Published online Mar 21, 2020. doi: 10.3748/wjg.v26.i11.1185
Peer-review started: December 20, 2020
First decision: February 18, 2020
Revised: February 20, 2020
Accepted: February 21, 2020
Article in press: February 21, 2020
Published online: March 21, 2020
Processing time: 91 Days and 8.2 Hours
Prolonged postoperative ileus (PPOI) is a prolonged state of “pathological” gastrointestinal (GI) tract dysmotility. There are relatively few studies examining the influence of preoperative nutritional status on the development of PPOI in patients who underwent GI surgery. The association between preoperative albumin and PPOI has not been fully studied. We hypothesized that preoperative albumin may be an independent indicator of PPOI.
To analyze the role of preoperative albumin in predicting PPOI and to establish a nomogram for clinical risk evaluation.
Patients were drawn from a prospective hospital registry database of GI surgery. A total of 311 patients diagnosed with gastric or colorectal cancer between June 2016 and March 2017 were included. Potential predictors of PPOI were analyzed by univariate and multivariable logistic regression analyses, and a nomogram for quantifying the presence of PPOI was developed and internally validated.
The overall PPOI rate was 21.54%. Advanced tumor stage and postoperative opioid analgesic administration were associated with PPOI. Preoperative albumin was an independent predictor of PPOI, and an optimal cutoff value of 39.15 was statistically calculated. After adjusting multiple variables, per unit or per SD increase in albumin resulted in a significant decrease in the incidence of PPOI of 8% (OR = 0.92, 95%CI: 0.85-1.00, P = 0.046) or 27% (OR = 0.73, 95%CI: 0.54-0.99, P = 0.046), respectively. Patients with a high level of preoperative albumin (≥ 39.15) tended to experience PPOI compared to those with low levels (< 39.15) (OR = 0.43, 95%CI: 0.24-0.78, P = 0.006). A nomogram for predicting PPOI was developed [area under the curve (AUC) = 0.741] and internally validated by bootstrap resampling (AUC = 0.725, 95%CI: 0.663-0.799).
Preoperative albumin is an independent predictive factor of PPOI in patients who underwent GI surgery. The nomogram provided a model to screen for early indications in the clinical setting.
Core tip: Prolonged postoperative ileus (PPOI) is a common postoperative complication in patients who undergo gastrointestinal surgery. There are relatively few studies examining the influence of a patient’s preoperative nutritional status on the development of PPOI. This study found and further confirmed that preoperative albumin was an independent predictor of PPOI. We further established a nomogram to accurately quantitate the probability of PPOI occurrence. This nomogram can be used to screen for early indications in the clinical setting.
- Citation: Liang WQ, Zhang KC, Li H, Cui JX, Xi HQ, Li JY, Cai AZ, Liu YH, Zhang W, Zhang L, Wei B, Chen L. Preoperative albumin levels predict prolonged postoperative ileus in gastrointestinal surgery. World J Gastroenterol 2020; 26(11): 1185-1196
- URL: https://www.wjgnet.com/1007-9327/full/v26/i11/1185.htm
- DOI: https://dx.doi.org/10.3748/wjg.v26.i11.1185
Prolonged postoperative ileus (PPOI) is a prolonged state of “pathological” gastrointestinal (GI) tract dysmotility beyond the expected time frame[1]. Some degree of GI tract dysmotility usually occurs in all patients following intra-abdominal surgery, especially major abdominal surgery. As one of the most common postsurgical complications, PPOI occurs in 3%-30% of patients following various types of abdominal operations[2-4]. Clinically, manifestations of POI are characterized as the absence of bowel movement, nausea, vomiting, abdominal distension, and intolerance to oral intake[1,5-8]. Following abdominal surgery, PPOI causes substantial physical discomfort and increases the length of hospital stay, thereby increasing health-care costs. According to a retrospective review of studies on postoperative ileus in more than 800000 US surgical patients, the median length of hospitalization for patients with postoperative ileus was 9.3 d compared to 5.3 d for those without, which resulted in increased costs of approximately $1.5 billion annually for the health-care system[9].
PPOI is a multifactorial process and several mechanisms have been suggested, including age, previous abdominal surgery, systemic opioids, and surgical technique[10-14]. To date, there are relatively few studies examining the influence of a patient’s preoperative nutritional status on the development of PPOI. Albumin is the most commonly used and reliable indicator of a patient’s nutritional status[15]. Hypoalbuminemia is a proven indicator of hospital stay, clinical outcomes, death, and a series of postoperative complications[15-17]. However, the association between preoperative albumin and PPOI has not been fully studied.
The aim of the present study was to investigate the clinical risk factors for PPOI and fully determine the independent relationship between preoperative albumin and PPOI in patients who undergo GI surgery. We plan to propose a nomogram for quantifying the presence of PPOI for early-warning in the clinical setting.
We conducted a retrospective observational cohort study at the Chinese PLA General Hospital. All data were collected from a prospective registry database of PPOI in our institution. Patients diagnosed with gastric or colorectal cancer between June 2016 and March 2017 were included if they met the following eligibility criteria: (1) Age ≥ 18 years; (2) Pathologic diagnosis of adenocarcinoma of the colorectum or stomach; (3) Totally resectable tumor; (4) Primary anastomoses; and (5) Informed consent obtained. Exclusion criteria included (1) emergency surgery, (2) conversion to laparotomy in laparoscopic plan and (3) multi-visceral resection. All of the enrolled patients were scheduled to receive standard surgical tumor excision performed by the same experienced surgical team. Individual written informed consent was obtained before surgery. This study was conducted in accordance with the Declaration of Helsinki. Ethics approval was obtained from the Institutional Review Boards of the Chinese PLA General Hospital (registration number: S2016-092-01).
