Published online Jul 28, 2021. doi: 10.3748/wjg.v27.i28.4504
Peer-review started: January 15, 2021
First decision: January 31, 2021
Revised: February 12, 2021
Accepted: June 7, 2021
Article in press: June 7, 2021
Published online: July 28, 2021
Processing time: 191 Days and 17.6 Hours
Severe acute respiratory syndrome coronavirus 2 (SARS-CoV-2) is considered the causative pathogen of coronavirus disease 2019 (COVID-19) and has become an international danger to human health. Although respiratory transmission and symptoms are still the essential manifestations of COVID-19, the digestive system could be an unconventional or supplementary route for COVID-19 to be trans
Core Tip: The coronavirus disease 2019 (COVID-19) pandemic has affected millions worldwide, with high lethality. In addition to typical respiratory symptoms of COVID-19, gastrointestinal and hepatic symptoms have been frequently observed in COVID-19 patients. The main purpose of this article is to focus on the manifestations, mecha
- Citation: Mohamed DZ, Ghoneim MES, Abu-Risha SES, Abdelsalam RA, Farag MA. Gastrointestinal and hepatic diseases during the COVID-19 pandemic: Manifestations, mechanism and management. World J Gastroenterol 2021; 27(28): 4504-4535
- URL: https://www.wjgnet.com/1007-9327/full/v27/i28/4504.htm
- DOI: https://dx.doi.org/10.3748/wjg.v27.i28.4504
Coronaviruses (CoV) is the largest group of spike-like viruses in the Nidovirales family. In the last two decades the CoVs have caused three worldwide outbreaks, with the most recent pandemic caused by severe acute respiratory syndrome coronavirus 2 (SARS-CoV-2) which has resulted in coronavirus disease 2019 (COVID-19). The first 21st century outbreak in Guangdong Province, China, in November 2002, caused by SARS-CoV (SARS-CoV-1), an extreme SARS, resulted in 8098 fatalities (9.6%) globally[1,2]. The second outbreak in Saudi Arabia in 2012 was caused by Middle East respiratory syndrome (MERS)-CoV, with 2521 (36%) confirmed deaths[3].
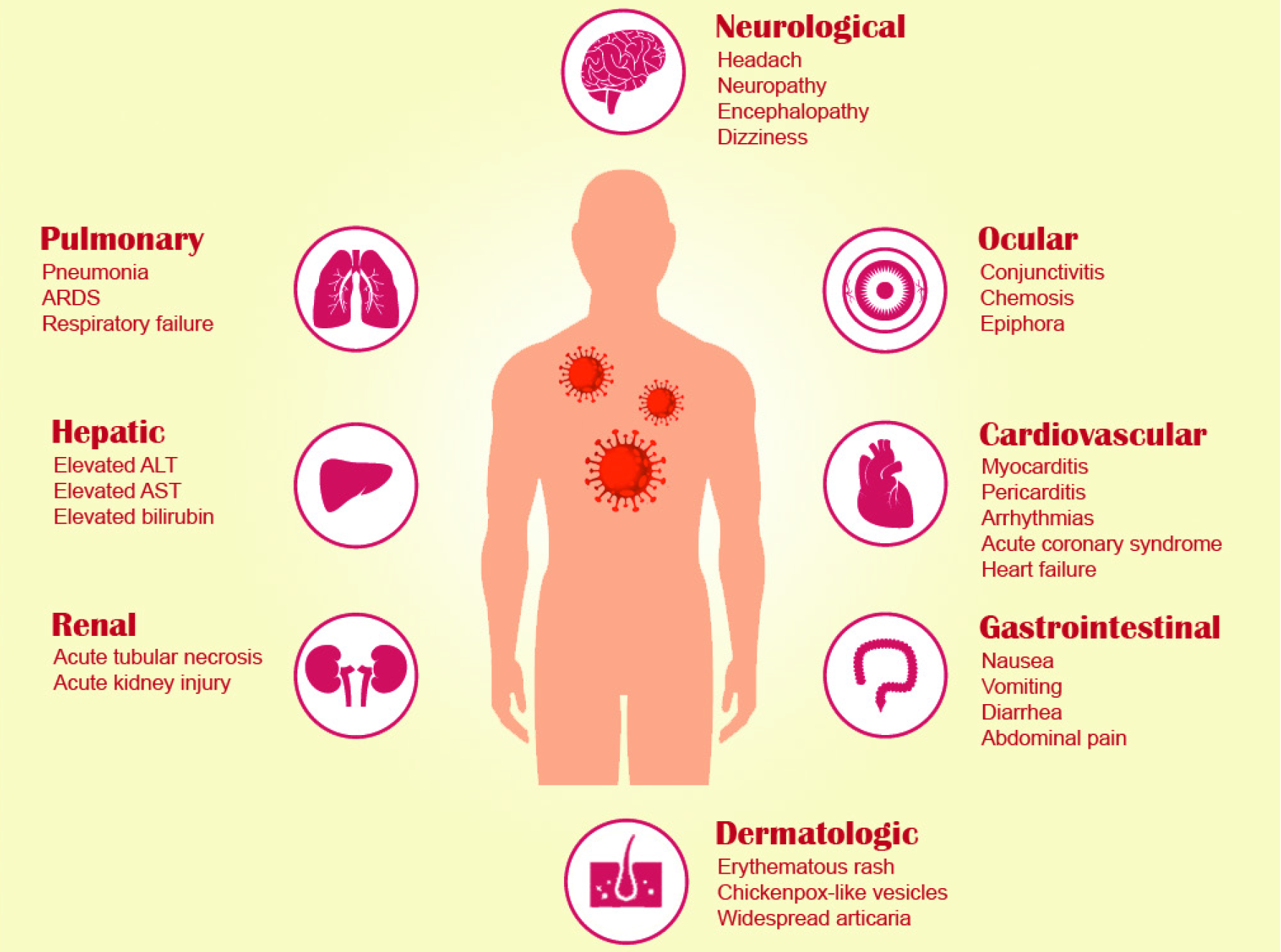
SARS-CoV-2 first originated in China in December 2019 and critically threatened worldwide health[4,5]. On February 12, 2021, 108 million cases and 2 million deaths have been recorded in over 219 countries and regions worldwide[6]. The lung is the primary organ involved in COVID-19 pneumonia, and most COVID-19 patients suffer typical respiratory symptoms (e.g., dyspnea, cough with sputum production, fatigue, and in severe cases, acute respiratory distress syndrome (ARDS), respiratory failure, and even death). On the other hand, extrapulmonary clinical manifestations of COVID-19 affect multiple other organs including cardiovascular (e.g., arrhythmias, myocarditis, pericarditis, acute coronary syndrome, and heart failure), renal (e.g., acute kidney injury and acute tubular necrosis), hepatic [e.g., elevated alanine aminotransferase (ALT), aspartate aminotransferase (AST), and bilirubin], gastrointestinal (e.g., diarrhea, nausea, vomiting, and abdominal pain), ocular (e.g., epiphora, conjunctivitis, and chemosis), dermatologic (e.g., erythematous rash, urticaria, and chickenpox-like vesicles), and neurological systems (e.g., headache, neuropathy, encephalopathy, cerebrovascular disorders, and dizziness)[7] (Figure 1). Considering all the previously mentioned data, this article examines the effects of gastrointestinal (GI) and hepatic symptoms, their associated mechanisms, and management caused by SARS-CoV-2 infection and provides a guide for clinical prevention and treatment (Figure 2).
For SARS-CoV-2, the incubation period is an average of 4-5 d, with most patients having symptoms before 14 d, although there have been instances where the incu
Overall, the case fatality ratio of COVID-19 is reported to be around 1-2% in patients aged 80 years, to over 15%[10]. Currently, WHO data show that most COVID-19 cases have mild to moderate signs of illness (80%), and 13.8% of cases have serious signs within 24-48 h with the following symptoms: shortness of breath; hypoxia < 300% and/or pulmonary infiltration > 50%; tachypnea > 30 breaths/min[11]. 6.1% of pa
It has been demonstrated that 51% of reported cases of COVID-19 are male patients. This may be due to the higher levels of estrogen in female COVID-19 patients which can reduce COVID-19 severity and mortality via the elevation in innate and humoral immunity[13-19]. Moreover, in vivo studies have demonstrated that there are higher levels of angiotensin-converting enzyme 2 (ACE2) expression in male kidneys than in female kidneys, which may explain the differences in the susceptibility and develop
Whether ACE2 expression varies in the lungs of male and female COVID-19 patients, is still unclear[20,21]. Furthermore, preclinical trials have suggested that ACE2 expression may increase vulnerability to COVID-19 in pregnant patients[22,23].
In the same way, COVID-19 varies between different ethnic groups in terms of severity and mortality. During the COVID-19 pandemic, American, Hispanic, and African communities have displayed higher rates of infection and hospitalization in comparison to Caucasian communities[24]. These discrepancies may be due to the higher occurrence of heart diseases, hypertension, obesity, diabetes, and asthma in minority groups[24].
Gastrointestinal symptoms including, nausea, anorexia, vomiting, diarrhea, and abdominal pain are common in COVID-19 patients (Table 1)[12,25-35]. In the SARS infection of 2002-2003, diarrhea was the main feature and appeared in 16%-73% of SARS patients mainly in the first week of infection[36]. Similarly, diarrhea is con
| Ref. | Patient number | Anorexia, nausea or vomiting, n (%) | Diarrhea, n (%) | Abdominal pain, n (%) |
| Kujawski et al[207], 2020 | 12 | Nausea: 3 (25) | 4 (33.3) | 2 (16.7) |
| Hajifathalian et al[44], 2020 | 1059 | Anorexia: 240 (22.7) | 234 (22.1) | 72 (6.8) |
| Nausea: 168 (15.3) | ||||
| Vomiting: 91 (8.6) | ||||
| Young et al[208], 2020 | 18 | NA | 3 (17) | NA |
| Tabata et al[209], 2020 | 104 | NA | 8 (9.6) | NA |
| Wölfel et al[210], 2020 | 9 | NA | 2 (22) | NA |
| Chen et al[26], 2020 | 99 | Nausea and vomiting: 1 (1) | 2 (2) | NA |
| Xu et al[27], 2020 | 62 | NA | 3 (8) | NA |
| Gritti et al[211], 2020 | 21 | NA | 5 (23.8) | NA |
| COVID-19 National Incident Room Surveillance Team[212] | 295 | Nausea: 34 (11.5) | 48 (16.3) | 6 (1) |
| COVID-19 National Emergency response Center[213] | 28 | NA | 2 (7) | 1 (4) |
| Sierpiński et al[214], 2020 | 1942 | NA | 470 (24.2) | NA |
| Wu et al[28], 2020 | 80 | Nausea and vomiting: 1 (1.25) | 1 (1.3) | NA |
| Wang et al[12], 2020 | 138 | Anorexia: 55 (39.9) | 14 (10.1) | 3 (2.2) |
| Nausea: 14 (10.1) | ||||
| Shi et al[29], 2020 | 81 | Anorexia: 1 (1) | 3 (4) | NA |
| Vomiting: 4 (5) | ||||
| Yang et al[30], 2020 | 50 | Vomiting: 2 (4) | NA | NA |
| Mo et al[31], 2020 | 155 | Anorexia: 26 (31.7) | 7 (4.5) | NA |
| Nausea: 3 (3.7) | ||||
| Vomiting: 3 (3.7) | ||||
| Qi et al[215], 2020 | 267 | Anorexia: 46 (14.2) | 10 (3.7) | NA |
| Nausea: 6 (2.2) | ||||
| Wen et al[216], 2020 | 417 | NA | 29 (7) | NA |
| Dan et al[217], 2020 | 305 | Anorexia: 101(50.2) | 146 (49.5) | 12 (6) |
| Nausea: 59 (29.4) | ||||
| Vomiting: 3 (2) | ||||
| Ma et al[218], 2020 | 81 | NA | 6(7.41) | NA |
| Luo et al[41], 2020 | 1141 | Anorexia: NA | 68 (6) | 45 (3.9) |
| Nausea: 134 (11.7) | ||||
| Vomiting: 119 (10.4) | ||||
| Liu et al[219], 2020 | 238 | Anorexia: 14 (9.2) | 14 (9.2) | 1 (0.7) |
| Nausea: 2 (1.3) | ||||
| Vomiting: 3 (2) | ||||
| Ai et al[220], 2020 | 102 | Anorexia: NA | 15 (14.3) | 3 (2.9) |
| Nausea: 9 (8.8) | ||||
| Vomiting: 2 (2) | ||||
| Zhao et al[221], 2020 | 75 | NA | 7 (9.3) | 1 (1.3) |
| Li et al[222], 2020 | 83 | NA | 7 (8.4) | 7 (8.4) |
| Lin et al[223], 2020 | 95 | Anorexia: 17 (17.8) | 23 (24.2) | 2 (2.1) |
| Nausea: 17 (17.9) | ||||
| Vomiting: 4 (4.2) | ||||
| Cholankeril et al[224], 2020 | 207 | Anorexia: NA | 22 (10.8) | 14 (7.1) |
| Nausea: 22 (10.8) | ||||
| Vomiting: NA | ||||
| Ferm et al[43], 2020 | 892 | Anorexia: 105 (11.8) | 177 (19.8) | 70 (7.8) |
| Nausea: 148 (16.6) | ||||
| Vomiting: 91 (10.2) | ||||
| Redd et al[225], 2020 | 318 | Anorexia: 110 (34.8) | 107 (33.7) | 46 (14.5) |
| Nausea: 84 (26.4) | ||||
| Vomiting: 49 (15.4) | ||||
| Kluytmans et al[226], 2020 | 86 | NA | 16 (18.6) | 5 (5.8) |
Among 204 COVID-19 patients in Hubei China, only 99 (48.5%) had gastrointestinal signs as their chief complaint. These COVID-19 patients had a diversity of digestive symptoms, including abdominal pain (0.4%), anorexia (83.8%), diarrhea (29.3%), and vomiting (0.8%)[38]. A retrospective study conducted by Guan et al[39] reported that among 1099 COVID-19 patients from 552 centers across China, 5% of cases had nausea and vomiting, and 3.8% of cases had diarrhea. A further study indicated that 32.5% of COVID-19 patients had at least one gastrointestinal tract (GIT) symptom. These symptoms included diarrhea (37.8%), anorexia (56.7%), abdominal pain (10.4%), nausea (16.5%), and vomiting (7.9%)[40]. Additionally, Luo et al[41] documented that 16% of 1141 cases, had at least one GIT symptom including, anorexia (98%), nausea, vomiting (66%), diarrhea (37%), and abdominal pain (25%). Furthermore, a research study conducted by Jin et al[42], which was the first COVID-19 study outside Wuhan, reported that the incidence of GIT symptoms was 11.4%; diarrhea was the most common of these symptoms (8.14%)[42].
In New York, GIT symptoms have been identified in a single-center-case series, which included 892 cases. The most common symptom was diarrhea (19.8%), accompanied by nausea (16.6%), abdominal pain (7.8%), vomiting (10.2%), and anorexia (11.8%)[43]. An additional retrospective study conducted in New York showed that of 1059 COVID-19 patients, 22% had diarrhea, 6% had nausea, 19% had vomiting, and 7% had abdominal pain[44]. In addition to these studies, the pooled prevalence of GI symptoms was found to be 17.6 % in a meta-analysis of 60 studies involving 4243 cases in 6 countries The most frequent symptom was anorexia (26.8%), accompanied by diarrhea (12.5%), nausea/vomiting (10.2%) and stomach pain/dis
Furthermore, Goyal et al[46] indicated that hyperlipidemia was found in 11.7% of patients affected by SARS-CoV-2 where 92 patients out of 756 with COVID-19, had elevated serum lipase levels, resulting in acute pancreatitis. Therefore, COVID-19 patients with hyperlipidemia have an approximately 3-fold higher risk of poor clinical outcomes, including the need for ICU admission, mechanical ventilation, and death[46].
The study by Gadiparthi et al[47] showed that the higher Glasgow Blatchford bleeding score was 7 and 11 in 2 of 3 patients on admission and represents a high risk of gastrointestinal bleeding (GIB) with a need for intervention of more than 50%. However, both young patients responded by carefully controlling hemodynamic parameters, levels of hematocrit or hemoglobin, transfusion of packed red blood cells as needed and medical therapy. Although GIB was resolved, two patients died due to respiratory failure[47].
Besides GIT symptoms, autopsies of COVID-19 patients showed that there are gross and microscopic changes in the GIT. Segmental dilation and stenosis of the small intestine, accompanied by mucosal shedding and necrosis was described at autopsy, whereas colitis, inflammatory infiltrates, and interstitial edema, were found on histopathology imaging[48,49]. Lui et al[50] have shown pneumatosis, intestinalis, pneumoperitoneum, ascites, and thickening bowel wall in addition to ileus on abdominal imaging. Therefore, abnormalities of the GIT can be obvious on imaging.
Based on these previous data, physicians must realize that the key characteristics of COVID-19 infection before respiratory symptoms may be digestive symptoms and, in rare instances may be the only COVID-19 symptoms to occur.
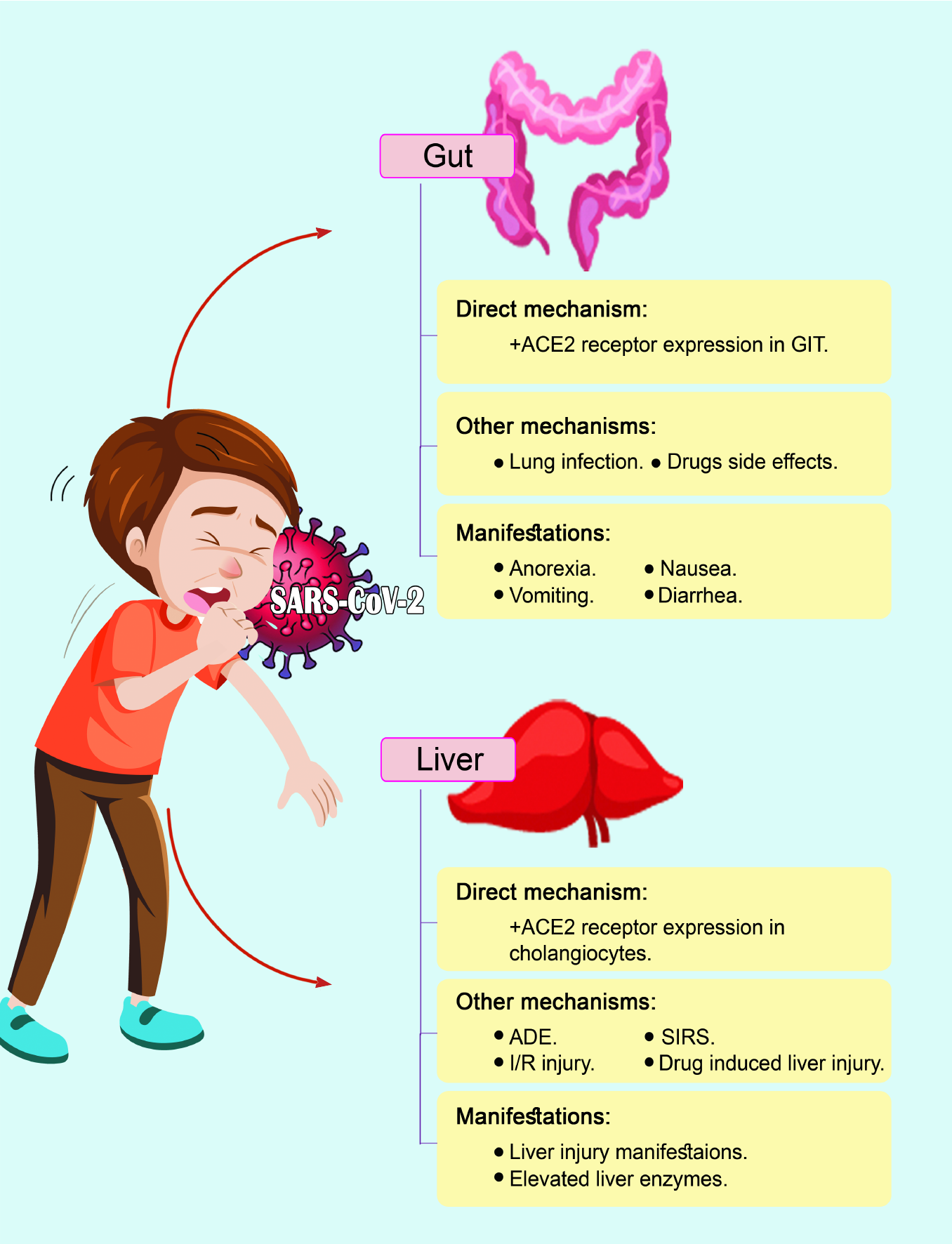
Direct infection of gastrointestinal cells: The entry of SARS-CoV-2 virus into host cells is the main part of cross-species transmission. All coronaviruses bind to receptors and mediate their entry via glycoprotein and spike proteins[51]. It has been recognized that ACE2 mainly contains receptors for SARS-CoV[52], and dipeptidyl peptidase 4, for MERS-CoV[53]. A plethora of studies has shown that the spike (S) protein of SARS-CoV-2 has a high host ACE2 affinity[54], and enters host cells via the ACE2 receptor[55] as shown in Figure 3.
