Published online Feb 14, 2020. doi: 10.3748/wjg.v26.i6.598
Peer-review started: October 15, 2019
First decision: December 4, 2019
Revised: December 20, 2019
Accepted: January 15, 2020
Article in press: January 15, 2020
Published online: February 14, 2020
Processing time: 122 Days and 2 Hours
The prognosis of gastric cancer continues to remain poor, and epigenetic drugs like histone deacetylase inhibitors (HDACi) have been envisaged as potential therapeutic agents. Nevertheless, clinical trials are facing issues with toxicity and efficacy against solid tumors, which may be partly due to the lack of patient stratification for effective treatments.
To study the need of patient stratification before HDACi treatment, and the efficacy of pre-treatment of HDACi as a chemotherapeutic drug sensitizer.
The expression activity of class 1 HDACs and histone acetylation was examined in human gastric cancer cells and tissues. The potential combinatorial regime of HDACi and chemotherapy drugs was defined on the basis of observed drug binding assays, chromatin remodeling and cell death.
In the present study, the data suggest that the differential increase in HDAC activity and the expression of class 1 HDACs are associated with hypo-acetylation of histone proteins in tumors compared to normal adjacent mucosa tissue samples of gastric cancer. The data highlights for the first time that pre-treatment of HDACi results in an increased amount of DNA-bound drugs associated with enhanced histone acetylation, chromatin relaxation and cell cycle arrest. Fraction-affected plots and combination index-based analysis show that pre-HDACi chemo drug combinatorial regimes, including valproic acid with cisplatin or oxaliplatin and trichostatin A with epirubicin, exhibit synergism with maximum cytotoxic potential due to higher cell death at low combined doses in gastric cancer cell lines.
Expression or activity of class 1 HDACs among gastric cancer patients present an effective approach for patient stratification. Furthermore, HDACi therapy in pre-treatment regimes is more effective with chemotherapy drugs, and may aid in predicting individual patient prognosis.
Core tip: Our study suggests that pre-treatment with histone deacetylase inhibitors (HDACi) in a pre-clinical model of gastric cancer increases acetylation, opens chromatin and favors synergistic binding of DNA-interacting chemotherapeutic drugs. This enhances the cytotoxic potential of chemotherapeutic drugs at low therapeutic doses, and reduces toxicity. The dose response studies using Fa plots and median curve analysis proposes valproic acid as the most synergistic and effective HDACi in combination with platinum-based drugs. Furthermore, HDAC expression, or activity-based patient stratification prior to HDACi therapy, has been put forth for better clinical outcomes of chemotherapeutic drugs in solid tumors.
- Citation: Amnekar RV, Khan SA, Rashid M, Khade B, Thorat R, Gera P, Shrikhande SV, Smoot DT, Ashktorab H, Gupta S. Histone deacetylase inhibitor pre-treatment enhances the efficacy of DNA-interacting chemotherapeutic drugs in gastric cancer. World J Gastroenterol 2020; 26(6): 598-613
- URL: https://www.wjgnet.com/1007-9327/full/v26/i6/598.htm
- DOI: https://dx.doi.org/10.3748/wjg.v26.i6.598
Gastric cancer (GC) is the 3rd leading cause of cancer deaths, 5th in terms of incidence in the world, and the most lethal cancer in Asia[1]. In India, it is one of the most aggressive cancers, ranking 5th in terms of incidence and mortality. The management of GC is multimodal, including surgical R0 resection, radiotherapy and chemotherapy. Based on multiple clinical trials, commonly used drug combinations for neoadjuvant and adjuvant chemotherapy (NACT and ACT) in GC involves drugs such as cisplatin, oxaliplatin and epirubicin, which exert their cytotoxic effects by binding to DNA[2,3]. Earlier reports have shown that inhibitors of chromatin remodelers, such as valproic acid and butyric acid, increase the efficacy of chemotherapeutic drugs[4-6].
Post-translational modifications of histone proteins are one of the major epigenetic mechanisms regulating chromatin conformations[7]. Acetylation of histones has been the most studied, and has been shown to positively correlate with chromatin relaxation. The dynamic equilibrium between histone acetyltransferases (HATs) and histone deacetylases (HDACs) dictates the acetylation levels and transcriptional status of chromatin[8]. Alterations in the levels of several histone acetylation marks such as H3K12ac, H3K18ac, H3K9ac and H4K16ac have been reported in multiple cancers, such as liver, kidney, prostate, breast and stomach[9]. Moreover, aberrant expression of HATs like CBP and p300, and HDACs like HDAC1 and HDAC2, has been observed in several malignancies[9]. These findings have led to the exponential growth in research on HAT inhibitors (HATi) and HDAC inhibitors (HDACi), and their anti-cancer properties. HATi, like E-7438 and EPZ-5676, are in phase II and phase I clinical trials, respectively; also, sodium butyrate is in phase II, and panobinostat and valproic acid (VPA) are in phase III clinical trials. Additionally, HDACi, like vorinostat (SAHA) and romidepsin, is now FDA approved for cancer treatment[10]. Importantly, studies have shown that HDACi can modulate cellular responses through different mechanisms, such as re-expression of tumor suppressors, transporters of chemotherapeutic drugs[11], enzymes associated with drug metabolism[12], and enhancing the levels of open chromatin[13]. HDACis have shown to have potential therapeutic benefits, predominantly in advanced hematologic malignancies, more so as combinatorial chemotherapy than as single agents; however, clinical responses are disappointing in solid tumors[14,15]. Marchio et al[16]’s study suggested the use of HDACi (SAHA) as a chemo-sensitizer that increases the efficiency of epirubicin in breast cancer. To overcome the limitation of HDACi in solid tumors, the identification of synergistic combinations of selective HDACi with other chemotherapeutic drugs, along with patient stratification based on HDAC levels, is a must and would help to both minimize toxicity and predict the response to treatment.
In the present study, we show a strong association between global histone hypo-acetylation with increased HDAC activity in human GC tissue samples and cell lines. Differential activity and expression levels of class 1 HDACs in patient samples and TCGA data highlight the importance of patient stratification for treatment with HDACi. Interestingly, the pre-treatment regime of HDACi followed by chemotherapeutic drugs exerts synergistic effects in GC cells and pre-clinical studies.
Paired frozen tissue samples from normal adjacent gastric mucosa (negative resection margin) and tumors were collected from GC patients through the tumor tissue repository of ACTREC and the Tata Memorial Hospital, Mumbai, India. The protocol was reviewed and approved by the institutional review board and ethics committee. Written informed consent was undertaken from all patients. Based on histopathological analysis by a blinded pathologist, the tumor content was found to be > 60% in all tumor samples.
The AGS GC cell line (CRL 1739) was procured from ATCC, and the HFE145 cell line was provided by DTS and AH. The cell lines were cultured in RPMI1640 (Invitrogen) media with 10% fetal bovine serum and maintained at 37 ºC with 5% CO2 and 100 U/mL penicillin, 100 mg/mL streptomycin (Himedia).
HDACi, sodium valproate (VPA; Sigma, P4543), trichostatin A (TSA; Sigma, T8552) and suberoylanilide hydroxamic acid (SAHA; Sigma, SML0061) were dissolved in ethanol to prepare stock solutions. The chemotherapeutic drugs cisplatin (Calbiochem, 232120), oxaliplatin (Sigma, O9512) and epirubicin (Calbiochem, 324905) were dissolved in DMSO to prepare stock solutions. As per the experimental requirements, cells were treated with different concentrations of HDACi and chemotherapeutic drugs.
Cell viability was quantified by MTT assay (Sigma, M2128) as per the manufacturer’s protocol. AGS cells (about 1000) were used for the assay, the absorbance was measured at 570 nm, and cell viability was expressed as the percentage of absorbance obtained compared to control cultures.
Histones were extracted, resolved on an 18% SDS-PAGE gel and transferred to a PVDF membrane[17]. Western blotting with the respective antibodies was carried out as per the manufacturer’s protocol (H3, Millipore #05-499; H4, Millipore #07-108; H3K9ac, Millipore #06-599; H3K18ac, Millipore #07-354; H3K27ac, Abcam #4729; H3K16ac, Millipore #07-329; γH2AX, Millipore #05-636; H3ac, Millipore #06-599; H4ac, Millipore #06-866). The signal was visualized using the ECL plus chemiluminescence kit (Millipore #WBKLS0500).
Nucleo-cytosolic fractions (NCF) from human GC tissues and cell lines were prepared as per the manufacturer’s instructions (Biovision, K332-100 and K331-100). Protein lysates, cell lines (50 µg) and tissues (100 µg) were used for calorimetric-based assays. The absorbance was measured at A440 and A405 for HAT and HDAC, respectively, and the average absorbance was plotted.
