-1.04) | 0.152 | 1.06 (1.04-1.07) | < 0.001 |
| Male sex | 2.50 (1.38-4.54) | 0.003 | - | - | 1.68 (1.23-2.30) | 0.001 |
| HCV genotype | - | - | - | - | ||
| 2 | Reference | - | ||||
| 1 | 2.25 (1.45-3.48) | < 0.001 | ||||
| Others/unknown | 1.57 (0.60-4.09) | 0.359 | ||||
| Cirrhosis | 3.75 (2.35-6.01) | < 0.001 | 7.54 (3.32-17.15) | < 0.001 | 1.72 (1.23-2.41) | 0.002 |
| Ever smoker | 1.53 (0.85-2.75) | 0.156 | - | - | - | - |
| Alcohol consumption | - | - | - | - | ||
| None | Reference | - | ||||
| Social | 1.24 (0.75-2.04) | 0.397 | ||||
| Significant | 2.69 (1.40-5.16) | 0.003 | ||||
| BMI, kg/m2 | - | - | - | - | 0.94 (0.89-0.99) | 0.024 |
| Diabetes mellitus | 0.99 (0.61-1.61) | 0.959 | - | - | 1.10 (0.76-1.60) | 0.612 |
| Albumin, g/dL | 0.76 (0.47-1.20) | 0.238 | 0.42 (0.24-0.71) | 0.001 | 0.42 (0.30-0.60) | < 0.001 |
| Total bilirubin, mg/dL | 0.96 (0.81-1.13) | 0.624 | 1.03 (0.99-1.08) | 0.156 | - | - |
| PT, INR | 2.53 (1.02-6.28) | 0.044 | 4.42 (1.48-13.20) | 0.008 | 2.69 (1.22-5.92) | 0.014 |
| AFP, log10 ng/dL | 2.10 (1.46-3.02) | < 0.001 | 1.48 (0.90-2.44) | 0.12 | 1.17 (0.86-1.60) | 0.32 |
The death/LT incidence decreased independently after antiviral treatment with IBT (HR, 0.28; 95%CI: 0.19-0.43; P < 0.001) or DAA (HR, 0.19; 95%CI: 0.10-0.35; P < 0.001) (Table 3, model 1). In contrast, older age (HR, 1.05; 95%CI: 1.03-1.06; P < 0.001), male sex (HR, 1.70; 95%CI: 1.24-2.33; P < 0.001), the presence of cirrhosis (HR, 1.88; 95%CI: 1.34-2.64; P < 0.001), lower body mass index (HR, 0.94; 95%CI: 0.89-0.99; P = 0.019), lower albumin level (HR, 0.35; 95%CI: 0.25-0.51; P < 0.001), and prolonged PT (HR, 2.34; 95%CI: 1.06-5.16; P = 0.034) independently increased mortality/LT rates. Antiviral treatment, cirrhosis, albumin level, and PT were independently associated with the risk of decompensation (Table 3, model 1).
We established another multivariable model (model 2) by replacing the achievement of SVR instead of IBT or DAA treatment. A multivariate time-varying Cox regression analysis of the combined outcomes of the untreated and no SVR groups as a reference showed that SVR induced by either IBT or DAA significantly decreased the risk of HCC (HR, 0.41; 95%CI: 0.26-0.65; P < 0.001), decompensation (HR, 0.10; 95%CI: 0.04-0.29; P < 0.001), and death/LT (HR, 0.26; 95%CI: 0.17-0.39; P < 0.001) (Table 3, model 2).
The SVR cohort comprised 1,268 chronic HCV patients who achieved SVR with IBT or DAA (IBT-SVR group, n = 451; DAA-SVR group, n = 817). The SVR rates of the IBT and DAA groups were 67.5% and 95.3%, respectively. The baseline characteristics at the time of initiation of treatment of the IBT and DAA groups according to SVR are shown in Supplementary Table 1. The incidence rates of HCC, decompensation, and death/LT were significantly lower in the IBT-SVR group than in the IBT-no SVR group (Supplementary Table 2). In contrast, the incidence rate of death/LT was significantly lower in the DAA-SVR group than in the DAA-no SVR group, whereas the incidence rates of HCC and decompensation did not reach statistical significance, probably because of the short follow-up or rare incidence of decompensation (Supplementary Table 2).
In the SVR cohort (5880.4 patient-years), 30 patients developed HCC, 6 patients had decompensation, 35 patients died, and no patient underwent LT during follow-up. The cumulative incidence rates of HCC at 2 and 5 years were 0.6% and 1.6%, respectively for non-cirrhotic patients with SVR (n = 985), and 4.8% and 10.1%, respectively, for cirrhotic SVR patients (n = 283). The cumulative HCC risk according to the presence of cirrhosis, FIB-4 index, APRI score, and ALBI score were significantly different (Supplementary Figure 3). However, HBcIgG positivity (HR, 0.39; 95%CI: 0.05-2.89; P = 0.358) and the presence of fatty liver disease (HR, 0.16; 95%CI: 0.02-1.18; P = 0.072) were not significant risk factors for the development HCC.