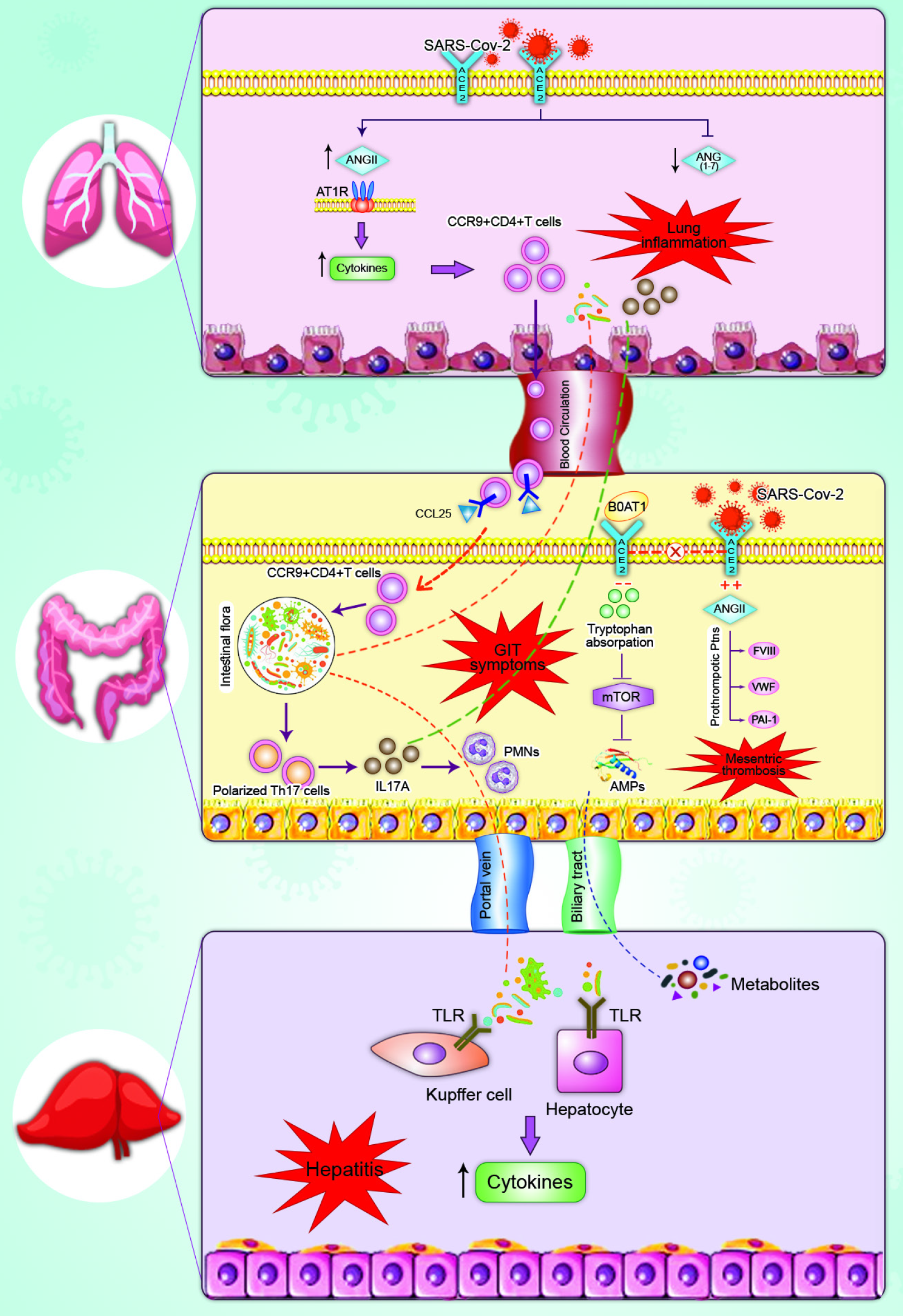
In small intestine cells, ACE2 is usually located and strongly expressed in type II epithelial cells[56,57]. It is considered an important enzyme in the renin-angiotensin system (RAS)[58,59] and plays an essential role in controlling inflammation and diarrhea[60]. Moreover, its deficiency leads to the accumulation of angiotensin II (ANG II)[61]. Subsequent studies[12,62-64] have proven that SARS-CoV-2, in COVID-19 patients, may invade the GIT via ACE2 receptors, causing digestive symptoms. It is now well acknowledged that the significant upsurge of ANG II level in COVID-19 patients is the cause of progression and severity of the disease.
Although in the mouse colon, the knockdown of ACE2 by the virus leads to an upsurge of ANG II levels; ACE2 does not primarily act via the intestinal RAS system but controls the homeostasis of intestinal amino acids, gut microbes and the expression of antimicrobial peptides[65]. Mice with blocked ACE2, for example, have significantly inhibited serum tryptophan that is important to the body’s niacin synthesis[66], therefore, inadequate intake of tryptophan or niacin results in pellagra[65]. It is well-recognized that tryptophan is absorbed on the lumen surface of intestinal epithelial cells via the B0AT1/ACE2 transport route and induces the mammalian target of rapamycin, which controls the consequently appearance of antimicrobial peptides, thereby having a direct influence on components of intestinal flora[65]. Ultimately, more than 90% of pellagra patients will develop colitis[67]. Hence, it is considered that SARS-CoV-2 attachment to the ACE2 in the GIT lowers the critical receptor level, disrupting the absorption of tryptophan and ultimately destroying the steady-state of the gut flora, resulting in diarrhea.
Furthermore, He et al[68] demonstrated that the pathological results of autopsies on COVID-19 patients indicated that proinflammatory cytokines (PICs) such as tumor growth factor-β1, monocyte chemokine-1 (MCP-1), tumor necrosis factor-α (TNF-α), interleukin (IL)-1β, and IL-6 were highly expressed in ACE2-expressing cells. There is an absence of PICs in cells that do not express ACE2. Numerous PICs produce the cytokine storm, which finally leads to multiple organ failure. Besides, plasma IL-2, recombinant human interferon (IFN)-induced protein-10, MCP-1, macrophage inflammatory protein-1A, IL-7, granulocyte colony-stimulating factor, IL-10, and TNF-α levels in COVID-19 patients were higher than those in healthy people[25]. Dendritic cells and macrophages show low levels of IFN and high levels of PICs and chemokines in the early stage of SARS-CoV infection[69-71]. Later, there is a rise in cytokine and chemokine levels, which causes severe tissue damage due to the large number of inflammatory cells, such as neutrophils and monocytes. In the same manner, SARS-CoV-2 can induce cells expressing ACE2 to release inflammatory cytokines, leading to the cytokine storm and multiple organ failure[72]. Furthermore, cytokine storms can have an effect on immune cells; lymphocytosis is known to be a common sign in severe cases of COVID-19, with a significant decline in the number of B, CD8+ and CD4+T cells as well as natural killer cells, which leads to lymph node necrosis, spleen atrophy, hepatomegaly, renal hemorrhage, necrosis, and degeneration of neurons[73].
Thromboembolic complications are being increasingly documented in COVID-19[74]. Acute mesenteric ischemia (AMI) has been reported in severe COVID-19 patients in addition to deep venous thrombosis and pulmonary embolism[75]. It has been demonstrated that ANG II stimulates the expression of tissue factor VIII (FVIII), von Willebrand factor, and plasminogen activator inhibitor-1 by endothelial cells, resulting in a state of hypercoagulation[76,77]. Histology of the small intestine secondary to mesenteric thrombosis revealed a prominent endothelium of the submucosa with evidence of direct viral invasion of endothelial cells along with diffuse endothelial swelling with mononuclear cell infiltrate. It is understood that there is stimulation of alternate and lectin complement trajectories [C5b-9 (membrane attack complex), C4d, and mannose-binding protein-associated serine protease 2] that destroy endothelial cells[78]. SARS-CoV-2 infection also resulted in an elevation of ANG II levels and other prothrombotic proteins, which led to AMI[78-81].
Gastrointestinal damage caused by lung infection: Any change in intestinal flora constituents affects the respiratory tract, via the common mucous immune system. Reciprocally, any damage to the respiratory tract mainly affects the digestive tract via immune regulation. This effect is known as the “gut-lung axis”[82,83]. Furthermore, SARS-CoV-2 cannot be detected in the stools of COVID-19 patients with digestive symptoms and thus GIT symptoms may not be affected by the direct damage caused by SARS-CoV-2. The entry of CD4+ T cells is essential to immunity and chronic en
Gastrointestinal symptoms caused by drug side effects: Diarrhea due to antibiotics is the most common unwanted side effect of antimicrobial agents, including cephalos
Proton pump inhibitors (PPIs) are mainly used as a therapy for patients with peptic ulcers and gastroesophageal reflux disease. By blocking the proton pump, PPIs decrease gastric acid released into the stomach. Although the decrease in stomach acid can be beneficial in patients with stomach diseases, it can make the gut more susceptible to COVID-19 infection[97-99]. SARS-CoV S protein was suggested to fuse with patients’ cells in neutral pH conditions. Additionally, Darnell et al[100] also established that highly acidic pH (1-3) and alkaline pH (12-14) may lead to inactivation of SARS-CoV, while, in the case of neutral pH, the virus remains stable[101]. Zhou et al[102] reported that, under the conditions of pH 1.0 and 2.0, SARS-CoV-2 was inacti
In general, the existence of comorbidities is associated with dramatically low outcomes in COVID-19 patients. This may cause consequences for the management of previous gastrointestinal patients[104]. Patients that have inflammatory bowel disease (IBD) and have been treated with immunosuppressive drugs are more at risk for regular and serious infections and may be more at risk for SARS-CoV-2 infection[105]. It was reported in the IBD registry in Wuhan that none of 318 patients (204 patients with ulcerative colitis and 114 patients with Crohn’s disease) had been infected with SARS-CoV-2 following precautions for COVID-19 control and prevention[106]. Thus, the use of biologics and immunosuppressants, diet therapy, deliberate postponement of elective and endoscopic surgery and provisions for personal safety are recommended[107].
Considerations and management of IBD medications during COVID-19 infection: It is currently not recommended that IBD patients stop immunosuppressant drugs[11]. This is because there is a risk of disease reactivation in patients who discontinue their treatment, leading to severe inflammation, surgery, increased risk of hospitalization, and infection[108,109]. Non-hormonal anti-inflammatory drug use should be prohibi
No evidence-based recommendations for immunosuppressed patients have been made by clinical parties. Table 2 shows the specific considerations for the treatment of IBD patients mainly based on specialist beliefs. These suggestions can be edited and updated as new evidence emerges[108,110]. Overall, the same therapies can be used by patients with moderate to serious Crohn’s disease or ulcerative colitis, regardless of COVID-19 infection[110]. IBD therapies can be restarted after 14 d for asymptomatic IBD patients with SARS-CoV-2 infection whose therapy has been stopped[110].
| Drug | Effects |
| Aminosalicylate acid derivatives (5-ASA) | No proof of increased risk of COVID-19 infection |
| Continue treatment even in the case of COVID-19 infection | |
| Corticosteroids | Their safety during COVID-19 infection is uncertain |
| They can be used at a low dose and for a short period to treat disease relapses | |
| Discontinue as soon as possible | |
| Ileo-cecal CD patients can be treated with Budesonide; IUC patients can be treated with Budesonide MMX | |
| Immunomodulators (Thiopurines and Methotrexate) | No proof of increased risk of COVID-19 infection |
| Accompanied by increased risk of other viral infection | |
| Not recommended to start with monotherapy | |
| Combination therapy with biologics should be maintained | |
| Recommendations in stopping | |
| Stable disease | |
| Sustained reduction in the case of elderly patients and/or significant comorbidities | |
| Symptom progression of COVID-19 infection | |
| Anti-TNF therapy | No proof of increased risk of COVID-19 infection |
| Infusion and dose intervals should be maintained | |
| Starting with monotherapy (adalimumab or certolizumab) | |
| Stop in the case of developing symptoms of COVID-19 | |
| Anti-IL-12/23p40 therapy (Ustekinumab) | No proof of increased risk of COVID-19 infection |
| Monotherapy is recommended | |
| Stop in the case of developing symptoms of COVID-19 | |
| Anti-a4b7 integrin therapy (Vedolizumab) | No proof of increased risk of COVID-19 infection |
| Monotherapy is recommended | |
| Stop in the case of developing symptoms of COVID-19 | |
| Janus Kinase inhibitors (tofacitinib) | Although there is no proof of increasing the risk of COVID-19 infection, it may inhibit the immune reaction against viral infections |
| Starting is not recommended | |
| Therapy should be maintained without elevating the dose | |
| Stop if symptoms of COVID-19 develop |
It is noteworthy that IBD patients should discontinue immunosuppressant drugs until COVID-19 is resolved, and medications can be resumed after complete resolution of COVID-19 symptoms or after two negative polymerase chain reaction (PCR) tests[111].
IBD surgery in the context of the COVID-19 pandemic: The new strain of corona
A number of articles have disclosed that SARS-CoV-2 RNA can be found in patients’ stool, suggesting that SARS-CoV-2 has the ability to be transmitted via the fecal-oral route[115-117]. It has been demonstrated that some COVID-19 patients had positive fecal but negative PCR tests[118]. Wang et al[117] revealed that of 153 (29%) patients with COVID-19, 44 showed positive stool virus. Moreover, Xiao et al[49] indicated that of 73 COVID-19 patients hospitalized in China, 39 (53.42%) showed SARS-CoV-2 RNA in stools[49]. Surprisingly, stool samples were positive in 17 (23.29%) patients but respiratory samples were negative and the period of positivity ranged from 1 to 12 d in stools. Xiao et al[49] demonstrated that infectious SARS-CoV-2 may be secreted from virus-infected gastrointestinal cells. Therefore, stool samples should be frequently tested using real-time-PCR and transmission-based precautions should be taken into consideration[119].
Furthermore, the probability of SARS-CoV-2 fecal-oral transmission has highlighted the importance of adequate hand hygiene, especially in certain areas. Strict measures must be taken to deal with the stools of COVID-19 patients and the sewage from hospitals. Therefore, patients with preexisting gastrointestinal diseases will be more worried when they are infected with SARS-CoV-2 in addition to potential fecal microbiota transplant donors.
Microecological preparation: The intestinal flora generates various vitamins, bile acids, immune factors, and fatty acids via the decomposition of food and participates in immune system regulation[120]. If there is dysfunction or damage in the intestinal flora and/or mucosa, the virus may further stimulate infection through this pathway. Other studies have stated that probiotics have the availability to manage diarrhea caused by rotavirus[121]. Also, lactic acid bacteria and Bifidobacteria can contribute to antiviral antibodies in the human body, accelerating virus removal. Therefore, pro
ACE2 inhibitors: As previously mentioned, ACE2 binds SARS-CoV-2 in cells. Therefore, to avoid SARS-CoV-2 infection the interaction between the receptor connection domain (RBD) of the virus and ACE2 could be blocked. A number of studies have shown that ACE2 inhibitors can regulate bowel metabolism, innate immunity, secretion of antibacterial peptides, and intestinal microbial homeostasis[122]. It was experimentally confirmed that the ACE2 pathway in epithelial cells of the small intestine trigger mTOR via nutrient stimulation and/or the tryptophan-nicotinamide pathway, thus affecting the composition of intestinal flora, and decreasing GIT indications in mice[60]. Azathioprine is an immunosuppressive agent similar to chloroquine. A recent study evaluated azathioprine and found that it could be regarded as an ACE2 inhibitor[123]. Additionally, in vitro studies have shown that Vaccinia virus can be inhibited by azathioprine[124]. With regard to molecular im
Diet and enteral nutrition: A number of COVID-19 patients have significant anorexia. To ensure effective therapy, the basic energy, absorption, enteral and peristalsis movement of the intestine, and normal function of GIT micro-organisms and mucosal immunity should be taken into account as well as the functioning of the GIT and patients’ nutrition[94]. Nutritional risk assessment should be performed in patients with serious COVID-19 and gastrointestinal symptoms[125]. Once the risk of enteral nutrition has been eliminated, enteral nutrition should immediately be restored. If the patient has a gastrointestinal disorder and cannot withstand enteral nutrition, paren
Hepatic injury is a frequent adverse event in both SARS-CoV and Middle East res
Data from a preprint meta-analysis involving 20 retrospective studies and 3428 COVID-19 patients showed that increased levels of COVID-19 were related to significant increases in the levels of ALT, AST, and bilirubin[130]. Recently, a plethora of studies reported increased serum ALT, AST, and GGT levels in severe compared with mild or non-severe COVID-19 patients[39,131]. A recent meta-analysis associated high marker admission levels with patient mortality[132]. Other studies have shown that the increase in these parameters resulted in a worse pulmonary computed tomo
According to a study conducted by Lei et al[132], AST was the first indicator to be considered high when patients were admitted to hospital and was correlated with the highest COVID-19 death rates. A recent study carried out by Guan et al[39], around 1100 Chinese patients reported elevated levels of serum AST and ALT of 18% and 26% in non-severe COVID-19 patients, respectively, compared with 56% and 28% in severe COVID-19 patients. Previous findings revealed the crucial role of immune-mediated systemic inflammation in liver dysfunction in patients with serious COVID-19[9]. Recently, Gordon et al[136] proposed that mitochondrial proteins may interact directly with the virus, providing a cause for the high level of AST in the liver profile[136]. Wang et al[137] also identified anomalies in liver enzymes, higher radiologic scores as well as a partial pressure differential between alveolar arterial oxygen, higher GGT, disease intensity, higher ferritin, lower CD4+ T and B cells and lower albumin. It is considered that total bilirubin, AST/ALT, and ALT/ALP ratios have helped to predict survival in cirrhotic COVID-19 patients[138]. Serum albumin levels were also markedly lower in patients who died of COVID-19[32]. The adverse path of COVID-19 patients has been shown to include elevated serum levels of IL-6, ferritin, procalcitonin, and C-reactive protein. In addition to reduced albumin content and platelet count, the parallel increase in the level of ferritin, ALT and IL-6 indicates a greater role for liver participation in COVID-19[32,134]. The CT imaging score for pulmonary lesions is known as a hepatic injury indicator. Therefore, patients must undergo careful monitoring of liver function in order to identify any liver insults at an early stage[133]. The incidence of hepatic abnormalities in COVID-19 patients are depicted in Table 3.