A number of studies have focused on PPOI. However, controversy existed regarding the diagnostic criteria. One systematic review and global survey clarified the terminology of PPOI and proposed a concise, clinically quantifiable definition[18]. PPOI was defined as two or more of the following criteria which were assessed on or after the fourth day of the postoperative period: nausea or vomiting for 12 h or more without relief, intolerance to a solid or semi-solid oral diet, persistent abdominal distension, absence of passage of both stool and flatus for 24 hours or more, and ileus noted on plain abdominal films or CT scans. We adopted this definition and two investigators independently assessed the diagnosis of PPOI.
Demographic and clinical data of the enrolled patients were determined from the electronic medical records of the prospective registry database before the assessment of PPOI. Baseline demographic data including sex, age, body mass index, and evidence of previous abdominal surgery were summarized during histories, physical examinations, and safety assessments. Serum chemistry, electrolytes, liver, and kidney function tests were monitored before surgery. This study drew attention to two indicators in the biochemistry tests. One was preoperative serum albumin, which is used as a measure of nutritional status[19], and the other was preoperative potassium, which is reported to play an essential role in autoregulation of intestinal smooth muscle[20]. The surgeons were blind to the enrollment of study participants, and the choice of surgical approach (laparoscopic or open surgery). Classification of the primary tumor, regional lymph nodes, and distant metastases was based on the 7th edition of the International Union Against Cancer TNM classification of malignant tumors system. Opioid medication was reported to induce opioid-related bowel dysfunction and was considered a risk factor for PPOI[8,21,22]. In this study, whether opioid analgesics were used after surgery in the enrolled group of patients was evaluated and determined by anesthesiologists who were blind to the study design.
Primary categorical variables were shown as number and percentage. Primary continuous variables were identified both as a continuous variable and as a categorical variable and expressed in two ways: mean ± SD and number and percentage, which were classified according to clinical cutoff values or statistical median. The optimal cutoff value of preoperative albumin was derived from receiver operating characteristic (ROC) curve analysis. Statistical heterogeneity was evaluated by the χ2 test (categorical variables) and one-way ANOVA test (continuous variables). Correlations between variables and PPOI were detected by univariate logistic regression models. Multiple regression analysis was performed to examine the independent association between preoperative albumin and PPOI in three models: for the crude model, no covariates were adjusted; model I, only sociodemographic data were adjusted; and model II, covariates in model I and other elected covariates were adjusted. The variables were considered in model II if the matched odds ratio changed at least 10% as a result of adding these covariates, which was described in previous studies[23]. A multivariable binary logistic regression model following a backward step-down selection process was applied to develop the nomogram. ROC curve analysis was performed to evaluate the discriminatory ability of the nomogram. Due to the limited number of patients, 500 bootstrap computer resamples were used to reduce overfit bias. Statistical analyses were performed using R software and Empower Stats software (X&Y Solutions, Inc.). Odds ratios (ORs) are presented with 95% confidence intervals (CIs) and P < 0.05 was considered significant.
A total of 311 patients in this study, consisting of 218 (10.10%) male patients and 93 (29.90%) female patients, were enrolled based on the inclusion and exclusion criteria. The average age at diagnosis was 58.98 years for the whole enrolled population. There was a similar proportion of gastric (n = 162, 52.09%) and colorectal (n = 149, 47.91%) patients. The overall PPOI rate was 21.54%. Mean preoperative albumin for the entire group was 39.77 ± 3.67 g/L. Other patient demographic data are shown in Table 1. An optimal cutoff value of 39.15 for preoperative albumin was revealed by the ROC curve analysis for PPOI prediction. The area under the ROC curve for the optimal cutoff value was 0.557 with a 95%CI of 0.479-0.635, which offered the optimum mix of sensitivity (0.552) and specificity (0.598) (Figure 1). Therefore, the cutoff value of 39.15 for preoperative albumin was used as an optimal margin in this study. Patient baseline characteristics between the lower preoperative albumin group (n = 135) and the higher preoperative albumin group (n = 176) are compared in Table 1. Mean values of preoperative albumin for the lower and higher group were 36.46 ± 2.33 and 42.30 ± 2.19 g/L, respectively. The low level of preoperative albumin was significantly associated with the older age group no matter if continuous or categorical variables (all P < 0.05). Although not statistically significant, patients in the lower preoperative albumin group tended to have more advanced neoplasia (III-IV stage). The level of preoperative albumin was significantly associated with PPOI, which was observed to be highly prevalent in the lower preoperative albumin group (27.41%) compared to the higher preoperative albumin group (17.05%).
