Published online May 7, 2020. doi: 10.3748/wjg.v26.i17.2064
Peer-review started: November 20, 2019
First decision: January 7, 2020
Revised: February 13, 2020
Accepted: March 11, 2020
Article in press: March 11, 2020
Published online: May 7, 2020
Processing time: 168 Days and 14.4 Hours
Epigallocatechin gallate (EGCG) is a polyhydroxy phenolic compound extracted from tea and its antitumor effect has received widespread attention. We explored the inhibitory effect of EGCG on dimethylhydrazine (DMH)-induced colorectal cancer (CRC) using a rat model, predicted the interaction between EGCG and CRC target genes using a database, and explained the EGCG associated target pathways and mechanisms in CRC.
To understand the inhibitory mechanisms of EGCG on CRC cell proliferation and identify its pharmacological targets by network pharmacology analysis.
DMH (40 mg/kg, s.c., twice weekly for eight weeks) was used to induce CRC in rats. After model establishment, the rats were administered with EGCG (50, 100, or 200 mg/kg, p.o., once daily for eight weeks) and killed 12 and 20 wk after the start of the experiment. Formation of aberrant crypt foci and tumor was studied by histological analysis. Using network pharmacology analysis, candidate and collective targets of EGCG and CRC were identified, and Gene ontology (GO) and Kyoto Encyclopedia of Genes and Genomes analyses were used to predict the pathways altered by EGCG.
At week 12, high-dose EGCG treatment significantly reduced the tumor formation rate, total number of tumors, cancerous and non-cancerous tumors, tumor volume, ascites formation, and aberrant crypt foci count. At week 20, all three doses of EGCG were effective. Seventy-eight collective targets of EGCG and CRC were identified, of which 28 genes were dysregulated in CRC. Kyoto Encyclopedia of Genes and Genomes and GO analyses showed that the dysregulated genes were enriched in hsa05210 (CRC), hsa04115 (p53 signaling pathway), and hsa04151 (PI3K-Akt signaling pathway), GO:0043124 (negative regulation of I-kappaB kinase/NF-kappaB signaling pathway), GO:0043409 (negative regulation of mitogen-activated protein kinase cascade), and GO:2001244 (positive regulation of intrinsic apoptotic signaling pathway) respectively.
EGCG inhibits the formation of DMH-induced CRC by regulating key pathways involved in tumorigenesis.
Core tip: Our research found that epigallocatechin gallate (EGCG) extracted from tea can inhibit the occurrence of colorectal cancer (CRC) in dimethylhydrazine-induced rat models and predicted the interaction between EGCG and CRC target genes using a bioinformatics database. The Cancer Genome Atlas database was used to further explore the significant differences of 28 genes among 78 intersection target genes and Kyoto Encyclopedia of Genes and Genomes and Gene ontology analyses were performed to explore the pathway and mechanism of EGCG on CRC.
- Citation: Wang Y, Jin HY, Fang MZ, Wang XF, Chen H, Huang SL, Kong DS, Li M, Zhang X, Sun Y, Wang SM. Epigallocatechin gallate inhibits dimethylhydrazine-induced colorectal cancer in rats. World J Gastroenterol 2020; 26(17): 2064-2081
- URL: https://www.wjgnet.com/1007-9327/full/v26/i17/2064.htm
- DOI: https://dx.doi.org/10.3748/wjg.v26.i17.2064
Colorectal cancer (CRC) has now become one of the most common malignancies of the digestive tract in the world[1]. In 2018, there were 1096601 new cases of colon cancer and 704376 new cases of rectal cancer in 185 major countries all over the world. In the same year, about 551269 and 310394 people died of colon and rectal cancer, respectively[2]. Even so, the morbidity and mortality of CRC have been declining slowly and steadily, which could be because of emergence of advanced detection methods, such as colonoscopy, flexible sigmoidoscopy, fecal occult blood test, fecal DNA examination, and barium enema X-ray examination. These have become part of routine cancer screening to detect the occurrence of early intestinal cancer and even polyps[3-5]. Treatment is as important as diagnosis. Although we have many effective treatment methods for CRC, including surgery, chemotherapy, radiotherapy, targeted therapy, and the recently popular immunotherapy techniques, these treatment methods are associated with many adverse effects that might necessitate treatment interruption or cause disease progression[6].
Green tea has gained popularity in the daily health care[7,8]. It contains more than ten effective polyphenols, including catechin, gallic acid, caffeic acid, epicatechin, gallocatechin, catechin gallate, epigallocatechin, epicatechin gallate, and epigallocatechin gallate (EGCG)[8]. When combined with traditional radiation and chemotherapeutic agents, synergistic effects with reduced toxicity have been reported[9]. EGCG, the most abundant polyphenol (50%) in green tea, has the highest activity[10]. Recent studies have shown that EGCG can inhibit tumor cell proliferation and metastasis and promote tumor cell apoptosis through various signaling pathways in vivo and in vitro[11-13]. EGCG probably exerted an anticancer effect, i.e., it halted the proliferation of PC-9 and A431 cells by arresting their cell cycle in the G2/M and G0/G1 phases, respectively[14,15]. EGCG also inhibits the proliferation of LoVo cells and induces their apoptosis by shifting the cell population from the S and M/G2 phases to the G1 phase[16]. It inhibits the growth of CRC cells by inhibiting the activation of receptor tyrosine kinases. In addition, EGCG inhibits the DNA methyltransferase of tumor cells through various signaling pathways, inhibit tumor cell proliferation, and promote apoptosis[17-19].
In this study, we aimed to investigate the inhibitory effects of EGCG on dimethylhydrazine (DMH)-induced CRC in rats. Through network pharmacology analysis, we also aimed to identify EGCG-associated targets and potential pathways in CRC.
Eighty six-week-old male Wistar rats weighing 180-200 g were purchased from Shanghai Laboratory Animal Center, CAS (SLACCAS) (production license number: SCXK 2007-0011). These rats were reared under SPF conditions (Jiangsu Pharmaceutical Biotechnology Co., Ltd.), fed cobalt-60 radiation-sterilized granulated feed, and adapted to laboratory conditions for two weeks. DMH [Aladdin Reagent (Shanghai) Co., Ltd.] was first prepared into a solution at a concentration of 4 mg/mL. EDTA (27 mg) was added to this solution, and the pH was adjusted to 6.5 with 0.1 mol NaOH. EGCG (Hangzhou Hetian Biotechnology Co., Ltd.) was dissolved in distilled water to a concentration of 50 mg/mL. For histological analysis, 4% (w/v) methylene blue solution was diluted in five volumes of distilled water (0.8% w/v). All experiments were conducted in accordance with the requirements of the Chinese Affidavit of the Approval of Animal Ethics and Welfare.
Eighty SPF Wistar rats were randomly divided into five groups as follows: Group 1, Vehicle (vehicle; saline, s.c., twice weekly) group (n = 10); Group 2, DMH (40 mg/kg, s.c., twice weekly) control group (n = 10); Group 3, DMH (40 mg/kg, s.c., twice weekly) + low-dose EGCG (50 mg/kg, p.o., once daily) group (n = 20); Group 4, DMH (40 mg/kg, s.c., twice weekly) + medium-dose EGCG (100 mg/kg, p.o., once daily) group (n = 20); Group 5, DMH (40 mg/kg, s.c., twice weekly) + high-dose EGCG (200 mg/kg, p.o., once daily) group (n = 20). DMH injection and EGCG administration both continued for eight weeks. Biweekly body weight measurements were performed to adjust the dosage of DMH. Five rats in each group were killed at week 12 and week 20.
Measurement of body weight and other physiological parameters: Each rat was weighed every two weeks to observe the change in body weight. Ascites formation was determined. The changes in anal and intestinal erosion, redness, swelling, and stool in rats were also recorded and observed. Tumor volume was measured using a Vernier caliper according to the following formula:
Gross tumor volume (mm3)[20]: V = (W2 × L)/2 (V is tumor volume, W is tumor width, L is tumor length).
Tumor formation rate and tumor inhibition rate were also calculated using the following formulas:
Tumor formation rate (%) = (number of tumorigenic rats)/(total number of rats) × 100%.
Tumor inhibition rate (%)[21] = (C-T)/C × 100% (C is average weight of control group tumor, and T is average weight of experimental group tumor).