| Ref. | Patient number | ALT (U/L) | AST (U/L) | TB (mg/dL) |
| Zhou et al[32], 2020 | 191 | ↑59 (31%) | None | NA |
| Shu et al[227], 2021 | 545 | ↑41 (7.5%) | ↑35 (10.1%) | ↑189 (34.7%) |
| Huang et al[25], 2020 | 41 | NA | ↑15 (37%) | NA |
| Huang et al[228], 2020 | 36 | 4 (13.3%) | 18 (58%) | ↑4 (12.9%) |
| Chen et al[26], 2020 | 99 | ↑28 (28%) | ↑35 (35%) | ↑18 (18%) |
| Ai et al[64], 2020 | 102 | ↑20 (19.6%) | ↑26 (25.5%) | NA |
| Xu et al[27], 2020 | 62 | ↑3 (3.75%) | ↑3 (3.75%) | NA |
| Yang et al[3], 2020 | 168 | ↑9 (8%) | ↑18 (17.3%) | ↑7 (6.4%) |
| Wu et al[28], 2020 | 80 | ↑3 (3.75%) | ↑3 (3.75%) | NA |
| Yao et al[229], 2020 | 40 | ↑21 (52.5%) | ↑16 (40%) | |
| Xu et al[230], 2020 | 355 | ↑91 (25.6%) | ↑102 (28.7%) | ↑10 (25%) |
| Cai et al[231], 2020 | 298 | ↑39 (13.1%) | ↑25 (8.4%) | ↑66 (18.6) |
| Richardson et al[1], 2020 | 5700 | ↑2176 (39.0%) | ↑3263 (58.4%) | ↑24 (8.1%) |
| NA | ||||
| Fan et al[1], 2020 | 40 | ↑27 (18.2%) | ↑32 (21.6%) | ↑9 (6.1%) |
| Guan et al[39], 2020 | 355 | ↑158 (21.3%) | ↑168 (22.2%) | ↑76 (10.5%) |
Morphological findings regarding the interpretation and description of liver parenchymal changes accompanying COVID-19 are rare, and most are seen at auto
Moreover, the study by Ramachandran et al[144] showed that in hospitalized COVID-19 patients, elevated aminotransferases were associated with higher mecha
| Ref. | Outcome of patients | SARS-CoV-2 patients with hypertransaminasemia (n = 20) | COVID-19 patients without hypertransaminasemia (n = 125) | P value |
| Ramachandran et al[169], 2020 | Shock | 9 (45%) | 38 (30.4%) | 0.207 |
| Mechanical ventilation | 10 (50%) | 30 (24%) | 0.028 | |
| Died | 10 (50%) | 46 (36.8%) | 0.324 | |
| Length of stay in days, median (IQR) | 7 (4.3, 10.3) | 7 (5, 10) | 0.78 |
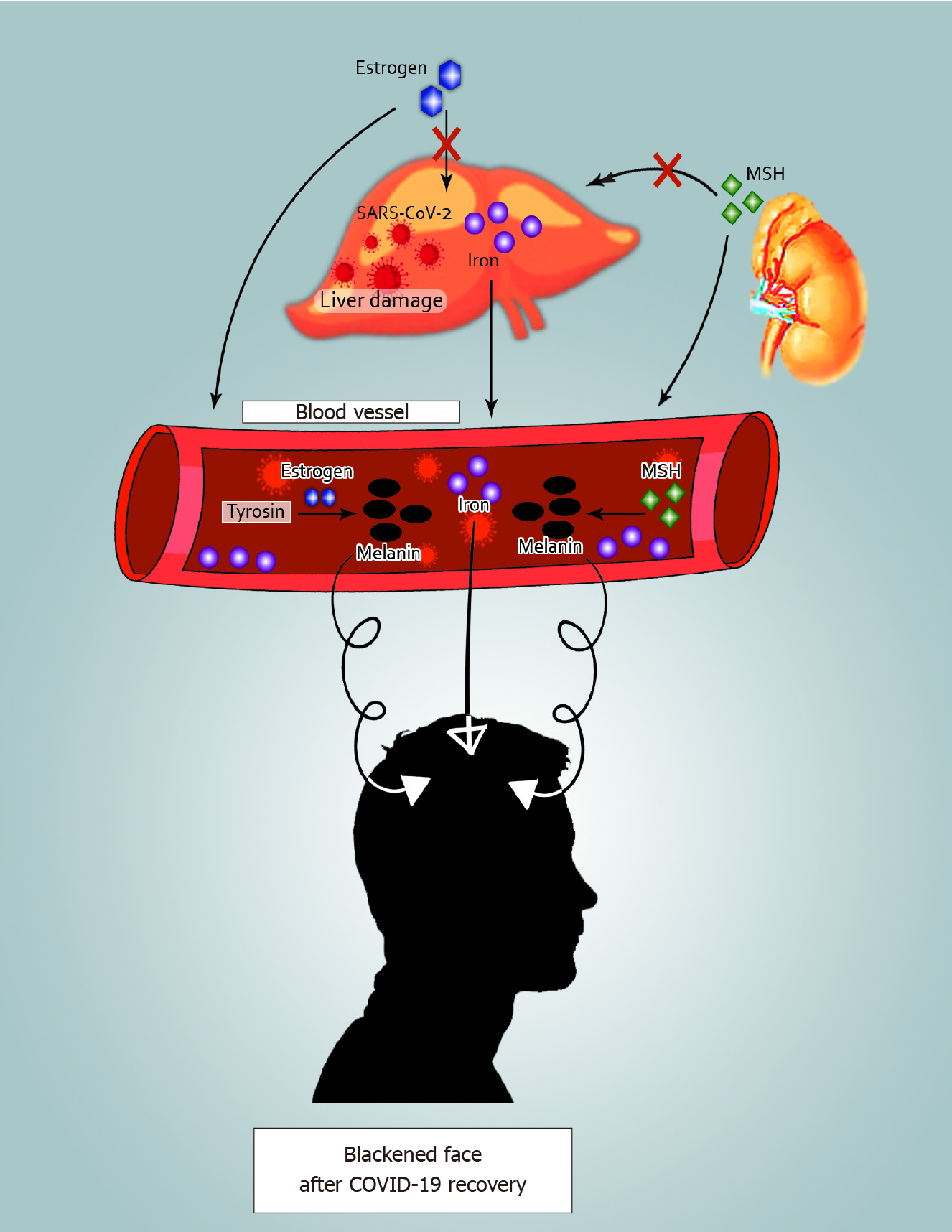
In China, some patients recovering from extreme COVID-19 were confirmed to have experienced special manifestations including a darkened face and pigmentation during treatment. The key causes of a darkened face and pigmentation were multiple organ injury, in particular liver injury[145,146]. Abnormal hepatic function can easily lead to pigmentation via the following three pathways: (1) Liver dysfunction can prevent estrogen from being inactivated[147]. The rise in estrogen decreases thiamine inhibition of tyrosinase in vivo, thus increasing the transformation of tyrosine into melanin[148]; (2) Abnormal liver function can cause hypofunction of adrenocortical hormones. The liver does not metabolize melanocytic stimulatory hormone produced by the anterior pituitary gland, which causes greater melanin secretion[149]; and (3) Liver damage can cause bleaching of the face due to iron in the blood that provides the facial skin[150,151] (Figure 4).
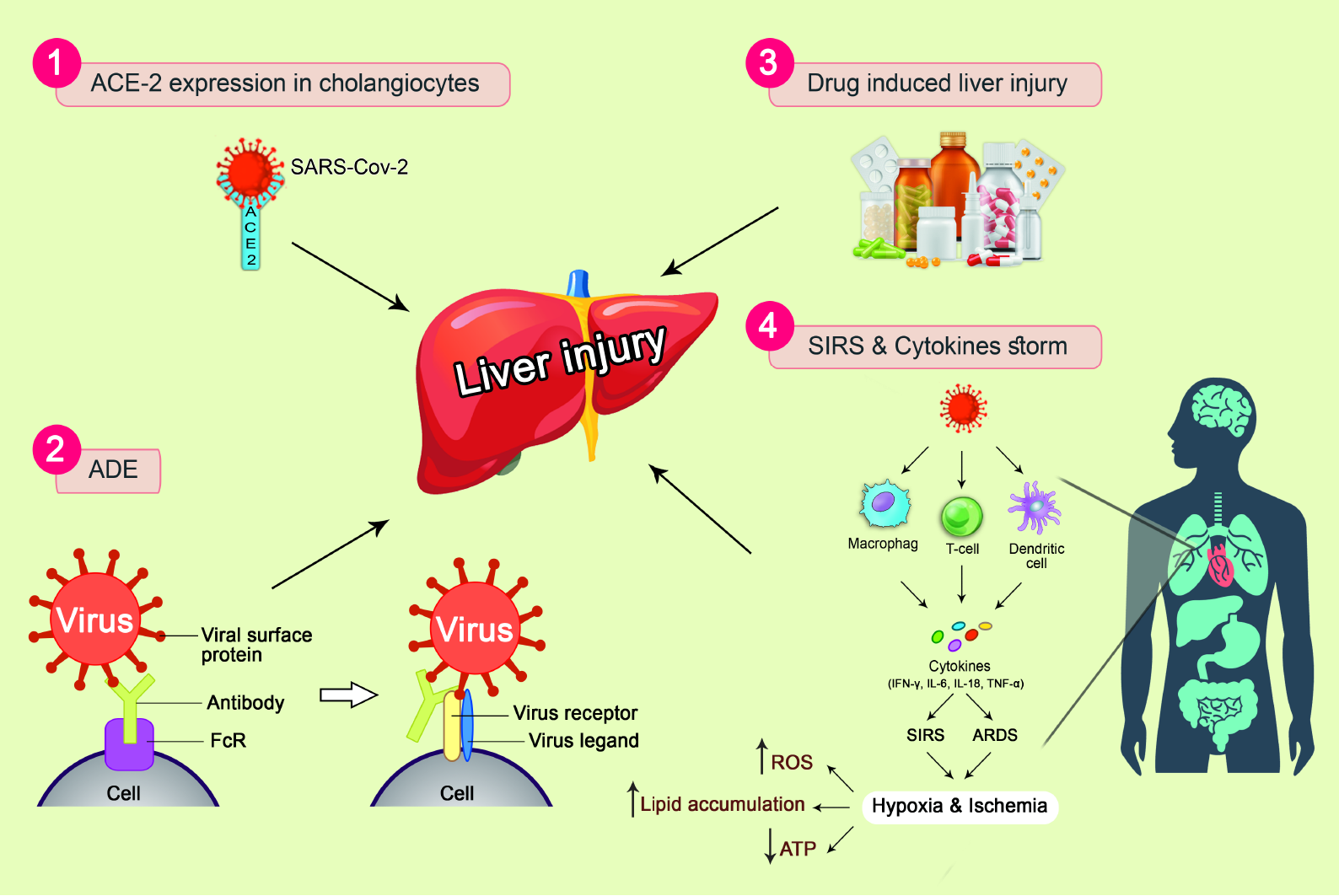
Direct damage: It is established that SARS-CoV-2 enters the host cells via its binding to ACE2 on the surface of the host cell by the S protein[152,153]. However, the expression of ACE2 levels in liver tissue was estimated to be approximately 0.31% and its expression in bile duct cells was 20 times higher than that in hepatocytes on the basis of single cell sequencing and animal model analysis[154]. Furthermore, elevated levels of γ-glutamyl transferase and alkaline phosphatase were observed in COVID-19 patients[155], and inconsistent with biliary epithelial cells injury, approximately 10% of COVID-19 patients have high levels of total bilirubin. Thus, suggesting that SARS-CoV-2 can bind to cholangiocytes expressing ACE2, resulting in their injury (Figure 5).
Antibody-dependent enhancement: Antibody-dependent enhancement of infection (ADE) may arise in patients with SARS[156]. ADE indicates that the interplay between the virus-based antibody and the CR and/or FC receptor complements increases the ability of the virus to reach macrophages, granulocytes, and monocytes (Figure 5). The virus frequently replicates in the aforementioned cells, leading to an increase in virus production and worsening of infection. Previous findings have indicated that SARS-CoV antibodies activate ADE, triggering SARS-CoV in immune cells that do not have ACE2 expression or harm the immune system[157]. Whether ADE can help SARS-CoV-2 infect immune cells through a non-ACE2-dependent pathway and participate in SARS-CoV-2 hepatic injury is an issue of concern.
Systemic inflammatory response syndrome and cytokine storms: Research has re
Ischemia and hypoxia reperfusion injury: COVID-19 patients have various degrees of hypoxia, as more than 40% of cases required oxygen supplementation[30]. Complications such as aspiration and multiple organ failure can cause hypoxia, ischemia and subsequent shock. The suppression of cell survival signal transduction and hepatocyte death may be caused by ischemia and hypoxia, ATP depletion in hepatocytes, lipid accumulation, and glycogen consumption (Figure 5). Furthermore, the respiratory distress syndrome can cause oxidative stress that increases the production of reactive oxygen species (ROS). The ROS and lipid peroxidation products can induce redox-sensitive transcription factors and then release various pro-inflammatory factors leading to liver damage. These changes can exaggerate the ischemia of hepatocytes, influence the excretion of toxic metabolites and eventually stimulate liver injury. Hypoxia is also one of the main causes of liver injury in patients with serious COVID-19[161].
Drug hepatotoxicity: In China, the occurrence of drug-induced hepatic damage including, traditional Chinese patent medicines[162,163], antitumor drugs, antibiotics, antimalarial drugs, and anti-tuberculosis drugs[164,165], is second only to fatty liver disease and viral hepatitis (alcoholic and nonalcoholic). Several COVID-19 patients have a fever and consequently, use antipyretic and analgesic drugs. Therefore, a drug overdose can cause hepatic damage.
Recently, this was observed with abidol, lopinavir, ritonavir and other antiviral medications used to control COVID-19. A recent study published in JCI[166] demon
Due to the global spread of chronic liver disorders, correlations among patients presenting with hepatic illness and COVID-19 need to be examined. A preliminary analysis showed that 2%–11% of patients had hepatic disease and COVID-19[170]. In a study of 1099 COVID-19 patients, hepatitis B infection occurred in 23 (2.1%) patients. Serious cases of hepatitis B infection were more likely (2.4% vs 0.6%) than milder cases[39]. SARS patients with hepatitis B virus (HBV) or hepatitis C virus (HCV) infection were more vulnerable to serious hepatitis, likely because viral replication was in
Moreover, patients with liver cirrhosis may be more susceptible to infection due to their systemic disease[172], thus, preventing COVID-19 is extremely important. None of 111 patients with COVID-19 in Wuhan had decompensated cirrhosis due to preventive measures. In contrast to this, 17% of 101 patients with COVID-19 deve
COVID-19 in liver transplant recipients: The worldwide spread of COVID-19 raises additional challenges for organ transplants. Transplant recipients with preoperative decompensated organs and chronic illness tend to contract respiratory viruses. Liver transplant patients, who are exposed to more people, during their wait for trans
COVID-19 causes mainly transient and indirect liver injuries, which can be caused by hypoxia, systemic inflammatory reactions, and medication. Thus, hepatic damage should be treated by elimination of the basic etiology in the COVID-19 patients. Correction of hypoxia by oxygen supplementation or mechanical ventilation, renal replacement treatment for cytokine storm, and restoration of intravascular effectiveness can enhance liver injuries in the event of septic shock[181]. Also early identification and reduction in the dosage of drugs inducing hepatic damage is crucial. Hepatoprotective anti-inflammatory medicines including L-ornithine-L-aspartate can be used in extreme cases as an adjuvant treatment[182]. It is worth noting that therapeutic drugs may be hepatotoxic, especially in chronic liver disease (CLD) patients. Furthermore, patients treated with immunosuppressive agents should be closely monitored due to drug interactions. Recommendations for the management of CLD, AIH, and immunosuppressed patients during a pandemic are summarized in Tables 5, 6 and 7[183,184].
| Selected recommendations | |
| To limit nosocomial spread | (1) Decrease in-person visits via other alternatives such as virtual platforms |
| (2) Symptom investigation before entering hospitals to identify COVID-19 patients | |
| (3) Reduce staffing to essential staff only | |
| (4) Reduce the frequency of screening and laboratory examinations | |
| (5) Adhere to recommended PPE by HCW and patients | |
| (6) Maintain proper social distancing in hospitals | |
| (7) Postpone unnecessary or elective operations | |
| Management of CLD patients with COVID-19 | (1) These patients should be admitted to hospital early |
| (2) Prioritization of COVID-19 testing for patients with cirrhosis, CLD patients taking immunosuppressive agents and acute decompensated patients | |
| (3) Repeated LFTs are advisable | |
| (4) Early registration in clinical trials as much as possible | |
| (5) COVID-19 patients with NAFLD should be kept under supervision | |
| (6) Screening of hepatitis B surface antigen should be taken into consideration | |
| (7) Drug-induced liver injury should be monitored | |
| (8) These patients can receive 2-3 g/d of acetaminophen, while limiting the use of NSAIDs when possible | |
| (9) HBV prophylaxis should be considered before starting immunosuppressive agents | |
| (10) Stopping Remdesivir in decompensated liver disease patients with ALT more than 5 times the upper limit of normal | |
| Management of chronic viral hepatitis (HCV and HBV) | (1) Despite COVID-19 status, treatment continuity of chronic HCV and HBV is recommended |
| (2) In the absence of flare, HBV treatment should be stopped | |
| (3) For uninfected individuals, HCV and HBV treatment should be continued according to guidelines | |
| Management of HCC | (1) HCC treatment should be continued according to guidelines; however, it can be delayed if necessary |
| (2) In the case of COVID-19 patients, delaying elective transplants and resection surgery, and stopping immunotherapy are advisable | |
| (3) Early admission to hospital is recommended for HCC patients | |
| Management of pre- and post-transplant recipients | (1) Screening donor and recipient for COVID-19 is suggested |
| (2) For donors testing positive for COVID-19, transplantation surgery should be postponed | |
| (3) Prioritization of patients with short-term prognosis | |
| (4) For post-transplant patients, a reduction in immunosuppressive dose can be considered for moderate COVID-19 cases, while for mild COVID-19 cases, the dose should not be reduced | |
| (5) For post-transplant recipients, vaccination against pneumonia and influenza is advisable |
| Selected recommendations | |
| Virtual platforms are recommended to minimize in-person visits as much as possible | |
| COVID-19 testing is advised in cases of acute deterioration and liver failure | |
| Patients with low risk of complications (patients on chronic immunosuppressive therapy) | (1) Frequent patient-provider communications should be closely supervised; (2) Virtual platforms should be used to decrease contact; and (3) Ensure enough drug supply and refills to decrease running out of medications |
| Patients with moderate risk of complications (symptomatic disease without cirrhosis) | (1) Empiric therapy via virtual healthcare platforms as much as possible; and (2) Preventing liver biopsy whenever possible |
| Patients with high risk of complications (acute flare, decompensated cirrhosis) | (1) Reduce invasive procedures as much as possible; (2) In the case of COVID-19 patients, if lymphopenia develops, dose reduction of antimetabolites is recommended; and (3) In the case of infection, corticosteroids should be attenuated |
| Suggestions | |
| (1) Starting with corticosteroids and immunomodulators should proceed; and (2) Risk benefit ratio assessments should be carried out | |
| Patients on immunosuppressive treatment and not infected with COVID-19 | Decreases or adjustment of doses is not advisable |
| Patients infected with COVID-19 on immunosuppressive drugs | (1) Reduce corticosteroids dose after specialist physician (consider tapering to prevent adrenal insufficiency); and (2) Decreasing the doses of cyclosporine, mycophenolate, and azathioprine is recommended in severe COVID-19 (especially patients with lymphopenia) |
| Patients requiring initiation of immunosuppressive agents | Starting treatment is suggested in these patients regardless of COVID-19 status |
Although millions of people have remained at their houses to decrease the transmi
For example, the Chinese National Health Commission determined that approximately 3300 health workers were infected in early March[185]. It is well-known that endoscopic staff are at high risk of airborne droplet infection, conjunctiva contact and surface contact contamination[182,186]. Airway suction and other cough-induced procedures result in an increased risk of SARS-CoV-2 transmission[182]. It must be noted that the possible risk of exposure to feces removal is not confined to upper endoscopy[35].