AGS cells treated with chemotherapeutic drugs with or without different combinations of HDAC inhibitors were washed in chilled PBS and lysed in nuclei isolation buffer (10 mmol/L HEPES pH 7.9, 1.5 mmol/L MgCl2, 10 mmol/L KCl, 0.5 mmol/L DTT, 0.1% v/v NP-40, 2 mmol/L EDTA, 1 mmol/L EGTA, 0.15 mmol/L spermine, 0.5 mmol/L spermidine, 1 mmol/L sodium orthovanadate, 10 mmol/L sodium fluoride, 10 mmol/L β-Glycerophosphate, 0.2 mmol/L PMSF). The lysate was centrifuged at 5000 rpm for 10 min at 4 ºC. The nuclear pellet obtained was lysed in 200 µL 5 mol/L urea-2 mol/L NaCl solution to estimate the DNA concentration at 260 nm. DNA concentration was adjusted to 1 µg/mL, and an equal volume was taken to measure the concentration of DNA-bound cisplatin, oxaliplatin and epirubicin at 220, 205 and 254 nm, respectively, as per European Pharmacopoiea 5.5[18]. The absorbance was considered to be directly proportional to the amount of DNA bound to drug. The mean absorbance of three independent experiments was plotted for the chemotherapeutic drugs.
Fraction-affected (FA) curves, a method for growth inhibition analysis, was carried out with cell survival percentage values obtained through three independent MTT assays. Fraction-affected values representing the percentages of cell death were calculated using the following formula:
FA value = 1—(% cell survival/100). FA values ranged from 0.01 to 0.99); CompuSyn software was used to further assist in these calculations, which is based on the Chao Tally’s algorithm[19]. FA values and respective doses of the drugs were used to generate FA curves.
The median effect plot shows the combination index (CI) on the Y-axis and FA values on the X-axis. For a particular FA value, CI values range from 0 to 1; CI < 0.8, CI = 0.8-1.2, and CI > 1.2 represents the synergistic, additive or antagonistic nature of drug combinations, respectively. FA values and total doses of drug combinations (chemotherapeutic drugs and HDACi) were used to generate median effect plots with the help of CompuSyn software[19].
Purified nuclei from control and VPA-treated AGS cells were subjected to micrococcal nuclease (MNase) digestion to analyze chromatin organization as per the published protocol[20].
RNA was extracted by TRIzol method (Invitrogen, 5596026), and cDNA was synthesized as per the manufacture’s protocol (Thermo scientific, K1632). Quantitative PCR using SYBER green (Agilent Tech, 600882) was employed, and the primers used are listed in Table 1. Data analysis was performed using ΔΔCt-based calculations, and fold changes were plotted for the different HDAC genes. The data were statistically analyzed by performing student t-tests.
| Gene | Forward primer, 5`-3` | Reverse primer, 5`-3` |
| RPS13 | GCTCTCCTTTCGTTGCCTGA | ACTTCAACCAAGTGGGGACG |
| HDAC1 | ATATCGTCTTGGCCATCCTG | TGAAGCAACCTAACCGATCC |
| HDAC2 | GGGAATACTTTCCTGGCACA | ACGGATTGTGTAGCCACCTC |
| HDAC3 | TGGCATTGACCCATAGCCTG | GCATATTGGTGGGGCTGACT |
| HDAC4 | TCGCTACTGGTACGGGAAAAC | AGAGGGAAGTCATCTTTGGCG |
| HDAC5 | ACTGTTCTCAGATGCCCAGC | TGGTGAAGAGGTGCTTGACG |
| HDAC6 | AGTGGCCGCATTATCCTTATCC | ATCTGCGATGGACTTGGATGG |
| HDAC7 | TTCCTGAGTGCAGGGGTAGT | CATCGCCAGGAGGTTGATGT |
| HDAC8 | ATAACCTTGCCAACACGGCT | CTTGGCGTGATTTCCAGCAC |
| HDAC9 | ACTGAAGCAACCAGGCAGTC | TTCACAGCCCCAACTTGTCC |
| HDAC10 | CTGGCCTTTGAGGGGCAAAT | CAGCAGCGTCTGTACTGTCA |
| HDAC11 | CCGGAAAATGGGGCAAAGTG | TAAGATAGCGCCTCGTGTGC |
| p27-CDKN1B | TTGGGGCAAAAATCCGAGGT | TGTGTTTACACAGCCCGAAGT |
| p21-CDKN1A | GCGACTGTGATGCGCTAATG | GAAGGTAGAGCTTGGGCAGG |
| p16-CDKN2A | ACTTCAGGGGTGCCACATTC | CGACCCTGTCCCTCAAATCC |
Different phases of the cell cycle were determined by flow cytometry as previously described in[20]. The data were analyzed using ModFit LN 2.0 software.
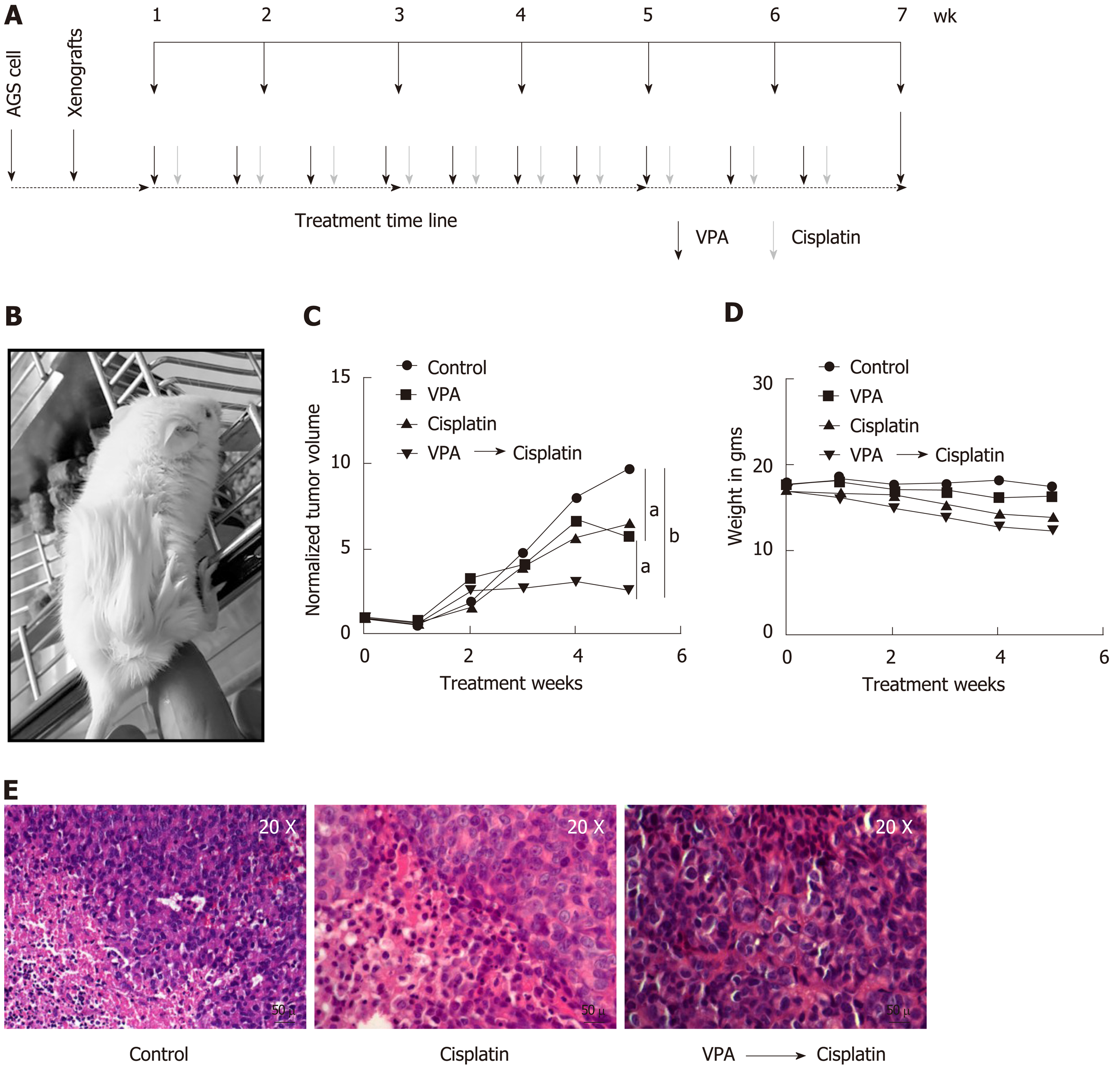
To study the in vivo therapeutic potential of a pre-treatment regime of VPA and cisplatin in an AGS cell-based xenograft model was developed after approval from the institutional animal ethics committee. AGS cells (approximately 5 × 106) were subcutaneously injected with matrigel (50 μL) in NOD-SCID mice (4-6 wk). After one round of serial transplantation, the mice bearing tumors of approximately 6-10 mm maximum length were randomly divided into four groups: (1) Control; (2) VPA (300 mg/kg/d); (3) Cisplatin (2 mg/kg/d); and (4) Combinatorial pre-treatment group (VPA followed by cisplatin, with doses as mentioned above). Drugs or saline (control group) were administered intraperitoneally twice a week at an interval of 2 d for 6 wk. For the combinatorial regime, cisplatin was administered following 24 h of VPA treatment.