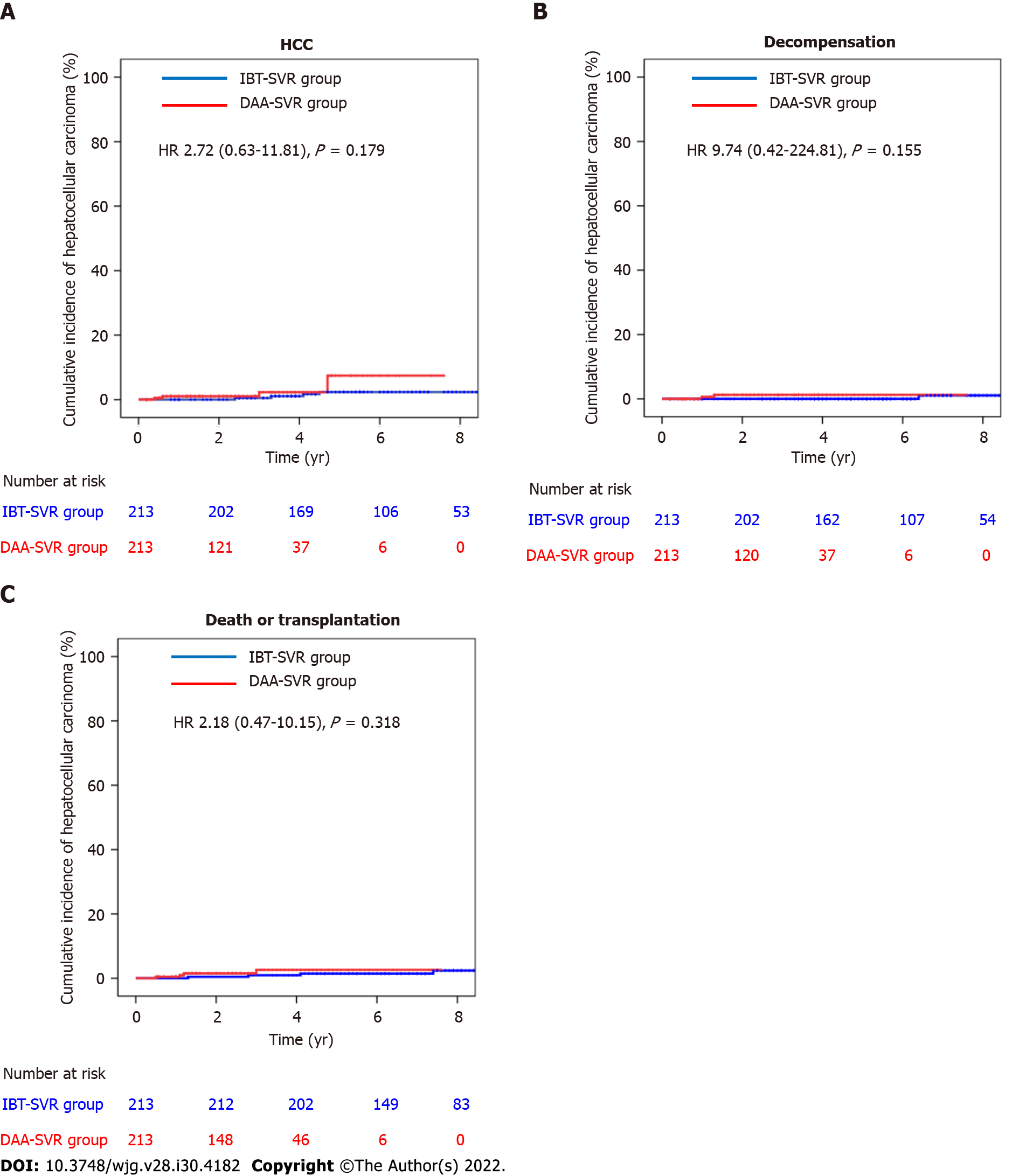
Comparisons of the clinical characteristics and outcomes of the IBT-SVR and DAA-SVR groups before and after PS matching (Table 4) yielded 213 matched pairs of patients from the IBT-SVR and DAA-SVR groups with no significant between-group differences in all baseline variables. Before PS matching, the DAA-SVR group had a significantly higher risk of HCC than the IBT-SVR group (HR, 3.53; 95%CI: 1.47-8.49; P = 0.005) (Supplementary Figure 4A), whereas the risks of decompensation (HR, 2.45; 95%CI: 0.33-18.37; P = 0.382) and death/LT (HR, 1.44; 95%CI: 0.62-3.34; P = 0.400) did not reach statistical significance (Supplementary Figure 4B and C). However, after PS matching, the DAA-SVR group showed no significant differences in the risks of HCC (HR, 2.72; 95%CI: 0.63-11.81; P = 0.179), decompensation (HR, 9.74; 95%CI: 0.42-224.81; P = 0.155), and death/LT (HR, 2.18; 95%CI: 0.47-10.15; P = 0.318) compared with the IBT-SVR group (Figure 3).
| Variable | Before adjustment | PS matched | ||||||
| IBT-SVR group (n = 451) | DAA-SVR group (n = 817) | P value | SMD | IBT-SVR group (n = 213) | DAA-SVR group (n = 213) | P value | SMD | |
| Age, yr | 51.0 ± 10.6 | 59.1 ± 11.4 | < 0.001 | 0.741 | 53.9 ± 9.8 | 53.7 ± 11.4 | 0.823 | 0.022 |
| Male sex | 209 (46.3) | 394 (48.2) | 0.52 | 0.036 | 102 (47.9) | 97 (45.5) | 0.627 | 0.047 |
| HCV RNA, xlog10 IU/mL | 5.72 (4.77-6.37) | 6.04 (5.26-6.52) | < 0.001 | 0.278 | 5.84 (4.91-6.53) | 6.07 (5.21-6.50) | 0.439 | 0.073 |
| HCV genotype | < 0.001 | 0.406 | 0.731 | 0.058 | ||||
| 1 | 167 (37.0) | 464 (56.8) | 95 (44.6) | 101 (47.4) | ||||
| 2 | 278 (61.6) | 342 (41.9) | 116 (54.5) | 111 (52.1) | ||||
| Others | 6 (1.3) | 11 (1.3) | 2 (0.9) | 1 (0.5) | ||||
| Entry-to-treat, year | 0 (0-0.3) | 0.2 (0-2.1) | < 0.001 | 0.654 | 0.1 (0-0.4) | 0.1 (0-0.4) | 0.78 | 0.125 |
| Diagnosis status | 0.001 | 0.192 | 0.901 | 0.012 | ||||
| Chronic hepatitis | 373 (82.7) | 612 (74.9) | 174 (81.7) | 173 (81.2) | ||||
| Compensated cirrhosis | 78 (17.3) | 205 (25.1) | 39 (18.3) | 40 (18.8) | ||||
| FIB-4 index | < 0.001 | 0.294 | 0.363 | 0.071 | ||||
| ≤ 1.45 | 156 (34.8) | 156 (19.9) | 69 (32.7) | 59 (29.1) | ||||
| 1.45–3.25 | 155 (34.6) | 326 (41.6) | 74 (35.1) | 85 (41.9) | ||||
| ≥ 3.25 | 137 (30.6) | 302 (38.5) | 68 (32.2) | 59 (29.1) | ||||
| APRI score | 0.562 | 0.096 | 0.108 | 0.048 | ||||
| < 2.0 | 383 (85.1) | 676 (83.9) | 186 (87.7) | 170 (82.1) | ||||
| ≥ 2.0 | 67 (14.9) | 130 (16.1) | 26 (12.3) | 37 (17.9) | ||||
| ALBI score | 0.958 | 0.044 | 0.339 | 0.054 | ||||
| ≤ -2.60 (Grade 1) | 360 (80.0) | 639 (79.9) | 172 (81.1) | 172 (82.7) | ||||
| > -2.60 (Grade 2 or 3) | 90 (20.0) | 161 (20.1) | 40 (18.9) | 36 (16.3) | ||||
| Ever smoker | 228 (50.8) | 15 (37.5) | 0.045 | 0.162 | 95 (44.6) | 93 (43.7) | 0.527 | 0.019 |
| Alcohol intake | < 0.001 | 0.669 | 0.816 | 0.058 | ||||
| None | 171 (38.0) | 19 (47.5) | 98 (46.0) | 104 (48.8) | ||||
| Social | 245 (54.4) | 13 (32.5) | 88 (41.3) | 85 (39.9) | ||||