A prospective study conducted by Johnston et al[187], revealed the quantification of unrecognized bacteria to endoscopists exposed to biological samples leading to infection transmission. In this investigation, 227 endoscopic face shields were examined for colony-forming units (CFUs), and 1-15 CFU, 91/227 (40%); 16-30 CFU, 6/227 (2.6%); 30 or more CFU, 6/227 (2.6%) significantly increased after endoscopy. Similarly, in 1999, a research study carried out by Mohandas and Gopalakrishnan[188] in a tertiary care hospital in India of 786 endoscopies (149 lower and 162 therapeutic endoscopies) concluded that the splash rate to the skin of the forearms, feet, and face was 9.5%, while the splash rate to the eyes was 4.1%.
The duodenoscope is the most complex medical equipment that undergoes dis
In COVID-19 patients single-use duodeno-scopes may be important. However, they are not accessible worldwide and have cost-related constraints[202]. Several societies have advocated the use of room negative pressure, particularly for COVID-19 patients, or if endoscopy is urgently needed[203]. Intraprocedural changes such as minimal verbal communication, avoiding procedures in patients with inadequate bowel preparation, and avoiding spillage of GI contents via the biopsy channel should be introduced[204]. A previous study reported the use of a “double gauze technique”; one for the endoscopists and the other for technicians in a controlled fashion to prevent the “whip” effect of accessories and spillage of GIT secretions[204] Institutional requirements for minimum staff involved in the procedure have been developed[205]. Thus, the risk of exposure among endoscopy personnel is diminished. Endo
Although there is a theoretical risk of fecal-oral transmission of COVID-19, the actual risk of transmission is extremely low. Despite this risk, endoscopy units have been functioning and reopening with no significant outbreaks noted.
COVID-19 can lead to lead to acute respiratory infection, and a high rate of morbidity and mortality. The main signs of COVID-19 are respiratory system reactions, and gastro-intestinal symptoms are also very common. COVID-19 patients with GIT symptoms are more probably associated with severe complications such as ARDS and liver damage, with a poor prognosis. Hence, during diagnosis and treatment of the disease, GIT symptoms should be taken into consideration as well as virus transmi
Manuscript source: Invited manuscript
Specialty type: Gastroenterology and hepatology
Country/Territory of origin: Egypt
Peer-review report’s scientific quality classification
Grade A (Excellent): A, A
Grade B (Very good): B, B
Grade C (Good): C, C, C
Grade D (Fair): 0
Grade E (Poor): 0
P-Reviewer: Perisetti A, Song B, Vij M, Watanabe T S-Editor: Fan JR L-Editor: Webster JR P-Editor: Yuan YY
| 1. | Raoult D, Zumla A, Locatelli F, Ippolito G, Kroemer G. Coronavirus infections: Epidemiological, clinical and immunological features and hypotheses. Cell Stress. 2020;4:66-75. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 199] [Cited by in RCA: 218] [Article Influence: 43.6] [Reference Citation Analysis (0)] |
| 2. | de Wilde AH, Snijder EJ, Kikkert M, van Hemert MJ. Host Factors in Coronavirus Replication. Curr Top Microbiol Immunol. 2018;419:1-42. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 160] [Cited by in RCA: 246] [Article Influence: 41.0] [Reference Citation Analysis (0)] |
| 3. | European Centre for Disease Prevention and Control (ECDC). Risk Assessment Guidelines for Infectious Diseases Transmitted on Aircraft (RAGIDA) Middle East Respiratory Syndrome Coronavirus (MERSCoV). Stockholm: ECDC; 2020. [Cited 1 January 2021]. Available from: www.hpsc.ie/A-Z/EmergencyPlanning/PortHealth/File,4661,en.pdf. |
| 4. | Zhu N, Zhang D, Wang W, Li X, Yang B, Song J, Zhao X, Huang B, Shi W, Lu R, Niu P, Zhan F, Ma X, Wang D, Xu W, Wu G, Gao GF, Tan W; China Novel Coronavirus Investigating and Research Team. A Novel Coronavirus from Patients with Pneumonia in China, 2019. N Engl J Med. 2020;382:727-733. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 18987] [Cited by in RCA: 17646] [Article Influence: 3529.2] [Reference Citation Analysis (0)] |
| 5. | Wu YC, Chen CS, Chan YJ. The outbreak of COVID-19: An overview. J Chin Med Assoc. 2020;83:217-220. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 878] [Cited by in RCA: 720] [Article Influence: 144.0] [Reference Citation Analysis (0)] |
| 6. | Worldometers. info. COVID-19 CORONAVIRUS PANDEMIC. 2021. [Cited 1 January 2021]. Available from: https://www.worldometers.info/coronavirus/#countries. |
| 7. | Johnson KD, Harris C, Cain JK, Hummer C, Goyal H, Perisetti A. Pulmonary and Extra-Pulmonary Clinical Manifestations of COVID-19. Front Med (Lausanne). 2020;7:526. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 112] [Cited by in RCA: 117] [Article Influence: 23.4] [Reference Citation Analysis (0)] |
| 8. | Lauer SA, Grantz KH, Bi Q, Jones FK, Zheng Q, Meredith HR, Azman AS, Reich NG, Lessler J. The Incubation Period of Coronavirus Disease 2019 (COVID-19) From Publicly Reported Confirmed Cases: Estimation and Application. Ann Intern Med. 2020;172:577-582. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 4286] [Cited by in RCA: 3370] [Article Influence: 674.0] [Reference Citation Analysis (1)] |
| 9. | Li Q, Guan X, Wu P, Wang X, Zhou L, Tong Y, Ren R, Leung KSM, Lau EHY, Wong JY, Xing X, Xiang N, Wu Y, Li C, Chen Q, Li D, Liu T, Zhao J, Liu M, Tu W, Chen C, Jin L, Yang R, Wang Q, Zhou S, Wang R, Liu H, Luo Y, Liu Y, Shao G, Li H, Tao Z, Yang Y, Deng Z, Liu B, Ma Z, Zhang Y, Shi G, Lam TTY, Wu JT, Gao GF, Cowling BJ, Yang B, Leung GM, Feng Z. Early Transmission Dynamics in Wuhan, China, of Novel Coronavirus-Infected Pneumonia. N Engl J Med. 2020;382:1199-1207. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 11224] [Cited by in RCA: 9319] [Article Influence: 1863.8] [Reference Citation Analysis (0)] |
| 10. | Wu Z, McGoogan JM. Characteristics of and Important Lessons From the Coronavirus Disease 2019 (COVID-19) Outbreak in China: Summary of a Report of 72 314 Cases From the Chinese Center for Disease Control and Prevention. JAMA. 2020;323:1239-1242. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 11409] [Cited by in RCA: 11506] [Article Influence: 2301.2] [Reference Citation Analysis (0)] |
| 11. | Scientific Research. Report of the WHO-China Joint Mission on Coronavirus Disease 2019 (COVID-19). Geneva, Switzerland: WHO; 2020. [cited 1 January 2021]. Available from: https://www.scirp.org/reference/referencespapers.aspx?referenceid=2725513. |
| 12. | Wang D, Hu B, Hu C, Zhu F, Liu X, Zhang J, Wang B, Xiang H, Cheng Z, Xiong Y, Zhao Y, Li Y, Wang X, Peng Z. Clinical Characteristics of 138 Hospitalized Patients With 2019 Novel Coronavirus-Infected Pneumonia in Wuhan, China. JAMA. 2020;323:1061-1069. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 14113] [Cited by in RCA: 14767] [Article Influence: 2953.4] [Reference Citation Analysis (0)] |
| 13. | Jaillon S, Berthenet K, Garlanda C. Sexual Dimorphism in Innate Immunity. Clin Rev Allergy Immunol. 2019;56:308-321. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 280] [Cited by in RCA: 380] [Article Influence: 63.3] [Reference Citation Analysis (0)] |
| 14. | Marriott I, Huet-Hudson YM. Sexual dimorphism in innate immune responses to infectious organisms. Immunol Res. 2006;34:177-192. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 187] [Cited by in RCA: 199] [Article Influence: 10.5] [Reference Citation Analysis (0)] |
| 15. | Schröder J, Kahlke V, Staubach KH, Zabel P, Stüber F. Gender differences in human sepsis. Arch Surg. 1998;133:1200-1205. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 433] [Cited by in RCA: 437] [Article Influence: 16.2] [Reference Citation Analysis (0)] |
| 16. | García-Durán M, de Frutos T, Díaz-Recasens J, García-Gálvez G, Jiménez A, Montón M, Farré J, Sánchez de Miguel L, González-Fernández F, Arriero MD, Rico L, García R, Casado S, López-Farré A. Estrogen stimulates neuronal nitric oxide synthase protein expression in human neutrophils. Circ Res. 1999;85:1020-1026. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 75] [Cited by in RCA: 76] [Article Influence: 2.9] [Reference Citation Analysis (0)] |
| 17. | Oertelt-Prigione S. The influence of sex and gender on the immune response. Autoimmun Rev. 2012;11:A479-A485. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 280] [Cited by in RCA: 322] [Article Influence: 23.0] [Reference Citation Analysis (0)] |
| 18. | Klein SL. Sex influences immune responses to viruses, and efficacy of prophylaxis and treatments for viral diseases. Bioessays. 2012;34:1050-1059. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 168] [Cited by in RCA: 205] [Article Influence: 15.8] [Reference Citation Analysis (0)] |
| 19. | Scully EP, Haverfield J, Ursin RL, Tannenbaum C, Klein SL. Considering how biological sex impacts immune responses and COVID-19 outcomes. Nat Rev Immunol. 2020;20:442-447. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 466] [Cited by in RCA: 615] [Article Influence: 123.0] [Reference Citation Analysis (0)] |
| 20. | Liu J, Ji H, Zheng W, Wu X, Zhu JJ, Arnold AP, Sandberg K. Sex differences in renal angiotensin converting enzyme 2 (ACE2) activity are 17β-oestradiol-dependent and sex chromosome-independent. Biol Sex Differ. 2010;1:6. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 181] [Cited by in RCA: 201] [Article Influence: 13.4] [Reference Citation Analysis (0)] |
| 21. | Sampson AK, Moritz KM, Denton KM. Postnatal ontogeny of angiotensin receptors and ACE2 in male and female rats. Gend Med. 2012;9:21-32. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 55] [Cited by in RCA: 57] [Article Influence: 4.4] [Reference Citation Analysis (0)] |
| 22. | Hussein J. COVID-19: What implications for sexual and reproductive health and rights globally? Sex Reprod Health Matters. 2020;28:1746065. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 108] [Cited by in RCA: 99] [Article Influence: 19.8] [Reference Citation Analysis (0)] |
| 23. | Brosnihan KB, Neves LA, Joyner J, Averill DB, Chappell MC, Sarao R, Penninger J, Ferrario CM. Enhanced renal immunocytochemical expression of ANG-(1-7) and ACE2 during pregnancy. Hypertension. 2003;42:749-753. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 102] [Cited by in RCA: 100] [Article Influence: 4.5] [Reference Citation Analysis (0)] |
| 24. | Garg S, Kim L, Whitaker M, O'Halloran A, Cummings C, Holstein R, Prill M, Chai SJ, Kirley PD, Alden NB, Kawasaki B, Yousey-Hindes K, Niccolai L, Anderson EJ, Openo KP, Weigel A, Monroe ML, Ryan P, Henderson J, Kim S, Como-Sabetti K, Lynfield R, Sosin D, Torres S, Muse A, Bennett NM, Billing L, Sutton M, West N, Schaffner W, Talbot HK, Aquino C, George A, Budd A, Brammer L, Langley G, Hall AJ, Fry A. Hospitalization Rates and Characteristics of Patients Hospitalized with Laboratory-Confirmed Coronavirus Disease 2019 - COVID-NET, 14 States, March 1-30, 2020. MMWR Morb Mortal Wkly Rep. 2020;69:458-464. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 1712] [Cited by in RCA: 1699] [Article Influence: 339.8] [Reference Citation Analysis (0)] |
| 25. | Huang C, Wang Y, Li X, Ren L, Zhao J, Hu Y, Zhang L, Fan G, Xu J, Gu X, Cheng Z, Yu T, Xia J, Wei Y, Wu W, Xie X, Yin W, Li H, Liu M, Xiao Y, Gao H, Guo L, Xie J, Wang G, Jiang R, Gao Z, Jin Q, Wang J, Cao B. Clinical features of patients infected with 2019 novel coronavirus in Wuhan, China. Lancet. 2020;395:497-506. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 35178] [Cited by in RCA: 30116] [Article Influence: 6023.2] [Reference Citation Analysis (3)] |
| 26. | Chen N, Zhou M, Dong X, Qu J, Gong F, Han Y, Qiu Y, Wang J, Liu Y, Wei Y, Xia J, Yu T, Zhang X, Zhang L. Epidemiological and clinical characteristics of 99 cases of 2019 novel coronavirus pneumonia in Wuhan, China: a descriptive study. Lancet. 2020;395:507-513. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 14869] [Cited by in RCA: 12976] [Article Influence: 2595.2] [Reference Citation Analysis (1)] |
| 27. | Xu XW, Wu XX, Jiang XG, Xu KJ, Ying LJ, Ma CL, Li SB, Wang HY, Zhang S, Gao HN, Sheng JF, Cai HL, Qiu YQ, Li LJ. Clinical findings in a group of patients infected with the 2019 novel coronavirus (SARS-Cov-2) outside of Wuhan, China: retrospective case series. BMJ. 2020;368:m606. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 1298] [Cited by in RCA: 1263] [Article Influence: 252.6] [Reference Citation Analysis (0)] |
| 28. | Wu J, Liu J, Zhao X, Liu C, Wang W, Wang D, Xu W, Zhang C, Yu J, Jiang B, Cao H, Li L. Clinical Characteristics of Imported Cases of Coronavirus Disease 2019 (COVID-19) in Jiangsu Province: A Multicenter Descriptive Study. Clin Infect Dis. 2020;71:706-712. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 393] [Cited by in RCA: 413] [Article Influence: 82.6] [Reference Citation Analysis (1)] |
| 29. | Shi H, Han X, Jiang N, Cao Y, Alwalid O, Gu J, Fan Y, Zheng C. Radiological findings from 81 patients with COVID-19 pneumonia in Wuhan, China: a descriptive study. Lancet Infect Dis. 2020;20:425-434. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 2493] [Cited by in RCA: 2310] [Article Influence: 462.0] [Reference Citation Analysis (0)] |
| 30. | Yang X, Yu Y, Xu J, Shu H, Xia J, Liu H, Wu Y, Zhang L, Yu Z, Fang M, Yu T, Wang Y, Pan S, Zou X, Yuan S, Shang Y. Clinical course and outcomes of critically ill patients with SARS-CoV-2 pneumonia in Wuhan, China: a single-centered, retrospective, observational study. Lancet Respir Med. 2020;8:475-481. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 6231] [Cited by in RCA: 6659] [Article Influence: 1331.8] [Reference Citation Analysis (0)] |
| 31. | Mo P, Xing Y, Xiao Y, Deng L, Zhao Q, Wang H, Xiong Y, Cheng Z, Gao S, Liang K, Luo M, Chen T, Song S, Ma Z, Chen X, Zheng R, Cao Q, Wang F, Zhang Y. Clinical characteristics of refractory COVID-19 pneumonia in Wuhan, China. Clin Infect Dis. 2020;. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 486] [Cited by in RCA: 601] [Article Influence: 120.2] [Reference Citation Analysis (0)] |
| 32. | Zhou F, Yu T, Du R, Fan G, Liu Y, Liu Z, Xiang J, Wang Y, Song B, Gu X, Guan L, Wei Y, Li H, Wu X, Xu J, Tu S, Zhang Y, Chen H, Cao B. Clinical course and risk factors for mortality of adult inpatients with COVID-19 in Wuhan, China: a retrospective cohort study. Lancet. 2020;395:1054-1062. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 17476] [Cited by in RCA: 18199] [Article Influence: 3639.8] [Reference Citation Analysis (0)] |
| 33. | Lee IC, Huo TI, Huang YH. Gastrointestinal and liver manifestations in patients with COVID-19. J Chin Med Assoc. 2020;83:521-523. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 144] [Cited by in RCA: 129] [Article Influence: 25.8] [Reference Citation Analysis (2)] |
| 34. | Sultan S, Altayar O, Siddique SM, Davitkov P, Feuerstein JD, Lim JK, Falck-Ytter Y, El-Serag HB; AGA Institute. AGA Institute Rapid Review of the Gastrointestinal and Liver Manifestations of COVID-19, Meta-Analysis of International Data, and Recommendations for the Consultative Management of Patients with COVID-19. Gastroenterology. 2020;159:320-334.e27. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 309] [Cited by in RCA: 293] [Article Influence: 58.6] [Reference Citation Analysis (1)] |
| 35. | Gu J, Han B, Wang J. COVID-19: Gastrointestinal Manifestations and Potential Fecal-Oral Transmission. Gastroenterology. 2020;158:1518-1519. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 954] [Cited by in RCA: 949] [Article Influence: 189.8] [Reference Citation Analysis (1)] |
| 36. | Ruan Q, Yang K, Wang W, Jiang L, Song J. Clinical predictors of mortality due to COVID-19 based on an analysis of data of 150 patients from Wuhan, China. Intensive Care Med. 2020;46:846-848. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 2604] [Cited by in RCA: 3193] [Article Influence: 638.6] [Reference Citation Analysis (0)] |
| 37. | Liang W, Feng Z, Rao S, Xiao C, Xue X, Lin Z, Zhang Q, Qi W. Diarrhoea may be underestimated: a missing link in 2019 novel coronavirus. Gut. 2020;69:1141-1143. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 250] [Cited by in RCA: 269] [Article Influence: 53.8] [Reference Citation Analysis (0)] |
| 38. | Pan L, Mu M, Yang P, Sun Y, Wang R, Yan J, Li P, Hu B, Wang J, Hu C, Jin Y, Niu X, Ping R, Du Y, Li T, Xu G, Hu Q, Tu L. Clinical Characteristics of COVID-19 Patients With Digestive Symptoms in Hubei, China: A Descriptive, Cross-Sectional, Multicenter Study. Am J Gastroenterol. 2020;115:766-773. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 1160] [Cited by in RCA: 1205] [Article Influence: 241.0] [Reference Citation Analysis (0)] |
| 39. | Guan WJ, Ni ZY, Hu Y, Liang WH, Ou CQ, He JX, Liu L, Shan H, Lei CL, Hui DSC, Du B, Li LJ, Zeng G, Yuen KY, Chen RC, Tang CL, Wang T, Chen PY, Xiang J, Li SY, Wang JL, Liang ZJ, Peng YX, Wei L, Liu Y, Hu YH, Peng P, Wang JM, Liu JY, Chen Z, Li G, Zheng ZJ, Qiu SQ, Luo J, Ye CJ, Zhu SY, Zhong NS; China Medical Treatment Expert Group for Covid-19. Clinical Characteristics of Coronavirus Disease 2019 in China. N Engl J Med. 2020;382:1708-1720. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 19202] [Cited by in RCA: 18877] [Article Influence: 3775.4] [Reference Citation Analysis (7)] |