| Characteristics | Total cohort | Subgroups of preoperative albumin | P value | |
| n = 311 | < 39.15, n = 135 | ≥ 39.15, n = 176 | ||
| Sex | 0.364 | |||
| Female | 93 (29.90%) | 44 (32.59%) | 49 (27.84%) | |
| Male | 218 (70.10%) | 91 (67.41%) | 127 (72.16%) | |
| Age (yr) | 58.98 ± 10.98 | 60.52 ± 10.77 | 57.80 ± 11.03 | 0.030 |
| Categorical variable | 0.007 | |||
| < 60 | 156 (50.16%) | 56 (41.48%) | 100 (56.82%) | |
| ≥ 60 | 155 (49.84%) | 79 (58.52%) | 76 (43.18%) | |
| Body mass index (kg/m2) | 24.43 ± 3.23 | 24.14 ± 3.11 | 24.65 ± 3.31 | 0.173 |
| Categorical variable | 0.136 | |||
| < 25 | 174 (55.95%) | 82 (60.74%) | 92 (52.27%) | |
| ≥ 25 | 137 (44.05%) | 53 (39.26%) | 84 (47.73%) | |
| Previous abdominal surgery | 0.178 | |||
| No | 252 (81.03%) | 114 (84.44%) | 138 (78.41%) | |
| Yes | 59 (18.97%) | 21 (15.56%) | 38 (21.59%) | |
| Preoperative albumin (g/L) | 39.77 ± 3.67 | 36.46 ± 2.33 | 42.30 ± 2.19 | < 0.001 |
| Preoperative K+ (mmol/L) | 3.96 ± 0.32 | 3.94 ± 0.34 | 3.98 ± 0.31 | 0.306 |
| Categorical variable | 0.948 | |||
| < 3.96 | 155 (49.84%) | 67 (49.63%) | 88 (50.00%) | |
| ≥ 3.96 | 156 (50.16%) | 68 (50.37%) | 88 (50.00%) | |
| Type of cancer | 0.701 | |||
| Gastric cancer | 162 (52.09%) | 72 (53.33%) | 90 (51.14%) | |
| Colorectal cancer | 149 (47.91%) | 63 (46.67%) | 86 (48.86%) | |
| Surgical technique | 0.868 | |||
| Endoscopic surgery | 215 (69.13%) | 94 (69.63%) | 121 (68.75%) | |
| Open surgery | 96 (30.87%) | 41 (30.37%) | 55 (31.25%) | |
| Tumor stage | 0.094 | |||
| I-II | 133 (43.46%) | 65 (48.87%) | 68 (39.31%) | |
| III-IV | 173 (56.54%) | 68 (51.13%) | 105 (60.69%) | |
| Postoperative opioid analgesic | 0.983 | |||
| No | 191 (61.41%) | 83 (61.48%) | 108 (61.36%) | |
| Yes | 120 (38.59%) | 52 (38.52%) | 68 (38.64%) | |
| PPOI | 0.028 | |||
| No | 244 (78.46%) | 98 (72.59%) | 146 (82.95%) | |
| Yes | 67 (21.54%) | 37 (27.41%) | 30 (17.05%) | |
Univariate analysis was performed to determine the clinical predictors of PPOI after the initial evaluation (Table 2). As shown, preoperative albumin was a protective factor of PPOI. The rate of PPOI in the group with lower preoperative albumin was 27.41% and the rate in the higher group was 17.05%, which indicated a significant association between the level of preoperative albumin and PPOI. Patients with a higher level of preoperative albumin (≥ 39.15) had a 46% reduced risk of PPOI (OR = 0.54, 95%CI: 0.32-0.94, P = 0.029) than those with the lower-level (< 39.15). Patients in the advanced stage (III-IV) were approximately three times more likely to experience PPOI than those in the non-advanced stage (I-II) (incidence of PPOI 30.06% vs 11.28%, OR = 3.33, 95%CI: 1.81-6.25, P < 0.001), which indicated that PPOI was significantly associated with advanced tumors. In addition, patients with clinical use of postoperative analgesia were observed to be at great risk of PPOI (OR = 3.04, 95%CI: 1.74-5.30, P < 0.001). Other associations are shown in Table 2, and no firm correlations were observed between other clinical features.
| Characteristics | Without PPOI | With PPOI | Univariate | P value |
| (n = 244) | (n = 67) | OR (95%CI) | ||
| Sex | 0.362 | |||
| Female | 76 (81.72%) | 17 (18.28%) | Ref. | |
| Male | 168 (77.06%) | 50 (22.97%) | 1.33 (0.72, 2.46) | |
| Age (yr) | 0.867 | |||
| < 60 | 123 (78.58%) | 33 (21.15%) | Ref. | |
| ≥ 60 | 121 (78.06%) | 34 (21.94%) | 1.05 (0.61, 1.80) | |
| Body mass index (kg/m2) | 0.886 | |||
| < 25 | 136 (78.16%) | 38 (21.84%) | Ref. | |
| ≥ 25 | 108 (78.83%) | 29 (21.17%) | 0.96 (0.56, 1.66) | |
| Previous abdominal surgery | 0.803 | |||
| No | 197 (78.17%) | 55 (21.83%) | Ref. | |
| Yes | 47 (79.66%) | 12 (20.34%) | 0.91 (0.45, 1.84) | |
| Preoperative albumin (g/L) | 0.029 | |||
| < 39.15 | 98 (72.59%) | 37 (27.41%) | Ref. | |
| ≥ 39.15 | 146 (82.95%) | 30 (17.05%) | 0.54 (0.32, 0.94) | |
| Preoperative K+ (mmol/L) | 0.701 | |||
| < 3.96 | 123 (79.35%) | 32 (20.65%) | Ref. | |
| ≥ 3.96 | 121 (77.56%) | 35 (22.44%) | 1.11 (0.65, 1.91) | |
| Type of cancer | 0.424 | |||
| Gastric cancer | 130 (80.25%) | 32 (19.75%) | Ref. | |
| Colorectal cancer | 114 (76.51%) | 35 (23.49%) | 1.25 (0.73, 2.14) | |
| Surgical technique | 0.839 | |||
| Endoscopic surgery | 168 (78.14%) | 47 (21.86%) | Ref. | |
| Open surgery | 76 (79.17%) | 20 (20.83%) | 0.94 (0.52, 1.70) | |
| Tumor stage | < 0.001 | |||
| I-II | 118 (88.72%) | 15 (11.28%) | Ref. | |
| III-IV | 121 (69.94%) | 52 (30.06%) | 3.33 (1.81, 6.25) | |
| Postoperative opioid analgesic | < 0.001 | |||
| No | 164 (85.86%) | 27 (14.14%) | Ref. | |
| Yes | 80 (66.67%) | 40 (33.33%) | 3.04 (1.74, 5.30) |
Our findings suggest that PPOI may be affected by a multitude of factors. Multiple regression analysis was performed to further assess the independent effect of preoperative albumin on PPOI. Three models were constructed as follows: A crude model (no variable was adjusted), model I (baseline demographics were adjusted) and model II (variables identified by the criteria described in the method were adjusted). As shown in Table 3, when preoperative albumin was regarded as a continuous variable, per unit or per standard deviation (SD) increase in albumin reduced the incidence of PPOI, although this result was just short of being significant in the crude model and model I. In model II, per unit or per SD increase in albumin significantly reduced the incidence of PPOI by 8% (OR = 0.92, 95%CI: 0.85-1.00, P = 0.046) and 27% (OR = 0.73, 95%CI: 0.54-0.99, P = 0.046) respectively. However, when preoperative albumin was regarded as a categorical variable, an independent and significant association between different levels of preoperative albumin and PPOI was firmly found in the three models. Patients with a high level of preoperative albumin (≥ 39.15) were less likely to experience PPOI with the crude model (OR = 0.54, 95%CI: 0.32-0.94, P = 0.029), model I (OR = 0.53, 95%CI: 0.31-0.92, P = 0.025) and model II (OR = 0.43, 95%CI: 0.24-0.78, P = 0.006).