Tumor size and weight was monitored once a week by measuring two perpendicular tumor diameters with a caliper, and volume was calculated as V = π / 6 × length × width2. The outcome of the different treatment regimens was statistically validated by performing unpaired t-tests. The animals were sacrificed, tissue was fixed in formalin, and 4-6 μm sections were processed with hematoxylin and eosin staining for histopathological analysis.
Normalized human gastric adenocarcinoma (n = 415) data (Z-score, cutoff > 1.5) of HDAC1, HDAC2, HDAC3 were downloaded from the cBioPortal website (http://www.cbioportal.org/)[21,22]. The samples were categorized into high and low expression groups according to the Z-score, and then tabulated.
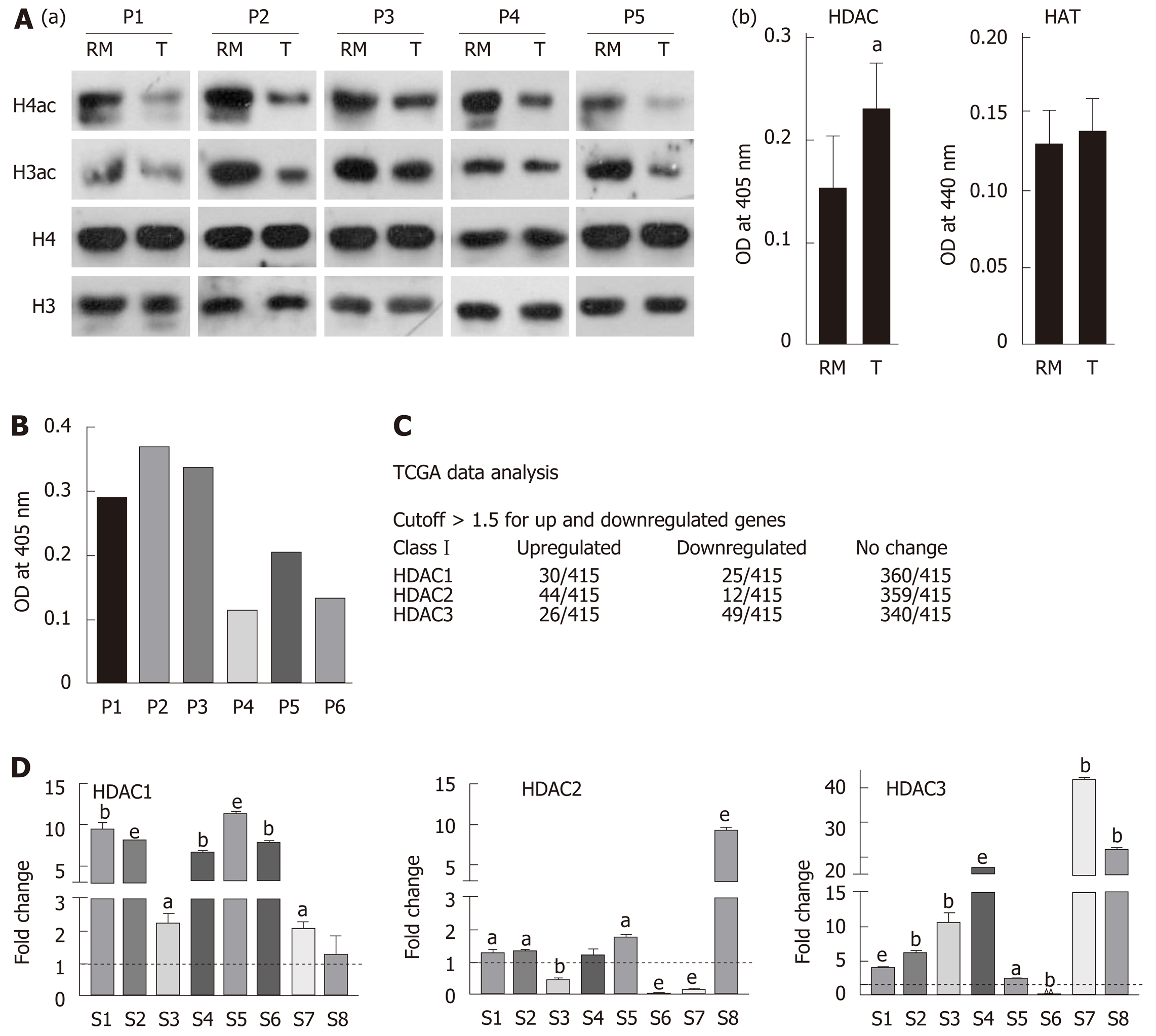
Histones were prepared from paired tumor and negative resection margin (RM) tissues, and subjected to immunoblot analysis to assess the level of acetylation using anti-acetyl lysine antibodies [Figure 1A(a)]. Immunoblot analysis showed low levels of histone H3 and H4 acetylation in the tumor tissues compared to RM tissues. This observed loss in acetylation levels of histone H3 and H4 could be the result of low histone acetyl-transferase (HAT) or high histone deacetylase (HDAC) activity in tumor tissues. Therefore, NCF was used to assess HAT and HDAC activity using calorimetric assays. Tumor and RM tissues showed differential levels of HAT and HDAC activity; however, all the tumor tissues showed high HDAC activity compared to their paired RM tissues, but HAT activity did not show any consistent pattern (Supplementary Figure 1). Further, statistical analysis showed a significantly higher level of HDAC activity in tumor compared to negative RM (P < 0.001) [Figure 1A(b)]; however, no significant difference was found in HAT activity. Taken together, our data suggested an inverse correlation between HDAC activity and histone acetylation in GC.
The major reasons for HDACi failure in solid tumors may be attributed to expression or activity heterogeneity in class 1 HDACs of patients. To understand the need of patient stratification for HDACi therapy, we analyzed HDAC activity and found that it significantly differs among GC patients (Figure 1B). TCGA was analyzed for the expression of class 1 HDAC viz HDAC1, HDAC2 and HDAC3 in gastric adenocarcinoma patients (n = 415) versus control (n = 35), and categorized into high and low expression groups according to the Z-score. HDAC1, HDAC2 and HDAC3 were upregulated in only approximately 24% of gastric adenocarcinoma patients (Figure 1C). Further, in coherence with activity, expression levels also showed differentially increased levels of HDAC 1 and 3 compared to normal gastric tissue (Figure 1D). Altogether, these data provide evidence for patient stratification, and for tailoring the dose of HDACi for (re)sensitizing tumors to the anti-proliferative effects of chemotherapeutic drugs at reduced doses with minimal side effects.
It has been hypothesized that HDACi mediates chromatin relaxation, which may enhance the amount of chemotherapeutic drugs bound to DNA. To understand this, histone acetylation and class 1 HDAC levels were determined in AGS and HFE145 cell lines. A decrease in site-specific histone acetylation at H3K9ac, H3K18ac, H3K27ac was observed [Figure 2A(a)], along with increased HDAC activity [Figure 2A(b)] and levels of class 1 HDACs 1, 2 and other HDACs 8, 10, 11 [Figure 2A(c)] in transformed AGS cells. The AGS cell line reflects the human gastric tumor state, and was therefore used for further studies. Dose response curves for chemotherapy drugs and HDACi showed IC50s of 12 µmol/L, 10 µmol/L and 0.2 µmol/L for cisplatin, oxaliplatin and epirubicin, respectively (Supplementary Figure 2A), whereas for HDACi, the IC50 concentrations of VPA, TSA and SAHA were found to be 4 mmol/L, 2 µmol/L and 0.01 µmol/L, respectively (Supplementary Figure 2B). Further, we confirmed HDACi activity by treating AGS cells with IC50 doses for 24 h, and assessing HDAC activity and histone acetylation levels. A decrease in HDAC activity (Supplementary Figure 2C) with hyperacetylation of histones H3 and H4 (Supplementary Figure 2D) were observed upon HDACi treatment. Taken together, our data confirm that the HDACi used in our study is functionally active, and alters both HDAC activity and histone acetylation.