| 40. | Zhang H, Liao YS, Gong J, Liu J, Xia X, Zhang H. Clinical characteristics of coronavirus disease (COVID-19) patients with gastrointestinal symptoms: A report of 164 cases. Dig Liver Dis. 2020;52:1076-1079. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 44] [Cited by in RCA: 38] [Article Influence: 7.6] [Reference Citation Analysis (3)] |
| 41. | Luo S, Zhang X, Xu H. Don't Overlook Digestive Symptoms in Patients With 2019 Novel Coronavirus Disease (COVID-19). Clin Gastroenterol Hepatol. 2020;18:1636-1637. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 234] [Cited by in RCA: 233] [Article Influence: 46.6] [Reference Citation Analysis (0)] |
| 42. | Jin X, Lian JS, Hu JH, Gao J, Zheng L, Zhang YM, Hao SR, Jia HY, Cai H, Zhang XL, Yu GD, Xu KJ, Wang XY, Gu JQ, Zhang SY, Ye CY, Jin CL, Lu YF, Yu X, Yu XP, Huang JR, Xu KL, Ni Q, Yu CB, Zhu B, Li YT, Liu J, Zhao H, Zhang X, Yu L, Guo YZ, Su JW, Tao JJ, Lang GJ, Wu XX, Wu WR, Qv TT, Xiang DR, Yi P, Shi D, Chen Y, Ren Y, Qiu YQ, Li LJ, Sheng J, Yang Y. Epidemiological, clinical and virological characteristics of 74 cases of coronavirus-infected disease 2019 (COVID-19) with gastrointestinal symptoms. Gut. 2020;69:1002-1009. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 944] [Cited by in RCA: 870] [Article Influence: 174.0] [Reference Citation Analysis (0)] |
| 43. | Ferm S, Fisher C, Pakala T, Tong M, Shah D, Schwarzbaum D, Cooley V, Hussain S, Kim SH. Analysis of Gastrointestinal and Hepatic Manifestations of SARS-CoV-2 Infection in 892 Patients in Queens, NY. Clin Gastroenterol Hepatol. 2020;18:2378-2379.e1. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 46] [Cited by in RCA: 60] [Article Influence: 12.0] [Reference Citation Analysis (0)] |
| 44. | Hajifathalian K, Krisko T, Mehta A, Kumar S, Schwartz R, Fortune B, Sharaiha RZ; WCM-GI research group*. Gastrointestinal and Hepatic Manifestations of 2019 Novel Coronavirus Disease in a Large Cohort of Infected Patients From New York: Clinical Implications. Gastroenterology. 2020;159:1137-1140.e2. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 99] [Cited by in RCA: 107] [Article Influence: 21.4] [Reference Citation Analysis (0)] |
| 45. | Cheung KS, Hung IFN, Chan PPY, Lung KC, Tso E, Liu R, Ng YY, Chu MY, Chung TWH, Tam AR, Yip CCY, Leung KH, Fung AY, Zhang RR, Lin Y, Cheng HM, Zhang AJX, To KKW, Chan KH, Yuen KY, Leung WK. Gastrointestinal Manifestations of SARS-CoV-2 Infection and Virus Load in Fecal Samples From a Hong Kong Cohort: Systematic Review and Meta-analysis. Gastroenterology. 2020;159:81-95. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 1113] [Cited by in RCA: 1132] [Article Influence: 226.4] [Reference Citation Analysis (1)] |
| 46. | Goyal H, Sachdeva S, Perisetti A, Mann R, Inamdar S, Tharian B. Hyperlipasemia and Potential Pancreatic Injury Patterns in COVID-19: A Marker of Severity or Innocent Bystander? Gastroenterology. 2021;160:946-948.e2. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 29] [Cited by in RCA: 25] [Article Influence: 6.3] [Reference Citation Analysis (0)] |
| 47. | Gadiparthi C, Perisetti A, Sayana H, Tharian B, Inamdar S, Korman A. Gastrointestinal Bleeding in Patients with Severe SARS-CoV-2. Am J Gastroenterol. 2020;115:1283-1285. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 42] [Cited by in RCA: 39] [Article Influence: 7.8] [Reference Citation Analysis (0)] |
| 48. | Tian Y, Rong L, Nian W, He Y. Review article: gastrointestinal features in COVID-19 and the possibility of faecal transmission. Aliment Pharmacol Ther. 2020;51:843-851. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 594] [Cited by in RCA: 583] [Article Influence: 116.6] [Reference Citation Analysis (0)] |
| 49. | Xiao F, Tang M, Zheng X, Liu Y, Li X, Shan H. Evidence for Gastrointestinal Infection of SARS-CoV-2. Gastroenterology. 2020;158:1831-1833.e3. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 1985] [Cited by in RCA: 1995] [Article Influence: 399.0] [Reference Citation Analysis (1)] |
| 50. | Lui K, Wilson MP, Low G. Abdominal imaging findings in patients with SARS-CoV-2 infection: a scoping review. Abdom Radiol (NY). 2021;46:1249-1255. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 33] [Cited by in RCA: 38] [Article Influence: 9.5] [Reference Citation Analysis (0)] |
| 51. | Li F. Structure, Function, and Evolution of Coronavirus Spike Proteins. Annu Rev Virol. 2016;3:237-261. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 2002] [Cited by in RCA: 1840] [Article Influence: 204.4] [Reference Citation Analysis (0)] |
| 52. | Kuhn JH, Li W, Choe H, Farzan M. Angiotensin-converting enzyme 2: a functional receptor for SARS coronavirus. Cell Mol Life Sci. 2004;61:2738-2743. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 168] [Cited by in RCA: 184] [Article Influence: 8.8] [Reference Citation Analysis (0)] |
| 53. | Raj VS, Mou H, Smits SL, Dekkers DH, Müller MA, Dijkman R, Muth D, Demmers JA, Zaki A, Fouchier RA, Thiel V, Drosten C, Rottier PJ, Osterhaus AD, Bosch BJ, Haagmans BL. Dipeptidyl peptidase 4 is a functional receptor for the emerging human coronavirus-EMC. Nature. 2013;495:251-254. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 1418] [Cited by in RCA: 1591] [Article Influence: 132.6] [Reference Citation Analysis (0)] |
| 54. | Wan Y, Shang J, Graham R, Baric RS, Li F. Receptor Recognition by the Novel Coronavirus from Wuhan: an Analysis Based on Decade-Long Structural Studies of SARS Coronavirus. J Virol. 2020;94. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 3085] [Cited by in RCA: 2915] [Article Influence: 583.0] [Reference Citation Analysis (0)] |
| 55. | Zhou P, Yang XL, Wang XG, Hu B, Zhang L, Zhang W, Si HR, Zhu Y, Li B, Huang CL, Chen HD, Chen J, Luo Y, Guo H, Jiang RD, Liu MQ, Chen Y, Shen XR, Wang X, Zheng XS, Zhao K, Chen QJ, Deng F, Liu LL, Yan B, Zhan FX, Wang YY, Xiao GF, Shi ZL. A pneumonia outbreak associated with a new coronavirus of probable bat origin. Nature. 2020;579:270-273. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 15248] [Cited by in RCA: 14131] [Article Influence: 2826.2] [Reference Citation Analysis (1)] |
| 56. | Hamming I, Timens W, Bulthuis ML, Lely AT, Navis G, van Goor H. Tissue distribution of ACE2 protein, the functional receptor for SARS coronavirus. A first step in understanding SARS pathogenesis. J Pathol. 2004;203:631-637. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 3643] [Cited by in RCA: 4149] [Article Influence: 197.6] [Reference Citation Analysis (0)] |
| 57. | Zou X, Chen K, Zou J, Han P, Hao J, Han Z. Single-cell RNA-seq data analysis on the receptor ACE2 expression reveals the potential risk of different human organs vulnerable to 2019-nCoV infection. Front Med. 2020;14:185-192. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 1286] [Cited by in RCA: 1530] [Article Influence: 306.0] [Reference Citation Analysis (0)] |
| 58. | Crackower MA, Sarao R, Oudit GY, Yagil C, Kozieradzki I, Scanga SE, Oliveira-dos-Santos AJ, da Costa J, Zhang L, Pei Y, Scholey J, Ferrario CM, Manoukian AS, Chappell MC, Backx PH, Yagil Y, Penninger JM. Angiotensin-converting enzyme 2 is an essential regulator of heart function. Nature. 2002;417:822-828. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 1324] [Cited by in RCA: 1331] [Article Influence: 57.9] [Reference Citation Analysis (0)] |
| 59. | Kuba K, Imai Y, Rao S, Gao H, Guo F, Guan B, Huan Y, Yang P, Zhang Y, Deng W, Bao L, Zhang B, Liu G, Wang Z, Chappell M, Liu Y, Zheng D, Leibbrandt A, Wada T, Slutsky AS, Liu D, Qin C, Jiang C, Penninger JM. A crucial role of angiotensin converting enzyme 2 (ACE2) in SARS coronavirus-induced lung injury. Nat Med. 2005;11:875-879. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 2710] [Cited by in RCA: 2645] [Article Influence: 132.3] [Reference Citation Analysis (0)] |
| 60. | Hashimoto T, Perlot T, Rehman A, Trichereau J, Ishiguro H, Paolino M, Sigl V, Hanada T, Hanada R, Lipinski S, Wild B, Camargo SM, Singer D, Richter A, Kuba K, Fukamizu A, Schreiber S, Clevers H, Verrey F, Rosenstiel P, Penninger JM. ACE2 links amino acid malnutrition to microbial ecology and intestinal inflammation. Nature. 2012;487:477-481. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 787] [Cited by in RCA: 990] [Article Influence: 76.2] [Reference Citation Analysis (0)] |
| 61. | Imai Y, Kuba K, Rao S, Huan Y, Guo F, Guan B, Yang P, Sarao R, Wada T, Leong-Poi H, Crackower MA, Fukamizu A, Hui CC, Hein L, Uhlig S, Slutsky AS, Jiang C, Penninger JM. Angiotensin-converting enzyme 2 protects from severe acute lung failure. Nature. 2005;436:112-116. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 1791] [Cited by in RCA: 2000] [Article Influence: 100.0] [Reference Citation Analysis (0)] |
| 62. | Fu L, Wang B, Yuan T, Chen X, Ao Y, Fitzpatrick T, Li P, Zhou Y, Lin YF, Duan Q, Luo G, Fan S, Lu Y, Feng A, Zhan Y, Liang B, Cai W, Zhang L, Du X, Li L, Shu Y, Zou H. Clinical characteristics of coronavirus disease 2019 (COVID-19) in China: A systematic review and meta-analysis. J Infect. 2020;80:656-665. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 709] [Cited by in RCA: 641] [Article Influence: 128.2] [Reference Citation Analysis (0)] |
| 63. | Shi JH, Wang YR, Li WB, Gang R, Liu X, Xu L, Luo QF. A single-center descriptive study of COVID-19 inpatients in Wuhan, analysis of digestive system performance and disease severity in 54 cases. Zhonghua Xiaohua Zazhi. 2020;40:167-170. [DOI] [Full Text] |
| 64. | Zhao Y, Zhong SP, Li F, Liu GQ, Wang XF, Liu ZJ. Analysis of clinical characteristics and risk factors of gastrointestinal symptoms of COVID-19 patients in Xinyang, Henan province. Zhonghua Xiaohua Zazhi. 2020;40:E011. [DOI] [Full Text] |
| 65. | Stratigos JD, Katsambas A. Pellagra: a still existing disease. Br J Dermatol. 1977;96:99-106. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 51] [Cited by in RCA: 40] [Article Influence: 0.8] [Reference Citation Analysis (0)] |
| 66. | Darby WJ, McNutt KW, Todhunter EN. Niacin. Nutr Rev. 1975;33:289-297. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 23] [Cited by in RCA: 15] [Article Influence: 0.3] [Reference Citation Analysis (0)] |
| 67. | Segal I, Ou Tim L, Demetriou A, Paterson A, Hale M, Lerios M. Rectal manifestations of pellagra. Int J Colorectal Dis. 1986;1:238-243. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 17] [Cited by in RCA: 16] [Article Influence: 0.4] [Reference Citation Analysis (0)] |
| 68. | He L, Ding Y, Zhang Q, Che X, He Y, Shen H, Wang H, Li Z, Zhao L, Geng J, Deng Y, Yang L, Li J, Cai J, Qiu L, Wen K, Xu X, Jiang S. Expression of elevated levels of pro-inflammatory cytokines in SARS-CoV-infected ACE2+ cells in SARS patients: relation to the acute lung injury and pathogenesis of SARS. J Pathol. 2006;210:288-297. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 330] [Cited by in RCA: 323] [Article Influence: 17.0] [Reference Citation Analysis (0)] |
| 69. | Cheung CY, Poon LL, Ng IH, Luk W, Sia SF, Wu MH, Chan KH, Yuen KY, Gordon S, Guan Y, Peiris JS. Cytokine responses in severe acute respiratory syndrome coronavirus-infected macrophages in vitro: possible relevance to pathogenesis. J Virol. 2005;79:7819-7826. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 314] [Cited by in RCA: 350] [Article Influence: 17.5] [Reference Citation Analysis (0)] |
| 70. | Lau SKP, Lau CCY, Chan KH, Li CPY, Chen H, Jin DY, Chan JFW, Woo PCY, Yuen KY. Delayed induction of proinflammatory cytokines and suppression of innate antiviral response by the novel Middle East respiratory syndrome coronavirus: implications for pathogenesis and treatment. J Gen Virol. 2013;94:2679-2690. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 272] [Cited by in RCA: 305] [Article Influence: 25.4] [Reference Citation Analysis (0)] |
| 71. | Law HK, Cheung CY, Ng HY, Sia SF, Chan YO, Luk W, Nicholls JM, Peiris JS, Lau YL. Chemokine up-regulation in SARS-coronavirus-infected, monocyte-derived human dendritic cells. Blood. 2005;106:2366-2374. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 372] [Cited by in RCA: 372] [Article Influence: 18.6] [Reference Citation Analysis (0)] |
| 72. | Gu J, Korteweg C. Pathology and pathogenesis of severe acute respiratory syndrome. Am J Pathol. 2007;170:1136-1147. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 416] [Cited by in RCA: 405] [Article Influence: 22.5] [Reference Citation Analysis (0)] |
| 73. | Cao X. COVID-19: immunopathology and its implications for therapy. Nat Rev Immunol. 2020;20:269-270. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 972] [Cited by in RCA: 1118] [Article Influence: 223.6] [Reference Citation Analysis (0)] |
| 74. | Lodigiani C, Iapichino G, Carenzo L, Cecconi M, Ferrazzi P, Sebastian T, Kucher N, Studt JD, Sacco C, Bertuzzi A, Sandri MT, Barco S; Humanitas COVID-19 Task Force. Venous and arterial thromboembolic complications in COVID-19 patients admitted to an academic hospital in Milan, Italy. Thromb Res. 2020;191:9-14. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 1298] [Cited by in RCA: 1535] [Article Influence: 307.0] [Reference Citation Analysis (0)] |
| 75. | Bhayana R, Som A, Li MD, Carey DE, Anderson MA, Blake MA, Catalano O, Gee MS, Hahn PF, Harisinghani M, Kilcoyne A, Lee SI, Mojtahed A, Pandharipande PV, Pierce TT, Rosman DA, Saini S, Samir AE, Simeone JF, Gervais DA, Velmahos G, Misdraji J, Kambadakone A. Abdominal Imaging Findings in COVID-19: Preliminary Observations. Radiology. 2020;297:E207-E215. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 181] [Cited by in RCA: 227] [Article Influence: 45.4] [Reference Citation Analysis (0)] |
| 76. | Parry AH, Wani AH, Yaseen M. Acute Mesenteric Ischemia in Severe Coronavirus-19 (COVID-19): Possible Mechanisms and Diagnostic Pathway. Acad Radiol. 2020;27:1190. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 60] [Cited by in RCA: 67] [Article Influence: 13.4] [Reference Citation Analysis (0)] |
| 77. | Panigada M, Bottino N, Tagliabue P, Grasselli G, Novembrino C, Chantarangkul V, Pesenti A, Peyvandi F, Tripodi A. Hypercoagulability of COVID-19 patients in intensive care unit: A report of thromboelastography findings and other parameters of hemostasis. J Thromb Haemost. 2020;18:1738-1742. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 926] [Cited by in RCA: 952] [Article Influence: 190.4] [Reference Citation Analysis (0)] |
| 78. | Pons S, Fodil S, Azoulay E, Zafrani L. The vascular endothelium: the cornerstone of organ dysfunction in severe SARS-CoV-2 infection. Crit Care. 2020;24:353. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 360] [Cited by in RCA: 348] [Article Influence: 69.6] [Reference Citation Analysis (0)] |
| 79. | Miesbach W, Makris M. COVID-19: Coagulopathy, Risk of Thrombosis, and the Rationale for Anticoagulation. Clin Appl Thromb Hemost. 2020;26:1076029620938149. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 259] [Cited by in RCA: 262] [Article Influence: 52.4] [Reference Citation Analysis (0)] |
| 80. | The Lancet Haematology. COVID-19 coagulopathy: an evolving story. Lancet Haematol. 2020;7:e425. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 74] [Cited by in RCA: 89] [Article Influence: 17.8] [Reference Citation Analysis (0)] |
| 81. | Galanis N, Stavraka C, Agathangelidis F, Petsatodis E, Giankoulof C, Givissis P. Coagulopathy in COVID-19 infection: a case of acute upper limb ischemia. J Surg Case Rep. 2020;2020:rjaa204. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 17] [Cited by in RCA: 18] [Article Influence: 3.6] [Reference Citation Analysis (0)] |
| 82. | Budden KF, Gellatly SL, Wood DL, Cooper MA, Morrison M, Hugenholtz P, Hansbro PM. Emerging pathogenic links between microbiota and the gut-lung axis. Nat Rev Microbiol. 2017;15:55-63. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 622] [Cited by in RCA: 1013] [Article Influence: 112.6] [Reference Citation Analysis (0)] |
| 83. | Domínguez-Díaz C, García-Orozco A, Riera-Leal A, Padilla-Arellano JR, Fafutis-Morris M. Microbiota and Its Role on Viral Evasion: Is It With Us or Against Us? Front Cell Infect Microbiol. 2019;9:256. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 48] [Cited by in RCA: 59] [Article Influence: 9.8] [Reference Citation Analysis (0)] |
| 84. | Wang J, Li F, Wei H, Lian ZX, Sun R, Tian Z. Respiratory influenza virus infection induces intestinal immune injury via microbiota-mediated Th17 cell-dependent inflammation. J Exp Med. 2014;211:2397-2410. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 256] [Cited by in RCA: 367] [Article Influence: 33.4] [Reference Citation Analysis (0)] |
| 85. | Papadakis KA, Prehn J, Nelson V, Cheng L, Binder SW, Ponath PD, Andrew DP, Targan SR. The role of thymus-expressed chemokine and its receptor CCR9 on lymphocytes in the regional specialization of the mucosal immune system. J Immunol. 2000;165:5069-5076. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 262] [Cited by in RCA: 252] [Article Influence: 10.1] [Reference Citation Analysis (0)] |