| Models | Crude model | P value | Model I | P value | Model II | P value |
| Preoperative albumin (g/L) | OR (95%CI) | OR (95%CI) | OR (95%CI) | |||
| Continuous variable | ||||||
| Per unit | 0.95 (0.88, 1.02) | 0.174 | 0.95 (0.88, 1.02) | 0.155 | 0.92 (0.85, 1.00) | 0.046 |
| Per SD (3.67) | 0.83 (0.63, 1.09) | 0.174 | 0.82 (0.62, 1.08) | 0.155 | 0.73 (0.54, 0.99) | 0.046 |
| Categorical variable | ||||||
| < 39.15 | Ref. | Ref. | Ref. | |||
| ≥ 39.15 | 0.54 (0.32, 0.94) | 0.029 | 0.53 (0.31, 0.92) | 0.025 | 0.43 (0.24, 0.78) | 0.006 |
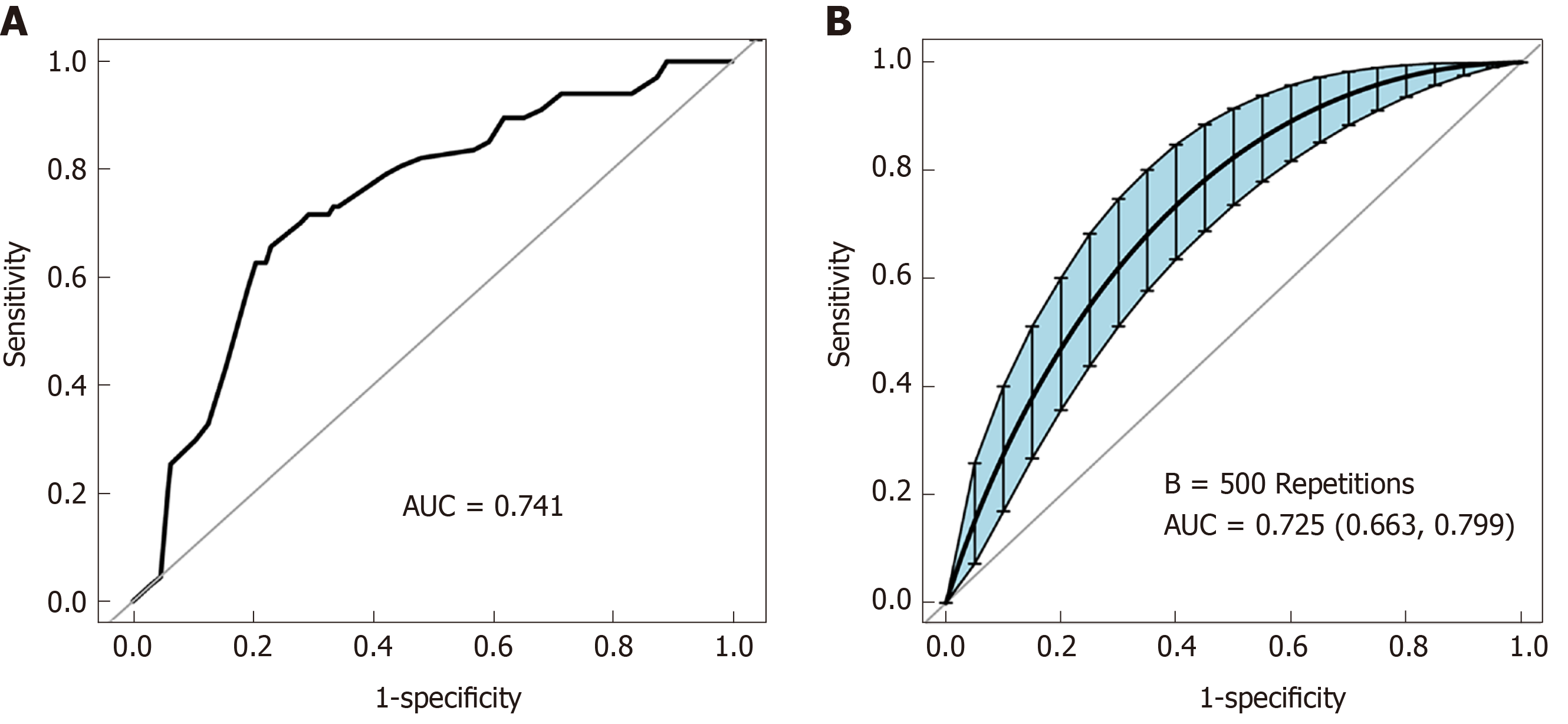
To further accurately quantitate the probability of PPOI occurrence, we established a nomogram to predict PPOI by a backward step-down selection. Five variables, including cancer type, tumor stage, postoperative analgesia, sex, and preoperative albumin, were selected to form the nomogram (Figure 2). A ROC curve was drawn to illustrate the diagnostic ability of the nomogram and the AUC was 0.741 (Figure 3A). Internal validity is a way to measure if the nomogram is sound, and 500 random bootstrap resamplings were performed to reduce overfit bias. Figure 3B shows that the AUC of the random bootstrap resampling was 0.725 (95%CI: 0.663-0.799), which indicated that the nomogram had satisfactory sensitivity.