AGS cells treated with HDACi and chemotherapeutic drugs at IC50 values in three different combinations (Figure 2B): (1) Pre- (24 h HDACi treatment followed by 24 h chemotherapeutic drug treatment); (2) Concurrent (24 h HDACi and chemo-therapeutic drug together); and (3) Post- (24 h chemotherapeutic drug treatment followed by 24 h HDACi treatment) showed that the quantity of DNA-bound chemotherapeutic drugs significantly increased with pre- followed by concurrent regimes. However, post-treatment did not show any significant increase compared to control for all three HDACis (Figure 2C).
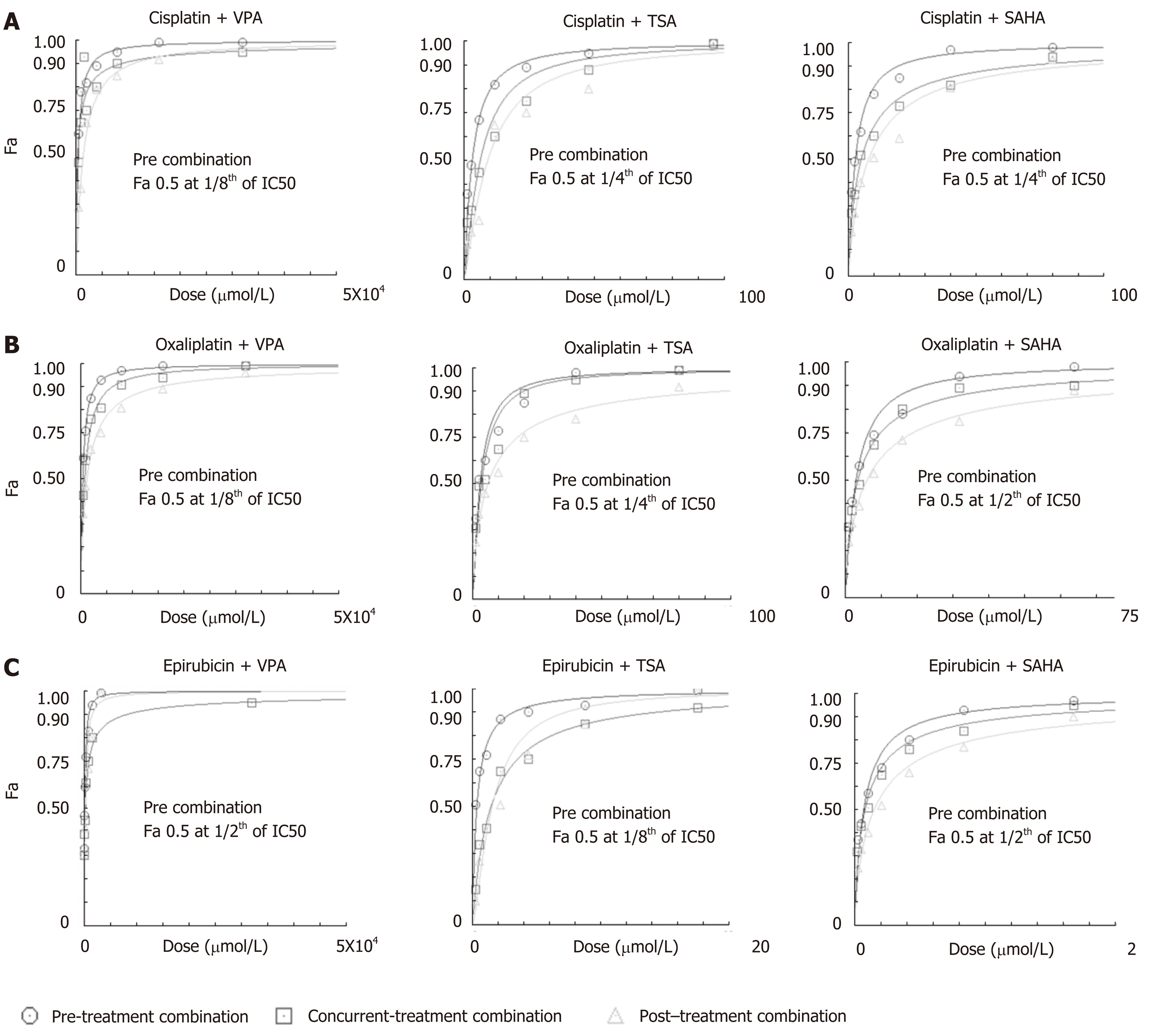
The effect of regime-specific combinatorial treatment of HDACi and chemotherapeutic drugs on cell death was studied using FA plot analysis (Figure 3)[23]. MTT assays were performed using a fixed constant ratio of chemotherapeutic drugs, and in three different combination regimes: Concurrent, pre- and post- (Supplementary Table 1). The data showed that pre-treatment with the three HDACis led to more cell death compared to concurrent or post-treatment in combination with cisplatin (Figure 3A), oxaliplatin (Figure 3B) and epirubicin (Figure 3C). Further, the combined doses of chemotherapeutic drugs and HDACi required to achieve FA values of 0.5, 0.75 and 0.95 was analyzed (Table 2). The pre-treatment regime of VPA with cisplatin required lesser combined doses to achieve FA 0.5, 0.75 and 0.95 compared to both concurrent and post-treatment regimes. However, pre-treatment of TSA or SAHA with cisplatin could only attain FA values of 0.5 and 0.75 at a lower combined dose than concurrent or post-treatment combinations. In the case of oxaliplatin, pre-treatment with only VPA attained FA values of 0.5, 0.75 and 0.95; whereas, TSA and SAHA achieved only FA values of 0.5 at lower combined doses than the concurrent or post-treatment regimes. In the case of epirubicin, pre-treatment with TSA was found to be most effective, achieving FA values of 0.5 and 0.75 at lesser doses, followed by SAHA. Overall, cell death by cisplatin and oxaliplatin is effectively enhanced with VPA, and epirubicin with TSA, in the pre-treatment combination regimes.
| Drugs | Treatment sequence and result of FA and median effect plot analysis | FA | |||
| 0.5 | 0.75 | 0.95 | |||
| Cisplatin and VPA | Pre | Dose of Cis, VPA (µmol/L) | 0.965, 386.359 | 2.711, 1084.65 | 15.361, 6144.61 |
| CI | 0.352 | 0.471 | 0.784 | ||
| Concurrent | Dose of Cis, VPA (µmol/L) | 0.705, 282.387 | 3.874, 1549.74 | 67.690, 27076.0 | |
| CI | 0.257 | 0.673 | 3.454 | ||
| Post | Dose of Cis, VPA (µmol/L) | 3.254 ,1301.90 | 9.362, 3745.16 | 55.260, 22104.1 | |
| CI | 1.188 | 1.627 | 2.820 | ||
| Oxaliplatin and VPA | Pre | Dose of Oxa, VPA (µmol/L) | 0.788, 394.469 | 2.094, 1047.04 | 10.796, 5398.39 |
| CI | 0.404 | 0.453 | 0.552 | ||
| Concurrent | Dose of Oxa, VPA (µmol/L) | 1.533, 766.633 | 4.305, 2152.92 | 24.406, 12203.2 | |
| CI | 0.786 | 0.933 | 1.249 | ||