| Significant1 | 34 (7.6) | 8 (20.0) | 27 (12.7) | 24 (11.3) | ||||
| Fatty liver disease on imaging study | 77 (22.8), n = 337 | 131 (22.4), n = 586 | 0.863 | 0.039 | 34 (24.3) | 35 (24.8) | 0.917 | 0.013 |
| Anti-HBc IgG positivity | 25 (8.6), n = 290 | 62 (10.5), n = 592 | 0.386 | 0.062 | 12 (7.8) | 15 (9.7) | 0.546 | 0.062 |
| BMI, kg/m2 | 23.7 ± 3.0 | 23.9 ± 3.1 | 0.429 | 0.047 | 23.9 ± 2.9 | 23.9 ± 3.4 | 0.983 | 0.005 |
| Diabetes mellitus | 61 (13.6) | 122 (14.9) | 0.495 | 0.049 | 26 (12.2) | 22 (10.3) | 0.54 | 0.055 |
| Hypertension | 80 (17.7) | 236 (28.9) | < 0.001 | 0.267 | 45 (21.1) | 44 (20.7) | 0.905 | 0.012 |
| Cardiovascular disease | 3 (0.7) | 24 (2.9) | 0.007 | 0.162 | 2 (0.9) | 3 (1.4) | 0.315 | 0.058 |
| Cerebrovascular disease | 2 (0.4) | 19 (2.3) | 0.012 | 0.172 | 1 (0.5) | 3 (1.4) | 0.315 | 0.141 |
| Laboratory study | ||||||||
| WBC, × 103/mm3 | 4.99 (3.90-6.10) | 5.40 (4.40-6.63) | < 0.0010.297 | 0.24 | 5.26 (4.25-6.35) | 5.35 (4.50-6.52) | 0.787 | 0.027 |
| Hemoglobin, g/dL | 13.6 (12.6-14.9) | 13.8 (12.8-14.8) | 0.218 | 0.061 | 13.9 (12.8-14.9) | 13.9 (12.8-14.9) | 0.916 | 0.01 |
| PLT, × 103/mm3 | 177 (136-220) | 173 (130-216) | 0.178 | 0.055 | 176 (136-222) | 180 (139-219) | 0.401 | 0.083 |
| Albumin, g/dL | 4.2 (4.0-4.5) | 4.2 (4.0-4.4) | 0.664 | 0.082 | 4.3 (4.0-4.5) | 4.3 (4.0-4.5) | 0.734 | 0.031 |
| Total bilirubin, mg/dL | 0.7 (0.6-1.0) | 0.7 (0.6-1.0) | < 0.001 | 0.072 | 0.8 (0.6-1.0) | 0.7 (0.6-1.0) | 0.85 | 0.009 |
| ALP, IU/L | 80 (63-113) | 122 (78-250) | 0.012 | 0.612 | 90 (65-126) | 96 (72-183) | 0.524 | 0.072 |
| AST, IU/L | 50 (30-86) | 46 (30-76) | < 0.001 | 0.156 | 50 (32-83) | 42 (27-85) | 0.899 | 0.012 |
| ALT, IU/L | 54 (29-105) | 36 (22-70) | 0.756 | 0.297 | 49 (29-85) | 37 (21-91) | 0.915 | 0.009 |
| Creatinine, mg/dL | 0.8 (0.7-1.0) | 0.8 (0.7-1.0) | 0.02 | 0.09 | 0.8 (0.7-1.0) | 0.8 (0.7-0.9) | 0.176 | 0.069 |
| PT, INR | 1.03 (0.98-1.08) | 1.04 (0.99-1.10) | 0.434 | 0.111 | 1.03 (0.98-1.08) | 1.04 (0.87-1.09) | 0.67 | 0.038 |
| AFP, ng/dL | 3.3 (2.3-6.3) | 3.8 (2.3-6.5) | 0.142 | 5.8 (4.9-6.5) | 6.1 (5.2-6.5) | 0.278 | 0.034 | |
According to the multivariable-adjusted analysis, the DAA-SVR group did not show any independent differences in the development of HCC (HR, 2.03; 95%CI: 0.76-5.43; P = 0.160) or death/LT (HR, 1.38; 95%CI: 0.55-3.49; P = 0.494) compared with the IBT-SVR group (Table 5). Covariates independently associated with a higher incidence of HCC were older age (HR, 1.05; 95%CI: 1.01-1.10; P = 0.025), genotype 1 (HR, 3.02; 95%CI: 1.18-7.68; P = 0.021), and the presence of cirrhosis (HR, 3.18; 95%CI: 1.33-7.68; P = 0.009) after adjustment for the antiviral treatment regimen, sex, alcohol consumption, diabetes mellitus, albumin level, PT, and AFP level (Table 5). Additionally, covariates independently associated with death/LT risk were the presence of cirrhosis (HR, 2.97; 95%CI: 1.42-6.20; P = 0.004) and PT (HR, 5.27; 95%CI: 1.01-27.53; P = 0.049).