| 86. | Stenstad H, Ericsson A, Johansson-Lindbom B, Svensson M, Marsal J, Mack M, Picarella D, Soler D, Marquez G, Briskin M, Agace WW. Gut-associated lymphoid tissue-primed CD4+ T cells display CCR9-dependent and -independent homing to the small intestine. Blood. 2006;107:3447-3454. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 134] [Cited by in RCA: 143] [Article Influence: 7.5] [Reference Citation Analysis (0)] |
| 87. | Crowe CR, Chen K, Pociask DA, Alcorn JF, Krivich C, Enelow RI, Ross TM, Witztum JL, Kolls JK. Critical role of IL-17RA in immunopathology of influenza infection. J Immunol. 2009;183:5301-5310. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 261] [Cited by in RCA: 293] [Article Influence: 18.3] [Reference Citation Analysis (0)] |
| 88. | Zhang D, Li S, Wang N, Tan HY, Zhang Z, Feng Y. The Cross-Talk Between Gut Microbiota and Lungs in Common Lung Diseases. Front Microbiol. 2020;11:301. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 294] [Cited by in RCA: 272] [Article Influence: 54.4] [Reference Citation Analysis (0)] |
| 89. | Tripathi A, Debelius J, Brenner DA, Karin M, Loomba R, Schnabl B, Knight R. The gut-liver axis and the intersection with the microbiome. Nat Rev Gastroenterol Hepatol. 2018;15:397-411. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 872] [Cited by in RCA: 970] [Article Influence: 138.6] [Reference Citation Analysis (0)] |
| 90. | Hu Y, Lu GZ, Xu J. Discussion on symptoms of digestive tract caused by novel coronavirus infection. Zhonghua Chuanranbing Zazhi. 2020;38:E022-E022. [DOI] [Full Text] |
| 91. | Zhang FC, Yin ZB, Tang XP. Clinical analysis of 260 cases of SARS in Guangzhou. Zhonghua Chuanranbing Zazhi. 2003;21:84-88. |
| 92. | Leung WK, To KF, Chan PK, Chan HL, Wu AK, Lee N, Yuen KY, Sung JJ. Enteric involvement of severe acute respiratory syndrome-associated coronavirus infection. Gastroenterology. 2003;125:1011-1017. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 127] [Cited by in RCA: 295] [Article Influence: 13.4] [Reference Citation Analysis (1)] |
| 93. | Fang D, Ma JD, Guan JL, Wang MR, Song Y, Tian DA, Li PY. A single-center, descriptive study on the digestive system of COVID-19 inpatients in wuhan. Zhonghua Xiaohua Zazhi. 2020;40:E005-E005. |
| 94. | Chinese Society of Gastroenterology. Expert consensus on diagnosis and treatment of covid-19 digestive system. Zhongguo Yishi Zazhi. 2020;100. [DOI] [Full Text] |
| 95. | Kociolek LK, Gerding DN. Breakthroughs in the treatment and prevention of Clostridium difficile infection. Nat Rev Gastroenterol Hepatol. 2016;13:150-160. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 100] [Cited by in RCA: 112] [Article Influence: 12.4] [Reference Citation Analysis (0)] |
| 96. | Perisetti A, Gajendran M, Goyal H. Putative Mechanisms of Diarrhea in COVID-19. Clin Gastroenterol Hepatol. 2020;18:3054-3055. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 26] [Cited by in RCA: 28] [Article Influence: 5.6] [Reference Citation Analysis (0)] |
| 97. | Strand DS, Kim D, Peura DA. 25 Years of Proton Pump Inhibitors: A Comprehensive Review. Gut Liver. 2017;11:27-37. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 219] [Cited by in RCA: 401] [Article Influence: 50.1] [Reference Citation Analysis (0)] |
| 98. | Robinson M. Review article: pH, healing and symptom relief with rabeprazole treatment in acid-related disorders. Aliment Pharmacol Ther. 2004;20 Suppl 6:30-37. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 18] [Cited by in RCA: 17] [Article Influence: 0.8] [Reference Citation Analysis (0)] |
| 99. | Xiao X, Chakraborti S, Dimitrov AS, Gramatikoff K, Dimitrov DS. The SARS-CoV S glycoprotein: expression and functional characterization. Biochem Biophys Res Commun. 2003;312:1159-1164. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 283] [Cited by in RCA: 301] [Article Influence: 14.3] [Reference Citation Analysis (0)] |
| 100. | Darnell ME, Subbarao K, Feinstone SM, Taylor DR. Inactivation of the coronavirus that induces severe acute respiratory syndrome, SARS-CoV. J Virol Methods. 2004;121:85-91. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 476] [Cited by in RCA: 457] [Article Influence: 22.9] [Reference Citation Analysis (0)] |
| 101. | Chan KH, Sridhar S, Zhang RR, Chu H, Fung AY, Chan G, Chan JF, To KK, Hung IF, Cheng VC, Yuen KY. Factors affecting stability and infectivity of SARS-CoV-2. J Hosp Infect. 2020;106:226-231. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 139] [Cited by in RCA: 172] [Article Influence: 34.4] [Reference Citation Analysis (0)] |
| 102. | Zhou L, Niu Z, Jiang X, Zhang Z, Zheng Y, Wang Z, Zhu Y, Gao L, Wang X, Sun Q. Systemic analysis of tissue cells potentially vulnerable to SARS-CoV-2 infection by the protein-proofed single-cell RNA profiling of ACE2, TMPRSS2 and Furin proteases. 2020 Preprint. Available from: bioRxiv:2020.2004.2006.028522. [DOI] [Full Text] |
| 103. | Ramachandran P, Perisetti A, Gajendran M, Jean-Louis F, Bansal P, Dwivedi AK, Goyal H. Pre-hospitalization proton pump inhibitor use and clinical outcomes in COVID-19. Eur J Gastroenterol Hepatol. 2020;. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 26] [Cited by in RCA: 21] [Article Influence: 7.0] [Reference Citation Analysis (0)] |
| 104. | Yang J, Zheng Y, Gou X, Pu K, Chen Z, Guo Q, Ji R, Wang H, Wang Y, Zhou Y. Prevalence of comorbidities and its effects in patients infected with SARS-CoV-2: a systematic review and meta-analysis. Int J Infect Dis. 2020;94:91-95. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 2672] [Cited by in RCA: 2509] [Article Influence: 501.8] [Reference Citation Analysis (2)] |
| 105. | Mao R, Liang J, Shen J, Ghosh S, Zhu LR, Yang H, Wu KC, Chen MH; Chinese Society of IBD; Chinese Elite IBD Union; Chinese IBD Quality Care Evaluation Center Committee. Implications of COVID-19 for patients with pre-existing digestive diseases. Lancet Gastroenterol Hepatol. 2020;5:425-427. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 218] [Cited by in RCA: 241] [Article Influence: 48.2] [Reference Citation Analysis (0)] |
| 106. | An P, Ji M, Ren H, Su J, Kang J, Yin A, Zhou Q, Shen L, Zhao L, Jiang X, Xiao Y, Tan W, Lv X, Li J, Liu S, Zhou J, Chen H, Xu Y, Liu J, Ding Y. Protection of 318 Inflammatory Bowel Disease Patients from the Outbreak and Rapid Spread of COVID-19 Infection in Wuhan, China. SSRN Electronic J. 2020;. [RCA] [DOI] [Full Text] [Cited by in Crossref: 23] [Cited by in RCA: 23] [Reference Citation Analysis (0)] |
| 107. | Danese S, Cecconi M, Spinelli A. Management of IBD during the COVID-19 outbreak: resetting clinical priorities. Nat Rev Gastroenterol Hepatol. 2020;17:253-255. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 76] [Cited by in RCA: 90] [Article Influence: 18.0] [Reference Citation Analysis (0)] |
| 108. | British Society of Gastroenterology (BSG). British Society of Gastroenterology (BSG) advice for management of inflammatory bowel diseases during the COVID-19 pandemic. 2020. [cited 22 March 2020]. Available from: https://www.bsg.org.uk/covid-19-advice/bsg-advice-for-management-of-inflammatory-bowel-diseases-during-the-covid-19-pandemic/. |
| 109. | Lamb CA, Kennedy NA, Raine T, Hendy PA, Smith PJ, Limdi JK, Hayee B, Lomer MCE, Parkes GC, Selinger C, Barrett KJ, Davies RJ, Bennett C, Gittens S, Dunlop MG, Faiz O, Fraser A, Garrick V, Johnston PD, Parkes M, Sanderson J, Terry H; IBD guidelines eDelphi consensus group; Gaya DR, Iqbal TH, Taylor SA, Smith M, Brookes M, Hansen R, Hawthorne AB. British Society of Gastroenterology consensus guidelines on the management of inflammatory bowel disease in adults. Gut. 2019;68:s1-s106. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 1402] [Cited by in RCA: 1565] [Article Influence: 260.8] [Reference Citation Analysis (0)] |
| 110. | Rubin DT, Abreu MT, Rai V, Siegel CA; International Organization for the Study of Inflammatory Bowel Disease. Management of Patients With Crohn's Disease and Ulcerative Colitis During the Coronavirus Disease-2019 Pandemic: Results of an International Meeting. Gastroenterology. 2020;159:6-13.e6. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 144] [Cited by in RCA: 172] [Article Influence: 34.4] [Reference Citation Analysis (0)] |
| 111. | Queiroz NSF, Barros LL, Azevedo MFC, Oba J, Sobrado CW, Carlos AS, Milani LR, Sipahi AM, Damião AOMC. Management of inflammatory bowel disease patients in the COVID-19 pandemic era: a Brazilian tertiary referral center guidance. Clinics (Sao Paulo). 2020;75:e1909. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 22] [Cited by in RCA: 21] [Article Influence: 4.2] [Reference Citation Analysis (0)] |
| 112. | Lei S, Jiang F, Su W, Chen C, Chen J, Mei W, Zhan LY, Jia Y, Zhang L, Liu D, Xia ZY, Xia Z. Clinical characteristics and outcomes of patients undergoing surgeries during the incubation period of COVID-19 infection. EClinicalMedicine. 2020;21:100331. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 809] [Cited by in RCA: 882] [Article Influence: 176.4] [Reference Citation Analysis (1)] |
| 113. | SAGES. SAGES and EAES recommendations regarding surgical response to COVID-19 crisis. 2020. [cited 29 March 2020]. Available from: http://www.sages.org/recommendations-surgical-response-covid-19/. |
| 114. | CBCD. Posicionamento do CBCD quanto ao COVID-19. 2020. [cited 1 April 2020]. Available from: http://www.cbcd.org.br/cbcdnews/2020/posicionamento-do-cbcd-quantoao-covid-19/. |
| 115. | Holshue ML, DeBolt C, Lindquist S, Lofy KH, Wiesman J, Bruce H, Spitters C, Ericson K, Wilkerson S, Tural A, Diaz G, Cohn A, Fox L, Patel A, Gerber SI, Kim L, Tong S, Lu X, Lindstrom S, Pallansch MA, Weldon WC, Biggs HM, Uyeki TM, Pillai SK; Washington State 2019-nCoV Case Investigation Team. First Case of 2019 Novel Coronavirus in the United States. N Engl J Med. 2020;382:929-936. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 4155] [Cited by in RCA: 3822] [Article Influence: 764.4] [Reference Citation Analysis (1)] |
| 116. | Yeo C, Kaushal S, Yeo D. Enteric involvement of coronaviruses: is faecal-oral transmission of SARS-CoV-2 possible? Lancet Gastroenterol Hepatol. 2020;5:335-337. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 546] [Cited by in RCA: 554] [Article Influence: 110.8] [Reference Citation Analysis (0)] |
| 117. | Wang W, Xu Y, Gao R, Lu R, Han K, Wu G, Tan W. Detection of SARS-CoV-2 in Different Types of Clinical Specimens. JAMA. 2020;323:1843-1844. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 1894] [Cited by in RCA: 2658] [Article Influence: 531.6] [Reference Citation Analysis (0)] |
| 118. | Chen L, Lou J, Bai Y, Wang M. COVID-19 Disease With Positive Fecal and Negative Pharyngeal and Sputum Viral Tests. Am J Gastroenterol. 2020;115:790. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 97] [Cited by in RCA: 105] [Article Influence: 21.0] [Reference Citation Analysis (0)] |
| 119. | Wu Y, Guo C, Tang L, Hong Z, Zhou J, Dong X, Yin H, Xiao Q, Tang Y, Qu X, Kuang L, Fang X, Mishra N, Lu J, Shan H, Jiang G, Huang X. Prolonged presence of SARS-CoV-2 viral RNA in faecal samples. Lancet Gastroenterol Hepatol. 2020;5:434-435. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 1050] [Cited by in RCA: 1150] [Article Influence: 230.0] [Reference Citation Analysis (0)] |
| 120. | Li C, Liu P, Guo SS, Zhao ZG. Study on the mechanism and treatment of COVID-19, SARS and MERS with gastrointestinal symptoms. Zhonghua Xiaohua Zazhi. 2020;40. [DOI] [Full Text] |
| 121. | Wilkins T, Sequoia J. Probiotics for Gastrointestinal Conditions: A Summary of the Evidence. Am Fam Physician. 2017;96:170-178. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 5] [Cited by in RCA: 5] [Article Influence: 0.6] [Reference Citation Analysis (0)] |
| 122. | Diagnosis and Treatment Protocol for Novel Coronavirus Pneumonia (Trial Version 7). Chin Med J (Engl). 2020;133:1087-1095. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 374] [Cited by in RCA: 531] [Article Influence: 106.2] [Reference Citation Analysis (0)] |
| 123. | Chen Y, Guo Y, Pan Y, Zhao ZJ. Structure analysis of the receptor binding of 2019-nCoV. Biochem Biophys Res Commun. 2020;. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 546] [Cited by in RCA: 513] [Article Influence: 102.6] [Reference Citation Analysis (0)] |
| 124. | Damaso CR, Oliveira MF, Massarani SM, Moussatché N. Azathioprine inhibits vaccinia virus replication in both BSC-40 and RAG cell lines acting on different stages of virus cycle. Virology. 2002;300:79-91. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 11] [Cited by in RCA: 13] [Article Influence: 0.6] [Reference Citation Analysis (0)] |
| 125. | Hersberger L, Bargetzi L, Bargetzi A, Tribolet P, Fehr R, Baechli V, Geiser M, Deiss M, Gomes F, Kutz A, Kägi-Braun N, Hoess C, Pavlicek V, Schmid S, Bilz S, Sigrist S, Brändle M, Benz C, Henzen C, Nigg M, Thomann R, Brand C, Rutishauser J, Aujesky D, Rodondi N, Donzé J, Stanga Z, Mueller B, Schuetz P. Nutritional risk screening (NRS 2002) is a strong and modifiable predictor risk score for short-term and long-term clinical outcomes: secondary analysis of a prospective randomised trial. Clin Nutr. 2020;39:2720-2729. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 52] [Cited by in RCA: 141] [Article Influence: 23.5] [Reference Citation Analysis (0)] |
| 126. | Xu L, Liu J, Lu M, Yang D, Zheng X. Liver injury during highly pathogenic human coronavirus infections. Liver Int. 2020;40:998-1004. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 622] [Cited by in RCA: 575] [Article Influence: 115.0] [Reference Citation Analysis (0)] |
| 127. | Kukla M, Skonieczna-Żydecka K, Kotfis K, Maciejewska D, Łoniewski I, Lara LF, Pazgan-Simon M, Stachowska E, Kaczmarczyk M, Koulaouzidis A, Marlicz W. COVID-19, MERS and SARS with Concomitant Liver Injury-Systematic Review of the Existing Literature. J Clin Med. 2020;9. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 69] [Cited by in RCA: 78] [Article Influence: 15.6] [Reference Citation Analysis (0)] |
| 128. | Richardson S, Hirsch JS, Narasimhan M, Crawford JM, McGinn T, Davidson KW; the Northwell COVID-19 Research Consortium; Barnaby DP, Becker LB, Chelico JD, Cohen SL, Cookingham J, Coppa K, Diefenbach MA, Dominello AJ, Duer-Hefele J, Falzon L, Gitlin J, Hajizadeh N, Harvin TG, Hirschwerk DA, Kim EJ, Kozel ZM, Marrast LM, Mogavero JN, Osorio GA, Qiu M, Zanos TP. Presenting Characteristics, Comorbidities, and Outcomes Among 5700 Patients Hospitalized With COVID-19 in the New York City Area. JAMA. 2020;323:2052-2059. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 6024] [Cited by in RCA: 6518] [Article Influence: 1303.6] [Reference Citation Analysis (0)] |
| 129. | Cai Q, Huang D, Yu H, Zhu Z, Xia Z, Su Y, Li Z, Zhou G, Gou J, Qu J, Sun Y, Liu Y, He Q, Chen J, Liu L, Xu L. COVID-19: Abnormal liver function tests. J Hepatol. 2020;73:566-574. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 623] [Cited by in RCA: 661] [Article Influence: 132.2] [Reference Citation Analysis (0)] |
| 130. | Parohan M, Yaghoubi S, Seraji A. Liver injury is associated with severe coronavirus disease 2019 (COVID-19) infection: A systematic review and meta-analysis of retrospective studies. Hepatol Res. 2020;50:924-935. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 103] [Cited by in RCA: 88] [Article Influence: 17.6] [Reference Citation Analysis (0)] |
| 131. | Bloom PP, Meyerowitz EA, Reinus Z, Daidone M, Gustafson J, Kim AY, Schaefer E, Chung RT. Liver Biochemistries in Hospitalized Patients With COVID-19. Hepatology. 2021;73:890-900. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 113] [Cited by in RCA: 143] [Article Influence: 35.8] [Reference Citation Analysis (0)] |
| 132. | Lei F, Liu YM, Zhou F, Qin JJ, Zhang P, Zhu L, Zhang XJ, Cai J, Lin L, Ouyang S, Wang X, Yang C, Cheng X, Liu W, Li H, Xie J, Wu B, Luo H, Xiao F, Chen J, Tao L, Cheng G, She ZG, Zhou J, Wang H, Lin J, Luo P, Fu S, Ye P, Xiao B, Mao W, Liu L, Yan Y, Chen G, Huang X, Zhang BH, Yuan Y. Longitudinal Association Between Markers of Liver Injury and Mortality in COVID-19 in China. Hepatology. 2020;72:389-398. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 318] [Cited by in RCA: 309] [Article Influence: 61.8] [Reference Citation Analysis (0)] |
| 133. | Xie H, Zhao J, Lian N, Lin S, Xie Q, Zhuo H. Clinical characteristics of non-ICU hospitalized patients with coronavirus disease 2019 and liver injury: A retrospective study. Liver Int. 2020;40:1321-1326. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 198] [Cited by in RCA: 213] [Article Influence: 42.6] [Reference Citation Analysis (0)] |
| 134. | Fan Z, Chen L, Li J, Cheng X, Yang J, Tian C, Zhang Y, Huang S, Liu Z, Cheng J. Clinical Features of COVID-19-Related Liver Functional Abnormality. Clin Gastroenterol Hepatol. 2020;18:1561-1566. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 559] [Cited by in RCA: 556] [Article Influence: 111.2] [Reference Citation Analysis (0)] |
| 135. | Zhang B, Zhou X, Qiu Y, Feng F, Feng J, Jia Y, Zhu H, Hu K, Liu J, Liu Z, Wang S, Gong Y, Zhou C, Zhu T, Cheng Y, Deng H, Tao F, Ren Y, Cheng B, Gao L, Wu X, Yu L, Huang Z, Mao Z, Song Q, Zhu B, Wang J. Clinical characteristics of 82 death cases with COVID-19. 2020 Preprint. Available from: medRxiv:2020.2002.2026.20028191. [RCA] [DOI] [Full Text] [Cited by in Crossref: 73] [Cited by in RCA: 75] [Article Influence: 15.0] [Reference Citation Analysis (0)] |