In the present study, we evaluated the association between clinical features and the presence of PPOI in patients who underwent gastric or colorectal surgery. We found that patients in an advanced tumor stage and who were administered postoperative opioid analgesics were more likely to develop PPOI. This study found and further confirmed that preoperative albumin was an independent predictor of PPOI. An optimal cutoff value of 39.15 was statistically calculated and may be regarded as a warning sign before GI surgery. The level of preoperative albumin, whether a continuous or categorical variable, was significantly associated with the presence of PPOI. We further established a nomogram to accurately quantitate the probability of PPOI occurrence. This nomogram was confirmed to have good diagnostic performance and was internally validated. These findings, if further confirmed in prospective studies, would help identify individuals at high risk of PPOI before surgery.
Preoperative fasting is usually advised for patients before GI. Resections with primary anastomosis during GI surgery always result in combined stress and damage to the GI tract[24,25]. PPOI, a stage of disturbed GI motility, is one of the postoperative complications. An observation study conducted over a period of fourteen years and consisting of 2400 consecutive colorectal cancer patients showed that the incidence of PPOI was 14.0%[10]. A systematic review and meta-analysis consisting of 54 studies and comprising 18983 patients revealed a PPOI incidence of 10.3%[26]. In the present study, the incidence of PPOI was 21.54% for the whole population. The difference in the rate of PPOI can be partly attributed to the eligibility criteria and controversial definition. The duration of prolonged periods of ileus is an important factor in defining PPOI, and in previous studies varied from 3 to 7 d[6,10,14,27]. In the present study, we adopted the criteria from the results of a systematic review and global survey, and the fourth day of postoperative ileus was considered as a prolonged state[18].
Analysis of risk factors for PPOI can assist in early warning and immediate treatment. The insights of risk factors gained from this study may be of assistance to the clinical practitioner. It is now well established from a variety of studies that older age, laparoscopic approach, inflammatory response, and opioid-related dysmotility are significant risk factors for PPOI[8,12,27,28]. In the present study, we found that patients in an advanced tumor stage were more likely to develop PPOI. Tumor stage is a comprehensive factor, and the advanced stage is usually associated with increasing wound size, high operative difficulty and a long operation duration[4,29]. Opioids are very effective for the treatment of acute and chronic pain and are commonly prescribed for pain relief after surgery. One of the most common adverse effects of opioid therapy is opioid-induced bowel dysfunction[22,30]. In the present study, the incidence of PPOI was significantly lower in patients without postoperative use of opioid drugs. Similar conclusions, that reducing opioid-based analgesics shortened the duration of PPOI, were also drawn in previous studies[21,31]. In addition, opioid receptor-specific antagonist drugs, such as methylnaltrexone and alvimopan, are being investigated and hold promise for preventing or resolving the clinically relevant problem of PPOI[8,32,33].
There is a growing body of literature that recognizes serum albumin as a marker of nutritional status and that hypoalbuminemia is correlated with a higher rate of postoperative complications[17,19,34]. The present study mainly focused on the relationship between preoperative albumin and the presence of PPOI. Strong evidence was found of a significant positive correlation between the decrease in preoperative albumin and PPOI. This result was confirmed in different adjusted multivariate models using albumin as a continuous or categorical variable. Albumin is a protein synthesized by the liver and helps maintain plasma osmolality. It does not leak into other tissues and carries various substances throughout the body, including hormones, vitamins, and enzymes[35,36]. Hypoalbuminemia is often observed in patients with GI cancer due to diet deficiencies, weakened liver function and GI bleeding[37,38]. Low levels of preoperative albumin are always associated with more extracellular fluid filling the spaces between tissues[39], which could result in surgical site infection and anastomotic leakage[17]. Furthermore, albumin catabolism is a major source of tumor growth, and a lower level of albumin indicates the severity and duration of GI cancers[40]. Preoperative albumin was also demonstrated to be a reliable and reproducible predictor of surgical risk and postoperative complications[16,41]. Thus, the level of preoperative albumin may reflect the overall preoperative physical condition and the function of GI anastomosis after resection.
To quantitate the probability of PPOI occurrence, a nomogram was established by backward step-down selection. Preoperative albumin and the other four independent variables were filtered out. The AUC value (0.741) showed the good performance of this nomogram. We developed the prediction model using the entire data set and then used resampling techniques to evaluate the performance. Resampling techniques, generally referred to as “internal validation”, are recommended as a prerequisite for prediction model development, particularly if data are limited[42]. This nomogram could be identified as an important early warning sign of PPOI in patients who underwent GI surgery.
Our study had several strengths. First, previous studies mainly focused on investigating variables that were only determined during or after surgery when predicting PPOI. Our study firstly demonstrated preoperative albumin as a predictive indicator that would be available before surgery. Second, this study established a nomogram for quantifying the presence of PPOI, and it was further validated by internal bootstrap.
The present study also has several limitations. First, this study was retrospective in nature which may not bring out many variables in a clinical setting such as complications and blood requirement during surgery, total procedure time, bowel handling and manipulation during the procedure and the resultant release of local inflammatory mediators. Thus, further studies with a prospective and multi-centered design are warranted. Second, due to the small number of patients and the prospectively collected registry database, it was difficult to develop external validation of the nomogram. In order to make up for this limitation, we sought to validate this model using bootstrap.
In conclusion, this retrospective cohort study suggests that PPOI is a common postoperative complication in patients who undergo gastric or colorectal surgery. Advanced tumor stage, use of opioid analgesics and lower preoperative albumin level were the main risk factors for PPOI. Preoperative albumin is an independent predictive factor for PPOI in patients undergoing GI surgery. In the present study, an easy-to-use nomogram was established to quantify the presence of PPOI, which may serve as an early warning sign of PPOI in clinical practice. Due to the retrospective nature of this study, caution should be exercised in proposing the nomogram in clinical practice.