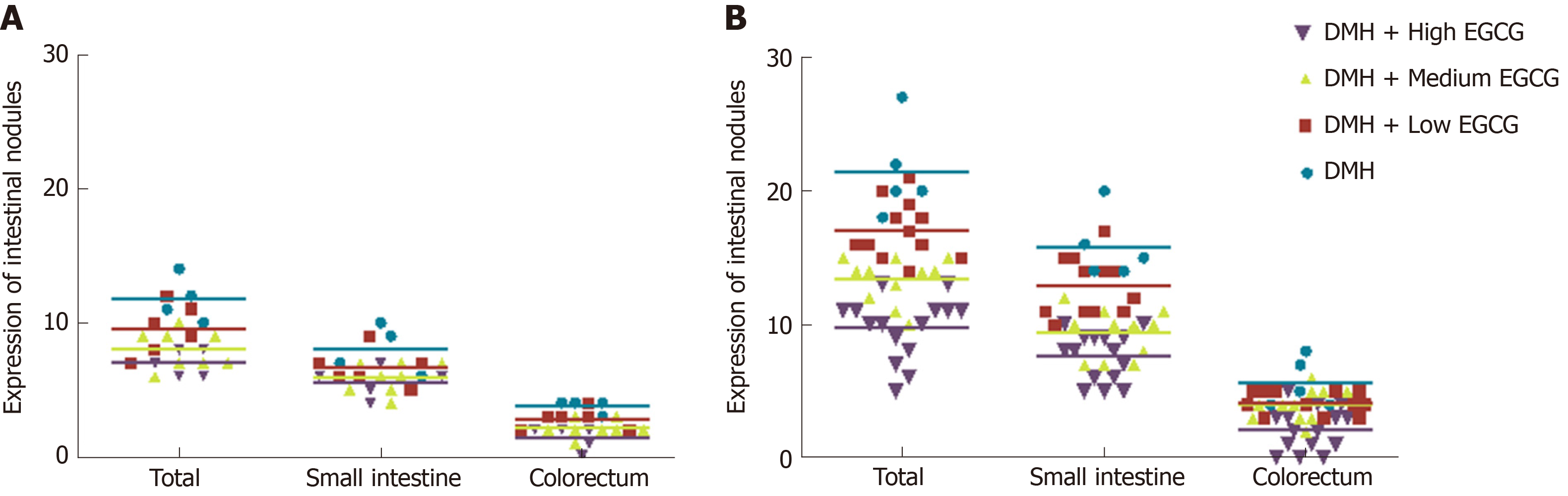
Counting of aberrant crypt foci: The intestinal tissues of rats were isolated and washed with physiological saline. The whole intestine was opened up longitudinally, spread out flat, fixed in 10% neutral buffered formalin solution for 24 h, and stained with 0.2% methylene blue for 3-5 min. The intestinal tract was divided into small intestinal and colorectal parts. The number and size of aberrant crypt foci (ACF) were observed under an optical microscope at 40-200 × magnification.
Histopathology: The intestinal tissues were fixed in 95% ethanol for 24 h, dehydrated with a gradient ethanol series, and embedded in paraffin in blocks. The blocks were sliced into 3 μm thickness using a microtome, dewaxed with xylene, hydrated with a gradient ethanol series, and stained with hematoxylin and eosin. The histopathological changes in small intestinal and colorectal parts were observed by a double blinded histopathologist under an optical microscope.
The data of Canonical SMILES on EGCG was obtained from the PubChem database (https://pubchem.ncbi.nlm.nih.gov/). We finally found totally 87 targets of EGCG through Swiss Target Prediction Database (http://www.swisstargetprediction.ch/).
About 7787 targets of CRC were acquired from the Online Mendelian Inheritance in Man Database (http://omim.org/) and GeneCard (https://www.genecards.org/).
Using the collective targets of EGCG and CRC, a protein–protein interaction (PPI) regulatory network was established through Cytoscape (http://www.cytoscape.org/) and String (https://string-db.org/).
The expression profile of CRC was retrieved from The Cancer Genome Atlas (TCGA) portal (http://tcgadata.nci.nih.gov), and the dysregulated genes were analyzed using the edger R package.
To clarify the function and biological pathways of differentially expressed methylation loci from our data, Kyoto Encyclopedia of Genes and Genomes (KEGG, http://www.kegg.jp/) and the Gene ontology (GO, http://www.geneontology.org) pathway enrichment analyses were performed using the scripts in R. P < 0.05 in GO terms and KEGG was considered significantly enriched by genes.
All data were analyzed by the χ2 test or Fisher's test using SPSS 20.0 (IBM, Armonk, NY, United States), and are represented as the mean ± SD. Multiple groups were compared by one-way ANOVA and P < 0.05 was considered to indicate a significant difference.
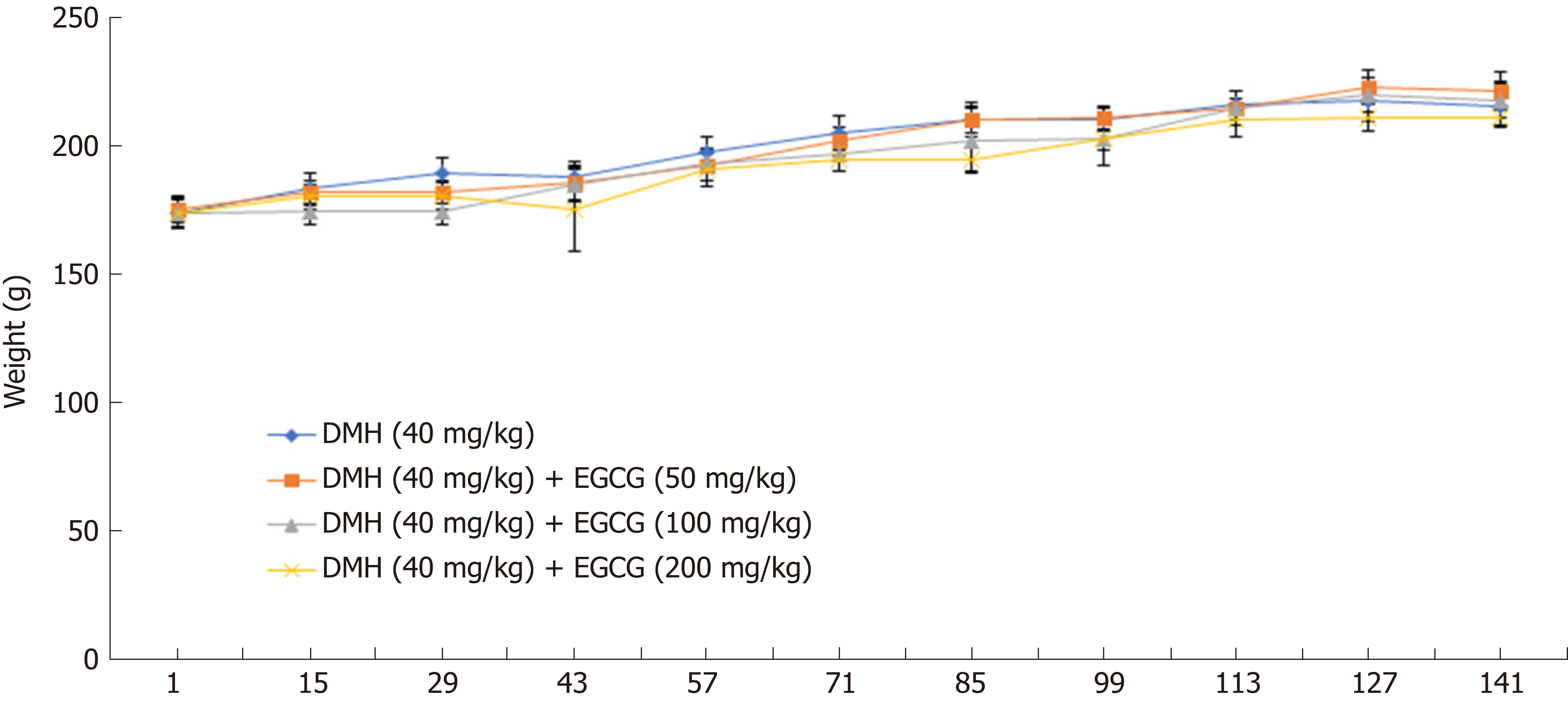
The average weight of the rats in all five groups was at the same baseline. Biweekly body weight measurements showed that rats in the DMH group (Group 2) significantly gained weight compared with rats in the DMH + high-dose EGCG group (Group 5) especially at week 6 (P < 0.01) and week 12 (P < 0.05). Ascites formation in rats in the DMH group (Group 2) at week 6 was significantly (P < 0.01) higher than that in the DMH + high-dose EGCG group (Group 5) (Table 1; Figure 1).
One rat in the DMH group (Group 2) died from an ulcer at the subcutaneous injection site; two rats died in the DMH + low-dose EGCG group (Group 3) from unexplained weight loss; and one rat in the DMH + medium-dose EGCG group died from a fight with other rats (Group 4). All dead rats pathologically manifested intestinal flatulence and small intestine dilatation, accompanied by ascites formation, ileal hemorrhage, necrosis, or colon darkening.