| Variable | Hepatocellular carcinoma | Death/transplantation | ||
| aHR (95%CI) | P value | aHR (95%CI) | P value | |
| IBT-SVR group | Reference | - | Reference | - |
| DAA-SVR group | 2.03 (0.76-5.43) | 0.16 | 1.38 (0.55-3.49) | 0.494 |
| Age, yr | 1.05 (1.01-1.10) | 0.025 | - | - |
| Male sex | 2.89 (0.97-8.61) | 0.057 | - | - |
| HCV genotype | - | – | ||
| 2 | Reference | - | ||
| 1 | 3.02 (1.18-7.68) | 0.021 | ||
| Cirrhosis | 3.18 (1.33-7.68) | 0.009 | 2.97 (1.42-6.20) | 0.004 |
| Ever smoker | 0.72 (0.26-1.99) | 0.545 | - | - |
| Alcohol consumption | - | - | ||
| None | Reference | - | ||
| Social | 2.62 (0.92-7.60) | 0.072 | ||
| Significant | 2.51 (0.71-8.82) | 0.152 | ||
| BMI, kg/m2 | - | - | - | - |
| Diabetes mellitus | 1.03 (0.42-2.52) | 0.952 | 2.16 (0.98-4.78) | 0.057 |
| Albumin, g/dL | 0.53 (0.19-1.49) | 0.228 | - | - |
| Total bilirubin, mg/dL | - | - | - | - |
| PT, INR | 4.12 (0.89-19.17) | 0.071 | 5.27 (1.01-27.53) | 0.049 |
| AFP, log10 ng/dL | 1.07 (0.49-2.34) | 0.858 | - | - |
We analyzed the incidence rates of HCC, decompensation, and all-cause death/LT in a large, prospective, Asian cohort including 2054 patients with chronic HCV infection. During a median follow-up period of 4.1 years, the risks of HCC, decompensation, and all-cause mortality were significantly lower after treatment with IBT or DAA compared with no treatment.
After statistical adjustment, including a time-varying Cox analysis and PS-matched analysis, the risks of HCC, decompensation, and all-cause mortality were not significantly different regardless of whether SVR was achieved after IBT or DAA treatment; however, the follow-up duration of the DAA group was shorter than that of the IBT group. After achieving SVR, independent factors associated with HCC risk were no treatment, age, male sex, HCV genotype 1, cirrhosis, alcohol consumption, PT, and pretreatment AFP level, whereas independent factors associated with all-cause mortality were cirrhosis and PT.
There are limited studies of the association between antiviral treatment (IBT or DAA) and all-cause mortality, especially those involving Asian cohorts. Many studies focused on liver-related mortality rather than all-cause mortality as the primary outcome after SVR achievement. However, extrahepatic mortality should be considered for patients with HCV because HCV is associated with increased cardiovascular disease events[20] and extrahepatic malignancies such as bile duct cancers and diffuse large B-cell lymphoma[21,22]. Moreover, Tada et al[23] reported that IBT could reduce all-cause mortality and HCC risk even in patients who did not achieve SVR. However, the effect of DAA treatment in the absence of SVR on clinical outcomes is unknown. Therefore, this study focused on the effect of treatment on the outcomes including all-cause mortality in the entire cohort, and the effect of SVR induced by IBT and DDA on the outcomes in the SVR cohort.
Recent 5-year follow-up studies after DAA treatment showed that SVR is associated with a gradual but significant reduction in liver fibrosis in terms of FIB-4, METAVIR, transient elastography, and Child-Pugh score[24,25], whereas another study showed that induced SVR was associated with a reduced risk of clinical disease progression in patients with Child-Pugh A cirrhosis but not in patients with Child-Pugh B/C cirrhosis[26]. In this context, our study showed an approximately 85% reduction in the risk of decompensation after IBT or DAA treatment in Child-Pugh A patients, with decompensated cirrhotic patients being excluded from enrollment.
This study demonstrated that treatment with IBT and DAA resulted in risk reductions of 72% (median follow-up, 94.8 mo) and 81% (median follow-up, 42 mo) for all-cause mortality, respectively, compared with no treatment after multivariate adjustment. Similar to previous studies[27], our results showed that even IBT, with its probable adverse events and lower SVR rates, had long-term beneficial effects on mortality. Regarding the benefit of DAA treatment, Butt et al[28] (n = 6790), using the Electronically Retrieved Cohort of HCV Infected Veterans (ERCHIVES) data in the United States in 2017, reported that DAA treatment and the achievement of SVR reduced all-cause mortality by 57% and 43%, respectively, within the first 18 mo of treatment. A prospective cohort study performed in France (n = 9895; follow-up, 33.4 mo) reported a 52% reduction in the risk of all-cause mortality in the DAA-treated group compared with the untreated group in 2019[11]. Compared with these studies, our results showed a higher reduction in the risk of all-cause mortality with DAA treatment. This may be related to the low proportion of patients with advanced fibrosis in the DAA group in our cohort. Additionally, the follow-up duration of this study was relatively longer than the durations used for those studies.
During this study, the achievement of SVR by IBT or by DAA resulted in a 59% reduction in the risk of HCC compared with no treatment or no SVR after multivariable adjustment, similar to previous studies[29,30]. Nonetheless, our results also showed that the HCC risk remained after SVR achievement. Despite the achievement of SVR, the absolute risk of HCC remains high for patients with cirrhosis; therefore, according to international guidelines, they should be enrolled in an HCC surveillance program[2-4]. However, there is little evidence of the benefits of HCC surveillance for non-cirrhotic chronic HCV patients with SVR, because of the low residual HCC risk. During this study, the non-cirrhotic group had an HCC risk of 0.26 per 100 person-years (5-year cumulative incidence of 1.6%). In particular, the low FIB-4 (5-year cumulative incidence of 0.3%) group had a very low risk of HCC during this study. These results are similar to those of the retrospective REAL-C cohort study that targeted Eastern Asians (5-year cumulative incidence: 1.35 for the non-cirrhotic group and 0.13 for the low FIB-4 group)[30]. These results support the European guidelines, which do not recommend HCC surveillance for fibrosis 0 to 2[4]. However, more long-term follow-up data after achieving SVR are needed.
Identifying the risk factors associated with HCC after SVR is important to the development of a reasonable surveillance strategy. An East Asian retrospective study suggested that among the cirrhotic DAA-SVR group, age older than 60 years, ALBI scores of 2 or 3, and pretreatment AFP > 10 ng/mL were associated with HCC risk; however, among the non-cirrhotic group, only AFP > 10 ng/mL was significant[30]. Additionally, there are many factors associated with HCC risk after SVR, such as clinical factors (age, sex, presence of diabetes, HCV genotype 3, history of IBT), laboratory parameters (platelet count, AFP), and fibrosis stage (determined by histology or estimation by Fibroscan®, FIB-4, APRI, or the presence of esophageal varices) before DAA treatment and at follow-up (determined by Fibroscan®, FIB-4, APRI, alanine aminotransferase, AFP, albuminemia, or VITRO score)[31]. In our study, age, cirrhosis, and HCV genotype 1 (compared with genotype 2) were independent risk factors for HCC after SVR. Therefore, it is necessary to test the HCV genotype even in the therapeutic era of pangenotypic regimens to develop a follow-up strategy after SVR according to the regional distribution of HCV genotypes. These factors should be considered when establishing an optimal HCC prediction model.