| 136. | Gordon DE, Jang GM, Bouhaddou M, Xu J, Obernier K, White KM, O'Meara MJ, Rezelj VV, Guo JZ, Swaney DL, Tummino TA, Hüttenhain R, Kaake RM, Richards AL, Tutuncuoglu B, Foussard H, Batra J, Haas K, Modak M, Kim M, Haas P, Polacco BJ, Braberg H, Fabius JM, Eckhardt M, Soucheray M, Bennett MJ, Cakir M, McGregor MJ, Li Q, Meyer B, Roesch F, Vallet T, Mac Kain A, Miorin L, Moreno E, Naing ZZC, Zhou Y, Peng S, Shi Y, Zhang Z, Shen W, Kirby IT, Melnyk JE, Chorba JS, Lou K, Dai SA, Barrio-Hernandez I, Memon D, Hernandez-Armenta C, Lyu J, Mathy CJP, Perica T, Pilla KB, Ganesan SJ, Saltzberg DJ, Rakesh R, Liu X, Rosenthal SB, Calviello L, Venkataramanan S, Liboy-Lugo J, Lin Y, Huang XP, Liu Y, Wankowicz SA, Bohn M, Safari M, Ugur FS, Koh C, Savar NS, Tran QD, Shengjuler D, Fletcher SJ, O'Neal MC, Cai Y, Chang JCJ, Broadhurst DJ, Klippsten S, Sharp PP, Wenzell NA, Kuzuoglu-Ozturk D, Wang HY, Trenker R, Young JM, Cavero DA, Hiatt J, Roth TL, Rathore U, Subramanian A, Noack J, Hubert M, Stroud RM, Frankel AD, Rosenberg OS, Verba KA, Agard DA, Ott M, Emerman M, Jura N, von Zastrow M, Verdin E, Ashworth A, Schwartz O, d'Enfert C, Mukherjee S, Jacobson M, Malik HS, Fujimori DG, Ideker T, Craik CS, Floor SN, Fraser JS, Gross JD, Sali A, Roth BL, Ruggero D, Taunton J, Kortemme T, Beltrao P, Vignuzzi M, García-Sastre A, Shokat KM, Shoichet BK, Krogan NJ. A SARS-CoV-2 protein interaction map reveals targets for drug repurposing. Nature. 2020;583:459-468. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 3145] [Cited by in RCA: 3188] [Article Influence: 637.6] [Reference Citation Analysis (0)] |
| 137. | Wang Y, Liu S, Liu H, Li W, Lin F, Jiang L, Li X, Xu P, Zhang L, Zhao L, Cao Y, Kang J, Yang J, Li L, Liu X, Li Y, Nie R, Mu J, Lu F, Zhao S, Lu J, Zhao J. SARS-CoV-2 infection of the liver directly contributes to hepatic impairment in patients with COVID-19. J Hepatol. 2020;73:807-816. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 353] [Cited by in RCA: 457] [Article Influence: 91.4] [Reference Citation Analysis (0)] |
| 138. | Sarin SK, Choudhury A, Lau GK, Zheng MH, Ji D, Abd-Elsalam S, Hwang J, Qi X, Cua IH, Suh JI, Park JG, Putcharoen O, Kaewdech A, Piratvisuth T, Treeprasertsuk S, Park S, Wejnaruemarn S, Payawal DA, Baatarkhuu O, Ahn SH, Yeo CD, Alonzo UR, Chinbayar T, Loho IM, Yokosuka O, Jafri W, Tan S, Soo LI, Tanwandee T, Gani R, Anand L, Esmail ES, Khalaf M, Alam S, Lin CY, Chuang WL, Soin AS, Garg HK, Kalista K, Batsukh B, Purnomo HD, Dara VP, Rathi P, Al Mahtab M, Shukla A, Sharma MK, Omata M; APASL COVID Task Force; APASL COVID Liver Injury Spectrum Study (APCOLIS Study-NCT 04345640). Pre-existing liver disease is associated with poor outcome in patients with SARS CoV2 infection; The APCOLIS Study (APASL COVID-19 Liver Injury Spectrum Study). Hepatol Int. 2020;14:690-700. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 217] [Cited by in RCA: 216] [Article Influence: 43.2] [Reference Citation Analysis (1)] |
| 139. | Xu Z, Shi L, Wang Y, Zhang J, Huang L, Zhang C, Liu S, Zhao P, Liu H, Zhu L, Tai Y, Bai C, Gao T, Song J, Xia P, Dong J, Zhao J, Wang FS. Pathological findings of COVID-19 associated with acute respiratory distress syndrome. Lancet Respir Med. 2020;8:420-422. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 5228] [Cited by in RCA: 5782] [Article Influence: 1156.4] [Reference Citation Analysis (2)] |
| 140. | Liu Q, Wang RS, Qu GQ, Wang YY, Liu P, Zhu YZ, Fei G, Ren L, Zhou YW, Liu L. Gross examination report of a COVID-19 death autopsy. Fa Yi Xue Za Zhi. 2020;36:21-23. [RCA] [PubMed] [DOI] [Full Text] [Cited by in RCA: 129] [Reference Citation Analysis (0)] |
| 141. | Sonzogni A, Previtali G, Seghezzi M, Alessio MG, Gianatti A, Licini L, Zerbi P, Carsana L, Rossi R, Lauri E, Pellegrinelli A, Nebuloni M. Liver and COVID 19 Infection: A Very Preliminary Lesson Learnt from Histological Post-mortem Findings in 48 patients. 2020 Preprint. Available from: medRxiv:2020040438. [DOI] [Full Text] |
| 142. | Boettler T, Newsome PN, Mondelli MU, Maticic M, Cordero E, Cornberg M, Berg T. Care of patients with liver disease during the COVID-19 pandemic: EASL-ESCMID position paper. JHEP Rep. 2020;2:100113. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 312] [Cited by in RCA: 334] [Article Influence: 66.8] [Reference Citation Analysis (1)] |
| 143. | Li Y, Xiao SY. Hepatic involvement in COVID-19 patients: Pathology, pathogenesis, and clinical implications. J Med Virol. 2020;92:1491-1494. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 60] [Cited by in RCA: 91] [Article Influence: 18.2] [Reference Citation Analysis (0)] |
| 144. | Ramachandran P, Perisetti A, Gajendran M, Chakraborti A, Narh JT, Goyal H. Increased Serum Aminotransferase Activity and Clinical Outcomes in Coronavirus Disease 2019. J Clin Exp Hepatol. 2020;10:533-539. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 11] [Cited by in RCA: 13] [Article Influence: 2.6] [Reference Citation Analysis (0)] |
| 145. | SOHU News. Doctor blackened after successful rescue? Expert: It could be multiple organ damage [in Chinese]. 2020. [cited 14 January 2021]. Available from: https://www.sohu.com/a/389423079_160789. |
| 146. | Zhu Y. Skin darkens in patients with COVID-19: don’t ignore the medical reasons. Health J. 2020;9:1-2. |
| 147. | Liu X, Xue R, Yang C, Gu J, Chen S, Zhang S. Cholestasis-induced bile acid elevates estrogen level via farnesoid X receptor-mediated suppression of the estrogen sulfotransferase SULT1E1. J Biol Chem. 2018;293:12759-12769. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 31] [Cited by in RCA: 32] [Article Influence: 4.6] [Reference Citation Analysis (0)] |
| 148. | Jee SH, Lee SY, Chiu HC, Chang CC, Chen TJ. Effects of estrogen and estrogen receptor in normal human melanocytes. Biochem Biophys Res Commun. 1994;199:1407-1412. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 84] [Cited by in RCA: 82] [Article Influence: 2.6] [Reference Citation Analysis (0)] |
| 149. | Burra P. Liver abnormalities and endocrine diseases. Best Pract Res Clin Gastroenterol. 2013;27:553-563. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 59] [Cited by in RCA: 63] [Article Influence: 5.3] [Reference Citation Analysis (0)] |
| 150. | Wang CY, Babitt JL. Liver iron sensing and body iron homeostasis. Blood. 2019;133:18-29. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 116] [Cited by in RCA: 223] [Article Influence: 31.9] [Reference Citation Analysis (0)] |
| 151. | Graham RM, Chua AC, Herbison CE, Olynyk JK, Trinder D. Liver iron transport. World J Gastroenterol. 2007;13:4725-4736. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in CrossRef: 99] [Cited by in RCA: 119] [Article Influence: 6.6] [Reference Citation Analysis (3)] |
| 152. | Xu X, Chen P, Wang J, Feng J, Zhou H, Li X, Zhong W, Hao P. Evolution of the novel coronavirus from the ongoing Wuhan outbreak and modeling of its spike protein for risk of human transmission. Sci China Life Sci. 2020;63:457-460. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 1252] [Cited by in RCA: 1316] [Article Influence: 263.2] [Reference Citation Analysis (0)] |
| 153. | Letko M, Marzi A, Munster V. Functional assessment of cell entry and receptor usage for SARS-CoV-2 and other lineage B betacoronaviruses. Nat Microbiol. 2020;5:562-569. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 1933] [Cited by in RCA: 2216] [Article Influence: 443.2] [Reference Citation Analysis (0)] |
| 154. | Chai X, Hu L, Zhang Y, Han W, Lu Z, Ke A, Zhou J, Shi G, Fang N, Fan J, Cai J, Lan F. Specific ACE2 Expression in Cholangiocytes May Cause Liver Damage After 2019-nCoV Infection. 2020 Preprint. Available from: bioRxiv:2020. [DOI] [Full Text] |
| 155. | Zhang Y, Zheng L, Liu L, Zhao M, Xiao J, Zhao Q. Liver impairment in COVID-19 patients: A retrospective analysis of 115 cases from a single centre in Wuhan city, China. Liver Int. 2020;40:2095-2103. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 293] [Cited by in RCA: 310] [Article Influence: 62.0] [Reference Citation Analysis (0)] |
| 156. | Tirado SM, Yoon KJ. Antibody-dependent enhancement of virus infection and disease. Viral Immunol. 2003;16:69-86. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 236] [Cited by in RCA: 238] [Article Influence: 10.8] [Reference Citation Analysis (0)] |
| 157. | Wang SF, Tseng SP, Yen CH, Yang JY, Tsao CH, Shen CW, Chen KH, Liu FT, Liu WT, Chen YM, Huang JC. Antibody-dependent SARS coronavirus infection is mediated by antibodies against spike proteins. Biochem Biophys Res Commun. 2014;451:208-214. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 307] [Cited by in RCA: 324] [Article Influence: 29.5] [Reference Citation Analysis (0)] |
| 158. | Wong CK, Lam CW, Wu AK, Ip WK, Lee NL, Chan IH, Lit LC, Hui DS, Chan MH, Chung SS, Sung JJ. Plasma inflammatory cytokines and chemokines in severe acute respiratory syndrome. Clin Exp Immunol. 2004;136:95-103. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 871] [Cited by in RCA: 959] [Article Influence: 45.7] [Reference Citation Analysis (0)] |
| 159. | Channappanavar R, Perlman S. Pathogenic human coronavirus infections: causes and consequences of cytokine storm and immunopathology. Semin Immunopathol. 2017;39:529-539. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 1895] [Cited by in RCA: 1791] [Article Influence: 223.9] [Reference Citation Analysis (0)] |
| 160. | Mahallawi WH, Khabour OF, Zhang Q, Makhdoum HM, Suliman BA. MERS-CoV infection in humans is associated with a pro-inflammatory Th1 and Th17 cytokine profile. Cytokine. 2018;104:8-13. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 396] [Cited by in RCA: 448] [Article Influence: 64.0] [Reference Citation Analysis (0)] |
| 161. | Tian D, Ye Q. Hepatic complications of COVID-19 and its treatment. J Med Virol. 2020;92:1818-1824. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 88] [Cited by in RCA: 87] [Article Influence: 17.4] [Reference Citation Analysis (0)] |
| 162. | Li X, Li X, Huang N, Liu R, Sun R. A comprehensive review and perspectives on pharmacology and toxicology of saikosaponins. Phytomedicine. 2018;50:73-87. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 81] [Cited by in RCA: 146] [Article Influence: 20.9] [Reference Citation Analysis (0)] |
| 163. | Li X, Li X, Lu J, Huang Y, Lv L, Luan Y, Liu R, Sun R. Saikosaponins induced hepatotoxicity in mice via lipid metabolism dysregulation and oxidative stress: a proteomic study. BMC Complement Altern Med. 2017;17:219. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 22] [Cited by in RCA: 34] [Article Influence: 4.3] [Reference Citation Analysis (0)] |
| 164. | Pan X, Zhou J, Chen Y, Xie X, Rao C, Liang J, Zhang Y, Peng C. Classification, hepatotoxic mechanisms, and targets of the risk ingredients in traditional Chinese medicine-induced liver injury. Toxicol Lett. 2020;323:48-56. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 25] [Cited by in RCA: 36] [Article Influence: 7.2] [Reference Citation Analysis (0)] |
| 165. | Iorga A, Dara L. Cell death in drug-induced liver injury. Adv Pharmacol. 2019;85:31-74. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 36] [Cited by in RCA: 55] [Article Influence: 9.2] [Reference Citation Analysis (0)] |
| 166. | Shehu AI, Lu J, Wang P, Zhu J, Wang Y, Yang D, McMahon D, Xie W, Gonzalez FJ, Ma X. Pregnane X receptor activation potentiates ritonavir hepatotoxicity. J Clin Invest. 2019;129:2898-2903. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 21] [Cited by in RCA: 35] [Article Influence: 5.8] [Reference Citation Analysis (0)] |
| 167. | Zha BS, Wan X, Zhang X, Zha W, Zhou J, Wabitsch M, Wang G, Lyall V, Hylemon PB, Zhou H. HIV protease inhibitors disrupt lipid metabolism by activating endoplasmic reticulum stress and inhibiting autophagy activity in adipocytes. PLoS One. 2013;8:e59514. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 55] [Cited by in RCA: 60] [Article Influence: 5.0] [Reference Citation Analysis (0)] |
| 168. | Cao R, Hu Y, Wang Y, Gurley EC, Studer EJ, Wang X, Hylemon PB, Pandak WM, Sanyal AJ, Zhang L, Zhou H. Prevention of HIV protease inhibitor-induced dysregulation of hepatic lipid metabolism by raltegravir via endoplasmic reticulum stress signaling pathways. J Pharmacol Exp Ther. 2010;334:530-539. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 22] [Cited by in RCA: 20] [Article Influence: 1.3] [Reference Citation Analysis (0)] |
| 169. | Shen T, Liu Y, Shang J, Xie Q, Li J, Yan M, Xu J, Niu J, Liu J, Watkins PB, Aithal GP, Andrade RJ, Dou X, Yao L, Lv F, Wang Q, Li Y, Zhou X, Zhang Y, Zong P, Wan B, Zou Z, Yang D, Nie Y, Li D, Wang Y, Han X, Zhuang H, Mao Y, Chen C. Incidence and Etiology of Drug-Induced Liver Injury in Mainland China. Gastroenterology. 2019;156:2230-2241.e11. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 245] [Cited by in RCA: 384] [Article Influence: 64.0] [Reference Citation Analysis (0)] |
| 170. | Zhang C, Shi L, Wang FS. Liver injury in COVID-19: management and challenges. Lancet Gastroenterol Hepatol. 2020;5:428-430. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 1348] [Cited by in RCA: 1295] [Article Influence: 259.0] [Reference Citation Analysis (4)] |
| 171. | Huang Y, Gao Z. Study of the relationship SARS and hepatitis virus B. Zhongguo Linchuang Ganbingxue Zazhi. 2003;6:342-343. |
| 172. | Strnad P, Tacke F, Koch A, Trautwein C. Liver - guardian, modifier and target of sepsis. Nat Rev Gastroenterol Hepatol. 2017;14:55-66. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 236] [Cited by in RCA: 446] [Article Influence: 55.8] [Reference Citation Analysis (0)] |
| 173. | Xiao Y, Pan H, She Q, Wang F, Chen M. Prevention of SARS-CoV-2 infection in patients with decompensated cirrhosis. Lancet Gastroenterol Hepatol. 2020;5:528-529. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 42] [Cited by in RCA: 43] [Article Influence: 8.6] [Reference Citation Analysis (0)] |
| 174. | Kumar D, Tellier R, Draker R, Levy G, Humar A. Severe Acute Respiratory Syndrome (SARS) in a liver transplant recipient and guidelines for donor SARS screening. Am J Transplant. 2003;3:977-981. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 119] [Cited by in RCA: 115] [Article Influence: 5.2] [Reference Citation Analysis (0)] |
| 175. | Michaels MG, La Hoz RM, Danziger-Isakov L, Blumberg EA, Kumar D, Green M, Pruett TL, Wolfe CR. Coronavirus disease 2019: Implications of emerging infections for transplantation. Am J Transplant. 2020;20:1768-1772. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 122] [Cited by in RCA: 127] [Article Influence: 25.4] [Reference Citation Analysis (0)] |
| 176. | Gori A, Dondossola D, Antonelli B, Mangioni D, Alagna L, Reggiani P, Bandera A, Rossi G. Coronavirus disease 2019 and transplantation: A view from the inside. Am J Transplant. 2020;20:1939-1940. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 54] [Cited by in RCA: 55] [Article Influence: 11.0] [Reference Citation Analysis (0)] |
| 177. | Qin J, Wang H, Qin X, Zhang P, Zhu L, Cai J, Yuan Y, Li H. Perioperative Presentation of COVID-19 Disease in a Liver Transplant Recipient. Hepatology. 2020;72:1491-1493. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 85] [Cited by in RCA: 88] [Article Influence: 17.6] [Reference Citation Analysis (0)] |
| 178. | Fraser J, Mousley J, Testro A, Smibert OC, Koshy AN. Clinical Presentation, Treatment, and Mortality Rate in Liver Transplant Recipients With Coronavirus Disease 2019: A Systematic Review and Quantitative Analysis. Transplant Proc. 2020;52:2676-2683. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 24] [Cited by in RCA: 36] [Article Influence: 7.2] [Reference Citation Analysis (0)] |
| 179. | D'Antiga L. Coronaviruses and Immunosuppressed Patients: The Facts During the Third Epidemic. Liver Transpl. 2020;26:832-834. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 479] [Cited by in RCA: 492] [Article Influence: 98.4] [Reference Citation Analysis (0)] |
| 180. | Webb GJ, Marjot T, Cook JA, Aloman C, Armstrong MJ, Brenner EJ, Catana MA, Cargill T, Dhanasekaran R, García-Juárez I, Hagström H, Kennedy JM, Marshall A, Masson S, Mercer CJ, Perumalswami PV, Ruiz I, Thaker S, Ufere NN, Barnes E, Barritt AS 4th, Moon AM. Outcomes following SARS-CoV-2 infection in liver transplant recipients: an international registry study. Lancet Gastroenterol Hepatol. 2020;5:1008-1016. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 138] [Cited by in RCA: 197] [Article Influence: 39.4] [Reference Citation Analysis (0)] |
| 181. | Yang RX, Zheng RD, Fan JG. Etiology and management of liver injury in patients with COVID-19. World J Gastroenterol. 2020;26:4753-4762. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in CrossRef: 58] [Cited by in RCA: 51] [Article Influence: 10.2] [Reference Citation Analysis (2)] |
| 182. | Tang JW, Li Y, Eames I, Chan PK, Ridgway GL. Factors involved in the aerosol transmission of infection and control of ventilation in healthcare premises. J Hosp Infect. 2006;64:100-114. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 454] [Cited by in RCA: 370] [Article Influence: 19.5] [Reference Citation Analysis (0)] |