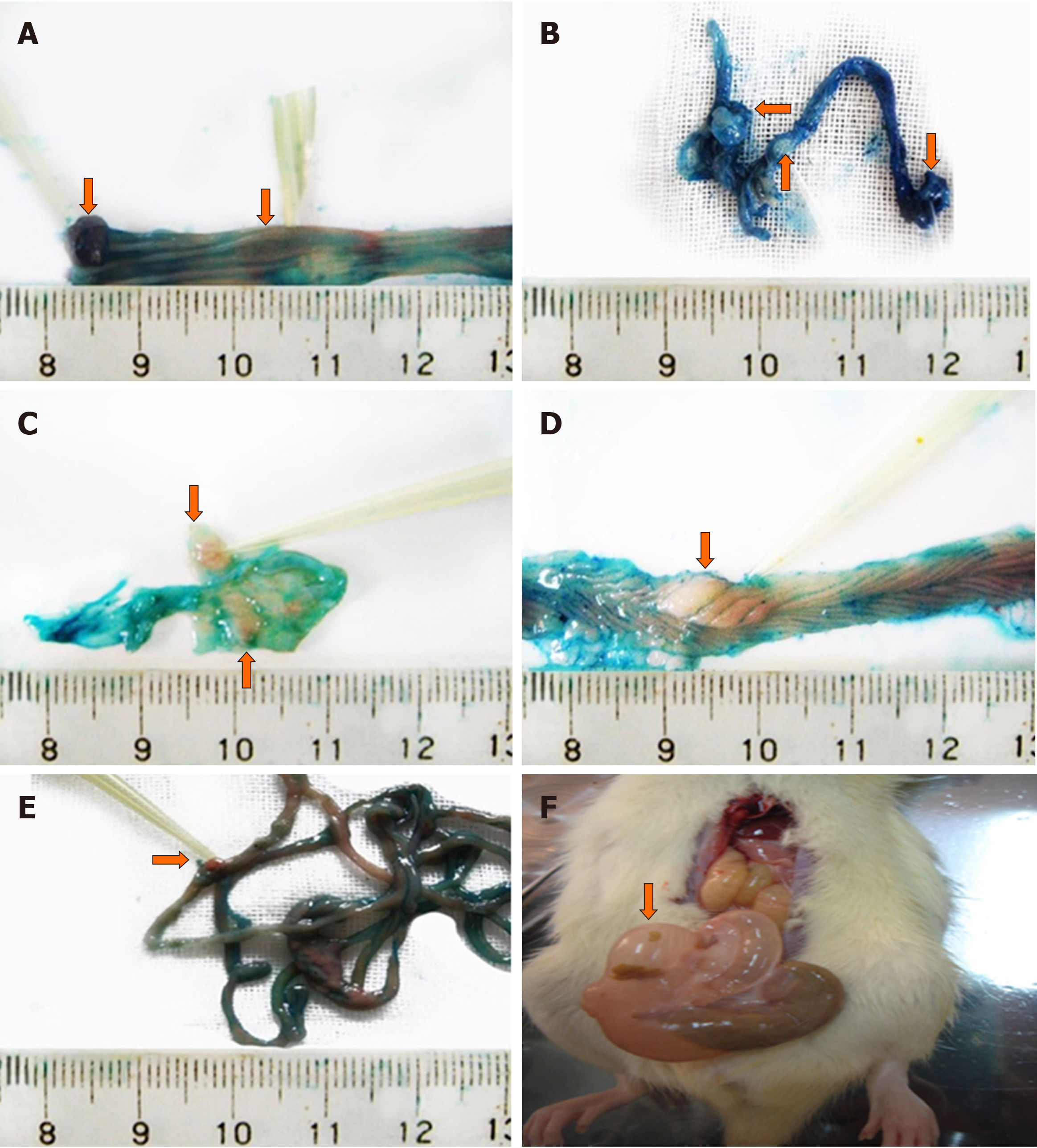
The stools of rats in the vehicle group (Group 1) were normal. Those of rats in the other groups were thin or loose. Four rats in the DMH group (Group 2) presented moderate anal erosion with ascites or bloody ascites, and abundant mesenteric nodules, intestinal wall stiffness, and fold disorder or enlargement in the intestinal mucosa were observed (Figure 2).
Tumor volumes in Groups 4 and 5 were significantly (P < 0.05) smaller than those in the DMH group (Group 2), as shown in Table 2. The tumor volumes in the DMH + medium-dose EGCG group (Group 4) and DMH + high-dose EGCG group (Group 5) were significantly (P < 0.05) smaller than that in the DMH group (Group 2).
| Group | No. of rat | ACF | |||||
| Small intestine | P value | Colorectum | P value | ||||
| 1 | 10 | 0 | - | 0 | - | ||
| 2 | 10 | 5.0 ± 2.0 | - | 2.8 ± 0.9 | - | ||
| 3 | 18 | 4.9 ± 2.8 | 0.93 | 1.8 ± 1.0 | 0.03 | ||
| 4 | 19 | 3.9 ± 1.5 | 0.23 | 1.6 ± 0.5 | 0.01 | ||
| 5 | 20 | 3.9 ± 1.8 | 0.25 | 1.1 ± 0.3 | < 0.01 | ||
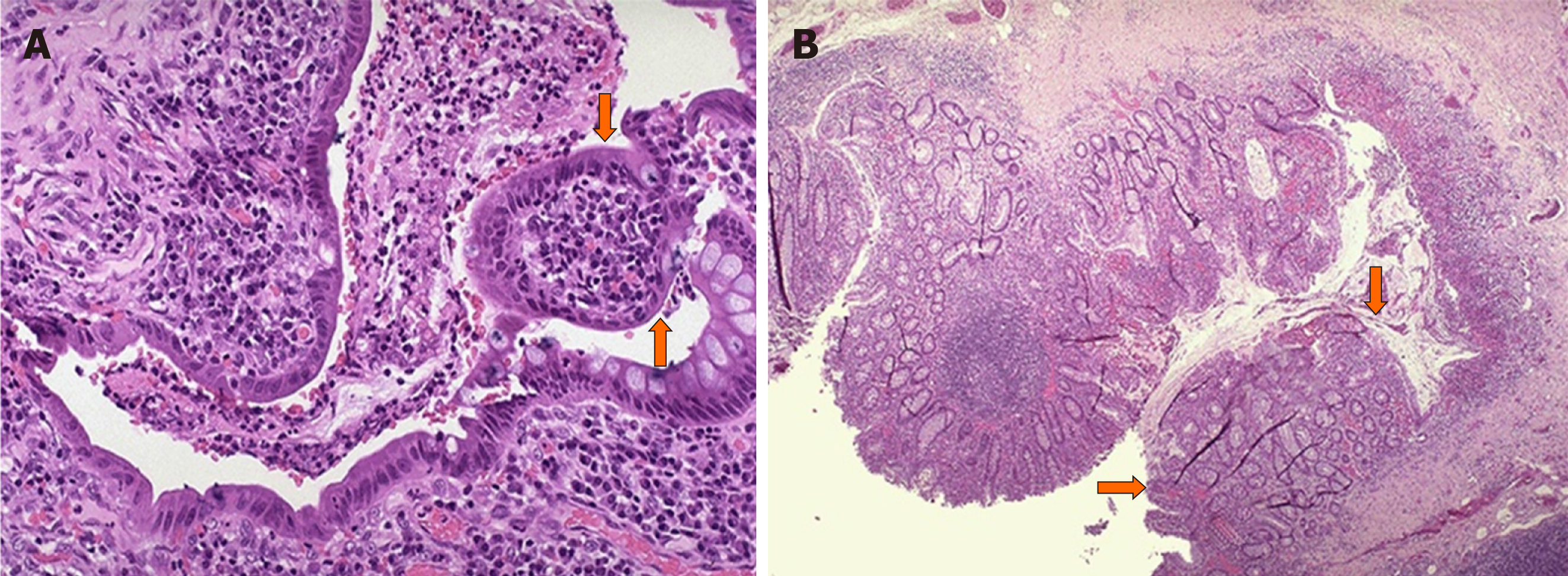
Observation under a light microscope showed that the intestinal mucosa of the vehicle group (Group 1) was intact, with a few orderly crypts, and no malignant tumor was observed. However, varying degrees of abundant ACF appeared in Groups 2-5, presenting as small abscesses with mucosal vascular congestion and inflammatory cell infiltration (Figure 3). The ACF count for the DMH + high-dose EGCG group (Group 5) was significantly (P < 0.01) lower than that in the DMH group (Group 2) (Table 2).