| Post | Dose of Oxa, VPA (µmol/L) | 2.362, 1181.47 | 8.451, 4225.64 | 71.917, 35958.6 | |
| CI | 1.211 | 1.83182 | 3.68265 | ||
| Epirubicin and TSA | Pre | Combined dose (µg) | 0.030, 0.300 | 0.093, 0.931 | 0.623, 6.231 |
| CI | 0.645 | 0.727 | 1.222 | ||
| Concurrent | Combined dose (µg) | 0.129, 1.299 | 0.410, 4.105 | 2.835, 28.356 | |
| CI | 2.789 | 3.207 | 5.563 | ||
| Post | Combined dose (µg) | 0.137, 1.375 | 0.301, 3.010 | 1.122, 11.220 | |
| CI | 2.952 | 2.351 | 2.201 | ||
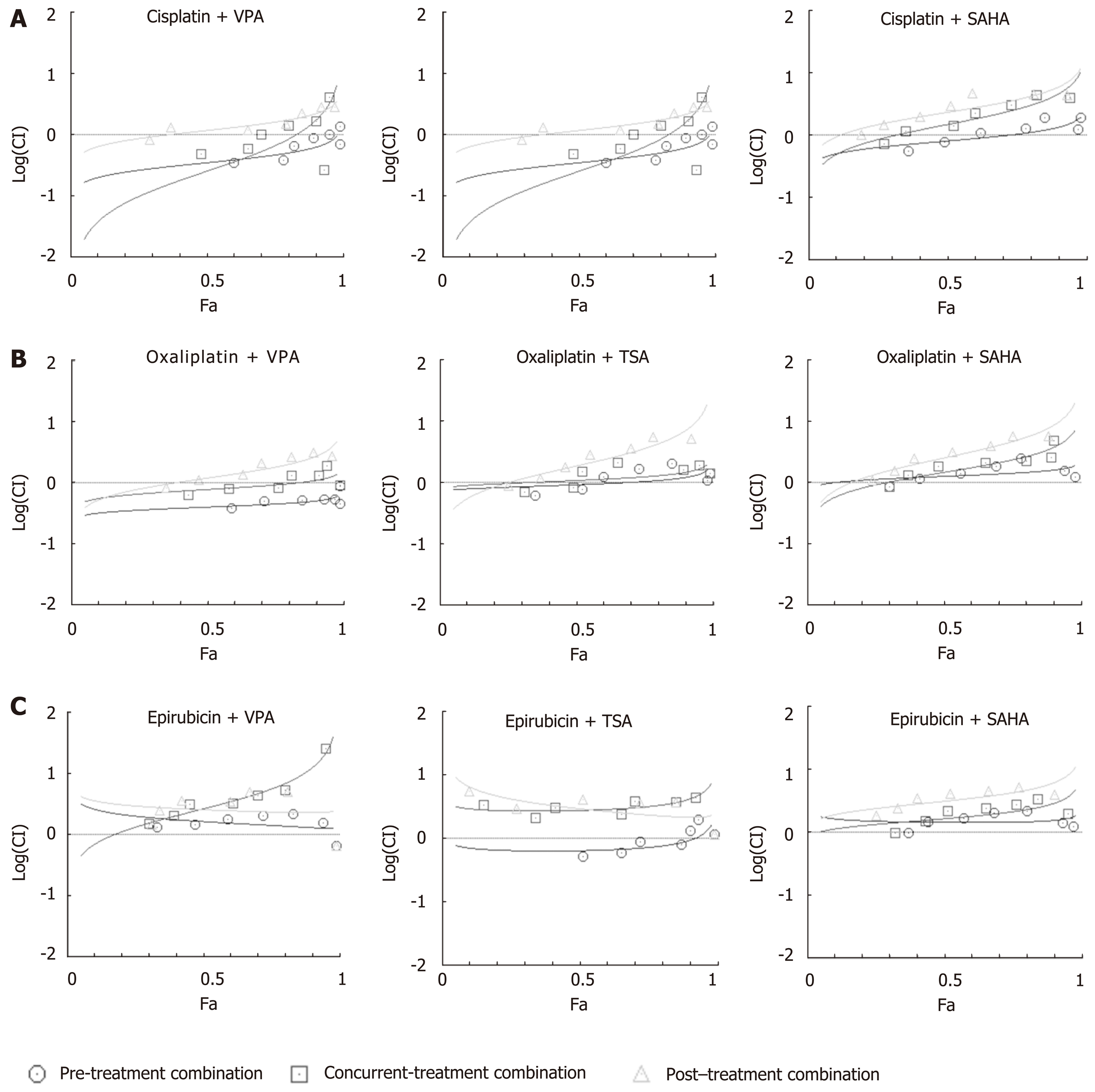
In order to assess which combination regimes of chemotherapeutic drugs and HDACi have a synergistic effect, median effect plot was generated using the combined doses of drugs and FA values. The data were quantitatively analyzed using CI at FA levels of 0.5, 0.75 and 0.95 (Figure 4 and Table 2). At an FA value of 0.5, concurrent and pre-combination regimes of VPA with cisplatin or oxaliplatin, pre-combination of TSA or SAHA with cisplatin, and pre-combination of TSA and epirubicin showed synergistic effects, whereas all other combination regimes showed antagonistic effects. In continuation, at an FA value of 0.75, pre-treatment and concurrent combination regimes of VPA or TSA with cisplatin or oxaliplatin showed synergistic effects; however, all other combinations showed additive or antagonistic effects. Further, at an FA level of 0.95, only pre-combination of VPA with cisplatin or oxaliplatin showed synergistic effects; however, all other combinations showed antagonistic effects. In conclusion, post-treatment with VPA, TSA or SAHA did not have any synergistic effect when combined with cisplatin, oxaliplatin or epirubicin. VPA was found to have a more synergistic effect in the pre-treatment combination regime with cisplatin and oxaliplatin.
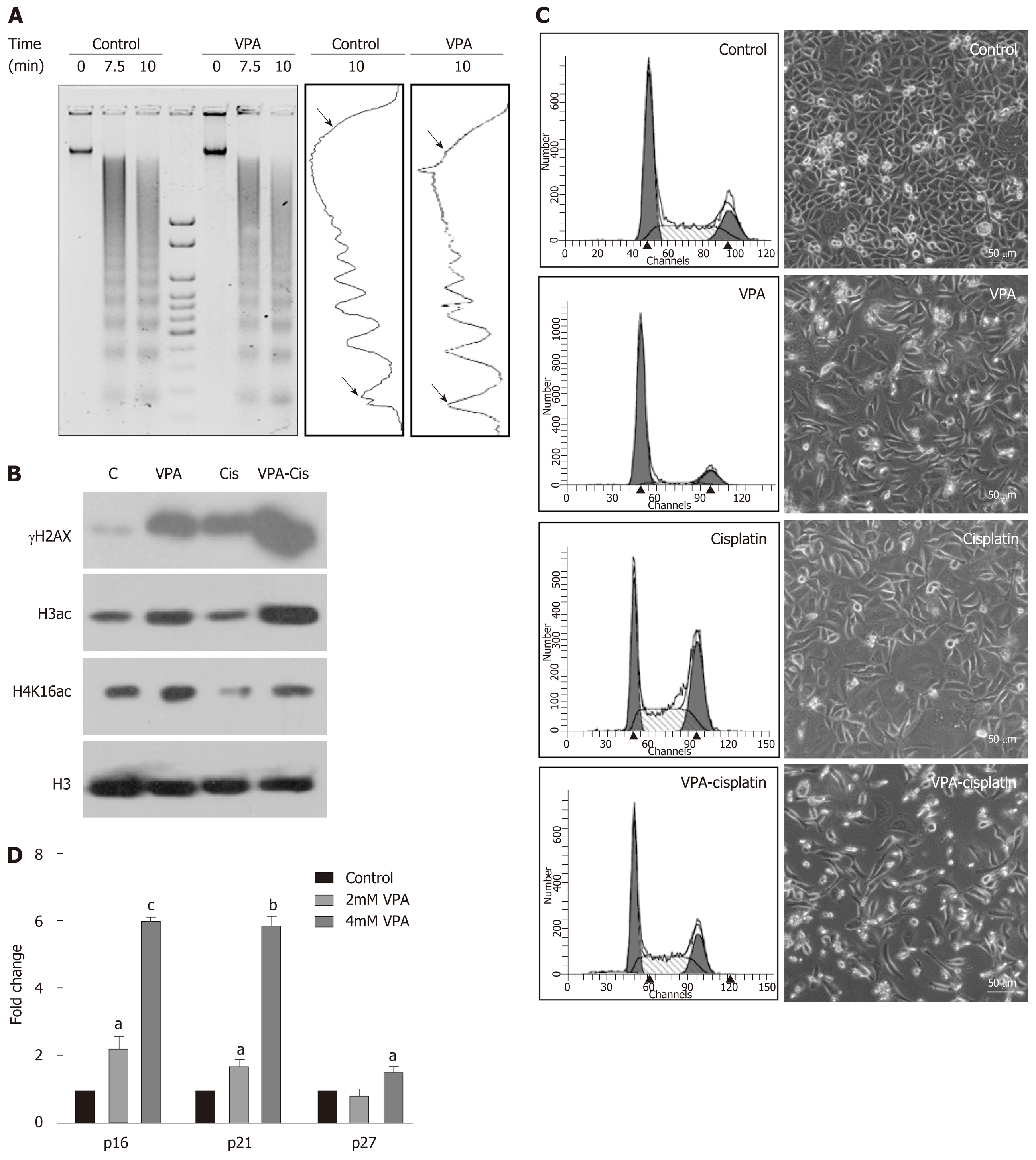
The synergistic effect of the pre-treatment combination regime on DNA damage, histone acetylation and cell cycle was investigated following treatment of AGS cells with IC25 doses of VPA and cisplatin either alone or in combination. MNase assays after VPA treatment suggested an increased intensity of mono- and di-nucleosomes, with a decrease in high molecular weight DNA, indicating chromatin relaxation in VPA-treated cells compared to control (Figure 5A). In coherence, levels of H3Kac and H4K16ac increase after VPA treatment (Figure 5B). H4K16ac is reported to prevent higher order chromatin organization, and therefore its increase indicated open chromatin[24]. The increased acetylation and relaxation of chromatin further correlated with an increased level of γH2AX after combinatorial treatment compared to VPA and cisplatin alone (Figure 5B). Interestingly, H4K16ac decreased after cisplatin treatment alone, indicating the compaction of chromatin. This was likely an outcome of G2/M arrest post-cisplatin treatment, which may lead to poor drug binding to chromatin. VPA treatment, on the other hand, arrests the cells in G1 phase, leading to an open chromatin conformation to enhance drug binding (Figure 5C).