Previous studies have suggested a similar risk of HCC development with IBT and DAA treatment[32]. Interestingly, a recent meta-analysis suggested that IBT is better than DAA for preventing the occurrence of HCC in chronic hepatitis C patients after achieving SVR[33]. Biologically, IFN family members, especially class I IFN (IFN-α, IFN-β), have important anti-HCC effects[32]. However, some investigators have suggested that the sudden decrease in viral load caused by DAA treatment causes immune distortion, thus deregulating the antitumor response and releasing precancerous foci from immune surveillance[34,35]. Although it can be explained theoretically, there is little clinical evidence regarding the difference in the HCC risk for patients with DAA-SVR and IBT-SVR. Our results showed significant clinical differences between the IBT-SVR and DAA-SVR groups. Both the multivariable analysis and PS matching analysis showed that the effect of IBT-SVR was not different from that of DAA-SVR on the HCC incidence and all-cause mortality. Recently, some studies reported different effects of IBT-SVR and DAA-SVR on the incidence of diabetes[36] and hematologic malignancies[37]. Therefore, long-term studies of the effects of DAA-SVR are warranted.
To meet the 2030 HCV elimination target of the WHO[1], active testing and enhanced linkage to treatment are important. In this study, 30.1% of patients remained untreated, and this percentage should be reduced. Indeed, the untreated group showed a higher mortality rate of 23.7% (88 Liver related deaths and 59 non-liver related deaths) during the median follow-up of 5.6 years. In the untreated group, 3 patients received IBT after HCC diagnosis, and 19 patients received DAA treatment after HCC diagnosis or a decompensation event. The majority of the untreated patients (78.8%, 457/619) were enrolled in the Korean HCV cohort before June 2015, when the first DAA was introduced, and 46.7% (289/619) died or were lost to follow-up before June 2015. Although DAA therapy is covered by the National Health Insurance System, 30% of the drug price is an out-of-pocket expense of the patients, equaling to approximately 3000 United States dollars. Even in the DAA era, the main reasons for non-treatment are the relatively high price of DAA, old age, and extrahepatic or advanced hepatic malignancy.
One of the strengths of our study was that it was a long-term study involving a prospective, Asian cohort that included both IBT and DAA groups. This study used a well-established protocol, guidelines and database, and the data, including death outcomes, were verified under government guidance. To date, few reports have evaluated and compared clinical outcomes (HCC, death, and decompensation) after IBT and DAA treatment, especially in Asia, where HCV is less prevalent than HBV.
This study had several limitations. First, because it was an observational study, the findings may show selection and confounding biases. Despite this limitation, it would be useful for comparing the effectiveness of IBT and DAA because of the relatively low incidence of clinical events. Second, we used clinical and radiological criteria to diagnose cirrhosis; therefore, some patients with advanced fibrosis may have been included in the non-cirrhotic group. However, we attempted to correct this point using a non-invasive liver fibrosis biomarker. Third, there was a disparity in the follow-up periods of the IBT and DAA groups because of the later introduction of DAA (since 2015 in Korea). Additional long-term follow-up studies evaluating the outcomes of patients with SVR are warranted. Forth, the presence of anti-HBc IgG in HCV patients has been implicated in HCC development, and a non-negligible risk of HBV inactivation during DAA treatment (0.91%)[38]. However, approximately 30% of our patients were not tested for anti-HBc IgG, and none of the anti-HBc IgG-positive patients were tested for HBV DNA during DAA treatment. Likewise, the role of fatty liver in the outcomes of HCV patients was not completely evaluated owing to missing data. Finally, compared with the IBT era, diagnostic imaging modalities and treatment options for HCC have improved in the DAA era. Therefore, these points could have affected the clinical outcomes of the IBT and DAA groups.
This study showed that antiviral treatment significantly reduced the incidences of HCC, decompensation, and mortality in an Asian population, regardless of the use of IBT or DAA. After achieving SVR, age, the presence of cirrhosis, and HCV genotype 1 were indicators of worse clinical outcomes. Therefore, an adequate HCC surveillance strategy after SVR that considers age, the presence of cirrhosis, and genotype should be developed. Additional studies evaluating the long-term DAA outcomes of SVR patients are also warranted.
Sustained virologic response (SVR) with either interferon-based therapy (IBT) or direct acting antivirals (DAA)-induced SVR significantly reduces the incidence of hepatocellular carcinoma (HCC) and long-term mortality in patients with hepatitis C virus (HCV) infection.
Prospective studies of the long-term outcomes for patients with HCV infection after treatment with IBT or DAA are limited in many Asian countries.
We aimed to elucidate the incidences of HCC and death/transplantation based on treatment with IBT or DAA. And, we aimed to compare the outcomes of the SVR to IBT and DAA. Finally, we aimed to investigate outcome-determining factors after SVR.
The cohort included 2054 viremic patients (mean age, 57 years; 46.5% male; 27.4% with cirrhosis) prospectively enrolled at seven hospitals between 2007 and 2019. They were classified as the untreated group (n = 619), IBT group (n = 578), and DAA group (n = 857).
Compared to the untreated group, the incidences of HCC and death/transplantation were significantly lower in the IBT group and the DAA group. SVR induced by IBT or DAA did not show significant differences in the risk of HCC and all-cause mortality. After achieving SVR, age, presence of cirrhosis, and genotype 1 HCV infection were indicators of worse clinical outcomes.
Treatment and SVR with either IBT or DAA significantly reduced the incidences of HCC and mortality in the Asian prospective cohort.
This study was a long-term study involving a prospective, Asian cohort that included both IBT and DAA groups. This study used a well-established protocol, guidelines and database, and the data, including death outcomes, were verified under government guidance.
We deeply appreciate the clinical research coordinators (Lee DE, Kim NH, Jeong DH, Na SM, Kang HY, Jo AR, Han SH, Chung ES, Jeong HY, and Lee HM) for their dedication to this study and the officials of the Korea Disease Control and Prevention Agency (Lee MS, Seong JH, Lee JK, Kee MK, Chung HM, Choi BS, Kim KS, Kang C, Kim SS, and Jee YM) for their strong support of this study.