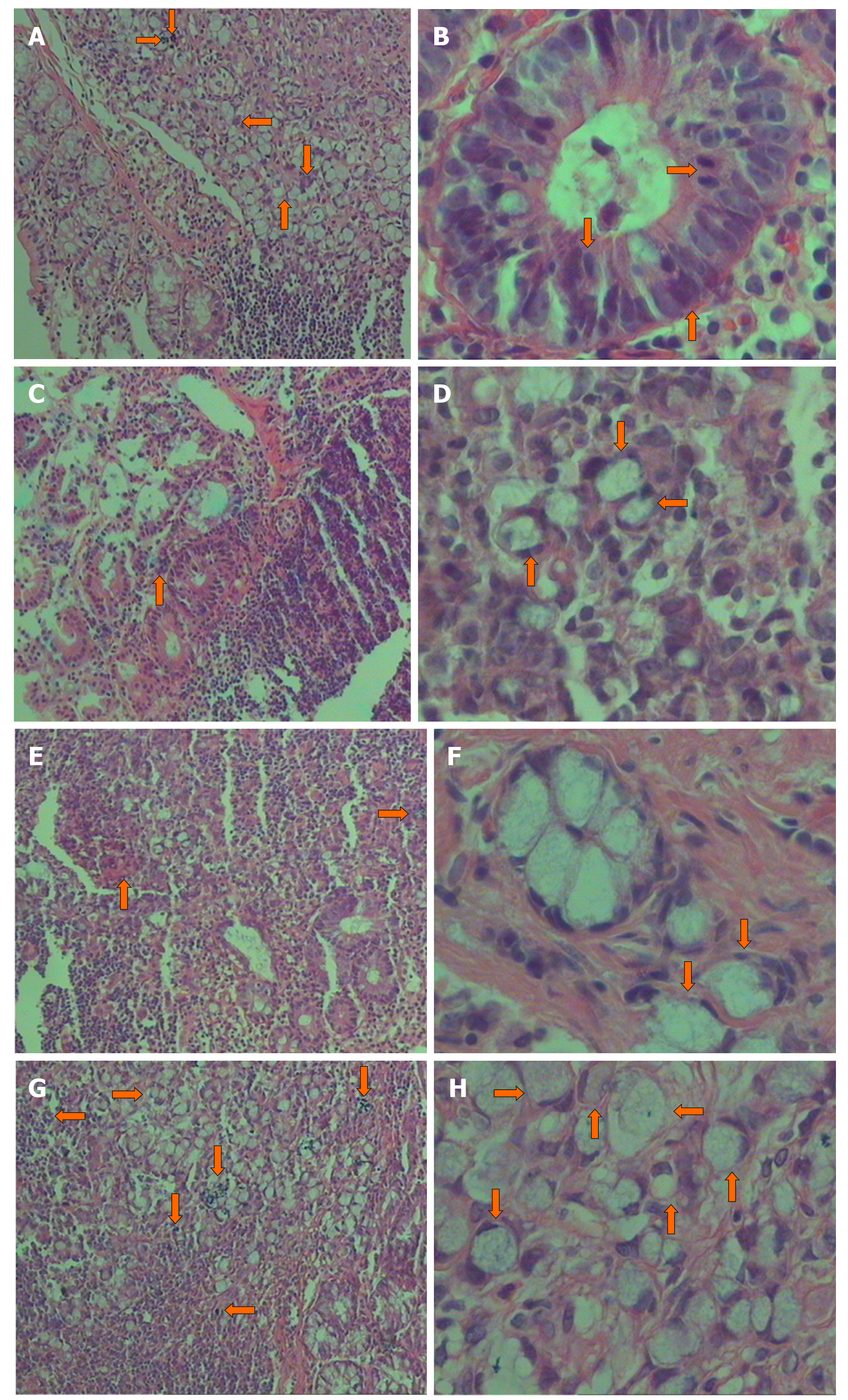
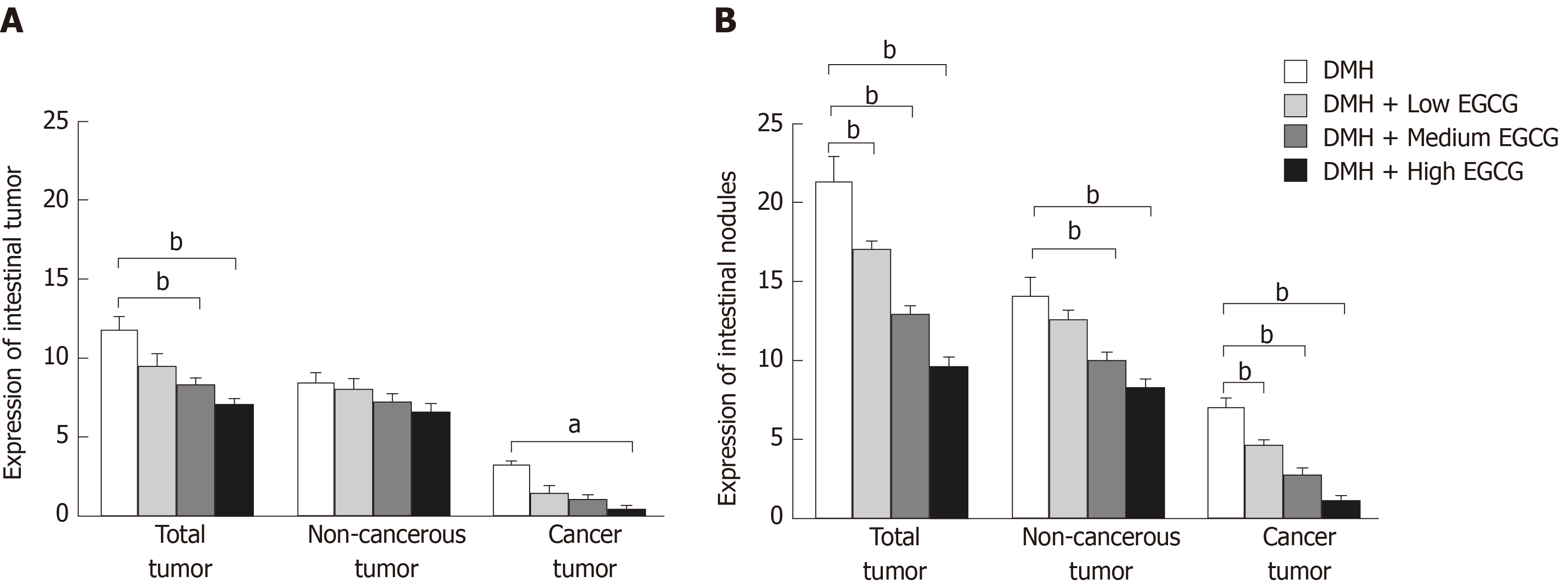
Histopathological analysis showed that 811 intestinal tumors of different sizes were found in the intestines of all rats, among which 178 were cancerous tumors (21.9%) and 633 were non-cancerous tumors (78.1%) determined through pathological examination (Figure 4).
At week 12, the total tumor formation rates in the DMH group (Group 2), DMH + low-dose EGCG group (Group 3), DMH + medium-dose EGCG group (Group 4), and DMH + high-dose EGCG group (Group 5) were 50.13%, 44.27%, 40.18%, and 32.75%, respectively. The total tumor formation rates in Group 4 and Group 5 were significantly (P < 0.01) lower than that in Group 2. It was lower in Group 5 than in Group 3 (P < 0.05) as well. The number of cancerous tumors was lower in Group 5 than in Group 2 (P < 0.05). Tumor inhibition rates in Group 3, Group 4, and Group 5 were 10.26%, 12.19%, and 13.43%, respectively. There was no significant difference in the tumor inhibition rate between each of these three groups (P > 0.05).
At week 20, the tumor formation rates in Groups 2-5 were 77.84%, 63.13%, 51.96%, and 40.12%, respectively. The total tumor formation rates in Group 3-5 were significantly (P < 0.01) reduced compared with that in Group 2. The incidence of cancerous and non-cancerous tumors in Group 3-5 was lower than that in Group 2 (P < 0.01). Group 5 had a statistically significant lower tumor formation rate than Group 3 (P < 0.05). Tumor inhibition rates in Groups 3-5 were 19.33%, 23.58%, and 34.53%, respectively. The tumor inhibition rate in Group 5 was significantly higher than that in Group 3 (P < 0.05) (Table 3, Figure 5 and Figure 6).