Gatekeeper tumor suppressor genes are known to be repressed in GC by an HDAC-mediated mechanism[25,26]. Levels of tumor suppressors were assessed after IC25 and IC50 treatment of VPA for 24 h. A two-fold increase in p16, p21 and p27 was observed, suggesting the involvement of tumor suppressor-mediated cell cycle arrest, and the observed effects of cell death (Figure 5D). Taken together, results indicate that the pre-treatment regime of VPA opens chromatin, increases the expression of tumor suppressor genes, and enhances cisplatin binding to chromatin, ultimately leading to more cell death.
The pre-treatment regime of HDACi, VPA alone or combined with cisplatin was further explored in vivo with a xenograft tumor model using AGS cells (Figure 6A and B). After 3 wk of treatment, a change in tumor volume was observed in VPA, cisplatin, and VPA followed by cisplatin-treated groups. In vivo, cisplatin and VPA alone showed a similar decrease in tumor volume, however a 3X decrease in tumor volume was observed in the combinatorial treatment group at the end of 5 wk (Figure 6C). Thus, the pre-treatment regime showed a synergistic anticancer effect in the xenograft tumor model. Drug toxicity, as assessed by a decrease in weight, was minimal, with a 15% weight loss in the combination group, indicating better drug tolerance (Figure 6D). Histopathological examination of different tumor tissues showed decreased levels of viable cells (20%-55%) in the case of the combinatorial treatment group compared to the control group (70%-90%). Interestingly, infiltration of inflammatory cells was also observed in the combination group. Also, a greater number of mitotic cells was observed in cisplatin alone and the combinatorial group. Moreover, pleomorphic, hyperchromatic nuclei were observed in the combinatorial-treated group compared to control, suggesting chromatin-associated alterations after drug treatments (Figure 6E). The preclinical study warrants use of the pre-treatment regime in clinical trials for better HDACi therapy success in solid tumors.
Histone acetylation has a significant effect in modulating chromatin architecture and transcription[8]. Thus, acetylation marks and their modifiers have been studied in cancer for their diagnostic, prognostic and therapeutic potential[9]. The clinical application of HDACi has been disappointing in solid tumors, and the major factor for failure in diverse clinical trials is the concurrent treatment regime, limited combination chemotherapeutic studies, and no patient stratification.
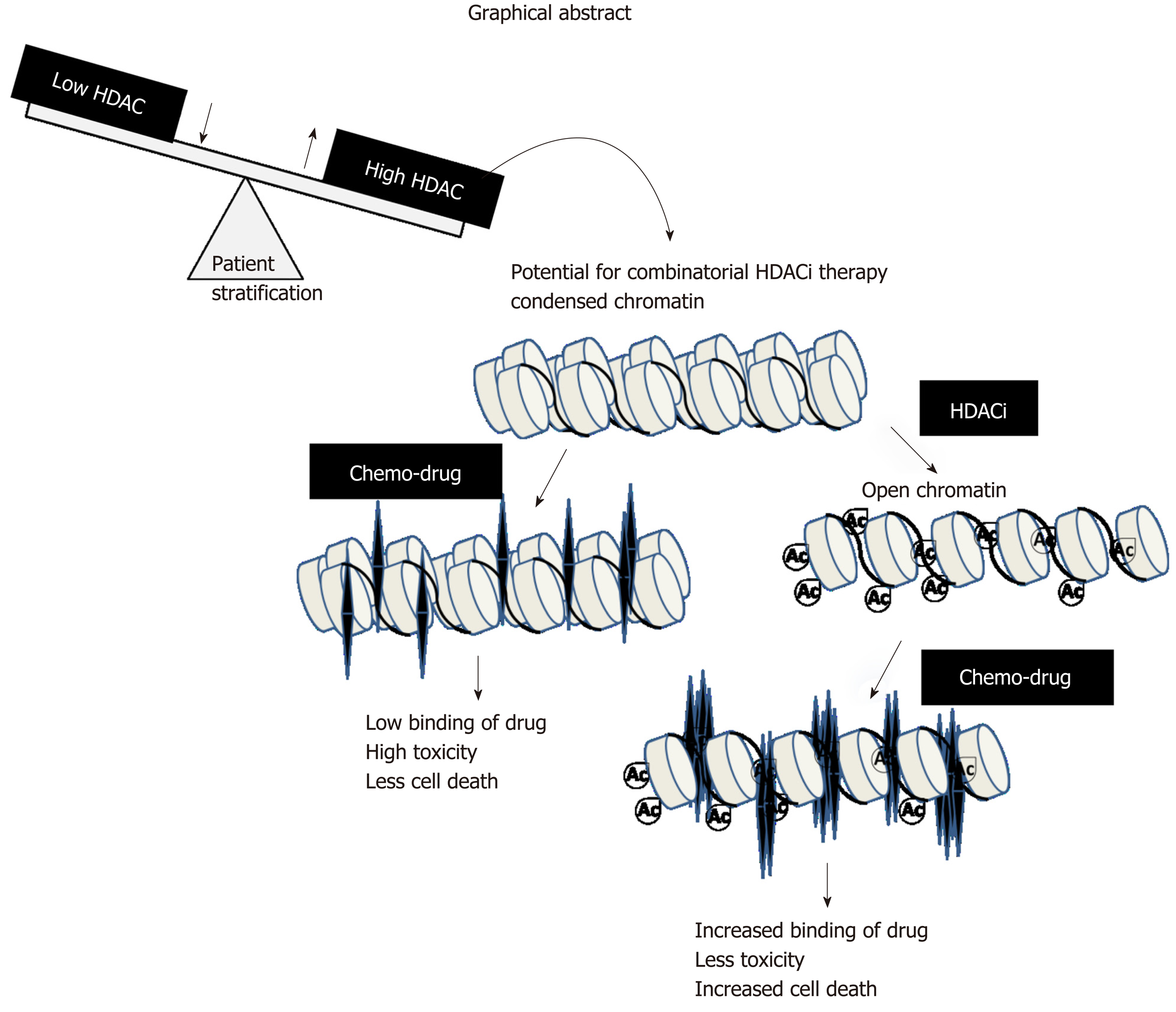
Our data suggest that sensitizing GC cells or in vivo xenografts containing increased levels of Class 1 HDACs with pre-HDACi treatment results in histone hyper-acetylation and relaxed chromatin organization. Increased accessibility of chromatin to DNA-interacting drugs induces DNA damage and cell death. Increases in cell death upon combination of two or more drugs do not form the basis of pre-clinical or clinical studies until the combination shows synergistic effects. All pre-treatment combinations of HDACi and chemotherapeutic drugs showed higher percentages of cell death at low combined doses; however, only VPA with oxaliplatin or cisplatin was found to be best, due to their synergistic effects across FA values from 0.5 to 0.95. Thus, in pre-clinical and clinical settings, the pre-treatment approach with HDACi would allow for a low dosage of chemotherapeutic drugs with similar dose-related cell toxicities. Earlier, Mutze et al[27] showed the importance of HDACi-SAHA pre-treatment to sensitize GC cell lines. The synergistic effect was further recapitulated in a pre-clinical in vivo model, wherein a decrease in tumor volume was observed in the pre-treatment group with low toxicity compared to the cisplatin only and VPA groups. Body weight in the VPA-treated group was not altered compared to the VPA/cisplatin-treated group. This suggests that VPA alone as a sensitizer has no appreciable side effects in the pre-clinical study. Earlier studies have shown that thioredoxin (Trx) levels play an important role in determining HDACi-induced cell death in cancer cells[28]. Normal cells have relatively higher levels of Trx, and therefore could account, in part, for the low toxicity observed in the pre-clinical protocol. Moreover, the cells are arrested in the G1 phase as opposed to the G2/M phase in the case of cisplatin treatment; the G1 phase with more open chromatin thus favors enhanced cisplatin binding and more cell death as suggested by increased γH2AX levels. A recent phase II study in GC with vorinostat as a first-line therapy with capecitabine and cisplatin did not meet its expected outcome[29]. In this study, vorinostat was administered concurrent with chemotherapeutic drugs, suggesting vorinostat had insufficient time to enforce a chromatin modulatory effect, leading to weak drug binding. The prerequisite for changing chromatin organization by HDACi in the case of multiple solid tumors is strengthened by phase I clinical trials with VPA and the topoisomerase II inhibitor epirubicin[30]. In this study, a 48 h pre-exposure of VPA was found to be essential to obtain synergistic outcomes with epirubicin. The pre-treatment combinatorial chemotherapy approach could achieve a positive outcome with acceptable toxicities in heavily treated and previously anthracycline-resistant tumors.