| 183. | Lau G, Ward JW. Synthesis of Liver Associations Recommendations for Hepatology and Liver Transplant Care During the COVID-19 Pandemic. Clin Liver Dis (Hoboken). 2020;15:204-209. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 14] [Cited by in RCA: 12] [Article Influence: 2.4] [Reference Citation Analysis (0)] |
| 184. | Kushner T, Cafardi J. Chronic Liver Disease and COVID-19: Alcohol Use Disorder/Alcohol-Associated Liver Disease, Nonalcoholic Fatty Liver Disease/Nonalcoholic Steatohepatitis, Autoimmune Liver Disease, and Compensated Cirrhosis. Clin Liver Dis (Hoboken). 2020;15:195-199. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 27] [Cited by in RCA: 38] [Article Influence: 7.6] [Reference Citation Analysis (0)] |
| 185. | The Lancet. COVID-19: protecting health-care workers. Lancet. 2020;395:922. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 617] [Cited by in RCA: 719] [Article Influence: 143.8] [Reference Citation Analysis (0)] |
| 186. | Wang C, Horby PW, Hayden FG, Gao GF. A novel coronavirus outbreak of global health concern. Lancet. 2020;395:470-473. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 4848] [Cited by in RCA: 4388] [Article Influence: 877.6] [Reference Citation Analysis (1)] |
| 187. | Johnston ER, Habib-Bein N, Dueker JM, Quiroz B, Corsaro E, Ambrogio M, Kingsley M, Papachristou GI, Kreiss C, Khalid A. Risk of bacterial exposure to the endoscopist's face during endoscopy. Gastrointest Endosc. 2019;89:818-824. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 100] [Cited by in RCA: 100] [Article Influence: 16.7] [Reference Citation Analysis (0)] |
| 188. | Mohandas KM, Gopalakrishnan G. Mucocutaneous exposure to body fluids during digestive endoscopy: the need for universal precautions. Indian J Gastroenterol. 1999;18:109-111. [PubMed] |
| 189. | Agency for Healthcare Research and Quality. Preventable tragedies: superbugs and how ineffective monitoring of medical device safety fails patients. Rockville: Agency for Healthcare Research and Quality, 2016. |
| 190. | Costerton JW, Lewandowski Z, Caldwell DE, Korber DR, Lappin-Scott HM. Microbial biofilms. Annu Rev Microbiol. 1995;49:711-745. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 3777] [Cited by in RCA: 3125] [Article Influence: 104.2] [Reference Citation Analysis (0)] |
| 191. | Pajkos A, Vickery K, Cossart Y. Is biofilm accumulation on endoscope tubing a contributor to the failure of cleaning and decontamination? J Hosp Infect. 2004;58:224-229. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 126] [Cited by in RCA: 134] [Article Influence: 6.4] [Reference Citation Analysis (0)] |
| 192. | Leung JW, Ling TK, Chan RC, Cheung SW, Lai CW, Sung JJ, Chung SC, Cheng AF. Antibiotics, biliary sepsis, and bile duct stones. Gastrointest Endosc. 1994;40:716-721. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 94] [Cited by in RCA: 87] [Article Influence: 2.8] [Reference Citation Analysis (0)] |
| 193. | Rerknimitr R, Fogel EL, Kalayci C, Esber E, Lehman GA, Sherman S. Microbiology of bile in patients with cholangitis or cholestasis with and without plastic biliary endoprosthesis. Gastrointest Endosc. 2002;56:885-889. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 10] [Cited by in RCA: 37] [Article Influence: 1.6] [Reference Citation Analysis (0)] |
| 194. | Nelson DB, Muscarella LF. Current issues in endoscope reprocessing and infection control during gastrointestinal endoscopy. World J Gastroenterol. 2006;12:3953-3964. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in CrossRef: 94] [Cited by in RCA: 73] [Article Influence: 3.8] [Reference Citation Analysis (0)] |
| 195. | Kovaleva J, Peters FT, van der Mei HC, Degener JE. Transmission of infection by flexible gastrointestinal endoscopy and bronchoscopy. Clin Microbiol Rev. 2013;26:231-254. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 283] [Cited by in RCA: 313] [Article Influence: 26.1] [Reference Citation Analysis (0)] |
| 196. | Rutala WA, Weber DJ. Outbreaks of carbapenem-resistant Enterobacteriaceae infections associated with duodenoscopes: What can we do to prevent infections? Am J Infect Control. 2016;44:e47-e51. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 57] [Cited by in RCA: 57] [Article Influence: 6.3] [Reference Citation Analysis (0)] |
| 197. | Reprocessing Guideline Task Force, Petersen BT, Cohen J, Hambrick RD 3rd, Buttar N, Greenwald DA, Buscaglia JM, Collins J, Eisen G. Multisociety guideline on reprocessing flexible GI endoscopes: 2016 update. Gastrointest Endosc. 2017;85:282-294.e1. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 135] [Cited by in RCA: 137] [Article Influence: 17.1] [Reference Citation Analysis (0)] |
| 198. | U.S. Food and Drug Administration (FDA). Supplemental Measures to Enhance Duodenoscope Reprocessing: FDA Safety Communication. New Hampshire Avenue: FDA; 2015. [cited 1 January 2021]. Available from: https://www.fda.gov/. |
| 199. | U.S. Food and Drug Administration (FDA). Reprocessing medical devices in health care settings: validation methods and labeling guidance for industry and Food and Drug Administration staf. New Hampshire Avenue: FDA; 2015. [cited 10 January 2021]. Available from: https://www.fda.gov/. |
| 200. | Rutala WA, Weber DJ. Guideline for disinfection and sterilization in healthcare facilities. Atlanta, GA: CDC; 2019. [cited 5 January 2021]. Available from: https://www.baidu.com/link?url=iza_ELF480aY01ogG5yoAQfQWGItZhSDXCauPcrUM32TMiqpwSXk1wjDdspbGOAhZAuZPb_v3cSiaCU_384Edhw4voHdo0-S9CXSoCsClzhxAFKj1dCVEouuSjo8kyVA&wd=&eqid=9a54e1460017380c0000000260b771a4. |
| 201. | Food and Drug Administration (FDA). Brief Summary of the Gastroenterology and Urology Panel of the Medical Devices Advisory Committee Meeting – February 26, 2016. Rockville, MD: FDA; 2016. [cited 1 January 2021]. Available from: https://www.fda.gov/. |
| 202. | Muthusamy VR, Bruno MJ, Kozarek RA, Petersen BT, Pleskow DK, Sejpal DV, Slivka A, Peetermans JA, Rousseau MJ, Tirrell GP, Ross AS. Clinical Evaluation of a Single-Use Duodenoscope for Endoscopic Retrograde Cholangiopancreatography. Clin Gastroenterol Hepatol. 2020;18:2108-2117.e3. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 78] [Cited by in RCA: 67] [Article Influence: 13.4] [Reference Citation Analysis (0)] |
| 203. | Gralnek IM, Hassan C, Beilenhoff U, Antonelli G, Ebigbo A, Pellisè M, Arvanitakis M, Bhandari P, Bisschops R, Van Hooft JE, Kaminski MF, Triantafyllou K, Webster G, Pohl H, Dunkley I, Fehrke B, Gazic M, Gjergek T, Maasen S, Waagenes W, de Pater M, Ponchon T, Siersema PD, Messmann H, Dinis-Ribeiro M. ESGE and ESGENA Position Statement on gastrointestinal endoscopy and the COVID-19 pandemic. Endoscopy. 2020;52:483-490. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 259] [Cited by in RCA: 296] [Article Influence: 59.2] [Reference Citation Analysis (0)] |
| 204. | Franzini TAP, Kotinda APST, Moura DTH, Badana MLV, Medeiros MS, Lima PGR, Mello BF, Kayano RP, Carmona MJC, Rocha MC, Campos AF, McCarty TR, Guimarães T, Moura MLDN, Thompson CC, Moura EGH. Approach to Endoscopic Procedures: A Routine Protocol from a Quaternary University Referral Center Exclusively for Coronavirus Disease 2019 Patients. Clinics (Sao Paulo). 2020;75:e1989. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 7] [Cited by in RCA: 9] [Article Influence: 1.8] [Reference Citation Analysis (0)] |
| 205. | Smith ZL, Das KK, Kushnir VM. Anesthesia-administered sedation for endoscopic retrograde cholangiopancreatography: monitored anesthesia care or general endotracheal anesthesia? Curr Opin Anaesthesiol. 2019;32:531-537. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 6] [Cited by in RCA: 20] [Article Influence: 3.3] [Reference Citation Analysis (0)] |
| 206. | Chen X, Liu Y, Gong Y, Guo X, Zuo M, Li J, Shi W, Li H, Xu X, Mi W, Huang Y; Chinese Society of Anesthesiology; Chinese Association of Anesthesiologists. Perioperative Management of Patients Infected with the Novel Coronavirus: Recommendation from the Joint Task Force of the Chinese Society of Anesthesiology and the Chinese Association of Anesthesiologists. Anesthesiology. 2020;132:1307-1316. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 157] [Cited by in RCA: 151] [Article Influence: 30.2] [Reference Citation Analysis (0)] |
| 207. | Kujawski S, Wong K, Collins J, Epstein L, Killerby M, Midgley C, Abedi G, Ahmed N, Almendares O, Alvarez F, Anderson K, Balter S, Barry V, Bartlett K, Beer K, Ben-Aderet M, Benowitz I, Biggs H, Binder A. First 12 patients with coronavirus disease 2019 (COVID-19) in the United States. BMJ. 2020;. [RCA] [DOI] [Full Text] [Cited by in Crossref: 35] [Cited by in RCA: 38] [Article Influence: 7.6] [Reference Citation Analysis (0)] |
| 208. | Young BE, Ong SWX, Kalimuddin S, Low JG, Tan SY, Loh J, Ng OT, Marimuthu K, Ang LW, Mak TM, Lau SK, Anderson DE, Chan KS, Tan TY, Ng TY, Cui L, Said Z, Kurupatham L, Chen MI, Chan M, Vasoo S, Wang LF, Tan BH, Lin RTP, Lee VJM, Leo YS, Lye DC; Singapore 2019 Novel Coronavirus Outbreak Research Team. Epidemiologic Features and Clinical Course of Patients Infected With SARS-CoV-2 in Singapore. JAMA. 2020;323:1488-1494. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 1346] [Cited by in RCA: 1366] [Article Influence: 273.2] [Reference Citation Analysis (0)] |
| 209. | Tabata S, Imai K, Kawano S, Ikeda M, Kodama T, Miyoshi K, Obinata H, Mimura S, Kodera T, Kitagaki M, Sato M, Suzuki S, Ito T, Uwabe Y, Tamura K. Clinical characteristics of COVID-19 in 104 people with SARS-CoV-2 infection on the Diamond Princess cruise ship: a retrospective analysis. Lancet Infect Dis. 2020;20:1043-1050. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 179] [Cited by in RCA: 163] [Article Influence: 32.6] [Reference Citation Analysis (0)] |
| 210. | Wölfel R, Corman VM, Guggemos W, Seilmaier M, Zange S, Müller MA, Niemeyer D, Jones TC, Vollmar P, Rothe C, Hoelscher M, Bleicker T, Brünink S, Schneider J, Ehmann R, Zwirglmaier K, Drosten C, Wendtner C. Virological assessment of hospitalized patients with COVID-2019. Nature. 2020;581:465-469. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 4682] [Cited by in RCA: 4810] [Article Influence: 962.0] [Reference Citation Analysis (0)] |
| 211. | Gritti G, Raimondi F, Ripamonti D, Riva I, Landi F, Alborghetti L, Frigeni M, Damiani M, Micò C, Fagiuoli S. IL-6 signalling pathway inactivation with siltuximab in patients with COVID-19 respiratory failure: an observational cohort study. BMJ. 2020;. [DOI] [Full Text] |
| 212. | COVID-19 National Incident Room Surveillance Team. COVID-19, Australia: Epidemiology Report 7 (Reporting week ending 19:00 AEDT 14 March 2020). Commun Dis Intell (2018). 2020;44. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 12] [Cited by in RCA: 17] [Article Influence: 3.4] [Reference Citation Analysis (0)] |
| 213. | COVID-19 National Emergency Response Center, Epidemiology and Case Management Team, Korea Centers for Disease Control and Prevention. Early Epidemiological and Clinical Characteristics of 28 Cases of Coronavirus Disease in South Korea. Osong Public Health Res Perspect. 2020;11:8-14. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 90] [Cited by in RCA: 93] [Article Influence: 18.6] [Reference Citation Analysis (0)] |
| 214. | Sierpiński R, Pinkas J, Jankowski M, Zgliczyński WS, Wierzba W, Gujski M, Szumowski Ł. Sex differences in the frequency of gastrointestinal symptoms and olfactory or taste disorders in 1942 nonhospitalized patients with coronavirus disease 2019 (COVID-19). Pol Arch Intern Med. 2020;130:501-505. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 21] [Cited by in RCA: 28] [Article Influence: 5.6] [Reference Citation Analysis (0)] |
| 215. | Qi D, Yan X, Tang X, Peng J, Yu Q, Feng L, Yuan G, Zhang A, Chen Y, Yuan J. Epidemiological and clinical features of 2019-nCoV acute respiratory disease cases in Chongqing municipality, China: a retrospective, descriptive, multiple-center study. BMJ. 2020;. [DOI] [Full Text] |
| 216. | Wen Y, Wei L, Li Y, Tang X, Feng S, Leung K, Wu X, Pan X-F, Chen C, Xia J. Epidemiological and clinical characteristics of COVID-19 in Shenzhen, the largest migrant city of China. BMJ. 2020;. [DOI] [Full Text] |
| 217. | Dan F, Jingdong MA, Jialun G, Muru W, Yang S, Dean T, Peiyuan LI. Manifestations of digestive system in hospitalized patients with novel coronavirus pneumonia in Wuhan, China: a single-center, descriptive study. Zhonghua Xiaohua Zazhi. 2020;12:E005. [DOI] [Full Text] |
| 218. | Ma L, Xie W, Li D, Shi L, Mao Y, Xiong Y, Zhang Y, Zhang M. Effect of SARS-CoV-2 infection upon male gonadal function: A single center-based study. BMJ. 2020;. [DOI] [Full Text] |
| 219. | Liu L, Liu W, Zheng Y, Jiang X, Kou G, Ding J, Wang Q, Huang Q, Ding Y, Ni W, Wu W, Tang S, Tan L, Hu Z, Xu W, Zhang Y, Zhang B, Tang Z, Zhang X, Li H, Rao Z, Jiang H, Ren X, Wang S, Zheng S. A preliminary study on serological assay for severe acute respiratory syndrome coronavirus 2 (SARS-CoV-2) in 238 admitted hospital patients. Microbes Infect. 2020;22:206-211. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 85] [Cited by in RCA: 109] [Article Influence: 21.8] [Reference Citation Analysis (0)] |
| 220. | Ai J, Chen J, Wang Y, Liu X, Fan W, Gaojing Q, Zhang M, Pei S, Tang B, Yuan S, Li Y, Wang L, Huang G, Pei B. The cross-sectional study of hospitalized coronavirus disease 2019 patients in Xiangyang, Hubei province. 2020 Preprint. Available from: medRxiv:2020. [DOI] [Full Text] |
| 221. | Zhao Z, Xie J, Yin M, Yang Y, He H, Jin T, Li W, Zhu X, Xu J, Zhao C, Li L, Li Y, Mengist HM, Zahid A, Yao Z, Ding C, Qi Y, Gao Y, Ma X. Clinical and Laboratory Profiles of 75 Hospitalized Patients with Novel Coronavirus Disease 2019 in Hefei, China. 2020 Preprint. Available from: medRxiv:2020. [DOI] [Full Text] |
| 222. | Li K, Wu J, Wu F, Guo D, Chen L, Fang Z, Li C. The Clinical and Chest CT Features Associated With Severe and Critical COVID-19 Pneumonia. Invest Radiol. 2020;55:327-331. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 882] [Cited by in RCA: 797] [Article Influence: 159.4] [Reference Citation Analysis (0)] |
| 223. | Lin L, Jiang X, Zhang Z, Huang S, Fang Z, Gu Z, Gao L, Shi H, Mai L, Liu Y, Lin X, Lai R, Yan Z, Li X, Shan H. Gastrointestinal symptoms of 95 cases with SARS-CoV-2 infection. Gut. 2020;69:997-1001. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 630] [Cited by in RCA: 658] [Article Influence: 131.6] [Reference Citation Analysis (0)] |
| 224. | Cholankeril G, Podboy A, Aivaliotis VI, Pham EA, Spencer SP, Kim D, Ahmed A. Association of Digestive Symptoms and Hospitalization in Patients With SARS-CoV-2 Infection. Am J Gastroenterol. 2020;115:1129-1132. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 35] [Cited by in RCA: 32] [Article Influence: 6.4] [Reference Citation Analysis (0)] |
| 225. | Redd WD, Zhou JC, Hathorn KE, McCarty TR, Bazarbashi AN, Thompson CC, Shen L, Chan WW. Prevalence and Characteristics of Gastrointestinal Symptoms in Patients With Severe Acute Respiratory Syndrome Coronavirus 2 Infection in the United States: A Multicenter Cohort Study. Gastroenterology. 2020;159:765-767.e2. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 241] [Cited by in RCA: 283] [Article Influence: 56.6] [Reference Citation Analysis (0)] |
| 226. | Kluytmans-van den Bergh MFQ, Buiting AGM, Pas SD, Bentvelsen RG, van den Bijllaardt W, van Oudheusden A, van Rijen MML, Verweij JJ, Koopmans MPG, Kluytmans JAJW. SARS-CoV-2 infection in 86 healthcare workers in two Dutch hospitals in March 2020: a cross-sectional study with short-term follow-up. 2020 Preprint. Available from: medRxiv:2020. [DOI] [Full Text] |
| 227. | Shu L, Wang X, Li M, Chen X, Ji N, Shi L, Wu M, Deng K, Wei J, Cao Y, Yan J, Feng G. Clinical characteristics of moderate COVID-19 patients aggravation in Wuhan Stadium Cabin Hospital: A 571 cases of retrospective cohort study. J Med Virol. 2021;93:1133-1140. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 14] [Cited by in RCA: 19] [Article Influence: 3.8] [Reference Citation Analysis (0)] |
| 228. | Huang Y, Yang R, Xu Y, Gong P. Clinical characteristics of 36 non-survivors with COVID-19 in Wuhan, China. 2020 Preprint. Available from: medRxiv:2020. [DOI] [Full Text] |
| 229. | Yao N, Wang SN, Lian JQ, Sun YT, Zhang GF, Kang WZ, Kang W. [Clinical characteristics and influencing factors of patients with novel coronavirus pneumonia combined with liver injury in Shaanxi region]. Zhonghua Gan Zang Bing Za Zhi. 2020;28:234-239. [RCA] [PubMed] [DOI] [Full Text] [Cited by in RCA: 39] [Reference Citation Analysis (0)] |
| 230. | Xu S, Fu L, Fei J, Xiang H-X, Xiang Y, Tan Z-X, Li M-D, Liu F-F, Li Y, Han M-F, Li X-Y, Yu D-X, Zhao H, Xu D-X. Acute kidney injury at early stage as a negative prognostic indicator of patients with COVID-19: a hospital-based retrospective analysis. 2020 Preprint. Available from: medRxiv:2020. [DOI] [Full Text] |
| 231. | Cai Q, Huang D, Ou P, Yu H, Zhu Z, Xia Z, Su Y, Ma Z, Zhang Y, Li Z, He Q, Liu L, Fu Y, Chen J. COVID-19 in a designated infectious diseases hospital outside Hubei Province, China. Allergy. 2020;75:1742-1752. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 252] [Cited by in RCA: 332] [Article Influence: 66.4] [Reference Citation Analysis (0)] |