Prolonged postoperative ileus (PPOI) is a prolonged state of “pathological” gastrointestinal (GI) tract dysmotility. PPOI is an essential contributor to increased hospitalization costs and prolonged hospitalization.
It is well known that preoperative albumin affects the outcome and mortality of patients undergoing any surgical procedure. We hypothesized that preoperative albumin may be an independent indicator of PPOI.
The present study aimed to investigate the association between preoperative albumin and PPOI to establish a nomogram for clinical risk evaluation.
A total of 311 patients diagnosed with gastric or colorectal cancer were retrospectively included. We performed univariate and multivariable logistic regression analyses to determine the relationship between variables and PPOI, and a nomogram to quantify the presence of PPOI was developed and internally validated.
The overall PPOI rate was 21.54%. Patients in an advanced tumor stage and who were administered postoperative opioid analgesics were more likely to develop PPOI. This study found and further confirmed that preoperative albumin was an independent predictor of PPOI. A nomogram was established to accurately quantitate the probability of PPOI occurrence. This nomogram was confirmed to have a good diagnostic performance and was also internally validated.
Preoperative albumin is an independent predictive factor of PPOI in patients who underwent GI surgery. An easy-to-use nomogram was established to quantify the presence of PPOI, which may serve as an early warning sign of PPOI in clinical practice.
Due to the retrospective nature of this study, caution should be exercised in proposing the nomogram in clinical practice.
We are very grateful to Liu Jie (General Hospital of Chinese PLA), Li Haibo PhD (Peking University Hospital of Stomatology) and Li Wenjing PhD (Peking Union Medical College) for their help in statistical analysis.
Manuscript source: Invited manuscript
Specialty type: Gastroenterology and hepatology
Country of origin: China
Peer-review report classification
Grade A (Excellent): 0
Grade B (Very good): B
Grade C (Good): 0
Grade D (Fair): 0
Grade E (Poor): 0
P-Reviewer: Tandon RK S-Editor: Wang J L-Editor: Webster J E-Editor: Ma YJ
| 1. | Bragg D, El-Sharkawy AM, Psaltis E, Maxwell-Armstrong CA, Lobo DN. Postoperative ileus: Recent developments in pathophysiology and management. Clin Nutr. 2015;34:367-376. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 192] [Cited by in RCA: 238] [Article Influence: 23.8] [Reference Citation Analysis (1)] |
| 2. | Pavoor R, Milsom J. Postoperative ileus after laparoscopic colectomy: elusive and expensive. Ann Surg. 2011;254:1075; author reply 1075-1075; author reply 1076. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 3] [Cited by in RCA: 5] [Article Influence: 0.4] [Reference Citation Analysis (0)] |
| 3. | Tevis SE, Carchman EH, Foley EF, Harms BA, Heise CP, Kennedy GD. Postoperative Ileus--More than Just Prolonged Length of Stay? J Gastrointest Surg. 2015;19:1684-1690. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 59] [Cited by in RCA: 95] [Article Influence: 9.5] [Reference Citation Analysis (0)] |
| 4. | Artinyan A, Nunoo-Mensah JW, Balasubramaniam S, Gauderman J, Essani R, Gonzalez-Ruiz C, Kaiser AM, Beart RW. Prolonged postoperative ileus-definition, risk factors, and predictors after surgery. World J Surg. 2008;32:1495-1500. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 201] [Cited by in RCA: 195] [Article Influence: 11.5] [Reference Citation Analysis (0)] |
| 5. | Chan DC, Liu YC, Chen CJ, Yu JC, Chu HC, Chen FC, Chen TW, Hsieh HF, Chang TM, Shen KL. Preventing prolonged post-operative ileus in gastric cancer patients undergoing gastrectomy and intra-peritoneal chemotherapy. World J Gastroenterol. 2005;11:4776-4781. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in CrossRef: 20] [Cited by in RCA: 23] [Article Influence: 1.2] [Reference Citation Analysis (0)] |
| 6. | Kronberg U, Kiran RP, Soliman MS, Hammel JP, Galway U, Coffey JC, Fazio VW. A characterization of factors determining postoperative ileus after laparoscopic colectomy enables the generation of a novel predictive score. Ann Surg. 2011;253:78-81. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 113] [Cited by in RCA: 138] [Article Influence: 9.9] [Reference Citation Analysis (0)] |
| 7. | van Bree SH, Nemethova A, Cailotto C, Gomez-Pinilla PJ, Matteoli G, Boeckxstaens GE. New therapeutic strategies for postoperative ileus. Nat Rev Gastroenterol Hepatol. 2012;9:675-683. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 65] [Cited by in RCA: 71] [Article Influence: 5.5] [Reference Citation Analysis (0)] |
| 8. | Becker G, Blum HE. Novel opioid antagonists for opioid-induced bowel dysfunction and postoperative ileus. The Lancet. 2009;1198-1206. [RCA] [DOI] [Full Text] [Cited by in Crossref: 84] [Cited by in RCA: 76] [Article Influence: 4.8] [Reference Citation Analysis (0)] |
| 9. | Iyer S, Saunders WB, Stemkowski S. Economic burden of postoperative ileus associated with colectomy in the United States. J Manag Care Pharm. 2009;15:485-494. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 207] [Cited by in RCA: 226] [Article Influence: 14.1] [Reference Citation Analysis (0)] |
| 10. | Chapuis PH, Bokey L, Keshava A, Rickard MJ, Stewart P, Young CJ, Dent OF. Risk factors for prolonged ileus after resection of colorectal cancer: an observational study of 2400 consecutive patients. Ann Surg. 2013;257:909-915. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 180] [Cited by in RCA: 157] [Article Influence: 13.1] [Reference Citation Analysis (0)] |