| Group | 12 wk | 20 wk | ||||||
| Total | Non-cancerous | Cancerous | Diameter (mm3) | Total | Non-cancerous | Cancerous | Diameter (mm3) | |
| 1 | 0 | 0 | 0 | 0 | 0 | 0 | 0 | 0 |
| 2 | 11.75 ± 1.71 | 8.50 ± 1.29 | 3.25 ± 0.5 | 13.91 ± 1.89 | 21.40 ± 3.44 | 14.20 ± 2.49 | 7.20 ± 1.30 | 17.62 ± 2.21 |
| 3 | 9.50 ± 1.87 | 8.00 ± 1.55 | 1.5 ± 1.05 | 12.22 ± 1.67 | 17.08 ± 2.15b | 12.67 ± 2.23 | 4.76 ± 0.87b | 17.13 ± 1.66 |
| 4 | 8.38 ± 1.19b | 7.25 ± 6.39 | 1.13 ± 0.84 | 10.92 ± 2.01a | 13.36 ± 1.69b | 10.08 ± 1.47b | 2.91 ± 1.30b | 13.60 ± 1.27a |
| 5 | 7.80 ± 1.00bc | 6.60 ± 1.14 | 0.4 ± 0.55a | 6.04 ± 1.23bd | 9.73 ± 2.34bd | 8.40 ± 1.84bc | 1.33 ± 0.90bc | 8.15 ± 1.72bd |
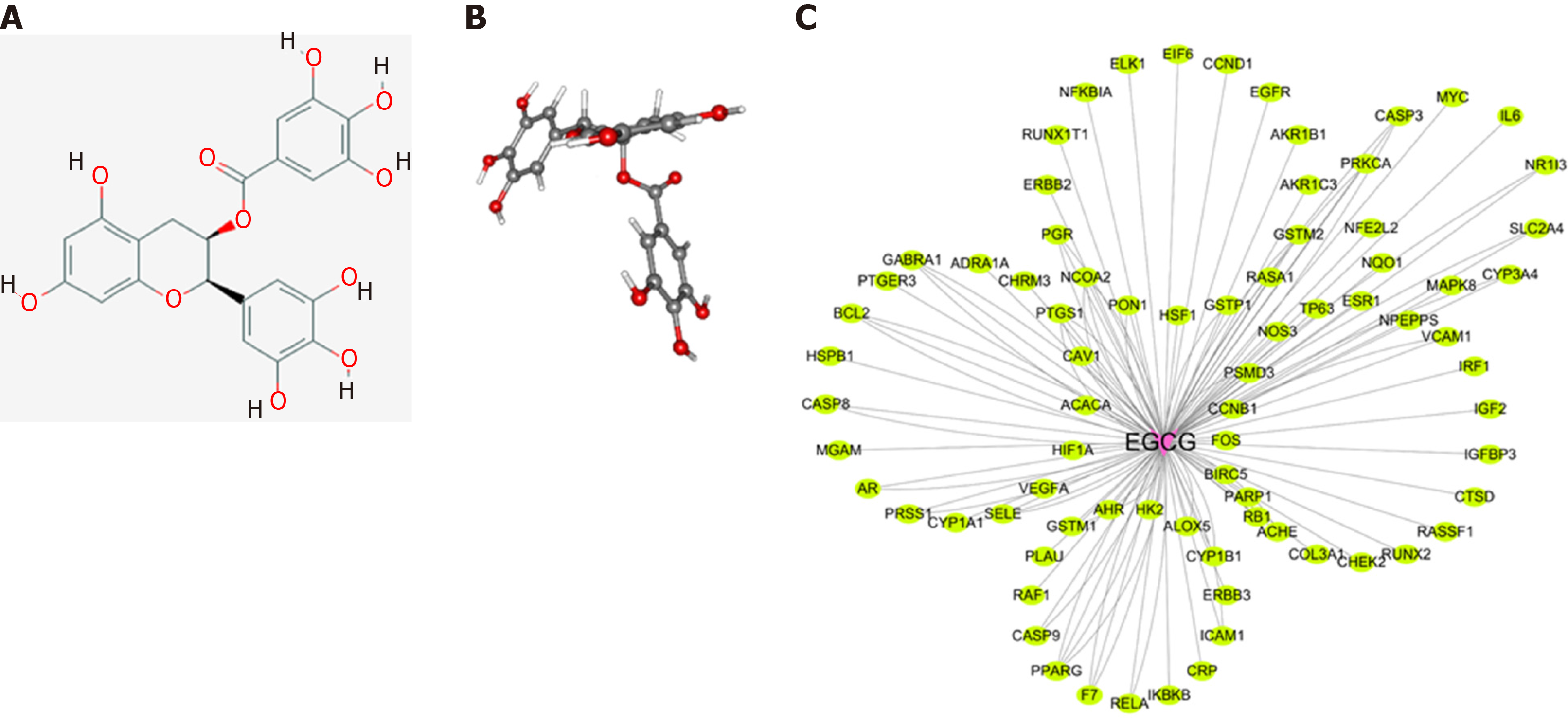
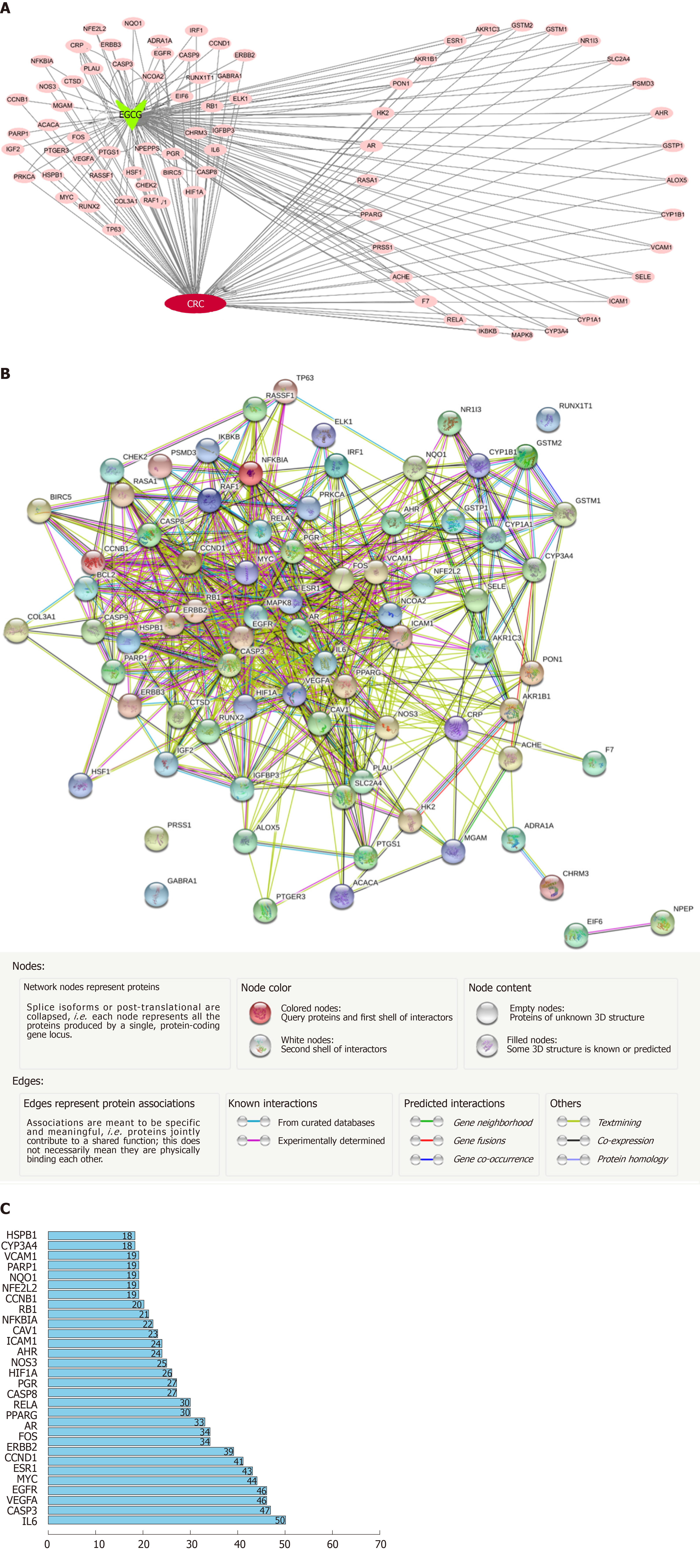
The 2-D and 3-D structures are shown in Figure 7A and 7B, respectively. Based on the Traditional Chinese Medicine Systems Pharmacology Database and Analysis Platform databases, 78 target genes of EGCG were constructed into a protein–protein regulatory network composed of 88 connected nodes (Figure 7C).
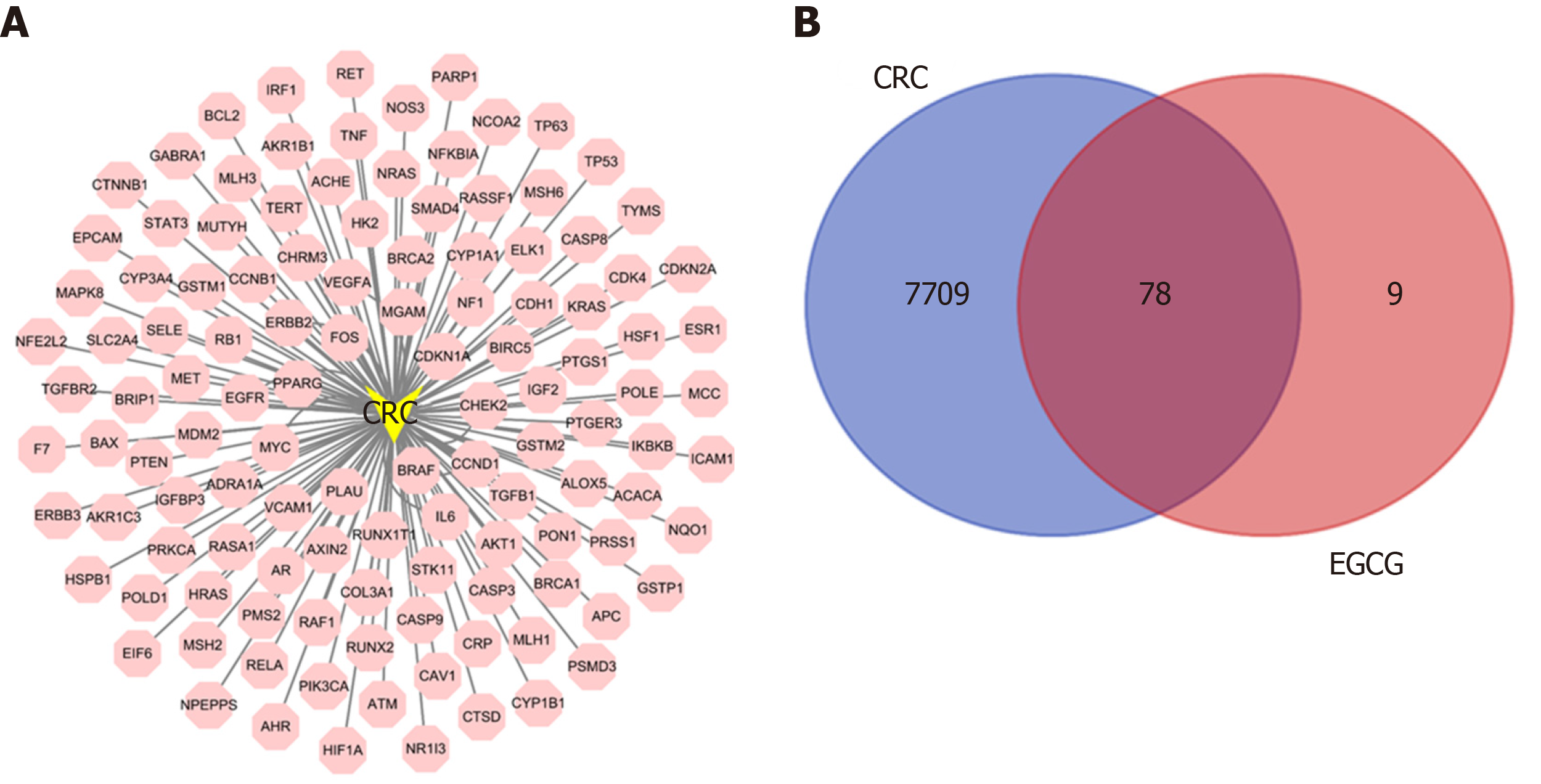
Online Mendelian Inheritance in Man and GeneCard databases were used to identify 7787 targets of CRC for constructing a protein–protein regulation network (Figure 8A). Thus, 78 collective targets of EGCG and CRC were obtained (Figure 8B) and used to construct the protein–protein regulation network (Figure 9A). Then, the 78 target genes were incorporated into a PPI network using String (Figure 9B); the top 30 genes are shown in Figure 9C.