Provenance and peer review: Unsolicited article; Externally peer reviewed.
Peer-review model: Single blind
Specialty type: Gastroenterology and hepatology
Country/Territory of origin: South Korea
Peer-review report’s scientific quality classification
Grade A (Excellent): 0
Grade B (Very good): B, B
Grade C (Good): C
Grade D (Fair): 0
Grade E (Poor): 0
P-Reviewer: Li J, China; Tamori A, Japan; Zarębska-Michaluk D, Poland S-Editor: Yan JP L-Editor: A P-Editor: Yan JP
| 1. | World Health Organization. Interim guidance for country validation of viral hepatitis elimination. World Health Organization, 2021. Available from: https://apps.who.int/iris/handle/10665/341652. |
| 2. | Korean Association for the Study of the Liver (KASL). 2017 KASL clinical practice guidelines management of hepatitis C: Treatment of chronic hepatitis C. Clin Mol Hepatol. 2018;24:169-229. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 28] [Cited by in RCA: 32] [Article Influence: 4.6] [Reference Citation Analysis (0)] |
| 3. | Ghany MG, Morgan TR; AASLD-IDSA Hepatitis C Guidance Panel. Hepatitis C Guidance 2019 Update: American Association for the Study of Liver Diseases-Infectious Diseases Society of America Recommendations for Testing, Managing, and Treating Hepatitis C Virus Infection. Hepatology. 2020;71:686-721. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 348] [Cited by in RCA: 528] [Article Influence: 105.6] [Reference Citation Analysis (0)] |
| 4. | European Association for the Study of the Liver; Clinical Practice Guidelines Panel: Chair; EASL Governing Board representative; Panel members. EASL recommendations on treatment of hepatitis C: Final update of the series☆. J Hepatol. 2020;73:1170-1218. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 567] [Cited by in RCA: 778] [Article Influence: 155.6] [Reference Citation Analysis (0)] |
| 5. | Fried MW, Shiffman ML, Reddy KR, Smith C, Marinos G, Gonçales FL Jr, Häussinger D, Diago M, Carosi G, Dhumeaux D, Craxi A, Lin A, Hoffman J, Yu J. Peginterferon alfa-2a plus ribavirin for chronic hepatitis C virus infection. N Engl J Med. 2002;347:975-982. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 4847] [Cited by in RCA: 4748] [Article Influence: 206.4] [Reference Citation Analysis (0)] |
| 6. | Bang CS, Song IH. Impact of antiviral therapy on hepatocellular carcinoma and mortality in patients with chronic hepatitis C: systematic review and meta-analysis. BMC Gastroenterol. 2017;17:46. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 39] [Cited by in RCA: 48] [Article Influence: 6.0] [Reference Citation Analysis (0)] |
| 7. | Lee YB, Nam JY, Lee JH, Chang Y, Cho H, Cho YY, Cho EJ, Yu SJ, Kim HY, Lee DH, Lee JM, Hwang SG, Kim YJ, Yoon JH. Differential Effect of HCV Eradication and Fibrosis Grade on Hepatocellular Carcinoma and All-cause Mortality. Sci Rep. 2018;8:13651. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 6] [Cited by in RCA: 8] [Article Influence: 1.1] [Reference Citation Analysis (0)] |
| 8. | Dolmazashvili E, Abutidze A, Chkhartishvili N, Karchava M, Sharvadze L, Tsertsvadze T. Regression of liver fibrosis over a 24-week period after completing direct-acting antiviral therapy in patients with chronic hepatitis C receiving care within the national hepatitis C elimination program in Georgia: results of hepatology clinic HEPA experience. Eur J Gastroenterol Hepatol. 2017;29:1223-1230. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 60] [Cited by in RCA: 64] [Article Influence: 8.0] [Reference Citation Analysis (0)] |
| 9. | Bachofner JA, Valli PV, Kröger A, Bergamin I, Künzler P, Baserga A, Braun D, Seifert B, Moncsek A, Fehr J, Semela D, Magenta L, Müllhaupt B, Terziroli Beretta-Piccoli B, Mertens JC. Direct antiviral agent treatment of chronic hepatitis C results in rapid regression of transient elastography and fibrosis markers fibrosis-4 score and aspartate aminotransferase-platelet ratio index. Liver Int. 2017;37:369-376. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 169] [Cited by in RCA: 195] [Article Influence: 24.4] [Reference Citation Analysis (0)] |
| 10. | Afdhal N, Everson GT, Calleja JL, McCaughan GW, Bosch J, Brainard DM, McHutchison JG, De-Oertel S, An D, Charlton M, Reddy KR, Asselah T, Gane E, Curry MP, Forns X. Effect of viral suppression on hepatic venous pressure gradient in hepatitis C with cirrhosis and portal hypertension. J Viral Hepat. 2017;24:823-831. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 89] [Cited by in RCA: 99] [Article Influence: 12.4] [Reference Citation Analysis (0)] |
| 11. | Carrat F, Fontaine H, Dorival C, Simony M, Diallo A, Hezode C, De Ledinghen V, Larrey D, Haour G, Bronowicki JP, Zoulim F, Asselah T, Marcellin P, Thabut D, Leroy V, Tran A, Habersetzer F, Samuel D, Guyader D, Chazouilleres O, Mathurin P, Metivier S, Alric L, Riachi G, Gournay J, Abergel A, Cales P, Ganne N, Loustaud-Ratti V, D'Alteroche L, Causse X, Geist C, Minello A, Rosa I, Gelu-Simeon M, Portal I, Raffi F, Bourliere M, Pol S; French ANRS CO22 Hepather cohort. Clinical outcomes in patients with chronic hepatitis C after direct-acting antiviral treatment: a prospective cohort study. Lancet. 2019;393:1453-1464. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 362] [Cited by in RCA: 462] [Article Influence: 77.0] [Reference Citation Analysis (0)] |