| 11. | van Bree SH, Bemelman WA, Hollmann MW, Zwinderman AH, Matteoli G, El Temna S, The FO, Vlug MS, Bennink RJ, Boeckxstaens GE. Identification of clinical outcome measures for recovery of gastrointestinal motility in postoperative ileus. Ann Surg. 2014;259:708-714. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 100] [Cited by in RCA: 128] [Article Influence: 11.6] [Reference Citation Analysis (0)] |
| 12. | Moghadamyeghaneh Z, Hwang GS, Hanna MH, Phelan M, Carmichael JC, Mills S, Pigazzi A, Stamos MJ. Risk factors for prolonged ileus following colon surgery. Surg Endosc. 2016;30:603-609. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 105] [Cited by in RCA: 129] [Article Influence: 12.9] [Reference Citation Analysis (0)] |
| 13. | Reichert M, Weber C, Pons-Kühnemann J, Hecker M, Padberg W, Hecker A. Protective loop ileostomy increases the risk for prolonged postoperative paralytic ileus after open oncologic rectal resection. Int J Colorectal Dis. 2018;33:1551-1557. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 12] [Cited by in RCA: 13] [Article Influence: 1.9] [Reference Citation Analysis (0)] |
| 14. | Sugawara K, Kawaguchi Y, Nomura Y, Suka Y, Kawasaki K, Uemura Y, Koike D, Nagai M, Furuya T, Tanaka N. Perioperative Factors Predicting Prolonged Postoperative Ileus After Major Abdominal Surgery. J Gastrointest Surg. 2018;22:508-515. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 22] [Cited by in RCA: 37] [Article Influence: 5.3] [Reference Citation Analysis (0)] |
| 15. | Hennessey DB, Burke JP, Ni-Dhonochu T, Shields C, Winter DC, Mealy K. Preoperative hypoalbuminemia is an independent risk factor for the development of surgical site infection following gastrointestinal surgery: a multi-institutional study. Ann Surg. 2010;252:325-329. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 203] [Cited by in RCA: 237] [Article Influence: 15.8] [Reference Citation Analysis (1)] |
| 16. | Blomberg J, Lagergren P, Martin L, Mattsson F, Lagergren J. Albumin and C-reactive protein levels predict short-term mortality after percutaneous endoscopic gastrostomy in a prospective cohort study. Gastrointest Endosc. 2011;73:29-36. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 81] [Cited by in RCA: 90] [Article Influence: 6.4] [Reference Citation Analysis (0)] |
| 17. | Shimura T, Toiyama Y, Hiro J, Imaoka H, Fujikawa H, Kobayashi M, Ohi M, Inoue Y, Mohri Y, Kusunoki M. Monitoring perioperative serum albumin can identify anastomotic leakage in colorectal cancer patients with curative intent. Asian J Surg. 2018;41:30-38. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 19] [Cited by in RCA: 30] [Article Influence: 3.3] [Reference Citation Analysis (0)] |
| 18. | Vather R, Trivedi S, Bissett I. Defining postoperative ileus: results of a systematic review and global survey. J Gastrointest Surg. 2013;17:962-972. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 273] [Cited by in RCA: 361] [Article Influence: 30.1] [Reference Citation Analysis (0)] |
| 19. | Lehmann S, Ferrie S, Carey S. Nutrition Management in Patients With Chronic Gastrointestinal Motility Disorders: A Systematic Literature Review. Nutr Clin Pract. 2019;. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 7] [Cited by in RCA: 18] [Article Influence: 3.0] [Reference Citation Analysis (0)] |
| 20. | Kuruba R, Fayard N, Snyder D. Epidural analgesia and laparoscopic technique do not reduce incidence of prolonged ileus in elective colon resections. Am J Surg. 2012;204:613-618. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 19] [Cited by in RCA: 13] [Article Influence: 1.0] [Reference Citation Analysis (0)] |
| 21. | Koo KC, Yoon YE, Chung BH, Hong SJ, Rha KH. Analgesic opioid dose is an important indicator of postoperative ileus following radical cystectomy with ileal conduit: experience in the robotic surgery era. Yonsei Med J. 2014;55:1359-1365. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 19] [Cited by in RCA: 20] [Article Influence: 1.8] [Reference Citation Analysis (0)] |
| 22. | Panchal SJ, Müller-Schwefe P, Wurzelmann JI. Opioid-induced bowel dysfunction: prevalence, pathophysiology and burden. Int J Clin Pract. 2007;61:1181-1187. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 238] [Cited by in RCA: 238] [Article Influence: 13.2] [Reference Citation Analysis (0)] |
| 23. | Kernan WN, Viscoli CM, Brass LM, Broderick JP, Brott T, Feldmann E, Morgenstern LB, Wilterdink JL, Horwitz RI. Phenylpropanolamine and the risk of hemorrhagic stroke. N Engl J Med. 2000;343:1826-1832. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 506] [Cited by in RCA: 458] [Article Influence: 18.3] [Reference Citation Analysis (0)] |
| 24. | Anup R, Balasubramanian KA. Surgical stress and the gastrointestinal tract. J Surg Res. 2000;92:291-300. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 35] [Cited by in RCA: 33] [Article Influence: 1.3] [Reference Citation Analysis (0)] |
| 25. | Thomas S, Balasubramanian KA. Role of intestine in postsurgical complications: involvement of free radicals. Free Radic Biol Med. 2004;36:745-756. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 34] [Cited by in RCA: 29] [Article Influence: 1.4] [Reference Citation Analysis (0)] |
| 26. | Wolthuis AM, Bislenghi G, Fieuws S, de Buck van Overstraeten A, Boeckxstaens G, D'Hoore A. Incidence of prolonged postoperative ileus after colorectal surgery: a systematic review and meta-analysis. Colorectal Dis. 2016;18:O1-O9. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 155] [Cited by in RCA: 147] [Article Influence: 16.3] [Reference Citation Analysis (0)] |