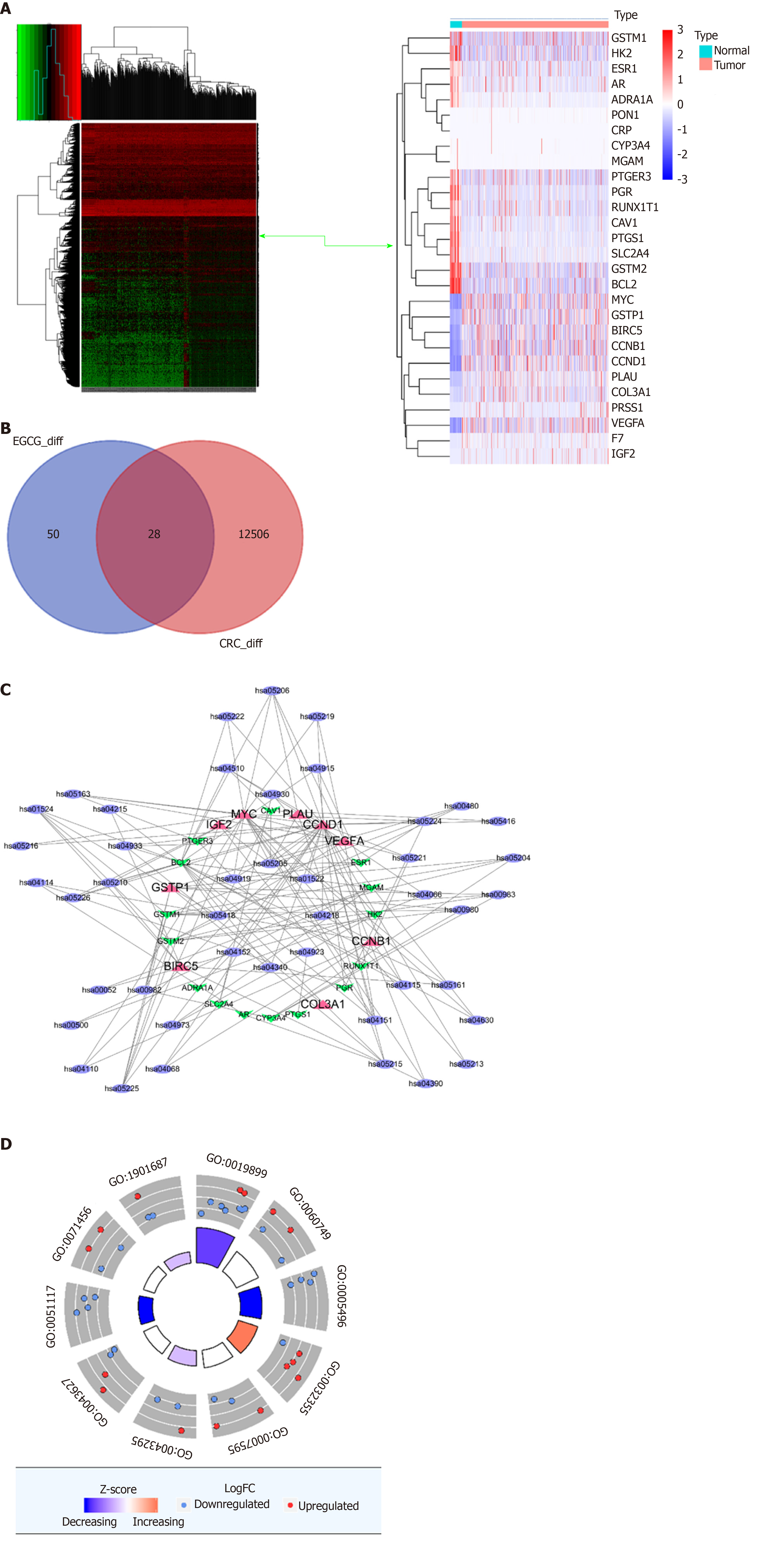
To narrow down the targets of EGCG in CRC, we then analyzed the dysregulated CRC genes in TCGA (Figure 10A) and found that 28 dysregulated genes in CRC were involved in the collective targets of EGCG and CRC (Figure 10B).
KEGG and GO analyses: KEGG analysis showed that the dysregulated genes were enriched in hsa05210 (CRC), hsa04115 (p53 signaling pathway), and hsa04151 (PI3K-Akt signaling pathway) (Figure 10C). GO analysis showed that the dysregulated genes were enriched in GO:0043124 (negative regulation of I-kappaB kinase/NF-kappaB signaling pathway), GO:2001244 (positive regulation of intrinsic apoptotic signaling pathway), and GO:0043409 [negative regulation of mitogen-activated protein kinase (MAPK) cascade] (Figure 10D).
The need for alternative treatments for CRC is mounting. Because green tea, and especially, EGCG in green tea have shown promise in cancer treatment, we sought to evaluate the anticancer mechanisms of EGCG in experimentally induced CRC in rats. EGCG was effective in reducing tumor induction, tumor volume, ascites formation, and ACF counts at all three doses, with the high dose (200 mg/kg) being the most effective.
In our study, we observed that except for the vehicle group rats, the stools of rats in other groups showed different degrees of abnormalities. An abnormal stool sample was characterized by thin consistency, shapelessness, looseness, and having one or more concave holes. Such abnormal stools were noted mostly in the DMH group, and the abnormality of stools was least obvious in the EGCG high-dose group.
In the DMH group, four rats had moderate erosion in the anus. Anatomical analysis revealed varying degrees of ascites or bloody ascites, a large number of tumors of varying sizes in the mesentery, and disordered and thickened intestinal folds in all groups except the control group. The average body weight for all five groups of rats was the same at baseline. Since week 6, the body weight was significantly reduced in the EGCG high-dose group (Group 5) compared with that in the DMH group (P < 0.01). At week 12, the body weight in Group 2 was higher than that in the other groups. At 8 wk, Groups 3 and 4 presented a sharp rise in body weight, and at week 20, body weights in both these groups were higher than that in Group 2. At week 12, the ascites production and tumor formation in the DMH group were greater than those in the other groups, and at this time, the rats in the DMH group were in the compensatory stage, and there was no significant loss of weight caused by cachexia. Anatomical analysis revealed that the DMH group had more ascites than the other groups at week 12. After that, rats in the DMH group gradually developed cachexia, had poor nutritional status, and showed fatigue. Weight loss due to cachexia was more prominent than overall weight gained due to increase in tumor mass; therefore, weight loss in the DMH group was significant. Groups 3 and 4 showed lower ascites and tumor formation and better health status than Group 2, indicating the anticancer effect of EGCG. In Group 5, the tumor suppression effect of EGCG was significant, and there was a reduction in tumor and ascites formation, resulting in lower body weights in this group.
Morphological observation of ACF revealed that the intestinal epithelial cells in the DMH group were larger, more in number, and presented with crypts that changed from round to elliptical shape, increasing the crypt’s peripheral space. The size of the nucleus of the mucosal epithelial cells around ACF increased, the proportion of nucleoplasm increased, the number of goblet cells decreased, and the population of cells in the mitotic phase increased. These changes are the cytological features of precancerous lesions. The process of intestinal carcinogenesis involves conversion of normal glandular epithelium to ACF, adenoma, and finally to tumor. All three stages are characterized by abnormal cell proliferation, inflammatory cell infiltration, and gene mutations[22,23]. It is currently recognized that the pathogenesis of CRC follows the pathway of ACF → adenoma → CRC[24]. Therefore, observing the cell morphology of ACF is crucial for judging whether it is a precancerous lesion. This process is consistent with the occurrence of human CRC.