| 12. | Nahon P, Layese R, Bourcier V, Cagnot C, Marcellin P, Guyader D, Pol S, Larrey D, De Lédinghen V, Ouzan D, Zoulim F, Roulot D, Tran A, Bronowicki JP, Zarski JP, Riachi G, Calès P, Péron JM, Alric L, Bourlière M, Mathurin P, Blanc JF, Abergel A, Serfaty L, Mallat A, Grangé JD, Attali P, Bacq Y, Wartelle C, Dao T, Thabut D, Pilette C, Silvain C, Christidis C, Nguyen-Khac E, Bernard-Chabert B, Zucman D, Di Martino V, Sutton A, Roudot-Thoraval F, Audureau E; ANRS CO12 CirVir Group. Incidence of Hepatocellular Carcinoma After Direct Antiviral Therapy for HCV in Patients With Cirrhosis Included in Surveillance Programs. Gastroenterology. 2018;155:1436-1450.e6. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 185] [Cited by in RCA: 181] [Article Influence: 25.9] [Reference Citation Analysis (0)] |
| 13. | Sahakyan Y, Lee-Kim V, Bremner KE, Bielecki JM, Krahn MD. Impact of direct-acting antiviral regimens on mortality and morbidity outcomes in patients with chronic hepatitis c: Systematic review and meta-analysis. J Viral Hepat. 2021;28:739-754. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 13] [Cited by in RCA: 28] [Article Influence: 7.0] [Reference Citation Analysis (0)] |
| 14. | Vallet-Pichard A, Mallet V, Nalpas B, Verkarre V, Nalpas A, Dhalluin-Venier V, Fontaine H, Pol S. FIB-4: an inexpensive and accurate marker of fibrosis in HCV infection. comparison with liver biopsy and fibrotest. Hepatology. 2007;46:32-36. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 1288] [Cited by in RCA: 1609] [Article Influence: 89.4] [Reference Citation Analysis (0)] |
| 15. | Loaeza-del-Castillo A, Paz-Pineda F, Oviedo-Cárdenas E, Sánchez-Avila F, Vargas-Vorácková F. AST to platelet ratio index (APRI) for the noninvasive evaluation of liver fibrosis. Ann Hepatol. 2008;7:350-357. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 63] [Cited by in RCA: 65] [Article Influence: 3.8] [Reference Citation Analysis (0)] |
| 16. | Johnson PJ, Berhane S, Kagebayashi C, Satomura S, Teng M, Reeves HL, O'Beirne J, Fox R, Skowronska A, Palmer D, Yeo W, Mo F, Lai P, Iñarrairaegui M, Chan SL, Sangro B, Miksad R, Tada T, Kumada T, Toyoda H. Assessment of liver function in patients with hepatocellular carcinoma: a new evidence-based approach-the ALBI grade. J Clin Oncol. 2015;33:550-558. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 1962] [Cited by in RCA: 2009] [Article Influence: 200.9] [Reference Citation Analysis (0)] |
| 17. | Korean Liver Cancer Association; National Cancer Center. 2018 Korean Liver Cancer Association-National Cancer Center Korea Practice Guidelines for the Management of Hepatocellular Carcinoma. Gut Liver. 2019;13:227-299. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 234] [Cited by in RCA: 241] [Article Influence: 40.2] [Reference Citation Analysis (0)] |
| 18. | Garcia-Tsao G, Friedman S, Iredale J, Pinzani M. Now there are many (stages) where before there was one: In search of a pathophysiological classification of cirrhosis. Hepatology. 2010;51:1445-1449. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 359] [Cited by in RCA: 392] [Article Influence: 26.1] [Reference Citation Analysis (0)] |
| 19. | Lee J, Lee JS, Park SH, Shin SA, Kim K. Cohort Profile: The National Health Insurance Service-National Sample Cohort (NHIS-NSC), South Korea. Int J Epidemiol. 2017;46:e15. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 363] [Cited by in RCA: 731] [Article Influence: 104.4] [Reference Citation Analysis (0)] |
| 20. | Chaudhari R, Fouda S, Sainu A, Pappachan JM. Metabolic complications of hepatitis C virus infection. World J Gastroenterol. 2021;27:1267-1282. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in CrossRef: 38] [Cited by in RCA: 40] [Article Influence: 10.0] [Reference Citation Analysis (3)] |
| 21. | Mahale P, Torres HA, Kramer JR, Hwang LY, Li R, Brown EL, Engels EA. Hepatitis C virus infection and the risk of cancer among elderly US adults: A registry-based case-control study. Cancer. 2017;123:1202-1211. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 55] [Cited by in RCA: 81] [Article Influence: 10.1] [Reference Citation Analysis (0)] |
| 22. | Masarone M, Persico M. Hepatitis C virus infection and non-hepatocellular malignancies in the DAA era: A systematic review and meta-analysis. Liver Int. 2019;39:1292-1306. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 30] [Cited by in RCA: 31] [Article Influence: 5.2] [Reference Citation Analysis (0)] |
| 23. | Tada T, Kumada T, Toyoda H, Kiriyama S, Tanikawa M, Hisanaga Y, Kanamori A, Kitabatake S, Yama T, Tanaka J. Viral eradication reduces all-cause mortality in patients with chronic hepatitis C virus infection: a propensity score analysis. Liver Int. 2016;36:817-826. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 54] [Cited by in RCA: 55] [Article Influence: 6.1] [Reference Citation Analysis (0)] |
| 24. | Flisiak R, Zarębska-Michaluk D, Janczewska E, Łapiński T, Rogalska M, Karpińska E, Mikuła T, Bolewska B, Białkowska J, Flejscher-Stępniewska K, Tomasiewicz K, Karwowska K, Pazgan-Simon M, Piekarska A, Berak H, Tronina O, Garlicki A, Jaroszewicz J. Five-Year Follow-Up of Cured HCV Patients under Real-World Interferon-Free Therapy. Cancers (Basel). 2021;13. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 11] [Cited by in RCA: 6] [Article Influence: 1.5] [Reference Citation Analysis (0)] |
| 25. | Poordad F, Castro RE, Asatryan A, Aguilar H, Cacoub P, Dieterich D, Marinho RT, Carvalho A, Siddique A, Hu YB, Charafeddine M, Bondin M, Khan N, Cohen DE, Felizarta F. Long-term safety and efficacy results in hepatitis C virus genotype 1-infected patients receiving ombitasvir/paritaprevir/ritonavir + dasabuvir ± ribavirin in the TOPAZ-I and TOPAZ-II trials. J Viral Hepat. 2020;27:497-504. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 6] [Cited by in RCA: 5] [Article Influence: 1.0] [Reference Citation Analysis (0)] |