| 27. | Huang DD, Zhuang CL, Wang SL, Pang WY, Lou N, Zhou CJ, Chen FF, Shen X, Yu Z. Prediction of Prolonged Postoperative Ileus After Radical Gastrectomy for Gastric Cancer: A Scoring System Obtained From a Prospective Study. Medicine (Baltimore). 2015;94:e2242. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 18] [Cited by in RCA: 24] [Article Influence: 2.4] [Reference Citation Analysis (0)] |
| 28. | Wehner S, Vilz TO, Stoffels B, Kalff JC. Immune mediators of postoperative ileus. Langenbecks Arch Surg. 2012;397:591-601. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 47] [Cited by in RCA: 56] [Article Influence: 4.3] [Reference Citation Analysis (0)] |
| 29. | Vather R, Josephson R, Jaung R, Robertson J, Bissett I. Development of a risk stratification system for the occurrence of prolonged postoperative ileus after colorectal surgery: a prospective risk factor analysis. Surgery. 2015;157:764-773. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 87] [Cited by in RCA: 112] [Article Influence: 11.2] [Reference Citation Analysis (0)] |
| 30. | Pappagallo M. Incidence, prevalence, and management of opioid bowel dysfunction. Am J Surg. 2001;182:11S-18S. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 403] [Cited by in RCA: 388] [Article Influence: 16.2] [Reference Citation Analysis (0)] |
| 31. | Delaney CP. Clinical perspective on postoperative ileus and the effect of opiates. Neurogastroenterol Motil. 2004;16 Suppl 2:61-66. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 74] [Cited by in RCA: 75] [Article Influence: 3.6] [Reference Citation Analysis (0)] |
| 32. | Xu LL, Zhou XQ, Yi PS, Zhang M, Li J, Xu MQ. Alvimopan combined with enhanced recovery strategy for managing postoperative ileus after open abdominal surgery: a systematic review and meta-analysis. J Surg Res. 2016;203:211-221. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 44] [Cited by in RCA: 39] [Article Influence: 4.3] [Reference Citation Analysis (0)] |
| 33. | Nair A. Alvimopan for post-operative ileus: What we should know? Acta Anaesthesiol Taiwan. 2016;54:97-98. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 6] [Cited by in RCA: 8] [Article Influence: 0.9] [Reference Citation Analysis (0)] |
| 34. | Lu PH, Wei MX, Shen W, Li C, Cai B, Tao GQ. Is preoperative serum albumin enough to ensure nutritional status in the development of surgical site infection following gastrointestinal surgery? Ann Surg. 2011;254:663-4; author reply 664. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 2] [Cited by in RCA: 2] [Article Influence: 0.1] [Reference Citation Analysis (0)] |
| 35. | Kmieć Z. Cooperation of liver cells in health and disease. Adv Anat Embryol Cell Biol. 2001;161:III-XIII, 1-151. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 50] [Cited by in RCA: 210] [Article Influence: 9.1] [Reference Citation Analysis (0)] |
| 36. | Lu CY, Wu DC, Wu IC, Chu KS, Sun LC, Shih YL, Chen FM, Hsieh JS, Wang JY. Serum albumin level in the management of postoperative enteric fistula for gastrointestinal cancer patients. J Invest Surg. 2008;21:25-32. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 19] [Cited by in RCA: 16] [Article Influence: 0.9] [Reference Citation Analysis (0)] |
| 37. | Nassri A, Zhu H, Wang DH, Ramzan Z. Serum Albumin at Diagnosis is an Independent Predictor of Early Mortality in Veteran Patients with Esophageal Cancer. Nutr Cancer. 2018;70:1246-1253. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 3] [Cited by in RCA: 3] [Article Influence: 0.4] [Reference Citation Analysis (0)] |
| 38. | SAFE Study Investigators, Finfer S, Bellomo R, McEvoy S, Lo SK, Myburgh J, Neal B, Norton R. Effect of baseline serum albumin concentration on outcome of resuscitation with albumin or saline in patients in intensive care units: analysis of data from the saline versus albumin fluid evaluation (SAFE) study. BMJ. 2006;333:1044. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 127] [Cited by in RCA: 145] [Article Influence: 7.6] [Reference Citation Analysis (0)] |
| 39. | Doweiko JP, Nompleggi DJ. Reviews: The Role of Albumin in Human Physiology and Pathophysiology, Part III: Albumin and Disease States. Jpen-Parenter Enter. 2016;15:476-483. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 176] [Cited by in RCA: 183] [Article Influence: 5.4] [Reference Citation Analysis (0)] |
| 40. | Kratz F. Albumin as a drug carrier: design of prodrugs, drug conjugates and nanoparticles. J Control Release. 2008;132:171-183. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 1512] [Cited by in RCA: 1617] [Article Influence: 95.1] [Reference Citation Analysis (0)] |
| 41. | Bendersky V, Sun Z, Adam MA, Rushing C, Kim J, Youngwirth L, Turner M, Migaly J, Mantyh CR. Determining the Optimal Quantitative Threshold for Preoperative Albumin Level Before Elective Colorectal Surgery. J Gastrointest Surg. 2017;21:692-699. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 9] [Cited by in RCA: 13] [Article Influence: 1.6] [Reference Citation Analysis (0)] |
| 42. | Collins GS, Reitsma JB, Altman DG, Moons KG. Transparent reporting of a multivariable prediction model for individual prognosis or diagnosis (TRIPOD): the TRIPOD statement. BMJ. 2015;350:g7594. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 1604] [Cited by in RCA: 2306] [Article Influence: 230.6] [Reference Citation Analysis (0)] |