To determine if the anticancer effects of EGCG are dose-dependent, we administered EGCG at three doses: 50 mg/kg, 100 mg/kg, and 200 mg/kg. From the data, we can see that with the extension of EGCG treatment duration and the increase in dose, the effect of EGCG in inhibiting tumor formation increased. Surprisingly, only the minimum dose of EGCG of 50 mg/kg had a significant inhibitory effect on tumor formation.
We used a network pharmacology approach to identify 78 collective targets of EGCG and CRC. After constructing the PPI network, we found that MYC, epidermal growth factor receptor (EGFR), vascular endothelial growth factor A, CASP3, IL6, and others had a close association with CRC. Overexpression of EIF5A2 promotes invasiveness of CRC by upregulating MTA1 through C-myc to induce epithelial-mesenchymal transition[25]. Besides, EGFR was reported to mediate the re-activation of MAPK signaling, which increases the sensitivity of BRAF mutant CRC to vemurafenib[26]. Vascular endothelial growth factor A, CASP3, and IL6 are closely related to CRC[27-29]. Masahito’s experiments investigated the effects of EGCG on VEGF/vascular EGFR (VEGFR) axis activity and the expression of hypoxia-inducible factor (HIF)-1α, suggesting that the factor promotes angiogenesis by increasing VEGF levels in CRC. EGCG treatment decreased the mRNA expression of HIF-1α, VEGF, EGF, insulin-like growth factor (IGF)-1, IGF-2, and Heregulin within 3 h in SW837 CRC cells, whereas it decreased the expression of VEGFR-2, p-VEGFR-2, p-ERK, p-IGF-1 receptor, and p-Akt proteins within 6 h[30]. This suggests that EGCG can inhibit HIF-1α and several major growth factors related to the VEGF/VEGFR axis to arrest the growth of CRC cells. Therefore, it is necessary to find the target genes of EGCG and CRC intersection through network pharmacology. In the future, we will conduct cellular molecular research on relevant target genes and target pathways through cell experiments, so as to further explore the molecular mechanisms of EGCG in the treatment of CRC.
In conclusion, we have successfully induced CRC in rats using DMH and found that EGCG at low and high doses can inhibit tumor formation. Moreover, the effect of EGCG positively correlates with dose and treatment duration. This reveals the clinical treatment potential of EGCG for CRC. Through bioinformatics analysis, we found 78 target genes that were common to EGCG and CRC. Through KEGG and GO analyses, we found that EGCG could regulate the signaling pathways of CRC, p53, and PI3K-Akt and I-kappaB kinase/NF-kappaB signal pathways, apoptosis signal pathways, and MAPK cascades, respectively. Our findings reveal the potential mechanisms of action and pathways of EGCG in CRC. Further in vitro studies using cell lines would be beneficial to verify the target genes and pathways of EGCG in CRC. One limitation of our study is that our CRC induction was short because of the fact that the tumor formation rate was low. Future studies can be conducted using azoxymethane, which is more potent and stable than DMH and has ability to induce significant tumor formation within a short time.
“Tea polyphenols” is a general term for more than 30 polyphenols in tea, of which epigallocatechin gallate (EGCG) is the main compound. In recent years, a large number of studies have demonstrated that EGCG is a highly effective, biologically active, non-toxic, naturally extracted compound. Its antitumor effect and mechanism of action are constantly being clarified, and they have become a hot topic of research today. However, most of the existing studies only use cell experiments to explain its inhibitory effect.
The aim of our research was to explore whether EGCG can inhibit the occurrence of dimethylhydrazine (DMH)-induced rat intestinal cancer in vivo in rats. We combined bioinformatic data analysis technology to explain the common targets and significant differential genes of EGCG and colorectal cancer (CRC) for the first time and analyze the mechanism of EGCG in CRC at the molecular gene level.
To, using animal experiments and network pharmacological analysis, discover the inhibitory mechanism of EGCG on CRC cell proliferation and clarify its pharmacological targets.
DMH (40 mg/kg) was used to induce CRC in rats twice weekly for 8 wk. Then, different doses of EGCG were administered (50, 100, or 200 mg/kg for 8 wk). Rats were euthanized at week 12 and week 20 to observe aberrant crypt foci and tumor formation. We used bioinformatic analysis to predict the pathways altered by EGCG in CRC.
We discovered that different doses of EGCG had inhibitory effects on aberrant crypt foci and tumors in rats and, as the dose increased, EGCG was more effective in the suppression of tumor formation. Using bioinformatics, we identified 78 target genes that were common to EGCG and CRC and 28 of the 78 genes were identified to have significant differential expression. Using KEGG and GO analyses, we found that EGCG regulated CRC, p53, PI3K-Akt, I-kappaB kinase/NF-kappaB, apoptosis, and mitogen-activated protein kinase signaling pathways.
EGCG can inhibit DMH-induced CRC tumor formation in rats. Moreover, the effect of EGCG positively correlated with dose and treatment duration. This reveals the clinical treatment potential of EGCG for CRC. Our findings reveal the potential mechanisms of action and pathways of EGCG in CRC.
In the future, azoxymethane, which is more potent and stable than DMH, could be used as it has the ability to induce tumor formation in a short time. Additionally, in vitro studies using cell lines would be beneficial to verify the target genes and pathways of EGCG in CRC.
Manuscript source: Unsolicited manuscript
Specialty type: Gastroenterology and hepatology
Country/Territory of origin: China
Peer-review report’s scientific quality classification
Grade A (Excellent): 0
Grade B (Very good): B, B
Grade C (Good): 0
Grade D (Fair): 0
Grade E (Poor): 0
P-Reviewer: Farhat S, Mastoraki A S-Editor: Dou Y L-Editor: Wang TQ E-Editor: Ma YJ
| 1. | Ferlay J, Soerjomataram I, Dikshit R, Eser S, Mathers C, Rebelo M, Parkin DM, Forman D, Bray F. Cancer incidence and mortality worldwide: sources, methods and major patterns in GLOBOCAN 2012. Int J Cancer. 2015;136:E359-E386. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 20108] [Cited by in RCA: 20499] [Article Influence: 2049.9] [Reference Citation Analysis (20)] |
| 2. | Bray F, Ferlay J, Soerjomataram I, Siegel RL, Torre LA, Jemal A. Global cancer statistics 2018: GLOBOCAN estimates of incidence and mortality worldwide for 36 cancers in 185 countries. CA Cancer J Clin. 2018;68:394-424. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 53206] [Cited by in RCA: 55751] [Article Influence: 7964.4] [Reference Citation Analysis (132)] |
| 3. | Malvezzi M, Carioli G, Bertuccio P, Boffetta P, Levi F, La Vecchia C, Negri E. European cancer mortality predictions for the year 2018 with focus on colorectal cancer. Ann Oncol. 2018;29:1016-1022. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 128] [Cited by in RCA: 147] [Article Influence: 24.5] [Reference Citation Analysis (0)] |
| 4. | Currie A, Askari A, Nachiappan S, Sevdalis N, Faiz O, Kennedy R. A systematic review of patient preference elicitation methods in the treatment of colorectal cancer. Colorectal Dis. 2015;17:17-25. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 20] [Cited by in RCA: 22] [Article Influence: 2.2] [Reference Citation Analysis (0)] |
| 5. | Jin H, Tan X, Liu X, Ding Y. The study of effect of tea polyphenols on microsatellite instability colorectal cancer and its molecular mechanism. Int J Colorectal Dis. 2010;25:1407-1415. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 12] [Cited by in RCA: 15] [Article Influence: 1.0] [Reference Citation Analysis (0)] |
| 6. | Singh PP, Sharma PK, Krishnan G, Lockhart AC. Immune checkpoints and immunotherapy for colorectal cancer. Gastroenterol Rep (Oxf). 2015;3:289-297. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 21] [Cited by in RCA: 52] [Article Influence: 5.2] [Reference Citation Analysis (0)] |
| 7. | Yang CS, Wang H, Li GX, Yang Z, Guan F, Jin H. Cancer prevention by tea: Evidence from laboratory studies. Pharmacol Res. 2011;64:113-122. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 170] [Cited by in RCA: 170] [Article Influence: 12.1] [Reference Citation Analysis (0)] |
| 8. | Du GJ, Zhang Z, Wen XD, Yu C, Calway T, Yuan CS, Wang CZ. Epigallocatechin Gallate (EGCG) is the most effective cancer chemopreventive polyphenol in green tea. Nutrients. 2012;4:1679-1691. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 297] [Cited by in RCA: 347] [Article Influence: 26.7] [Reference Citation Analysis (0)] |