| 26. | Krassenburg LAP, Maan R, Ramji A, Manns MP, Cornberg M, Wedemeyer H, de Knegt RJ, Hansen BE, Janssen HLA, de Man RA, Feld JJ, van der Meer AJ. Clinical outcomes following DAA therapy in patients with HCV-related cirrhosis depend on disease severity. J Hepatol. 2021;74:1053-1063. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 50] [Cited by in RCA: 99] [Article Influence: 24.8] [Reference Citation Analysis (0)] |
| 27. | Yoshida H, Arakawa Y, Sata M, Nishiguchi S, Yano M, Fujiyama S, Yamada G, Yokosuka O, Shiratori Y, Omata M. Interferon therapy prolonged life expectancy among chronic hepatitis C patients. Gastroenterology. 2002;123:483-491. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 176] [Cited by in RCA: 164] [Article Influence: 7.1] [Reference Citation Analysis (0)] |
| 28. | Butt AA, Yan P, Simon TG, Abou-Samra AB. Effect of Paritaprevir/Ritonavir/Ombitasvir/Dasabuvir and Ledipasvir/Sofosbuvir Regimens on Survival Compared With Untreated Hepatitis C Virus-Infected Persons: Results From ERCHIVES. Clin Infect Dis. 2017;65:1006-1011. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 27] [Cited by in RCA: 28] [Article Influence: 4.0] [Reference Citation Analysis (0)] |
| 29. | Kanwal F, Kramer J, Asch SM, Chayanupatkul M, Cao Y, El-Serag HB. Risk of Hepatocellular Cancer in HCV Patients Treated With Direct-Acting Antiviral Agents. Gastroenterology. 2017;153:996-1005.e1. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 523] [Cited by in RCA: 685] [Article Influence: 85.6] [Reference Citation Analysis (0)] |
| 30. | Tanaka Y, Ogawa E, Huang CF, Toyoda H, Jun DW, Tseng CH, Hsu YC, Enomoto M, Takahashi H, Furusyo N, Yeh ML, Iio E, Yasuda S, Lam CP, Lee DH, Haga H, Yoon EL, Ahn SB, Wong G, Nakamuta M, Nomura H, Tsai PC, Jung JH, Song DS, Dang H, Maeda M, Henry L, Cheung R, Yuen MF, Ueno Y, Eguchi Y, Tamori A, Yu ML, Hayashi J, Nguyen MH; REAL-C Investigators. HCC risk post-SVR with DAAs in East Asians: findings from the REAL-C cohort. Hepatol Int. 2020;14:1023-1033. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 52] [Cited by in RCA: 49] [Article Influence: 9.8] [Reference Citation Analysis (0)] |
| 31. | Negro F. Residual risk of liver disease after hepatitis C virus eradication. J Hepatol. 2021;74:952-963. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 15] [Cited by in RCA: 32] [Article Influence: 8.0] [Reference Citation Analysis (0)] |
| 32. | Nagaoki Y, Imamura M, Aikata H, Daijo K, Teraoka Y, Honda F, Nakamura Y, Hatooka M, Morio R, Morio K, Kan H, Fujino H, Kobayashi T, Masaki K, Ono A, Nakahara T, Kawaoka T, Tsuge M, Hiramatsu A, Kawakami Y, Hayes CN, Miki D, Ochi H, Chayama K. The risks of hepatocellular carcinoma development after HCV eradication are similar between patients treated with peg-interferon plus ribavirin and direct-acting antiviral therapy. PLoS One. 2017;12:e0182710. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 37] [Cited by in RCA: 42] [Article Influence: 5.3] [Reference Citation Analysis (0)] |
| 33. | Ma L, Liu J, Wang W, Yang F, Li P, Cai S, Zhou X, Chen X, Zhuang X, Zhang H, Cao G. Direct-acting antivirals and interferon-based therapy on hepatocellular carcinoma risk in chronic hepatitis-C patients. Future Oncol. 2020;16:675-686. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 6] [Cited by in RCA: 6] [Article Influence: 1.2] [Reference Citation Analysis (0)] |
| 34. | Llovet JM, Villanueva A. Liver cancer: Effect of HCV clearance with direct-acting antiviral agents on HCC. Nat Rev Gastroenterol Hepatol. 2016;13:561-562. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 54] [Cited by in RCA: 67] [Article Influence: 7.4] [Reference Citation Analysis (0)] |
| 35. | Llorens-Revull M, Costafreda MI, Rico A, Guerrero-Murillo M, Soria ME, Píriz-Ruzo S, Vargas-Accarino E, Gabriel-Medina P, Rodríguez-Frías F, Riveiro-Barciela M, Perales C, Quer J, Sauleda S, Esteban JI, Bes M. Partial restoration of immune response in Hepatitis C patients after viral clearance by direct-acting antiviral therapy. PLoS One. 2021;16:e0254243. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 10] [Cited by in RCA: 12] [Article Influence: 3.0] [Reference Citation Analysis (0)] |
| 36. | Butt AA, Yan P, Aslam S, Shaikh OS, Abou-Samra AB. Hepatitis C Virus (HCV) Treatment With Directly Acting Agents Reduces the Risk of Incident Diabetes: Results From Electronically Retrieved Cohort of HCV Infected Veterans (ERCHIVES). Clin Infect Dis. 2020;70:1153-1160. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 10] [Cited by in RCA: 19] [Article Influence: 4.8] [Reference Citation Analysis (0)] |
| 37. | Ioannou GN, Green PK, Berry K, Graf SA. Eradication of Hepatitis C Virus Is Associated With Reduction in Hematologic Malignancies: Major Differences Between Interferon and Direct-Acting Antivirals. Hepatol Commun. 2019;3:1124-1136. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 10] [Cited by in RCA: 15] [Article Influence: 2.5] [Reference Citation Analysis (0)] |
| 38. | Kanda T, Lau GKK, Wei L, Moriyama M, Yu ML, Chuang WL, Ibrahim A, Lesmana CRA, Sollano J, Kumar M, Jindal A, Sharma BC, Hamid SS, Kadir Dokmeci A, Mamun-Al-Mahtab, McCaughan GW, Wasim J, Crawford DHG, Kao JH, Ooka Y, Yokosuka O, Sarin SK, Omata M. APASL HCV guidelines of virus-eradicated patients by DAA on how to monitor HCC occurrence and HBV reactivation. Hepatol Int. 2019;13:649-661. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 86] [Cited by in RCA: 68] [Article Influence: 11.3] [Reference Citation Analysis (0)] |