Decades of research involving HDACi and chemotherapeutic drugs have failed to take into account the HDAC levels or activity status of patients, resulting in inappropriate HDACi dose administration. The Neck-V-CHANCE trial will be the first clinical trial to test the efficacy of HDACi pre-treatment. In this ongoing head and neck cancer trial, valproic acid will be administered 2 wk prior to the administration of cisplatin and cetuximab. However, this trial also does not stratify patients based on HDAC levels[31]. Our HDAC activity correlative data linking HDAC expression with histone acetylation, and the presence of heterogeneous HDAC activity and transcript levels in patients, provides sufficient evidence for categorizing the patients for HDACi therapy. Weichert et al[32] showed that approximately 71% (209/293) of GC patients are positive for the expression of either all three or one of the three class 1 HDAC isoforms. Also, Mutze et al[27] highlights that high expression of HDAC1/2 in GC does not relate to response and overall survival. The latest publication by Jiang et al[33] showed an association between high HDAC1 (60% patients) with larger tumor size, tumor grade, lymph node metastasis and lymphovascular invasion, making it an independent prognostic factor for GC. Our in silico TCGA data analysis suggests that 24% of GC patients have higher expression levels of class 1 HDACs. This explains the need for prior assessment of class 1 HDAC levels HDACi therapy patient stratification. This will also assist in defining the dose of HDACi in pre-treatment regimes with DNA-interacting chemotherapeutic drugs for better therapeutic potential.
In conclusion, our results establish a strong rationale for exploring pre-treatment regimes in stratified patients groups with HDACi in clinical trials. The proposed mechanism is through the attainment of open chromatin architecture, and the accumulation of activating histone marks for enhanced binding of DNA-interacting chemotherapeutic drugs (graphical abstract, Figure 7). This worthwhile strategy may become more successful in overcoming the limitations of epi-drugs in solid tumor treatment, and may increase therapeutic outcomes with minimal chemotherapeutic toxicity in the clinic.
The authors are thankful to departments of Flow Cytometry and Mr. Mahesh Pawar, Laboratory Animal Facility, ACTREC, Ms. Kanchan Hariramani and Mr. Narendra Varma, trainees at Gupta laboratory for their assistance during experimental work. The authors are also thankful to members of Gupta laboratory for critical reading of the manuscript.
Alterations of the epigenome play an important role during the process of gastric carcinogenesis. Therefore, drugs like histone deacetylase inhibitors (HDACi) are being explored for their anti-tumor activity.
Identify alterations in the epigenetic milieu of gastric cancer, and check whether the concomitant usage of HDACi with chemotherapeutic drugs increases the drug’s efficacy.
This study aimed to reveal the most optimal combination of chemotherapeutic drugs, as well as HDACi type, dose and regime (pre, post and concurrent). The biochemical mechanism of action was investigated, and the combination was tested in an in vivo system.
This study utilized paired gastric cancer human samples, along with the gastric adenocarcinoma cell line AGS and immortalized normal counterpart HFE145. The efficacy of several chemotherapeutic agents and HDACi was tested in the AGS cell line, and the final combination was tested in an animal model of gastric cancer.
Gastric cancer patients showed differential HDAC activity and levels. Furthermore, pretreatment of valproic acid followed by cisplatin favors an open chromatin conformation via increased histone acetylation. These changes increase the binding of cisplatin to DNA at lower concentrations. In vivo studies suggest a better response with pretreatment regimes that do not cause toxicity.
This study described that decreased histone acetylation in human gastric cancer tumor samples may be attributed to differential/elevated histone deacetylase activity and expression. Additionally, pre-treatment with HDACi was the most optimal regime that maximally enhanced the cell killing potential of chemotherapeutic drugs. This was achieved by increased intercalation of the drug in chromatin post-HDACi treatment. The pre-treatment of HDACi valproic acid and cisplatin was able to decrease tumor volume in vivo compared to cisplatin alone.
This pre-clinical study provides evidence that pre-treatment of HDACi followed by standard chemotherapeutic agents enhances the effectiveness of the drug. Hence, clinical testing of such combinations may be explored for better management of gastric cancer.
Manuscript source: Invited Manuscript
Specialty type: Gastroenterology and hepatology
Country of origin: India
Peer-review report classification
Grade A (Excellent): A
Grade B (Very good): 0
Grade C (Good): 0
Grade D (Fair): 0
Grade E (Poor): 0
P-Reviewer: Ebrahimifar M S-Editor: Zhou JJ L-Editor: Filipodia E-Editor: Zhang YL
| 1. | Bray F, Ferlay J, Soerjomataram I, Siegel RL, Torre LA, Jemal A. Global cancer statistics 2018: GLOBOCAN estimates of incidence and mortality worldwide for 36 cancers in 185 countries. CA Cancer J Clin. 2018;68:394-424. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 53206] [Cited by in RCA: 55666] [Article Influence: 7952.3] [Reference Citation Analysis (132)] |
| 2. | Denny WA. DNA-intercalating ligands as anti-cancer drugs: prospects for future design. Anticancer Drug Des. 1989;4:241-263. [PubMed] |
| 3. | Martínez R, Chacón-García L. The search of DNA-intercalators as antitumoral drugs: what it worked and what did not work. Curr Med Chem. 2005;12:127-151. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 348] [Cited by in RCA: 348] [Article Influence: 17.4] [Reference Citation Analysis (0)] |
| 4. | Catalano MG, Fortunati N, Pugliese M, Poli R, Bosco O, Mastrocola R, Aragno M, Boccuzzi G. Valproic acid, a histone deacetylase inhibitor, enhances sensitivity to doxorubicin in anaplastic thyroid cancer cells. J Endocrinol. 2006;191:465-472. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 81] [Cited by in RCA: 91] [Article Influence: 4.8] [Reference Citation Analysis (0)] |
| 5. | Niitsu N, Kasukabe T, Yokoyama A, Okabe-Kado J, Yamamoto-Yamaguchi Y, Umeda M, Honma Y. Anticancer derivative of butyric acid (Pivalyloxymethyl butyrate) specifically potentiates the cytotoxicity of doxorubicin and daunorubicin through the suppression of microsomal glycosidic activity. Mol Pharmacol. 2000;58:27-36. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 20] [Cited by in RCA: 20] [Article Influence: 0.8] [Reference Citation Analysis (0)] |
| 6. | Wittenburg LA, Bisson L, Rose BJ, Korch C, Thamm DH. The histone deacetylase inhibitor valproic acid sensitizes human and canine osteosarcoma to doxorubicin. Cancer Chemother Pharmacol. 2011;67:83-92. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 47] [Cited by in RCA: 49] [Article Influence: 3.5] [Reference Citation Analysis (0)] |
| 7. | Bannister AJ, Kouzarides T. Regulation of chromatin by histone modifications. Cell Res. 2011;21:381-395. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 3359] [Cited by in RCA: 3991] [Article Influence: 285.1] [Reference Citation Analysis (0)] |
| 8. | Yang XJ, Seto E. HATs and HDACs: from structure, function and regulation to novel strategies for therapy and prevention. Oncogene. 2007;26:5310-5318. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 697] [Cited by in RCA: 747] [Article Influence: 41.5] [Reference Citation Analysis (0)] |
| 9. | Füllgrabe J, Kavanagh E, Joseph B. Histone onco-modifications. Oncogene. 2011;30:3391-3403. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 204] [Cited by in RCA: 210] [Article Influence: 15.0] [Reference Citation Analysis (0)] |
| 10. | Dhanak D, Jackson P. Development and classes of epigenetic drugs for cancer. Biochem Biophys Res Commun. 2014;455:58-69. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 65] [Cited by in RCA: 57] [Article Influence: 5.2] [Reference Citation Analysis (0)] |
| 11. | Chen JH, Zheng YL, Xu CQ, Gu LZ, Ding ZL, Qin L, Wang Y, Fu R, Wan YF, Hu CP. Valproic acid (VPA) enhances cisplatin sensitivity of non-small cell lung cancer cells via HDAC2 mediated down regulation of ABCA1. Biol Chem. 2017;398:785-792. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 23] [Cited by in RCA: 36] [Article Influence: 4.5] [Reference Citation Analysis (0)] |