| 9. | Suganuma M, Saha A, Fujiki H. New cancer treatment strategy using combination of green tea catechins and anticancer drugs. Cancer Sci. 2011;102:317-323. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 119] [Cited by in RCA: 115] [Article Influence: 7.7] [Reference Citation Analysis (0)] |
| 10. | Matsunaga K, Klein TW, Friedman H, Yamamoto Y. Legionella pneumophila replication in macrophages inhibited by selective immunomodulatory effects on cytokine formation by epigallocatechin gallate, a major form of tea catechins. Infect Immun. 2001;69:3947-3953. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 47] [Cited by in RCA: 42] [Article Influence: 1.8] [Reference Citation Analysis (0)] |
| 11. | Zhu BH, Zhan WH, Li ZR, Wang Z, He YL, Peng JS, Cai SR, Ma JP, Zhang CH. (-)-Epigallocatechin-3-gallate inhibits growth of gastric cancer by reducing VEGF production and angiogenesis. World J Gastroenterol. 2007;13:1162-1169. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in CrossRef: 67] [Cited by in RCA: 70] [Article Influence: 3.9] [Reference Citation Analysis (0)] |
| 12. | Adhami VM, Siddiqui IA, Ahmad N, Gupta S, Mukhtar H. Oral consumption of green tea polyphenols inhibits insulin-like growth factor-I-induced signaling in an autochthonous mouse model of prostate cancer. Cancer Res. 2004;64:8715-8722. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 220] [Cited by in RCA: 216] [Article Influence: 10.8] [Reference Citation Analysis (0)] |
| 13. | Kim R, Emi M, Tanabe K, Arihiro K. Tumor-driven evolution of immunosuppressive networks during malignant progression. Cancer Res. 2006;66:5527-5536. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 328] [Cited by in RCA: 332] [Article Influence: 17.5] [Reference Citation Analysis (0)] |
| 14. | Okabe S, Suganuma M, Hayashi M, Sueoka E, Komori A, Fujiki H. Mechanisms of growth inhibition of human lung cancer cell line, PC-9, by tea polyphenols. Jpn J Cancer Res. 1997;88:639-643. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 140] [Cited by in RCA: 140] [Article Influence: 5.0] [Reference Citation Analysis (0)] |
| 15. | Ahmad N, Feyes DK, Nieminen AL, Agarwal R, Mukhtar H. Green tea constituent epigallocatechin-3-gallate and induction of apoptosis and cell cycle arrest in human carcinoma cells. J Natl Cancer Inst. 1997;89:1881-1886. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 563] [Cited by in RCA: 520] [Article Influence: 18.6] [Reference Citation Analysis (0)] |
| 16. | Tan X, Hu D, Li S, Han Y, Zhang Y, Zhou D. Differences of four catechins in cell cycle arrest and induction of apoptosis in LoVo cells. Cancer Lett. 2000;158:1-6. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 55] [Cited by in RCA: 54] [Article Influence: 2.2] [Reference Citation Analysis (0)] |
| 17. | Fang MZ, Wang Y, Ai N, Hou Z, Sun Y, Lu H, Welsh W, Yang CS. Tea polyphenol (-)-epigallocatechin-3-gallate inhibits DNA methyltransferase and reactivates methylation-silenced genes in cancer cell lines. Cancer Res. 2003;63:7563-7570. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 4] [Cited by in RCA: 4] [Article Influence: 0.2] [Reference Citation Analysis (0)] |
| 18. | Chen C, Shen G, Hebbar V, Hu R, Owuor ED, Kong AN. Epigallocatechin-3-gallate-induced stress signals in HT-29 human colon adenocarcinoma cells. Carcinogenesis. 2003;24:1369-1378. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 172] [Cited by in RCA: 174] [Article Influence: 7.9] [Reference Citation Analysis (0)] |
| 19. | Jin H, Gong W, Zhang C, Wang S. Epigallocatechin gallate inhibits the proliferation of colorectal cancer cells by regulating Notch signaling. Onco Targets Ther. 2013;6:145-153. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 34] [Cited by in RCA: 48] [Article Influence: 4.0] [Reference Citation Analysis (0)] |
| 20. | Faustino-Rocha A, Oliveira PA, Pinho-Oliveira J, Teixeira-Guedes C, Soares-Maia R, da Costa RG, Colaço B, Pires MJ, Colaço J, Ferreira R, Ginja M. Estimation of rat mammary tumor volume using caliper and ultrasonography measurements. Lab Anim (NY). 2013;42:217-224. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 256] [Cited by in RCA: 352] [Article Influence: 29.3] [Reference Citation Analysis (0)] |
| 21. | Long L, Li Q. The effect of alkaloid from Oxytropis ochrocephala on growth inhibition and expression of PCNA and p53 in mice bearing H22 Hepatocellular Carcinoma. Yakugaku Zasshi. 2005;125:665-670. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 12] [Cited by in RCA: 9] [Article Influence: 0.5] [Reference Citation Analysis (0)] |
| 22. | Xi HQ, Zhang KC, Li JY, Cui JX, Zhao P, Chen L. Expression and clinicopathologic significance of TUFM and p53 for the normal-adenoma-carcinoma sequence in colorectal epithelia. World J Surg Oncol. 2017;15:90. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 8] [Cited by in RCA: 10] [Article Influence: 1.3] [Reference Citation Analysis (0)] |
| 23. | Ferlay J, Shin HR, Bray F, Forman D, Mathers C, Parkin DM. Estimates of worldwide burden of cancer in 2008: GLOBOCAN 2008. Int J Cancer. 2010;127:2893-2917. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 11128] [Cited by in RCA: 11831] [Article Influence: 845.1] [Reference Citation Analysis (4)] |
| 24. | Ezuka A, Sakai E, Kawana K, Nagase H, Kakuta Y, Uchiyama S, Ohkubo H, Higurashi T, Nonaka T, Endo H, Takahashi H, Nakajima A. Association between factors associated with colorectal cancer and rectal aberrant crypt foci in humans. Oncol Lett. 2015;10:3689-3695. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 5] [Cited by in RCA: 6] [Article Influence: 0.6] [Reference Citation Analysis (0)] |
| 25. | Zhu W, Cai MY, Tong ZT, Dong SS, Mai SJ, Liao YJ, Bian XW, Lin MC, Kung HF, Zeng YX, Guan XY, Xie D. Overexpression of EIF5A2 promotes colorectal carcinoma cell aggressiveness by upregulating MTA1 through C-myc to induce epithelial-mesenchymaltransition. Gut. 2012;61:562-575. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 129] [Cited by in RCA: 139] [Article Influence: 10.7] [Reference Citation Analysis (0)] |
| 26. | Corcoran RB, Ebi H, Turke AB, Coffee EM, Nishino M, Cogdill AP, Brown RD, Della Pelle P, Dias-Santagata D, Hung KE, Flaherty KT, Piris A, Wargo JA, Settleman J, Mino-Kenudson M, Engelman JA. EGFR-mediated re-activation of MAPK signaling contributes to insensitivity of BRAF mutant colorectal cancers to RAF inhibition with vemurafenib. Cancer Discov. 2012;2:227-235. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 660] [Cited by in RCA: 815] [Article Influence: 62.7] [Reference Citation Analysis (0)] |
| 27. | Coissieux MM, Tomsic J, Castets M, Hampel H, Tuupanen S, Andrieu N, Comeras I, Drouet Y, Lasset C, Liyanarachchi S, Mazelin L, Puisieux A, Saurin JC, Scoazec JY, Wang Q, Aaltonen L, Tanner SM, de la Chapelle A, Bernet A, Mehlen P. Variants in the netrin-1 receptor UNC5C prevent apoptosis and increase risk of familial colorectal cancer. Gastroenterology. 2011;141:2039-2046. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 27] [Cited by in RCA: 29] [Article Influence: 2.1] [Reference Citation Analysis (0)] |
| 28. | Rahbari NN, Kedrin D, Incio J, Liu H, Ho WW, Nia HT, Edrich CM, Jung K, Daubriac J, Chen I, Heishi T, Martin JD, Huang Y, Maimon N, Reissfelder C, Weitz J, Boucher Y, Clark JW, Grodzinsky AJ, Duda DG, Jain RK, Fukumura D. Anti-VEGF therapy induces ECM remodeling and mechanical barriers to therapy in colorectal cancer liver metastases. Sci Transl Med. 2016;8:360ra135. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 140] [Cited by in RCA: 189] [Article Influence: 23.6] [Reference Citation Analysis (0)] |
| 29. | Tseng-Rogenski SS, Hamaya Y, Choi DY, Carethers JM. Interleukin 6 alters localization of hMSH3, leading to DNA mismatch repair defects in colorectal cancer cells. Gastroenterology. 2015;148:579-589. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 72] [Cited by in RCA: 70] [Article Influence: 7.0] [Reference Citation Analysis (0)] |
| 30. | Shimizu M, Shirakami Y, Sakai H, Yasuda Y, Kubota M, Adachi S, Tsurumi H, Hara Y, Moriwaki H. (-)-Epigallocatechin gallate inhibits growth and activation of the VEGF/VEGFR axis in human colorectal cancer cells. Chem Biol Interact. 2010;185:247-252. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 85] [Cited by in RCA: 91] [Article Influence: 6.1] [Reference Citation Analysis (0)] |