| 12. | Terranova-Barberio M, Roca MS, Zotti AI, Leone A, Bruzzese F, Vitagliano C, Scogliamiglio G, Russo D, D'Angelo G, Franco R, Budillon A, Di Gennaro E. Valproic acid potentiates the anticancer activity of capecitabine in vitro and in vivo in breast cancer models via induction of thymidine phosphorylase expression. Oncotarget. 2016;7:7715-7731. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 49] [Cited by in RCA: 60] [Article Influence: 6.7] [Reference Citation Analysis (0)] |
| 13. | Marchion DC, Bicaku E, Daud AI, Sullivan DM, Munster PN. Valproic acid alters chromatin structure by regulation of chromatin modulation proteins. Cancer Res. 2005;65:3815-3822. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 151] [Cited by in RCA: 151] [Article Influence: 7.6] [Reference Citation Analysis (0)] |
| 14. | Oronsky B, Oronsky N, Scicinski J, Fanger G, Lybeck M, Reid T. Rewriting the epigenetic code for tumor resensitization: a review. Transl Oncol. 2014;7:626-631. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 34] [Cited by in RCA: 32] [Article Influence: 2.9] [Reference Citation Analysis (0)] |
| 15. | Ahuja N, Easwaran H, Baylin SB. Harnessing the potential of epigenetic therapy to target solid tumors. J Clin Invest. 2014;124:56-63. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 104] [Cited by in RCA: 113] [Article Influence: 10.3] [Reference Citation Analysis (0)] |
| 16. | Marchion DC, Bicaku E, Daud AI, Richon V, Sullivan DM, Munster PN. Sequence-specific potentiation of topoisomerase II inhibitors by the histone deacetylase inhibitor suberoylanilide hydroxamic acid. J Cell Biochem. 2004;92:223-237. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 145] [Cited by in RCA: 142] [Article Influence: 7.1] [Reference Citation Analysis (0)] |
| 17. | Khan SA, Amnekar R, Khade B, Barreto SG, Ramadwar M, Shrikhande SV, Gupta S. p38-MAPK/MSK1-mediated overexpression of histone H3 serine 10 phosphorylation defines distance-dependent prognostic value of negative resection margin in gastric cancer. Clin Epigenetics. 2016;8:88. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 30] [Cited by in RCA: 28] [Article Influence: 3.1] [Reference Citation Analysis (0)] |
| 18. | Alexander S, Swatson WS, Alexander H. Pharmacogenetics of resistance to Cisplatin and other anticancer drugs and the role of sphingolipid metabolism. Methods Mol Biol. 2013;983:185-204. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 4] [Cited by in RCA: 3] [Article Influence: 0.3] [Reference Citation Analysis (0)] |
| 19. | Chou TC. Drug combination studies and their synergy quantification using the Chou-Talalay method. Cancer Res. 2010;70:440-446. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 3361] [Cited by in RCA: 4251] [Article Influence: 283.4] [Reference Citation Analysis (0)] |
| 20. | Sharma AK, Bhattacharya S, Khan SA, Khade B, Gupta S. Dynamic alteration in H3 serine 10 phosphorylation is G1-phase specific during ionization radiation induced DNA damage response in human cells. Mutat Res. 2015;773:83-91. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 16] [Cited by in RCA: 17] [Article Influence: 1.7] [Reference Citation Analysis (0)] |
| 21. | Cerami E, Gao J, Dogrusoz U, Gross BE, Sumer SO, Aksoy BA, Jacobsen A, Byrne CJ, Heuer ML, Larsson E, Antipin Y, Reva B, Goldberg AP, Sander C, Schultz N. The cBio cancer genomics portal: an open platform for exploring multidimensional cancer genomics data. Cancer Discov. 2012;2:401-404. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 9144] [Cited by in RCA: 12699] [Article Influence: 976.8] [Reference Citation Analysis (0)] |
| 22. | Gao J, Aksoy BA, Dogrusoz U, Dresdner G, Gross B, Sumer SO, Sun Y, Jacobsen A, Sinha R, Larsson E, Cerami E, Sander C, Schultz N. Integrative analysis of complex cancer genomics and clinical profiles using the cBioPortal. Sci Signal. 2013;6:pl1. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 8187] [Cited by in RCA: 11182] [Article Influence: 931.8] [Reference Citation Analysis (0)] |
| 23. | Chou TC, Talalay P. Quantitative analysis of dose-effect relationships: the combined effects of multiple drugs or enzyme inhibitors. Adv Enzyme Regul. 1984;22:27-55. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 5258] [Cited by in RCA: 5690] [Article Influence: 138.8] [Reference Citation Analysis (0)] |
| 24. | Shogren-Knaak M, Ishii H, Sun JM, Pazin MJ, Davie JR, Peterson CL. Histone H4-K16 acetylation controls chromatin structure and protein interactions. Science. 2006;311:844-847. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 1370] [Cited by in RCA: 1399] [Article Influence: 73.6] [Reference Citation Analysis (0)] |
| 25. | Richon VM, Sandhoff TW, Rifkind RA, Marks PA. Histone deacetylase inhibitor selectively induces p21WAF1 expression and gene-associated histone acetylation. Proc Natl Acad Sci USA. 2000;97:10014-10019. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 878] [Cited by in RCA: 881] [Article Influence: 35.2] [Reference Citation Analysis (0)] |
| 26. | Herold C, Ganslmayer M, Ocker M, Hermann M, Geerts A, Hahn EG, Schuppan D. The histone-deacetylase inhibitor Trichostatin A blocks proliferation and triggers apoptotic programs in hepatoma cells. J Hepatol. 2002;36:233-240. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 104] [Cited by in RCA: 109] [Article Influence: 4.7] [Reference Citation Analysis (0)] |
| 27. | Mutze K, Langer R, Becker K, Ott K, Novotny A, Luber B, Hapfelmeier A, Göttlicher M, Höfler H, Keller G. Histone deacetylase (HDAC) 1 and 2 expression and chemotherapy in gastric cancer. Ann Surg Oncol. 2010;17:3336-3343. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 50] [Cited by in RCA: 57] [Article Influence: 3.8] [Reference Citation Analysis (0)] |
| 28. | Ungerstedt JS, Sowa Y, Xu WS, Shao Y, Dokmanovic M, Perez G, Ngo L, Holmgren A, Jiang X, Marks PA. Role of thioredoxin in the response of normal and transformed cells to histone deacetylase inhibitors. Proc Natl Acad Sci USA. 2005;102:673-678. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 383] [Cited by in RCA: 398] [Article Influence: 19.9] [Reference Citation Analysis (0)] |
| 29. | Yoo C, Ryu MH, Na YS, Ryoo BY, Lee CW, Kang YK. Vorinostat in combination with capecitabine plus cisplatin as a first-line chemotherapy for patients with metastatic or unresectable gastric cancer: phase II study and biomarker analysis. Br J Cancer. 2016;114:1185-1190. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 24] [Cited by in RCA: 38] [Article Influence: 4.2] [Reference Citation Analysis (0)] |
| 30. | Münster P, Marchion D, Bicaku E, Schmitt M, Lee JH, DeConti R, Simon G, Fishman M, Minton S, Garrett C, Chiappori A, Lush R, Sullivan D, Daud A. Phase I trial of histone deacetylase inhibition by valproic acid followed by the topoisomerase II inhibitor epirubicin in advanced solid tumors: a clinical and translational study. J Clin Oncol. 2007;25:1979-1985. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 166] [Cited by in RCA: 169] [Article Influence: 9.4] [Reference Citation Analysis (0)] |
| 31. | Caponigro F, Di Gennaro E, Ionna F, Longo F, Aversa C, Pavone E, Maglione MG, Di Marzo M, Muto P, Cavalcanti E, Petrillo A, Sandomenico F, Maiolino P, D'Aniello R, Botti G, De Cecio R, Losito NS, Scala S, Trotta A, Zotti AI, Bruzzese F, Daponte A, Calogero E, Montano M, Pontone M, De Feo G, Perri F, Budillon A. Phase II clinical study of valproic acid plus cisplatin and cetuximab in recurrent and/or metastatic squamous cell carcinoma of Head and Neck-V-CHANCE trial. BMC Cancer. 2016;16:918. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 38] [Cited by in RCA: 51] [Article Influence: 5.7] [Reference Citation Analysis (0)] |
| 32. | Weichert W, Röske A, Gekeler V, Beckers T, Ebert MP, Pross M, Dietel M, Denkert C, Röcken C. Association of patterns of class I histone deacetylase expression with patient prognosis in gastric cancer: a retrospective analysis. Lancet Oncol. 2008;9:139-148. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 277] [Cited by in RCA: 299] [Article Influence: 17.6] [Reference Citation Analysis (0)] |
| 33. | Jiang Z, Yang H, Zhang X, Wang Z, Rong R, Wang X. Histone deacetylase-1 as a prognostic factor and mediator of gastric cancer progression by enhancing glycolysis. Hum Pathol. 2019;85:194-201. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 10] [Cited by in RCA: 12] [Article Influence: 1.7] [Reference Citation Analysis (0)] |